Abstract
Ultrahigh molecular weight polyethylene (UHMWPE) are widely used as a biomaterial for manufacturing of prostheses, implants and other biomedical components with complex geometry and shapes. Continuous loading of these components subject to heavy stress and deformation resistance should have enhanced mechanical and chemical properties. This paper aims to improve the mechanical and thermal aspects of the conventional UHMWPE by supplementing reduced Graphene oxide (rGO) in varying weight percentages to develop polymer bio nanocomposite samples. The effect of green synthesized GO nanoparticles in the UHMWPE polymer was investigated for biomedical application. The rGO was distributed in a UHMWPE matrix using a unique and optimized technique to create high-performance nanocomposites. The proposed UHMWPE filled with different loading (0, 0.5, 1.0,1.5, 2.0, and 3.0 wt.%) of rGO was produced by Ultrasonication in an acetone medium. The findings suggest that evenly distributed rGO layers were present throughout the polymer matrix. This, in turn, indicates a good connection between the fillers and matrix by Scanning electron microscopy (SEM) and Energy Dispersive X-Ray Analysis (EDAX), resulting in better composite capabilities. The layers of rGO-aided lamellar arrangements and microfibers between the crystals were observed in the results. The Microhardness of the bio nanocomposite (1 Wt.% rGO/UHMWPE) with 1wt.% rGO in the UHMWPE matrix increased by 2.8% compared to an unfilled polymer. At the same rGO concentration, the bio nanocomposite had a crystallization degree of 46.96%. To achieve optimal performance, rGO content of 1wt.% was added to the sample, which is ideal in many situations where good mechanical and thermal qualities are required during operation. The outcomes reveal that the rGO supplement primarily boosts the thermo-mechanical performances of the modified bio-nano composites for orthopedic products.
Introduction
Biomaterial research has been one of the fastest growing and most active fields in recent years due to its usefulness and societal interest. Polymeric biomaterial has emerged at the tip of developing new tailored-made bio-nano composites. Polymer matrix composites have become an alternative to metal/metal matrix composites due to their high biocompatibility, mechanically strong chemically stabilized structure adaptability, and durability concerns.1,2 It can be used for drug delivery, tissue engineering, cancer therapy, biosensors, artificial joints, and many other things.3–5 The hybrid and supramolecular approaches are recently used in polymeric biomaterial research, resulting in the significant contemporary development of polymer science and engineering. Polymer morphologies and structural features can be fine-tuned to fulfill various medical application needs. The natural and inorganic nanoparticles application for polymeric biomaterials may be mixed to obtain new and improved properties that can enhance the performance and efficiency of the developed materials. Bioinspired and adaptive polymer systems were also created to improve the required material design. It has been remarked that numerous studies have been published on the improvement of the bio nanocomposites properties needed for mechanical and tribological applications. For biomedical applications, such as Bearings, Pins, Coatings, Wires, and Artificial joints, polymer matrix composite materials have emerged as a viable alternative to metals. Also, their alloys consist of enhanced benefits of low density, simplicity of manufacturing, significant shock absorption capacity, wear resistance and high vibration self-lubrication.6–8 Many studies have been conducted to enhance the polymer characteristics of UHMWPE since its debut in the 1960s as a preferred material for implant-bearing components. Because of its clinical performance over the past few decades, UHMWPE is commonly used for artificial joints.9,10 Firdous et al. 11 studied the Polarimetric characterization of UHMWPE for bone substitute biomaterials. The findings show that UHMWPE is a robust and compact compatible material. This research aims to establish an optical evaluation approach for bone graft substitute biomaterials within the medical business.
In contrast, UHMWPE seems to have a weak load-bearing capability, a low Young's modulus, and an anti-fatigue capability. 12 As a result, attempts have been devoted to enhancing UHMWPE tribological properties. In contrast, UHMWPE's thermo-mechanical properties are little affected by Vitamin E incorporation.13,14 Another common technique is the addition of fillers material to recover mechanical, tribological, and biocompatibility properties.15–17 Inorganic fillers such as kaolin, 18 zirconium,9,19,20 and nano zinc oxide 21 were utilized for this purpose.
In order to improve the processability of UHMWPE, Wu et al. 22 used differential scanning calorimetry to study the crystallization and melting behaviour of UHMWPE and its composites filled with CaCO3, acrylic acid-modified CaCO3(AA-CaCO3), and PE wax coated CaCO3(PEW@ CaCO3). Adding CaCO3, AA- CaCO3, and PEW@ CaCO3 raises the crystallization temperatures (Tcs) and crystallinity of UHMWPE owing to the heterogeneous nucleation of fillers. The PEW@ CaCO3 demonstrates better improvement in processability of UHMWPE than AA-CaCO3. This is because the PEW macromolecular chain of PEW@ CaCO3 is more compatible with the UHMWPE than the AA-CaCO3. Khasraghi and Rezaei 23 explored the effect of MWCNTs (1wt.%) in UHMWPE and HDPE blends. The two variants of HDPE were used, HDPE 5218 and HDPE 3840 for this study. It was found that the mechanical characteristics of the blend samples made with HDPE 5218 were somewhat enhanced due to the incorporation of MWCNT. The composites crystallization was improved by adding MWCNT to 20 wt.% of the UHMWPE blend but slowed at higher UHMWPE contents. UHMWPE/HDPE/MWCNT nanocomposites were found to be less elastic than their blends. In the study of Dubey et al., 24 the effect of Graphene nanoplatelets (GNPs)/hydroxyapatite (HA) in PMMA bone cement is used to produce a biopolymer composite. The amount of each nanofiller applied is between 0 and 0.5 wt.%. The results reveal that a sample containing 1.5 wt.% of GNP/HA/PMMA led to an increase in flexural strength of 49.28% and an increase in flexural modulus by 30.80%. Furthermore, it has a compression modulus of 57.41% and a compression strength of 31.7%. Thus, finds suitable to be used in various biomedical industries. Liang et al. 25 explored the growth of organic montmorillonite (OMMT) clay-filled UHMWPE nanocomposite by Ultrasound-assisted melt mixing. It showed that melt mixing with ultrasonic and extensional deformation was beneficial in silicate layer exfoliation, filler distribution, and dispersion. The material's thermal stability, tensile strength, storage modulus, and complicated viscosity were all observed to improve with the addition of the right amount of OMMT but worsened with the addition of ultrasound treatment by ultrasound-assisted vane mixer (UAVM).
In addition to this, Hussain.et al. 26 studied the new material developed from nano Al2O3/Vitamin-C filled UHMWPE Polymer matrix bio nanocomposite as a biomaterial for artificial cartilage in three variations (0.5,1 and 2 wt.%) of nano Al2O3 was analyzed with 2 wt.% Vitamin C in UHMWPE. A higher wear resistance against Ti6Al4V alloy has been noted with 2wt.% Al2O3 under Human serum environment due to film formation on the developed bio nanocomposite surface. Suner. et al. 27 explored UHMWPE/GO biomaterial for joint application using a hot press moulding technique (185°C under 15 Mpa). The addition of GO improved the oxidative resistance, thermal stability, wettability, and mechanical properties of the polymer matrix. 0.5 wt.% of GO showed the optimal physical and mechanical properties for the developed biomaterial concerning unfilled one. Chen et al. 28 also developed the GO (0-1wt.%) based UHMWPE biomaterial having porous structure by utilizing NaCl which gets evaporated under higher temperature forming porous surface in UHMWPE/GO composite by Hot-press molding. The combination of GO and NaCl improves wear resistance and reduces friction in the UHMWPE polymer matrix. The effect of Alumina Toughened Zirconia (ATZ) was examined by Duraccio et al. 29 comprising ATZ 20–80 wt.% within the UHMWPE matrix using several blending methods followed by compression molding and found good cells interaction with improved mechanical properties. Additionally, the composite with the least quantity of ATZ (2.5 wt.%) exhibits significant yield stress improvement. The supplement of 2.5 wt.% ATZ in the UHMWPE matrix enhances hydrophobicity and cell interaction compared to a pristine one, expanding UHMWPE's use for biomedical applications. Bhusari et al. 17 described a novel method for covalently grafting polyethylene chains with GO nanosheets. Processability was enhanced by employing melt-extrusion and injection moulding methods with appropriate loading of modified GO. A Dual hybrid technique was utilized to blend HDPE-UHMWPE for composite development with a small amount of surface-modified GO. A 60/40 ratio of the polymer matrix having 1 wt.% content of GO (surface modified) resulted in mechanical properties. As a result of this research, tensile strength and elastic modulus increased by 65 MPa and 908 MPa, respectively. Suresha et al. 30 explored the behavior of copper nanoparticles (n-Cu) and CNTs mixed with UHMWPE. The improvement in sliding wear behavior has been reported by use of nano-fillers in very limited quantity i.e. (0.1–0.5 wt.%). In addition, the CNTs (1-D filler) decrease the interface surface area., increases UHMWPE interchange, and inhibit interlocking with a furrowing of developed composite with respect to steel counterpart, lowering both, the coefficient of friction (µ) and explicit wear rate (Ks). UHMWPE with 0.5 wt.% CNTs beat the other combinations in terms of tribological performance. A decline in the Ks value has been reported with the increase of abrading distance as well as the coarseness of the emery paper. Also, UHMWPE/CNTs beats both pristine and nano Cu-reinforced UHMWPE in terms of abrasion resistance.
Prior work summary for the supplement of nanomaterials in polymer composite.

