Abstract
Most of the polymers made from fossil fuels end up as waste material and this environment-damaging situation has revealed the need to take some precautions such as the use of eco-friendly, biodegradable materials obtained from renewable sources as an alternative. Biopolymers are being evaluated as alternatives to traditional polymers especially in the automotive industry due to their better-understood properties such as mechanical and physical behavior through the studies conducted. On the other hand, when the literature studies are evaluated, it can be seen that the production of foam materials, which are focused on lightness and carbon dioxide (CO2) emission limitations that are important and essential for the automotive sector, is an area that is also researched and studied for biopolymers. Several studies are carried out with both physical and chemical foaming agents on this subject in recent years. In this study, it is aimed to develop light, environmentally friendly, high-performance polylactic acid (PLA) based polymeric composite foams that can be used in the automotive industry by using twin screw extruder and compression molding methods. For this purpose, polyamide 6 (PA6) and polypropylene (PP) polymers were used with PLA and by adding 1 wt.%, 1.5 wt.% and 2 wt.% chemical blowing agents to the polymer mixtures, the physical, thermal, mechanical, morphological properties and changes in these properties were investigated. The most suitable chemical blowing agent ratio was found to be 1.5 wt.% for PLA/PP and PLA/PA6 mixtures.
Keywords
Introduction
While recycling activities started to become popular in the mid-1990s, the fact that oil prices, which had been stable for many years, started to rise rapidly in the early 2000s, also reflected on the prices of petroleum based polymer raw materials. These developments have revealed the need to develop raw materials that can be utilized as a replacement to traditional polymers. The development of biopolymers has become one of the most significant investigations of the last decade. Particularly, in the automotive, health, electronics and transportation areas the properties of these materials have been proved to be very improved besides the need of the reduction of CO2 emissions. As a result, it can be said that this new class of composites has allowed the apparition of a recent category of biocomposites with enhanced properties using various additives and polymers.1,2
One of the most largely utilized biopolymer is polylactic acid which is an aliphatic polyester used in various areas and has similar properties compared to traditional polymers with higher mechanical parameters such as high hardness and high tensile strength and ease of processing.1–3 However, outside of its benefits, some shortcomings can also be observed such as low toughness, high price, low thermal properties.4–6 In order to overcome this situation, various polymers and additives can be used for the development of composite materials.
The amelioration of the weak properties of pure polymers can be possible with the production of polymer mixtures which is the common way and in some situations, more developed properties can be obtained compared to pure polymers. Among the engineering plastics, PP and PA6 having improved properties can be mixed with PLA and preferred to generate a recent category of biocomposites with higher features such as especially mechanical strength chemical resistance.7–9
PA6 and PP polymers have been widely used for years in various areas and their mixtures with PLA as a biopolymer have become important. While PA6 is a polymer mainly used for its high mechanical strength, good abrasion, rigidity and stability, PP is well-known and widely used for its enhanced processability, impact resistance, stiffness and also low cost properties.10–12
In recent works, due to the increase of CO2 gas emission and according to the regulations, foaming studies appeared for all class of polymers including biopolymers, and especially the foaming behavior of PLA, in order to obtain lightweight structures. However, during foaming processes cell gathering for PLA foams can be observed owing to the slow crystallization speed and the poor melt strength, engendering a low foaming tendency of PLA.13,14 Final foaming properties such as cell density, closed-cell foams with constant structure or a growth speed which is more controlled can be enhanced importantly during extrusion and injection processes especially with the use of foaming agents.15–18 Improving PLA-based foamed composites has become an important issue, particularly in the automotive sector for the production of lightweight parts. For this purpose, by working on the production of foam systems of composite structures of PLA with both biopolymers and petroleum based polymers, on the one hand, PLA’s weak properties are improved with composite structures, on the other hand, the preparation of foam structures helps both to gain lightness and to reduce costs and CO2 emissions with the use of less raw materials.15,16
In this study, it was aimed to prepare biopolymer mixtures as a replacement to traditional polymers that can be used in the automotive industry and to produce polymeric composite foams by using chemical foaming agents by compression molding method. Although the lightness issue has gained importance in recent years, studies have started with industrial processes such as injection and extrusion, but there are difficulties and limitations in carrying out small-scale research studies as these equipments require high investment, cost and labour. Therefore, the propose of this study is to carry out foaming studies using compression molding due to both ease of process and easy accessibility, and to evaluate the properties such as mechanical and morphological features of foam structures prepared using chemical foaming agents. The use of compression molding method in the manufacturing of PLA-based polymer composite foam structures developed in this study, a different foam preparation technology was developed by making lightening studies with the chemical foaming method, and both weight and CO2 emissions reduction can be achieved in accordance with the regulations. In this sense, the present work aims to provide chemical foaming of the blends by adding different ratios of foaming agent to the mixtures prepared with the use of PP and PA6 polymers together with PLA and to determine the most appropriate foaming agent ratio through the characterization of the thermal, density, mechanical and microstructure features of the materials.
Experimental procedure
Materials
PLA through the tradename Luminy L175 was supplied from Total Corbion/China (density: 1.24 g/cm3 and melt flow index: 8 g/10 min (210°C, 2.16 kg)). PP, through the tradename Buplen was provided from Lukoil Petrochemical Company/Russia (density: .91 g/cm3 and melt flow index: 7 g/10 min (230°C, 2.16 kg)). PA6 (Domamid 27) was purchased from Ultra Polymers/Germany (density: 1.14 g/cm3). The chemical blowing agents (CBA) used with PLA/PP and PLA/PA6 mixtures (Hyd 812) and Hyd 822) were supplied from Clariant.
Preparation of polymer blends
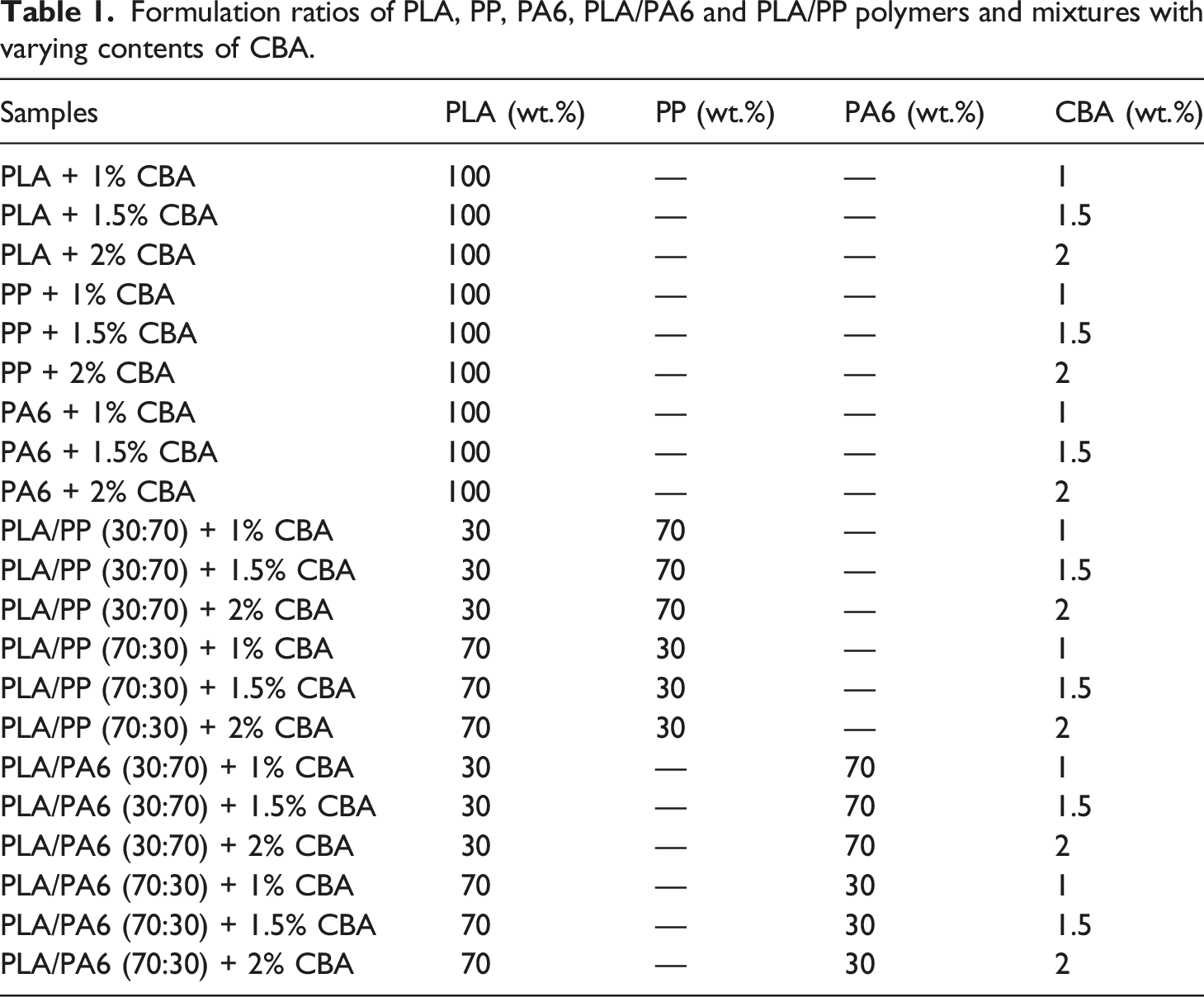
Formulation ratios of PLA, PP, PA6, PLA/PA6 and PLA/PP polymers and mixtures with varying contents of CBA.
Characterization methods
Density analysis
Density measurements were carried out using a precision balance (Shimadzu/AUX321) composed of a density measurement part. The detection of the density (
Molecular spectroscopy analysis
The measurements of Fourier transform infrared (FT-IR) spectroscopy were carried out through a spectrophotometer (Thermoscientific/Nicolet-IS50). The functional groups in the polymeric structures were obtained with measurements through a diamond crystal (4000-600 cm−1).
