Abstract
The improvement of properties has been pursued through the addition of an immense variety of fillers in polymers. In this work we studied the effect of lamellar zirconium phosphate (ZrP) on the physico-mechanical properties of polyamide-6 (PA-6). The ZrP was synthesized and incorporated into PA-6 matrix at 1, 2 and 3 wt.%. Structural, thermal, relaxometric, tensile, melt flow and rheologic characteristics were assessed. Thermogravimetry indicated a low influence of the filler on thermal stability. The deconvolution of the melting peaks indicated transformation of the PA-6 α to γ phase and increase in crystallinity degree. Sample with 1 wt.% of ZrP showed an additional peak at higher relaxation time indicating restriction of polymer molecular mobility. By infrared, amine and carbonyl indices evaluation suggested ZrP interfered on the PA-6 hydrogen bonds. The elastic modulus increased slightly for all composition. Melt flow rate decreased with the amount of ZrP. The composite with 1 wt.% of ZrP exhibited the highest storage modulus and shear thinning effect noticed in the complex viscosity.
Keywords
Introduction
Polymeric microcomposites were thought as engineering materials of high and ultra performance. These kinds of materials are characterized by high amount of filler and limitation concening its dimension. The advent of nanotechnology brought changes in the concept of polymeric composites. Polymeric nanocomposites use fillers where one of the dimensions is below 100 nm and filler content extremely lower than that of microcomposites. With nanoscale size, inorganic and organic fillers have brought a huge contribution in the construction of new materials with functionalities such as antifungal, antimicrobial, waterproofing, radiation barrier, transport of molecules, drug delivery and so on. Among the most common inorganic fillers in nature, silicates are the most abundant. They are presented in the cationic or anionic form represented by montmorillonite and layered double hydroxide, respectively. Recently, Chatterjee et al. 1 published an review on layered double hydroxide based bionanocomposites. Nazir et al. 2 synthesized and modified cobalt-aluminum layered double hydroxide in order to produce a material with ability to remove pollutants from industrial waste water. Ultrafiltration membrane based on cellulose acetate, α-aminophosphonate modified montmorillonite and Ag-TiO2 nanoparticles for treatment of hazardous textile wastewater was experienced by Abdel-Karim et al. 3 A kind of catalyst supported on montmorillonite chemically modified with different mass fractions of Fe2O3 was built for degradation of bisphenol-A. 4 Silver nanoparticles show a large range of usage due to its wide spectrum of properties. Salem et al. 5 performed the green synthesis of silver nanoparticles. They spread it onto cotton fabrics and concluded that an effective activity against pathogenic Gram-positive and Gram-negative bacteria was attained. Silver nanoparticles were immobilized into cellulose nanocrystal and incorporated into carboxymethyl cellulose. The set was impregnated in paper for packaging application. The authors highlighted the improvement of properties in particular the sample with 7 wt.% of immobilized matter where the tensile strength increased about 1.26 times and the best antibacterial activities against E.coli and S.aureus were achieved. 6 Ortega et al. 7 produced silver nanoparticles through green synthesis procedure. They prepared starch composite films containing different amounts of it. The sample containing 71.5 ppm of nanoparticles decreased water vapor permeability while tensile resistance increased indicating reinforcement of the polymer matrix. Packaging films based on chitosan/essential oils/silver nanoparticles were developed by Shankar et al. 8 The authors revealed that the composite presented strong antimicrobial activity against Escherichia coli, Listeria monocytogenes, Salmonella Typhimurium, and Aspergillus niger besides an increase of tensile strength after addition of silver nanoparticles. As nanoscale material, zinc oxide acquires multiples properties permiting its integration into diversified sectors of the nanotechnology. Green synthesis of zinc oxide nanoparticles was experienced and its action as antimicrobial agent was tested against Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa, E. coli, and Candida albicans. The microorganisms activities were reduced at low concentration of zinc nanoparticles. 9 A species of fungus - Penicillium chrysogenum – was experienced for synthesizing zinc and copper oxides. The nanoparticles provided antimicrobial activities against bacteria Gram positive and negativeia. 10 For odontological application, zinc oxide nanoparticles were tested as antimicrobial against cariogenic bacteria and effective anti-plaque. The authors concluded that it had an effective inhibition against Streptococcus mutans and anticaries agent. 11 Yang et al. 12 synthesized a catalyst based on the functionalization of magnetic graphene oxide with tannic acid/Fe3+ and silver nanoparticles. The authors emphasized its antimicrobial capability and recyclability. Chemical inertia, non-toxicity, barrier to ultraviolet radiation are some qualities of titanium oxide nanoparticles. It find application in cosmetics, paints, plastics, papers, healthy and care sectors. Thipperudrappa et al. 13 prepared a glass fiber epoxy composite incorporated with nanoparticles of titanium and zinc oxides. The incorporation of nanofillers improved impact, tensile and flexural strengths. The synergistic effect on mechanical, thermal and electrical properties of the addition of nanodiamond and titanium oxide in pitch-derived carbon foam composites were studied by Khan et al. 14 Improvement of properties were achieved derived from connection between nanofillers and carbon foam. Montmorillonite was filler into hydrogeld of pullulan matrix in order to develop adsorbent for removing dye in wastewater treatment. 15 Our research group has developed works in which zirconium and titanium phosphates were incorporated into addition and condensation polymers to build materials with improved properties. Biodegradable blends based on poly(vinyl alcohol)/poly(N-vinyl-2-pyrrolidone) (PVAl/PVP) filled with octadecylamine-containing zirconium phosphate were studied. The authors registered the presence of multiphase systems in that amine provided variable polymer molecular motion of PVAl and PVP phases. 16 Similar work was published where to same blend was filled with pristine lamellar zirconium phosphate. The authors reported that PVAl thermal properties were reduced and the reinforcing effect of ZrP. 17 Quinelato et al. 18 studied the influence of polycaprolactone/titanium phosphate compound on the properties of recycled polypropylene. It was deduced that polymer intercalation inside titanium phosphate galleries, TiP acted as reinforcing agent and phase separation system were attained. Albitres et al. 19 investigated the effect of titanium phosphate modified with two types of amines on the nanocomposites based on poly(ethylene terephthalate). The authors registered that the presence of amines induced the lowering of PET glass transition temperature (Tg) and heat crystallization temperature (Tch) and the increase of its crystallinity degree (Xc), cold crystallization temperature (Tcc). Herein, PA-6 composites filled with 1–3 wt.% of lamellar zirconium phosphate were studied. The effect of nanofiller on structural, thermal, relaxometric, tensile, melt flow and rheological characteristics were evaluated. The composite was thought as potential material for packaging sector.
Experimental
Materials
Polyamide-6 (density = 1.13 g/cm3) was provided by Radici Plastics. For zirconium phosphate synthesis, the zirconium (IV) oxide chloride octahydrate (ZrOCl2.8H2O) and phosphoric acid (H3PO4) were purchased from Vetec.
Synthesis of layered zirconium phosphate
Lamellar zirconium phosphate (ZrP) was synthesized by the direct precipitation reaction method. 20 Phosphoric acid and zirconium oxide chloride (P:Zr ratio equal 18) were kept under reflux for 48 h. The precipitates were centrifuged and washed with distilled water to attain a pH of 6. The material was frozen at −80°C, 24 h, and lyophilized for 4 days for releasing water.
Nanocomposite preparation
Mono-screw extruded melt blending nanocomposites based on PA-6 with a ZrP (1–3 wt.%) were processed. The processing window corresponded to three heating zones (210–230oC), and a screw speed of 60 rpm was used. Each composition was processed twice to improve homogeneity. Compression molded plates with dimensions of (140 x 140 x 1 mm) were obtained at 200°C, 6000 lbf for 5 min, and cooled in another press.
Thermogravimetry analysis (TGA)
The thermogravimetric analysis was carried out in a TA Q500 thermogravimetric analyzer from 30 to 700°C, at 10°C/min, with nitrogen as carrying gas (flow rate 60 mL/min). Tonset, Tmax, and Tfinal degradation temperatures were assessed.
Differential scanning calorimetry (DSC)
Calorimetric data were evaluated in a PerkinElmer DSC-7 calorimeter (flow rate of carrying gas was 50 mL/min). Two heating and one cooling cycles were applied. The first heating cycle was performed from 0 to 250°C, at a 10°C/min heating rate, and kept molten for 0.7 min. A cooling cycle to 0°C at 10°C/min was applied next. The third heating cycle was experienced under the same conditions as the first one. The melting (Tm) and crystallization (Tc) temperatures were measured. The degree of crystallinity (Xc) was estimated by the ratio between the melting enthalpy of the sample and melting enthalpy of 100% PA-6 (190 J/g) of the second heating cycle. The value was normalized according to the filler amount in each nanocomposite.21,22
Hydrogen nuclear magnetic resonance time domain (1HNMRTD)
Hydrogen nuclear resonance in the time domain was conducted in a Maran Ultra 23 NMR equipment. From the domain curve, the relaxation time (T1H) of the pristine PA-6 and nanocomposites were assessed, 27°C, in time intervals of 10 s.
Fourier transform infrared spectroscopy (FTIR)
Infrared spectroscopy was performed in Varian Excalibur equipment. The spectra were obtained by ATR assembly, 4000-1140 cm−1, 50 scans, resolution of 2 cm−1. The variable bands at 3295 cm−1 (amine) and 1634 cm−1 (carbonyl) and an invariable band (1460 cm−1) were selected to determine the amine and carbonyl indices. 23
Tensile test
The tensile test was performed in a Universal test equipment (INSTRON) according to ASTM D638, using samples with dimensions of 0,8 by 10 cm and 1 mm of thickness. The elastic modulus, stress and elongation at the yield and break were determined and expressed considered the average of five specimens.
Melt flow rate
The melt flow rate (MFR) was performed according ASTM 1238 – method B using a dynisco plastometer at 235oc using a load of 2.16 kg.
