Abstract
This study presents an alternative to reducing solid waste, improving the concept of green composites. So, the use of fibers from pine cone as reinforcement in acrylonitrile butadiene styrene (ABS) composites filaments as a potential for 3D pen was evaluated. The effect of the treatment chemical (alkaline and bleaching) was studied. A thermokinetic mixer processed the ABS/pine cone fibers (2 and 5% wt.) composites. After, the filaments were prepared by mini extruder and printing 3D pen. Filaments were characterized by Scanning Electron Microscopy (SEM), Thermogravimetry (TGA), and Infrared Spectroscopy (FTIR). Filaments revealed homogeneous diameters. The addition of 2 and 5% wt. fiber not significantly influenced the filament’s diameter and density. On the other hand, thermal stability and morphological analysis influenced the type of fiber (raw, treated, and bleached). The addition of bleached fibers to ABS increased composites’ thermal stability compared to other fibers (treated and raw). Also, inserting bleached fibers was perceptible a uniformly distributed and embedded throughout the wire cross-section compared to treated and raw fiber added to ABS due to good interfacial bonding. Results indicated that fibers were hydrogen-bonded to ABS chains and increased the filament’s density. So, it is possible to affirm that the addition of fibers from pine cone to ABS thermally improved and can be a low-cost feedstock for printing 3D pen applications. Despite the low concentration of natural fiber on the composites investigated in this work, the successful obtainment of ABS reinforced with biodegradable natural fiber, compromising neither its thermal properties nor its processability and printability, opens the possibility for future work investigation into a composite with larger fiber content.
Keywords
Introduction
One of the most versatile additive manufacturing (AM) techniques, three-dimensional (3D) printing, is a relatively recent approach, which has been the focus of research due to its ability to model quickly, allowing complex development 3D objects with high precision and reduced cost compared to the traditional manufacturing process.1–3 Although it is a promising processing technology, 3D printing has its limitations, that is, the limited availability of material and documentation.
The most widely investigated 3D processing technologies are fused deposition modeling (FDM), stereolithography (SLA), selective laser sintering (SLS), direct-write (DW), and binder jetting (BJ) processes. Among these printing methods, FDM stands out for its reliability, cost-effectiveness in producing 3D objects with good resolution, wide material customization, simple fabrication process, and low-cost features; it is commonly used in producing conceptual models, engineering components, and prototypes.4–7 FDM is a very flexible technology being easily integrated into computer-aided design (CAD) software packages, where the filament is melted, extruded through a nozzle, and deposited on a build sheet as per the desired path to complete the part represented in the 3D CAD model. Commercially available FDM machine possesses an upper-temperature limit of the heating element/chamber equal to 300°C. Thus, high melting point material cannot be processed by this technique. Only a few thermoplastic materials such as acrylonitrile-butadiene-styrene (ABS), high-density polyethylene (HDPE), nylon, polylactic acid (PLA), and their blends hold the correct thermal and rheological properties to be quickly processed by FDM. 8
Based on this limitation, most researchers are focused on developing composites materials such as fiber-reinforced composites, which possess properties from their components (matrix and reinforcement). At present, reinforcements such as glass and carbon are widely used in the manufacturing industry, particularly in the transportation sector, replacing components previously made of metals. Although these composites possess superior properties, i.e light weight structures, corrosion resistance, high dimensional stability, they are non-biodegradables, and their recycling is a global concern. Natural fibers are great candidates to replace these materials due to their low cost, low density, abundance, low abrasive wear, and biodegradable nature. Polymer matrix reinforced with agricultural waste, such as coffee, 9 pineapple peels, 10 cotton, 11 nutshell, 12 palm, 13 rice husks, 14 fruit stones, 15 coconut shells, 16 pine cone, 17 bamboo, 18 rice straw 19 has been showing promising results. Among these, the pine cone stands out as one of the most common residues in North America and is vastly explored worldwide. The conversion of this inexpensive and widely available resource to various value-added materials is quite promising. 20 Pine cone fibers are a cellulosic material readily available and can be used as reinforcement in a thermoplastic-based composite because they mainly consist of cellulose, hemicelluloses, and lignin macromolecules which provide a hydrophilic character.17,21 The chemical composition of pine and its constituents (wood, bark, leaves, cones, seeds, and resin) varies according to the Pinus tree and many other factors and can be compared to other natural fibers. 22 Although pine cone is an inexpensive and widely available source, composites reinforced with pine cone have shown increased mechanical properties (such as Brinell hardness, tensile strength) compared to other natural fibers as an acorn. 23
The main component of natural fibers is the cellulose, a natural polymer rich in the hydroxyl group, and the remaining components (hemicellulose and lignin) are molecule rich in carboxyl, hydroxyl, and other functional groups. Thus, natural fibers are usually classified as hydrophilic materials. On the other hand, the polymer matrix tends to be apolar, exhibiting significant hydrophobicity. Consequently, there are significant problems of adhesion between the fiber and the matrix that can compromise the composite application, as reported by Oksman, 24 when investigating the effects on the properties of polypropylene (PP) reinforced with flax fibers. The PP reinforced with natural fiber showed lower tensile and impact strengths compared to glass fiber reinforced composite. This behavior could be attributed to the poor adhesion between the flax fiber and the matrix.
A general strategy to produce a good quality natural fiber reinforced thermoplastic filament is to alter the fiber surface with chemical treatment. The chemical treatment improves the fiber’s compatibility by removing non-cellulosic components to enhance its mechanical and thermal properties. In this context, pine cone stands out being a promising source of cellulose fiber. Its great potential for usage as reinforcement in polymers matrix was reported by García et al., 25 according to the authors, pine cone has shown a cellulose content as high as 88.99 wt% after blenching treatment, the high cellulose concentration of pine cone fibers resulted from the progressive removal of lignin and hemicellulose, made it suitable for a subsequent cellulose nanocrystal isolation.
This research purposes an alternative for pine cone (Pinus elliottii) fibers as reinforcing in acrylonitrile-butadiene-styrene (ABS), obtaining eco-friendly composite filament for use in pen 3D printing never-before-explored, aiming all the advantages offered by the natural fiber in the composite, and opening the possibility for future mechanical properties investigation. The fiber surface treatment and content on the thermal and microstructure properties of the composite filaments were evaluated.
Materials and methods
Materials
To obtain the composite’s filaments, the fibers from pine cone (Pinus elliottii) were kindly donated by a farm producer, and the acrylonitrile-butadiene-styrene (ABS) was provided by Ineos Styrolution of Brazil (GP-35). Firstly, the pine cone (Pinus elliottii) were dried at 80° C for 24 h, the fiber was then crushed and sieved (35 mesh) in a mill Willye, model TE-650 (Tecnal Equipamentos Científicos Ltda., Piracicaba, Brazil).
Obtaining of treated fibers from pine cone
Fibers untreated from pine cone were pre-treated with a NaOH solution (4% w/v) for 1 h under constant stirring at room temperature. After that, the solution was filtered using a vacuum filter, and the remained fibers were washed with distilled water.
Obtaining bleached fibers from pine cone
Treated fibers were previously dried at 80 ºC for 24 h. Subsequently, they were bleached with a 1:1 solution of NaOH (1 mol. L−1) and H2O2 30% (v/v) at 70°C for 1 h. The fibers were filtered to remove the excess reagents, and all the steps were repeated one more time. Subsequently, the fibers were washed with distilled water until neutral pH and dried at 60°C for 24 h, obtaining bleaching fibers.
