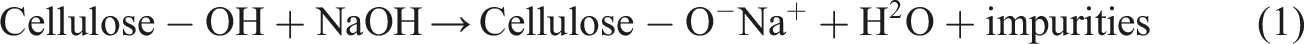Abstract
This study was aimed at evaluating the individual and combined effects of organically modified nanoclay cloisite 20A and alkali treatments on the valorization of totally waste-based bagasse fiber/recycled polypropylene nanocomposites. FTIR spectra indicated the reduction of bagasse fibers’ hydroxyl moieties due to hemicellulose removal resulting from alkali treatment. There were also trace diminutions in C-O peak intensities due to the degradation of little amounts of lignin. X-ray Scattering revealed the intercalation of nanoclay in the polymer matrix. The improved interaction of fiber-polymer interfaces brought about by alkali treatment was also confirmed by scanning electron microscopy. The physical properties of the nanocomposites were improved due to the barrier properties of nanoclay against water ingress. The flexural strength and modulus of elasticity increased by both individual and combined treatments; however, the impact strength decreased by the individual treatments. Thermogravimetric analysis demonstrated that the temperatures of first and second stages of composites’ thermal degradation increased due to the treatments via the formation of a carbonized char layer thermally insulating the deeper composite layers. Differential scanning calorimetry showed some slight increases in the melting temperature, melting enthalpy, crystallisation temperature, crystallisation enthalpy, and crystallinity index of the treated composite formulations due to the nucleating effect of nanofillers. The overall results confirmed that the combination of nanoclay cloisite 20A and NaOH treatments could significantly improve the overall properties of the studied composites. This was due to some interesting synergistic effects of the given treatments on the nanocomposites converting the bagasse/recycled PP composites to high performance materials of choice.
Keywords
Introduction
Incorporation of natural fibers in to polymer composites has attracted much attention in recent years due to environmental restraints, and the fact that natural fibers are biodegradable, cost effective, and widely available.1,2 Reprocessing plastic scraps and adding them in to wood plastic composites (WPC) as the polymer matrix reduce the adverse effects of dumping plastics in nature. These inexpensive wastes not only hold a large share of municipal wastes across the globe but also are suitable for WPC production due to their low melting and processing temperature being below the degradation temperature of natural fibers (<200°C).3,4 Water absorption is a major drawback in wide application of WPCs which is affected by the kind and amount of WPC constituents. The hydrophilic nature of natural fibers introduces phenomenal problems in WPCs which are poor interfacial adhesion of the given fibers with the hydrophobic plastic matrix as well as providing a proper condition for fungal attack. Thus, the surface of natural fibers need to be treated in order to reduce their hydrophilicity leading to enhanced stress transfer from the polymer matrix to the fibers and the resulting improved physico-mechanical performance. Sodium hydroxide (NaOH) can remove the surface impurities of fibers and increase their unevenness, thus opening and making accessible more hydroxyl and other functional groups on cellulosic fiber surfaces.5–7 Sodium hydroxide can also react with reachable –OH moieties, thus facilitating other chemical reactions
8
as given:
Bagasse fiber (sugar cane residue) alkali treatment with 1% NaOH has been reported to improve tensile, flexural, and impact strength properties as well as the aspect ratio (fiber length to diameter) of the manufactured biocomposites. Scanning electron microscopy (SEM) micrographs showed improved interfacial adhesion among the alkali treated bagasse fiber and the polymer matrix. 9 The existing archival publications demonstrated that NaOH treatment led to increased rates of fiber surface fibrillation, thus providing more accessible reaction sites on fiber surfaces for subsequent chemical reactions; this improved the tensile strength and elongation at break of the polypropylene bagasse fiber composites.10,11
Nanotechnology is rapidly on the growth in different industries.12–20 One of the cheapest and widely available naurally occurring mineral fillers, which can be used to improve the overall properties of WPC, is nanoclays (i.e., montmorillonite). They are one to two layer aluminosilicate groups, having exchangeable cations (e.g., Na+ and Ca2+) which can fill the spaces between the layers. The exchangeable cations can also be replaced by organic cations like quaternary ammonium salts and produce organophilic clay holding a much higher compatibility and chemical affinity to bond with polymers. 21 Wang et al. (2001) stated that the impact of nanoclay on composites depended on its shape, size, aspect ratio, type, rate and quality of dispersion, and interfacial adhesion. They also reported that the addition of nanoclay could improve thermomechanical properties as well as dimensional stability of the studied composites. 22 Wu et al. (2007) found out that the incorporation of only 2 wt% nanoclay into pine flour/high density polyethylene (HDPE) composites could increase flextural and tensile strengths by 4.4% and 1.2%, respectively; however, water absorption and thichness swelling decreased by 5%–7%. They also profferred that the temperature of glass transition, moduli of storage and loss, and cystallinity degree of the studied composites dramatically increased due to nanoclay. This brought about improved thermal stability and fire retardancy for the studied pine flour/HDPE composites. 23
To the best of the authors’ knowledge, the present study, for the first time, addresses the synergistic effects of alkali treatment and organically modified nanoclay addition on structure-property relationships of fully waste-based environmentally compatible bagasse fiber/recycled polypropylene composites through a wide range of analyses including water absorption, thickness swelling, flexural and impact strengths, modulus of elasticity, SEM, FTIR, XRD,TGA, and DSC tests.
Materials and methods
Materials
The bagasse fiber used in this study was purchased from Khuzesta Dash Laleh Nasr Co., Iran. Polypropylene (PP) grade V30 G with melt flow index (MFI): 16 g/10 min and density: 0.9 g.cm-3 was purchased from Maroon Petrochemical Co., Iran. Maleic anhydride grafted ploypropylene (MAPP) with MFI: 10 g/10 min and density: 0.95 g.cm−3 was purchased as the compatibiliser from Aria Polymer Pishgam, Iran. Organically modified montmorillonite grade cloisite 20A with bulk density: 0.118 g/cc and average particle size: <10 μm was provided from Southern Clay Co., USA. Sodium hydroxide (99%) was purchased from Merck Co., Germany.
Treatment of sugarcane bagasse fibers
The sugar cane bagasse fibers were first soaked in hot water for 1 hour. The fibers were then air dried for 72 h and placed in an oven (103°C for 24 h) to be well died. The fibers were then processed in a rotary chipper, and the 30–100 mesh size portion of the fibers were selected for further experiments. Alkali treatment of bagasse fibers was conducted by soaking the fibers in 5wt% NaOH at room temperature for 2 h. Thereafter, the fibers were washed and rinsed with distilled water, air dried for 3 days, and then placed in an oven at 103°C for 24 h. The so-treated bagasse fibers were sealed in plastic bags for further WPC manufacture.
Preparation of recycled PP
Vigin PP was extruded through a single screw extruder (barrel length: 1.5 m) equipped with five thermal zones manufactured by Pouya Polymer Amirkabir Iran Co., Iran and cut in to recycled PP granules coming out of the extruder. Extrusion was carried out at 190°C, and the produced granules were pulverized and sealed in plastic bags for further WPC manufacture.
Composite processing
Composition and codes of composite formulations.
