Abstract
Poly (sulphone) (PSu) was chemically grafted with PVC via Friedel-Crafts alkylation reaction at 85°C for 2 h under N2 atmosphere. Further, it was treated with 2-Methylimidazole (2MI) in the presence of Cu2O nanoparticle catalyst in N-methylpyrrolidone (NMP) medium for 2 h under nitrogen atmosphere with stirring. Thus synthesized PSu-g-PVC-g-2MI/Cu2O nanocomposite was characterized by FT-IR, 1H-NMR spectroscopy, DSC, TGA, XPS, SEM, EDX, HR-TEM, WCA and mechanical testing. The FT-IR spectrum showed a peak at 534 cm−1 corresponding to the metal-oxide stretching. The methyl proton signal of 2MI from the resultant polymer appeared at 2.40 ppm in the 1H-NMR spectrum. The glass transition (Tg) temperature and degradation temperature (Td) of the PSu-g-PVC-g-2MI/Cu2O nanocomposite system was determined as 169.2°C and 628°C respectively. The surface water contact angle of the resultant polymer nanocomposite system was found to be 48.1° and confirmed the hydrophilic nature of the membrane. The XPS showed the presence of Cu2p at 930.2 eV derived from Cu2O nanocatalyst. The experimental results were deeply analyzed and compared with the results of the other research teams.
Introduction
The quality of environment particularly, the water is disturbed by the various activities of human beings. The industrialization leads to the emission of various oxides of C, N and S. When these pollutants are mixed with water they became inseparable from water and threaten the human’s life on the earth. Recently, various methodologies have been developed to purify the water. Among them, membrane filtration is a process being utilized recently. Hence, the membrane is useful for desalination purpose. The membrane material must have the following requirements: presence of voids, hydrophilicity, antimicrobial properties, enough tensile strength with low cost. Currently, PSu, polyimide (PI), Poly (vinylidenefluride) (PVDF), Poly (ethersulphone) (PES) are used as membrane materials. Unfortunately, they are expensive and does not have hydrophilicity. Till the investigation of low cost membrane material, it is necessary to use the structurally modified and already existing membrane material. In order to increase the tensile strength of PSu, graphiticoxide was added in different % weight. 1 The influence of methylester moiety on the pervaporation activity of PSu was tested by Yoshikawa et al. 2 The micro-structural properties of PSu were improved by making blend with Poly (phenylenesulphide). 3 The anion exchange capacity of PSu membrane was improved by cross linking with tetramethylene diamine. 4 The gas separation activity of PSu was improved by grafting with amine functionalized lignocelluloses. 5 In 2020, a review article on functionalized PSu was published by Serbamescu and co-workers. 6 Poly (dopamine) functionalized PSu was reported with improved fouling resistance. 7 The pyrazole grafted graphiteoxide was functionalized with PSu with enhanced dye removal from waste water. 8 Primary amine functionalized PSu was reported by Guiver et al. 9 The zwitter ion functionalized PSu membrane with improved antifouling properties were reported. 10 The alkaline stability of PSu membrane was improved by grafting reaction with benzimidazolium ion. 11 Similarly, PSu was functionalized with silane, 12 benzoxazine, 13 phosphonated poly (pentafluorostyrene), 14 zwitter ionic grapheneoxide 15 and functionalized layered silicate 16 to improve certain properties of PSu membrane. There is no literature available on the PVC grafted PSu via FC alkylation reaction. This urged the authors to do the present research work. Moreover, it has further chlorine atom to replacement. The main aim of preparing the material is for water filtration particularly for desalination purpose.
PVC is a thermoplastic polymer with readily replaceable chlorine atom. PVC is a flame retardant polymer but its commercial applications are less due to lack of hydrophilicity and hydro-functionality. The domestic application of PVC can be increased by functionalization reaction. Methyacrylicacid was grafted onto PVC. 17 PVC was modified with trimethoxysilyl propane thiol. 18 In 2019, PVC was structurally modified by thiourea. 19 PVC was functionalized with SF6. 20 Likewise, PVC was functionalized with azide, 21 vinyl functionalized silane rubber, 22 maleamates, 23 thiourea 24 and epoxidized novolac. 25 2MI functionalized PVC report is not available. This motivated the authors to do the present investigation.
2MI is an aromatic hetero cyclic colorless compound which is freely soluble in polar solvents and is used as a building block for the drug molecules. Due to the presence of C=N like Schiff base structure, it has antimicrobial properties. Imidazole containing polymer was synthesized for biological application.26,27 2MI was chemically grafted onto carboxymethylcellulose, 28 poly (aryletherketone), 29 poly (arylethersulphone), 30 PS31,32 and PVC. 33 The literature report reveals that the antimicrobial activities of polymers were not done properly. This is properly done in the present research work. 2MI functionalized PSu-g-PVC was done and studied the thermal, mechanical, structural, morphological and wettability of the same in the present investigation.
Recently, PSu based nanocomposites are playing vital role in the membrane field due to their improved thermal, mechanical and surface properties. In 2019, PSu/silica modified zeolite nanocomposite was prepared with enhanced mechanical and antibacterial properties. 34 PSu/graphite composite was prepared with improved autohesive property. 35 Dooher et al. 36 prepared the PSu/MWCNT nanocomposite with excellent thermal properties. The PSu/Cu2O nanocomposite report is not available in the literature.
The novelty of the present research work is chemical grafting of 2MI onto PSu-g-PVC system via FC alkylation reaction in order to import the antimicrobial property to the membrane. Further structural modification is possible due to the availability of Cl atom on the PVC backbone. The surface water contact angle of the resultant membrane was highly enhanced by the 2MI. The nanocatalyst Cu2O is acting as filler and influences the thermal property of the membrane. Due to the filling effect, the tensile strength of the resultant membrane was highly activated.
Experimental
Chemicals and materials
Poly (sulphone) (PSu), Poly (vinylchloride) (PVC), 2-methylimidazole (2MI), N-methyl-pyrrolidone (NMP) was purchased from Sigma Aldrich, USA. Acetone, NaBH4, Cu2O bulk powder was purchased from local suppliers. Double distilled water was used.
Synthesis of Cu2O nanoparticles
The Cu2O nanoparticle was prepared by chemical reduction method. 5.0 g of Cu2O bulk powder was dispersed in 100 mL double distilled water under ultrasonic irradiation for 2 h. Then, 5 g of NaBH4 in 50 mL water was added with Cu2O dispersant with vigorous stirring. The stirring was continued for 5 h. A similar procedure was followed for the synthesis of V2O5 nanoprticles from bulk powder. 37 At the end, a brownish black precipitate was obtained. The contents were filtered and the precipitate was washed with excess acetone for 2–3 times. The dried mass was stored under N2 atmosphere till further uses.
