Abstract
Ring opening polymerization (ROP) of ε-caprolactone (CL) was carried out by bulk polymerization technique using stannous octoate (SO) as a catalyst and in the presence of macro or molecular initiator for 5 h under inert atmosphere with mild stirring. The % yield of poly(ε-caprolactone) (PCL) obtained was calculated and compared based on the chemical structure of macro and molecular initiators. Various analytical techniques such as FT-IR, 1H-NMR, XPS, GPC, DSC, TGA, SEM and WCA were used to characterize the PCL. The FT-IR spectrum showed the C=O stretching around ∼1725 cm−1. The aromatic proton signal of isoeugenol (IE) initiator appeared at 7–8 ppm in the proton NMR spectrum. The experimental results obtained were compared with the literature values.
Introduction
Poly(ε-caprolactone) (PCL) is a bio-medically important thermoplastic polymer with good bio-compatibility and bio-degradability. PCL is synthesized by ring opening polymerization (ROP) in the presence of an initiator. Generally, stannous octoate (SO) is used as a catalyst. For the ROP of CL various organic and inorganic initiators were used. Among the organic initiators, macro and molecular initiators are recently used to initiate the ROP of CL. In 2018, Cotton macro initiator initiated ROP of CL was reported by Agathian et al. 1 Assya et al. 2 reported the nano-fibrillated cellulose initiated ROP of CL. 2 Sugih and research team utilized silylated starch as an initiator for the ROP of CL. 3 PVA was used as a macro initiator for the ROP of CL. 4 Guargum surface initiated ROP of CL was reported by Assimi et al. 5 Lignin was used as a macro initiator for the ROP of CL. 6 Organic acid catalyzed ROP of CL was carried out in the presence of cellulose fiber as an initiator. 7 Silk fiber was used as an initiator for the ROP of CL. 8 PCL was grafted onto cellulose nanocrystals. 9 Similarly, mmPEG, 10 PEG, 11 Chitosan, 12 amphiphilic PEG 13 and PEI14,15 was sued as an initiator for the ROP of CL. The literature survey indicates that the ROP of CL was initiated by –OH or –NH2 group of various natural and synthetic high molecular weight macro initiator.
Apart from the high molecular weight macro initiators, recently low molecular weight molecular initiators are also used as an initiator for the ROP of CL. In 2018, mercaptohexanol was used as a chemical initiator for the ROP of CL. 16 Propargyl alcohol and dibenzocyclooctynol was used as an initiator for the ROP of CL. 17 Yttrium salicylaldimine complex, 18 N-hydroxysuccinimide, 19 gallicacid, 20 penicillin, 21 alanine, 22 barium mercaptoacetate, 23 Ag mercaptoacetate 24 , eosiny, 25 mercaptosuccinate 26 were used as a chemical initiator for the ROP of CL. The literature also indicated the preparation of PCL/glass fiber composites.27,28 There is no comparative study of both macro and molecular initiator initiated ROP of CL report is not available in the literature. The key idea behind the present research work is while changing the molecular weight of the initiator from macro to molecular level, the surface morphology, thermal properties and the physico-chemical properties of PCL is changed. Based on the nature of application, one can select the initiator for the ROP of CL. In the present research work, the above task was taken as a challenge and successfully studied the effect of macro and molecular chemical initiator on the structure-property relationship of PCL. This is the novelty of the present research work.
Experimental
Materials
Stannous octoate (SO) and ε-caprolactone (CL) were freshly purchased from Acros chemicals, USA and used as such. CHCl3 solvent and dietylether non-solvent were purchased from Aldrich chemicals, USA. Ascorbicacid (AA) and isoeugenol (IE) were purchased from Showa chemicals, Japan. Chitosan with the Mw of 12,000 g/mol was purchased from Lancaster, UK. Monomethoxy polyethyleneglycol (mmPEG, Lancaster, Mw-10,000 g/mol) and linear Polyethyleneimine (PEI, Lancaster, Mw-25,000 g/mol) were used as received.
Synthesis of PCL
A typical ring opening polymerization procedure was performed as follows based on the standard literature report.24,25 0.10 mM of SO (catalyst) was taken in a 25 mL capacity two way necked round bottom flask (RBF) and 100 mM of CL (monomer) and 0.02 g of AA (initiator) were charged and mixed well for 120 s. After mixing, the temperature was raised to 413 K for bulk polymerization under nitrogen purging. After 3600 s of ROP, highly viscous liquid was obtained. Now the liquid was cooled, dissolved in chloroform and re-precipitated by the addition of excess of diethylether non-solvent. Further, the sample was washed with de-ionized water and methanol to remove the un-reacted reactants and the same washing procedure was repeated for three times. The polymer samples were subjected to freeze drying. The dried samples were collected, weighed and stored in a vial. The ROP was carried out under the following experimental conditions. The Monomer/Catalyst = 1000 and the Monomer/Initiator [M0/I0] = 100. Reactions are mentioned in Figure 1. The same procedure was followed for the other initiating systems too.