Tensile test
Tensile tests of mixtures were conducted using a universal testing device (Shimadzu/AGS-X) (force: 10 kN, tensile speed: 5 mm/min, 20°C). Various parameters related to tensile properties were analyzed according to ASTM D638 as the result of five replicate measurements.
Thermal analysis
Thermal evaluations of polymer mixtures were performed with a TA Instrument/DSC25 differential scanning calorimeter. Thermal parameters such as the melting temperature (Tm), the glass transition temperature (Tg) and the cold crystallization temperature (Tc) of the different mixtures and polymers, heat-cool-heat techniques was applied under nitrogen gas at constant flow rate. The samples were analyzed between 20°C to 250°C at a heating speed of 5°C/min. The crystallinity ratio (
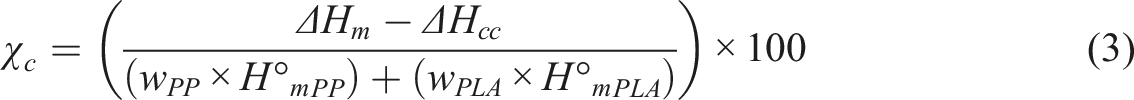
In equation (3) which was used for PLA/PP mixtures,
Morphological analysis
A Carl Zeiss (Gemini 300) scanning electron microscopy (SEM) was utilized in order to evaluate the microstructure of polymer mixtures (5 kV and ×1000 magnification). Before to imaging, the polymer mixtures were covered with 15 nm Gold/Palladium (Au/Pd) alloy using a coating device (Leica/ACE600) to prevent charging artefacts. Microscope pictures of the polymeric foam composites with a magnification of ×25 were detected using a Carl Zeiss/Discovery V12 microscope.
Results and discussions
Density change of polymer mixtures
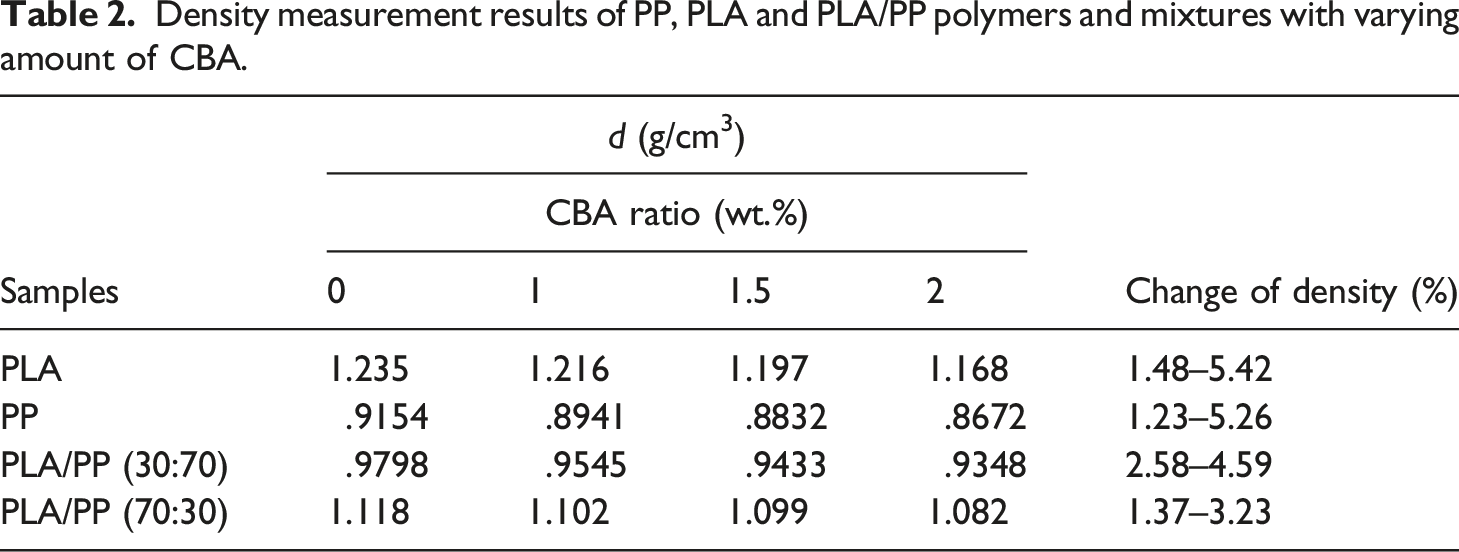
Density measurement results of PP, PLA and PLA/PP polymers and mixtures with varying amount of CBA.
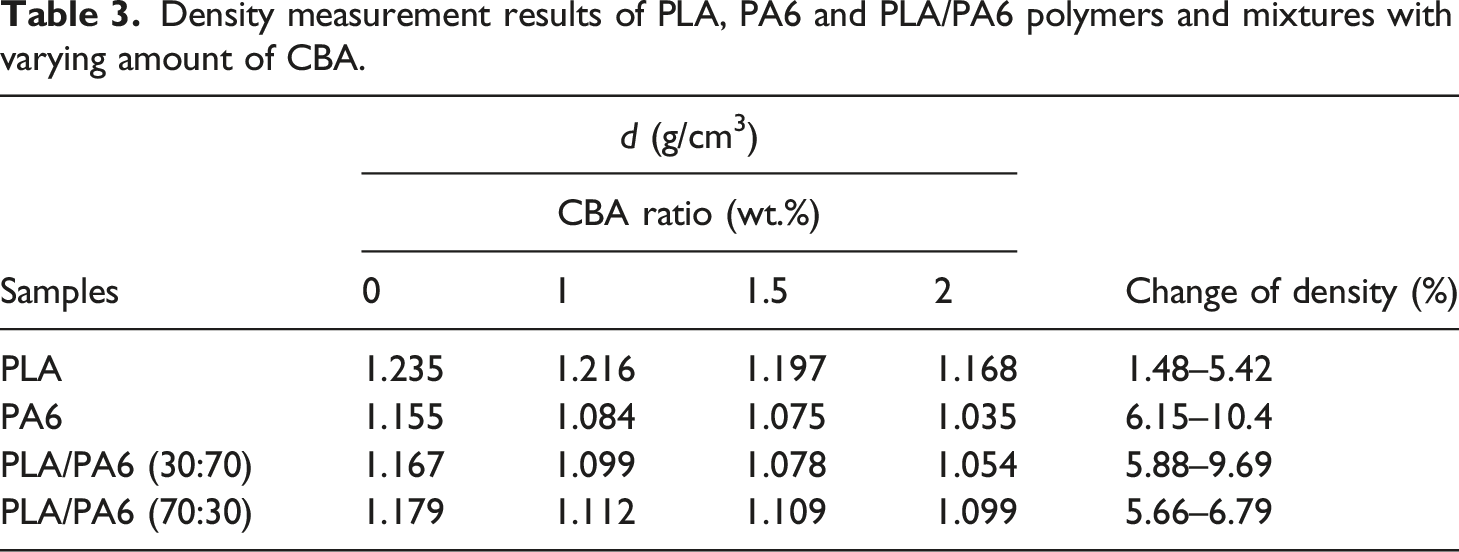
Density measurement results of PLA, PA6 and PLA/PA6 polymers and mixtures with varying amount of CBA.
