Abstract
The objective of this research is to examine the potential use of melamine derivatives namely melamine cyanurate (MC), melamine phosphate (MP), and melamine polyphosphate (MPP) as flame retardant additives in chicken feather (CF) containing thermoplastic polyurethane (TPU). The performances of the composites are evaluated using limiting oxygen index (LOI), vertical UL-94 (UL-94 V), and mass loss calorimeter (MLC) tests. The V0 rating is achieved with the addition of 10 wt% MC, 20 wt% MP, and MPP. The performances of the additives are ranked in the ascending order of MC, MP, and MPP in LOI test. MP and MPP show distinct better performance than MC in the condensed phase. However, the gas phase action of MC is much better. than MP, and MPP. The lowest pHRR (103 Kw/m2), THE (39 MJ/m2), and THE/TML ratio (1.4 MJ/m2g) are observed with the use of 20 wt% MPP, MP, and MC, respectively.
Introduction
Feather based fibers including chicken feather (CF) are used as biomass filler in polymers. CF containing composites have good thermal and sound deadening characteristics with light weight, and remarkable mechanical properties. With these outstanding characteristics, feather containing composites find applications especially in the automotive, and construction industries.1–4 The mechanical properties of the CF containing thermoplastic polyurethane (TPU) based composites were investigated in the literature. With the addition of CF, the improvements in elastic modulus and tensile strength were observed.5–7 Although the flame retardant performance of CF containing composites with different matrix materials are tried to increase,8–12 no effort is seen in the literature to improve that of TPU based composites. In the literature, various commercially available flame retardant additives including melamine derivatives were used to improve the flame retardant performance of neat and filled TPU based composites.13–15
Melamine derivatives are generally classified into nitrogen containing flame retardant additives. They are also classified into intumescent systems, phosphorus or boron containing flame retardant additives depending upon the parent acid type. Melamine derivatives are considered as environmentally friendly flame retardant additives owing to the reasonable non-toxic degradation products, and eligibility for recycling. 16 Accordingly, they can be good choice for producing environmentally friendly TPU based biocomposites. The promising flame retardant performances of melamine derivatives in neat TPU take into consideration as well.
In the literature, melamine derivatives are used either as main flame retardant additive or their synergistic effect in TPU.17–26 Liu et al. used 15 wt% melamine polyphosphate (MPP) in TPU. They found that limiting oxygen index (LOI) value increased from 20.8 to 26.1% and vertical UL-94 (UL-94 V) rating increased from burn to clamp (BC) to V2. 19 Yu et al. observed enhancement in LOI value with the use of 20 wt% melamine cyanurate (MC) and melamine pyrophosphate. 21 Tabuani et al. found that the addition of 23 wt% MC reduced the peak heat release rate (pHRR) at about 68%, whereas no improvement in LOI value was observed. 22 It is thought that commercially available melamine derivatives including MC, melamine phosphate (MP), and MPP can exert better flame retardant efficiency in the presence of char forming filler (CF) in TPU. In the current study, the effect of filler amount on the thermal and flammability characteristics of CF containing TPU composites is investigated. The properties of the composites are examined via thermogravimetric analysis (TGA), LOI, UL-94 V, and mass loss calorimeter (MLC) tests.
Experimental studies
Materials
TPU (PearlthaneTM Clear 15N85-CF) was bought from Brenntag (Istanbul, Turkey). It has hardness and density of 86 (Shore A) and 1.11 g/cm3, respectively. CF was obtained from local sources located in Bursa, Turkey. Melamine derivatives with the commercial names of MelapurTM MC25 (density: 1.7 g/cm3), Melapur® MP (density: 1.74 g/cm3), and Melapur 200 (density: 1.85 g/cm3) were purchased from BASF AG (Germany).
Production of the composites
Prior to the grinding process, CF was washed with hot soapy water at 60 oC to take out the oil and other undesirable substances. CF was dried at 80oC for 16 h after the washing process. CF with quills was pulverized with blade grinding (Fritsch Pulverisette 19, Germany). Powder CF was used in the composite production. CF, and melamine derivatives were dried in a blast oven at 80°C for 24 h prior to the extrusion process. The compounding process was performed in twin screw extruder (Gülnar Makina, Istanbul, Türkiye) with the temperature profile of 50, 165, 170, 175, 170, 165°C from the hopper to die at 100 r/min. The samples for flammability test were shaped by injection-molding machine (DSM Xplore 12 mL Micro-Injection Molder, Netherlands) under the processing conditions of 190oC barrel temperature and 30oC mold temperature. MLC samples were manufactured with hydrolytic hot-press (GULNAR MAKINA, Istanbul, Turkey) at 160°C in 3 min. The flame retardant efficiencies of melamine derivatives were investigated in constant CF loading of 20 wt%. Melamine derivatives were used in three different concentrations of 5, 10 and 20 wt%. For sample coding, the abbreviations TPU, CF, MC, MP, and MPP were used for thermoplastic polyurethane, chicken feather, melamine cyanurate, melamine phosphate and melamine polyphosphate, respectively. The abbreviation, TPU/CF/10 MC, shows the sample containing 20 wt% CF and 10 wt% MC.