Synthesis of PSu-g-PVC
5.0 g of PSu was dissolved in 50 mL NMP at 85°C for 2 h. The clear solution of PSu was added with 5 g PVC in 15 mL NMP at 85°C. Anhydrous AlCl3 (3.0 g) was added and the Friedel-Crafts (FC) alkylation reaction
38
was carried out under nitrogen atmosphere with vigorous stirring for the next 2 hours. At the end, 250 mL of acetone was added and the PSu-g-PVC precipitate was filtered. The reaction is given in Scheme 1. The precipitate was washed with excess acetone and dried at 85°C for overnight. The obtained product was named as PSu-g-PVC (PSu2 system). Synthesis of PSu-g-PVC-g-2MI/Cu2O nanocomposite.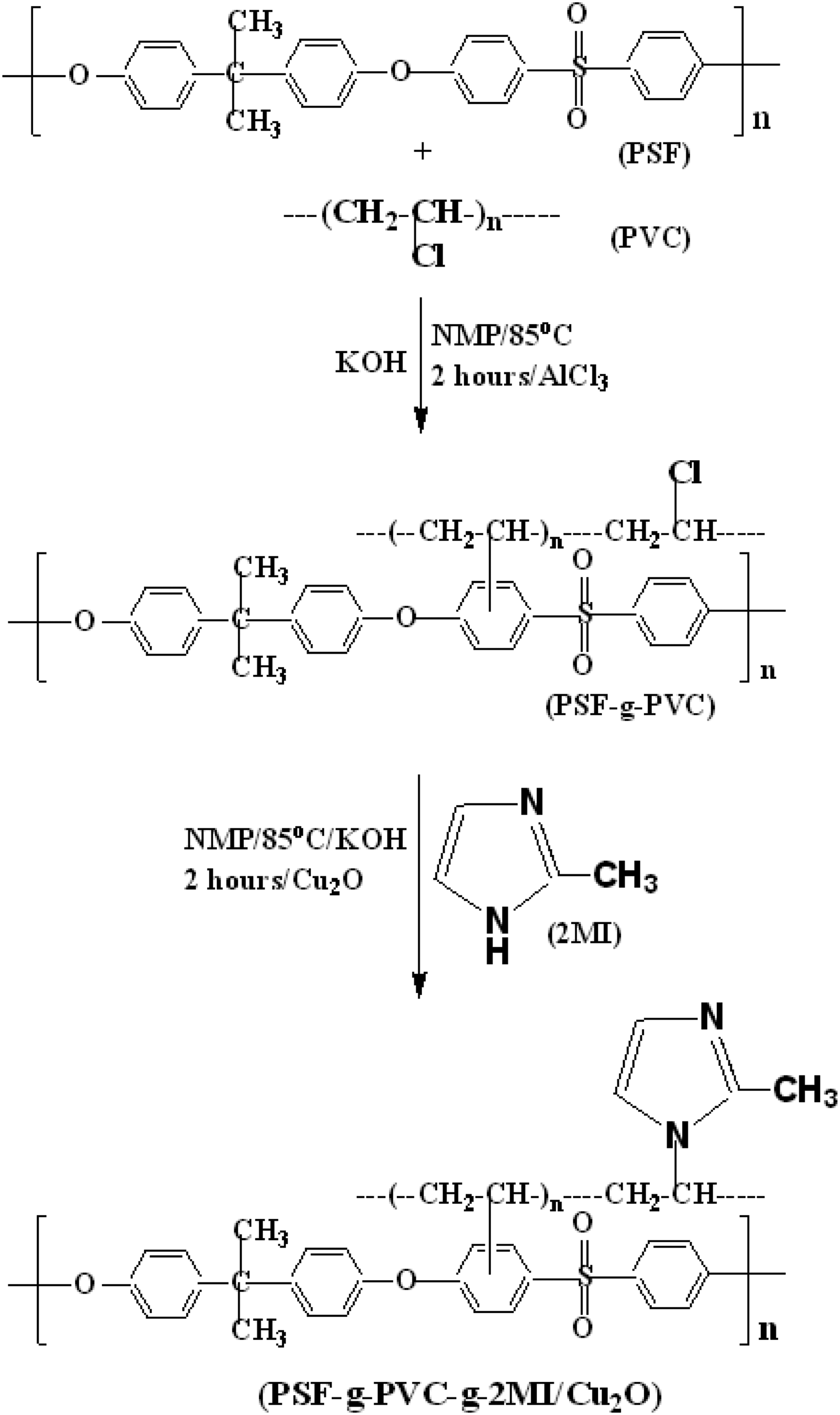
Synthesis of PSu-g-PVC-g-2MI/Cu2O nanocomposite
3.0 g of PSu2 system was dissolved in 25 mL NMP at 85°C under vigorous stirring. After the complete dissolution, 3.0 g of 2MI and 5 mol% of Cu2O nanoparticles were added. 39 The reaction was allowed to continue for another 2 h (Scheme-1). Finally, the contents were filtered by the addition of 200 mL acetone. After filtration, the precipitate was washed with acetone and dried at 85°C for overnight. The dried mass was named as PSu-g-PVC-g-2MI/Cu2O nanocomposite (PSu3 system) and stored in a zipper lock cover under nitrogen atmosphere.
Characterization
The FT-IR spectra of the polymer samples were recorded from 400 - 4000 cm−1 with the help of a Shimadzu 8400 S, (Japan) instrument by the KBr pelletization method. 2 mg of sample was mixed with 200 mg of spectral grade KBr and made into a disc under the pressure of 7 tons. The high resolution transmission electron microscope image was recorded using JEM-200 CX, Japan, instrument. The dried sample was subjected to vacuum and Au coated prior to image measurement. The Kyowa DMs-200, Japan instrument was used to test water contact angle (WCA) measurement. The WCA was measured in six places of the sample and the average one was taken. The DSC and TGA were measured using a Universal V4.4 A (TA Instruments, USA) under nitrogen atmosphere at the heating rate of 10 K min−1 from room temperature to 373 K by taking ∼10 mg of sample in a Pt crucible. In DSC the second heating scan of the sample was used by deleting the previous thermal history of the sample. For TGA measurement, air atmosphere was used and measured from room temperature to 900oC. 1H-NMR (500 MHz) spectra was obtained using Varian, Unity Inova-500 NMR instrument at room temperature in CDCl3 solvent. 5 mg of polymer sample was dissolved in CDCl3 solvent and the 1H-NMR spectrum was recorded. The SEM and EDX were recorded on JSM 6300, JEOL model, USA. The binding energy of the polymer samples were determined by XPS, Thermo Scientific, Theta Probe, UK instrument. The tensile strength was determined by Universal Tensile Tester, Deepak Polyplast, India.
Antimicrobial study
Antimicrobial analysis was done using standard agar well diffusion method. 40 Each bacterial isolate was suspended in Brain Heart Infusion (BHI) broth and diluted to ∼105 colony forming unit per mL. They were flood-inoculated onto the surface of Media (Mueller Hinton Agar for Bacteria and Sabouraud’s Dextrose agar for Fungi). Five-millimeter diameter wells were cut from the agar using a sterile cork-borer and 30 µL (5 µg compound in 500 µL DMSO) of the sample solution were poured into the wells. The plates were incubated for 18 h at 37°C for bacteria and room temperature for fungi. Antimicrobial activity was calculated by measuring the diameter of the zone of inhibition in mm against the test microorganisms and the solvent. DMSO was used as solvent control. Ciprofloxacin (5 µg) disc was used as reference antibacterial agent and Amphotericin B (5 µg) disc was used as reference antifungal agent. Tests were carried out in triplicates.
Results and discussion
Characterization
Figure 1(a) presents the FT-IR spectrum of PSu1 systems with aromatic C-H stretching (3034, 3088 cm−1), aliphatic C-H stretching (2874, 2986 cm−1), C-H bending vibration (1471 cm−1), SO2 stretching (1264 cm−1),
41
C-O-C stretching (1008 cm−1) and aromatic wagging (618, 709, 853 cm−1). On structural modification with PVC via FC alkylation reaction it showed the above mentioned peaks in Figure 1(b) with one new peak. The Cl ion stretching was noted at 485 cm−1. On further structural modification with 2MI it showed three new peaks like C=N stretching (1696 cm−1),
42
C-N stretching (1365 cm−1) and Cu-O stretching (554 cm−1) in Figure 1(c). Hence, the appearance of new peaks confirmed the structural modification reaction in PSu. FT-IR spectrum of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.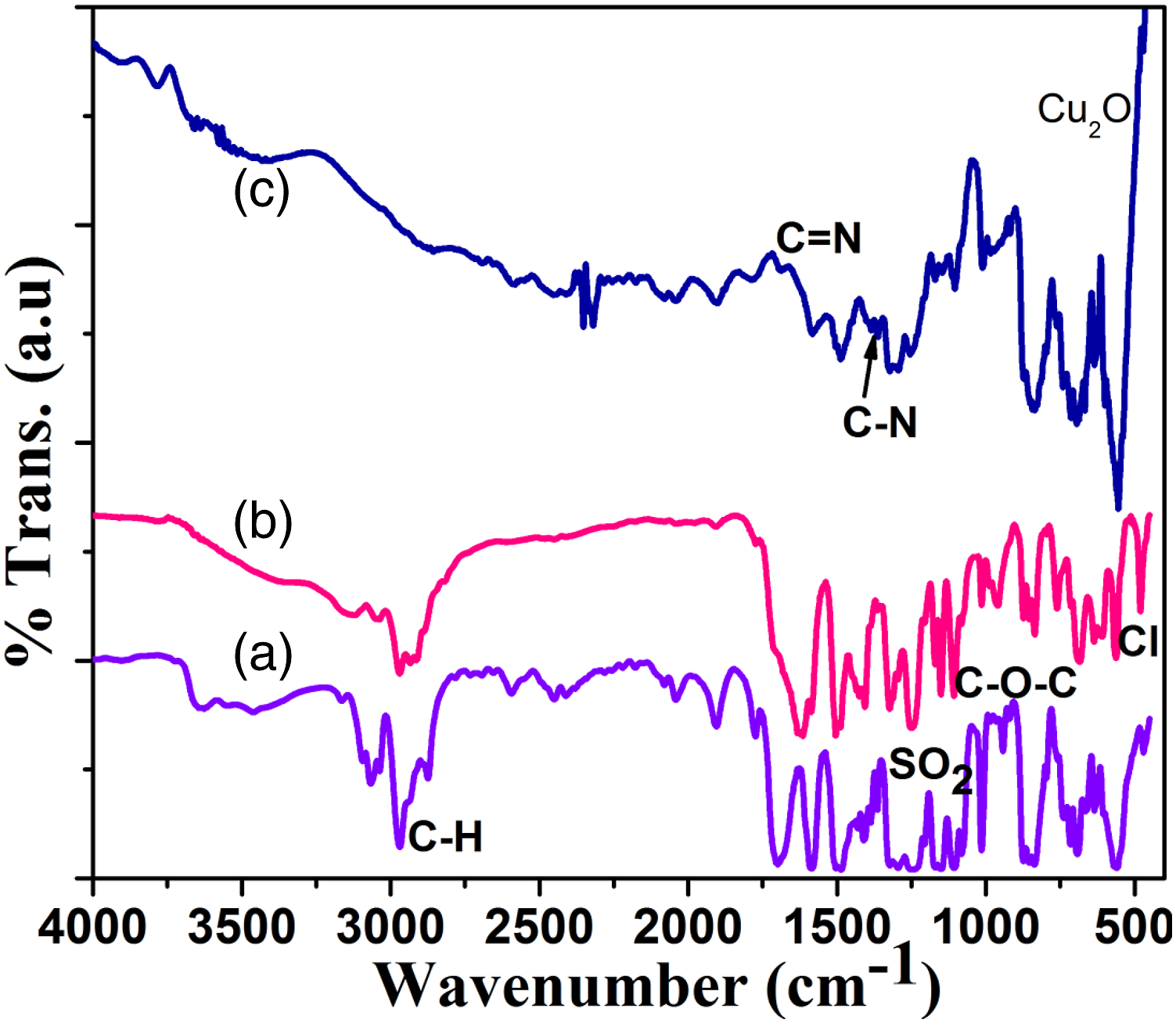
The Tg of PSu was determined from DSC (second heating scan) as 187.6°C (Figure 2(a)).
