Abstract
Synthesized polystyrene (PSt) with a molecular weight of 2100 g/mol, hexamethylene diisocyanate (HMDI), 6-hexylamino benzamide (6B), and N1-(6-aminohexyl)-N4-(6-benzoylaminohexyl)terephthalamide (6T6B) are used to make a copolymer of thermoplastic elastomers. The prepared polymers’ inherent viscosity results (0.4–1.1 dL/g) support the polymer’s high mass. The presence of a monodentate urea group in the polymer chain is confirmed by FT-IR tests. The temperature dependence of FT-IR confirms that the synthesized copolymer has a space length dependent reversible crystallinity. Data from differential scanning calorimetry (DSC) also shows that the hard segment crystallization is strong and reversible in nature. The XRD results show that the polymer is semi-crystalline. The TGA analysis confirmed that the synthesized copolymers are thermally stable up to 290°C. The presence of hydrogen bonds in polymer chains is thermally reversible. The polymer’s solvent resistivity is excellent due to its high crystallinity.
Keywords
Introduction
Polyurethanes (PU) are used for various applications due to its awesome properties. Most of the polyurethane used as fibers, coating materials, foams, etc.1–3 and present of different type of soft and hard segments is responsible for the PU chain flexibility which is essential for the above said applications.4–7 To date, thermoplastic polymer is being synthesized by varying hard and soft segments for different applications.6,8–10 There hasn’t been much research into thermoplastic polyurethane (TPU) with various functional groups, and the existence of a functional group in TPU can decide its applications and uses. 11 In comparison to other processes, the reaction between an amine and isocyanates is quicker, which can result in the incorporation of hard segment (HS) in the polymer chain, which can produce valuable properties.12–15 According to a previous paper, the presence of an amide moiety in the polymer chain is responsible for the polymer solubility without significantly altering its thermal stability, and that’s why this form of polymer is used in a variety of applications. 16 Polymer systems can benefit from segmented block copolymers with uniform hard segments, which can provide a variety of desirable properties. The uniform hard segment is crystallized fast and can crystallize completely The segmented block copolymer’s thermal and mechanical properties would be affected by the complete crystallization of the HS. The uniform hard segment polymer would have stronger mechanical properties than the randomized hard segment. 17 A triblock copolymer with crystallizable amide hard segments at the chain ends is a new addition to the field of segmented block copolymers. The triblock thus made showed an aggregation of HS in the melt, 18 which leads to complete (100%) crystallization of HS in a uniform thickness. Compared to multiblock copolymers, this custom-made triblock copolymer had a higher storage modulus and excellent tensile strength. 19 The polymer would have an excellent solvent resistance due to the full crystallization of the hard segment. 20 Polystyrene-based compound with a high Tg used in a variety of applications, including electrical and electronic industries. Polystyrene is a hazy substance with pitiful solvent resistivity and a high strain rate. It is reacted with semi-crystalline material to prevent these disadvantages. 21 Semi-crystalline materials often exhibit excellent solvent resistance and dimensional stability even above the material’s Tg. Incorporating semi-crystalline nature into the polystyrene matrix requires challenging laboratory conditions such as high temperature, high vacuum, and so on. However, inserting a urethane group into polystyrene is relatively simple, and the polymer can be synthesized in relatively mild conditions. As a result, the focus of this article is on the synthesis and characterization of a thermoplastic polymer of styrene with hard segments based on urethane-urea-amide. The polystyrene’s solvent resistivity is improved by the segmented hard segments 6B and 6T6B. The different ratio of hard and soft segmented copolymer are prepared, characterized and tested for thermal stability, swelling property and solvent resistivity.
Experimental
Materials
1,6-Hexamethylenediisocyanate (HMDI), Styrene, Potassium bromide, phenyl benzoate, 1,6-hexamethyldiamine and phenol are bought from Merk chemicals. The solvents chloroform, acetone, dimethyl formamide, 1,1,2,2-tetrachloroethane, Dichloro methane, ethyl acetate, benzene, hexane, tetrahydrofuran, toluene, methanol, 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) and Deuterated trifluoroacetic acid (TFA-d) are brought from Aldrich chemicals. The hard segment 6B and 6T6B is synthesized and used for triblock copolymer preparation. 22 OH-PSt-OH is synthesized and used as a soft segment. 23
Synthetic procedure of block copolymer
Different moles of synthesized hard and soft segments are used for the synthesis of segmented copolymer which is given in the Table 1. Thermoplastic elastomer is prepared according to the Figure 1. The procedure for the preparation of the polymer A1 is given here as a representative for the series of triblock copolymers. Di-hydroxy terminated polystyrene having weight average molecular weight of 2100 g/mol (6.3 g, 0.003 mol) is taken in a round bottom flask, stirred mechanically under inert atmosphere at 120°C. At this temperature HMDI (1.01 g, 0.006 mol) is added and stirring is continued for 3 h. After this the temperature of the medium raised to 150°C and 1.15 g (0.006 mol) of hexylamine benzamide (6B) is added and stirred for 2 h. The entire synthesized thermoplastic polymer is transparent, while being semi-crystalline.
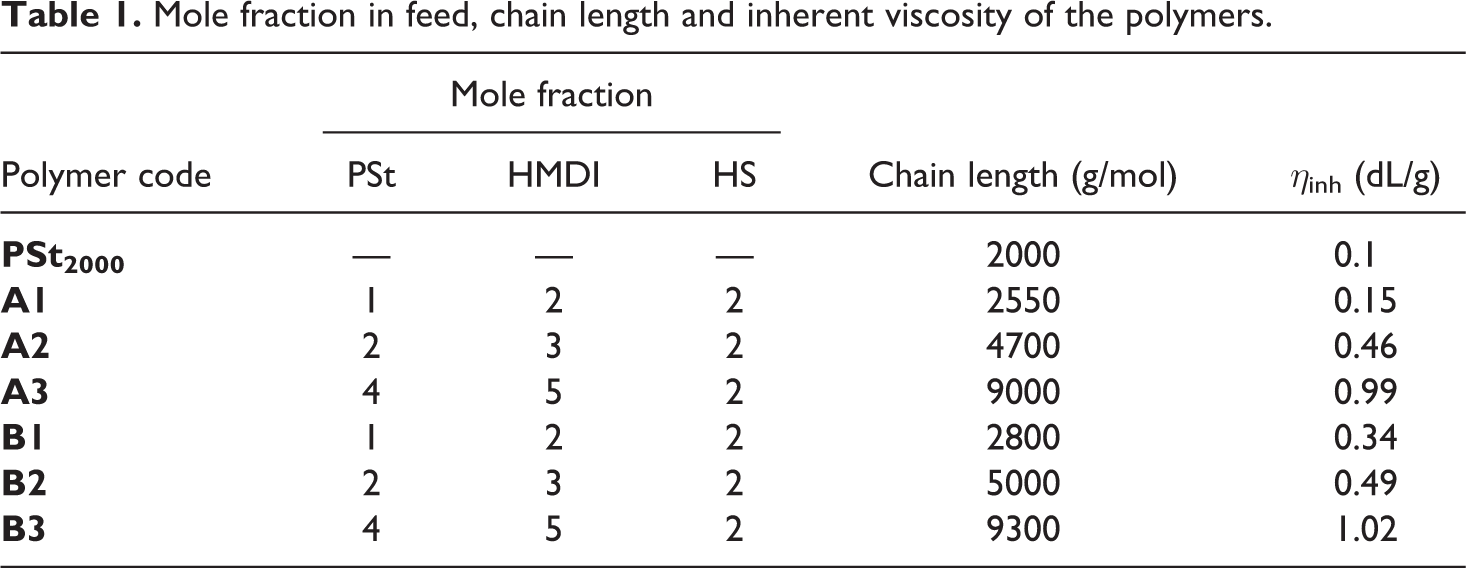
Mole fraction in feed, chain length and inherent viscosity of the polymers.
