Abstract
Present investigation elucidates the effect of surface treatment on the mechanical response of carbon fiber (CF)/polyimide (PI) composite loaded with a range of graphene oxide (GO) content. Incorporation of 0.5 wt% GO in CF/PI exhibited 21.1% improvement in flexural strength. The state of interface and its impact on the tensile behavior at various testing temperatures have been discussed. Scanning electron microscope (SEM) analysis of fractured surfaces has been carried out to comprehend various interfacial strengthening and failure micromechanisms. Surface-treated CFs were incorporated into PI matrix to fabricate a high-performance multiscale composite. To improve the stress transfer between PI and CFs, GO was also grown on fibers to produce GO-grown short CFs. The synergic reinforcing effects of combined fillers were analyzed on the fracture surface of composites through SEM.
Introduction
Carbon fibers (CFs)-reinforced polymer composites with their favorable stiffness-to-weight and strength-to-weight ratios are displacing traditional metal materials in a great many of high-performance structural applications, such as aerospace and automotive industries. 1 –4 However, the quality of fiber–matrix interface makes a critical contribution to the performance of composites. 5 –7 A good quality interface is essential to ensure efficient load transfer from the hot matrix to the reinforcement, which helps to reduce stress concentrations and increase bulk mechanical performance. However, applications of CFs-reinforced polymer composites are restricted owing to the poor interfacial adhesion between CFs and matrix resin, which is attributed to the nonpolar, low surface energy and chemically inert surface of CFs. 8,9 Fiber-reinforced polymer composites exhibit strength properties that make them a favored class of materials for a wide range of applications where the reinforced fiber composites fail to deliver the desired results. Another special feature of such reinforcement is their ability to drape or conform to curved surfaces without wrinkling. By combinations of different fibers in weft and warp or employing different weaves, composites of tailored properties can be fabricated. 1 –3 CF-reinforced composites are used in aircraft industries since more than five decades, and this was initiated from trim tabs, rudders, spoilers, doors, and so on. 4,5 CF is very much preferred as reinforcement due to very important properties such as high specific strength in two directions, thermal conductivity, thermal and thermo-oxidative resistance, low expansion coefficient, self-lubricity, and so on. However, CFs are chemically inert in nature and hence have poor wettability and adsorption with most of the polymer matrices which results in weak interface. 6,7 The interface between fiber and matrix is an important aspect in controlling the overall properties of the composite. A strong interface increases the structural stability of the composites and transfers the stress efficiently from matrix to fiber. For enhancing fiber–matrix interfacial adhesion, chemical bonding and mechanical interlocking are the most effective ways. Various surface treatment methods such as chemical, electrochemical, thermal, cold plasma, rare earth solution, and radiation have been employed so far to improve the adhesion between the CFs and various matrices. Significant efforts are currently devoted to the development of nanoparticle/polymer composite motivated by the processing of individual particles into a solid material for device integration or by the design and control of polymer properties. 8,9 Improvements of thermal, mechanical, or electromagnetic behavior among others have given rise to interesting studies. 10 Only a few weight percent of inorganic particles dispersed in the polymer matrix are necessary to observe a modification of mechanical properties. 11 On the contrary, polymers can be added in small quantities to inorganic powders in order to extend the use of inorganic materials, as in the case of microwave absorbents. 12
Experimental
Materials
Polyimide (PI) YS-20 grade powder (Shanghai Synthetic Resin Institute, China) of particles size of 75 mm and specific density of 1.4 g/cm3 was used for composites fabrication by means of a hot press molding technique, which is the most common technique for the sintering of PI composites.
Surface treatment
Reagents, including anhydrous alcohol, tetrabutyl titanate, triethanolamine, triethylene tetramine, and ethyl silicate, were purchased from Shanghai Linfeng chemical reagent company (Shanghai,China).
Tetrabutyl titanate and triethanolamine were first put into a four-necked flask and mixed at 60°C for 30 min. Then, anhydrous alcohol was added and reacted for 4 h. At the same time, the mixed solution of anhydrous alcohol and deionized water was slowly dropped. After adjusting pH value to 8–9.