43
The data is given in Table 1. After the grafting with PVC via FC alkylation reaction, the Tg was increased to 247.7°C (Figure 2(b)). This is due to the increase in molecular weight after the chemical grafting reaction via FC alkylation reaction. 169.2°C was noted as a Tg for the PSu3 system (Figure 3(c)). This low Tg can be explained on the basis of existence of traces amount of NMP solvent. The tertiary nitrogen atom present in the 2MI can interact with the NMP solvent through secondary forces of attraction. In over all comparison, the PSu2 system exhibited the highest Tg value due to the increase in molecular weight and absence of traces of NMP solvent. DSC second heating scan of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system. DSC, TGA and WCA data. TGA thermogram of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.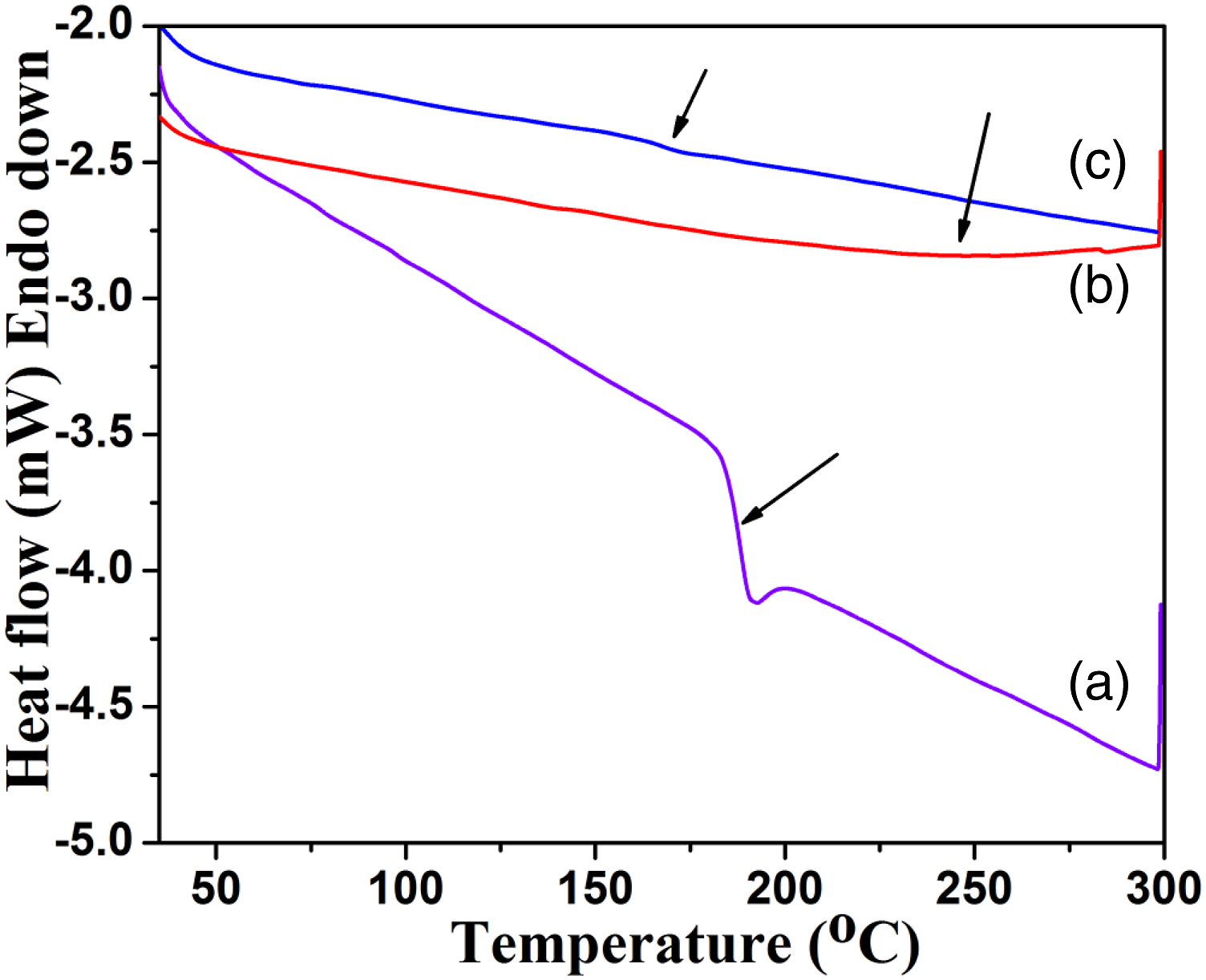
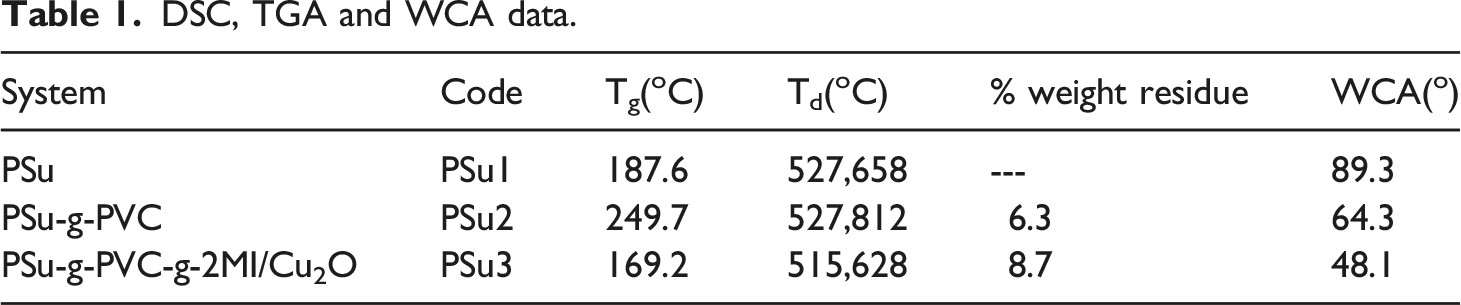
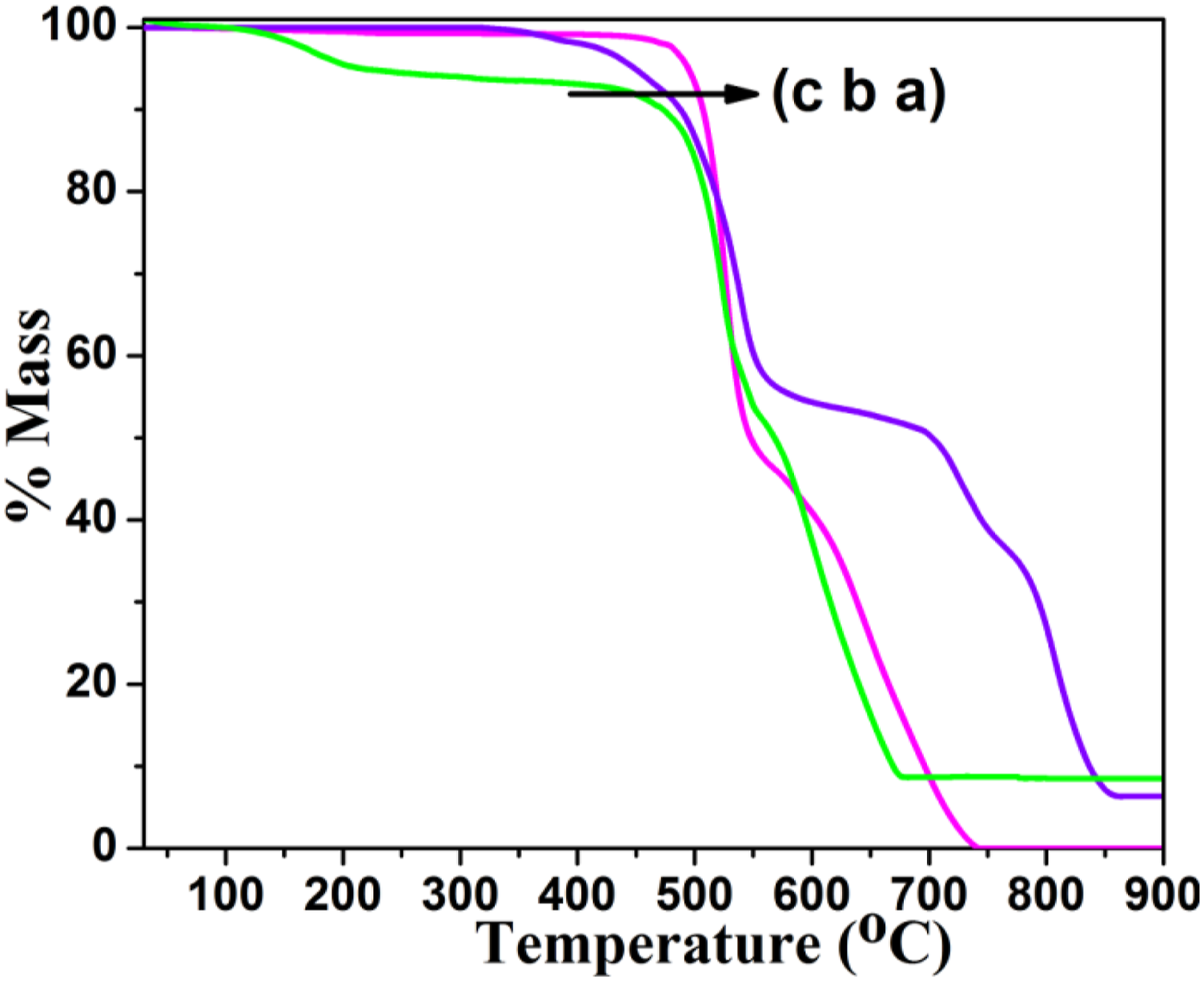
The thermal stability of PSu before and after functionalization reaction was tested by TGA under air atmosphere at the heating rate of 10oC/min. Figure 3(a) represents the TGA of PSu1 system with two step degradation process. The major weight loss appeared at 527°C is ascribed to the degradation of aromatic ether segments. The aromatic sulphone degradation was noticed at 658°C (without any char formation). Figure 3(b) is associated with the TGA of PSu2 system with three step degradation process as mentioned earlier. The aromatic ether segments degraded around 527°C. The grafted PVC degraded at 725°C. The aromatic sulphone segment degraded around 812°C. The increase in thermal stability confirmed the chemical grafting of PVC on to PSu system through FC alkylation reaction. Figure 3(c) denotes the TGA of PSu3 system with three step degradation process. The minor weight loss around 176°C is