Characterization methods
TGA tests were performed for melamine derivatives, and the composites using Hitachi-High Tech STA-7300 instrument with a heating rate of 10°C/min from room temperature up to 800°C under the nitrogen atmosphere. LOI and UL 94 V tests were carried out according to the standards of ASTM D2863, and ASTM D3801, respectively. The dimensions of LOI and UL-94 V samples were carried out on the test bars with the dimensions of 130 × 13 × 3.2 mm3, and 130 × 6.5 × 3.2 mm3, respectively. MLC test was carried out on square specimens (100 × 100 × 3 mm3) using Mass Loss Cone with thermopile attachment (Fire testing Technology, U.K) under the heat flux of 35 kW/m2 according to ISO 13,927 standard. The microstructures of the residual chars remained after the mass loss calorimeter test were examined with SEM (FEI Quanta 400F). The sample surfaces were covered with gold with a sputter-coater to achieve the conductivity.
Results and discussions
Thermal decomposition of the additives
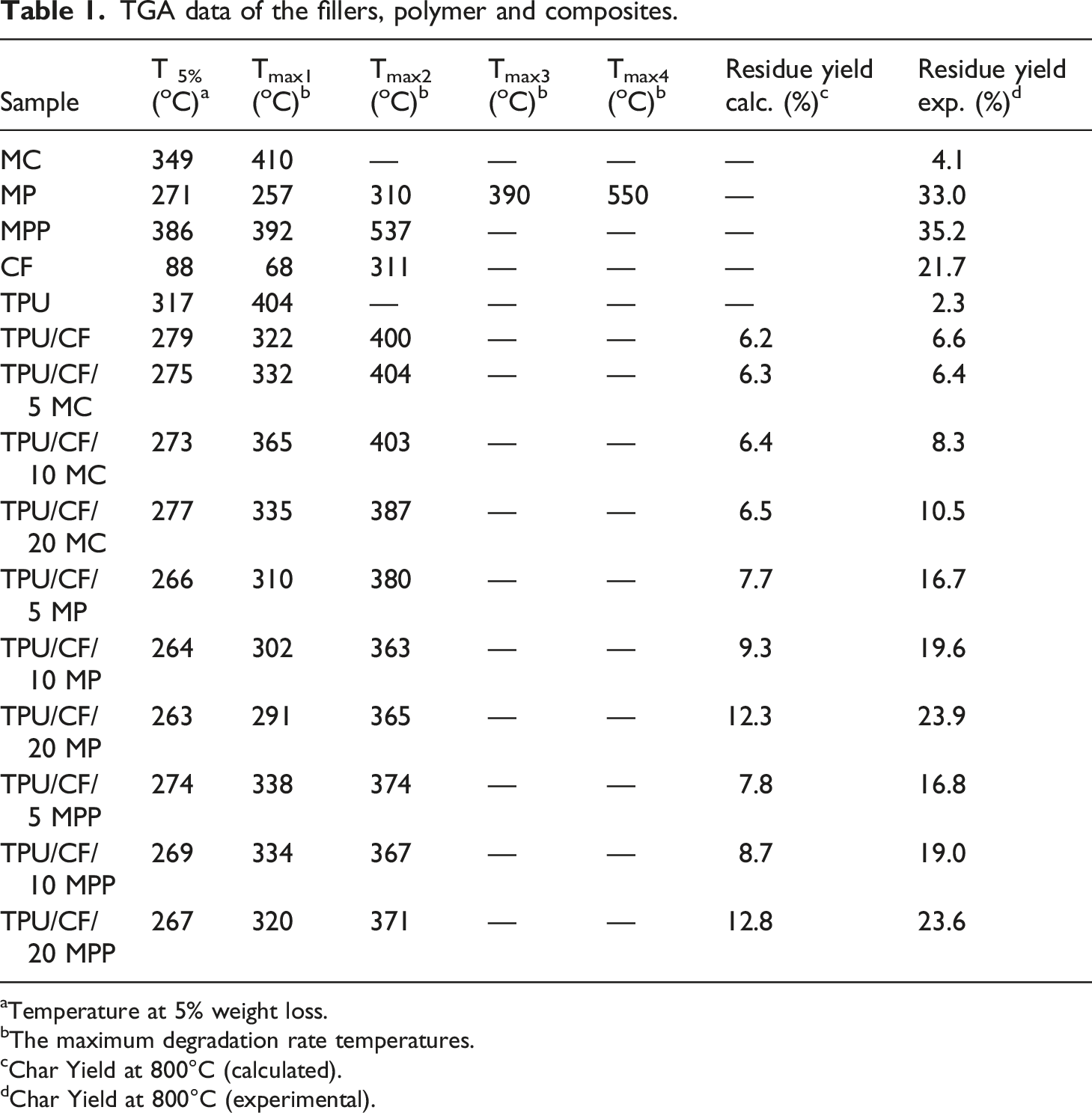
Thermal decomposition characteristics of the additives are analyzed by TGA under nitrogen atmosphere. TGA and DTGA graphs of the additives, and the related data are given in Figure 1 and Table 1, respectively. The decomposition of CF takes place in two steps at 68 and 311oC with leaving 21.7% residue. The first step