Abstract
Although the construction of superwettability materials for oil-water separation has become a research hotspot, it is still a thorny issue due to its easy pollution to the environment during the preparation process. In this paper, we reported the functional PP/EPDM TPV film (with a contact angle of oil and water is 0° and 157.9±2.9°) as superhydrophobic and superoleophilic materials with reproducible processing characteristics at high temperature; moreover, during the molding process the etched aluminum foil was used as a template for the preparation of functional TPV film which was used in the efficient separation treatment of oily wastewater. It should be noted that a novel method was proposed for the continuous oil-water separation via a pressure response through valve, which was different from the traditional gravity-driven method and the permeable oil-water separation. The flexible PP/EPDM TPV film was rolled up and loaded into the through valve and the gap between films was used as the pressure response channel of oil-water separation. The novel functional TPV film used in the continuous oil-water separation was recyclable, environmentally friendly, and easy to industrialize, indicating the great potential in practical applications.
Keywords
Introduction
Superhydrophobic surfaces present a great high water contact angle (WCA >150o) as well as a low water sliding angle (WSA <10o).1,2 This phenomenon can be widely found in nature, the most well-known was the “lotus effect,”3–4 which inspired the research of superhydrophobic surfaces around the world. Moreover, the superhydrophobic surfaces have also attracted considerable attention owing to their emerging usage in industrial applications and in our daily life, which can be used to achieve self-cleaning,5,6 oil-water separation,7,8 anti-corrosion, 9 and low-drag. 10 Generally, the superhydrophobic/superoleophilic surfaces have been fabricated by two strategies: constructing a micro-nano rough structure on the hydrophobic matrix, or subsequently modifying the rough structure surface using the low-surface energy materials. 11 Based on these two principles, numerous methods have been successfully applied in preparing superhydrophobic surfaces including chemical vapor deposition, 12 templating method,13,14 electrospinning technique, 15 sol-gel process,16,17 UV light curable reaction, 18 and other methods 19,20 to develop the waterproof surfaces. There has been plenty of work for the templating method to develop the waterproof surfaces because of the low cost and the simple operation. 21 Thus, it was desirable to integrate a superhydrophobic material on the basis of the template method with higher efficiency of oil-water separation, excellent durability and recyclability.
In recent years, superhydrophobic materials have been widely used in oil-water separation due to their superwettability to oil and repellency to water. 22 In particular, the membrane separation method was considered to be an alternative technology for the separation of oil-water mixtures due to its simplicity, high efficiency, 23 low cost, outstanding flexibility, 24 and environmental friendliness. 25 Hereinto, Yuan et al. fabricated the thermo-responsive polyvinylidene fluoride (PVDF)/poly (stearyl methacrylate) (PSMA) composite membranes with a highly hydrophobic surface 26 ; Du et al. prepared a TiO2 coated stainless steel mesh membrane with the dual superhydrophobicity, which can be used to separate heavy and light immiscible oil-water mixtures 27 ; Yang et al. fabricated the cellulose acetate (CA)/SiO2-TiO2 hybrid microsphere composite aerogel films. 28 Even if these superhydrophobic materials for oil-water separation have achieved great success in the laboratory, there are still limitations in the industry to the high cost of the material itself and the difficulty of industrialization. Moreover, the preparation of most membranes requires a large number of chemical reagents, 29 the recovery process is still time-consuming, 30 and the necessary pretreatment may cause secondary pollution. 31
To solve the aforementioned problems and according to the related research of H. Yildirim Erbil, we only need to produce two different sizes of scale roughness on a surface to obtain a superhydrophobic surface. 32 Therefore, thermoplastic vulcanizates (TPVs) will be successfully applied to the preparation of superhydrophobic/superoleophilic films with high processing properties and recyclability. TPVs have the same elastic and mechanical properties as conventional vulcanized rubber while it can also be reprocessed or recycled like a thermoplastic at the elevated temperature. 33 Now, TPVs are irreplaceable in the industrial field, especially in cable, automotive seals and home appliances applications because of their excellent processing performance. 34 Due to its structural characteristics, ethylene-propylene-diene terpolymer (EPDM) rubber is widely used in automotive materials with excellent aging resistance, such as ozone resistance, heat resistance, and weather resistance. 35 Polypropylene (PP) is a non-toxic, odorless, thermoplastic with excellent electrical properties and high-frequency insulation. 36 On this basis, TPV prepared with EPDM/PP as the matrix is the most important and versatile TPV product, and industrialization has been commercially mature. In this sense, applying TPV materials to superhydrophobic/superoleophilic materials through certain modifications has a distinct meaning.
In this work, based on the template method, a common and feasible approach to fabricate the superhydrophobic/superoleophilic PP/EPDM TPV films by using etched aluminum foil as the template was researched. The whole process was easy to operate without low-surface-energy fluorine-containing chemical reagents, which were simple, environmentally friendly, and easy to scale up. Based on the superhydrophobic and superoleophilic properties, the films could easily remove and recycle the oil spills from the water surface. As a result, the prepared superhydrophobic/superoleophilic TPV film was applied to a new type of oil-water separation device assembled in the laboratory, which were driven by the pressure response of oil-water separation valves, showing the high oil-water separation. Moreover, the functional TPV films with good corrosion resistance to organic solvents, reusability and mechanical durability were ideal materials for oil-water separation.
Experimental
Materials
Polypropylene (K8303 type) was commercially supplied by Yanshan Petrochemical Co., Ltd., China, with 17.8 (mol%) of ethylene and a melt flow rate (measured at 230°C and 2.16 kg) of 1.39 g 10 min−1. EPDM rubber [EP33 type, with a diene component 5-ethylidene-2-norbornene, diene content of 8.1 wt.%, ethylene content of 52 wt.%] was purchased from Japan Synthetic Rubber Co., Ltd., Japan. Sulfur was obtained from Hengye Zhongyuan Chemical Co., Ltd., China, used as a vulcanization agent. N-Cyclohexyl-2-benzothiazole sulfonamide (CZ) and tetramethylthiuram disulfide (TMTD) were all obtained from Northeast Auxiliary Chemical Industry Co., China, used as accelerators. Zinc oxide (ZnO) was obtained from NewLe Qinshi Zinc Co., Ltd., China, used as an activator. Stearic acid obtained from Wanyou Co., Ltd., China, used as an activator. Poly (1,2-dihydro-2,2,4-trimethyl-quinoline, antioxidant RD) was obtained from Shengao Chemical Co., Ltd., China, used as an antioxidant. Aluminum foil (0.1 mm thickness, 1060 type) was commercially obtained from Suzhou Hairun Metal Materials Co., Ltd., China, used as a template. W10 grade metallographic sandpaper was commercially obtained from Shanghai Grinding Wheel Co., Ltd., China. N-hexane for oil-water separation was received from Tianjin Fuyu Fine Chemical Co., Ltd., China.