corresponding to the removal of physisorbed NMP solvent. Both the aromatic ether and aromatic sulphone were degraded at 515 and 628°C respectively. When compared with the literature report 44 the present work yielded higher Td. In over all comparison, the PSu2 system exhibited the highest degradation temperature due to the PVC grafting. In 2016, Banerjee et al. 45 reported the thermal stability of PEEK as 215°C. When compared with the literature the present system yielded a good result.
The XPS of PSu1 system is given in Figure 4(a) with C1s (285 eV), O1s (529 eV) and S2p (167.5 eV). This is in accordance with literature report.
45
After the structural modification with PVC (Figure 4(b)) two more new peaks appeared corresponding to Cl2p (200 eV derived from PVC) and Al2p (86.6 eV derived from the AlCl3 catalyst). Figure 4(c) is owing to the PSu3 system with N1s (396.7 eV) and Cu2p (930.2 eV, derived from Cu2O nanocatalyst). The important point noted here is even after the grafting with 2MI, some amount of Cl from PVC is available for further reaction. XPS of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.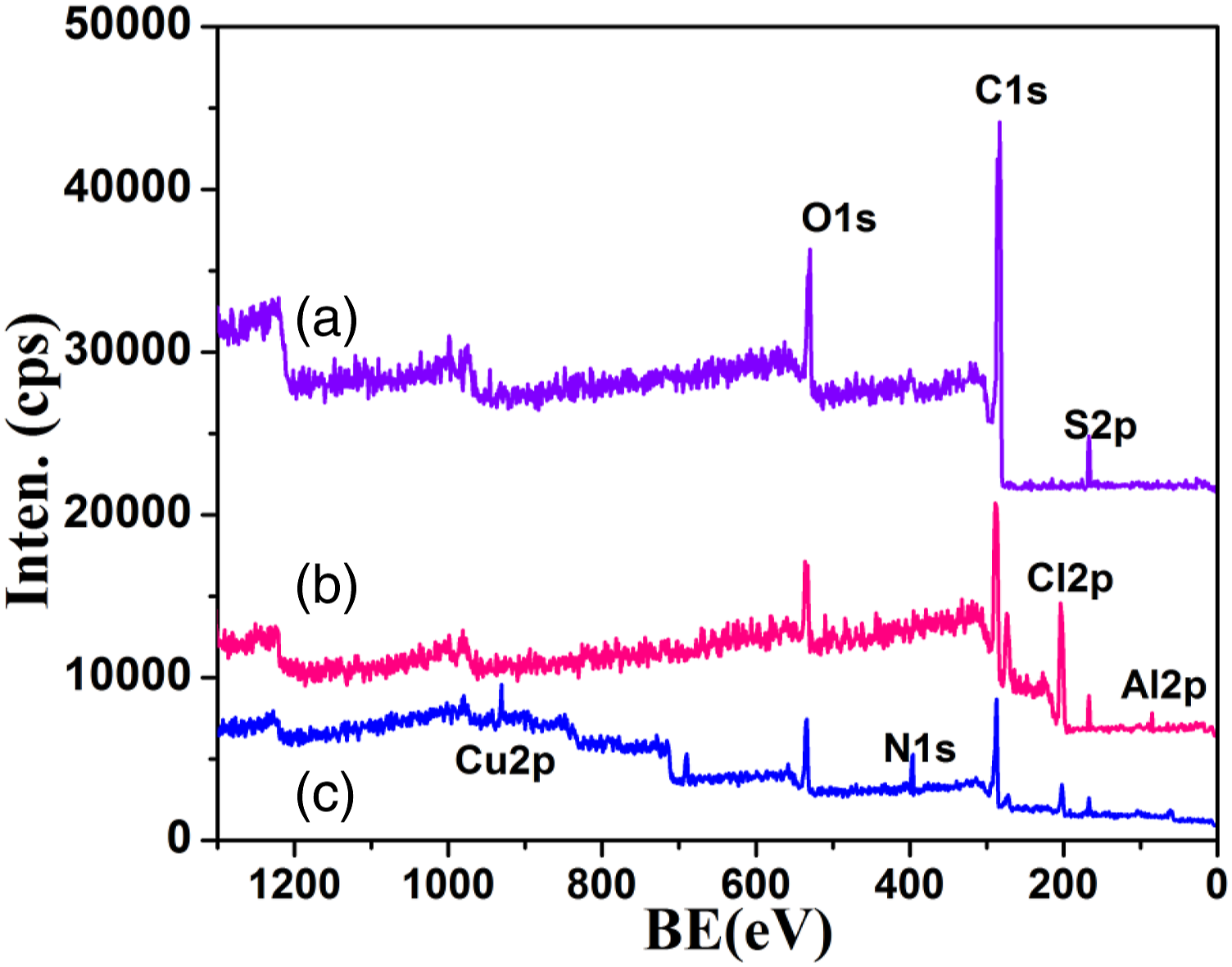
The surface morphology of PSu1 system is given in Figure 5(a) with PSu pattern. After grafting with PVC (Figure 5(b)), the surface morphology was completely changed to distorted spherical morphology. At the same time the PSu3 system (Figure 5(c)) showed more number of voids
46
and suited for the water filtration application. The diameter of the voids varied from 5 to 10 um. Figure 5(d) indicates the surface morphology of PSu3 system with the scale bar of 1um. Here one can see the dispersion of Cu2O nanoparticles (yellow color circled portion) on the PSu-g-PVC-g-2MI backbone. Again one of the targets was achieved. SEM image of (a)PSu, (b)PSu-g-PVC, (c,d)PSF-g-PVC-2MI/Cu2O nanocomposite system.
