Abstract
In the present work, nanocomposite polymer electrolyte films were prepared by solution casting technique using nanosized fumed silica to polyethylene oxide (PEO)-based polymer electrolytes containing ammonium bifluoride (NH4HF2). The ionic conductivity of 1.19 × 10−5 S cm−1 has been observed at room temperature (25°C) for 3 wt% fumed silica in PEO-10 wt% NH4HF2 polymer electrolytes after which the conductivity was observed to decrease. Furthermore, the addition of high dielectric constant plasticizer propylene carbonate (PC) in the optimized composition of nanocomposite polymer electrolytes has increased the number of charge carriers by the large dissolution of ionic salt, amorphous content, and hence the ionic conductivity. Maximum ionic conductivity obtained at room temperature was found to be 1.55 × 10−4 S cm−1 in the case of PEO-10 wt% NH4HF2-3 wt% fumed silica-0.3 (ml) PC polymer electrolytes which is five orders of magnitude higher than that of the polymer host material. Temperature-dependent ionic conductivity, activation energy, and dielectric constant studies have been described for all the compositions of polymer electrolytes. Ionic conductivity and dielectric constant studies were determined from impedance data. Polymer electrolytes containing both fumed silica and PC highlight that there is no phase transition in the polymer electrolyte and temperature dependence of ionic conductivity in the temperature range is of almost Arrhenius type. The lowest activation energy value for the highest conducting polymer electrolyte was found to be 0.172 eV. Change in melting temperature, % crystallinity (χc), and mechanical properties have also been observed in polymer electrolytes containing fumed silica as well as PC as studied by Differential Scanning Calorimetry/Thermogravimetric Analysis (DSC/TGA) and universal testing machine, respectively.
Introduction
Polymer electrolytes have been intensively studied because of their possible application as electrolytes in solid-state electrochemical devices such as fuel cells, batteries, supercapacitors, sensors, and electrochromic windows. Polymer electrolytes also have various advantages such as electrochemical stability, light weight, good processability, flexibility, ease of fabrication in thin-film form, and ability to form effective electrode–electrolyte contacts.1–6 Crystalline morphology of polyethylene oxide (PEO) is its main drawback which hinders the ion transportation and hence limits its ionic conductivity at room temperature (RT).7,8 Polymers have been complexed with different ionic salts to provide ionic conduction. The protonic transport in polymer electrolytes generally involves motion of the groups like H+, NH4+, H3O+, and so on. Ammonium salts such as ammonium thiocyanate (NH4SCN), ammonium nitrate (NH4NO3), ammonium chloride (NH4Cl), ammonium hexafluorophosphate (NH4PF6), ammonium acetate (CH3COONH4), and so on, have been reported as good proton donors to the polymer matrix.9–14
Unfortunately, low ionic conductivity of polymer electrolytes at room temperature limits their practical applications. Different approaches and significant work have been carried out to increase the ionic conductivity as well as amorphous phase of polymer electrolytes with the addition of ionic salts—which plays an important role for fulfilling the criterion of faster ion transport and high ionic conductivity, the addition of low molecular weight plasticizers—which increases the free volume required for transport by penetrating between polymer chains and addition of nanofillers—which provides the favorable conducting pathways for ion migration on the surface of nanofiller. One of the promising approaches to improve the ion transport and mechanical properties of polymer electrolytes is to introduce nanofillers such as Al2O3, SiO2, or other ceramics as reported earlier.15–18 Also, depending on the concentration and particle size of the nanofiller, the dispersion of filler may improve the mechanical strength as well as the ionic conductivity of polymer electrolytes.19,20 The increase in ionic conductivity due to the presence of nanosized fumed silica may be due to minimizing the concentrations of PEO crystalline domains without affecting the PEO flexibility and mechanical stability over the wide temperature range. 21 The addition of nanofiller has been observed to increase the polymer amorphicity, improve chain mobility, change the interfacial resistance between electrolyte and electrode, and so on.22,23 The addition of nanofiller has also been observed to enhance the mechanical properties with an increase in Young’s modulus and yield strength as well.24,25
On the other hand, response to another approach, that is, to enhance the ionic conductivity as well as to increase amorphous content of a polymer electrolyte is to incorporate low molecular weight as well as high dielectric constant plasticizers, is also available in the literature.26–31 The addition of plasticizers may change the texture of polymer matrix into a softer and more flexible form which may result in enhancement on the chemical and mechanical stabilities of polymeric films.32–34
No doubt, plasticized polymer electrolytes have been found to possess high ionic conductivity and better transport properties yet the excess use of solvent may deteriorate the mechanical properties of these polymer electrolytes. However, limited reports are available in the literature representing the combined effect of plasticizer and nanofiller concentration on the electrical and structural properties of polymer electrolytes.35–38
The present research work is devoted to enhance the ionic conductivity along with improved mechanical properties of polymer electrolytes by a combined effect on the addition of plasticizer with the optimized salt and nanofiller concentration. Therefore, an attempt has been made to optimize and study the ion-conducting behavior and mechanical properties of PEO-ammonium bifluoride (NH4HF2)-nanosized fumed silica nanocomposite polymer electrolytes by incorporating different concentrations of plasticizer; propylene carbonate (PC).
Experimental work
Preparation of polymer electrolyte membranes
PEO (Aldrich, average molecular weight 5 × 106), NH4HF2 (Aldrich), PC (Merck), and fumed silica (Aldrich) with average particle size 7 nm have been used as the starting materials for the preparation of polymer electrolytes by solution casting method using methanol (Merck) as a solvent. PEO, NH4HF2, fumed silica, and PC taken in the stoichiometric ratio were dissolved in methanol and stirred for 8–10 h to obtain a homogeneous solution. The formed homogeneous solution was then poured in polypropylene dishes kept under dried atmosphere to evaporate the solvent slowly and dehydrated at 45°C in an oven for several hours to allow the complete evaporation of the solvent until solvent-free polymer electrolyte membranes were formed. For the formation of PEO-10 wt% NH4HF2 polymer electrolytes, the amount of NH4HF2 was taken to be 50 mg and the amount of PEO was taken to be 450 mg. Freestanding, mechanically stable, and transparent membranes of polymer electrolytes so obtained were carefully peeled off from the Petri dishes and stored in the vacuum desiccators for further drying before characterizations. The membranes having optimum properties seemed to have good mechanical strength and are much easy to handle.