Characterization of the fibers
The morphology of the raw, treated, and bleached fibers were analyzed by Scanning electron microscopy (SEM) through a microscope (model TR-3000, HITACHI Ltd., Tokyo, Japan) with tungsten filament, secondary electron detector, 5 kV, and low-vacuum. A double face 3 M tape fixed the foam samples on brass support.
The physical structures of the raw, treated, and bleached fibers were evaluated by X-ray diffraction using a Shimadzu diffractometer (model XDR-6100, Kyoto, Japan). The measuring conditions were: CuKa radiation with graphite monochromator, 30 kV voltage, and 40 mA electric current. The patterns were obtained in 10–50o angular intervals with 0.05 steps and 1 s of counting time.
The chemical structures of the raw, treated and bleached fibers from pine cone were analyzed by attenuated total reflectance (ATR) in a spectrometer (Perkin Elmer® Inc, model Spectrum 100, Massachusetts, USA) by a transmittance mode, covering wavenumbers from 4500 to 400 cm−1 with a spectral resolution of 4 cm−1.
Preparation of composites filaments
The raw, treated fibers and bleached fibers from pine cone were mixed with ABS in a thermokinetic mixer model MH-50 H (MH Equipamentos Ltda., Guarulhos, SP, Brazil) at a high-speed rate rotation (5250 rpm) for 55 s, in which fibers were responsible for 2 and 5 wt% of the composition. The homogenized mixture was then milled in a knife mill (Plastimax Indústria e Comércio Ltda, Barueri, Brazil) and dried at 50 ºC for 2 h.
ABS’s filaments reinforced with different percentages of raw, treated, and bleached fibers from pine cone (2 and 5 wt%) obtained using a mini-extruder (brand Weellzoom, model B Desktop, Guangdong Prov, China). The processing temperature of the filaments was in the range of 190–195°C. The mini-extruder (Weellzoom) has only one temperature control option, so different temperatures were not used throughout the extrusion chamber. After, filament diameters were measured on each test specimen at the interval of 50 mm using a caliper rule. In all, 10 measurements for each filament sample were performed. The density was calculated as the ratio of mass per unit volume. Composite filaments were tested in the printing 3D pen (3DPEN-2, China).
Characterization ABS/fibers from pine cone filaments composites
The microstructural analysis of the filaments’ surface and cross-section was performed by scanning electron microscopy technique, using a microscope HITACHI with tungsten filament operating at 5 kV, employing a low-vacuum technique and secondary electron detector at different magnifications (i.e.50×, 200×). Samples were dispersed on brass support and fixed with a double face 3 M tape. Samples cryo-fractured to expose the internal structure for high-resolution imaging.
Thermal analyses were performed to evaluate the stability of the fibers, ABS, and composites filaments. Thermogravimetric analysis was conducted using a simultaneous TGA/DSC thermal analyzer, Model SDT Q600 (TA Instruments). Experiments were carried out using a specimen weight of 5 mg under nitrogen as purging gas, utilizing a heating rate of 10 ºC.min−1, from 30 ºC to 600 ºC.
Results
Characterization of the fibers
The obtaining of bleached fibers from pine cone demonstrates a difference in the color fibers, size fibers, and morphology, as shown in Figure 1. The alkali treatment changed the color of the fibers from reddish-brown to dark brown, and a yellowish color was observed in the fibers bleached with H2O2.
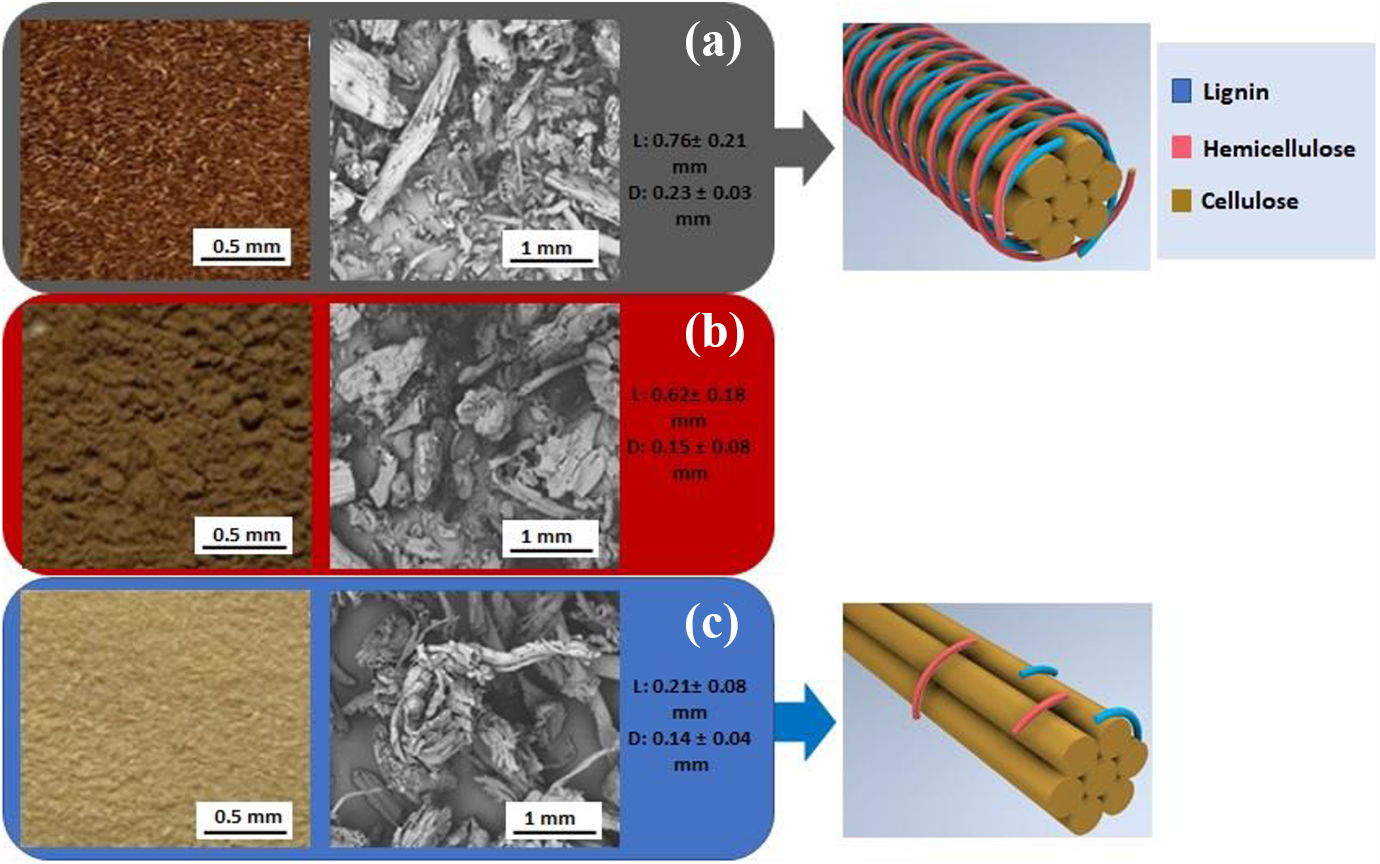
Visual aspect and SEM of the fibers: (a) raw fibers; (b) treated fibers; (c) bleached fibers.
