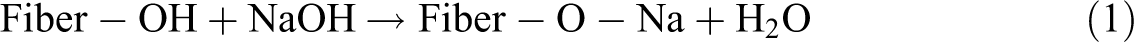Abstract
The goal of this research is to investigate the thermal, chemical, and tensile properties of chemical modification of sugarcane bagasse (SCB)-filled polypropylene (PP) and recycled acrylonitrile butadiene rubber (NBRr). The composites with different SCB loading (5, 15, and 30 per hundred resin) were prepared using a heated two-roll mill at temperature of 180°C.Thermal and the tensile properties of the modified SCB composite have shown improvement. The silane-treated composites have higher thermal stability compared to treated NaOH. The degradation temperature at 70% weight (T 70%) of NaOH and silane composite increase by 6% and 15%, respectively. Meanwhile, the tensile strength and Young’s modulus for the both treatment showed an improvement of 20% and 25% for NaOH and 30% and 32% for silane compared to untreated composites, respectively. The chemical properties were investigated using Fourier transform infrared analysis. The modification SCB fiber has improved the adhesion and interfacial bonding between SCB fiber and PP/NBRr matrices.
Keywords
Introduction
Due to the hydrophobic effect between the hydrophilic fiber and hydrophobic matrices, many studies on chemical modification or treatment had been carried out to remove the hydroxyl groups from lignin and cellulose of natural fiber. Removal of such hydroxyl group may improve strength and fitness of fiber as well as adhesion between matrices and fiber composites. 1 For example, alkaline treatment, silane treatment, acetylation treatment, peroxide treatment, and other chemical treatments had been studied and applied on treating the natural fiber. The kenaf core wood had been studied by previous researchers using alkaline treatment such as NaOH to remove the lignin and hemicellulose. 2 Thermal properties of oil palm mesocarp fibers has shown higher crystallinity and thermal stability compared to untreated fiber. 3 On the other hand, to improve the mechanical properties and decrease the water and oil absorption of composite, silane treatment had been employed to polypropylene (PP)/acrylonitrile butadiene rubber (NBRr)/rice husk powder (RHP) composite. 4
Alkaline treatment is one of the most common chemical treatments used for natural fibers for thermoplastics and thermosets. Alkaline treatment modifies the surface of fiber by disrupting hydrogen bonding of cellulose in the network structure, thus increasing surface roughness. 5 This treatment will eliminate a certain amount of lignin, wax, oils, and impurities covering the external surface of the fiber cell wall, depolymerizes cellulose, and exposes the short length crystallites. Addition of aqueous sodium hydroxide (NaOH) to natural fiber will ionize the hydroxyl group to alkoxide 6 : The alkaline treatment equation is shown in equation (1)
Thus, alkaline treatment will directly affect the cellulosic fibril and degree of polymerization of lignin and hemicellulosic compounds. 7,8 It was found that alkaline treatment has two effects on the fiber: It is capable of increasing surface roughness and increases the number of possible reaction sites exposed on the fiber surface. There explains the reinforcement phenomenon of mechanical properties with NaOH-treated fiber. 3,8 –10 However, higher concentration of alkaline will undergo fiber degradation and delignification phenomenon. 11
Meanwhile, silane is a chemical compound that has a chemical formula of SiH4. Silanes are used as coupling agents to decrease the number of cellulose hydroxyl groups at the surface of fiber. Hydrolysable alkoxy group with the presence of moisture is able to lead to the formation of silanols. The silanol forms stable covalent bonds to the cell wall that are chemisorbed onto the fiber surface by reacting with the hydroxyl group of the fiber. Therefore, the hydrocarbon chains provided by the application of silane prevent the swelling of the fiber by producing a cross-linked network caused by covalent bonding between the matrix and the fiber. The reaction of silane with fiber can be seen in Figure 1. 12
Reaction mechanism during the silane treatment of sisal fiber. 12
In general, interaction of silane coupling agents with natural fillers may be processed through the following steps 13 : (1) Hydrolysis: the silane monomers are hydrolyzed in the presence of water and catalyst (normally acid or base), liberating alcohol and yielding reactive silanol groups. (2) Self-condensation: During the hydrolysis process, the concomitant condensation of silanols (aging) also takes place. The condensation should be minimized at this stage to leave the silanols free for being adsorbed to the hydroxyl groups in the natural fillers. For the bulking treatment of fillers, the condensation should also be controlled to retain a small molecular size of monomers or oligomers to diffuse into the cell walls. The condensation rate of silanols is controllable by adjusting the pH of the hydrolysis system. An acidic pH environment is usually preferable to accelerate the hydrolysis rate of silanes, but it slows down the condensation rate of silanols. (3) Adsorption: The reactive silanols monomers or oligomers are physically adsorbed to hydroxyl groups of natural fillers by hydrogen bonds on the fiber surface (surface coating) and/or in the cell walls (cell wall bulking), which depends on the molecular size of silanol monomers/oligomers formed. The free silanols also adsorb and react with each, thereby forming a rigid polysiloxane structures linked with stable Si–O–Si bond. (4) Grafting: Under heating conditions, the hydrogen bonds the silanols and the hydroxyl groups of fillers can be converted into the covalent Si–O–C bonds and liberating water. The residual silanol groups in the fibers will further condense with each other. The bonds of Si–O–C may not be stable toward hydrolysis; however, this bond is reversible when the water is removed at a raised temperature.
Research had been done using silane treatment in surface modification of natural fiber composites, and the results show that silane coupling agents were found to be effective in modifying natural fiber–polymer matrix interface and improving the interfacial strength. The mechanical properties of composite after treatment had improved compared to untreated composite. It was proven that the interaction between the silane coupling agent modified fiber and the matrix was better than treated using alkaline. It then led to composites with higher tensile strength from silane-treated than alkaline-treated fiber. 14,15 Silane treatment also shown improvement on thermal stability of the composites.
Therefore, in this research, two types of treatment had been proposed: NaOH and 3-aminopropyltrimethoxysilane (3-APS) to compare the chemical and thermal tensile properties and morphology of fiber and the PP/recycle NBRr/sugarcane bagasse (SCB) powder composite after treated by the alkaline treatment and the silane treatment.
Experiment
Material
PP used in this work was supplied by TitanPro Polymers (M) Sdn, Bhd, Johor, Malaysia (code 6331) having a melt flow index and density of 14 g/10 min at 230°C and 0.9 g cm−3, respectively. The recycled NBRr glove with a density of 0.98 g cm−3 was obtained from Juara One Resources Sdn, Bhd, Penang, Malaysia. NaOH and 3-aminopropyltriethoxysilane (3-APS silane) was supplied by Alfa Aesar (M) Sdn. Bhd. (Malaysia). NBRr with particle size 300–150 μm was used in this research. The SCB was collected from KilangGulaFelda Perlis Sdn. Bhd. Perlis, Malaysia. The SCB was dried at 80°C for 4 h in the oven and grinded into 300–150 μm particle size prior to its usage.
SCB treatment
NaOH treatment
NaOH pellets and acetic anhydride were brought from Alfa Aesar (M) Sdn. Bhd. Five percent of NaOH pellets was mixed with 1000 ml of distilled water. One hundred grams of SCB was added into the NaOH aqueous solution and stirred for 2 h. After that, the SCB was rinsed with distilled water for several times to remove the alkalinity. Next, 3 ml of acetic anhydride were added and mixed with the SCB powder followed by drying them in oven at a temperature of 60°C for 48 h. After 48 h, the treated SCB were sieved to get a particular size of 150–300 μm. Table 1 presents the chemical specification of NaOH used in this research.
