Abstract
With the application of hot-pressing technology, polyamide 6 (PA6) was successfully directly adhered to the aluminum alloy. The relationship between the bonding property and microstructures of PA6 and anodized AA5754 aluminum alloy hybrid were explored in the paper. The results showed that the honeycomb-like porous oxide film was prepared by anodic oxidation, which resulted in the decreased surface energy of the aluminum alloy, so that PA6 easily wetted on the aluminum alloy surface, and the maximum tensile shear strength and fracture work reached 8.24 MPa and 9.27 kJ m−2, respectively. Improved bonding strength was attributed to micro- and nanoscale honeycomb-like microstructures and large roughness on the anodized aluminum alloy surface.
Introduction
Recently, the world has been confronted with increasingly serious problems such as energy consumption and environmental protection with the booming automobile industry, hence the lightweight materials and their processing technology are becoming more and more important for the auto industry. Lightweight materials mainly involve high-strength steel, aluminum alloy, plastic, and so on. Among the materials, plastic–metal hybrid (PMH) products meet the comprehensive performance requirements of vehicle parts; therefore, the PMH technology has attracted many researchers’ interests at present.
So far, several PMH technologies have been developed including compression molding, injection molding, and other fusion bonding methods, of which multiple molds and/or foreign applied loads have been additionally utilized to join plastic to metal. Yousefpour et al. 1 summarized the influence and application of welding technology and corresponding manufacturing methods of various thermoplastic materials on welding performance and quality. Abibe et al. 2 realized the connection of glass fiber-reinforced polyamide 66 (PA66/FG30%) and aluminum AA2024-T351 by using injection riveting technology and discussed the mechanical, microstructure, and thermal properties of the joint. Zhou et al. 3 studied high-modulus carbon fiber-reinforced polymer (CFRP)/AZ31 magnesium alloy sheet hybrids prepared by the hot-pressing process, and the results showed that the CFRP/AZ31 composite has higher tensile strength, flexural rigidity, and specific modulus than the traditional magnesium matrix hybrid. Yeh and Hsu 4 treated A5052 aluminum alloy sheet surface with chemical etching and atmospheric plasma, respectively, which was used to bond with polycarbonate (PC)/ acrylonitrile butadiene styrene (ABS) during injection molding. They found the chemical change affects bonding strength significantly for the hybrids, and the bonding strength is improved 2.5 times after the two-step process of chemical etching and plasma treatment. Liu et al. 5 fabricated aluminum alloy/PA66 joints by friction lap welding (FLW). X-ray photoelectron spectroscopy was used to analyze the chemical bonds at the interface of aluminum alloy/PA66, which shows the chemical bond is a key factor to obtain good joint strength for the PMH hybrid. In addition to these technologies, the coupling agent such as silane has been successfully used as a primer coating to direct bond polymers such as the production of laptop shell. However, the adhesive properties of coupling agent bonded products are susceptible to the environment temperature, acid and alkali conditions, usually resulting in interface bonding failure. Meanwhile, to further improve bonding performance of the PMH part, a variety of metal surface treatments such as chemical etching, anodizing, sandblasting, plasma treatment, and laser irradiation are widely adopted to treat the metal surface. Honkanen et al. 6 reported the effect of silane treatment on the bonding properties of thermoplastic polyurethane and stainless steel hybrids and found that the hybrid failure mode depends on the silane layer thickness. For example, hybrids with a thick silane layer fail mainly in the silane layer and a thinner layer cohesively in plastic. Byskov-Nielsen et al. 7 investigated the mechanical strength of the joint relies on the laser-irradiated metal surface morphology, and the appropriate surface structuring can increase the stable bonding strength by several orders of magnitude. Zhang et al. 8 used to join aluminum alloy and carbon fiber-reinforced plastic. The results indicate that the pretreatment of A7050 with the laser process could improve the shear strength of the dissimilar joint to a large extent. Pan et al. 9 annealed and anodized seawater aged AA5083 aluminum alloy sheet and then jointed with carbon fiber/epoxy laminate. The reports reveal the porosity and pore size of the anodic oxide film decrease with the application of annealing technology, which results in weak interface between metal and composite laminate. He et al. 10 studied the effect of anodizing temperature on the surface chemical composition, microstructure, and morphology of Ti6Al4V matrix and concluded the nanoscale honeycomb pore structure helps to improve the bonding strength of epoxy resin and Ti6Al4V hybrid. Nagatsuka et al. 11 reported the carbon fiber-reinforced PA6 and A5052 aluminum alloy is successfully jointed by the FLW. They found that the surface of A5052 aluminum alloy covered with a layer of aluminum hydroxide increases the tensile shear strength of the joint. Bergmann and Stambke 12 bonded the glass fiber-reinforced nylon 6 (PA6-GF45) to the sandblasted aluminum alloy surface through a laser-based welding process and indicated there is no correlation between the shear strength of the joint and the surface roughness parameter Ra. Arkhurst et al. 13 discovered annealing treatment plays a significant role in the joint strength of AZ-MG alloy with carbon fiber-reinforced polyurethane. Among these metal surface treatment methods, anodic oxidation has been developed as one of the most promising surface treatment methods to produce relatively uniform microstructure oxidation film, resulting in excellent stable bonding strength with polymers and their composites.