Molecular spectroscopic analysis of polymer mixtures
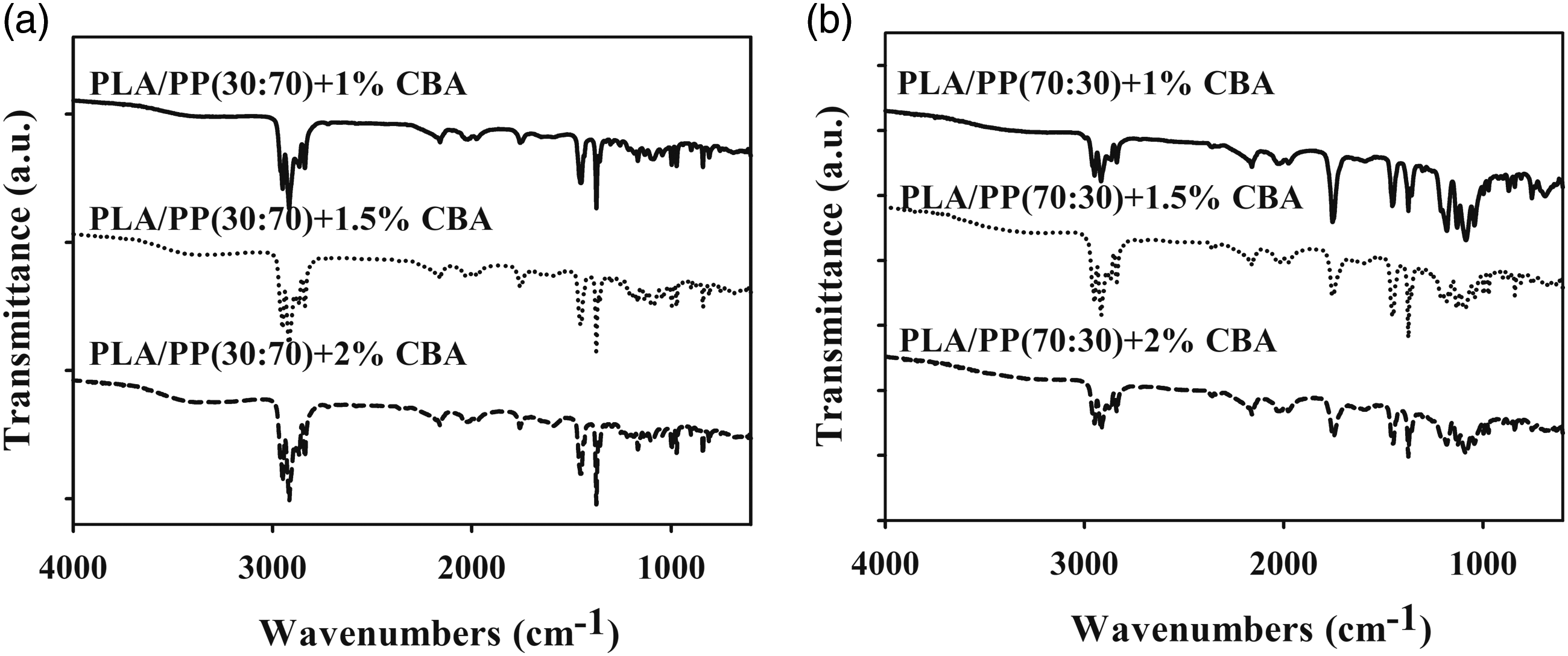
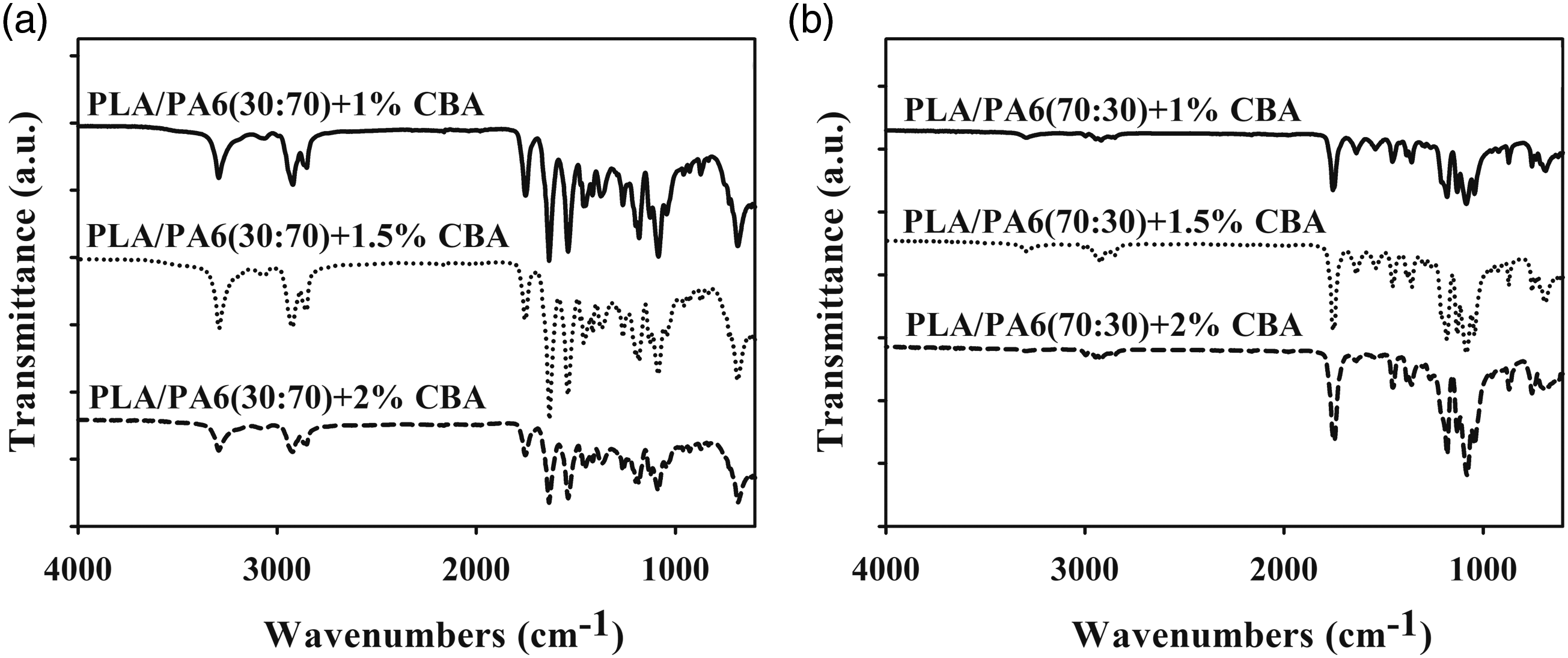
The FT-IR results of neat PP, PA6 and PLA polymers with PLA/PA6 and PLA/PP polymer mixtures containing different ratios of CBA in (1, 1.5 and 2 wt.%) were given in Figures 1–3. Evaluation of the FT-IR spectrum of pure PLA containing CBA (Figure 1(a)) reveals that an asymmetric CH3 peak at of 1080 cm−1, a symmetric C-O-C stretched peak detected at 1180 cm−1 and C=O stretched peak obtained at 1750 cm−1 can be sighted. It could be expressed that the characteristic peaks of neat PP with CBA (Figure 1(b)) at 1376 cm−1, 1455–1453 cm−1 and 2950–2838 cm−1 were related to -CH3 bending, C-H bending and C-H stretching, respectively. The characteristic peaks of pure PA6 containing CBA (Figure 1(c)) (1632 cm−1, 3300 cm−1 and 2931-2865 cm−1) are observed related to N-H stretching, C=O stretching and C-H stretching. Also, the intensities of the C=O stretch at the 1750 cm−1 band, the C-O-C stretch at the 1180 cm−1 band, and the asymmetric CH3 peak at the 1080 cm−1 band of PLA are significantly different from PLA/PA6 and PLA/PP polymer mixtures with varying amount of PLA (Figures 2 and 3). These FT-IR spectra indicated that the PLA, PP and PA6 polymers in the polymer mixtures continued to exhibit their specific properties observed in their neat form and did not give highly compatible structures in the mixture.
23
As could be seen in Figures 1–3, since CBAs used with PP, PA6 and PLA polymers with PLA/PP and PLA/PA6 polymer mixtures do not have characteristic peaks other than the C-H peaks present in polymers, these differences did not occur in the spectrum of neat polymers containing CBA.7,24,25 FT-IR results of (a) PLA, (b) PP and (c) PA6 polymers containing different ratios of CBA. FT-IR results of (a) PLA/PP (30:70) and (b) PLA/PP (70:30) polymer mixtures containing different ratios of CBA. FT-IR results of (a) PLA/PA6 (30:70) and (b) PLA/PA6 (70:30) polymer mixtures with varying content of CBA.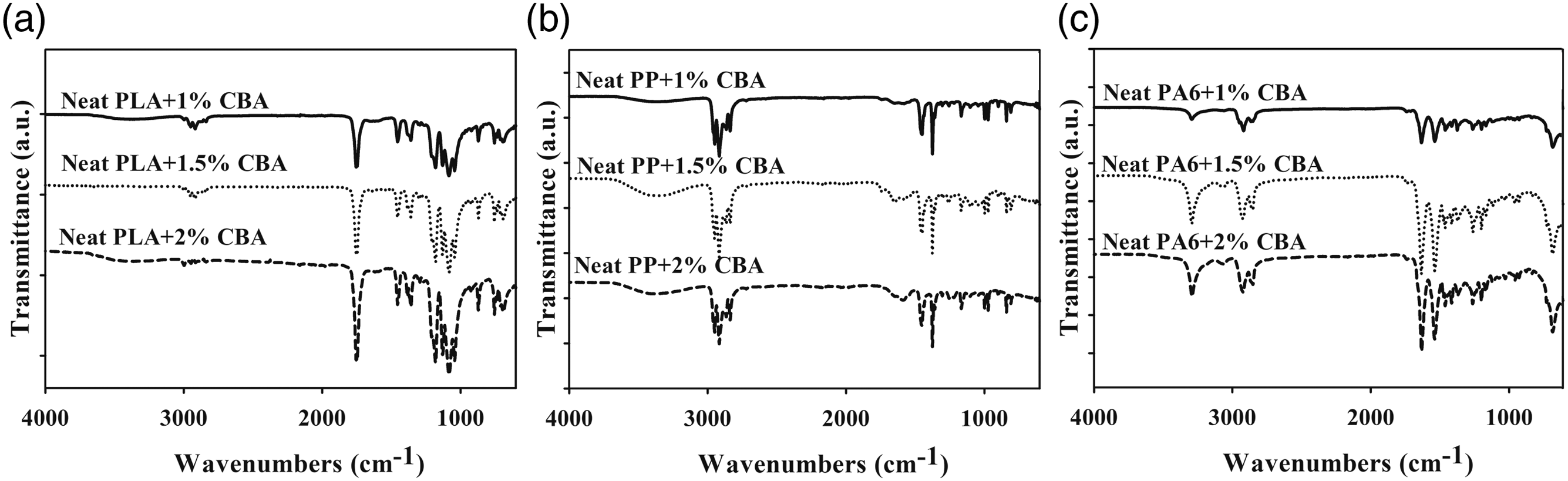
Mechanical properties of polymer mixtures
Tensile test parameters of PLA, PP and PLA/PP polymers and mixtures with varying content of CBA.