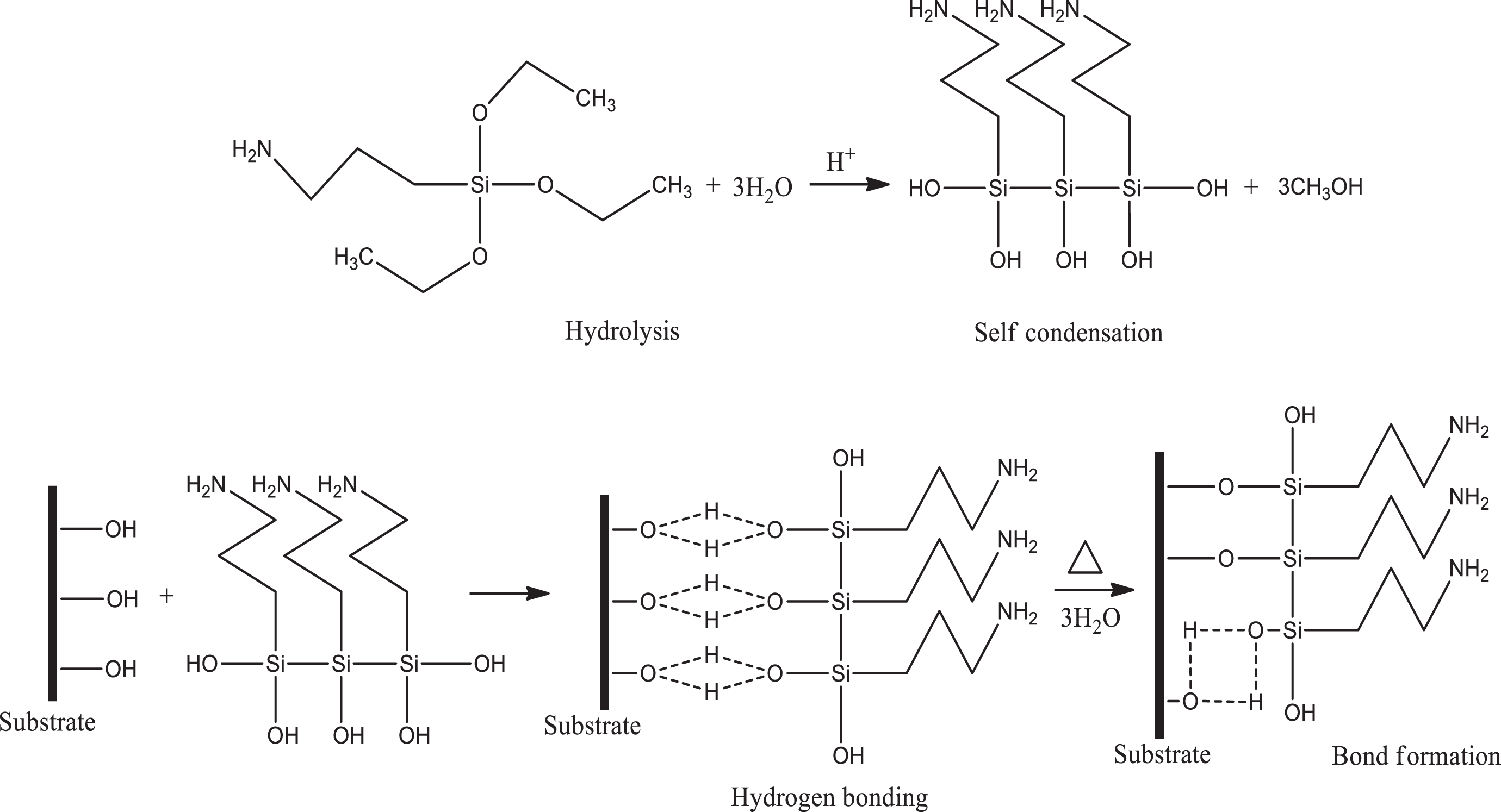
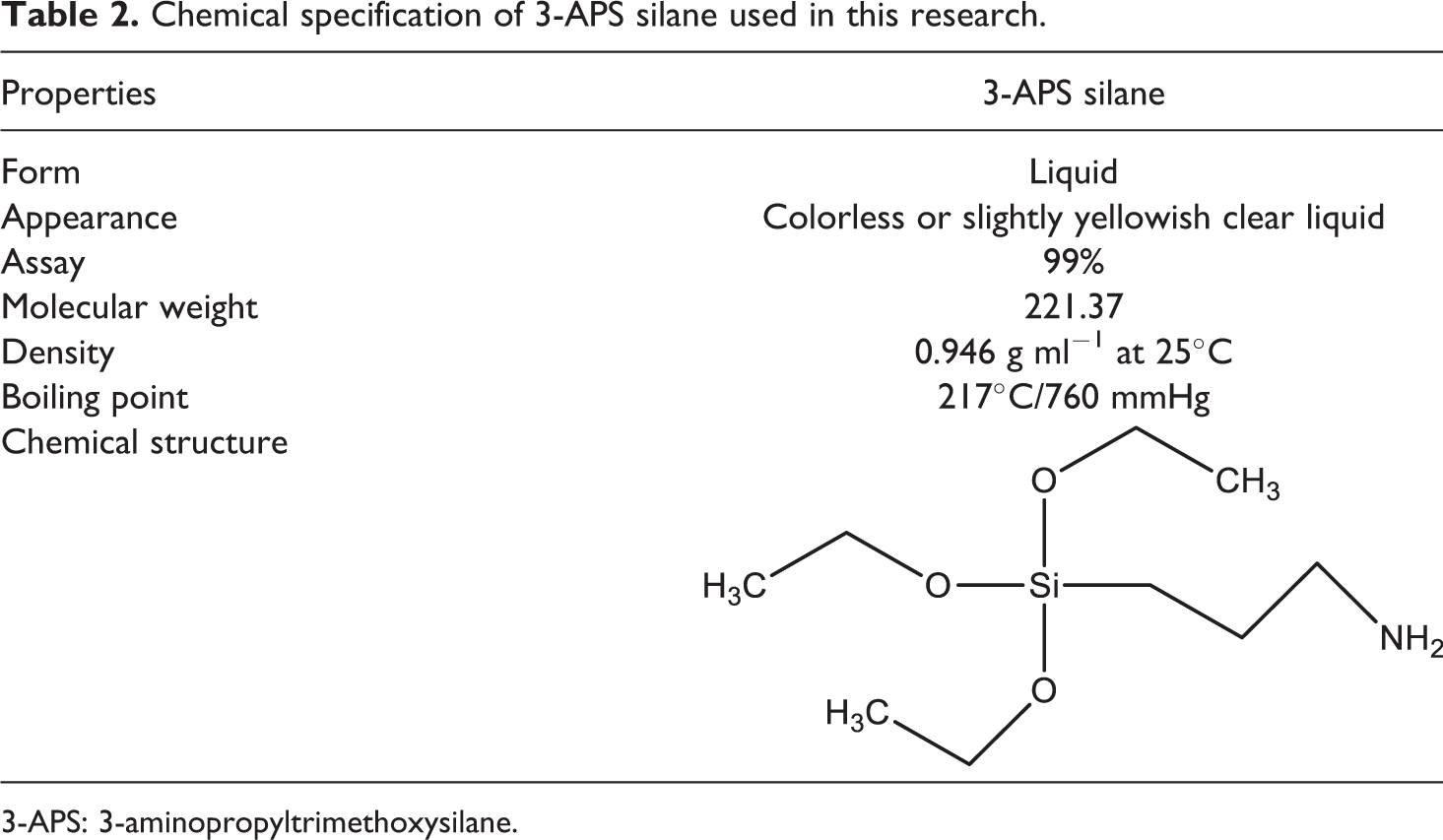
Chemical specification of 3-APS silane used in this research.

3-APS: 3-aminopropyltrimethoxysilane.
3-APS treatment
Firstly, SCB filler was immersed in a mixture of water and ethanol (40/60 volume, respectively) and stirred for 1 h at 60°C. After that, the SCB was collected using a filter. Five percent of 3-APS was first introduced into 1000 ml of the mixture water/ethanol and was allowed to stand for 1 h. The pH of the solution was maintained at 4 with the addition of acetic acid. Then, 100 g of SCB was added into the solution and the solutions were continuously stirred for 1.5 h. The filler was filtered and dried in the vacuum oven at 80°C for 24 h. All treated SCB particles were crushed using grinder to ensure homogenous size of 150–300 μm. Chemical specification of 3-APS silane used in this research is shown in Table 2.
Chemical specification of 3-APS silane used in this research.
3-APS: 3-aminopropyltrimethoxysilane.
Preparation of composites
The PP and NBRr loading in the fabrication of PP/NBRr/SCB composites were fixed at 70 and 30 phr, respectively. Meanwhile, the SCB filler loading was manipulate at 5, 15, and 30 phr. Same formulation is used in treated NaOH and silane composites. The composites were prepared by melt mixing technique using a heated two-Roll mill mixer at 180°C with the rotor speed of 15 r min−1. PP was charged into the mixing for 4 min prior to NBRr. At 6 min, SCB filler was added and the mixing process was continued for another 3 min for a total mixing time of 9 min. Then, the compound materials are shaped in 1 mm thickness sheet by compression molding followed by cool pressing. An electrically heated hydraulic press model GT-7014-A300C with force of 15 MPa was used for this purpose. The electrically heated hydraulic press was heated up to 180°C for 7 min preheating, 2 min compressing, and 2 min of cooling down, resulting total preparation time is 11 min. After the cooling process, the samples are stored at room temperature for 24 h. The samples are ready to been cut into the dumbbell shape using a Wallace Die Cutter and the sample will be taken to test.
Characterizations
Thermogravimetric analysis
Thermogravimetric analysis (TGA) of the composites is carried out with Perkin Elmer Pyris 6 TGA analyzer at Universiti Malaysia Perlis, Malaysia. The sample’s weight of about 15–16 mg is scanned at a heating rate of 10°C min−1 and from 30°C to 500°C. The scan is carried out using nitrogen flowing rate of 50 ml min−1. The degradation temperature at 70% weight, T 70% and final degradation temperature T deg of the composites are analyzed.
Differential scanning calorimetry
The crystallization behavior and melting characteristics of the composites were studied using differential scanning calorimetry (DSC) model Perkin-Elmer DSC-7 analyzer based on ASTM D3418-03. The samples of about 4 mg average weight were encapsulated in a hermetically sealed aluminum pan. The samples then were heated from 30°C to 220°C using nitrogen flowing rate of 50 ml min−1 at a heating rate of 10°C min−1 and held at 220°C for 5 min to remove the thermal history. The sample was then cooled from 220°C to 30°C with cooling rate of 10°C min−1 and then reheated back. The degree of crystallinity of PP/NBRr is calculated using the following equation (2)
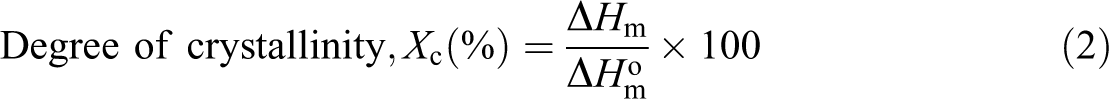
where ΔH
m is the melting enthalpy of the composite, and
Fourier transform infrared analysis
Fourier transform infrared (FTIR) analysis was done to evaluate the chemical composition composite. For sample in solid and sheet form, the sample has been cut into small rectangular sheets and is use directly with the FTIR machine. FTIR spectra are record by plotting wavelength against absorbance. The spectrum resolution is 4 cm−1 and the scanning range is from 550 to 4000 cm−1.
Tensile test
The tests are performed at room temperature on a Universal Testing Machine model Instron 3366 at Universiti Malaysia Perlis, Malaysia. The initial jaw separation distance of the tensile tester was 50 mm and the testing is carried out at a cross head speed of 5 mm min−1. The tests recorded the tensile properties of sample via tensile strength, elongation at break (E b), and Young’s modulus.
Morphological properties
Studies on the morphology of the composites were performed using a scanning electron microscope (SEM) machine (FESEM ZEISS SUPRA36VP-24-58) at Universiti Malaysia Perlis, Malaysia. All samples were coated with a thin platinum layer using Sputter Coater Polaron (Universiti Malaysia Perlis, Malaysia). SC 515 to avoid electrostatic charging and poor image resolution during the examination. The morphology of tensile fracture surface samples was taken from the fracture samples from the tensile test.
Result and discussion
TGA and derivative thermogravimetry
TGA and derivative thermogravimetry (DTG) had been carried out to determine characteristic of the decomposition and the thermal stability of materials. The T 70%, residue weight at 600°C, and DTG peak degradation temperature had been summarized in Table 3. Meanwhile, Figures 2 and 3 show the TGA and DTG graph of untreated, NaOH, and silane PP/NBRr/SCB composites at composition 70/30/15 phr, respectively, for better view.