This article mainly focused on the bonding strength of the anodized aluminum alloy jointed with PA6 by hot-press molding technology. The mechanical properties of the joint samples and their failure mechanisms are explored and analyzed by tensile shear strength, the contact angle test, atomic force microscopy (AFM), energy-dispersive X-ray spectrometry (EDX-S), and scanning electron microscopy (SEM), respectively.
Experimental
Materials
An aluminum alloy (grade A5754) used in the experiment was provided by Southwest Aluminum Group Co., Ltd (China), which chemical components are 2.3% Mg, 0.5% Mn, 0.4% Si, 0.4% Fe, 0.3% Cr, 0.2% Zn, 0.1% Cu, 0.05% Ti, and the rest aluminum, and its rectangular specimen dimension was 100 × 25 × 2 mm3. PA6 (grade YH800) was produced by Hunan Yueyang Chemical Co., Ltd (China).
Sample preparation
The rectangular aluminum alloy samples were ground with 200#, 400#, 600#, and 800# abrasive papers successively, polished with SiO2 polishing paste, ultrasonically cleaned with distilled water and acetone, and next treated with sodium hydroxide solution, nitric acid solution, and water in sequence. After the surface-treated sample was dried, a 5 wt% of the phosphoric acid solution was prepared as an electrolyte and the ampere density was set at 8 A dm−2 during anodic oxidation processing. The oxidation time was set to 40 min and the oxidation temperature was set to room temperature. The PA6 was dried in a drying oven at 100°C for 8 h, and the dried PA6 was injection molded as dumbbell specimen. The PA6 and aluminum alloy single-overlap joint is prepared via micro-nano hot-press technology (MNHPT) and the MNHPT processing parameters are listed in Table 1. The schematic diagram of hot-pressing setup and self-made mold is shown in Figure 1(a) and (b), respectively. The schematic diagram of single-overlap joint specimen is shown in Figure 2, and the overlapping area is 10 × 12.5 mm2. The labeled samples with different treatments are listed in Table 2.
The parameters of the micro-nano pressing technology.
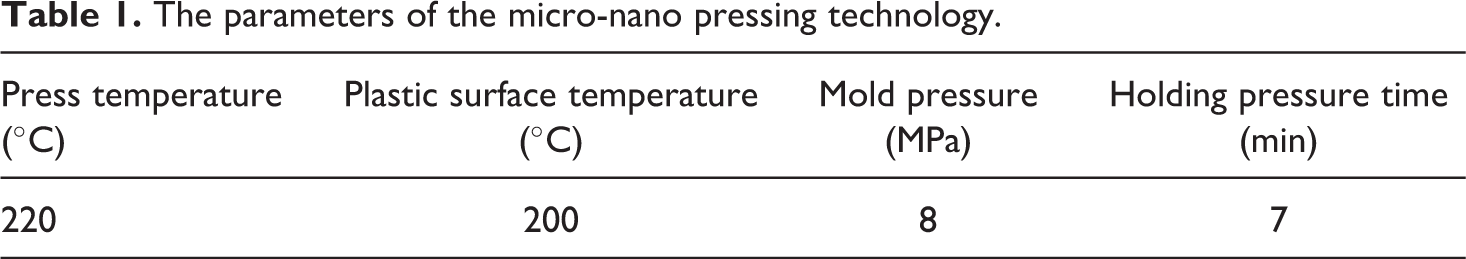
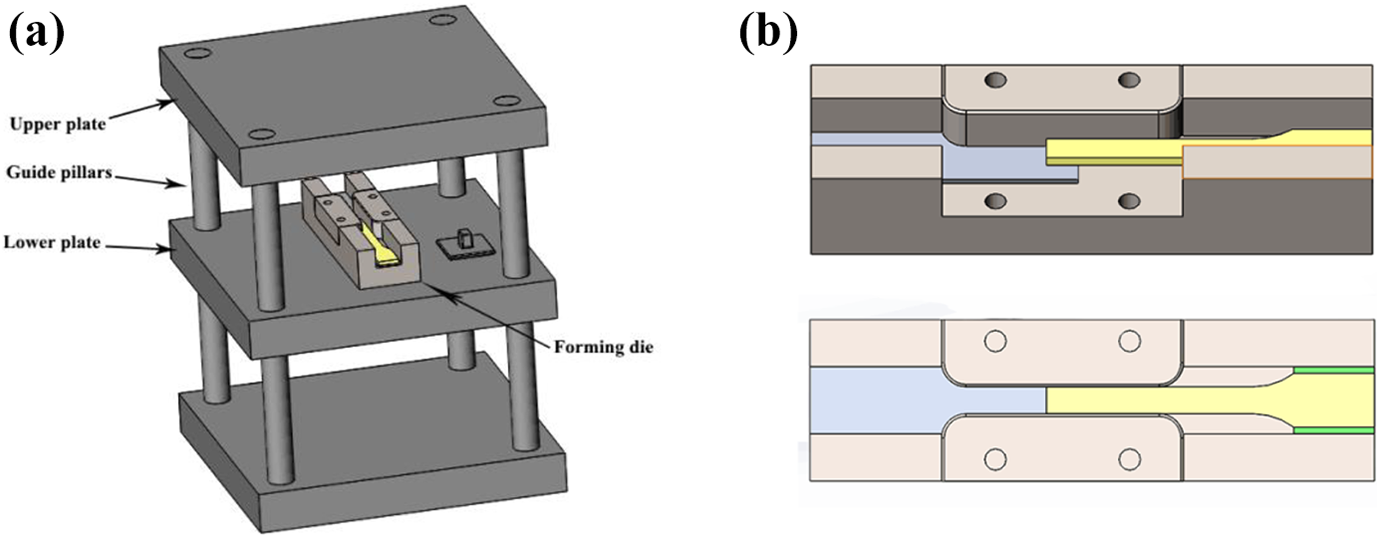
The schematic diagram of (a) hot-pressing setup and (b) self-made mold.
