Abstract
The current trends in biodegradable food packaging include the use of materials such as biopolymers which should be free of toxic metals to ensure their quality and use in multiple applications. However, these samples can contain zinc given its presence in the materials used to make them. In this study, a method to determine the concentration of zinc (Zn) in thermoformed and biodegradable flexible films samples based on Cassava and in their raw materials (flour, starch and fique) using flame-atomic absorption spectroscopy is described. Prior, an acid digestion with nitric acid under reflux was required. The method was standardized by means of the evaluation of statistical parameters. The method was sufficiently lineal (R2 = 0.999) in a working range from 0.1 to 1.0 mg Zn/L with detection and quantification limits of 0.03 and 0.82 mg/L, respectively. The method was found to be precise and accurate, and could therefore be used to measure Zn content at levels well below safe limits.The precision of the method was evaluated using intermediate precision and repeatability which showed coefficients of variation less than 6.7% and 4.7%, respectively. The percentages of recovery ranged from 96.5% to 98.2%. The method was successfully applied for the determination of Zn in the studied biopolymers samples and the results obtained support the method’s suitability for determining the presence of the metal. Zinc concentrations in thermoformed, flexible films and flour were below 2.36, 2.14 and 2.01 mg/L, respectively, indicating that these polymers could be used for food containers.
Introduction
Biopolymers are currently used in food packaging materials, food coatings and encapsulation of matrices for obtaining functional foods. 1 This is due not only to their potential to replace conventional polymers and other materials widely used in food packaging with a high carbon footprint, but also to the intrinsic properties and characteristics of these bio-based resources (e.g. edibility, antioxidant or antimicrobial properties).2–4
Biopolymer preparation includes the extraction from biomass (plants riches in starch, cellulose, etc.), synthesis from bio-derived monomers (based on the composition: polysaccharides, proteins, or lipids) and produced from microorganisms through fermentative processes of different bioresources. 5 According to their application, biopolymers properties can be modified; they can be laminated, improved with nano-inclusions or formed as composites.
Cassava (Manihot esculenta Crantz) is an exceptional source of the basic compositional units (carbohydrates, starch) required for obtaining of thermoplastic starch, a category of biopolymers. 6 Starch is a naturally occurring biopolymer that, once plasticized, can be thermally processed and handled on traditional extrusion, injection-molding, blow-molding, or thermoforming equipment. Biodegradable thermoformed and flexible films can be obtained from cassava flours by adding moulding substances such as: fique fiber, gelatin, poly(butylene adipate-co-terephthalate), glycerol, natural extracts, polylactic acid, plasticizer, cellulose, pullulan.7–10 As result, biodegradable alternatives made partially or entirely from renewable resources are created.
Prior to identify the application of biopolymer packaging materials is very important to define their composition and properties to ensure the related quality criteria and functionality in the operational environment. Numerous studies have evaluated the potential use of biopolymer packaging for a range of food products.2,5,11–14 The detection de heavy metals is a one of the key analysis to be evaluated in the biopolymers, especially if these have been designed to be food contact materials, since their persistence over time in the environment10,15,16 may result in their accumulation on the natural resources and become a contamination factor in the manufacturing process of biopolymers. The presence of these substances in packaging materials at concentrations not permissible and in their bioavailability chemical form are toxic to living beings due to the heavy metals are not biodegradable or metabolizable.16,17
In addition, the manufacture of biodegradable materials may produce or increase heavy metal contamination due to the raw material (natural or synthetic) used for their processing or cross-contamination coming from the pretreatment activities of the material (washing, drying and storage including utensils, water and equipment required) during manufacturing.18,19
Among the heavy metals, zinc (Zn) is one of the most typical elements in the earth’s crust that can enter to the body in diverse ways such as through the digestive tract, the lungs, by exposure, by manufacture process, by metal fume and can cause fever, dyspnea, cough, pneumonitis. 20 The Environmental Protection Agency (EPA) has stated that Zn is not classifiable as a “human carcinogen” due to the lack of information. 20
On the other hand, Zn accumulation can take place in the tissues of the cortex of cassava tubers and can increase in plants and tubers that are grown in periurban areas and industrial zones on soils contaminated by irrigation using wastewater and sewage sludge18,21 or fortified soils. 22 Thermoformed and flexible films are materials that can be used as food containers, and their quality and safety must be ensured in their processing and manufacturing as well as handling and storage. The safety of these kind of biopolymers is estimated by the quantity of substances that migrate to food and fulfilling of legal requirements. To the best of our knowledge, there are not any references regarding the presence of Zn in thermoformed and biodegradable flexible films.