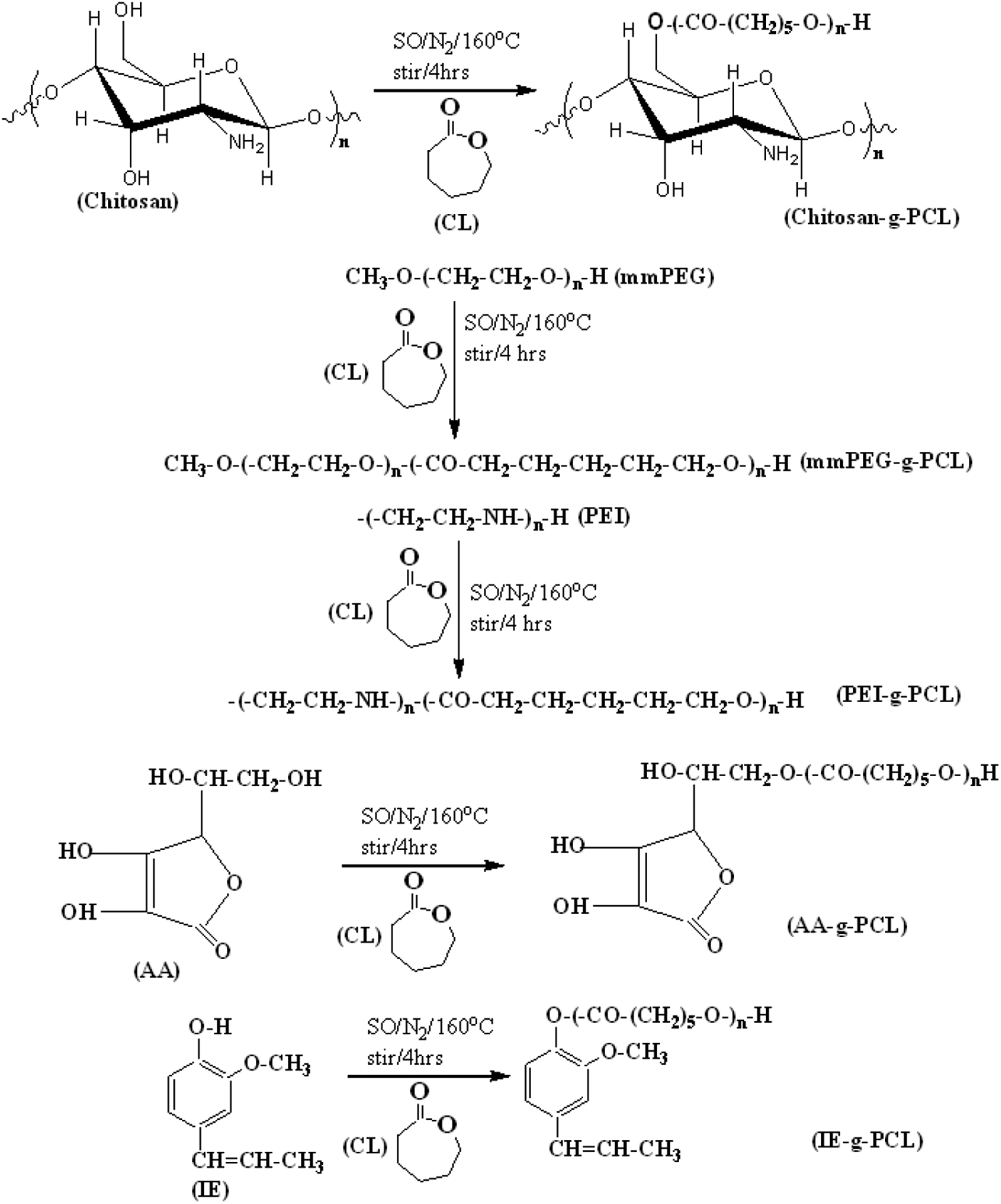
Synthesis of PCL.
Characterizations
The FT-IR spectra of the TPP samples were recorded with the help of Perkin Elmer Spectrum 100 series instrument by KBr pelletization method from 400 to 4000 cm−1. 3 mg of PCL sample was grinded with 200 mg of spectral grade KBr and made into a disc under the pressure of 7 tons. 1H-Nuclear Magnetic Resonance (NMR) (500 MHz) spectra were obtained using an NMR apparatus (Varian, Unity Inova-500 NMR) at room temperature in CDCl3 solvent. DSC was measured by using Universal V4.4A TA Instruments (simultaneous DSC and TGA analyzer) under nitrogen atmosphere at the heating rate of 10 K/min from room temperature to 373 K. The second heating scan of the sample was considered in order to delete the previous thermal history of the sample. The same instrument was used for TGA at the heating rate of 10 K/min under air atmosphere. A Waters 2690 GPC instrument was used to determine the Mn and Mw of the polymer samples by using tetrahydrofuran (THF) as an eluent at room temperature at the flow rate of 1 mL min−1 against polystyrene (PS) standard. The WCA of the polymer samples was measured by Kyowa DMs-200, Japan instrument. The WCA of the samples were measured for six times and the average one is considered here. The XPS of the polymer samples were measured by Thermo Scientific, Theta Probe, UK instrument. The SEM image of the polymer sample was recorded with the help of Hitachi S-3700 N SEM, Japan model instrument.
Results and discussion
% yield report
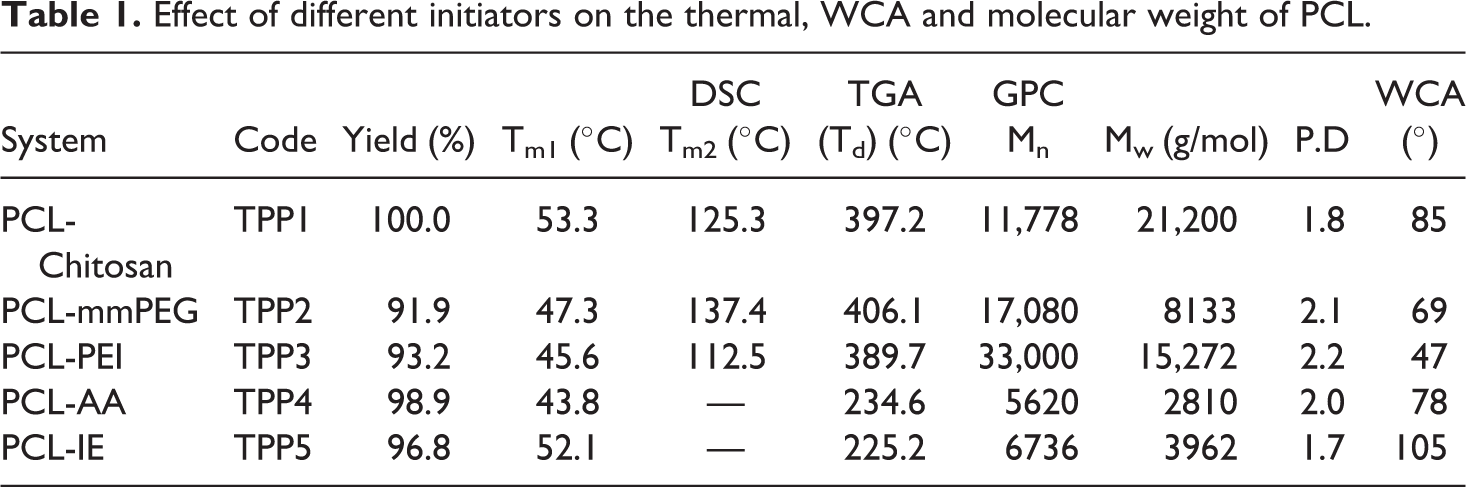
The ring opening polymerization (ROP) efficiency of an initiator is predicted based on the % yield of the resultant product. In the current research work, the authors used Chitosan (contains –NH2, primary alcohol and secondary alcohol as functional groups), mmPEG (contains primary alcoholic group as a functional group), PEI (contains primary and secondary amine as functional groups), AA (contains primary, secondary and tertiary alcoholic groups as functional groups) and IE (contains one phenolic functional group). In the case of Chitosan, both the –NH2 and –OH groups per repeating unit were involved in the ROP of CL. It is well known that the –OH group is more effective toward the ROP of CL than the amino group. Among the alcoholic groups present in the Chitosan cyclic structure, the secondary alcoholic group is more effective toward the ROP of CL than the primary alcoholic group due to the stericful structure of secondary alcoholic group. Hence, the order of efficiency toward the ROP of CL is 2° OH > 1° OH > amine functional groups. In the case of mmPEG, one primary alcoholic OH group per polymer chain was involved in the ROP of CL. Among the initiators used, the Chitosan system yielded 100% PCL (Table 1) because of active involvement of both –OH and –NH2 groups in the ROP of CL. At the same time, the mmPEG system yielded 91.9% PCL (Table 1) because of very active involvement of only one –OH group per chain in the ROP of CL. In overall comparison, all the systems yielded above 90% yield because of active involvement of –OH group in the ROP of CL. In the present research work, two types of initiators such as macro initiator (Chitosan, mmPEG and PEI) and molecular initiator (AA and IE) were used. Among these, the molecular initiators exhibited excellent initiating capability toward the ROP of CL. This is associated with the smaller size without any steric effect of molecular initiators. In the case of macro initiators particularly, Chitosan contains lot of initiating groups and all the initiating groups were participated in the ROP of CL. Moreover, the Chitosan is in the form of fiber. Generally, the molecular initiators yielded the highest % yield than the macro initiators except Chitosan. When compared with the literature report, 29 the present system yielded higher % yield.