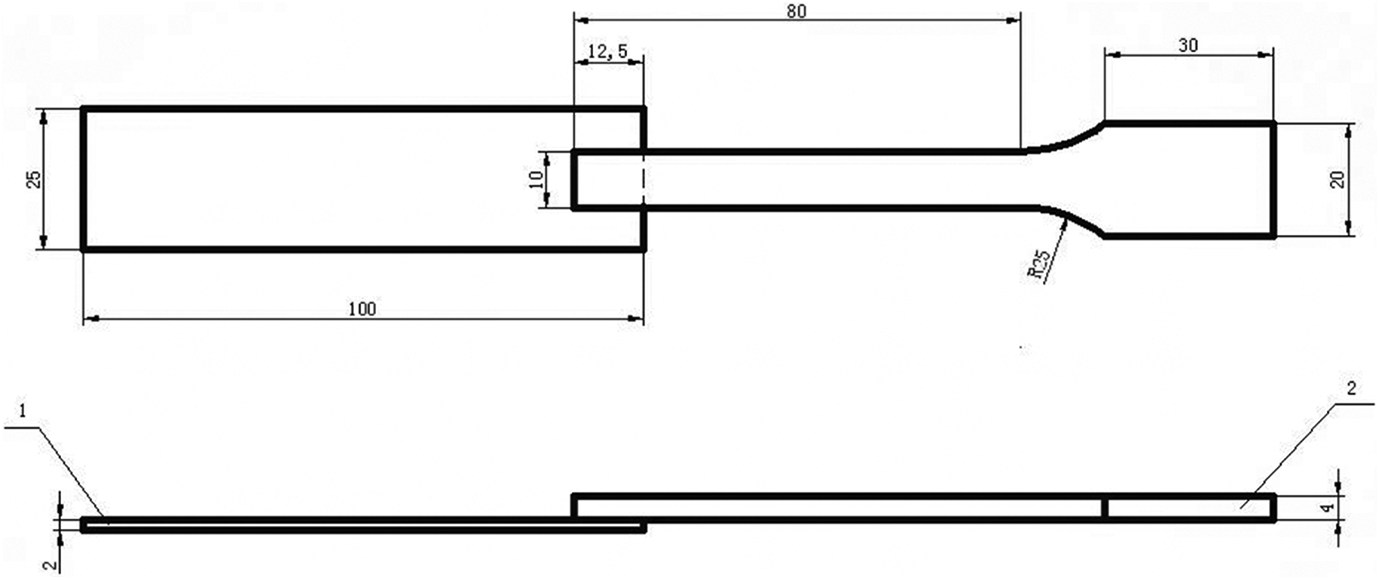
The schematic diagram of single-lap joint sample with dimensions: 1-aluminum alloy sheet and 2-a half of plastic dumbbell specimen.
The aluminum alloy samples with different treatments.
PA6: polyamide 6.
Tensile shear test
The tensile shear tests of single-lap joint samples were carried out using a universal testing machine (CMT 6104, MTS Systems Corporation, China) at a moving rate of 2 mm min−1 and room temperature. The load–displacement curves and shear tensile strength were recorded, and the work of fracture was calculated by the failure area according to the load–displacement curve.
Structure and morphology investigation
The surface-treated aluminum alloy sheets were observed by SEM (JSM-6460LV, JEOL, CO., LTD., Japan). The topograph of the surface-treated aluminum alloy sheet was subjected to AFM (Park NX10, Park Systems, Korea) in the non-contact mode to explore surface microstructure and morphology, and the scanning area was 5 × 5 μm2 in the selected overlapping area. After the tensile shear fracture failure, the failure surface of aluminum alloy samples was observed under a scanning electron microscope.
Surface chemical component analysis
To analyze the chemical component of the aluminum alloy surface, the surface-treated aluminum alloy samples were subjected to EDX-S test using Cu target Kα ray with a power of 3 Kw, the tube current and voltage of 20 mA and 35 KV, respectively, and the scanning speed of 0.02° s−1.