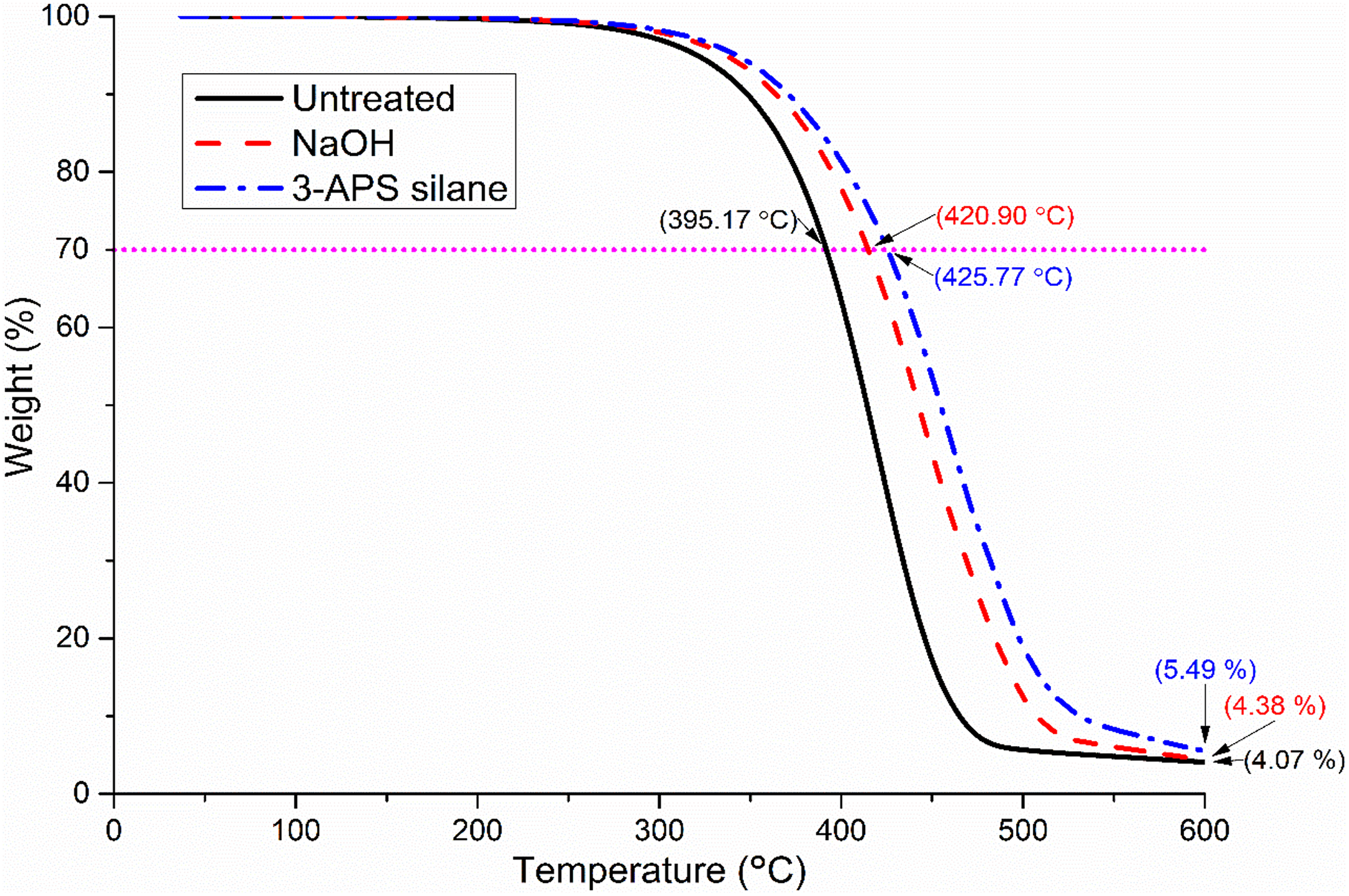
TGA curve of untreated, NaOH, and silane PP/NBRr/SCB composites at composition 70/30/15 phr.
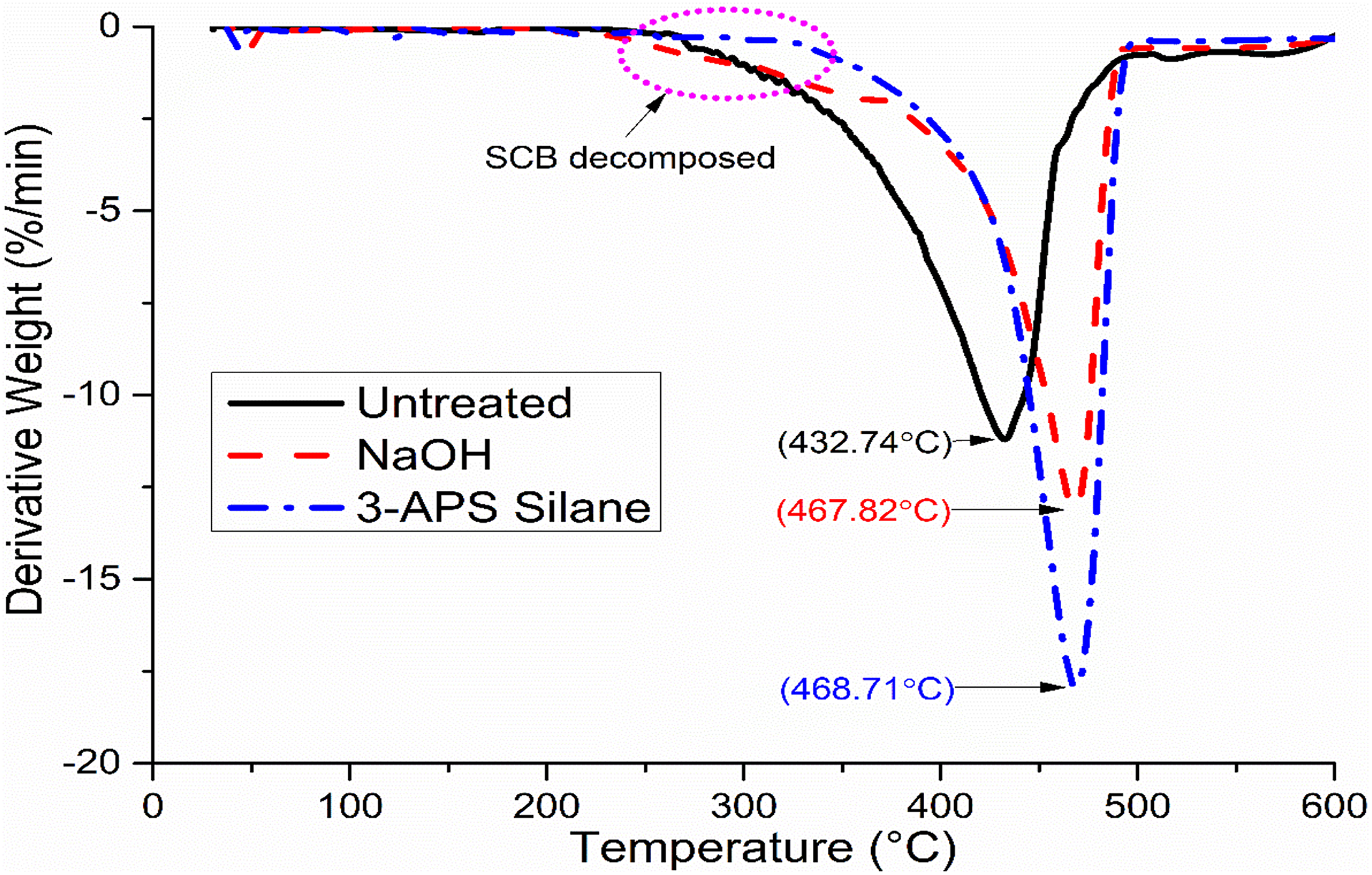
DTG curve of untreated, NaOH, and silane PP/NBRr/SCB composites at composition 70/30/15 phr.
Thermal properties of PP/NBRr/SCB composites.
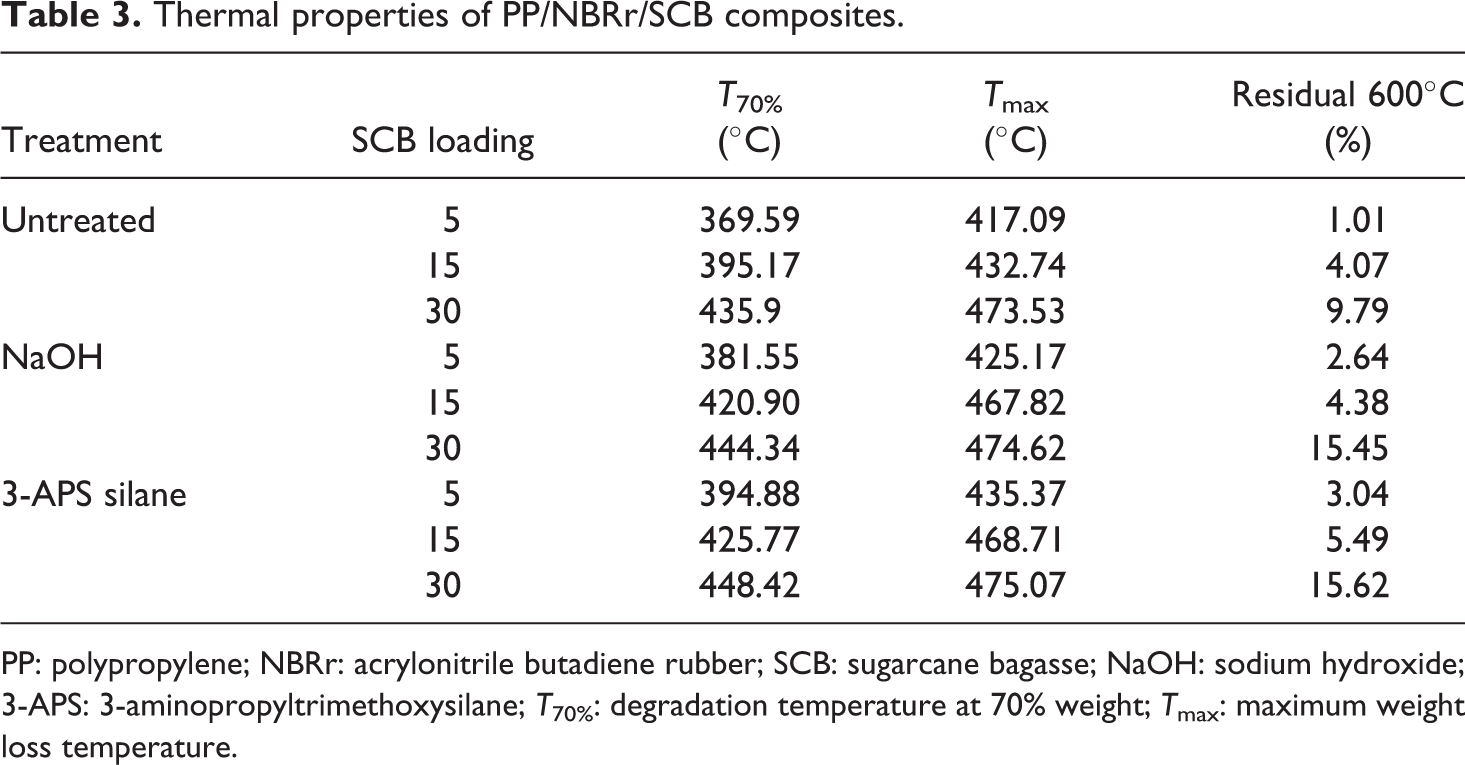
PP: polypropylene; NBRr: acrylonitrile butadiene rubber; SCB: sugarcane bagasse; NaOH: sodium hydroxide; 3-APS: 3-aminopropyltrimethoxysilane; T 70%: degradation temperature at 70% weight; T max: maximum weight loss temperature.