In Colombia, there is not a current regulation that sets the maximum level of Zn in these samples. Different techniques have been used to quantify metals in different samples, such as flame-atomic absorption spectroscopy (F-AAS) which stands out as a conventional technique. F-AAS is a good alternative for the determination of elements such as zinc due to its high sensitivity and low limits of detection, in the order of μg/L. 23
This paper presents the quantification of zinc in thermoformed and biodegradable flexible films based on cassava, as well as in their raw materials, using the standardized technique of F-AAS on samples previously digested with nitric acid.
Experimental
Materials
The materials used were thermoformed (MBRA-383, MPER-183, CM 523-7, CM 7951-5, CM 4574-7, NATAIMA 31, HMC 1) and biodegradable flexible films (SM 707-17, SM 1498-4 and CM 7138-7, all pristine and hydrolyzed samples). Biopolymers samples were prepared from flour and starch of various species of cassava and fiber fique as material of reinforcement and supplied by CYTIBIA research group of the University of Cauca.24,25
Experiments were conducted upon samples previously cut manually or macerated (Sieve Newark USA Standard Series No. 18) into pieces of suitable size (≤1.135 mm) and dried at 70°C up to 4.5 h.
Reagents
Zn standard solution (1000 mg/mL, Titrisol Merck). Working solutions of Zn (0.1–1.0 mg/L for zinc) were prepared by dilution of a stock standard solution (500 mg/mL ZnCl2 in 0.2% HNO3 in a 100 mL volumetric flask) with water. Nitric acid: 65% (Merck). All of chemicals used were of analytical-reagent grade and were used as received without further purification. Millipore Milli-Q water was used throughout.
Sample digestion procedure
Organic impurities present in samples were removed through an acid digestion. Concentrated nitric acid (20 mL) was added to 1.0 g of the sample and then heated on a hot plate under reflux at 50°C for 3 h or 50°C for 45 min for the thermoformed and biodegradable flexible films, respectively. After cooling, digested samples were finally filtered through a Gooch crucible (Schott Duran glass porosity 2) and stored at 4°C until further analysis.
Standardization of F-AAS method
Evaluated statistical quality parameters for the standardization of F-AAS method for the quantification of Zn in thermoformed and biodegradable flexible films were linearity, precision through the repeatability and intermediate precision, accuracy, sensitivity, limit of detection (LOD) and limit of quantitation (LOQ). 26
In order to evaluate the linearity, a calibration curve of Zn concentration in the range from 0.1 to 1.0 mg/L including six solutions of known Zn concentrations was prepared. Intermediate precision involved one analyst and eight calibration curves of Zn (0.1–1.0 mg/L) analyzed over eight days in the same laboratory. While repeatability was carried out at the same day from five calibration curves of zinc in the same range of concentrations. The sensitivity of the method was carried out by comparison of the slope of the calibration curve used to assess intermediate precision with those of repeatability.
LOD and LOQ were estimated from the standard deviation (SD) of the intercept and average of the slopes of three calibration curves with Zn concentrations ranging from 0.2 to 1.0 mg/L. 27
In the study of accuracy, the addition of known amounts (10, 20 and 60 mL) of a stock solution of Zn to thermoformed (CM 4574-7) before the digestion treatment was used.
Stability was assayed as an additional parameter. Aliquots of a standard solution of Zn (1.0 mg/L) and thermoformed (CM 523-7) were analyzed by F-AAS over 10 consecutive days.