Characterization studies
The electrical conductivity of polymer electrolytes was measured by complex impedance spectroscopy using a computer interfaced Hioki (Japan) 3532-50 LCR HiTester in the 40 Hz–5 MHz frequency range by placing polymer electrolyte film between the two electrodes of stainless steel sample holder under spring pressure. The conductivity was also measured in the 30–100°C temperature range by keeping the sample holder containing polymer electrolyte in a temperature-controlled furnace. Ionic conductivity and dielectric constant studies of polymer electrolytes were measured as a function of salt, fumed silica, plasticizer concentrations as well as temperature variations. Differential Scanning Calorimetry/Thermogravimetric Analysis (DSC/TGA) studies were undertaken by using Perkin Elmer (Pyris Diamond; USA) calorimeter at the heating rate of 10°C per minute in the temperature range of 0–400°C. Mechanical properties of different polymer electrolytes were measured by using Hounsfield (UK) H25KS universal testing machine (UTM). The conductivity values of different polymer electrolytes are calculated by using the equation: σ = (1/Rb)l/A; where l and A are the thickness and area of the polymer electrolytes.
Results and discussions
Composition dependence of ionic conductivity
Impedance spectroscopic technique has been used to study Nyquist plots (Z′ vs. Z″) for different polymer electrolytes. The real component of complex impedance Z′ is plotted on the abscissa and the imaginary component of the complex impedance Z″ is plotted on the ordinate. Rb is the bulk resistance which can be determined by the low-frequency intercept of the semicircle and the high-frequency intercept of inclined line on the x-axis.39,40 It is noted from Figure 1 that the plots show two regions: one is a high-frequency semicircle and the other is the low-frequency spike region. Low-frequency region indicates the bulk effect of blocking electrodes, that is the interface between the electrode and electrolyte is due to the migrations of ions. High-frequency semicircle is related to the conduction process in the bulk of the complex and can be used to obtain the bulk resistance (Rb) as shown in Figure 1. It is also observed from Figure 1 that the diameter of the high-frequency semicircle gradually decreases with the increase in PC concentrations. The appearance of a semicircle with a very small diameter for PEO-10 wt% NH4HF2-3 wt% nanosized fumed silica containing 0.3 (ml) PC is observed, which suggests that only resistive component of polymer prevails41,42 due to the maximum migration of ions at this concentration. The ionic conductivity values of PEO-10 wt% NH4HF2-3 wt% nanosized fumed silica nanocomposite polymer electrolytes at different concentrations of PC were determined by Nyquist plots obtaining the value of Rb and using the relation: σ = (1/Rb)l/A as represented in Figure 1 and Table 1. Therefore, it is concluded that the plasticizing solvent helps to increase the number of charge carriers by the large dissolution of ionic salt resulting in increased conductivity. 43 As PC molecules are of small size, they can easily penetrate in between the polymer chain. This results in an enhancement in the segmental motion of polymer chains as well as the creation of more free volume by penetrating between polymer chains so as to provide an easy path for the migration of cations. Hence, incorporation of PC in the above system is considered to be one of the best approaches, which plays a significant role in the enhancement of the amorphous phase as well as better dissociation of ionic salt. It was also experienced that when the concentration of PC was increased above 0.3 ml, the films became sticky for which measurements became difficult.
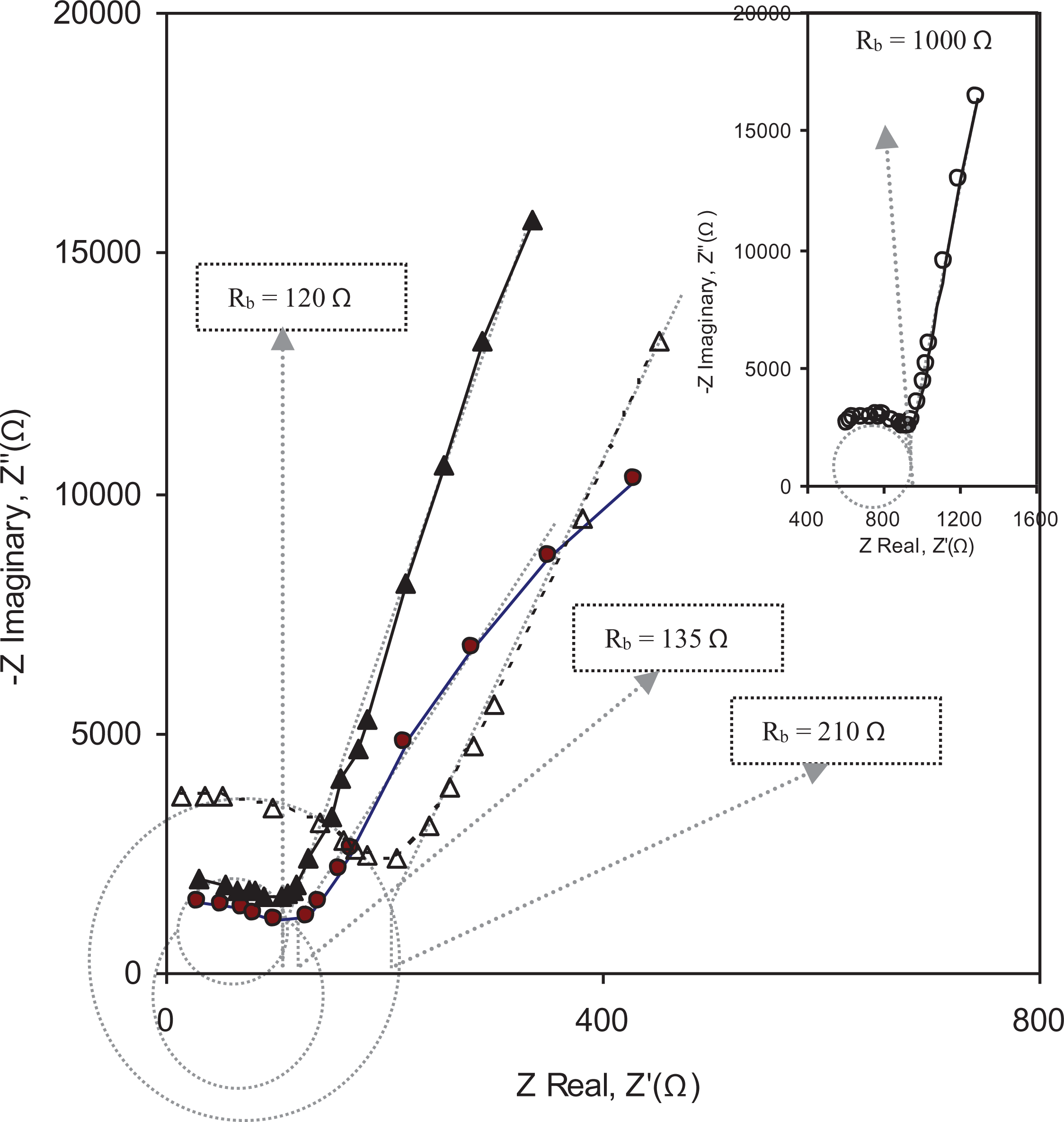
Nyquist plot for PEO-10 wt% NH4HF2-3 wt% fumed silica containing (0 wt%: inset (ο), 0.1 ml (Δ), 0.2 ml (•), and 0.3 ml (▴)) PC.
