Abstract
Bone substitutes are biomaterials applied in surgical therapy to repair many cases of a damaged bone. More than two million bone surgeries using biomaterials are realised each year worldwide. In this line of research, the researchers of this project study developed a bioresorbable synthetic bone substitute designated by (β-TCP/DCPD)-PHBV which is a mixture of the two materials: β-TCP/DCPD and PHBV in a 60/40 weight fraction. This research study attempts to categorize the (β-TCP/DCPD)-PHBV and evaluate its ability on triggering bone regeneration by making bone implants on 12 New Zealand white male rabbits. The RESEARCHERS performed multi-scale studies namely; a uniaxial compression test, an examination by scanning electron microscopy, and analysis with infrared spectroscopy in order to get the characteristics. The neoformed bone is examined by SEM coupled with energy dispersive spectroscopy (EDS). In addition to the salient mechanical benefits; attenuation of the modulus of elasticity and limitation of brittleness, the biomaterial has also proven to be effective in giving rise to new bone tissues having a suitable mineral composition.
Keywords
Introduction
It is generally recognized that bio-orthopedic researchers, physicians, and biomaterial specialists are widening their scope of research studies to develop functional biomaterials for bone implants with advantageous biological and mechanical properties.
In fact, bioresorbable bone implants are the most preferable solutions to deal with many clinical complications. In particular, they are largely used in dental rehabilitation and bone reconstruction in cases of missing or partially damaged skeletal tissues.1,2
In order to fulfill this mission, scaffolds for bone regeneration must undoubtedly meet specific criteria, namely biocompatibility and bioresorbability in the medium term at a rate compatible with remodeling processes as well as without disturbing body tissues and organs. 3
It is subtle that these biomaterials are classified into organic or inorganic materials, where calcium phosphate (CaP) bioceramics stand for the majority of inorganic scaffolds while natural or synthetic biopolymers form the majority of organic scaffolds. 4 Recent research studies have shown that therapeutic composites, such as phosphocalcic ceramic materials embedded in biodegradable biopolymers, have promised orthopedic uses in bone tissue regeneration.5–8 The main advantages of these materials lie in the combination of the rheological and mechanical qualities of the polymer with the biocompatibility, bioactivity, and osteoconductivity of the bioceramic, which enhances bone regrowth.9,10 In this research study, researchers developed and analysed a biomaterial that would support a bone regeneration mechanism. To do so, they chose to combine two materials with distinct properties. The first selected constituent is Poly (hydroxybutyrate-co-hydroxyvalerate) (PHBV) which is well recognized as a biological product and is categorized as linear aliphatic polyester. 11 Over the past few decades, this material has been highly regarded by bone regeneration clinicians thanks to its biocompatibility, as well as its rheological and ecological properties. 12 As this material can be produced from renewable resources, it is also highly promoted by the biomedical industry given its potential applications in tissue engineering and in controlled release devices.
Likewise, PHBV is a radiolucent material which gives Poly (hydroxybutyrate-co-hydroxyvalerate) (PHBV) the particularity of simulated medical monitoring. However, this material does not have the required bioactivity.11,13 In order to overcome this deficiency, researchers preferred its alloy with a bioactive phosphocalcic ceramic. Indeed, this choice is based on and explained by their specific properties namely biocompatibility; the ability to promote an intermediate layer between the repaired bone and its surface, as well as its more or less rapid bioresorbability depending on its granulometry and porosity. Not only do these properties valid the choice but also the chemical side does. Their compositions are widely similar to those of the mineral phase of the bone. 14 Regarding its fragility, this material must not be used in bones under high loads. 15
Materials and procedures
Materials
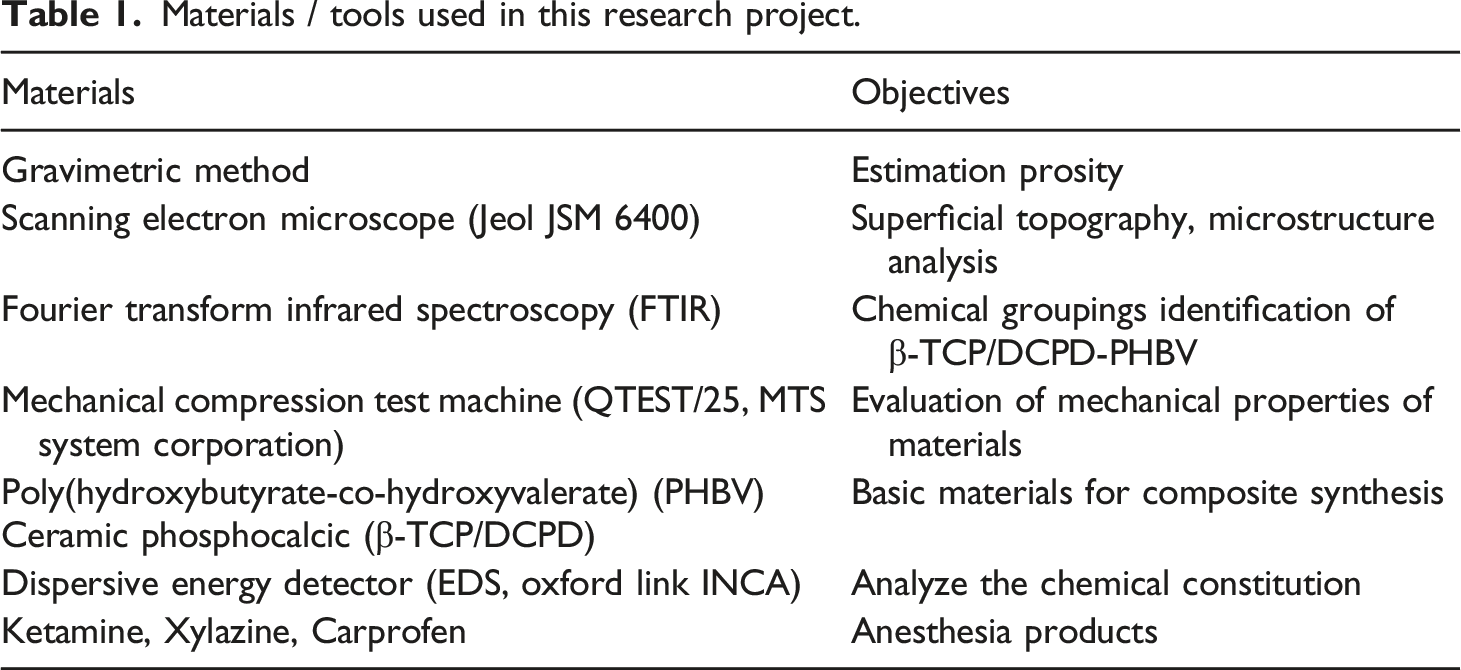
Materials / tools used in this research project.