Determination of Zn in biopolymer samples
The digested samples were analyzed for the presence of Zn using atomic absorption spectroscopy (Thermo AA S4) with flame and a deuterium background corrector. The air acetylene at a rate of 1.2 mL/min and 0.5 nm slit was the flame used. A single Zn hallow cathode lamp was used as light source at a wavelength of 213.9 nm. The digested samples (5 mL) were analyzed in triplicates and expressed in mg/L after extrapolation from the standard curve. No matrix interferences were detected.
Statistics
The results of the standardization and implementation were statistically analyzed using SPSS v. 11.5, and Microsoft Office Professional Plus 2016. Shapiro-Wilk and Levene tests were applied. One way ANOVA, Pearson correlation, the coefficient of determination (R2) and coefficient of variation of response factors (CV) were calculated for each calibration curve. 28
Results and discussion
Zn calibration curve obtained using six concentrations (ranging from 0.1 to 1.0mg/L) of the standard for Zn was linear. Figure 1 depicts the maximum absorbance at 213.9 nm vs. concentration of Zn with relative standard deviations between 0.2% and 4.8%, and a correlation coefficient value equal to 0.999.
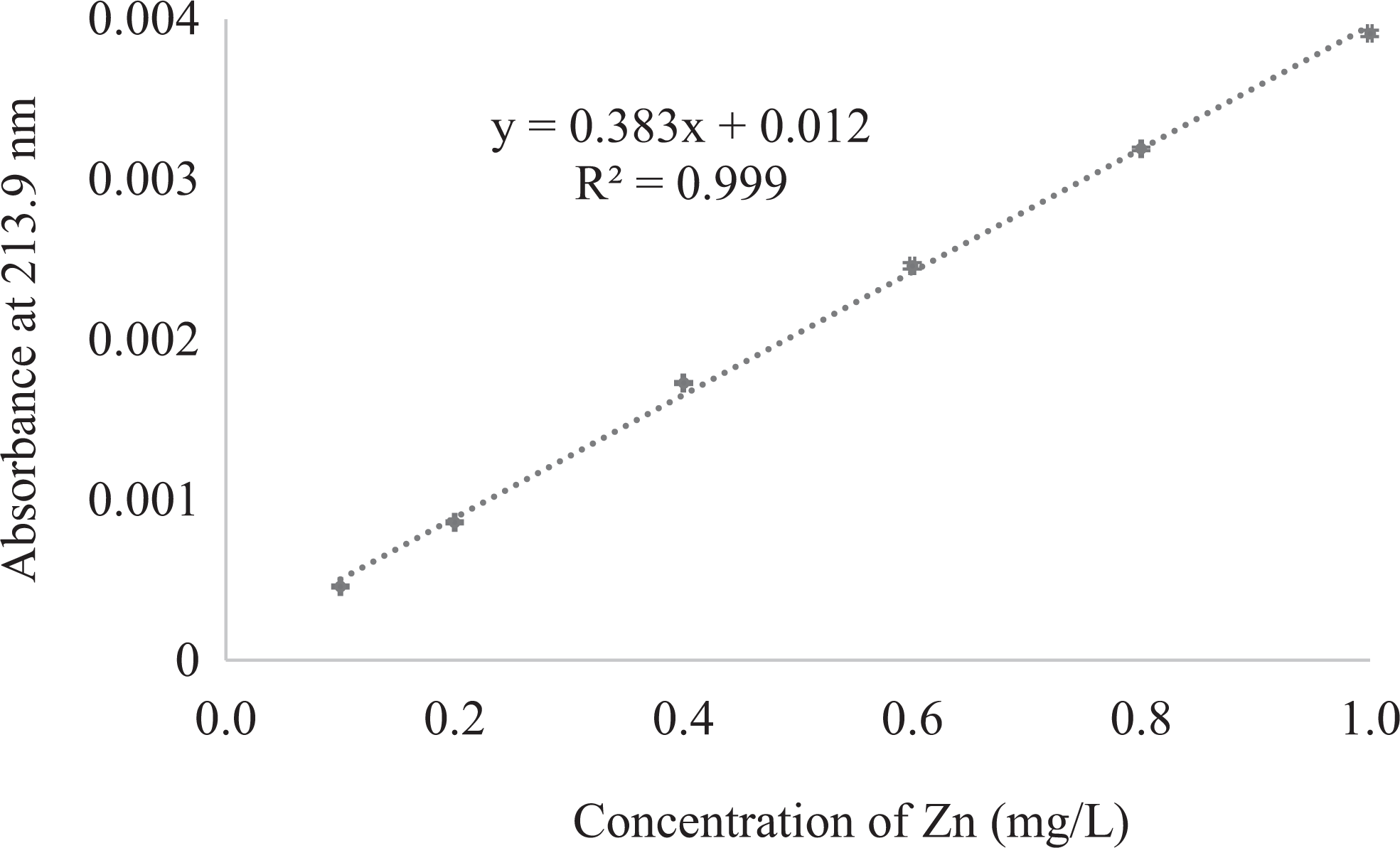
Calibration curve for Zn standard solutions absorption expressed as mean (n = 4). Source: The authors.
The results of absorbance, standard deviation of absorbance and CV for each Zn standard solution considered at the level of intermediate precision and repeatability are gathered in Table 1. Eight and five calibration curves were statistically analyzed for intermediate precision and repeatability, respectively, using the Shapiro-Wilk and Levene