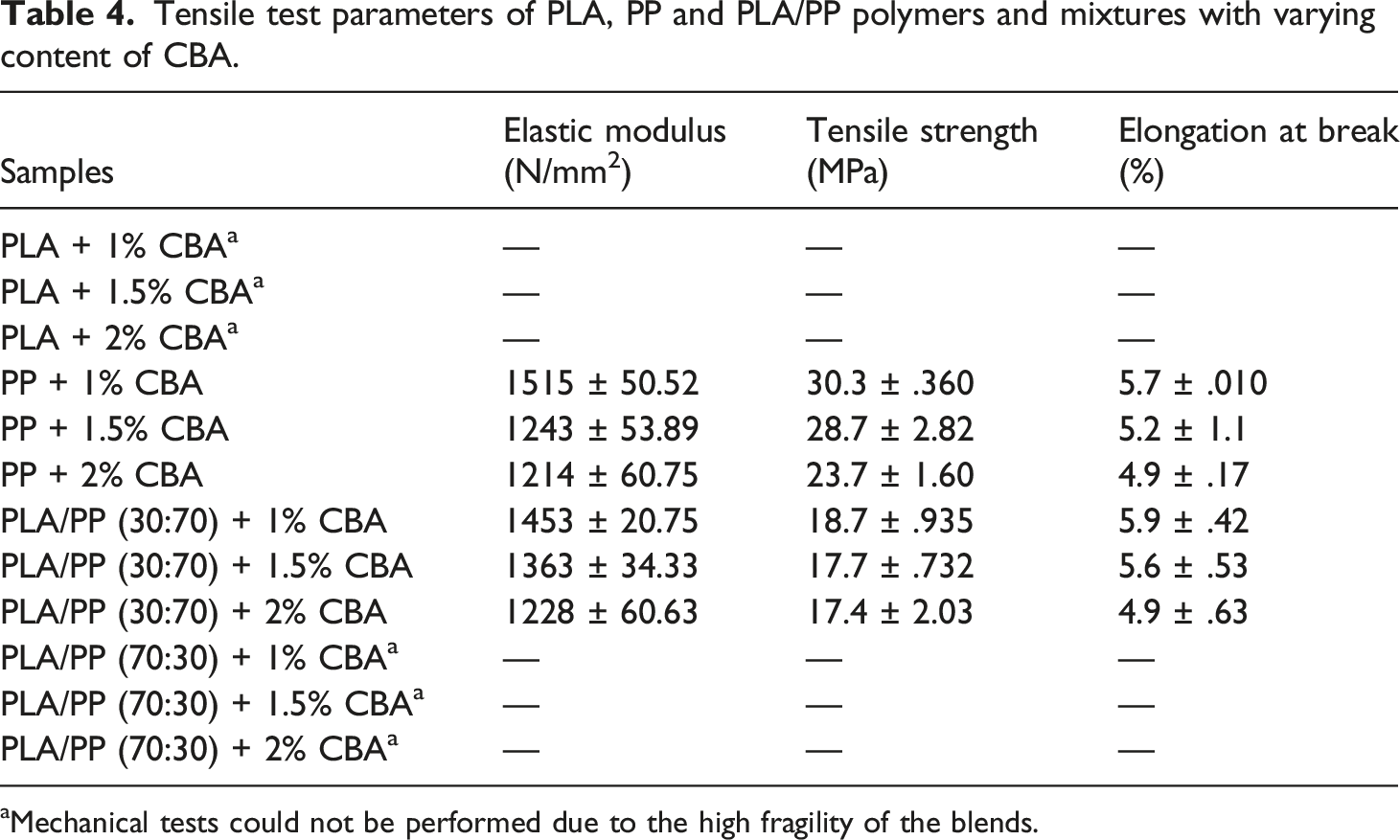
aMechanical tests could not be performed due to the high fragility of the blends.
Tensile test results of PA6 polymer and PLA/PA6 mixtures containing different ratios of CBA.
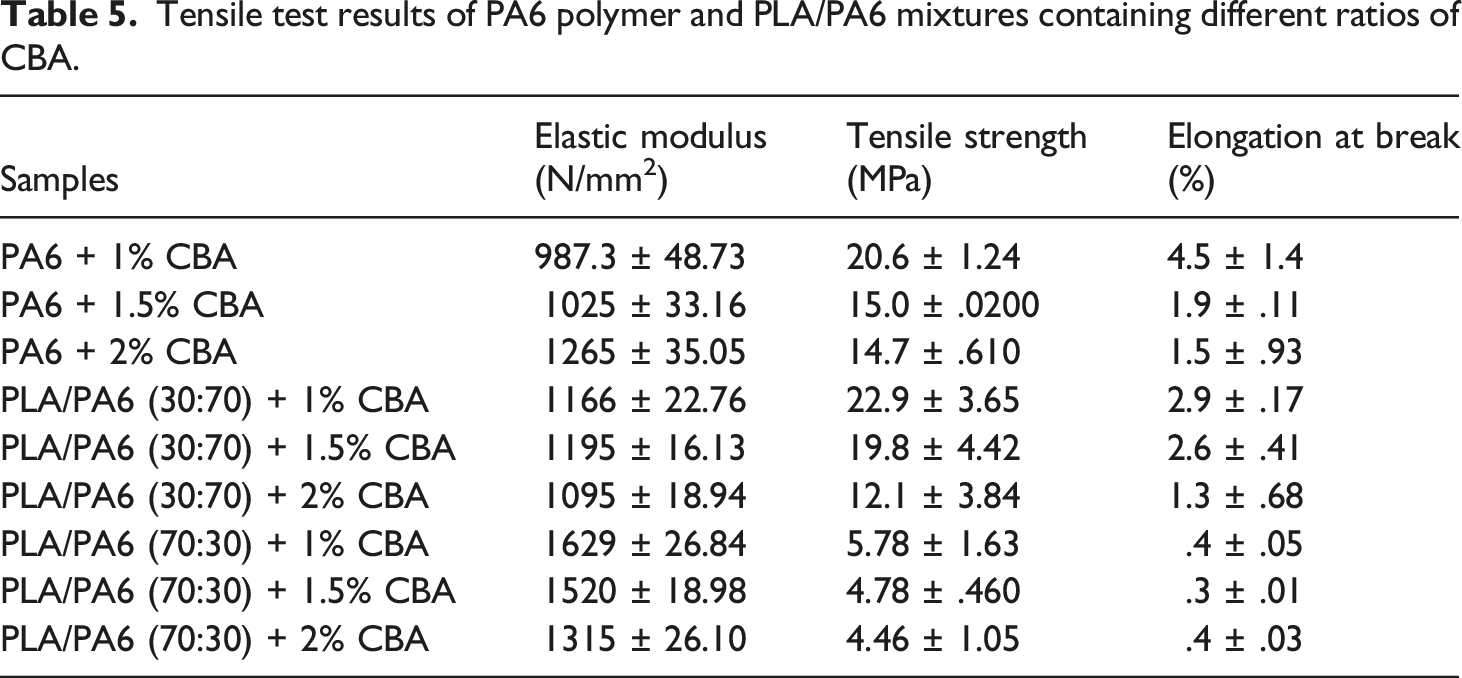
The variations in the tensile parameters of PLA/PP (30:70) polymer mixtures with varying content of CBA.
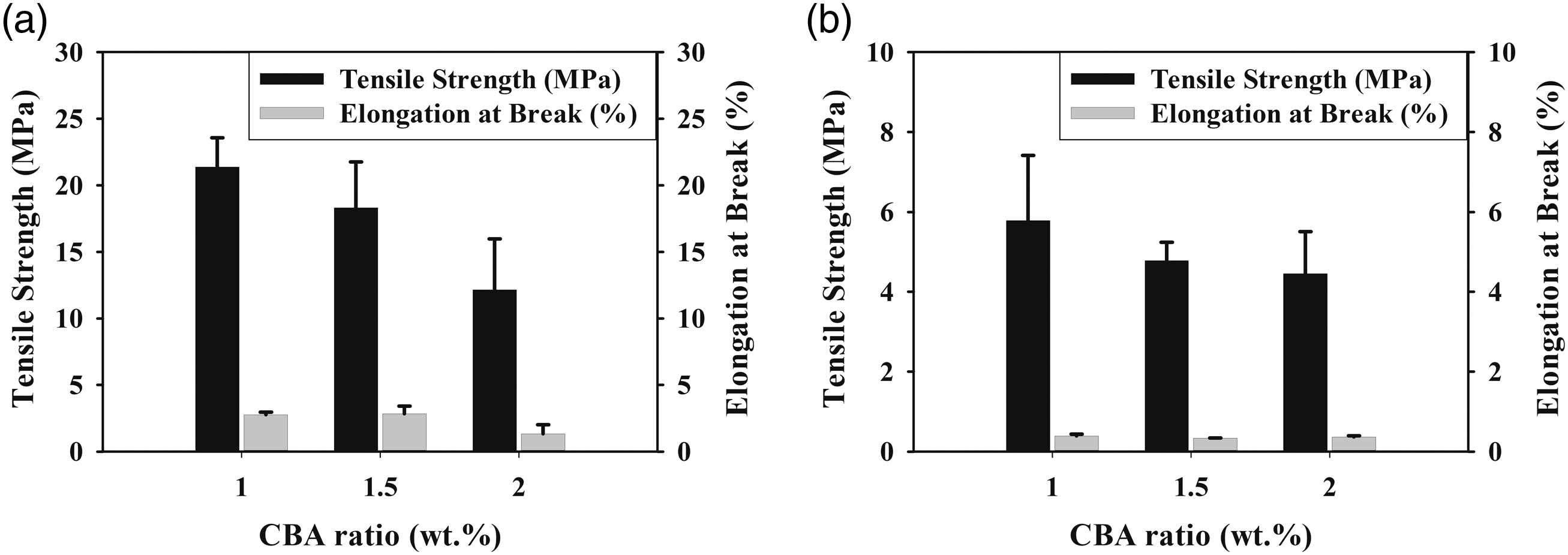
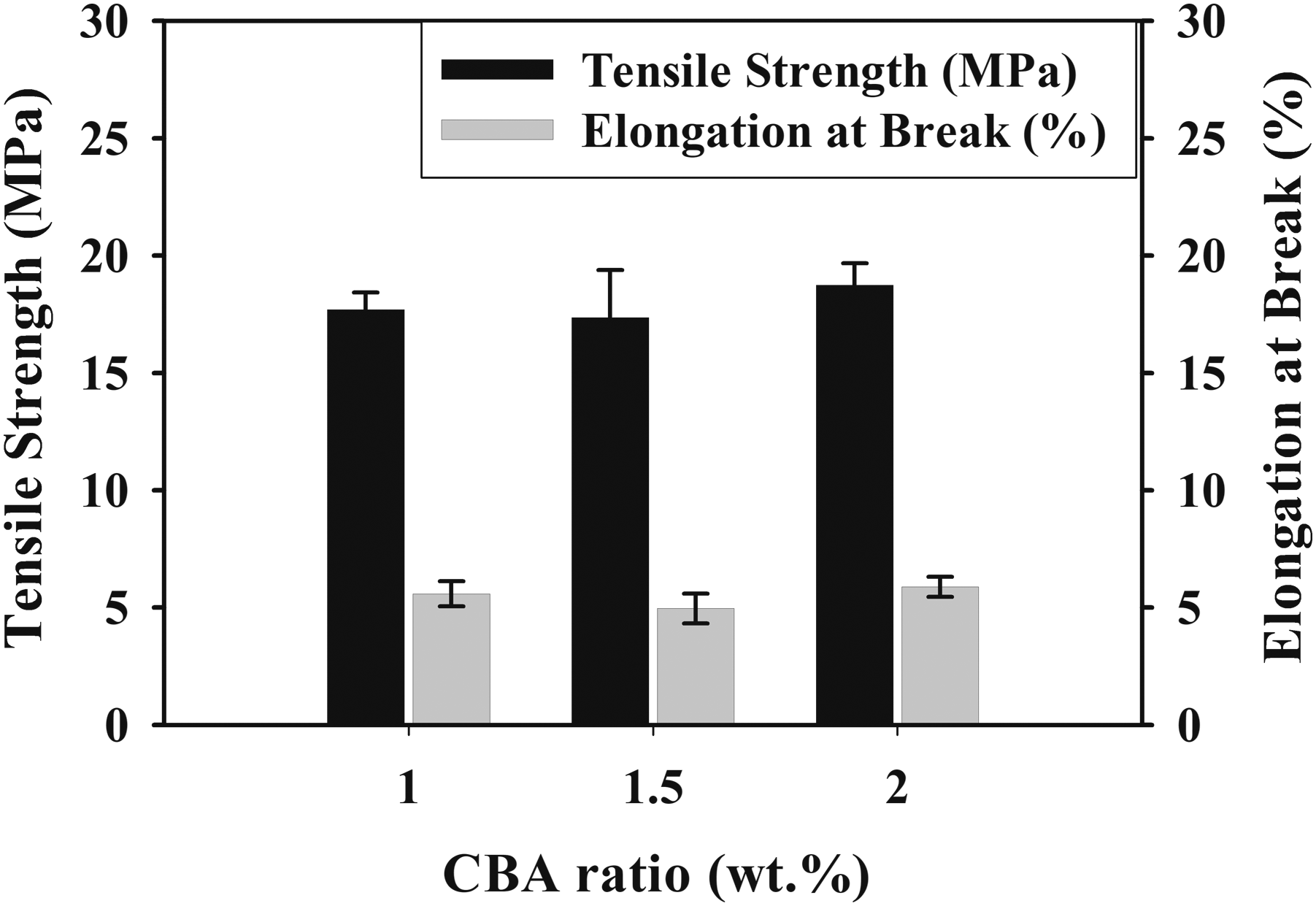
The variations in the tensile parameters of (a) PLA/PA6 (30:70) and (b) PLA/PA6 (70:30) polymer mixtures with varying content of CBA.

SEM and stereo microscope images of (a) PP + 1% CBA, (b) PP + 1.5% CBA and (c) PP + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively.

SEM and stereo microscope images of (a) PLA/PP (30:70) + 1% CBA, (b) PLA/PP (30:70) + 1.5% CBA and (c) PLA/PP (30:70) + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively.
It was observed that the tensile test results of PA6 polymer and PLA/PA6 polymer mixtures with varying content of CBA decreased similarly to PLA/PP polymer mixtures. Table 5, Figures 8 and 9 reveal that the materials become more brittle due to the cellular structure formed in the mixtures and that the elongation at break and tensile strength values especially decrease as the CBA ratio in polymer mixtures increases. While the reduction in the tensile strength values of PA6 mixtures containing different CBA was between 20.09% and 42.80%, this decrease varied between .2% and 47.14% in PLA/PA6 (30:70) polymer mixtures. On the other hand, it was seen that the reduction in tensile strength values varied between 65.68% and 73.57% for PLA/PA6 (70:30) mixtures and the structure became extremely brittle as a consequence of the mechanical properties that were severely weakened by the addition of CBA to these mixtures which contains a high percentage of PLA. It can be concluded that the decrease in mechanical properties that occur in general, the rise of the amount of cells depending on the CBA ratio used in the mixtures and the decrease in the shell thicknesses may cause the weakening in the mechanical properties. In this sense, the decrease in the shell layer (Figures 9 and 10) as a result of the evolutions of the cell diameter and cell numbers, the stress concentrations created by the cells may generate a drop in the tensile strength of the mixtures. These results are in correlation with the literature and especially the lower internal gas pressure of smaller cells generate higher mechanical properties.26,27 SEM and stereo microscope images of (a) PA6 + 1% CBA, (b) PA6 + 1.5% CBA and (c) PA6 + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively. SEM and stereo microscope images of (a) PLA/PA6 (30:70) + 1% CBA, (b) PLA/PA6 (30:70) + 1.5% CBA and (c) PLA/PA6 (30:70) + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively. SEM and stereo microscope images of (a) PLA/PA6 (70:30) + 1% CBA, (b) PLA/PA6 (70:30) + 1.5% CBA and (c) PLA/PA6 (70:30) + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively.