The EDX of PSu1 system is shown in Figure 6(a) with S, C and O elements. The PSu2 system showed C (68.6%), O (14.2%), S (0.87%), Cl (10.14%) and Fe (6.15%) (Figure 6(b)). The EDX of PSu3 system (Figure 6(c)) showed C (80.5%), N (2.19%), O (14.08%), S (2.86%), Cl (0.15%) and Cu (0.16%). The % content of N, Cl and Cu confirmed the chemical grafting of 2MI onto PSu2 system via FC alkylation reaction. EDX of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.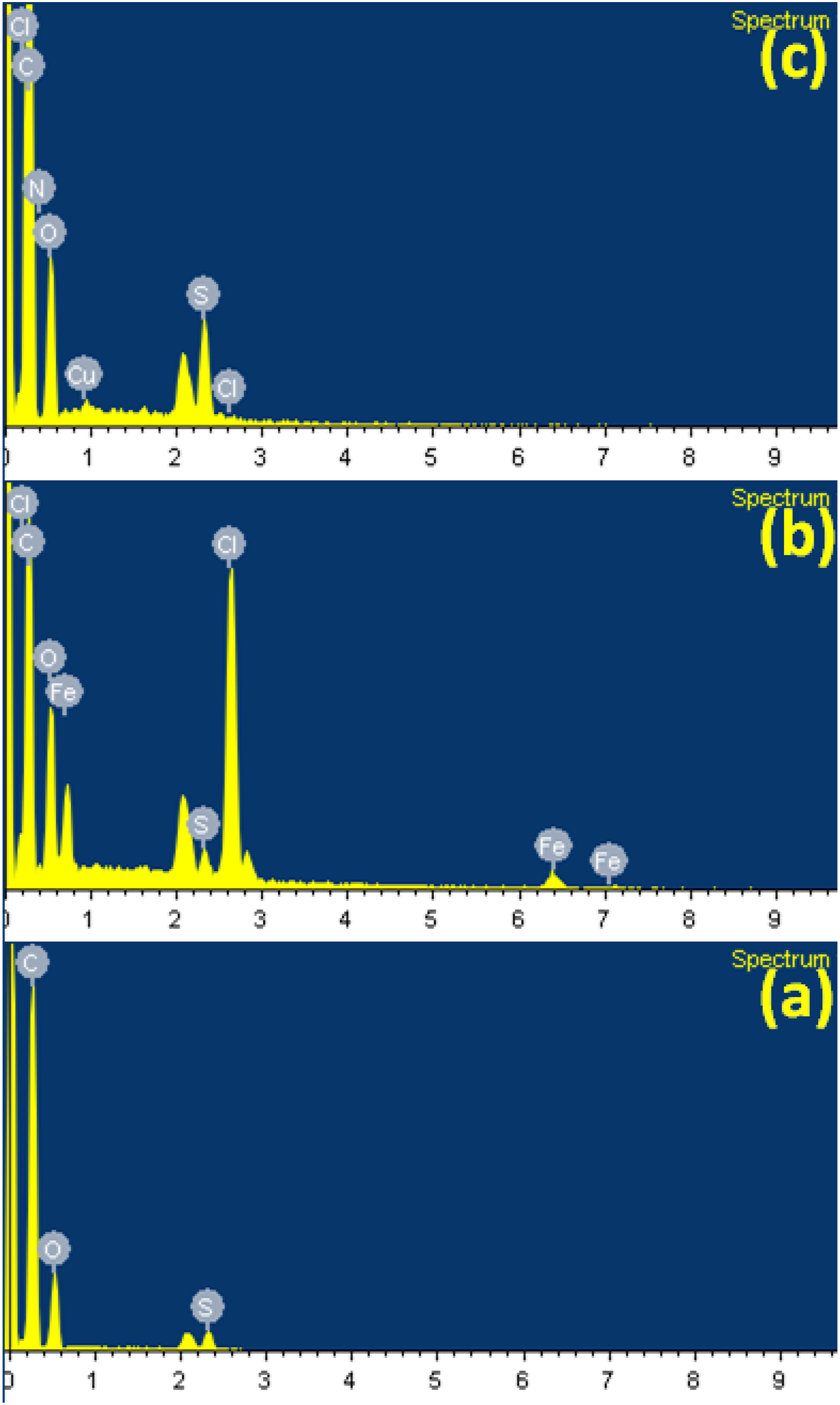
The pristine PSu (PSu1 system) showed the WCA of 89.3o (Table 1, Figure 7(a)). The PSu2 system showed (Figure 7(b), Table 1) the WCA of 64.3o. The decrease in WCA is due to the decrease in rigidity and the introduction of Cl group. The PSu3 system exhibited the WCA of 48.1o (Figure 7(c), Tbale-3). The introduction of two nitrogen atoms drastically reduced the WCA. This is a very important criterion for a membrane material. Hence, one more achievement was made after the structural modification with 2MI. The PSu/poly (dopamine) blend system exhibited the WCA of 53.2o.
47
When compared with the literature report the present system yielded good result due to the presence of C=N like structure. WCA image of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.
The size and internal structure of Cu2O nanoparticle was confirmed by HR-TEM (Figure 8). Figure 8(a) showed the nanorod and nanobelt like morphology, indicated by the arrow mark. Figure 8(b) showed various crystal planes, indicated by a circled portion. This confirmed the crystalline nature of the Cu2O nanoparticles. The length and breadth of the crystal planes are varied from 5–8 nm and <1 nm respectively. Due to its smaller size, it acted as an active catalyst and filler. HR-TEM images (a,b) of PSu-g-PVC-g-2MI/Cu2O nanocomposite system.
The chemical structure of PSu1 system was confirmed by 1H-NMR (Figure 9(a)). The methyl proton signals appeared around 1.6 ppm. The aromatic protons of PSu appeared from 6.7 to 7.9 ppm.
