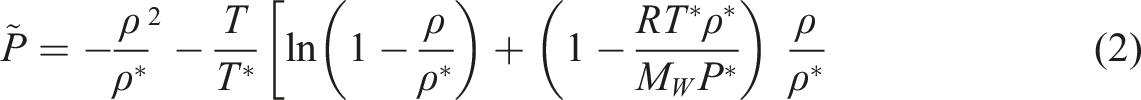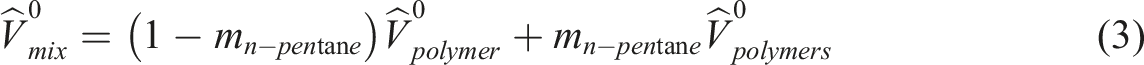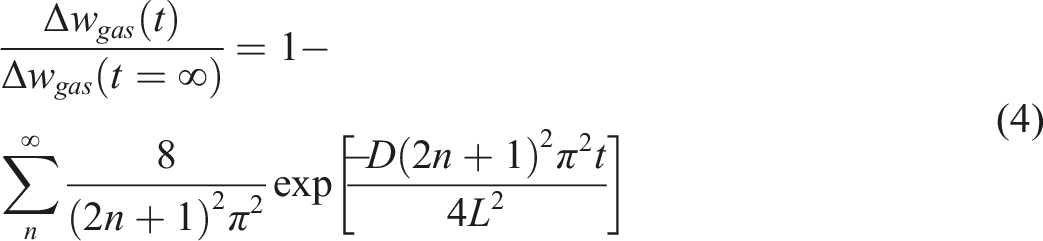Abstract
In this work, we investigated the effect of Gelatin on the solubility and diffusivity of normal pentane in poly (vinyl alcohol) (PVA)/Gelatin (Gel) blends. The processibility of PVA was enhanced after adding the gelatin as plasticizer to PVA which gelatin could extend the process widow of PVA with destroying the hydrogen bonding of the polymer matrix. Therefore, the PVA/Gel blends with different concentrations were prepared by simple solution-casting method. The intermolecular suitable interactions in PVA/Gel blends were confirmed using the FTIR, XRD and SEM/EDX analyzes. In the following, the effect of various factors like temperature, pressure and gelatin content on the thermodynamic parameters like specific free volume, solubility and diffusivity was investigated. It was shown that there was a relationship between the specific free volume of the blends with the solubility and diffusivity of normal pentane in blends. The specific free volume of blends was determined by applying the PVT data and the Sanchez-Lacombe (SL) equation of state, in which it was enhanced with increasing of the temperature and gelatin in blends, respectively. Also, the solubility and diffusivity of normal pentane in samples were determined by the magnetic suspension balance (MSB) system. The results presented that the solubility and diffusivity of n-pentane improved with increasing of gelatin content in blend, pressure and temperature decrement, respectively.
Introduction
Lately, different blowing agents due to their economical obtainability, are being used in foaming process of polymers.1–3 Poly (vinyl alcohol) is the polar biopolymer which has good properties like, physical adsorption and solvent resistance.4–6 PVA has a strong molecular hydrogen bonding which cause the small processing window. Plasticizers are widely used as significant additives for the processing of PVA to widen its processing window. In this case they interfered in hydrogen bonding of the PVA and changed the crystalline regions of the polymer matrix.7-8 The porous PVA/plasticizers systems may be used as a foam or scaffold in tissue engineering applications. 4 Gelatin is a water-soluble biopolymer and acts as a good plasticizers which is a suitable chose for combination with the poly (vinyl alcohol). 9 The PVA/Gel foams could be produced via the bath foaming technique. The batch foaming process contains three main stages: Production of polymer/blowing agent mixture in high pressure, rapid pressure drop and finally cell nucleation and growth. 12 In the recent studies, two points of view should be considered; the thermodynamic study and the morphological investigations. In thermodynamic study, some important parameters like the solubility and diffusivity of different gases in polymers and operation conditions like temperature and pressure must be investigated.10–13 Between different systems for calculating the solubility and diffusivity, using the Magnetic Suspension Balance system is common method, which consists of a calculating chamber, and a balance located outside the chamber.14–16 From the thermodynamic point of view, there is no study in literature about measuring solubility and diffusivity of normal pentane in poly (vinyl alcohol)/Gelatin. However, some related thermodynamic studies were done using different polymer/blowing agent systems. In this case, Kiran et al. investigated the solubility and diffusivity of CO2 and N2 in polymers and also the glass and melting temperatures of the polymers were determined at high pressures. 17 Also in another work, using free volume theory, Ushiki et al., measured solubility and diffusivity of CO2 in molten state polycaprolactone. 18 The solubility of CO2 in ionic polyimides were investigated and it was found that solubility is weakly related to the theoretical surface area, and the diffusivity is correlated to the fractional free volume. 19 Some researchers focused on the solubility comparison of different gases in polymers.20–22 For example, Sebok et al., 23 worked on the Diffusivity, permeability and solubility of H2, Ar, N2, and CO2 in poly(tetrafluoroethylene) in detemonrd temperature range. 23 In this work also the pre-exponential factors and activation energies of the diffusivity were reported. Other similar studies were done in the case of solubility and diffusivity measurements of different blowing agents in polymers. 23 Maghami et al., considered the effects of temperature and pressure on the permeability, diffusivity and solubility of some polymers. 24 They also investigated the validity of the model by comparing the experimental data with the literature. 24 In another study, Andersons et al., estimated the diffusivity of gases in closed-cell polyurethane foams. 25 In this work, the gas diffusivities are predicted by determining the gas content in foams at various times. 25 In some studies about the polymer foaming processes, the measurements of the solubility and diffusivity of the gases as blowing agents in the polymer were performed prior to the foaming process.26–28 The reason for this is to investigate the effect of gas solubility on its foaming properties. By increasing the gas solubility and diffusivity in polymer, the final cellular structure of produced foam was more homogeneously, due to the accessibility of more gas during the foaming process.29–30 For instance, Azimi et al., investigated on the solubility, diffusivity and bubble nucleation of supercritical CO2 in Polystyrene. 31 They measured the thermodynamic parameters for PS/CO2 system and in the following, the foaming process of PS was detected. It was concluded that with increasing the solubility and diffusivity parameters, some foaming characteristics like average cell density and sizes improved. 31
In this work the solubility and the diffusivity of normal pentane (n-pentane) in poly (vinyl alcohol)/Gelatin blends, were calculated at different temperature and pressure by using the MSB system. The application of PVA/Gel blends is in tissue engineering in which the PVA/Gel blends foams or scaffolds were produced via the batch foaming process. We used PVA/Gel blends as polymer and n-pentane as the blowing gent. Therefore cconsidering the application in tissue engineering, which is related to our next research work, it is important to determine the solubility and diffusivity of n-pentane in the poly (vinyl alcohol)/Gelatin blends. So that the higher the values of these thermodynamic parameters, the final scaffold structure will be open cell with higher porosity that will be useful in tissue engineering applications. Therefore, in the present study, we have studied the effect of various factors like temperature, pressure and gelatin content on the thermodynamic parameters like specific free volume of polymer/gas, solubility and diffusivity.