Effect of different initiators on the thermal, WCA and molecular weight of PCL.
FT-IR spectral study
The FT-IR spectrum gives an idea about the functional groups present in the polymer systems. The FT-IR spectrum of Chitosan initiated ROP of CL is shown in Figure 2(a). The important peaks are discussed here. Chitosan contains –NH2, primary alcoholic group and secondary alcoholic groups. The system exhibits NH stretching (3566 cm−1), OH stretching (3435 cm−1), CH symmetric and asymmetric stretchings (2849 and 2944 cm−1, respectively), C=O stretching (1725 cm−1), 22 C–O–C stretching (1173 cm−1) and C–H out of plane bending vibration (OPBV) (725 cm−1). The NH, OH and C–O–C stretchings are corresponding to Chitosan. This confirmed the chemical grafting of PCL onto Chitosan matrix. Figure 2(b) represents the FT-IR spectrum of mmPEG initiated ROP of CL. This system exhibits C–H stretchings (2848, 2940 cm−1), C=O stretching (1726 cm−1), C–O–C stretching (1189 cm−1) and C–H OPBV (725 cm−1). The C–O–C stretching is corresponding to the mmPEG whereas the C=O, C–H OPBV and C–H stretchings are corresponding to PCL. This confirmed the formation of mmPEG end capped PCL chains. The PEI initiated ROP of CL is indicated in Figure 2(c). The N–H and O–H stretchings of PEI and PCL are merged and appeared at 3454 cm−1. 22 The C–H stretchings of PCL (2858, 2933 cm−1), C–H stretchings of PEI (3020 cm−1), C=O stretching (1747 cm−1), C–N stretching (1383 cm−1), 22 C–N–C stretching (1049 cm−1), C–H OPBV of PEI and PCL are observed at 826 and 883 cm−1, respectively. An important point noted here is the intensity of C=O stretching of PCL appeared at 1747 cm−1 is compressed due to the long PEI chains. Appearance of C–N stretching and the merged N–H and O–H stretchings confirmed the PEI-PCL block copolymer formation. Moreover, there is no availability of free –NH2 group. It means the primary amino group involved in the ROP of CL. Figure 2(d) shows the FT-IR spectrum of AA initiated ROP of CL. The important peaks are characterized below. Here one can observe –OH stretching (3465 cm−1), C–H stretchings (2858, 2952 cm−1), C=O stretchings (1727 cm−1) and C–H OPBV (725 cm−1). A broad peak around 1200 cm−1 is corresponding to the ester C–O–C linkage. 23 An interesting point noted here is the C=C stretching of AA is not observed here due to the keto-enol tautomerism at a given pH. The above mentioned peaks confirmed the AA initiated ROP of CL. Figure 2(e) denotes the FT-IR spectrum of IE initiated ROP of CL. In this system also the above mentioned –OH stretching (3323 cm−1), C–H stretchings (2839, 2940 cm−1), C=O stretching (1757 cm−1), C=C stretching (1626 cm−1), C–O–C stretching (1030 cm−1) and C–H OPBV (645 cm−1). Here the C=C stretching is corresponding to the IE unit whereas the C=O stretching is due to the PCL chains. Hence, Figure 2(a) to (e) confirmed the ROP of CL by different initiating groups like –OH and –NH2.