is attributed to the loss of physically absorbed water. The second step is caused by the degradation of keratin with successive cleavage, crosslinking, and cyclization reactions.27,28 MC directly volatiles as an intact molecule and undergoes endothermic decomposition into melamine and cyanuric acid in a single step. The presence of cyanuric acid favors the condensation of melamine and gives 4.1% residue. During the decomposition, volatile water, ammonia, carbon dioxide, melamine and cyanic acid are mainly formed.16,29,30 The thermal decomposition of MP occurs in four steps with leaving 33% residue yield. In the first and second steps, MP turns into melamine pyrophosphate and MPP structures via dehydration reactions, respectively. In the third and fourth steps, the decomposition of MPP occurs via dehydration and deamination reactions simultaneously. Volatile products of water, ammonia and melamine formation takes place.29,31 MPP decomposes in two steps at 392 and 537oC and leaves 35.2% residue. Similar degradation characteristic of MPP is observed as in the case of MPP formed after the decomposition of MP. TGA/DTGA curves of the additives. TGA data of the fillers, polymer and composites. Temperature at 5% weight loss. The maximum degradation rate temperatures. Char Yield at 800°C (calculated). Char Yield at 800°C (experimental).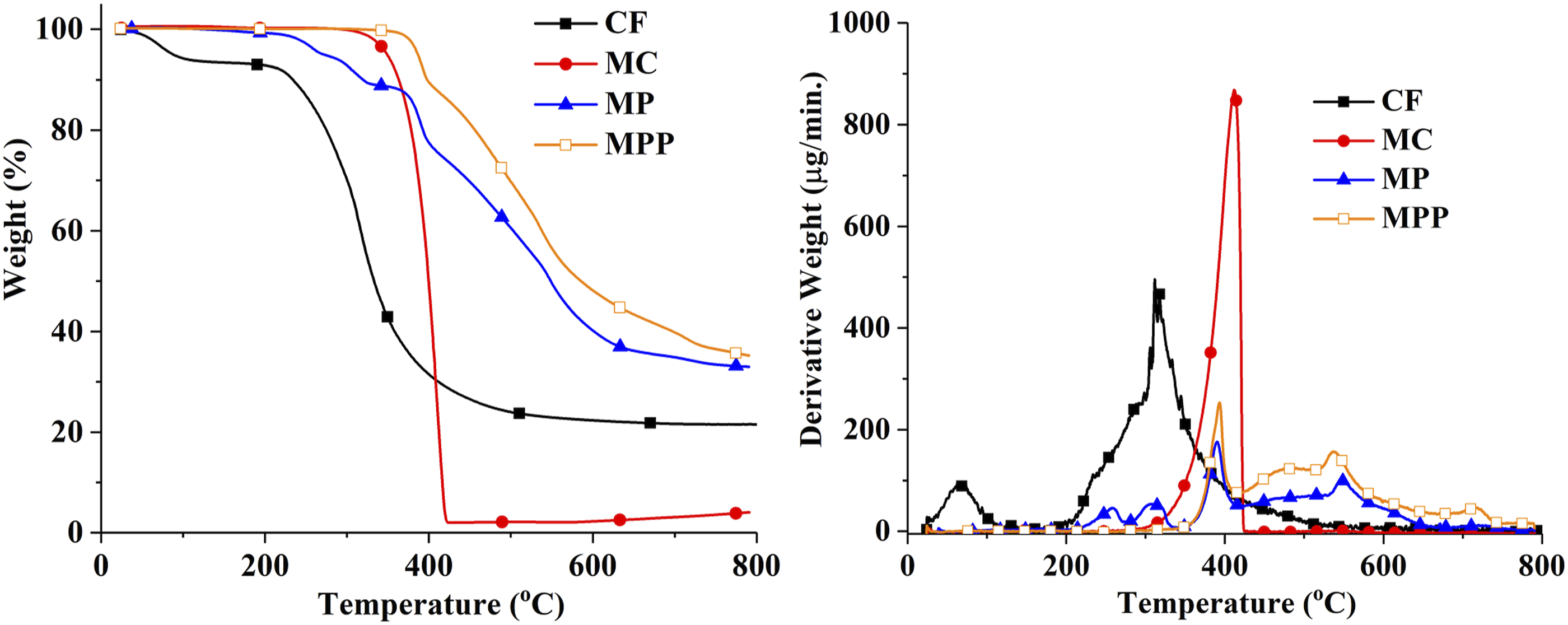