This color change can be associated with chromophore groups’ formation because the perhydroxyl ions (HOO−) generated by the dissociation of hydrogen peroxide in alkaline media are responsible decolorization of the fiber. 26 These ions attacks remove the chromophore compounds or the remaining hemicellulose and lignin in the delignification process. Thus the cellulose becomes whiter, producing better physical and thermal characteristics.27,28 Rayung et al. 29 cite that the bleaching of fiber with hydrogen peroxide can remove surface impurities and create a rougher fiber surface. The use of H2O2 as an oxidizing bleaching agent causes discoloration of fiber. According to Rosa et al., 30 this change in tonality is qualitative indicative that the expected removal of amorphous substances, such as wax, fatty substances, lignin, and hemicellulose, might have been achieved. It is worthwhile to highlight that the same effect has been observed in different research groups.31,32
The treatment made in the fibers also caused a change in morphology and size of fibers. The pine cone fiber raw’s surface morphology is tubular with a circular section (Figure 1a). Similar behavior was observed by Arrakhiz et al. 17 when investigating the mechanical and thermal properties of polypropylene reinforced with pine cone fibers.
The pinecone fiber treated with an alkaline solution showed a rougher surface topography than the untreated fiber (Figure 1b). Previous reports have shown that this change in the surface topography results in a better fiber-matrix interface adhesion and an enhancement of the mechanical properties. Another effect of the alkaline treatment on fiber is an increase in the amount of cellulose exposed on the fiber surface, increasing the possible reaction sites.33,34
The roughness of the surface increases significantly in the bleached fiber (Figure 1c). The bleached fibers’ surfaces are smoother and contain fewer aggregates compared to the surface of the untreated and treated fibers. Changes in the bleached fiber surface occur due to removing surface impurities, lignin, and hemicellulose, as has also been previously reported.27–29,35,36 Also, it was observed by the SEM micrographs of bleached fiber is the existence of pores on the fiber surface as presented in Figure 1c. Bleached fiber showed a wrinkled surface due to the disintegration of lignocellulose.
Similar micrographs were observed by Arnata et al. 37 to produced cellulose from sago frond with alkaline delignification and bleaching on various types of bleach agents. Meng et al. 26 evidenced that after some structural breakdown and transformation, buckwheat straw showed a wrinkled surface, owing to the disintegration of lignocellulose when investigating the effects of alkaline hydrogen peroxide (AHP) treatment. Li et al. 38 also evidenced by similar micrographs during the hydrogen peroxide (H2O2) bleaching process of the mulberry stem came from a sericulture base.
Therefore, it is possible to conclude that a larger amount of amorphous substance was removed by bleaching the fiber with hydrogen peroxide, causing a change in the physical structure. 29
Figure 2 shows X-ray diffraction patterns of raw, treated, and bleached. From these results, was possible observed three main peaks in 15.3°, 22.12°, and 34.25°, corresponding to lattice planes of peaks (110), (200), and (004) corresponding for cellulose I.39,40 These patterns are typical of semicrystalline materials with a broad crystalline hump and an amorphous peak.41,42 Table 1 shown the CrI (crystallinity index) was found to be about 26.04, 29.27, and 58.81% for the raw, treated, and bleached, respectively.
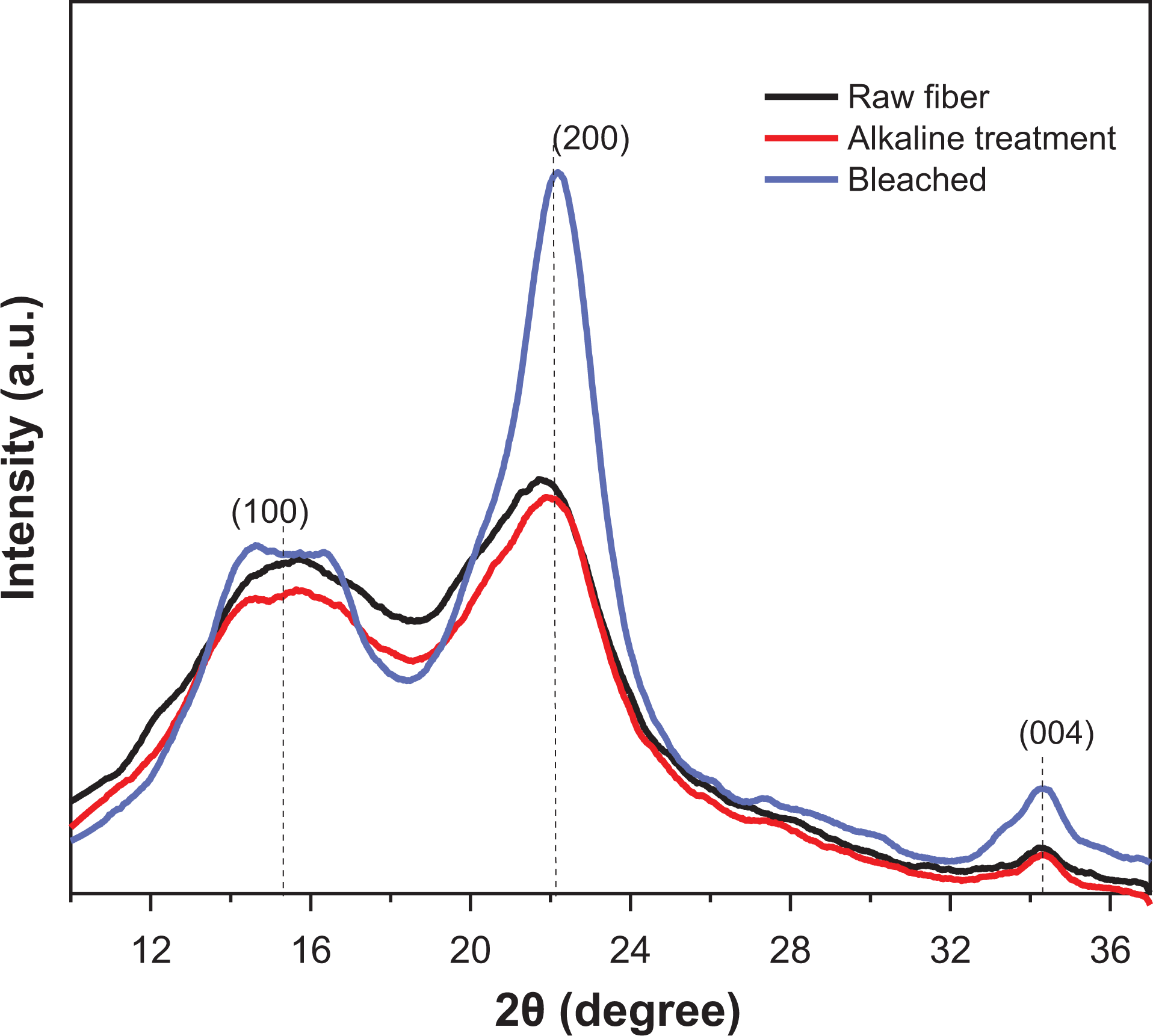
X-ray diffractograms of raw, treated, bleached fibers from a pine cone.
CrI of raw, treated, bleached fibers from pine cone.

