Abstract
This study comprehensively investigated the effect of iron-oxide nanoparticles (NPs) on weathering durability and thermal resistance of wood-plastic composites (WPCs). The hydrophobic nature of NPs improved the dimensional stability of WPCs. The small size of NPs deposits the voids in the matrix, which helps to increase the mechanical properties, even after weathering test. The decrease of modulus of rupture (MOR) reached up to 16% for control samples, while it was 2% for IO40-1. Despite the intensive weathering conditions, NPs cover the surface of materials like a UV shield, improving WPCs' UV resistance. Moreover, the increase in the wood and NPs content limited the UV degradation, which resulted in lower color changes. The attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR) analysis also proved that there were nearly no changes in the characteristic bands of polymer (2916 and 2846 cm−1) and wood (1512 cm−1). The microscopic investigation, however, revealed the deterioration on the surface of WPCs after weathering exposure. Even though UV degradation is inevitable, iron oxide NPs significantly preserved the WPCs surface. However, there were also crack formations, which were also inhibited. On the other hand, NPs decelerated the thermal degradation by acting as a heat barrier. Thermogravimetric analysis (TGA) analysis revealed an increase in the degradation temperature. Limit oxygen index (LOI) values also increased up to 27.6%, which demonstrated an improvement against flammability.
Introduction
Due to increased environmental concerns, designers, engineers, and manufacturers seek green construction materials. WPCs have been among the most preferred wood-based composites in recent years. Although the first use of WPCs was in decking, today, they can be used in different areas from fences to siding, car parts to construction. The WPCs are applied mainly outdoors, where severe conditions shorten the service life. However, long-term performance is desired due to raw material shortages, climate change, etc.
Most of the shortcomings can be eliminated by encapsulating wood fiber with a polymer; however, outdoor conditions, such as UV lights, humidity, exhaust gases, rain, wind, etc., inevitably cause degradation. 1 Chromophoric groups in wood and polymers absorb UV light.2,3 Then, the scission reactions occur, resulting in a shortage of polymer chains, which induces crack formation.4,5 Therefore, conserved wood fibers with encapsulation become visible and are exposed to UV lights and humidity. Finally, this phenomenon occurs recurrently, depleting the materials' service life and causing economic losses. Furthermore, the application range of WPCs, which are highly sensitive to flame, is limited since the non-coal-forming thermal plastics used in the WPCs cause a noticeable wick effect by creating high combustion heat. The structural integrity begins to be disrupted above the polymer's glass transition temperature. 6 There are more studies recently focusing on the synergy between fire retardants and surface modification and improving fire retardant properties and effects for WPCs.7,8
The high surface/volume ratio describes “nanomaterials” as one of plastics' most popular reinforcement agents. 9 Their increased interaction improves the interface between the polymer and filling materials. 10 Therefore, nanoparticles (NPs) are crucial in enhancing materials' technological properties, such as mechanical, physical, thermal, weathering, and biological properties. 11 Therefore, SiO2, TiO2, and ZnO NPs have been chiefly preferred nanomaterials for reinforcement. Their non-toxicity and high thermal stability are the main reasons why they are chosen for many applications. Besides that, high durability, opacity, low toxicity, and low cost are attributed to iron oxide NPs, which are prominent for plastic composites. 12 Despite the limited number of studies, recent investigations revealed the potential of iron oxide NPs for wood-based composites. Gul et al. enhanced the dimensional and thermal stability of middle-density boards with the reinforcement of iron-oxide NPs. 13 Some researchers also revealed the weathering-reducing effect of iron oxide NPs. Zhang et al. reinforced WPCs with iron oxide NP pigments (red, yellow, blue, and black). 14 The coloration decreased on the surface of WPCs, mainly red and black, which were better light stabilizers due to their inorganic status. Xu et al. also stated that iron oxide NPs imparted a light stabilizer effect on WPCs after the hygrothermal aging test. 12 However, there were also some losses in mechanical properties. In their study in which they simulated seawater conditions, Jiang et al. revealed the positive effect of iron oxide pigments on the structural integrity, morphological stability, and thermal durability of PVC-based composites. 15
As explained above, NPs impart superior properties to materials, which is the main reason for their preference as reinforcement agents. Although iron oxide NPs are commonly evaluated in gas detectors, catalysts, and magnetic and absorbing materials, 16 there are a limited number of studies about iron oxide-reinforced WPCs. This study comprehensively discussed the effect of iron oxide (Fe2O3) reinforcement on water uptake and dimensional stability, mechanical resistance, weathering durability, and thermal degradation of WPCs. The weathering performance of iron oxide-reinforced WPCs was also investigated after 840 h of artificial weathering. Moreover, the surface of WPCs exposed to weathering was characterized with ATR-FTIR, scanning electron microscopy (SEM), and light microscopy (LM) analysis. The thermal properties were also investigated with TGA analysis and LOI test.
Experimental
Materials and WPCs production
Pine wood flour (WF) (Pinus sylvestris L.) as a filler and high-density polyethylene (HDPE) as a thermoplastic polymer were obtained from commercial suppliers to produce the WPCs. The fine-grain polymer (∼200 mesh) and 40-60 mesh of WF were preferred to manage a homogeneous mixture. The melt flow index (MIF) and density of the polymer were 5.5g/10 min (190°C/2.16 kg) and 0.965g/cm3, respectively. Maleic anhydride-grafted polyethylene (MAPE) (Licocene PE MA 4351 Fine Grain) was added as a coupling agent to the matrix to improve the bonding, whose softening point and density were 123°C and 0.99g/cm3, respectively. Red iron oxide NPs (18-38 nm) (Nanografi, Ankara, Turkiye) with a 5.24g/cm3 density were used as reinforcement agents.
The rate of components (%).
Water uptake and dimensional stability
The water uptake test was performed according to ASTM D570.
17
The dimensional stability was evaluated with the changes in the thickness swelling of WPC specimens. The specimens with dimensions of 50 mm × 50 mm × 4 mm were entirely soaked in the water at 20 ± 1°C. The measurements were taken after 1, 3, 7, 14, and 28 days. The surface of the specimen was cleaned and dried before the measurement. Five specimens were used for the test of each group. The values of WA as percentages were calculated with equation (1):
Accelerated UV weathering test
The artificial weathering test was conducted in a QUV/spray-accelerated weathering tester (Q-Panel Lab. Products) according to ASTM G154-12a. 18 The temperature was set to 60°C in the test chamber, and 313 nm fluorescent UV lamps (UVB) were preferred for UV exposure. The WPCs specimens were exposed to 8 h of UV irradiation and 4 h of condensation (50°C) cycle during the 840 h. Three replicates were exposed to weathering tests for each group. The deformation in the WPCs samples caused by the accelerated UV weathering test was determined with the color measurement, ATR-FTIR spectroscopy, light and scanning electron microscopic, modulus of rupture, and modulus of elasticity (MOE) tests.
