Abstract
The objective of this research was to comprehensively compare the effects of two different types of nanoclay, namely layered double hydroxide (LDH) and halloysite nanotube (HNT) on the physical, mechanical, and dynamic mechanical properties of compression-molded composite panels fabricated from reed flour (RF) and polyvinyl chloride (PVC). To achieve the desired properties in the composites, the clay nanoparticles were modified with surfactant (mLDH and mHNT) before usage. The results showed that the composite specimens with mLDH exhibited higher tensile and flexural properties (strength and moduli) than with mHNT at low content. Compared with the maximum flexural strength and tensile modulus of 21.56 MPa and 2186.16 MPa for the specimens made with mHNT, the highest flexural strength and tensile modulus were found in the specimens incorporated with mLDH (23.05 MPa and 2227.44 MPa). Moreover, at high content, the composite specimens with mHNT presented greater hydrophobicity. The comparative analysis exhibited that that the water uptake of the composites including mHNT (5.03%) was approximately 15% lower than that of the mLDH (5.73%) based composite. The DMTA results indicated that the composite specimens with mLDH demonstrated better molecular restriction and larger storage modulus than with mHNT. Besides, the loss-tangent (tan δ) peak was shifted to a higher temperature for the samples including both mLDH and mHNT than without ones. The specimens made with mLDH had the highest glass transition temperature values (70.67°C) compared with 70.12°C for the specimens treated with mHNT. Morphological observations showed that the nanoparticles were predominantly dispersed uniformly within the polymer matrix. Overall, it is found that the addition of 3 phc mLDH clay was the most effective in the composite formulation; it has significantly enhanced the properties of the wood-plastic composites.
Keywords
Introduction
Wood-plastic composites (WPCs) is a new generation of composite materials and also the most promising sector in the field of both composite and plastic industries. 1 WPCs, which are made mainly from a combination of lignocellulosic materials (in different shapes of flour/fiber such as wood, kenaf, bagasse, hemp, reed, sisal, etc.) and thermoplastic polymers (including PP, PE, PVC, and etc.). They are used in different outdoor and indoor applications such as decking, fencing, railing, industrial flooring, docks, landscaping timbers, and automobile parts. Despite their extensive use in many fields, they also have some shortcomings, including relatively low modulus, low impact resistance, high moisture uptake, low thermal stability, and substantial creep.2–5 To alleviate these drawbacks, using nano-sized reinforcement as an ideal candidate has been widely concerned in recent years.6–9
Organically modified nanoclays are among the most studied nano-scale materials, because of their extraordinary mechanical and unique thermal properties. Moreover, they are relatively inexpensive, environment friendly, commercially available, and exhibited a layered morphology with a high aspect ratio and large specific surface area environment-friendly.10–12 The incorporation of nanoclay into the polymer matrices demonstrated synergistic improvements in composite properties even at very low amounts.13–15 The most desirable nanoclays are montmorillonite, bentonite, kaolinite, layered double hydroxide (LDH), and halloysite nanotube (HNT).10,11 Among them, LDH is a new class of anionic lamellar (two-dimensional nanomaterial with the general formula [M1−x2+Mx3+ (OH)2]x+[An−x/n.mH2O]x−, which are composed of positively charged brucite-type layers and intercalated water and anions in the interlayer space. They have attracted increasing attention due to their potential applications as catalysts, anionic exchangers, UV adsorbents, fire retardants, and polymer additives.16–23 HNT is an aluminosilicate with the chemical formula of Al2Si2O5(OH)4 and hollow tubular structure (one dimensional), which can be formed as a result of hydrothermal or weathering processes. It is chemically similar to the kaolinite group of clay and exhibits a hollow nanotubular structure with a large aspect ratio similar to that of the carbon nanotube. The diameter of HNT can be ranging from 100 to 300 nm with an approximate length of 1–3 µm. Due to nanosize, surface area, low cost, natural availability, and biocompatibility, HNT in polymer composites have been developed for numerous exciting applications.24–33
The positive effects of organ-modified montmorillonite (OMMT) as the most famous and widely used nanoclay on the characteristics of the thermoplastic composites filled with the lignocellulosic fillers were reported in previous research studies.34–44 But, the impacts of other types of nanoclay such as LDH and HNT on the technological properties of WPCs have not been comprehensively explored. Furthermore, there is little information available on the performance aspects of fiber-reinforced composites employing LDH or HNT.45–49 Therefore, the present paper focuses on characterizing and comparing the physical, mechanical, and dynamic mechanical properties of reed flour/PVC composites in the presence of LDH and HNT nanoparticles. Besides, the Zn-F layered double hydroxide modified with Sodium dodecyl sulfate [LDH (Zn-Fe)@SDS], and functionalized HNT nanoparticles with tetraethyl orthosilicate (HNT@TEOS) were synthesized and characterized by Fourier transform infrared (FTIR).
Experimental
Materials
PVC (Poliran PV S-6058) was supplied by Bandar Imam Petrochemical Company (Iran), with a bulk density of 550–610 g/L and a viscosity of 85–92 cm 3 /g. The reed used as a lignocellulosic filler was directly provided from Hawizeh Marshes, which is located in the Khuzestan province of southern Iran. After debarking it was chopped and by grinding changed to flour. The reed flour (RF) was dried at 80°C for 24 h and then was sieved. The particle size of RF was selected 40–60 mesh. Then, it was dried again to ensure that the moisture content is less than 3 wt%, and then stored in sealed plastic bags before compounding. Halloysite nanotube (HNT) was purchased from Sigma Aldrich (Taufkirchen, Germany). The molecular weight, relative density, diameter, pore size, and cation exchange capacity of HNT were 294.19 g/mol, 2.53 g/cm3, 30–70 nm, 1.26–1.34 mL/g, and 8 meq/g, respectively. Sodium dodecyl sulfate (SDS) and tetraethyl orthosilicate solution (TEOS) were obtained from Merck (Germany).
Methods
Synthesis of LDH (Zn-Fe)
Firstly, 2.975 g of Zn(NO3)2.6H2O (2 mol), and 2.02 g of FeCl3.6H2O (1 mol) were dissolved in 25 mL of pre-boiled deionized water using magnetic stirring. Then, this solution was added to 50 mL of a pre-boiled deionized water droplet while the pH of the solution was adjusted to 9.5–10 with adding of NaOH solution (1 mol L−1). After that, the provided precipitate was refluxed at 70°C for 70 h in an oil bath under stirring. Finally, the brown precipitate was centrifuged and washed several times with deionized water and dried in an oven at 50°C. This LDH was synthesized for comparing the structure after modification.
In situ synthesis of LDH (Zn-Fe)@SDS
Firstly, 17.31 g of SDS (2 mol) was dissolved in pre-boiled deionized water. Then, the pH of the solution was adjusted to 9.5–10 with adding of NaOH solution (1 mol L−1). In another beaker, 17.85 g of Zn(NO3)2.6H2O (2 mol) and 12.12 g of FeCl3.6H2O (1 mol) were dissolved in 100 mL of pre-boiled deionized water using magnetic stirring. Then, this solution was added to the above SDS solution droplet while the pH of the solution was adjusted to 9.5–10 with adding of NaOH solution (1 mol L−1). After that, the provided brown precipitate was refluxed at 70°C for 72 h in an oil bath under stirring. Finally, the brown precipitate was centrifuged and washed several times with deionized water and dried in an oven at 50°C. The synthesis procedure is presented in Figure 1.
Synthesis of tetra ethyl otwrthosilicate-modifid HNT
The tetraethyl orthosilicate solution (TEOS) was selected to modify the surface of as received HNT. Firstly, 0.5 ml of acetic acid was dissolved into 200 ml of water to obtain 3 pH. Then, tetraethyl orthosilicate (3.324 ml) was mixed with 90 ml of methanol and added to the prepared aqueous solution. The obtained solution was stirred at 60°C using a magnetic bar and then 19.564 g of HNT was added to the solution and stirred for 48 h. The solution was centrifuged and the obtained precipitate (HNT@TEOS) was dried in a vacuum oven at 40°C for 24 h. The synthesis procedure is presented in Figure 1.