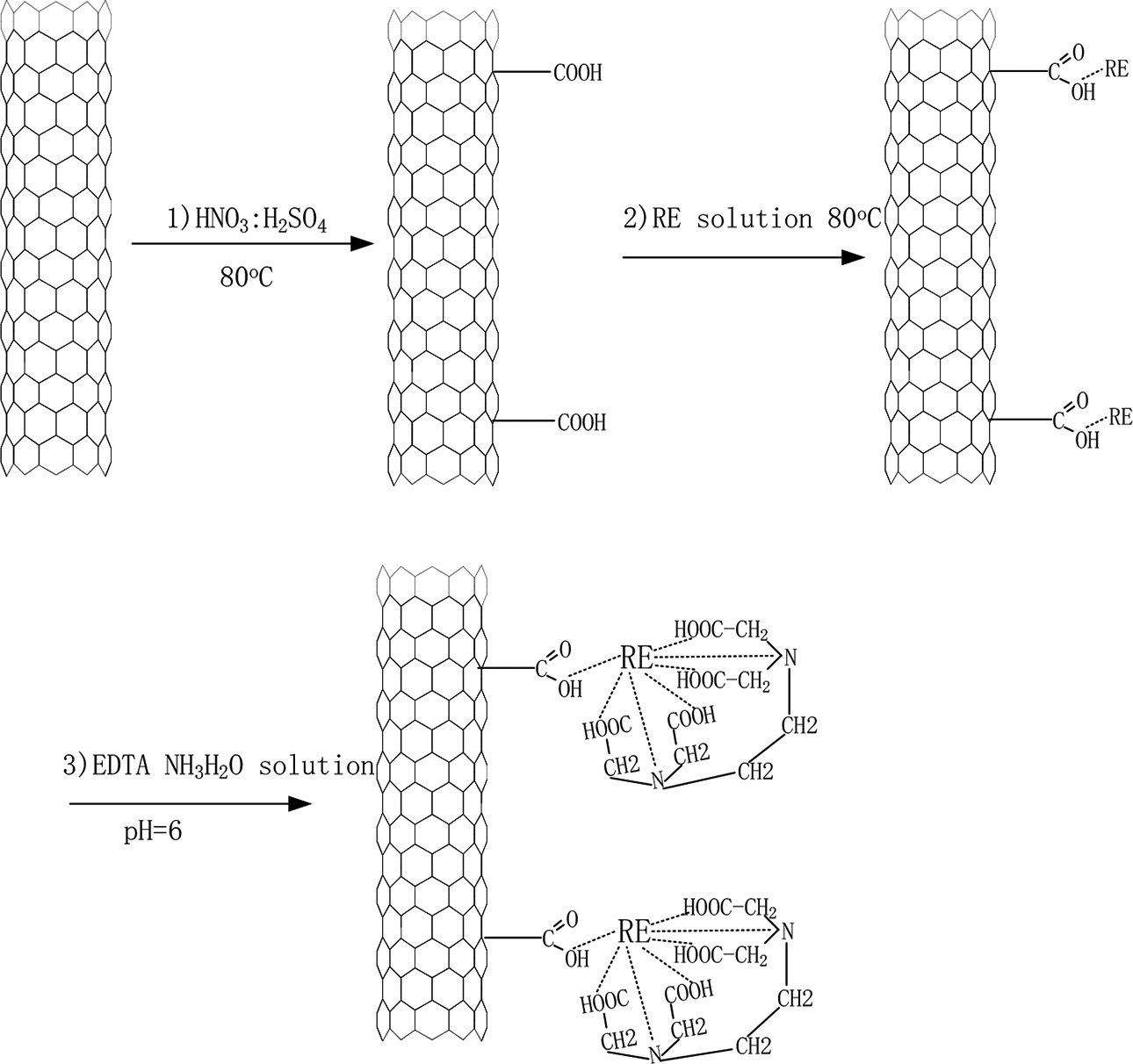
CFs were dried at 100°C in vacuum for 24 h until reaching a constant weight then immersed in sodium hydroxide (NaOH) solutions at 2%, 5%, and 10%, for 24 h at room temperature.
Graphene oxide (GO) aqueous dispersion was diluted to 0.25 g/L with deionized water. Subsequently, the solution was adding a 0.1 M NaOH solution until the pH value reached 10 and then sonicated (60 W, 40 kHz) for 20 min to form a uniform GO suspension. In general, the nanoparticles repel each other and have no tendency to flocculate in the solutions when they have a large negative or positive zeta potential. The GO was deposited on the treated CF.
Preparation of PI composites
Before mixing, the PI pellets were dried under vacuum at 80°C for at least 12 h and fillers were dried under vacuum at 300°C for the same time. PI composites were prepared in a Sigma high-temperature internal mixer (Shanghai Hangtian Equipment Corporation, Shanghai, China) equipped with two counter-rotating rotors (the rotors are of Sigma type) at 320°C, with a speed of 100 rpm and a mixing time of 10 min with CF of 5 vol%, 10 vol%, 15 vol%, and 20 vol%.
Results and discussion
Tensile properties
Figures 1 and 2 show tensile properties of CF-filled PI composites as a function of filler content together with CF surface treatment. The tensile properties provide an excellent measure of the degree of reinforcement provided by the fiber to the composites. It can be seen in Figure 1 that untreated composites show slight increment in tensile strength up to 10 vol%, then it continuously decreased with increasing filler content. The increased trend indicates a considerable reinforcing effect from these fibers. Since the CF is highly hydrophilic in nature, its strong interfiber hydrogen bonding allows them to cling together, thus resisted dispersion of the fiber, which leads to weak interfacial bonding with consequent problems such as poor stress transfer, small void spaces, and debonding in the resulting composites.
