Abstract
Biodegradable polyurethane with excellent mechanical property finds a lot of applications in the biomedical field. In this study, multi block copolymer poly(urethane-urea-amide) (PUUA) is prepared using polyethylene oxide, 1,6-hexamethylene diisocyanate and diamide (N1, N4-bis(6-aminohexyl)benzene-1,4-dicarboxamide (6T6) or
Introduction
In a recent year, polymer materials used in important applications such as biodegradable, engineering materials, etc. Conventional polymers such as natural rubber, polystyrene were replaced today with segmental block copolymers. The segmented block copolymers have different chemical entities in its chain and have excellent properties. Segmented block copolymers generally consist of a flexible soft and crystallisable hard segments. The phase separation takes place due to the presence of flexible soft segments and partly crystallized hard segments.1,2 The phase separation (in solids) could be facilitated when the segmented block copolymers using monodispersed crystallizable hard segments.2–4 The monodispersed hard segment can crystallize fast and completely. Aliphatic diisocyanates are the main source for prepare polyurethane and it is already being used as
Experimental section
Materials
1,6-Hexamethylene diisocyanate, deuterated triflouroacetic acid (TFA-d), 1,1,2,2 tetrachloroethane and phenol, polyethylene glycol (1000 g/mol), Dimethyl Acetamide (DMAc) and Dibutyltin dilaurate (DBTDL) were purchased from Aldrich Chemicals. The hard segments, 6A6 and 6T6 are synthesized by the known route. 16
Characterization
Bruker FT-NMR 320 MHz instrument used for measuring 1H NMR spectra using TFA-d as a solvent. Alpha Bruker FT-IR instrument along with alpha-T heating accessory (between 30°C to 120°C) having resolution of 4 cm−1 is used for recording FT-IR spectrum. The sample preparation and the calculation presented in our earlier report. 17 Molecular weight of the polymer is evaluated using inherent viscosity methods at 0.1 dL/g concentration using the mixture of (1:1 molar ratio) phenol/ 1,1,2,2-tetrachloroethane as solvent at room temperature. Perkin Elmer DSC 7 instrument has PE 7770 computer and TAS-7 software is used for measuring DSC spectra of the polymer. 10–15 mg of the sample heated at the rate of 20°C/min under nitrogen atmosphere. Tm and Tc were obtained from the second heating and first cooling curve of the DSC graph respectively. Dupont 951 instrument used for measuring thermo gravimetric analysis (TGA). TGA recorded from 30 to 600°C using about 10 mg samples under nitrogen atmosphere condition. Stress-strain curves obtained using compression moulded bar using Pack Test UTM model KC-3000 fitted with 500 N load cell. The stain rate of the test is 0.4 s−1 equal to the speed of 60mm/min. Strain at 0.1–0.25% has been used to calculate E-modulus of the polymer.
Preparation of poly(urethane-urea-amide) based on 6A6
Polyethylene oxide (PEO) of average molecular weight 1000 g/mol (7.5 g, 0.015 mol) taken into a 100 ml RB flask fitted with a nitrogen inlet, mechanical stirrer and kept in an oil bath. The reaction carried by without usage of a solvent. The PEO melts at 40°C, when the temperature attained 40°C, 1,6-hexamethylene diisocyanate (2.52 g, 0.03 mol) added into the reaction medium. After adding 1,6-hexamethylene diisocyanate, the reaction mixture was stirred for 2 h, and then the temperature raised to150°C. After 2 h, 1.74 g (0.015 mol) of 6A6 dissolved in DMAC is then added. To this mixture, DBTDL added and the stirring continued for another 1 h. The solvent removed under vacuum and the polymer obtained is colorless rubbery material. 1H NMR (δ, ppm): 4.4 (t, CH2 connected to the oxygen present in the urethane), 3.8 (m, CH2 connected to the ether), 3.6(t, CH2 connected to amide), 3.2 (b, –C
Preparation of PUUA based on 6T6
Similar procedure which is given in the section 2.3 is adopted for preparing Poly(urethane-urea-amide) has 6T6 as a hard segment. PEO (7.5 g, 0.015 mol), 1,6-hexamethylene diisocyanate (2.52 g, 0.03 mol) and 6T6 (2.71 g, 0.015 mol) used for preparing PUUA-6T6 polymer. The obtained polymer is color less rubbery material. 1H NMR (δ, ppm): 8.0 (t, aromatic proton), 4.3 (t, CH2 connected to the oxygen present in the urethane), 3.8 (m, CH2 connected to the ether), 3.6(t, CH2 connected to amide), 3.3 (b, –C
Biodegradation of the polymer
Biodegradation studies carried out using a compression moulded bar of dimension 10 × 10 × 2 mm. A piece of the polymer sample dipped in pH 9.2 and pH 7.0 solution for 4 weeks. After the required time, the polymer sample dried in vacuum at 70°C for 24 h. The dried sample is then subjected to molecular weight determination using the inherent viscosity method. From the inherent viscosity of the samples before and after treatment, the biodegradable nature of the polymer studied.