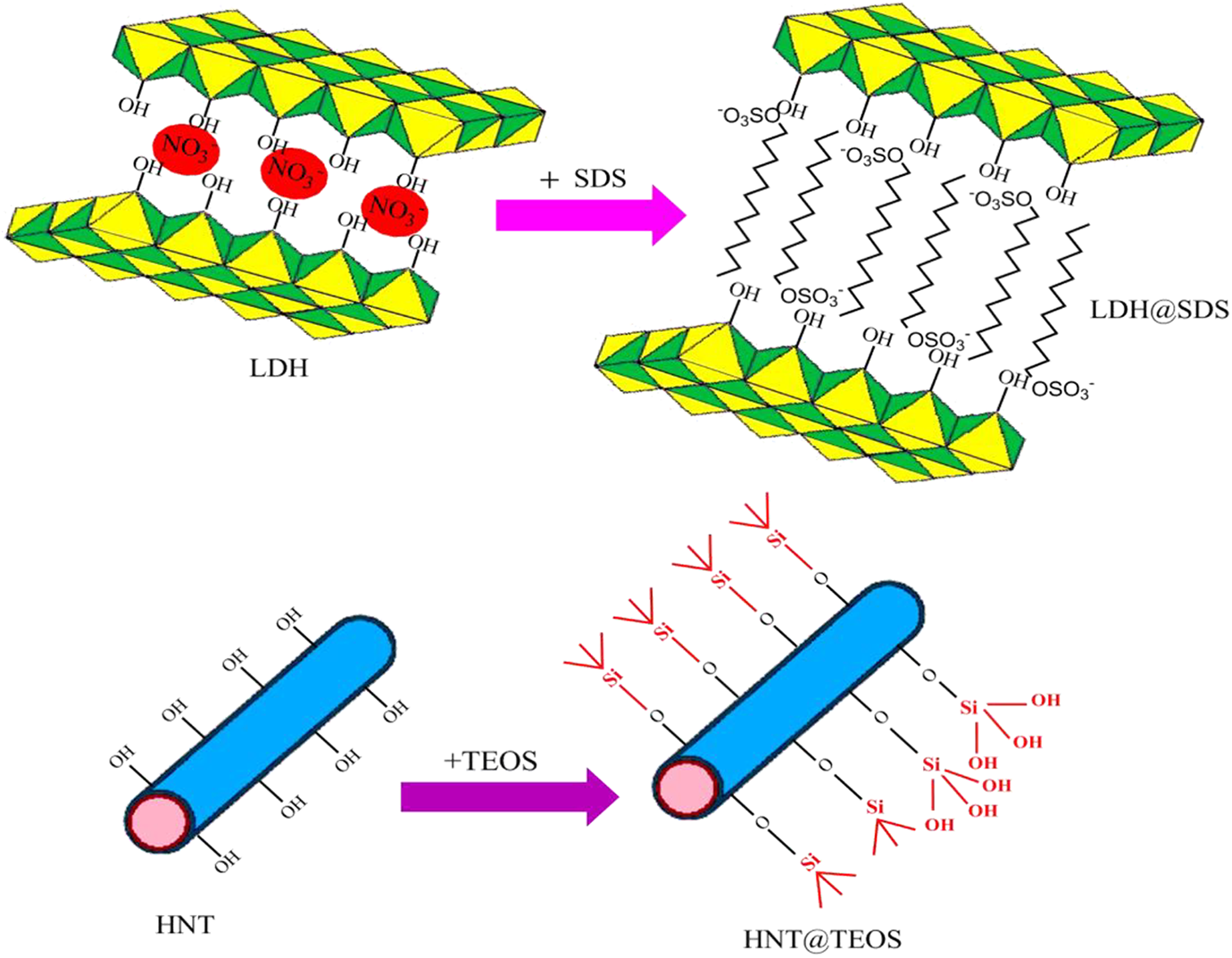
The synthesis procedure of modified LDH and HNT nanoparticles.
Manufacturing method of WPC panel
RF was dried in an oven at 103 ± 2°C for 24 h to achieve a moisture level below 3%. The RF, PVC, and nanoparticles were mixed in a co-rotating twin-screw extruder (Dr.Collin GmH, D-8017 Ebersberg, Germany). The melt temperature at the die was 175°C and the rotation speed was 60 rpm. The extruded strands were cut into granules (5–10 mm) before hot-pressing. The resulting granules were molded into composite sheets in a compression hot press (Toyo Seike Mini-test Press, model WCH, Japan), which was used to prepare the composites at 180°C at 5 MPa for 6 min, followed by 8 min cold pressing at room temperature. The dimension of the mold is 100 × 100 × 6 mm. Finally, the specimens were conditioned at a temperature of 23°C and relative humidity of 50% according to ASTM D 618. The average density of the WPC panels was 0.93 g/cm3. Various blending formulations and their mass ratios are listed in Table 1.
Formulations of the fabricated WPC specimens.

* Parts per hundred of compounds (RF+PVC).
Measurements
Fourier transform infrared spectroscopy
Fourier transform infrared-Attenuated total reflection spectroscopy (FTIR-ATR) assessment was recorded using a Bruker Vertex 70v spectrometer (Germany) throughout the range from 400–4000 cm−1 using a 4 cm−1 resolution.
X-ray diffraction
The crystalline structure of both LDH and HNT particles, and manufactured composite samples containing nanoclay were studied with help of powder X-ray diffraction (XRD). XRD analyses were performed in a Siemens D5000 diffractometer (Munich, Germany) using Cu-Ka radiation (λ= 0.1540 nm) at ambient temperature. The incidence angle was varied from 5° to 80° at a scanning rate of 1°/min with intervals 0.02°/s. The generator was operated at 40 kV and 40 mA. The interlayer spacing (d00) of clay was calculated under the Bragg equation: d00 = n λ/2sin θ.
Mechanical testing
The tensile and flexural properties of the composites were evaluated according to ASTM D638 and ASTM D790, respectively, on an Instron universal testing machine (model 4486, England) at a test speed of 5 mm/min. The measurements were performed at ambient conditions and relative humidity of approximately 50%. The sample size of 100 × 13 × 5 mm3 and the specimens with the dimension of 167 × 10 × 3 mm3 were used for flexural and tensile tests, respectively. At least six specimens were tested for each set of experiments and the mean values were reported.
Dynamic mechanical testing
Dynamic mechanical analysis of the test specimens, with dimensions of 30 × 10 × 1 mm3, have been performed on a Mettler Toledo machine (DMA Instruments, Model Stare System, Swiss) to characterize the storage modulus (E′), loss modulus (E″) and loss-tangent (tan δ). The test specimens were subjected to a sinusoidal displacement at constant strain amplitude with a frequency of 1 Hz in the temperature range of 20–120°C and at a heating rate of 5°C/min.
Physical testing
The water absorption and thickness swelling test was carried out following ASTM D570. The samples dimensions of 20 × 20 × 20 mm3 were immersed in distilled water at 23 ± 1°C for 60 days. Before tests, the weight and thickness of each specimen were measured. Samples were removed at certain periods, wiped with tissue paper to remove the excess water on the surface, and immediately their wet mass and thickness were measured. At least 10 specimens were tested for each set of experiments and the mean values were reported.
Scanning electron microscopy
The flexural specimens were frozen in liquid nitrogen for 5 min and broken manually. The fracture surfaces were subsequently dried, sputter-coated with gold, and then observed with a field emission scanning electron microscope (Tescan Mira 3 XMU, Czech Republic) at an accelerated voltage of 15 kV.
Result and discussion
Characterization of modified LDH and HNT nanoparticles
The synthesized LDH@SDS and HNT@TEOS were subjected to Fourier transform infrared spectroscopy (FTIR) analysis. Figure 2 presents the FT-IR spectra of LDH, SDS, and LDH@SDS particles. The broadband centered at 3450 cm−1 is attributed to the stretching vibration of the OH groups in the LDH structure and the adsorbed water. The vibration at 1645 cm−1 in LDH and LDH@SDS was ascribed to the (H–O–H) bending vibration of the adsorbed water. Some new bands were detected in the FTIR spectra of LDH@SDS. The double peaks at 2918 and 2845 cm−1 were due to the asymmetric and symmetric CH2 stretching modes, respectively which are confirmed the presence of dodecyl sulfate in LDH@SDS composites. The new band at 1405 cm−1 was also related to the C–H bending vibration band. The adsorption bands at 1060, 1195, and 1216 cm−1 are related to the S–O asymmetric stretching of SDS. The absorption bands due to the asymmetric (1224 cm−1) and symmetric (1060 cm−1) sulfate S=O stretching vibrations were also observed. So, the intercalation of the SDS anions into the interlayer of LDH is confirmed by these new peaks.