Contact angle test
The contact angle was explored on the aluminum alloy surface, and the water and diiodomethane were used as test liquids individually to describe the surface wettability of the surface-treated aluminum alloy sample. The surface free energy parameters of the test liquids are shown in Table 3. According to the Owens–Wendt–Rabel–Kaelble theory proposed by Owens and Wendt, 14 the surface free energy was calculated by the formula as follows

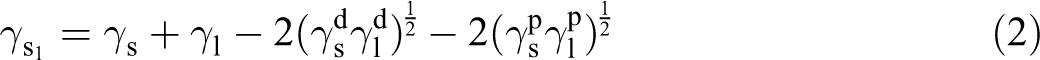
Combine equations (1) and (2) with the Young equation to get
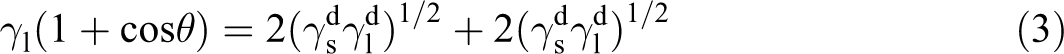
where
Surface free energy parameters of the test liquids.

Results and discussion
Surface topography
To observe the surface topography of aluminum alloy samples, the SEM and AFM photos are displayed in Figures 3 and 4, respectively. As shown in Figure 3(a) and (b), there are almost no micropores on the polished aluminum alloy surface, which presents the polishing lines and traces on the polished aluminum alloy surface. This relatively flat of the polished aluminum alloy surface plays little roles in joining plastic resin to aluminum alloy surface. As shown in Figure 3(c) and (d), there are many relatively uniform dispersed needle-shaped honeycomb-like micropores with obvious boundaries on the anodized film, and the average pore size range is from 100 nm to 140 nm, which obviously provide a good micro-nano interlocking structure for aluminum alloy and plastic interface bonding.

SEM images of the polished and/or anodized aluminum alloy surfaces: (a and b) P-Al and (c and d) P/A-Al.
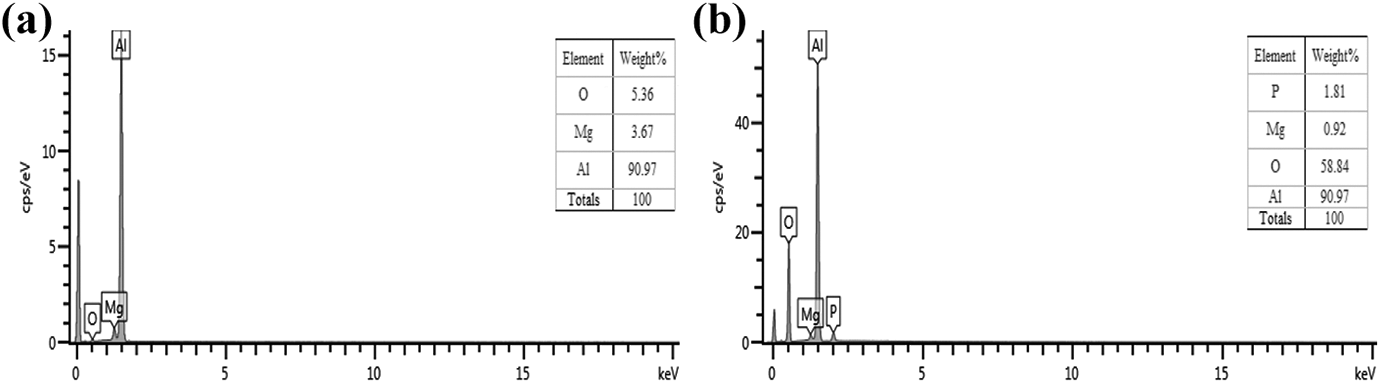
The quantitative analysis of EDX-S elements of the different surface-treated aluminum alloy samples: (a) P-Al and (b) P/A-Al.
The EDX-S element analysis of aluminum alloy surfaces after polishing and anodizing treatments is shown in Figure 4. For the polished aluminum alloy surface, only three elements (O, Mg, and Al) were detected, while for the anodized aluminum alloy surface, the P element was observed due to the anodized phosphate residues, and obviously the oxygen element percentage of the anodized aluminum alloy surface increases greatly and reaches 58.84%. Compared with the P-Al/PA6 joint, the tensile shear strength and fracture work of the P/A-Al/PA6 hybrid sample are greatly improved, which can be further indicated that the oxide layer formed on the aluminum alloy surface is beneficial to improve the interface bonding strength to some extent. In related reports, Arkhurst et al. 15 found that the oxide layer introduced on the surface of the magnesium alloy by annealing has a significant influence on the joint strength of the Mg alloy/CFRP hybrid.
To clearly investigate the microstructure of the aluminum alloy surface, the AFM observation was applied to confirm nanoscale honeycomb porous film on the anodized aluminum alloy surface, as displayed in Figure 5. The detected AFM parameters for the surface-treated metal samples are listed in Table 4. The roughness (Ra) is around 2.3 nm and parallel scratch depth is around 16 nm on the P-Al surface. After phosphoric acid anodized, as shown in Figure 5(c), the mean surface roughness (Ra) is around 51 nm, which is 22.2 times against that of P-Al sample, and the hole depth reaches up to around 220 nm. It can be concluded the anodizing treatment increases the surface roughness of the aluminum alloy to provide a micro-nano interlocking structure for the metal to plastic bonding, accordingly improving the bonding strength of PMH.