Rheology
The rheological behavior was evaluated in a TA rheometer model AR-2000 using parallel plate geometry of 25 mm diameter, at 280oC. The experiments were performed in nitrogen atmosphere in dynamic shear. Deformation scanning tests were performed to observe the region of linear viscoelasticity of the materials in the frequency of 1 Hz and moduli (storage and loss) and complex viscoty were determined.
Water absorption
The uptake of water was performed following the ASTM D570. Each sample with 2.6 × 7.8 cm and 1 mm of thickness was heat conditioned at 80°C for 24 h, weighed and immersed in water. The gain of water was monitored until constant mass value was attained. For each specimen, the percentage change in mass (ΔM%) was determined, according to equation (1). The result was expressed by the average of three specimens.
Results and discussion
Thermogravimetry
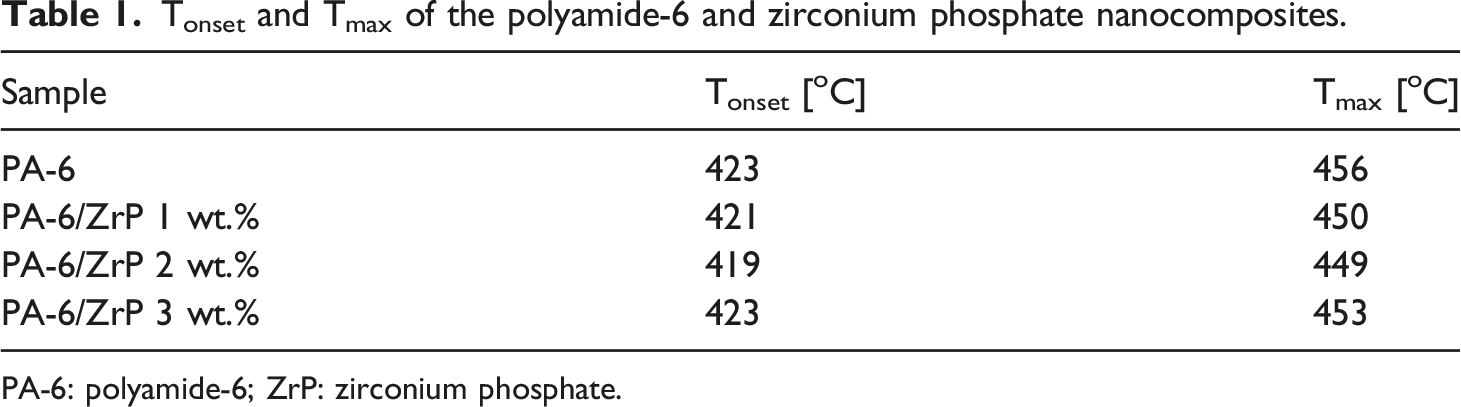
Figure 1 shows the TGA loss mass and derivative curves. All curves presented one degradation step almost superimposed attributed to the degradation of PA-6 chains. Table 1 displays the initial degradation temperature (Tonset) and the temperature where the degradation rate is maximum (Tmax). The Tonset decreased slightly only for 1–2% of ZrP. Tmax decreased for all samples. Erem et al. prepared nanocomposites of polyamide-6 incorporated with zinc oxide. They detected the decrease of thermal stability with the nanofiller amount.
24
We hypothesized that the slight lowering of PA-6 thermal stability could be associated to a possible catalytic effect of ZrP. (a) Loss mass and (b) derivative curves of PA-6 and PA-6/ZrP composites. PA-6: polyamide-6. Tonset and Tmax of the polyamide-6 and zirconium phosphate nanocomposites. PA-6: polyamide-6; ZrP: zirconium phosphate.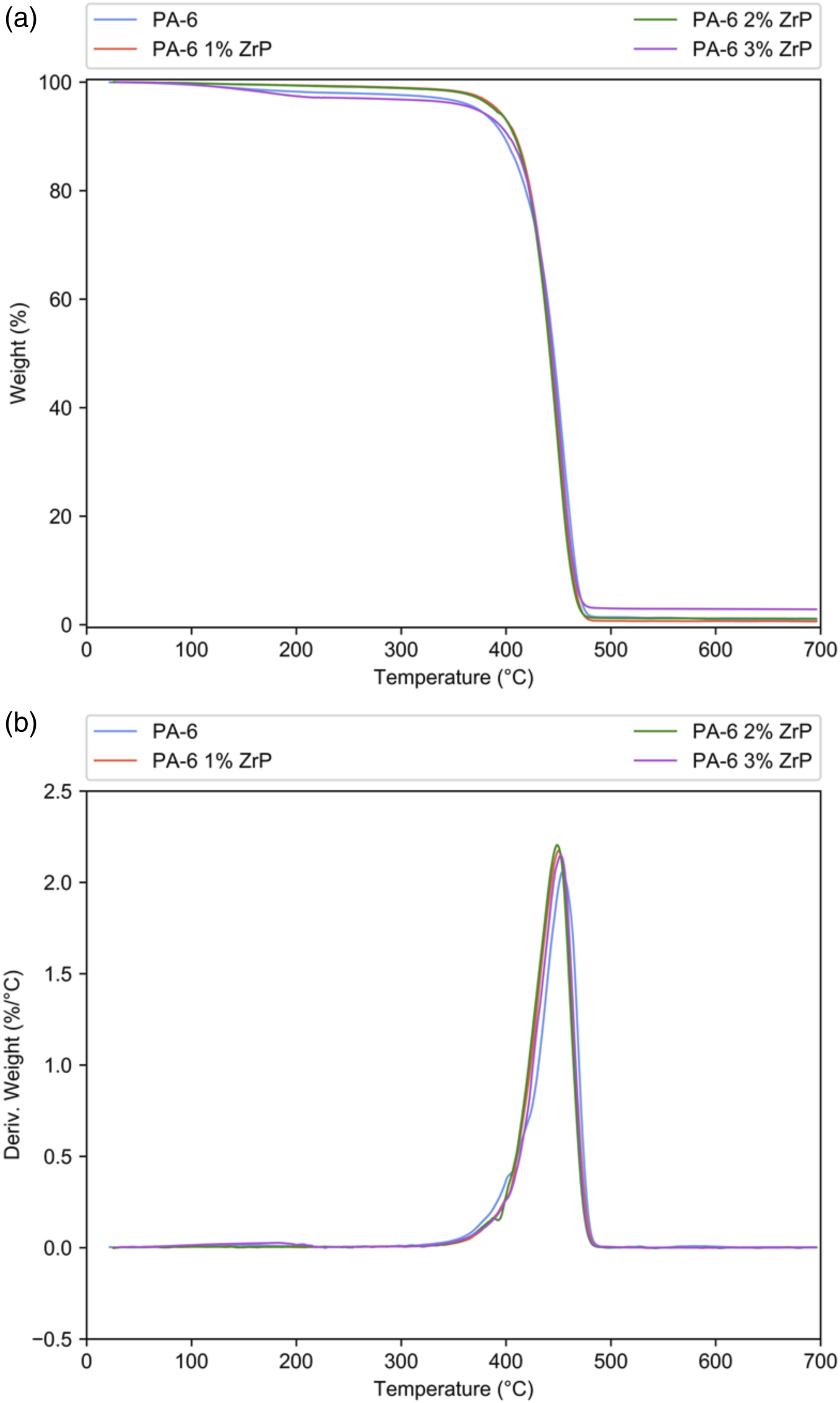
Differential scanning calorimetry
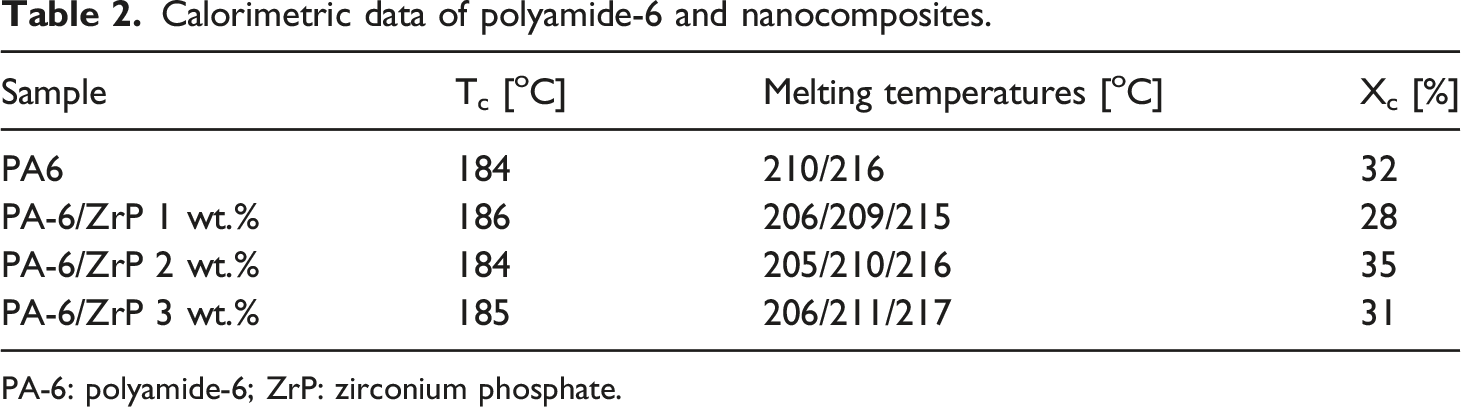
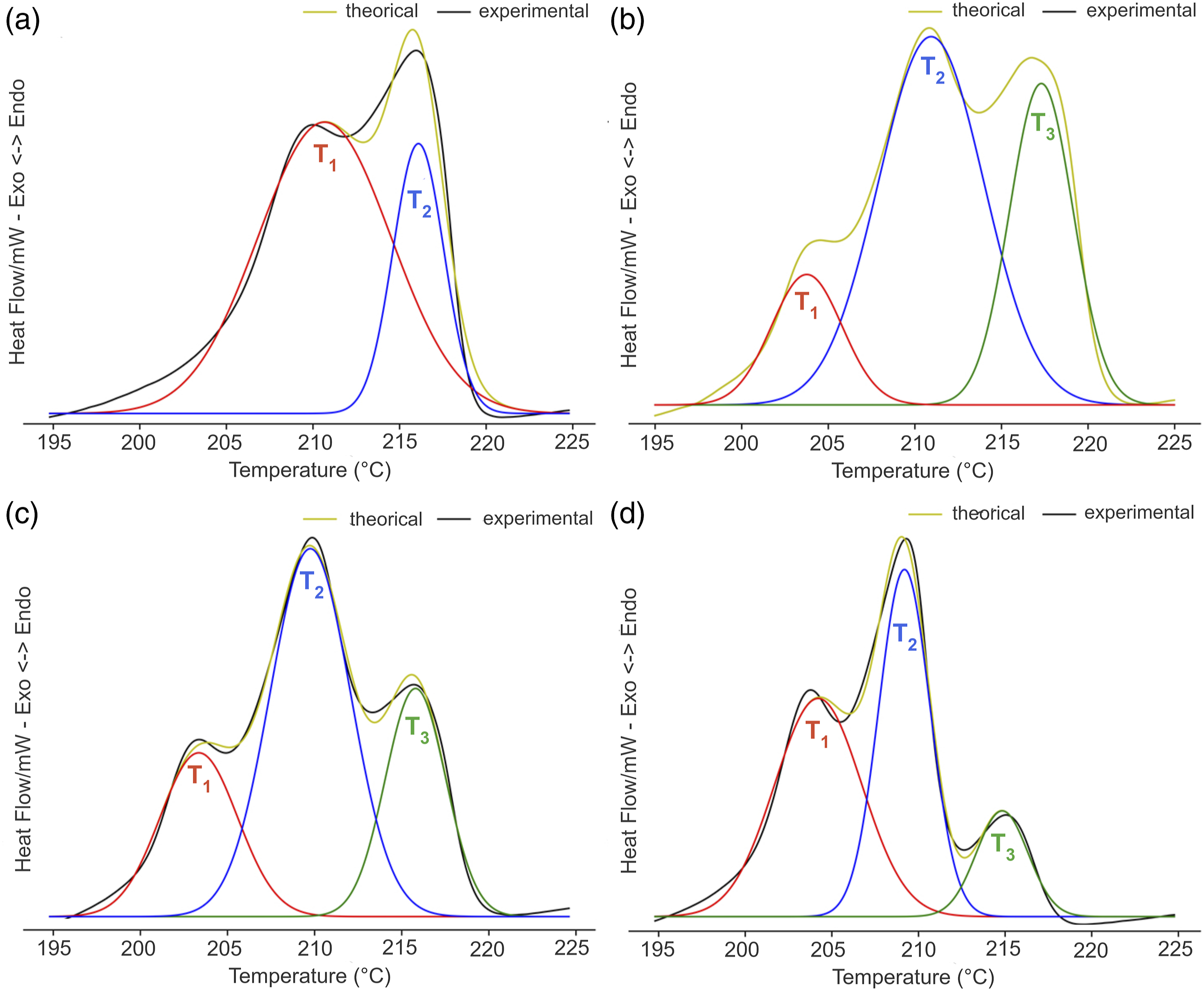
Figure 2 exhibits the sample’s crystallization and melting temperatures (Tc and Tm) curves. Only one exothermic peak was noticed for Tc. On the contrary, PA-6 and nanocomposites presented two and three endothermic peaks, respectively. The two melting peaks revealed by PA-6 were considered representative of α-crystalline arrangement with different crystal sizes. They were named as T1 and T2 being the first peak related to the higher temperature and the second as the lower one. All composites present