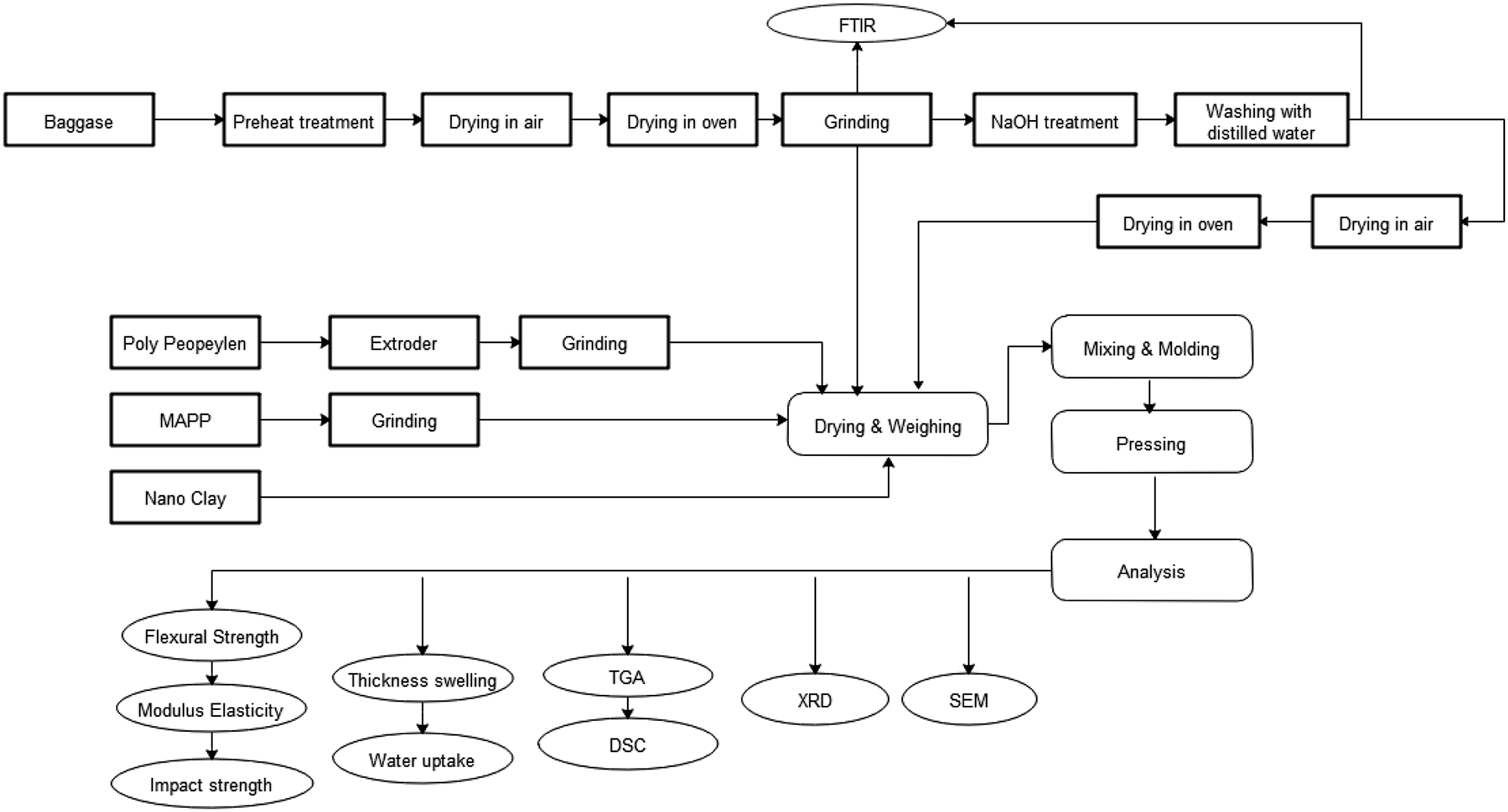
Schematic flow chart of the experimental process.
Composites characterization
Water absorption and thickness swelling tests were carried out according to ASTM D7031-11 standard specifications with five replications and calculated based on equations (2) and (3).
Where: mw and mO are sample wet and oven-dried weights
Where: tw and tO are sample wet and oven-dried thicknesses
Mechanical tests included unnotched impact and bending strengths which were conducted according to ASTM D256 and ASTM D790 specifications, respectively, using an Instron testing machine model 4489 with five sample replications.
The functional groups of the untreated and alkali treated fibers were determined and analysed by a Fourier Transform Infrared Spectroscopy (FT-IR) Tensor 27 IR spectrophotometer (Bruker Co., Germany). The transmittance range of the scan was 4000–370 cm−1 for each composite sample.
SEM was conducted on the fractured surfaces of the composites to analyse the interfacial adhesion and mophology of the treated and untreated bagasse fiber/PP nanoclay composites by a MIRA III model SEM (Tescan Co., Czech Republic). The fractured surfaces were coated with a thin layer of gold before scanning.
Small-Angle X-ray Scattering (SAXS) analysis for WPC samples was conducted with a Phillips model PW17 C (Germany) operating with scattering angle, 2θ, scanned from 5° to 90° via Cu Kα (λ = 0.154 nm) radiation. XRD analysis was performed at 2 
Where: d is interplanar distance,
Thermogravemetric analysis (TGA) can determine the percent by mass ratio of a solute using heat and stoichiometry ratios. TGA analyses of the WPCs were conducted using a thermal gravimetric analyzer (STA 504 thermal analysis, Co., Bahr, Germany). The WPC samples were heated up at a rate of 10 °C/min from 25 to 600°C.
Differential scanning calorimetry (DSC) can measure major thermoplastic properties, including melting temperature (Tm), melting enthalpy (ΔHm), crystallization temperature (Tc), crystallization enthalpy (ΔHc), and crystallinity rate (Xc). Thermal analysis of the WPC samples was carried out on a differential scanning Q600, Co.,TA, USA. All DSC measurements were performed with 15 ± 0.2 mg of each composite sample under a nitrogen atmosphere with a flow rate of 20 mL/min.
The statistical analyses were conducted in a completely randomized factorial design using SPSS software. One way analysis of variance (ANOVA) was employed, and the means were grouped by Duncan’s Multiple Range Test (DMRT) at 95% confidence level.