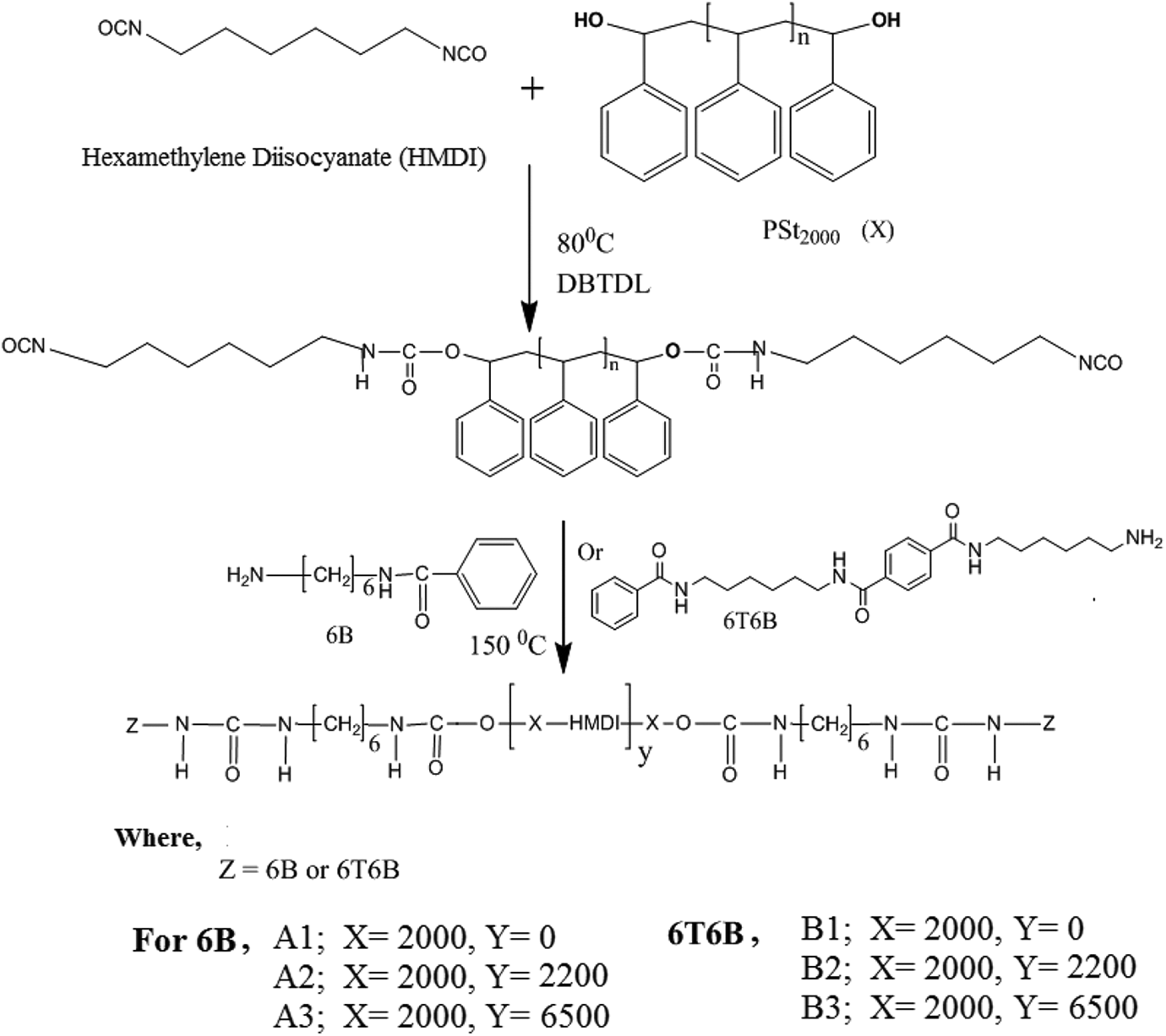
The synthetic route of semi-crystalline copolymer based on 6B and 6T6B.
6B-based TPE (A1): Yield: 7.8 g (92%).1H-NMR (TFA-d, δ, ppm): 6.6–7.6 (b, aromatic proton), 4.4 (t, C
6T6B-based TPE (B1): Yield: 9.2 g (91%).1H-NMR (δ, ppm): 6.7–8 (b, aromatic proton), 4.5 (t, C
FT-IR spectra
Alpha Bruker FT-IR, with a resolution of 4 cm−1 is used for the analysis. A small drop of a polymer solution (HFIP (1 g/L)) is placed on an ironed KBr pellet and dried in air. A thin film thus formed will have stable morphology and is used for the measurements at normal temperature. 24 Similar specimen is prepared and used for recording FT-IR at different temperature using the Alpha Bruker instrument connected with alpha-T at 30–120°C. The degree of crystallinity found out by using the known procedure. 23
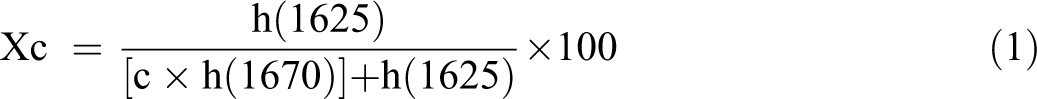
The amorphous and crystalline amide peak height (h) are related by the factor “C” which is having the value of 2.4. 23
Inherent viscosity measurements
The inherent viscosity of the synthesized polymer at a strength of 0.1 dL/g in a 1:1 molar solution of phenol/1,1,2,2-tetrachloroethane is found at 25°C by capillary Ubbelohde by adopting the known procedure. 23
1H-NMR
Bruker FT-NMR spectrophotometer (400 MHz) used to record proton NMR spectra of the synthesized polymer at RT here TFA-d used as a solvent and tetramethylsilane (TMS) used as an interior allusion.
DSC
DSC spectra of the synthesized polymer are recorded on a Perkin Elmer DSC 7 apparatus equipped with a PE 7770 computer and TAS-7 software.
TGA
Thermal response of the synthesized polymer found out using DuPont 951 thermo gravimetric analyzer under inert atmosphere.
Swelling ratio of polymer
The polymer is prepared as bars with the help of compression molded instrument and placed in desiccators with a layer of dematerialized water for 28 days at room temperature. The weight gain of the polymer gives the swelling ratio. 23
Solvent resistivity
A compression molded polymer sample of the dimension 10 × 10 × 2 mm is employed for the solubility measurements. The copolymer sample is weighed and dipped in the solvents (50 mL) which is shaken for 60 min. The solvent is then decanted and the residue in the flask is dried for 24 h at 70°C. The weight loss thus obtained is then used solvent resistivity calculation.
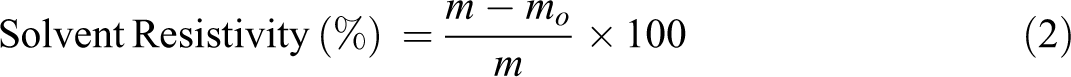
where, m and mo is the weight of dry substance (mg) before and after solvent treatment respectively.
Results and discussion
Synthesis of polyurethane-urea amide (PUUA)
Thermoplastic copolymer is made in two steps; first, the pre-polymer is made by melting 1 mole of Polystyrene with 2 moles of diisocyanate, then adding 6B or 6T6B to the pre-polymer. The obtained polymer is characterized using 1H NMR technique and the peak value confirms the formation of the desired product. The 1H NMR spectrum of the triblock copolymers A1 and B1 are presented in the Figure 2. Similar type of NMR spectrum is obtained for other copolymers in the series A and B. The existence of hydrogen atoms linked to urethane, urea, aromatic, and aliphatic groups in the polymer chain confirms that the target groups were present. 18 The concentration of the hard segment is varied by increasing the soft segment length. In the copolymer, the hard segment weight percentage is varied from 27% to 8% for series A and 55–13% for series B. Table 1 displays the mole ratio in the feed. The product thus formed is given in the Figure 1.