an additional endothermic peak at lower temperature (named T3) attributed to the γ-crystalline arrangement. Table 2 highlights the values of Tc, Tm, and crystallinity degree (Xc). The Tc values revealed a negligible variation, and the melting temperatures were within the experimental error of the DSC technique. Xc varied randomly with ZrP amount. If considered the variation of Tc insignificant we could argued that the variation in Xc was related to the nanofiller dispersion in polymer matrix. Szakács et al.
25
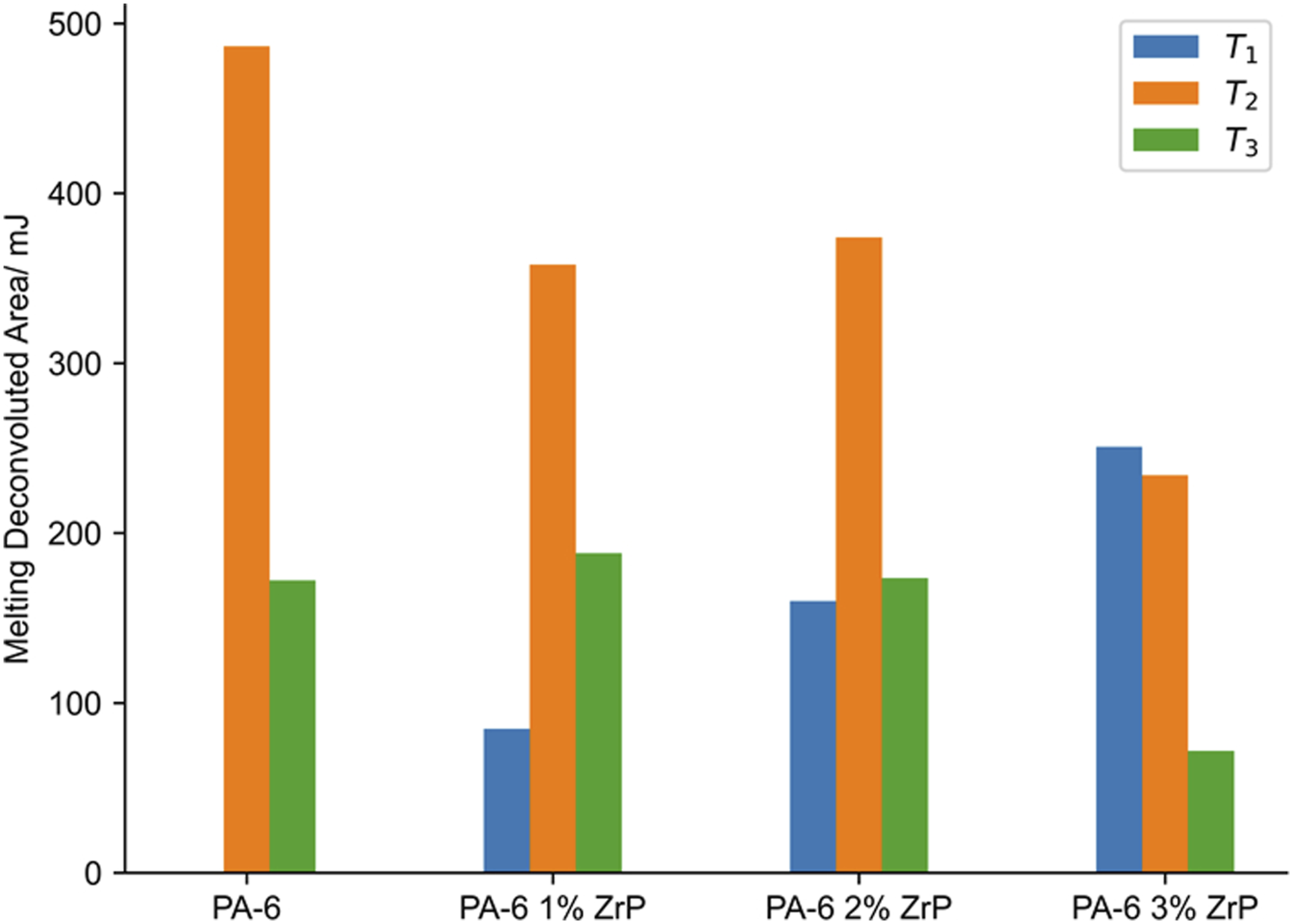
studied hybrid PA-6 composites containing carbon nanotubes and microfibers (basalt and carbon). They reported that basalt (inorganic matter rich in magnesium and iron) did not have a nucleating effect, while the role of nanotubes as nucleation agents was limited by its concentration building α-crystalline PA-6. To investigate the relationship between the amount of ZrP and the appearance of new crystal order, we performed the deconvolution of the melting peaks. Figure 3 displays the deconvolution curves of the samples. T1 melting peak (α-form) trended to decrease while the T3 (γ-form) increased with ZrP amount. Figure 4 presents the comparative bar plot of the crystalline phase’s stability as a function of the ZrP amount. Three crystal structures of PA-6 were noted. T3 appeared at lower temperature while T1 and T2 varied according to the ZrP content. At 1% of ZrP, the T2 portion diminished due to the transformation from α to γ-crystalline phase. Concomitantly, T2 refined its structure, changing to T1 form. The T3 peak increased with 2% of ZrP, while a slight decrease in T1 was noticed. At 3% of ZrP, T3 peak became more evident while T1 and T2 peaks decreased. The literature has reported that inorganic salts can affect the PA-6 phase stability, promoting the conversion of the α-crystalline form into γ-one. Karacan and Baysal
26
registered the effect of cupric chloride on the thermal stabilization of PA-6 as carbon fiber precursor. Freitas et al.
27
reported the outcome of octadecylamine-modifying ZrP on crystalline phase conversion of PA-6. Wang et al.
28
presented the effect of the amount of organic montmorillonite on morphologies, thermal and mechanical properties of polyamide-6 composite foam. Composites based on PA-6/clinoptilolite revealed the effect of the zeolite as both nucleating and intercalating agents, modifying the alpha to gamma form crystal arrangement.