Procedures
Biocomposite synthesis
Firstly, the polymer and the phospho-calcium ceramic were thoroughly proportioned (60:40), mixed in a flat bottomed boiling flask, and brought with the solvent chloroform to a temperature of 70°C under regular agitation. Once the polymer was melted, the chloroform was removed from the medium using a rotary evaporator. The recovered product was finally dried and molded into cylindrical pellets.
Porosity estimate
The porosity of the scaffold (β-TCP/DCPD)-PHBV with fraction (60:40) was estimated using the gravimetric method according to the following equation (1).9,16
The parameters P, DS, and DM represent the porosity, skeletal density and scaffold density, respectively. Thus, the skeletal density of the (β-TCP/DCPD)-PHBV composite is estimated from that of the PHBV polymer and the β-TCP/DCPD material, while DM is calculated from the volume and mass of the (β-TCP/DCPD)-PHBV according to the following equation.
The formula below was used to calculate the skeleton density (Ds) of the (β-TCP/DCPD)-PHBV scaffold, where DC and DP indicate the densities of β-TCP/DCPD and PHBV, respectively.
Superficial topography
The superficial topography of (β-TCP/DCPD)-PHBV scaffolds was examined using the scanning electron microscope (Jeol JSM 6400).
Chemical groupings identification of (β-TCP/DCPD)-PHBV (60:40)
The distinct functional chemical groups of (β-TCP/DCPD)-PHBV are identified by Fourier transform infrared spectroscopy. The traditional method for this analysis is to combine the biomaterial with KBr and compact it into cylindrical granules which are then exposed to FTIR. This analysis brings to the surfaces the transmission curves linked to chemical groups. 17
Compressive mechanical behavior
The mechanical properties of (β-TCP/DCPD)-PHBV are evaluated using a mechanical compression test machine (QTEST/25, MTS System Corporation). This is an uniaxial compression type test with a traverse speed of 0.2 mm/min. The specimens are cylindrical in shape, with dimensions of 13 mm in diameter and 5.1 mm in height, respectively. The primary characteristics derived from this mechanical examination are the Young’s modulus and the Mechanical strength.
Bone Implantation and neoformed bone examination
In order to estimate the biological effect of the biomaterial on bone tissue, 12 New Zealand white rabbits were involved. They are about 8 months old and weigh between 1.5 kg and 2 kg. They were obtained from the animal care unit and received anesthesia as a preparatory phase to surgery. It consists of injecting two different doses of products, the first being ketamine (keaminol, Intervet International GmbH) and the second is xylazine (Rompun, Bayer Healthcare, Puteaux, France). After 15 minutes, Carprofen (Rimadyl, Pfizer, Paris, France) was applied at an additional dose of (4 mg/kg) for local anesthesia.
First, a skin and subcutaneous incision is made on the medial part of the tibia. Next, a 1 cm longitudinal bone defect is established at the mid-diaphysis. The cavity created is occupied by the (β -TCP/DCPD)-PHBV pellet (Figure 1). The operated limb is immobilized with a stainless steel fixation system appropriate for this surgical application. Bone implanted with β-TCP/DCPD-PHBV and immobilized with a mini metalical fixator.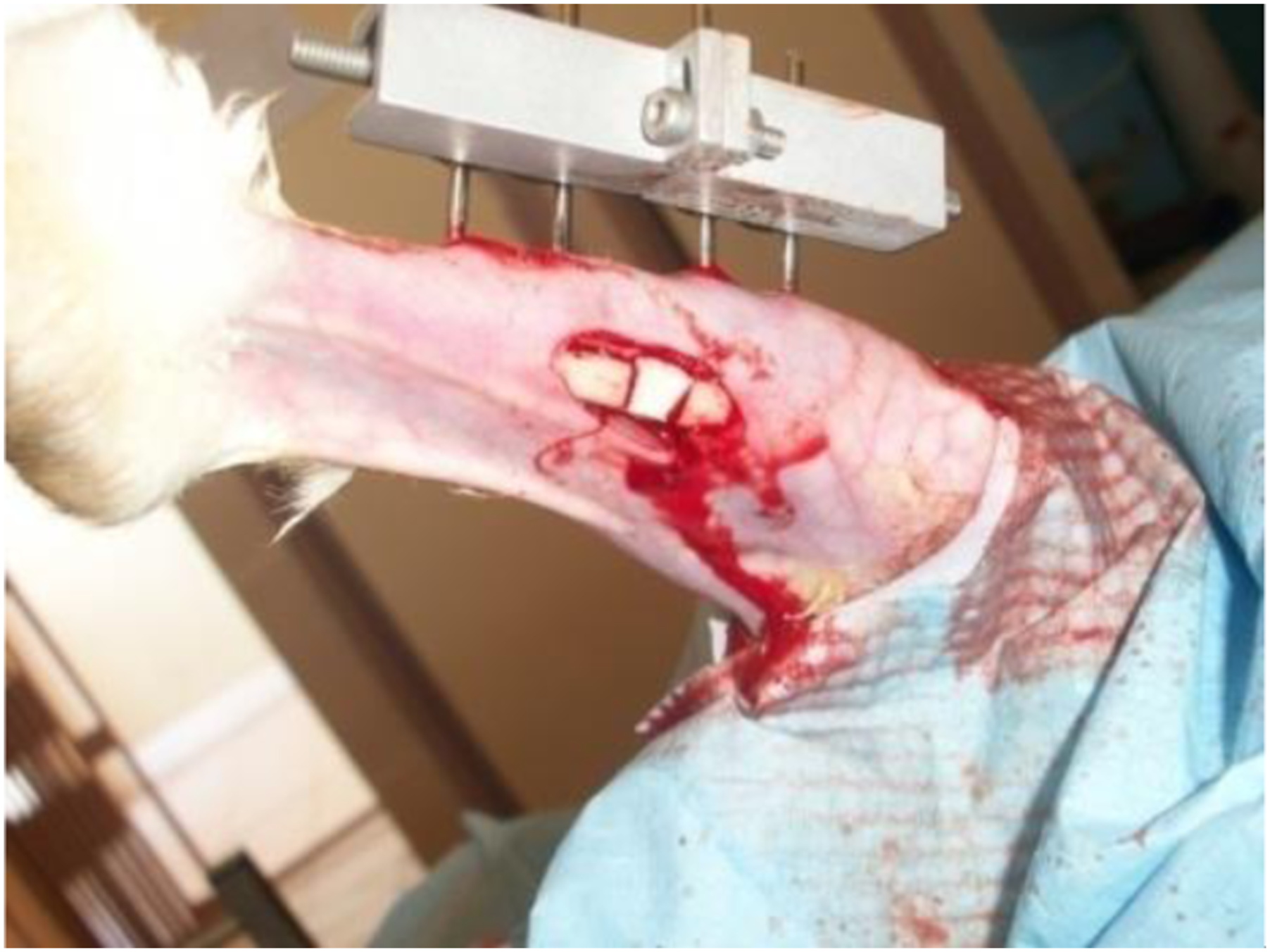
The recovered animals were examined in relation to the morphological makeup of the neoformed bone three months after the surgery. SEM (Jeol JSM 6400) was employed for this purpose with a Dispersive Energy Detector (EDS, Oxford Link INCA) operating at 10 Kv.The primary objective of the electronic system known as the EDS is to analyze the chemical constitution of the samples.