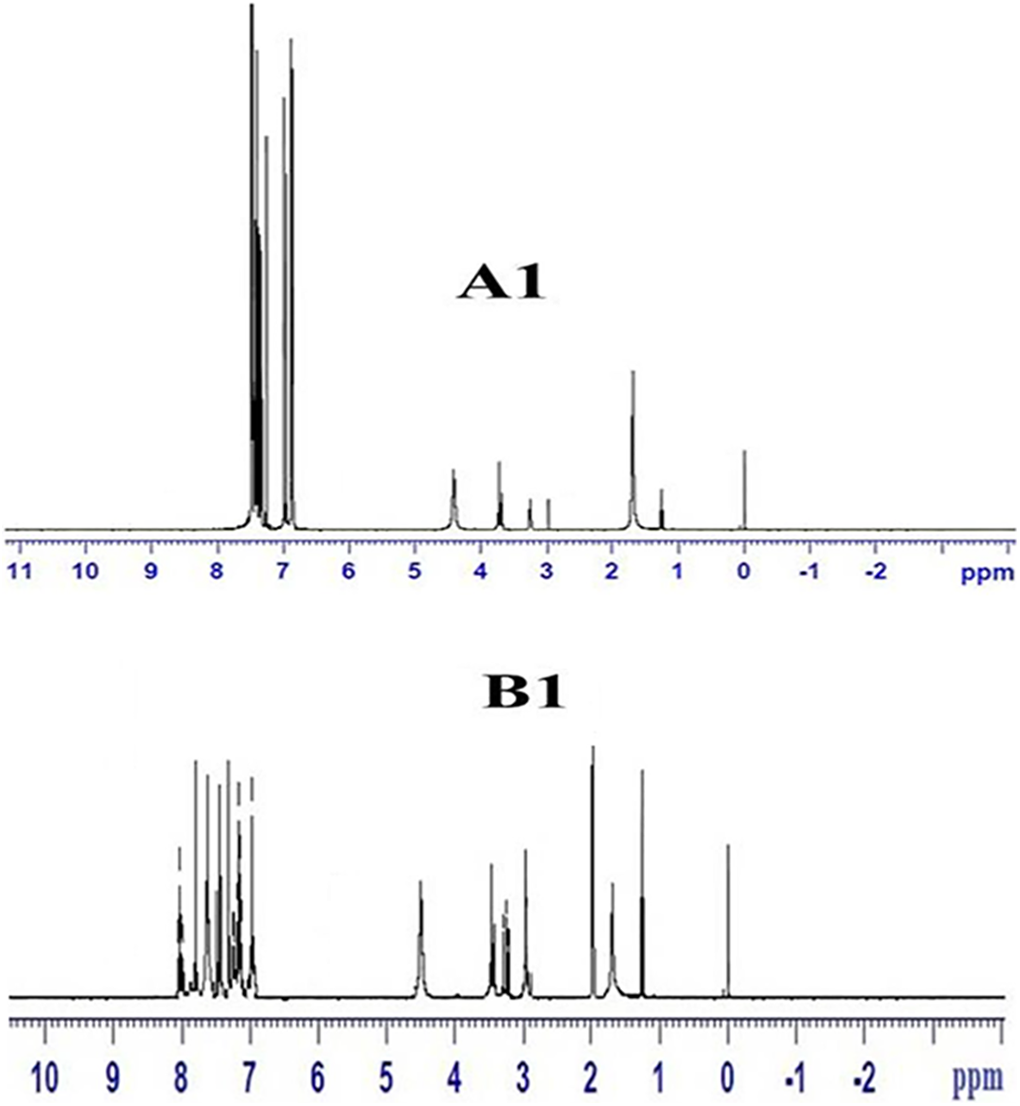
1H NMR spectrum of the copolymers A1 and B1.
Molecular weight measurements
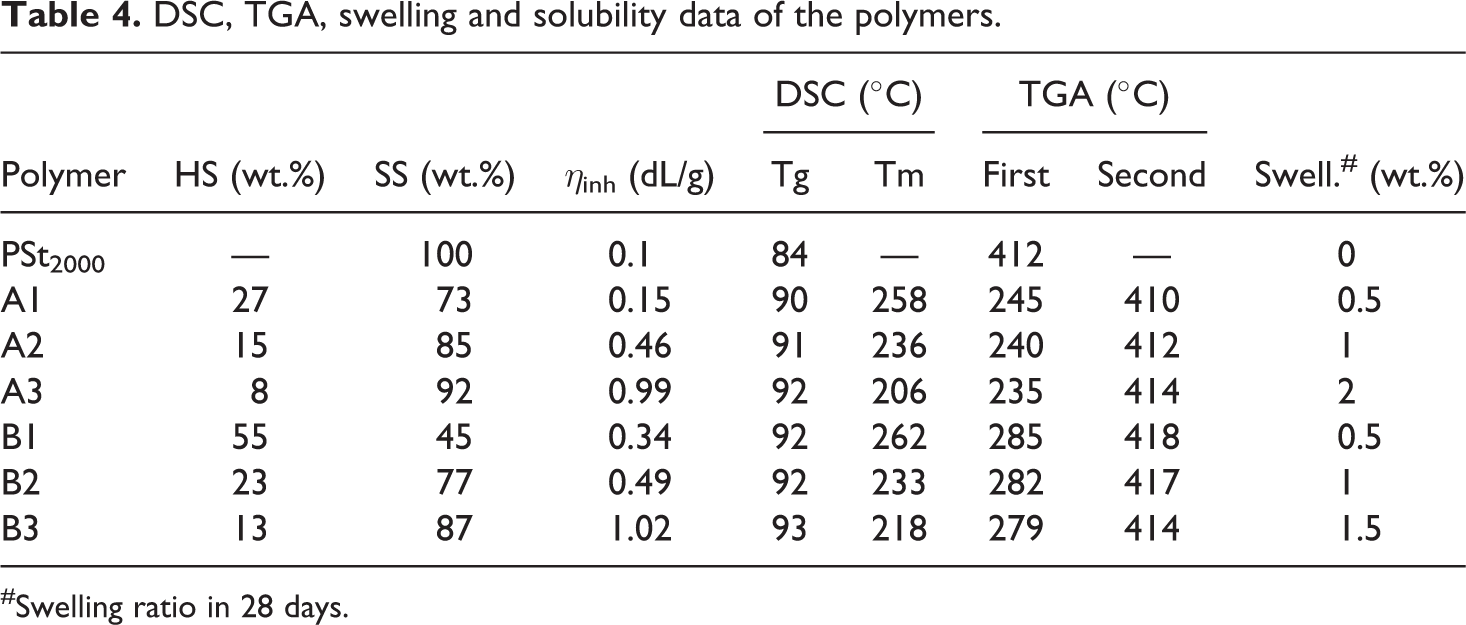
It is hard to find the molecular weight of the synthesized polymer by using GPC due to its hydrogen bond forming existence, the inherent viscosity (ηinh) approach was used to determine the molecular weight. A 1:1 molar mixture of phenol/1,1,2,2 tetrachloroethane is used as a solvent for this purpose. The ηinh value of hydroxyl terminated polystyrene is 0.1 dL/g. Inherent viscosity of the synthesized copolymer A1, A2, A3, B1, B2 and B3 are given in the Table 1. Copolymer A3 and B3 have the highest intrinsic viscosity values of 0.99 and 1.02(dL/g), respectively, compared to the other copolymers. The presence of soft segment length in the polymer chain affects the molecular weight of the polymer; this is reflected in the Figure 3. The molecular weight of the prepared triblock copolymer is linearly increasing with the increase in the soft segment length.
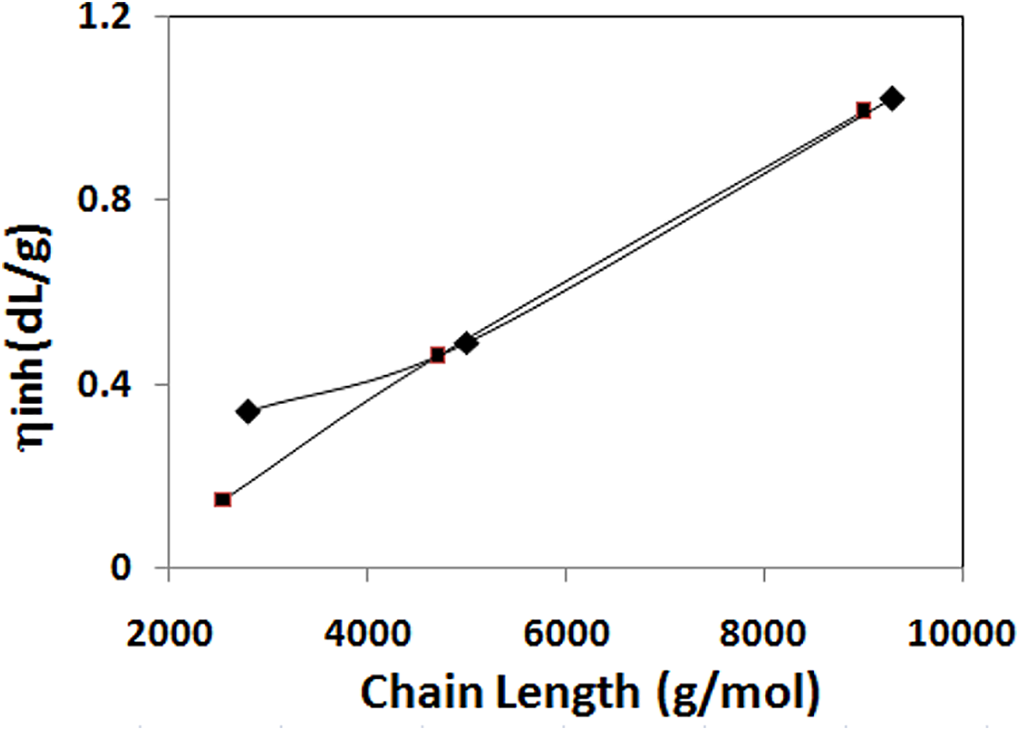
Effect of chain length on the molecular weight of the polymer: ▪, 6B; ♦, 6T6B.