The increase of CrI from raw to bleached was ascribed to the progressive removal of amorphous noncellulosic materials (lignin, hemicellulose, ashes, and extractives).43,44 El Halal et al. 45 studied barley fibers using similar treatments and found results related to this work (15.4, 22.7, and 34.5), CrI was found 38.9, 56.4, and 73.2 for the barley raw, alkaline treatment, and bleached, respectively. Pereira et al. 36 applied alkaline and bleaching treatment on pineapple crown fibers and also found increasing in CrI, values observed were 60.1%, and after the bleaching treatments were 84.34%.
FTIR analysis has been widely analyzed to investigate chemical and structural changes occurring in biomass during bleaching treatment from the raw fiber. Several absorption bands were observed and the presence of cellulose, hemicellulose, and lignin in the fiber was confirmed, as shown in Figure 3. The broadband at 3336 cm−1, it is possible to observe broadband for all, in this region is due to the OH-stretching vibrations arising from hydrogen bonding in cellulose and water absorbed, 46 while the band 2880 cm−1 the bending of CH2 hemicellulose component. 47 The vibrations at 2930 cm−1 were attributed to the C-H vibrations in aromatic methoxyl and methylene groups of the side chains. 48
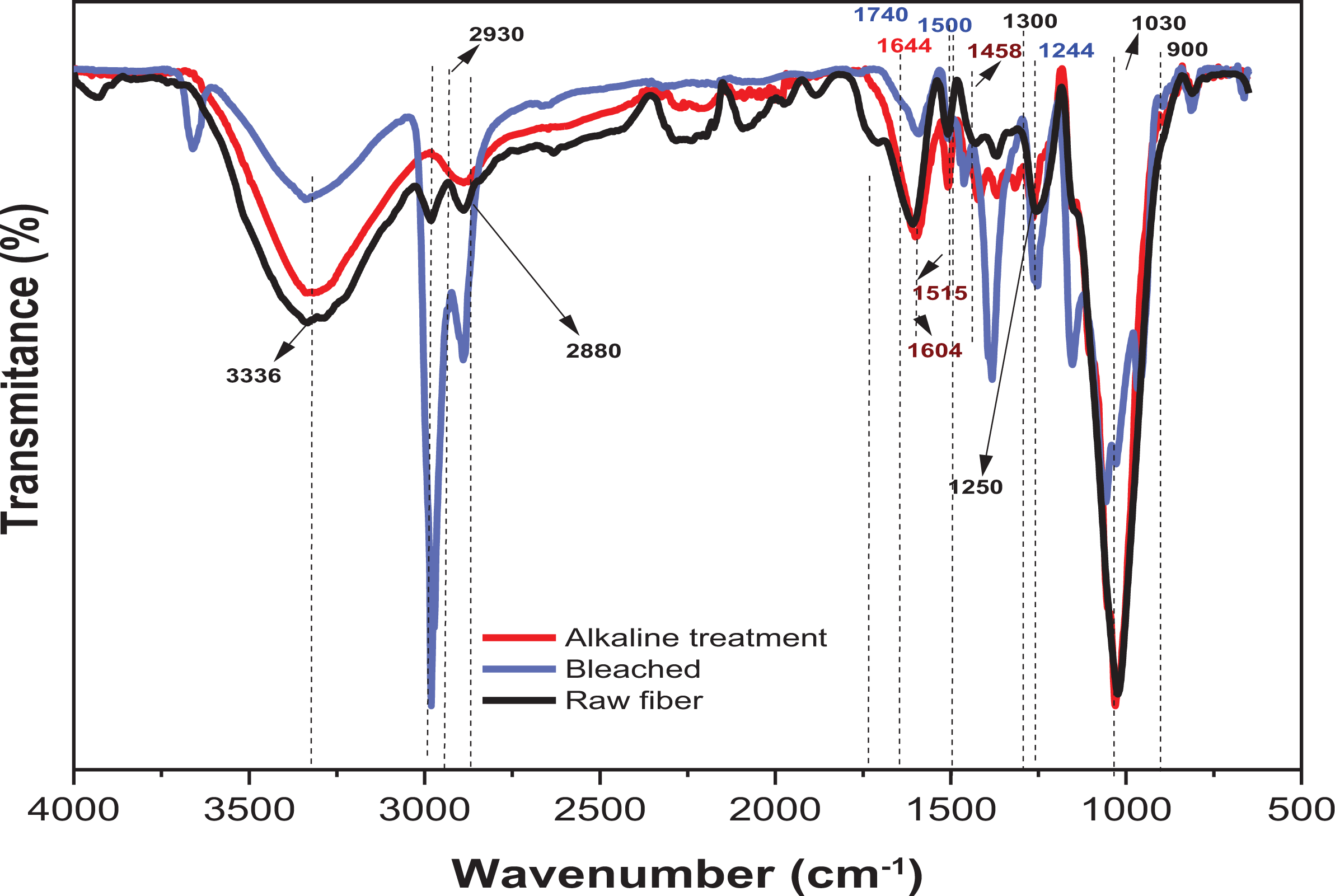
FTIR of raw, NaOH treatment, bleached fibers from a pine cone.
After chemicals treatments, the band 1244 cm−1, 1174 cm−1, and 1500 cm−1 are accentuated or disappear, which can be attributed to eliminating or reducing hemicellulose, mainly lignin. The peak 1174 cm−1 is attributed to the stretching of the C=O bond in carbonyl, ester acetyl groups in the xylan components of hemicellulose, and lignin structure. The peaks 1500 cm−1 and 1244 cm−1 are attributed to the C=C bond in aromatic rings in lignin and the C-C-O stretching cellulose, hemicellulose, and lignin. 49
The peak at 900 cm−1 corresponds to the glycosidic bonds present in the cellulose. 50 The peak at 1030 cm−1 is due to the C–O–C from lignin or hemicelluloses 51 or the C–O stretching and C–H rocking vibrations of the pyranose ring. 52 These peaks’ intensity is sharply weakened after treatment chemical (alkaline and bleaching) because of removing the main hemicelluloses material.51,53
The bands in the region at 1604 cm−1, 1515 cm−1, and 1458 cm−1 relating to the lignin macromolecules are observed in nature. 52 However, the mentioned band was not detected in bleaching, suggesting that lignin and hemicellulose were successfully removed or reduce after alkaline and bleaching treatments. Consequently, the FTIR results are in accord with other kinds of literature, in which the extraction of cellulose from natural fibers showed similar spectra.36,54