Preparation of PP/EPDM TPV
Commercially available PP and EPDM, as described above, were used for the preparation of TPVs. The formulations for crosslinking the EPDM system were expressed in parts per hundred EPDM by weight (phr). The sulfur-containing accelerating system recipe the following ingredients: 100 phr EPDM, 1.0 phr sulfur, 2.0 phr CZ, 0.5 phr TMTD, 5.0 phr ZnO, 1.5 phr stearic acid, and 1.5 phr antioxidant RD.
Detailed preparation procedures have been reported in our previous studies. 37 The weight ratio of PP/EPDM was fixed at 50/50.
Preparation of aluminum foil template
First of all, the aluminum (Al) foil (3 × 10 cm2) is polished mechanically by the W10 grade metallographic sandpaper to remove the oxide film rapidly, then cleaned ultrasonically in ethanol to remove the superfluous aluminum and dried thoroughly at 26°C. Second, the Al foil was immersed into the 5.4 wt.% HCl solution for a period of time; and then sonicated with the deionized water. Finally, the etched Al foil templates were dried in a vacuum oven (30°C) for 5 min to achieve roughness.
Preparation of Superhydrophobic/Superoleophilic PP/EPDM TPV Film
The PP/EPDM TPV film was modified in a simple step to achieve superhydrophobic properties where the etched Al foil was used as a template. The abovementioned TPV was placed on the surface of the etched Al foil template, then preheated in a flat vulcanizer (SKZ401, SKZ Industrial, Co., Ltd., China) at 180°C for 8 min. Second, the TPV was molded with the etched Al foil template at the pressure of 2 MPa at 180°C for 3 min and then cooled at room temperature for 5 min. Finally, the superhydrophobic/superoleophilic PP/EPDM TPV film was obtained after stripping the Al foil template by hand.
For simplicity, the untreated PP/EPDM TPV film was named as P/E film and the PP/EPDM TPV film modified with the etched Al foil template was named P/E-Al film. And the preparation process of superhydrophobic/superoleophilic PP/EPDM TPV film was exhibited in Figure 1. Experimental scheme of fabrication of superhydrophobic TPVs.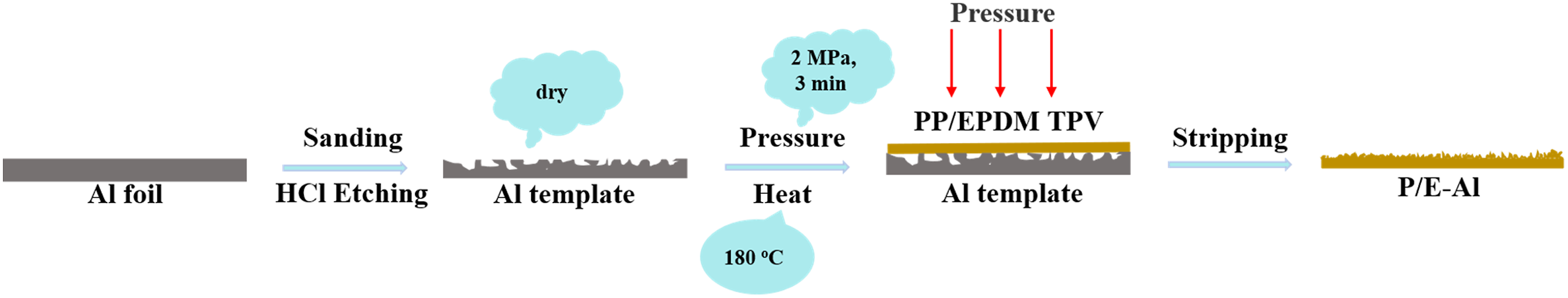
Instruments and characterization
Superhydrophobic/Superoleophilic property
The superhydrophobicity of the film was evaluated by water contact angle (WCA) measurement and water slide angle (WSA) measurement with a surface analyzer (LSA100, LAUDA Scientific, Lauda-Königshofen, Germany). The WCA values were measured by dropping 5 μL droplets of water on each sample under ambient condition and the reported average WCAs were determined by measuring at five different positions. The WSA value was obtained using 20 μL water droplets according to the automatic tilting stage (ATS) in a tilt speed with 60 degree/min and repeated three times. Moreover, the surface free energy (SFE) was obtained by the surface analyzer under "Direct Input Mode." In order to evaluate the solvent resistance of PP/EPDM TPV superhydrophobic films, hexane, toluene, acetone, ethylene glycol, petroleum ether, HCl, and NaOH solutions were used to test its the wetting performance.
Oli/water separation experiments
Light oil (n-hexane) was used to test the separation efficiency of oil-water mixture. Figure 7 showed a typical laboratory self-made equipment illustrating the oil-water separation process driven by pressure difference. For better identification, the 100 ml of light oil (n-hexane) was dyed with oil red O and added to 250 ml water before the test to prevent the solvent from evaporating too quickly. It can be seen that the phase separation of oil-water mixture was formed due to the different densities and surface energy. The external pressure required for oil-water separation was provided by a water-circulation multifunction vacuum pump.
When testing the oil-water separation efficiency, the rolled P/E-Al film was put into the through valve and set at the oil-water interface. Theoretically, the red oil in the oil-water mixture could pass through the valve composed by P/E-Al film and flow into the filter bottle under the appropriate external pressure; however, the water could not flow past the gap between the rolled P/E-Al film and remained in the beaker. In addition, the separation test was repeated for 25 cycles to research the repeatability of the P/E-Al film. The below equation could be utilized to calculate the film separation efficiency rate through the ratio of oil volume (Ro) after the separation process
V was the total volume of liquid collected after the separation and Vo was the oil volume in the collected liquid.