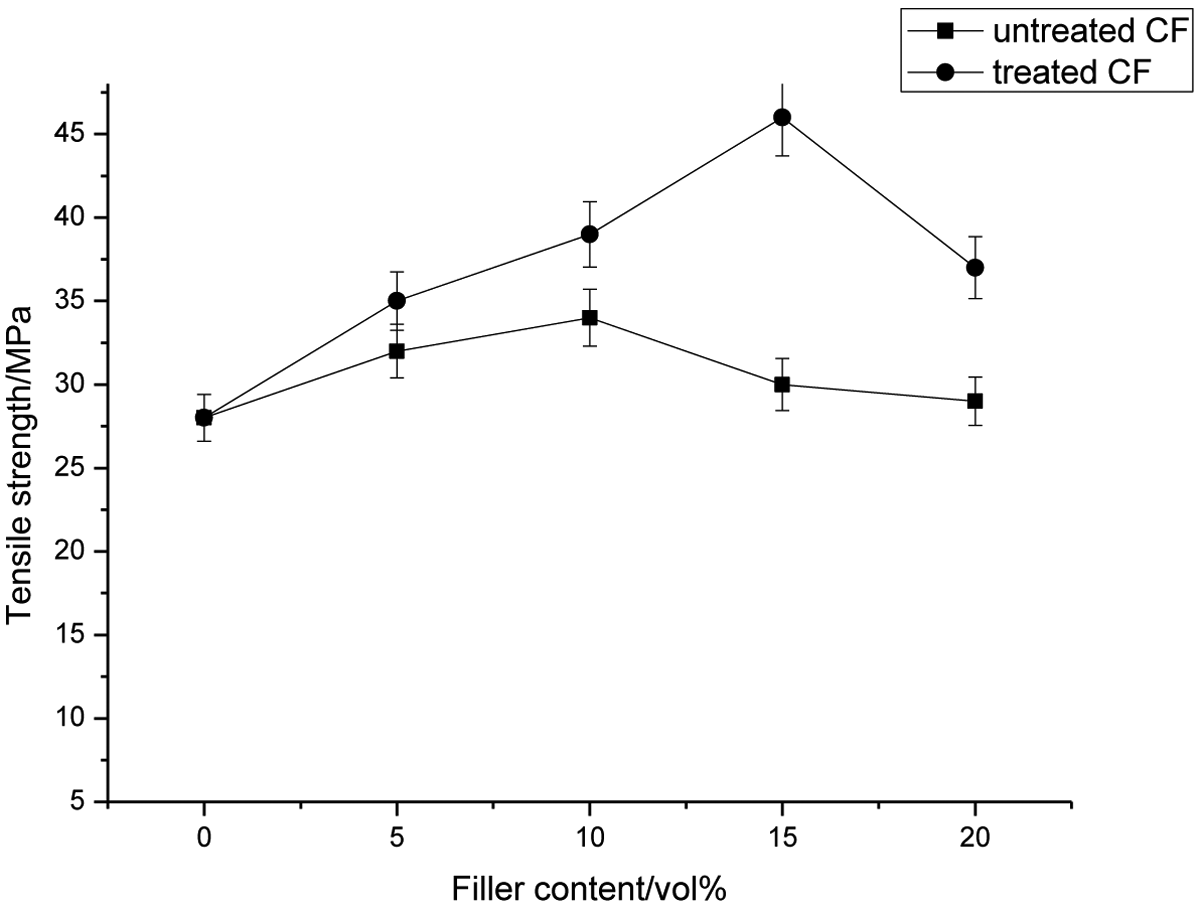
Effect of CF content on tensile strength of CF-filled PI composites. CF: carbon fiber; PI: polymide.
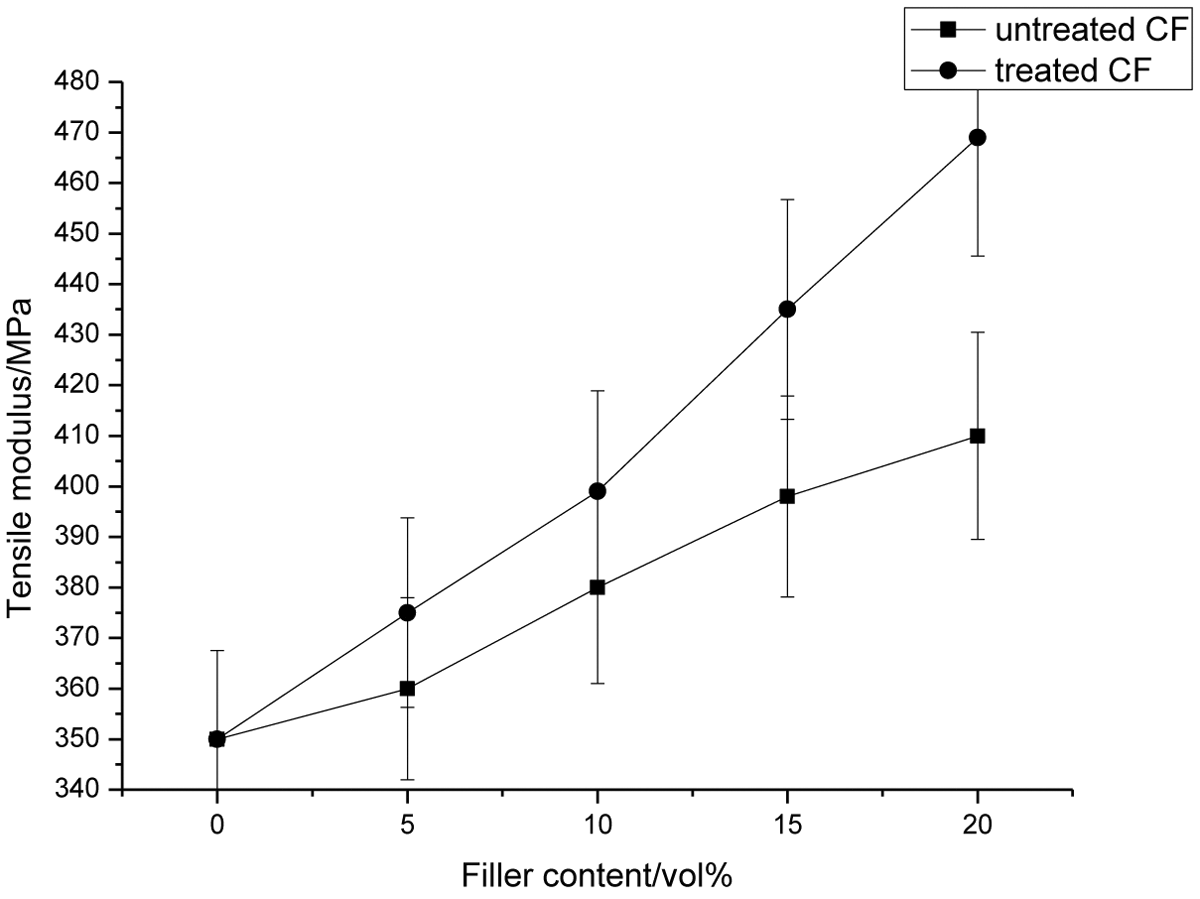
Effect of CF content on Young’s modulus of CF-filled PI/GO composites. CF: carbon fiber; PI: polyimide; GO: graphene oxide.