Result and discussion
Polyurethane urea-amide based on two monodispersed hard segments (extender) namely, 6A6 and 6T6 synthesized and characterized. The reaction route of the PUUA is given in the Figure 1 and the mole ratio used for making the polymer is given in Table 1. The product formation confirmed by 1H NMR spectroscopic method using deuterated triflouroacetic acid as a solvent. The NMR spectrum of the PUUA-6A6 and PUUA-6T6 shown in the Figure 2 and Figure 3. Expected peak for the PUUA have seen in NMR spectrum and is accordance with the reported values of similar type of systems. 18
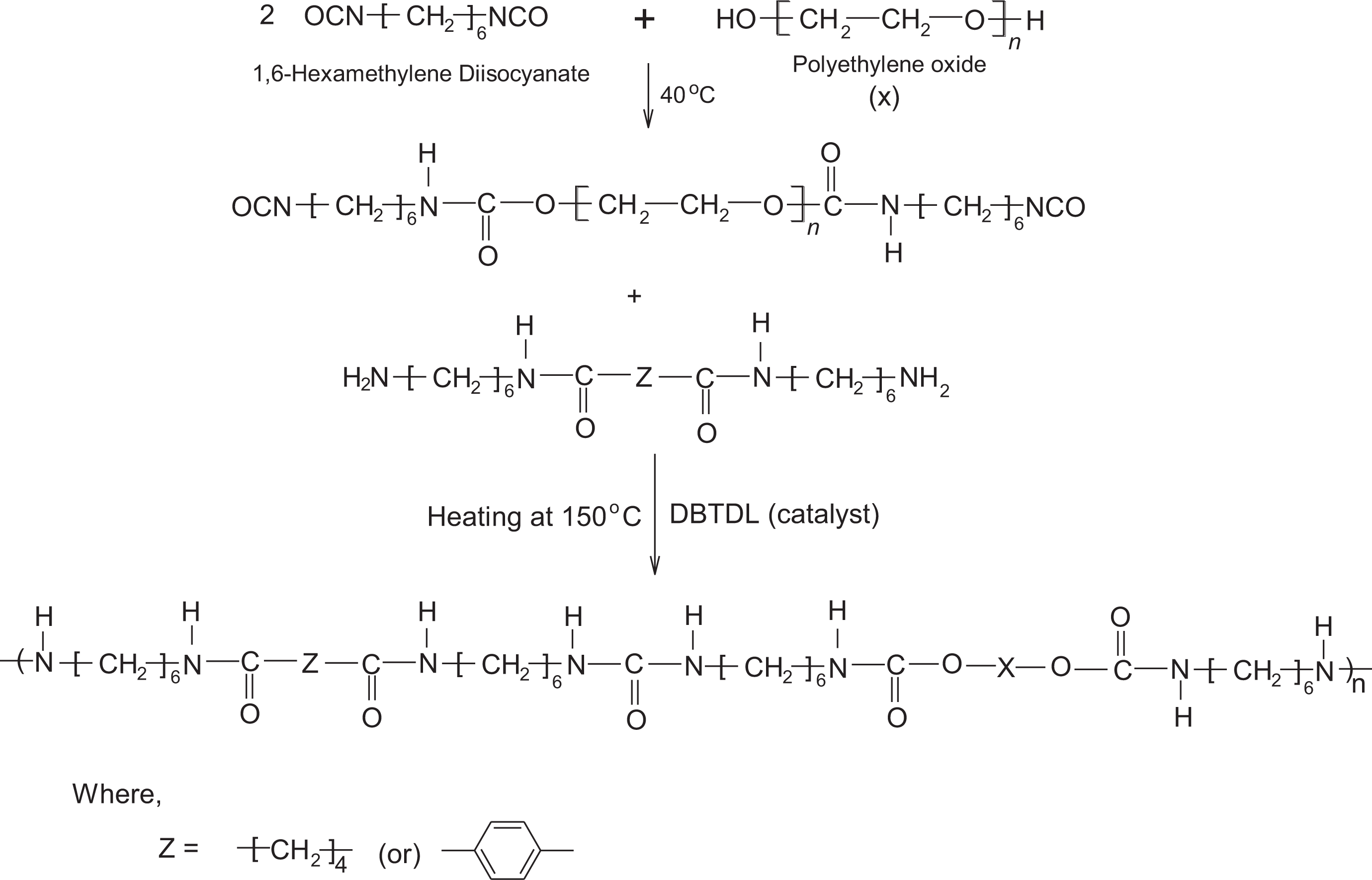
Synthetic route of PUUA.
Repeating length, mole fraction in feed and the inherent viscosities of the polymers.

* X = Adipate or Terephthalate.
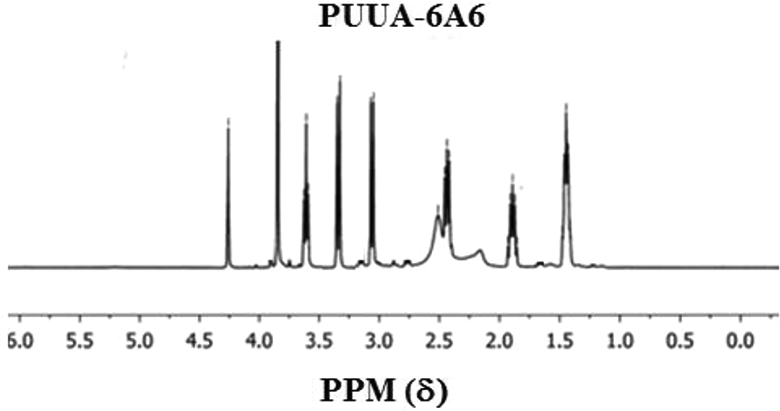
1H NMR spectrum of PUUA-6A6.