Color measurement
The color change on the surface of WPCs was determined with a Minolta CM-600d spectrophotometer (Konica Minolta) equipped with an integrating sphere, according to ISO 7724. 19 Standard illuminant D65 was preferred. The color measurements were performed in an area of 8 mm2 in the 400–700 nm wavelength range. Six measurements were obtained from each sample's surface. The color measurement was taken at 24, 48, 72, 120, 168, 336, 504, 672, and 840th h of the artificial weathering test.
ATR-FTIR spectroscopy analysis
The chemical change of the WPCs samples exposed to accelerated weathering was investigated with the ATR technique using a Thermo Nicolet iS50 FTIR (Thermo Fisher Scientific Co.) spectrometer equipped with a single-bounce diamond crystal and a deuterated triglycine sulphate detector. The surfaces of the specimens were in contact with the ATR crystal so that the surface could absorb the evanescent wave. The resultant attenuated radiation produced an ATR spectrum similar to a conventional absorption spectrum. 20 The ATR-FTIR spectra of the samples before and after weathering test were acquired in the range of 800–4000 cm−1 with a resolution of 4 cm−1. Each spectrum was collected through 64 scans in the absorbance mode. Two measurements were carried out and averaged to produce one spectrum.
Microscopic analysis
The surface of WPCs was investigated with an LM (Zeiss Stemi 305) equipped with a (camera) (Zeiss AxiocAM erC 5-s) and SEM (Zeiss Evo LS10). The surface of WPCs was gold-coated before SEM analysis (Emitech, SC7620).
Mechanical properties
A three-point bending test was performed to determine the MOR and MOE of the WPC specimens in a universal test machine (Marestek, Istanbul, Turkiye) according to ASTM D790. 21 The eight specimens with 127 mm × 12.7 mm × 5 mm dimensions were tested for each group. The tension side was the part of the specimens exposed to the light.
Thermal properties
The thermal stability of WPCs at a temperature between 30°C to 600°C was investigated with Perkin–Elmer STA 6000 thermogravimetric analyzer 4000 (USA). The heating rate was set to 10°C/min under nitrogen. Two samples were tested for each group.
The need for oxygen of WPCs during flammable combustion was determined with the LOI test according to ASTM D2863-19. 22 The five samples with 127 mm × 12.7 mm × 5 mm were prepared for each group.
Statistical analysis
The data were statistically examined based on the analysis of variance (ANOVA). The differences between groups were investigated using the Duncan test (p < 0.05).
Results and discussion
Water uptake and dimensional stability
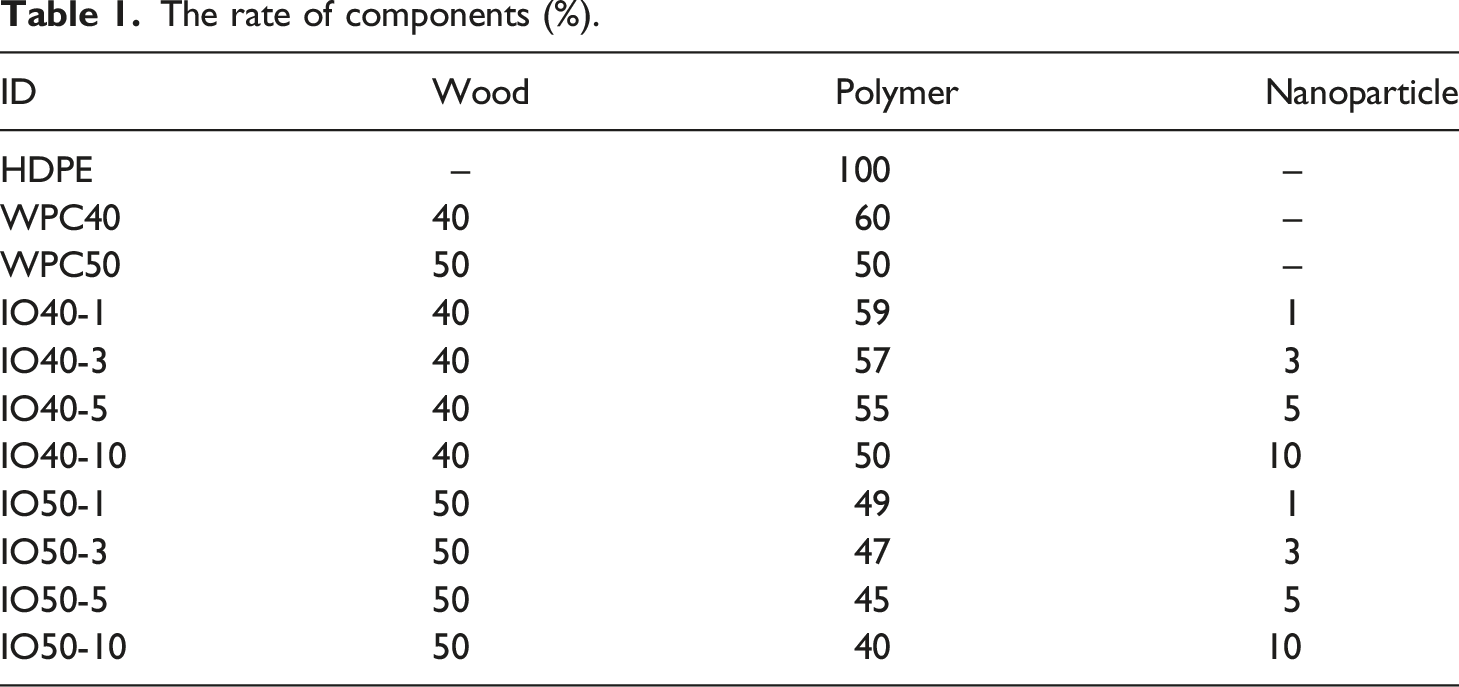
The effect of iron oxide NPs on the water uptake and dimensional stability of WPCs was investigated, as seen in Figure 1. WF is a hydrophilic material, contrary to HDPE, which negatively affects water uptake and dimensional stability.
23
However, iron oxide NPs improved the dimensional stability of WPCs, which is significantly essential outdoors. NPs could penetrate the deepest point of wood cells thanks to their small size and change wood fibers' surface character, which makes wood fiber stronger against water uptake.
