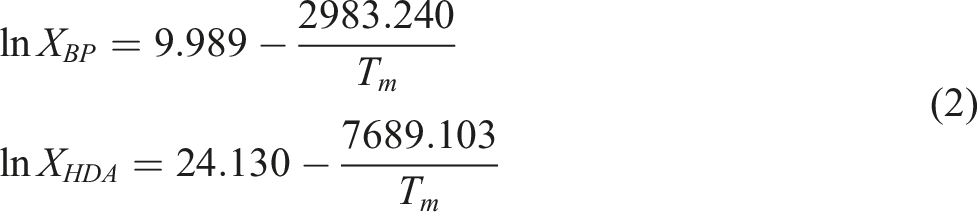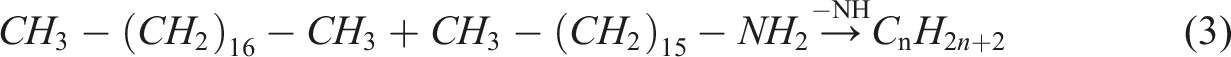Abstract
Binary paraffin has attracted much attention because it can control the phase change temperature range and has good flexibility. To improve the latent heat effect, a modified binary paraffin
Keywords
Introduction
Thermal energy storage (TES) technology is an effective mean to recover the rich thermal energy and improve the energy utilization rate. Thermal energy storage can be roughly divided into two types: thermal and latent heat. Due to the outstanding characteristics of the phase transition process, the latent thermal storage using phase-change materials (PCMs) is preferred. The PCMs can store and release a certain amount of latent energy1–3 at an almost constant temperature during its phase transition, effectively preventing rapid thermal fluctuations during phase transitions and ultimately improving indoor thermal comfort by stabilizing the indoor surface temperature. To meet the requirement of a living environment, the phase transition temperature shall be kept in 22–26°C. 4 Paraffin is a promising TES candidate, 5 which has high potential heat, good thermal reliability, low volume change and low over-cooling during phase transition. However, most phase transition temperature of paraffin exceeds the required room temperature range, and the phase transition interval is relatively fixed, which cannot meet the heat storage and temperature control requirements of specific temperature conditions. 6 In addition, it has defects of low thermal conductivity and easy leakage, limiting its wide applications.
In order to broaden the application field of paraffin as PCMs, researchers committed have been trying to adjust its phase transition temperature. Usually two or more organic phase-change materials are mixed in a specific ratio to achieve the required phase-change temperature and control the phase transition interval.7–9 Shilei and Sari10,11 prepared a binary low co-melting mixture, which has a suitable phase transition temperature, high phase transition potential heat and stable thermal performance, and has a great potential for building energy saving applications. Yu 12 prepared a paraffin-stearic acid binary phase-change material by melting and mixing different materials. It is found that when the mass ratio of paraffin to stearic acid was 17:8, the minimum phase-change temperature was 52.5°C. The highest phase transition temperature can reach 68°C by expanding the phase-change temperature interval. The pure paraffin showed a low thermal conductivity of approximately 0.2 W/(m·K). To improve the thermal conductivity of pure PCMs, various thermal conductivity materials were used, including mixing graphite (EG) matrix 13 and saturated aluminum foam 14 with PCMs. Nourani and Liu et al.15,16 dispersed thermal conductive material nanoparticles into paraffin and found that the improvement of thermal conductivity of paraffin by nanoparticles. To overcome the leakage of PCM, researchers prepared a stable phase-change materials, a porous material, 17 to encapsulate the organic PCMs, and coating a layer of film with stable performance on the surface can effectively solve the leakage problem. 18 The above research shows that the thermal properties of phase-change materials can not fully meet the actual production requirements, and when it is limited to the carrier, the conductivity of PCMs is very low, which cannot produce the expected working temperatures. In the actual thermal energy application research, to maintain the temperature balance of the environment, Taeyeon Kim et al. 19 added PCMs to the exterior wall, which can delay the indoor peak temperature by 3 h. A large number of research results show that adding PCMs to exterior walls, roofs and floors can significantly reduce the amplitude of indoor temperature fluctuations.20,21
In this study, a new high sub-thermal phase-change material hexadecylamine and binary paraffin were mixed into new PCMs under the action of nanoparticles. Mixing nanoparticles to improve the heat conduction coefficient of the PCMs. 22 The thermal properties of phase-change materials were tested by characterization. In addition, it was investigated that the thermal conductivity of different mass fractions of nano-graphite, nano AlN and copper powder were mixed with PCMs respectively, and adsorbed to ceramsite carrier. To prevent leakage, ceramsite is used as adsorption carrier, and AlN + benzene emulsion secondary package is used. The thermal stability is tested through a high and low temperature cycle. This co-crystal PCMs improve the latent heat value of binary paraffin, enhance the heat transfer rate of phase-change materials, and improve shape stability and can be used repeatedly. The new materials can be used in greenhouses and buildings, to improve energy utilization, efficiency.