Thermal decomposition of the composites
TGA and DTGA curves of the composites and the relevant data are given in Figure 2 and Table 1, respectively. Pristine TPU decomposes in one step with leaving 2.3 wt% residue. CF containing TPU composite decomposes in two steps at 322oC (shown as shoulder) and 400oC. The first step is attributed to the decomposition of CF. The second step arises mainly from the decomposition of TPU. The initial thermal stability (T5%) of TPU reduces by approximately 40oC with the addition of 20 wt% CF owing to the low thermal stability of it. The residue yield increases to 6.6 wt% owing to the thermally stable decomposition products of CF. TGA and DTGA graphs of the composites.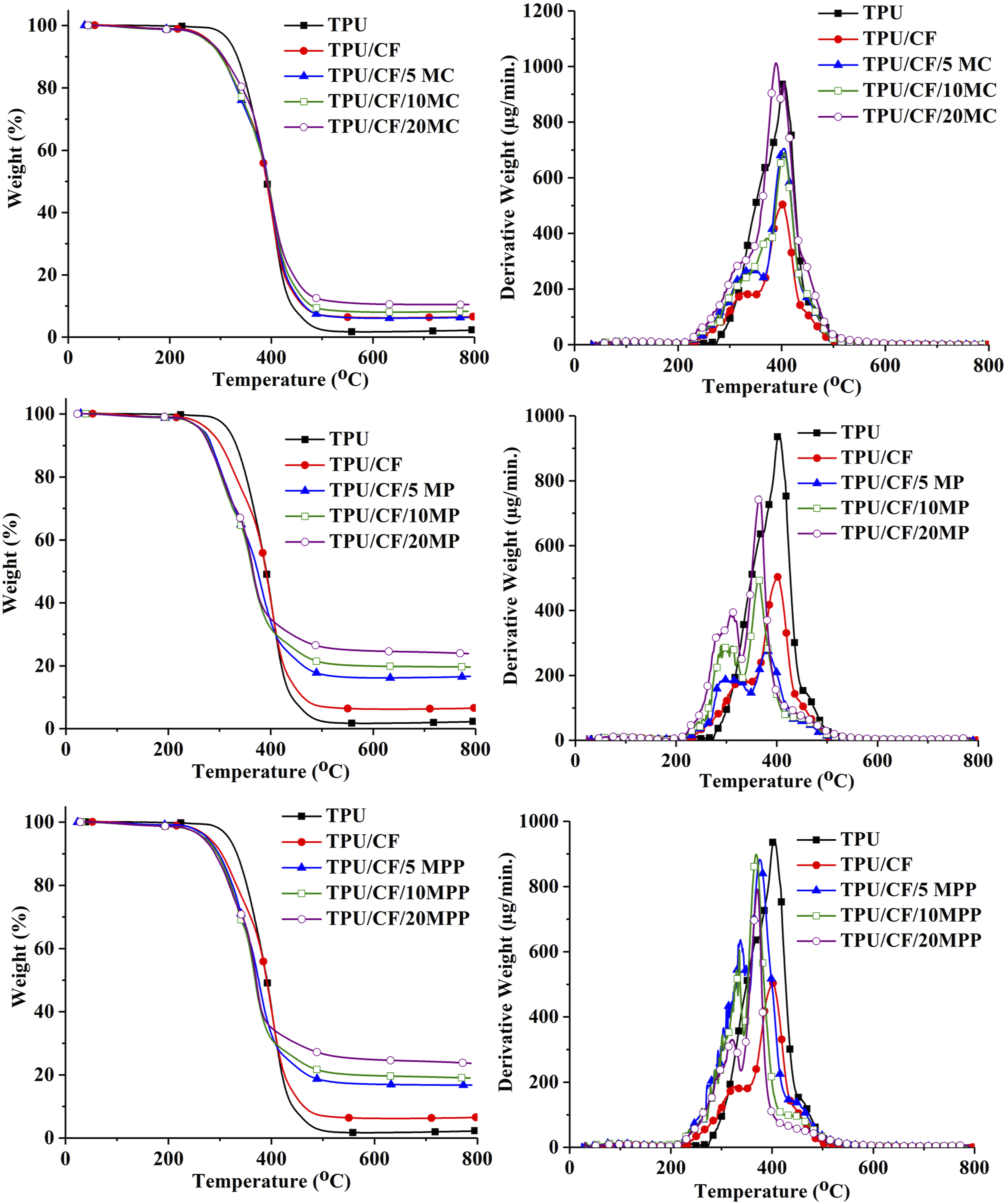
All melamine derivative containing composites decompose in two steps. In the case of MC and MPP, the degradation of CF takes place in the first step. In the second step, the decompositions of MC, MPP, and TPU occur simultaneously. In the case of MP, the decompositions of MP and CF take place simultaneously in the first step. In the second step, the decompositions of MPP, which is formed after the decomposition of MP, and TPU take place simultaneously. The addition of MC has negligible effect on T5% whereas the distinct reduction is observed in T5% value with the addition of MP, and