Experimental
Material
In this work, poly (vinyl alcohol) with weight average molecular weight of 90,000 g/mol was prepared from Kuraray, Japan. Also, the gelatin was bought from the Rankem Ranbaxy Fine Chemicals, Ltd. (Mumbai, India) and n-pentane was used as the blowing agent.
Preparation of PVA/Gel/CNT blends
The PVA/Gel blends with different concentrations of gelatin were prepared by the solution-casting method. Both PVA and Gel, with different compositions, were dissolved in the distillated water at 90 °C to make PVA/Gelatin composites. The final solutions was transferred onto a glass Petri dish and kept in an oven at 80 °C for 4 h. The water was evaporated, and the films of PVA/Gelatin composites keep on the Petri dish.
Characterization and methods
The XRD analysis and the sample crystallinity were studied by a DX-1000 diffractometer (Dandong Fangyuan Instrument Co., Ltd., China), the Cu Kα generator was worked at 40 kV and 25 mA, with a scanning rate of 1°/s. In this case the crystallinity of PVA and the blends were determined as:
The thermodynamic parameters like solubility and diffusivity data for gases were calculated by the MSB system.
31
The MSB contains of a calculating chamber and a balance located outside under special conditions. In chamber with high pressure and temperature, the sample is hooked up to a holdup magnet. In this system, an electromagnet is attached to the hook of the balance and located outside the chamber. In the following, without any contact, the sample weight could be conveyed to the balance. The PVT data were obtained by a high-pressure apparatus (GNOMIX, Inc., USA). For determining the specific volume of our samples with n-pentane, the Sanchez-Lacombe (SL) equation of state was used
31
:
The analytical solution of the Fick’s second diffusion equation was given by
31
:
Results and discussion
Figure 1 shows the X-ray patterns for PVA and PVA70/Gel 30 samples. In the case of PVA, a large and distinct peak at 2θ = 19.5° is observed, indicating the semicrystalline nature of PVA. By using equation (1), the crystalline fraction is determined as 62.3%. Though, the addition of 30% Gelatin to PVA (PVA70/Gel30) cause reduction in intensity of the mentioned peak and the crystalline fraction was decreases to 11.2%. Basically, the crystallinity of PVA is significantly affected by the plasticization effect of Gelatin. The recent result clearly confirmed that Gelatin suitably interacted with the PVA chains and destroy the hydrogen bonding in PVA matrix. The XRD analysis for Poly vinyl alcohol and PVA70/Gel 30.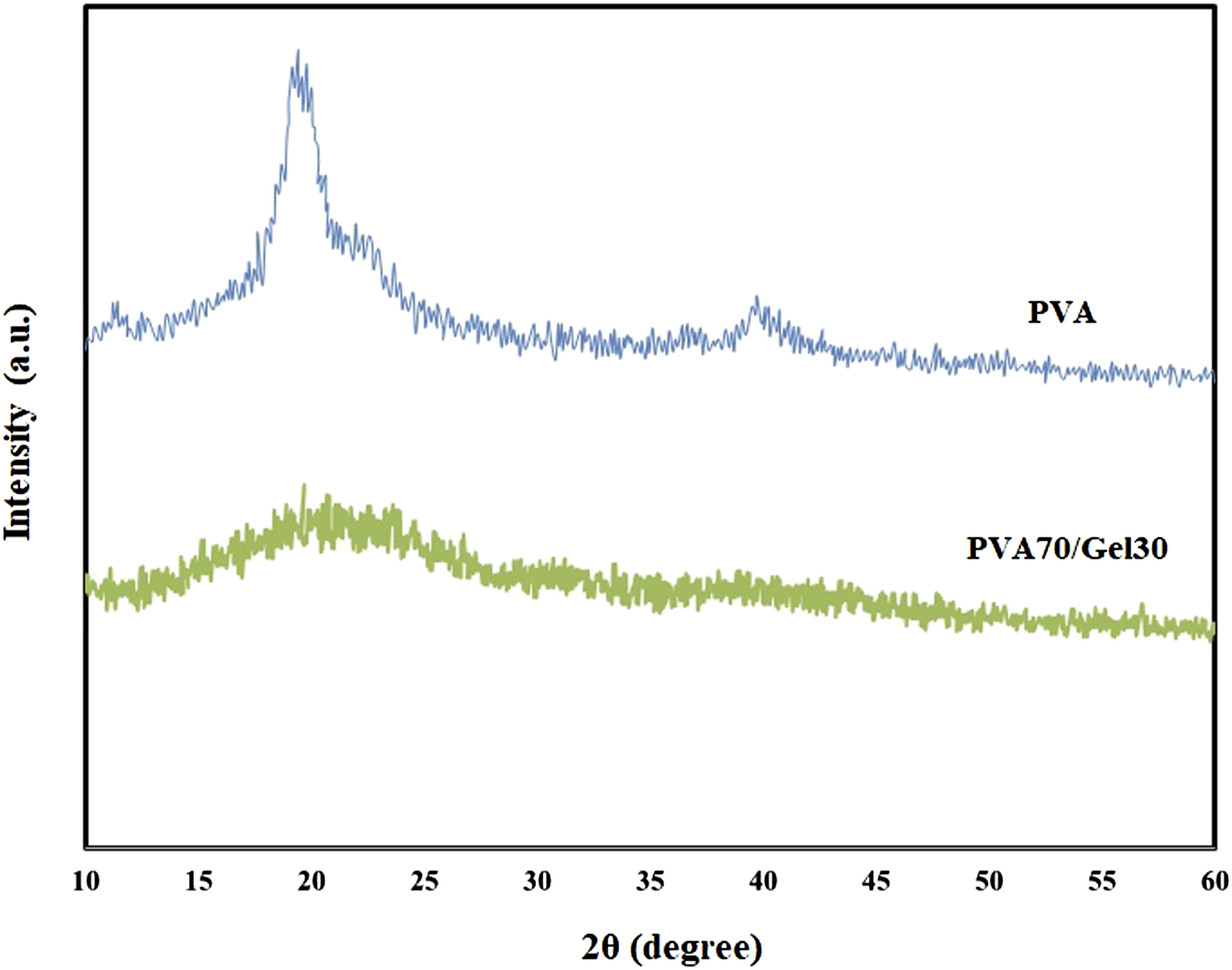
The interaction between PVA and Gelatin molecules also was conducted by the FTIR analysis in Figure 2. It is shown that PVA exhibits a wide band of O-H bonding in 3000–3600 cm−1 range. While, such bands for the PVA/Gel blends move to higher wavenumbers in which, by increasing the Gelatin content to 30% in composite, the wave number increases roughly by 15 cm−1. The related interaction between polymer chains of PVA and other plasticizers has been reported in literature.
7
Considering the results of the XRD and FTIR analysis, the clear destruction in PVA crystallinity could be occurred and Gelatin diffuse into PVA chains and disturb the hydrogen bonding of PVA. As it was mentioned, PVA is a semi-crystalline bio polymer and the solubility and diffusivity of gases in PVA is hard compared with other amorphous polymer. The FTIR analysis for Poly vinyl alcohol/Gel blends.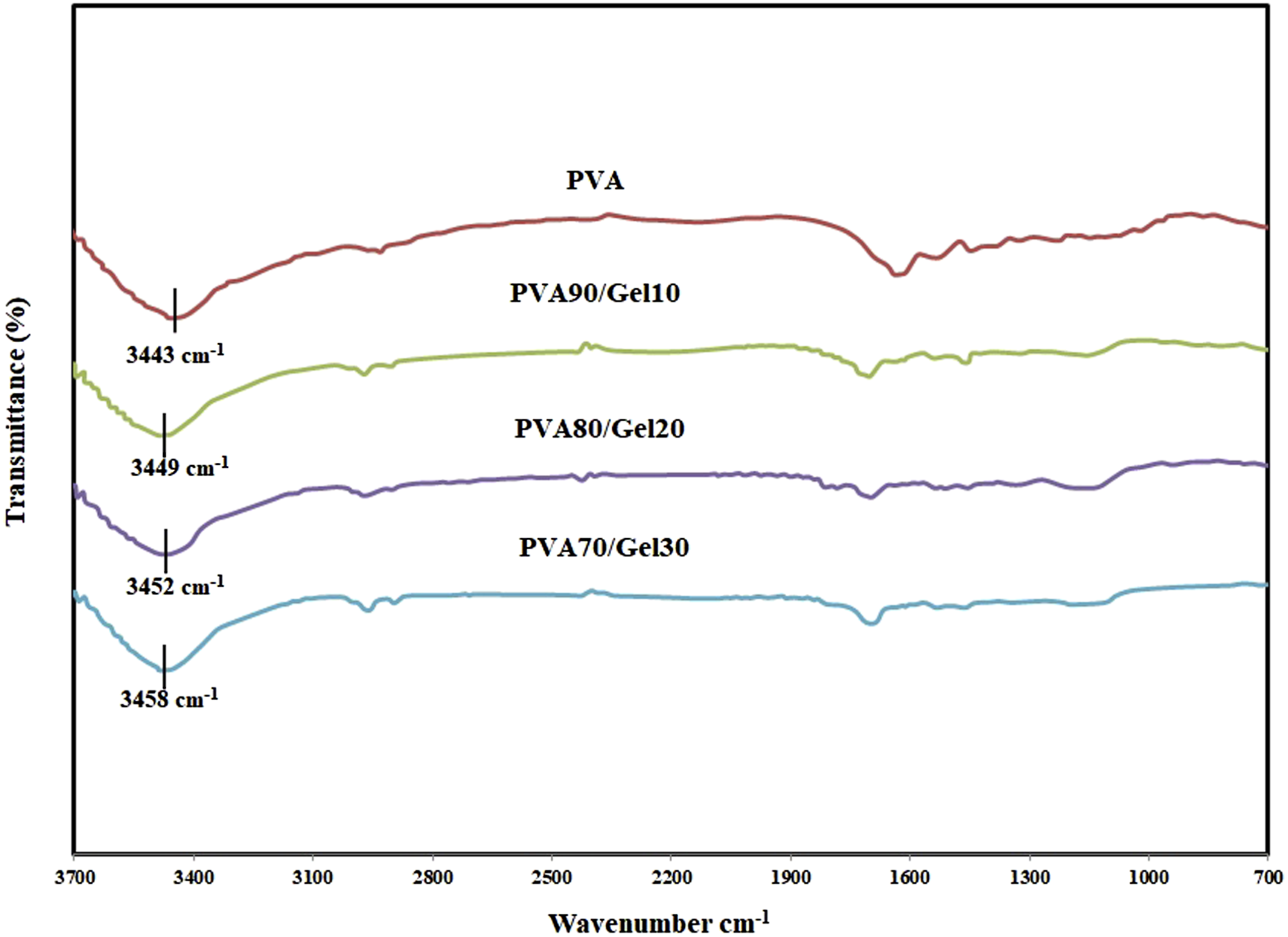
The characteristic parameters of samples.