FT-IR spectra
FT-IR is the powerful tool to study the hydrogen bonding in PUUA systems. The C=O and N-H group absorption value depend on the extent of hydrogen bond formation. Three carbonyl groups present in our system, namely, urethane, urea and amide which can form hydrogen bonds.25–27 The FT-IR spectra of the synthesized polymer at normal temperature is given in the Figure 4. The peak at 3310–3330 cm−1 is due to crystalline N-H groups. The carbonyl group in the amorphous urethane appears around 1740 cm−1 and the carbonyl of the crystalline urethane appear around 1710 cm−1. The carbonyl group of the free amide and the crystalline urea (in the monodentate crystalline form) is appearing at 1685–1667 cm−1. The hydrogen bonded amide group shows the absorption around 1625 cm−1. The peak appeared at 2917 and 2852 cm−1 is corresponds to asymmetric –CH2 and symmetric –CH2 stretching. Temperature will alter the properties of polyurethane-urea-amide and hence the effect of temperature on the crystallinity of the hard segment is found out by in situ FT-IR technique. The research was conducted at temperatures ranging from 30°C to 120°C. The height of the peak about 1630 cm−1 steadily decreases as the temperature rises; this decrease can be seen even as the temperature rises by 10°C. The peak intensity continues to decrease up to the tested high temperature of 120°C, indicating that as the temperature rises, the hydrogen bonds in the synthesized polymer chain gradually break. The gradual formation of hydrogen bonds occurs when the temperature is reduced from 120°C to 30°C. The cooling curve of the temperature dependent FT-IR spectra of A1 and B1 are given in the Figure 5. A carbonyl group of the hydrogen bonded urethane stretching appears at 1735 cm−1 and utmost intensity also observed in this region due to loss of electron density for the formation of hydrogen bonds.15,23 The peak around 1745 cm−1 is corresponds to non-bonded urethane carbonyl stretching. The stretching at 1710 cm−1 and at 1660–1690 cm−1 corresponds to amorphous amide carbonyl and hydrogen bonded urea carbonyl. The heating and cooling curves of the polymers showed an isosbestic point, which clearly proves that there is two types of the absorbing species in the copolymer chain.28,29 The two absorbing species are hydrogen bonded and nonhydrogen bonded hard segment.
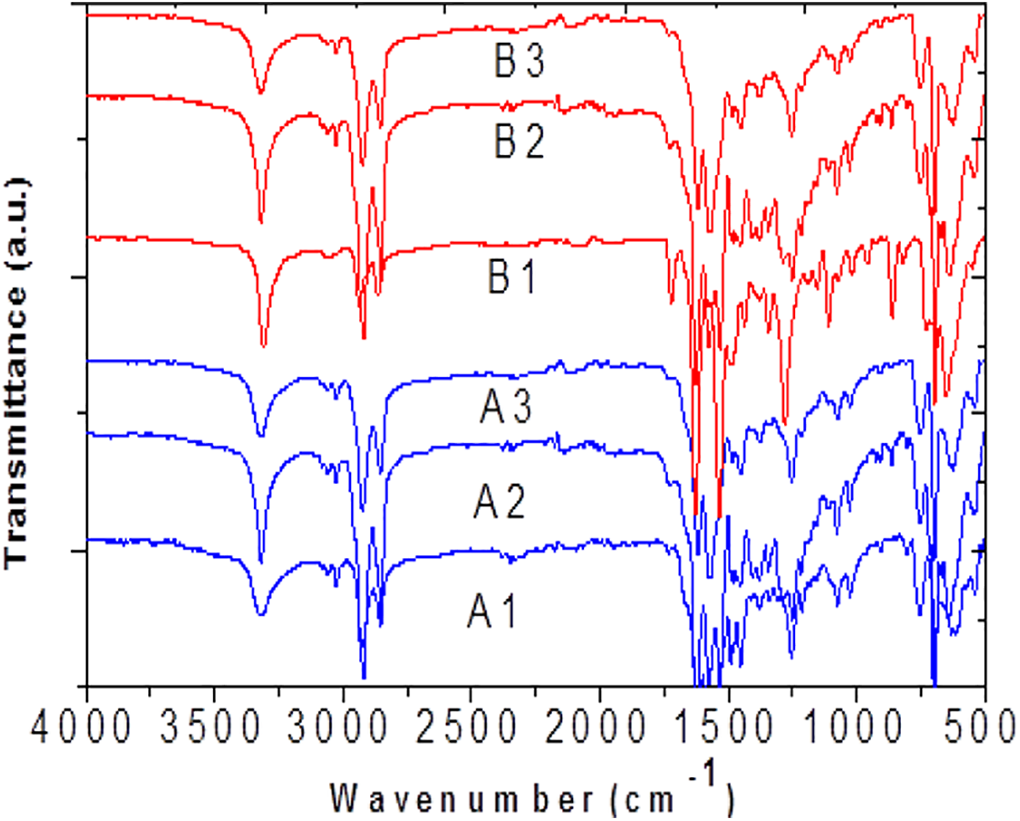
FT-IR spectrum of the six copolymers at room temperature.
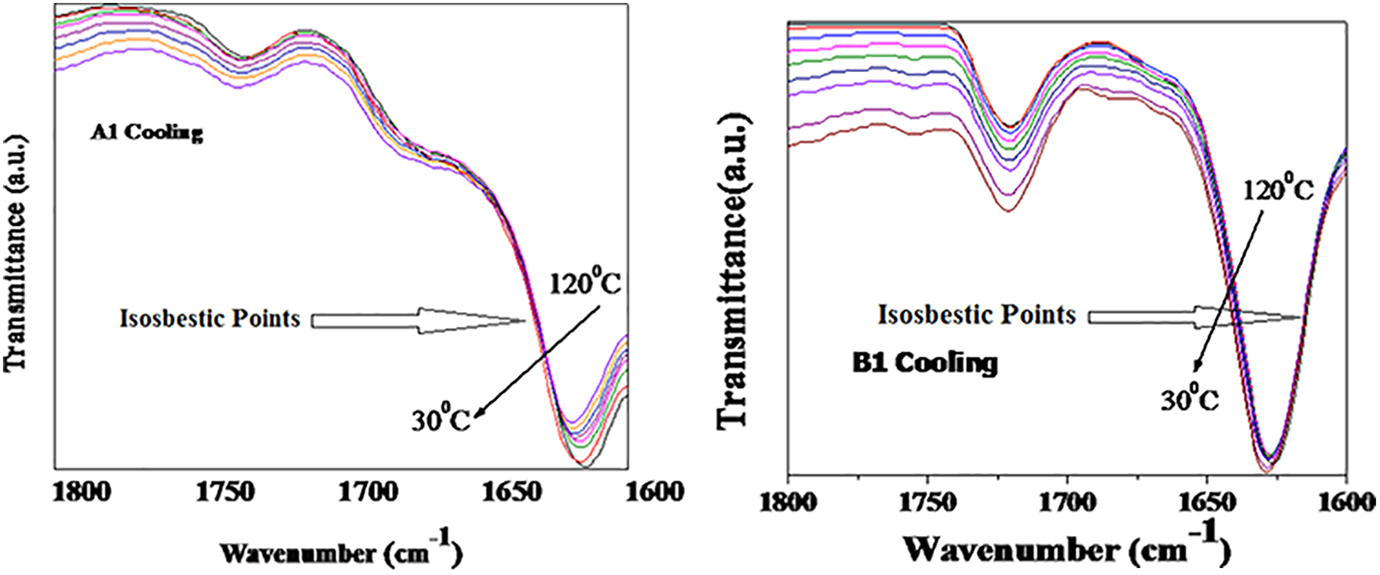
Temperature dependent FTIR spectrum Cooling curve of copolymer A1 and B1 at different temperatures.
