Abstract
This work intends to develop an effective heavy metal-free modifier having properties comparable to traditional stabilizers and flame retardants, simultaneously being environmentally friendly and may be superior in many aspects. The important requirement focused on is: how to increase thermal stability and flame retardancy of flexible poly(vinyl chloride). Due to the typical materials now used with poly(vinyl chloride), which increases health and environmental concerns, utilizing a novel heavy metal-free additive will make poly(vinyl chloride) substantially safer. We have used an artificial silicate for this aim, which proved to be an efficient flame retardant and surprisingly showed excellent heat stabilizing effect. Thermal stability of flexible poly(vinyl chloride) was tested by both discoloration and hydrochloric-acid release methods. Fire properties were tested by limiting oxygen index (L.O.I) method. And penetration resistance of poly(vinyl chloride) has been tested by vicat test. The improvement rate in the thermal stability time during the dehydrochlorination test was 110.7% at 160°C and 90.1% at 180°C. Also, the improvement rate in the L.O.I test was 22.2%, which increased from 21.6% for the poly(vinyl chloride) to 26.4% when 5wt.% of artificial silicate was added. The artificial silicate also increases the thermal stability of the poly(vinyl chloride) and maintains its appearance of more saturated chromatic saturated (brighter). In addition, artificial silicate increases the resistance to hot penetration of poly(vinyl chloride). Where the resistance of poly(vinyl chloride) increased by 8.9% after adding 5wt.% of artificial silicate.
Introduction
Fire accidents account for a sizable fraction of the millions of annual automobile mishaps resulting in thousands of casualties around the world. Consider that in the United States alone, there were 1708 car fires in 2019, destroying 223,000 vehicles and deaths 4045 people. Vehicle fires accounted for 15.1% of all fires that occurred.1–4 There are many causes of car fires, and the most important ones are design flaws, poor maintenance, arson, hybrid vehicles, catalytic converters that overheat, overheated engines, spilled fluids, electrical system failures, and fuel system leaks. Car collision causes only a small percentage of fires, while design flaws or other components cause the most significant proportion of fires.5,6 Basically, plastic is one of the non-metallic materials used to construct automobile interiors, such as dashboards, seats, roofs, doors, fuel tanks, vehicle frames, control systems, insulated wires, and other components. In addition to its unique properties. Unfortunately, despite their unique properties, plastics are highly flammable.7–10 For instance, fuel that drips onto hot metal parts can assist generate sparks, which can subsequently cause ignition of plastic parts, leading to rapid flame spread from the engine compartment to the automobile cabin, which is totally constructed of plastic and rubber. In addition, the existence of scraping in the insulating cover of the wires that make up the electrical connections will aid in generating electric sparks, which, in turn, will cause the plastic parts to catch fire.11–13 Therefore, the safety standards in the automotive industry are not limited to the impact or corrosion resistance of bodywork, the quality of wheels, lights, electrical parts, etc., but also the resistance of non-metallic parts (plastics and elastomers) during emergencies, especially fire. Because of this, it is necessary to improve its thermal resistance at high temperatures and ensure that it has the best mechanical properties.14–17