Data of conductivity, activation energy, pre-exponential factor (σo), and regression parameter (R 2 ) of PEO-10 wt% NH4HF2-3 wt% fumed silica containing different concentrations of PC.
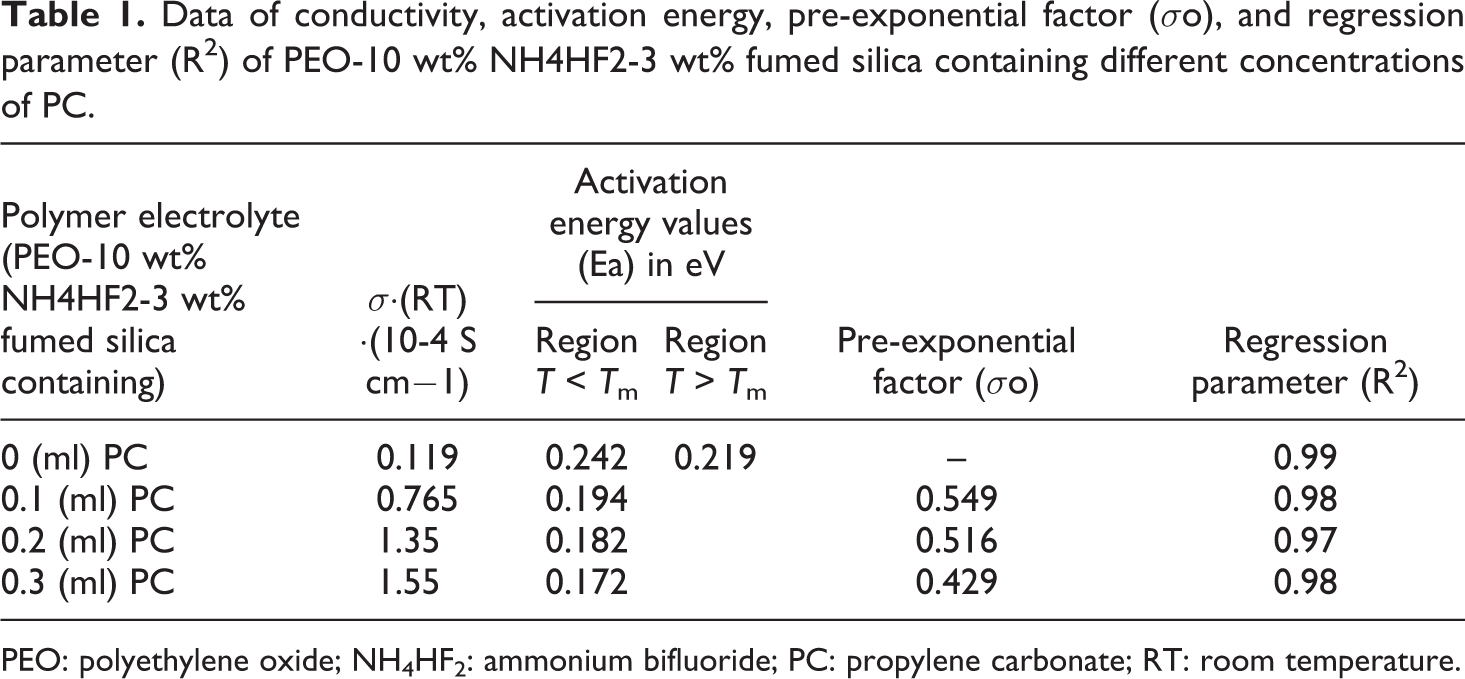
PEO: polyethylene oxide; NH4HF2: ammonium bifluoride; PC: propylene carbonate; RT: room temperature.
Temperature dependence of ionic conductivity
The effect of PC concentrations on the ionic conductivity of nanocomposite polymer electrolytes over a wide temperature range was studied as shown in Figure 2. From Figure 2, the natural logarithm of conductivity (ln σ) versus 1000/T for different polymer electrolytes confirms the increase in conductivity with the increase in temperature for all the composition of polymer electrolytes, which suggests the existence of large free volume around the polymer chain causing more mobility of ions and polymer segmental motion. For PEO-NH4HF2-fumed silica polymer electrolyte, it is observed that the plot follows Arrhenius type behavior with two regions (T < Tm and T > Tm) and two activation energies below and above melting temperature. For this composition, the sharp change in the conductivity values with temperature may be attributed to the transitions from the semi-crystalline phase to the amorphous phase. For other compositions