Results and discussion
The Superhydrophobic/Superoleophilic Property of the PP/EPDM TPV films
In order to better investigate the effect of surface structure on the surface wettability and stability, Al foils surfaces etched with different time were prepared as templates to prepare PP/EPDM TPV films with superhydrophobic/superoleophilic properties. The surface morphologies were presented in Figure 2. And the water static contact angle (WCAs) and oil contact angle (OCAs) of PP/EPDM TPV hydrophobic film molded by series etched Al foils were shown in Table 1. FE-SEM images of series Al foil template and P/E-Al. (a) Top surface of primary Al foil; (b) top surface of P/E; (c-c’) top surface of etched Al foil for 10 min; (d-d’) top surface of P/E-Al; (e) cross-section of etched Al foil for 10 min; (f) cross-section of P/E-Al. The Superhydrophobic/Superoleophilic property of PP/EPDM TPV films molded with series etched Al foil.
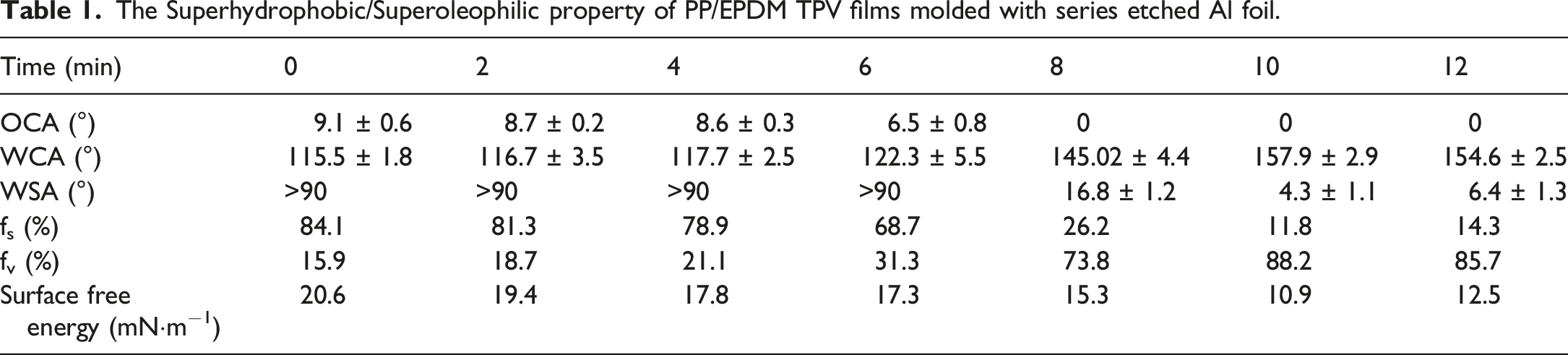
The WCA, OCA, and WSA of untreated PP/EPDM TPV films were found to be 108.8°± 1.6°, 9.2°± 0.4°, and >90°. As shown in Table 1, it was obvious that the OCAs data of all PP/EPDM TPV molded films were below 90°, indicating that the modified PP/EPDM TPV films were lipophilic to n-hexane; surprisingly, the lipophilic behavior of the prepared TPV film was changed into the superoleophilic behavior when the etching time was greater than 8 min. Corresponding to this variation, the WCAs behavior was also changed from hydrophobic to superhydrophobic, indicating the excellent superhydrophobic and superoleophilic behavior. Furthermore, it was worth noticing that PP/EPDM TPV surface molded with Al foil etched for 10 min has the relatively lowest surface energy.
A stable interface is very important for the successful design of superhydrophobic material. In this section, the Cassie–Baxter equation was used in the data fitting to further characterize the superwettability of the film. The equation was as follows
θr, θs, and θv were the WCA of the molded PP/EPDM TPV films, untreated PP/EPDM TPV surface and air, respectively. Although fv was represented the percentage of air portion in the water contact area and fs was represented the percentage of the TPV portion, typically, fs + fv=1.
It was generally believed that a solid surface with lower SFE showed better water repellency. Moreover, according to Cassie’s model, an increase in the fv of a superhydrophobic solid surface can achieve a higher WCA. The SFE and the calculated fv results of series molded PP/EPDM TPV films were also shown in Table 1. The data showed that the PP/EPDM TPV film surface molded with Al foil etched for 10 min possessed both the highest fv and the lowest SFE, which was important for its applications in oil-water separation.
The top surface morphologies of primary Al foil and P/E film were shown in Figure 2(a) and (b), demonstrating that the primary Al surface was smooth while the top surface of P/E was relatively flat. Parts c-c’ and d-d’ displayed the top-view FE-SEM images of the Al template and P/E-Al. The surface of the Al template was much rougher than the original Al surface. After 10 min of etching, a large amount of microsteps with the size of several microns were formed in the surface. As compared to those untreated Al, these dislocation defects that existed on the Al surface were prone to be corroded and dissolved by acid etchants first owing to the higher surface energy. These preferential corrosion effects finally resulted in the formation of microstep structures with inconsistent height, which were also well represented in the cross-section of Figure 2(e). It can also be found that the surface of P/E-Al was not directly copied the surface microstructure of the template during the molding process. In contrast, a large number of fiber-like microstructures were protruded out from the surface. As indicated in Figure 2(f), the rough structure can be clearly seen from the cross-section of the P/E-Al, which further verified the plastic deformation of the TPV surface. These structural characteristics provided the strong evidence for the following experimental verification.