Incorporating different nanomaterials has observed a tremendous improvement in property enhancement in various types of the polymer matrix. This work targets the supplement of Graphene and its derivatives because of its enormous success in today's areas of study and the ease with which it may be modified without causing significant difficulty. The Graphene was successfully isolated using graphite in 2004, and much work has been devoted to obtaining its massive amounts.10,42 Both GO and rGO are by-products of the oxidation of graphite. It implies that GO possesses several oxygen-containing functional groups on its basal and layer edges. Both distribution and dispersion of Graphene sheets must be tailored throughout the nanocomposite manufacturing process. It is mainly performed to enhance the surface and mechanical characteristics of the nanocomposites. Researchers nowadays are concentrating on investigating the achievements of Graphene and Graphene derivatives in a diverse range of industrial and biomedical products. Several recent research studies have been published exploring the adaptation of Graphene in solar devices, food industries, drug delivery, biomedical fields, automobile industries, and many more. These investigations have shown that adding carbon nanoparticles to the UHMWPE matrix enhanced its hardness as well as tribological properties.43–46 More research needs to be done to better understand the influence of Graphene/Graphene-derived fillers on the properties of the polymer matrix. Research on Graphene and its derivatives for biomedical applications has proven the range of its potential as a biomaterial throughout the last decade. Chemical or physical reduction processes transform GO into other Graphene derivatives, such as rGO. Much research has been done on biomedical nanocomposites based on nanofillers. Reinforcement materials such as graphite, Graphene oxide (GO), Graphene nanoparticles (GNPs), carbon nanotubes (CNTs), nanodiamonds (NDs) and other forms of Carbon-based nanomaterials (CBNs) have been extensively investigated in polymers such as PMMA, PEEK, PLA, UHMWPE, HDPE, and LDPE, and others, for mechanical, thermal, and tribological applications. The harmless chemical reaction of hydrothermal reduction was used to produce rGO, and Ultrasonication was used to achieve a surfactant-free dispersal of the rGO. Optimization of ultrasonic settings was utilized to create a stable rGO dispersion that could be used to deliver the anti-cancer drug Paclitaxel with more efficiency than previously possible.
47
Apart from this, very little research has been done on how rGO affects polymer matrixes. Kang et al., Priyadarsini et al., and Lee et al. have identified the increased Osteogenesis impact rGO most desired property for bioimplant development.48–50 As a result, further research is required to explore its relevance to implant development. Bhattacharyya et al.
51
developed a reduced GO-based UHMWPE composite using the in-situ reduction method in the presence of phenylhydrazine, a toxic substance, but the composites show high strength and creep resistance. Mindivan and Çolak also evaluated the different loading of rGO (0.1, 0.3, 1.0, and 2.0 wt.%) synthesized from the green route and found good results.
52
Generally, the rGO is synthesized by using phenyl hydrazine as a reduction agent. Hydrazine derivatives are strong and most used reducing agents but highly toxic and instability. These limit its large-scale application. With the growing concern of the biomedical sector, the green route has been used in various applications in biomedical. Especially in biomaterial industries, to develop more compatible and long-lasting implants. Therefore, recently greener materials (amino acid, plant extracts, microorganisms, proteins, hormones etc.) were used to substitute toxic hydrazine used in GO reduction and their yields.53–55 The procedure of green synthesis of rGO (Figure 1) consists of the following set of procedures: Green synthesis of rGO.
According to the above state of the art, there is very little work on supplementing reduced Graphene oxide (rGO) in UHMWPE. The incorporation of rGO has shown better results as desired by different kinds of biomaterials, biomedical devices (sensors), tissue engineering, coating, bone implants, etc. The inquiry into the influence of greenly derived rGO on mechanical qualities is still in its early stages. It demands more focus from the scientific community, biomedical society, and industrial sectors. Few of them have investigated the incorporation of carbon-based materials with Hap nanoparticles in UHMWPE. This research aims to improve the physio-mechanical properties of UHMWPE by using biocompatible nanofillers (rGO). As indicated in the literature review, most work exists to supplement Graphene, GO, GNPs, Hap, CNT's, MWCNT's, SWCNT's, ND's nanofiller materials in the matrix phase. By incorporating rGO (green) into the UHMWPE polymer matrix, the author attempts to overcome the mechanical performance limitation of the UHMWPE articulating material (Hip, Knee, Shoulder, finger, etc.). The present paper evaluated an extensive range of wt.% (0, 0.5,1,1.5,2, and 3) to better understand the synthesized bio-nano composites. Furthermore, mechanical properties (tensile, flexural, and hardness) are assessed and elaborated along with structural, morphological, and thermal investigation. Through FESEM, Fracture surface analysis of tensile samples, Fourier transform infrared spectroscopy (FTIR), X-ray powder diffraction (XRD), and Differential scanning calorimetry (DSC), respectively. The current research aims to develop a cost-effective green bio nanocomposite with minimal loading and enhanced characteristics. It could be a forward step to aid the healthcare industry in fabricating economic and durable implants and regular minimization surgery. This form of rGO-incorporated implants opens up new possibilities in the biomedical sector to develop liner implant materials with high strength, biocompatibility, wear resistance, and drug-delivery capacity, primarily for knee, shoulder, fingers, and hip joint assemblies.