tests. Regarding to intermediate precision, significant differences were not found at any concentration level between the eight calibration curves and CVs values less than 6.7% evidence a good intermediate precision of the method. The linear equation for the average calibration curve was y = 0.3945 (±0.0132)× + 0.022 (±0.040) with R2 of 0.989 and the Pearson correlation coefficient was equal to 0.994. In the case of repeatability, the obtained CVs (less than 5.0%) indicate that the studied spectroscopic method is repeatable with an average linear equation of y = 0.3826 (±0.0091)× + 0.017 (±0.0083) and a R2 of 0.988; so it can be affirmed that at all Zn concentrations in the five calibration curves there was low variability in relation to the mean of the population. Pearson correlation coefficient was equal to 0.993. In general, the dispersion of the data was less than 0.005 mg/L of Zn. All these results demonstrate that the precision of the method studied is adequate according to the acceptance criteria set by Instituto Nacional de Metrologia, Normalização, e Qualidade Industrial. 29
Intermediate precision and repeatability study of Zn.
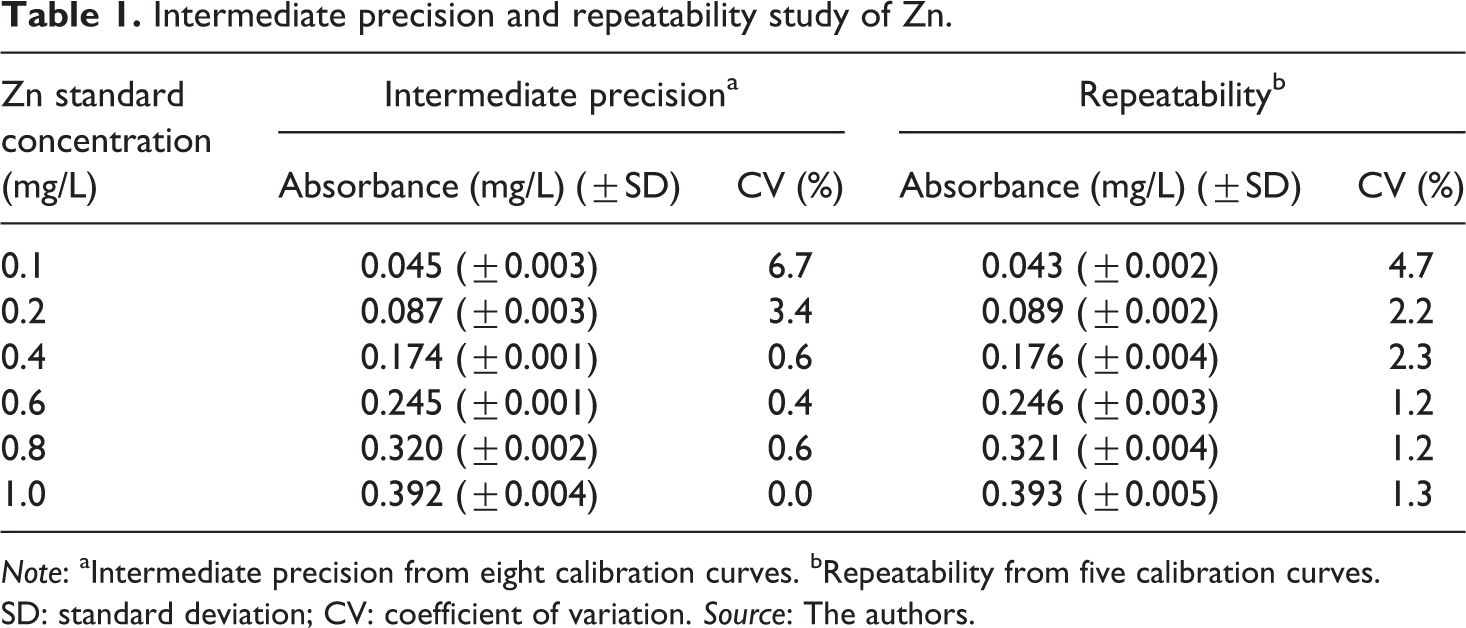
Note: aIntermediate precision from eight calibration curves. bRepeatability from five calibration curves. SD: standard deviation; CV: coefficient of variation. Source: The authors.
Sensitivity is an important parameter in method optimization. The sensitivity of the method was estimated based on the calibration curve´s steepest slope analyzed for precision. The considered values correspond to 0.3945 (±0.0132) and 0.3826 (±0.0091) for intermediate precision and repeatability, respectively. Thus, the method resulted to be more sensitive if the analysis is performed on the same day. The sensitivity of the method was 0.3826 suggesting that the proposed method for sample preparation is appropriate.