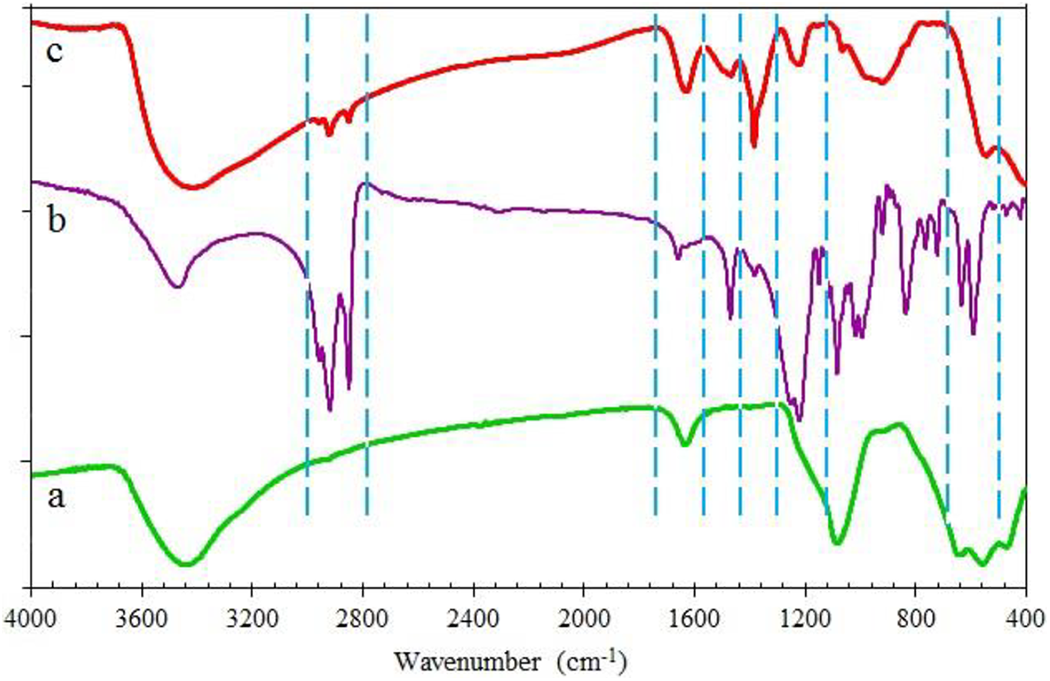
FTIR spectra of (a) LDH, (b) SDS, and (c) LDH@SDS.
Figure 3 illustrates the FTIR spectra of HNT, TEOS, and HNT@TEOS clays. The new absorption peaks at 2982, 2941, and 2905 cm−1 in HNT@TEOS are attributed to C–H stretching vibrations (asymmetric and symmetric) of CH2 in TEOS which appeared in the HNT@TEOS structure. So, it confirms TEOS was successfully coated on the HNTs. Interlayer water was indicated by the bending vibration at 1633 cm−1. The absorption peak at 1305 cm−1 is related to the stretching vibration of the C–N band in HNT. The band observed at 915 cm−1 denoted the bending vibration of the inner hydroxyl group. Stretching vibration of Si–O observes at around 1095 and 1032 cm−1. The absorption peak at 795 cm−1 is strong in HNT@TEOS due to the 793 cm−1 strong peak in TEOS. The bands centered at 1048 cm−1 and 798 cm−1 correspond to SiO4 tetrahedral symmetric and asymmetric stretch vibrations, respectively. Two stretching absorptions of Al2OH are shown at 3695 and 3621 cm−1. Also, the bending vibration of Al–OH is shown at 905 cm−1.
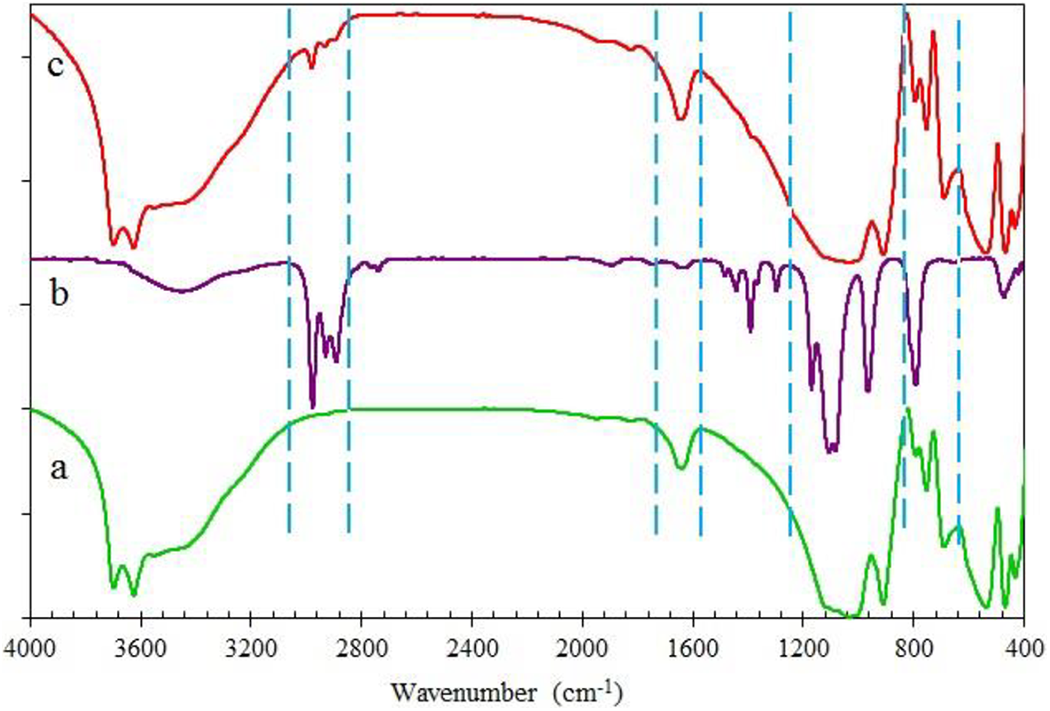
FTIR spectra of (a) HNT, (b) TEOS, and (c) HNT@TEOS.