TGA and DTG curves of raw, treated, and bleached from pine cone fibers are shown in Figures (4a and 4b). The three components exhibit different behaviors in the TG and DTG profiles. From Figure 4(a), the thermal decomposition of nature is divided into three main stages. For all curves, the first stage in the temperature range 25–140°C, this weight loss is mainly attributed to evaporation adsorbed and volatiles.47,51 Due to differences in the chemical structure among cellulose, hemicelluloses, and lignin, they presented different thermal stabilities. Stage third in the temperature range 200–400° C, as indicated in Figure 4(b), is related to the decomposition of hemicellulose and cellulose degradation, and stage second stage only appears to raw fibers in the temperature range 200–300° C is related to range to the decomposition of hemicellulose. 55 attributed this event related to decarbonylation and decarboxylation, and produced gas products such as CO, CO2, and CH4; the glycan backbone glycoside bond of hemicellulose was destroyed. After bleaching treatment in Figure 4(b), the second event disappeared, showing efficient alkaline treatment to remove the hemicellulose fraction. The main degradation peak also did not vary with treatment; it is peak attributed to cellulose, in the temperature range the 200−600°C, related to the final degradation of lignin with the respective cellulose and hemicelluloses chars. 56
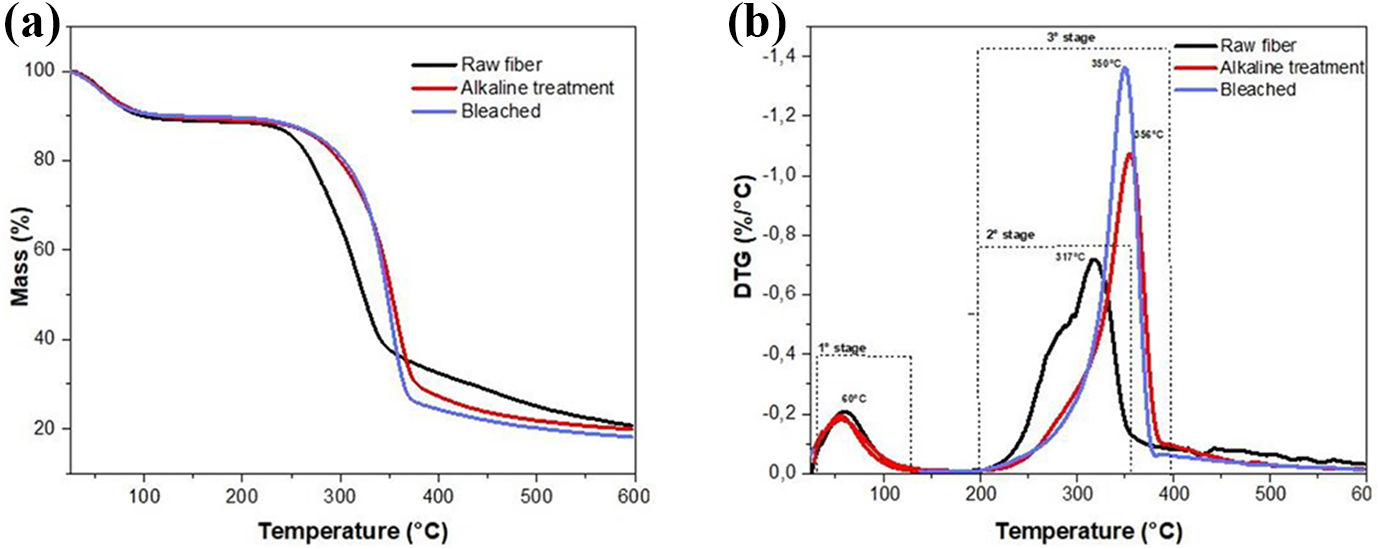
TGA and DTG of raw, NaOH treatment, bleached fibers from a pine cone.
For all samples, the third stage is related to the degradation of cellulose. The DTG curve showed that the maximum temperature of mass loss rate in the third stage of in nature, NaOH treatment, and bleached occurred at 317, 356, and 350°C, respectively. Also, the thermal stability of fibers bleached increases after chemical treatments.
Preparation of composites filaments
Composite filaments have been successfully manufactured to ensure that printing parameters, such as extrusion pressure and viscosity, do not affect the quality of the material obtained. 57 Table 2 evidences a slight variation in the filament diameters, as well as in processing temperature.
Composition, diameter, density, and processing temperature of filaments.
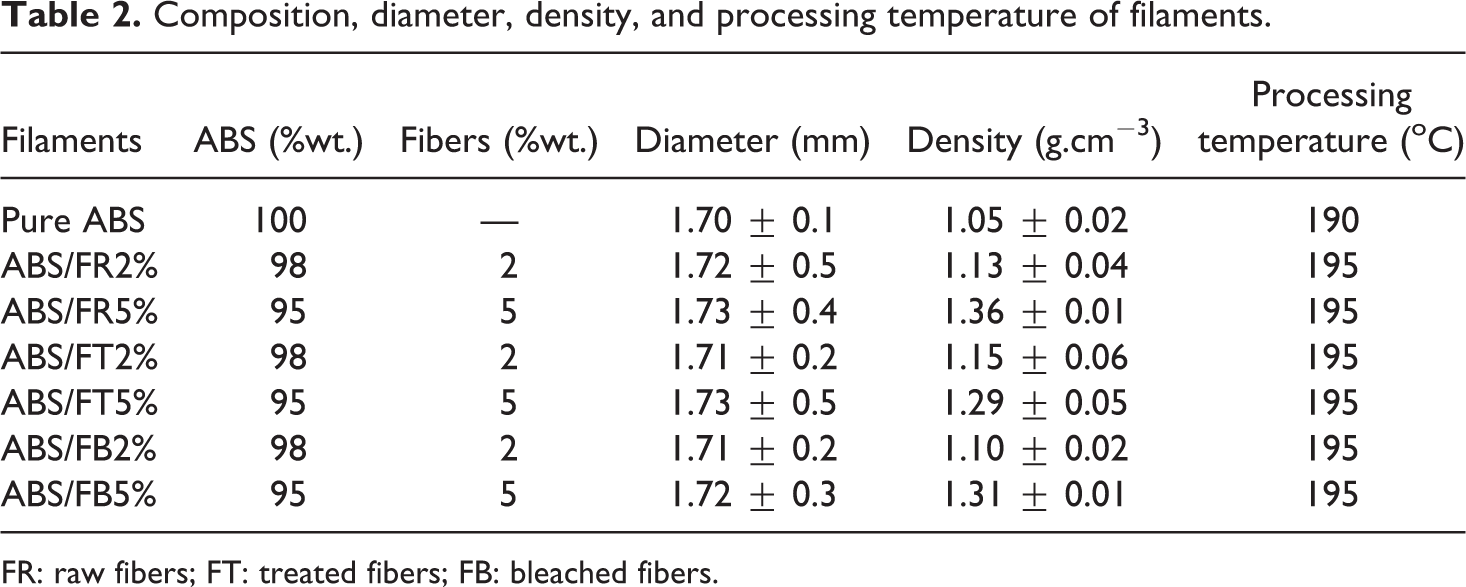
FR: raw fibers; FT: treated fibers; FB: bleached fibers.
The filament diameter produced varied for non-reinforced ABS from 1.605 to 1.615 (standard deviation of 0.005) mm, and reinforced filament showing no trend with fiber content from 2.6 to 3.1 (standard deviation varying from 0.08 to 0.23) mm. The relatively gentle coiling process allowed sufficient time for the filament to solidify and retain the intended geometrical profile.
It was observed in the filaments a variation in color due to the amount of fibers reinforced to the ABS and the type of fiber. Extruded composite filament with raw fiber has a color like wood (ABS/FR), as shown in Figure 5. A similar color was observed in extruded composite filament with treated fiber (ABS/FT). However, a color change was noted in extruded composite filament with bleached fiber (ABS/FB). Osman et al. 19 have observed similar diameters when investigating ABS-rice straw composite filament by FDM.

Extruded composites 3D filament for a 3D pen.