24
As seen in Figure 1, IO40-10 has the highest resistance against water absorption. Water uptake and dimensional stability of WPCs.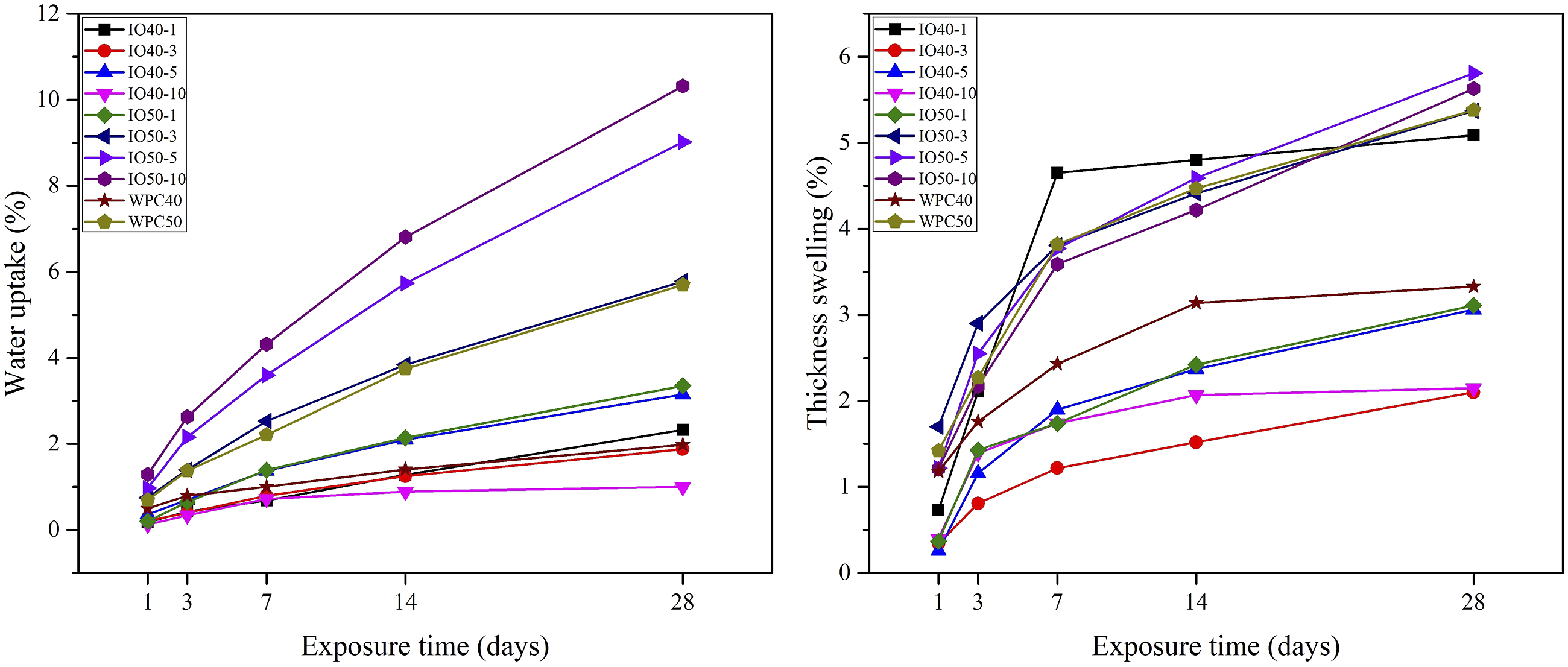
NPs gathered on the microvoids of fibers and inhibited water transfer, which limited water uptake. Moreover, the micro/nanomaterials create a rough surface, which helps to reduce the surface energy due to the entrapment of air between the water drop and the surface. 25 However, increased NP content with WF inevitably reduced the resistance due to decreased polymer as a bonding material. Moreover, iron-oxide NPs tend to agglomerate in the higher range due to the magnetic interaction, which could also maximize the water pathway, resulting in increased water uptake and dimensional stability.
Mechanical properties
The effect of iron oxide NPs on the mechanical properties was investigated, as seen in Figure 2. The inconsistency between WF and polymer resulted in a loss of MOR. At the same time, the natural fibers improved the MOE due to low deformation capacity in the elastic zone.
26
The increase in the WF content reduced the MOR to 22%, while the improvement reached 144% for MOE compared to the only polymer. Meanwhile, the NPs fill the micro gaps between the components, improving the matrix's load transfer. Similar to dimensional stability, as NP content increased, the polymer decreased, negatively influencing mechanical properties due to the bonding role in the matrix.
27
Therefore, the highest MOR of 10% was obtained from IO40-1. Meanwhile, the van der Waals forces enhance the interaction between the iron oxide NPs.