The essential kinds of plastic and those that are used in automotive parts are as follows: (1) Polypropylene (PP), which can either be used on its own or blended with rubber to make thermoplastic elastomers. (2) Polystyrene (PS), also known as polystyrene. Polypropylene has strong thermal resistance, and when mixed with other substances, it will have low gloss and density in addition to high stiffness and toughness as well as good recyclability features. (2) Acrylonitrile butadiene styrene (ABS), the parts created from it are defined by their high impact strength, remarkable toughness, and strong resistance to cracking. These characteristics are in addition to its high resistance to breaking. (3) Polyurethanes are characterized by their exceptionally high impact resistance and stiffness. (4) Polycarbonate (PC), a material that is both highly resistant to impact and highly resistant to the effects of the weather. (5) Styrene maleic anhydride copolymer (SMA), which possesses a unique resilience to heat.18–20 (6) Poly(vinyl chloride), more commonly referred to as PVC, is a material utilized extensively in producing plastic components for automobiles. This is because PVC possesses many desirable qualities, including a high level of flame retardancy, in addition to an affordable price and excellent mechanical properties. However, poly(vinyl chloride) starts to release HCl from its molecular structure when the temperature rises, turning it into a dangerous substance. This is one of the reasons why it is classified as a hazardous material. In addition to its impacts on both human health and the environment, the release of HCl from the poly(vinyl chloride) structure will severely affect the entirety of this polymer’s functional capabilities.15,21,22
Now the biggest challenge in the industry is not to find an alternative for poly(vinyl chloride), but to improve the performance of this material, for example, making poly(vinyl chloride) significantly safer by using a unique heavy metal-free additive considered the best and fastest solution to the pollution and degradation problems, because the traditional chemicals now being utilized with poly(vinyl chloride) are raising the risks to both human health and the environment. Therefore, the best way to do that is by developing an efficient, environmentally friendly heavy metal free working as a flame retardant-stabilizer agent such as artificial silicate; with properties equivalent to traditional materials and could be better than it in many aspects in terms of engineering properties and not only at the environmental level. Artificial silicate (also called nanocement type A) is a special mineral admixture added to the concrete mix to make it more resistant to water leakage-, freezing-, heat-, and acid/alkali, among other things. Artificial silicate is composed of various nano oxides and nano carbides compounds, such as Aluminum oxide; Titanium dioxide; Iron (III) oxide; Calcium Carbonate, and nanotubes or fibers, as well as copolymers such as styrene-butadiene; vinyl acetate. The copolymer content is between 10% and 20%. These components vary according to the type of artificial silicate.15,23 The effectiveness of artificial silicate as a fire retardant and as a heat stabilizer has been demonstrated to be outstanding. Additionally, it has demonstrated substantial promise in the stabilization process of plasticized poly(vinyl chloride).
Experimental procedure
Materials
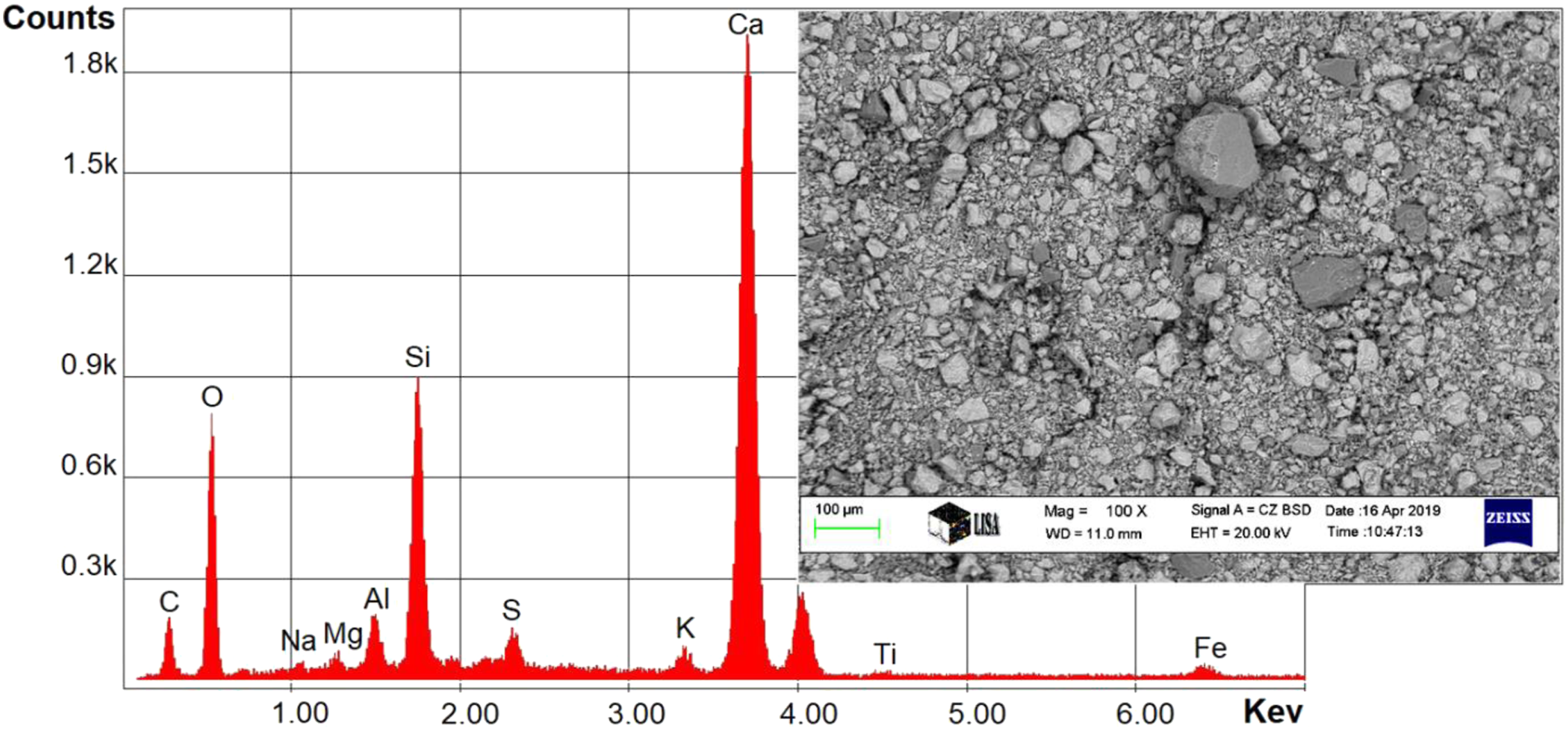
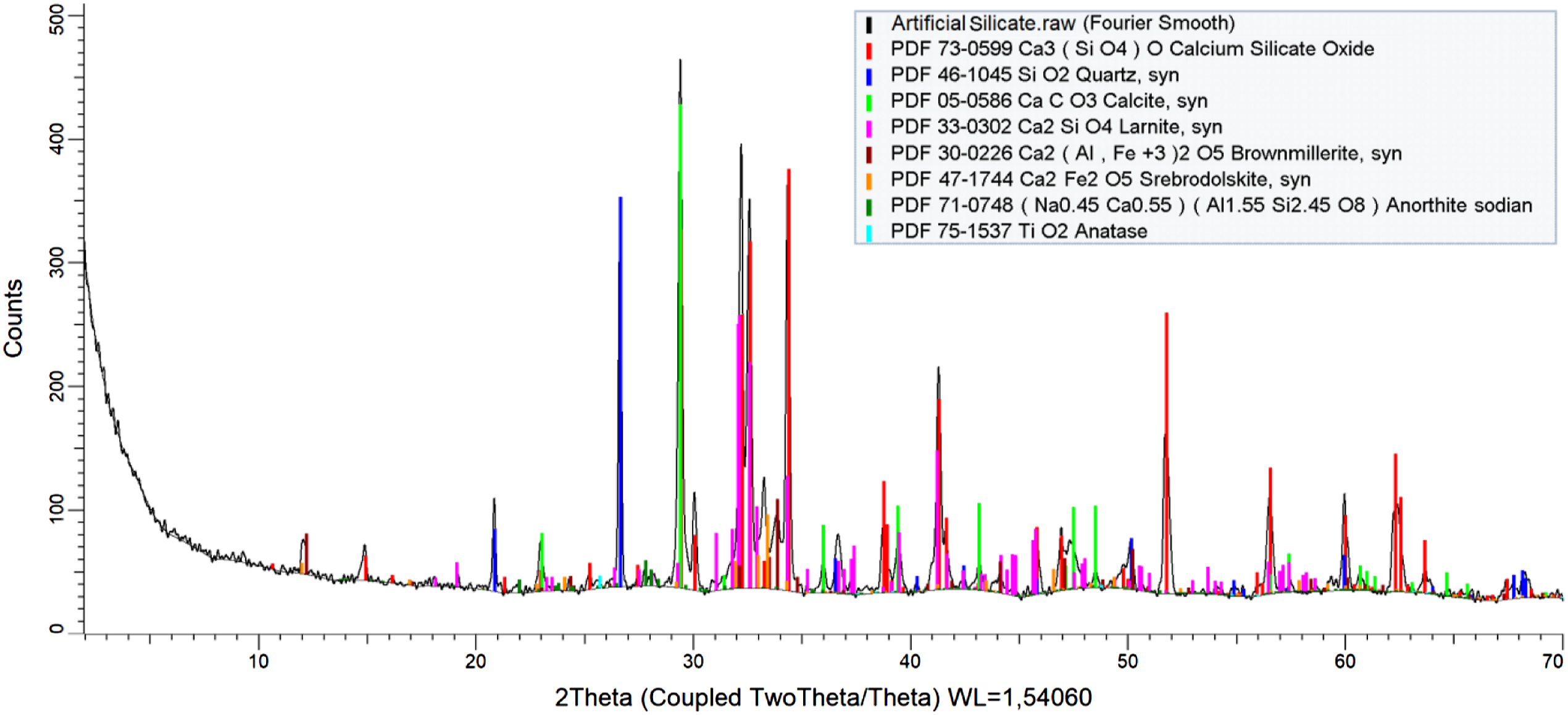
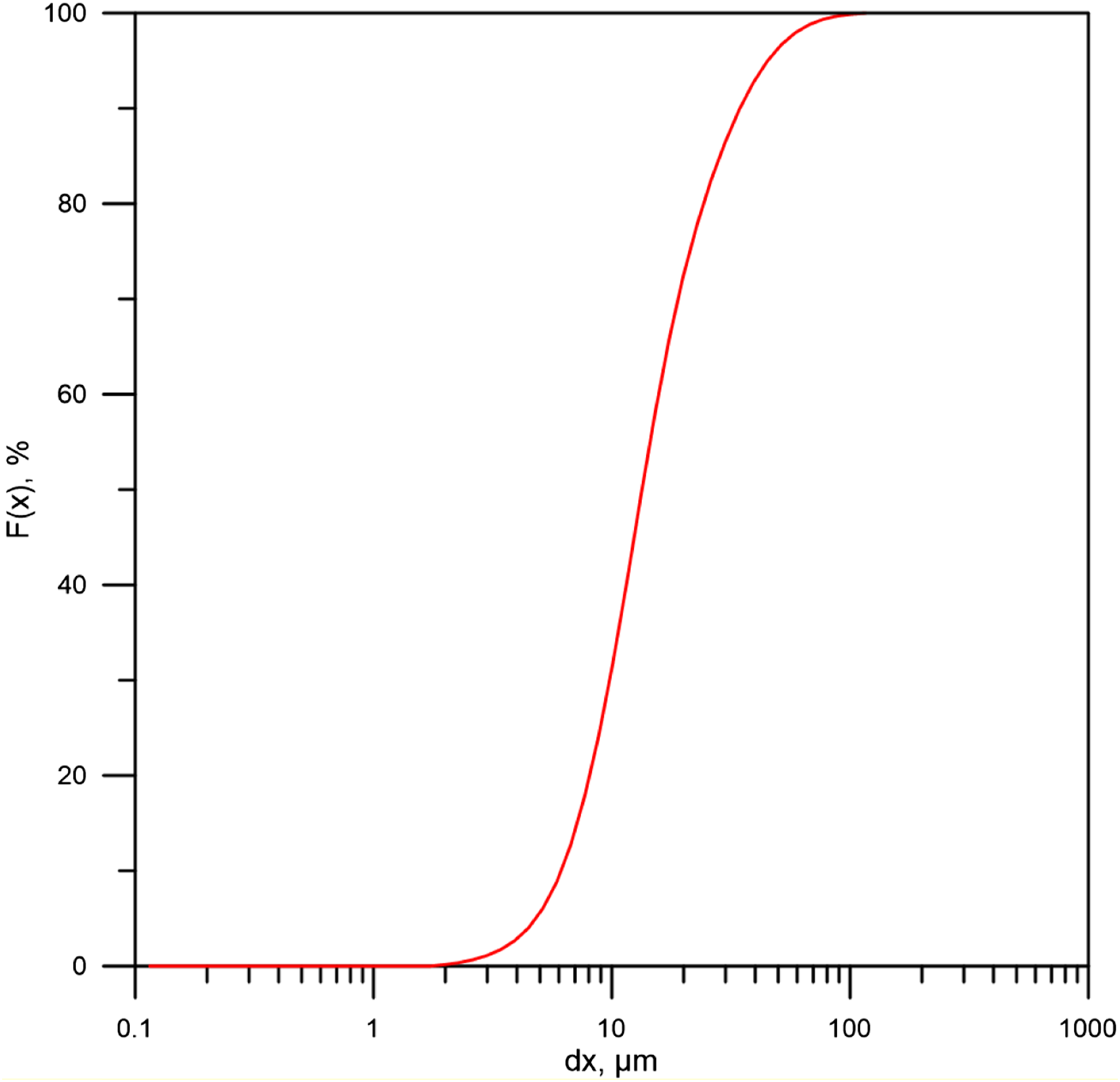
The materials used are listed in the Table 1. 1. Poly(vinyl chloride) powder type S-5070 (under trademark Ongrovil®) produced and supplied by BorsodChem Zrt, Hungary. 2. Plasticizer DOP (Di-Octyl Phthalate) supplied by DEZA, a. s. CO, Valašské Meziříčí, Czech Republic. 3. Calcium-Zinc-based stabilizer (under trademark Newstab-50), which supplied by Betaquímica CO, Barcelona, Spain. 4. Wax-E (under trademark Licowax®E) supplied by Clariant International Ltd, Muttenz Switzerland. 5. Artificial silicate, supplied by Bioekotech Hungary Kft. Analysis of SEM- EDAX for Artificial silicate is shown in Figure 1. This test was conducted at the University of Miskolc, Hungary, using a Carl Zeiss EVO MA10 SEM. Figure 2 represents the XRD analysis for artificial silicate. XRD analysis was performed using Bruker D8 Advance instrument at the University of Miskolc, Hungary. The particle size distribution of the artificial silicate is shown in the Figure 3. This analysis has been accomplished by the HORIBA LA-950V2 laser particle size analyzer. Materials used and their percentages. SEM- EDAX analysis for artificial silicate. XRD analysis for artificial silicate. Particle size distribution of artificial silicate.