Experiment
Preparation of multi-compound phase change materials
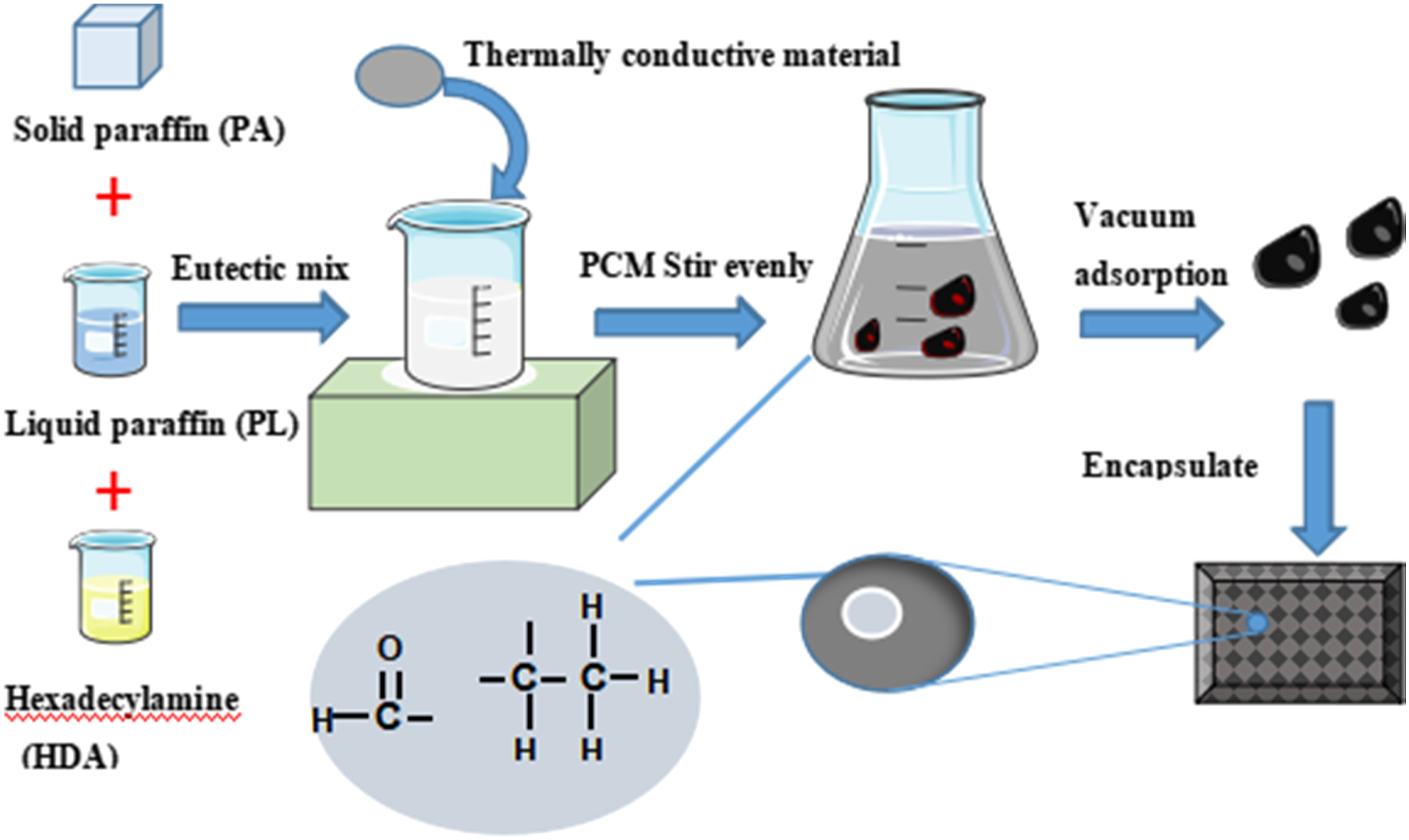
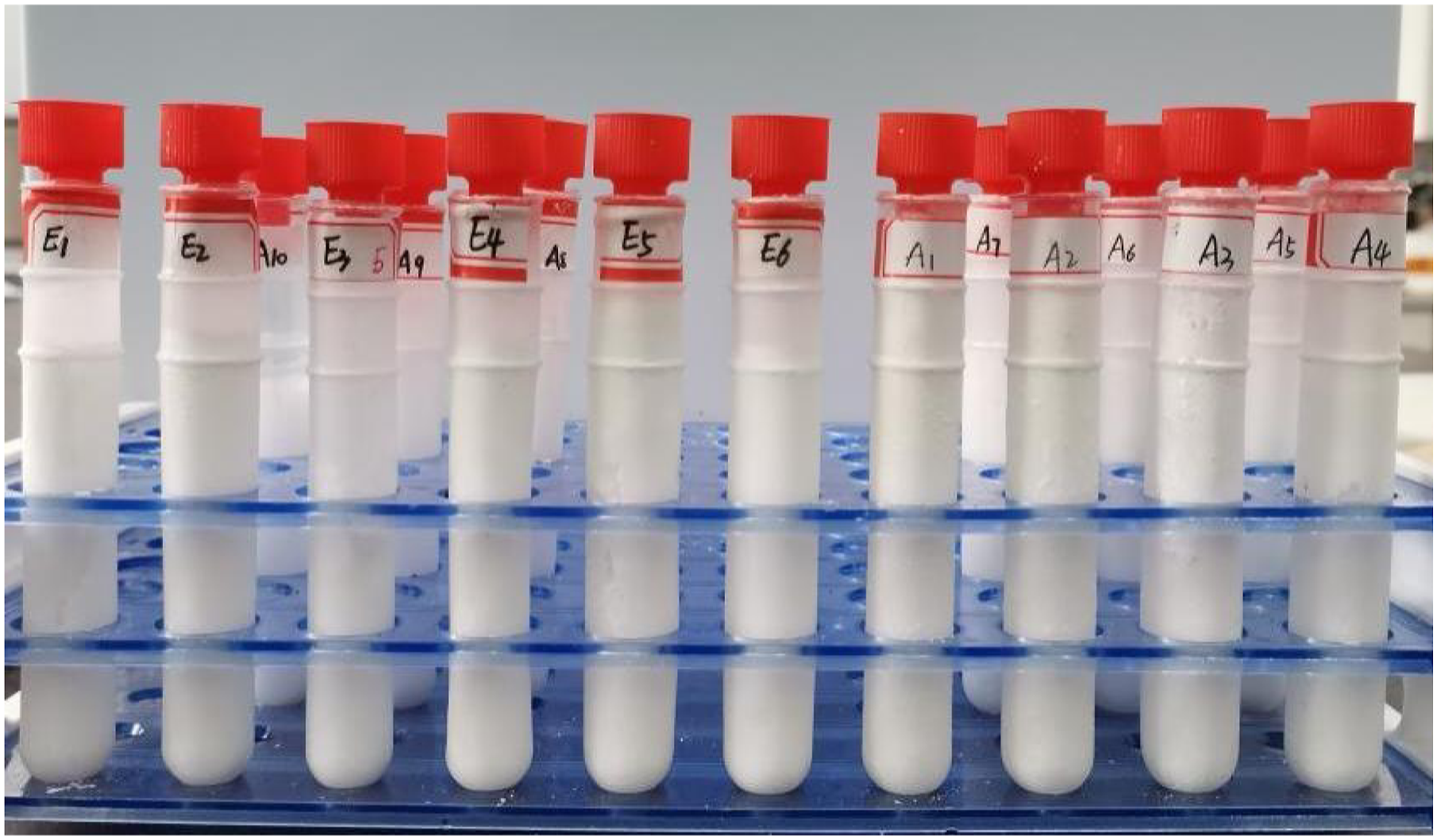
The process of preparing multiple composite phase-change materials (CPCMs) by fusion mixing method is as follows, and the preparation process is shown in Figure 1. (1) Paraffin (PA) (phase-change temperature TP = 51°C, phase-change latent Δ H = 221.37 J/g) and Hexadecylamine (HDA) (TP = 45.5°C, Δ H = 265.29 J/g) are put together into a constant temperature drying box and melt it to liquid for standby. (2) Weigh PA and liquid paraffin (PL) with different mass fractions and melt and mix them in a constant temperature magnetic stirrer at 70°C to form binary paraffin (BP) phase-change material. The phase-change temperature range is 25°C–54°C. (3) Place PA, PL and HDA in a beaker in a 70°C water bath in a stirrer, set the mixing speed of 20 r/min, and pour them into a test tube after mixing evenly. The sample of multiple phase-change material is shown in Figure 2. A mixture of #5 BP (PA accounts for 50% of BP) and HDA is mixed evenly. The mass fraction of BP added is 10%–90%, the step is 10%, and the number is A1 ∼ A9. Because BP has the advantages of low melting point, large phase transition range and low cost, a group of PA: PL: HDA mass ratios are added, which are 35:30:35, 40:30:30, 50:15:35, 50:18:32, 50:20:30 and 50:25:25 respectively, numbered E1 ∼ E6. (4) BP (5) Attach the mixture to porous ceramsite (with particle size of 1–8 mm and main components of SiO2, Al2O3 and Fe2O3), place in the reactor, heat in a water bath and vacuum, stirred at 120 r/min, adsorb under negative pressure for 1 h, and take out after saturation, so as to obtain the CPCMs. (6) Encapsulation of part of the CPCMs are added to the styrene acrylic emulsion and nano AlN solution, stirring continuously to the surface of each ceramsite, then extracted and put in the air for natural curing, and finally package and shape the mixtures. Preparation process of multi-component CPCMs. Sample diagram of multi-component PCMs.
Test and characterization of multi-component CPCMs
(1) The fluctuation of the material temperature in a complete cycle was recorded by the temperature recorder. (2) The phase-change temperature and phase-change latent heat values of the multi-component PCMs were tested using a DSC. A PCMs sample is preferably placed in a DSC, and the heating temperature range is 25–60°C under a nitrogen constant gas stream, and the heating rate is 5°C/min. The experimental data is processed to produce DSC graphs that can analyze the phase-change temperature of the PCMs (the intersection of the front edge maximum slope line and the peak baseline) and the phase-change latent heat (the area enclosed by phase peaks). (3) SEM is mainly used to perform micro-structural analysis of blank ceramsite and CPCMs after ceramics. The control and composite shaped phase-change materials were polished into powder, and the sample was performed on the sample, 300 times under the electron microscope, and the samples were observed using SEM, and different micro photos were obtained. (4) The crystal structure analysis of each pure substance and the mixed PCMs were tested using FTIR, with a 4000–400 cm−1 test frequency and 4 cm−1 resolution. Each sample was scanned 6 times. First put the pure potassium bromide sheet and the sample flakes in the samples holders, in which the former was placed in the background area, the latter was placed in the test optical path, which was covered to ensure that the test environment is stable. (5) The powder sample was spread on the sample frame, and the material was scanned with a D/MAX-2400 X-ray diffraction tester (XRD). The scanning step was 0.02°, and the scan range was 5–80°. (6) The samples after adsorbed CPCMs and the packaged PCMS were placed in a high and low temperature cooling cycle incubator for a certain period of time to conduct speedy melting and solidifying experiments. Repeat the process, compare changes of samples mass and thermal parameters at the cycles of 1, 100, 200, 300, 400, and 500.