The T 70% increases with the increase of SCB filler content. The presence of SCB filler is found to improve the thermal stability of the composites. 16 The increasing of thermal stability of the composites was due to the presence of high cellulose fiber content in the SCB. The T 70% untreated and treated composites (NaOH and silane composites) will have higher values due to enhancement of the interaction between filler and matrix by NaOH and silane that leads to better dispersion of SCB particles in the PP/NBRr matrices. 9,17 The result is supported by the tensile properties result in earlier section. The thermal stability of the silane treated composites was also improved by higher tensile strength that is caused from stronger covalent bonding when compared to hydrogen bonding from NaOH treatment.
Based on Figure 3, it can be noticed that two curves were observed, which is due to the occurrence of two stages of decomposition in the composite. The first stage was associated with decomposition of SCB filler. Hemicelluloses and lignin of SCB filler were decomposed at temperature ranges of 220°C to 340°C. The second stage degradation was primarily due to NBRr and PP. Results from Table 3 show that the untreated PP/NBRr/SCB composite has the lower maximum weight loss temperature (T max) compared to NaOH and silane composites. The incompatibility of polar SCB filler and non-polar PP/NBRr matrices had reduced the thermal properties and temperature needed to degrade the materials. However, silane composite has higher T max followed by NaOH composites. This may be due to the better interaction and adhesion between silane-treated SCB filler and PP/NBRr matrices. Therefore, higher temperature was required to break the bonding formed and adhesion between filler and matrices.
Meanwhile, the char residue of samples after heating can show thermal stability as well. The silane-treated composite had higher char percentage than NaOH followed by untreated composite. It may be due to better filler and matrices interaction of treated composite that resulted in the formation of char layer. The formation of char layer at the materials will provide better thermal protection and thus improve thermal stability. 18
The thermal stability of the PP/NBRr matrix reinforced with SCB was improved by NaOH and silane treatment. The T 70% of NaOH and silane composite increases by 3–6% and 6–15%, respectively. Moreover, the T max of composite also increases using treated SCB in PP/NBRr composite by 3% for NaOH and 11% for silane composites. Meanwhile, the char residual at 600°C increases with increasing the SCB loading in the composites.
Differential scanning calorimetry
The thermal properties of temperature of untreated, NaOH-treaded, and silane-treated PP/NBRr/SCB composites using differential scanning calorimetry (DSC) have been analyzed in Table 4. The crystalline temperature, T c; melting temperature, T m; enthalpy of melting, ΔH m, and the percent of crystallinity, X c, of composites were recorded. The highest T c and T m were for PP/NBRr/SCB composite treated with silane, followed by NaOH composites, and finally untreated composites. This is due to the incompatible composite without treatment caused by non-polar hydrophobic matrices and polar hydrophilic fiber which leads to the poor interfacial properties 19 and thus had the lowest melting point and crystalline point. Furthermore, the untreated PP/NBRr/SCB composite had the lowest percentage of crystallinity, because the fiber between the polymers block the polymer chain and cannot pack together closely to form crystalline.
Thermal properties of temperature of untreated, NaOH, and silane-treated PP/NBRr/SCB from DSC.
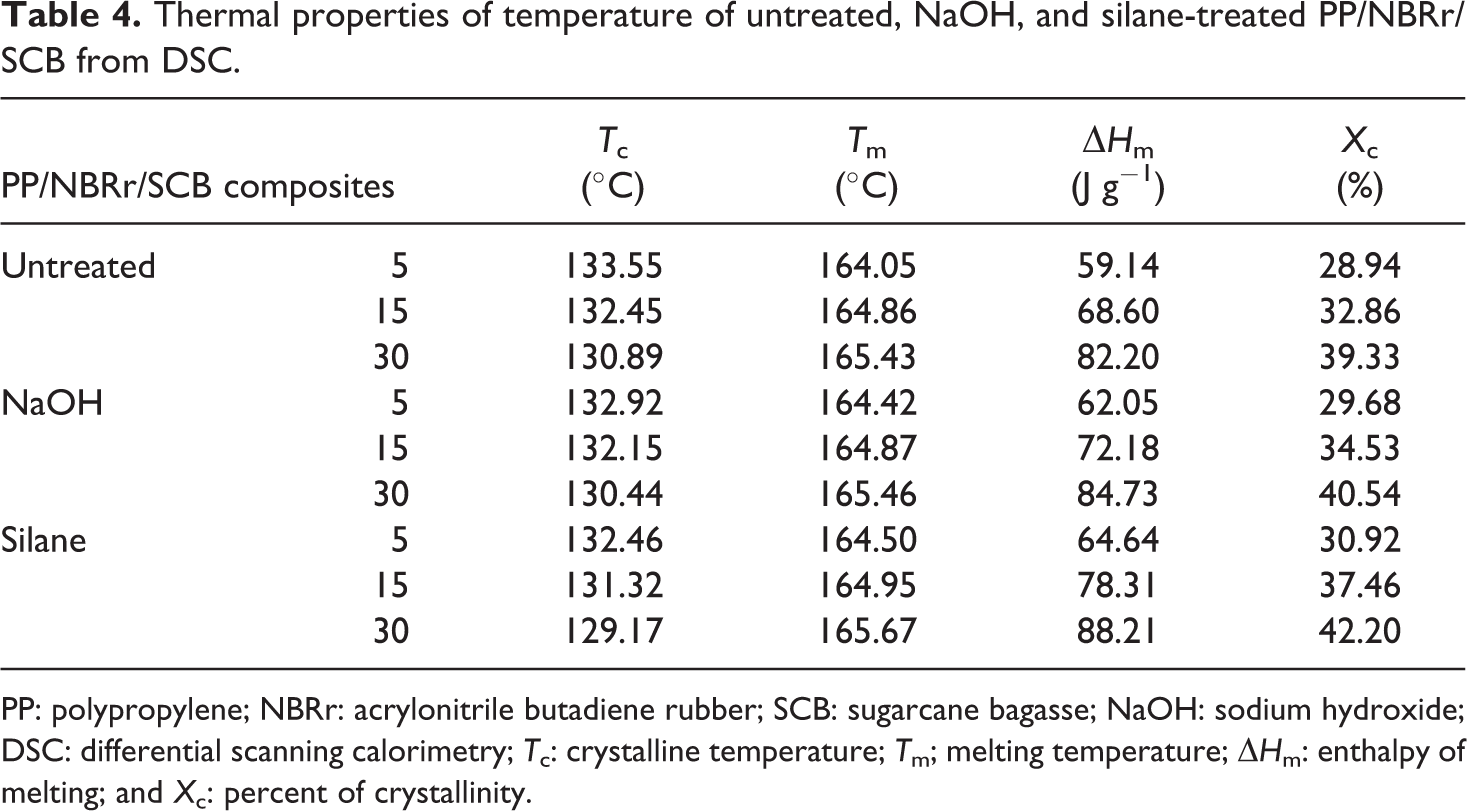
PP: polypropylene; NBRr: acrylonitrile butadiene rubber; SCB: sugarcane bagasse; NaOH: sodium hydroxide; DSC: differential scanning calorimetry; T c: crystalline temperature; T m; melting temperature; ΔH m: enthalpy of melting; and X c: percent of crystallinity.
However, in NaOH composites, the surface treatment of SCB filler has been found to remove impurities and increase the surface functionality of the SCB filler, causing enhanced surface affinity with PP/NBRr which further increases the crystallinity. Meanwhile, the silane treatment composites played an important role in surface wetting of SCB filler by PP/NBRr matrices, which leads to further improvement in crystallinity (%). Similar observations were reported by Pilla et al. 20 on polylactide-pine wood flour composites. Furthermore, Huda et al. 21 reported that surface treatment of the natural fibers using silane increases the nucleation density and enhances the crystallinity (%) of the polylactic acid (PLA) in the composites.
The 3-APS silane-treated composite has higher increase by 0.09–0.45°C for T m, 6.0–12.5 J g−1 for ΔH m, and 2.8–5.9% for X c compared to NaOH increase by 0.01–0.37°C for T m, 2.5–9.9 J g−1 for ΔH m, and 1.2–4.7% for X c. This is due to the increase in the nucleation density of the treated SCB filler, which increases the nucleation density and which enhances the crystallinity percentage. Figures 4 and 5 show the DSC curves of cooling and melting PP/NBRr/SCB composites at 70/30/15, respectively.