Materials and Methodology
The materials and fabrication techniques used in developing nanocomposite materials are described in the following section. The first section describes the specifications and properties of various materials that have been used in the development of nanocomposite materials. The second section describes the two-step procedure, which includes nano powder development and hot compaction of nano powders.
Materials
The essential polymer matrix was procured via Merck (Sigma-Aldrich) in the form of UHMWPE powder (white colour) with an average molecular weight of 3 x 106–6 x 106 g/mol and a density of 0.94 g/mL at ambient temperature. In this, a melting temperature of 198–201°C and reduced Graphene oxide (rGO) nanoparticles synthesized from the green route with vitamin C and having CAS number 7782425 having average numbers of layers 2–6 along with 0.48 g/mL density at 25°C having the purity of about 98–99% was purchased from Nano_researchlab, Jharkhand India. Acetone having CAS number 67641 along with 58.08 g/mol Molecular weight and 784 kg/m3 density (Purity % > 99%) was purchased from Rankem (LR Lab Reagent).
Methodology
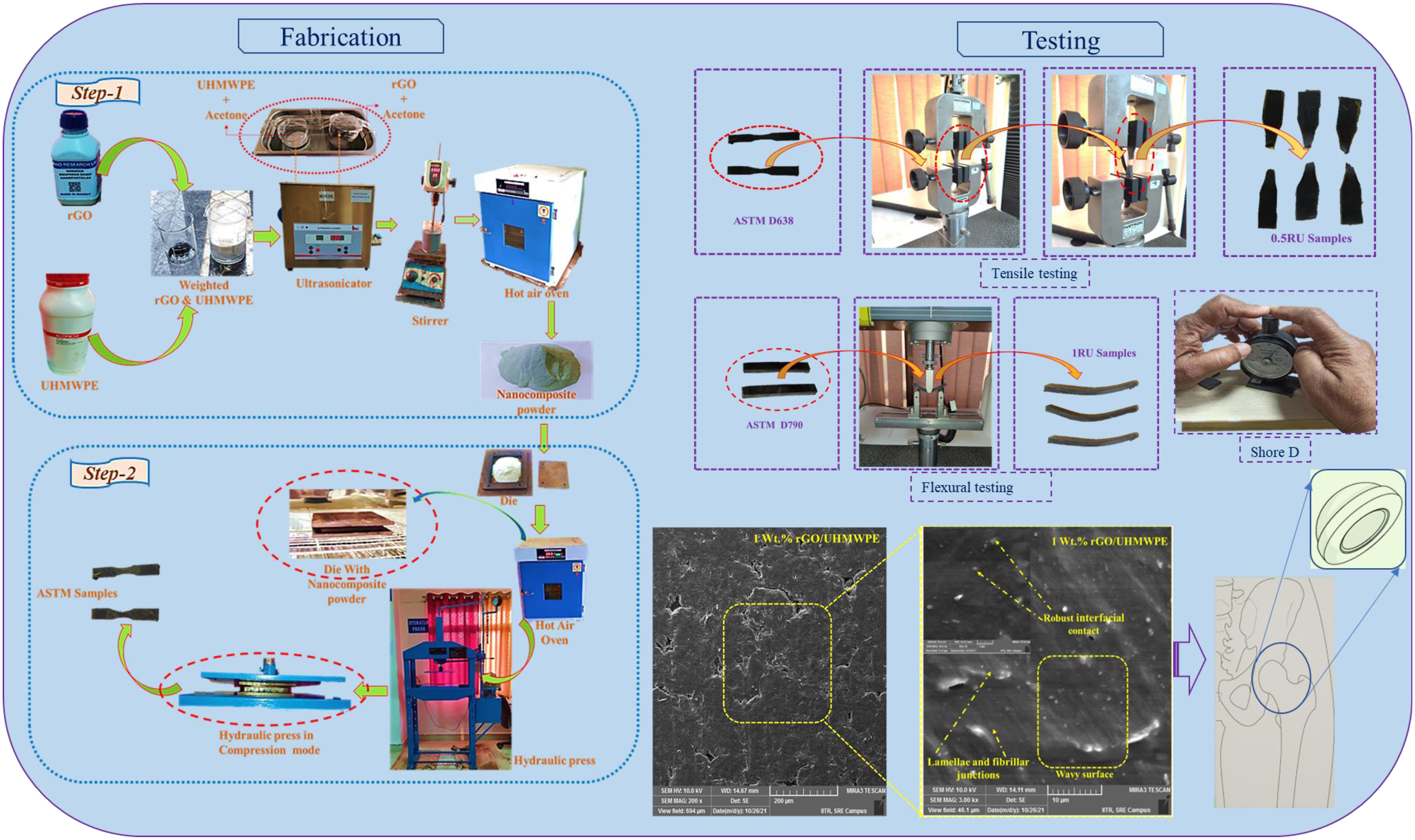
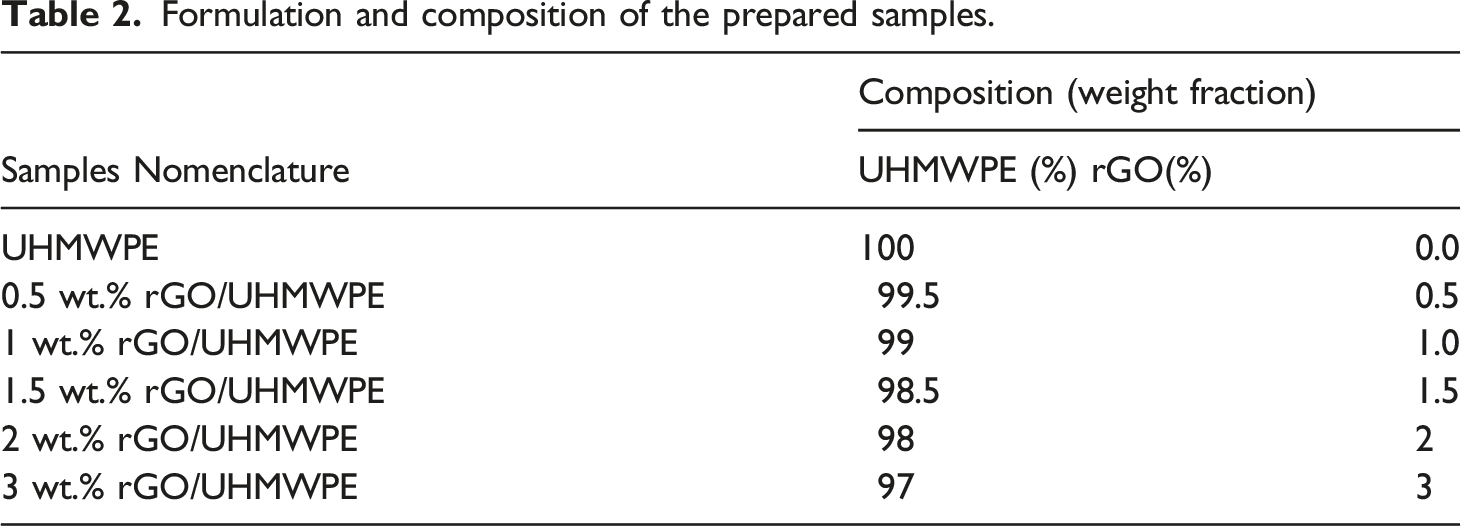
The developed bio nanocomposite was prepared by adopting a two-step process. The first step involves preparing nanocomposite powders using Ultrasonication and stirring inside a liquid media. The whole fabrication process is represented most simply in Figure 2. The reinforcing phase and UHMWPE powder were formerly weighed on a digital weight indicator, according to Table 2, which depicts the sample formulation. To assess rGO incorporation, (0.5, 1, 1.5, 2, and 3) wt.% rGO was introduced to the polymer matrix. Schematic view of the sample fabrication procedure. Formulation and composition of the prepared samples.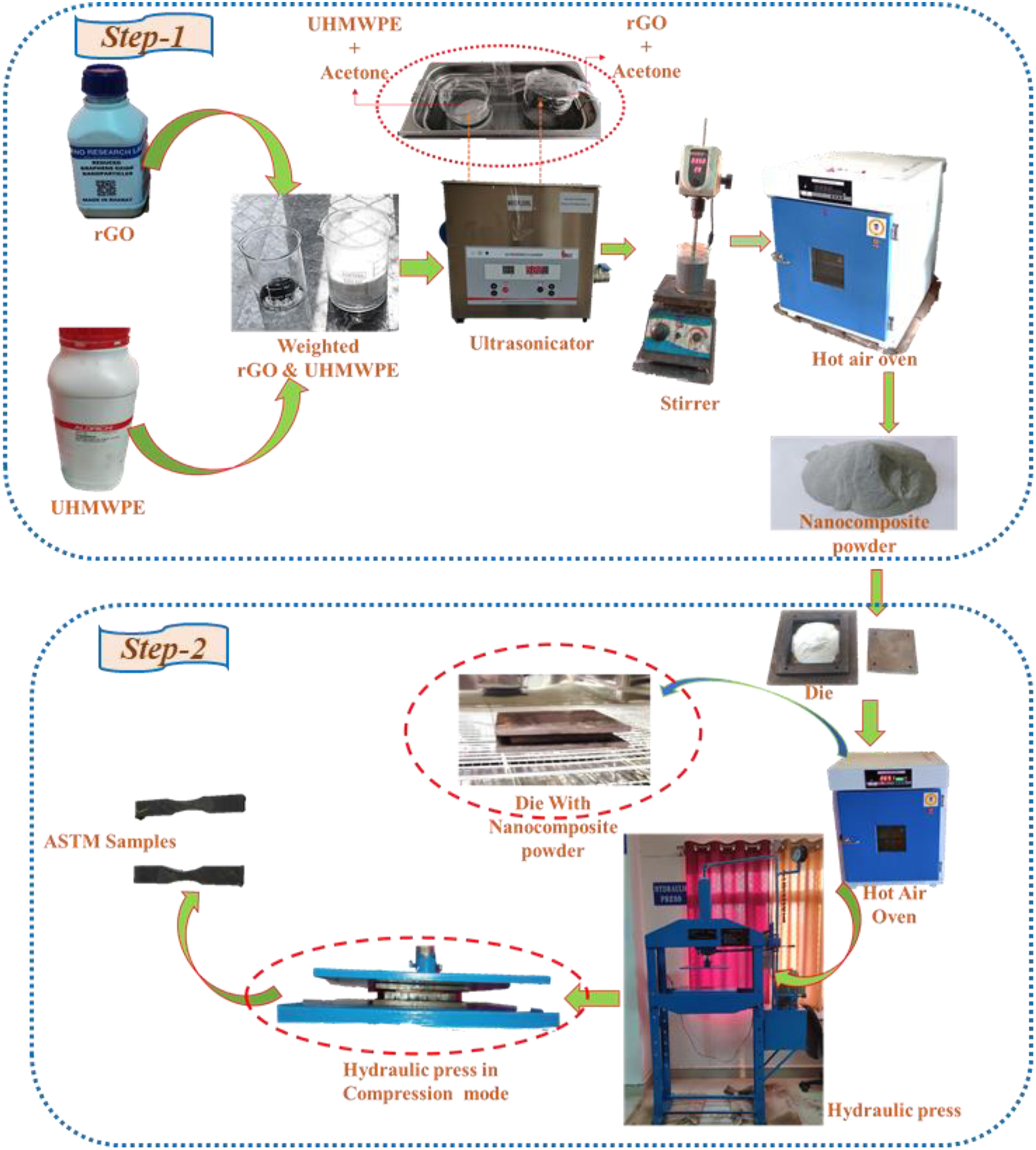
A suspension of rGO nanoparticles was prepared by Ultrasonication for 60 min at 50 W in 30 mL of acetone. A UHMWPE-50 mL acetone combination was then added to the resultant mixture, ultrasonicated for another hour to get the final version. The mixture was then stirred for another 60 min at 75 C to remove its solvent media, whereas the stirring operation avoided any deposition. To achieve effective solvent extraction, the resultant powder was heated in a hot air oven for 50 h at 80°C. Finally, the obtained powder mixture is ball milled for around 2 h to blend and prevent any particle deformation, if any, properly.
The second step involved transferring the final dry powders to the Mild steel die, which were hot-pressed at 210°C with 100 bars of pressure for 1 h. They were then cool to room temperatures to deliver the nanocomposite with desired shape (10x10 cm square blocks of 4 mm thickness). In the whole process, silicon oil is used as a lubricating medium to remove the composite from the die surface easily. At last, the different ASTM Standard samples were cut according to the standard specification. A pristine UHMWPE sample is fabricated using the same two-step process described above to compare with the newly produced nanocomposites.
Characterization of rGO/UHMWPE bio nanocomposite
Mechanical characterization
The developed samples of bio nanocomposite's tensile, flexural, and Shore D hardness tests were determined using ASTM standards. The following sections describe in-depth info about mechanical performances.
Tensile testing
Each batch is made and tested using three Dumbbell form specimens (ASTM D638) with a (t = 3.2 + 0.4 mm, g = 7.62 mm), where (t) represents thickness in mm and (g) stands for gauge length in mm. In order to perform the experiments, we used a Universal Testing Machine (UTM) of the INSTRON® brand (Model 3365) at a 10 mm/min crosshead speed in a uniaxial direction using a load cell of 5-KN. Also, to increase the accuracy of the test conducted, the mean value of results was recorded.
Flexural testing
For each batch, three 80x10x4 mm3 cuboidal shape samples are created and tested in accordance with ASTM D790 standards. At a 2 mm/min speed, a three-point bending method is employed in UTM. In accordance with the aforementioned specification, the span length is adjusted to 16x4 mm, which dictates that the span length must be 16 times the thickness of the sample, i.e. (4 mm). The tensile and flexural testing process can be understood in Figure 3 and Figure 4. Tensile testing of developed bio nanocomposite samples. Flexural testing of developed bio nanocomposite samples.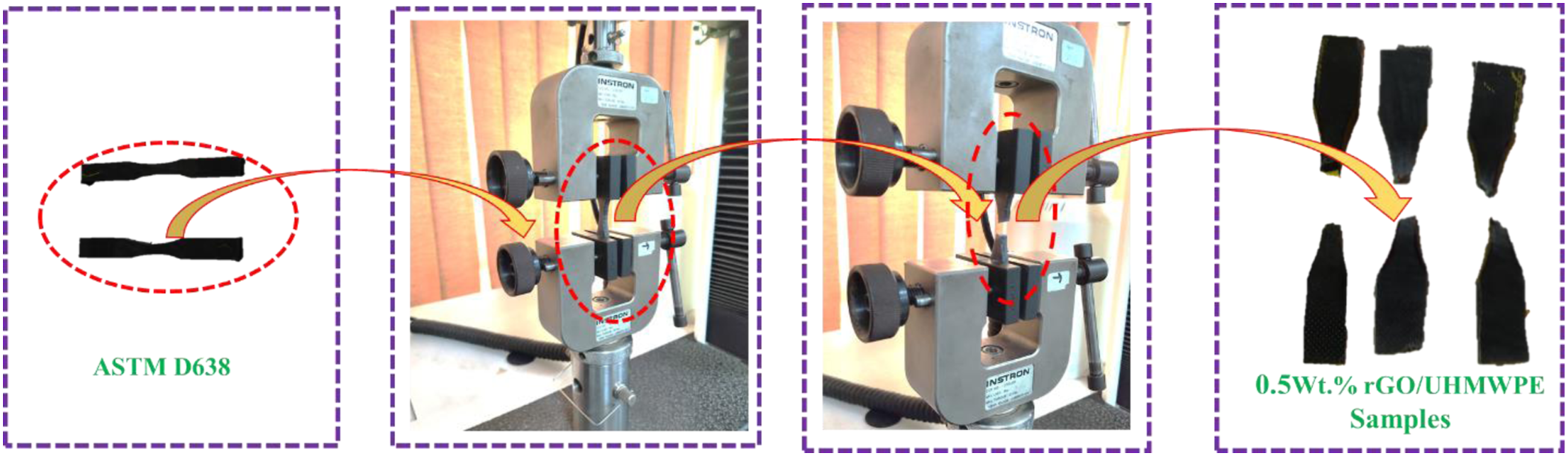
Shore D hardness
Composite samples were tested for hardness using a hardness tester (DUROMETER Shore D- ASTM D2240, MILHARD) with Shore hardness type-D. The given hardness value is an average of nine measurements taken across samples of each kind. In this test, the intender is brought in contact with each piece, and the readings indicated on the dial were recorded.