Multi-eutectic system mechanism and results forecast
A phase-change medium of eutectic system mixed in the proportion of co-crystal points is stable and, like the phase transition performance of pure material, has a determined single melting point and phase change latent heat, which is lower than that of each individual component.
23
On the basis of the second law of thermodynamics and the phase equilibrium theory, Zhang
24
deduced the liquid phase equation of the N element co-crystal system
Equation (1) is for the binary co-crystal system, which is also applicable for the calculation of N-element co-crystal system.
25
In this paper, the thermal performance test of HDA and BP was conducted. DSC test results showed that the melting temperatures of HDA and BP were 45.5°C and 25.5°C, respectively. Substitute the thermal property parameters corresponding to BP and HDA into equation (1) to obtain the BP
The co-crystal equilibrium phase diagram of the BP Eutectic phase diagram of multi-component PCMs.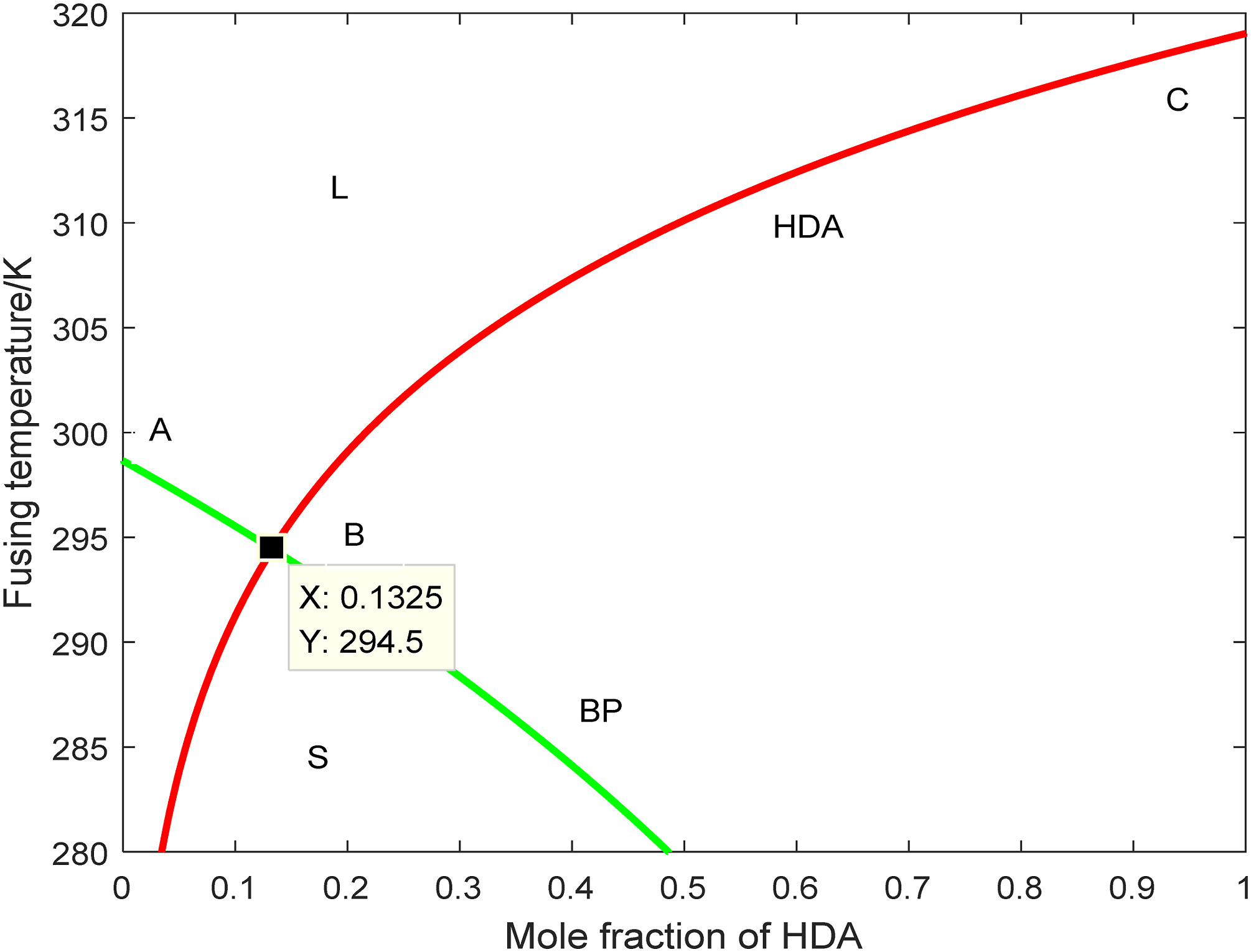
As can be seen in Figure 3, the quantity ratio of the theoretical material of BP
Results and analysis
Multi-component PCMs heat storage and release characteristics