Characterization ABS/fibers from pine cone filaments composites
Morphological analysis
Figure 6 evidence micrographs obtained by SEM, showing the cross-section of composite filaments filled with different different fiber (raw, treated, and bleached) weight fraction, as well as pure ABS filament. It was possible to note the round shape of the filament cross-section, whose nominal diameter was between 1.70 to 1.73 mm. As observed, the pure ABS filament presents a solid cross-section without visible voids. However, the addition of the fibers leads to visible porosities in the filament, which get increasingly larger as the fiber content increases. Besides, non-impregnated matrix particles appear in the composite filaments. Also, it was observed potential stress concentration regions, voids, and small pores are formed around these particles, which could affect the properties by generating the possibility of microcrack formation. These voids can be caused by fibers pulling out after the cryo-fractured process. In the magnified micrographs (2000×), it is noticeable that fiber is uniformly distributed and embedded throughout the wire cross-section, and some voids are caused by fibers pulling out after the cryo-fractured process.

SEM micrographs of cross-section of (a1–a2) ABS; (b1–b2) ABS/FR2%; (c1–c2) ABS/FR5%; (d1–d2) ABS/FT2%; (e1–e2) ABS/FT5%; (f1–f2) ABS/FB2%; (g1–g2) ABS/FB5% composites filaments.
In the magnified micrographs (200×) of Figure 6 (a2–g2) was perceptible that bleached fiber is uniformly distributed and embedded throughout the wire cross-section when compared to treated and untreated fiber added to ABS. Thus, it is clearly shown that the ABS involved the bleached fibers in the magnified images, indicating good interfacial bonding between the bleached fiber and the ABS matrix.
Nguyen et al., 58 present similar micrographs to reinforce a melt-stable lignin waste-stream from biorefineries as a renewable feedstock, with acrylonitrile-butadiene rubber and acrylonitrile-butadiene-styrene (ABS) polymer to synthesize a renewable matrix having excellent 3D-printable. 58
Osman et al. 19 have observed similar micrographs when investigating ABS-rice straw (RS) composite filament by FDM. The micrographs show how the RS was distributed throughout the matrix and illustrate an increasing amount of porosity as RS increased. 19
Sang et al. 59 also presented similar micrographs reinforcing treated basalt fiber (KBF) to polylactide (PLA) for 3D-printed feedstock. It was also clearly seen that the PLA encompassed the fibers showed good interfacial bonding between KBF and PLA matrix. 59
Rahimizadeh et al. 60 evidenced similar micrographs with short glass fibers from scrap turbine blades reclaimed and mixed with polylactic acid (PLA) through a double extrusion process to produce composite feedstock with recycled glass fibers for fused filament fabrication (FFF) 3D printing. 60
Thermal characterization
The thermal stability of ABS, FR, FT, FB, and its filament composites were evaluated by thermogravimetric analysis, as can be observed in Figure 7 all TG/DTG curves. The curves related to FR, FT, and FB show two events of weight loss; the first one from room temperature up to 130 ºC is attributed to water evaporation, while the second one relates to the degradation of hemicellulose, lignin, and cellulose. 17
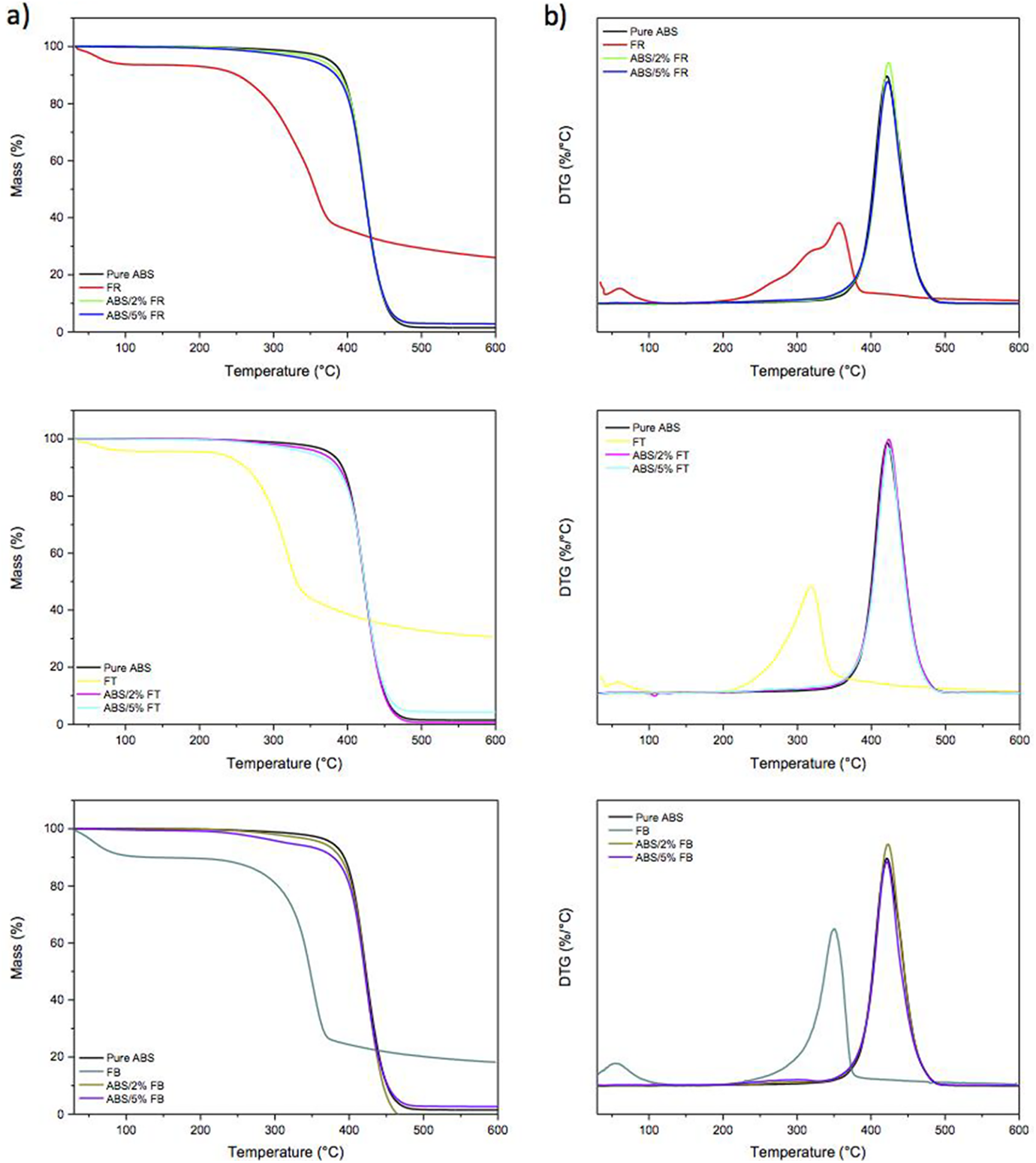
a) TG and b) DTG curves for ABS, FR, FT, FB, and its composites filaments.
Two main steps in the degradation of ABS/fiber composites were observed. The first step, between 245–399oC, can be attributed to the fiber degradation. The second degradation event occurs above 420oC, and it is related to ABS degradation that leads to the formation of a charred residue. 61 The type fiber influenced in curve causing the highest amount of char. Besides, both degradation T onset and temperature of the maximum rate of weight loss of the composites are very close to the values shown by pure ABS, except for ABS/5%FB, which exhibited T onset closer to its reinforcing fiber. Additionally, ABS samples incorporated with the fibers showed residue mass at 600 ºC directly proportional to the fibers’ content. It has been previously reported by Martins et al. 61 that fiber composites based on ABS and acrylic fiber, cotton fiber, and waste textile fiber had a similar effect on the degradation behavior of ABS, where the composites with fiber addition up to 30 wt.% had little effect on the ABS matrix degradation behavior. Table 3 summarizes the results extracted from TGA, DTG, and DSC curves.