The stability of the P/E-Al surface under various environmental conditions
It is well-known that the superior wettability of superhydrophobic surfaces often leads to irreversible damage when they are exposed to the complexity and harshness external conditions. Maintaining stability and durability in all kinds of harsh environments is an urgent and unsolved problem for the superhydrophobic surface used in the outdoor applications. Evaluating and examining the stability and durability of the P/E-Al surface is meaningful. The environmental stability, heat resistance, and mechanical resistance of the prepared P/E-Al surface were investigated and the results were shown in Figure 3. The chemical stability of the P/E-Al surface under various environmental conditions. The samples were (a) immersed into HCl solution (pH = 1) and NaOH solution (pH = 14); (b) immersed into toluene, acetone, n-hexane, ethylene glycol, and petroleum ether for different time; (c) placed at room condition for different time, and(d) heat treatment at various temperatures.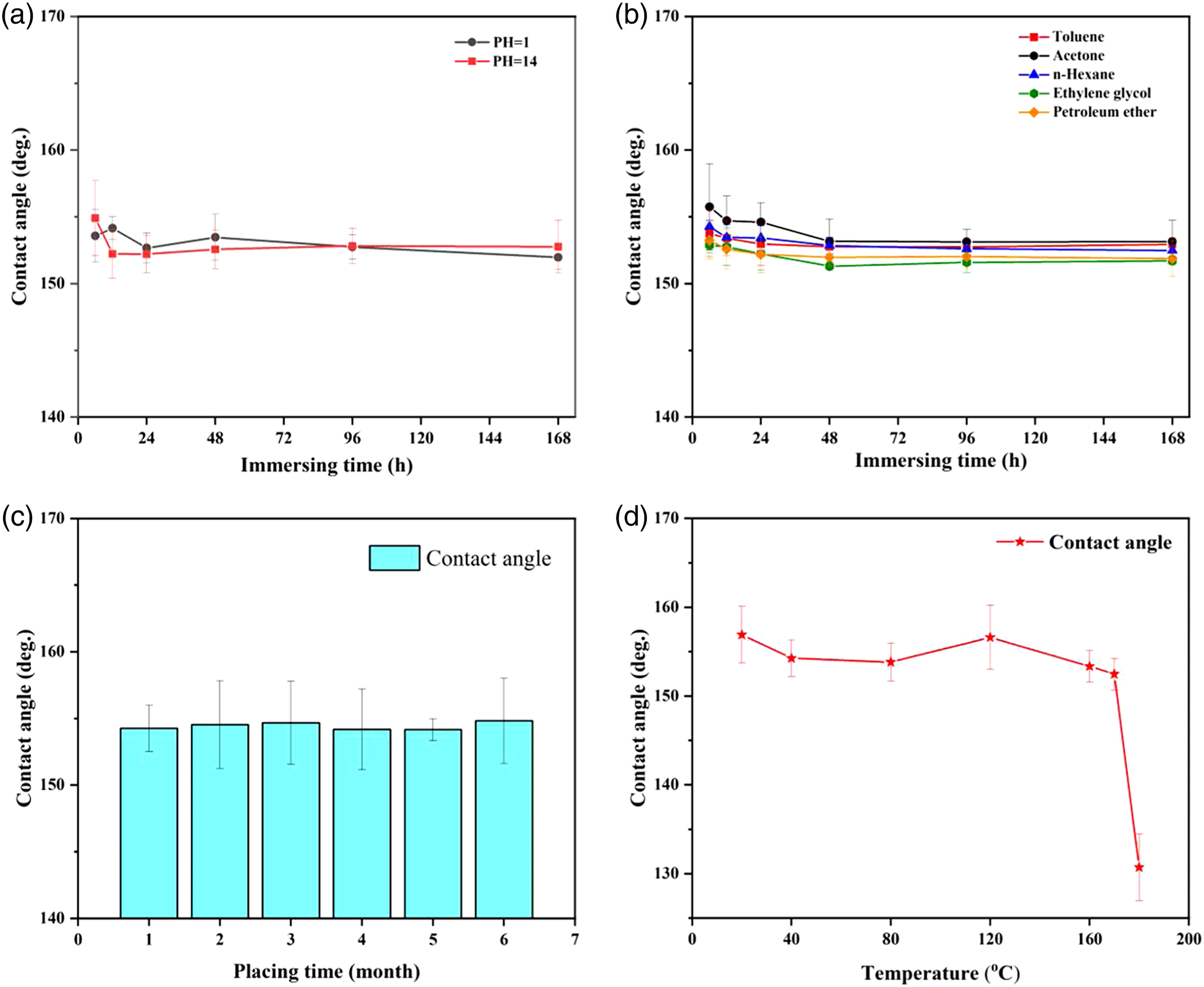
From Figure 3, the environmental stability of the P/E-Al film was assessed under different severe conditions in order to evaluate the resistance of the TPV films to the various solutions/solvents including aqueous solutions with different pH (e.g., HCl solution (pH = 1) and NaOH solution (pH = 14)) and organic solvents (e.g., toluene, acetone, n-hexane, ethylene glycol, and petroleum ether). After immersing the P/E-Al film in the above solution/solvent for a period of time the WCAs were measured and presented in Figure 3(a) and Figure 3(b). As can be seen, the WCAs were always unchanged and remained beyond 150o; even after being immersed into solutions or solvents for as long as 7 days (168 h), the surface of the prepared TPV film can still exhibit the strong water repellency, which revealed that the immersing time has only slight influence on the WCA values and the P/E-Al film was highly stable for chemical reagents. Figure 3(c) showed the WCAs as a function of storage time under indoor conditions. The results showed that the film retained superhydrophobic behavior even after being positioned at room condition for 6 months, indicating the long-term stability to atmospheric environment. Meanwhile, the thermal stability was evaluated by heat treatment in the oven at various temperatures (20–180°C) for 2 h, and the WCA values after heat treatment measurement were shown in Figure 3(d). It can be seen that the WCAs had no obvious changes when the temperature was 170°C below; however, when the heat treatment temperature was increased to 180°C, the WCAs was suddenly decreased from 153.7° to 134.1°, indicating that the film has excellent high temperature resistance especially when the ambient temperature was 170°C below. The fiber-like structures on the surface would be shrunken and curled when the melting point of PP 172°C was exceeded, and the fine and rough microstructure of the film was destroyed gradually, leading to the weakening superhydrophobic behavior.