Figure 4 shows the temperature fluctuation of PCMs in a complete process of heat storage and release. The BP Heat storage and release curve of multi-component CPCMs.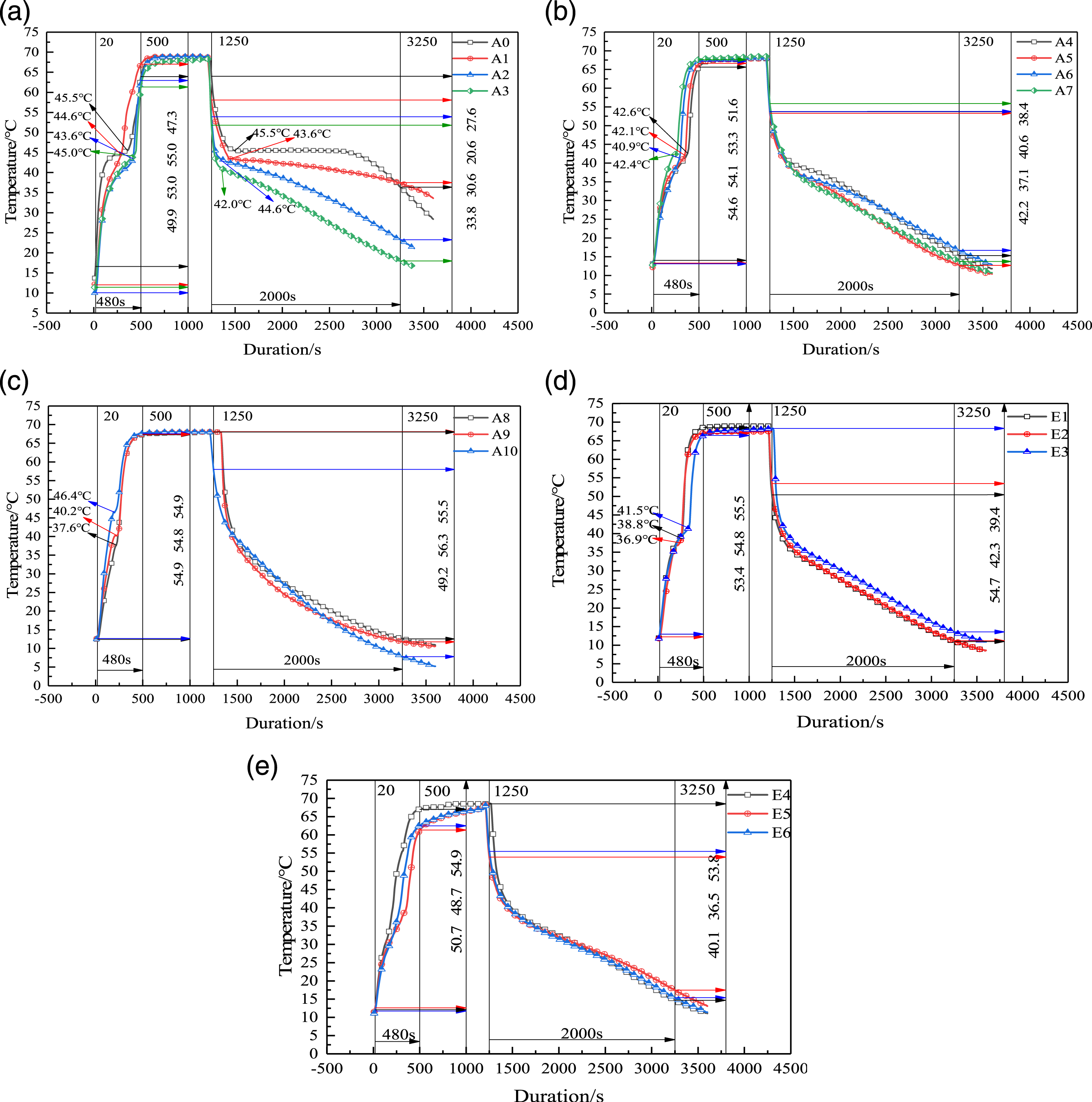
In order to more intuitively analyze the heat storage and release rate, the heating and cooling rates of multi-component PCMs are shown in Figure 5. The heating speed of A1 and A9 is faster, and E1 to E6 shows a descending trend as the increase of PA, and locally bulges at E4. The cooling rate of A1 ∼ A9 shows an upward curve with the increase of BP. With the proportion of PL gradually increases, the temperature changes faster. The heating and cooling rate of A9 is the fastest, which is consistent with the results of theoretical analysis. The heating and cooling rate of E4 is also appropriate. Statistics of temperature rise and fall rate of multi-component PCMs.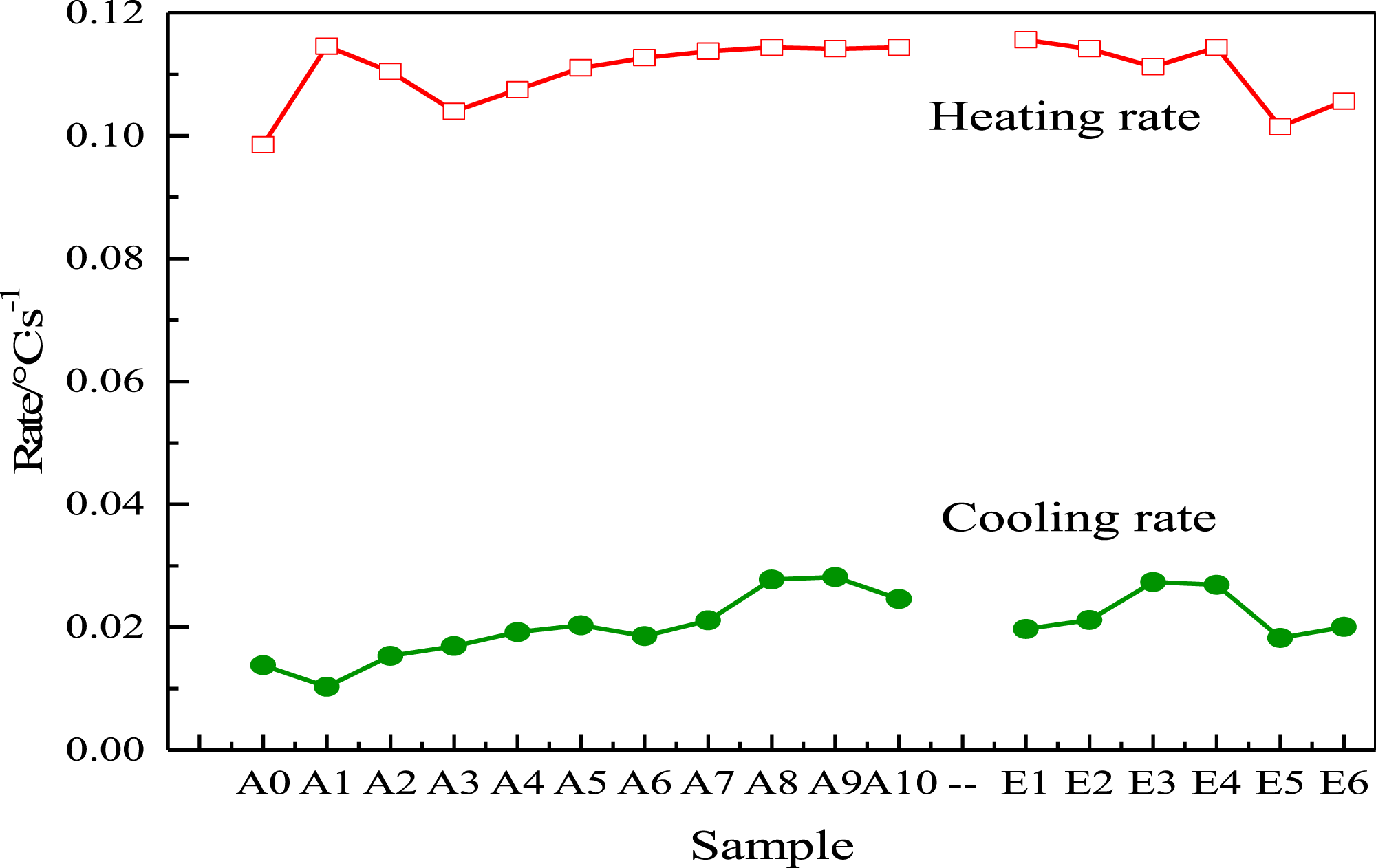
Multi-component PCMs DSC analysis
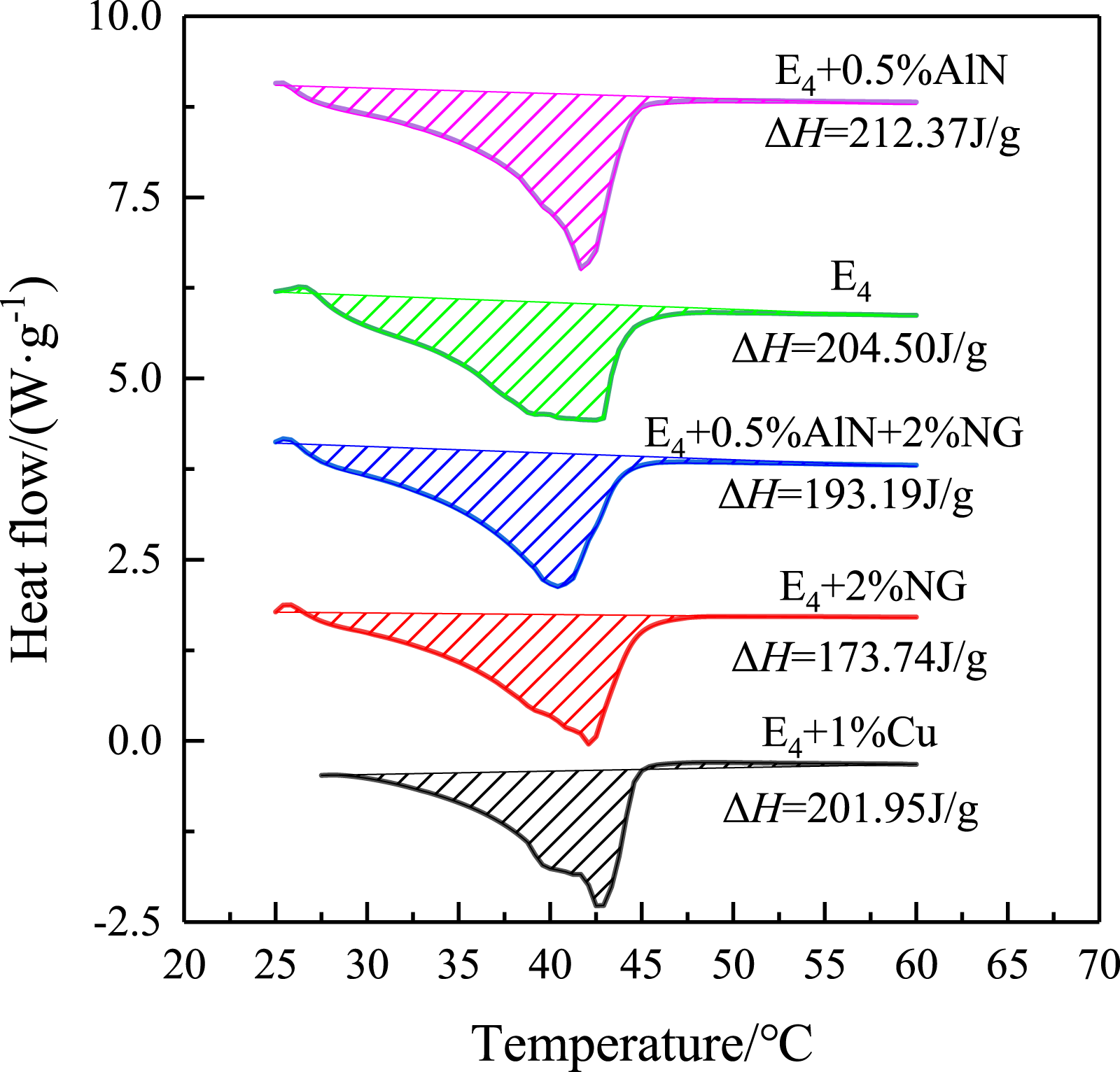
The DSC curves of A0 (HDA), A10 (# 5 BP), A9, and E4 multi-component PCMs are shown in Figure 6. From the figure, A0 has a high latent heat value, the phase-change temperature is also higher than 45.5°C, which is consistent with the heat temperature curve. A9 phase-change temperature is much in