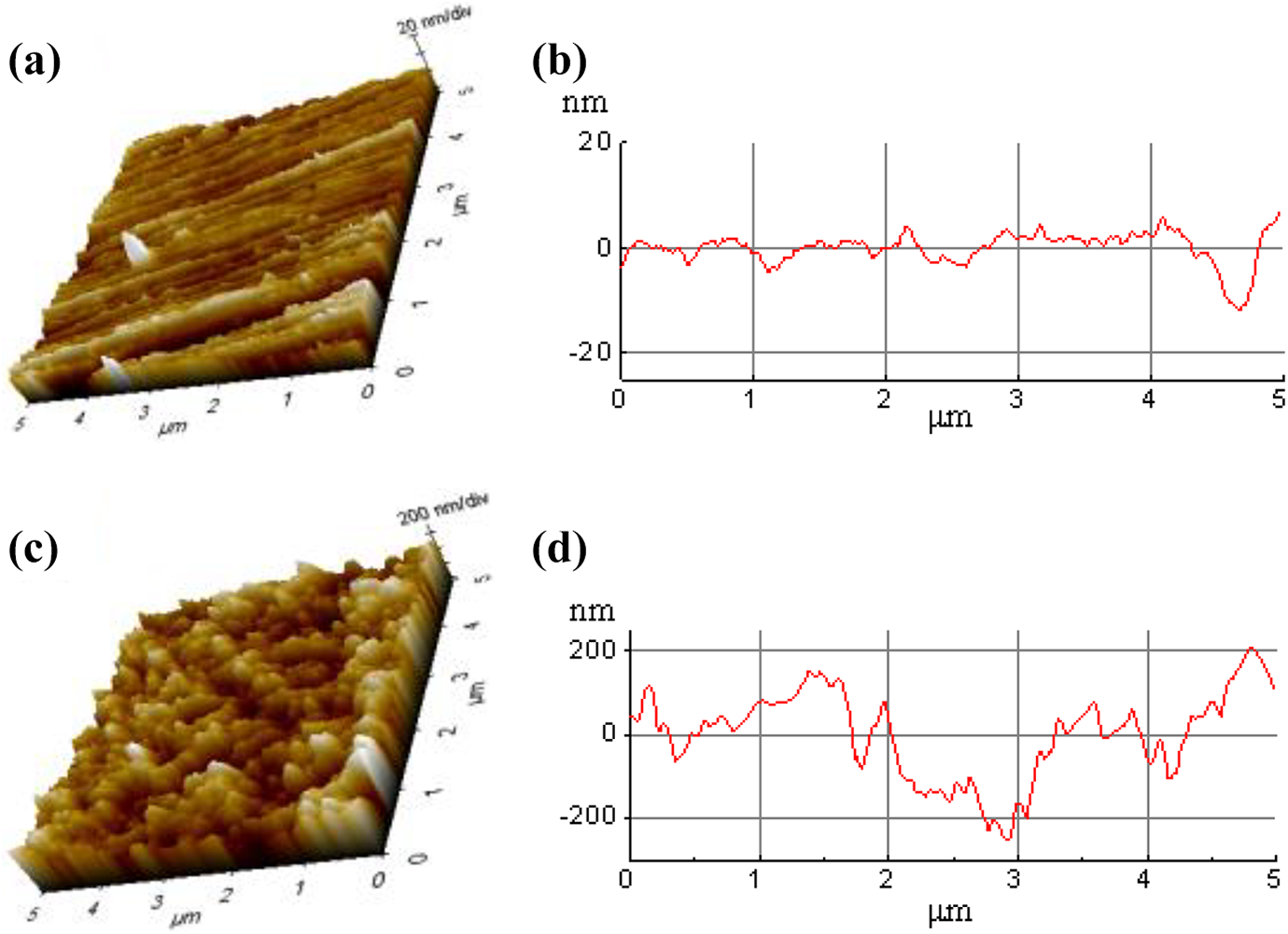
The 3D microstructures of the surface-treated aluminum alloy samples: (a and b) P-Al and (c and d) P/A-Al.
The AFM parameters of the surface-treated aluminum alloy samples.
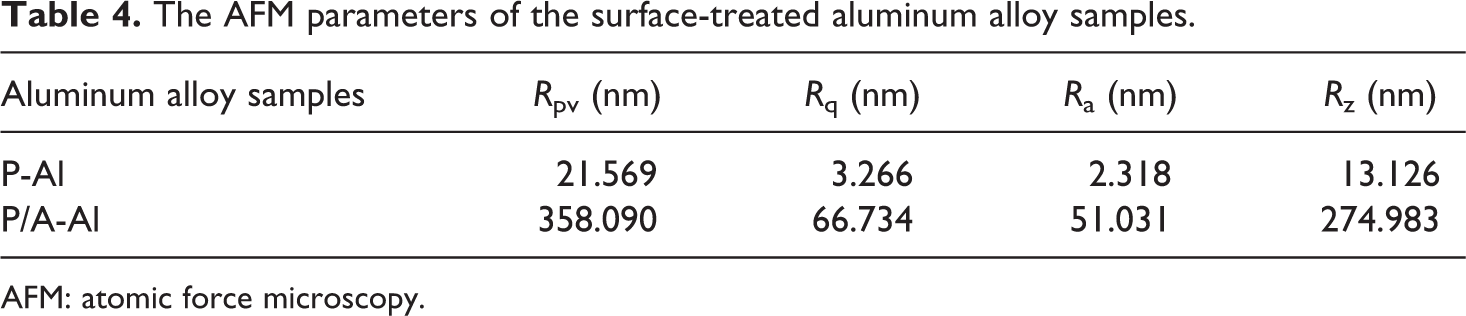
AFM: atomic force microscopy.