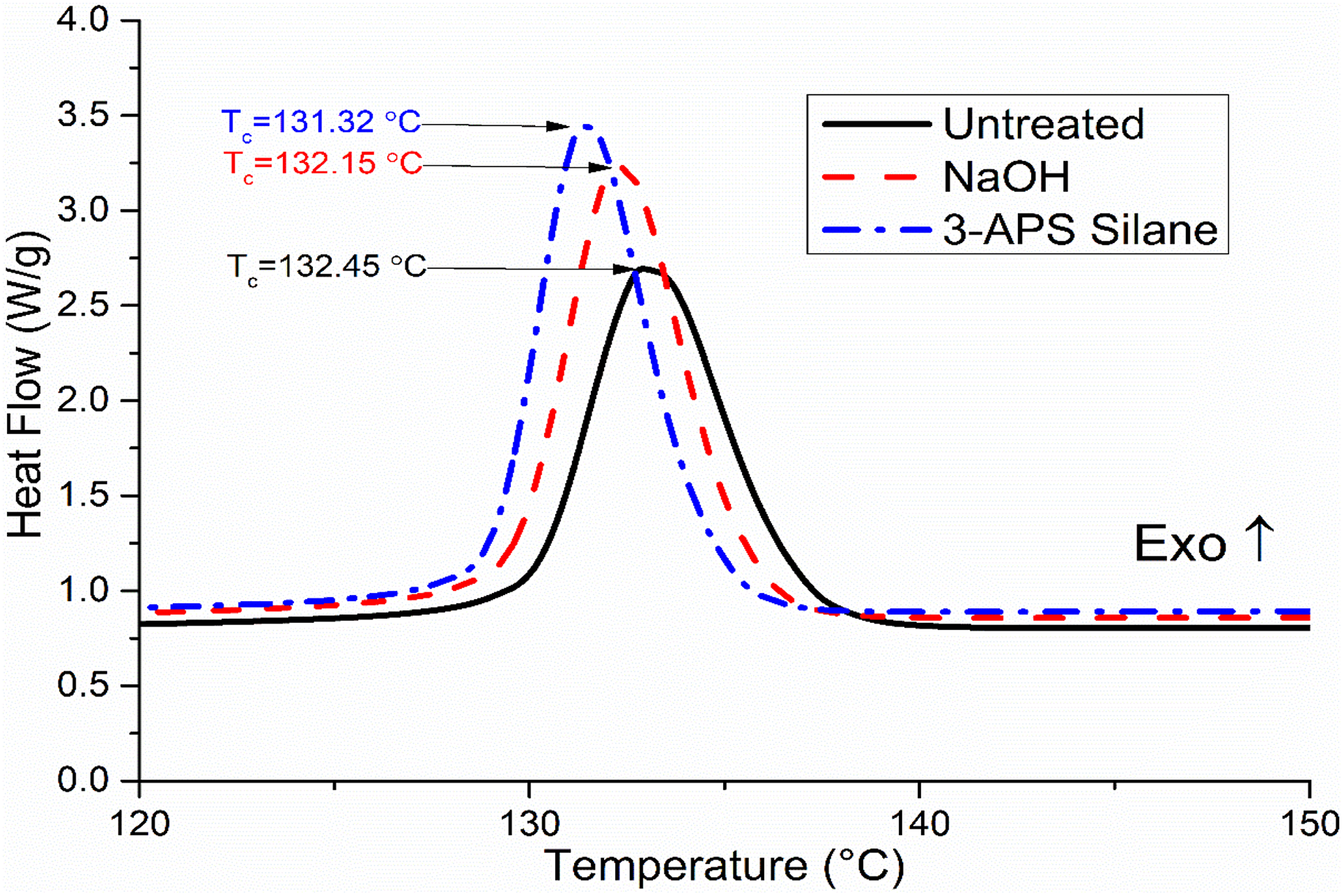
DSC curves of cooling PP/NBRr/SCB composites at 70/30/15.
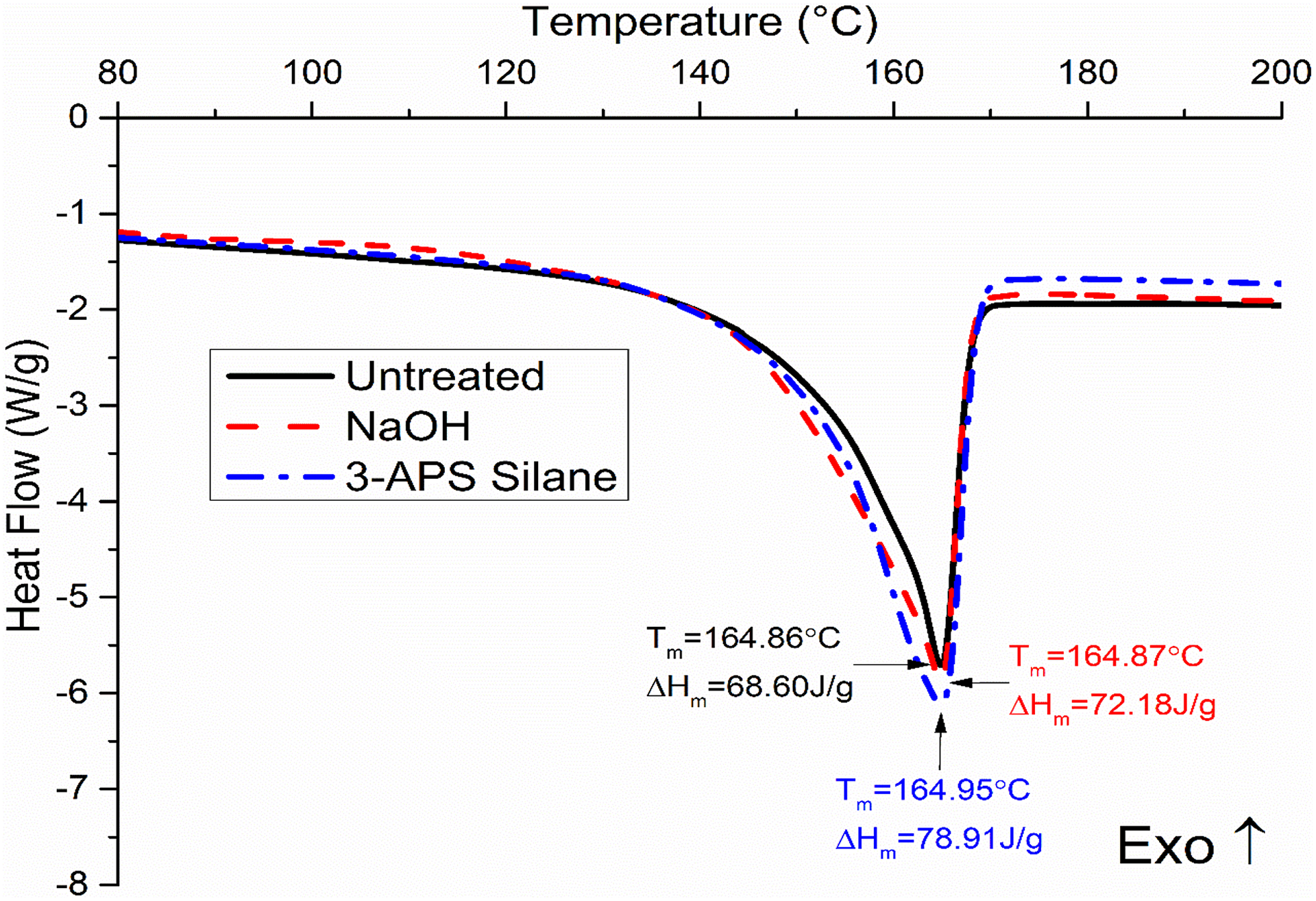
DSC curves of melting PP/NBRr/SCB composites at 70/30/15.
FTIR spectroscopy
Table 5 presents the infrared characteristics modes of vibrations for the major absorption peaks in all materials. Figure 6 shows the FTIR spectra of SCB filler and PP/NBRr/SCB composites with and without treatments. The untreated composite shows that broad absorption band in the 3359.8 cm−1 was related to the hydrogen bonding (OH) stretching vibration. With NaOH and silane treatment, the intensity of the band decreases. 22 The vibration peak at 2916.2 cm−1, corresponding to the stretching of the C–H aliphatic group, was included in almost all natural fibers. This peak decreased due to the removal of hemicelluloses. 23 It is also clearly noticeable that, with NaOH treatment, the band around 1731.9 cm−1 corresponding to non-cellulosic components (pectin, lignin, and hemicelluloses) disappeared. The peak at 1654.8 cm−1 would be due to the presence of lignin. 24 The 1454 cm−1 band was attributed to the CH2 bending of cellulose. The peak at 1253.6 cm−1, which is C=O stretch of acetyl group of lignin, was reduced. This was due to the partial removal of lignin from the fiber surface. Alkali treatment is expected to reduce the hydrogen bonding in cellulose hydroxyl groups by the removal of the carboxyl group. 25 Meanwhile, the silane composite that showed bands around 1162 and 1105 cm−1 was assigned to the stretching of the –Si–O–cellulose and –Si–O–Si bond, respectively. The large band around 1047 cm−1, present in the spectrum of untreated composite, was attributed to –Si–OH groups. This band disappeared after surface modification and was replaced by a wide band around 1020 cm−1, characteristic of –Si–O–Si– moieties. 17 Similar finding was reported by Zafar et al. 26 Figure 7 shows the schematic representation of the PP/NBRr/SCB filler interface: (a) untreated composite, (b) NaOH composite, and (c) silane composite.