Results and discussion
Composites physical properties
Hydrophilicity and polarity of lignocellulosic fibers as well as the hydroxyl and carboxyl moieties existing on fiber surfaces lead to the formation of hydrogen bonding between water molecules and natural fibers.24,25 Figure 2 shows the percentages of water absorption and thickness swelling in the studied nanocomposites after 3 weeks of immersion in water. There is the same water absorption and thickness swelling trends in the studied composte formulations. Physical properties of bio-nanocomposite formulations.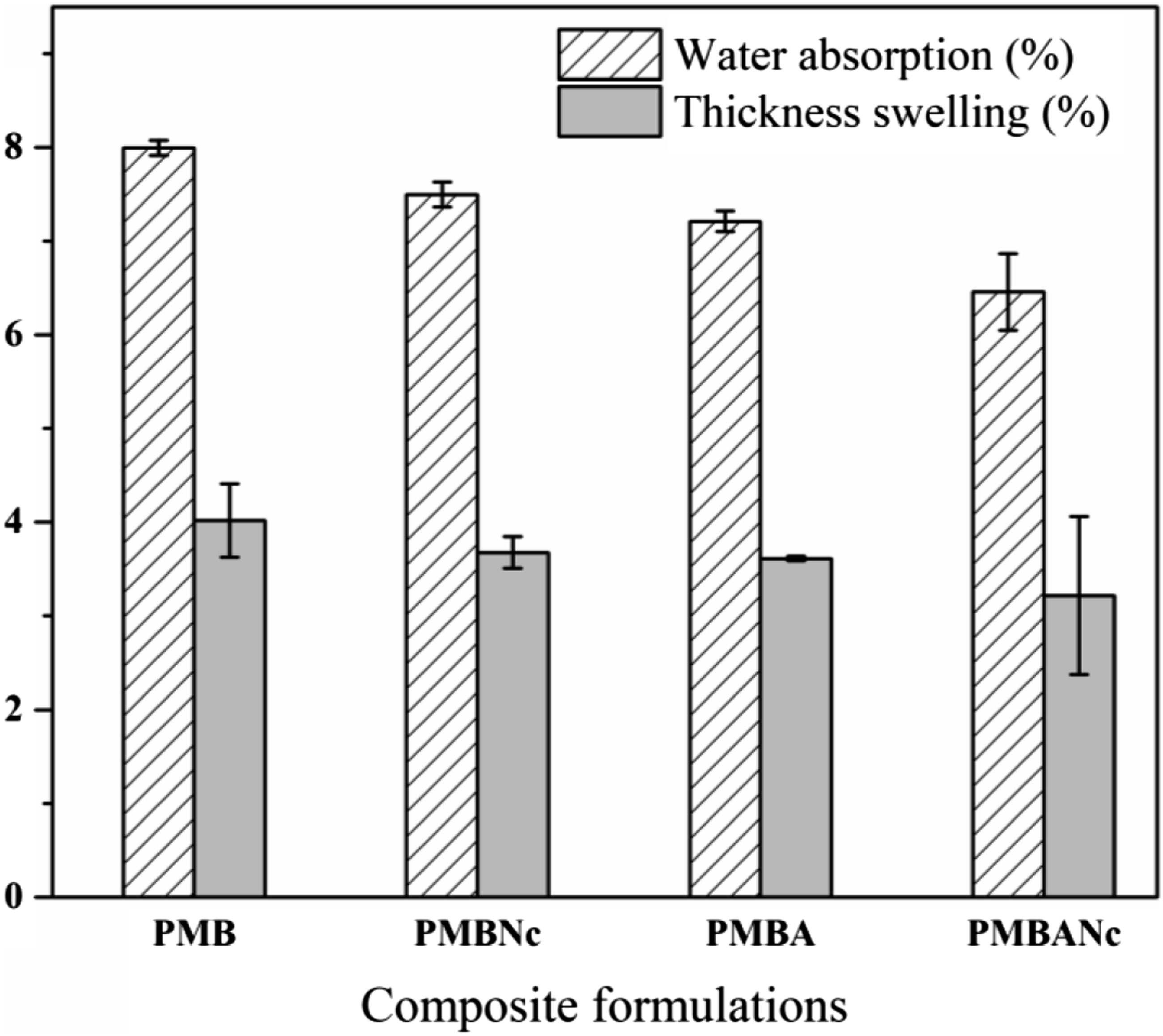
PMB showed the highest percentages of water absorption and thickness swelling, being about 8% and 4%, respectively. The Addition of 4wt% nanoclay cloisite 20A to composite formulation (PMBNc) could slightly decrease water absorption and thus thickness swelling by 6.22% and 8.39%, respectively, which were statistically non-significant. This is due to the barrier properties of nanoclay particles preventing from water ingress in to composites as well as the fact that they also fill the micro and nano voids present within the composites, thus reducing both of the mentioned physical properties.26–28 Archival publications show that proper dispersion of nanoclay, hydrophobic nature of silicates present in the given nano, increased crystallinity rate due to the addition of nanoparticles can also lead to the immobilization of water and vapor molecules and thus reduced water absortion and thickness swelling rates in PMBNc composites.29,30 However, Yeh and Gupta 31 reported that the addition of >2% nanoparticles could increase composites’ water absorption rates due to the coagulation and agglomeration of nano fillers.
Alkali treatment reduced water absorption rate from 8% (PMB) to 7.21% (PMBA). Also, thickness swelling decreased by 10% in PMBA composite formulation, as compared to the reference one (PMB). This is due to the reduction of polar hydroxyl groups by alkali treatment which creates stronger bonds between the now less polar fibers and the apolar polymer matrix.32–34 SEM micrographs (Figure 3) also confirm the better wettability and adhesion of NaOH treated bagasse fibers and the polymer (PMBA) vs. the reference composite formulations (PMB). SEM micrographs of fracture surfaces of composites, (a) PMB, (b) PMBNc, (c) PMBA, (d) PMBANc.
Alkali treatment also releases the fibers entangled in fiber bundles through dissolving the surface hemicellulose, pectin, and aliphatic compounds (e.g., waxes) on fiber bundles,35–37 thus increasing not only the efficient fiber surface area to bond with the polymer but also the fiber distribution in the polymer matrix. Furthermore, the NaOH treated fiber surfaces grow rougher, thus enhancing fiber-matrix wettability by providing added fiber sites for mechanical entanglement.35–38 Statistical analysis revealed that there were no significant differences between the water absorption and thickness swelling rates of PMBA and those of the PMBNc at 95% confidence level, whereas significant differences were observed when the given properties of PMBA composites were compared to those of the reference (PMB). PMBANc composite showed the highest reduction rates of water absorption and thickness swelling (∼20%). This can be attributed to the combined positive effects of nanoclay and alkali treatments on the mentioned physical properties. Except for thickness swelling rates of PMBA and PMBNc composites, there were significant differences between the water absorption and thickness swelling rates of PMBANc and all the other composite formulations at 95% confidence level.
Composites mechanical properties
Flexural strength
Flexural strength is defined as a material’s potential to resist deformation under static load. This represents the highest stress exerted on the material at the moment of rupture. 39 The flexural strength of composites depends on the properties of each composite material as well as the interfacial adhesion between the matrix and reinforcing and/or filler agents.
Figure 4 displays the flexural strengths of the studied composite formulations. As observed, the given strength slightly increased in PMBNc samples (29.67 MPa) as compared to that of the reference one; however, the given increase was not statistically significant at 95% confidence level. This increase can be attributed to miscellaneous reasons including the high aspect ratio of nanoclay, formation of intercalation and exfoliation nanoclay structures, filling the empty spaces and void in composite structure, thus introducing a denser and more rigid texture in nanocomposites,40–42 strong interaction between the polymer and silicate layers due to formation of hydrogen bonds in the gallery spaces of nanoclay;31,43–45 furthermore, heterogeneity and high ratio of surface to volume of the organically modified nanoclay cloisite 20A improve the reinforcing effect of nanoclay in the studied composites by increasing the interfacial area and the resulting interactions between the polymer matrix and bagasse fibers.
23
The existing literature demonstrates that the content of nanoclay addition in composite formulations plays a key role in the overall effects of the nanoparticles; it has been reported that agglomeration of nanoparticles leads to impaired interfacial adhesion between the phases and the resulting weak mechanical performance in nanocomposites.