The mechanical abrasion resistance of the superhydrophobic surface is one of the most significant factors in the daily applications. In order to evaluate the durability of the P/E-Al, the resulted samples were subjected to multiple intimate peeling tests using sticky adhesive tape. First, the adhesive tape was tightly adhered to a rough P/E-Al surface and then the tape was pressed hard by hand to ensure the tight adhesive between the tape and the substrate. Finally, the tape was quickly peeled off from the surface to complete the whole peeling process. The photographs of the peeling process were shown in Figure 4(a)-(c) and the relationship between the WCAs and peeling times was shown in Figure 4(d). From the Figure 4(d), the WCAs kept beyond 145◦ even after 10 peeling cycles, and the superhydrophobic behavior tended to decrease slowly. The above results indicated that the TPV film surfaces were highly stable even after contacting with the adhesive tape tightly, which was crucial to the industrialization. The peeling tests applied on the P/E-Al surface by using adhesive tape. (a–c) The photographs of the sample before and after the peeling test. (d) The relationship between the peeling times and WCAs.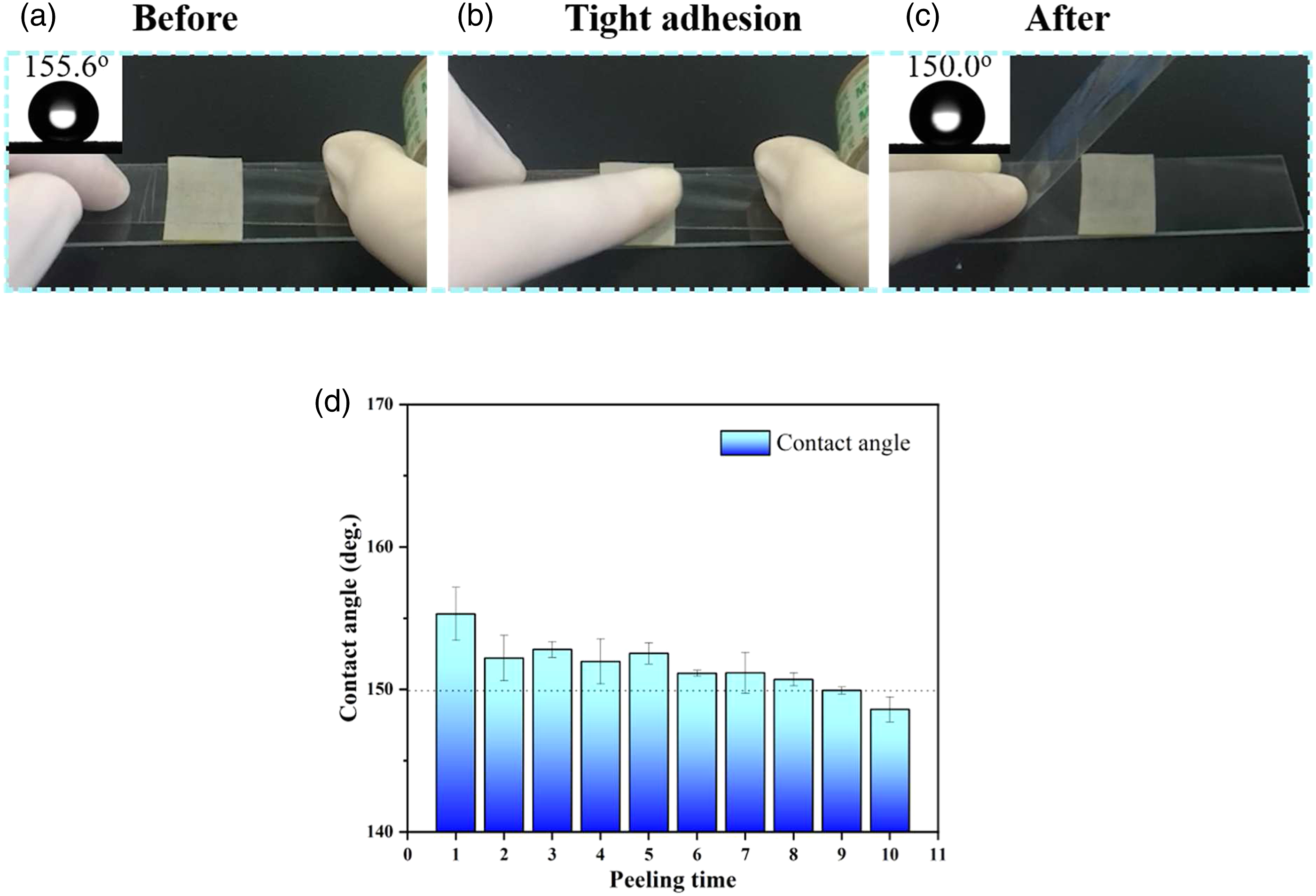
Wetting and self-cleaning properties of the P/E-Al film
The superhydrophobic surface always remains dust-free due to the low WCA hysteresis, which is called the self-cleaning property of the material. Water droplets rolled off the surface of the material quickly, and can remove the contaminants that accumulated on the surface according to this unique behavior. To confirm the self-cleaning ability of the prepared P/E-Al obviously, various liquids (water, coffee, pure milk, and green tea) and dusty were used as contaminants. As shown in the small image embedded in the upper right corner of Figure 5(a)-(d), the above droplets can maintain a spherical shape stably on the surface of the P/E-Al material without wetting, indicating the excellent repelling properties for these liquids. Interestingly, all the above droplets could quickly roll off the surface of the P/E-Al without leaving obvious stains, showing the excellent low adhesion and the remarkable self-cleaning behavior. Simultaneously, the dust on the surface was easily carried away by the moving water droplet, resulting in a completely clean surface, which can be observed in Figure 5(e)-(g), and the excellent superhydrophobic self-cleaning performance was fully verified. Superhydrophobic self-cleaning property of the P/E-Al against various liquids and dusty. Pollution: (a) water (dyed blue), (b) coffee, (c) pure milk, (d) green tea, (e–g) dusty.