XRD analysis
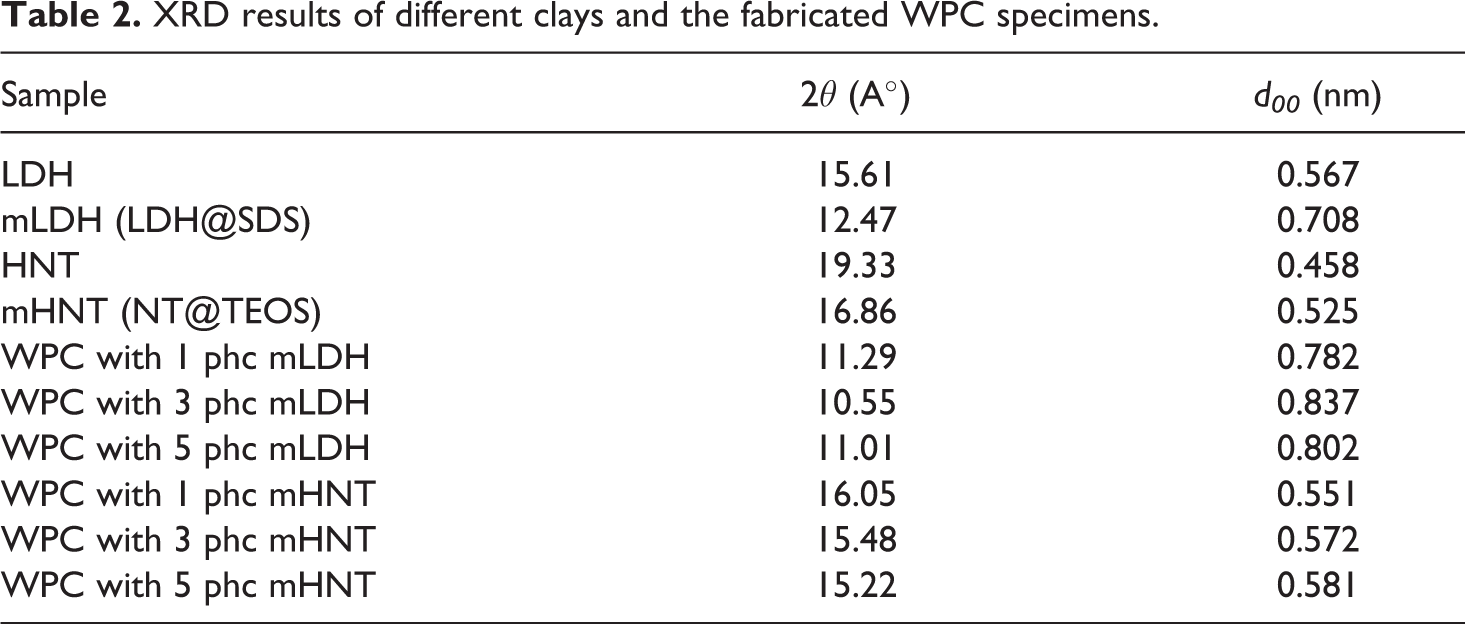
The X-ray scattering intensities for both LDH and HNT particles and the WPC specimens filled with various amounts of nanoclay are listed in Table 2. Consistent with Bragg’s equation, the interlayer spacing of LDH and HNT is equal to d003 = 0.567 nm (2θ = 15.61°) and d003 = 0.458 nm (2θ = 19.33°), respectively. After the modification of nanoparticles, the interlayer spacing has increased to d003 = 0.708 nm (2θ = 12.47°) for mLDH (LDH@SDS), and to d003 = 0.525 nm (2θ = 16.86°) for mHNT (HNT@TEOS). However, mLDH had a higher d-spacing than that of mHNT. The increase in the d-spacing of the clays indicates that the organic surfactants had diffused between the clay sheets and modification was successfully done. Besides, the loaded of both LDH and HNT clay into the composite specimens showed a peak shift to lower diffraction angle (2θ), which implied an increase in d-spacing of silicate layers and it is easier for the polymer chains to intercalate through the clay layers. The increase of the interlayer distance might result from the stronger shear during processing when the filler was introduced.10–14 The reduced peak intensity is attributed to the low concentration of clay in the samples. Similar observations have been recorded by previous studies.34–50
XRD results of different clays and the fabricated WPC specimens.
Mechanical analysis
The tensile strength and modulus of the fabricated WPC specimens as functions of the clay type and content are presented in Figure 4. As can be seen, tensile properties were improved with the incorporation of both mLDH and mHNT clays. The tensile strength and modulus values were observed to be 22.94 MPa and 1895.74 MPa for samples made without nanoclay. More specifically, the addition of 3 phc mLDH into the formulations increased the tensile strength and modulus 32.09 MPa and 2227.44 MPa respectively, which are 39.88% and 17.5% higher than the control composite. This result is consistent with the general observation that the introduction of nano-sized particles into a polymer matrix increases its tensile properties.34–44 The enhancement is attributed to the high aspect ratio, as well as the surface area of stiff silicate layers in the polymer matrix that result in a higher extent of interaction with the polymer chains, and good interfacial adhesion between the nanoclay particles and the polymer matrix, so that the mobility of polymer chains is restricted under loading.34–44 Nevertheless, with a further increase in the mLDH clay loadings (from 3 to 5 phc), a decrease in the strength and modulus of the samples was observed. The possible reason for this kind of behavior may be attributed to the agglomeration of the clay or the formation of interconnected masses of the clay at the fracture points, which led to induced local stress concentrations, hence initiating micro-cracks in the WPCs.46,48 The dispersion of clay into the produced WPC specimens supported through FE-SEM observations (Figure 6).
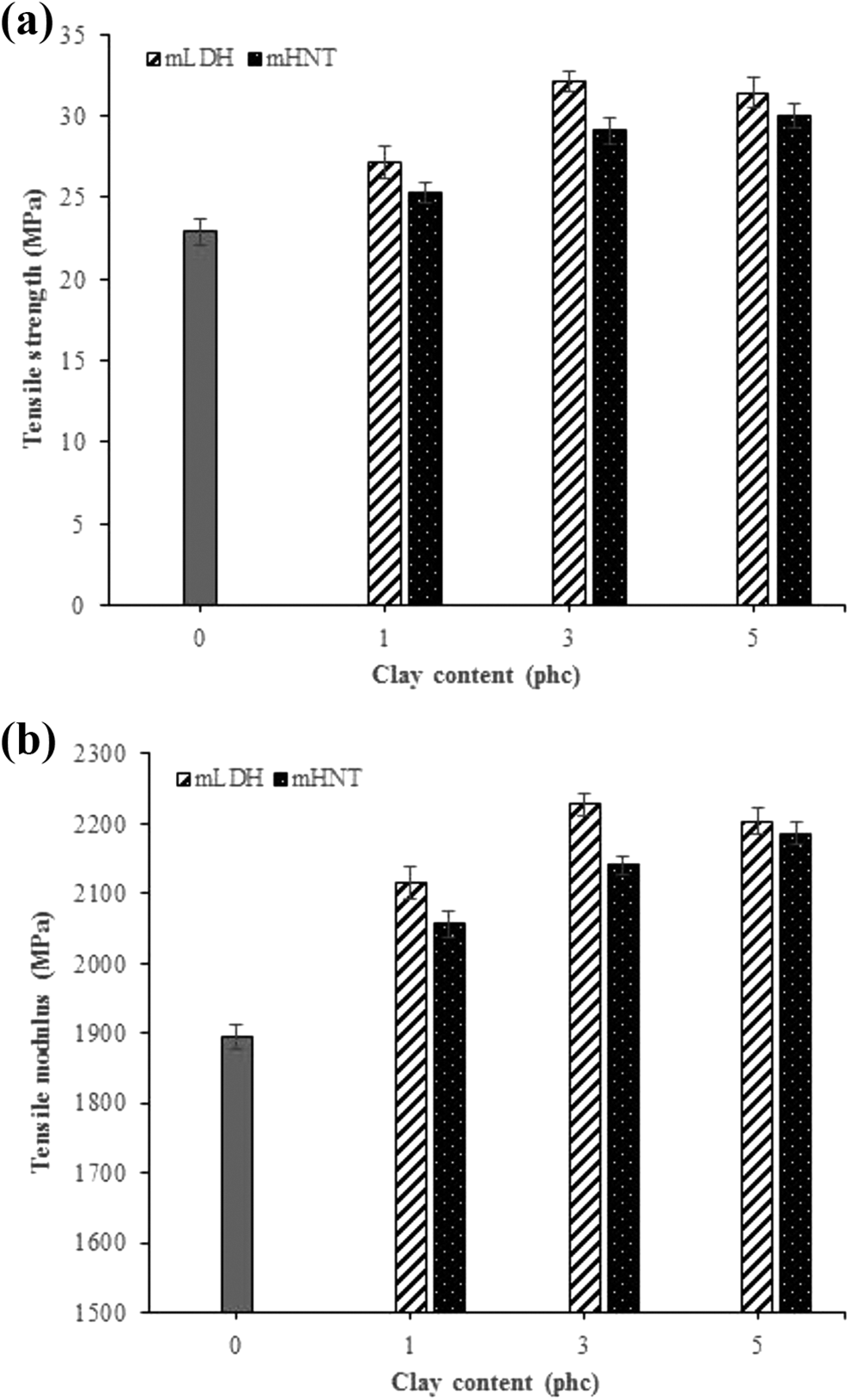
Effects of mLDH and mHNT contents on (a) tensile strength and (b) tensile modulus of the fabricated WPC specimens.