40
When nanoclay concentration is increased, the solid nano particles may adversely affect the absorption of fracture energy rendering the nanocomposites more brittle; this is attributed to the harder and denser structure of nanos as well as their restrictiing effect on natural fiber and polymer entanglement and interlocking. Also, the nanofillers would attract much more compatibilizer and prevent from the proper bonding of the compatibilizer with wood fibers, undermining the mechanical strength of nanocomposites.31,41 Flexural Strengths of bio-nanocomposite formulations.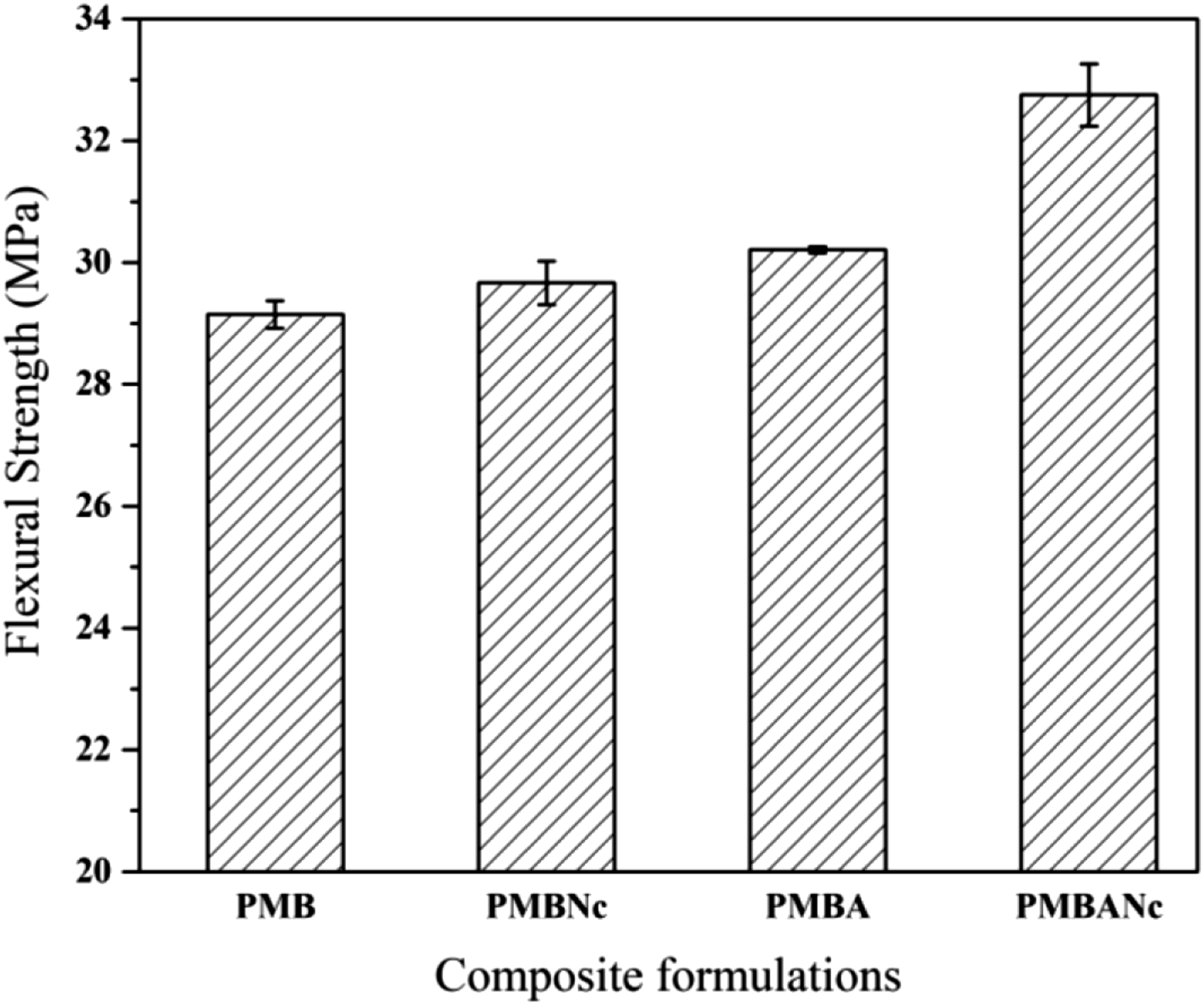
Figure 4 shows that the flexural strength of NaOH treated bagasse fiber composites (PMBA) increased (30.21 MPa) as compared to that of the reference, being statistically significant at 95% level. This is probably due to the dissolution of fibers’ surface impurities such as pectin and waxes, and likewise the improved wettability (polymer encapsulation), interfacial adhesion, and distribution of alkali treated bagasse fibers in the polymer matrix; 46 this can also be observed in SEM microgrphs of the fractured surfaces of PMBA composites (Figure 3).
The combined effects of alkali treatment and nanoclay introduced a marked increase (11%) in the flexural strength of PMBANc composites versus PMB ones, which was statistically different with those of the other studied formulations. The given treatments improved the interfacial adhesion between the bagasse fibers and the polymer, thus increasing flexural strength in PMBANc formulations. This can be attributed to the formation of a more homogeneous composite structure due to the synergistic effects of the combined treatments; this leads to better stress transfer, thus fewer stress concentration points in the given nanocomposite materials when a static load (stress) is introduced on the given samples.
Modulus of elasticity
Modulus of elasticity (MOE) is a measure to describe stiffness in solid materials; in other words, it measures a material’s resistance against elastic deformation under stress. Figure 5 shows that alkali, nanoclay, and the combined treatments had positive effects on the MOE of composite formulations. As Figure 5 displays, there is an increase in the MOE of PMBNc nanocomposites (5248.34 MPa), as compared to that of the reference (5057.33 MPa). MOE of a composite is a function of the MOE values of the composite’s individual components.47,48 The MOE of nanocomposites depends on a variety of factors such as nanoclay content, surface to volume ratio, aspect ratio (nano filler length to diameter), formation of intercalation and exfoliation structures, strong entanglement of nanoclay with the polymer, and the nucleating effect of well-dispersed orgnically modified nanoclay particles in the polymer matrix, which generally improve the MOE and strength performance of nanocomposites, owing to the stiffness of nanoclay silicate layers.49,50 Studying the effects of nanoclay (Cloisite 20A) on wood fiber/HDPE composites, Lee et al. (2010) reported that the incorporation of nanoclay could increase MOE, such that the exfoliated composites had larger MOE values than the intercalated ones. Also, Compatibilizers hold the potential to reduce and near the surface energy of natural fibers to that of the polypropylene; this leads to better wettability and encapsulation of fibers by the polymer, thus increasing the entanglement and adhesion of nanoparticles with both composite phases.