After the fiber surface is treated, the tensile strength has increased up to 15 vol% and also shows higher strength than control composites at similar filler content with the 42% maximum improvement. At similar filler content, surface-treated CF-filled composites have the highest tensile strength. This improvement is due to the interaction through chemical bonds between the PI and the hydroxyl groups in the fiber, which would form covalent bonds and ester linkages, thus improving fiber–matrix bonding. Meanwhile, the propylene chain in PI would melt and form a single phase with the PI matrix. Since the fiber–matrix bonding is improved, the PI long chains of PI become compatible with the virgin matrix, lower the surface tension of the fiber, and increase its wettability with the matrix. Furthermore, the sufficient numbers of PI molecules allow better diffusion into the matrix polymer, which indicates easier entanglement with the polymer matrix. Without PI, the only adhesion mechanism is interdiffusion. With strengthening of the interfacial interaction between CF and matrix by PI, the resultant efficient stress transfer from the matrix to the fiber leads to enhance in tensile strength. For unmodified fiber-reinforced composites, the lack of adequate impregnation of prepolymer into fibers limits interfacial bonding between unmodified fiber surface and matrix, which could reduce the transmission of stress to the bulk of material through the interface and hence inducing the crack propagation at the weak interface. However, the tensile properties of the composites are significantly increased with increasing the modified coir fiber loading up to 15 vol%, followed by decreased at higher fiber content (20 vol%).
The use of surface treatment resulted in some increment of the tensile strength but the improvement is only until 10 vol% of filler content and then decreases with increasing filler content. The result indicates that the use of surface treatment was proved to be effective in enhancing dispersion, adhesion, and compatibility of systems consisting of a hydrophilic filler and a hydrophobic matrix through modification of polymer–filler interface.
In both cases, such as control composite and surface-treated CF-filled PI/GO composites, increasing filler content resulted in enhanced Young’s modulus since it represents the stiffness of the composites. The enhancement in modulus is easily understood because filler in fibrous form can carry more tensile load with increasing filler content. Besides that, filler is said to be much stiffer than polymer matrix and as a result, it adds stiffness to the composites. For overall trend, a better modulus of about 12–21% was observed at all filler content, when CF was treated, when compared with its counter parts. First, this improvement could be related to better adhesion between the fiber and the matrix by chemical interactions. Better adhesion yields to more restriction to deformation capacity of the matrix in the elastic zone and increasing modulus. Surface treatment increases the nucleation capacity of fibers for PI and alters the crystal morphology of PI around the fibers. Consequently, surface crystallization dominates over bulk crystallization and a transcrystalline can be formed around the fibers. From that, it can be seen that the effects of crystallites have much higher modulus when compared with the amorphous regions and resulted in the increase of the modulus.
During the test, all specimens suddenly ruptured in the fiber bundle area when the maximum drawing force was loaded. Figure 3 reveals that the tensile strength of CF/PI composites is gradually improved as GO content increases, indicating a better fiber/matrix interfacial bonding after adding GO.