It was also observed that the tensile properties have considerably increased when high mHNT content was incorporated in the composite formulation. This trend may be due to the increased d-spacing of the mHNT clay at high contents, causing a strong interaction in the interfacial region and enhanced the mechanical performance. 50 However, it is clearly illustrated that the samples filled with mLDH have higher tensile properties as compared with the samples containing mHNT. This could be explained by the fact that mLDH had larger interlayer spacing than mHNT; therefore, mLDH showed better dispersion in the polymer matrix. In this condition, stress could be transferred efficiently in the composite matrix and turn led to higher tensile properties.48,50
Figure 5 demonstrates the flexural strength and modulus of composites made with both kinds of clays at different loading levels. The trends in the flexural properties are the same as those of the tensile properties. Samples filled with 3 phc mLDH displayed the highest values among the other types of specimens. The flexural strength of the control samples was found to be 15.17 MPa, with a flexural modulus of 1285.66 MPa. The incorporation of 3 phc mLDH led to an increase in the flexural strength and modulus of about 51.94% and 15.57%, respectively. As evident from Figure 5, with an increase in mLDH content from 3 to 5 phc, the flexural properties are moderately decreased. This could be explained by the agglomeration of mLDH particles. The mechanical results indicate that the reinforcing efficiency of the clays as fillers in the hybrid composite is balanced by two opposite phenomena: dispersion of clays at low contents, which has a positive effect that could enhance the mechanical properties, and a negative effect attributed to the agglomeration of clays at high content, causing a decreased performance.14,36,46 On the other hand, the addition of mHNT up to 5 phc, increased the flexural strength and modulus 21.56 MPa and 1448.39 MPa respectively, which are 42.12% and 12.66% higher than the control composite. This result may be due to the better dispersion of particles and their adhesion in the interface area which occurred at a high content. However, the samples containing mLDH showed better flexural properties than mHNT, due to their larger interlayer spacing.
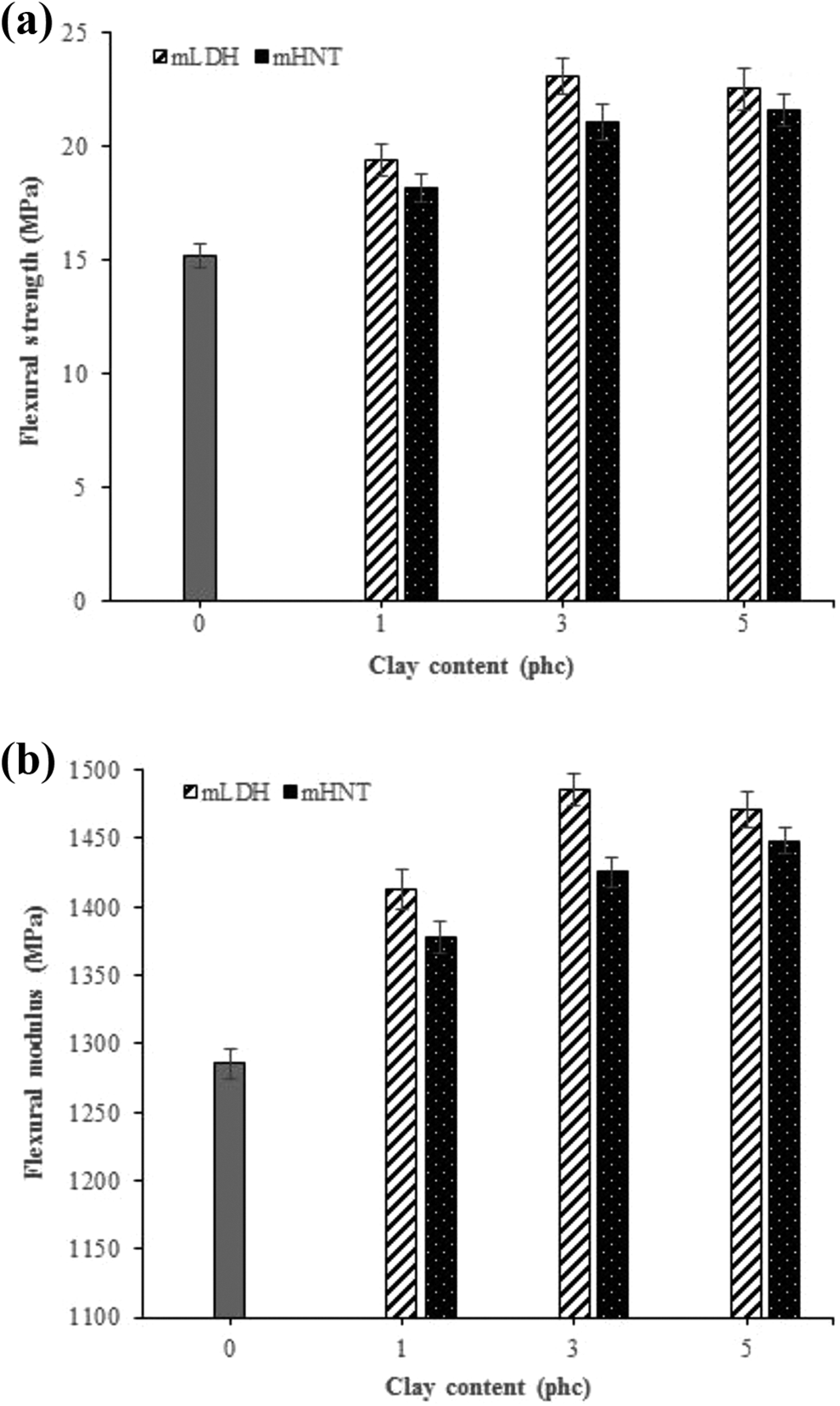
Effects of mLDH and mHNT contents on (a) flexural strength and (b) flexural modulus of the fabricated WPC specimens.
Morphological observation
Figure 6 shows the dispersion state of both mLDH and mHNT clays in the fabricated WPC specimens to validate the mechanical results. Figure 6(a) corresponds to WPC without nanoclay (control sample), showing some evidence of holes, fibers pull-out, and matrix cracking, implying inefficient adhesion between the RF and the polymer matrix. Hence, when stress is applied it causes the fibers to leave the matrix easily and makes micro-voids. The morphological surface of the specimens after treatment by nanoparticles can be observed in Figure 6(b) to (g). It is seen that both mLDH and mHNT particles were dispersed uniformly in the composites. This dispersion state verified that the interfacial adhesion between the components was distinctly improved and further contributed to the stress transfer from the RF to clay nanoparticles, and consequently enhanced the mechanical properties of composites. In the case of the sample containing LDH, the agglomeration that occurred at a higher fraction might be due to the improper dispersion and surface interaction between nanoparticles (Figure 6(d)).