51
Similar results have also been reported by other researchers.52,53 Modulus Elasticity of bio-nanocomposite formulations.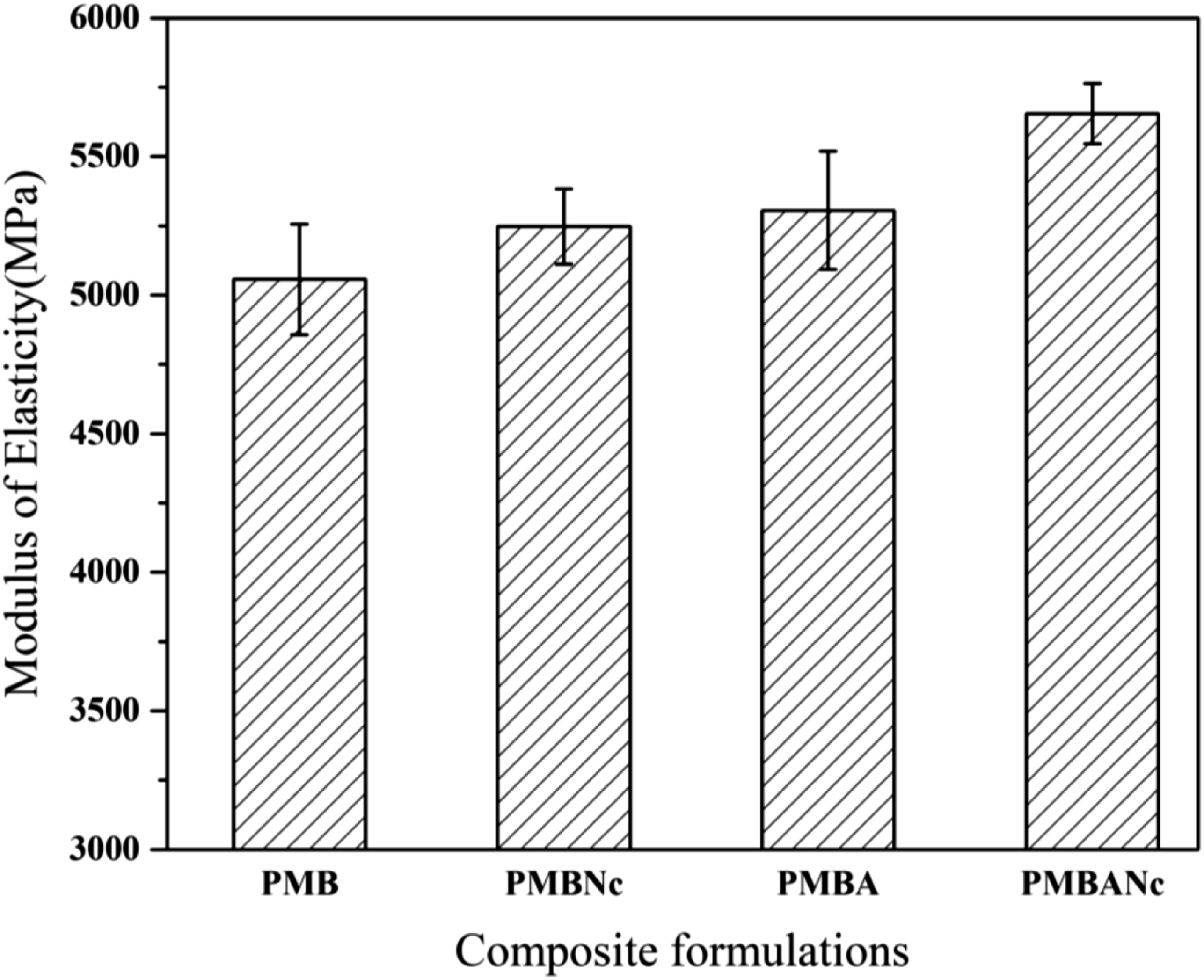
As Figure 5 depicts, there is an increase in the MOE value of PMBA composites (5306.33 MPa), compared to that of the reference. This is due to the reaction of NaOH with the hydroxyl moieties of bagasse fibers converting them to opened-up microfibrils; this phenomenon (i.e., fibrillation) introduces increases in the efficient surface area of fibers to bond with the polymer matrix, fiber wetting and encapsulation by the polymer, bagasse fiber distribution in the polymer, interfacial adhesion between the two phases. These are the reasons behind the enhanced mechanical strengths of alkali treated composites.51,54 Mercerization of bagasse fibers with sodium hydroxide also makes the fibers more rigid and fragile, engendering an additional increase in the MOE value of PMBA formulations. The combined treatment led to a statistically significant difference of the MOE value (5644.3 MPa) of PMBANc composites with those of the other composite formulations at 95% level. The combined treatment seems to have more efficiently increased the interfacial adhesion between the bagasse fibers and the recycled polypropylene. This probably led to facilitated stress transfer between the two mentioned phases; therefore, a larger stress load is needed should the composites be fractured.55,56 The interactive effect of nanoclay and alkali treatment could possibly improve particle dispersion in the resulting composite materials, which can be observed in the corresponding SEM micrographs (Figure 3).
Impact strength
Impact strength of a composite is affected by an array of factors such as the hardness of the reinforcing agent, the nature of the interfacial surface, and the frictional energy engaged in pulling out the fibers from the polymer.57,58 As shown in Figure 6, thers is a slight reductioin in the impact strength value of the nanoclay treated composites (7.24 kj/m2), as compared to that of the reference (7.41 kj/m2). This is in line with the results reported by Khanjanzadeh et al. (2011); they related the reduction of impact strength to the restriction of polymer chain movements due to the effect of nanoparticles, which undermines the ability of composites to absorb the applied impact energy.6,59 Furthermore, the formation of non-dispersed fiber bundles and voids would impair impact strength.45,60–62 The lowest impact strength was observed in alkali treated composites (PMBA), being 7.23 kj/m2; however, there was not a statistically significant difference between the mentioned value and those of the PMB and PMBNc composites. The sodium hydroxide treatment of bagasse fibers reinforces the interaction between wood-polymer phases, bringing about strong adhesion; thus, the composites would be fractured at relatively lower impact energy values. On the other hand, composites with weaker interfacial adhesion show higher impact strength values due to the formation of micro-cracks in the impact test. The mentioned micro-cracks propagate along the fiber/polymer interface and cause fiber/matrix debonding, imparting a higher energy-absorption capacity to the composites.63,64 However, the reduction of impact strength in the alkali treated composites may be due to the immobilization of the macromolecular polymer chains introduced by bagasse fibers; this undermines the ability of composite materials to adapt to the deformation, thereby causing the composites to be more brittle.65,66 As Figure 6 illustrates, there is an interesting improvement in the impact strength of PMBANc composites, which is statistically sifnificant at 95% confidence level, as compared to the impact strength values of the other composite formulations. As previously mentioned, there are contradictory results for the impact strength of WPC in the archival publications, which needs to be further studied. Also, the unnotched impact strength test is highly dependent on the surface properties of composite samples such that in case of the presence of a hard and rigid substance on the sample surface, the impact strength is expected to rise.