28
Moreover, their high surface area and energy may have resulted in accumulation, especially higher NPs content, which might also be a reason for poor mechanical properties. However, compared to control samples, whole NP-reinforced WPCs have higher mechanical properties. The mechanical properties of WPCs; AW: After weathering.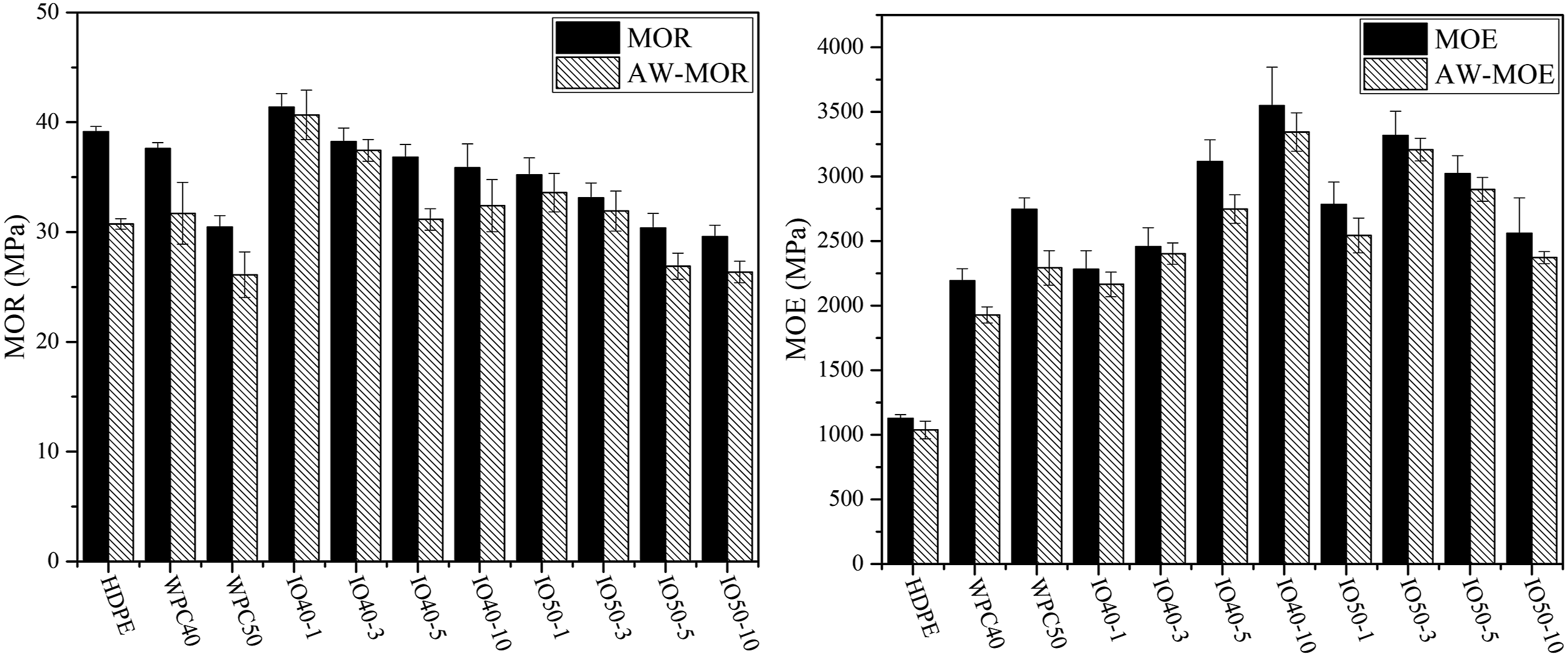
Furthermore, the MOR and MOE values were still higher after the weathering test than the control samples. However, WPCs without reinforcement did not resist degradation during the weathering exposure. The decrease in the MOR reached 16% for control samples, while it was under 2% for IO40-1. As explained above, UV light and humidity considerably degraded WPCs, limiting the load transfer. Meanwhile, iron oxide has absorption and/or reflection ability of UV light as well as a hydrophobic character, which reduces the degradation effect. Especially iron oxide content of up to 5% showed a significant protective effect. However, the increased NPs with decreasing polymer content were insufficient to reduce weathering effect. Therefore, the loss was inevitable for mechanical properties.
Color changes
The effect of iron oxide NPs on color change was investigated, as seen in Figure 3. Color change was inevitable, according to the data. However, it can be reduced via materials' UV absorption and/or reflection abilities. The highest color changes were obtained from control samples. The main ones responsible for the deterioration of the WPC surface are chromophoric groups, which are possessed of both wood and polymer. Lignin, a cell wall component of wood, is mainly responsible for less than 400 nm of UV light, causing the yellowish to the grayish surface of woody materials.29,30 On the other hand, iron oxide NP-reinforced WPCs significantly resisted weathering conditions and provided light stabilization. Meanwhile, inorganic iron oxide pigments have been recognized as having high UV and thermal resistance, low cost, and compatibility with plastics compared to organics.
12
Therefore, iron oxide NPs cover the surface of wood fibers and cut off UV light's access, making WPCs’ surface more resistant. The light stabilization effect increased with increased NPs content in the matrix. The highest iron oxide nearly inhibited the color changes on the surface of WPCs. The color changes of WPCs during 840 h of artificial weathering test.
A similar trend was observed for the lightness of WPCs’ surface. WPCs’ surface tends to be whitened as exposed to UV lights. The surface of WPCs was whitened during the weathering test, especially after 168 h, as seen in Figure 3. However, the light stabilization effect of iron oxide limited the whitening of the surface. Red and black iron oxide is recognized as an inorganic character, which expresses the color stabilization of WPCs’ surface.
Microscopic analyses
Light microscopy analysis
The effect of iron oxide NPs on the weathering performance of WPCs was investigated with light microscopy, as seen in Figure 4. The color changes were remarkable in light microscopy images. The WPCs without NPs were whitened and cracked (black arrows). No significant crack formation was observed on the polymer’s surface (HDPE), while roughness increased. As photooxidation degrades the polymer surface, a new layer reveals, which increases the surface roughness. Inconsistency between the materials also enhances photodegradation, which results in higher crack formation. Therefore, significant cracks were observed on the WPC50, which clearly indicates why it reduces mechanical properties. The LM images of weathered-WPCs; (a, b) HDPE, (c) WPC40, (d) WPC50, (e) IO40-1, (f) IO50-1, (g) IO40-10, (h) IO50-10.
On the other hand, iron oxide-reinforced composites significantly resisted extensive weathering conditions. Especially the surface character was well-preserved for IO40-1 and IO50-1. Meanwhile, there were cracks on the surface of WPCs containing a higher content of NPs and WF. As explained above, the decrease in the polymer content negatively weakened the adhesion between the materials. Moreover, the magnetic interactions between the iron oxide NPs might have adversely affected the structure. On the other hand, the color changes were significantly prevented compared to the crack formation.