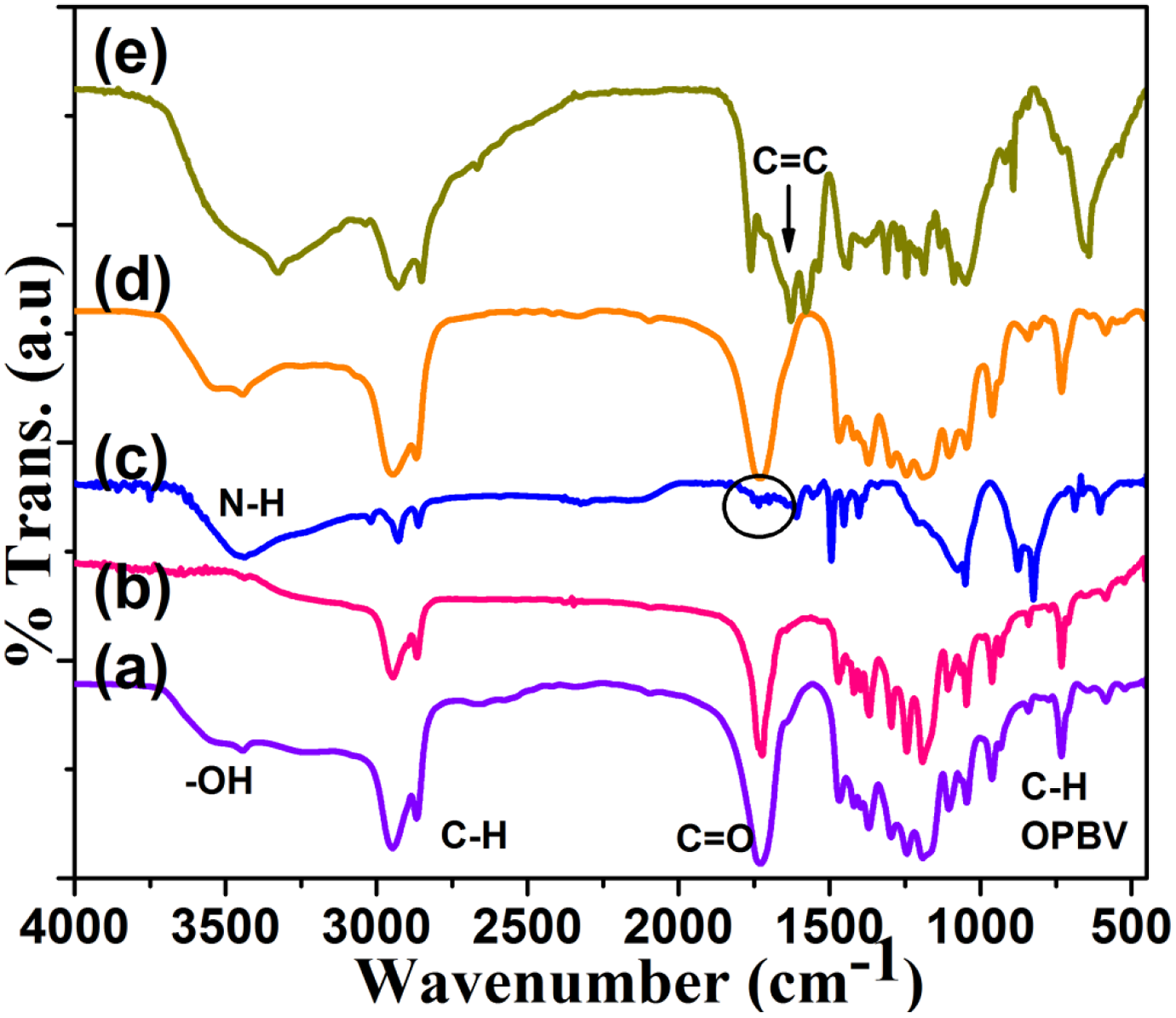
FT-IR spectrum of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
DSC analysis
Generally, the DSC provides an idea about the thermal transitions present in the polymer. The melting temperature (Tm) of PCL initiated with different initiators are given in Figure 3. Figure 3(a) shows the DSC heating scan of TPP1 system and the data is shown in Table 1. The Tm of PCL 25 was appeared at 53.3°C (Table 1) whereas the Tm of Chitosan appeared at 125.3°C. One more broad endothermic peak appeared at 88.3°C is ascribed to the Chitosan grafted with PCL chains. Appearance of these thermal transitions confirmed the diblock copolymer formation between Chitosan and PCL. The TPP2 system showed the Tm value of PCL at 47.3°C (Figure 3(b)) with Tg of mmPEG at 137.4°C. Here also the DSC result confirmed the diblock copolymer formation between mmPEG and PCL. The TPP3 system showed the Tm value of PCL at 45.6°C (Table 1) whereas the PEI exhibited the Tg value of 112.5°C (Figure 3(c)). Even though the PCL is grafted onto PEI, there appear two transitions corresponding to the Tm of PCL and Tg of PEI. Figure 4(d) indicates the DSC heating scan of TPP4 system. The Tm of PCL was observed at 43.8°C. The Tm of TPP5 system was determined as 52.1°C (Figure 3(e), Table 1). In overall comparison, the TPP1 system exhibited the highest Tm of PCL due to the following reasons: (i) presence of more number of highly active –OH groups for the ROP of CL and (ii) presence of intermolecular hydrogen bonding. The Tm of PCL in the present research work is in accordance with the literature reports.22,23 The molecular initiators produced PCL with single thermal transition whereas the macro initiator produced more than one thermal transition due to difference in molecular weights.
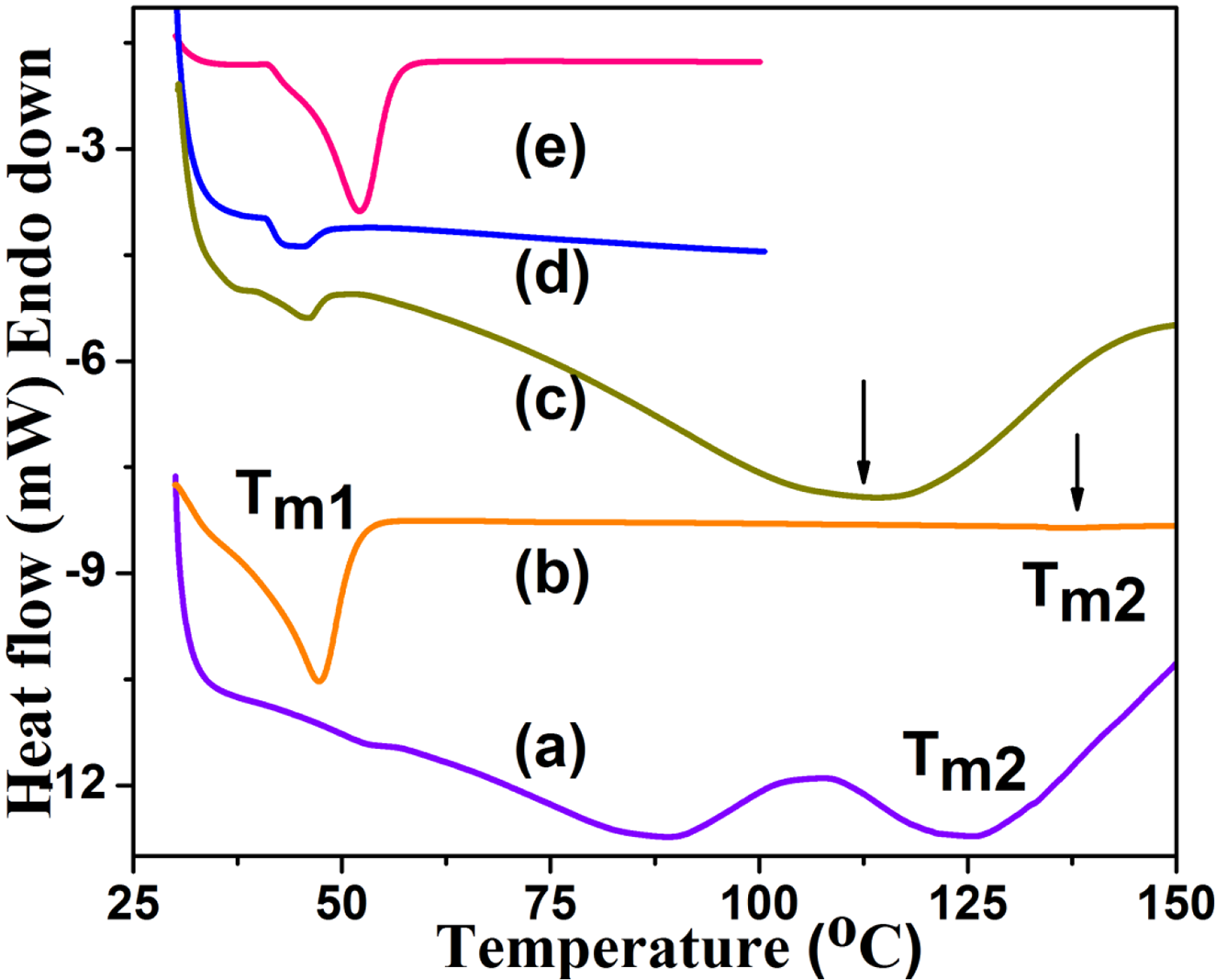
DSC heating scan of (a)TPP1, (b)TPP2, (c)TPP3, (d)TPP4, (e)TPP5.