advance, but the peak area is smaller and the latent heat effect is poor, and there are two clear peaks, indicating that it is not completely mixed in this ratio. The peak area of E4 is more protruding than A9. The starting temperature of endothermic peak is 25.0°C, and the ending temperature is 47°C. With the increase of HDA, the peak temperature is 44°C. The enthalpy of phase transition of E4 is calculated from the area of endothermic peak ΔH is 204.50 J/g. Due to the existence of HDA, the latent heat value of BP system increases significantly. The room temperature condition is still satisfied, and a solid-liquid phase peak appears at around 40°C. The three components can produce a good eutectic system under the ratio of E4, which shows that E4 has good heat storage effect. DSC curves of multi-component PCMs.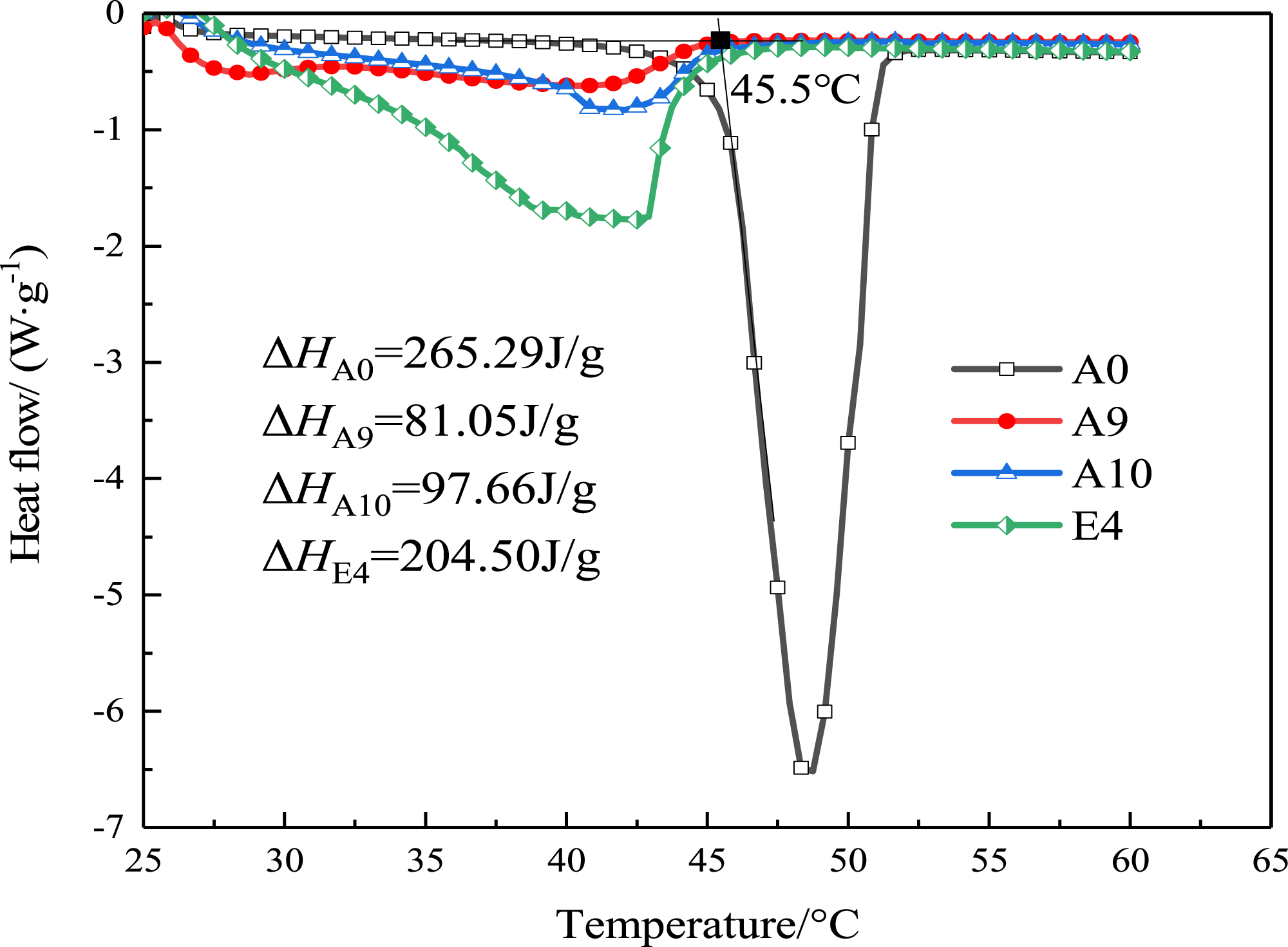
Thermal material analysis of modified multi-component PCMs in thermal conductivity
The PCMs and nanoparticles are combined to improve the effective pathway of the heat conductance coefficient of the PCMs. Due to the smaller particle size of the nanopowder, the Brown motion of the multivariate PCMs molecules can overcome the gravity effect, so the stability of the nanofluid is better.