41
The 1H-NMR spectrum of PSu2 system also showed the same proton signals but with some new peaks in Figure 9(b). The CH-Cl proton signal of PVC appeared at 5.38 ppm. The -CH and –CH2 proton signal of PVC appeared at 4.69 and 2.01 ppm respectively. The appearance of new peaks confirmed the chemical grafting o PVC onto PSu backbone via FC alkylation reaction. The 1H-NMR spectrum of PSu3 system is given in Figure 9(c) with some new peaks. Based on the peak integration method, the vinyl proton signal of 2MI appeared at 6.98 ppm was determined. The methyl proton signal of 2MI appeared at 2.40 ppm. Thus the 1H-NMR spectrum confirmed the chemical structure of PSu before and after the structural modification reaction. 1H-NMR spectrum of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system.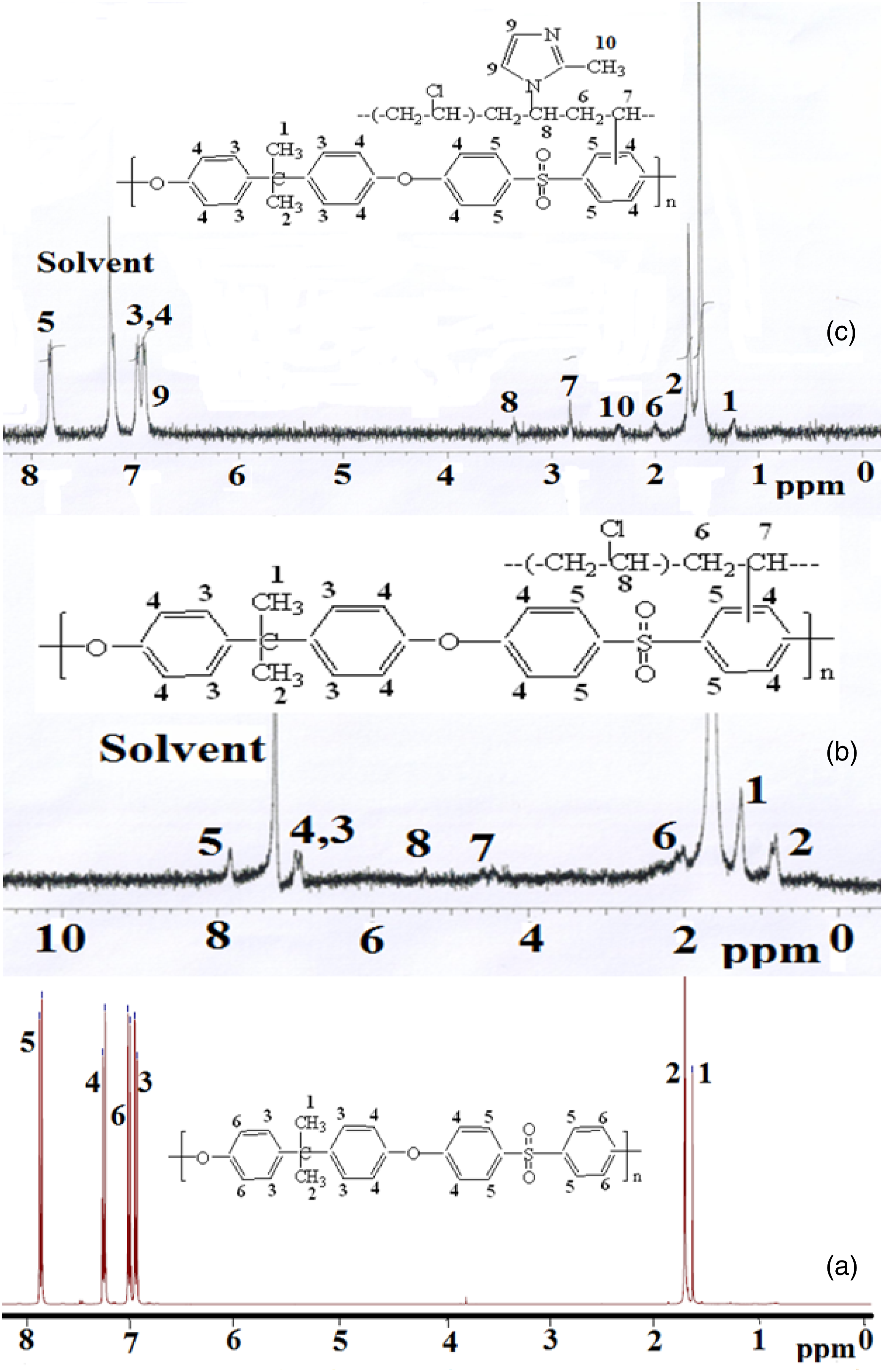
The main aim of the present research work is to increase the TS, hydrophilicity and antimicrobial activity of PSu. The TS of PSu1 system was determined as 2.18 MPa (Figure 10(a)) (Table 2). The PSu2 and PSu3 system exhibited the same as 2.32 MPa and 3.05 MPa (Table 2) respectively from Figure 10 b and c. The poly (n-butylacrylate) grafted PSu exhibited the tensile strength value of less than 2.5 MPa.
48