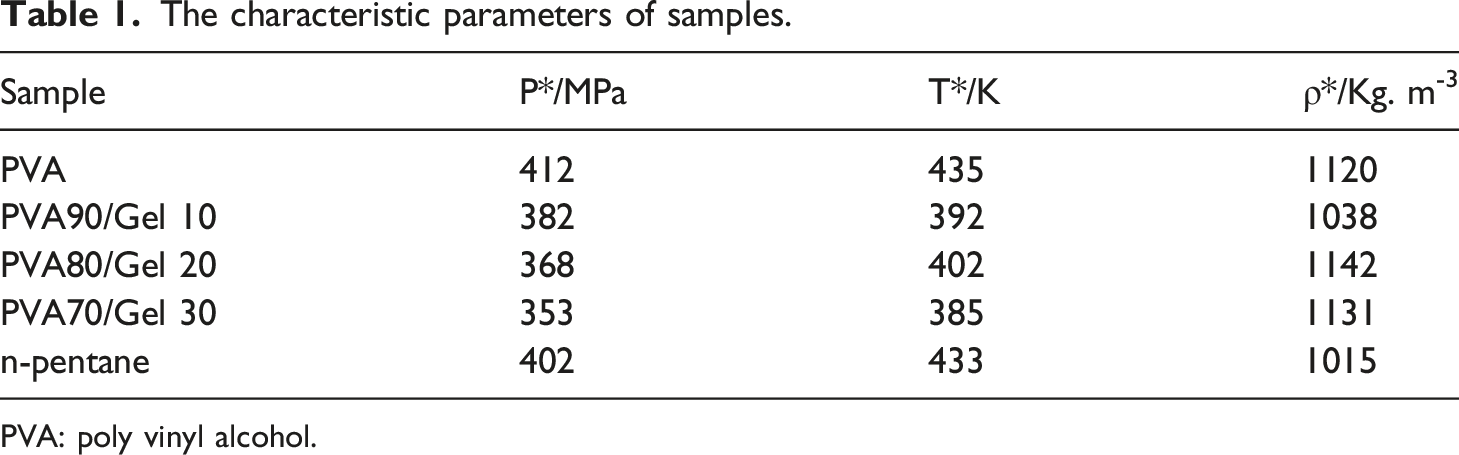
PVA: poly vinyl alcohol.
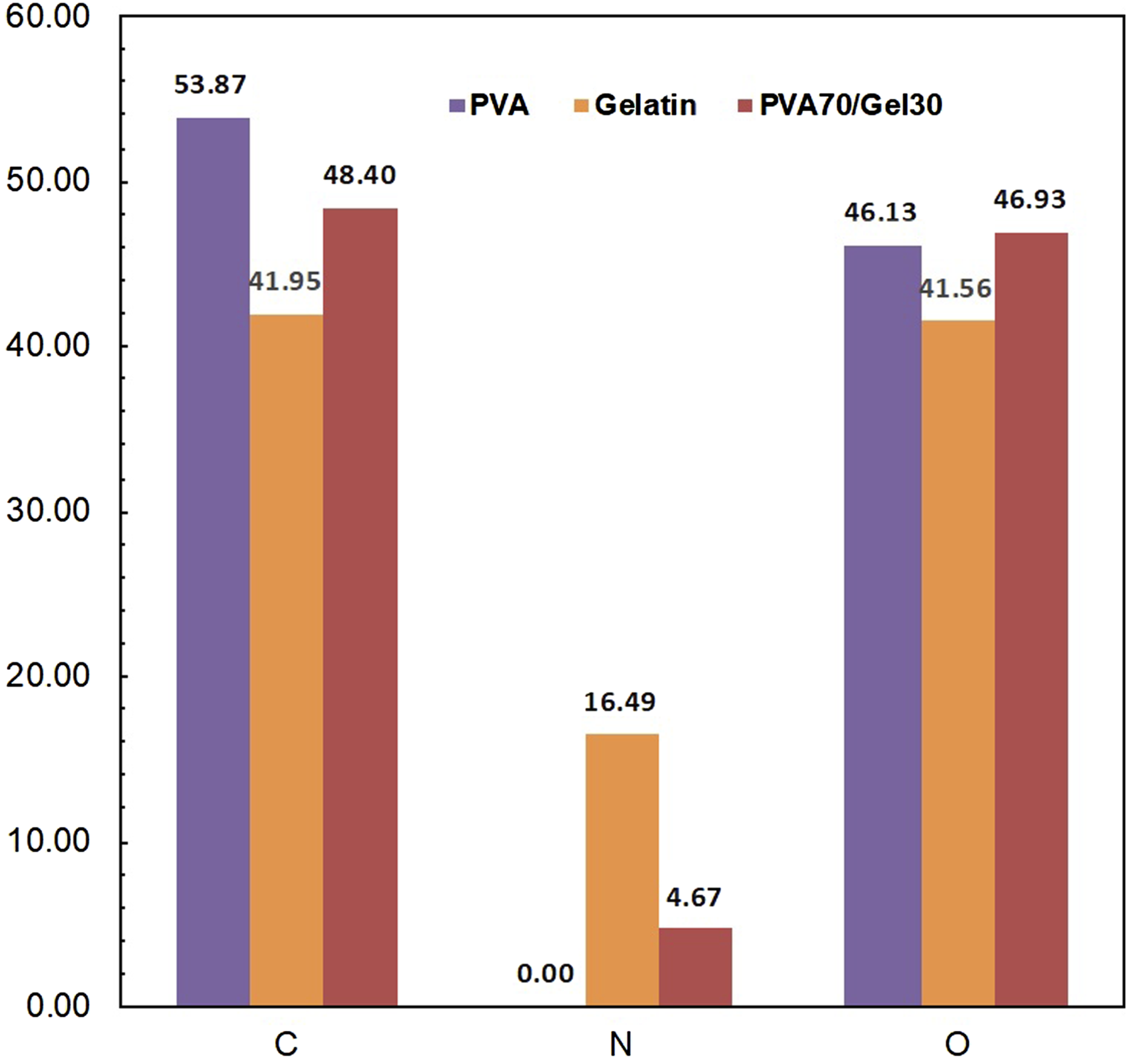
In the following the surface morphology/EDX analyzes were done for investigating the dispersion of gelatin in blend. Figure 3 shows the results of the FESEM images for samples. It is clear that, PVA70/Gel30 shows a smooth surface morphology which exhibits the uniform dispersion of gelatin in the PVA/Gel. Figure 4 shows the EDX data of surface molar percentages of for PVA, Gelatin and PVA70/Gel30 blend. EDX measurements illustrates the change in the structure of PVA after adding gelatin to the blend. It is should be noted that both polymers (PVA and Gelatin) individually contain carbon and oxygen elements in their structure while gelatin also has nitrogen element in its structure. In the following, Figure 5, represents the dot mapping images of the for PVA, Gelatin and PVA70/Gel30 blend. It