of polymer electrolytes containing fumed silica and PC, non-Arrhenius (curved) variation of conductivity with temperature determines the ion transport obeying Vogel–Tamman–Fulcher behavior. This indicates that the conductivity behavior of these polymer electrolytes follows the free volume law and hence the ion transport which is due to the segmental motion of polymeric chains. Also, the curved nature of temperature-dependent ionic conductivity plot seems to be applied mostly for amorphous polymer electrolytes. 44
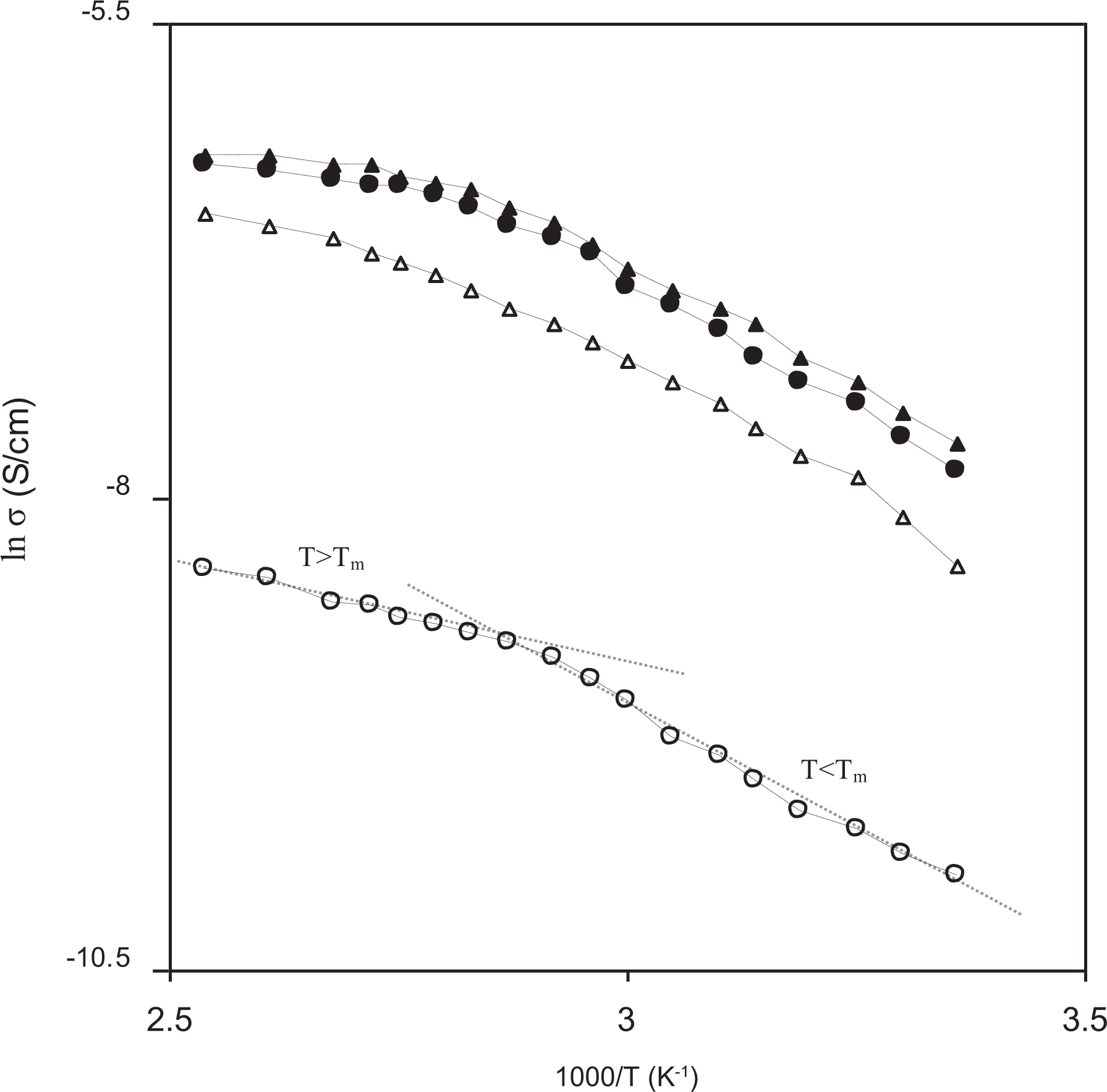
Variation of ln conductivity (S cm−1) with 1000/T (K−1) for PEO-10 wt% NH4HF2-3 wt% fumed silica containing (0 wt% (ο), 0.1 ml (Δ), 0.2 ml (•), and 0.3 ml (▴)) PC.
Activation energy measurement
Since the activation energy is considered as an energy barrier, which the ions have to overcome for a successful jump between the sites.
45
In the present study, the activation energy and other parameters of the ionic conduction are obtained for all the compositions from the slops of ln σ versus 1000/T and equation obeying Arrhenius behavior:
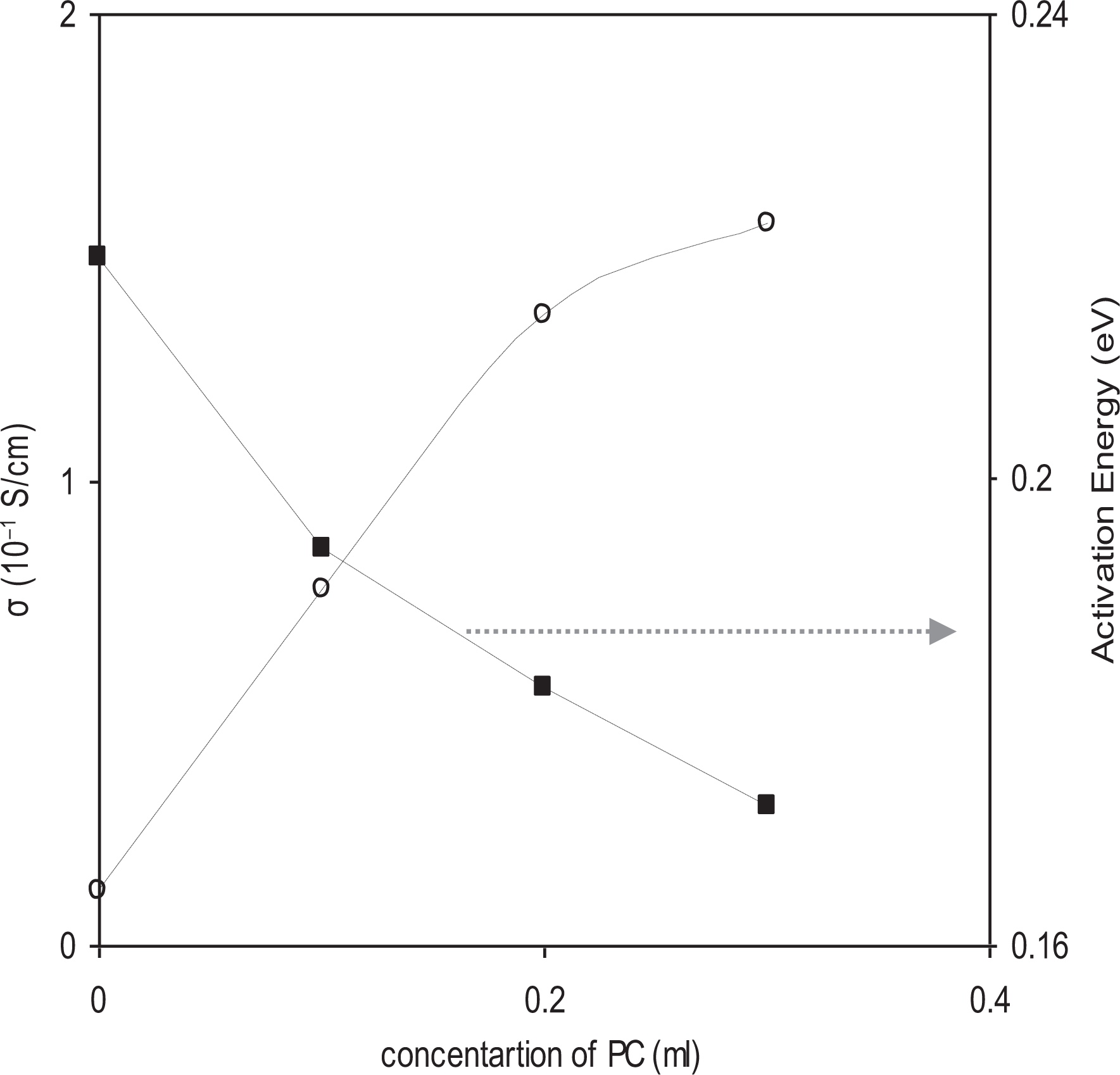
Variation of conductivity and activation energy as a function of concentration of plasticizer (PC) in PEO-10 wt% NH4HF2-3 wt% fumed silica polymer electrolytes at 300 K.