Oil-water separation performance
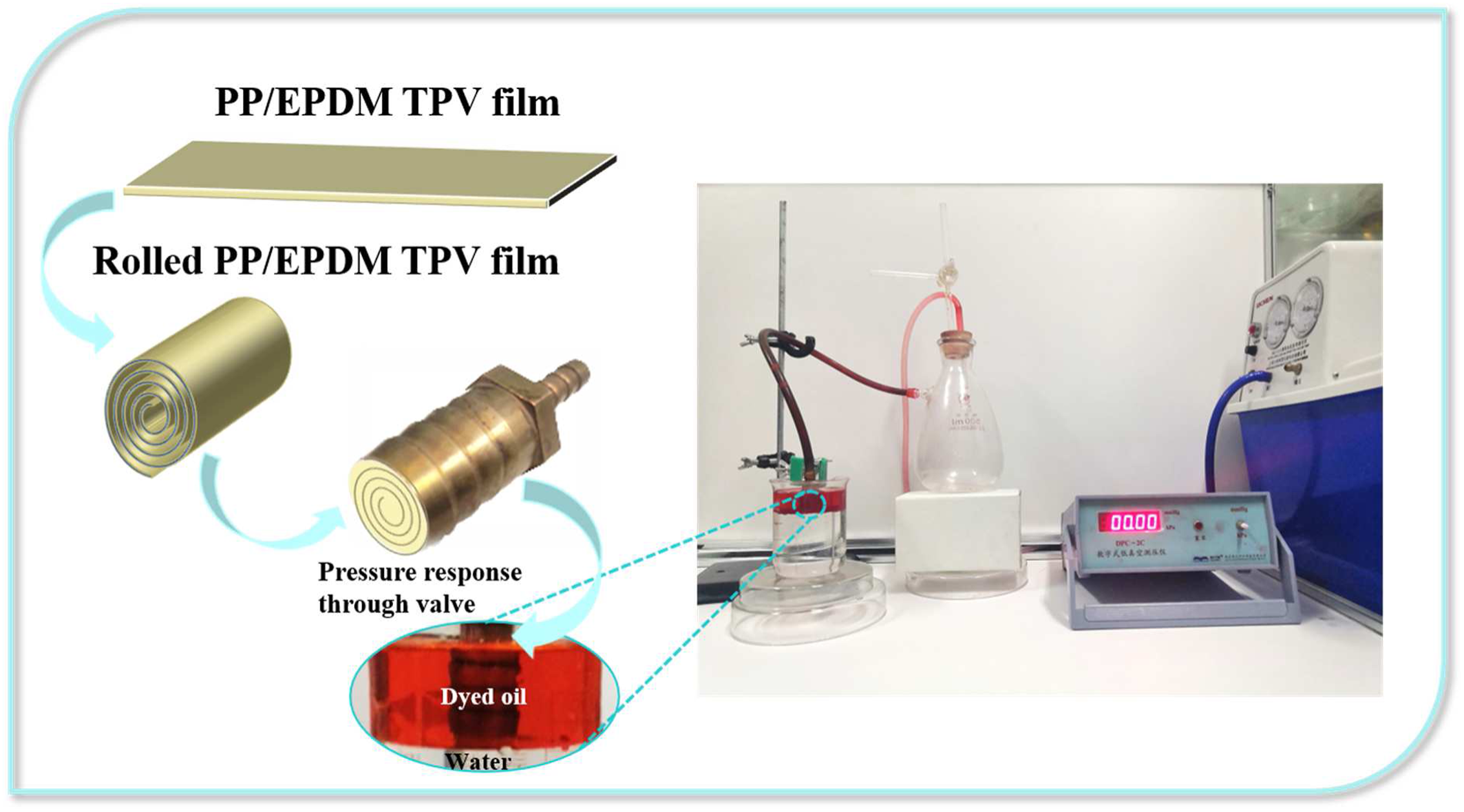
The superhydrophobic surface has incomparable advantages in oil-water separation, and the P/E-Al would be an ideal candidate for oil-water separation. Herein, a series of proof-of-concept studies were carried out to evaluate the separation capability of the film to oil-water mixture. The absorption abilities of P/E-Al were shown in Figure 6(a)-(c), the as-prepared sample was soaked with water and absorbed a small amount of oil (n-hexane was dyed red). When the film was used in contact with the red dyed n-hexane, it could absorb n-hexane instantaneously without being wet by the surrounding water. The above results indicated that the P/E-Al has potential applications in oil-water separation material. The separation device was shown in Figure 7, in our research, the pressure response through valve was used as a novel type of oil-water separation method with higher separation efficiency and a satisfactory recycling effect of industrial oily wastewater which was different from the traditional process by the traditional gravity-driven. In detail, the thickness and width of the TPV film was 250 μm and 22 mm while the diameter of the through valve was 13 mm. The 459 mm length of TPV film was used to rolled and plug into the valve carefully. The external pressure value of the liquid that can accurately flow into the flask was noted as Pm (kPa), the Pm that water could through was noted as Pm/w while the Pm of oil was defined as Pm/o. The ΔP (kPa) was defined as the difference between Pm/o and Pm/w. Photographs of the removal processes of n-hexane (colored with OR) (a–c) from water using the P/E-Al. Schemes illustrate the process of continuous oil-water separation via a pressure response through valve. 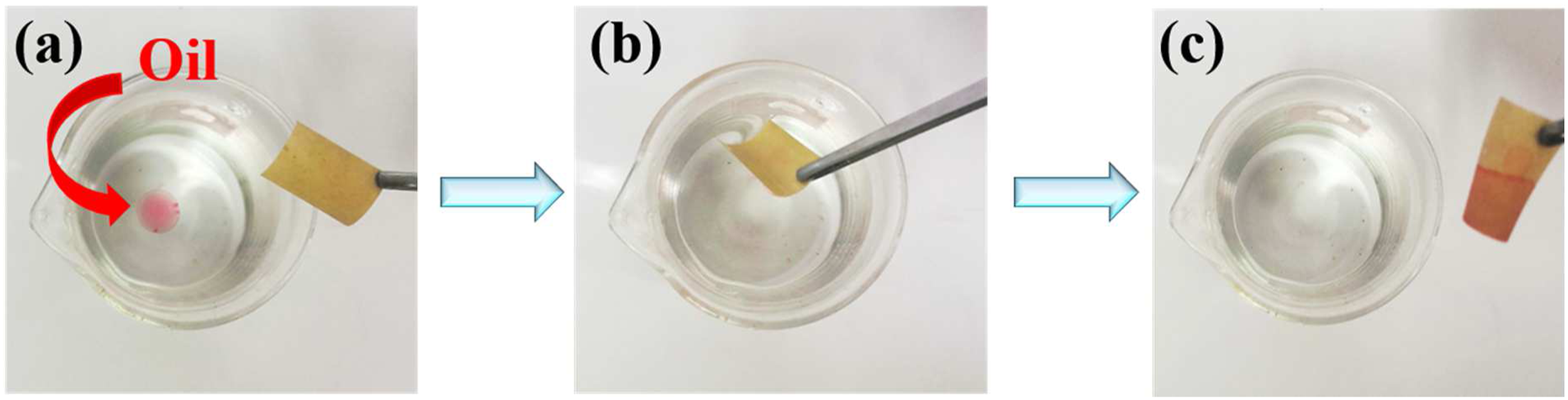
As shown in Figure 9(a), the through valve without any film was set as a blank testing. It was obvious that the Pm/o, Pm/w of P/E and P/E-Al were significantly different, and the Pm/w was greater than Pm/o under the same condition. The water critical pressures tested in our system were higher than 6.85 kPa when the P/E-Al was used in the through valve, demonstrating that water cannot pass through the valve under the pressure. More importantly, the highest ΔP of the rolled P/E-Al films can reach to 5.43 kPa, which was 323.2% higher than that of P/E under the same condition and 3878.6% higher than that of the blank through valve. In the above experiments, the through valve installed with P/E-Al film displayed different critical pressure response behavior according to the oil and the water, and this phenomenon was confirmed in supporting information (Supplemental