Mixing process
A high-speed fluid mixer type Mischtechnik MTI 10 had mixed all of the raw components listed in Table 1. Due to the shearing of particles between the components, the temperature rises progressively from ambient temperature to 150°C when the mixing process begins, rising from 600 r/min to 2700 r/min. For around 15 min, the combination spins at 2700 r/min as the temperature climbs to 150°C. It permits the plasticizer to penetrate all of the poly(vinyl chloride) particles, resulting in a homogenized structure and optimal manufacturing properties. After that, the mixing speed is reduced to 600 r/min, cooling water is circulated in the mixer’s jacket, and the temperature drops to 45°C, and the mixture remains at this temperature and speed until the mixing phase is done. The mixing process takes roughly 40 min to complete. Artificial silicate is applied to the basic formulation mixture of poly(vinyl chloride) by weight fraction (1, 3, and 5 wt.%) and mixed in a small electrical mixer to uniformly disperse and homogenize into sample powder mixes after the main mixing phase.
Samples preparation and tests
The ISO standards were followed for samples preparation and testing. The preparation and testing procedure have been completed at BorsodChem Zrt, Hungary. The tests are divided into two groups: (1) main tests and (2) sub-tests. Sub-tests depend on their implementation on the main test samples and are primarily analytical, as will be explained below.
Main tests
1. Static heat stability: Schwabenthan type 150U polymix roll mill were used to produced samples of static heat stability with a cross section dimensions 200 mm length, 15 mm width, and 1 mm thickness. Static heat stability test or heat stability testing oven has been done withen 2 h at 190°C by using a Stabilemetr PVC 03 device according to EN ISO 305 standard.
24
2. Limiting oxygen index: L.O.I samples were prepared by a Schwabenthan type 150U polymix roll mill as single sheets, then compressed using a hydraulic press to the desired shape with a cross section dimensions 80 mm length, 10 mm width, and 4 mm thickness. The L.O.I test was conducted following the ISO 4589-2 standard,
25
and a Stanton Redcroft FTA flammability unit was used for completing the test. 3. Dehydrochlorination: Schloemann type BT-50 twin-screw extrusion machine was used to prepare dehydrochlorination samples as a pellets shape with 3 mm diameter and 2 mm thickness. The standard method was used to estimate the dehydrochlorination rates by Metrohm 763 Thermomat at 160°C and 180°C. The Dehydrochlorination test was achieved according to ISO 182-3 standard.
26
4. The Vicat test: a disk shape samples with a cross section dimensions 3 mm thickness and 10 mm diameter were first prepared as by Schwabenthan type 150U polymix roll mill, and then pressed to the desired shape by a hydraulic press. The test was completed with a Ceast type 6520 instrument, according to ISO 306 standard.
27
5. Tensile test: ISO 527 standard was used to produce tensile samples from plates prepared by Schwabenthan type 150U polymix roll mill.
28
6. Chlorine content test: the milled powder samples were analyzed using XRF analyzer according to EN ISO 305:2019 standard.
29
Sub-tests
1. Color change analysis: Many of the color changes of the thermal stability test samples cannot be distinguished by the human eye, so the analysis of these samples was carried out by a datacolor spectraflash SF 300 spectrophotometer. This test was performed in accordance with the ISO 11,664-1 standard.