29
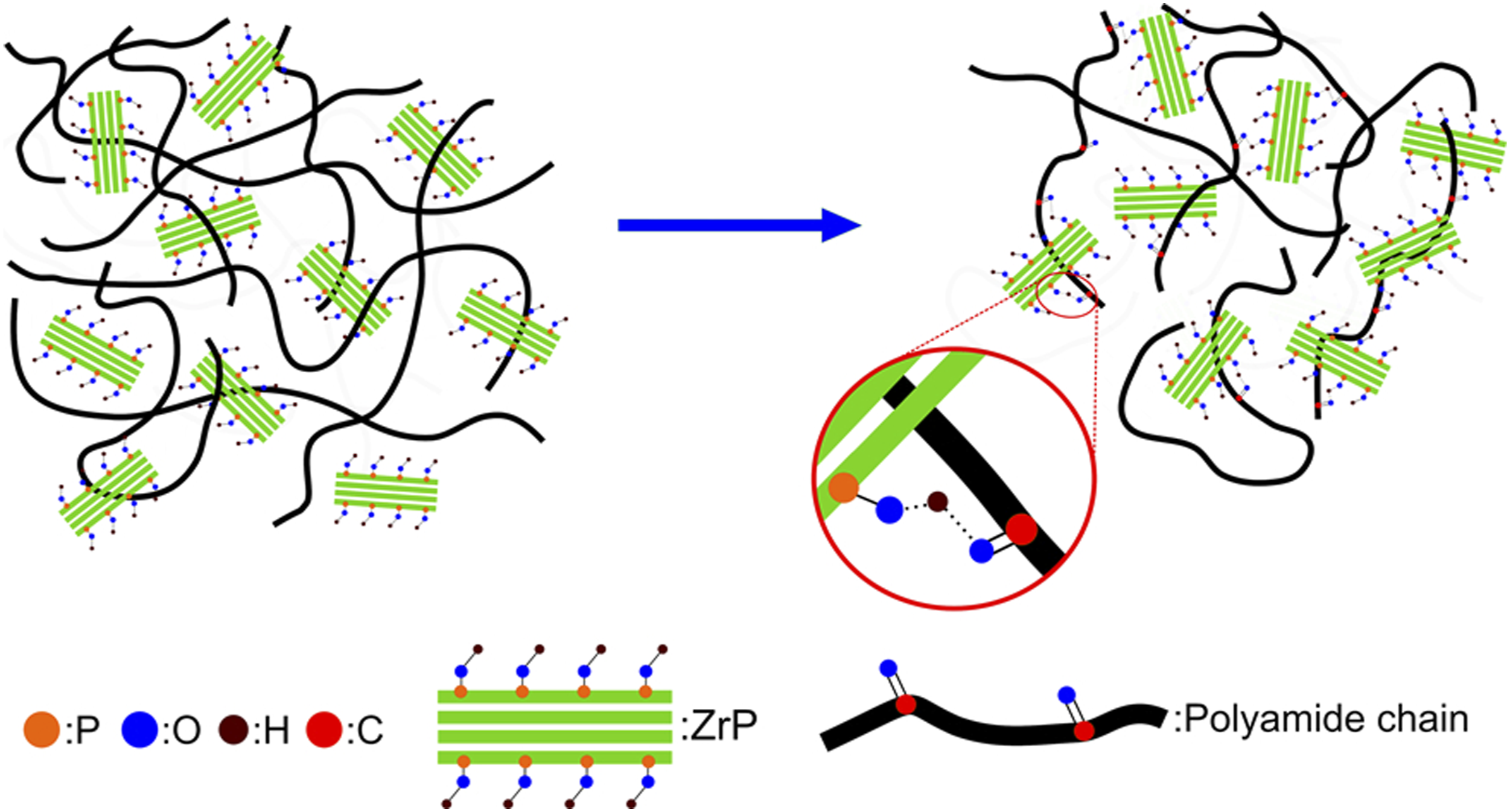
Herein, the PA-6 phase transformation could indicate that ZrP interposed among the PA-6 chains destroying its original intra and inter hydrogen bonds. We strongly believed that P-O-H group attached inside and outside the ZrP lamellae reacted with the PA-6 carbonyl group building a new hydrogen bond as represented in Figure 5. Crystallization and melting temperatures curves of polyamide-6 and nanocomposites. Calorimetric data of polyamide-6 and nanocomposites. PA-6: polyamide-6; ZrP: zirconium phosphate. Deconvoluted differential scanning calorimetry curves: a) PA-6, b) PA-6 1% ZrP, c) PA-6 2% ZrP and d) PA-6 3% ZrP. PA-6: polyamide-6; ZrP: zirconium phosphate. Effect of polyamide-6 crystalline phase with zirconium phosphate content. Effect of lamellar zirconium phosphate on the polyamide-6 hydrogen bonds.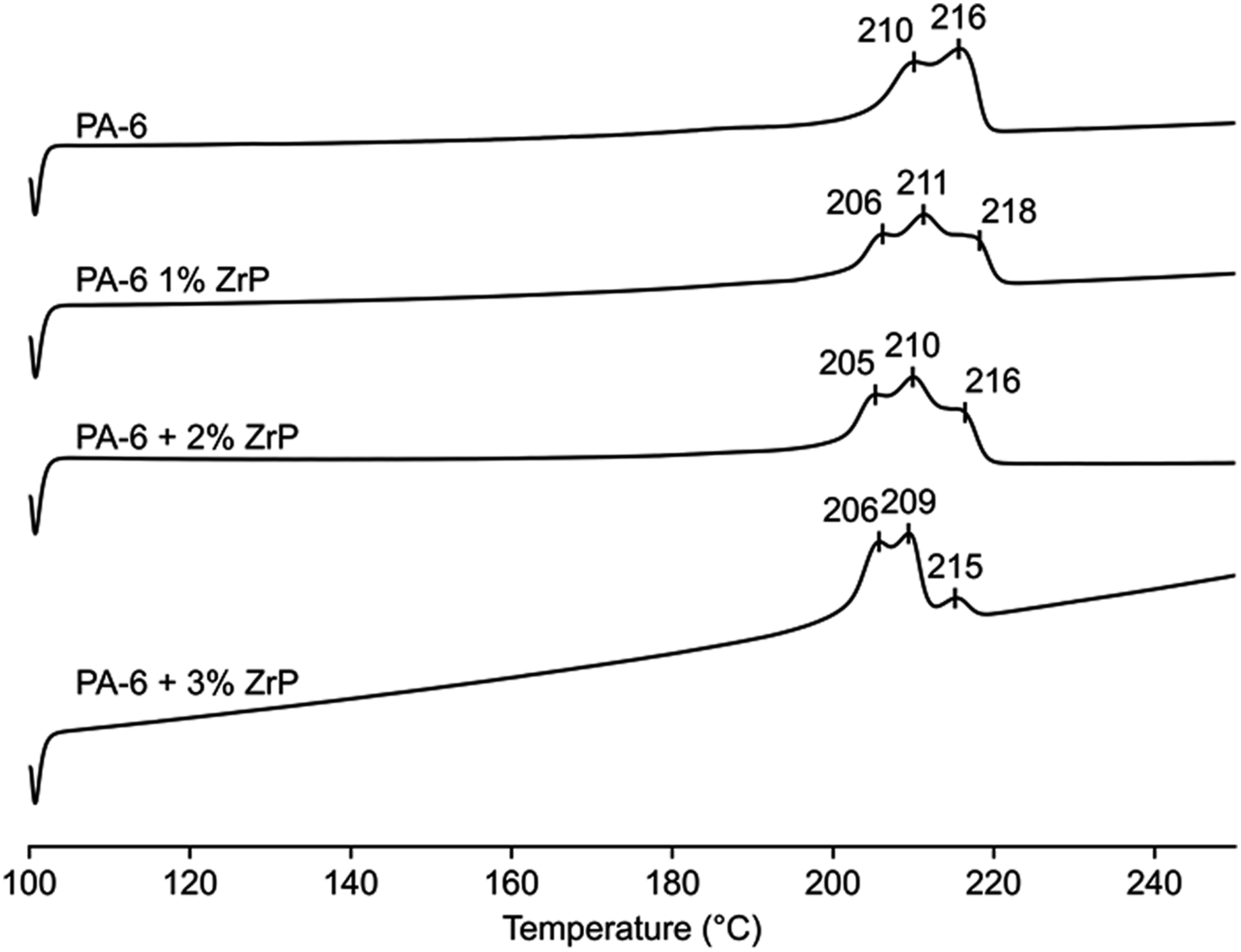
Hydrogen Nuclear Magnetic Resonance Time Domain (1HNMRTD)
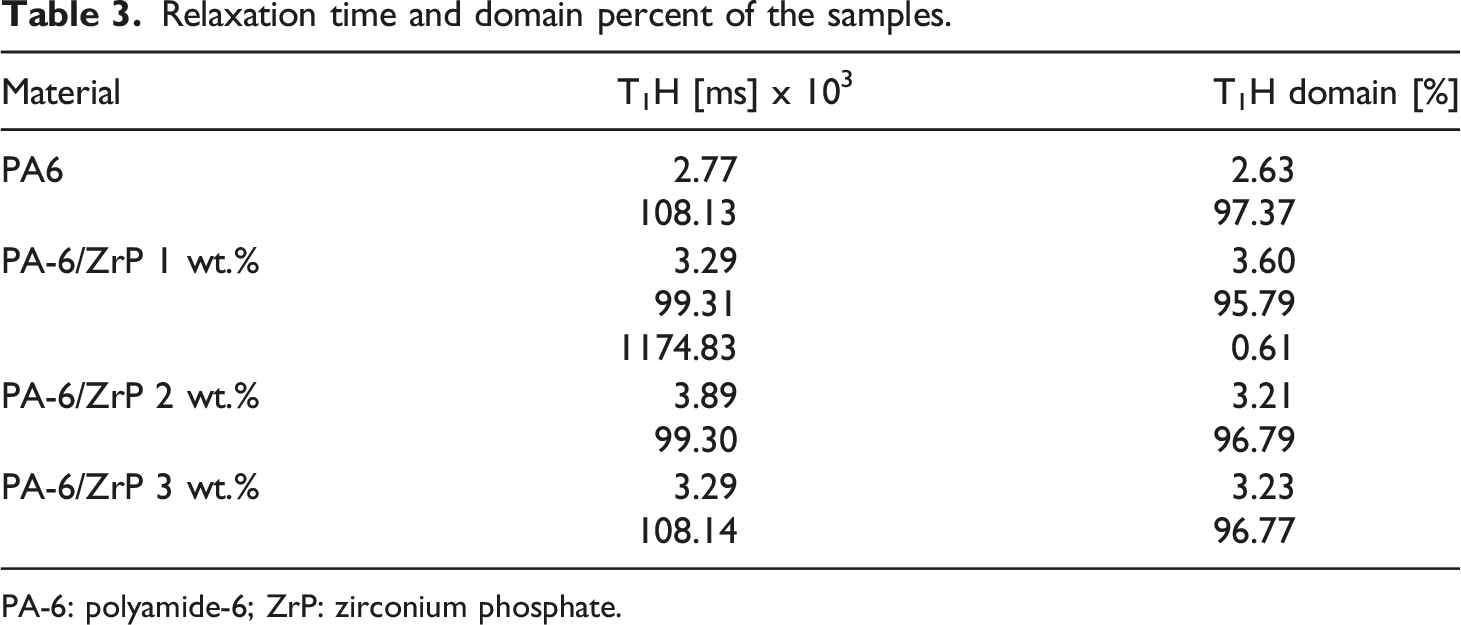
Figure 6 displays the 1HNMRTD domain curves. All presented similar profile but composites show peak base narrower than PA-6. The sample with 2 and 3% ZrP showed two domains ranging from 103 to 104 and 104–105 ms. The sample with 1 wt.% of ZrP revealed an additional domains above 106 ms. The domain at lower relaxation was associated with some residual water molecules and/or low molar mass impurity. The domains at highest molecular relaxation were related to the hydrogen of the PA-6 chains constricted among the PA-6 crystalline arrangement and possibly those involved in the interaction between polymer-filler. Table 3 displays the percentage domain and T1H for the samples. It was no observed variation on the domain percent. With 1 and 2 wt.% of ZrP, the T1H decreased, while with 3 wt.%, the value was recovered to the starting one. Freitas et al.