Scanning electron microscopy analysis
The effect of iron oxide NPs on the weathering performance of WPCs was investigated with SEM microscopy, as seen in Figure 5. The SEM images comprehensively revealed the weathering effect on the surface of WPCs. Although the crack formation was not determined via light microscopy analysis in detail, there were micro cracks on the surface of HDPE. The degradation as a result of scission reactions with the effect of UV lights was clearly seen—moreover, WPCs without NPs significantly degraded during weathering exposure. Especially the degradation of the polymer resulted in the emergence of WF on the surface of WPC50 (white circle). Therefore, water molecules get attached to -OH groups and swell fibers, which enhances the crack formations. Meanwhile, increased NPs content reduced the crack formation but could not be inhibited. However, the increase in the NPs content with WF adversely affected the surface character, which caused crack formation. The SEM images of weathered-WPCs; (a, b) HDPE, (c) WPC40, (d) WPC50, (e) IO40-1, (f) IO50-1, (g) IO40-10, (h) IO50-10.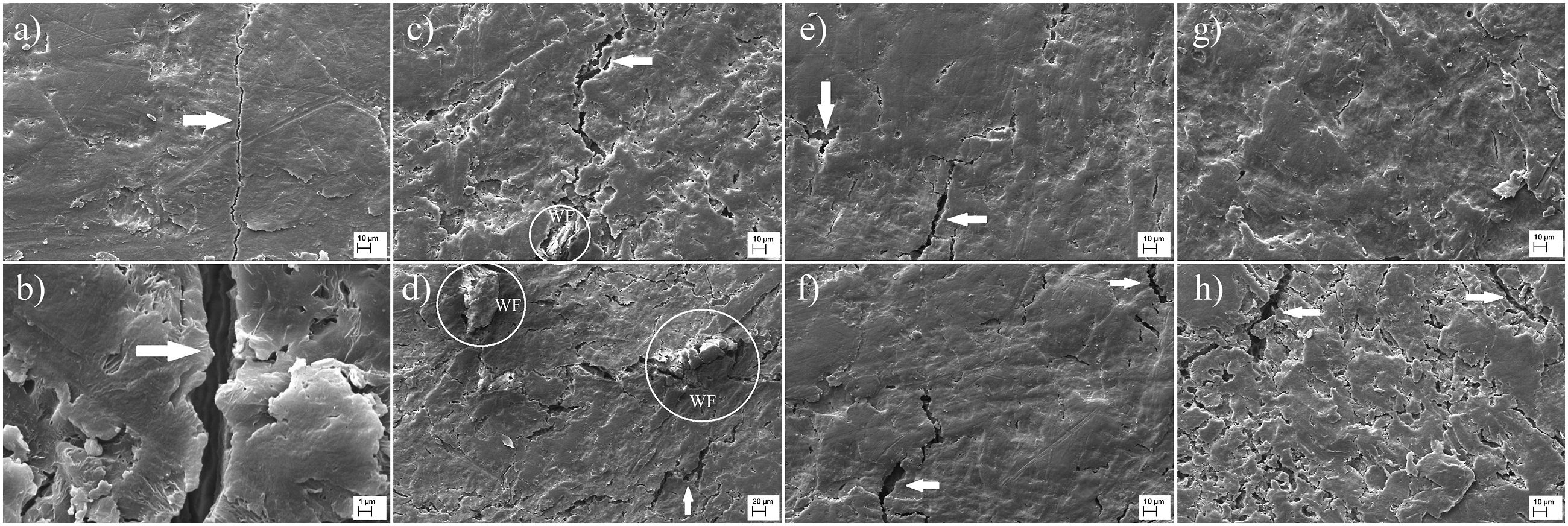
ATR-FTIR analysis
The effect of iron oxide NPs on the chemical changes on the surface of WPCs exposed to weathering conditions was investigated with ATR-FTIR analysis, as seen in Figures 6 and 7. The weathering conditions affected the intensities of wood and polymer-related bands. The bands at 2916 cm−1 and 2846 cm−1, recognized as characteristic bands of HDPE, were attributed to the asymmetric and symmetric C-H stretching of the polymer's chemical structure.31,32 The NPs did not affect these bands' intensities, indicating that iron oxide NPs physically dispersed in the polymer rather than bonding chemically. However, peak intensities were commonly weak due to hosting nanomaterials covering the active groups, indicating that NPs act as a shield. After weathering, these bands' intensities were higher for WPC40 and WPC50, while they were moderate for HDPE. UV lights triggered scission and crosslinking reactions in the chemical chain of HDPE.
33
As exposure time increased, the degraded surface was removed, and a new layer of polymer emerged, resulting in mild intensities of bands. However, micro-crack formations occurred on the surface of HDPE with increased UV exposure, also seen in SEM images. On the other hand, iron oxide NPs limited the degradation and preserved nearly all the surface chemistry with a barrier effect. However, there was also a slight change in these peaks' intensities due to oxidation reactions triggering the scission reactions, mainly responsible for decreasing mechanical properties, which were limited by the NP's UV shielding ability. The FTIR spectra of WPCs containing 40% WF. The FTIR spectra of WPCs containing 50% WF.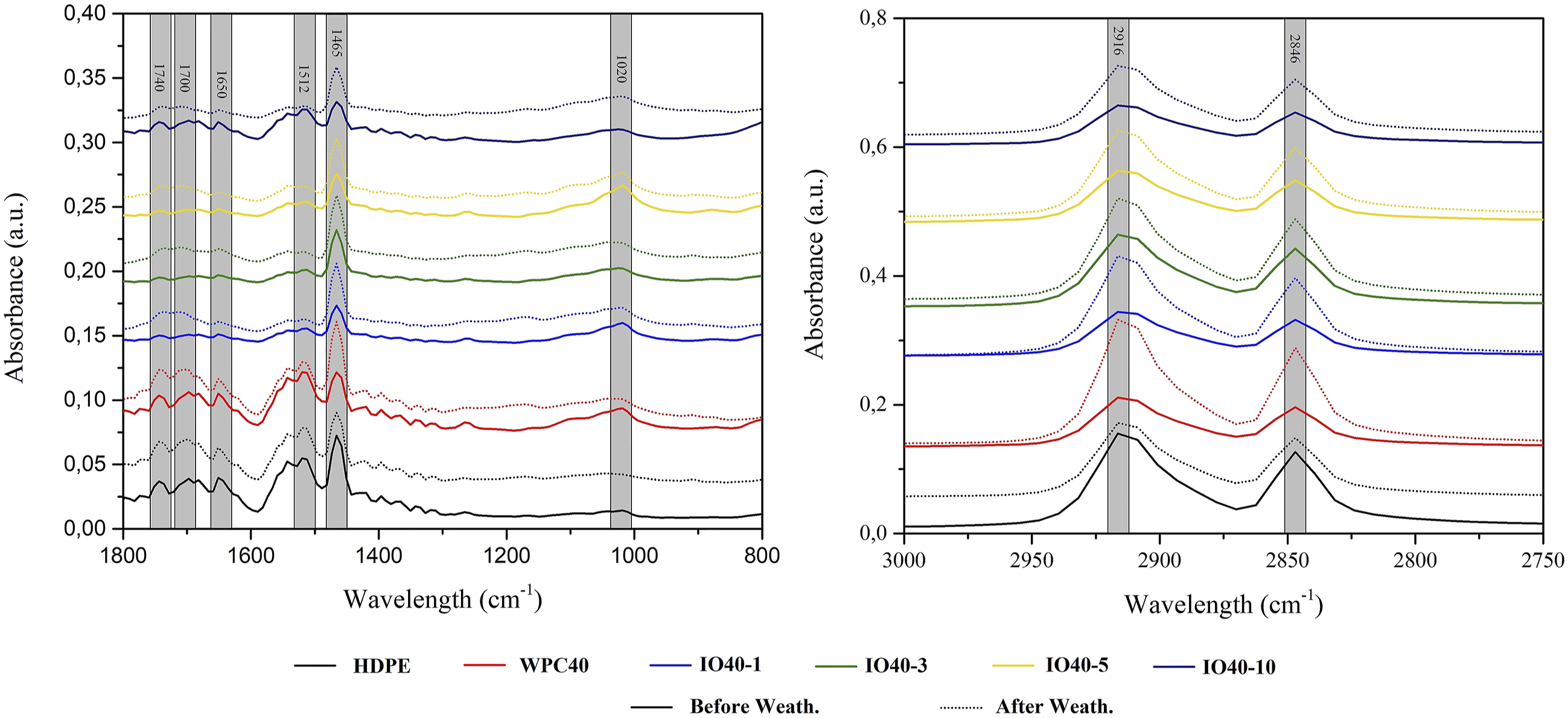