The values of LOD and LOQ of Zn were 0.03 mg/L and 0.82 mg/L. These parameters are suitable for routine monitoring of quality control of biopolymers or plastic materials with Zn levels higher than 0.82 mg/L.30,31
Accuracy of the method was assessed by recovery studies from three replicates determinations of CM4574-7 thermoformed sample spiked with three different levels of zinc standard solutions. This procedure considered sample preparation, digestion treatment and analysis of samples by F-AAS. Percentage recoveries of Zn between 96.5% and 98.2% were obtained indicating the method is accurate for Zn determination in these samples according to the criteria set by INMETRO. 29 Significant differences between percent recoveries of Zn and 100% recovery were detected using t student test which can be attributed to random errors in the stages of preparation of solutions and/or analyte loss during the digestion.
On the other hand, a high stability of Zn standard solution (1.0mg/L) and CM 523-7 thermoformed sample over time (ten consecutive days) was demonstrated. Maximum CVs values of 4.1% and 4.2%, respectively, were computed.
The standardized F-AAS method was applied to the quantification of Zn in biopolymers samples. Zn concentrations determined in the seven thermoformed samples are registered in Table 2. Values ranged from 0.81 to 2.36 mg/L. Observed variation of Zn concentration is associated to the composition of the raw materials employed for the production of biopolymers.
Zn concentrations in biodegradable thermoformed samples analyzed (n = 3).