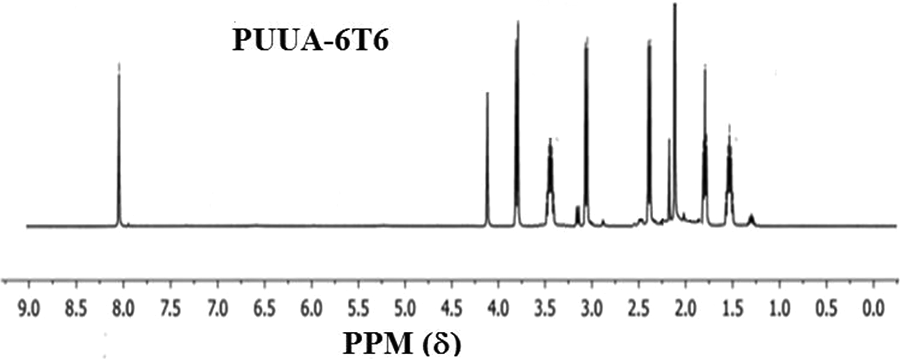
1H NMR spectrum of PUUA-6T6.
Molecular weight measurements
The PUUA copolymers can interact with the column used in the GPC instrument and the GPC analysis could not be done. The copolymer molecular weight can show by the inherent viscosity (ηinh) method in phenol/1,1,2,2-tetrachloroethane as a solvent. At a low concentration, the relative viscosity of the polymer related to the inherent viscosity. The inherent viscosity of PUUA-6A6 and PUUA-6T6 polymers is 2.51 and 2.76 dL/g (Table 1). The high inherent viscosity confirms the high molecular weight of the synthesized polymer. The inherent viscosity value of PUUA-6T6 is higher than PUUA-6A6. Both the polymers showed exceptionally high molecular weight, which further confirms the complete conversion of the monomers into polymers.
FT-IR spectra
The IR spectroscopy used to find the hydrogen bonding in polyurethanes and PUUA copolymers. The FT-IR spectroscopy of the PUUA shown in Figure 4 and the data is given in Table 2. The free and hydrogen bonded state of carbonyls groups absorbance reported in the literature.17–20 The peaks appear at 3303–3329 cm−1 is due crystalline N–H groups, at 1713–1719 cm−1 due to the C=O present in crystalline urethane, at 1623–1631 cm−1 and 1679–1693 cm−1 for crystalline and amorphous amide carbonyls respectively. The observed frequency at 1563 cm−1 is representing aromatic stretching. Asymmetric –CH2 stretching appeared at 2919 cm−1 and symmetric –CH2 stretching appears at 2867 cm−1.
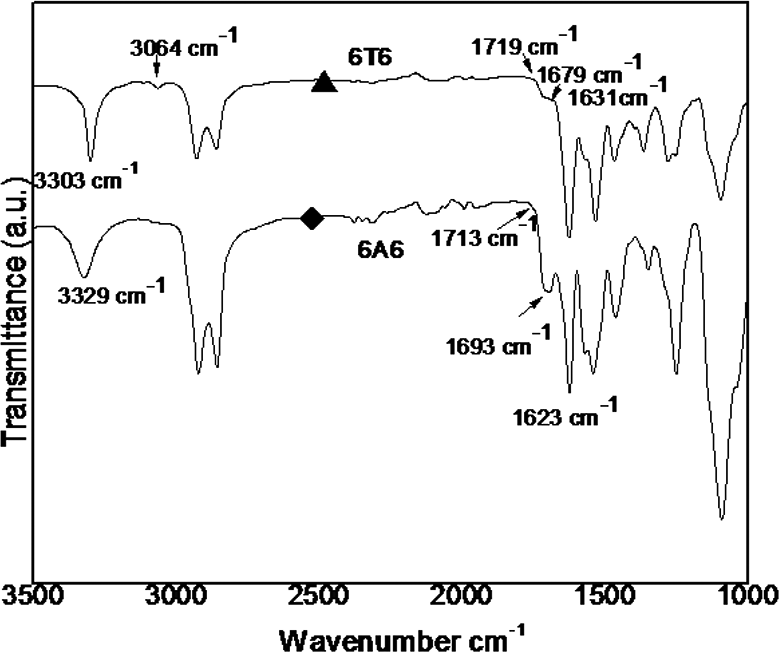
Room temperature of FT-IR spectrum of two copolymers.
Room temperature of FT-IR data of the PUUA copolymer.