Tensile shear strength test
The typical load–displacement curves of single-lap joint specimens are shown in Figure 6. It is observed that the displacement and maximum load is only 1.4 mm and 246 N, respectively, when the P-Al/PA6 interface debonding failed. The maximum load of P/A-Al/PA6 sample reaches sharply up to 1032 N with increased elongation at break. This result is due to the anodized rough topography helps polymer melt flow into the micro holes forming mechanical interlocking microstructure during cooling down to strengthen the A5754/PA6 hybrid.
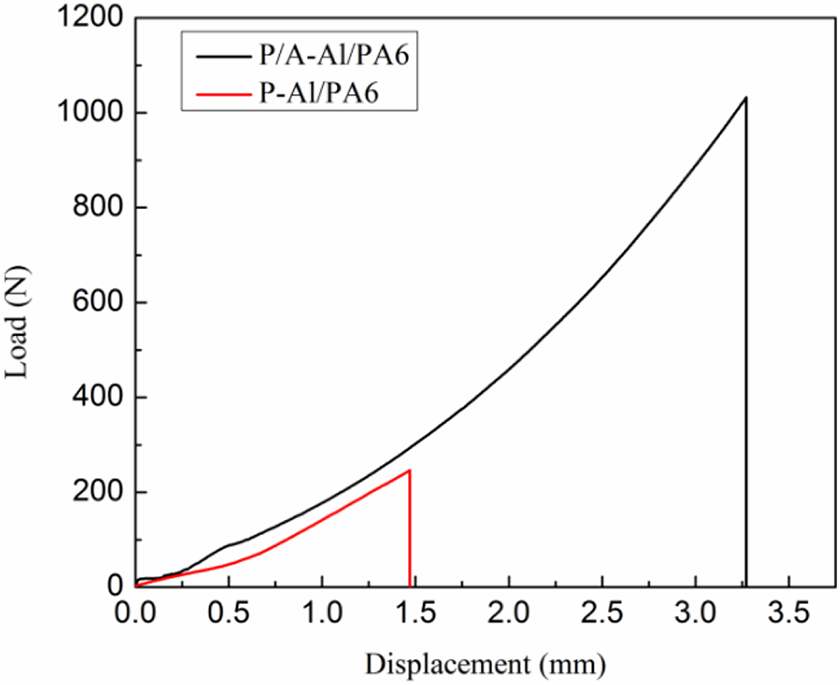
The typical load–displacement curves for single-lap joint specimens.
The tensile shear properties of aluminum alloy A5754/PA6 MNHPT samples are demonstrated in Figure 7. As can be seen from Figure 6, for the polished A5754 aluminum alloy plate, the shear strength and work of fracture are 1.97 ± 0.21 MPa and 1.23 ± 0.14 KJ m−2, respectively, against 8.24 ± 0.56 MPa and 9.27 ± 0.53 KJ m−2 of the anodized MNHPT sample, with corresponding increase percentages of 318% and 654%, respectively.
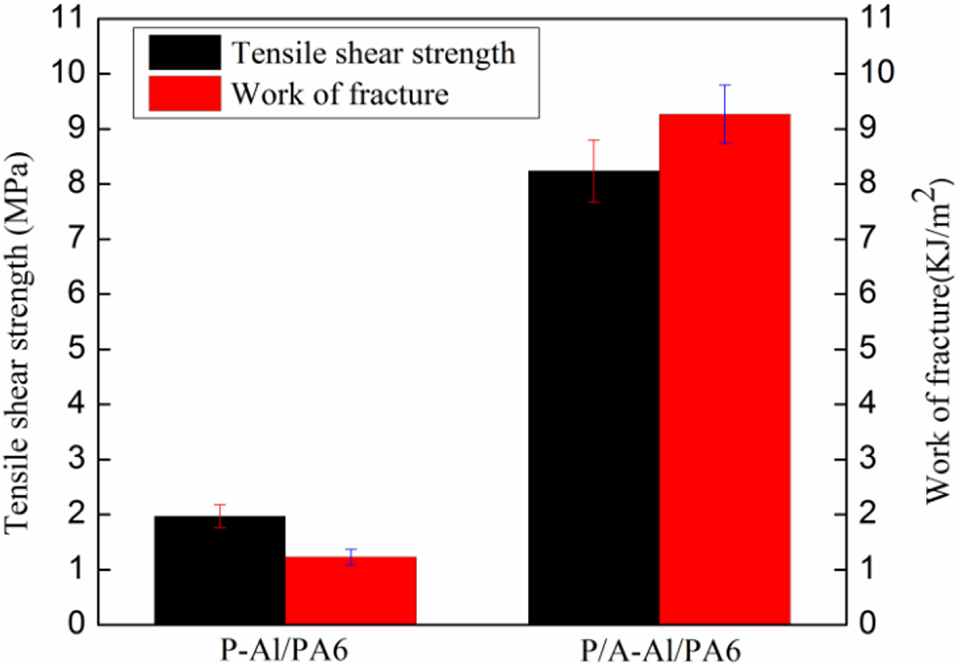
The tensile shear strength and fracture work of the different MNHPT samples.