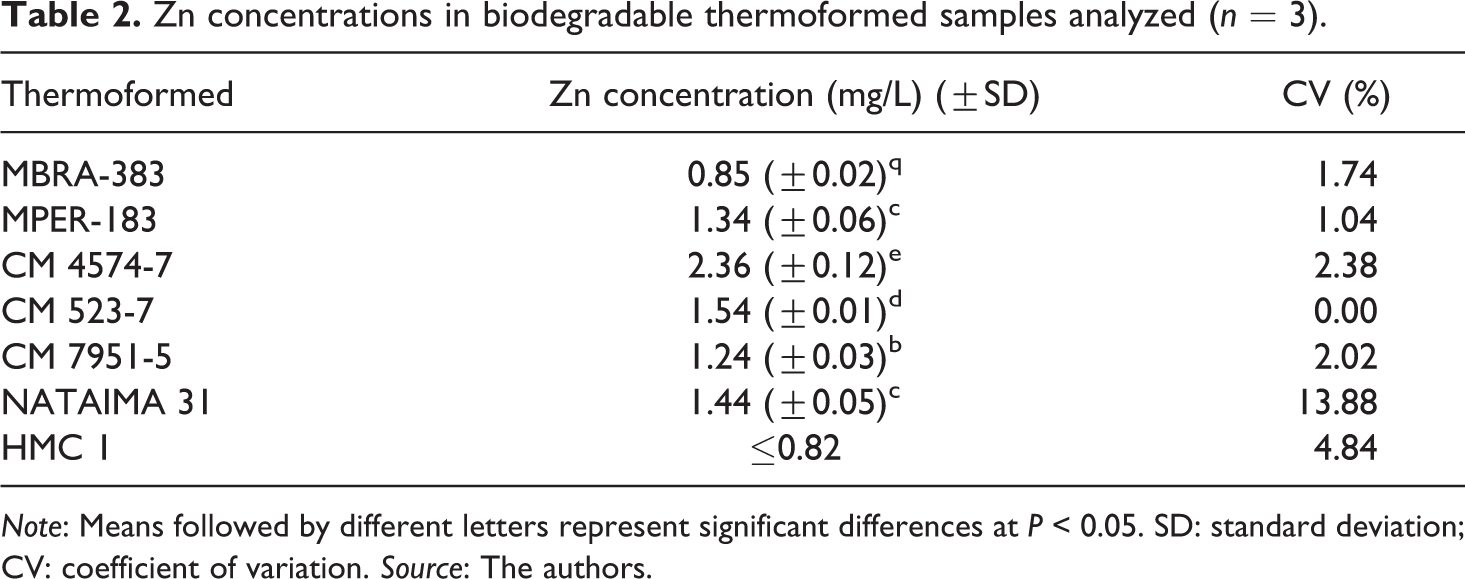
Note: Means followed by different letters represent significant differences at P < 0.05. SD: standard deviation; CV: coefficient of variation. Source: The authors.
In previous studies, the presence of others metals such as mercury, arsenic, selenium, lead and cadmium in these samples was detected.10,24,32–34 Levels of mercury up to 67.2 mg/L, arsenic below 1.9 mg/L, selenium up to 62.0 mg/L, lead up to 3.2 μg/L and cadmium ranging from 21 μg/L to 89 μg/L were found.
Zn concentrations determined in the flexible films in the pristine and hydrolyzed forms are presented in Table 3. Quantities of Zn ranged from 1.13 to 2.14 mg/L. Literature reports for these same samples the detection of mercury ≤6.4 mg/L, 24 selenium ≤5.4 mg/L, 33 while arsenic, 32 Lead 34 and Cadmium 10 were not detected. These findings may be associated to the cultivation areas which are naturally rich in these toxic metals. Several authors have reported that Zn uptake from soil is the highest in plants grown in areas with elevated heavy metal levels.18–20 Zn is released into the environment and can be introduced into the flour, starch and fique fiber by irrigation with contaminated water or due to soils contaminated.
Zn concentrations in biodegradable flexible films samples analyzed (n = 3).