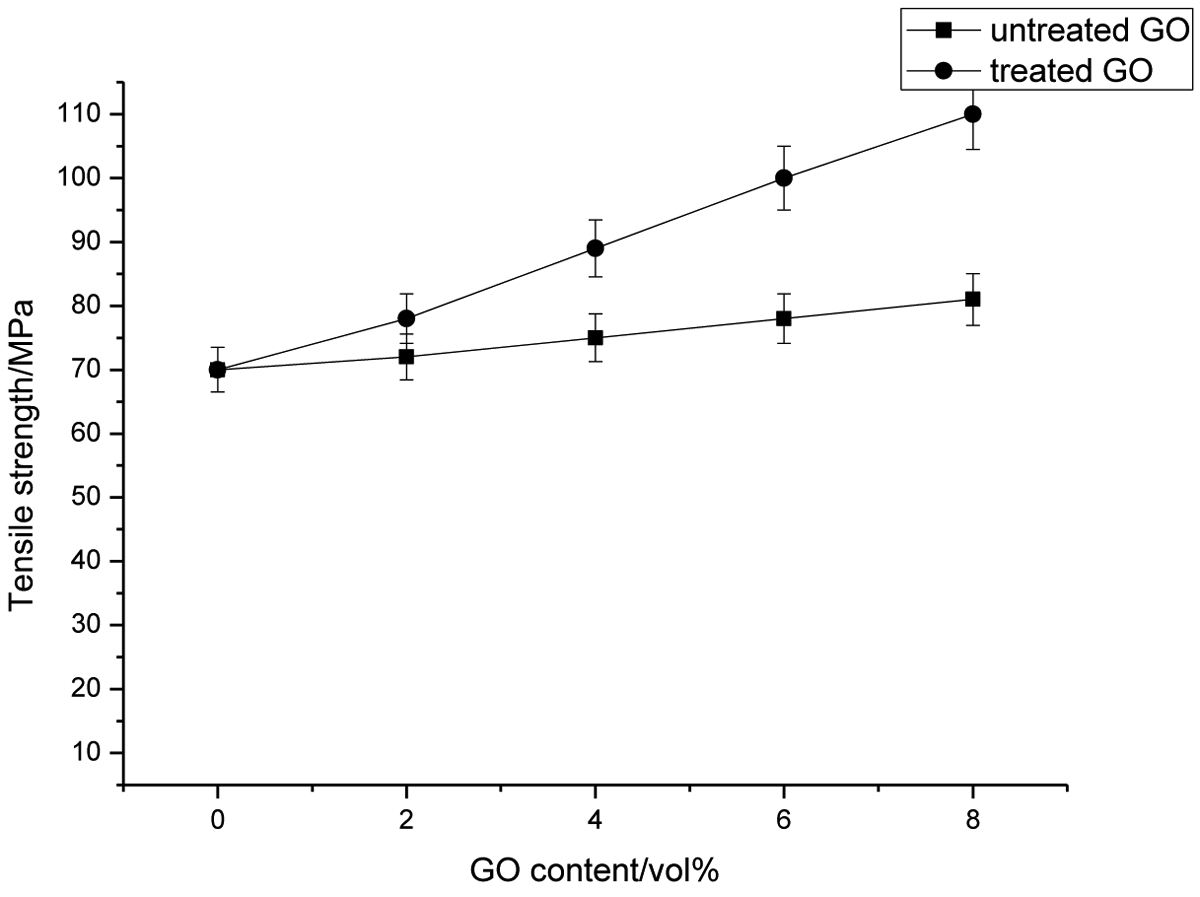
Effect of graphene oxide content on tensile strength of CF-filled PI/GO composites. CF: carbon fiber; PI: polyimide; GO: graphene oxide.
In general, all polymer composites with surface-treated fillers exhibit an increase of their tensile strengths as compared to untreated filler composites. Figure 4 illustrates the dependence of the tensile strength of the PI and all PI composite materials on the untreated and surface-treated of GO content. From the Figure 4, we can see that the tensile strength values for all the PI composites containing untreated and surface-treated GO particles increase with increasing the amount of filler loading. The result also indicates that all these PI composites showed a tensile strength higher than the PI. Chemical treatment of GO improves the interlocking between the filler and the PI matrix. As a result, the tensile strength values of PI composite materials containing surface-treated GO is significantly improved than the equivalent PI composites that included particles without coupling agent. This indicates that GO particles–matrix interfacial adhesion has improved leading to increased stress transfer efficiency from the PI matrix to the filler resulting in improvement of tensile strength values of the PI composite materials. For the PI composite systems containing surface-treated GO with the silane coupling agent, flexible interface layers were formed, and there was interfacial adhesion to some extent between the dispersed GO particles and the PI matrix. The higher tensile strengths of composites with surface-treated filler are attributed to restricted segmental motions in the neighborhood of organic–inorganic interfaces. Tensile modulus is a major parameter for characterizing the stiffness of materials. Figure 8(b) displays the dependence of the tensile modulus and elongation at break of the PI and PI matrixes on the untreated or surface-treated GO content. It is obvious that the tensile modulus of the all PI composites materials with untreated or surface-treated GO with the silane coupling agent increased significantly with the increase in filler loading, and the effect is pronounced in the case of composites with modified GO particles. Related to crystallinity of PI matrix, up 2.0 wt% GO loading the modulus should have decreased, but instead it increased. Incorporation of filler particles within the chains decreases the overall crystallinity probably by reducing the hydrogen bonding interaction between C=O and N–H groups on the PI chains, but above 2.0 wt% untreated or surface-treated GO content crystallinity tends to increase. The trend in the modulus could be explained by the opposing factors of decrease in crystallinity upon a certain filler content and reinforcing action of GO particles. A plausible explanation for the improvement in tensile modulus of PI composites including treated filler is also the fact that the interaction of PI chains with the organic part of the modified filler leads to immobilization or partially mobilization of polymer phases. In particular, the tensile modulus and tensile strength increased to 32% and 9%, respectively, upon addition of 8.0 wt% unmodified GO to PI matrix. In case of PI-modified GO composites, tensile modulus increased by 37% whereas tensile strength showed an increase of 14% when 8.0 wt% of modified GO was used. Tensile elongation at break may also reflect to a certain extent the tensile fracture toughness of polymer and polymer composite. This behavior may be due to the interaction between the GO particles and PI matrixes, reducing the segmental mobility of polymeric chains and formation of stress concentration sites. In PI, the presence of the polar amide groups allows a high degree of hydrogen bonding between the CO and NH groups in adjacent sections of the polyamide chains to be developed when chains are aligned. The GO particles acted as rigid filler, reducing the ability of the polymer matrix for extension. Incorporation of fillers that have poor adhesion to the polymer matrix seems to cause interruption in the alignment process of the chains during elongation to break measurements. With the enhancement in rigidity, by inclusion of untreated particles, the ductility of PI composites decreases; consequently, the composites break at lower elongation. During polymerization-molding process can appear few H bonds between GO surface OH groups and PI matrix which closes the inorganic component to PI chains. At the same time, GO particles have tendency to agglomerate caused by H bonds between the O=H species on GO surface and high surface energy, by which the dispersion into matrix is hindered. Due to the different nature of components, the filler particles are poorly attached to matrix. Thus, nonadherence and the stress transfer at the particle/PI interface are inefficient, and in the composites, the discontinuity in the form of debonding occurs. When filler loading is increased, more weak interfacial regions between the filler surface and the PI matrix are formed. The aggregated centers would probably act as stress centers which cause the decrease of elongation at break. The cracks are more easily induced in the weaker interfacial regions and the composites fracture at a lower degree of elongation with increasing filler content occurs. The GO particles tend to have better dispersion in matrix rather than unmodified filler that leads to enhanced interfacial interactions between particles and PI matrices which can improve elongation at break.