28
2. Thermal gradient analysis: During performing of L.O.I test, a thermal imaging camera type FLIR SC660 was used to ascertain the thermal gradient of heat affected zone (HAZ).
Results and discussion
When poly(vinyl chloride) in general begins to degrade at high temperatures, it begins to release hydrochloric acid (HCl). The degradation process of poly(vinyl chloride) is accompanied by visible and invisible color changes,
15
as shown in Figure 4, which represents the Tested samples by Static heat stability test at 190°C for flexible poly(vinyl chloride) containing artificial silicate. From the first sample, which represents pure flexible poly(vinyl chloride), we notice that the color turns yellow, orange, red, brown, and finally entirely black. The creation of polyene as a result of dehydrochlorination of poly(vinyl chloride) with the reaction of HCl could explain the yellowish color, where the development of conjugated polyene sequences double bonds causes the discoloration.30,31 This means that the high values of yellowing, the higher the polyene reactions will be, accompanied by an increase in double bonds, and the HCL release will continue to rise.
32
In case of pure poly(vinyl chloride), the color change is very rapid due to the low thermal resistance, where the color change started from transparent to pale yellow, then yellowish orange, then yellowish red, then pale red, then dark red, then brown, until it becomes completely dark.15,33–36 This behavior is very clear from Figure 5, which represented the yellowish-bluish diagram of poly(vinyl chloride) containing artificial silicate that the reactions of polyene are high for pure poly(vinyl chloride) (black curve), beginning with the exposure of the polymer to high test temperatures due to the violent release of chlorine and the formation of HCl, which leads to the formation of excessive double bonds.37–39 Static heat stability strips for flexible poly(vinyl chloride) containing artificial silicate tested at 190°C. Yellowish-bluish behavior of flexible poly(vinyl chloride) containing artificial silicate.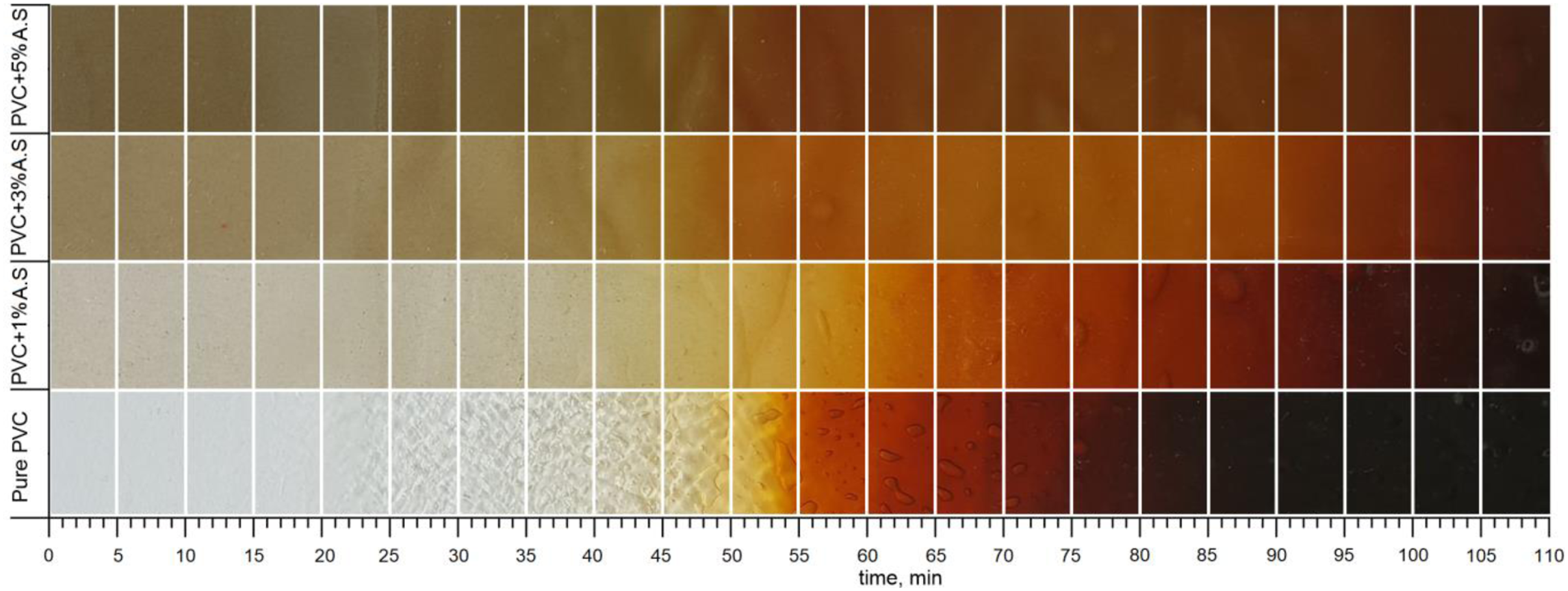
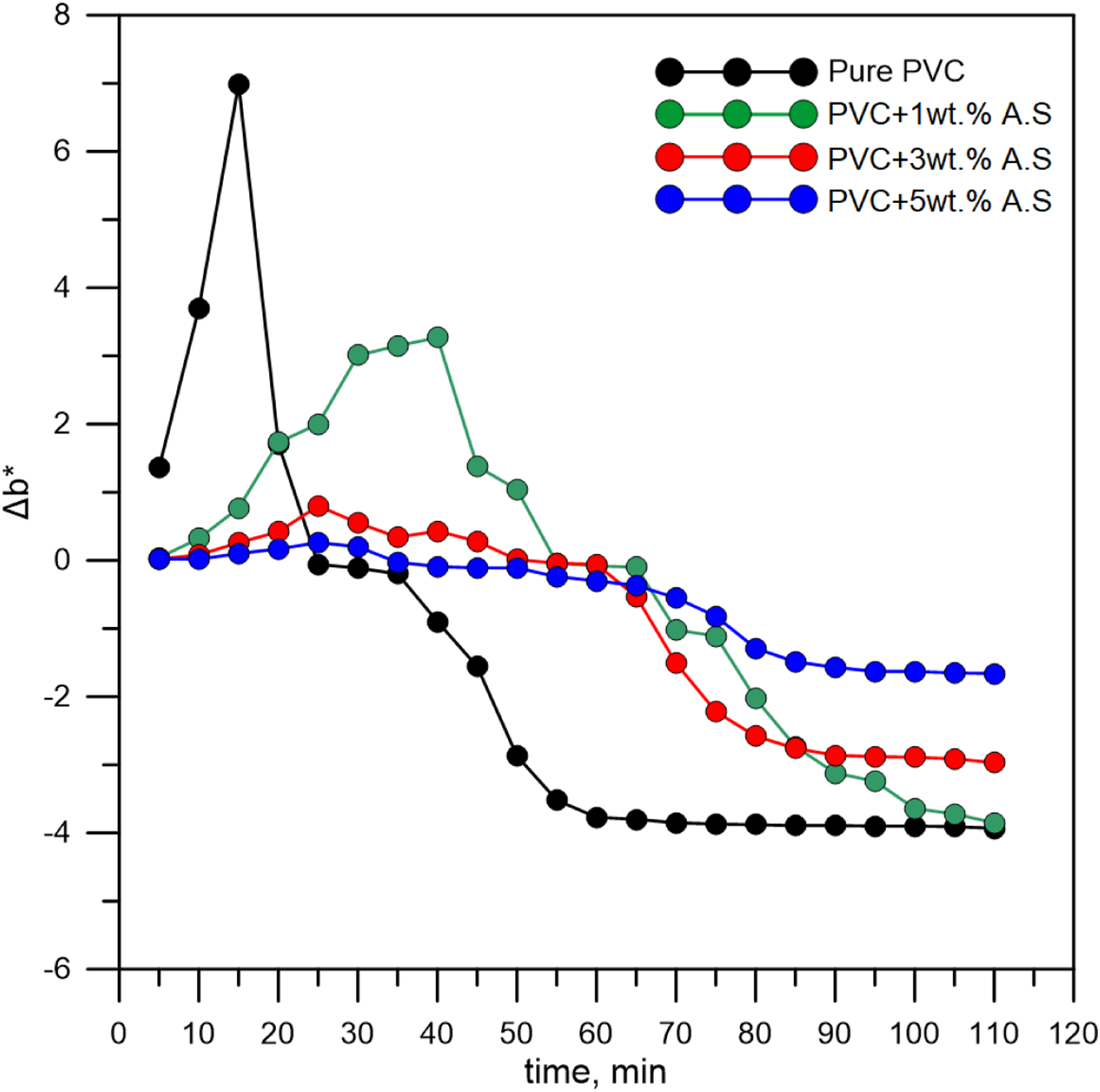
Poly(vinyl chloride)’s behavior changes after adding 1 wt.% of artificial silicate, as the yellowing intensity decreases, as shown in Figure 4. This means that the polyene has been slowed down and hindered by the artificial silicate. This behavior of artificial silicate indicates that it’s an ideal stabilizer for poly(vinyl chloride), where discoloration of poly(vinyl chloride) will be reduced thanks to the artificial silicate. As a result, the degradation process of poly(vinyl chloride) has been reduced by employing artificial silicate as an active filler, which improves the thermal resistance of poly(vinyl chloride) and enhances the effectiveness of the original stabilizer (Newstabe). Increasing the percentage of artificial silicate additives to 3 wt.% and 5 wt.% will improve the discoloration resistance of poly(vinyl chloride), as shown in Figure 4, where the yellowing is reduced significantly. When the percentage of artificial silicate increases, the color changes will not be easily recognized because of the dark color of the artificial silicate, so it is easier to trace these color changes through Figure 5. Where, after adding artificial silicate, the yellowing values decrease significantly (the green, red and blue curves), not only this, but the bluing values decrease as well. The high