TGA and DSC results for ABS, FR, FT, FB, and its composites: glass transition temperature (Tg), onset temperature (T onset ) of the degradation process, the temperature of the maximum rate of weight loss (T d ), mass loss temperature data from TG curves and amount of residue at 600 ºC.
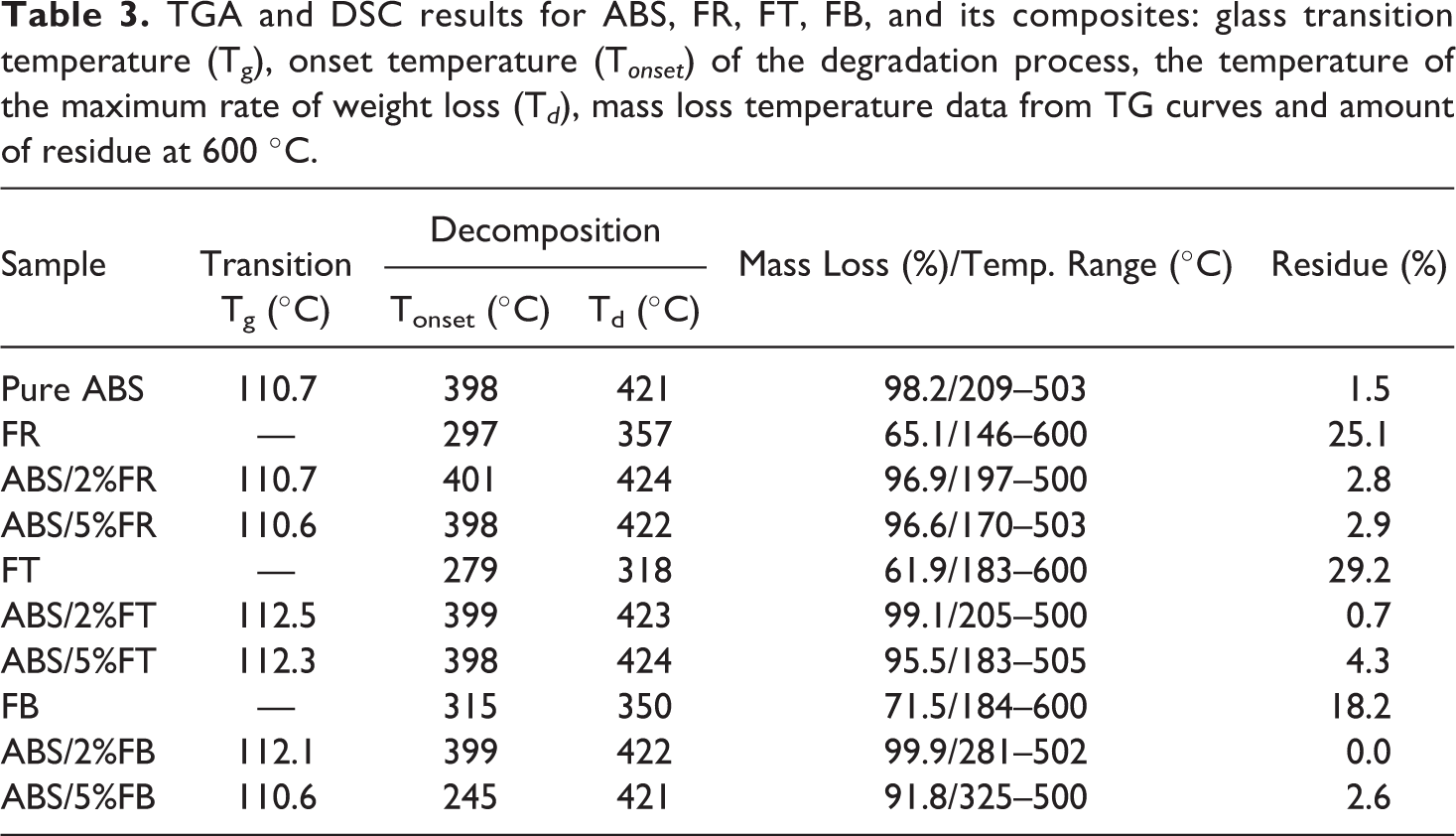
Figure 8 shows DSC curves for ABS and its filament composites. The shape of the DSC curves obtained for ABS and its composites shows the same profile. The step-change of heat flow in the range of 90–120 ºC indicates the materials’ glass transition temperature, which represents the transition from the glassy to rubbery phase.35,62 The neat ABS Tg was determined at 110.7 ºC, as previously reported. 62 The corresponding Tg of the composites was only slightly affected by the pine cone fibers’ addition; it was lower than the neat ABS only for ABS/5%FR and ABS/5%FB. The endothermic peak around 422 ºC relates to ABS decomposition, as stated by TG/DTG. Once the use of the studied fibers as a reinforcing agent did not interfere appreciably with ABS’s thermal properties, the insertion of these natural materials in the applied concentrations does not affect the processability and printability of ABS polymer.
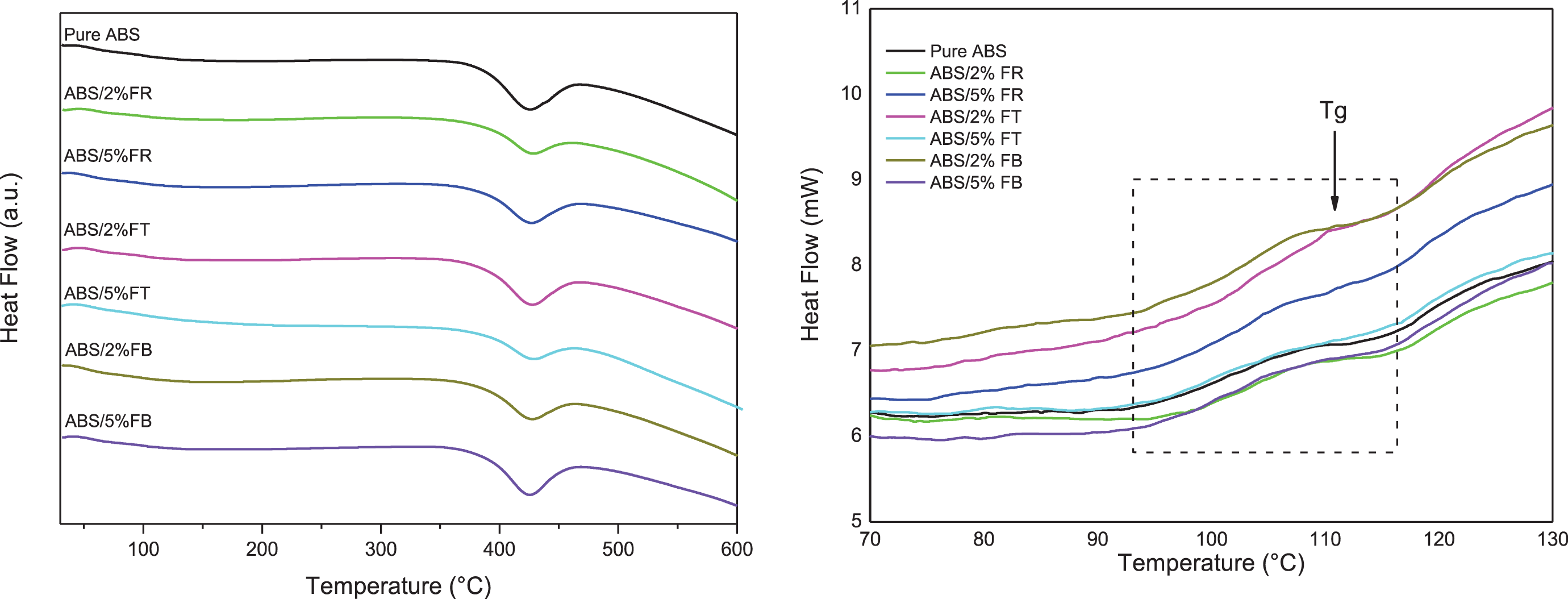
DSC curves for ABS and its composites.