Thermal parameters of polymer mixtures
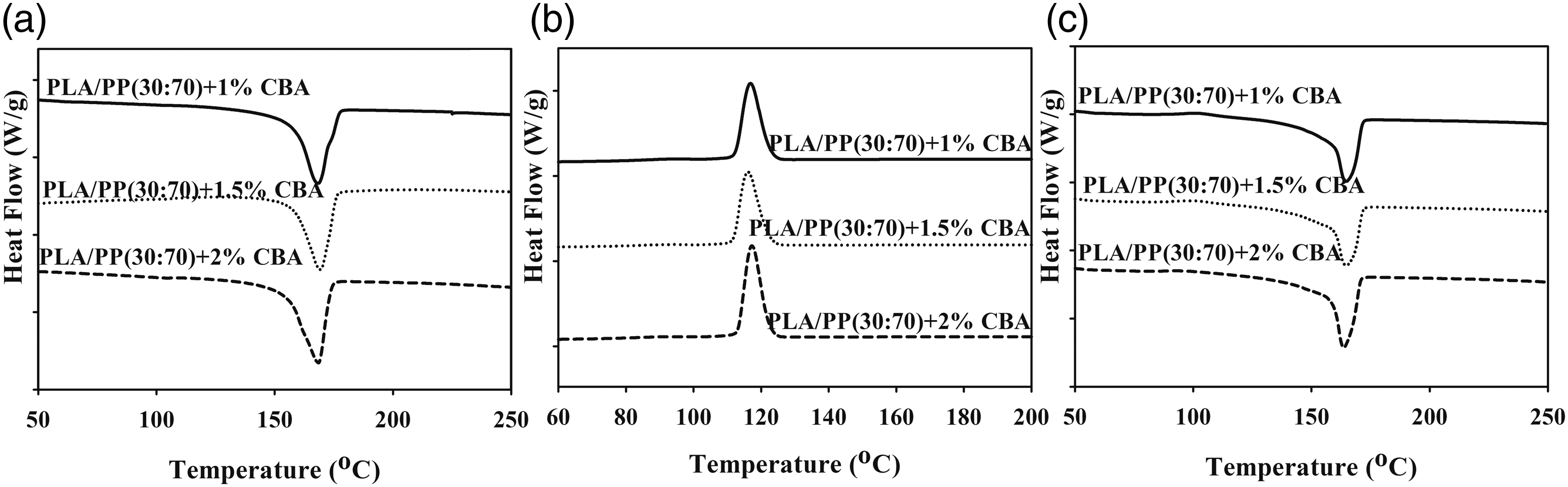
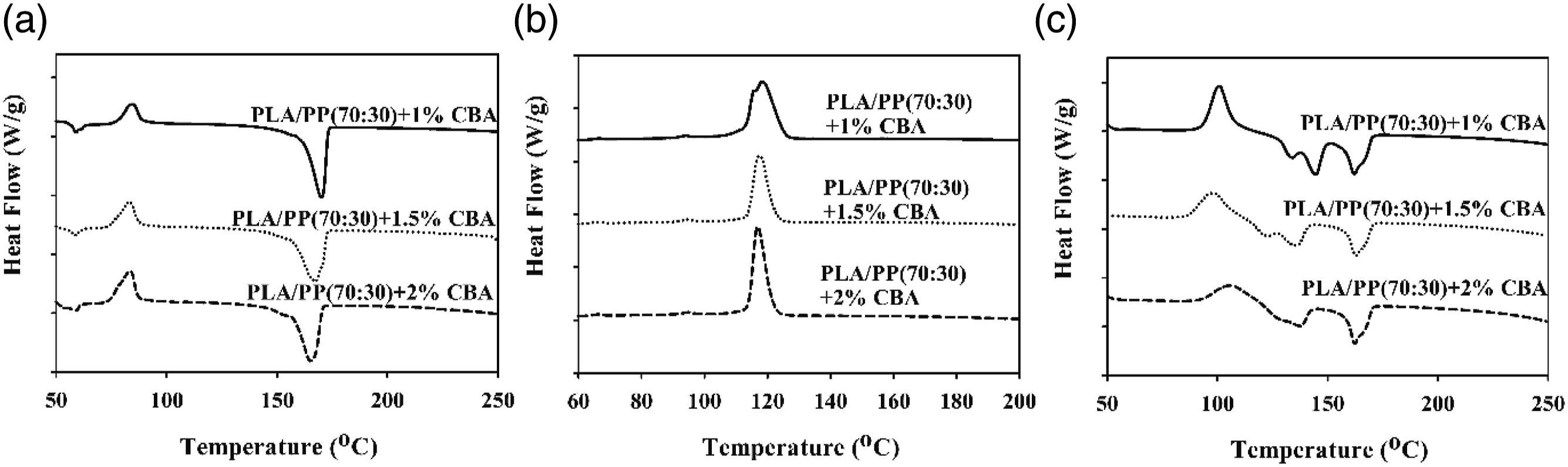
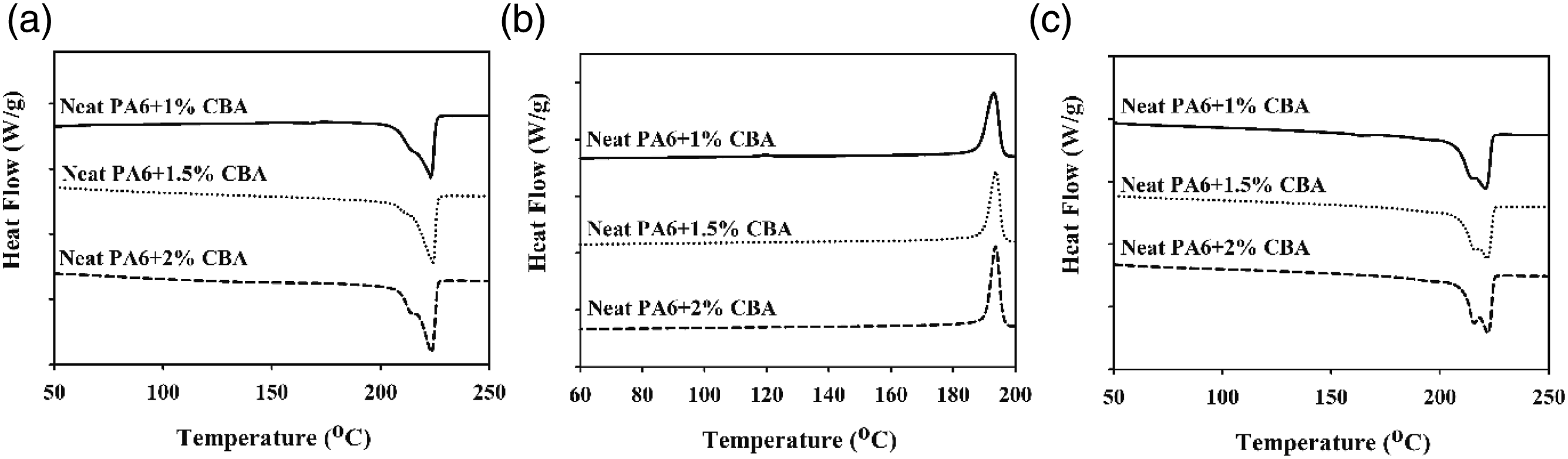
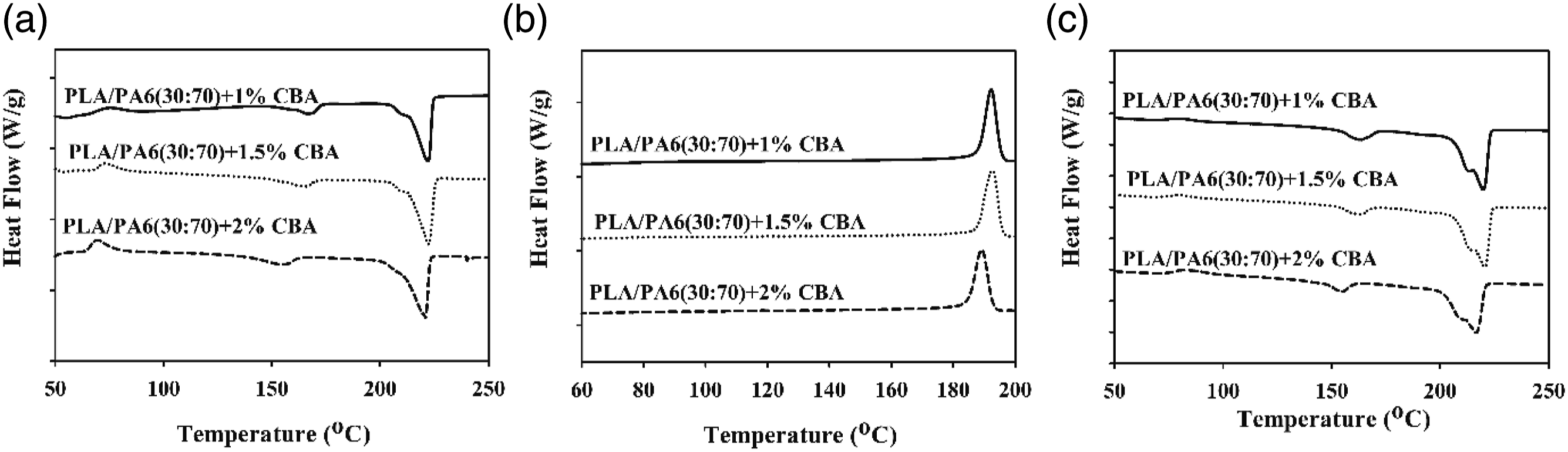
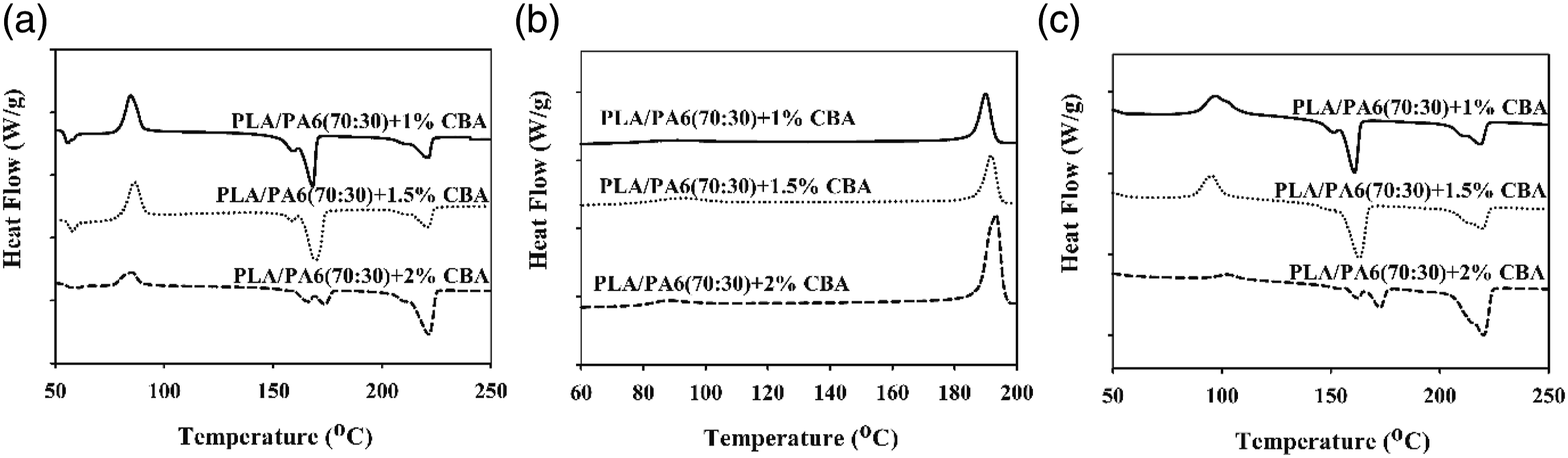
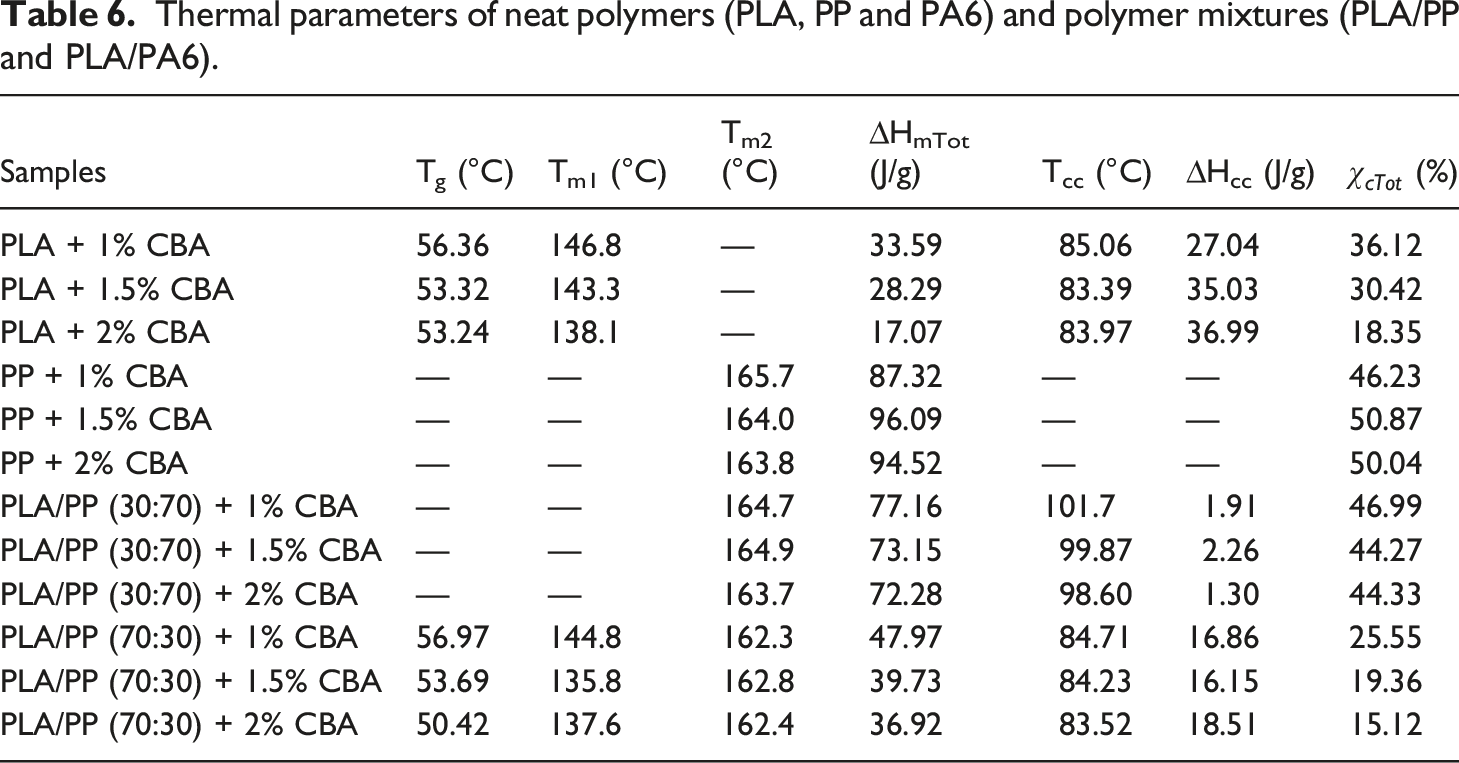
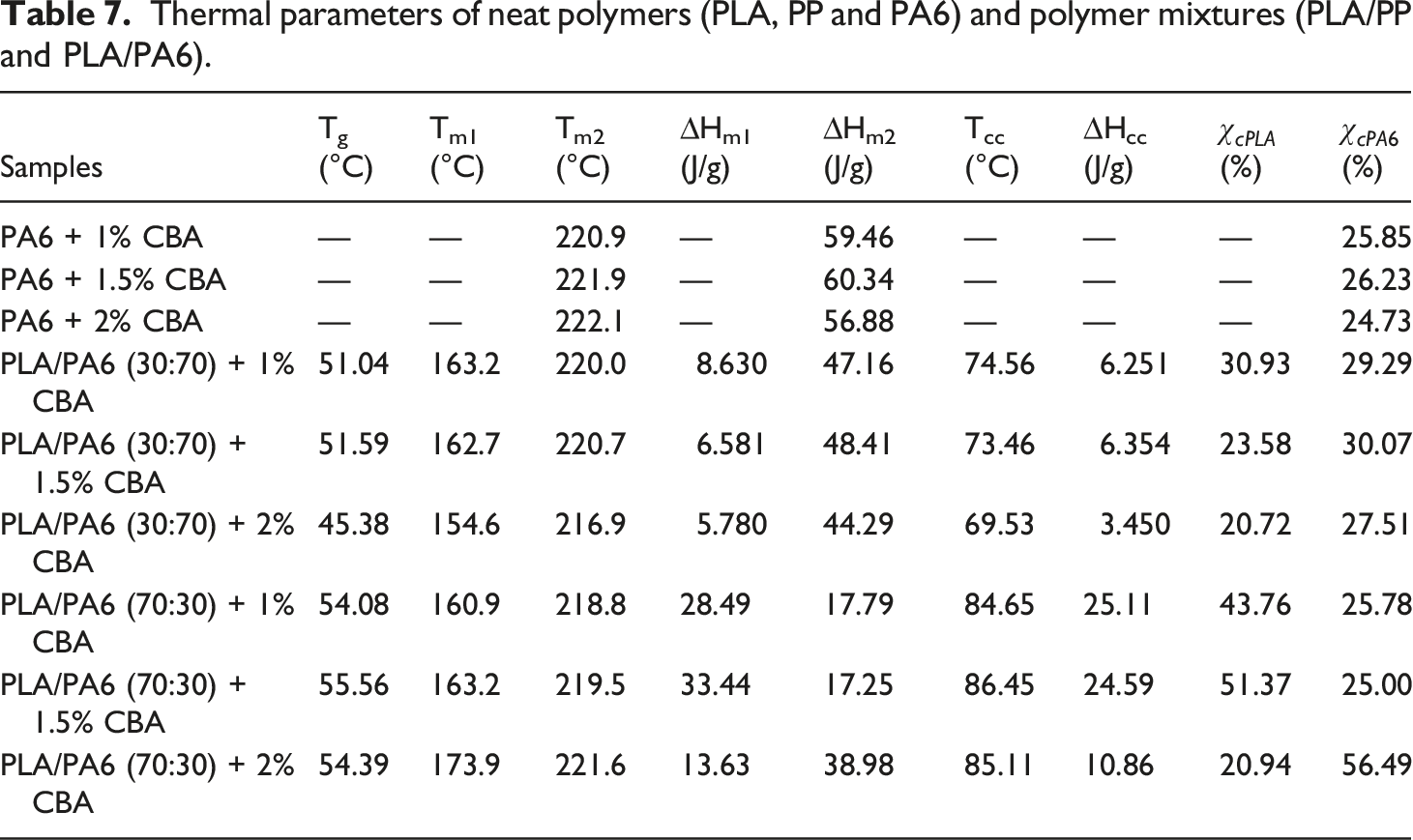
The thermal parameters of PLA/PP and PLA/PA6 polymer mixtures with varying ratios of CBA were characterized by using melting temperature (Tm1 and Tm2) (first and second heatings), Tg (first heating) and Tcc (first cooling) specified from the DSC thermograms. Melting enthalpy of melting (∆Hm), enthalpy of crystallization (∆Hcc) and crystallization ratio ( DSC thermograms of PP polymers containing different ratios of CBA; (a) first melting, (b) cooling, (c) second melting transitions. DSC results of PLA/PP (30:70) mixtures with varying content of CBA; (a) first melting (b) cooling, (c) second melting transitions. DSC results of PLA/PP (70:30) mixtures with varying content of CBA; (a) first melting, (b) cooling, (c) second melting transitions. DSC thermograms of PA6 polymers with varying content of CBA; (a) first melting, (b), cooling, (c) second melting transitions. DSC thermograms of PLA/PA6 (30:70) mixtures with varying content of CBA; (a) first melting, (b) cooling, (c) second melting transitions. DSC thermograms of PLA/PA6 (70:30) mixtures with varying content of CBA; (a) first melting, (b) cooling, (c) second melting transitions. SEM and stereo microscope images of (a) PLA + 1% CBA, (b) PLA + 1.5% CBA and (c) PLA + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively. Thermal parameters of neat polymers (PLA, PP and PA6) and polymer mixtures (PLA/PP and PLA/PA6). Thermal parameters of neat polymers (PLA, PP and PA6) and polymer mixtures (PLA/PP and PLA/PA6).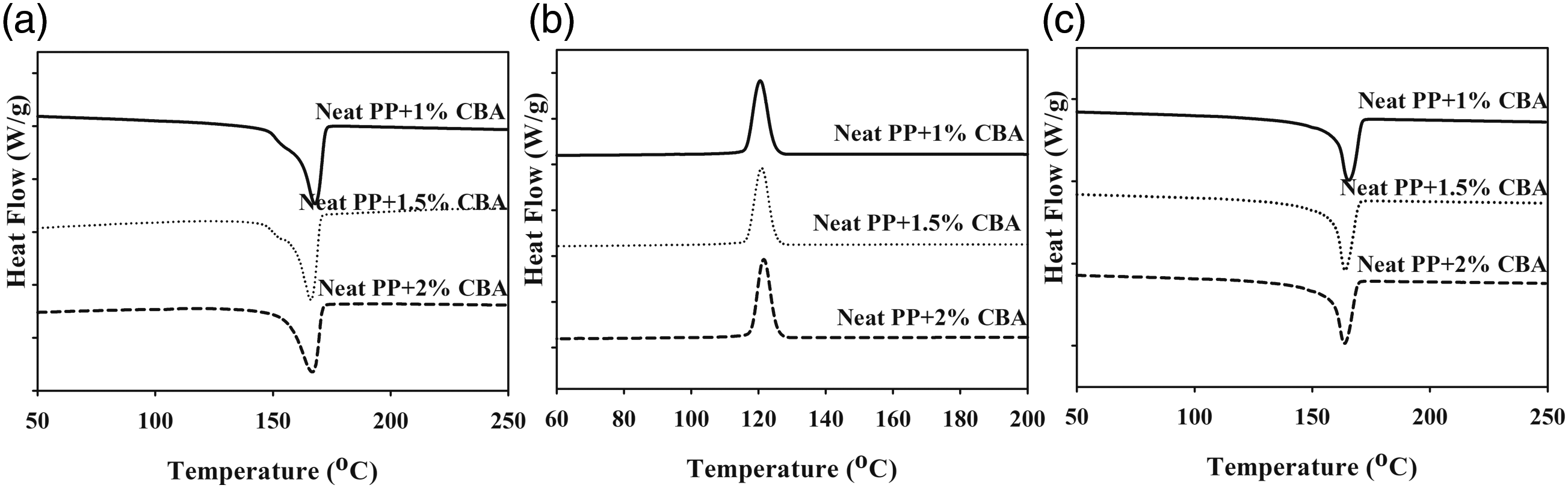

During the foaming process, PLA is exposed to an isothermal treatment at a definite temperature for a definite interval of time in order to ensure the saturation of the blowing agent. Meanwhile, PLA displays an unusual crystallization behavior with the effect of additives such as blowing agents, plasticizers or nucleating agents added to its structure, and deformations can be observed in the structure with the effect of thermal processing. With the impact of the heat and the foaming agent added to the structure, a new