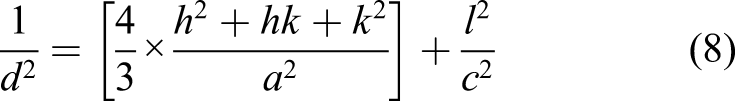Abstract
A simple wet-chemical synthesis method was developed to fabricate Zinc oxide micro-rod clusters. The synthesis process involved the rapid dilution of a zinc-bearing alkaline solution at 150°C in a convection oven on indium tin oxide (ITO) substrates. The synthesis was carried out by immersing an unseeded ITO substrate in a mixture of zinc nitrate hexahydrate (Zn(NO3)2·6H2O) and hexamethylenetetramine ((CH2)6N4) aqueous solution. The obtained sample was annealed at 400°C for 2 h. The structural, morphological and optical properties of the synthesized ZnO microstructures were investigated by X-ray diffraction, scanning electron microscopy and ultraviolet-visible spectroscopy, respectively. The ZnO rod clusters are hexagonal phase of the wurtzite structure. The crystal grain sizes of the films were found to be 72.6, 84.3 and 66.3 nm for the (100), (002) and (101) crystal planes, respectively. The optical bandgap of the ZnO was determined to be 3.147 eV.
Introduction
Zinc oxide (ZnO) is one of the most commonly used metal oxides with special physical and chemical properties for various applications. 1 ZnO is a wellknown semiconductor material with a wide and direct bandgap (3.37 eV) at room temperature. 2 It has a wurtzite crystal structure with a hexagonal lattice belonging to the P63mc space group. It can be considered as numerous planes composed of tetrahedrally coordinated O2− and Zn2+ ions, alternately stacked along the c-axis under ambient conditions. 3 Notably, the hexagonal wurtzite-type structure facilitates the growth of various one-dimensional (1D) ZnO structures. 4 ZnO can be synthesized as 1D structures, such as nanorods, nanosprings and nanoneedles; 2D structures, such as nanosheets and nanoplates; and 3D structures, such as nanoflowers and urchin-like pores. 5
Depending on the reaction conditions and reactants, the starting precursors can be engineered into miscellaneous nanostructures, such as particles, rods, tubes, thin films, plates, stars, wires, belts or hollow spheres. The morphology is a crucial parameter that controls the properties and functionality of the materials. 6 Materials with the desired properties can be designed for potential applications by controlling their morphologies; therefore, the morphology and property control of materials is highly important. 7 For example, nano and microstructured ZnO, which exhibits enhanced optical properties and a large surface area, is used for photodetector applications. 8
The growth of ZnO micro-rods/tubes with a hexagonal cross-sectional geometry provides a novel multifunctional platform for various applications, such as wave-guided whispering-gallery mode sensors (WGMs), low-dimensional transistors, micro-electromechanical systems, ultraviolet (UV) detectors, lasers and photovoltaics.9–12 Moreover, ZnO microor nano-rods/wires can be used as high-performance photocatalytic materials. 13
ZnO micro/nanostructures are usually prepared by various methods, such as hydrothermal synthesis, 14 microwave coating, 15 thermal chemical-reaction vapour-transport deposition, 16 modified chemical-vapour deposition, 17 spin coating, 18 vapour–liquid–solid (VLS)19,20 and flame-transport techniques.4,21,22
In our previous study, 23 micro-rods were synthesized at 150°C on glass substrates via a wet-chemical technique. The advantages of this technique over the aqueous-chemical method and chemical-bath deposition method were mentioned. Using an aqueous-chemical method, it was observed that the growth mechanism was much faster on the ITO substrate than on the glass substrate, and the growth of nano and micro rods was more supported by ITO-coated substrates, rather than glass substrates, in this particular case, which show high optical properties. 24