FTIR
FTIR analysis is efficient to analyze the chemical structure of pure ABS and composites, analyzing the existence of new covalent or hydrogen bonds. Figure 9 shows FTIR spectra of ABS and its composites filaments. The FTIR spectra of ABS and its composites show the same profile—the bands are attributed to the ABS matrix. The absorbance bands characteristics of ABS are in the range of 3100–2800 cm−1 (C-H stretching of CH, CH2, and aromatic rings), ∼2238 cm−1 (C≡N stretching), ∼1600 cm−1 (cis-1,4-C=C stretching), and ∼ 1450 to 1500 cm−1 (CH bending of aromatic ring). Finally, the bands at 966 cm−1 related to (trans-1,4-C=C stretching), 760, and 700 cm−1 related to the mono-sub phenyl group. 63
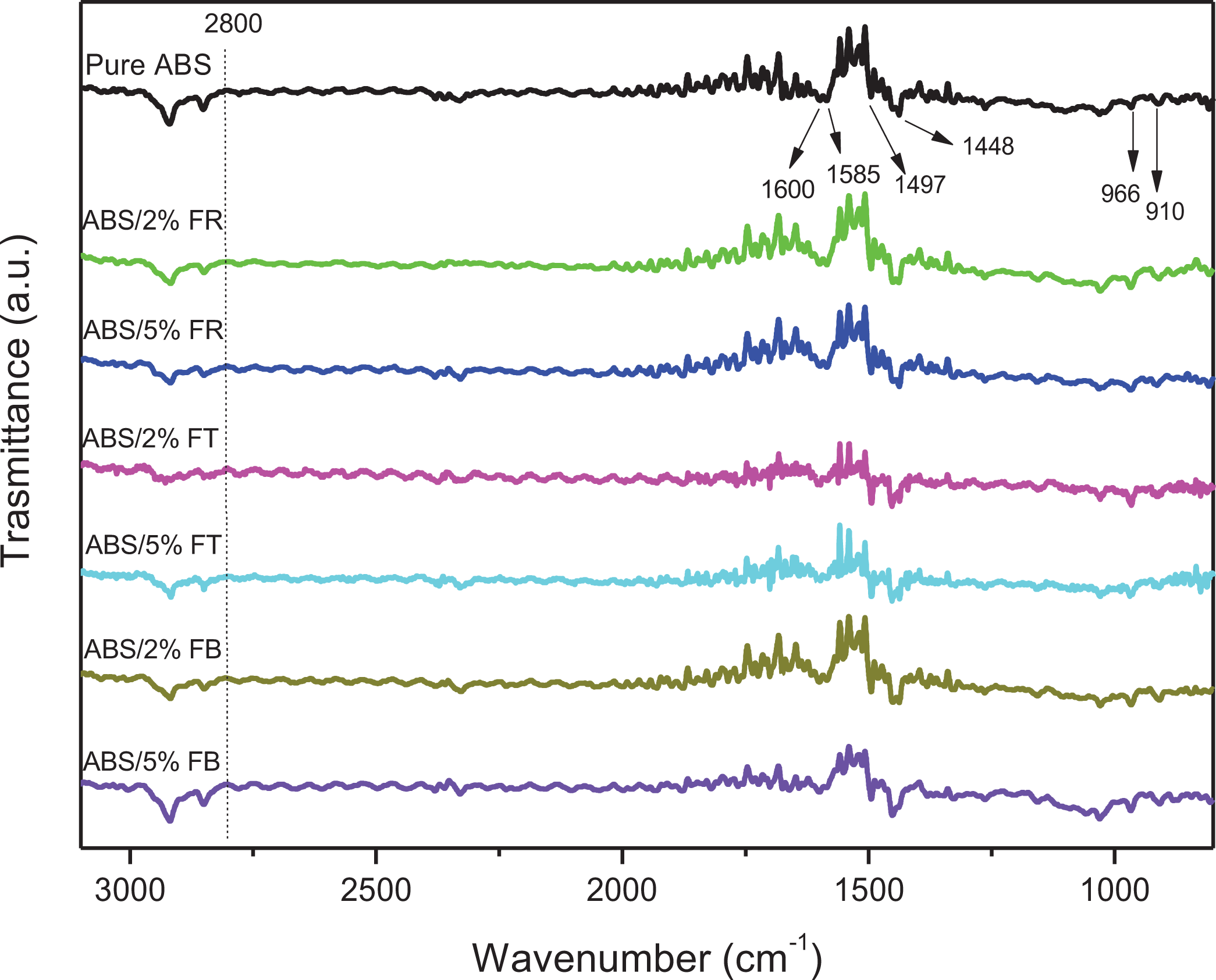
FTIR of ABS and fibers composites filaments in the range of 3100–800 cm−1.
Considering the composites, a change C=C peak frequency increased with the addition of fibers. It was also observed an increase in the peaks at 2930 cm−1, corresponding to C–H stretching vibrations. However, ABS/FB% exhibited hydrogen bonding slightly compared to the ABS/FR% as shown in the FTIR data. 64
Printing 3D pen
Composite filaments were tested in the printing 3D pen. All filaments presented satisfactory results (regarding visual appearance, shape, and stability). Figure 10 presents all 3D pen printable filaments with 2 and 5% wt. For future tests, the intention will be to reduce the fiber’s size to increase the composites’ amount of reinforcement. The literature shows many jobs with a low amount of reinforcement due to clogging caused by the printer nozzle.62,65,66

Printing 3D pen of composite filaments.
Conclusion
In this research, the effect of the treatment chemical of the fibers from pine cone as reinforcement in ABS composites filaments as a potential for the 3D pen was evaluated. Results showed that the type of fiber (raw, treated, and bleached) was influenced by thermal stability and morphological analysis of filaments and the amount of fibers inserted into the ABS. However, this fact not significantly influenced the diameter and density of the filament. The addition of bleached fibers to ABS increases composites’ thermal stability compared to other fibers (treated and raw). Besides, the addition of the bleached fibers to ABS showed a uniform distribution and embedded throughout the wire cross-section compared to treated and raw fiber added to ABS due to good interfacial bonding confirmed by FTIR technique, which indicated that fibers were hydrogen-bonded to ABS chains, causing an increase the filament’s density. The addition of fibers from pine cone to ABS thermally improved and can be a low-cost feedstock for printing 3D pen applications. Also, the ecological aspect of reusing industrial residue provides a second useful life.