shows the distribution of involved O, C and N elements. It is clear that the PVA modification by gelatin was clearly observed for existing elements (C, O and N) on PVA/Gel structure. The surface morphology for (a) poly vinyl alcohol, (b) Gelatin and (c) PVA70/Gel 30 blend. Surface molar composition for samples: Poly vinyl alcohol, Gelatin and PVA70/Gel 30. Dot mapping images of samples: (a) Poly vinyl alcohol, (b) Gelatin and (c) PVA70/Gel 30.

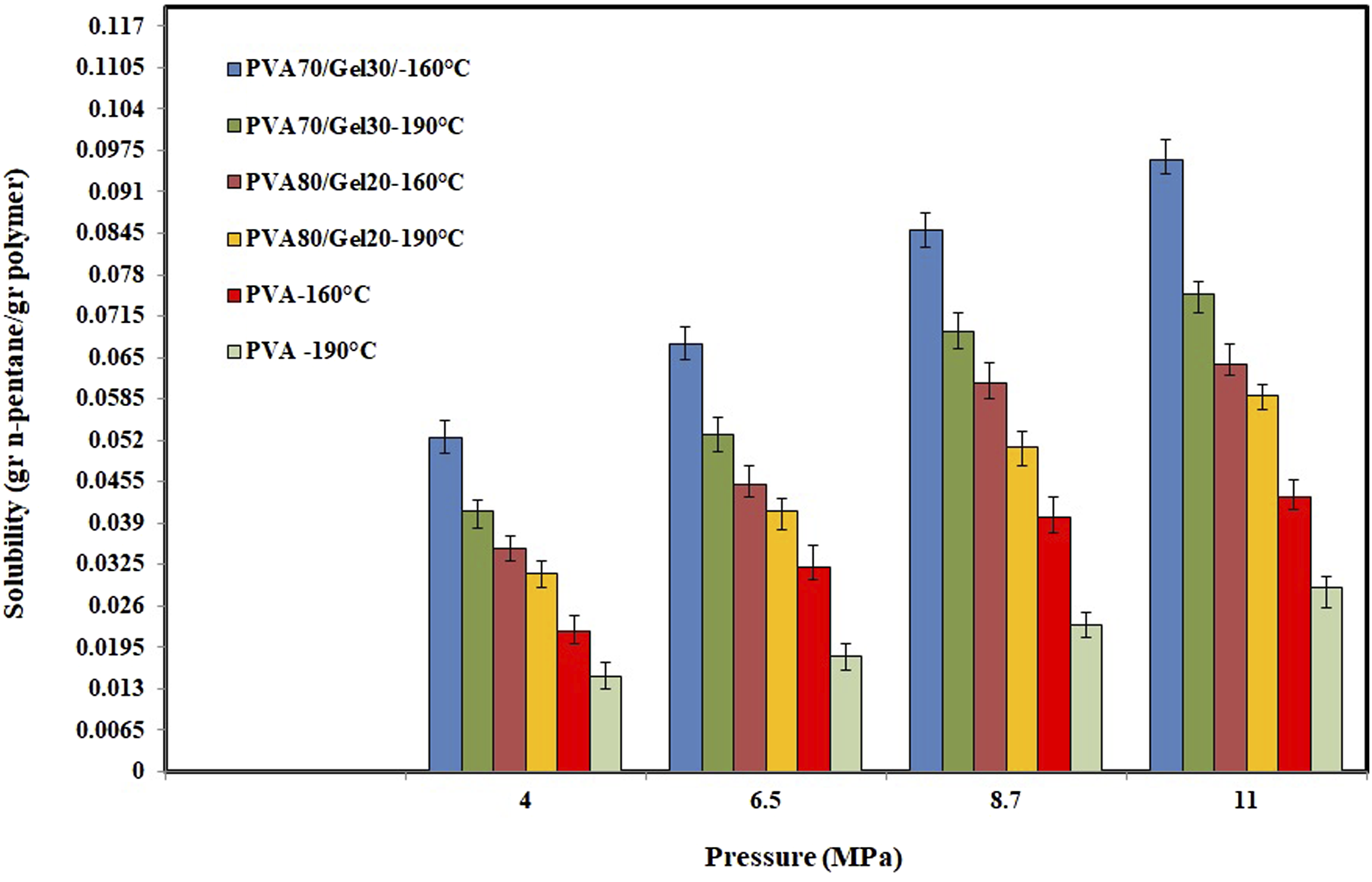
The solubility of n-pentane in samples at different pressures and temperatures is shown in Figure 6. To perform accurate measurements, the weight of dissolved n-pentane measured by MSB readout has to be corrected in consideration of the buoyancy effect. The true amount of dissolved n-pentane, w, was calculated by: The solubility of n-pentane in Poly vinyl alcohol/Gel samples at different temperatures and pressures.