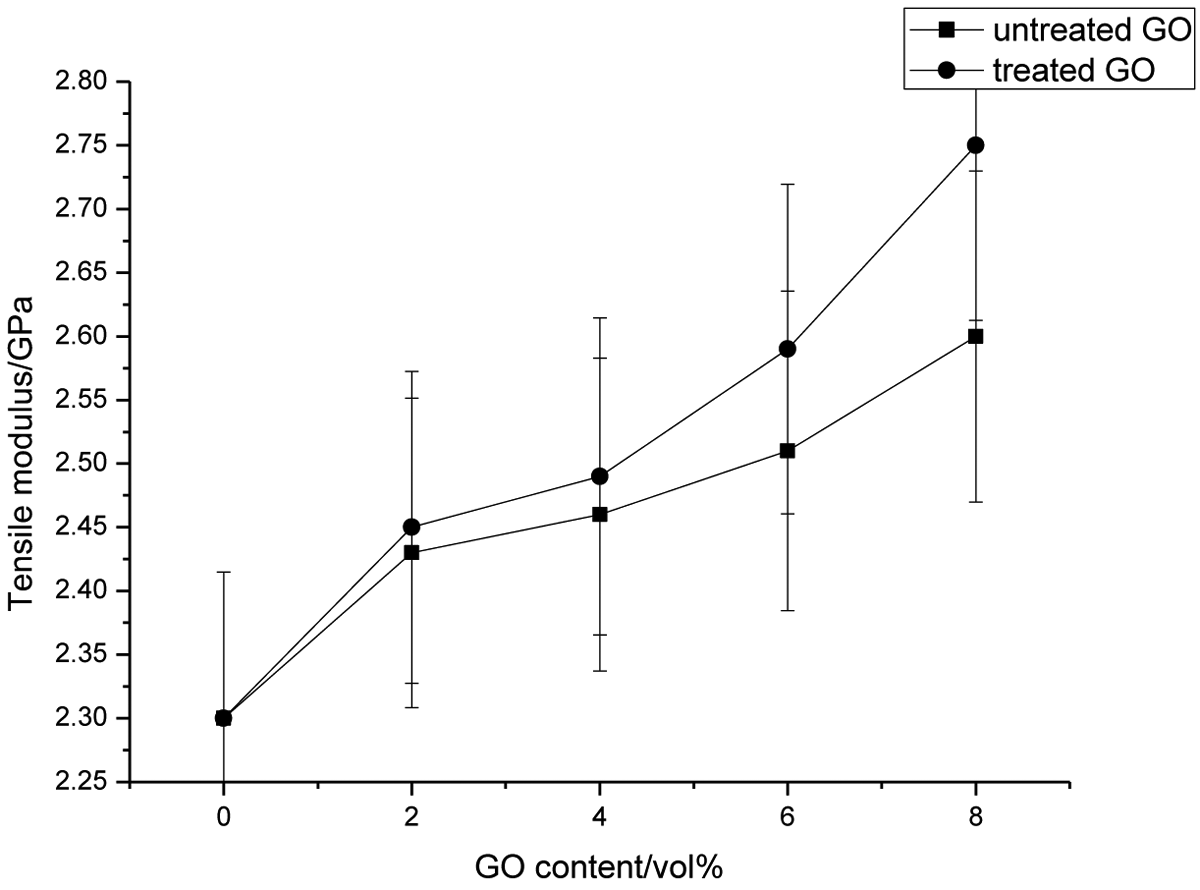
Effect of graphene oxide content on Young’s modulus of CF-filled PI/GO composites. CF: carbon fiber; PI: polyimide; GO: graphene oxide.