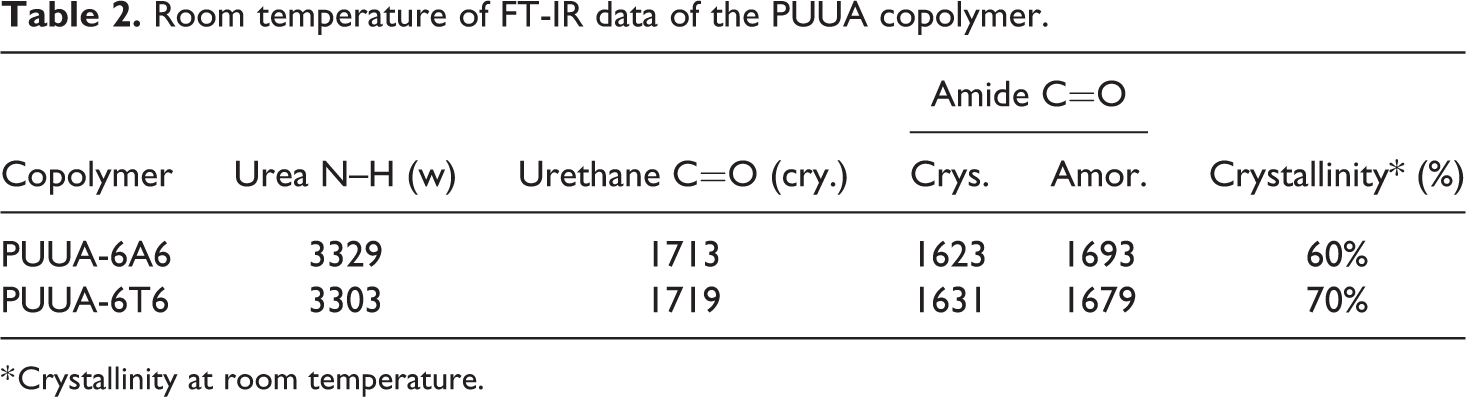
* Crystallinity at room temperature.
Due to the hydrogen bonding, the distance between the two carbonyl group of the amide unit present in the hard segments is less compared to the free carbonyl and is sensitive to the temperature. The distance between the hard segments is greater in the amorphous state compared to the crystalline state. In amide containing TPEs, calculation of the extent crystalline nature of the amide segment is found in the literature. 21 The absorption peak appears at 1630 (h(1630 cm−1)) and 1660–1670 (h(1670 cm−1)) will represent the crystalline and amorphous nature of the amide bond. The calculated crystallinity presented in the Table 2.
Temperature dependent infrared spectroscopy
An in situ temperature dependent FTIR spectrum is the powerful tool to study the effect of temperature on the crystalline nature of the HS. The temperature dependent spectrum of the PUUA-6T6 copolymer at various temperatures (40, 60, 80, 100 and 120°C) presented (40, 60, 80, 100 and 120°C) in Figure 5. In this graph increasing the temperature from 40 to 120°C, the crystallinity of polymer slightly decreases. The degree of crystallinity of the amide segments as a function of temperature for the PUUA-6T6 presented in the Figure 5. Crystallinity starts at 70% and decreases upon increasing the temperature to 120°C. Upon heating, intensity of amorphous amide peak increases, whereas intensity of crystalline amide decreases. Upon cooling, reverse situation takes place. The excellent crystallinity of the hard segment confirms the very least amount of hard segment dissolved in soft matrix. The crystalline nature of the hard segment creates the remarkable stability to the PUUA system under investigation. Figure 6 shows the effect of temperature on crystallinity of the studied PUUA. It clearly indicates that the hard segments crystallinity is reversible. At higher temperature, the distance between the hard segment increases and upon cooling, it shows closely packed crystalline structure. 22 These crystalline hard segments domains act as physical crosslink and therefore increase the polymer mechanical strength. The observed higher crystallinity of the hard segment is the effect of uniform crystallisable amide segments employed during the polymerization process. 21
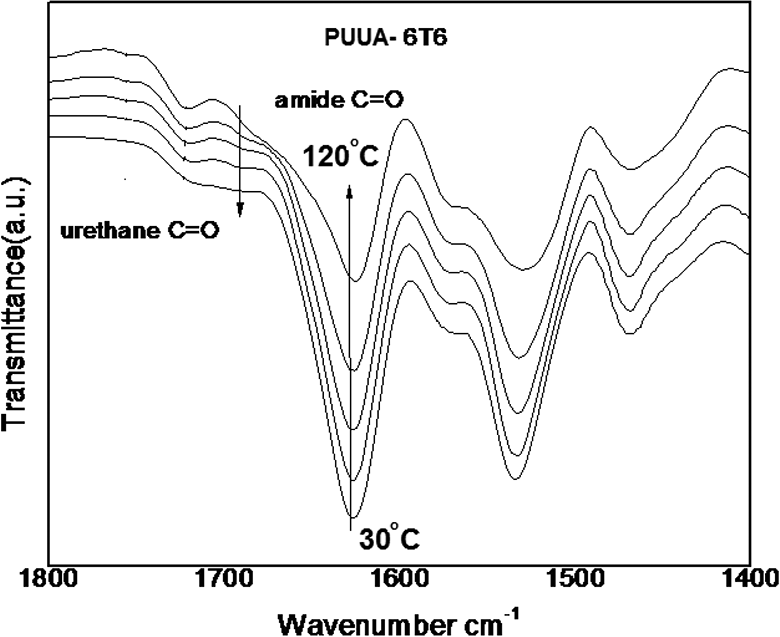
FT-IR spectra of PUUA-6T6 at different temperature.