FESEM analysis
At an accelerating voltage of 5–10 kV, a high vacuum field emission scanning electron microscope (MIRA3 TESCAN, USA) was utilized to analyze the UHMWPE/rGO Bio nanocomposite microstructure. Before the FESEM investigation, all test specimens and the pristine UHMWPE were placed in the machine tray. The polymer surface was conductive and coated with gold-coated using automated sputter coater equipment (Quorum Mini Sputter Coater, SC7620, UK). The samples were then magnified under an electron microscope to evaluate their structure and morphology.
Thermal characterization
Using a Differential Scanning Calorimetry (DSC2-00754) instrument, the melting temperature (Tm), the temperature at which melting begins (Tmi), and the temperature at which melting ends were all determined (Tmf). Also, after sintering/consolidation, the degree of crystallization (Xc) of UHMWPE/rGO Bio nanocomposites was also investigated. Heating each sample to 200°C for 5 min wiped off the thermal history of the produced nanocomposites. The test samples were then heated at 10°C/min from ambient temperature to 200°C. For DSC analysis, a test sample weight of 3.04 mg is employed. The aluminum pan was used during a DSC scan to hold the sample.
FTIR spectroscopy
The chemical and structural changes in the Bio nanocomposite were investigated using Fourier transform infrared spectroscopy (FTIR). The FTIR spectra of UHMWPE/rGO were collected at a wavenumber of 4000 to 400 cm-1 at a resolution of 4 cm-1 with two scans on every sample using a PerkinElmer FTIR Spectrum, which was manufactured in the United States of America. Before analysis, every powder sample was placed in the oven for about 24 h to eliminate any moisture content from the powder. The findings were recorded once the tests were completed.
X-Ray diffraction analysis
The UHMWPE/rGO Bio nanocomposite crystalline structure was determined using X-ray diffraction analysis. In this experiment, an X-ray diffractometer (Rigaku Ultima IV model, Japan) was used, which operated at 40 mA and 40 kV voltage and emitted (Cu K-alpha) radiation with a wavelength of =0.154 nm was used to perform the measurements. The dried samples were placed on a quartz substrate before being placed into the sample holder. The XRD spectra were collected at a 2θ range of 5–80° at a speed of 4°/min, with a step length of 0.02.
Results and discussions
The present work illustrates the mechanical performances such as tensile, flexural and hardness, thermal analysis (DSC) and morphological investigation of developed bio-nano composite, including the fractography study of tensile samples. These properties are highly desired for biomaterials application, specifically in the development of orthopedic components. The findings of mechanical testing of rGO modified UHMWPE are described in this section. Figure 5(a) demonstrates tensile stress-strain curves of the developed samples. Elastic modulus, yield strength, tensile strength, and the elongation at break calculated from the plots are also compared. Mechanical characteristics: (a) Tensile; (b) Flexural properties.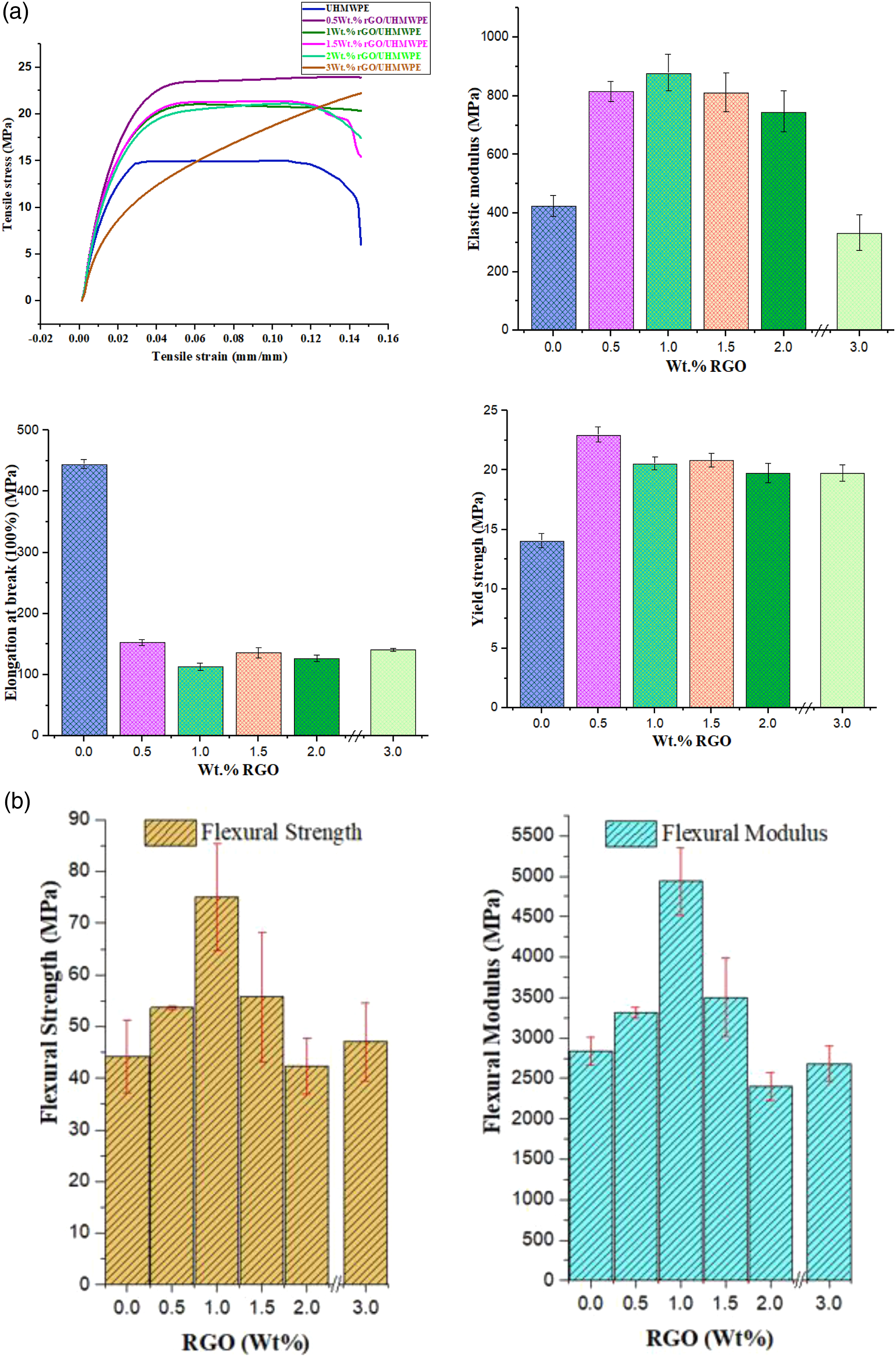
The results of mechanical testing of the developed samples.