chain arrangement takes place in such a way that the unmelted small and irregular crystals are transformed into new structures which are better packed with a more perfect crystal structures and a higher melting point. In this foaming process, pre-molten crystal structures return to their original forms and exhibit a low melting point.28,29 As can be seen in Figure 18(c), it has been shown in various studies that this double melting point formation occurs during the foaming of PLA polymer, and consistent results have been obtained in the literature.30,31 In other studies explaining this phase change of PLA during foaming, this situation is explained by PLA, which can be formed in various crystal types such as α, α′, β and ϒ according to the crystallization parameters, exhibits the α′ form similar to most frequent α form in the helical conformation. This form, which is more irregular gives a lower melting point and has lower thermal stability.32–36 DSC thermograms of PLA polymers containing different ratios of CBA; (a) first melting, (b) cooling, (c) second melting transitions.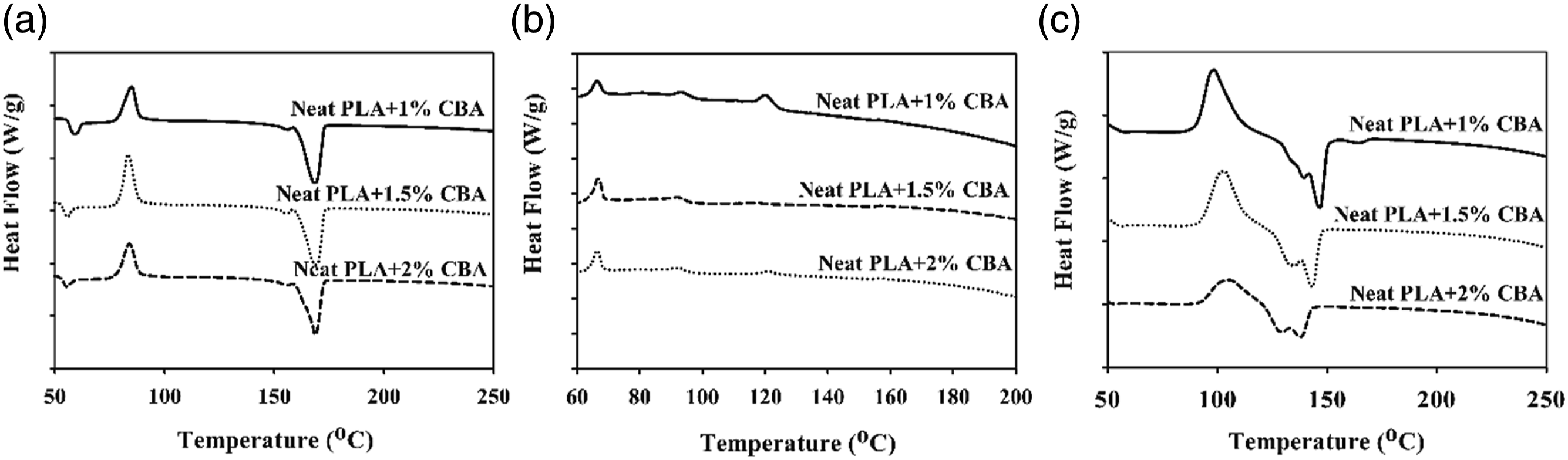
As observed in Figures 11 and 12 it can be seen that there no major variations in the structures of PP mixtures containing CBA and PLA/PP (30:70) mixtures with high PP ratio is detected. Conversely, in PLA/PP (70:30) mixtures where the PLA ratio is high, it can be seen that as the CBA ratio augments, the crystal structure changes and the melting peaks give two melting points which wander away from each other (Figure 13). It can be stated that the reason why PLA exhibits two-point melting behavior is related to the fact that the smaller and more irregular crystals do not have enough time in the heating and recrystallization mechanism to form crystals with higher structural perfection, and they form sequentially and exhibit remelting behavior at a relatively lower temperature. 37 This situation is also associated with the depletion in the crystallinity ratio of PLA as CBA is added.
When the melting and crystallization behavior values in Tables 6 and 7 were evaluated, it can be seen that as the ratio of CBA in the structures increases, no significant change in the Tm values of PP polymer in polymer mixtures is obtained, while a significant decrease was occurred in melting points of PLA polymer due to the phase separations formed in the structure with the addition of CBA. On the other hand, a decrease was observed in the Tg and Tc values of PLA in polymer mixtures as the CBA ratio increases. Table 6 displays the calculated total crystallinity ratios (XcTop) of PLA/PP mixtures without CBA and it can be seen that values in the range of 40–46% were obtained while by addition of CBA this ratio decreased to the level of 15%, especially in PLA and PLA/PP (70:30) polymer mixtures. This decrease showed that the crystal microstructure of PLA polymer was greatly affected by the use of blowing agent. This can be explained by the fact that CBA in the mixture creates a physical barrier in the matrix and prevents the crystal structure formation of the polymers. 38