The crystallinity of the synthesized thermoplastic elastomers varies with temperature and the copolymer’s molecular mass. Figure 6 shows the shift in crystallinity of A1, A2, A3, B1, B2, and B3 as a function of temperature. The transition in crystallinity is determined from 30°C to 120°C; crystallinity is partly affected at higher temperatures, and crystallinity is reversed when the temperature is lowered from 120°C to 30°C. Figure 7 shows a schematic diagram depicting the impact of temperature on the crystallinity of the hard part. At higher temperatures, the crystallinity decreases, and as the temperature drops, the crystallinity returns to its original state.
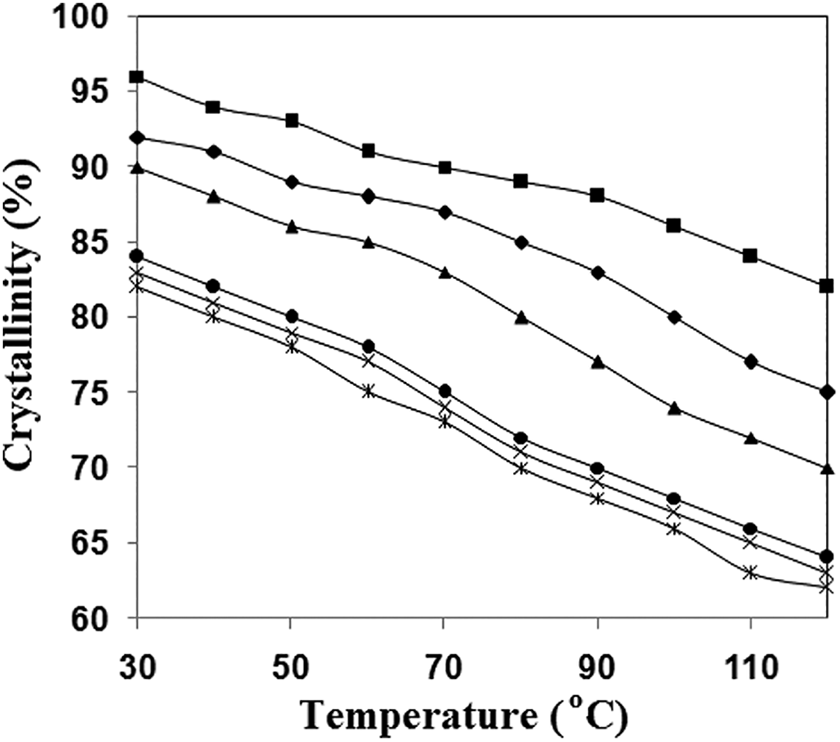
Effect of temperature on the crystallinity of the polymer series; •, A1; X, A2; х, A3; ▪, B1; ♦, B2; ▴, B3 (crystallinity is calculated using the cooling curve of FTIR).
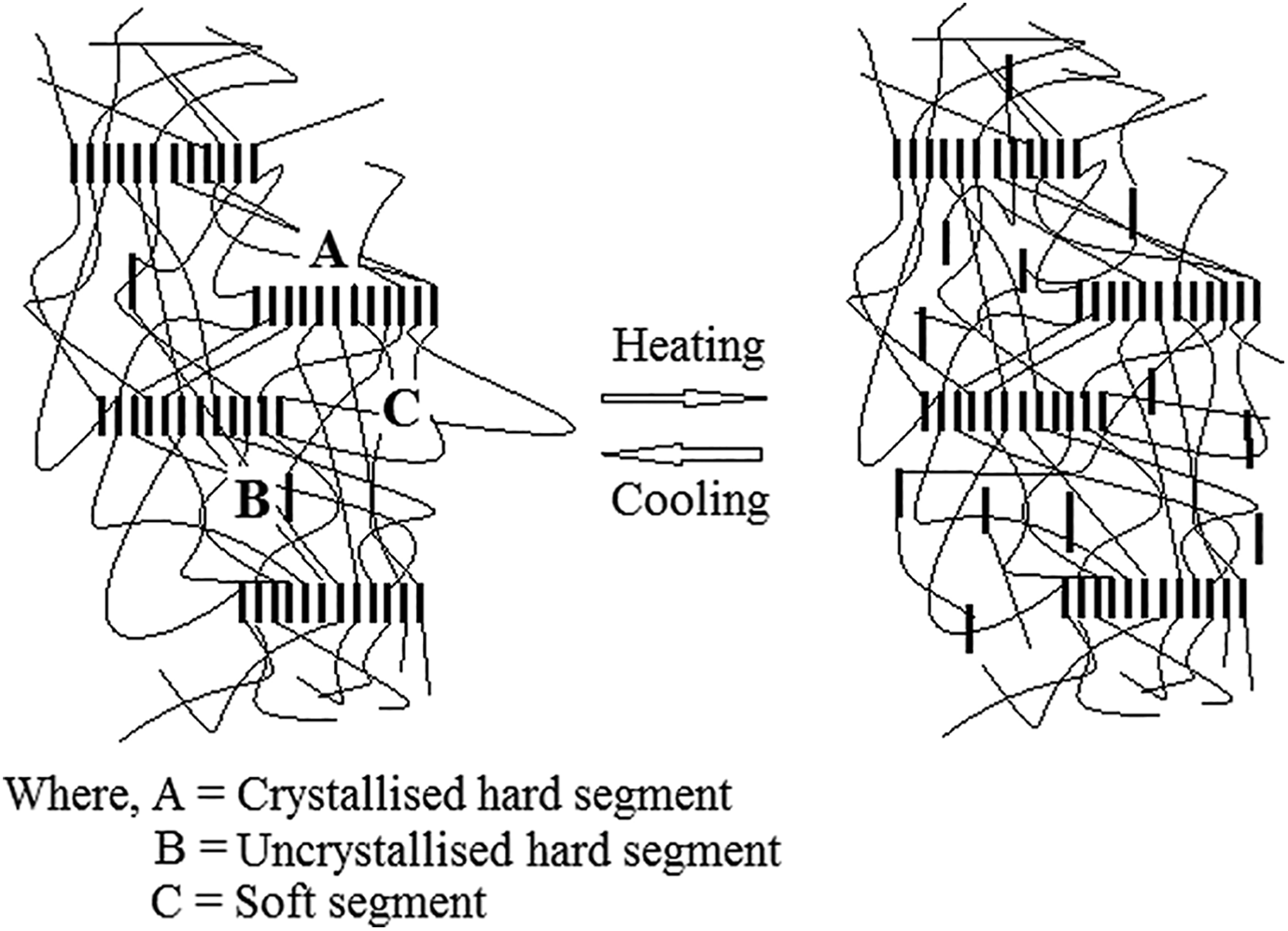
Schematic diagram representing the effect of temperature on the synthesized TPE.