Fracture failure analysis
The SEM images of A5754 aluminum alloy surfaces after tensile shear failure are shown in Figure 8, which are used to determine the fracture failure mode. Figure 8(a) and (b) shows a relatively flat failure surface with nearly no PA6 residues on the polished aluminum alloy surface, which indicates the interface bonding between PA6 and polished aluminum alloy mainly depends on the effect of van der Waals force. However, a great amount of PA6 residue on the anodized aluminum alloy surfaces was shown in Figure 8(c)ed and (d), which proved that the PA6 melt entered the nano and micro holes of the anodized surface to form the interlocking microstructures with corresponding strong A5754/PA6 hybrid. Therefore, it can be deduced that polymer resin cohesive failure mainly occurs on the anodized aluminum alloy surface, and layer interface failure exists on the polished aluminum alloy surface.

SEM images of different A5754/PA6 hybrids after failure tests: (a and b) P-Al/PA6 and (c and d) P/A-Al/PA6.
Contact angle measurement can be used to characterize the surface properties of common materials. The contact angles of the polished and/or anodized aluminum alloy surfaces are displayed in Figure 9. The contact angles of water and diiodomethane on the polished surface were 63.5° and 29.8°, respectively, as shown in Figure 9(a) and (b), and the corresponding values of the anodized surface were 102.2° and 50.2°, respectively, as shown in Figure 9(c) and (d). The higher contact angle means poorer wettability and lower surface free energy of the surface.
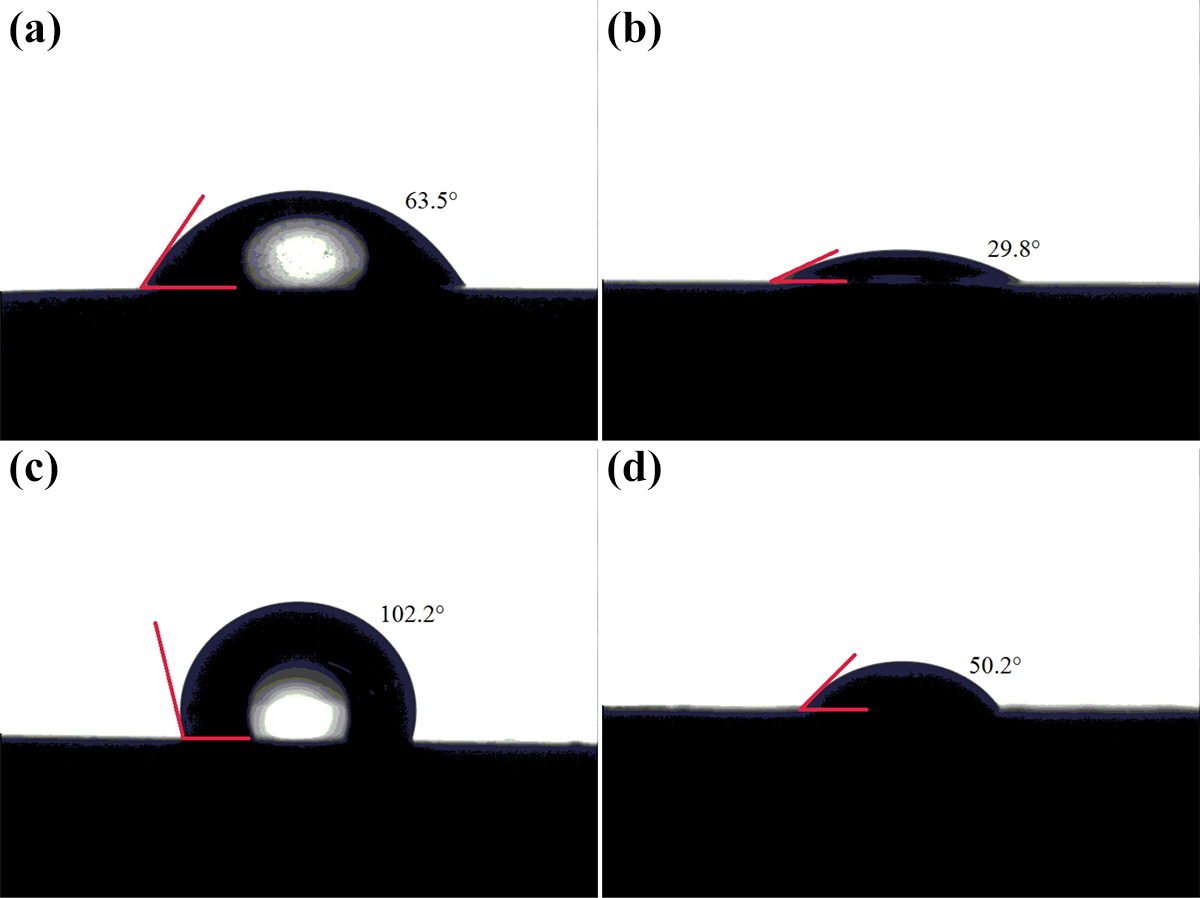
Contact angles of water and diiodomethane on the different treated surfaces: (a and b) polished surface and (c and d) anodized surface.