Study of dielectric constant
The dielectric property provides valuable information about the characteristics of ionic interaction of polymer electrolytes as well as an understanding of ion transport behavior. It is observed from Figure 4 that the value of the real part of permittivity ε′ which corresponds to an ordinary dielectric constant of the material is higher at lower frequencies. This may be due to the accumulation of a large number of charge carriers near electrodes.53,54 At high frequencies, the polarity reversal of the field occurs by which the dipoles cannot orient themselves along the direction of the field and hence no further ion diffusion takes place along the direction of the field. This results in a decrease in the value of the real part of permittivity (ε′) at high frequencies. 55 Also, the dielectric constant value has been observed to increase with the addition of additives in lower frequency region as shown in Figure 4 which indicates the increase in the number of mobile ions. 56 Higher the value of ε′ suggests better conductivity, which may be due to free charge motion and presence of charge effects within the polymeric system.
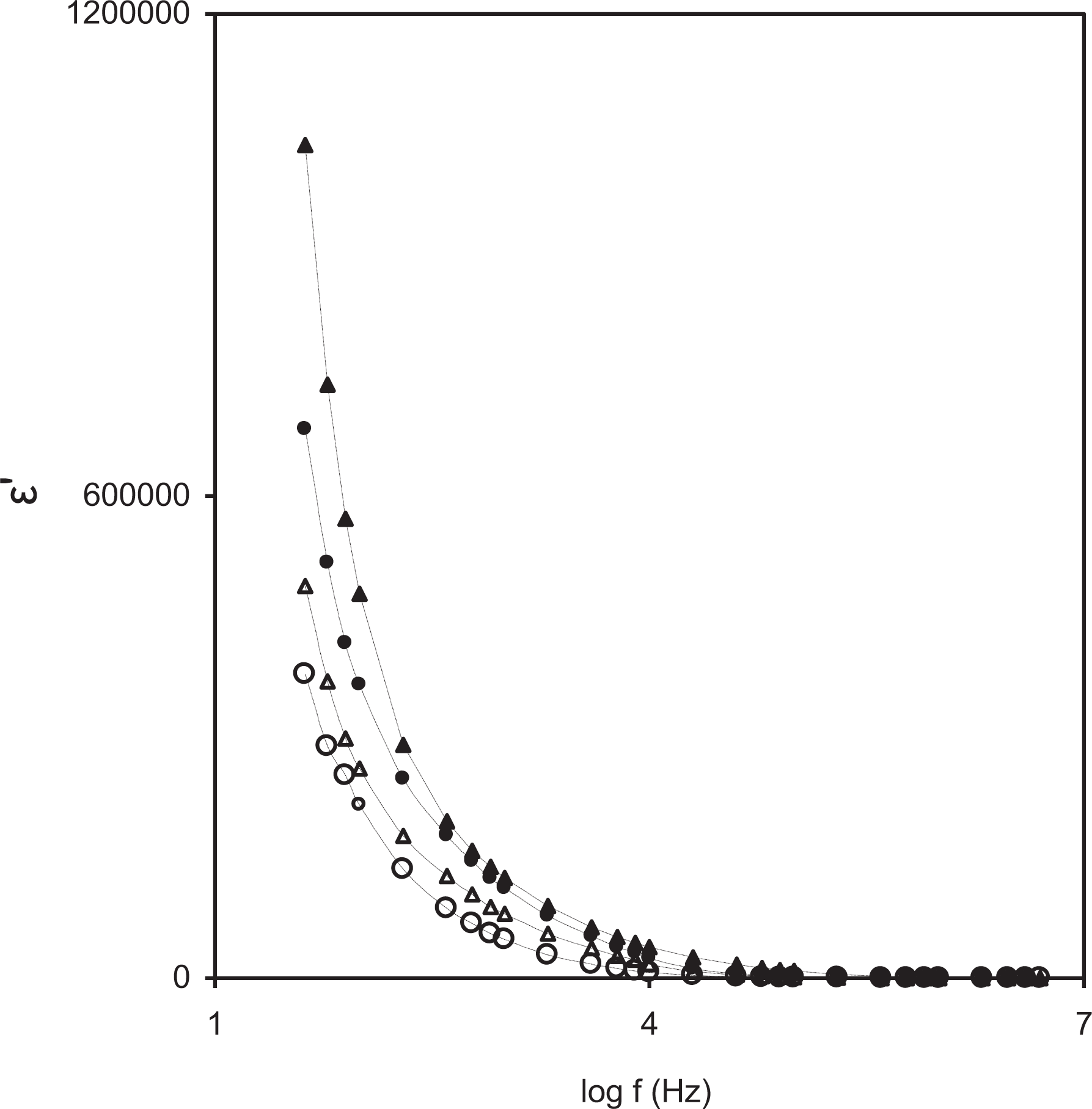
Variation of dielectric constant (∊′) as a function of log frequency for PEO-10 wt% NH4HF2-3 wt% fumed silica containing (0 wt% (ο), 0.1 ml (Δ), 0.2 ml (•), and 0.3 ml (▴)) PC.