XRD analysis
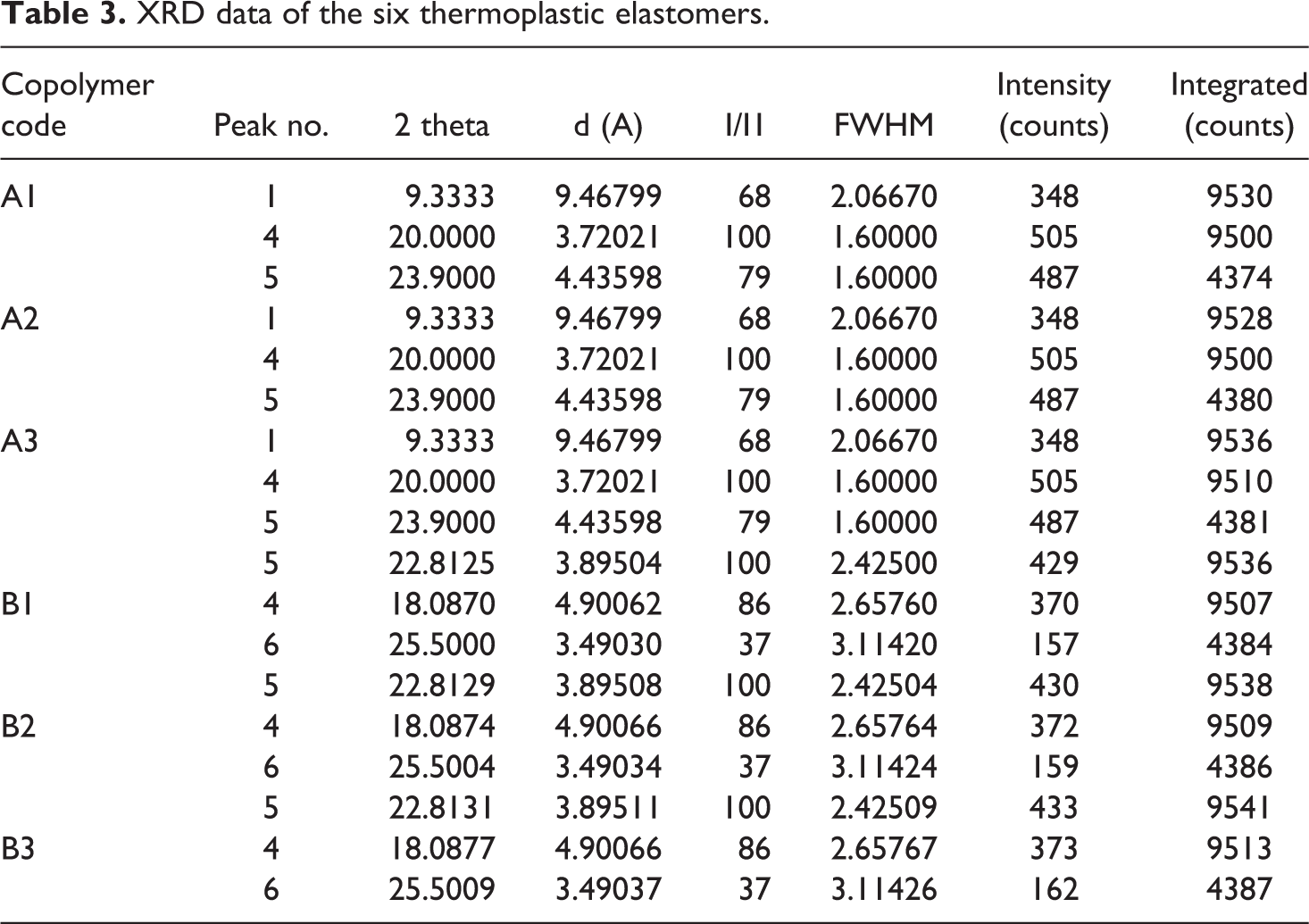
Figure 8 shows the XRD spectrum of the copolymers A1 and B1, with obtained values mentioned in Table 2, revealing three major peaks at 2–9, 20 and 23 for 6B and 18, 22 and 25 for 6T6B segmented copolymer. These values confirm that the synthesized copolymer is in the semi-crystalline form due to the presence of amorphous soft segment polystyrene present in the polymer chain. The XRD pattern clearly shows that all of the synthesized copolymers are both amorphous and semi-crystalline. The XRD pattern of all the copolymer doesn’t have a sharp peak, but there is a major three broad peak appear at same 2θ value for copolymer but the height of the peak is not the same. The height of the peak is gradually increasing from A1 to A3 and B1 to B3, this might be due to the varied hard segment concentration in the polymer network. The presence of hard segment 6B or 6T6B is responsible for the crystalline region present in the polymer chain, this kind of XRD pattern also observed in reported PUU polymer.23,30,31 The hard segment does not change the polystyrene’s amorphous nature to crystalline, but it may change the nature of the polymer chain, i.e. the polymer chain is not entirely amorphous. The presence of a higher amount of hard segment in the polymer network does not enhance more crystalline region due to the uniform hard segment used in the polymer preparation, as shown by the nearly identical XRD pattern obtained for prepared 6B or 6T6B segmented copolymer. The copolymers A3 and B3 have the highest hard segment concentration of all the copolymers, but their 2θ value (Table 2) varies little with A1 and A2 and B1 and B2.
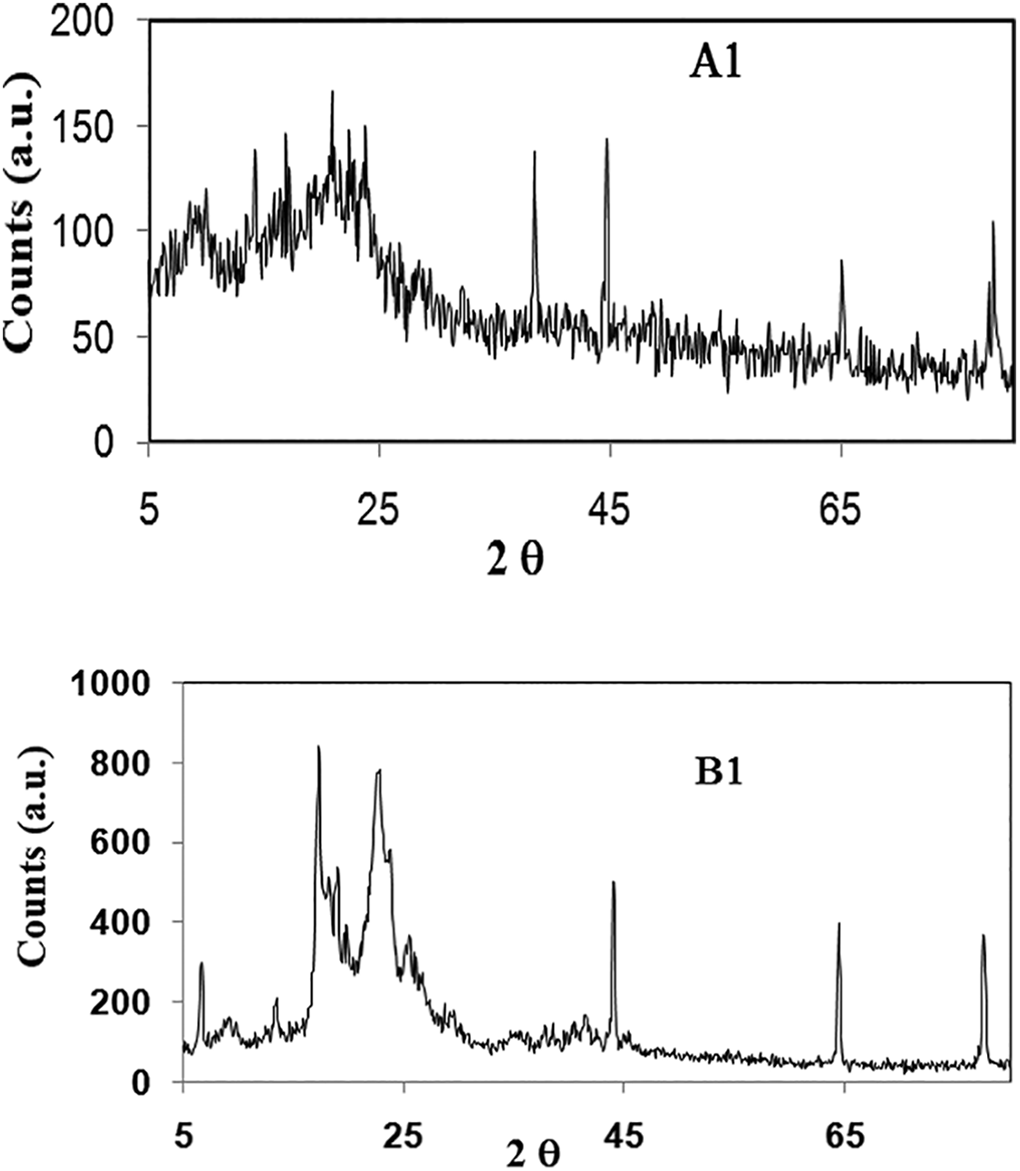
WAXS at room temperature of polymer A1 and B1.
FT-IR data of the multiblock copolymer at room temperature.