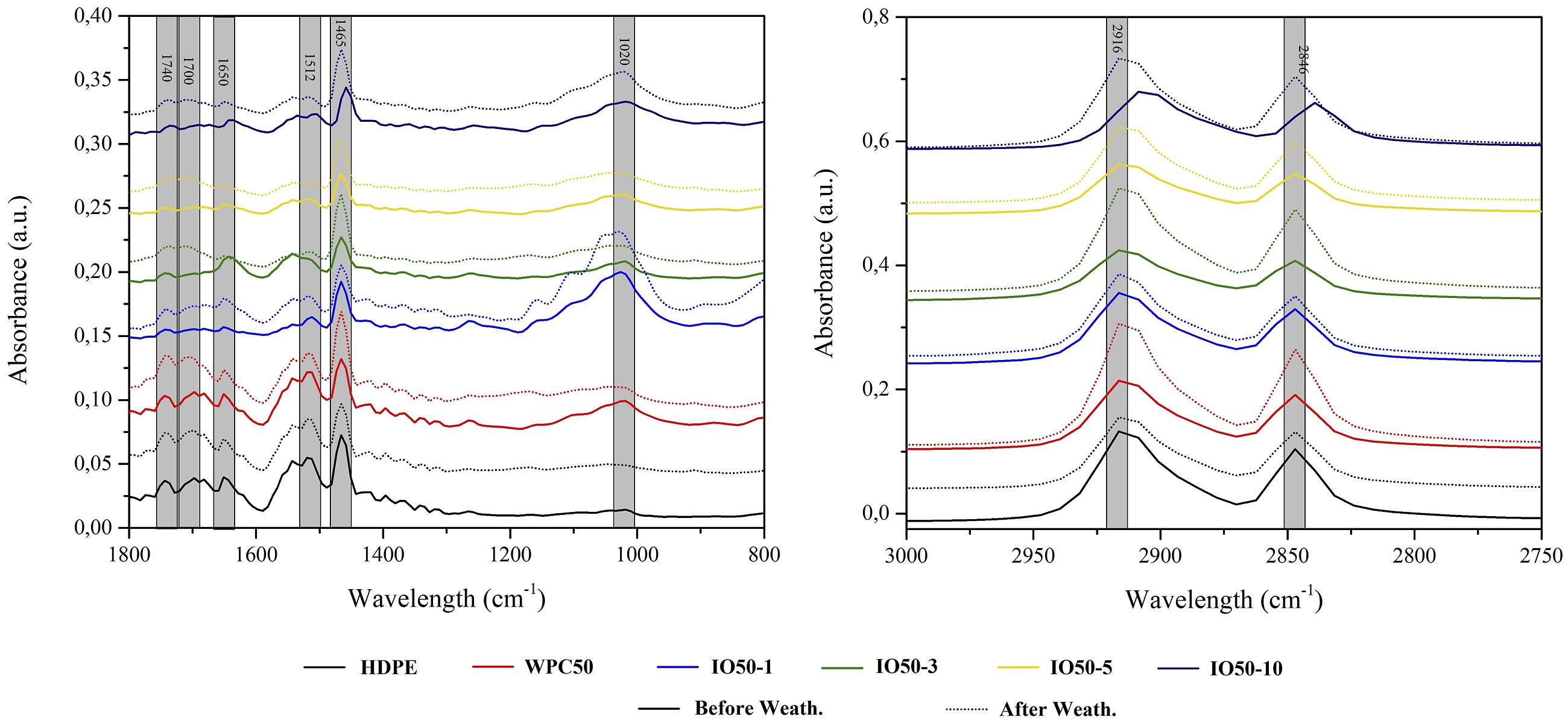
The spectra between 1800 cm−1 and 1600 cm−1 are identified in the carbonyl region. The band at 1740 cm−1 is associated with ester bonds, which is more intense for HDPE, WPC40, and WPC50, while there are no essential changes for WPCs containing NPs. The bands at 1700 cm−1 and 1650 cm−1 are related to the vinyl group and carboxylic acids, which were more moderate for NPs than for control samples.34,35 The UV lights trigger Norrish I and II reactions in the chemical chain of polymer in this region. Therefore, free radicals occur as a by-product of Norrish I, which causes scission reactions resulting in the shortening of the chain. Followed by Norrish I, carbonyl, and vinyl groups arise and enhance the cleavage of chemical chains, which increases the MOE due to the increased crystallinity of the polymer. Moreover, 1650 cm−1 also indicates water uptake in the cellulose responsible for fiber swelling. The polymer's chain shortening and cellulose degradation enhanced the crack formation, which limited the stress transfer and resulted in poor mechanical properties.
The characteristic lignin peak at 1512 cm−1 is responsible for the primary UV absorbance of wood. 1 Conjugated carbonyl groups, aromatic rings, and C=C bonds absorb UV light, which leads to color change from brown to black. 29 During the photodegradation, scission and crosslinking reactions compete with each other. The UV absorption by lignin’s chromophoric groups hinders the crosslinking reactions, which enhances the degradation intensity. Therefore, this band's intensities are higher for WPC40 and 50. However, the NPs in the polymer encapsulated the wood fibers and acted as a shield against UV lights, which inhibits and/or limits the degradation. However, with increasing wood content, the efficiency of NPs decreased. Especially the intensities of this band increased for IO50-10, although it was still lower than the control.
The band at 1465 cm−1 is attributed to both the amorphous polyethylene structure and the C-H deformation of hemicellulose and cellulose.32,36 As stated above, molecular chain shortening increased the chain mobility in the amorphous structure of HDPE, which decreased the band intensity for HDPE. On the other hand, the degradation of hemicellulose and cellulose increased the carbonyl moieties, sharpening the intensities of this band for WPC40 and 50. The increased carbonyl moieties revealed much more free-hydroxyl groups, which easily bond with water molecules. As a result, swelling fibers increased crack formation. This phenomenon was also monitored with microscopic analysis. As exposure time increased, carbonyl moieties leached and decreased the band intensity. However, NPs well-preserved the chemical structure as compared to control samples.