As observed in Figures 14–16 and Table 7, it is observed that the increased CBA ratio in PA6 mixtures containing CBA does not affect the structure of the materials and that their thermal behavior is similar to their pure state. Furthermore, it was determined that PLA in PLA/PA6 mixtures was less affected by CBA compared to PLA/PP mixtures. Thus, it can be stated that the blowing agent Hyd 822 exhibited more compatible behavior with PLA and PA6 polymers. Furthermore, the crystallinity values were not affected by CBA.
Microstructures of polymer mixtures
Scanning electron microscope and stereo microscope images of PLA, PP and PA6 polymers with PLA/PA6 and PLA/PP polymer mixtures containing CBA are given in Figures 17, 6, 7, 19, and 8–10. ×25 and ×1000 were the magnification ratios for stereo microscope and SEM images, respectively. From the evaluation of these images, it can be said that larger cells and denser porous structures, as demonstrated in Tables 2 and 3 concerning the density results, are formed as the CBA ratio increases in the majority of the mixtures, as expected. Similar results are obtained in the literature.39,40 Furthermore, the comparison of the cell wall and cell size of the binary mixtures cannot be interpreted precisely due to the incompatibility between CBA and polymers. PLA was foamed using a PP compatible foaming agent (Hyd 812) (Figure 17), while this agent could not be used for PA6 which is compatible with the foaming agent Hyd 822 due to its higher activation temperature. However, it was observed that the foaming agent Hyd 812 could not provide an effective foaming owing to the low melt strength of PLA polymer.
36
SEM and stereo microscope images of (a) PLA/PP (70:30) + 1% CBA, (b) PLA/PP (70:30) + 1.5% CBA and (c) PLA/PP (70:30) + 2% CBA for a magnification of ×25 (top) and ×1000 (bottom), respectively.
From SEM pictures of PP polymer and PLA/PP polymer mixtures (Figures 6, 7, and 19), due to the incompatibility between PLA and PP polymers, droplets in matrix morphology is observed. Furthermore, it has been observed that porous structures could be obtained by providing effective foaming, with the effect of using a CBA whose activation temperature is compatible with the melting and process temperature of the polymer. In addition, the effect of CBA ratio is especially seen in PP polymer and PLA/PP (30:70) polymer mixtures. However, successful and effective foaming could not be achieved in PLA/PP (70:30) polymer mixtures having a high PLA ratio.