67
Impact Strength of bio-nanocomposite formulations.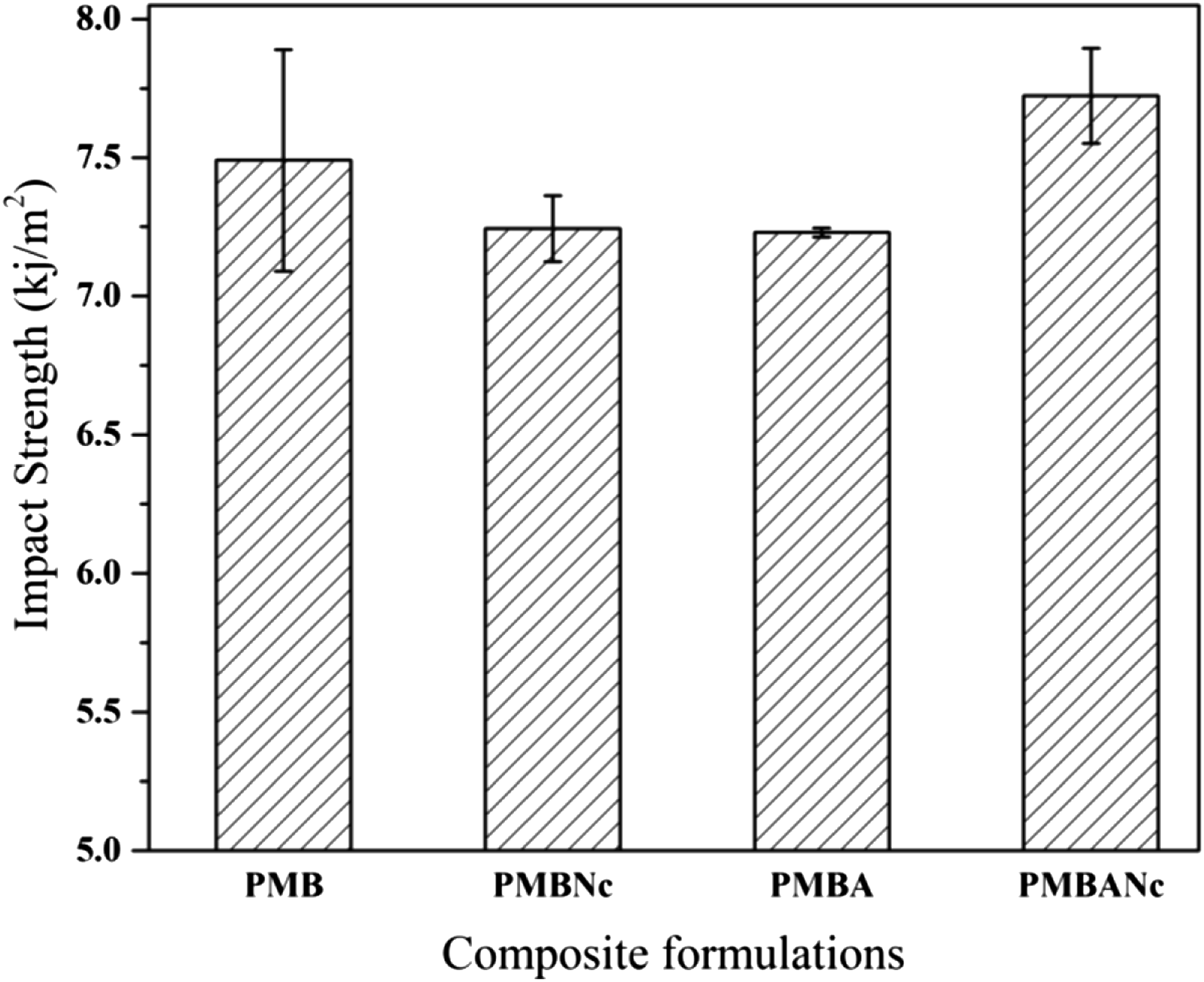
Bagasse fiber characterization
The effects of chemical treatment on bagasse fiber structure was analysed by Fourier transform infra-red spectroscopy (FTIR). Figure 7 displays the results of FTIR analysis of bagasse fibers prior to and after NaOH modification in the wavelength range of 500–4000 cm−1 The band at 3440 cm−1 shows the –OH groups stretching in bagasse fibers, which is observed at a lower intensity of infra-red absorbance in the treated fibers, as compared to that of the untreated fibers.68,69 This is ascribed to the reduction of hydroxyl moieties due to alkali treatment, rendering the fibers less hydrophilic and thus more chemically reactive towards the hydrophobic polymer matrix. There are also strong peaks at 2923 cm−1, which belong to the C-H stretching of aromatic methoxyl and methylene groups existing in lignin.70,71 The given peaks did not have significant differences in the alkali treated and treated fibers, demonstrating that the treatment had almost no effect on the removal of lignin polymer components. The absorption bands at 1738 cm−1 can be attributed to the vibrational stretching of unconjugated carbonyl functional groups (C = O), present in fiber hemicelluloses.72,73 As observed in Figure 7, the given band has disappeared in the alkali treated fibers due to hemicellulose removal by sodium hydroxide treatment. Furthermore, the increase in absorption intensity at 1245 cm−1 is relative to C-O stretching of acetyl groups in hemicelluloses,
74
which is eliminated in the alkali treated fibers. The peak at 1605 cm−1 can be related to the bending mode of the absorbed water and also some contributions from carboxylate moieties present in hemicelluloses structure, having transferred to 1590 cm−1; this can be referred to the removal of bound water from hemicellulose due to the given treatment.
75
The peak at 1432 cm−1 is for the stretching C-H bonds in the amorphous and crystalline parts of cellulose chains, having both transferred to 1429 cm−1 and increased in intensity; this can be due to the release of C-H groups from fiber surfaces after alkali treatment.
76
The peak at 1047 cm−1 relates to the stretching C-C and C-O bonds in hemicellulose xylan, and C-O-C bond in the glycosidic linkages of hemicelluloses,
77
having remarkably increased in intensity after alkali treatment. This can be ascribed to fibrillation phenomenon caused by NaOH treatment, which opens up the once-interwoven cellulose and hemicelluloses fibers, thereby making the above-mentioned stretching bonds accessible to absorb the infra-red light, introduced on the test specimens, in FTIR analysis. The vibrating peaks at 898 cm−1 are due to β-glycosidic bonds of glucose rings in cellulose, being symmetric in polysaccharides.78–80 FTIR spectra of untreated and alkali treated bagasse fibers.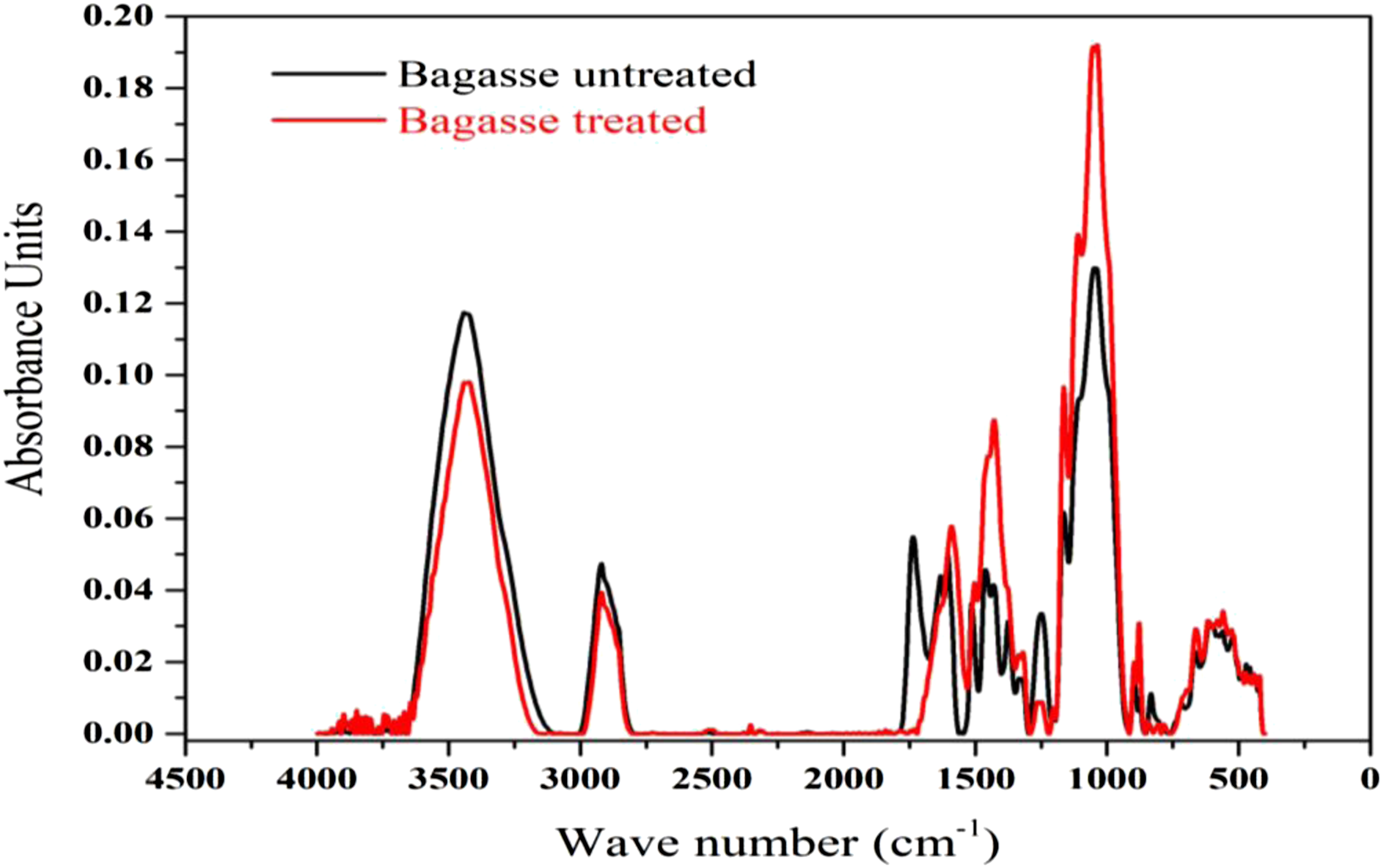
Morphological analysis
SEM is an analysis conducted on the fractured surfaces of composite materials to study the interfacial adhesion between the reinforcing agent and the polymer matrix; this is a qualitative measure showing the reasons behind the overall evolution of composite properties. As observed in Figure 3, there are some gaps and cavities in the fracture surface of PMB composites (Figure 3(a)) showing fiber pull-outs due to the wealk interfacial adhesion between bagasse fibers and the polymer. The incorporation of nanoclay in composite formulations incurred some alterations in the fracture surfaces of PMBNc composites (Figure 3(b)). Nanoclay particles can be observed in the given figure leading to better fiber-polymer interfacial adhesion, since the organically modified nano particle could make some linkages beween composite phases. Although few gaps are also seen, the fiber fractures observed in Figure 3(b) reveal the better fiber-polymer interlocking in the PMBNc composites than the PMB ones confirming the improved physical and mechanical (except for impact strength) performance of the nanoclay cloisite 20A treated composites versus the untreated counterparts. The effects of NaOH treatment on bagasse fibers are seen in Figure 3(c) (PMBA). The given composite shows a different fracture surface compared to that of the reference. There are fewer and smaller cavities but remarkably more fiber pull-outs in PMBA composites than the PMB ones demonstrating the better fiber-polymer bonding in the former composites than the latter ones. The dissolution of fiber surface impurities (e.g., pectin and waxes) could also improve the wettability and thus interfacial adhesion between fiber-polymer phases in the alkali treated composites versus the reference ones. 81 Furthermore, the alkali-induced fibrillation in PMBA composites could increase fiber surface areas bonding with the other composite components (i.e., compatibilizer and polypropylene).82–85 There is a marked difference in the fracture surfaces of PMBANc composites with those of the other composite formulations (Figure 3(d)). The whole surface is fairly uniform and smooth showing a good fiber-polymer wetting. There is almost no distinct cavities and gaps between the two phases. Also, The nanoclay particles are well dispersed through out the fractured surfaces. The fiber fracture observed in the given micrograph is another reason behind the significantly better overall properties of PMBANc composites than other formulations.