TGA history
The thermal stability of PCL initiated by various initiators can be tested through TGA. The TGA thermogram of TPP1 System is given in Figure 4(a) with two step degradation process. The minor weight loss around 296°C is ascribed to the degradation of linkage between Chitosan and PCL. The second major weight loss around 397.2°C is due to the degradation of PCL backbone. 29 Above 450°C, 94% weight residue was remained. The data is given in Table 1. The TGA thermogram of TPP2 system is given in Figure 4(b) with one minor (around 322°C due to the degradation linkage between mmPEG and PCL) and one major (406°C due to the degradation of PCL backbone) weight loss steps. Above 450°C 95.7% weight residue was seen. The TGA thermogram of TPP3 system is given in Figure 4(c) with PEI-PCL linkage degradation (278°C) and PCL backbone degradation (389°C). The % weight residue remained above 450°C is 94%. In comparison among the macro initiators, the mmPEG yielded PCL with highest Td. This confirmed the highly active participation of –OH group toward the ROP of CL. The TPP4 system yielded three step degradation process (Figure 4(d)). The minor weight loss below 175°C is due to the removal of moisture and unreacted CL monomer units. A major weight loss around 234.6°C is associated with the degradation of PCL backbone. The AA degradation was noticed around 390°C. Above 450°C, the system showed 93% weight residue remained. The TGA thermogram of TPP5 system is given in Figure 4(e) with three step degradation process. The minor weight loss below 200°C is due to the removal of moisture and degradation of linkage between PCL and IE. The major weight loss (around 225°C) is due to the degradation of PCL backbone. The next minor weight loss is due to the degradation of IE. Above 450°C the system exhibited 98.3% weight residue remained. In comparison among the molecular initiators, the AA initiator yielded PCL with the highest Td of 234.6°C. In overall comparison, the mmPEG yielded PCL with the highest Td. When compared with the literature report, 26 the present systems exhibited excellent results based on % residue remained above 450°C at the same time poor result based on Td of PCL. This depends on the nature of the initiator system. In the case of TGA analysis, apart from the molecular weight of the polymer, the hydrophilicity (measured by water contact angle) played vital role because of absorption of moisture from the environment. This is further discussed in the WCA chapter.
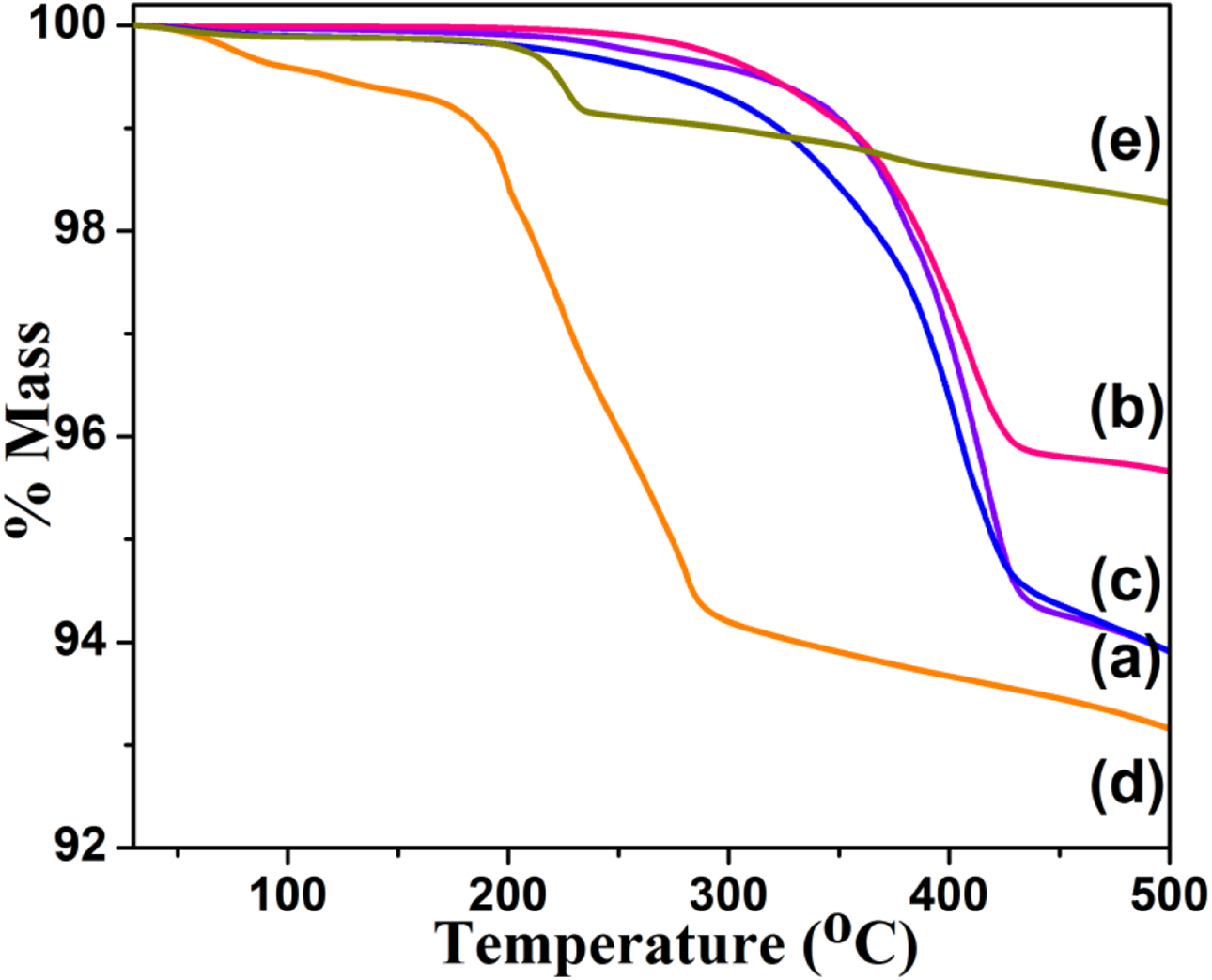
TGA trace of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
GPC analysis