When Figures 8–10 presenting the stereo microscope and SEM pictures of PA6 polymer and PLA/PA6 polymer mixtures are examined, due to the hydrogen bonds interactions between PLA and PA6, a more homogeneous structures is observed. In addition, it can be mentioned that the PA6 compatible Hyd 822 chemical blowing agent is very effective in the formation of porous structure in PA6 polymer, PLA/PA6 (30:70) and PLA/PA6 (70:30) polymer mixtures. When these results are compared with the PP-based mixtures, it was observed that the size of the pores formed in the PA6-based mixtures was larger.
Conclusions
In order to determine the most suitable chemical blowing agent ratio for PLA/PA6 and PLA/PP polymer mixtures at fixed polymer ratios (30:70 and 70:30), the mixtures were obtained using the extrusion and compression molding processes for CBA contents of 1, 1.5 and 2 wt.%. Then, the mechanical, density, FT-IR, DSC and microstructure analyzes were examined. Consequently, it can be stated that all test results were compatible with each other and with the literature. Although more cell formation and lower density values were obtained with increasing CBA ratios in the mixtures, weakening was observed especially in mechanical and thermal properties. While mechanical tests could not be performed on PLA/PP (70:30) polymer mixtures containing CBA, a significant decrease in mechanical values was observed in PLA/PA6 (70:30) polymer mixtures. For this reason, when the analysis results were interpreted according to the lightness, strength and thermal stability, the most appropriate CBA ratio for PLA/PA6 (30:70) and PLA/PP (30:70) polymer mixtures was determined as 1.5 wt.%. When the results of the mixtures produced and analyzed to determine the polymer and CBA ratios are evaluated, it is seen that the need for the use of compatibilizing agents arises in the mixtures prepared by using PLA biopolymer together with PP and PA6 polymers. It is thought that, in the foaming of the mixtures of biopolymer PLA prepared with petroleum-based polymers PP and PA6, with the use of compatibilizing agents, lighter but durable structures can be produced without seriously affecting the thermal and mechanical properties.
Footnotes
Acknowledgements
The author thank the Central Research Laboratory of Bursa Technical University for providing the facilities for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Scientific Research Projects Department of Bursa Technical University (contract number 190D001).