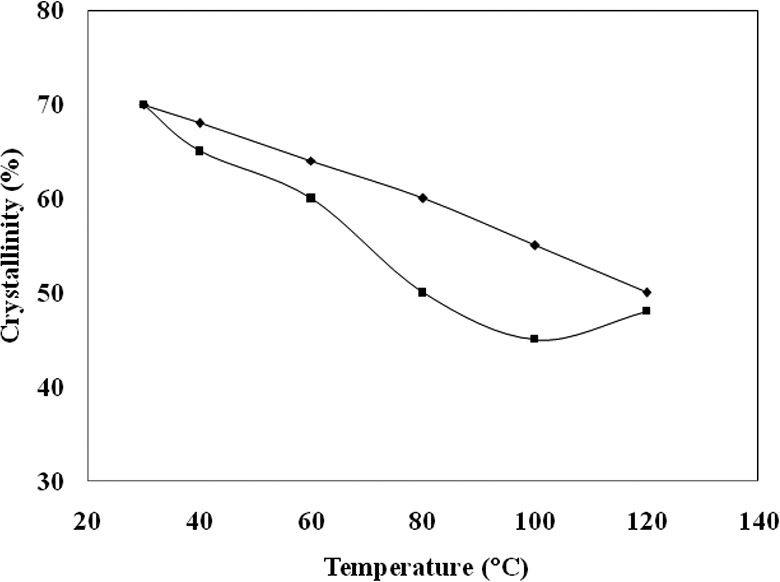
The effect of temperature on the crystallinity of PUUA-6T6 (♦;heating, ▪; cooling curve).
Thermal properties
DSC
DSC used to study the phase transition (crystalline or amorphous) of the constituent present in the polymer upon increase the temperature. The heating and cooling DSC spectrum of the PUUA-6A6 and PUUA-6T6 shown in Figure 7 and the results presented in Table 3. A classical thermoplastic materials, DSC obtained from our PUUA system. 23 The heating curve of both synthesized materials showed three distinct peaks around −45, 40 and 250°C represents the glass transition temperature SS (Tg) and the melting temperatures of the SS (Tms) and the melting of HS (Tmh). Similar to the heating curve, the cooling curve shows two distinct peaks around 0 and 220°C representing crystallization of the SS (Tcs) and crystallization of HS (Tch). In both the polymer system, the Tg of the PUUA is very close to the Tg of pure PEO which is −50°C. This remarkable value, suggesting that the hard segment employed in the synthesis of the PUUA is completely phased separated from the soft matrix leading to the pure PEO soft phase. When we compare the Tg value of PUUA-6A6 (−45.9°C) to that of the PUUA-6T6 (−38.6°C), in the former the crystallinity of hard segment is higher than the latter. However the observed Tg value considered as excellent for the PUUA systems which contain urethane linkage along with the amide linkage. The melting of soft segment appears at 41.9°C and 39.7°C for PUUA-6A6 and PUUA-6T6. The melting of soft segment is very close to each other also, the observed value is equal to the melting of soft segment PEO1000 which has the value of 40°C. The Tmh of PUUA-6A6 and PUUA-6T6 appears at 254 and 259°C. The Tm value depends on the concentration of the hard segment employed during the synthesis of PUUA. In both cases, the concentration of the hard segment is around 42% and the Tmh value is close to one another. The effect of type of HS, which is 6T6 or 6A6 on the crystallinity have little or no influence. The single melting point of HS with narrow window is due to the uniform sized HS employed during the polymerization process which produces uniform crystallite size.23–25
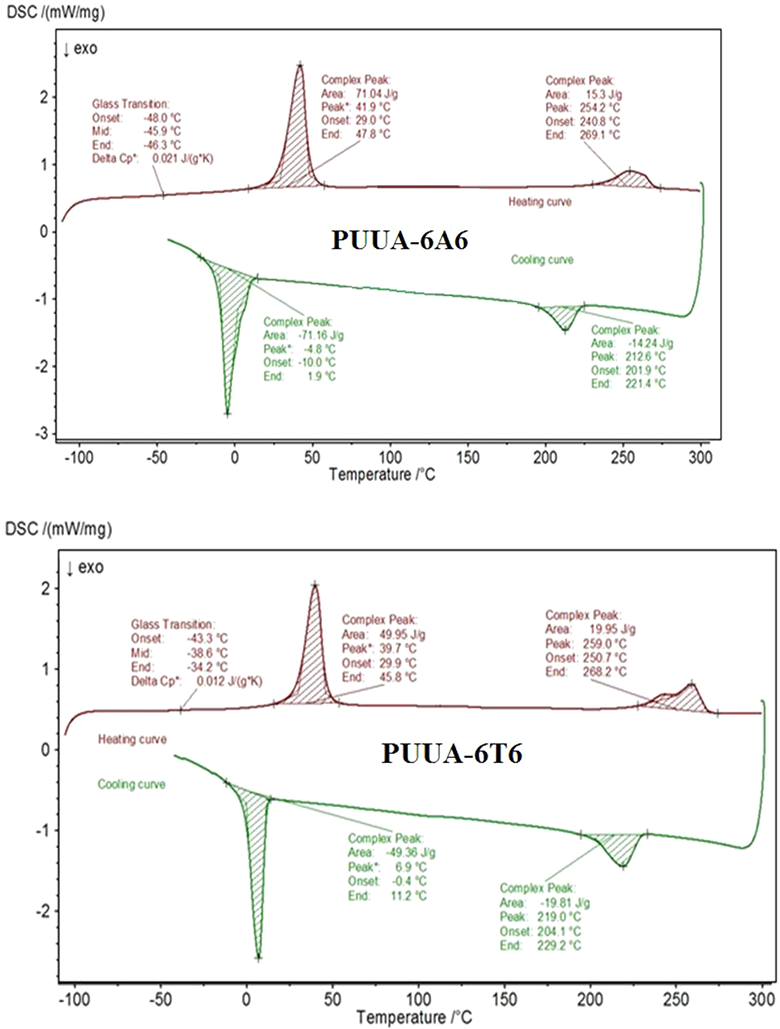
DSC heating and cooling curve of the PUUA.