values of bluish mean excessive degradation and combustion of the polymer, so it turns dark. The artificial silicate works to impede the synthesis of polyene and thus reduce the rate of degradation due to the slow release of HCl. The unique composition of artificial silicate, which includes a variety of compounds such as oxides and carbonates, is responsible for its behavior. These chemical compounds will synergistically interfere with the original stabilizer which is calcium-zinc-based stabilizer, producing a synergistic stabilizing action. This synergistic stabilization leads to an increase in the thermal stability of poly(vinyl chloride).
Poly(vinyl chloride) begins to lose its surface brightness and becomes dull over time, and this change in appearance can be accelerated by heat. In Figure 6, which represents the chroma for poly(vinyl chloride) containing artificial silicate, we can see that the intensity of the brightness is high for poly(vinyl chloride) represented by the black curve. Still, after which it drops very low as it becomes dull. This color change is caused by the release of HCl due to degradation. But after artificial silicate is added, this behavior changes as the polymer maintains a shiny appearance for a long time before it completely degrades. It is true that initially, the surface brightness after adding artificial silicate is lower than that of pure poly(vinyl chloride) (green, red, and blue curves) and this is due to the opaque appearance of the artificial silicate. Furthermore, the light-colored additives give a more bright appearance.
40
Difference in chroma for flexible poly(vinyl chloride) containing artificial silicate.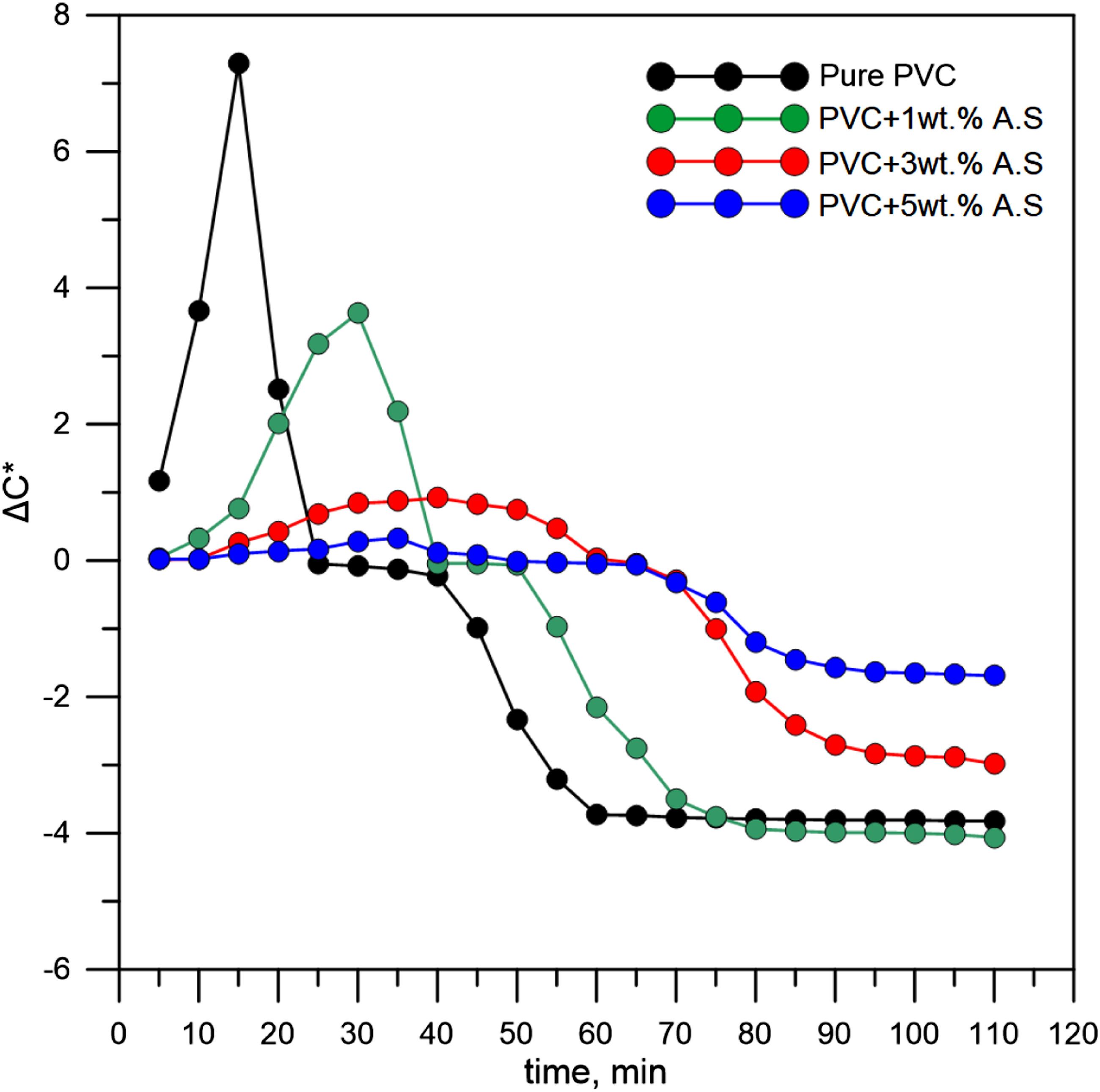
All materials, including plastic, when exposed to fires or high temperatures without flame, the effects of this exposure are not limited to the outer surface only but also extend to the inner layers located under the combustion area, which is called the heat-affected zone.41,42 This critical area which exposed to heat only, suffers significant structural changes, causing rapid failure if the flame retardation of the material is not improved. In Figure 7, which represents the thermal imaging of L.O.I test samples, we note that the heat-affected zone of poly(vinyl chloride) is comprehensive compared to the samples that contain specific percentages of artificial silicate, where the heat-affected zone decreases, with artificial silicate percentage increases, which is also accompanied by an increase in the value of L.O.I. The explanation for this is due to the high stabilizing action of poly(vinyl chloride) after adding artificial silicate, which comes from the interaction of this filler with the original stabilizer, results in a synergistic action that improves the flame retardation resistance, and finally rationing of the size of the heat-affected zone. Thermal imaging of L.O.I test samples for flexible poly(vinyl chloride) containing artificial silicate.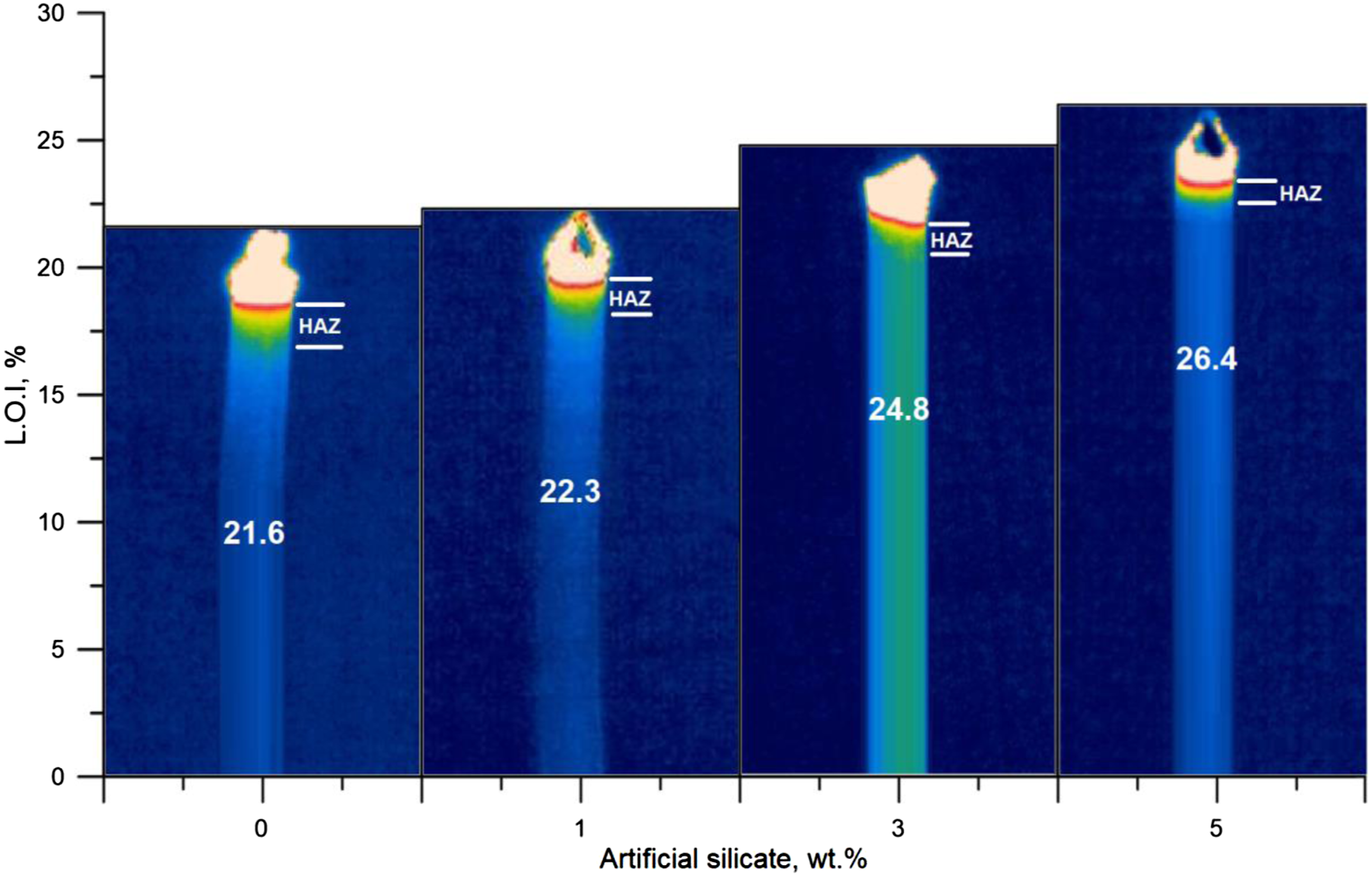