22
A mass fraction of 0.5%, 1%, 2%, and 5% of nano graphite, nano AlN, and copper powder were added to E4 respectively. The results of temperature change and DSC test were performed by melt mixing. The results obtained are shown in Figures 7 and 8. Statistical diagram of heat storage and release of modified multi-component CPCMs. DSC curves of multi-component CPCMs after modification.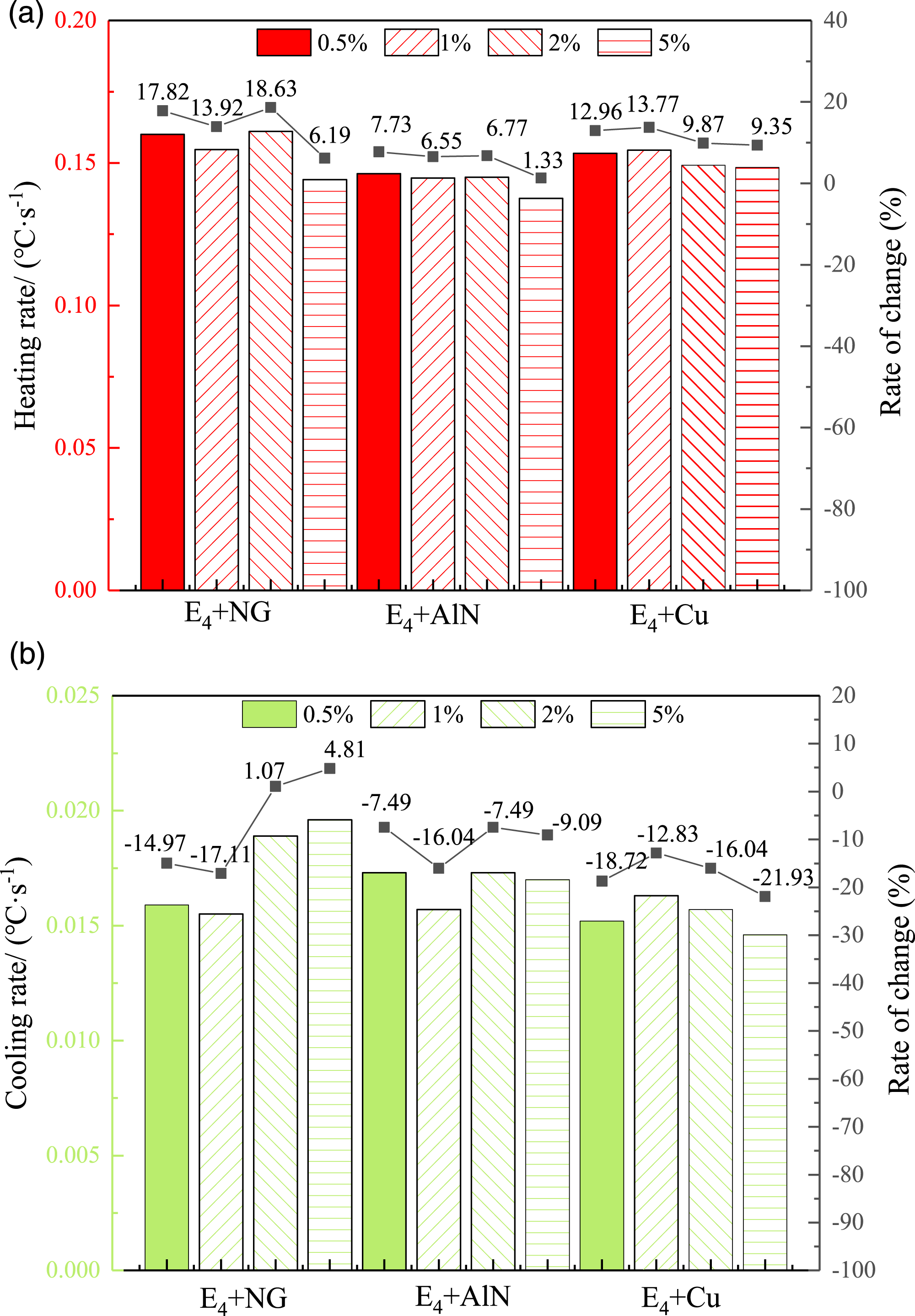
As shown in Figure 7, after adding thermal conductive materials, the heating rate of multi-component PCMs is accelerated, and the cooling rate is partially delayed, because the material itself has the characteristics of fast heat absorption and slow heat release. The same conclusion is reached for modified PCMs. It can be seen from the broken line diagram that the more heat conductive material reinforcement is added, the better. Adding nano graphite, nano AlN and copper powder, the mass fractions are 2%, 0.5% and 1% respectively. Compared with E4, the temperature change in unit time is the most obvious. However, the temperature change of the mixture of copper powder group slows down in the cooling stage. Due to the large particle size of copper powder, it is not evenly dispersed among multi-component PCMs molecules, which does not improve the heat release rate of PCMs. In order to better enhance the thermal conductivity of PCMs, nano graphite with mass fraction of 2% and nano AlN of 0.5% were added to E4. As a result, the improvement of comprehensive heating and cooling effect is the most significant. The two kinds of magnetic fluid PCMs with uniform particle size can be mixed into nano-sized PCMs.
The DSC graph of the modified mixture is shown in Figure 8. It can be seen that 0.5% of the nano AlN in the E4 ΔH is higher, and the mass fraction of nano AlN is small, which has little effect on the latent heat value, or even partially increases. Since nano AlN has strong metal polarity and small particle size, which is easy to adsorbed with the organic molecules, and part of the