In comparison with literature value, the PSu3 system exhibited the highest TS value. This is due to the perfect filling action of hydrophobic Cu2O nanoparticles. Actually, Cu2O nanoparticle was used as a catalyst. Hence, it acted both as a catalyst and filler. The inorganic hydrophobic Cu2O nanoparticle filled the voids between the polymer chains. As a result, the TS of PSu3 system was increased by 39.9%. Finally, one of the targets was achieved. Stress-Strain plot of (a)PSu, (b)PSu-g-PVC, (c)PSF-g-PVC-2MI/Cu2O nanocomposite system. TS data.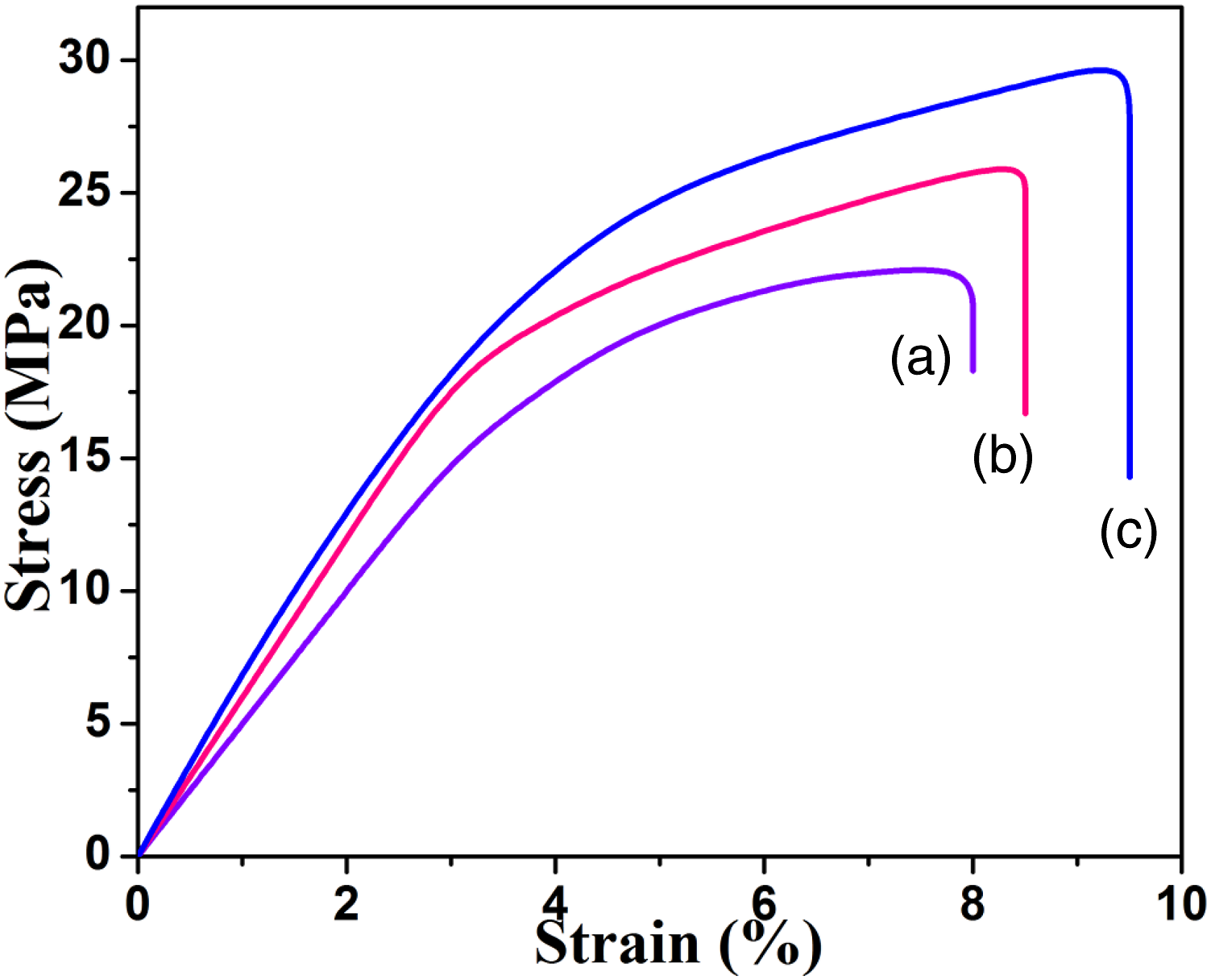

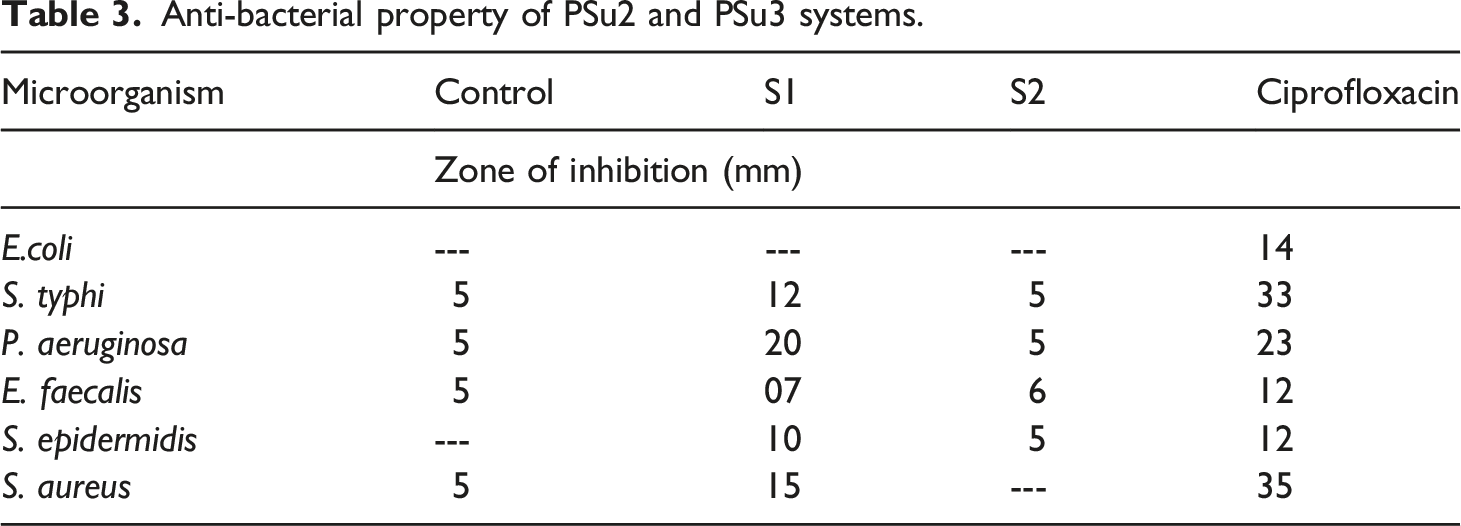
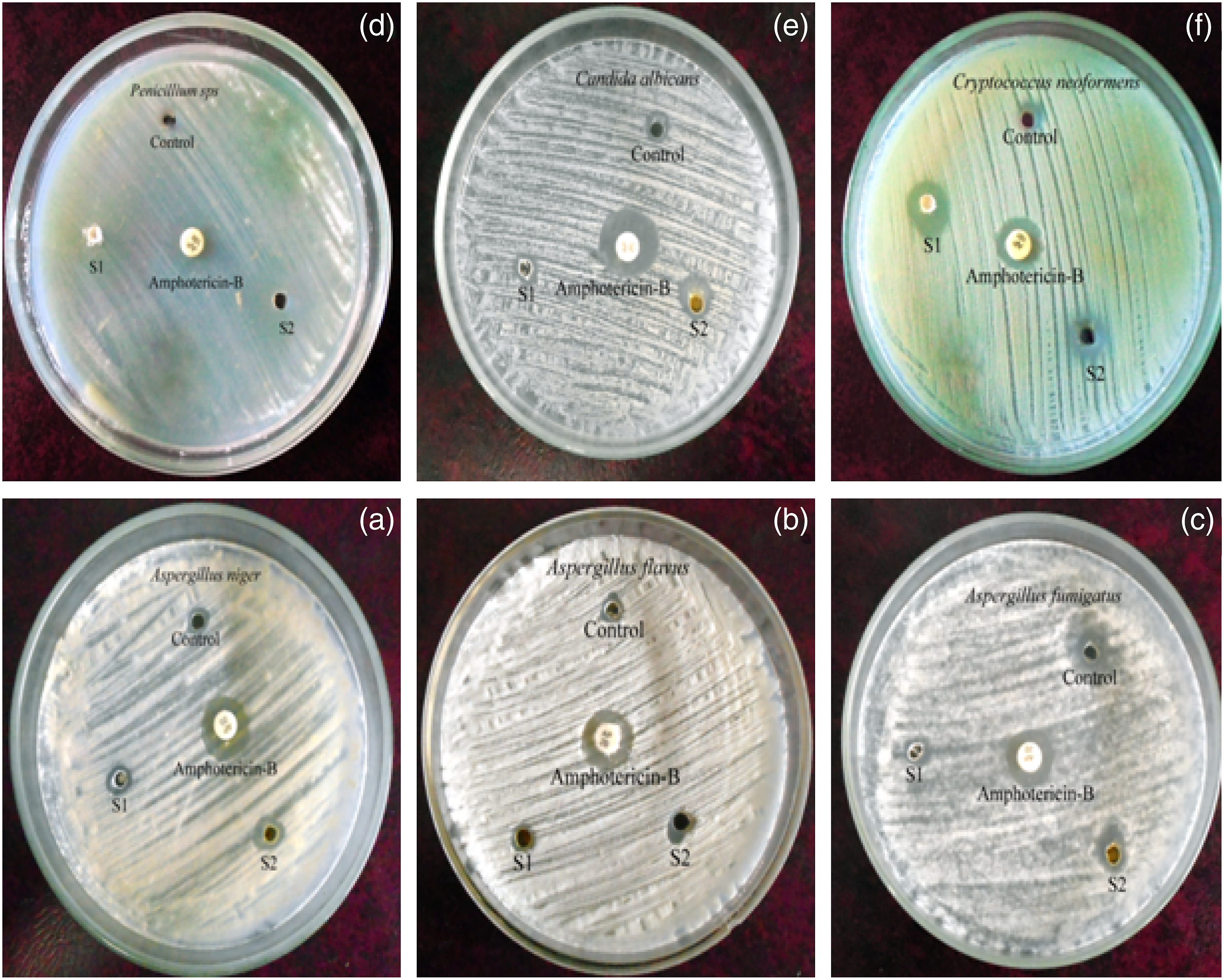
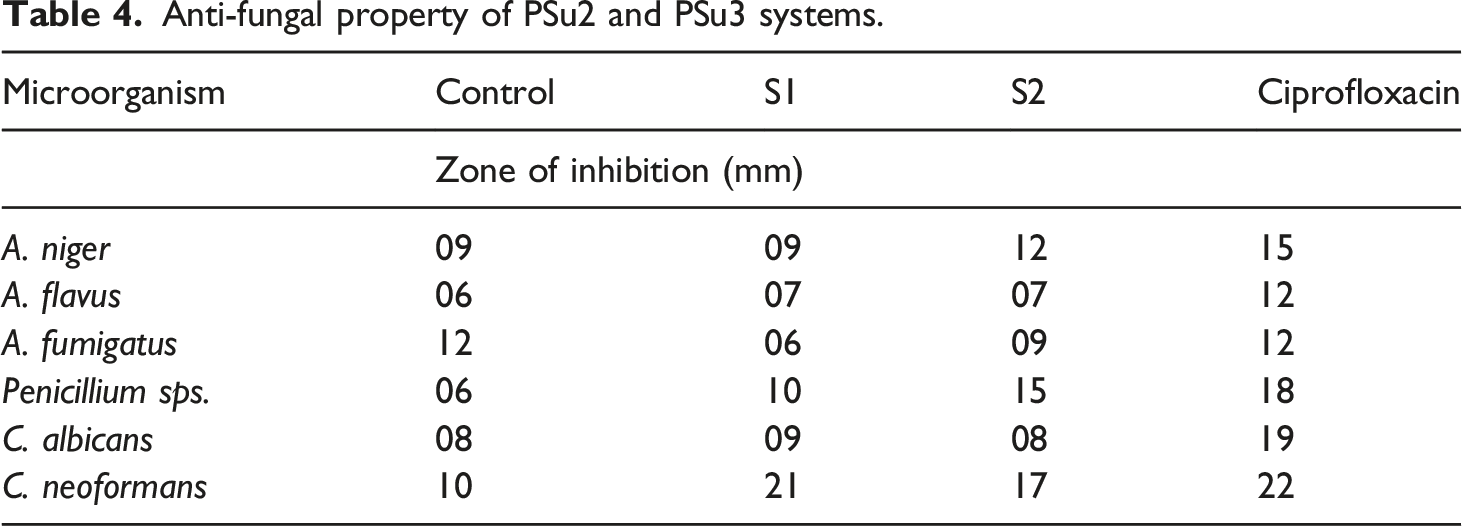
The drawback of PSu is absence of antibacterial and antifungal activities. In the present research work, due to the presence of C=N structure, mild antimicrobial property was imported to PSu. Water living bacterial systems were tested with PSu2 and PSu3 systems. Among the six bacterial systems considered, the PSu3 system exhibited 20 mm against P.aeruginosa (Figures 11(a)–(f)) (Table3). At the same time the PSu2 system exhibited 5 mm only. This confirmed that the PSu3 system has antibacterial activity due to the presence of C=N structure. The antifungal testing results declared that the PSu3 system showed the highest antifungal activity against C.neoformans (Figure 12 a-f) (21 mm) (Table 4). Again this proved that due to the presence of C=N structure in 2MI both the antifungal and antibacterial properties were introduced to PSu material. The PSu/GO/Ag nanocomposite system exhibited the antibacterial properties due to the presence of Ag nanoparticles.