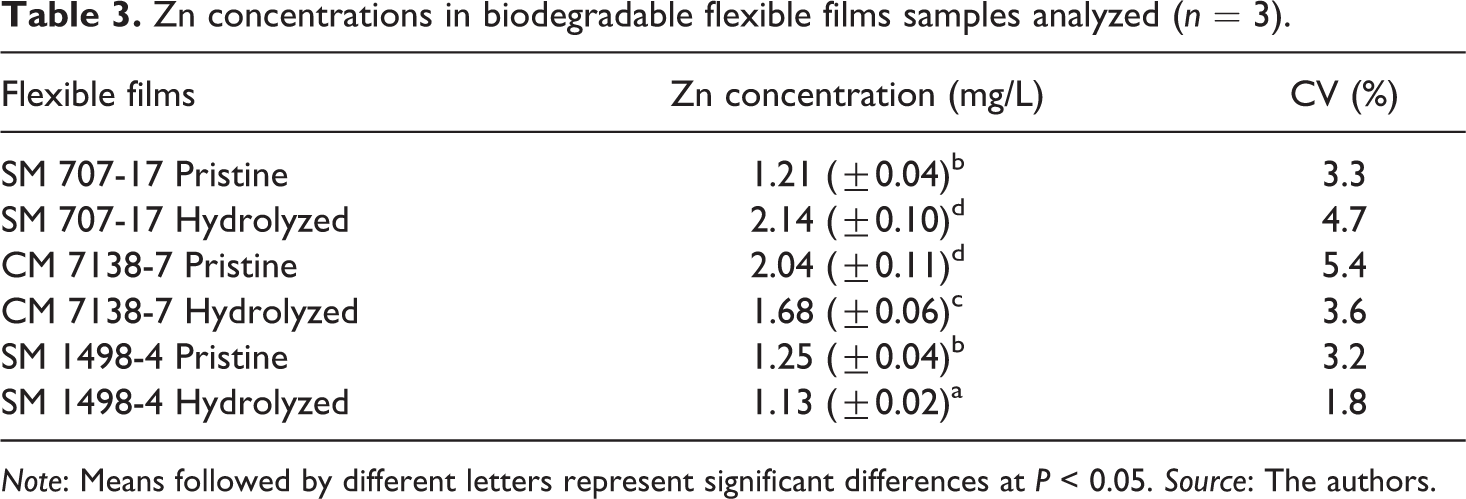
Note: Means followed by different letters represent significant differences at P < 0.05. Source: The authors.
Amounts of Zn found in the thermoformed (Table 2) and flexible films (Table 3) above the limit (1 ppm) authorized by BIS, IP, USP and other regulatory agencies, 30 indicate that Zn migration from the material would affect the quality of the final product and could negatively affect to consumers’ health. However, it is necessary to study the source, speciation and bioavailability of this metal in these samples thoroughly. In addition, Zn was quantified in raw materials (cassava flour and starch and fique fiber) used in the elaboration of biodegradable thermoformed samples. The measurements are shown in Table 4. Zn in the cassava starch could not be quantified (concentrations less than LOQ). The largest Zn contributions to CM 4574-7 and CM 523-7 thermoformed materials (≈85%) are made by flours (2.01 and 1.31 mg/L, respectively). Fique fiber presented a high Zn content (84.2 mg/L), hence this raw material is identified as a significant source of Zn for the thermoformed films.
Zn concentrations in the raw materials used in the production of the thermoformed samples (n = 3).