DSC data for the PUUA copolymers.
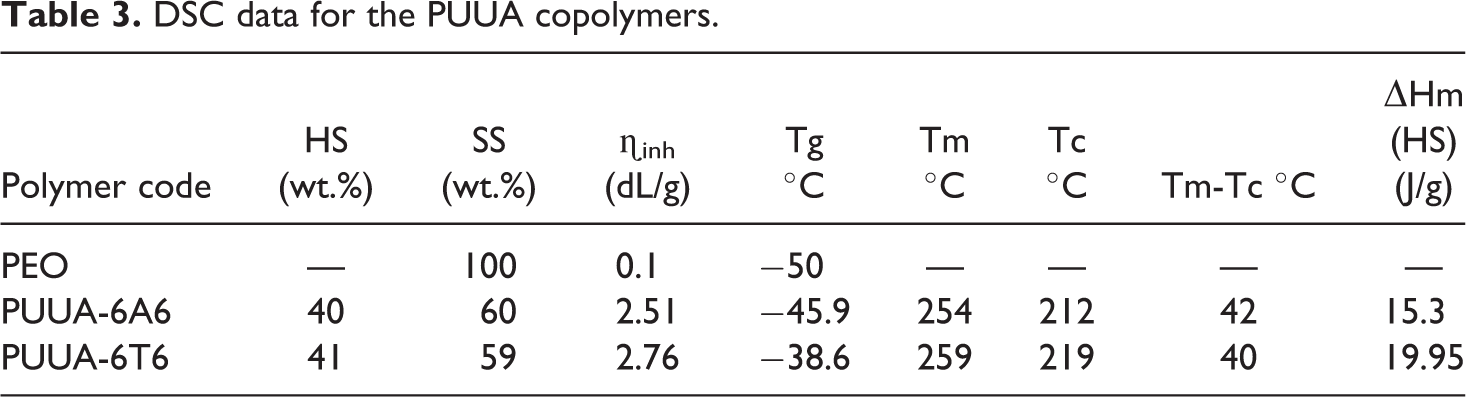
The Cooling curve of the PUUA showed two distinct peaks. In both the systems, the enthalpy of crystallization of soft segment (▵Hcs) is equal to the enthalpy of melting of the SS (▵Hms). This remarkable value, suggesting that the SS is completely phase separated from the HS. The mixing of hard and soft segment during melting and the phase separation during cooling is completely reversible without any phase mixing with each other. Similar to the SS, the enthalpy of melting and crystallization of the HS is similar confirms the complete crystallization of hard segment in the polymer when subjected to cooling. The complete crystallization of hard segment again due to the presence of the uniform crystallite size produced due to the uniform hard segment. The low under cooling value (Tmh-Tmc) further suggesting the hard segment crystallization is fast and complete. Generally the PU are seldom shown the crystallization peak upon cooling, but in our system, PUUA showed excellent crystallization peak with complete phase separation of hard from soft segment.
TGA
Thermal stability of the PUUA system studied using TGA analysis. The TGA curve of PUUA presented in Figure 8 and the data listed in Table 4. The TGA spectrum of the PUUA-6A6 confirms a single stage decomposition centered around 350°C and the PUUA-6T6 shows two stages decomposition. The first decomposition centered at 380°C and second decomposition centered around 500°C. The high first decomposition temperature of the PUUA confirms the high thermal stability of the synthesized polymers.
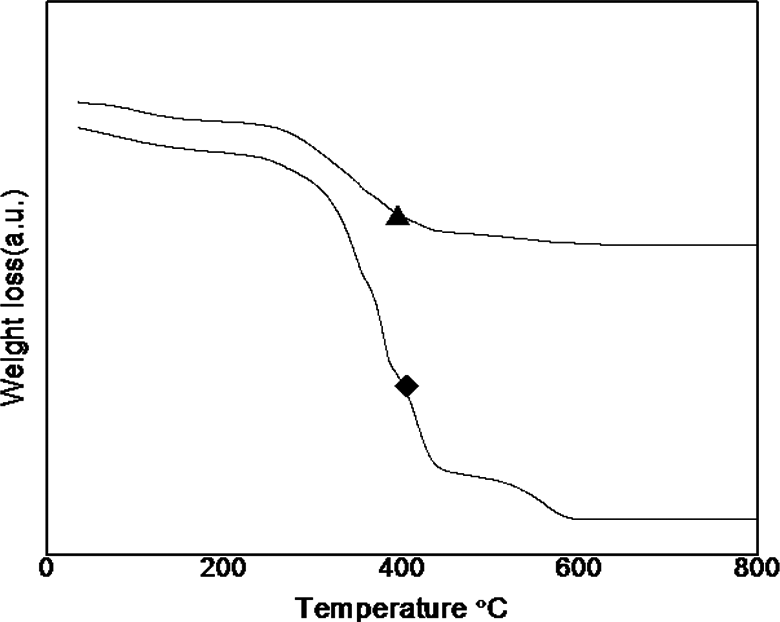
TGA thermogram of the polymers ♦, PUUA-6A6; ▴, PUUA-6T6.