The degradation of poly(vinyl chloride) is a sequential process that takes place in three steps: initiation, propagation, and termination. The speed of entry into each of these stages depends on the resistance of the polymer and the surrounding conditions. The degradation process begins with the release of HCl, resulting in the synthesis of conjugated polyene sequences. When polyene-forming reactions are allowed to continue, the polymer will lose its properties, and its resistance will begin to deteriorate, eventually leading to its complete combustion.43–46 The high inclination of the pure poly(vinyl chloride) curve (black) shown in Figure 6 refers to the fact that the loss of HCl is rapid because poly(vinyl chloride) has poor thermal resistance at high temperatures, resulting in the formation of allylic chloride groups in the structural segments of poly(vinyl chloride).47–49 After adding artificial silicate, the dehydrochlorination behavior of poly(vinyl chloride) has been changed, where the structure of poly(vinyl chloride) demonstrating greater stability at elevated temperatures, as shown in the green, red, and blue curves within Figure 8, which represent 1wt%, 3wt.%, and 5wt.% of artificial silicate additives, respectively. This indicates that the HCl release has been reduced due to the poly(vinyl chloride) structure being more stable. In addition, after adding artificial silicate, the degradation period was significantly extended, and this period expanded further with increased artificial silicate percentage. From Table 2, we notice the significant improvement in the stability time of poly(vinyl chloride). The magnitude of increase in stability time more than doubled when 5wt.% artificial silicate was added compared with their value in the case of pure poly(vinyl chloride). This progression is due to the increasing synergistic effect between artificial silicate and the original stabilizer. Dehydrochlorination of flexible poly(vinyl chloride) containing artificial silicate at 160°C. Stability time for flexible poly(vinyl chloride) containing artificial silicate.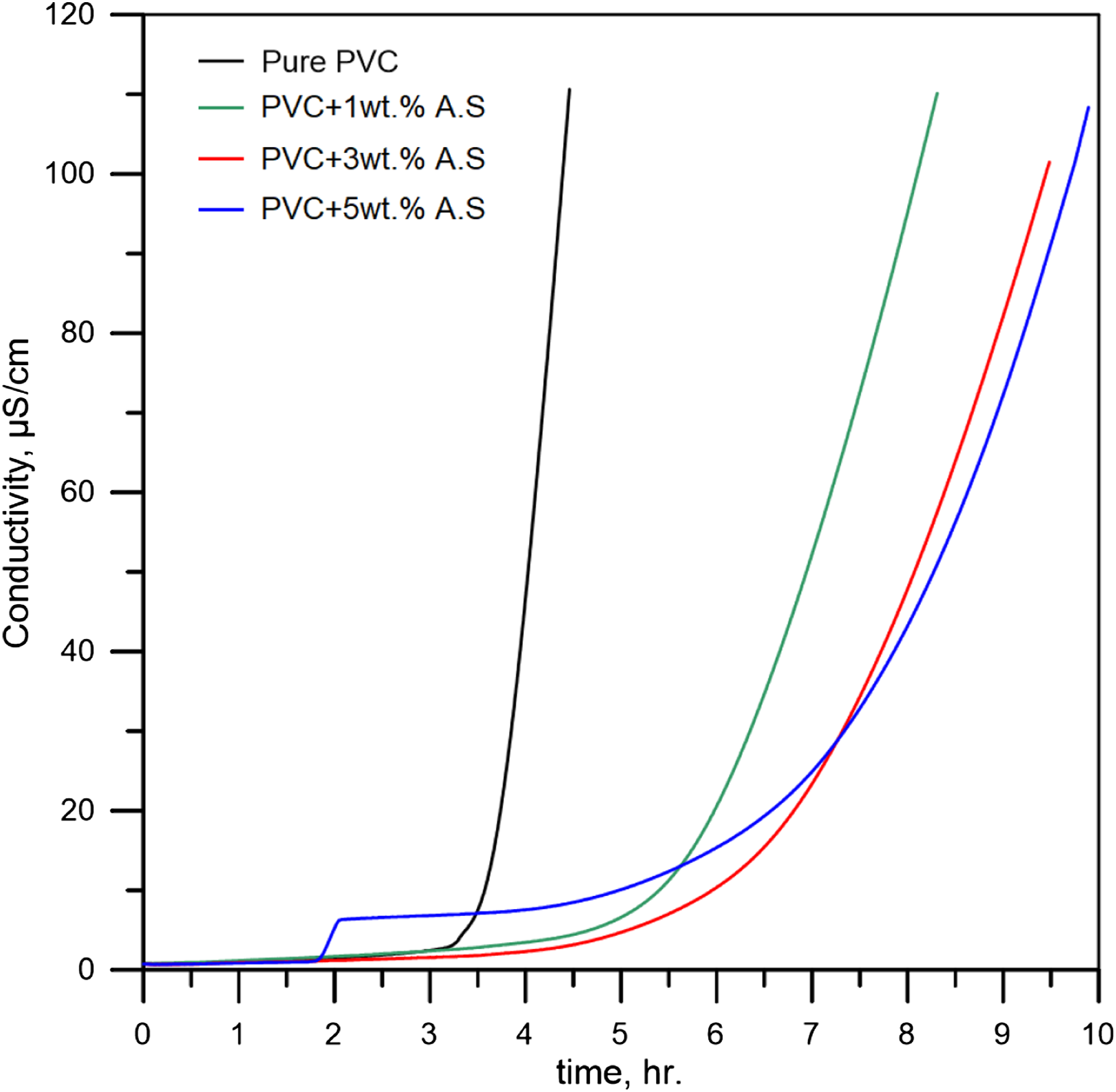
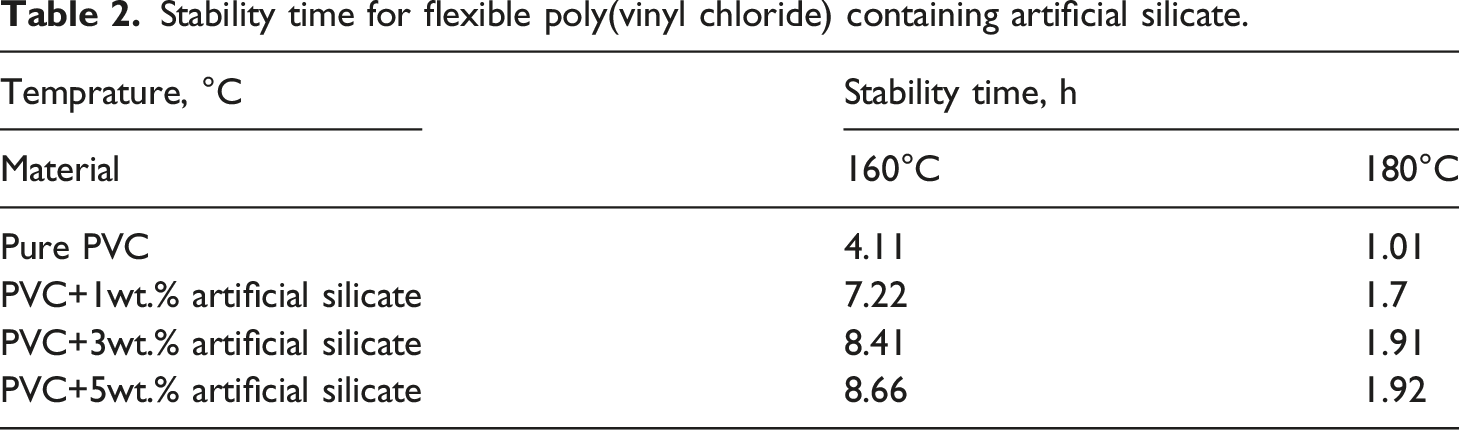
As we know, the increased temperature is an effective accelerator to increase the degradation rate and an abundance of HCl releasing, which is what we notice in Figure 9, which represents dehydrochlorination behavior of poly(vinyl chloride) containing artificial silicate at 180°C. Despite the steady rise in poly(vinyl chloride) degradation rate, which is evident from Figure 9 (black curve), artificial silicate is still an effective stabilizing additive. When referring to Table 2, we will notice that the increase in the stability time was (90.1%) when adding 5wt.% of artificial silicate compared to the pure poly(vinyl chloride). This behavior is also evident in Figure 9, where the artificial silicate extends the degradation period (green, red, and blue curves). Dehydrochlorination of flexible poly(vinyl chloride) containing artificial silicate at 180°C.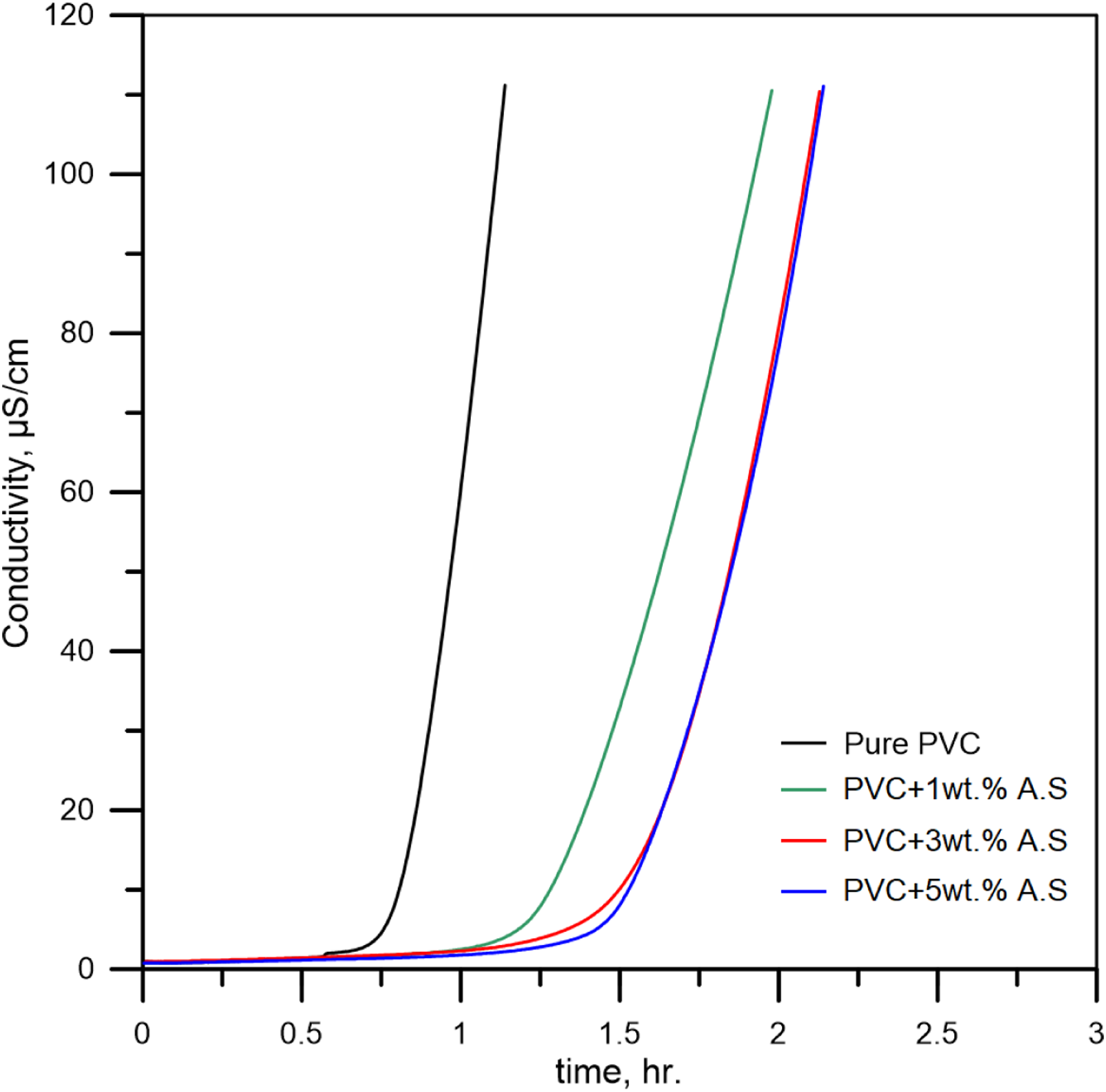
Many additives can be mixed with poly(vinyl chloride) to improve its properties, and one of these properties is softening point.
50
Figure 10 represents the Vicat test results for poly(vinyl chloride) containing artificial silicate. From this figure, the softening point of poly(vinyl chloride) has been shifted upwards after adding artificial silicate. The results indicated that the increase in the penetration resistance is a continuous and almost linear increase, which means there is a high affinity between artificial silicate. This behavior is due to the chemical structure of artificial silicate containing many active groups coming from many compounds, including aldehyde, ketone, amines, polyamides, and alcohols or aromatic or phenolic. All of these compounds can combine with poly(vinyl chloride) to form double and tie triple bonds, as well as aromatic rings, resulting in a more substantial material.