DSC/TGA studies
Figure 5 shows the reduction in melting temperature, % crystallinity (χc), and % weight loss in nanocomposite polymer electrolytes containing different concentrations of PC as studied by DSC and TGA, respectively. The data values related to the above parameters and their comparison are also represented in Table 2. In DSC thermogram, shifting of peak related to melting temperature of PEO with the addition of fumed silica as well as PC toward lower temperature side (65–55°C) has been observed, which corresponds to the reduction of crystalline phase and hence more polymer chain flexibility. The decrease in melting temperature as a function of plasticizer concentrations has also been studied earlier. 32 According to Pradhan et al., plasticizer can easily penetrate the polymer matrix as these molecules are relatively small in size as compared to that of polymer host molecules. Hence, the strong interaction between plasticizer molecule and polymer chain molecules may cause an increase in chain segmental mobility and hence ionic conductivity of nanocomposite polymer electrolyte on the addition of plasticizer concentrations. TGA provides information regarding thermal stability and thermal degradation of polymer electrolytes when heating of the sample occurs at a fixed rate. From the study as shown in Figure 5, nonsignificant weight loss (<1%) at 100°C is noted, which may be probably due to the evaporation of the residual solvent/moisture present (at the time of loading the sample) in the polymer electrolytes. After this temperature, minimal weight loss continued and weight loss (about 10%) observed at 200–250°C may be due to the melting of a new complex formed after the addition of fumed silica and boiling of PC. It is, therefore, suggested that the presence of additives adds some complexity to the thermal behavior of the polymer electrolytes. Endothermic peaks observed in the DSC thermogram correspond to the temperatures about which weight loss occurs in TGA. Rapid weight loss (>20%) corresponding to a higher temperature range is observed, which may be due to the structural decomposition of PEO.
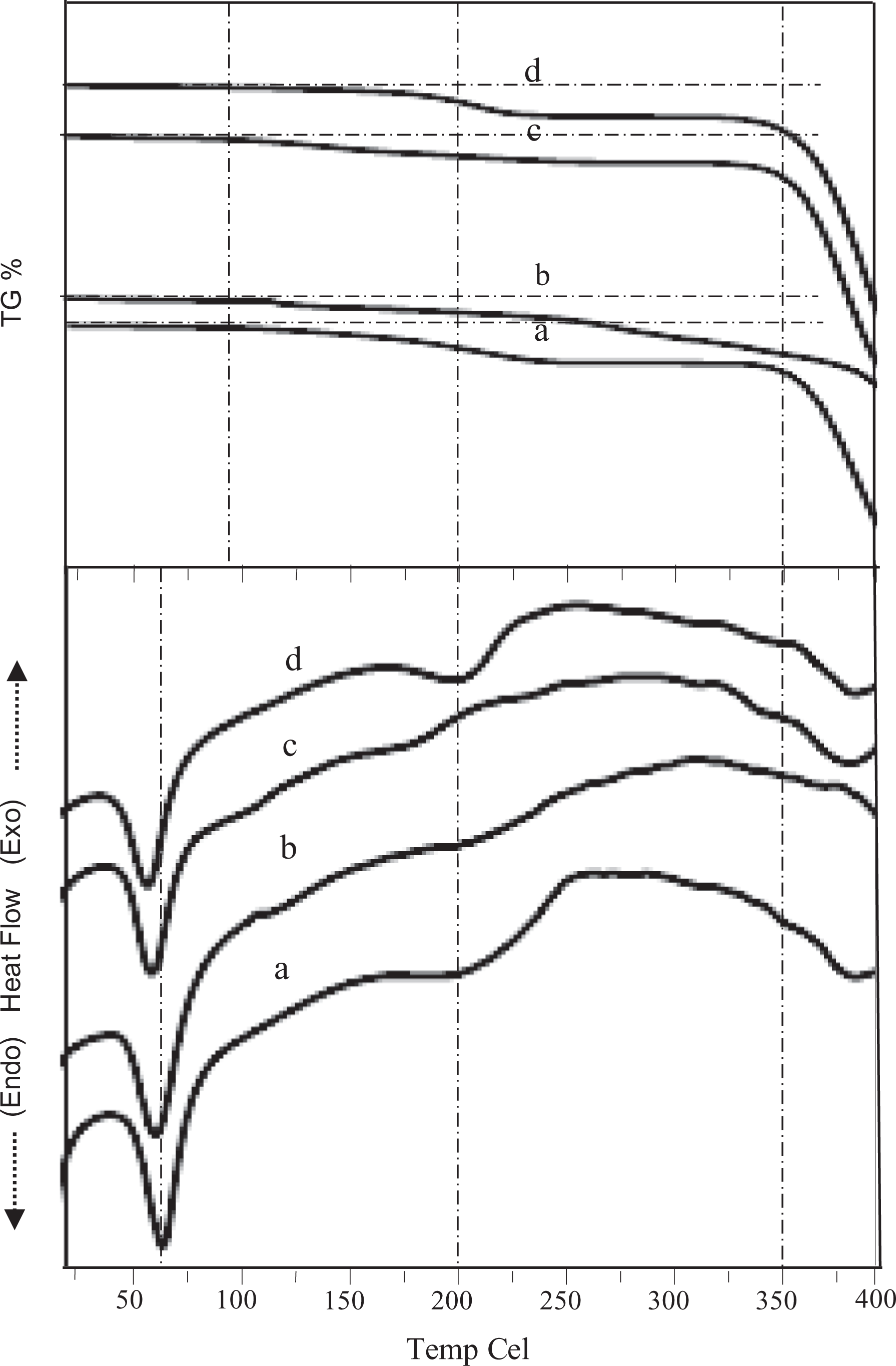
DSC/TGA plot for PEO-10 wt% NH4HF2-3 wt% fumed silica containing (0 wt% (a), 0.1 ml (b), 0.2 ml (c), and 0.3 ml (d)) PC.