In the present study, ZnO micro-rod clusters were prepared by a wet-chemical method and high-transmittance ZnO micro-rods were obtained with a faster process than the aqueous-chemical method. The values of certain parameters were changed for this study to obtain highly transparent films on ITO substrates with cluster morphology, which can be used for optoelectronic applications. For example, ZnO (doped or undoped) can be either used as a current spreading layer or an electrode in light-emitting diodes and solar cells, as well as a buffer layer. 25 In particular, ZnO, which exhibits enhanced optical properties and a large surface area per constant volume, is desirable; recently, photodetectors using micro or nanostructured ZnO with enhanced performance, such as high responsivity and gain, have been reported. 8 A UV photodetector with single ZnO microrods introduced an optical whispering gallery mode effect that enhanced photocurrent and photoresponsivity. 26 Also, it has been reported in the literature that a self-powered, low-cost, photoelectrochemical cell-structured UV light photodetector was assembled using ZnO/ZnS core–shell nanorod arrays as the photoanode; vertical ZnO nanorod arrays were uniformly grown on FTO glass by a hydrothermal method. 27 A flexible UV photodetector using ZnO nanorods was prepared via a facile CBD process and a high-performance UV photodetector using a ZnO@A-SWNT hybrid film was prepared via a facile and fast spin-coating process.28,29 Both of the two techniques are easy solution-based techniques similar to the technique we use.
Materials and method
This study used the same method as that in a previous study.
23
ZnO rods were grown by wet-chemical synthesis on ITO glass substrates at 150°C. Zn (NO3)2·6H2O and ((CH2)6N4) were used to prepare a bath solution (0.2 The synthesis chart of the ZnO rods.
In this study, zinc oxide rods were prepared using a simple wet-chemical method. This method is similar to CBD and hydrothermal methods. The CBD process is usually performed on a heating plate; thus, the solution is heated by conduction, and it rapidly evaporates at high temperatures. Conversely, hydrothermal processes are carried out using a hydrothermal reactor for synthesis at high temperatures. In this study, the solution evaporated very slowly because the reaction was carried out at 150°C in a convection oven without using a hydrothermal reactor. In this study, initially at low temperatures, a highly crystalline ZnO nucleation was obtained; whereas at higher temperatures, as the solution evaporated, the concentration increased, causing the ZnO clusters to grow, owing to the unseeded ITO substrate. Because the convection oven was used in the present study, the solution evaporated slowly at a high temperature (150°C). Thus, micro-rod clusters were deposited on the ITO.
First, ZnO nucleations were formed on the substrate. At high temperatures in the convection oven, the nanorods began to grow by the following reaction. Because there was no seed layer on the substrate, the cores grew randomly on the surface. The reactions are as follows
Next, an improved crystal structure is obtained by annealing the films.
The structural properties of the synthesized samples were studied using a PANalytical Empyrean X-ray diffractometer (XRD) operated at 45 kV and 40 mA with CuKα radiation (λ = 1.5406 Å; Scanning range: 10.0051–89.9811°(2Th); Scanning type: continuous; incidence angle: ∼50° (2Th); methodology: gonio mode; stage: flat; slit: 1/2–1; and scanning rate: 0,122,142 (°/s)). The morphologies of the samples were studied using an FEG Quanta 450 field-emission scanning electron microscope (FESEM). The optical properties of the samples were studied using a TETRA T80+ UV-Vis spectrometer.
Results
The XRD patterns of the synthesized ZnO micro-rods are shown in Figure 2. All XRD peaks matched well with those of the wurtzite (hexagonal) ZnO.
30
The pattern shows the (100), (002), (101), (102), (112), (110), (103) and (112) peaks of the wurtzite ZnO crystal structure (JCPDS No. 36–1451). The (100) peak of the ZnO nanostructures exhibited the maximum intensity. This indicates that the surface energy of the (100) plane is lower than that of the other planes.