latent heat contained in AlN is accumulated. The contrast curve can be found that the phase-change occurs after the addition of the thermal conductive material. The temperature and heat transfer peak are moved forward and accelerate its heat storage rate. There are no other peaks in the heating process, indicating that the modified multi-component PCMs do not experience crystallization. Adding 0.5% nano AlN as the modifier of E4 has good energy storage effect.
Analysis of chemical composition and crystal structure of multi-phase mixture
Chemical composition of PA, HDA and BP FTIR of HDA, PA and E4.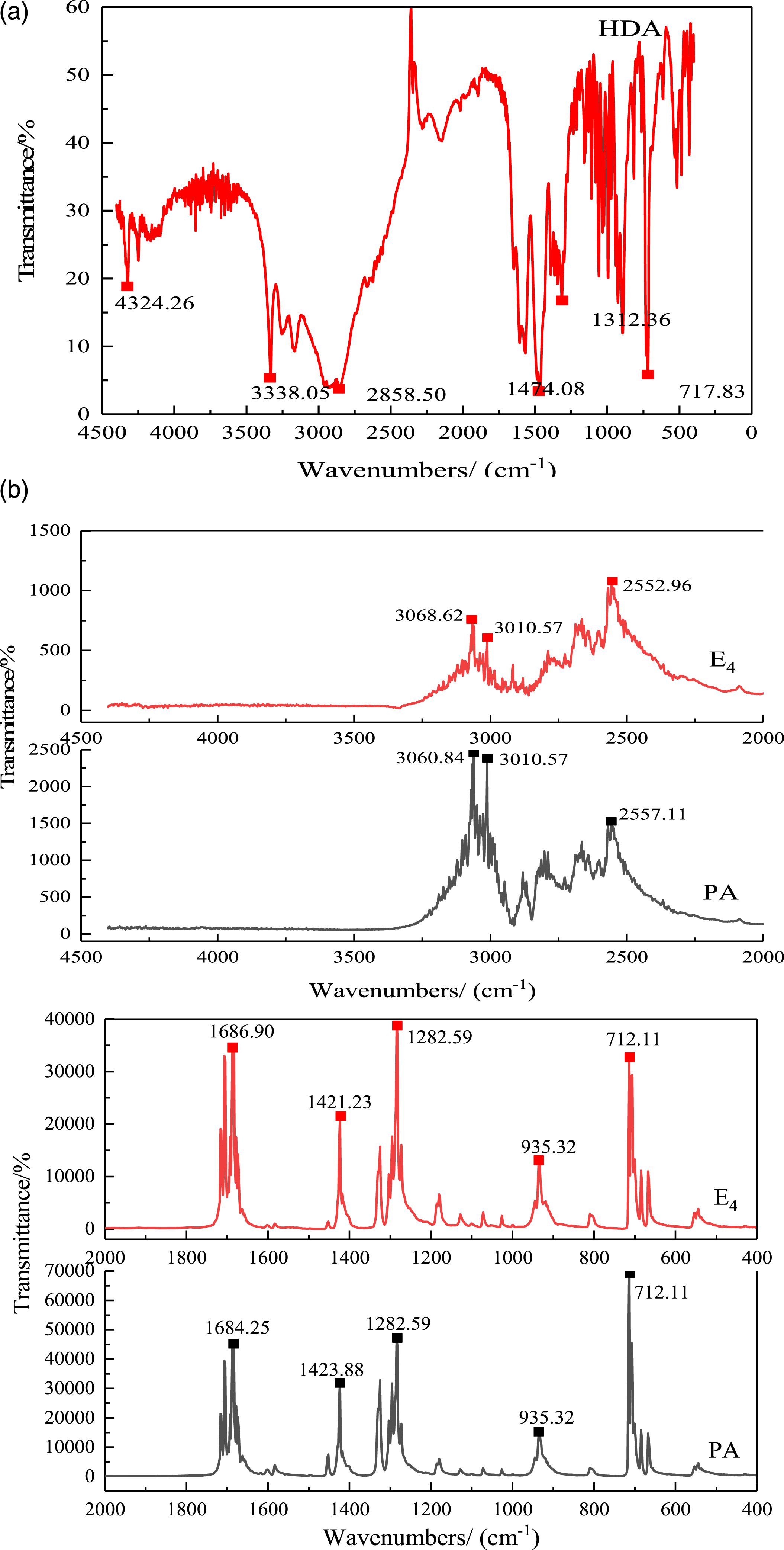
The crystal structure of the multi-component PCMs were analyzed using XRD, as shown in Figure 10. The strong diffraction peaks of organic PCM are located at 21.0° and 40.0°, for the PA XRD diagram of multi-component PCMs.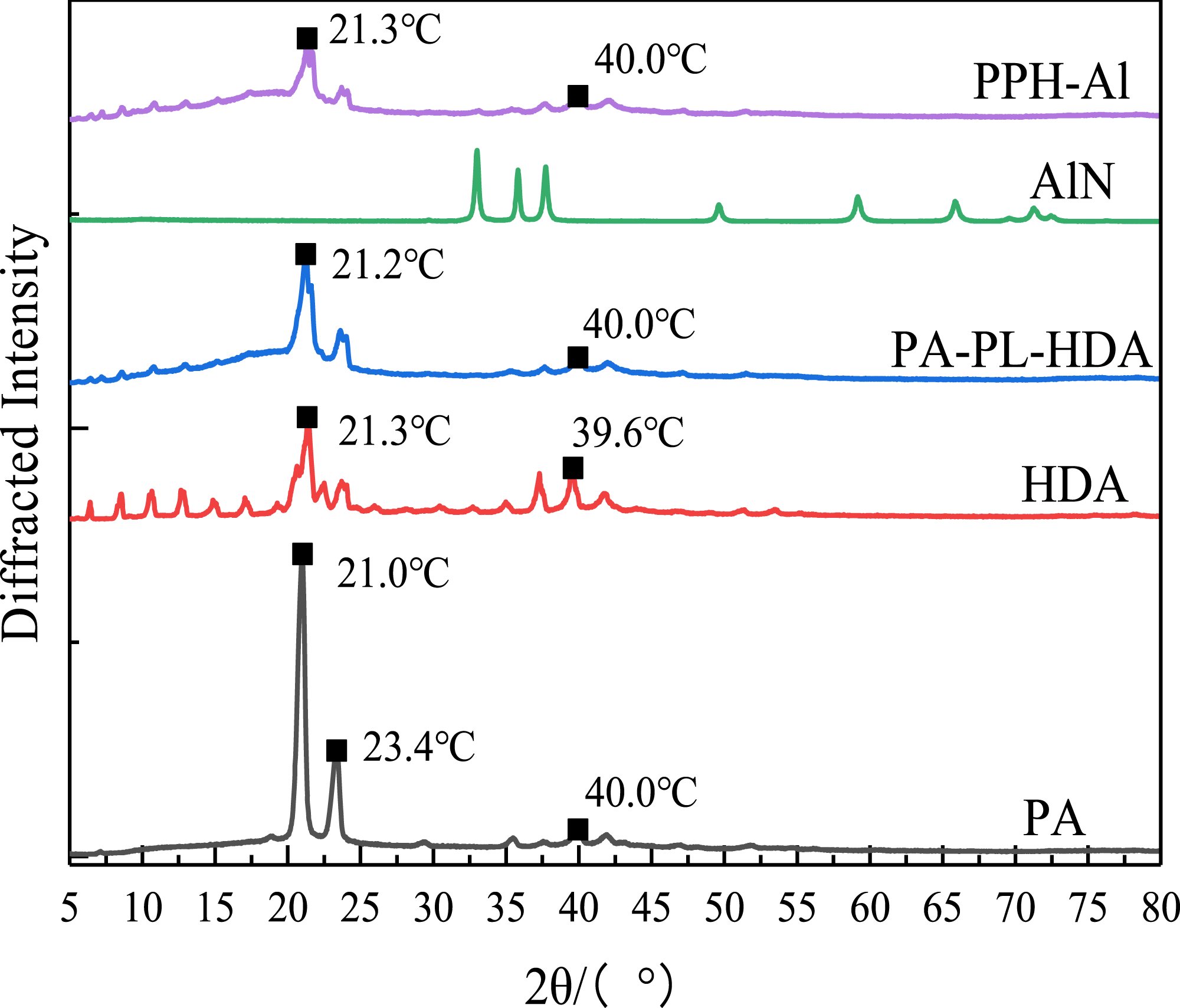
E4 (PA: PL: HDA = 50: 18: 32) is a mixture of organic carbon atoms where paraffin has a molecular formula C18H38 and fatty amine HDA has a molecular formula C16H35 N. The two system eutectic molecular formulas is
SEM law
Although the ceramsite has strong adsorption capacity, it is impossible to adsorb indefinitely. It is necessary to determine the suitable mass ratio of BP Seepage diagram of CPCMs sample. Exudation statistics of CPCMs samples.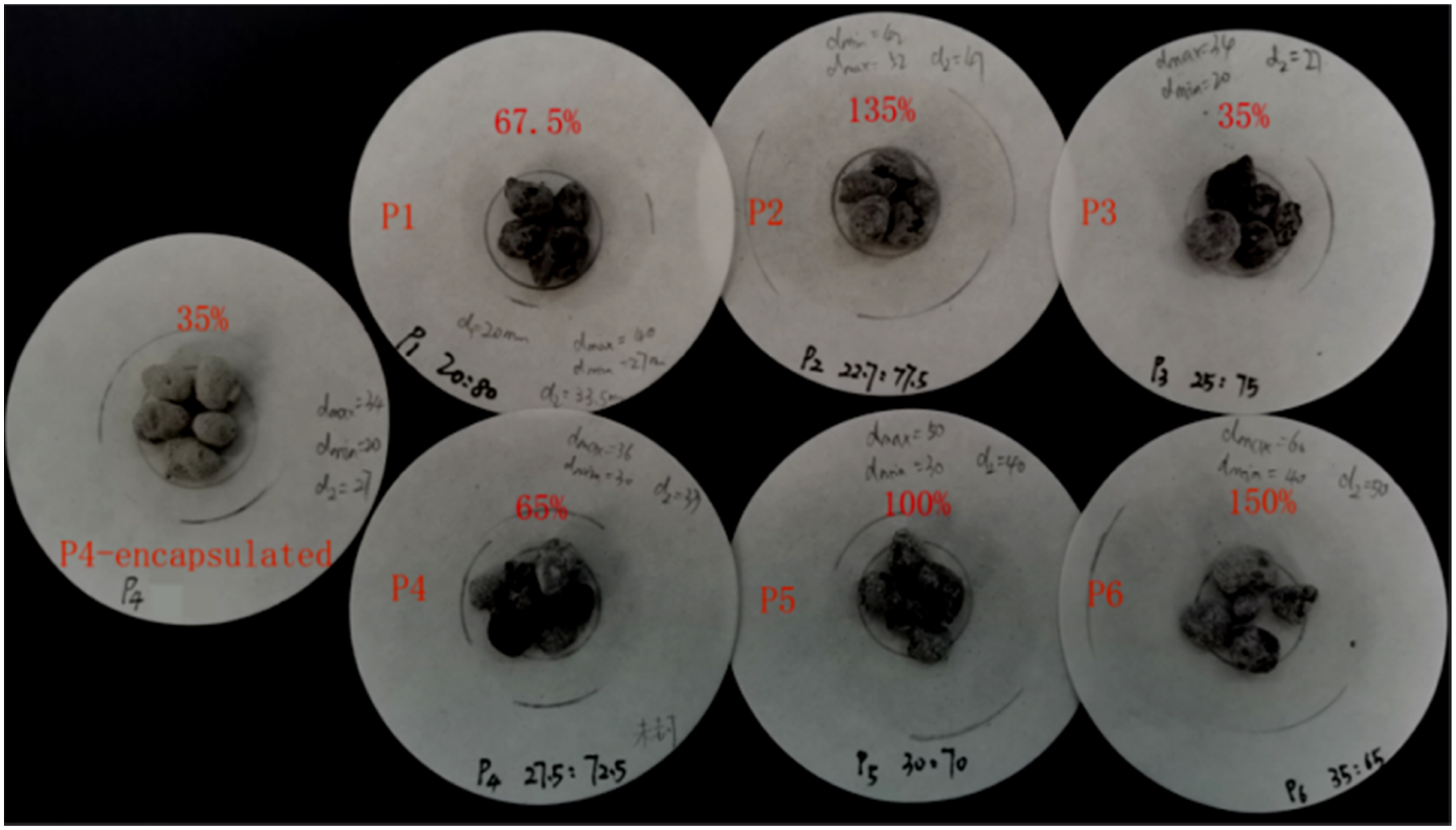
Figure 12 shows the microscopic surface morphology of ceramsite and CPCMs. Most of the appearance features of ceramsite are round and oval spheres, sintered from clay, dark red, and a hard shell on the surface. The microscopic morphology is as shown in Figure 12(a) and (b), which are magnified by 300 and 500 times respectively. It can be seen that the interior of the ceramsite is fine honeycomb like holes, and there are a large number of micropores on the hole wall, which are evenly dispersed. The hard shell and internal porous structure provide stereotyped support for the multi-component PCMs. SEM of multi-component composite PCMs.