The efficiency of initiators toward the ROP of CL was tested through GPC analysis. For the ROP of CL, two types of initiators were used such as macro and molecular initiators. Both the number average molecular weight (Mn), weight average molecular weight (Mw) and polydispersity (PD) of PCL confirmed the efficiency of initiators toward the ROP of CL. Figure 5(a) indicates the GPC trace of TPP1 system. It showed the Mw of 21200 g/mol with the PD value of 1.8 (Table 1). This is in accordance with the literature reports.23,29 Here, the primary alcoholic, secondary alcoholic and the primary amino groups were involved in the ROP of CL. The GPC trace of TPP2 system is shown in Figure 5(b). The mmPEG-PCL system exhibited the Mw of 17,080 with the P.D value of 2.1. In this case, the ROP of CL occurred at the chain end of mmPEG and hence there is no steric effect. The Mw and PD for PCL-PEI (TPP3) system is 33,600 g/mol and 2.2, respectively (Figure 5(c), Table 1). The PEI contains both primary and secondary amino groups. The chain end primary amino group of PEI involved in the ROP of CL without any steric effect whereas the secondary amino group experienced some amount of steric effect. In comparison among the macro initiators, the Chitosan macro initiator yielded the PCL with the Mw and degree of polymerization (DP) of 9200 g/mol and 80.7, respectively. This is due to the active involvement of three hydroxyl groups and one amino groups present in each repeating units of Chitosan. The PEI macro initiator yielded the PCL with the Mw and DP values of 8600 g/mol and 75.4, respectively. This is due to the ROP of CL by the –NH2 and –NH groups present in the linear PEI. In the case of mmPEG initiated ROP of CL, the Mw and DP of PCL obtained were calculated as 7080 g/mol and 62.5, respectively. This is due to the active participation of only one primary OH group toward the ROP of CL. Similarly, the GPC traces were recorded for the molecular initiators initiated ROP of CL and the data are given in Table 1. The TPP4 system exhibited the Mw and PD values as 5620 g/mol and 2.0 respectively. The TPP5 system showed the Mw of 6736 g/mol and the PD value of 1.7. In comparison, the IE initiator yielded the PCL with the Mw value of 6736 g/mol. Again this confirmed the active participation of –OH group toward the ROP of CL. In overall comparison, the macro initiators yielded the PD value of >2.0 and led to the conclusion of branching or cross linking reaction during the ROP of CL. The GPC study proved that based on the application, one can choose the correct initiator for the ROP of CL.
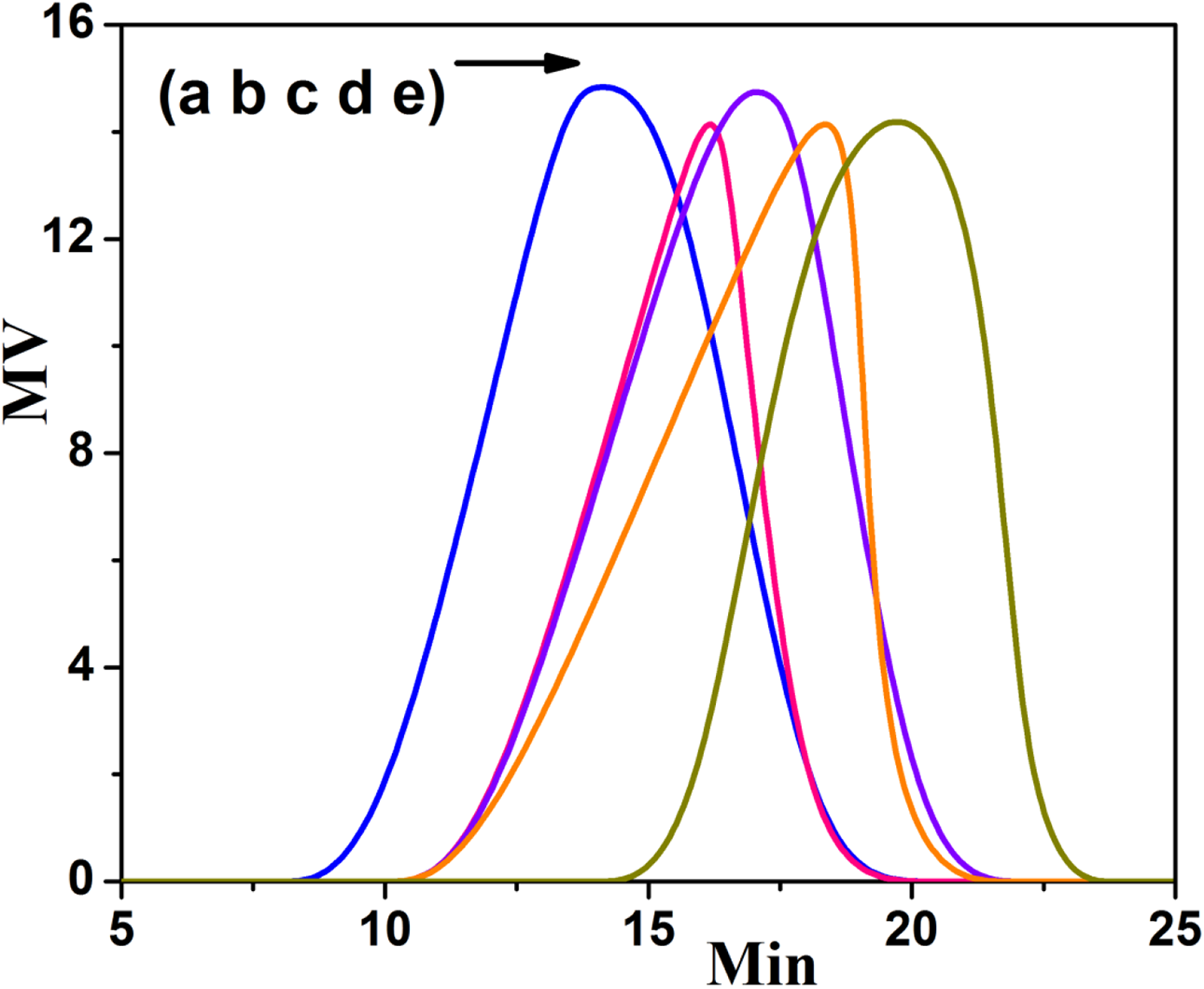
GPC trace of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
1H-NMR study
The 1H-NMR spectrum confirmed the chemical structure of PCL with different initiating systems. Figure 6(a) to (e) indicates the 1H-NMR spectra of PCL initiated by Chitosan, mmPEG, PEI, AA and IE respectively. The peaks and structure is clearly indicated in Figure 6. The methylene groups nearer to –OH and –CO2 groups are appeared at 4.2 and 2.4 ppm respectively.25,29 The aromatic protons of IE is appeared around 7.6 ppm. This confirmed the IE initiated ROP of CL. Thus the 1H-NMR spectrum confirmed the chemical structure of PCL formed during the ROP of CL initiated by various macro and molecular initiators.