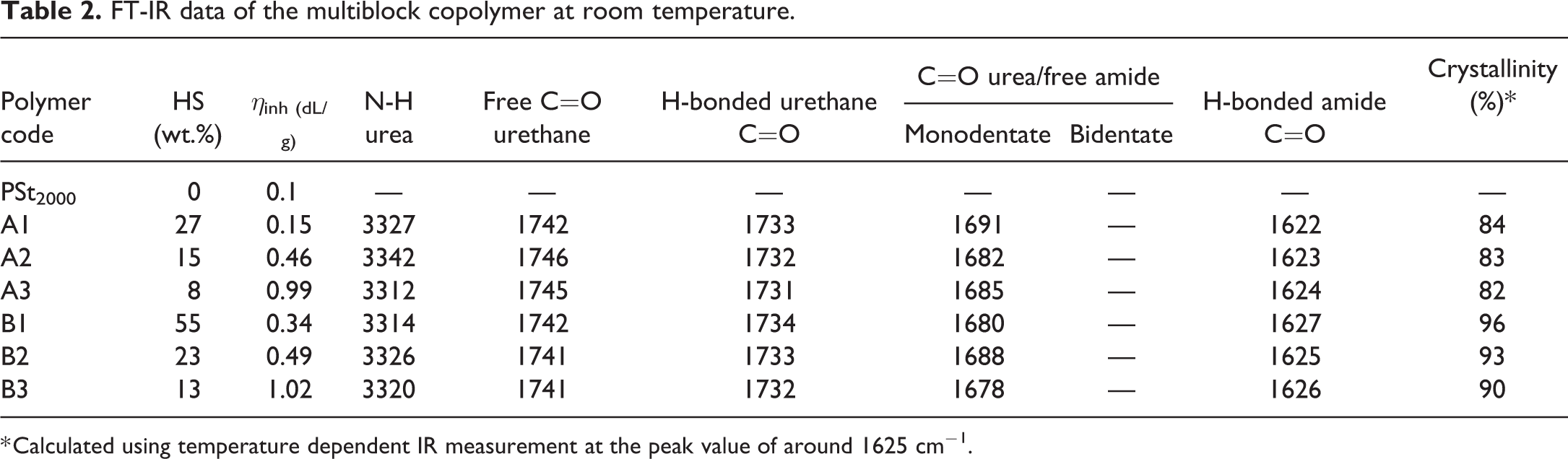
* Calculated using temperature dependent IR measurement at the peak value of around 1625 cm−1.
DSC
The DSC thermogram of the synthesized copolymer A3 and B3 are present in the Figure 9 as a representative of the synthesized series and obtained values are presented in the Table 3. The heating curve of the synthesized PUUA elastomers reveals two characteristic peaks, first peak shows Tg of soft phase and second peak corresponds to melting endothermic of hard segment. The presence of hard segments in the polymer chain influences the melting endotherm. The hard segment concentration affects the Tg of the synthesized polymer. Tg of A1, A2, A3 is 90°C, 91°C and 92°C respectively. Similarly, for the B series, the Tg values are 92°C, 92°C and 93°C for B1, B2 and B3 respectively. This clearly reveals the relationship between the hard segment content on the Tg of the soft segmented. Tg of the soft segment is increasing slightly with increasing the hard segment content in the copolymer. A small variation in the Tg upon increasing the hard segment content proves the complete crystallization of hard segment leaving behind the pure soft phase. The hard segment is little mixed with the soft phase. The melting temperature of the synthesized copolymer A1 is 258 whereas Tm of A2 and A3 is lesser than the Tm of A1. The melting temperature of B1 is 262 whereas B2 and B3 is 233 and 218 respectively. This shows the presence of soft segments content influence the Tm of polymer. As the concentration of the hard segment increases, the concentration of the soft segment decreases, thereby increasing the Tm of the polymer. This kind of change was absorbed commonly in segmented PUU-copolymer. 32 According to the heating curve, the cooling curve of the polymer reveals two peaks, one for hard segment crystallization and the other for soft segment Tg. The temperature difference between the melting and the crystallization of HS are close to the range of around 30°C, which shows the fast crystallization of hard segment.
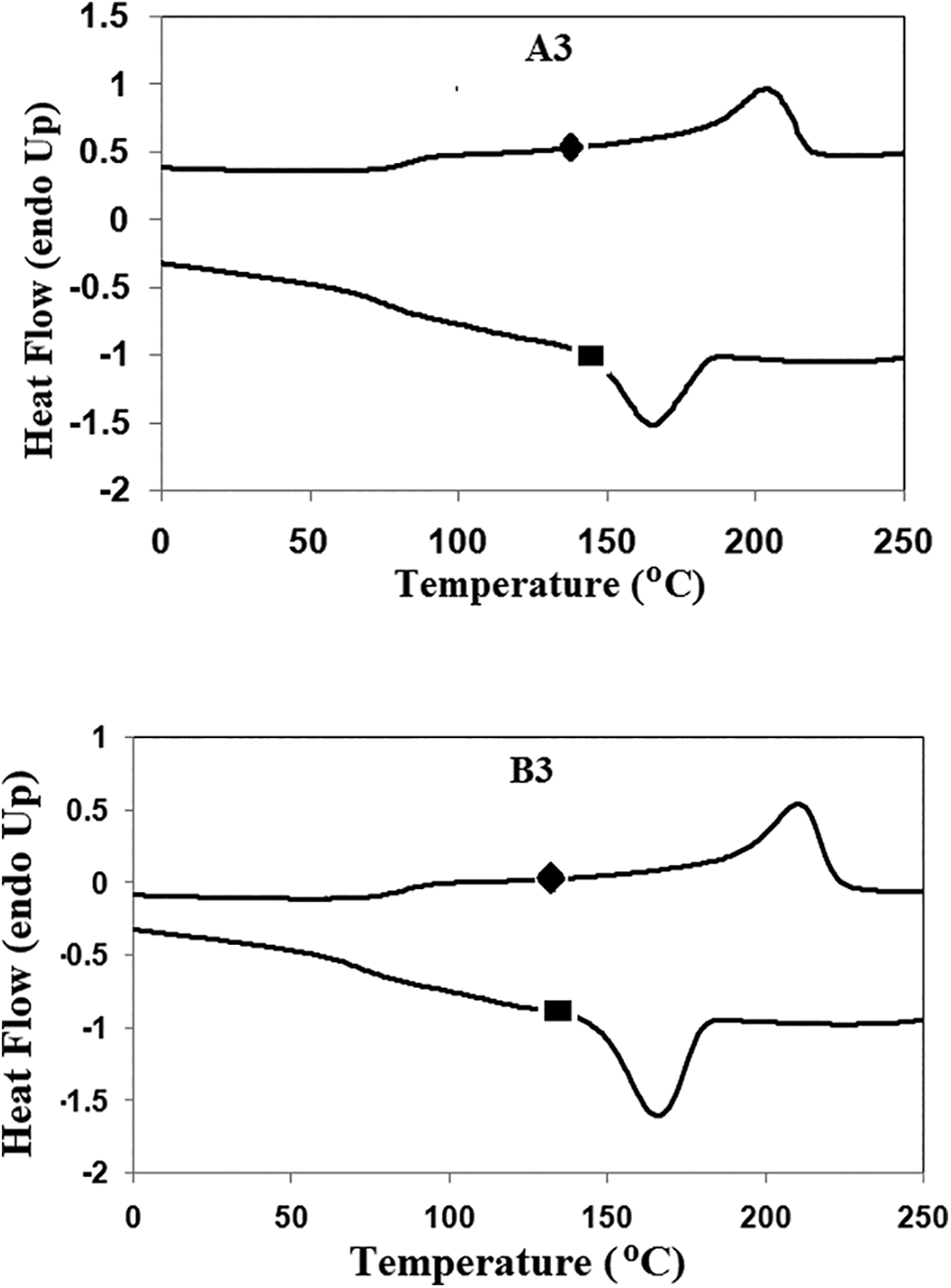
DSC heating curve (♦) and cooling curve (▪) of the polymer A3 and B3.
XRD data of the six thermoplastic elastomers.
TGA
The obtained TGA curve of the copolymer A1 and B1 is present in the Figure 10 and the obtained data are given in the Table 3. Similar type of the TGA curve is obtained for the other copolymer studied. All the materials showed two stage decomposition temperatures, first and second decomposition stage at 290°C and 400°C respectively. Polymer completely decomposed, above 400°C, hard segment content is responsible for the slight increase in the decomposition temperature of A1 and B1.