Small-Angle X-ray Scattering (SAXS) analysis
SAXS analysis of nanoclay and its composites.

As shown in Table 2, 2θ = 4,80° pertains to neat nanoclay with basal spacing of 18.33 A°. In PMWNc, PMWANc, PMBNc, and PMBANc formulations, the peaks were shifted to lower 2θ angles, and therefore higher interplanar d-spacing values than cloisite 20A demonstrating the formation of intercalation structures. The shifting of the peak to lower angle can be assigned to the increase in the interlayer spacing of silicate layers present in the structure of the incorporated nanoclay in the composite formulations. This reveals that the crystalline chains of polypropylene were intercalated into the silicate layers of the nanoclay. Diffraction peak shifting to a lower angle has also been reported for nano clay based HDPE biocomposite. 43
To note that the reported increases in the interplanar d-spacing values of the studied nanocomposites indicate the occurrence of only low intercalation (vs. exfoliation) structures in the given composites, since interplanar distances > 70 Å are attributed to total exfoliation phenomenon, which refer to 2θ values close to 0°. 14
Composites thermal properties
TGA
TGA analysis of bagasse fiber/polypropylene nanocomposites.
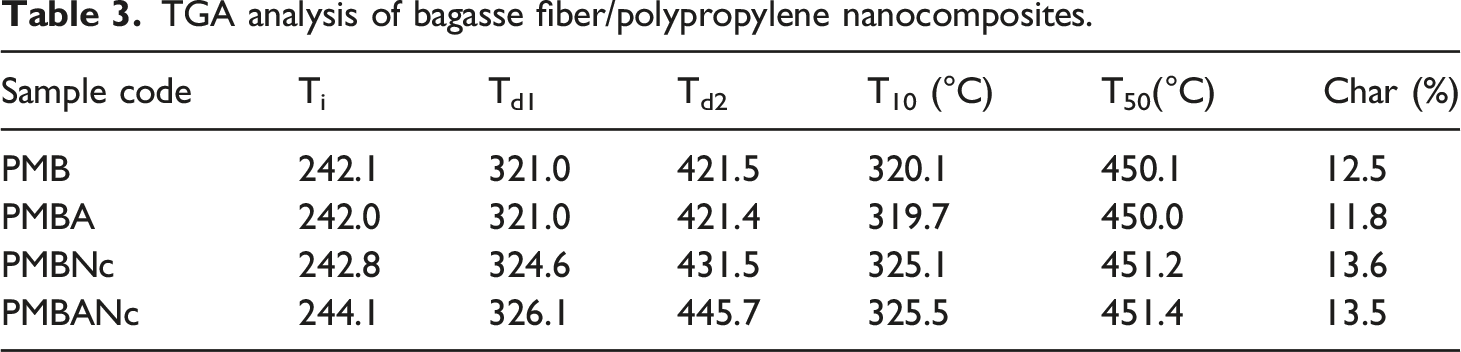
The TGA results reveal that the alkali treatment (PMBA) slightly reduced the fibers thermal behavior, which can be attributed to composites’ lignin content. The FTIR spectra demonstrate that lignin was not efficiently degraded through alkaline treatment.96,97 Alkali treatment is reported to impair thermal performance as a result of the interactive effects of transformation of cellulose, crystallinity, etc., which need be further studied. 86 However, the given treatment has also led to the improved themal stability of composites.74,98 Their results showed that alkali treatments degraded portions of hemicellulose and lignin from the fiber. Thus, the decomposition process mainly occurred on the cellulose, thereby raising the overall degradation temperature. Also, the alkali treated composites release water at higher temperatures as a result of better encapsulation of the finely separated fibers by the polymer matrix. This led to stronger polymer-fiber interfacial adhesion, thus downsizing the accessible surface area for moisture release. This engenders delay in moisture evaporation bringing about elevated thermal decomposition temperatures. 64 TGA results also indicate that the incorporation of nanocly improved the thermal properties of PMWNc, as compared to those of the PMW and PMWA composites. Different mechanisms can play a role in this regard, briefed as follows: The silicate layers of the studied nanoparticle expand the tortuous path, thus delaying the diffusion of degraded volatile compounds throughout the composites due to their high aspect ratio as well as formation of intercalation structure) 99 A char layer is formed on the external surfaces of composite materials by nanoclay/silica, and this leads to the insulation of the sub layer, therby preventing the nanocomposite from burning and the resulting higher degradation temperatures and better thermal stability) 100 Strong interaction between the nanoclay silicate layers and polypropylene chains can limit chain mobility and thus delay conveying free radicals formed in the course of polymer fragmentation brought about by thermal degradation, thus enhancing the thermal stability of nanocomposites)101,102 Rigid, impervious nanoclay layers may decrease heat conduction; thus, their presence in nanocomposite formulations not only limits the mobility of polymer chains but also inherently improves composites thermal performance) 103 The pores created by the crystalline structure of nanoclay cloisite 20A act as some thermal insulation sites leading to enhanced thermal performance.104–106
However, Lei et al. (2007) attested that the presence of 2 wt% nanoclay slightly decreased the degradation temperature of wood flour/HDPE nanocomposites. They attributed this to the released low-molecular-weight compounds, by which the applied nanoclay particles were modified to become organophilic (i.e., nanoclay). 107
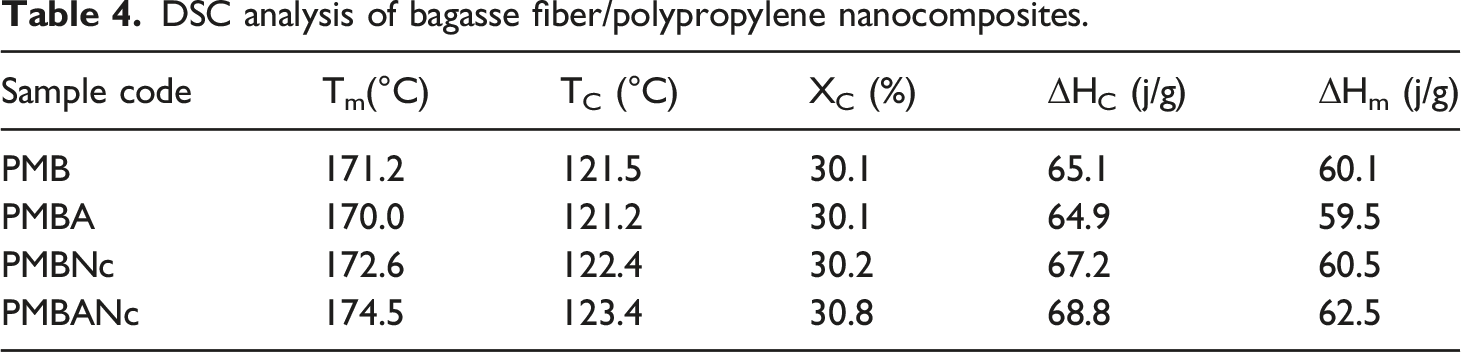
DSC analysis of bagasse fiber/polypropylene nanocomposites.