27
studied the effect of octadecylamine-modifying ZrP in the nanocomposites based on PA-6. They reported that variation on the T1H was associated with the interference of the ZrP on the PA-6 hydrogen bonds. We speculated that the samples with 1 and 2 wt.% of ZrP showed higher interaction due to the lowering of T1H. Also, we deduced that the sample with 1 wt.% of ZrP presented the higher interaction between PA-6 and nanofiller. Domain curves of the samples. Relaxation time and domain percent of the samples. PA-6: polyamide-6; ZrP: zirconium phosphate.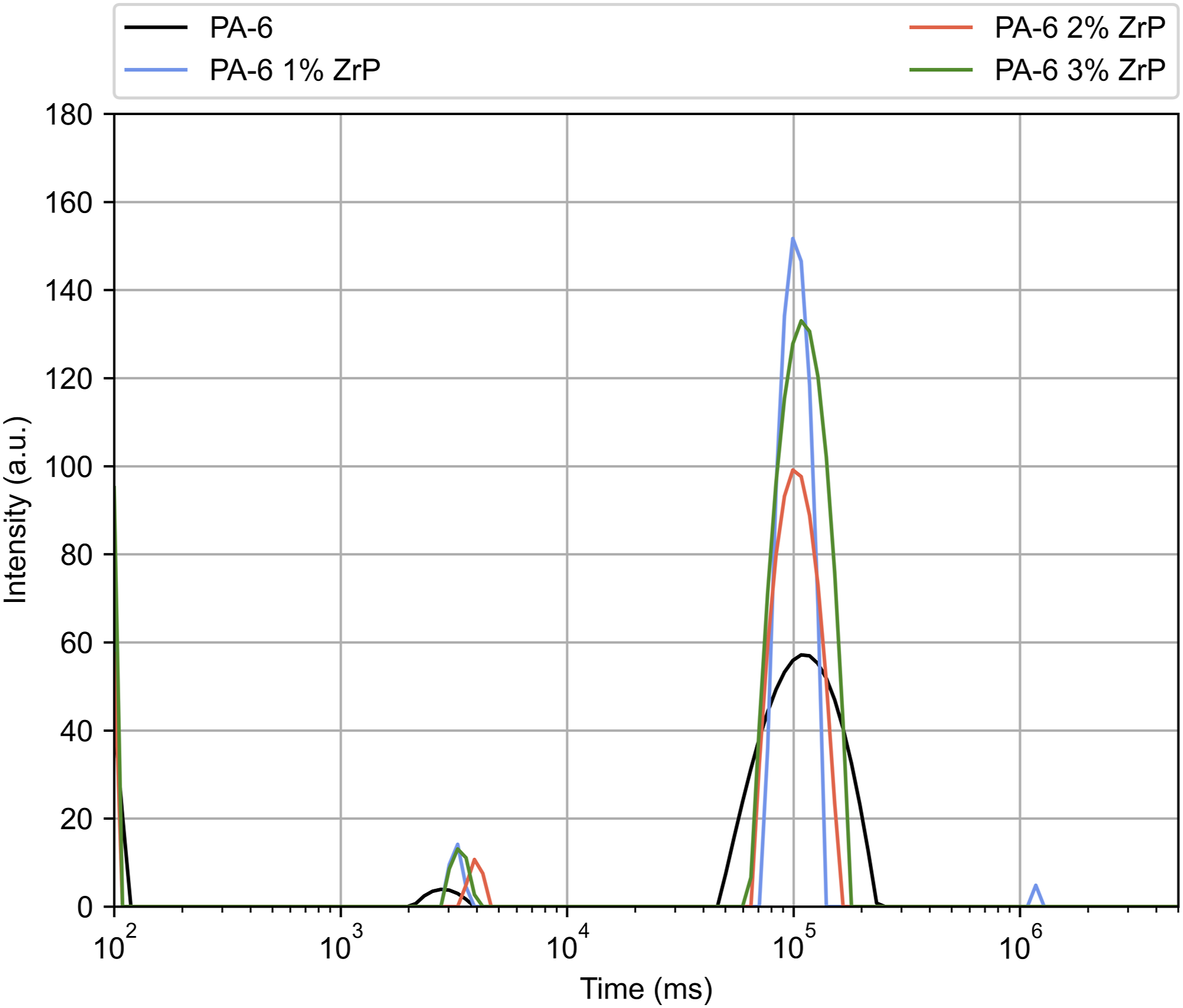
Fourier transform infrared spectrometry
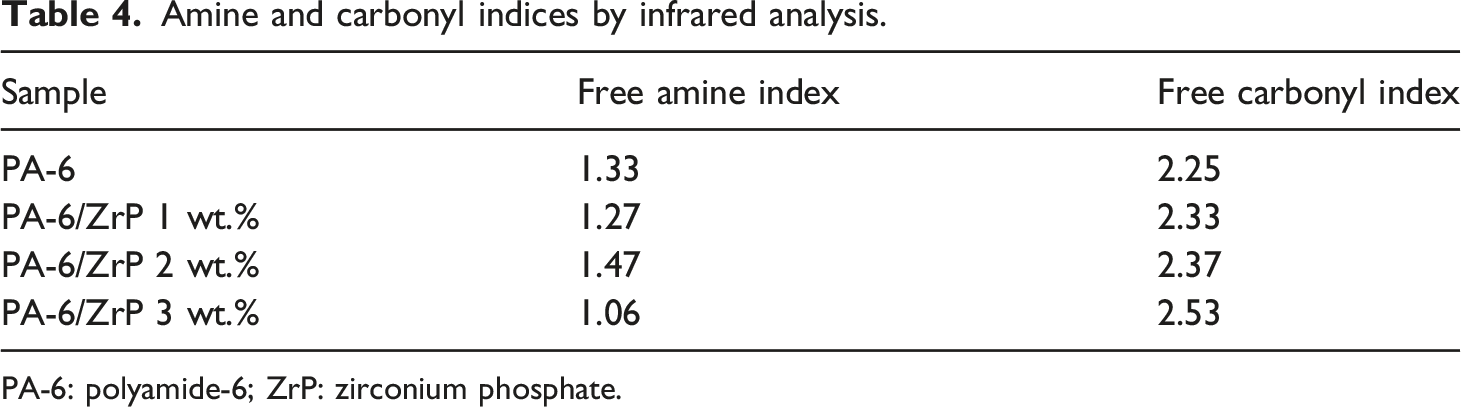
The spectra of the samples were allocated in two spectral regions (4000-2000 cm−1 and 1800-1140 cm−1, Figure 7). Modifications on the spectra were assessed making the calculation among some bands: 3294–3298 cm−1 (NH stretching), 2936–2926 cm−1 (CH stretching), 1636-1634 cm−1 (amide I C=O stretching), 1545-1543 cm−1 (amide II NH bending and CH stretching), 1462-1460 cm−1 (CH2 scissors vibration), 1373-1366 cm−1 (amide III and wag vibration) and 1204-1202 cm−1 (CH2 twist-wag vibration), The first ratio consisted of variable band at 3295 cm−1 (NH stretching) and an unchangeable one at 1460 cm−1 (CH2 scissors vibration). The second one was related to the variable band at 1634 cm−1 (amide I C = O stretching) and the band at 1460 cm−1. The ratios were named amine index and carbonyl index, respectively, and are displayed in Table 4. Except for the sample with 2 wt.% of ZrP, the amine index showed a tendency to decrease. On the contrary, the carbonyl index increased according to the ZrP content. Macossay and co-authors studied polyamide 6/functionalized graphene oxide nanofibers. Reinforcement in mechanical properties were associated to the hydrogen bond between the NH group from PA-6 with the functional groups in the graphene.
30
Strong interaction between PA-6 filled with organically modified zirconium phosphate was reported by Garcia et al.
31
and Freitas et al.