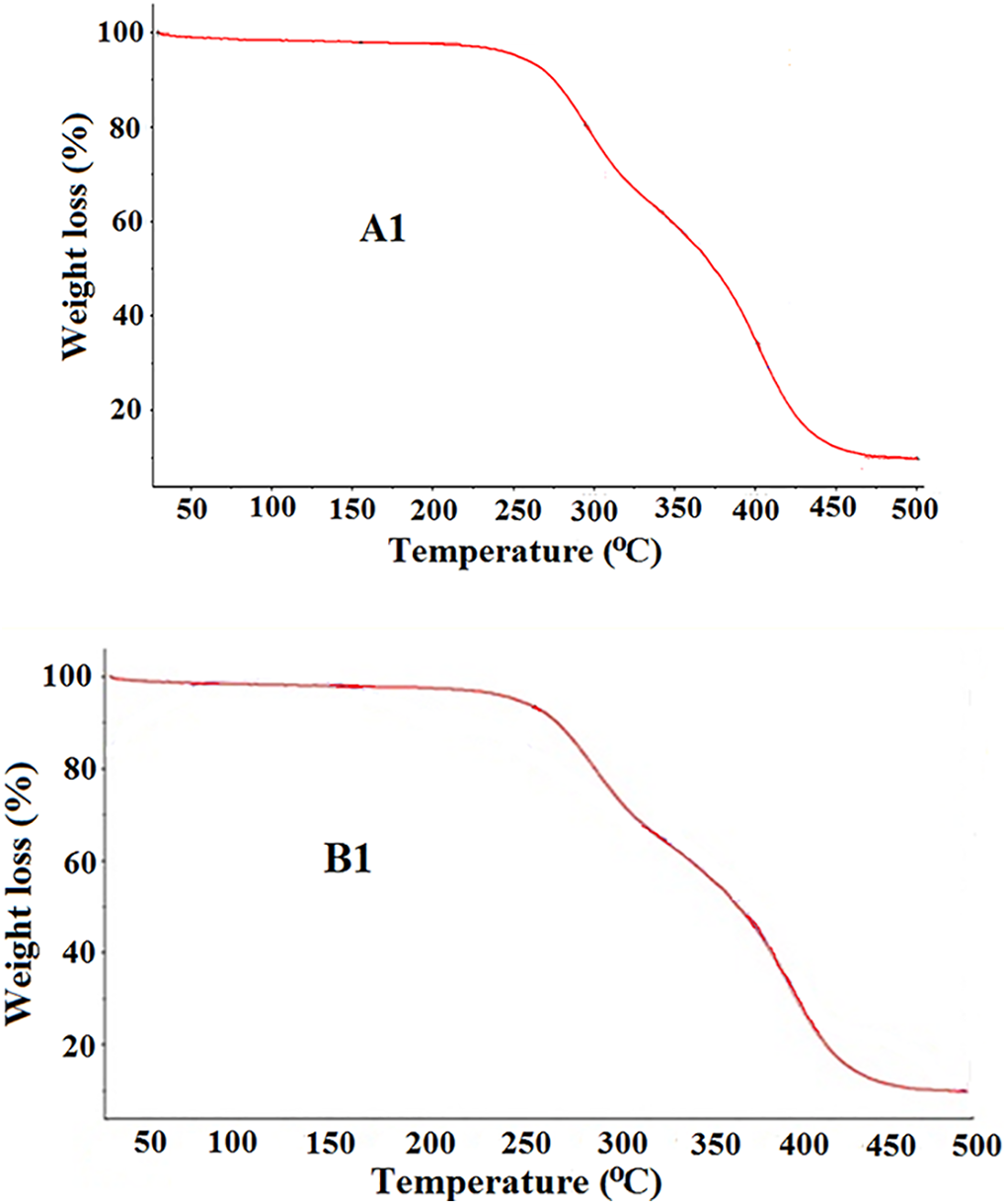
TGA thermogram of the copolymers A1 and B1.
Swelling ratio of polymer
The swelling behavior of the synthesized copolymer A1, A2, A3, B1, B2 and B3 is found and given in Table 4. Polymers are semi-crystalline in nature, hence undergoes physical crosslinks between the chain, but the percentage of crosslinks depends on the 6B and 6T6B hard segments used for the preparation of segmented copolymer. A polymer containing higher hard segment content shows less swelling behavior. The effect of HS content on the swelling ratio confirms that the swelling ratio increases with a decrease in the HS content on the copolymer.
DSC, TGA, swelling and solubility data of the polymers.
#Swelling ratio in 28 days.
Solvent resistivity
Solvent resistant of the triblock copolymer is measured in different solvents and the values are presented in the Table 5. The prepolymer, polystyrene is freely soluble in all tested solvents, whereas the triblock copolymers are seldom soluble. The drastic decrease in the solubility of the synthesized polymers is due to the presence of a crystallized amide segment in the copolymer chain. The crystallized amides are acting as a barrier for the solvent to attack the soft segment thereby acting as a physical crosslinks. The obtained solvent resistivity value is higher than or equal to the reported value. 31
Solubility data (%) of the copolymers at room temperature.
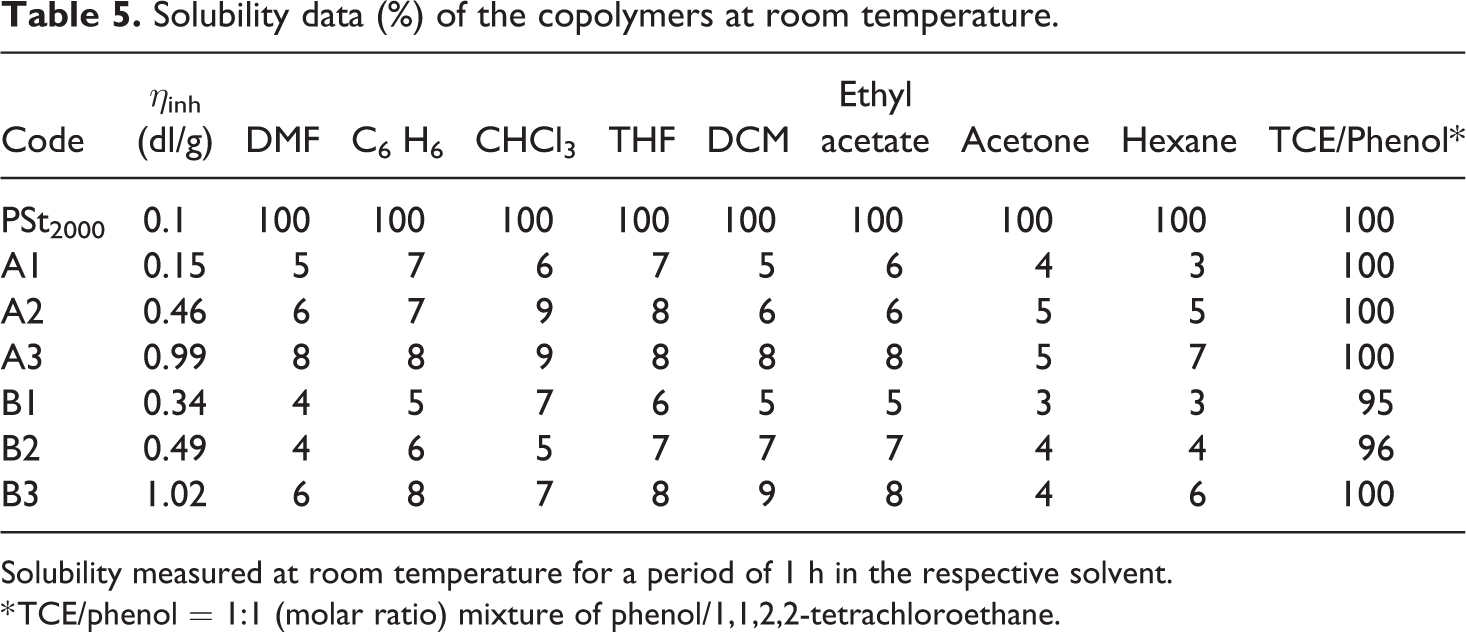
Solubility measured at room temperature for a period of 1 h in the respective solvent.
* TCE/phenol = 1:1 (molar ratio) mixture of phenol/1,1,2,2-tetrachloroethane.
Conclusion
PUUA is synthesized using polystyrene with the Mw value of 2100 g/mol, HMDI along with chain extender, 6-hexylamino benzamide or n-(aminohexyl)-n-6-benzoylaminohexyl hexylterephthalamide(6T6B). The different mole ratio of synthesized soft and hard segment used for the preparation of segmented copolymer. Crystallinity of the A1 and B1 is high, which contains higher hard segment content. Tg, Tc and Tm of the polymer depends on the hard and the soft segment ratio in the polymer chain. Solubility of the soft segment is lowered by introducing the hard segment. Temperature dependent FT-IR analysis of the prepared copolymer clearly pictured out the hydrogen bond present in the polymer chain. The presence of hydrogen bond in the polymer chain is temperature dependent; the entire polymer shows a higher number of hydrogen bonds at higher temperature. Crystallinity of the polymer gets reversed on cooling the polymer sample, this property of the polymer also confirmed by the FT-IR analysis. The XRD curve also reveals that the polymer is semi-crystalline in nature. The triblock copolymer showed two stage decomposition temperatures. The swelling behavior and solubility of the polymer is increasing with increase in soft segment content in the copolymer. The polymer showed excellent solvent resistivity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