31
XRD spectrum of ZnO micro-rod clusters.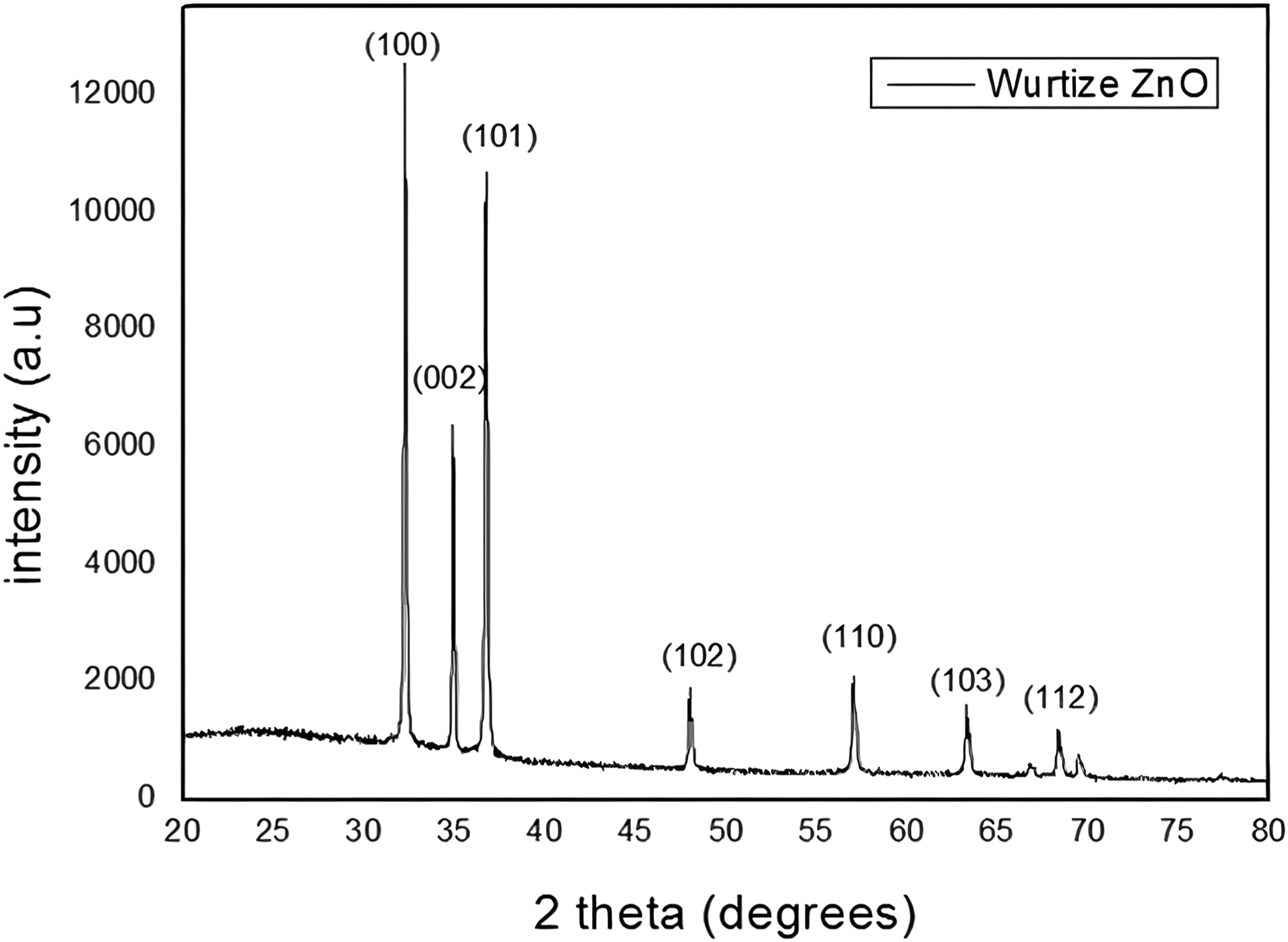
The structural parameters of ZnO micro-rods clusters.