Results
Micrographic structure
The fine β-TCP/DCPD particles appear to be randomly dispersed and well-integrated within the PHBV matrix (Figure 2). The surface of the biomaterial appears to have a rough appearance (Figure 3). Aggregate of β-TCP/DCPD-PHBV. Superficial roughness of (β-TCP/DCPD)-PHBV biomaterial.

Porosity and physical parameters
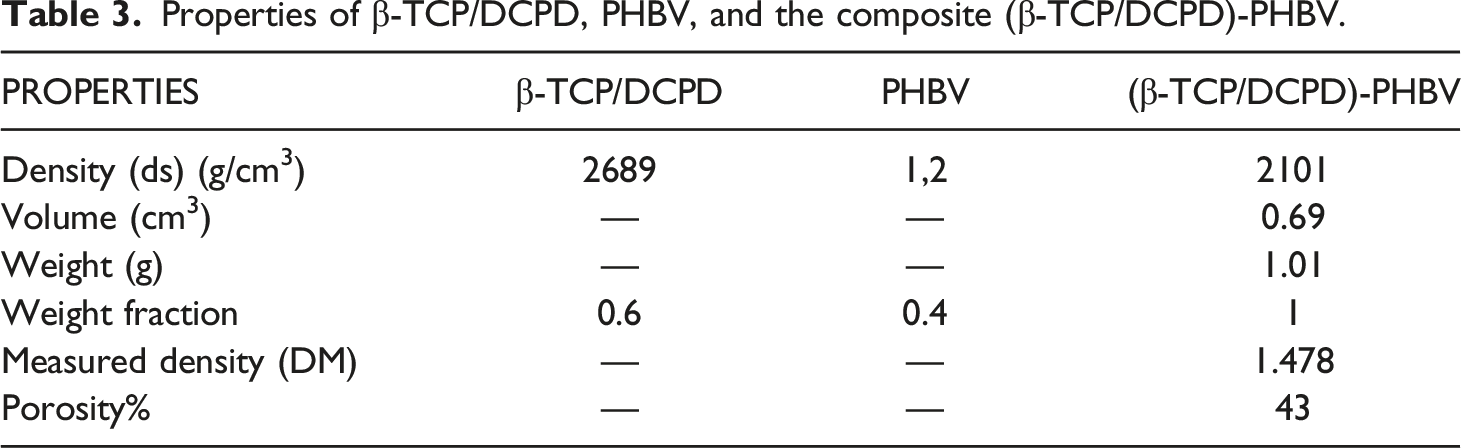
Properties of β-TCP/DCPD, PHBV, and the composite (β-TCP/DCPD)-PHBV.
IR specters of (β-TCP/DCPD)-PHBV (60:40)
The absorption bands deduced from the FTIR spectrum highlight the association of many functional chemical groups (Figure 4) including PO43− which absorbs at 458 Cm−1 as well as HPO42− located at 544 Cm−1. Similarly, the PHBV spectrum shows peaks at 1730 cm−1 and 3000 cm−1 which correspond to specific rotations around the carbon atoms. The first peak corresponds to the C-O stretching of the ester group presented in the highly ordered molecular chain and the adsorption band at 3000 cm−1 corresponds to the ester bond. IR spectrum of β-TCP/DCPD-PHBV (60/40).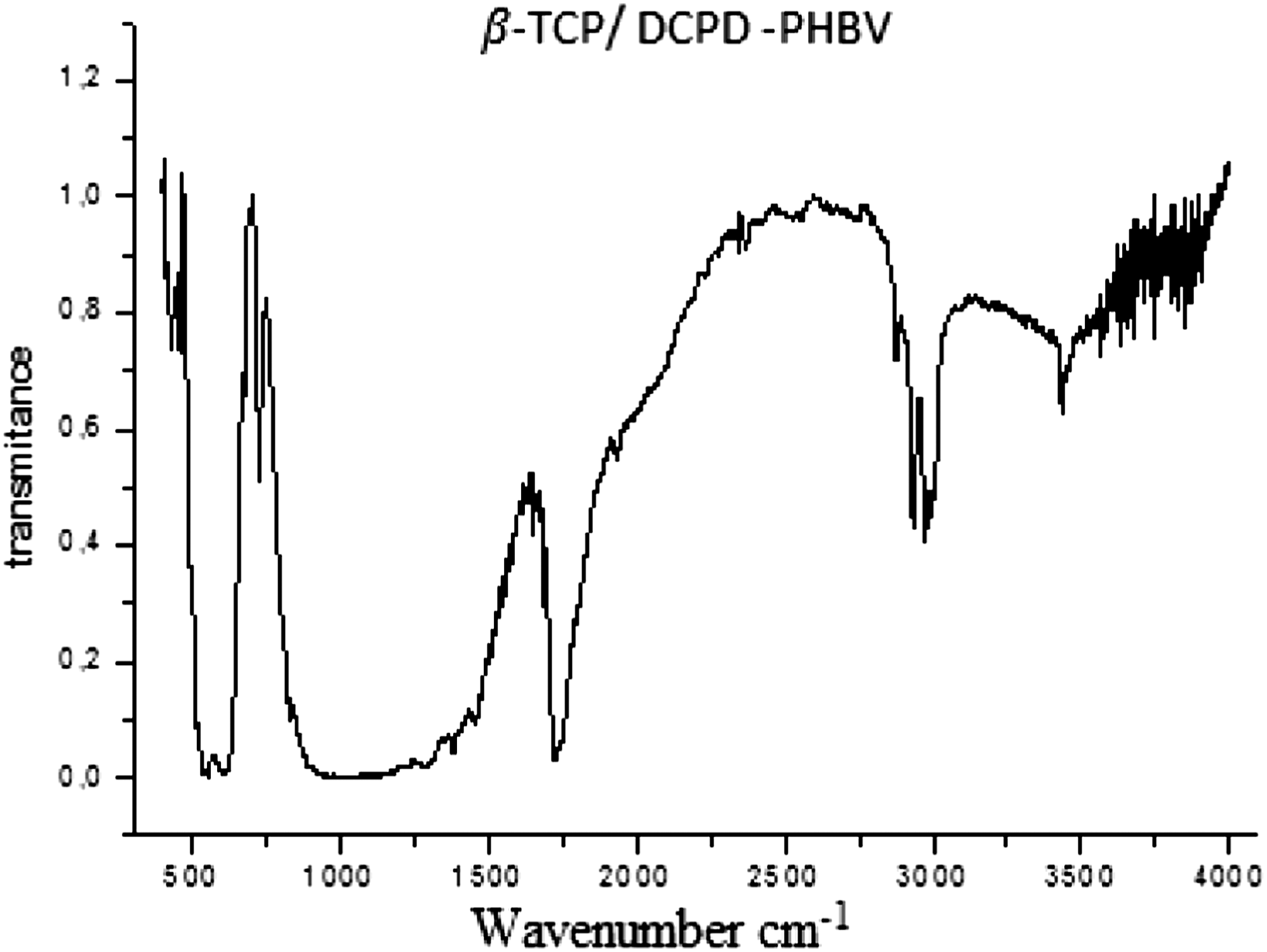
Mechanical property
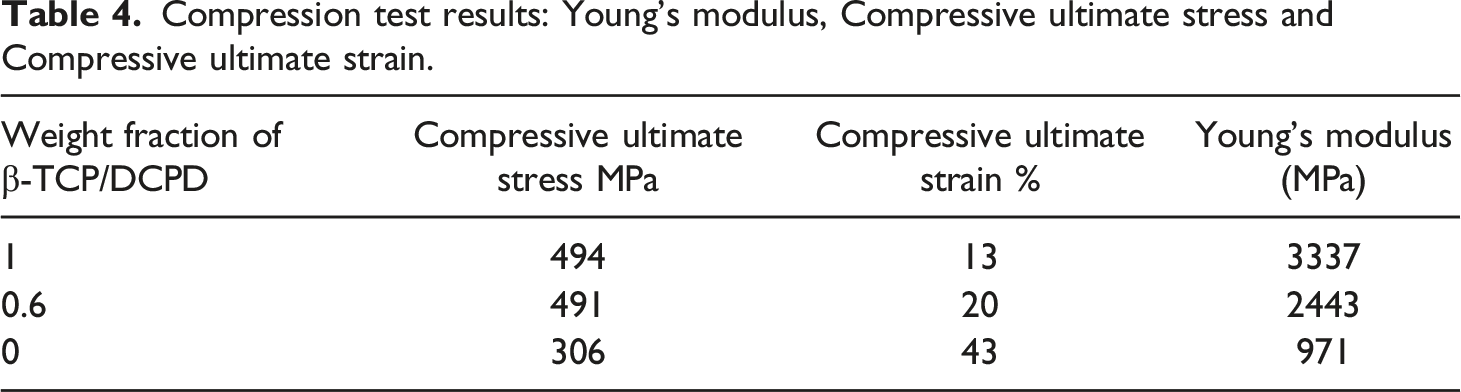