15
Vicat (heat distortion temperature) results for flexible poly(vinyl chloride) containing artificial silicate.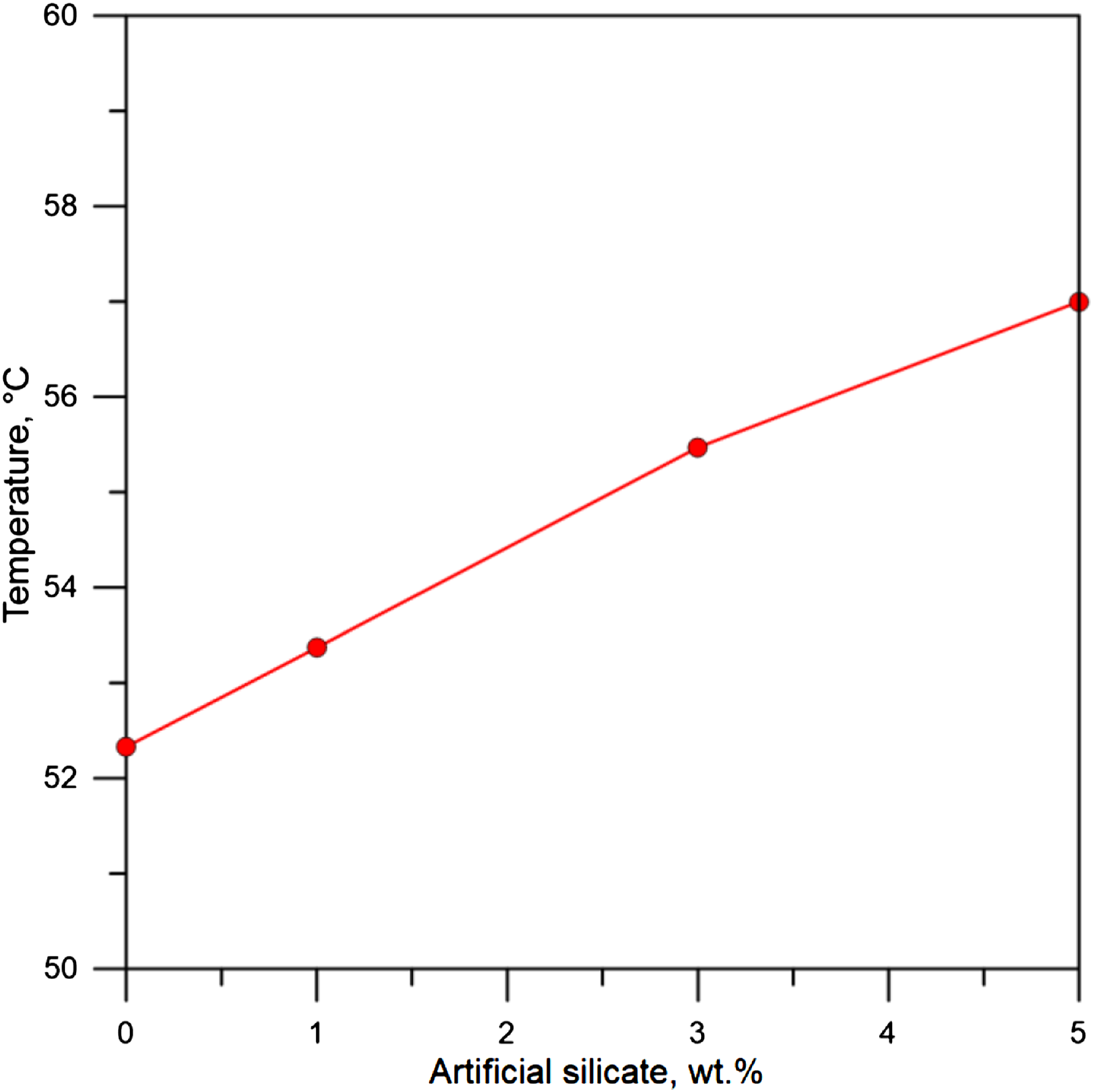
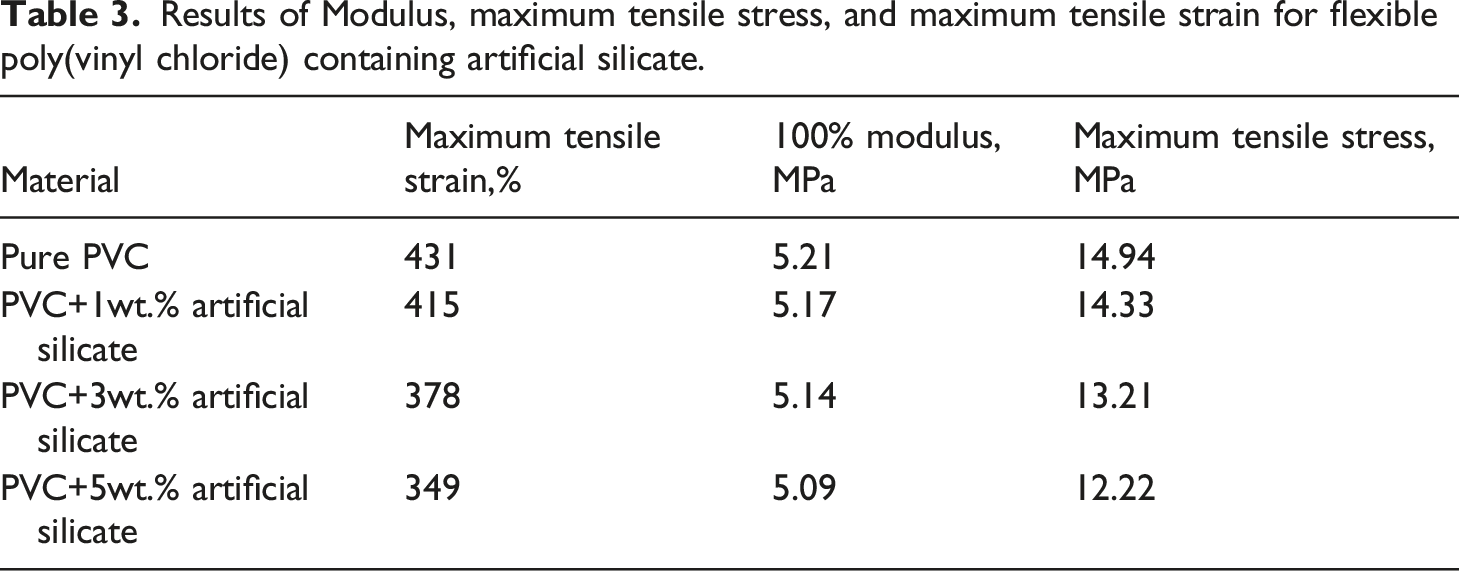
Despite the significant improvement in thermal properties of flexible poly(vinyl chloride) in general, but the side effect of adding artificial silicate is that the tensile strength has been reduced, which is clearly seen in Figure 11, which represents the tensile strength of flexible poly(vinyl chloride) containing artificial silicate. Table 3 represents the results of Modulus, maximum tensile stress, and maximum tensile strain for flexible poly(vinyl chloride) containing artificial silicate. This decrease is mainly caused by the rupture of the polymer surrounding the artificial silicate particles, and this is evident from Figure 12, which represents the SEM analysis of the flexible poly(vinyl chloride) samples before and after the tensile test. We clearly notice the polymer tearing around the granules, which contributes significantly to a sharply reduced tensile strength. From our point of view, perhaps this situation could be avoided if the grain size of the artificial silicate particles were much smaller. i.e. as nanoparticles. Tensile test curves for flexible poly(vinyl chloride) containing artificial silicate. Results of Modulus, maximum tensile stress, and maximum tensile strain for flexible poly(vinyl chloride) containing artificial silicate. SEM analysis for flexible poly(vinyl chloride) containing artificial silicate (a) before and (b) and (c) after tensile test.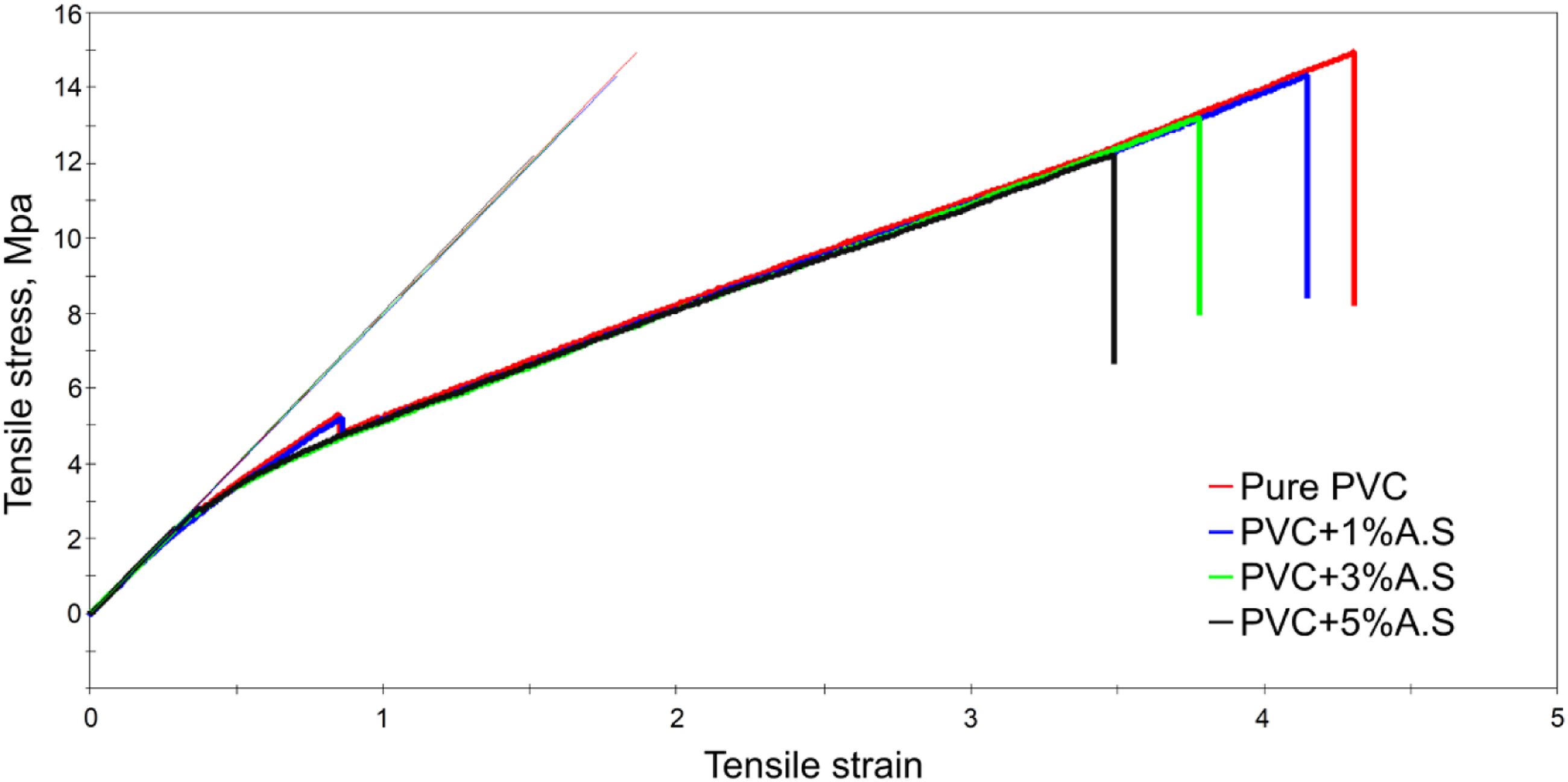

The other critical parameter that leads to the deterioration of the tensile property and at the same time works to maximize the thermal properties of poly(vinyl chloride) is that the artificial silicate reduces the percentages of chlorine and PVC contents simultaneously, as shown in Figure 13, which represents the results of chorine content test for flexible poly(vinyl chloride) before and after artificial silicate additions. Results of chorine content test for flexible poly(vinyl chloride).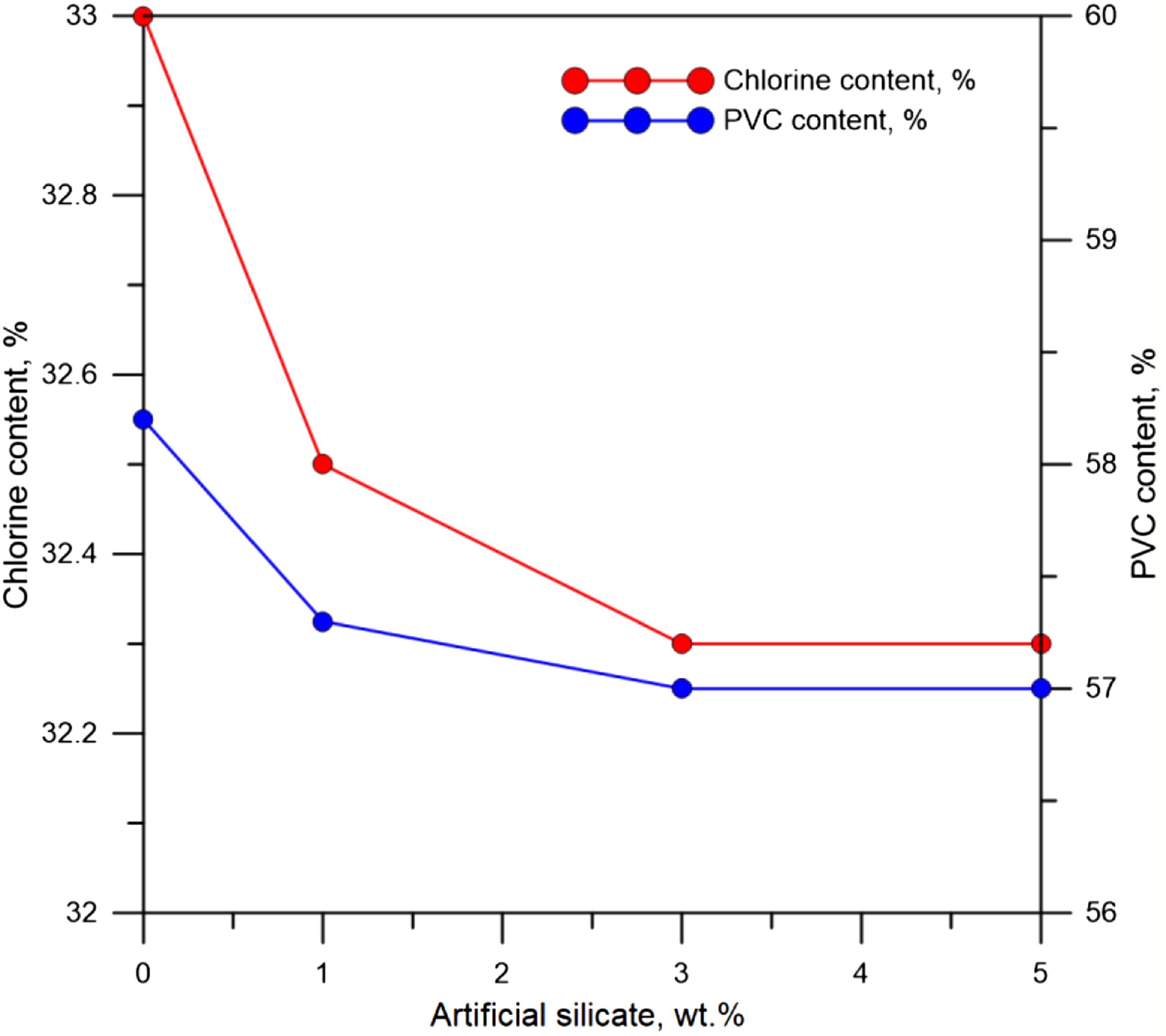
Conclusions
Enhancing the thermal stability and flame retardancy of poly(vinyl chloride) is considered the biggest challenge to guarantee the continued success of this unique polymer in industrial applications exposed to fires. Such enhancement aims not only to increase the thermal efficiency of poly(vinyl chloride) but also to make it safer to use by reducing and inhibiting the release of HCl upon decomposition. The other important thing is that the additives used to improve the properties of the polymer are as environmentally