The specific free volume calculated for different composites and are shown in Figure 7. The specific free volume is increased with increasing of temperature and n-pentane content. As well with adding Gelatin in blends, while other two parameters kept constant, the specific free volume of composites increased. The determined specific free volume of PVA/Gel/n-pentane systems.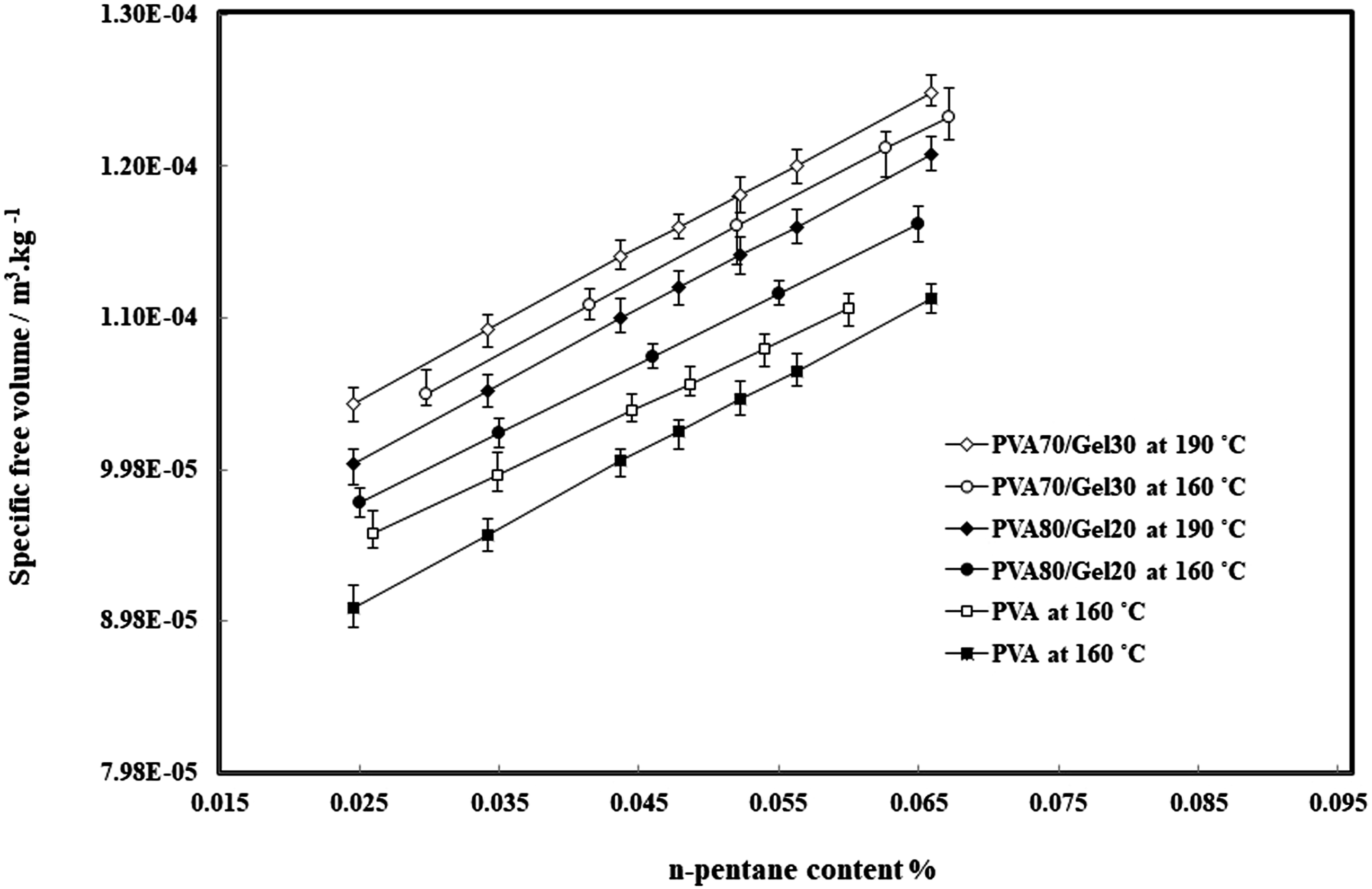
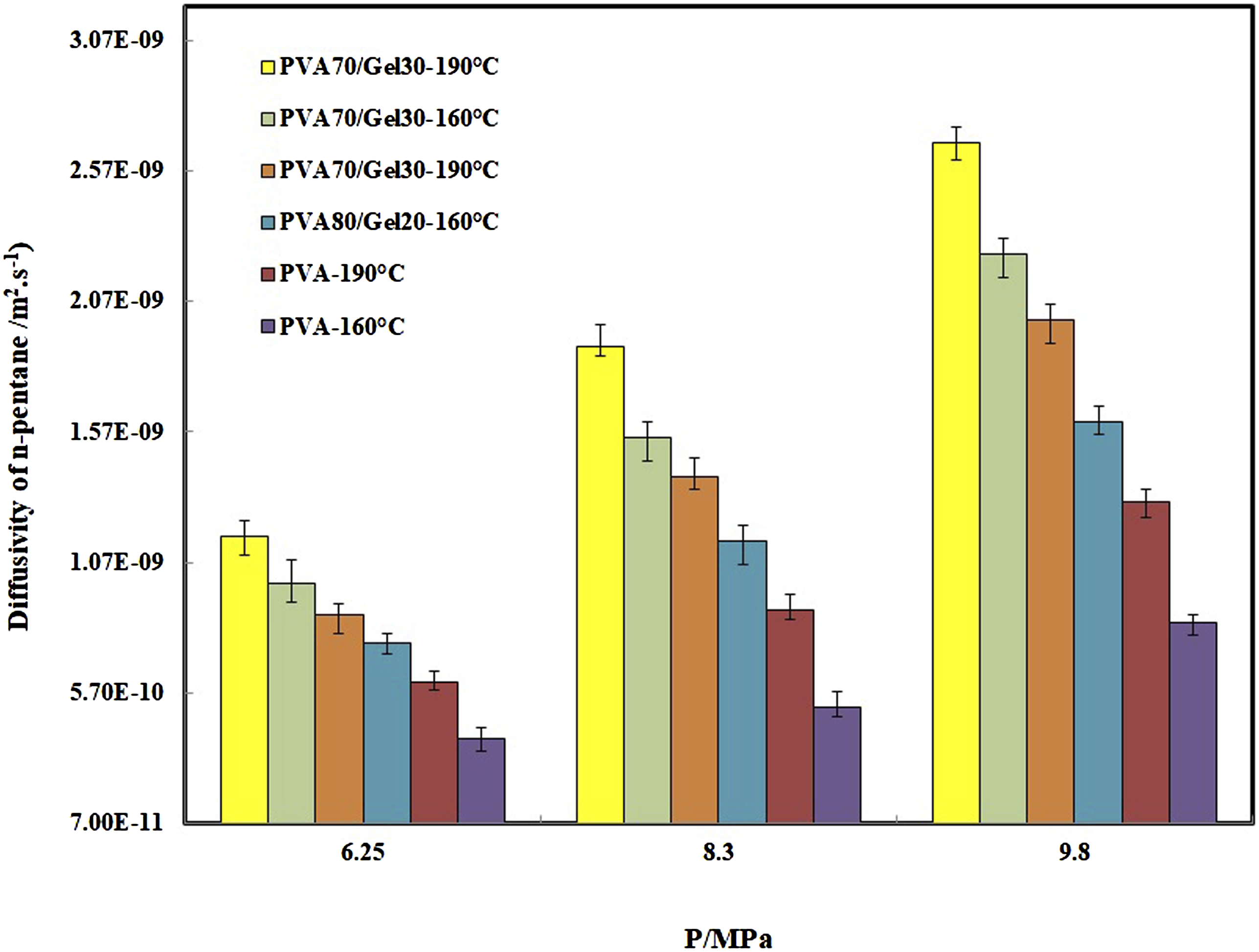
This behavior is due to destroying the crystalline structure in PVA due to presence of Gelatin in which will effect on the diffusivity results. The relationship between dissolved n-pentane in samples with time was calculated for all samples and for instance, is just shown for PVA/n-pentane in Figure 8. The symbols are the experimental data and the solid line signifies the determined value by using equation (3) and in the following the results of n-pentane diffusivity in samples are showed in Figure 9. In general, the diffusion coefficient of pentane in polymer depends on the concentration of pentane dissolved in the polymer. This is because the dissolved pentane induces swelling of the polymer and changes the specific volume of the polymer. Therefore, strictly speaking, since the swelling occurs during the diffusivity measurement, Fick’s second law cannot describe the diffusion process when the swelling effect is large. Thus, by making the stepwise change in pressure for diffusivity measurements small enough to make the swelling small, which in this study is 1 MPa, the mutual diffusion coefficient can be treated as a constant parameter during the measurement and given as a function of the n-pentane concentration of a final steady state. It is clear