Shore D hardness
The fluctuation of hardness values with increasing rGO content into the UHMWPE polymer matrix can be seen in Figure 6 and Table 4. Variations of hardness as a function of rGO addition. ShoreD hardness variations as a function of rGO addition.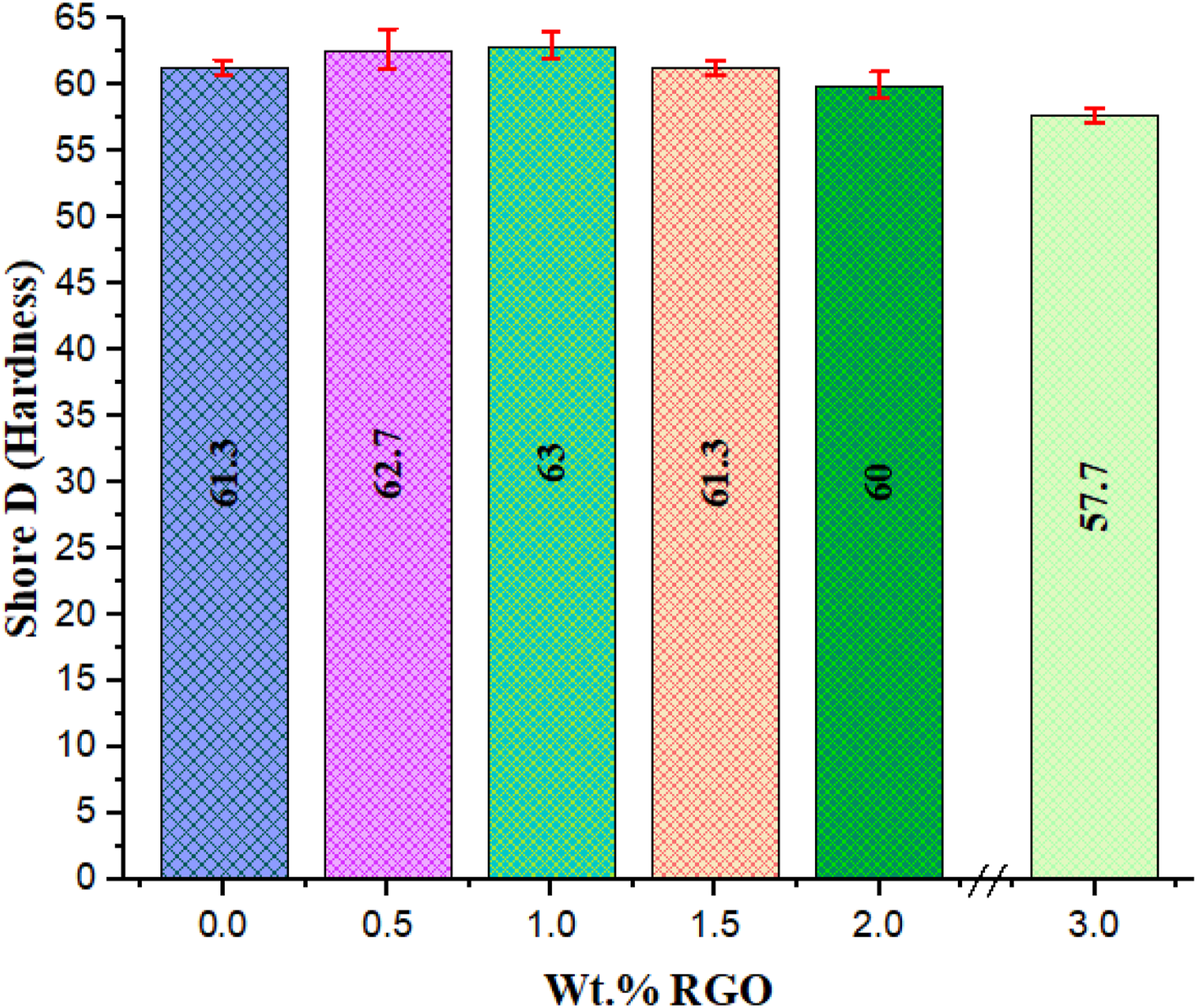
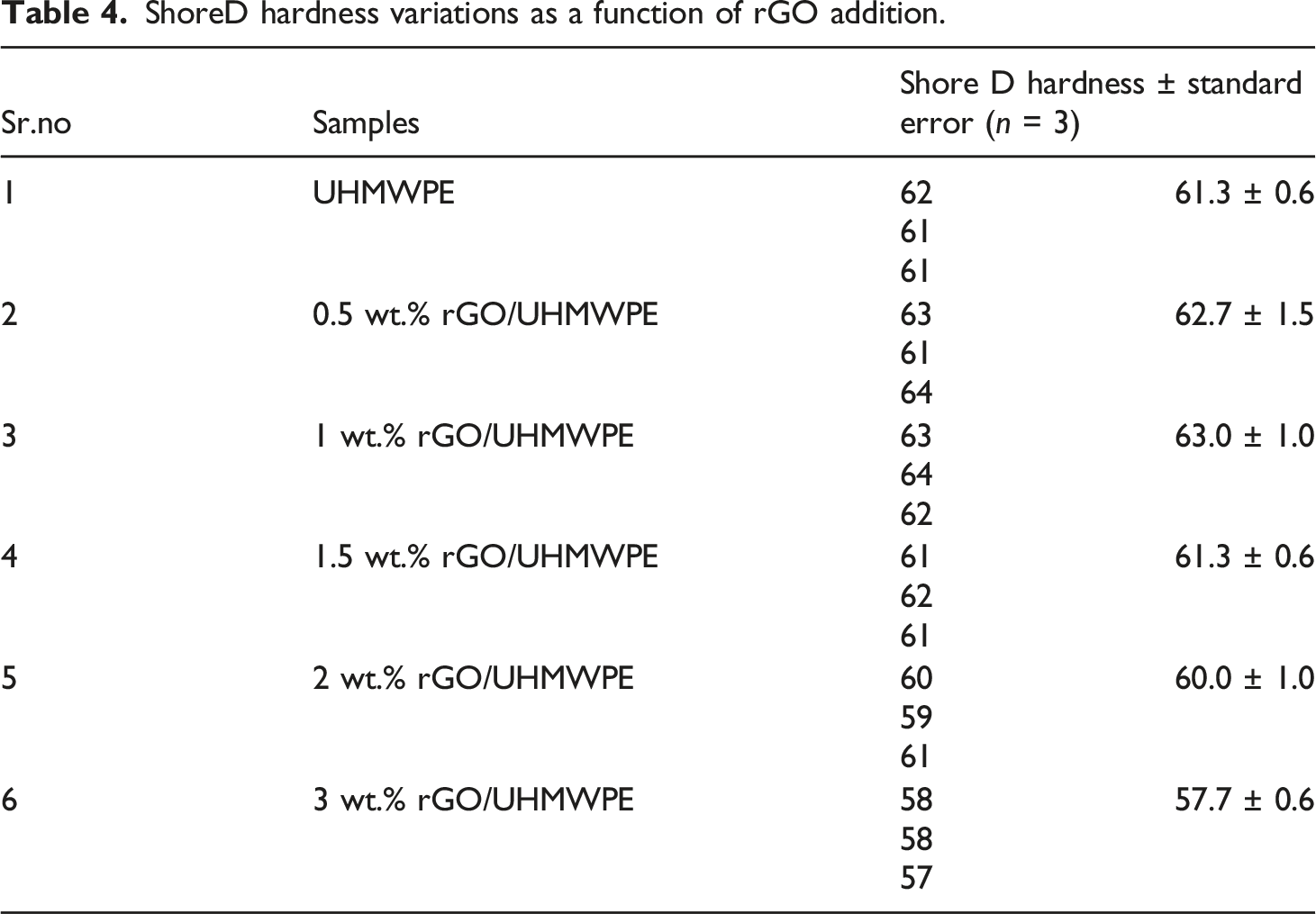
For lower wt. % values of filler incorporation, the hardness of the rGO-filled nanocomposites grew gradually with increasing rGO content. With 1.0 wt.% of rGO content in UHMWPE, the microhardness value of the material raised by around 2.8%, from 61.3 to 63.0. Thus, it could be claimed that small amounts of rGO might significantly increase the hardness of UHMWPE. This was attributed to rGO's outstanding mechanical qualities, which allowed it to sustain partial loads Archard's prediction and were critical for transferring load. According to, 59 increasing the hardness of UHMWPE or its composite would minimize the plastic contact regions between the plastic and the metal. As a result, we expect that UHMWPE-rGO composites containing coordination metal material will have superior friction performance when compared to unfilled UHMWPE. At last, EDS and XRD results show that rGO dispersion within the polymer matrix might improve indentation resistance. 53
There was a drop in hardness as the graphite concentration increased (>1 to 3 wt. %) owing to the decrease in the UHMWPE matrix used as the binding phase. A similar pattern was observed when graphite incorporated UHMWPE composite was tested from 0 to 30 wt. %. 69
The effect of rGO on polymeric structure
The processing parameters and rGO wt. %. presents directly impact the structure and morphology of the polymer matrix. The dispersion of rGO particles inside the polymer matrix is shown by correlating SEM Micrographs of rGO with various produced bio nanocomposites. Figure 7 and Figure 8 showed the SEM images of multi-layered reduced graphite oxide and the surfaces of rGO/UHMWPE bio nanocomposites with different content of rGO. Also, it has been observed that no voids and the wavy surface are observed with unfilled UHMWPE (Figure 9). It can be noted that the surface of rGO-0.5 wt. % and rGO- 1 wt.% nanocomposite was primarily flat, but it also had areas that appeared like wavy riverbed patterns. The bio nanocomposite micrographs further demonstrate that rGO was fully submerged in the UHMWPE matrix, showing that rGO and the matrix exhibit robust interfacial contact.
70
SEM images of reduced Graphene oxide nanoparticles. Scanning electron microscopy image of bio nanocomposites: (a) Nodule area with 0.5 wt. % rGO content; (b) (c) The lamellae and fibrillar junctions for rGO levels of 1 and 1.5 wt. %; (d) (e) for 2&3 wt. % rGO content with agglomeration zone. Scanning electron microscopy image of UHMWPE (pristine): illustrate basic pristine UHMWPE structure, i.e., lamellae and fibrillar junctions.


In the UHMWPE matrix, the rGO nanofillers are well distributed due to the absence of rGO clusters. The nanofillers are well distributed, as shown by the polymer matrix's rise in the nanofiller percentage. Furthermore, the surface morphology changes dramatically as the quantity of rGO grows above 1 wt.%. The morphology of the developed bio-nano composite appeared visibly rough and distorted in appearance. Consequently, the elevated amount of rGO has hampered polymer rearrangement and chain folding. The clusters get more prominent as the rGO concentration rises from 1.5, 2, and 3 wt. %. As a result, the approach used to create the UHMWPE nanocomposite is a practical methodology for achieving an appropriate dispersion of rGO. After the Ultrasonication of rGO, the reduced Graphene oxide is instantly incorporated with the UHMWPE powder using high-speed stirring. This phase is critical because it encourages the rGO layers on UHMWPE particles to be covered. The rGO layers may be dispersed by Ultrasonication; however, when the solvent is evaporated, the layers (rGO) which were not adequately deposited on the polymer surface prefers to become aggregated at different sites forming agglomeration. As a result, the powder blend was pulverized effectively for 120 min using a ball milling method to blend the nanocomposite powder well.
The fracture morphology of the tensile samples was examined using a scanning electron microscope with a 5 kV accelerating voltage (Figure 10 and Figure 11). The green colour marks represent an improvement in ductility (rGO filled) compared to the pristine sample (indicated by red marks). The dog-bone pieces were sliced parallel to the fracture surface 1–3 mm underneath it. Before SEM examination, each sample was gold plated to make it conductive. Observations during the tensile test revealed significant elongation before fracture development, a typical example of ductile failure. Before the specimen broke, it developed a fracture on its surface. The fracture surface micrographs exhibit ductile failure characteristics such as plastic deformation and fibrils on the surface caused by particle separation during the tensile test.71–73 Crack initiation is often followed by crack propagation, in which more fibrils experience severe plastic deformation until they burst. Figure 10 shows fewer fibrils than 1wt%. rGO content bio nanocomposite demonstrating lesser ductility. Also, no voids in the fracture surface reveal excellent interaction between filler and matrix components. An acute sharp fracture is seen with UHMWPE (pristine) in comparison to 1Wt.% rGO/UHMWPE nanocomposites. SEM Micrographs of UHMWPE bio nanocomposites: fractured (pristine UHMWPE) tensile dog bone sample. SEM Micrographs of UHMWPE bio nanocomposites: Fibril’s pull out in a fractured tensile dog bone sample with 1wt. % rGO concentration. 1 Wt.% rGO/UHMWPE fracture surfaces indicate excellent rGO-UHMWPE matrix bonding.