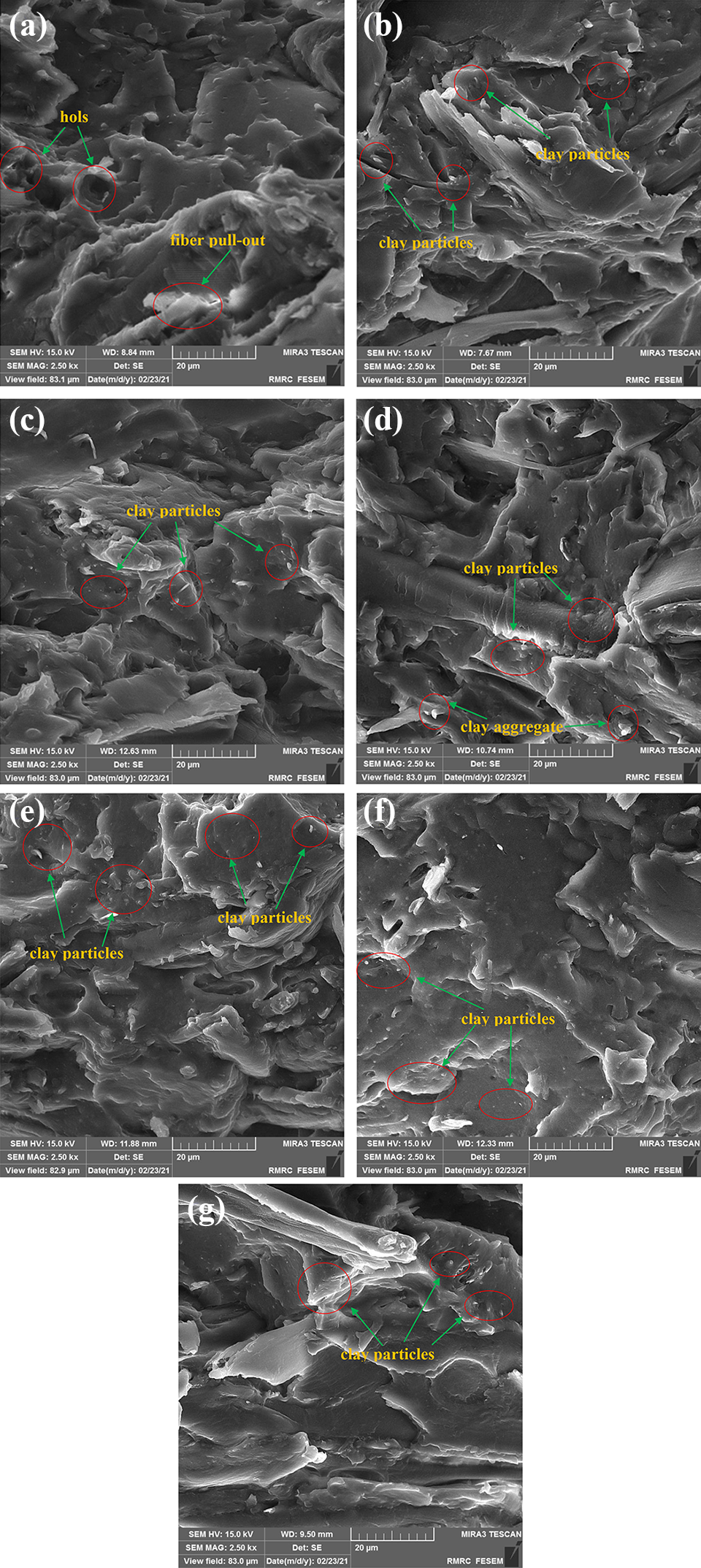
FE-SEM micrographs of the fabricated WPC specimens: (a) without nanoclay, (b) 1 phc mLDH, (c) 3 phc mLDH, (d) 5 phc mLDH, (e) 1 phc mHNT, (f) 3 phc mHNT, and (g) 5 phc mHNT.
Dynamic mechanical analysis
The storage modulus of the fabricated WPC specimens as functions of the clay type and content are illustrated in Figure 7. As shown, there are three apparent regions in all curves including a glassy high modulus region where the segmental mobility is restricted, a transition zone where a steep decrease is evident in storage modulus values within a narrow temperature range, and a rubbery region where a drastic deterioration of the modulus is leveled off against temperature. It can be observed that the storage modulus of the WPCs increased after the incorporation of nanoclay. Samples filled with 3 phc mLDH showed the highest values among the other types of specimens within 25–80°C. In general, before Tg, the specimen containing nanoclay stores more energy compared to the specimens without nanoclay. The increased storage modulus of the clay-containing samples is attributed to the strong chemical interaction between the polymer matrix and the filler which restricted the segmental mobility of the polymer chains in the vicinity of the nano-sized reinforcements. On further increasing the weight fraction of nanoclay, agglomeration place which decreases the effective stress transfer between the fiber and polymer matrix.8,51,52
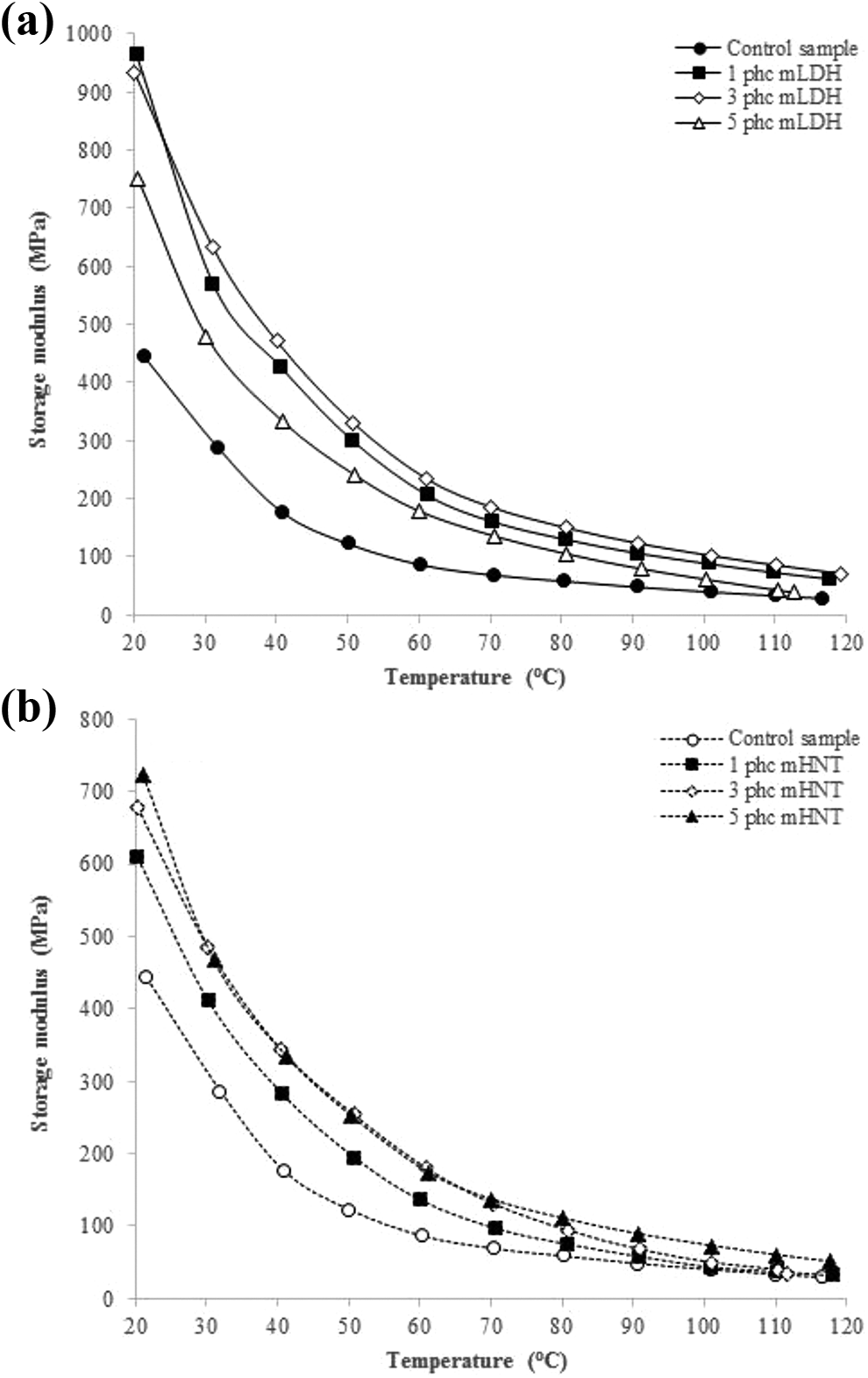
Effects of (a) mLDH and (b) mHNT contents on storage modulus of the fabricated WPC specimens.
As the temperature of samples reaches their glass transition, a sharp drop in the storage modulus is observed. It has been proven that the magnitude of the drop depends on the amount of filler in the samples.51,52 The level of the drop in storage modulus in the rubbery plateau, in addition to the nanoclay content, can be due to the dispersion of the filler in the polymer matrix. Namely, homogeneous dispersion and the presence of large agglomerated nanoparticles may reduce cross-linking and induce chain mobility.51,52 The extent of storage modulus in the rubbery plateau region is proportional to the degree of cross-linking. 8 It has been reported that the high surface area of nanoparticles can interact with polymer chains and influence segmental mobility. Also, the uniform dispersion of particles can allow tight bounds to polymers that are around the particles. Therefore, the well-dispersed nanocomposite requires more energy to begin the viscous flow.8,51,52 The results also demonstrate that the storage modulus of the samples including mHNT is lower compared to that of the samples filled with mLDH, most likely due to its smaller d-spacing as well as lesser mechanical strength.