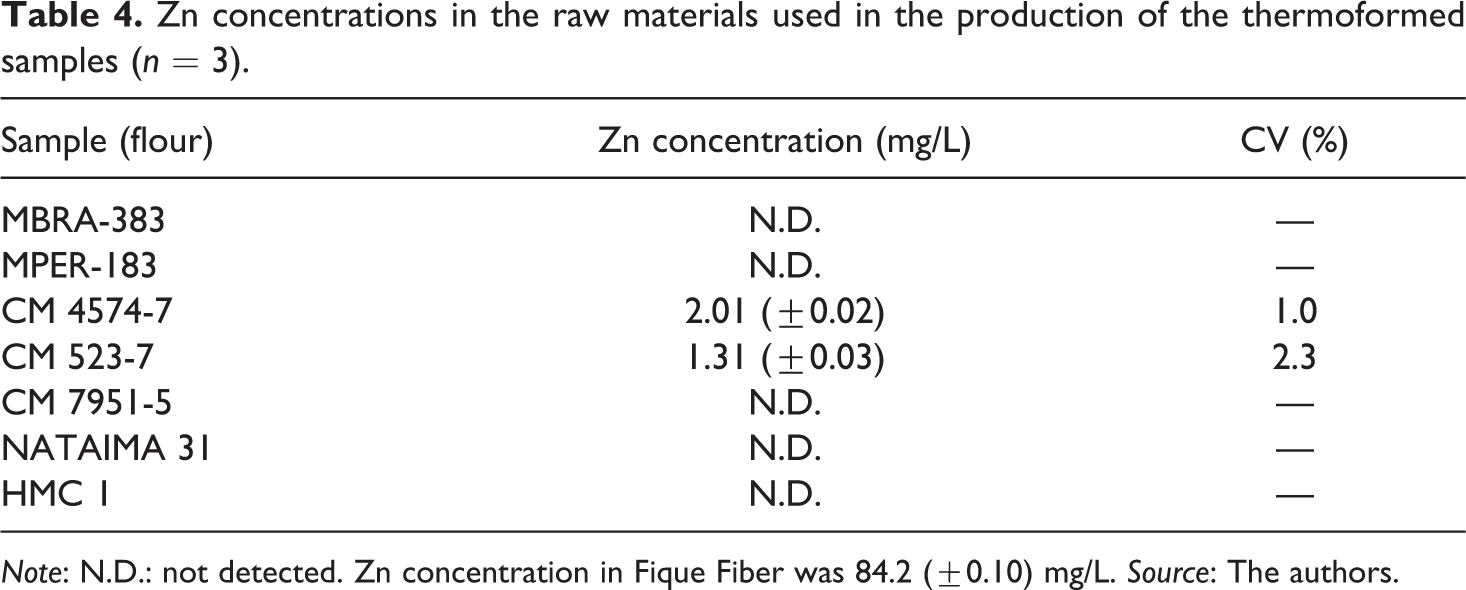
Note: N.D.: not detected. Zn concentration in Fique Fiber was 84.2 (±0.10) mg/L. Source: The authors.
Results shown in Tables 2 and 3 suggest that Zn found in thermoformed material comes principally from cassava flours and fique fiber. In these natural sources, Zn was probably accumulated during their cultivation, post-harvest, storage and transportation. On the contrary, the manufacturing process of biopolymers is discarded as a source of contamination.
In general, the Zn content found in the biopolymers studied was high, meaning that these materials could be probably be toxic and their toxicity must be evaluated before they are used as food wrapping material, food containers, or in food conservation35–37 without potential health risks to the consumer. Hence, the implementation of good farming and manufacturing practices are key to obtain heavy metals free materials and safe to be used as food packaging.
Conclusions
It is important to monitor the presence of Zn in biopolymeric materials that are widely usedas biodegradable food packing in order to estimate their potential migration and therefore to avoid the harmful or toxic effects of this metal on food or human health. The developed method of FAAS in this study resulted to be an acceptable analytical routine method for the quantification of Zn in the analyzed biopolymers based on cassava (precise, accurate and LOD and LOQ in order of mg/L).
Quantified Zn in the assayed thermoformed and flexible films and their raw materials led to infer that the flour and fique fiber are the main sources of this metal into the films, and the manufacturing process less likely, therefore, investigations that enable the speciation and bioavailability of zinc from the material to food are required to ensure the final use of the polymeric material.
Footnotes
Acknowledgments
We would like to acknowledge the University of Cauca (Industrial Analysis Unit, BICAMSA and QPN laboratories) and the Ministry of Agriculture and Rural Development for funding the projects on which this work is based.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universidad del Cauca (501100005682; BICAMSA and QPN laboratories) and by Ministerio de Agricultura y Desarrollo Rural de Colombia.