TGA data for the PUUA copolymers at different temperatures.
Tensile test
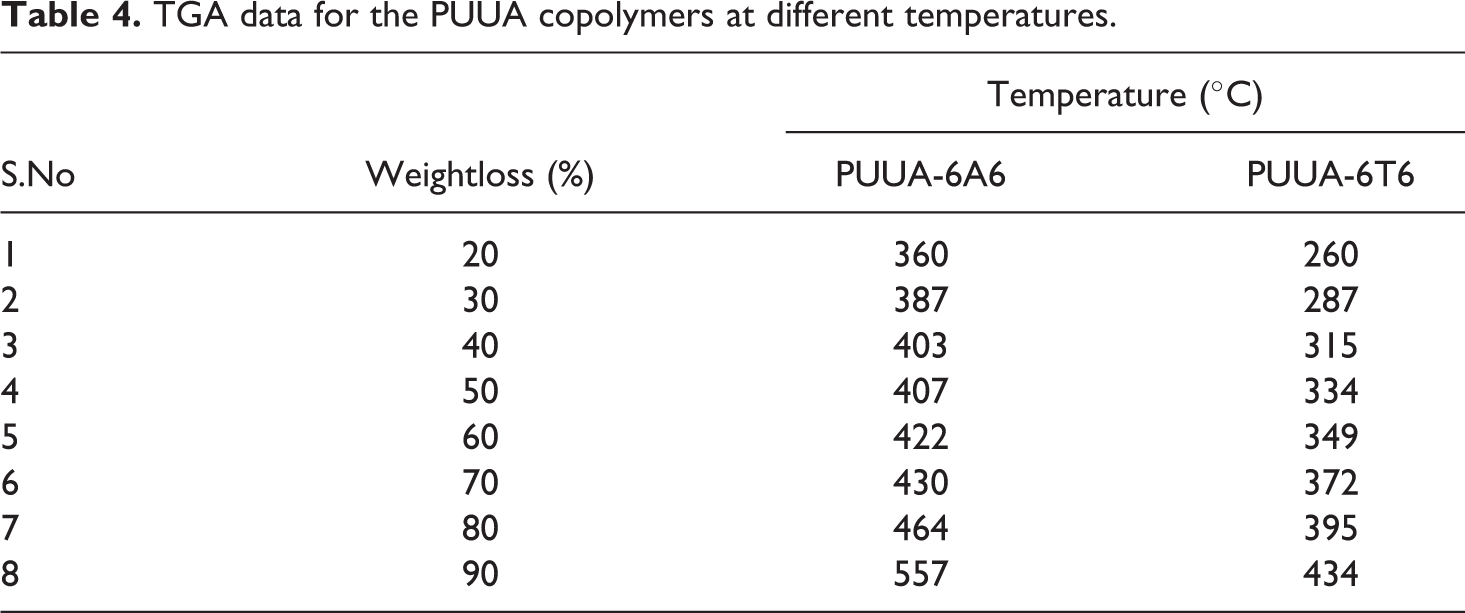
Tensile experiment studied to test the mechanical strength of the synthesized segmented block polymers. The polymers obtained from the reaction vessel made into sheet by solvent casting method. The obtained film then cut into a rectangular shape for the tensile experimental study. The stress-strain curve of the two polymers shown in the Figure 9. The derived data from the stress, strain curve presented in the Table 5. The tensile graph of both polymers showed strain induced crystallization effect. The strain induced crystallization reported by the several research groups on thermoplastic elastomers. The E-modulus value of PUUA-6T6 polymer is higher than the PUUA-6T6. The higher E-modulus value of PUUA-6T6 is due to the presence of aromatic ring which sandwiched between two hexamethylene groups in 6T6. However the elongation at break and the stress at break of PUUA-6A6 are higher than the PUUA-6T6 polymer system. The high percentage of elongation at break value is due to the high molecular weight of the polymer which produced during the polymerization process (ηinh value greater than 2.5 dL/g). In both the studied polymers, yield stress and yield strain are clearly visible due to the response of the crystalline structure of hard segment upon strain imposed into the polymer system. True fracture stress is the combination of fracture stress and the fracture strain. The true fracture stress of the PUUA-6A6 is higher than the PUUA-6T6 polymer due to the high fracture stress of the former. The observed values of the tensile experiment are in accordance with the published results of similar type of systems.17,23
Stress strain curve of the polymer: ▴, PUUA-6A6; ▪, PUUA-6T6.
Tensile properties for the PUUA block copolymers.

*σtrue was obtained by multiplying σbreak by the straining factor (=1 + (∊break/100)).