27
We attributed the lowering of the amine index to the action of ZrP collapsing of the intra and inter PA-6 hydrogen bond. We also believed that this breakage favored the formation of hydrogen bond among P-OH groups in ZrP with PA-6 carbonyl groups. The result is in agreement with calorimetry and nuclear magnetic resonance data. Samples fourier transform infrared spectroscopy at 4000–2000 and 1800-1140 cm−1. Amine and carbonyl indices by infrared analysis. PA-6: polyamide-6; ZrP: zirconium phosphate.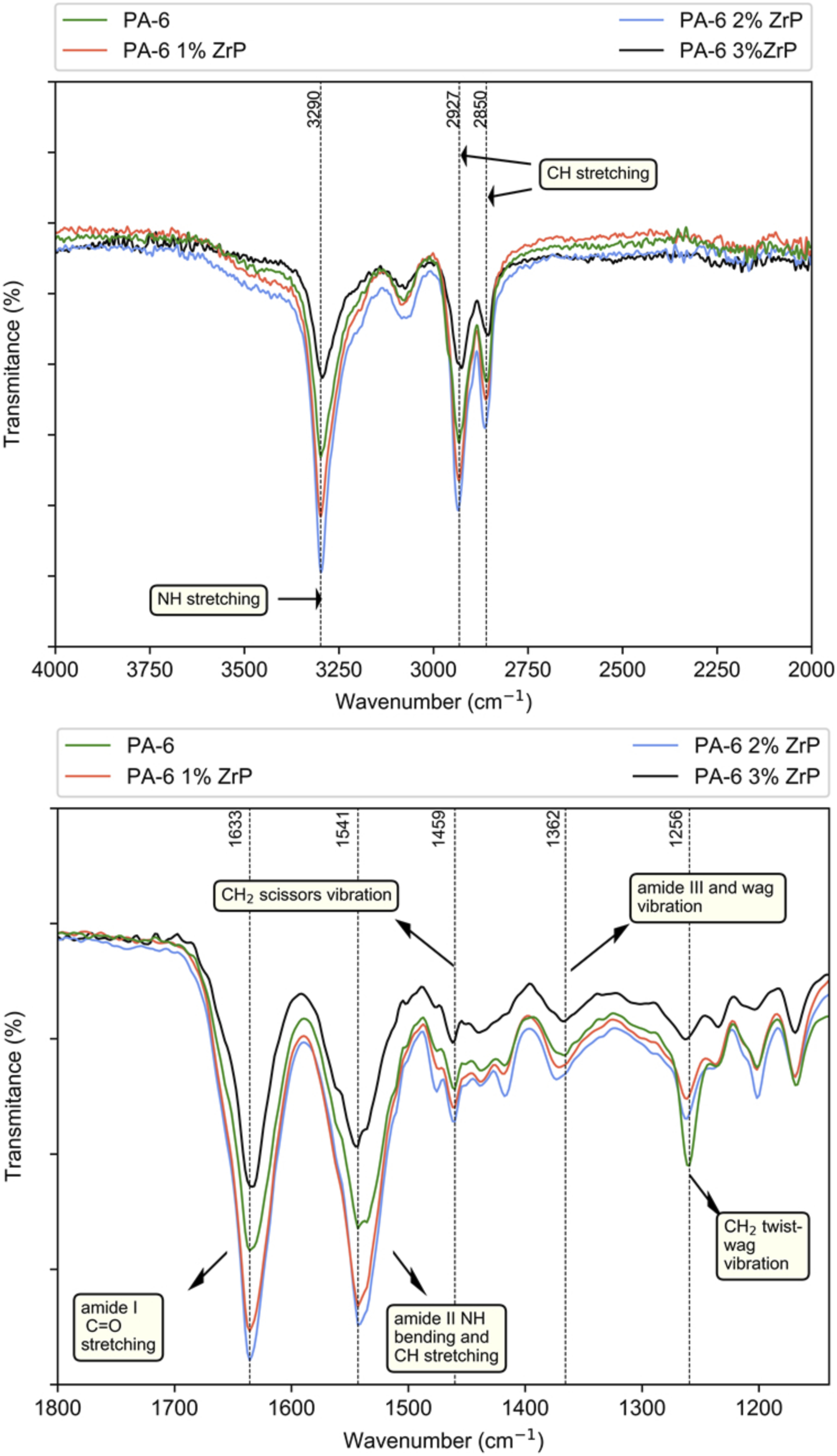
Mechanical proprties
Mechanical data of the samples.
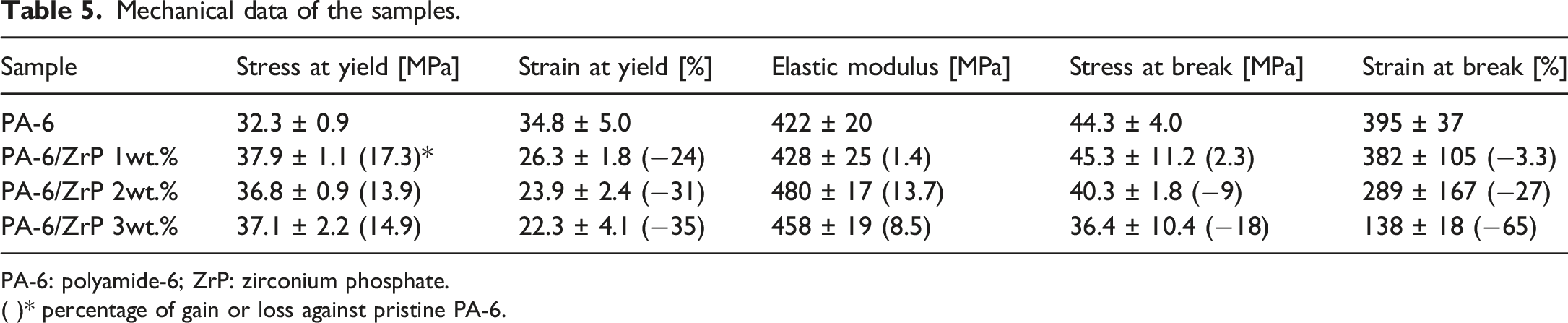
PA-6: polyamide-6; ZrP: zirconium phosphate.
( )* percentage of gain or loss against pristine PA-6.
Melt Flow Index (MFR)
Melt flow rate of the samples.

PA-6: polyamide-6; ZrP: zirconium phosphate.
( ) percentage change.
Water uptake
Figure 8 shows the samples water uptake versus time. It is well known that PA-6 possess great trend to absorb water because of hydrogen bonds. The water uptake kinetics of the samples was governed by the Fick’s Law. All curves presented a steeper portion related to water solubilty (lesser than 1 day). A less steep second portion associated to water diffusion occurred between 1–5 days. Following, the stabilization was noticed. For all composites, the water uptake diminished in the following order: PA-6/ZrP 1 wt.% and PA-6/ZrP 2 wt.% (≈−25%) < PA-6/ZrP 3 wt.% (≈−12.5%) < PA-6. Lei et al.
36
studied hygroscopicity in composites of PA-6/carbon fiber. They registered lesser water absorption due to the barrier effect of carbon fiber. Vasveld et al.
37
also reported a reduction of water absorption in composites of PA-6 filled with 10.6 wt.% of silicate. The diffusion coefficient was decreased around one third of unfilled PA-6. Herein, as deduced in infrared evaluation, we supposed that the zirconium phosphate particles restricted the ability of the amide groups to form hydrogen bonds among PA-6 chains and the access of the water molecules to link to amide groups. Then, the water uptake was lowered. The results are in agreement with calorimetry, nuclear magnetic resonance and infrared data. Water uptake for all samples.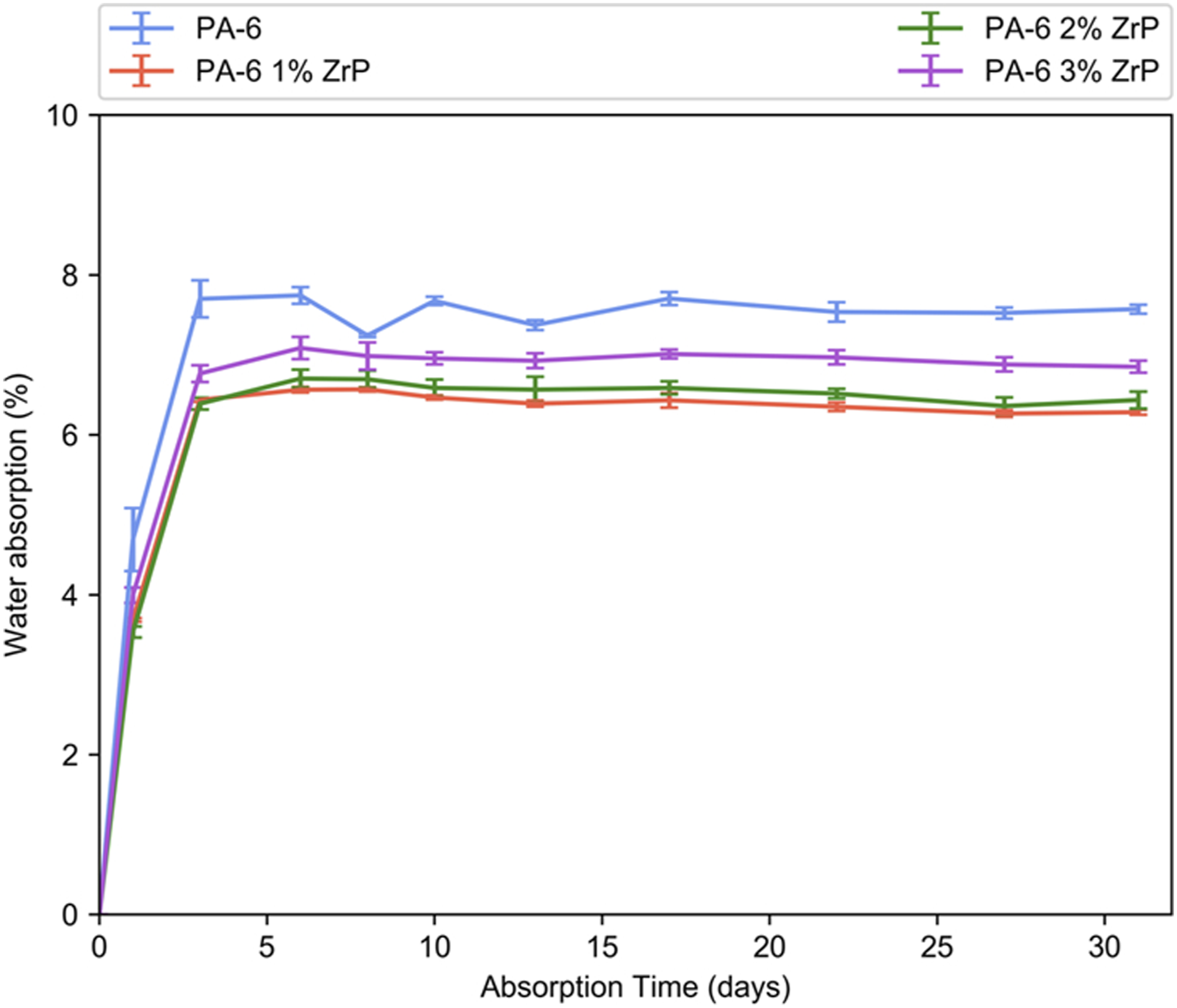