MPP. A correlation is observed between the decomposition temperatures of melamine derivatives and the reduction in T5% value. The thermal stabilities of melamine compounds are in the descending order of MC, MPP, and MP where the lowest T5% value is observed. Although the addition of MC has negligible effect on the maximum degradation of TPU (Tmax2), the addition of MP and MPP reduces the thermal stability of TPU. All melamine derivative containing composites have higher experimental char yield than the calculated ones. It is suggested that cyanic acid does not as effective as in reducing the thermal stability of TPU and improving the char yield owing to its volatile characteristic. A similar trend is observed in the literature with the use of MC in unfilled TPU. 22 Phosphoric and polyphosphoric acid formed during the degradation of MP and MPP tend to stay in the condensed phase and interact with CF and TPU for increasing the char yield via favoring carbonization reactions and reducing the thermal stability of TPU via accelerating the hydrolysis of the TPU. Supportive findings are observed in the literature with the use of phosphoric acid with CF,8,12 MPP,19,21,25 and melamine pentaerythritol phosphate 17 in TPU.
Mass loss calorimeter studies
MLC data of the composites.
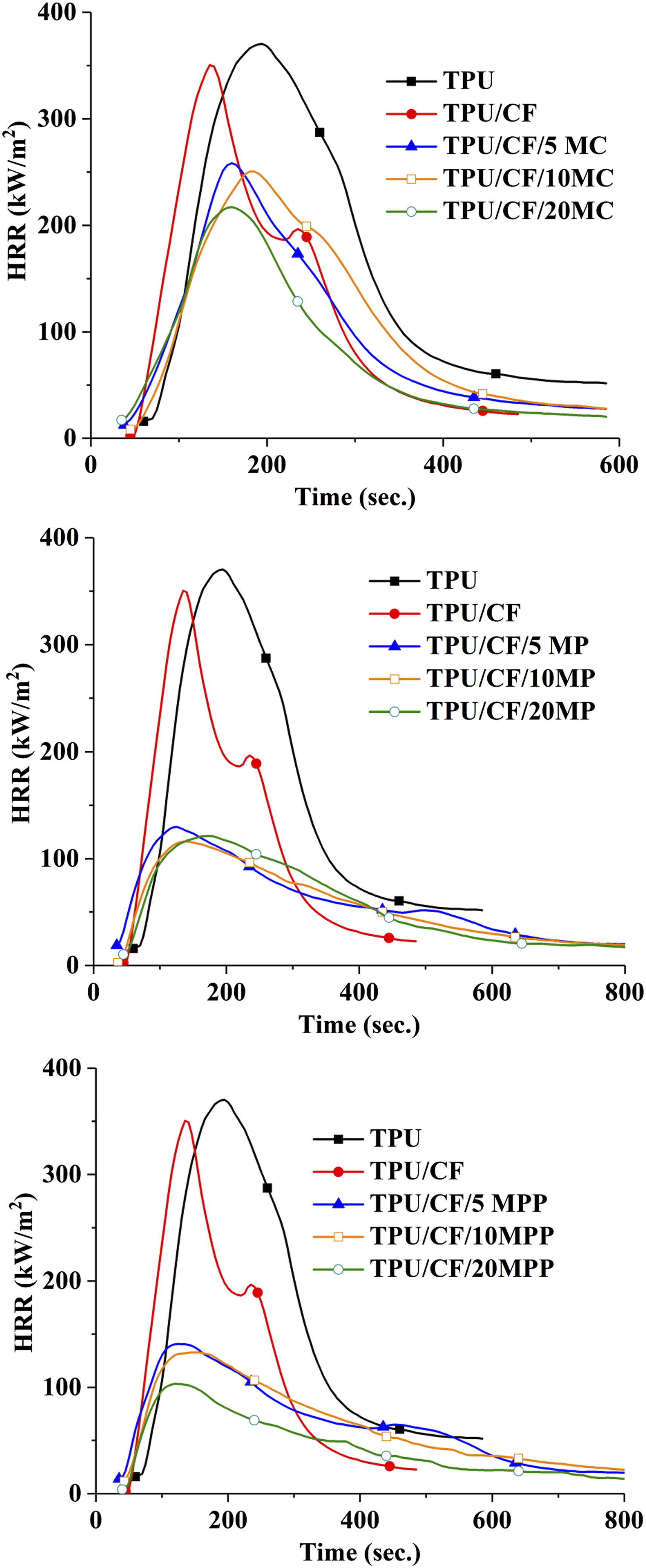
HRR curves of the composites.