that in Figure 9, the diffusion coefficients are increased with the temperature, pressure and Gelatin content, which the diffusivity of n-pentane in PVA is the lowest value and in PVA70/Gel30 is the highest at the same temperature and pressure. Also with addition of Gelatin due to decrement in the sample crystallinity (Figure 1), the specific free volume free volume in polymer increased (Figure 7) and n-pentane can diffuse into the polymer matrix easily. In this case, due to the loss of strong crystalline structures of PVA, n-pentane has not any barrier for diffusing into the polymer blends. At the same temperature and amount of pentane, the presence of gelatin in the blends causes greater mobility of polyvinyl alcohol chains and thus increases the specific free volume (Figure 7). There are also similar behavior for solubility and diffusivity of n-pentane in polymer, so that the value of these thermodynamic parameters increases with the addition of gelatin in blends. Therefore, there is the relationship between specific free volume, solubility and diffusivity parameters. Relative quantity of dissolved n-pentane versus time for Poly vinyl alcohol. The solid lines are predicted by equation (4). Diffusivity of n-pentane in Poly vinyl alcohol/Gel blends.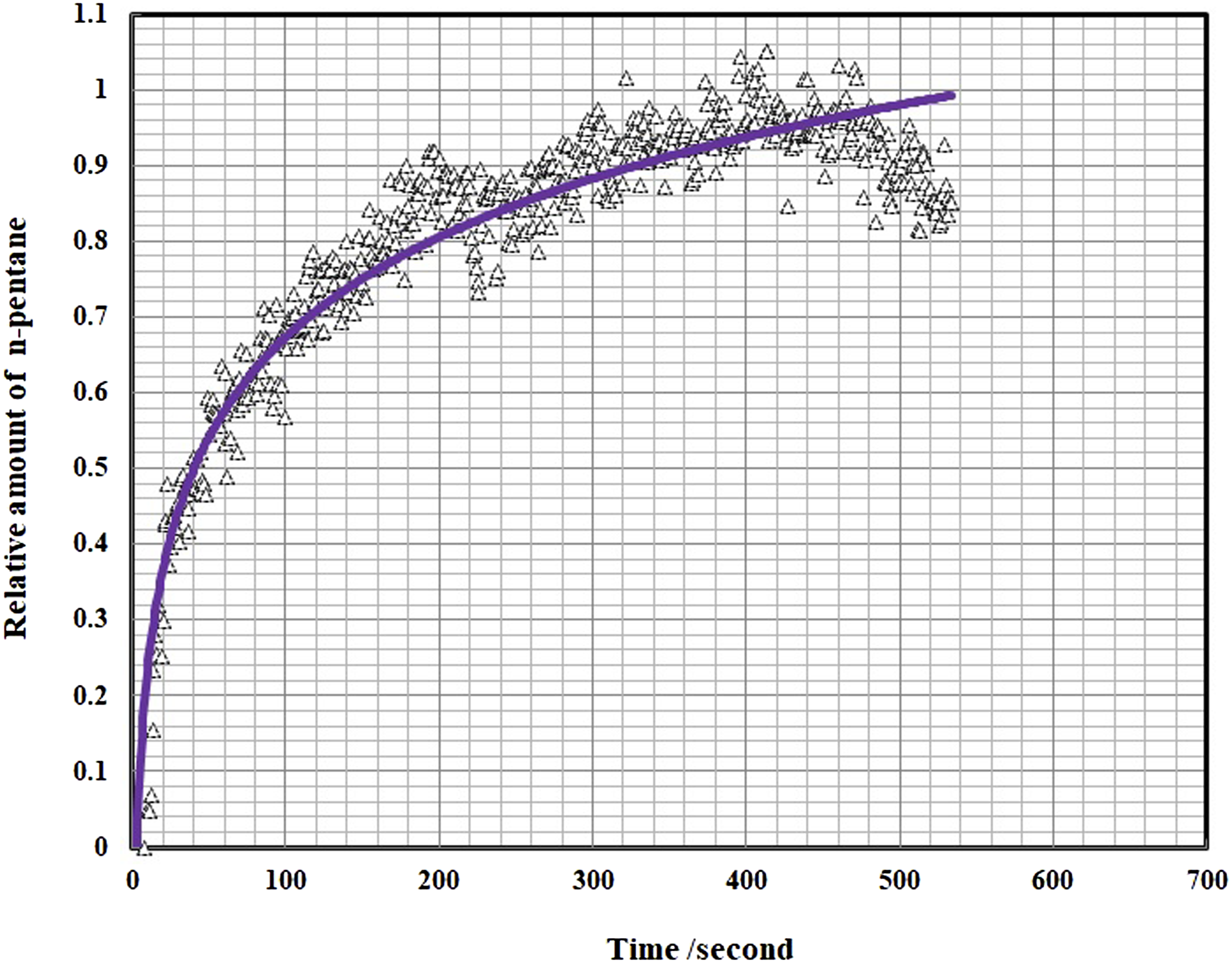
Conclusion
The solubility and the diffusivity of n-pentane in PVA/Gel blends were calculated at different temperature and pressure using the MSB system. Combining the XRD and FTIR results, it was shown the Gelatin as a plasticizer could suitably interact with PVA. In addition, the SEM/EDX analysis showed the suitable dispersion of gelatin in PVA/Gel blend. The solubility of n-penatne in PVA/Gel blends increased