Rheology
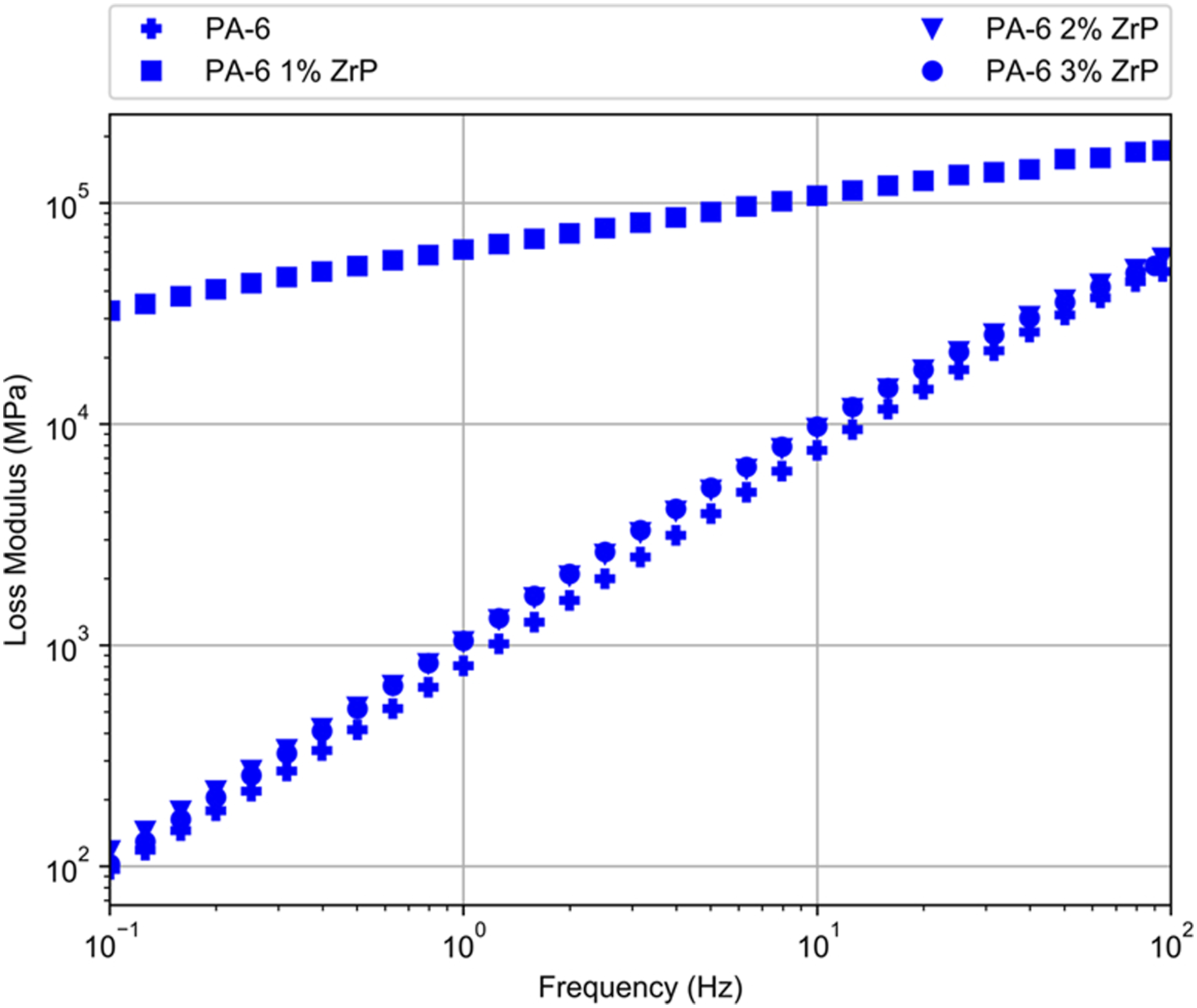
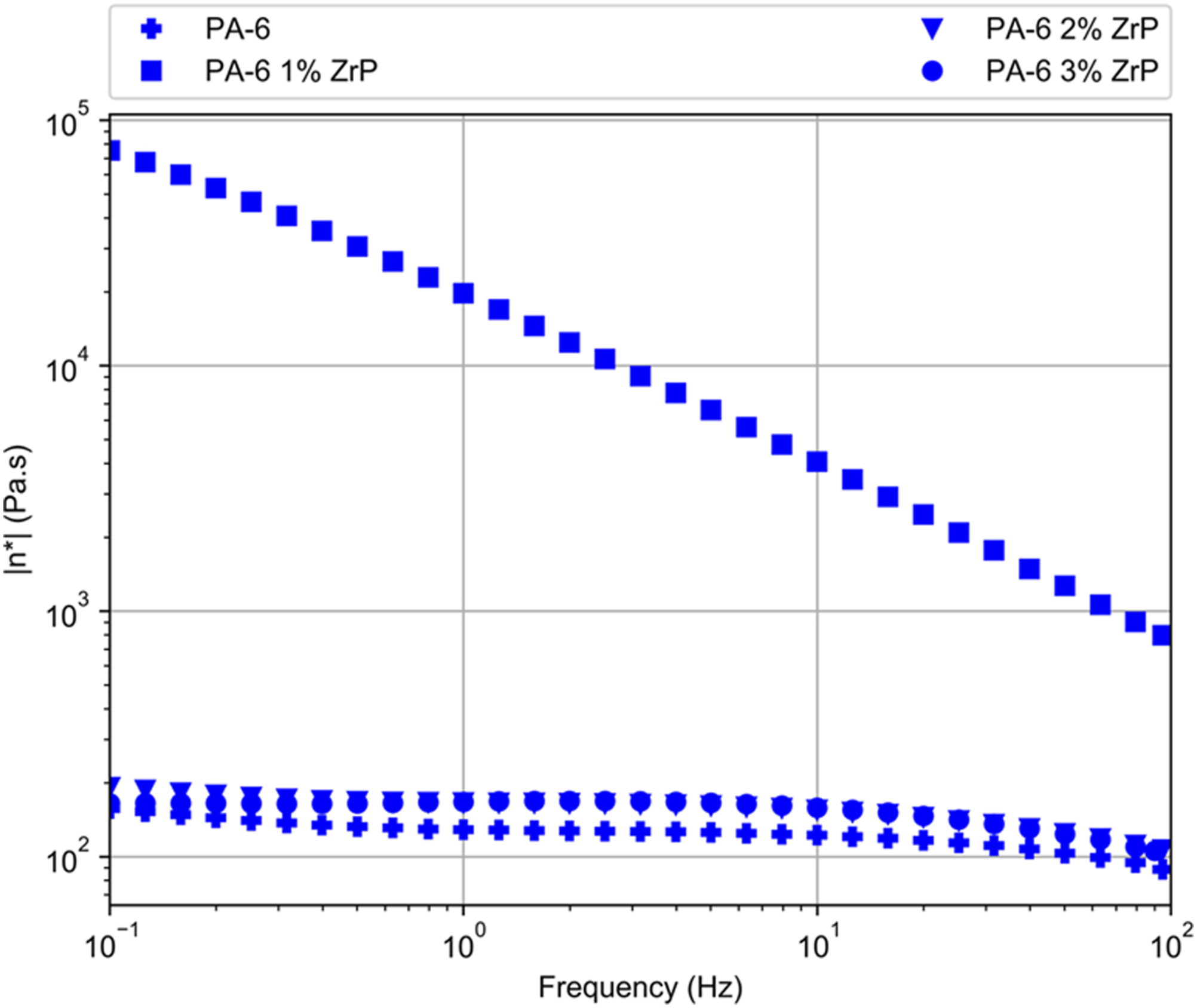
Figures 9–11 show the moduli and complex viscosity curves versus frequency, respectively. Storage modulus (G’) of PA-6/ZrP 1wt.% presented a peculiar behavior. In all frequency range, it increased gradually and its value was quasi three magnitude order higher than pristine PA-6 and two other composites. PA-6, PA-6/ZrP 2 wt.% and PA-6/ZrP 3 wt.% samples displayed behavior dependent on the frequency. Below 100 Hz, G’ curves were overlapped and the value increased continuously as function of frequency. After 100 Hz, G’ also increased with frequency but PA-6/ZrP 2wt.% and PA-6/ZrP 3wt.% curves seems to separate from PA-6 one exhibiting values slightly higher. Also, above 10° Hz, the G' of the composites filled with 2 and 3 wt.% of phosphate showed tendency to attain values in the vicinity those exhibited for the composite containing 1wt.% of ZrP. Loss modulus (G″) curve of PA-6/ZrP 1wt.% behave similarly to its G’ counterpart. For PA-6, PA-6/ZrP 2 wt.% and PA-6/ZrP 3 wt.%, the curves were overlapped in all frequency range. Also, for all them, G’’ increased with the frequency and if compared with PA-6/ZrP 1wt.% the values tend to be closer. Complex viscosity (η*) of PA-6/ZrP 1wt.% decreased in all frequency range. Although close to each other, PA-6/ZrP 2 wt.% and PA-6/ZrP 3wt.% curves detached slightly from its PA-6 counterpart. It is expected that the incorporation of nanoparticles in polymer matrices imparts enhancement of their physico-chemical properties. These characteristics are strongly dependent on the type, geometry, surface area, amount, distribution of the loaded filler. Besides these factors, the interconnected network, interfacial region, interplay among filler-filler, polymer-polymer and filler-polymer play important role in the composites rheological behavior. In their study on low molecular weight epoxy resin with carbon nanotubes (CNT, 0.5 wt.t%), Meeuw et al.