Char photographs of MC containing samples remained after MLC test.

Char photographs of MP containing samples remained after MLC test.

Char photographs of MPP containing samples remained after MLC test.

SEM images of selected char residues.
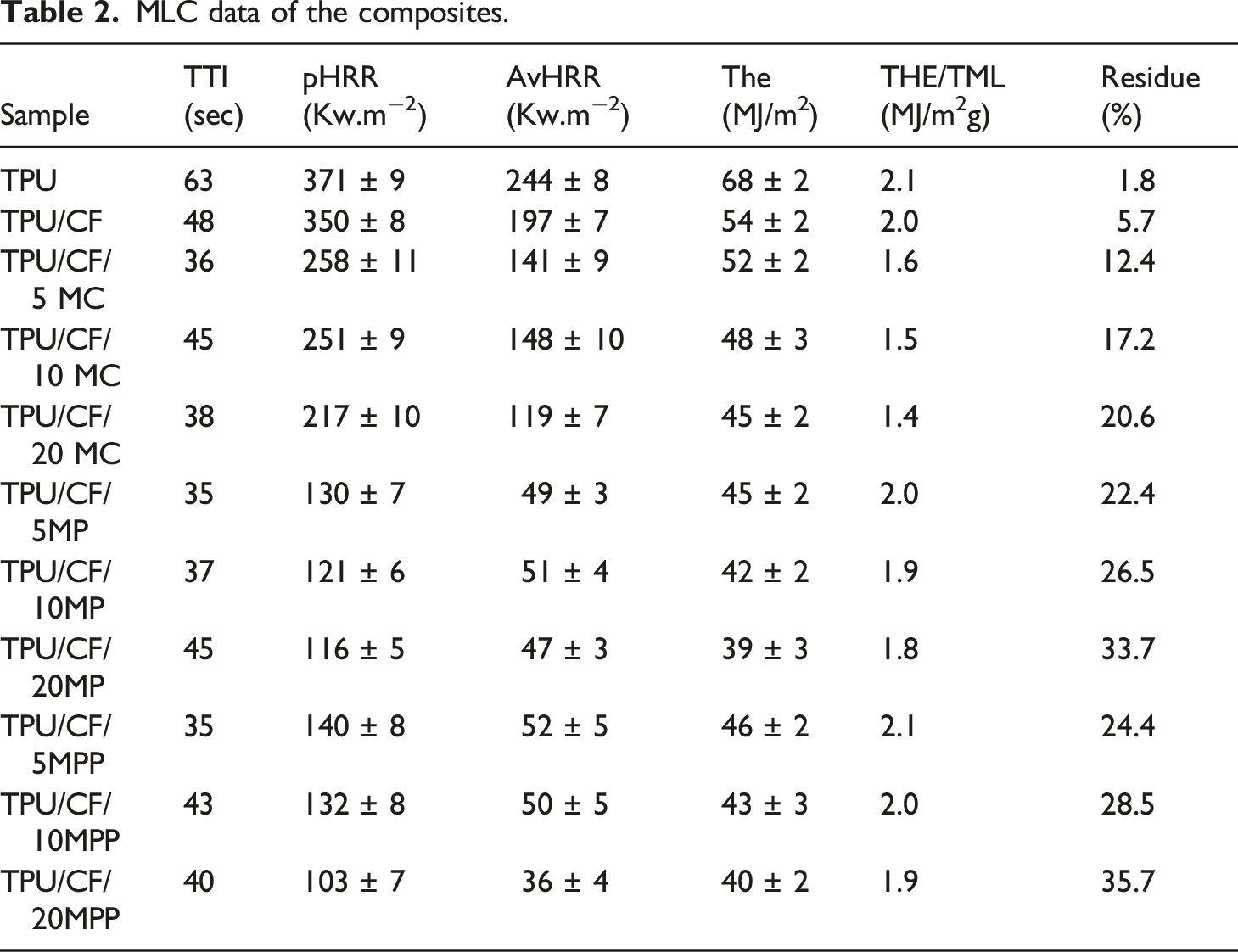
As given in Figure 3, TPU combusts readily giving one sharp HRR peak after ignition. It leaves 1.8% carbon-based residue after MLC test. TTI value is reduced with the incorporation of CF due to the lower thermal stability of CF compared to TPU. Thus, the required amount of combustible volatile compounds for ignition reaches in short time. With the addition of protein fibers, the decrease in TTI value is observed in epoxy resin, 9 and polypropylene. 12 With the addition of CF, little amount of char is formed and accumulated on the sides of aluminum foil as seen in Figure 4. Accordingly, the formed char structure does not exert any barrier effect and no significant change is observed in pHRR value. However, 14% reduction in THE value is observed owing to the enhanced char formation. No meaningful change is obtained in THE/TML value with the addition of CF. It is concluded that the inclusion of CF has no remarkable gas phase action.