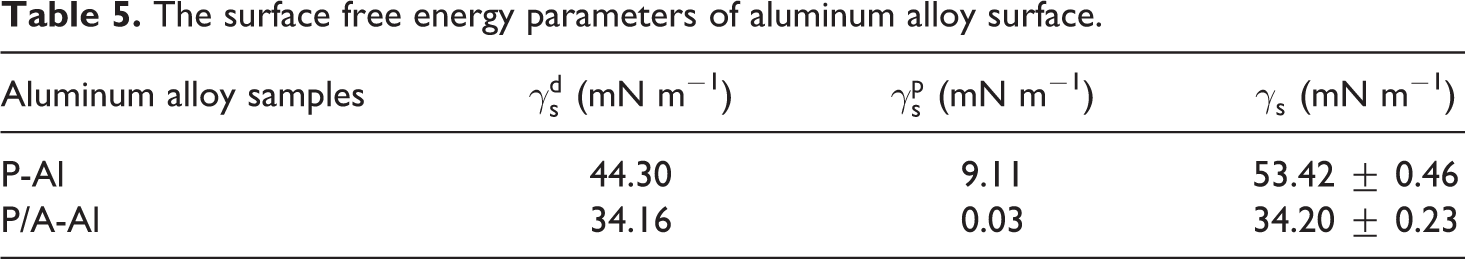
The energy parameters of the polished and anodized aluminum alloy surfaces were calculated according to Owens–Wendt–Rabel–Kaelble theory. As shown in Table 5, the free energy of the polished and anodized aluminum alloy surfaces is 53.42 ± 0.46 mN m−1 and 34.20 ± 0.23 mN m−1, respectively. When the free energy of the metal surface decreases, the surface energy polar and dispersion components significantly reduce. It is known that the plastic has generally low surface energy about 20–30 mN m−1, thus the surface energy difference decreases between anodized aluminum alloy and plastic surfaces. According to the wetting thermodynamics, a better wettability exists at the interface when two surfaces have similar surface energy. This is also confirmed by relevant research as follows. Anvar and Akbarzadeh 16 studied the structure, microstructure, and mechanical properties of roll welded AA5052 aluminum alloy/PP sheet and found the similar surface energy of plastic and metal improve the wettability of plastic on metal surface with corresponding good bonding property between the layers. With application of anodizing technology, the surface energy of the aluminum alloy decreases, which leads to the surface energy ratio between plastic and metal is nearly uniform, and hence the interfacial performance of the plastic and metal hybrid is improved consequently.
The surface free energy parameters of aluminum alloy surface.
The micrographs of the single-lap samples’ cross-sections are shown in Figure 10 to display the interface structure between aluminum alloy and PA6. Figure 10(a) shows gaps on the interface of the polished aluminum alloy and PA6, and the voids on the interface between PA6 and aluminum alloy result in a relatively low tensile shear strength of P-Al/PA6 sample with only 1.97 ± 0.35 MPa. However, PA6 bonded relatively tightly together with the anodized aluminum alloy surfaces shown as Figure 10(b), it can be observed that the anodized aluminum alloy surface is nearly completely jointed with PA6 resin. This good wettability makes the PA6 melt flow into the micro holes on the anodized surface to form the interlocking microstructure with the corresponding improved tensile shear property of P/A-Al/PA6 samples.

SEM micrographs of cross-sections for the hybrids: (a) P-Al/PA6 and (b) P/A-Al/PA6.
Conclusion
With the application of hot-pressing technology, PA6 was successfully directly adhered to the aluminum alloy, and microstructures of the PMH hybrids were explored. The main conclusions are as follows: The anodic oxidation on the aluminum alloy prepared a honeycomb porous membrane to easily bond the PA6 with aluminum alloy, resulted in increased tensile shear strength and work of fracture of the PMH hybrids. The fracture failure is dependent on the surface treatment of the aluminum alloy, thus P/A-Al/PA6 sample presented adhesive failure between aluminum alloy and PA6 interfaces against the debonding fracture of the P-Al/PA6 sample. The micro- and/or nanoscale honeycomb-like pores provided coherent networks to form interlocking structure for PMHs, and the average pore diameter range was from 100 nm to 140 nm, while surface roughness increased from 2.3 nm to 51 nm, compared to those of the polishing specimen. In addition, the surface quality of aluminum alloy is another important factor to determine the adhesion properties of the PA6 and aluminum alloy sheet. The large roughness of the anodized surface enlarged the contact angle tested by the deionized water, which decreased the free energy of the anodized surface and the surface energy difference between the metal and the plastic interface, so that the aluminum alloy surface easily wetted by nylon melt improved their joints’ bonding performance.
Footnotes
Acknowledgement
The authors thank research group for cooperation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the project of science and technology research program of the Chongqing Education Commission of China (No. CXQT19027), the project of Chongqing graduate student tutor group of Polymer material engineering and postgraduate innovation project of Chongqing University of Technology (No. ycx20192045).