EDS analysis's elemental mapping of bio nanocomposites demonstrates that oxygen was evenly distributed throughout the bio-nano composites (Figure 12(a) and (b)). The oxygen dispersion is vital since the filler, i.e., rGO, contains namely rGO, comprises functional groups that contain oxygen. Results from the EDS were compared to those from the pristine composite and the Element overlay for 1wt. % rGO Content bio nanocomposite reveals the homogeneous distribution of rGO in UHMWPE. As shown in eZAF Smart Quant Results and Sum Spectrum of EDAX analysis, the increase in oxygen suggests that rGO is well dispersed within the developed composite. The oxygen level also increases due to the presence of an oxygen-containing group rGO. EDX spectrum of (a) Pristine UHMWPE and (b) Developed bio nanocomposite with 1wt.% rGO content.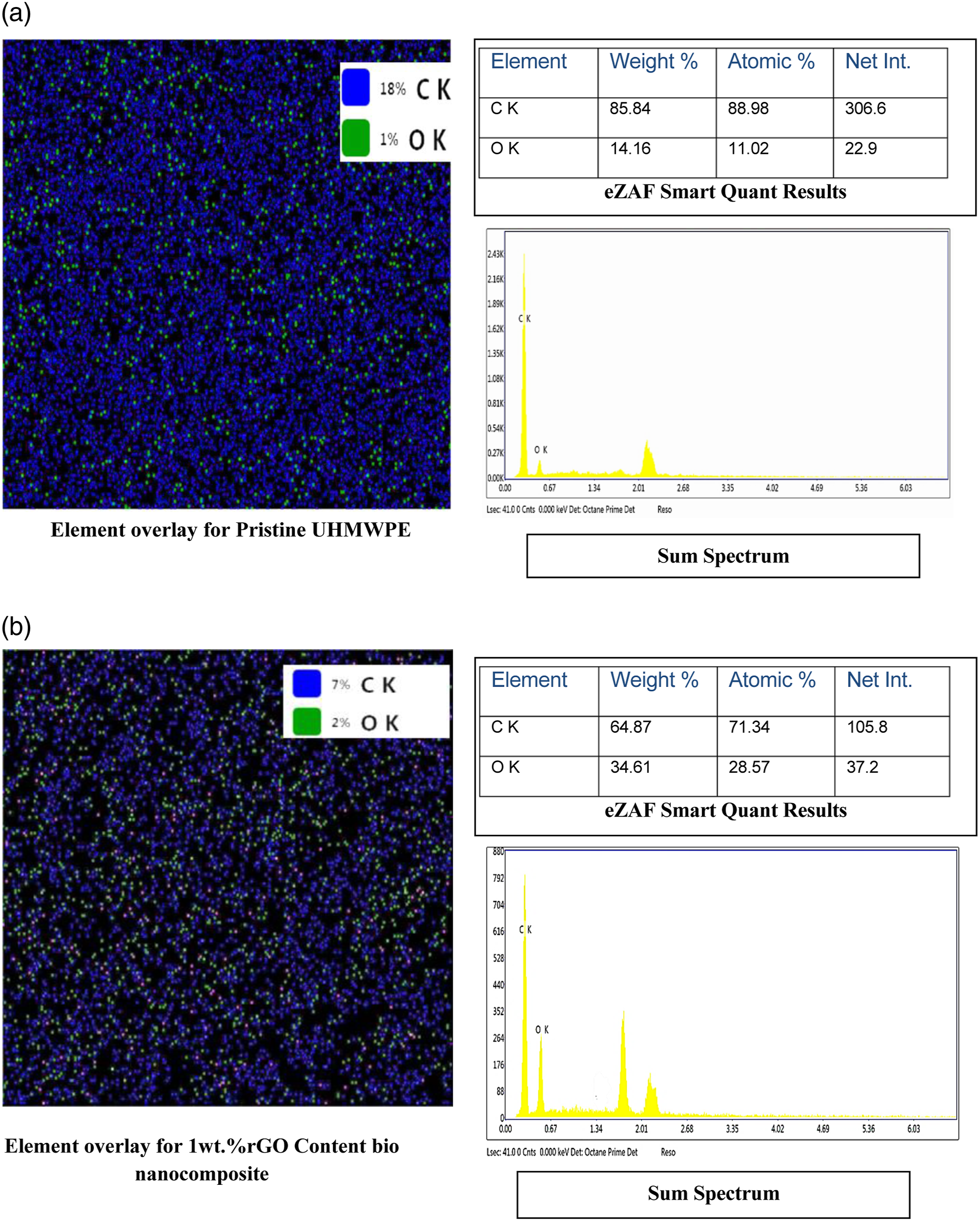
DSC analysis
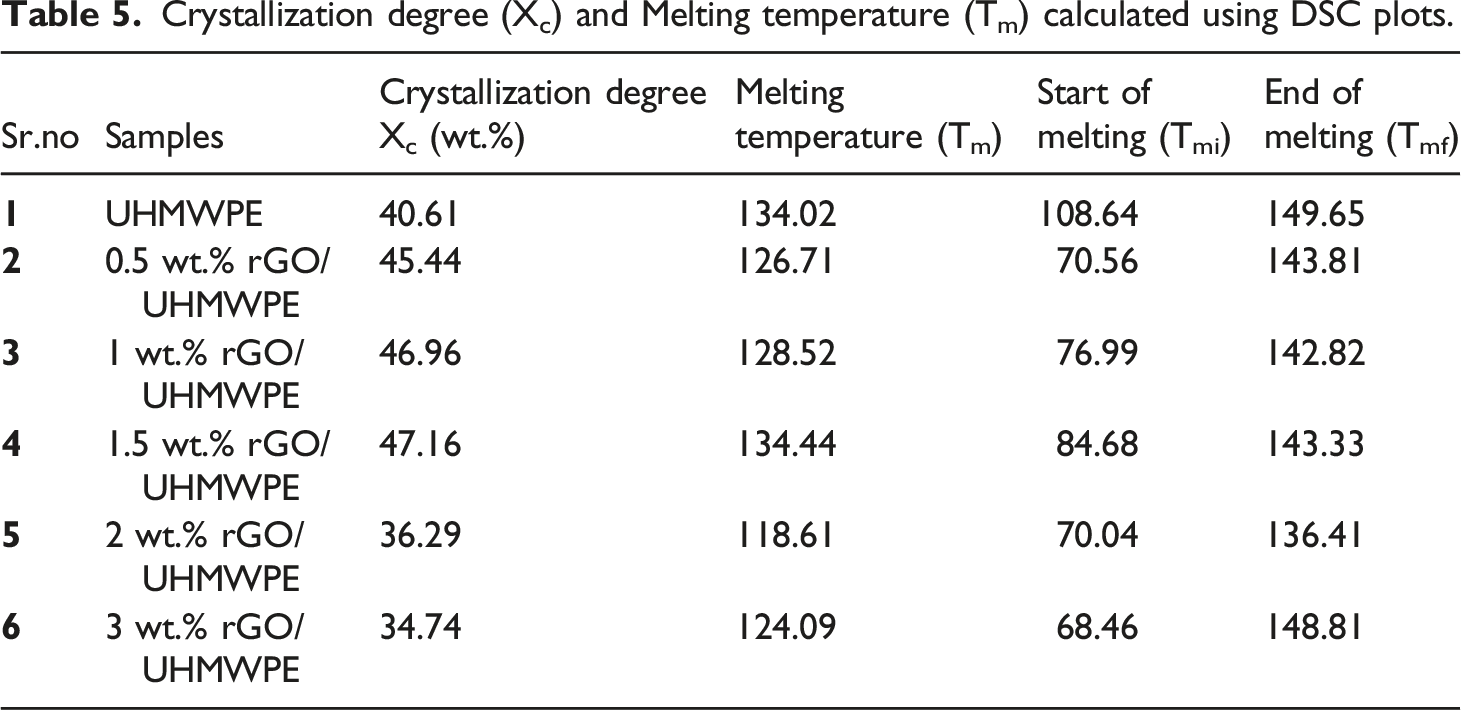
DSC analysis was used to determine microstructural changes after adding reinforcements to the matrix, and the melting temperature (Tm) and degree of crystallization (Xc) were measured. DSC endotherm graph (Figure 13) was used to compute the melting temperature (peak temperature in the DSC), and crystallinity was estimated using equation (1) Graphs showing endothermic DSC plots of samples for pristine UHMWPE and UHMWPE/rGO series composites.
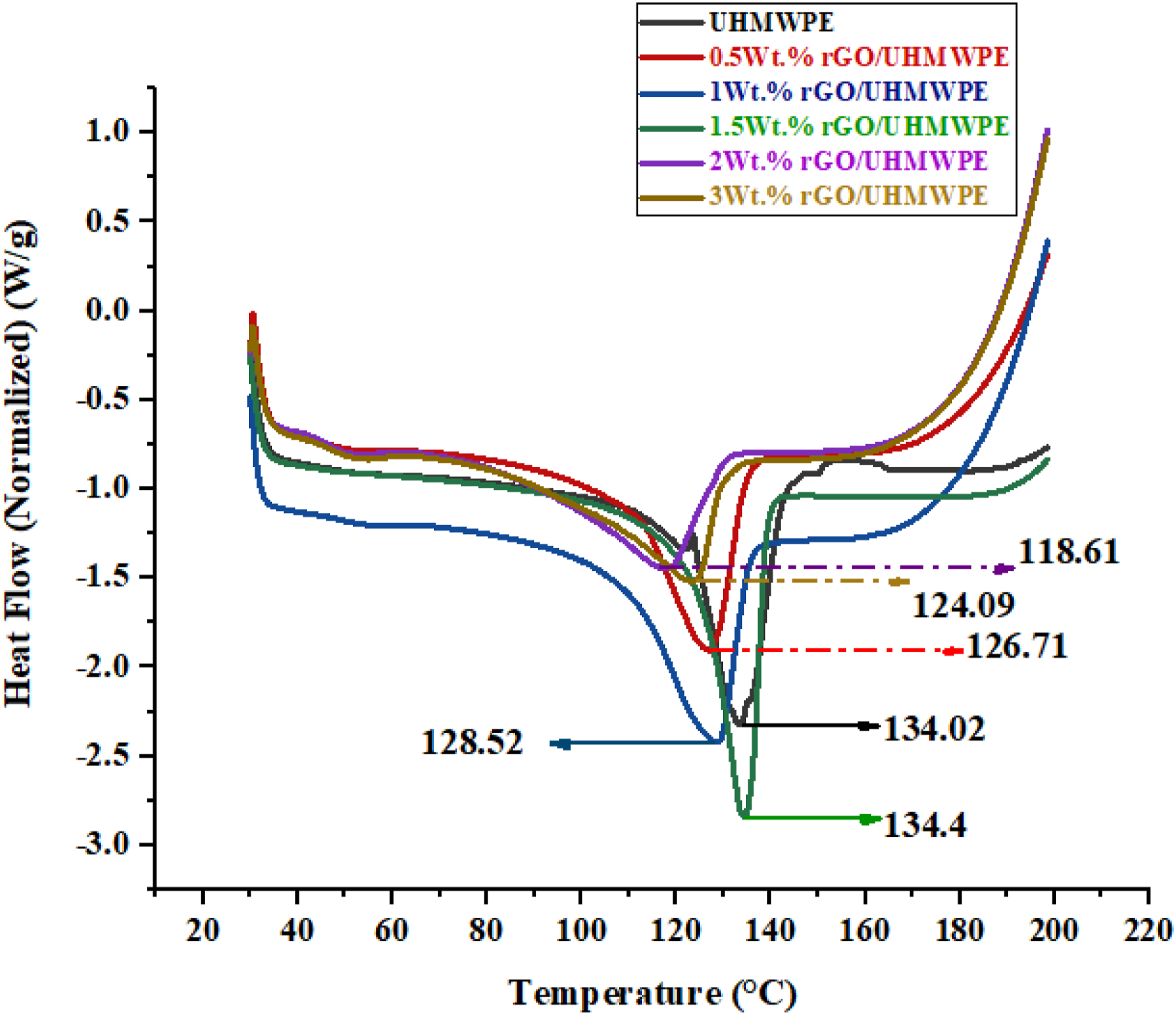
Crystallization degree (Xc) and Melting temperature (Tm) calculated using DSC plots.