The dislocation density (δ), which is defined as the length of dislocation lines per unit volume, is a measure of the number of defects in a crystal; therefore, a small δ value indicates a good crystal structure. 38
δ was estimated using equation (10)
The structural parameters of the film determined from the XRD data are listed in Table 1. The small δ value and large D value for the sample indicate that the crystal structure along the (002) direction is better than that along other orientations.
It is well known that the reaction temperature determines the formation and morphology of ZnO structures. 39 The formation of ZnO rod assemblies involves the initial precipitation of an ε-Zn (OH)2 intermediate, followed by a relatively slow-phase transformation process. 40 It is well known that supersaturation is a prerequisite for crystal growth in solution and is intimately connected with growth processes involved in the evolution of crystal morphology. 41
In general, Zn (II) ions are solvated by water and exist as monomeric hydroxyl species, such as Zn(OH−) (aq), Zn(OH)2 (aq) and Zn(OH)2- (aq). 41 The ZnO formation mechanism involves two steps: (1) nucleation, resulting from the decomposition of clusters of molecules with increasing temperature, and (2) particle growth, resulting from heterogeneous reactions, wherein particles combine to form nuclei on the substrate. 42 However, because no seed layer is formed on the substrate before synthesis, the rods are deposited on the ITO surface as clusters.
The SEM images of the micro-rod clusters are shown in Figure 3. These images were recorded at magnifications between 1k and 50k. The ZnO micro-rods grew randomly oriented as clusters because of the unseeded nature of the ITO substrate. On the other hand, the ZnO micro-rods exhibit a strong peak, corresponding to the (100), (002) and (101) planes of the wurtzite ZnO crystal structure, which confirms that the structures grow along the surface and have high crystalline.
43
SEM images of ZnO micro-rod clusters at (a) 1000, (b) 10,000, (c) 25,000 and (d) 50,000 magnifications.
The energy-dispersive X-ray spectroscopy (EDS) spectra of the micro-rod clusters are shown in Figure 4. The intensities and percentages of Zn and O are shown in the figure. It reveals that only Zn and O elements were present in the ZnO, and impurities could not be found within the EDS detection limit. The EDS spectrum of the ZnO reveals the atomic ratio of the elements: Zn/O: 53.67/46.33 (∼1.15; this means that there is little excess Zn in the ZnO structure). A compositional analysis using EDS was performed on ZnO micro-rod clusters. EDS spectrum of ZnO micro-rod clusters.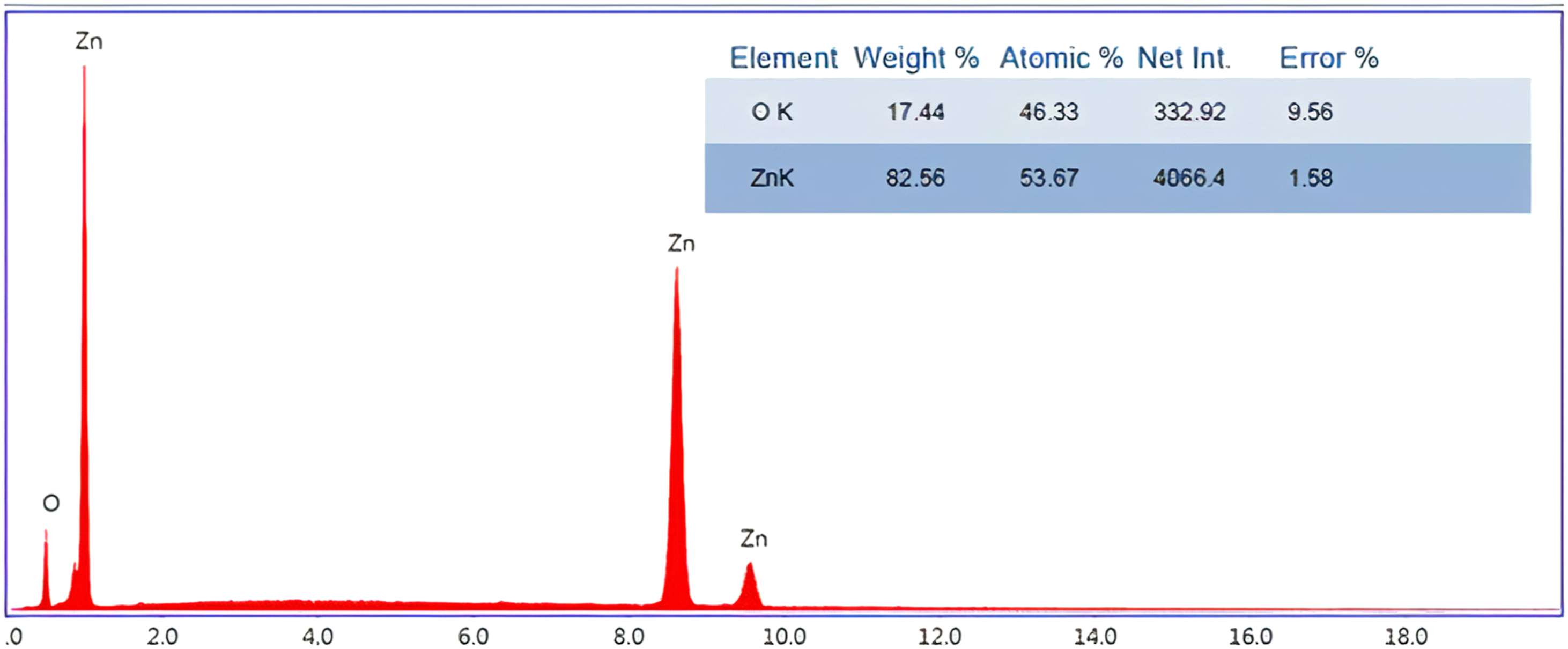