DSC
The results of DSC analysis including melting temperature (Tm), melting enthalpy (ΔHm), crystallization temperature (Tc), crystallization enthalpy (ΔHc), and degree of crystallinity (Xc) of all the studied composite formulations are given in Table 4.
The DSC results attest that the alkali treatments very slightly impaired the thermal properties of PMBA composites. This is due to lignin, which serves as a cementing agent between the fibers, thus preventing from the penetration of sodium hydroxide into these areas.96,97 The FTIR results (Figure 7) also confirmed that alkaline treatment could not efficiently remove lignin in the composites.
It has been reported that higher concentrations of alkali treatment would significantly weaken thermal stability due to the combined effects of transformation of cellulose and crystallinity, which need be further studied. Also, the decreased crystallinity of WPCs was attributed to the hindering effect of wood fibers on the formation of polymer crystals. 96 Kallakas et al. (2015) declared that Tc and Xc of PP/WF composites treated with 5wt% NaOH were higher than those of the untreated composites due to better nucleatin ability of the treated wood fibers, 109 which expedite polymer crystallisation, thereby increasing crystallisation temperature (Tc). 110 Furthermore, Alkali treatment can increase ΔHc by increasing crystallisation in heterogeneous nucleation spots. This, therefore, enhances crystallization rate (Xc) in alkali treated composites, as compared to that of the untreated ones. 110 The crystallinity index of coir fibers have been reported to improve by alkali treatment due to a partial degradation of cementing materials imparted by the rupture of ester linkages between polyuronic acid and lignin.108,111 PMBNc composites rendered significantly better thermal performance than PMB and PMBA composites.
The melting and crystallization temperatures of nanocomposites have been reported to increase by nanoclay incorporation into the composites. 112 This was attributed to the strong intyerlocking between the polymer molecules and the nanosilicate layers present in, and therefore the immobilization of polymer chains. These immobilised molecules of polymer are responsible for the crystallization process of nanocomposite. Therefore, the crystallization and melting temperatures of the polymer chains in nanocomposites shifted to higher values. 113 To note that polymer chains can crystallize either via a self-nucleation process (homogeneous nucleation) or by incorpotating a nucleating agent, namely nano reinforcing or filler particles (heterogeneous nucleation). 114 The enhanced crystallinity can be ascribed to the intercalation of polypropylene chains between MMT silicate layers and the possible interactions between them, where MMT nanoplatelets would serve as nucleating agents in PP/rice husk nanocomposite. 115 It has also been asserted that the impervious crystalline regions improve stress transfer and energy absorption, thus enhancing composites’ overall mechanical performance. 116 There are as well some contadicting results affirming that organoclay incorporation in to the polymer brought about an 18% decrease in the crystallininty rate of composites due to the creation of some disturbances in the crystalline structure of polymer matrix by the given nano fillers. 116 The results of DSC analysis conducted on PMBANc composites demonstrated that the combined nanoclay and alkali treatment treatments could markedly raise melting temperature, melting enthalpy, crystallisation temperature, crystallisation enthalpy, and crstallinity rate of the given composites (Table 4). This can be attributed to the degradation of certain amounts of wax, oil, lignin, and hemicellulose compounds, the least thermally stable portions of wood fibers by alkaline treatment. This leadsto the more and better accessibility of nanoclay/silica particles to the exposed cellulose fiber moieties, i.e., hydroxyl groups, and thus stronger interfacial adhesion and higher thermal performance due to the nucleating effect of nanoplatelets.
Conclusion
The individual and combined effects of alkali and nanoclay cloisite 20A treatments on structure-property relationships of totally waste-based recycled polypropylene/bagasse fiber composites were studied; the results are as follows: 1. Alkaline and nanoclay treatments had positive effects on the flexural strength and MOE of the studied composites, in particular when a combination of both treatments was applied; this was attributed on one hand to the modification of fiber surfaces by NaOH leading to improved polymer-fiber interfacial adhesion and on the other hand to the resulting better dispersion of organically modified nanoclay in composite structure. The individual and, more specifically, the combined treatments could improve the physical properties (i.e., water absorption and dimensional stability) of the given composites. 2. The impact strengths of PMBA and PMBNc composites were lower than the reference composites due to the impaired impact energy absorption of the strong fiber-polymer interfaces in the former composites and the restriction of polymer chain movements by nanoparticles in the latter composite. It was interestingly observed that the impact strength increased when the combined treatment was introduced; this was ascribed to the probable coagulation of nanoclays in the interface of NaOH-treated fibers-polymer, weakening fiber-polymer interfacial adhesion, and thus, the occurrence and propagation of micro-cracks in the impact test would possibly increase the energy-absorption capacity of PMBANc composites. The sodium hydroxide treatment of bagasse fibers reinforces the interaction between wood-polymer phases, bringing about strong adhesion. 3. TGA and DSC analyses revealed the improved thermal properties of nanoclay and combined treatments on composite materials. Nanoclay hindered the progress of heat towards the deeper layers of composites by forming a carbonized heat insulating char layer. Although alkali treatment had no distinct effect on PMBA composites, the combined treatment could significantly enhance thermal properties in PMBANc samples. 4. It can be safely concluded that totally waste-based bagasse fiber/recycled polypropylene composites could be significantly valorized for different applications by the synergistic effects of nanoclay/alkali treatments.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.