Figure 12(c) and (d) show the micro morphology of the CPCMs. Under the condition of vacuum and negative pressure, PCMs are uniformly wrapped in the pores by ceramsite, the internal adsorption is saturated, and there are partial PCMs adhesion on the outer wall. The capillary effect of ceramsite fixes the multi-component PCMs on the carrier to prevent its leakage. The CPCMs were combined with styrene acrylic lotion + AlN for secondary packaging to reduce BP—HDA leakage. The seepage diagram and micro-structure diagram show that the shape of the CPCMs is stable.
The rule of thermal stability
Thermal stability is one of the important parameters of CPCMs, which evaluates the durability of PCMs in practical applications. To analyse the thermal stability of CPCMs, the phase change temperatures, latent heat, quality, etc. of CPCMs are evaluated before and after 500 cycles of heating and cooling. Figure 13 shows a DSC curve before and after 500 cycles, and the relevant results are shown in Table 2. DSC diagram of multi-component CPCMs after cycling. Thermal parameters of CPCMs before and after 500 cycles.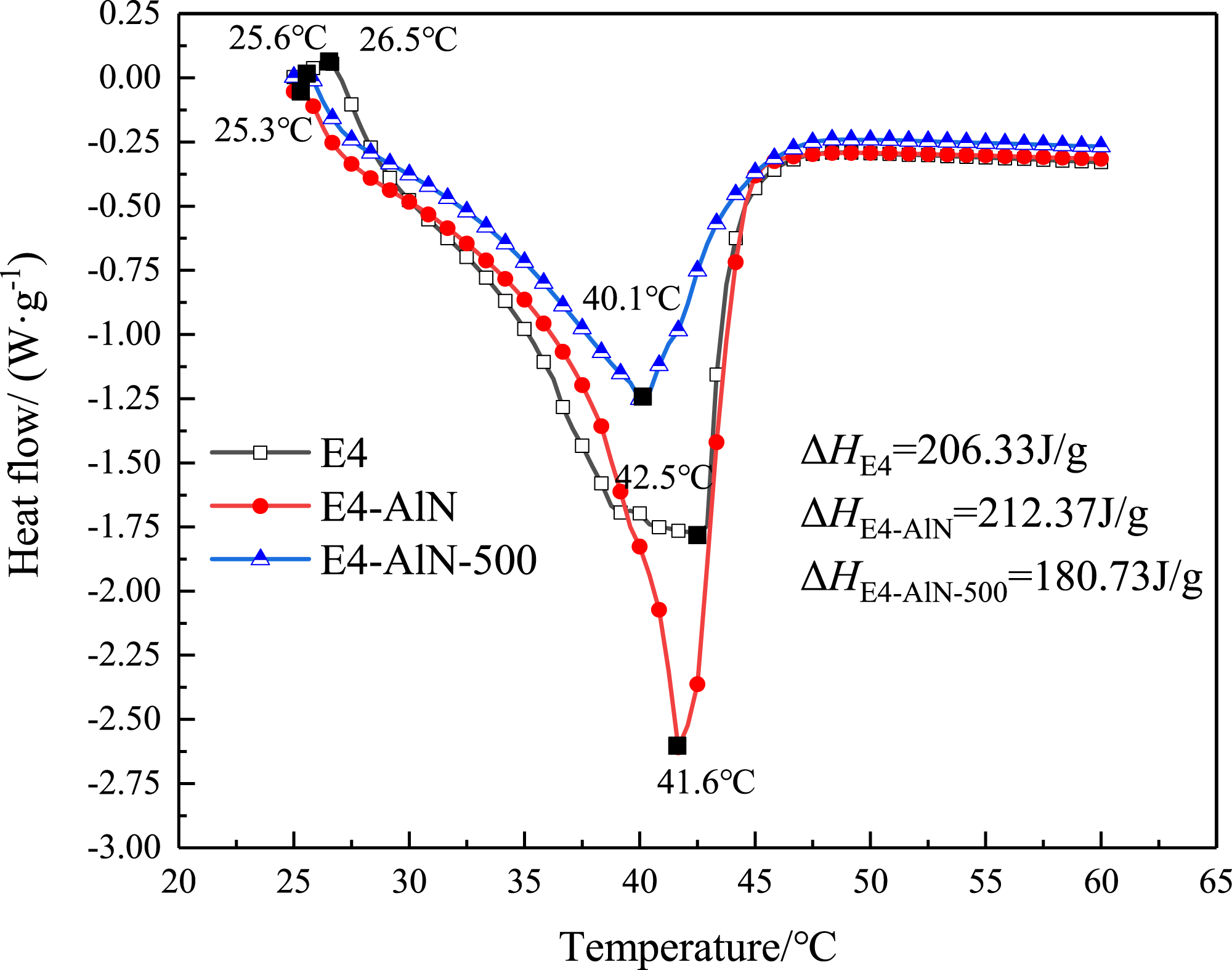

As shown in Figure 13, the melting temperature (Tm) before and after 500 cycles is 25.3°C and 25.6°C, respectively, and the corresponding ΔH is 212.37 J/g and 180.73 J/g. Comparing the values measured before and after the heat cycles, the difference between the melting temperature is 0.3°C, but the latent heating of CPCM after 500 thermal cycles changed 14.9%. A weight loss test was performed with a test temperature range from room temperature to 60°C. Because of the leaking, the weight of the CPCM reduced by 0.695% after 500 cycles. The material loss rate can be significantly reduced after adding a package. In ceramsite, the secondary package of AlN + benzene can prevent leaking and has good thermal reliability.
Conclusions
This paper prepared the BP (1) DSC and temperature curves indicated that PA, PL, and HDA with various mass fractions form an eutectic organics mixture. The latent heat value of BP increased with the addition of HDA. When PA, PL and HDA were mixed at a mass ratio of 50:18:32, the eutectic mixture had a high latent heat (204.50 J/g), and its phase-change temperature (26.5°C) was close to room temperature. To accelerate the storage rate, 0.5% AlN was added to the mixture and the rate per second can be increased by 7.73%, and the latent heat was 212.37 J/g, and the melting temperature was 1.2°C lower. It has good energy storage effect. (2) The results of FTIR and XRD confirmed that the BP (3) Taking into account the good adsorption capacity of ceramsite, the BP (4) By adsorption into ceramsite, the thermal stability of the eutectic mixture was improved. After 500 heating-cooling cycles, the phase change temperature and mass of CPCMs did not change significantly, and there was a small loss of phase change latent heat. In general, CPCMs have good thermal stability. In addition, a secondary package of nano AlN + benzene can further reduce the mass loss of CPCMs to 0.06%, because it can prevent the PCMs liquefaction on the ceramsite surfaces. In summary, the synthetic CPCMs have the advantages of appropriate phase-change temperature, high heat storage rate, thermal stability, good thermal conductivity and long service life, which can be used in building energy saving, greenhouse rear walls and many other fields.
Footnotes
Acknowledgement
This work was supported by Hebei Province key research and development plan project (21327209D). The authors greatly appreciate their financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hebei Province key research and development plan project (21327209D). The authors greatly appreciate their financial support.