Figure 5(a) shows the UV-Vis transmittance (%T) of the ZnO micro-rod clusters. The ZnO film exhibited high transmittance. The optical bandgap spectrum ((αhν)2 vs. hν) of the ZnO clusters is shown in Figure 5(b). One of the widely used techniques to determine the bandgap energy (Eg) of semiconductor materials is optical absorption spectroscopy.18,30,44 Alternatively, the band gaps of semiconductors can be determined electrically. However, optical spectroscopy is easy to implement. In several studies, Eg was calculated using the Tauc plot. The approximate optical bandgap value of the sample can be calculated from the UV-Vis absorbance data using the Tauc equation, as given below (a) % T-wavelength (nm), (b) (αhυ)2 versus (hυ) spectra of ZnO micro-rod clusters.

Transmittance (T%) and bandgap energy (Eg) values for ZnO micro-rods clusters.

Conclusion
In this study, a rapid and simple methodology was developed to synthesize highly crystalline and high-transmittance zinc oxide micro-rod clusters. They were obtained via a rapid and one-step wet-chemical synthesis at 150°C on unseeded ITO substrates in a convection oven. The microstructures possessed a wurtzite crystal structure. The crystal grain sizes of the films were found to be 72.6, 84.3 and 66.3 nm for the (100), (002) and (101) crystal planes, respectively. The optical bandgap of the ZnO was determined to be 3.147 eV. The originality of this study is the synthesis of high-transmittance zinc oxide microstructures on an ITO glass substrate by a rapid one-step process in a convection oven. The zinc oxide microstructures grown by this technique could be used for microscale device applications. Especially, these structures, which were easily obtained in a short time, can be exhibited high performance in various applications of optoelectronic technology such as photodetectors, gas sensors and solar cells. In particular, zinc oxide microrods with high porosity, grown by this facile novel technique could be potentially used for the fabrication of UV detectors because of their special properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.