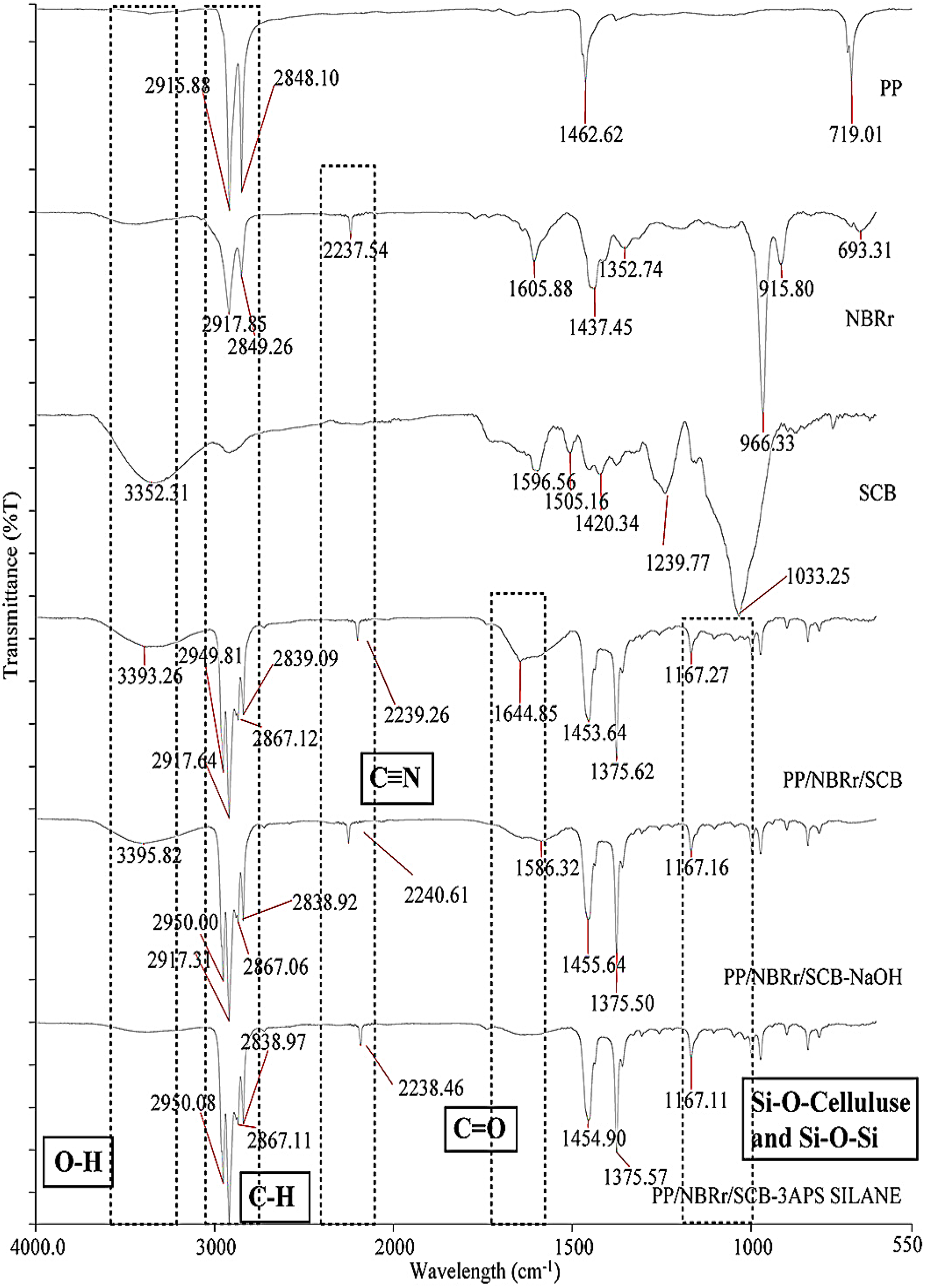
FTIR spectra of PP, NBRr powder, SCB powder, untreated, NaOH, and silane treatment for PP/NBRr/SCB composites at composition 70/30/15 phr.
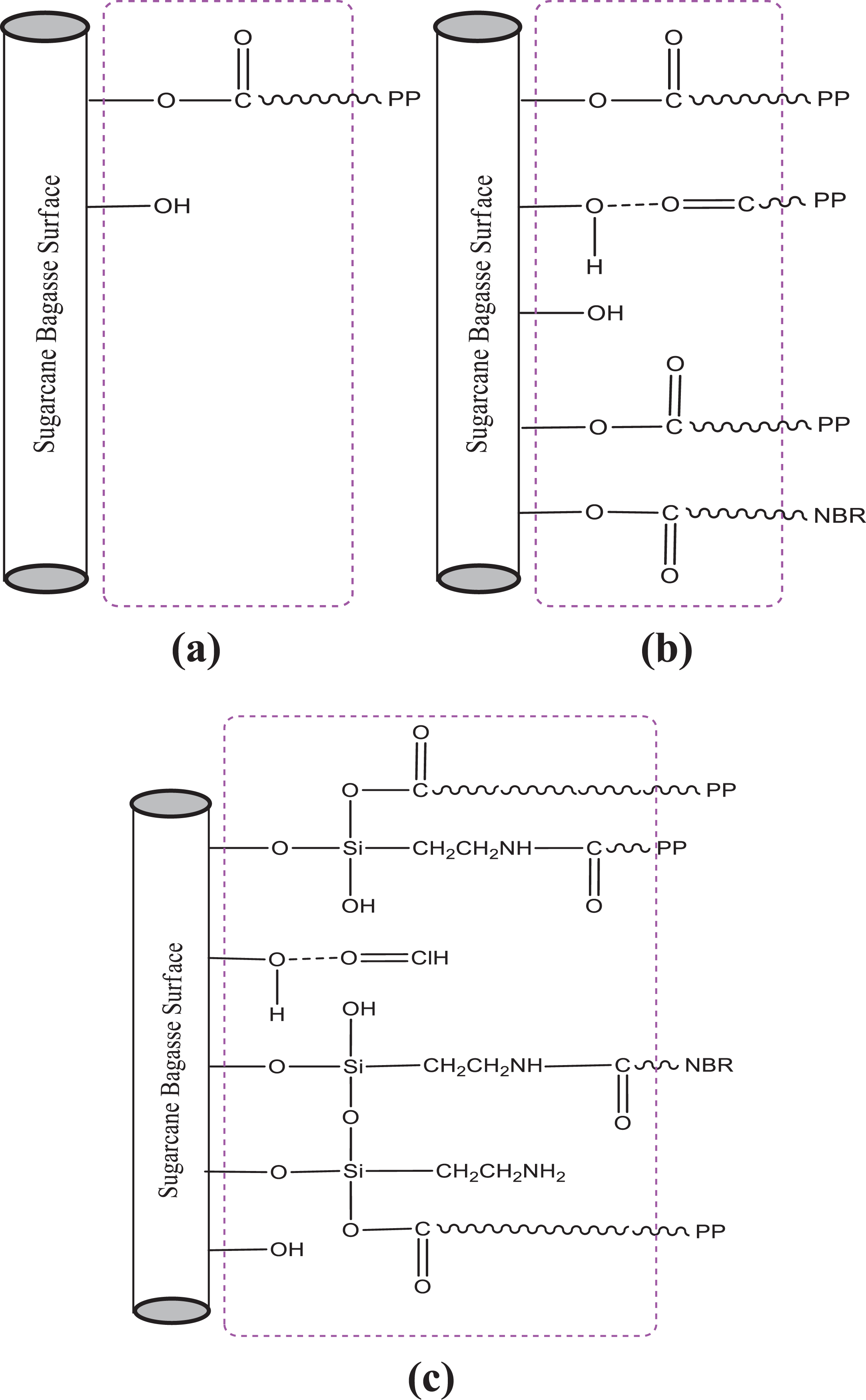
Schematic representation of the PP/NBRr/SCB filler interface: (a) untreated composite, (b) NaOH composite, and (c) silane composite.
Infrared characteristics modes and frequency range.

Tensile properties
Table 6 and Figures 8 to 10 show the tensile strength, Young’s modulus, and E b of PP/NBRr/SCB composites treated with NaOH and 3-APS silane. Based on the result, the untreated PP/NBRr/SCB composites have the lowest tensile strength compared to both treated composites. This is due to the incompatibility of hydrophobic matrices (PP and NBRr) and hydrophilic nature of SCB in the composite. 27 Thus, the interactions of interface between SCB filler and PP/NBRr matrices are weak. However, NaOH and silane treatment composites have improved the adhesion between PP/NBRr matrices and SCB filler, and thus resulted in higher tensile strength than untreated composite. The tensile strength for NaOH composites increase by 4–22% and 16–32% for 3-APS silane composites. The PP/NBRr/SCB composites treated with NaOH resulted in high tensile than untreated composite but lower than silane composite. The NaOH treatment is an evidence that treatment of filler by alkalization helps in improving the mechanical interlocking and chemical bonding between the PP/NBRr matrices and SCB filler resulting in better mechanical properties. 28 The alkali treatment enhances the fibers’ surface adhesion characteristics by removing natural and artificial impurities, as well as producing a rough surface topography. 29 The PP/NBRr/BSP composites treated with silane exhibit highest tensile strength when compared to NaOH composites. The result indicates that the use of silane as coupling agent was proven to be effective in enhancing dispersion, adhesion, and compatibility of systems consisting of hydrophilic filler and hydrophobic matrix through modification of polymer–filler interface. 30
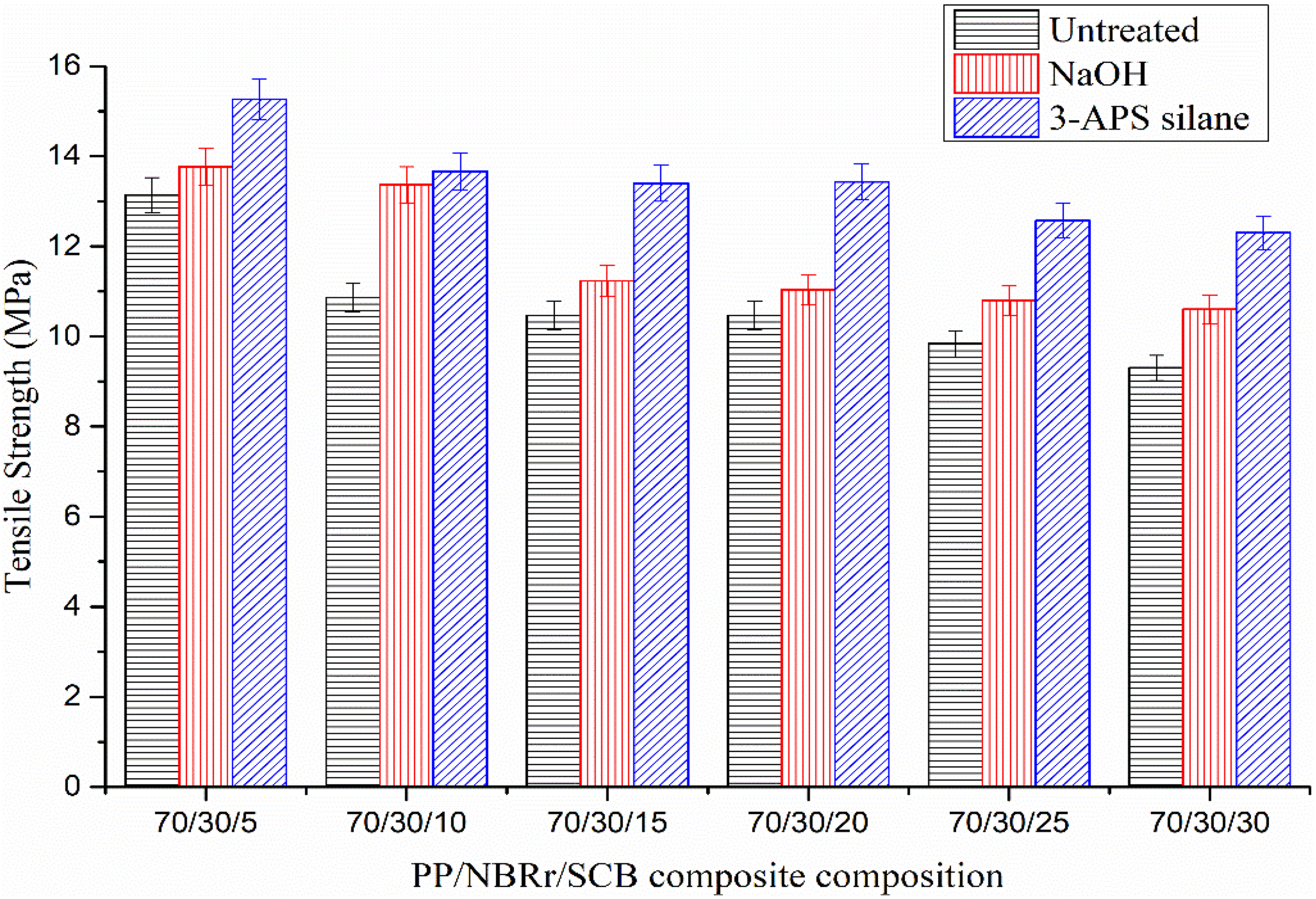
Tensile strength of PP/NBRr/SCB composites treated with NaOH and 3-APS silane.