Data of melting temperature Tm, melting enthalpy of polymer electrolyte (ΔHm), degree of crystallinity χc (%), tensile strength (MPa), and (%) elongation at break of PEO-10 wt% NH4HF2-3 wt% fumed silica containing different concentrations of PC.a
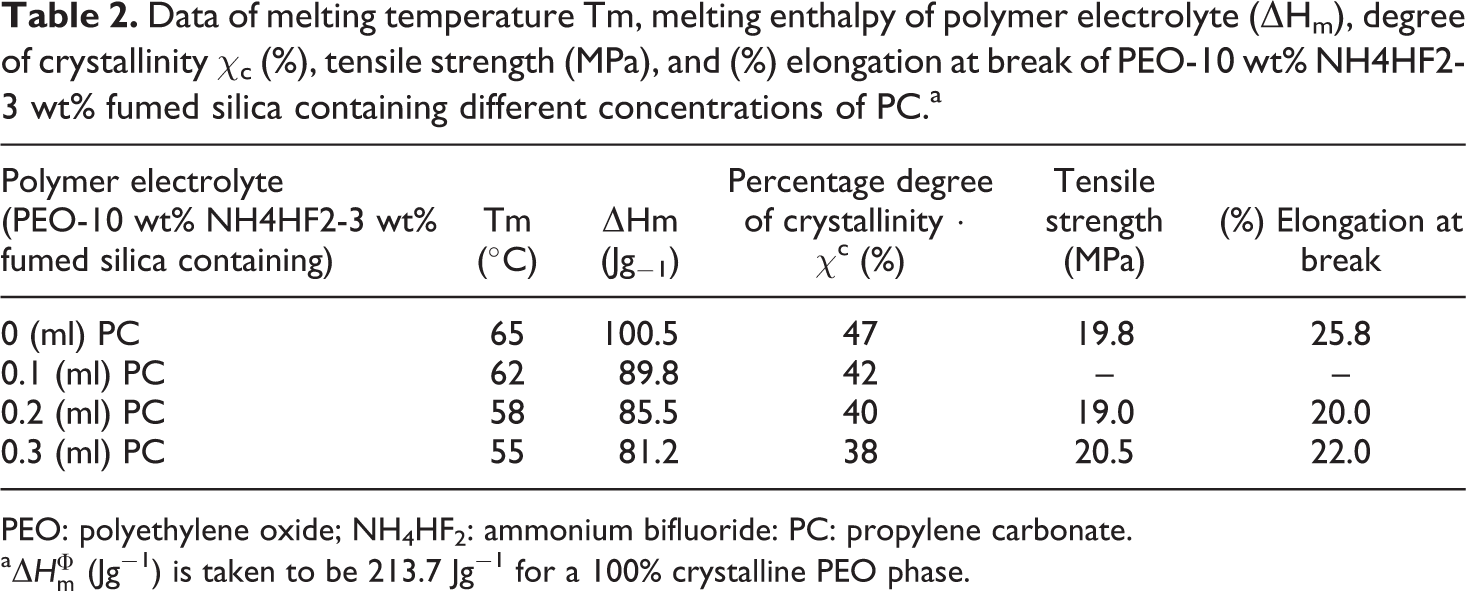
PEO: polyethylene oxide; NH4HF2: ammonium bifluoride: PC: propylene carbonate.
a
From the results of DSC studies, Figure 6 shows the variation of % crystallinity (χc) and melting temperature (Tm) versus PC concentrations of different polymer electrolytes. The percentage degree of crystallinity (χc) for different polymer electrolytes has been calculated from the equation:
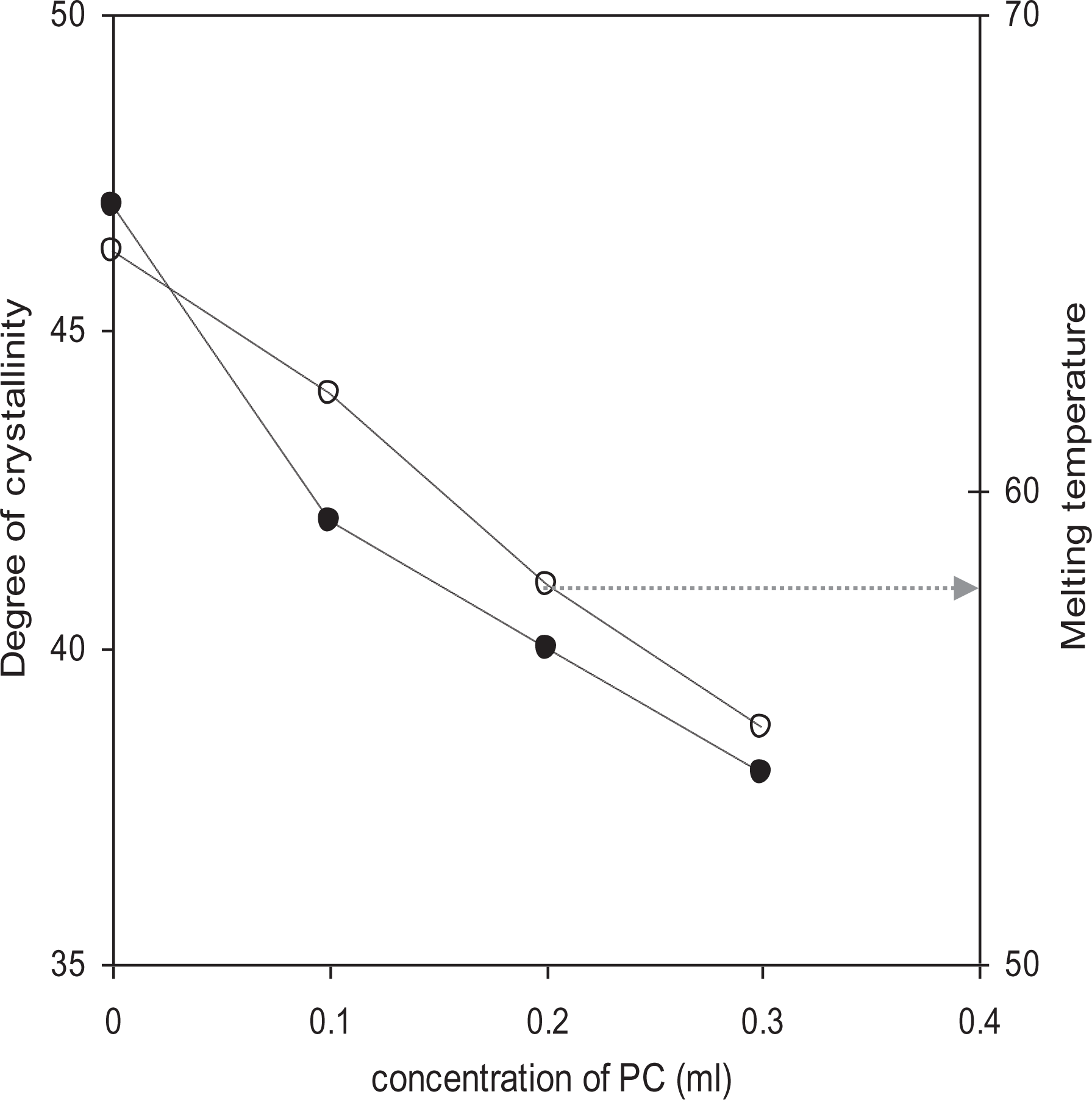
Variation of degree of crystallinity and melting temperature as a function of concentration of plasticizer (PC) in PEO-10 wt% NH4HF2-3 wt% fumed silica polymer electrolytes.