Biodegradation of polymer
Biodegradation studies carried out by dipping a piece of polymer sample in pH 9.2 and 7.0 solutions. Before biodegradation studies, the polymers have the ηinh values of 2.76 and 2.51 dL/g for PUUA-6T6 and PUUA-6A6. After 15 days, the polymer sample piece taken off from the solution and dried in vacuum oven at 70°C for a period of 24 h. After that, the inherent viscosity measured and found that 2.01 and 2.06 dL/g for the polymer PUUA-6T6 and PUUA-6A6 respectively. A similar procedure adopted after 28 days and the obtained inherent viscosity values are given in Table 6. Figure 10 shows biodegradability of the PUUA which shows that the viscosity of the polymer decreases with increase in time. The decrease in ηinh of the polymer with increasing in time is depending on the pH of the medium. Both the polymers showed drastic decrease in viscosity at neutral pH. The amide linkages are resistant to degradation at alkaline pH. This decrease in inherent viscosity suggested that the synthesized polymer can useful for biodegradation application.
Biodegradable data of PUUA copolymers.
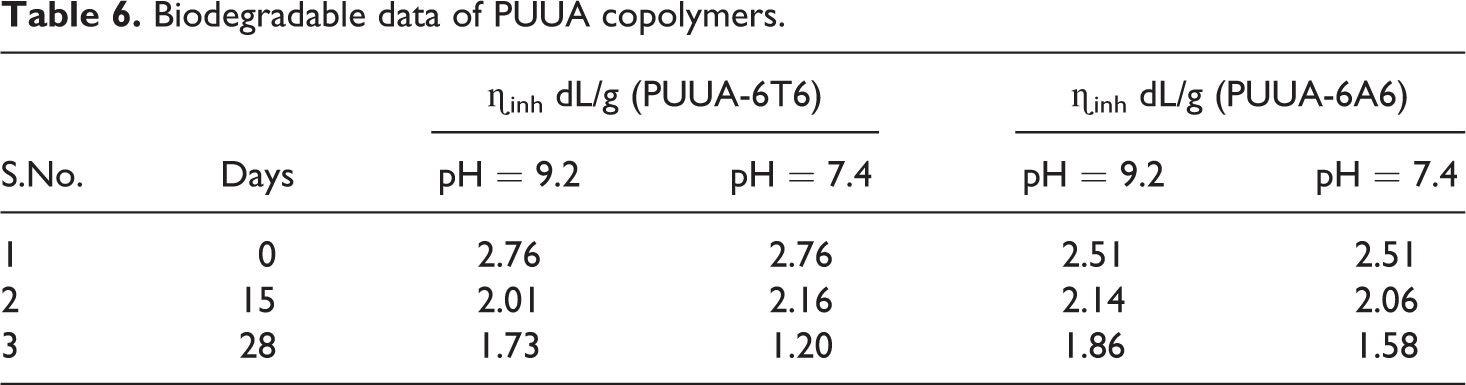
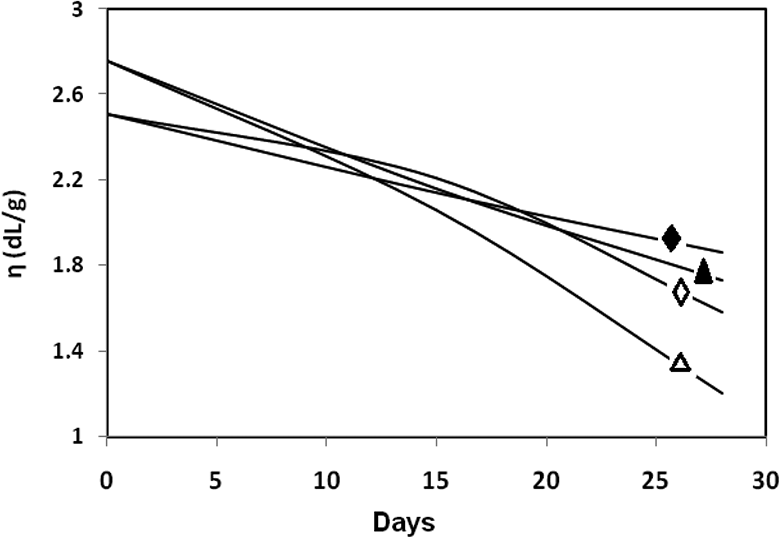
Biodegradability of polymer: ♦, PUUA-6A6; ▴, PUUA-6T6 for pH = 9.2 and ⋄, PUUA-6A6; Δ, PUUA-6T6 for pH = 7.4.
Conclusion
Polyurethane-urea-amide based on PEO, HMDI and hard segment (6A6 or 6T6) formed using a solution/melt polymerization method and characterized by NMR, FT-IR, DSC and TGA techniques. 1H NMR spectrum confirms the formation of PUUA copolymer. Using FT-IR techniques, the crystallinity of the HS measured and found the value greater than 70%. The HS crystallizes fast and fully. DSC data’s of the prepared copolymer shows three transitions, the Tg of SS, Tm of SS and the Tm of HS as for semi-crystalline material. Tm (HS) has high value in the case of 6T6 than 6A6 based PUUA; under cooling value of these polymers are very low due to fast crystallization of HS. These materials have very high thermal stability and it centered at 400°C. The mechanical strength of the polymers is very high due crystallized hard segment. Strain induced crystallization is found in our system. The biodegradability of polymers depending on the hard segment type and the duration of the study. Overall, the material highly suitable for biomedical application due to its high mechanical strength combined with biodegradable nature.