safe as possible and do not cause pollution or harm to health. The additive used in this article, which is artificial silicate, has proven its industrial and environmental efficiency. From an industrial point of view, the percentages added to artificial silicate improved the thermal properties tested for poly(vinyl chloride) as a whole. In addition, the HCl release decreased sharply after the addition of artificial silicate, which was reflected in a significant improvement in the thermal stability of poly(vinyl chloride) by significantly reducing the synthesis of polyene, which resulted in a decrease in the yellowing intensity associated with the synthesis of polyene. The artificial silicate also enables the poly(vinyl chloride) to maintain an acceptable, brighter appearance for a long time. The addition of artificial silicate resulted in a significant improvement in the poly(vinyl chloride)’s resistance to dehydrochlorination. The degradation period was extended by 121.8% and 88% than it was in the case of pure poly(vinyl chloride) at 160°C and 180°C, respectively. Also, the stability time has been increased from 4.11 h and 1.01 h in the case of pure poly(vinyl chloride) at 160°C and 180°C respectively, to 8.66 h and 1.92 h when 5% of artificial silicate was added at the same temperatures. This means that the rates of improvement in the time of stability were 110.7% and 90.1% at 160°C and 180°C, respectively, and it is a great improvement compared to that value in the case of pure poly(vinyl chloride). This proves that artificial silicate significantly inhibits the HCl release process and delays the entry of the poly(vinyl chloride) into the first step of degradation, in addition to slowing its entry into the rest of the steps, with the continued exposure of the polymer to the conditions of degradation. Artificial silicate works to start narrowing the heat affected zone effectively, allowing higher flame retardation levels. This is what we notice through the increase in the value of the L.O.I by 22.2% than before the addition. This improvement in flame retardancy will prevent the layers’ entry located below the combustion zone to advanced stages of degradation and combustion. The penetration resistance of pure poly(vinyl chloride) in the Vicat test improved by 9% after adding 5wt.% of artificial silicate. Decreased tensile strength of the flexible poly(vinyl chloride) due to the ruptures in the polymer layer surrounding the particles of artificial silicate. It is because of the large grain size of these particles.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors certify that they NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