The stress-strain curve shown in Figure 5 reflects the mechanical behavior of the (β-TCP/DCPD)-PHBV biomaterial subjected to a compression test. Furthermore, Table 4 summarizes the most determinant mechanical properties including Young’s modulus, ultimate compressive stress, and strain at break. Strain-stress curve of (β-TCP/DCPD)-PHBV (60/40). Compression test results: Young’s modulus, Compressive ultimate stress and Compressive ultimate strain.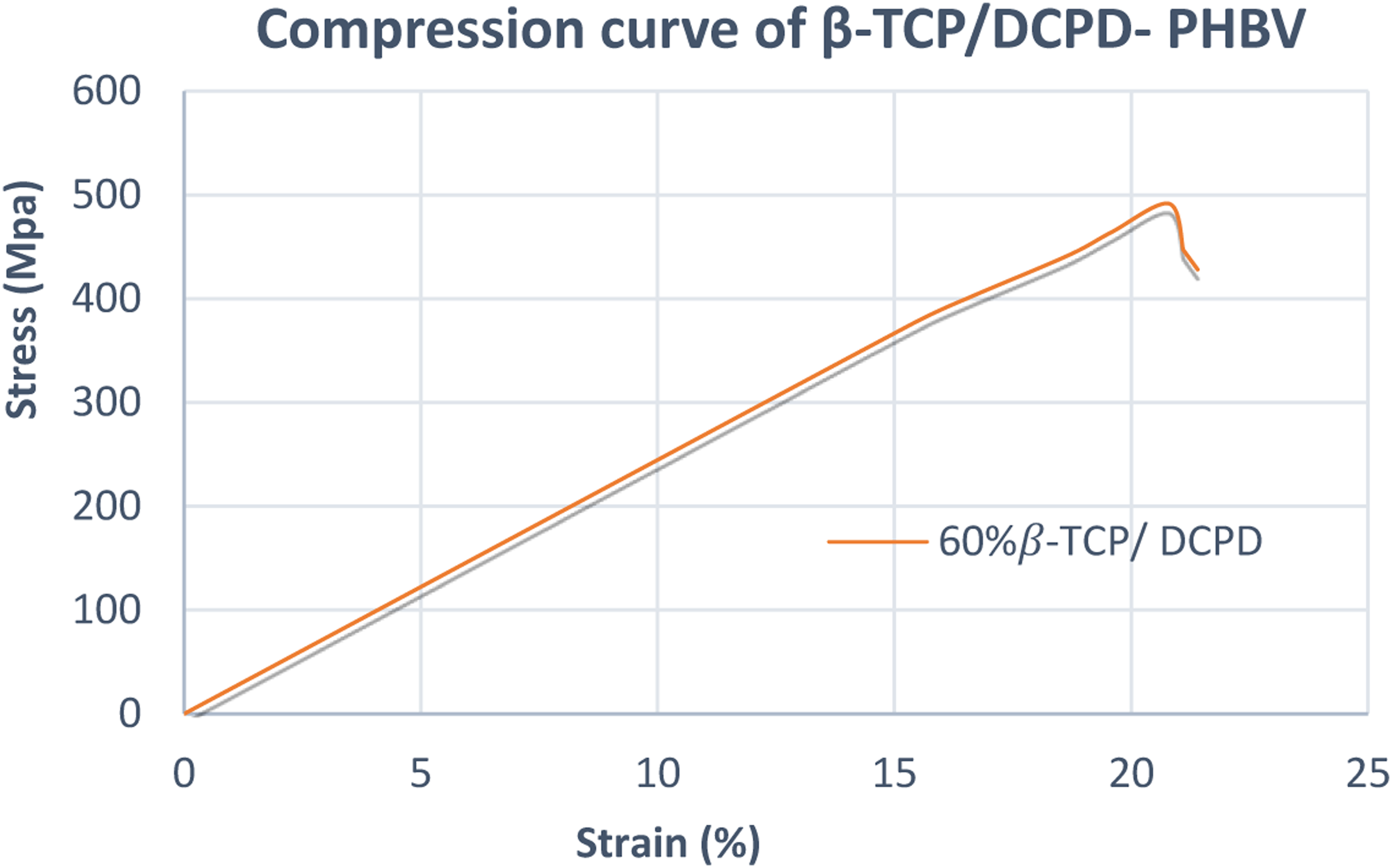
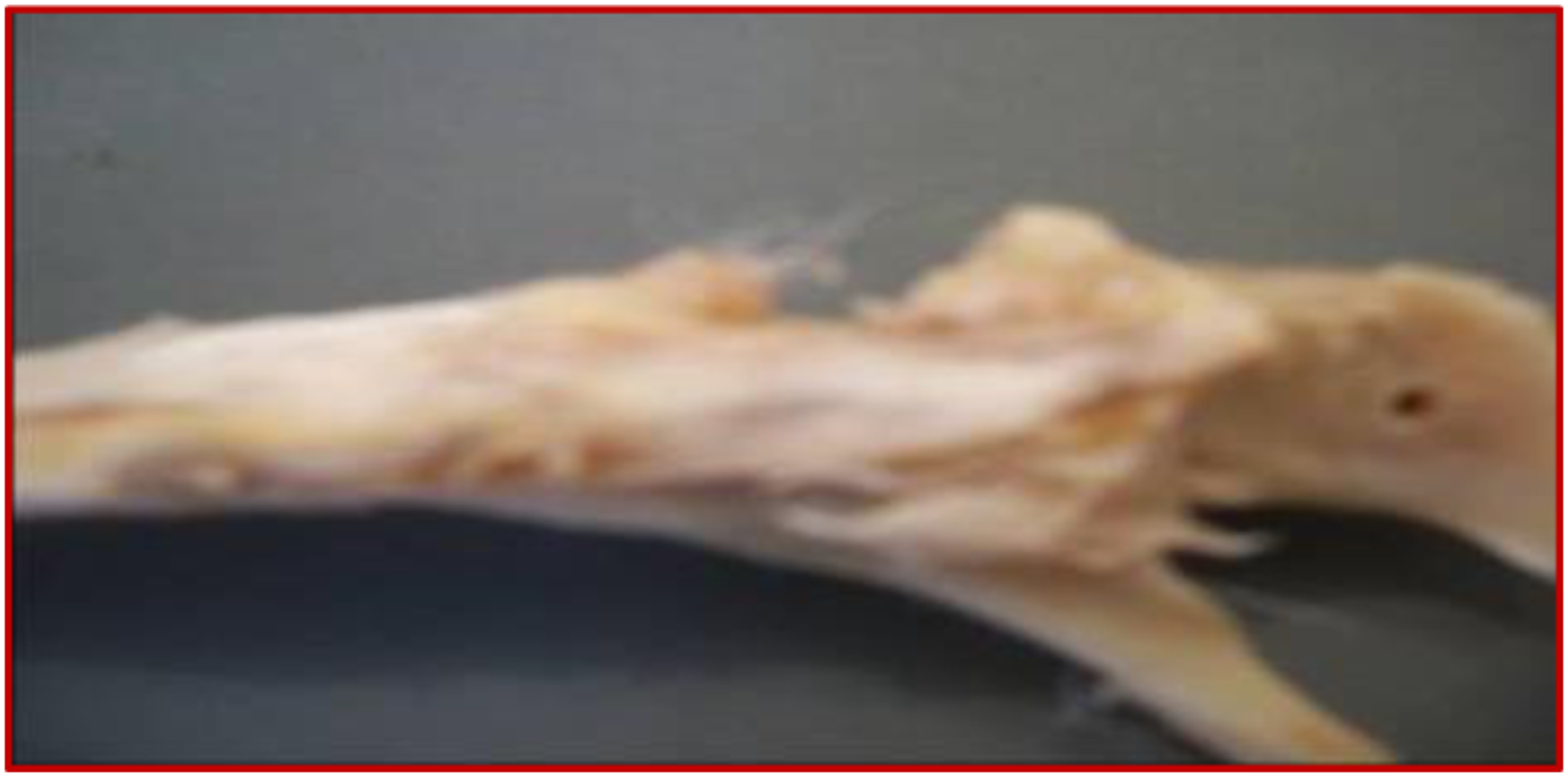
Remodeling of implanted bone
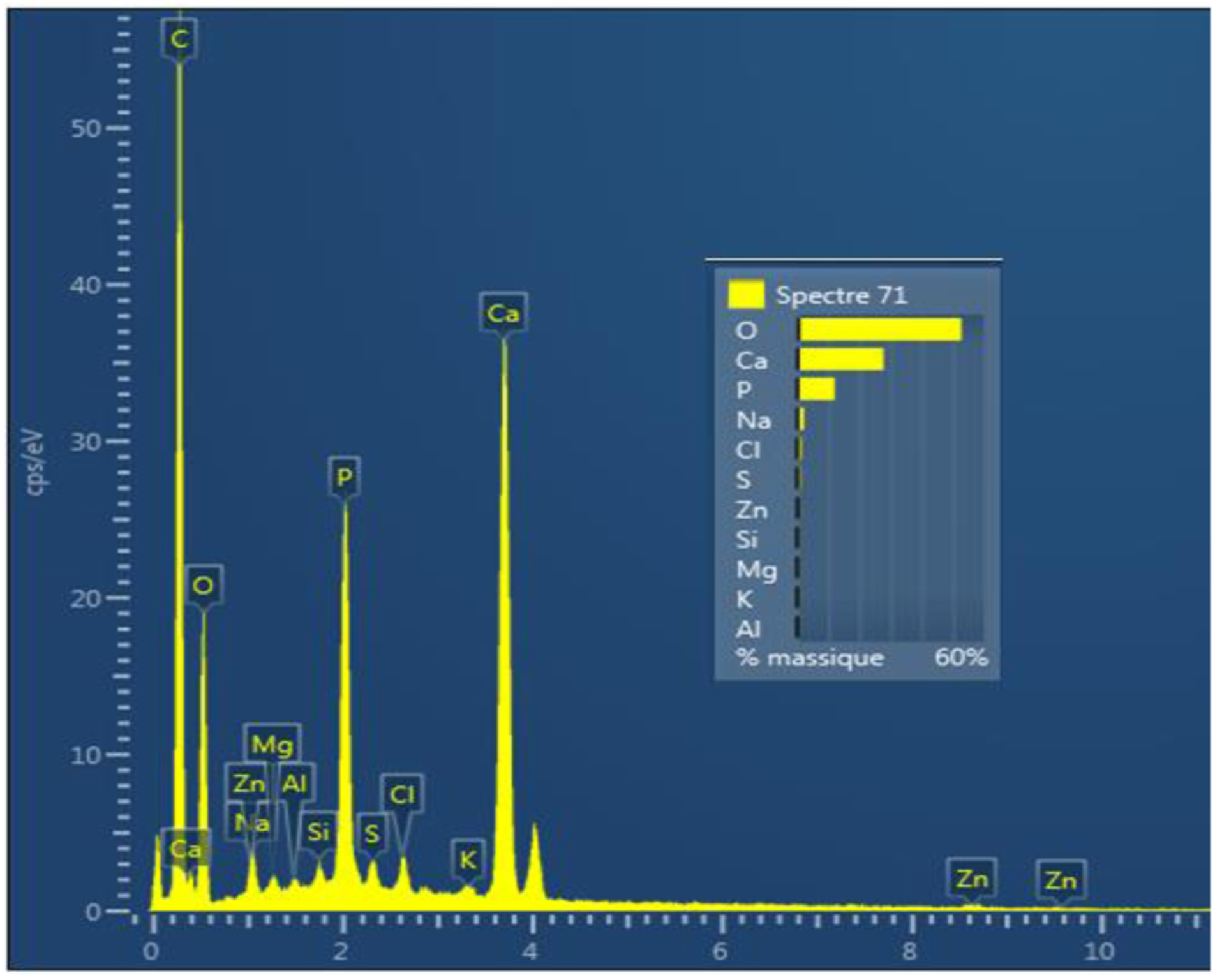
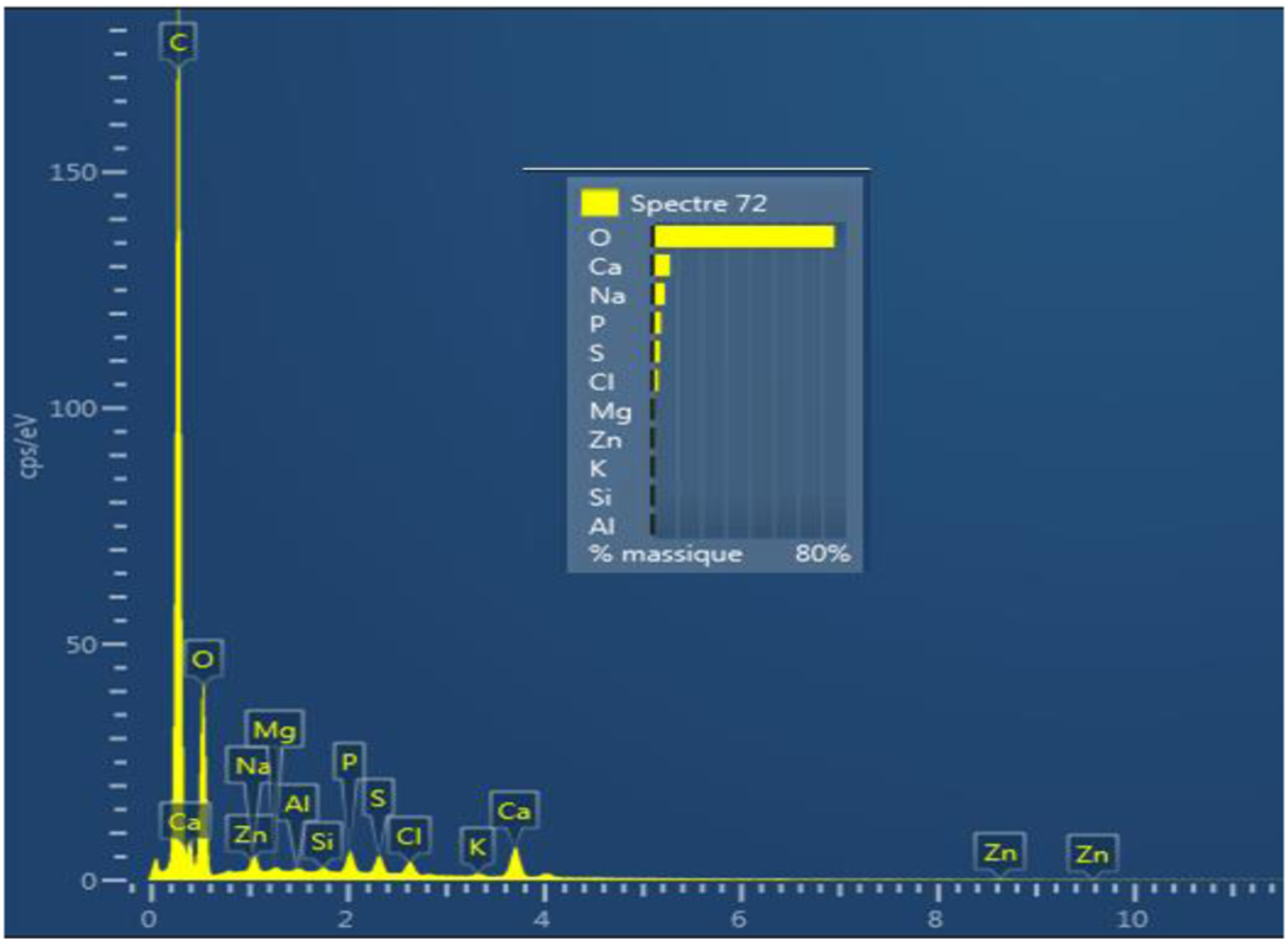
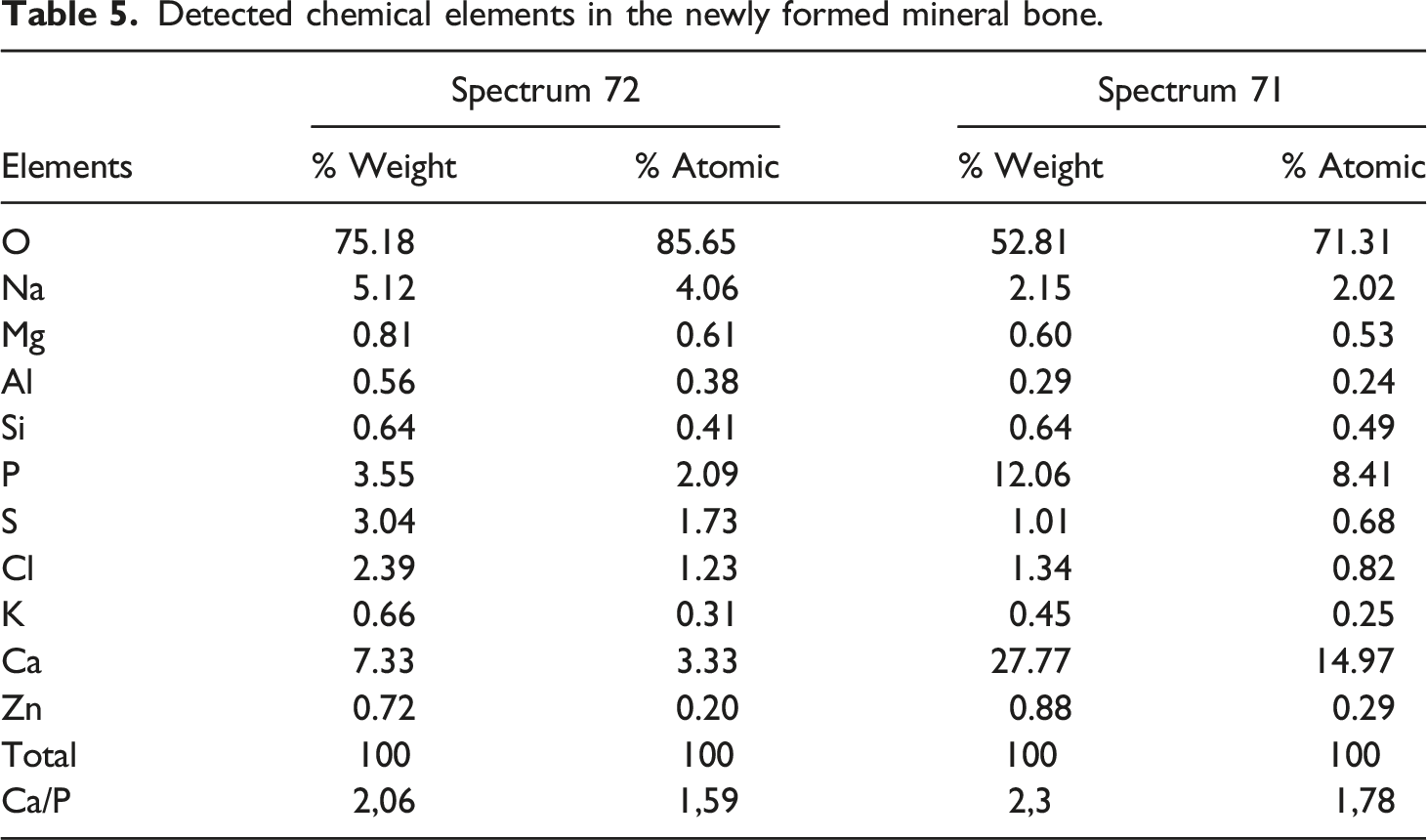
Figure 6 shows the almost complete disappearance of the biomaterial from the recovery process. Simultaneously, the cavity is filled with newly formed bone tissue with both a biological and a physiological processes that had taken 3 months to complete. The fifth table illustrates the mass and the atomic chemical ratio of the neoformed bone as established by energy dispersive X-ray spectroscopy (EDX) (Figures 7 and 8). The numerical photograph confirms the well-healed state of the operated diaphysis (Figure 9). The survival rate of the rabbits after the surgical manipulation is about 60%. Scanning electron microscopy observation of a newly formed bone (tibia, 12 weeks). EDX spectrum 72 of ground cortical bone (12 weeks) shows the corresponding characteristic peaks and chemical compositions of new bone growth. EDX spectrum 71 of ground cortical bone (12 weeks) revealed characteristic peaks and chemical compositions. Tibia bone diaphysis after remodeling (3 months).
Discussion
Over the past decades, tissue biotechnology has led to the development of new porous biomaterials for bone regeneration which were intended to temporarily support cell proliferation by degrading as they went along.