40
In the present research work due to the presence of C=N structure in 2MI, the antimicrobial properties were imported to PSu. Hence, the system reached the target of the present research work. Anti-bacterial activity of PSu2 (S1) and PSu3 (S2) systems against (a)E. coli, (b)S. typhi, (c)P. aeruginosa, (d)E. faecalis, (e)S. epidermidis, (f)S. aureus. Anti-bacterial property of PSu2 and PSu3 systems. Anti-fungal activity of PSu2 (S1) and PSu3 (S2) systems against (a)A.niger, (b)A.flavus, (c)A.fumigatus, (d)Penicillium sps., (e)C.albicans, (f)C.neoformans. Anti-fungal property of PSu2 and PSu3 systems.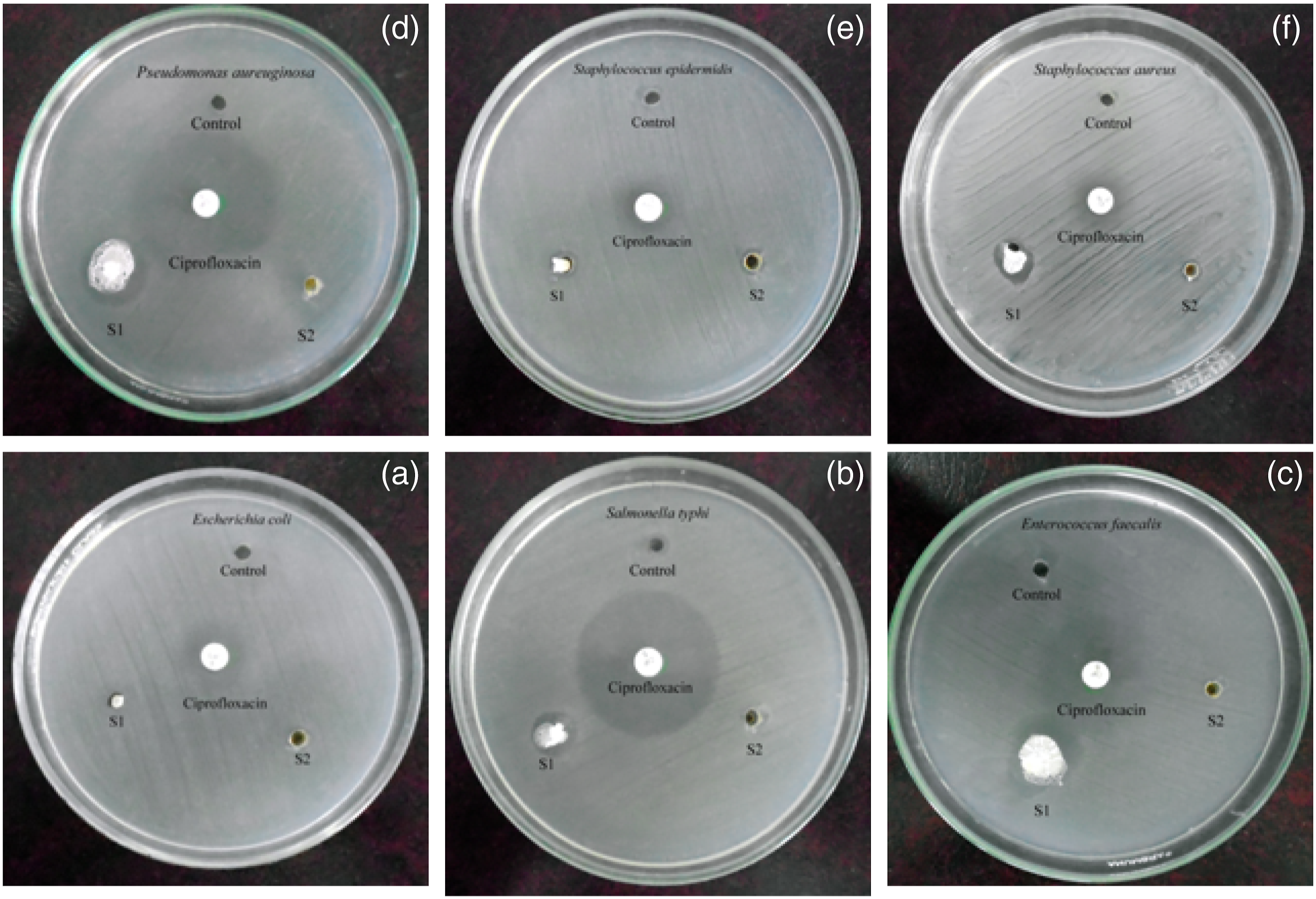
Conclusions
The important results of the present investigation was carefully analyzed and presented here as conclusion. The FT-IR spectrum showed the peaks corresponding to C-N stretching of 2MI, Cu2O stretching of Cu2O nanoparticle, chloride ion stretching of PVC and SO2 stretching of PSu and confirmed the chemical grafting of 2MI onto PSu-g-PVC backbone. Further, it was confirmed by 1H-NMR spectrum by noting vinyl proton signals around 7 ppm. The PSu3 system showed lower Tg value due to the presence of traces amount of NMP solvent. The TGA declared the enhanced thermal stability of PVC grafted aromatic sulphone unit of PSu. The XPS showed the Cu2p peak around 930 eV and confirmed the nanocomposite formation. The SEM image authenticated that the PSu3 system is a suitable candidate for the water filtration application due to the presence of more number of voids. The EDX showed that 0.15% Cu available in PSu3 system. The hydrophilicity of PSu3 system increased after the grafting with 2MI. The HR-TEM confirmed the presence of Cu2O nanoparticle with various crystal planes. The TS of PSu3 system was determined as 3.05 MPa due to the chemical grafting of PVC and 2MI/Cu2O. The PSu3 system exhibited excellent antibacterial and antifungal properties than the PSu2 system due to the presence of C-N like Schiff base structure. The main targets of the present investigation were achieved. The decrease in surface water contact angle and increase in tensile strength of the resultant membrane is the novelty of the present research work. Next our research team is going to concentrate on the improvement of antimicrobial properties of PVDF membrane.
Footnotes
Acknowledgements
We express our sincere thanks to Dr.N. Sundararajan, Associate Professor, Department of English, KCET, Madurai for his valuable help during the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s note
Mr.K. Dheepak Vishnu carried out the experimental work. Dr.R. Anbarasan did the characterization work and wrote the research paper. Prof.K.L. Tung corrected the content of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Ministry of Science and Technology (MOST) in Taiwan (Project number: 109-2221-E-002-102-MY3, 110-2622-E-002-014 and 111-2634-F-002-016), and “Advanced Research Center of Green Materials Science and Technology” (111L891801) from The Feature Area Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE), Taiwan (111L9006).