Fourier-transform infrared spectroscopy
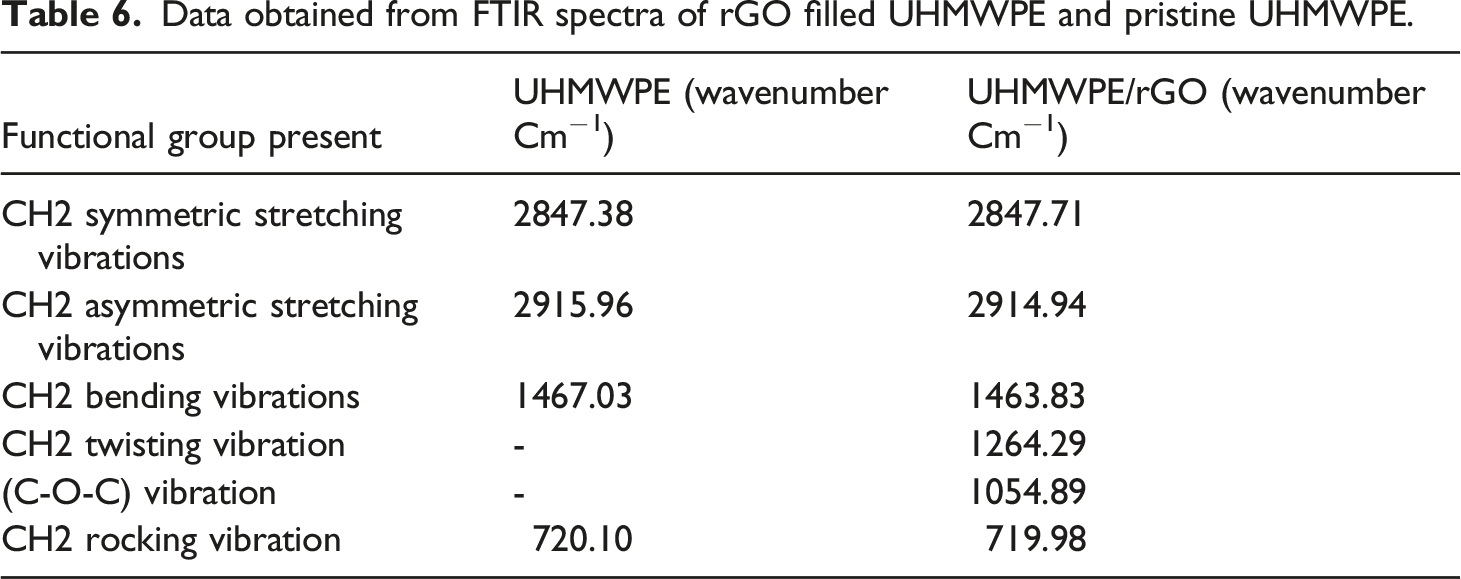
UHMWPE/rGO bio nanocomposite FTIR spectra were shown in Figure 14 and Table 6 for various groups, respectively. When applied to the unfilled UHMWPE, the CH asymmetric vibration, the CH symmetric vibration, the CH2 bending vibration, and the CH2 rocking vibration all have peaks of 2915.96 cm−1, 2847.38 cm−1, 1467.03 cm−1, 720.10 cm−1 respectively.
78
Since the infrared absorption peak position of rGO/UHMWPE composites does not shift with increasing rGO content. This indicates that rGO did not significantly affect the molecular structure of UHMWPE during the ultrasonic dispersion and hot-pressing process and that UHMWPE did not degrade during either step. Apart from that, the impact of CH2 twisting vibration (1264.29 cm−1) as well as the effect of (C-O-C) vibration 1054.89 cm−1 were statistically significant when rGO was present in a polymer matrix.
79
Because of the presence of rGO, the intensity of the peaks at 22915.96 cm−1, 2847.38 cm−1, 1467.03 cm−1, 720.10 cm−1,1260.15 cm−1, and 1054.79 cm−1 increased, perhaps indicating stronger interaction between filler and the Polymeric matrix. FTIR spectral analysis of UHMWPE (pristine) and UHMWPE/rGO bio-Nanocomposites. Data obtained from FTIR spectra of rGO filled UHMWPE and pristine UHMWPE.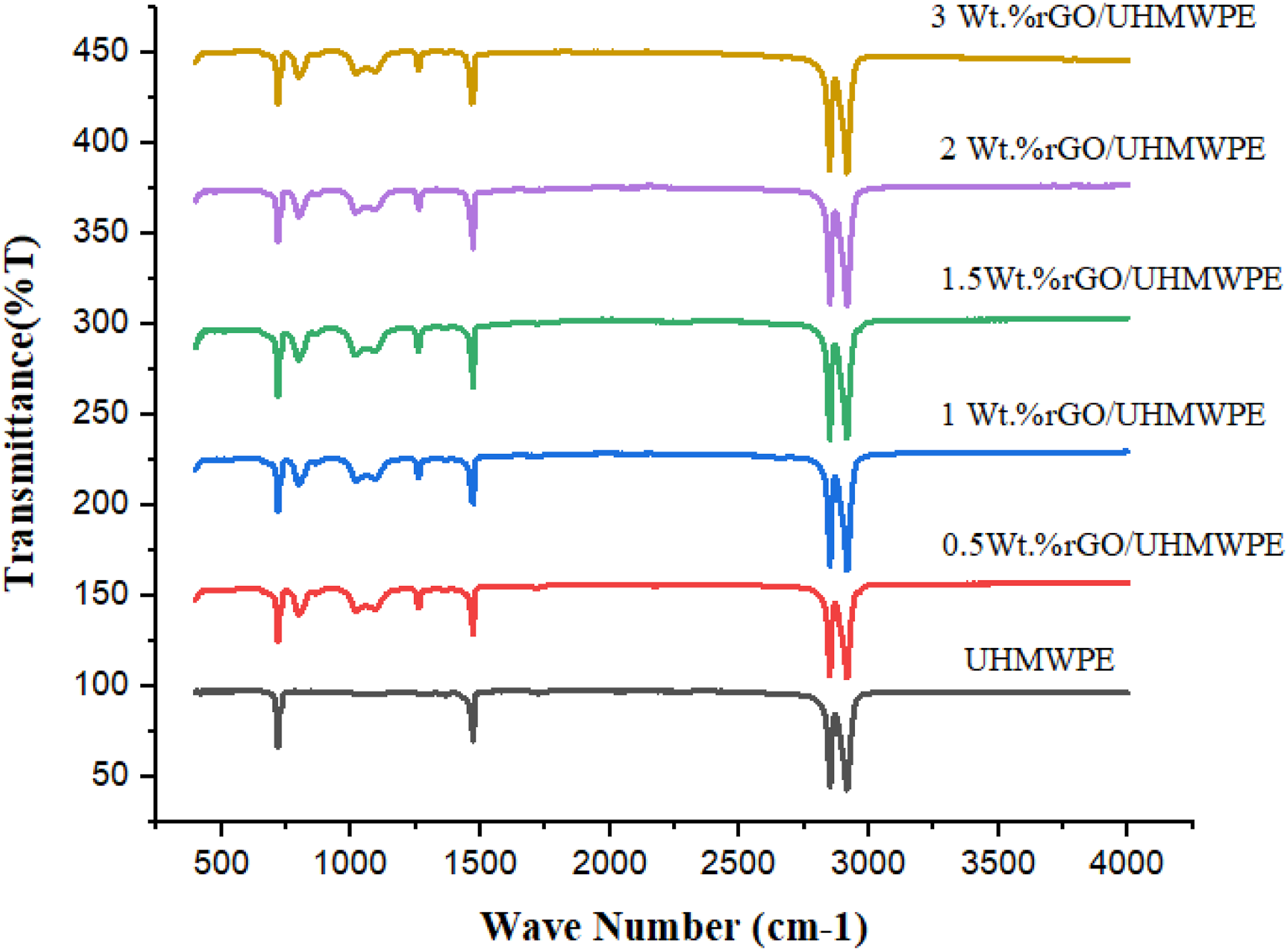
XRD analysis
Unfilled and rGO filled bio nanocomposites XRD diffraction patterns are depicted in Figure 15. Diffractograms of the orthorhombic crystal show two peaks at 2θ° = 21.46 and 23.84, which correspond to the (110) and (200) planes, respectively.