With the addition of MC in all concentrations, the shape of HRR curve stays as non-char forming material despite of the reduction in pHRR. As seen in Figure 4 with yellow circles, the char structure does not completely cover the burning surface and accumulates the sides of aluminum foil with the 5 wt% MC addition. Although the residue yield increases remarkably with the 10 wt% MC addition, almost half of the burning surface is still uncovered with the char. When the concentration of MC reaches to 20 wt%, the char covers completely the burning surface. However, the large cracks and holes are observed especially in the center of the residue. Accordingly, the protective function of the residue is not good enough to achieve the shape HRR curve of typical insulating material. pHRR, av HRR, and THE values are steadily reduced as the added amount of MC increases. pHRR values reduce at about 26, 28, and 38% with the addition of 5, 10 and 20 wt% MC, respectively. THE value reduces at about 4, 11, and 17% with the inclusion of 5, 10 and 20 wt% MC, respectively. The reduction in fuel source with the increasing MC amount and the increase in char yield are the main reasons of low THE values. THE/TML value gives information related with the gas phase action of the flame retardants additives. The reduction in this value indicates the gas phase action of the additive via fuel dilution and flame inhibition. In the case of MC, the gas phase action of MC is attributed to the fuel dilution via the liberation of intact MC molecule and the noncombustible gases of water, ammonia, carbon dioxide, melamine and cyanic acid.16,29,30 The lowest THE/TML values are observed with the use of MC. As it is well known fact that phosphorus based parent acids in melamine derivatives favor the condensation and charring reactions. It is proposed that more noncombustible volatiles are formed with the use of MC rather than staying in the condensed phase via favoring charring reactions.
With the addition of MP and MPP in all concentrations, the shape of HRR curve represents the typical insulating material due to the formation of compact char structure with intumescent character (see Figure 5 and Figure 6). In both additives, the sharp reduction in pHRR value is observed in only 5 wt% addition. pHRR is reduced at about 63% and %60 with 5 wt% MP and MPP addition, respectively. As the concentration increases, a slight change in pHRR value is observed. THE values reduce steadily as the added amount of MP and MPP increases. Similar factors as stated for MC cause reduction in THE value. THE values of MP and MPP containing samples are much lower than those of MC filled samples. The higher residue yield and the higher protective function of the char, which favors incomplete pyrolysis, cause the higher reduction in THE value. Although MP and MPP have the ability to form noncombustible gases (water, ammonia, melamine), slight reduction in THE/TML value is observed when the concentration of MP and MPP reaches to 10 wt% and 20 wt%, respectively. It thought that most of the noncombustible gasses act as blowing agent and stay in the residue structure. Accordingly, slight reduction in THE/TML value is observed. In brief, the findings in the MLC test show that MC exerts predominantly its flame retardant effect in the gas phase via fuel dilution and MP and MPP show their predominant flame retardant effect in the condensed phase via the enhanced char formation with intumescent character.