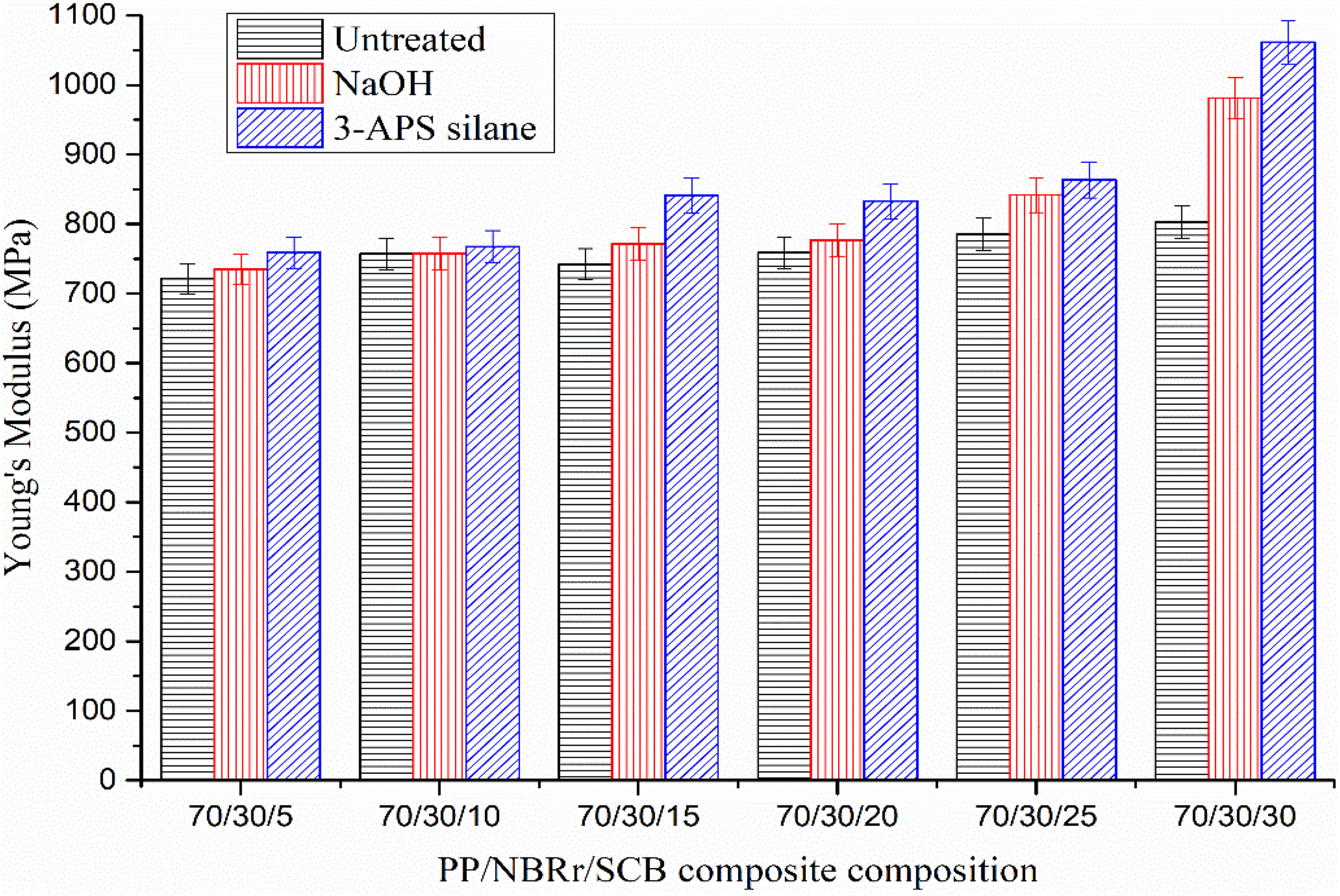
Young’s modulus of PP/NBRr/SCB composites treated with NaOH and 3-APS silane.
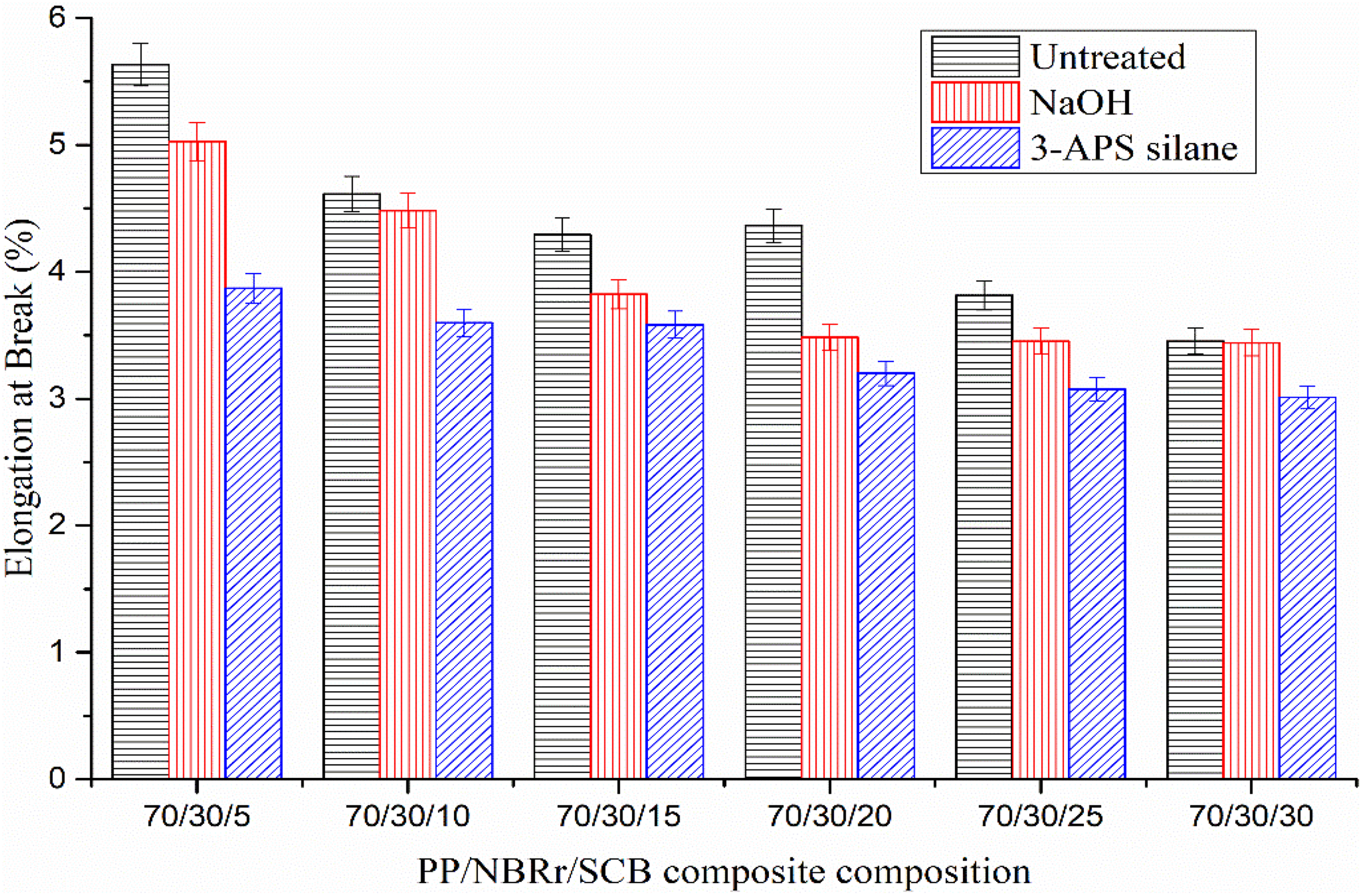
Elongation at break (E b) of PP/NBRr/SCB composites treated with NaOH and 3-APS silane.