The peak at 1020 cm−1 is related to the C-O stretches of carbonyl moieties for cellulose and hemicellulose. 37 The intensities of this peak decreased for control samples, while there were no changes for NPs. UV lights degraded hemicellulose and cellulose, removing them from the surface and reducing this band’s intensity. The encapsulated fibers were persevered by polymer-absorbed NPs against UV lights, enhancing photo-stabilization.
Thermal properties
TGA analysis
The effect of iron oxide NPs on the behavior of gradual temperature increase was investigated, as seen in Figure 8. The degradation temperature was also given in Table 2. The onset and endset temperature ranged between 189.82 to 211.26°C. The degradation occurred in three stages for WPCs. The first stage continued up to ∼170 to 180°C, in which humidity in the matrix was released. Then the second stage was triggered by the effect of increased temperature, in which the main cell wall components (cellulose, hemicellulose, lignin) initiate the degradation.38,39 As well-known, hemicellulose degradation starts first, followed by cellulose at nearly 220–250°C, and finally lignin above ∼270°C.39–41 On the other hand, the polymer lost a substantial part of its weight between 330 to 500°C.
40
Most of the structure turned into paraffinic and olefinic compounds owing to scission reactions in the structure of HDPE, which results in weight loss.
42
Above 550°C, there was no residue due to oxidization to volatile compounds.
43
TGA and DTGA curves of WPCs. TGA and LOI values of WPCs. Note: The letters show the differences (p < 0.05) between groups depending on the Duncan test.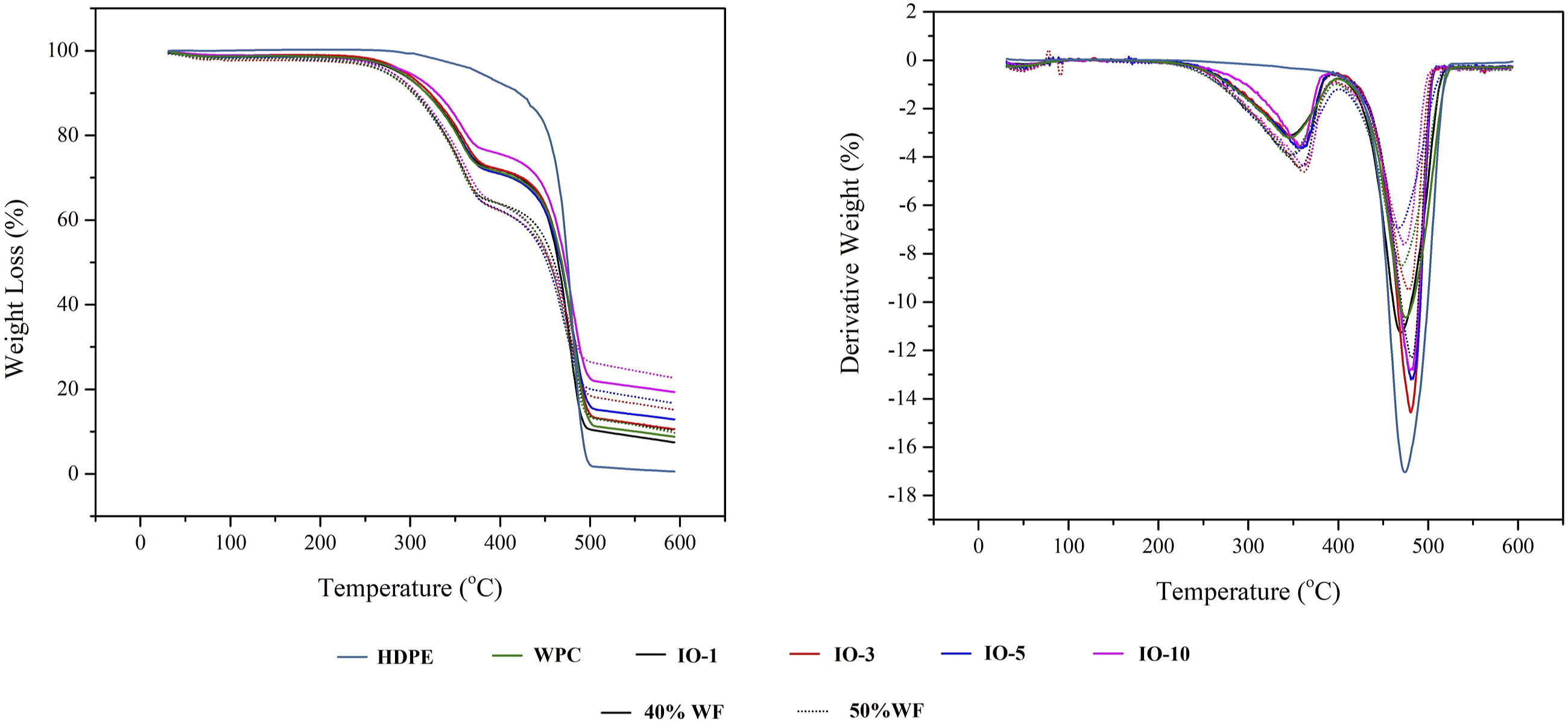
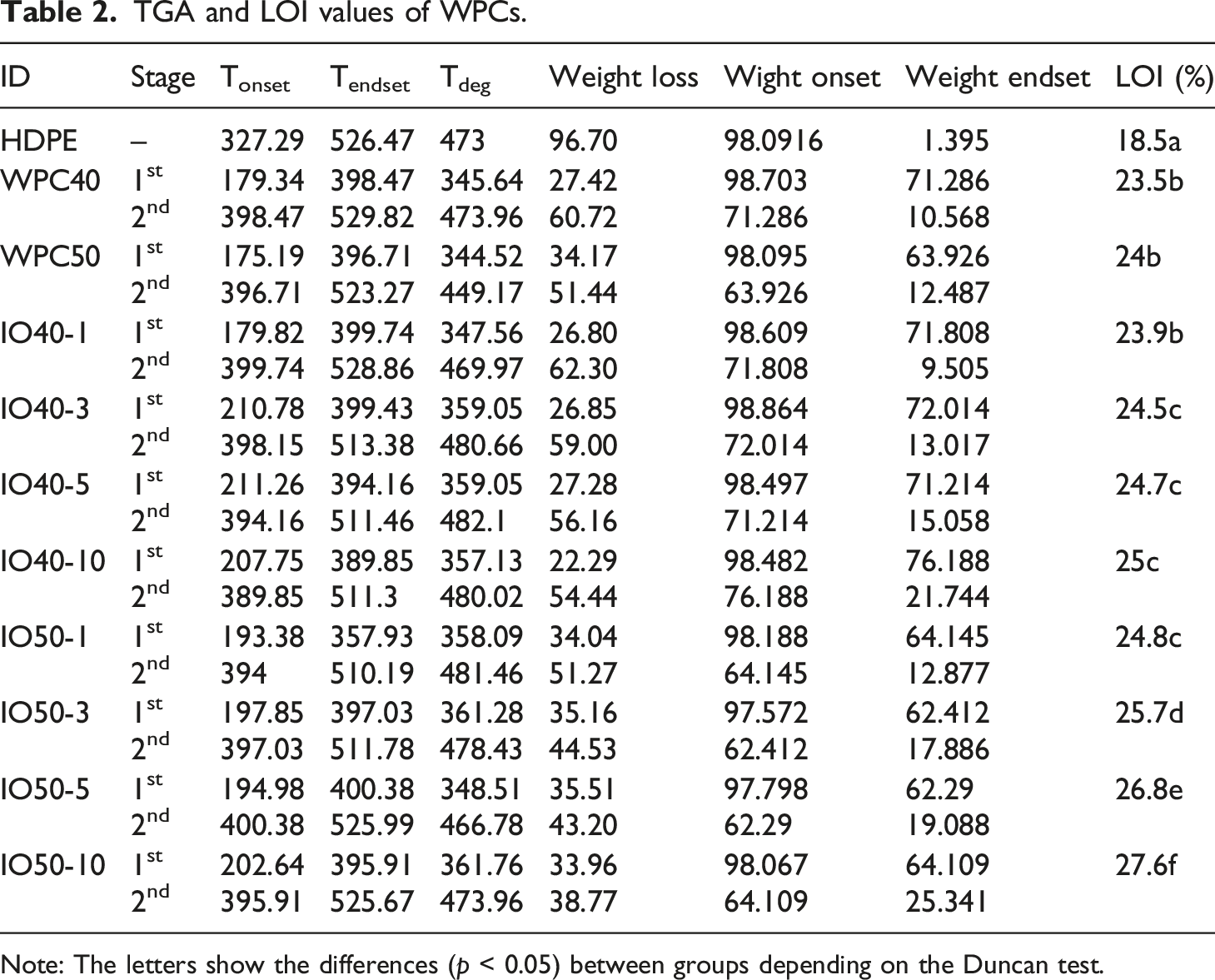
NPs dispersed in the polymer and acted as a heat barrier, cutting oxygen penetration from the surface to the core, which is crucial in preventing oxidation reactions. The barrier effect of iron-oxide NPs also diminished the heat transfer and reduced degradation velocity. Moreover, Jouyandeh et al. stated that the barrier effect increased with the NPs content. 44 The increase in the nanomaterials content closed the iron-oxide NPs, which covered the surface and created a relatively thick heat shield. On the other hand, lignocellulosic materials are recognized as having a heat-release rate-reducing effect. 45 Hence, IO50-5 slowed the degradation velocity, while it was fast for HDPE. However, IO50-10 had a slightly higher degradation velocity than IO50-5. Above the threshold content, NPs tend to agglomerate, increasing pathways for oxygen penetration, which could explain the slight increase in degradation. As a result, iron oxide slowed down the degradation and increased the residue formation, improving thermal stability.
LOI analysis
Oxygen is an essential component of combustion, which helps determine materials' fire resistance. The effect of iron oxide NPs on fire performance was determined with the LOI test, as seen in Table 2. The LOI values varied between 18.5 to 27.6%. When HDPE is exposed to heat, oxidation reactions occur in its chemical chains, revealing peroxy radicals and other oxidation by-products. 