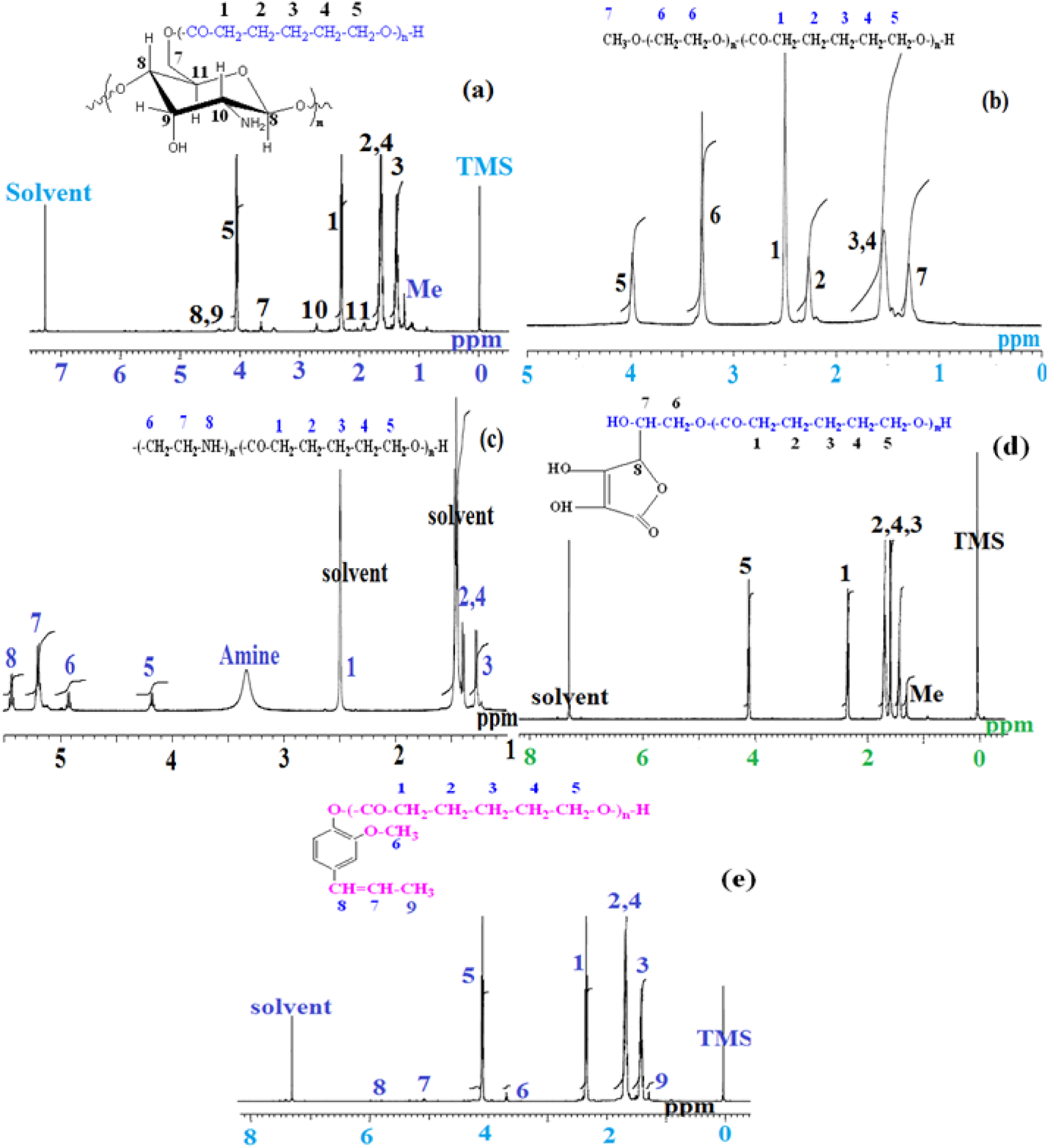
1H-NMR spectrum of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
SEM analysis report
The surface morphology of TPP1 system is shown in Figure 7(a) with broken stone like morphology. 29 Here, one can see the bigger size voids too. Voids present in the polymer chains are suitable for the drug loading applications in bio-medical engineering field. Figure 7(b) indicates the SEM image of TPP2 system with some molecular voids but with smaller size. The polymer with smaller sized micro voids is a suitable candidate for the membrane applications. The SEM image of TPP3 system is given in Figure 7(c) with some micro voids. In comparison among the macro initiators initiated ROP of CL, all the initiators exhibited almost the same surface morphology. The TPP4 system showed an entirely different surface morphology (Figure 7(d)). It looks like fiber with the length of ∼15 um and with the breadth of 2–3 um. The surface morphology of TPP5 system is given in Figure 7(e) with lesser number of micro voids. The size of the void was determined as <5 um. This study concludes that the surface morphology of PCL depends on the nature and molecular weight of initiator used. This also indicated that based on the application one can select the initiator for the ROP of CL.

SEM image of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
XPS analysis
The XPS gives the information on electrons present in the outer most level. The TPP1 system showed the C1s (284 eV), N1s (389 eV) and O1s (531 eV) levels 30 (Figure 8(a)). The N1s is derived from Chitosan macro initiator. Similarly, the TPP3 system also exhibited the N1s peak derived from the PEI macro initiator (Figure 8(c)). The other systems simply exhibited C1s and O1s only. In comparison, the TPP3 system has higher % of N derived from the PEI macro initiator. The appearance of N1s peak confirmed the ROP of CL initiated by PEI and Chitosan like macro initiators.