In this study, it can be seen from scanning electron microscopic (SEM) images in Figure 5 that some of the CFs were broken during attacking process and interfacial debonding between CF and PI matrix could be observed, which could absorb a part of impact energy. However, GO particles have lower mechanical property than CF, so they absorb less impact energy than CF. This can be used to explain why the impact strength of the composites is higher than that of the PI, and the impact strength of composite is higher than that of the particle composite at the same concentration. On the other hand, the filler may become agglomeration when its concentration is too high. SEM observations indicated that there was a considerable difference in the fiber/matrix interaction between the treated and untreated composites. It could be seen in the untreated system (Figure 5(a)) that the phenomenon of pullout occurred to a greater extent than treated systems (Figure 5(b)). This was probably due to the existence of voids between the matrix and fibers, which led to weak interfacial interactions. The treated systems showed better adhesion compared to the untreated one. For the treated CF composites, the fracture morphologies were obviously different. It was exhibited that lots of resin were tightly adhered to the fiber surface accompanied with the obvious resin deformation. The “separation” between fiber and matrix is clearly visible, suggesting a poor fiber/matrix adhesion, as shown in Figure 6. In contrast, the fracture surfaces of CF/nanofilled PI systems appear to become rougher.

SEM of impact fracture surface. SEM: scanning electron microscope.
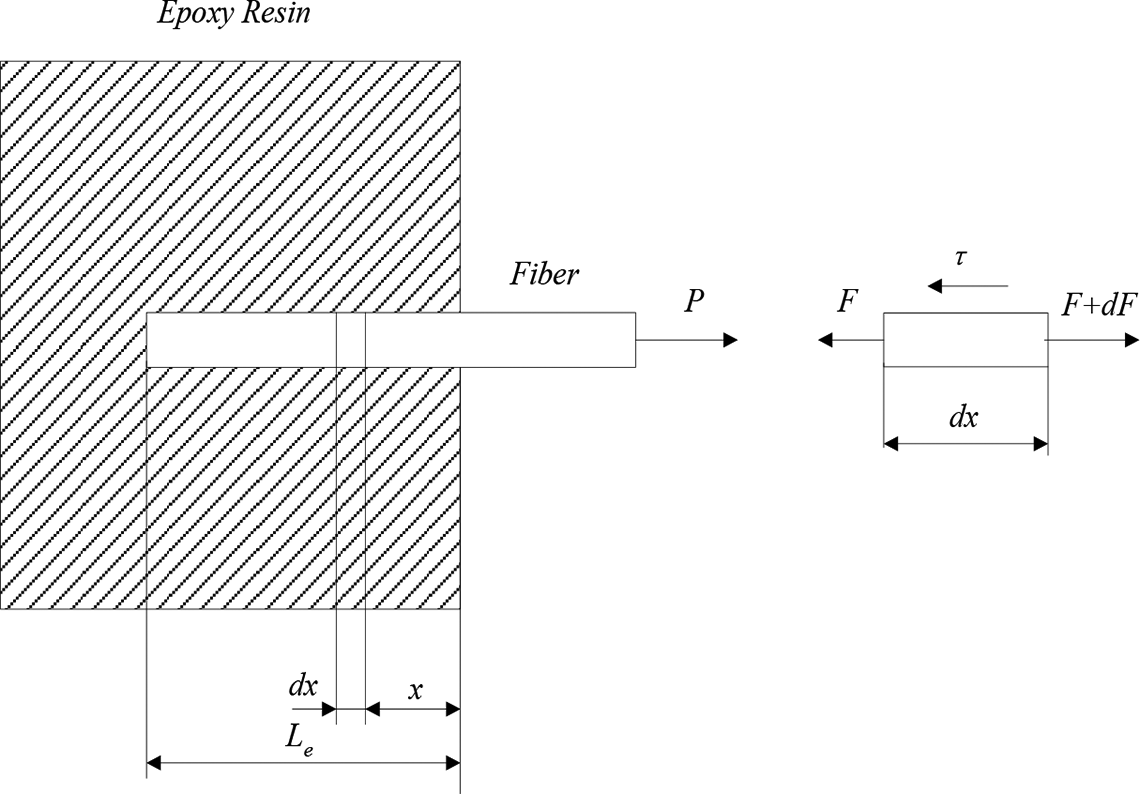
Greszczuk model of single fiber pull-out test.
Abundant plastic dimples and multibranched river lines (ductile characteristic of PI) have emerged; some matrix debris is still attached at the surface of the fibers; and the CFs combine strongly with the nanomodified matrix at their edges, with no gaps or separation caused by debonding. So a much better interfacial bonding is proved through the analysis of SEM images.
According to SEM images, the fracture surface of CF/PI composite sample is featured with quite regular cracks, formed by apparent cleavage between fibers. After introducing GO nanoparticles, the crack propagation paths become more branched and complicated, clearly manifesting the antishear phenomena. More interestingly, the number of debonded fibers in the fracture region decreases evidently, indicating a better stress transfer in the interphase layer around fibers. It is proved that the better interfacial adhesion of fiber reinforced polymers (FRPs) is obtained through filling matrix with GO.
The improvement in the mechanical properties is probably due to the increase in the wettability and roughness of GO/CF, which provides close molecular contact between the fibers and the matrix. In addition, chemical bonding between the nanoparticles and the resin matrix facilitated by the oxygen-containing functional groups in GO may form a strong chemical bond with the oxygen groups of the PI during the curing steps, which leads to a more intense interfacial bonding between the PI and GO in the interphase region. Furthermore, GO was firmly coated to the oxidized CF surface through covalent attachments, which leads to GO not easily detaching from the matrix. Therefore, electrochemical oxidation coupled with the GO can provide additional reinforcement to the interface to increase the material’s strength and toughness and decrease the interlaminar stress concentration, which effectively improves the mechanical properties of the composites. The exact structure differences and the coalescence and propagation of microcracks are illustrated in Figure 7.
The structure differences and the coalescence.