80
From XRD analysis it is clear that with the increase of rGO content up to 1.5wt.% the crystallinity shows increasing trends and then declines with further increase of rGO content. This result is similar to the study conducted by Li.et al. They utilized solution mixing to develop different carbon materials (Graphite particle (GP), Graphite nanoplatelets (GNP), and flaky Graphene oxide (GO) as filler, based nanocomposites. According to the results, heterogeneous nucleation and the effect of carbon materials that restrict crystal development are present throughout the UHMWPE crystallization process. Both of these factors work together to prevent crystal growth. The macromolecular chain motion of UHMWPE is slowed down because of the heterogeneous nucleation of carbon materials. This is the reason why crystallinity increases at lower wt.% of filler than at higher wt.% i.e. above 1.5 to 3wt.%.
81
Another study by Çolak.et al. also shows the same trend. The results of XRD analysis show an increase in crystallinity at lower content of reduced Graphene oxide (rGO), which further shows a decline at higher wt.% of rGO filler. Since, the low RGO content was able to operate as nucleation sites rather than causing disruptive rearrangement and chain folding throughout the crystallization process.
82
The nanoconfinement impact of Graphene nanoplatelets on the characteristics of matrix-grafted Graphene/polysulfide polymer nanocomposites is explored in the study conducted by Amangah et al., the results of DSC and XRD demonstrated that the addition of modified Graphene (mGO) up to 0.7 wt.% boosts the crystallinity of nanocomposites, but that the crystallinity diminishes beyond that. This shows that the crystallinity of the sample grows with the amount of mGO added, since mGO functions as a nucleating agent during the crystallization process. With the increase in filler loading, nanoconfinement effect of Graphene nanoplatelets came into play, resulting in a decrease of crystallinity.
83
With the incorporation of 3wt.%, a decline in intensities of both peaks as in the UHMWPE XRD diffraction pattern, was observed. It can be correlated with crystallinity alteration in the polymer matrix.
75
There will be less crystallinity and more disruption during the crystallization process due to the fillers' high rGO content (3 wt. %), making them ineffective as nucleation centers and causing chain folding and rearrangement disruptions. UHMWPE/rGO bio nanocomposite crystallite size data for 2θ = 23.84° is shown in Figure 15. Increasing the size of the crystalline structure in the UHMWPE/rGO bio nanocomposite will change the wear behaviour of the developed composite.
84
The XRD patterns of nanocomposites also did not show any unique diffraction peak, with the exception of orthorhombic crystal peaks of empty polymer, which were detected in the patterns of pristine polymeric material. That rGO had been exfoliated in the UHMWPE matrix was evident from this finding.
23
XRD plot for developed nanocomposites.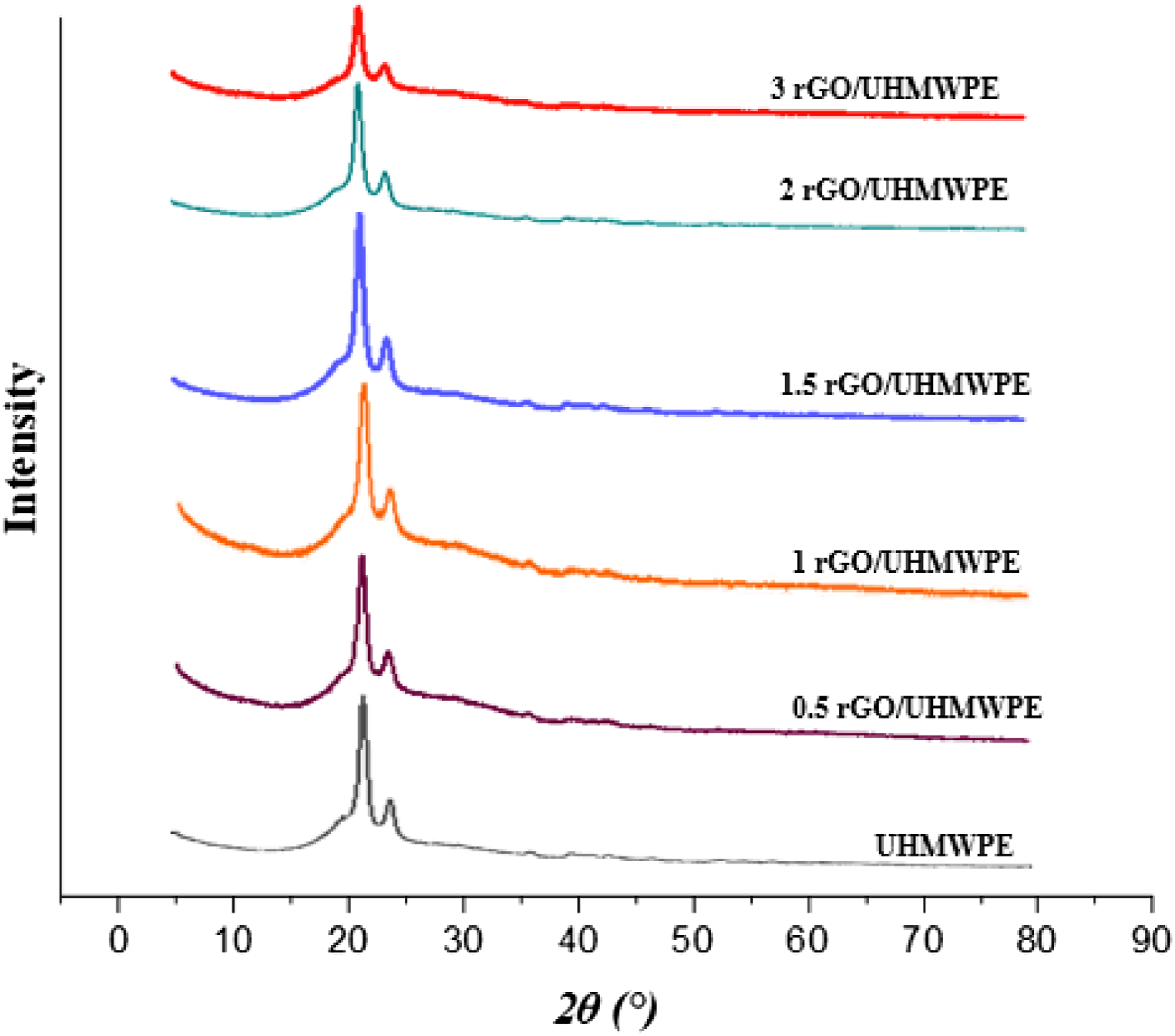
Conclusion
This paper presents an effort to apply rGO reinforced polymeric biomaterials to different types of contacting surfaces and joints in the human body. Due to various factors, including obesity, medical issues, accidents, sports activities, an unhealthy diet, and the natural deterioration of joints over time, surfaces and joints are subjected to a variety of loading conditions during the course of daily activities. These loading conditions change throughout an individual's life due to various factors, including age, gender, and physical condition. Green-produced reduced Graphene oxide was employed as reinforcement in UHMWPE. The bio nanocomposites were created using liquid-phase Ultrasonication, ball mill pulverization, and hot pressing in a hydraulic system. The study illustrates the effect of rGO-modified UHMWPE and obtained promising results. Based on the acquired findings, the following conclusions were drawn: • A series of Ultrasonication processes evenly disseminate the rGO sheets in the polymer matrix. Compared to other fillers cited in the literature, such as CNTs, MWCNTs, and more expensive nanofillers, rGO could improve UHMWPE's mechanical and thermal properties with only a small amount of filler content. • Increasing rGO concentration improved UHMWPE crystallinity. rGO improved UHMWPE/rGO bio nanocomposite's thermal performance. High-rGO content greater than 1wt.% shows a decline in crystallinity. • It was found that the rGO concentration of 0.5 wt.% and 1 wt.% gave the best tensile strength results. A good flexural response is also seen with 1wt.% of rGO Content. Furthermore, even small Wt.% of rGO can significantly boost the micro-hardness. There was a significant interaction response between the rGO lamellar structure and the UHMWPE matrix, resulting in better load transfer performance. • SEM and EDAX analysis show the good dispersion of rGO nanofillers for 1Wt.% sample. Well-coated nanofillers with polymer matrix are observed in the SEM image. Also, the EDAX shows the uniform distribution of rGO throughout the polymer matrix. Investigating the fractography of tensile (Dog-Bone) test specimens of rGO/UHMWPE Nanocomposite shows ductile fracture properties. It could be possible due to the lubricating effect of rGO. • The XRD diffraction pattern and FTIR demonstrate the excellent interaction between the polymer (UHMWPE) and the nanofiller (rGO). • The two most important factors affecting the success of UHMWPE/rGO composites were the free-space effect and the interaction-force effect. In thermal characteristics, these two elements are mutually offsite. In terms of mechanical capabilities, these two features mutually reinforce one other. As a result of these mechanical and thermal qualities, it could be inferred that rGO mixed UHMWPE is a potential material for the future of biomaterial implants due to its low cost and excellent thermal and mechanical properties.
This research demonstrates the effectiveness of rGO-reinforced nanocomposites and their potential for use in developing orthopedic components. It may be utilized as a linear acetabular material; however, it can also be used in knee, shoulder, and finger implants, biomedical sensors, thermal barriers, and various medical devices. This investigation was confined to mechanical tests such as tensile, flexural and Shore D hardness and some advanced characterization that yielded positive results as a preliminary screening on rGO-filled bio-nanocomposites. On the other hand, the shape and distribution of wear particles may have a considerable influence on the in-vivo performance of the material. Recent nanocomposite research has suggested that using smaller filler particles might provide more impressive results with lower filler concentrations and increased capabilities. This may be accomplished by dispersing the nano-fillers with other ceramic nano-fillers to offer high strength and lubricating effect while maintaining the requisite optimal qualities. Further research is needed to study the composite's viscoelastic behaviors, tribological characteristics, and toughness. These qualities might be investigated in the future to better understand the suggested nanocomposites.
Footnotes
Acknowledgment
The authors would like to acknowledge the kind support of the laboratories facilities available at Central Institute of Petrochemicals Engineering & Technology (CIPET), Lucknow and Indian Institute of Technology (IIT), Roorkee (Saharanpur Campus), India.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