material) and the oil-water interaction mechanism model of rolled P/E-Al in oil and water media was established in Figure 8(b)-(c). As can be seen from Figure 8(b), when the rolled hyperlipophilic (θ < 90°) film was immersed in the oil, the n-hexane diffuses rapidly on the surface of the P/E-Al film, resulting in capillary forces in the smaller gaps of rolled film. The oil could pass through the narrow gap between the rolled P/E-Al film surface with the low levels of Pm. For Figure 8(c), when a rolled superhydrophobic (θ > 150°) film was immersed in water, it was obvious that the negative capillary effect between the film gaps prevented water penetration, resulting in the higher Pm. Schematic diagram of the experimental process and the wetting mechanism of oil and water. (a) The process chart of oil-water separation efficiency experiment; (b) the wetting model of the rolled P/E-Al in oil; (c) the wetting model of the rolled P/E-Al in water.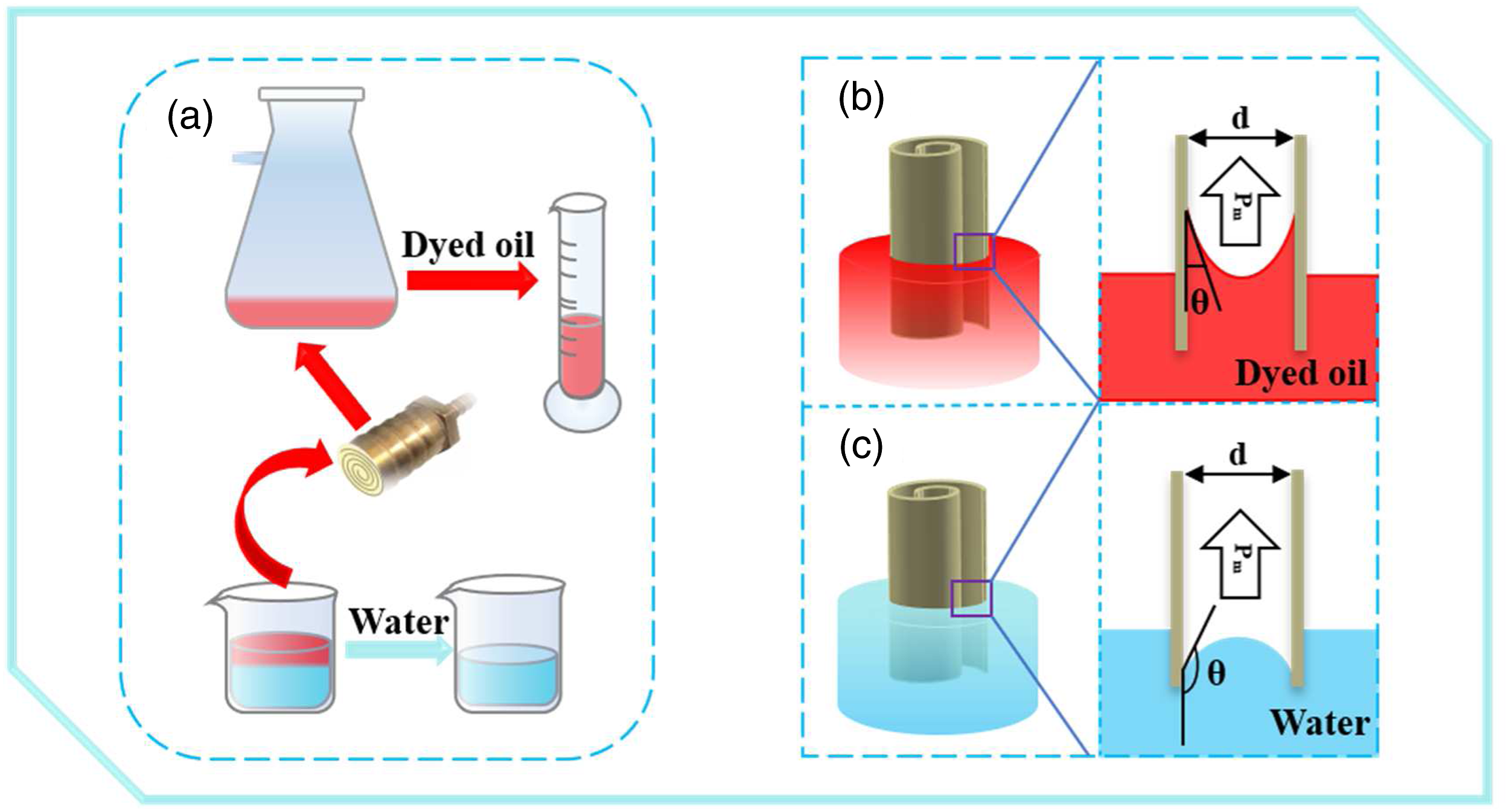
Compared with the P/E film that only has the good hydrophilicity and lipophilicity, the P/E-Al film exhibited the excellent oil-water separation efficiency when the external pressure was applied to the oil-water separation device. The experimental process of oil-water separation efficiency was shown in Figure 8(a) and supporting information (Supplemental material). During the separation process, the dyed n-hexane can easily pass through the valve and the water was relatively difficult to transfer because the Pm was set between the critical external pressure of oil and water. As shown in Figure 9(b), the separation efficiency of the P/E was decreased from100% to 77.24% when the Pm was 5 kPa; moreover, increasing the Pm, the oil-water separation efficiency was significantly decreased. Significantly, the oil permeating flux increased with increasing the water to oil ratio in the oil-water mixtures because of a gradual increment in external pressure. Nevertheless, from Figure 9(c), the separation efficiency of the P/E-Al with a slightly changed between 98.43% to 100%, and the separation efficiency could reach 100% when Pm was below 5 kPa, indicating the rolled P/E-Al could achieve excellent oil-water separation under proper oil-water separation pressure. More importantly, from Figure 9(d), after using for separation for 25 cycles, the separation flux and efficiency for n-hexane water mixture showed no obvious decreasing tendency when the Pm was set to 4 kPa; moreover, the flux and separation efficiency were still slightly changed between 39.61 ml/min to 49.82 ml/min and 98.27% to 100%. In general, the rolled P/E-Al displayed wonderful oil-water separation efficiency and recyclability. The experiment results of PE-Al film. (a) The Pm and ΔP of blank, P/E film and P/E-Al film; (b) the oil flux and separation efficiency of P/E under a serious of Pm; (c) the oil flux and separation efficiency of P/E-Al under a serious of Pm; (d) the oil flux and separation efficiency of P/E-Al versus the recycle number.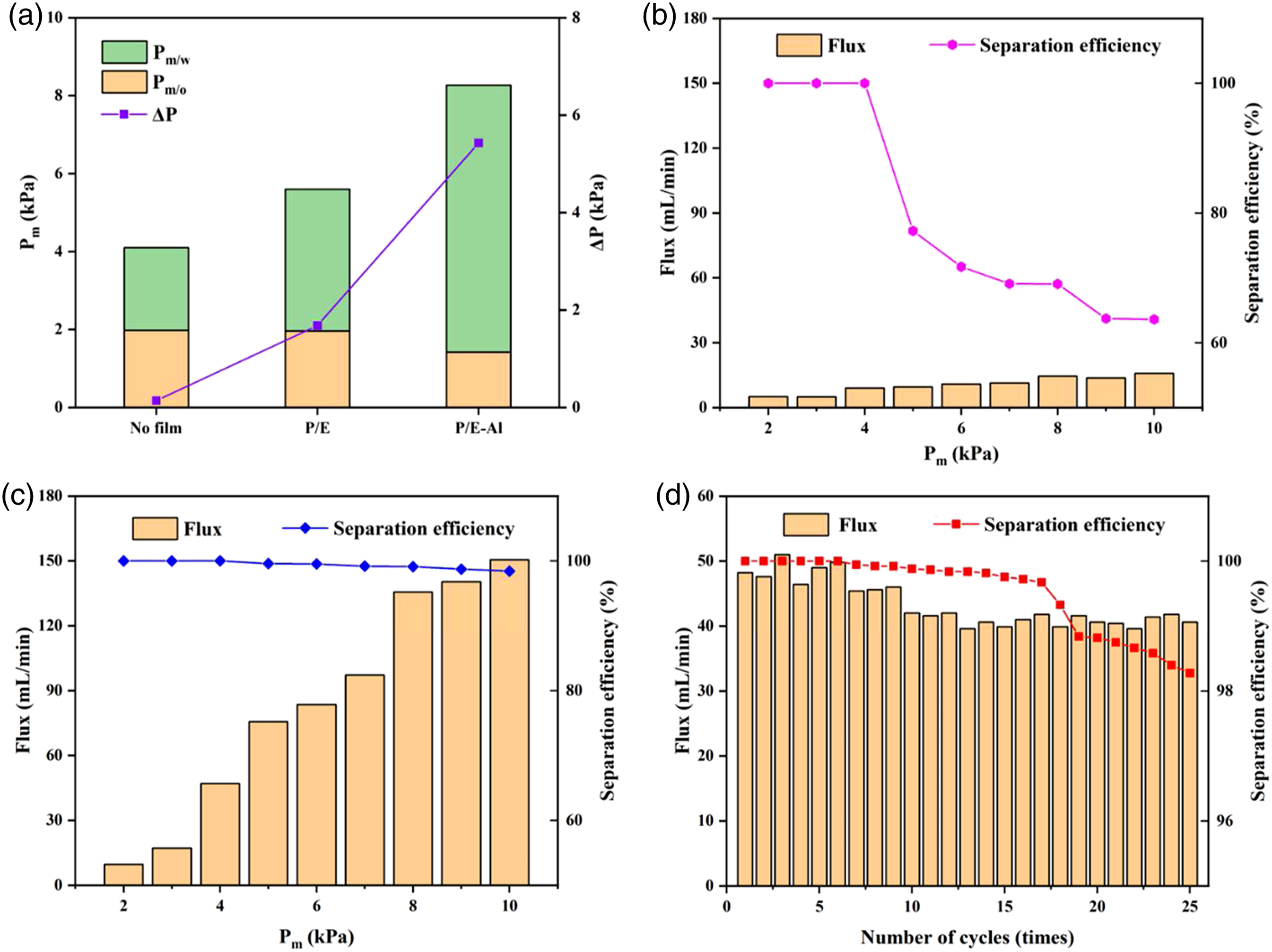