Flammability properties
The flammability properties of the composites are characterized using UL-94 V, and LOI tests. The related results are given in Figure 8. The proposed flame retardant actions are summarized in Figure 9. TPU burns to clamp (BC) and gets 22.8% LOI value. UL-94 V rating remains BC and LOI value slightly reduces to 21.9% with the addition of CF. UL-94 V rating turns to V2 with the addition of 5 wt% MC, whereas no meaningful change is observed in the LOI value. 10 and 20 wt% MC containing samples get the highest UL-94 rating of V0. It is observed that the outstanding UL-94 V rating performance of MC stems from the runaway mechanism. As a visual observation, the presence of MC favors the dripping as small droplets. These small droplets extinguish before reaching the cotton. However, the distinct improvement in LOI value is observed when the concentration of MC reaches to 20 wt%. LOI increases from 21.9% to 27.2%. The improvement in flammability properties in the presence of MC arises from the various flame retardant effects including runaway, heat sink action during the decomposition of MC, and the sublimation and condensation melamine, enhanced char formation, and fuel dilution via the volatilization of intact MC molecule and the formation of H20, NH3, CO2, C6H6N6, and HNCO.16,29,30 Flammability properties of the composites. Proposed flame retardant actions of melamine derivatives.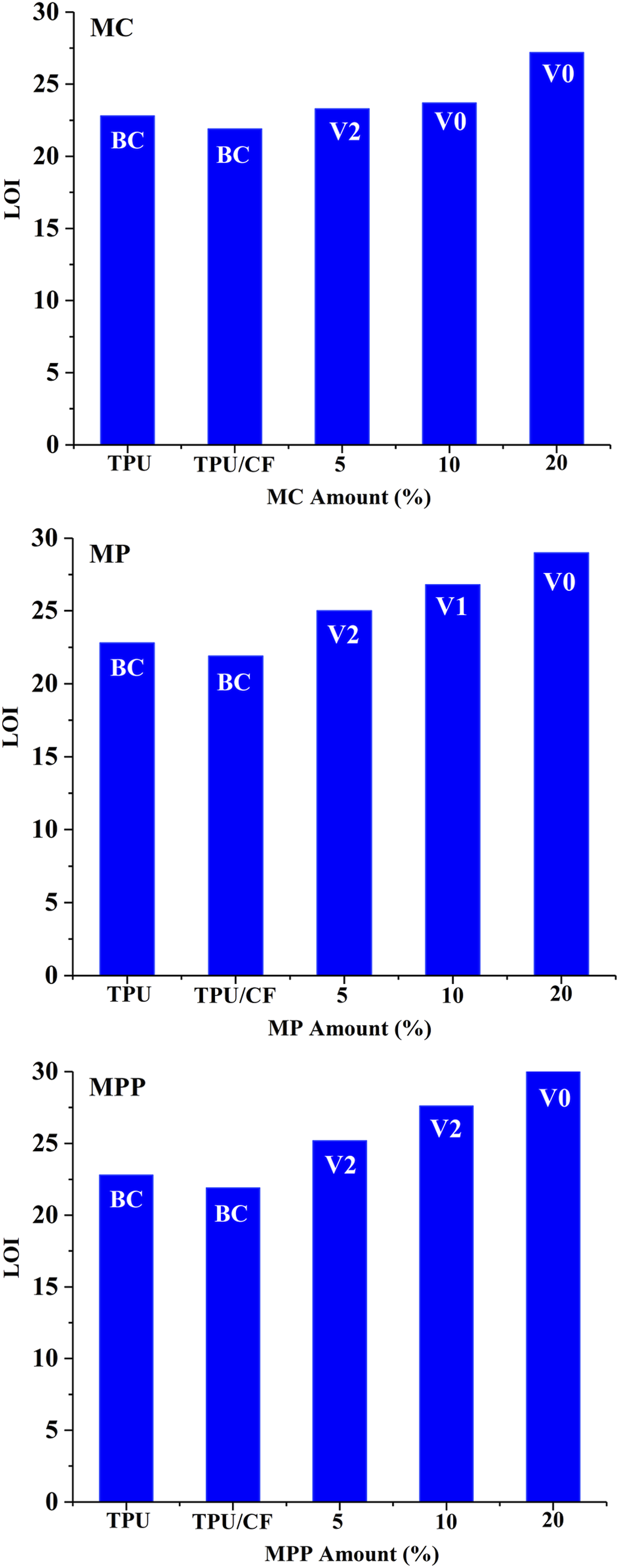

With the use of MP and MPP, 20 wt% additive is required to get the highest UL-94 V rating of V0. In both fillers, the steady increase in LOI value is observed as the added amount increases. The highest LOI values of 29% and 30.5% are achieved with the use of MP and MPP, respectively. Both fillers exert the same predominant flame retardant mechanisms of heat sink action, and enhanced intumescent char formation. In brief, melamine derivatives are ranked in ascending order of MPP, MP and MC according to the UL-94 V performance. According to the LOI performances, they are ranked in ascending order of MC, MP and MPP.
Conclusion
In the current study, the flame retardant performances of three melamine derivatives (MC, MP, MPP) in CF containing TPU biocomposites are evaluated. The effect of filler amount on thermal and fire retardant properties of the composites is investigated. According to the TGA results, all melamine derivative containing compounds reduce the initial thermal stability and favor char formation. According to the flammability test results, the highest performances in UL-94 V and LOI tests are observed with the use of MC and MPP, respectively. According to the MLC test results, MPP and MP containing samples exert lower pHRR and THE values than those of MC containing ones due to the formation of compact intumescent char structure. However, MC containing samples show better gas phase flame retardant action via fuel dilution with the formation of more noncombustible gases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Erciyes University Scientific Research Unit under grant no BAP- FDK- 2018-8383 and the program of TUBITAK 2211-C.