Mechanical property studies
The influence of nanosized fumed silica as well as plasticizer (PC) on the mechanical properties of polymer electrolytes has been studied by UTM. Figure 7 shows the change in tensile strength and (%) elongation at the break of polymer electrolytes containing fumed silica and PC. It is observed that the tensile strength of the polymer electrolytes is enhanced along with the appreciable improvement in plastic elongation of polymer matrix with the addition of additives. Mechanical strength of nanocomposite polymer electrolytes was also observed to maintain the mechanical integrity in the presence of PC. During a tensile test, a necked region that is elastic initially has been observed in polymer electrolytes containing PC which may be due to the ductile nature of the material. Maximum tensile strength of 19–21 MPa along with (%) elongation at break 20–22 has been observed in the nanocomposite polymer electrolytes containing PC, and the results are represented in Table 2. The enhancement in tensile strength along with ionic conductivity has been observed with the addition of optimal concentration of additives, which may be due to the strong interaction of additives with the polymer.47,59 Hence, the addition of additives in polymer electrolytes can greatly affect both electrical and mechanical properties.
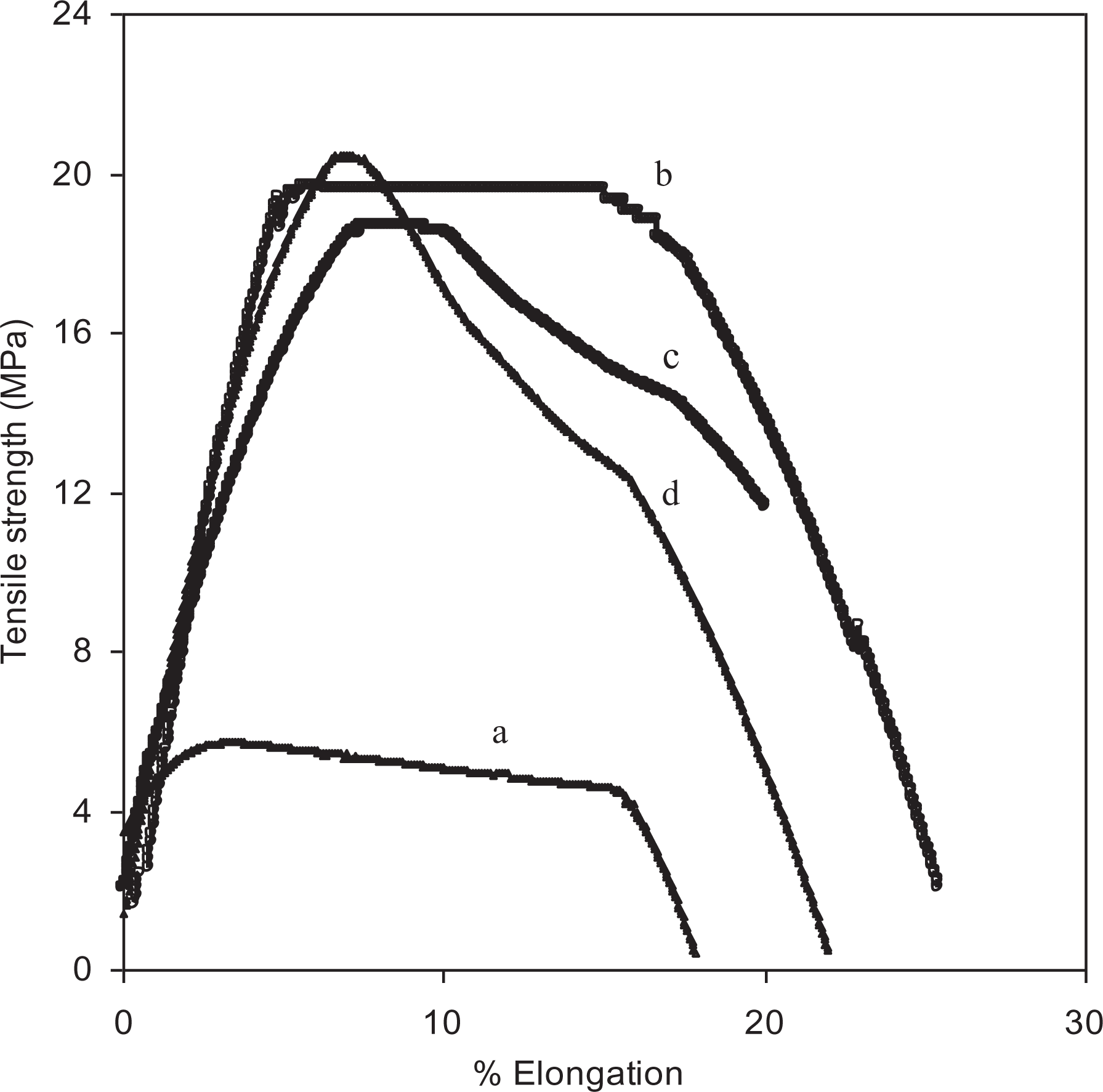
Variation of tensile strength with percent elongation (%) for PEO-10 wt% NH4HF2 (a), PEO-10 wt% NH4HF2-3 wt% fumed silica containing (0 ml (b), 0.2 ml (c), and 0.3 ml (d)) PC.
Conclusions
The influence of different concentrations of plasticizer (PC) on the electrical, thermal, and mechanical properties of nanocomposite polymer electrolytes with an optimized concentration of NH4HF2 and nano-sized fumed silica has been studied. Maximum ionic conductivity value of 1.55×10−4 S cm−1 has been obtained at room temperature for PEO-10 wt% NH4HF2-3 wt% fumed silica polymer electrolytes containing 0.3 (ml) PC exhibiting low activation energy value of 0.172 eV. Studies on temperature-dependent ionic conductivity, activation energy, and dielectric constant have also been described for all the compositions of polymer electrolytes. The reduction in melting temperature and % crystallinity with the addition of PC concentrations corresponds to more polymer chain flexibility and hence increase in more amorphous character. Minimal weight loss of about 10% is observed up to 200–250°C, which suggests that these polymer electrolytes are thermally stable up to this temperature range. The change in mechanical properties with the addition of nanosized fumed silica as well as optimizing the concentration of PC to polymer electrolytes has been observed to maintain mechanical integrity, which can greatly affect both electrical and mechanical properties. Hence, polymer electrolytes consisting of the simultaneous presence of nano-sized fumed silica as well as plasticizer (PC) have shown significant improvement in electrical, thermal, and mechanical properties. Further detailed studies on different parameters are underway to further explore ion dynamics in these polymer electrolytes.
Footnotes
Acknowledgements
The authors are grateful to Solid State Ionics Laboratory, Guru Nanak Dev University, Amritsar, for providing conductivity measurement facility; and to the Institute instrumentation Centre, IIT Roorkee, for providing DSC/TGA facility, and Mechanical Engineering Department, NIT Hamirpur, for providing necessary UTM facility.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