Tensile properties of PP/NBRr/SCB composites treated with NaOH and 3-APS silane.
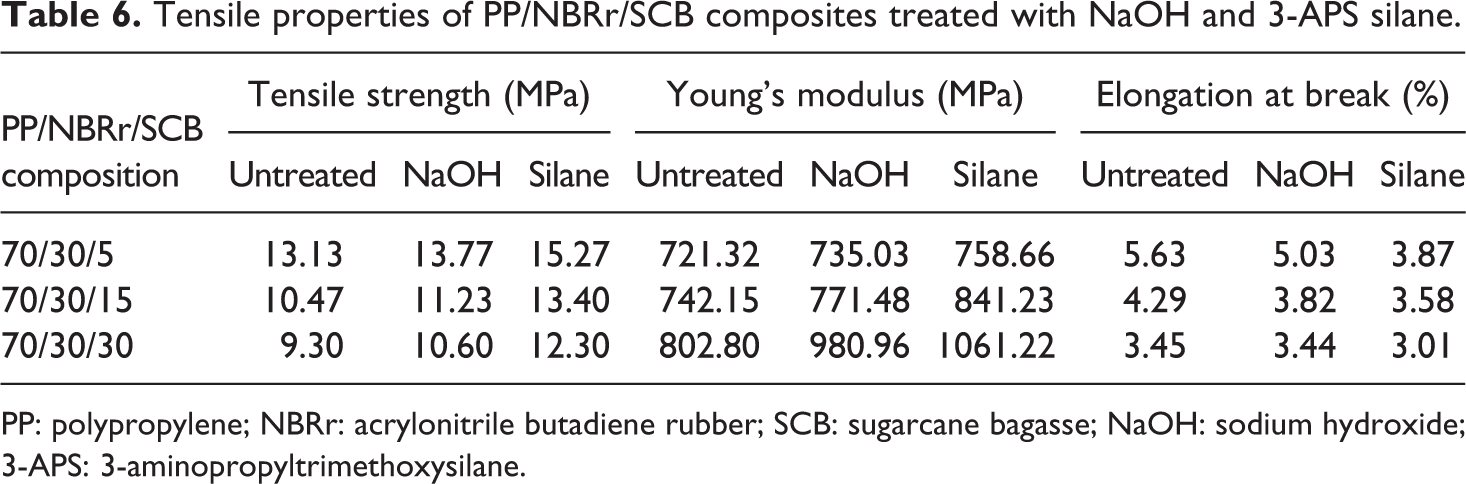
PP: polypropylene; NBRr: acrylonitrile butadiene rubber; SCB: sugarcane bagasse; NaOH: sodium hydroxide; 3-APS: 3-aminopropyltrimethoxysilane.
Figure 9 shows the Young’s modulus of PP/NBRr/SCB composites treated with NaOH and 3-APS silane. The results show that the Young’s modulus of all treated and untreated composites increases accordingly when the filler loading increased. The stiffness of the composites increased during the incorporation of SCB filler into the PP/NBRr matrices. The addition of SCB filler into the PP/NBRr matrices reduces the PP chains mobility and resulted in increasing more rigid composites. 4 Meanwhile, Young’s modulus for silane composites was higher compared to NaOH composites. The better interactions between the matrix and filler will be achieved with the addition of silane coupling agent or undergoing NaOH treatment. Similar finding of previous research had also shown that higher Young’s modulus in the surface of both NaOH-treated jute fibers and modified jute fibers with silane coupling agent was caused by higher rigidity of biocomposites. 9
Figure 10 shows the E b of the untreated, NaOH-treated, and silane-treated PP/NBRr/SCB composites. The results had shown that E b of all composites decreases with the increasing of fiber loading. This is because the increment of filler loading had hardened the composites and thus reduced their ductility. 31,32 The E b for PP/NBRr/SCB composites with silane was the lowest, followed by NaOH composites with decrement 12–31% for silane and 0.2–10% for NaOH, respectively. The lowest E b of silane-treated PP/NBRr/SCB composites was due to the presence of coupling agent that formed chemical bonding inside the composite, thereby enhancing the matrix and filler interior interaction. E b is maximum for untreated composites up to 5 phr of filler content, but then decreased steadily at higher filler content. The presence of NaOH and silane further decreased, even at the lowest filler content. For both the cases, once the composites become harder and stiffer, the E b is certainly lowered. The improved adhesion in the presence of bonding agent restricts the mobility of polymer segments, which finally results in a reduction in elongation. 33 Similar behavior has also been reported by many researchers. 34 –37 They found that the decrease in E b at lower filler content may be due to low E b of the filler, and this restricts the polymer molecules flowing past between each other. Incorporation of silane yield composites with the E b lower than that of NaOH-treated composites and untreated composites. The lower E b of the treated composites was associated with its higher stiffness as well as dramatic increase in Young’s modulus due to the rigidity of the composites. The increase in stiffness upon addition of silane made the composites more brittle. 17
Tensile properties of the PP/NBRr/SCB composite treated with NaOH and silane had improvement because of good adhesion of PP/NBRr matrices and SCB filler. The result of the tensile strength and Young’s modulus for both treatment showed an improvement of 4–20% and 0.3–25% for NaOH and 16–30% and 1.3–32% for silane compared to untreated composites, respectively. However, E b for NaOH and silane composites decreases by 0.2–10% and 12–31%, respectively. It is shown that the silane treatment results in better tensile properties than the NaOH treatment.
Morphological properties
Figures 11 to 13 indicate the scanning electron micrographs of the tensile fractured surfaces of PP/NBRr/SCB at 150× magnification for untreated composites, NaOH composites, and 3-APS silane-treated composites at the ratio of 70/30/5 and 70/30/30, respectively.

SEM micrographs of fractured surface of untreated PP/NBRr/SCB composites (×150) with SCB filler loading of (a) 5 and (b) 30 phr.

SEM micrographs of fractured surface of NaOH treated PP/NBRr/SCB composites (×150) with SCB filler loading of (a) 5 and (b) 30 phr.

SEM micrographs of fractured surface of 3-APS silane treated PP/NBRr/SCB composites (×150) with SCB filler loading of (a) 5 and (b) 30 phr.
Figure 11 shows SEM micrographs of fractured surface of untreated PP/NBRr/SCB composites (150×) with SCB filler loading of (a) 5 and (b) 30 phr. It clearly shows a gap between the NBRr and SCB filler with PP, which is the proof of poor adhesion between the filler and matrix. The SCB filler was noticeably pulled out from the matrix and exhibited more detachments of SCB filler and NBRr from the PP/NBRr matrices. This is because of the poor dispersion of the SCB filler by the PP/NBRr matrices. 38 Low adhesion between the phases gives rise to poor stress transfer across the interface. 39
Figure 12 shows SEM micrographs of fractured surface of NaOH-treated PP/NBRr/SCB composites (×150) with SCB filler loading of (a) 5 and (b) 30 phr. The surface roughness of lignocelluloses SCB before NaOH treatment was residual of lignin. Residual lignin was eliminated when NaOH treatment was applied. Removal of lignin will breakdown the fiber and lead to gaining of the effective surface area, and surface charge exposed further hydroxyl and carboxyl groups. 40 This is because alkaline will break the filler into finer filaments by reacting with binding materials of the fiber. Moreover, combination of sodium hydroxide with the composite will enhance the porosity and pore size on the fiber which will improve physical interlocking and causing better interfacial bonding between SCB filler and PP/NBRr matrices. In addition, the alkaline treatment can remove impurities from the surface of the filler and hence such roughening may produce an improvement in the wettability property with PP matrix. 8 Thus, the NaOH-treated composites had better adhesion than untreated composites. Hence, the results of tensile strength of NaOH-treated composites were found to be better than untreated composites and were supported by this testing.
Figure 13 shows SEM micrographs of fractured surface of 3-APS silane-treated PP/NBRr/SCB composites (150×) with SCB filler loading of (a) 5 and (b) 30 phr. It can be seen that treated PP/NBRr/BSP composites show better attachment of SCB filler and PP/NBRr matrices. As mentioned earlier, this is due to the better interaction between PP/NBRr/BSP with the addition of 3-APS silane than NaOH-treated composites and untreated composites. The addition of a suitable coupling agent improved interfacial adhesion between hydrophilic filler and hydrophobic matrices of the composite. 41 The adhesion between 3-APS-treated SCB filler and PP/NBRr matrices was powerful enough that the filler was rather broken under stress. Although there is SCB filler pull out, some of them are coated with the PP/NBRr matrices. Overall, this explained the effectiveness of improvement in tensile properties of the PP/NBRr/SCB composites with banana skin powder (BSP) filler treatment by 3-APS silane coupling agent.
Conclusion
As conclusion, the treated SCB showed improvement in tensile and thermal properties on PP/NBRr/SCB composites. The silane-treated composites have higher thermal stability compared to those treated with NaOH. The T 70% of NaOH and silane composites increases by 3–6% and 6–15%, respectively. Moreover, the T max of composite also increases using treated SCB in PP/NBRr composite by 3% for NaOH and 11% for silane composites. Meanwhile, the char residual at 600°C increases with increasing the SCB loading in the composites. Furthermore, tensile properties of the PP/NBRr/SCB composite treated with NaOH and silane had improvement because of good adhesion of PP/NBRr matrices and SCB filler. The result of tensile strength and Young’s modulus for both treatments showed an improvement of 4–20% and 0.3–25% for NaOH and 16–30% and 1.3–32% for silane compared to untreated composites, respectively. However, E b for NaOH and silane composites decreases by 0.2–10% and 12–31%, respectively. It is clear that the silane treatment shows better tensile properties than NaOH treatment. The SEM morphology showed better interaction between PP/NBRr/BSP with the addition of 3-APS silane. These composites are suitable in the application of building materials such as partition board and electrical cable jacket/outer insulator.
Footnotes
Acknowledgement
The authors would like to thank School of Environmental and School of Material, Universiti Malaysia Perlis for supported equipment in this research.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.