FTIR spectroscopic analysis
The Fourier-transform infrared (FTIR) technique was used to investigate the change in the surface chemistry of the CF and GO after surface treatment. Figure 8 shows the spectra of the two CFs, unmodified and modified fibers in the region from 4000 cm−1 to 400 cm−1. In addition, the spectrum of GO is shown for comparison (Figure 8(a)). In the spectrum of the untreated CF (Figure 8(b)), the broad and strong peaks at 3380 cm−1 and 2885 cm−1 were due to hydrogen-bonded –OH vibration and C–H stretching from the –CH2 group of the cellulose structure in the fiber, respectively. The peak at 1643 cm−1 is usually assigned to the stretching vibration of H–O–H due to absorbed water, which was probably collected during sample preparation. The peaks at 1432 cm−1 and 1373 cm−1 were ascribed to the bending vibration of –CH2. The peak observed at 1050 cm−1 was due to the C–O stretching of CF. Upon treatment with the NaOH solution, new absorption bands emerged at wave numbers of 2961 cm−1, 1238 cm−1, and 1065 cm−1, as shown in Figure 7(c); this was clearly not seen in the spectra of the corresponding untreated fiber. The appearance of a small absorption peak at 2961 cm−1 was found in the same region of the stretching vibration of the –CH2 and –CH3 groups at 2973 cm−1, 2923 cm−1, and 2887 cm−1. It was reported that the IR absorption band around 1200 cm−1 was attributed to C–O–Si, and this band was found to be present in GO (1246 cm−1) and in the treated fiber (1238 cm−1). It was clear that the band at 1050 cm−1 present in the untreated fiber was replaced by a broad band ranging from 1080 cm−1 to 1050 cm−1 after the treatment. This indicated that the C–O groups in the treated fiber had different local chemical environments.
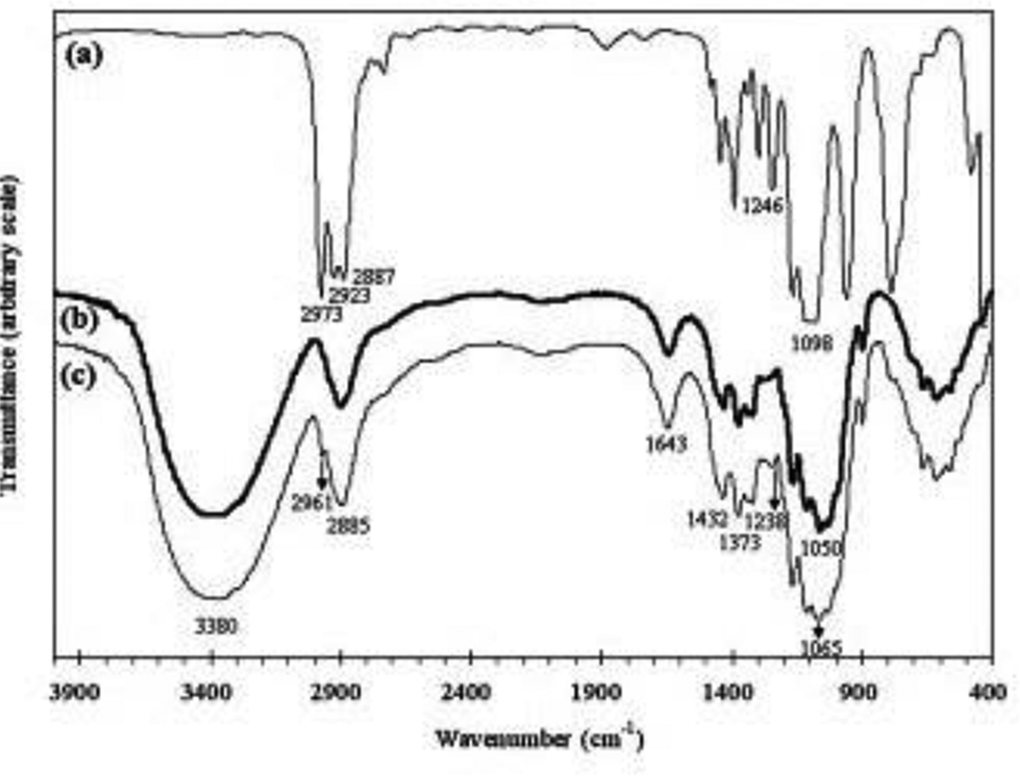
FTIR spectroscopic analysis. FTIR: Fourier-transform infrared.
Conclusion
The tensile properties provide an excellent measure of the degree of reinforcement provided by the fiber to the composites. At similar filler content, surface-treated CF-filled composites have the highest tensile strength. This improvement is due to the interaction through chemical bonds between the PI and the hydroxyl groups in the fiber, which would form covalent bonds and ester linkages, thus improving fiber–matrix bonding. The tensile strength values for all the PI composites containing untreated and surface-treated GO particles increase with increasing the amount of filler loading. Chemical treatment of GO improves the interlocking between the filler and the PI matrix.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the editor and anonymous referees for their insightful and constructive comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Grant from the National Natural Science Foundation of China (funding nos: 71472065), the Research Projects of the Social Science and Humanities of the Young Fund of the Ministry of Education (funding no. 14YJC630026), the Shanghai Pujiang Program (14PJC027), and the Key Projects of Shanghai Soft Science Research Program (17692107000).