38
revealed that the investigation of rheological behavior of CNT/polymer is complex owing to different networks involved such as entanglements of polymer chains, interconnected network of CNT and combined polymer/nanotube network. They emphasized the use of low molecular weight epoxy resin was to focus on filler network changes by using strain and frequency dependency of oscillatory shear flow. They pointed out the applied shear amplitude drove the changes in the network. Little or no variation was induced on CNT network states by applying frequency and shear rate. Thinking on dispersion and phase morphology strongly affetec the rheological and mechanical properties of nancomposites based on polymers and nanoclays, Gahleitner et al.
39
investigated rheology-morphology interactions in nanocomposites based on polypropylene-polyamide-6 (PP/PA-6) incorporated with two kinds of nanoclays (7–13 wt.%). Different behaviors were noticed for each polymer. At frenquency range (10–2–102 rad/s), PP complex viscosity continuously decreased. Sample curve of PP with addition of 7.5 wt.% of nanoclay overlapped the pristine PP indicating that polymer-polymer and filler-filler predominated. Pristine PA-6 complex viscosity decreased discretely along the frequency axis. Addition of 7.5 wt.% of nanoclay imparted high shear thinning effect on the polyamide flow. For blend incorporated with nanoclays, unexpected effect was noticed. Preferentially, nanoclays located in the PA-6 phase and the authors deduced that the main rheological effect was not ascribed to the improvement of the degree of dispersion but owing to the stronger PA-6/filler interactions. Kelnar et at.
40
investigated the action of PA-6 microfrils and organically modified layered montmorillonite (oMMT) when incorporated in high density polyethylene (HDPE). None effect in the HDPE viscosity was observed after addition of 3 and 5 wt.% of oMMT. With respect to PA-6, the incorporation of 0.5 and 1 wt.% of oMMT decreased the viscosity while the a drastic increase with the addition of 5 and 10 wt.% of nanoclay was noticed. In the whole range of frequency, the blend system HDPE/PA-6/oMMT (80/20/3) showed higher viscosity than its counterpart without nanoclay. The results were associated to several mixing protocols which included concurrent mixing and application of different masterbaches for both components and combinations. Nano zinc oxide (0–5 wt.%) was incorporated into low density polyethylene (LDPE) and PA-6 viewing the ability as antibacterial agent. Rheological study was performed at low degree of loaded filler (0.5 and 1 wt.%). LDPE exhibited Newtonian behavior at frequencies below 100 rad/s and afterwards pseudoplastic flow was noticed. After addition of those filler content, the viscosity curves were quasi overlapped and shear thinning occurred in the whole frequency. For PA-6 filled with 1 wt.% of ZnO, the authors reported that the Newtonian viscosity increase slightly (from 360 to 450 Pa.s). At these low content, ZnO particles showed no significant effect on the polymer flow. Thus, nano ZnO reinforced LDPE but there is no apparent effect on PA6 matrix.
41
Herein, when PA-6 was incorporated with 1 wt.% of phosphate the complex viscosity increased and decreased continuously in whole frequency range. This could be explained in terms of shear thinning flow where the filler interposed among macromolecular chains hampering their movements during the applied shear force. This reasoning could be endorsed by the detection of the peak at higher relaxation time (T1H = 1174 ms) in the 1HNMRTD analysis. At one composite, the polymer chain, dispersed phase and interfacial region relaxations regulated its elastic properties. Storage modulus of PA-6/ZrP 1wt.% was the highest among the composites and continuously increased in whole frequency range. We could argue that a kind of interconnected or network like structures between PA-6 and phosphate - polymer/filler interaction - existed. This could restrict the motion of polymer chains resulting in the slight pseudo-solid like behavior. These assumptions could be endorsed by Kiziltas et al.
42
in their work addressed to the melt rheology and crystallization behavior of the PA-6/Cellulose Composites. With respect to composites with 2 and 3 wt.% of phosphate, we assumed that worse filler dispersion and/or possibly particle-particle interactions reflected on a quasi independence of the complex viscosity at frequency range. The G’ enhancement occurred owing to the stiffness imparted by nanofiller. The findings corroborated those found by Alavitabari et al.
43
in their investigation on rheological properties of hybrid nanocomposites based on high density polyethylene reinforced filled with layered silicate and calcium carbonate. In summary, we deduced that in the PA-6/ZrP 1wt.% composite particle-particle interaction did not occur, the filler was well dispersed, polymer-filler interaction existed and thus an homogeneous matter was reached. In the composites PA-6/ZrP 2 wt.% and PA-6/ZrP 3wt.% the interactions polymer/polymer and filler/filler prevailed dictating their rheological behavior. Storage modulus against frequency of the samples. Loss modulus against frequency of the samples. Complex viscosity against frequency of the samples.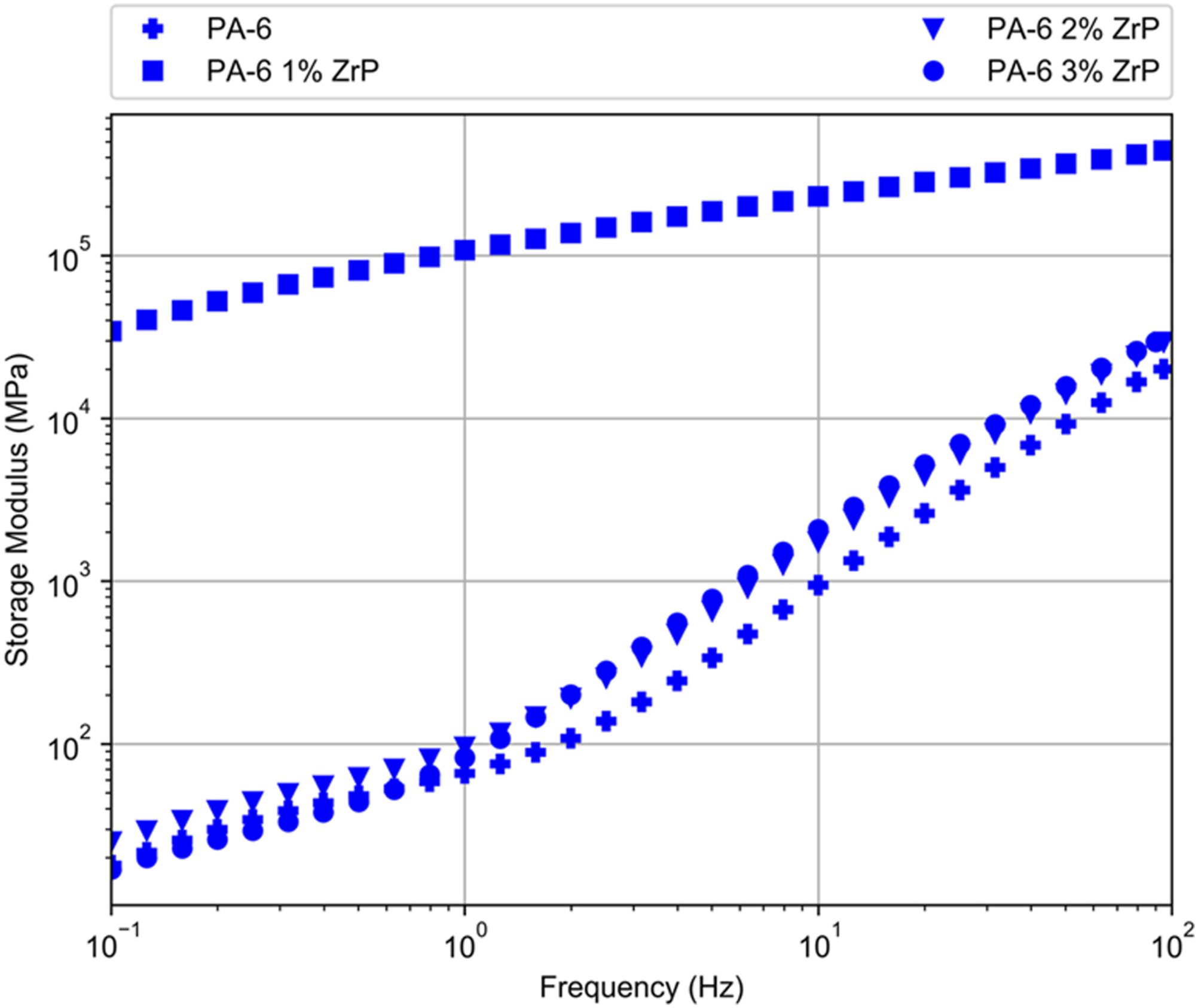
Conclusions
The effects of nanoscale lamellar zirconium phosphate on polyamide-6 physico-chemical properties were investigated. It was assigned a great influence on phase transformation and crystallinity degree. PA-6 molecular relaxation induced interaction among polymer and nanofiller. Infrared suggested the rupture of hydrogen bond among PA-6 chains and formation new one with P-OH groups of ZrP. The elastic modulus increased for all composition. Melt flow rate decreased with the amount of ZrP. The composite with 1 wt.% of ZrP exhibited the best storage modulus and complex viscosity values. Elastic modulus increased at all range of ZrP amount. The properties at yield showed increment in tensile strength and reduction of elongation. Elongation at break continuously decreased. MFR reduced in all composition. Water uptake showed maximum reduction around 25%. Composite with 1 wt.% of ZrP presented the best polymer/nanofiller interaction according to the rheology assessment.
Footnotes
Acknowledgements
The authors thank Conselho Nacional Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and Universidade Federal do Rio de Janeiro (UFRJ) for supporting this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.