Today, the requirements in terms of reliability and quality have contributed to the continuous optimization of manufacturing technologies. This opens the prospect of realizing new bio-resorbable bone implants possessing specific intrinsic properties.19,20 In the present research study, the biomaterial (β-TCP/DCPD)-PHBV is properly developed according to the procedures described in the materials and methods section. In this project several evaluation techniques were exploited to highlight these biomaterial qualities and characteristics. The obtained results reveal interesting characteristics which give us more enthusiasm to progress in more advanced research approaches.
In fact, the micro-morphological examination of the biomaterial is a fundamental approach to the development of bone regeneration materials. This examination seeks to establish the topographical characteristics such as the surface roughness of the scaffold and its porosity which have a direct impact on cell adhesion, migration, and differentiation.21–23
It is agreed that a scaffolding material convenient for the effective function of bone regeneration is provided with a highly porous structure. In the current research study, the porosity of the (β-TCP/DCPD)-PHBV biomaterial is estimated to be 43% (Table 3). In comparison with other research studies, this threshold seems adequate for a bone substitute to provide bone defect filling. Indeed, in animal experiments on rabbits, Tarvainen T. affirmed that a glassy carbon material with a porosity of 40% stimulates bone neoformation in a hollow tibial defect. 24
However, El-Ghannam AR. revealed that a silica/calcium phosphate scaffold with different porosities (51%, 47% and 43%) contribute to bone regeneration in femoral lesions. 25 Similarly, other results revealed that a combination of ceramics (hydroxyapatite β-tricalcium phosphate) having 50% porosity was able to remedy femoral defects in dogs. 26
Another important morphological parameter for the initiation of the bone regeneration phenomena is the bone implant surface roughness. Numerous research studies have been conducted on the role of this parameter and how it can affect the process of osteogenesis. These studies identified the bone implant surface roughness as the second-most crucial topographic determinant regulating the biomaterial reliability. The surface roughness does, in fact, contribute to the fluidity of contact between the implant and the bone tissue, according to their findings. As a corollary, cellular activity is increased on the rough surface.27–30 The (β-TCP/DCPD)-PHBV biomaterial level of surface roughness is depicted in Figure 3. The biomaterial surface appears to have sufficient relief which can promote cell attachment to its contact.
Chemical analyses by Fourier transform infrared spectroscopy revealed that PO43−and HPO42− are the dominant chemical groups of (β-TCP/DCPD)-PHBV (Figure 4). Numerous studies have shown that these chemical components can create strong bonds with the surrounding tissues. 31 They stand as a natural bond between living and non-living materials. 32
The performance of an implant is also conditioned by its mechanical properties. Several researchers have focused their studies on the different mechanical properties of the elaborated biomaterial and have aimed to improve these properties. Thus, Ma et al. created a nano-hydroxyapatite (nHA)/polyether (PEEk) composite with a high modulus and stiffness. 31 As for Singh et al., they studied the tensile, compressive and morphological properties of 3D printed functional prototypes of PLA-PEKK-HAp-CS. 33 Further mechanical studies on FDP-based polylactic acid (PLA) structures with biologically treated natural fibrosis were performed by Mangat and Singh. 34 As common overall results, the mechanical tests revealed sustained stability of the scaffold, and the biomaterial stunted the growth of fibroblast cells which demonstrated cell proliferation and differentiation in the interconnected specimens.
For the achievement of cellular integration, the biomaterial must undoubtedly have mechanical properties that are extremely similar to those of bone. 35 Both Compressive strength and stiffness present the two primary mechanical properties considered to assess bone implants (Table 4). In particular, the stiffness is one of the most important mechanical features and is crucial to the kinetics of bone remodeling. In practice, a moderate Young’s modulus is highly recommended since using implants with an Elastic modulus higher than cortical bone may impede the osseointegration process. However, the application of bone implants having a Young’s modulus lower than or close to that of cortical bone may stimulate bone tissue and, therefore, promote the integration of bone tissue into the implant.36,37
In this study, the combination of PHBV and phosphocalcic ceramic mitigated the brittleness of the resulting biomaterial by improving its ductility and reducing its Young’s modulus from 3337 MPa to 2443 MPa. Another advantage consists of the density and the elastic of the modulus of (β-TCP/DCPD)-PHBV which are relatively close to bone. 38 The mechanical strength also increases by 33%. In fact, this property brought the enhancement of the structural cohesion of the biomaterial which enables the extracellular matrix cells enough time to multiply in the host site. The majority of the manipulated subjects, with this product, were fully remodeled presenting approximately 60%. Figure 9 shows an appropriate structure of the remodeled diaphyseal bone. In fact, these results of surgical implantation also reflect the favorable effects of these mechanical advantages on the microenvironment of bone cell proliferation in the material.
Detected chemical elements in the newly formed mineral bone.
With the exception of a few microcrystals, Figure 6 illustrates the almost complete disappearance of the biomaterial which degrades during the consolidation process. As this degradation proceeds, the cavity will be occupied by neoformed bone. According to the findings of the latter investigation, the scaffold degradation was sufficient to allow the regulated chemical release of bioactive compounds. Additionally, it provides enough time for the development of new tissue to really be matured.
Despite advances in biomaterial science, estimating a definitive or even approximate biodegradation time for a bone implant is a complex challenge. Since biodegradation mechanisms combine physicochemical degradation (chain splitting and dissolution in a moist environment), enzymatic activity, and cellular degradation (inflammation, response to a foreign body). Adding that this phenomenon is influenced by the chemical composition of the biomaterial in terms of crystallinity and porosity, along other parameters that affect the biodegradation process. 40
Conclusion
In all, these research results show that the incorporation of β-TCP/DCPD (60/%) into the PHBV matrix (40%) ends by an improvement of the most critical characteristics, such as brittleness limitation, through the reduction of Young’s modulus compared to β-TCP/DCPD. The provenance of bioactive chemical groups from β-TCP/DCPD improves the bone implant efficiency. Morphological properties such as porosity and surface roughness are also crucial properties to enhance the effectiveness of the biomaterial and make it a suitable synthetic bone substitute capable of restoring bone architecture with biological and mechanical characteristics similar to those of uninjured bone. It is also deduced that the ultimate compressive stress is increased by 33% and the ultimate compressive strain is reduced by 41%.
Additional research investigation are required to provide more information on the chemical, physical and biological circumstances that have a direct influence on the kinetics of the degradation processes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