46 The oxygen reaction reduces the thermal stability of HDPE under ambient conditions, which is the main reason the polymer easily ignitions so that neat-HDPE is classified as “combustible material” (≤23) according to ISO 4589. 47 However, the carbonization of the wood fiber surface during the combustion limits oxygen penetration into the core and reduces the heat-release rate. Therefore, adding only WF to the polymer significantly improved the LOI values and categorized it as a “limited fire-retardant material.” Moreover, iron oxide nanomaterials restrained the oxidation reactions in HDPE, which reduced the weight loss of the polymer and improved the fire performance of WPCs. Meanwhile, Wang et al. also stated that metal oxide could promote char layer formation, which is crucial for inhibiting combustion. 48 However, NPs did not change the fire classification but significantly enhanced the LOI values. Furthermore, Dorigato et al. also highlighted that LOI values above 27 are known as “self-extinguishing”. 43
Conclusion
Nanomaterials have been preferred in most applications due to their superior properties. As stated above, iron-oxide NPs have been evaluated in various studies. However, there are a limited number of studies about iron-oxide NP-reinforced WPCs. In this study, the effect of iron-oxide NPs on the technological properties of WPCs was comprehensively investigated. NPs' hydrophilic nature improved WPCs' water uptake and dimensional stability, which are vital for outdoor applications. Meanwhile, there was an improvement for MOR of 10%, while MOE of 29% with the effect of the NPs despite the inconsistency between the wood and polymer. MOR and MOE of WPCs were still well-preserved even after weathering test. The decrease in MOR was 16% for control samples, while it was 2% for IO40-1. The magnetic interaction of NPs and reduced polymer adversely affected WPCs in higher wood and NPs content.
The color change is one of the main disadvantages of WPCs, which was limited by the ability of UV light absorption and/or reflection of iron oxide NPs. NPs inhibited UV light penetration with increasing content, thanks to covering the surface of the wood fiber. On the other hand, the microscopic analysis revealed significant cracks on the surface of the control samples, responsible for poor mechanical properties. NPs reduced the crack formation but could not inhibit it. The changes in the chemical structure of polymer and wood fibers enlighten the remarkable UV degradation. However, the chemical structure of the surface was well-preserved even through extensive weathering conditions. The ATR-FTIR spectra also depicted nearly no changes in the characteristic bands of wood (1512 cm−1) and polymer (2916 and 2846 cm−1), thanks to NPs.
NPs also acted as a heat shield, which reduced the degradation velocity during the gradual temperature increase and improved the degradation temperature. Moreover, the need for oxygen during the combustions for WPCs was also enhanced by up to 27.6%, which implied the improvement of the fire resistance of WPCs. Consequently, the improved dimensional stability, mechanical resistance, weathering durability, and thermal stability demonstrated that WPCs reinforced with iron oxide NPs could be considered for outdoor applications, such as decking, siding, furniture, playground, construction, etc.