The variation of loss modulus with temperature for the fabricated WPC specimens using both mLDH and mHNT clays is given in Figure 8. It can be seen that loss modulus increases with the inclusion of nanoclay and reaches a maximum and then decreases. The maximum heat dissipation occurs at the temperature where the loss modulus is maximum indicating a relaxation phenomenon.51,52 The increase in loss modulus is attributed to the increase in energy absorption caused by the addition of nanoclay. Expressly, the increase in loss modulus peak height with nanoclay content is likely to have occurred due to the inhibition of the relaxation process within the composite due to the enhancement in the number of chain segments and higher free volume resulting from nanoclay addition. 8 This result is consistent with the general observation that the introduction of nano-sized particles into a polymer matrix increases its loss modulus. This improvement is due to an increase in internal friction, promoting energy dissipation.8,51,52
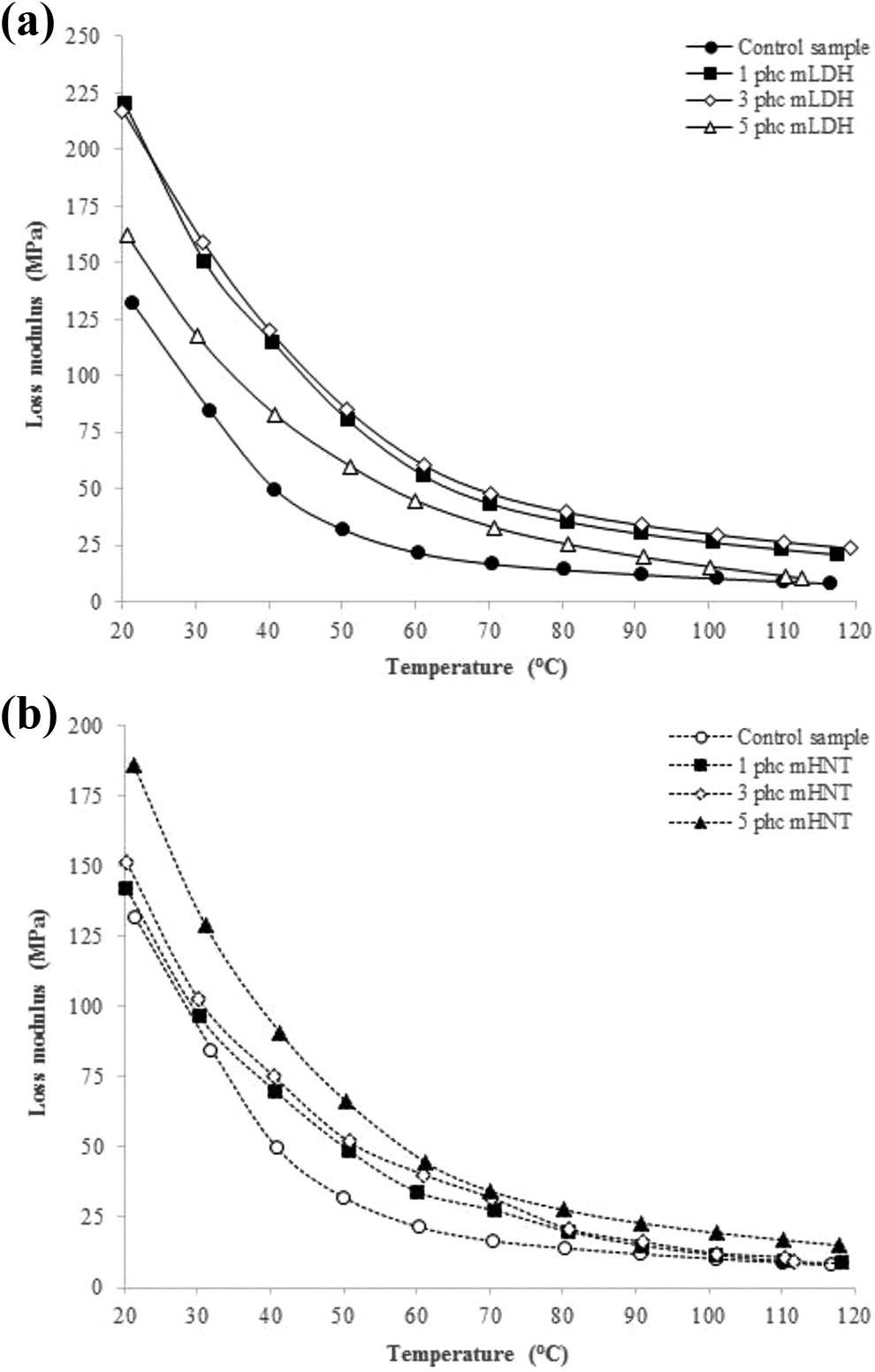
Effects of (a) mLDH and (b) mHNT contents on loss modulus of the fabricated WPC specimens.
Figure 9 represents the dependence of loss-tangent (tan δ) with temperature for the fabricated WPC specimens using both mLDH and mHNT clays. It can be seen that introduction of nanoparticles to the composite specimens shifts the Tg to higher temperatures. It has been reported that Tg rise in a nanocomposite is a result of strong interfacial interaction of particles with the polymer. Besides, nanoparticles can penetrate the spaces between the polymer chains and enhance the density of the composite which leads to a Tg increase.8,51,52 In a well-dispersed nanocomposite, segments can interact with all surfaces of the particles. Also, the number of nanoparticles located within a radius of few nanometers around polymer chains can have a determining role in the Tg of the nanocomposite. The height of the tan δ curve represents the mechanical loss.51,52 The amount of loss is proportional to the input energy required for the motion of the molecular chains when the polymer is close to the transition region. It can be observed that the tan δmax of clay-containing specimens is lower than specimens without nanoclay. The obtained result suggests that the specimen using both mLDH and mHNT has the highest elasticity and least energy of damping in comparison with control samples.
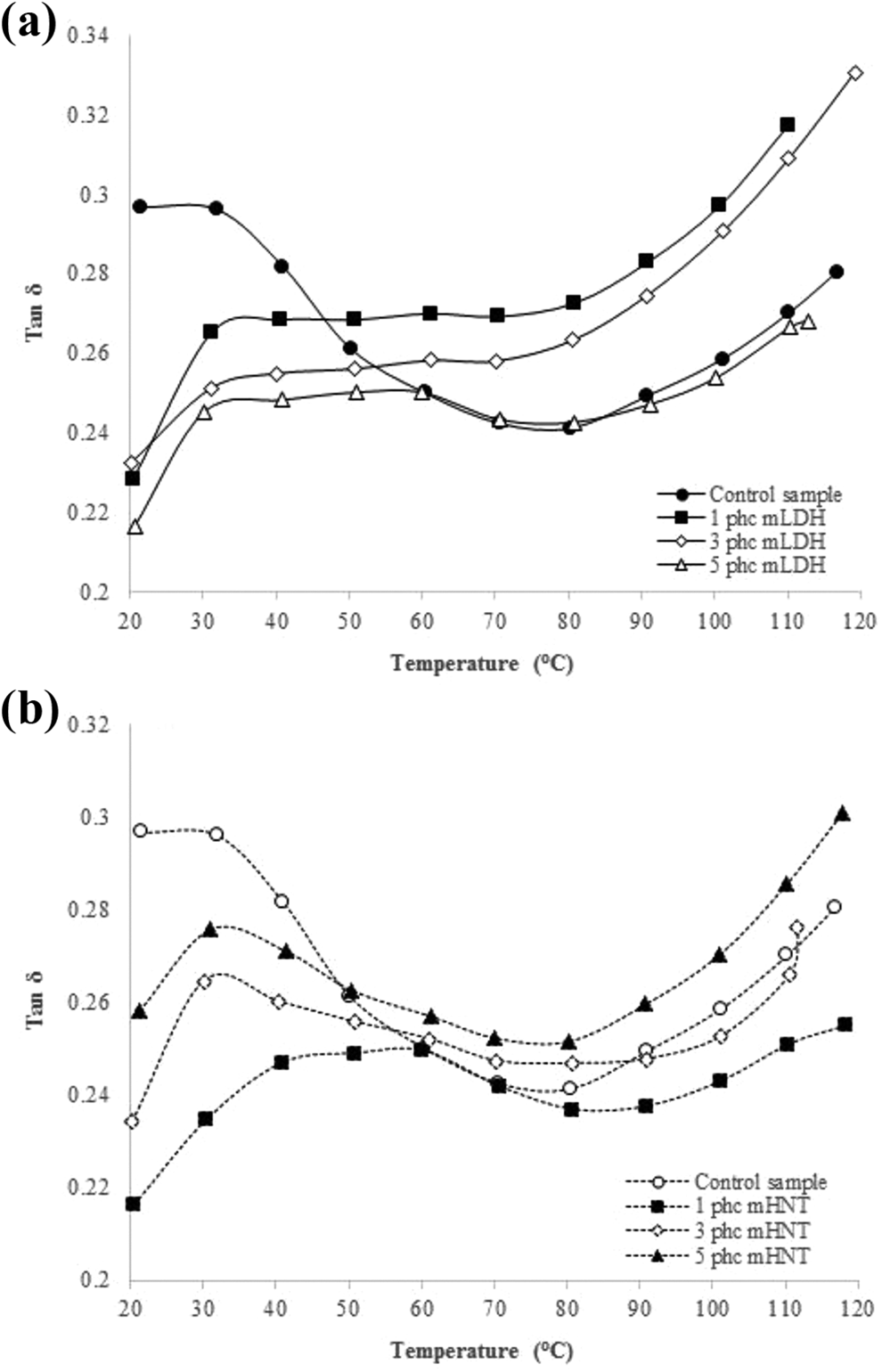
Effects of (a) mLDH and (b) mHNT contents on loss-tangent of the fabricated WPC specimens.