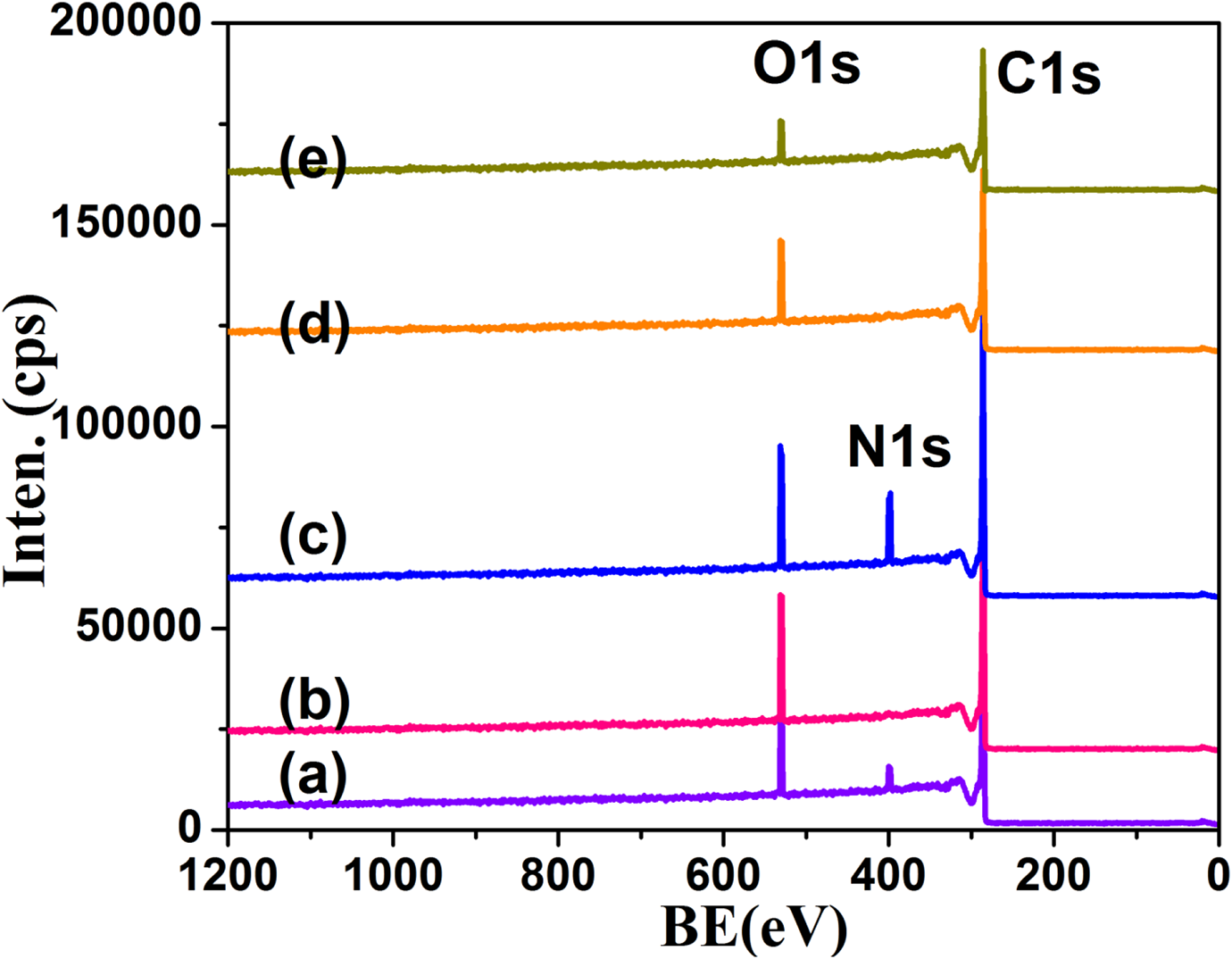
XPS of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
Water contact angle (WCA) measurement
The WCA measurement confirmed that the synthesized polymer is hydrophobic or hydrophilic in nature. Table 1 shows the data. The TPP1 system showed the WCA of 85° (Figure 9(a)). The TPP3 system showed the WCA of 47° (Figure 9(c)). In overall comparison, the TPP5 system showed the highest WCA of 105° and this confirmed the hydrophobic nature of the system. This can be explained as follows: i) due to the presence of bulky phenyl ring in the initiator led to the rigidity, ii) the presence of C=C makes the system more rigid, iii) the PCL is hydrophobic. As a result, the TPP5 system showed the highest WCA. This is in accordance with the literature report of WCA of PCL. 31 In the case of TPP3, it showed the WCA of 47° (Figure 9(e)) and this confirmed that still traces amount of –NH or –NH2 groups are freely available even after the ROP of CL due to steric effect. This leads to hydrophilic nature of TPP3 system. Moreover, NH or NH2 groups are less reactive toward the ROP of CL rather than the –OH groups.

WCA of (a) TPP1, (b) TPP2, (c) TPP3, (d) TPP4, (e) TPP5.
Conclusions
The important points are presented as conclusions. The ROP of CL was done by bulk polymerization method. The Chitosan based macro initiator yielded 100% PCL because of the involvement of 2-OH and 1-NH2 groups toward the ROP of CL. The TPP1 system produced the PCL with the highest Tm value of 53.3°C. The TPP2 system yielded the PCL with the highest Td value of 406.1°C. The TPP3 system produced the highest Mw of 33,000 g/mol. The TPP5 system exhibited the highest WCA of 105° due to the rigid nature of phenyl and allyl group. The 1H-NMR showed a small hump around 7.6 ppm for the phenyl proton of IE present in the TPP5 system. The molecular initiator (AA) produced PCL with fiber like morphology. The TPP1 and TPP3 systems showed N1s peak at 399 eV in the XPS due to the macro initiator. This study proved that while changing the initiator from macro to molecular level the thermal, surface morphology, molecular weight and WCA values were varied. Depends on the nature and application, the macro or molecular initiators are preferred. Next our team is going to test the drug release and membrane activities of PCL synthesized by various macro and molecular initiators.
Footnotes
Acknowledgements
Dr N. Sundararajan, Associate Professor, Department of English, KCET, Madurai is gratefully acknowledged for his valuable help during this manuscript preparation work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