Conclusions
We have successfully demonstrated a facile, environmental-friendly, and cost-effective method for the preparation of superhydrophobic/superoleophilic films to effectively separate oil-water mixtures. It was worth noting that the aluminum foil etched by hydrochloric acid was used as the template, and the optimal etching time was determined to obtain the aluminum foil with a fine and rough surface structure. Moreover, the obtained superhydrophobic/superoleophilic PP/EPDM TPV films were proved to exhibit excellent resistance when exposed to some extreme environments (strong acidic or alkaline, corrosive organic solvents, and heating or mechanical abrasion conditions), also kept durable super-liquid-repellency. Through the pressure response of the valve, a feasible and simple device for continuous oil-water separation was assembled, which can effectively separate the oil-water mixture. Furthermore, the oil-water separation efficiency was remarkably improved when the PP/EPDM TPV films were applied to the device. We proposed that this novel device with an ease to use mass production PP/EPDM TPV film has a broad application prospect for effective oil-water separation.
Supplemental Material
sj-mp4-1-jtc-10.1177_08927057221086832 - Supplemental material for Superhydrophobic polypropylene/ethylene-propylene-diene terpolymer thermoplastic vulcanizates for continuous oil-water separation
Supplemental material, sj-mp4-1-jtc-10.1177_08927057221086832 for Superhydrophobic polypropylene/ethylene-propylene-diene terpolymer thermoplastic vulcanizates for continuous oil-water separation by Xiu Shan, Linji Yang, Ping Huang and Zhaobo Wang in Journal of Thermoplastic Composite Materials
Supplemental Material
sj-mp4-2-jtc-10.1177_08927057221086832 - Supplemental material for Superhydrophobic polypropylene/ethylene-propylene-diene terpolymer thermoplastic vulcanizates for continuous oil-water separation
Supplemental material, sj-mp4-2-jtc-10.1177_08927057221086832 for Superhydrophobic polypropylene/ethylene-propylene-diene terpolymer thermoplastic vulcanizates for continuous oil-water separation by Xiu Shan, Linji Yang, Ping Huang and Zhaobo Wang in Journal of Thermoplastic Composite Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation, China (ZR2021ME028).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