Tan δ peak has also been known as the relaxation peak temperature because it indicates how effectively the material loses energy to molecular rearrangements and internal friction. 8 During the relaxation process, the applied mechanical energy transmits across the chains by segmental motions and conformational rearrangements.8,51,52 In other words, the change in the mobility of the chains which leads to the relaxation process causes a shift in Tg of the system. As evident from Figure 9, in the β and α relaxation region, the position of the peak is shifted to higher temperatures with the inclusion of nanoparticles. This suggests that the molecular motions in the unrestricted amorphous and the restricted crystalline phases of polymer chains are affected by the incorporation of nanoclay.
Physical analysis
The effect of incorporation of both mLDH and mHNT clays with different weight percentages on the water absorption and thickness swelling of the fabricated WPC specimens is depicted in Figure 10. It can be seen that the addition of nanoclay provided substantial improvements in the water-resistance performance. The water absorption and thickness swelling values were observed to be 8.57% and 5.43% for samples made without nanoclay. The addition of 5 phc mLDH into the specimens, significantly decreased the water absorption and thickness swelling by 5.73% and 3.05% respectively, which are 49.56% and 78.03% higher than the control composite. Moreover, the incorporation of the same loading level of mHNT improved the water-resistance of the produced WPC samples, which shows 5.01% for moisture uptake and 3.05% for dimensional stability. This reduction can be explained by the following two effects. In the first place, nanoclay acts as an efficient barrier against moisture uptake due to the generated tortuous pathway for a movement of water molecules diffusing through the composite samples. 38 The second reason is that nano-sized clay particles restrict intermolecular movements of the surrounding polymer matrix thus, making water penetration into the composite much more difficult.34–38 Besides, the results indicated that mHNT with having a smaller d-spacing than mLDH, caused lengthens the transfer of moisture into the composite and plays a more effective role in water repellency.
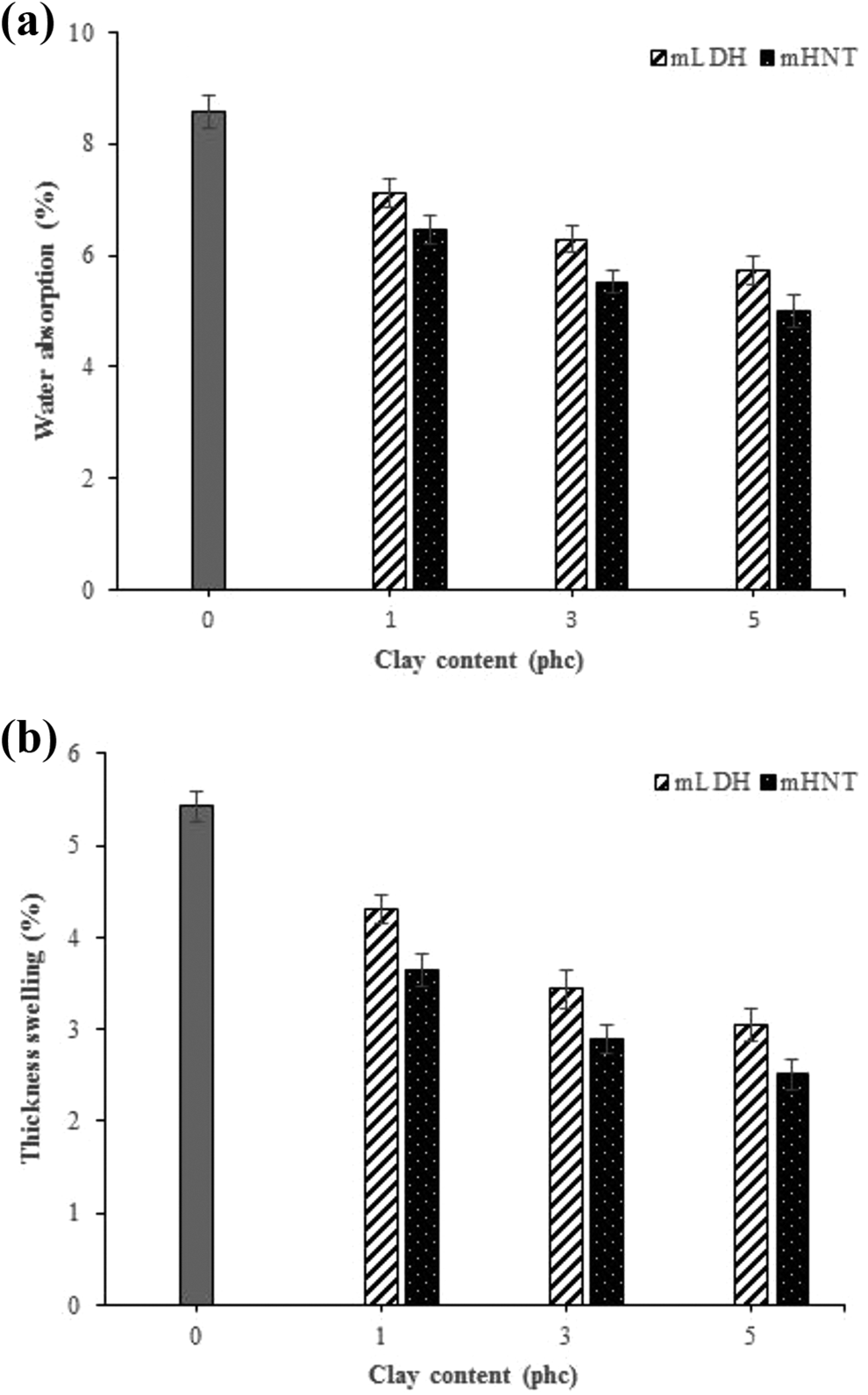
Effects of mLDH and mHNT contents on (a) water absorption and (b) thickness swelling of the fabricated WPC specimens.
Conclusions
In this paper, the effect of mLDH and mHNT at various loading levels on the physical, mechanical and dynamic mechanical characteristics of the produced WPCs have been investigated. The experimental results revealed that the samples including mLDH showed higher mechanical properties, but lower physical properties compared to the samples filled with mHNT. Besides, the dynamic modulus of the composites was greatly improved through the addition of both mLDH and mHNT. In general, the specimens filled with 3 phc mLDH showed the highest values of storage and loss modulus compared with the other ones. The tan δ peak signifying the glass transition temperature of composites shifted to a higher temperature, in the presence of both of mLDH and mHNT. This comparative study will help in the choice of appropriate nanoclay for use in practical applications of WPCs.
Footnotes
Acknowledgment
The authors wish to acknowledge the University of Zabol for financial support.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the University of Zabol, project code: PR-UOZ99-2.