with pressure and decreased with temperature. As well, it was concluded that there was relationship between the specific free volume of polymer with the solubility and diffusivity parameters which with increasing of the specific free volume, the solubility and diffusivity of n-pentane in blends increased. Due to the effective presence of gelatin in the blend, the PVA processability window is increased and in addition to increment in gas solubility and the specific free volume data, gas diffusivity into the polymer matrix, was also increased. The application of PVA/Gel blends is in tissue engineering in which the PVA/Gel blends foams or scaffolds were produced via the batch foaming process. High porosity in biopolymer scaffolds is very important in tissue engineering applications. In order to achieve high porosity, the solubility and diffusivity of the foaming agent like n-pentane in the polymer should be improved. In this case, more n-pentane could dissolve and diffuse into the PVA/Gel blends and the open cell structure with high porosity will be produced after the foaming process. In the present work we investigated the thermodynamic parameters nd obtained the solubility and diffusivity of n-pentane in PVA/Gel blends in different temperature and pressure ranges. The determined thermodynamic data in this study, will be used in further research work in the case of the PVA/Gel blends scaffolds.
Footnotes
Acknowledgements
The author would like to acknowledge the Iran National Science Foundation (INSF) for the financial support of this research project with project number of 99010405.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Iran National Science Foundation (INSF) [grant number 99010405].