Abstract
The polypropylene (PP) composites filled with flame retardants (including aluminum hydroxide (Al(OH)3), magnesium hydroxide (Mg(OH)2), and zinc borate (ZB)) were prepared, and the composites were separately loaded with polyolefin elastomer (POE) and nanometer calcium carbonate (nano-CaCO3). The tensile properties and the fire residue morphology of the specimens after burning for these three groups of composites were measured. The results were shown that the tensile fracture strength and the Young’s modulus increased while the tensile yield strength and the tensile elongation at break (
Introduction
Aluminum hydroxide (Al(OH)3) and magnesium hydroxide (Mg(OH)2) are halogen-free flame retardants.1,2 Mg(OH)2 is usually used with Al(OH)3 to improve the flame-retardant properties of polymers such as polypropylene (PP).3,4 In addition, Mg(OH)2 and Al(OH)3 can improve the thermal stability of polymer composites.5,6 Gwon et al.
7
studied the effect of hybridization of wood fibers and metal hydroxides on the thermal stability of PP-based plastic composites through thermogravimetric analysis and found that the thermal stability and the maximum weight loss rate (T-max) of the PP matrix decomposition increased with an increment of the contents of both the Al(OH)3 and the Mg(OH)2 fillers. In general, the improvement of the flame-retardant properties of a polymer needs a high concentration of Mg(OH)2 and Al(OH)3. In previous work, Liang and his colleagues
4
investigated the effects of the filler content (from 10 wt% to 70 wt%) on the flame-retardant properties of PP/Al(OH)3/Mg(OH)2 composites filled with nanometer calcium carbonate (nano-CaCO3); the results showed that the limiting oxygen index (LOI) of the composites increased approximately linearly with an increase of the filler concentration. However, the mechanical properties, such as the tensile, impact, and flexural properties, of the flame-retardant polymer composites are usually weakened in the case of high concentration of Mg(OH)2 and Al(OH)3.8–10 Therefore, it is quite meaningful to reveal the reinforcing and toughening mechanisms when the new flame-retardant polymer composites loaded with Mg(OH)2 and Al(OH)3 are prepared. Studies on the reinforcing and toughening of PP have been paid extensive attention in the past two decades.9–15 In particular, Liang et al.
The combustion or burning characteristics is also one of important flame-retardant properties of polymeric materials, while the residue surface morphology of specimens is closely related to the combustion or burning characteristics.16,18–22 However, there have been a few of studies on the residue surface morphology of specimens after combustion for PP composites loaded with one or more of Mg(OH)2, Al(OH)3, zinc borate (ZB), POE, and nano-CaCO3. 16 To improve the mechanical properties of PP/Mg(OH)2/Al(OH)3/ZB composites, the composites are filled with POE and nano-CaCO3, but there are also relatively few studies on the tensile properties of PP/Mg(OH)2/Al(OH)3/ZB/nano-CaCO3/POE composites.13,14 The objectives of this article are to investigate the effects of Mg(OH)2/Al(OH)3 content on the tensile properties, including Young’s modulus, tensile strength, and tensile elongation at break, and the fire residue surface morphology of such burned specimens for the PP composite systems filled separately with ZB, nano-CaCO3, and/or POE, and to understand the flame-retarded mechanisms and the reinforcing mechanisms.
Experiment
Materials
The PP supplied by the Guangzhou Petrochemical Works (China) was used as the matrix resin; its trademark was CJS-700G. The melt index (230°C, 2.16 kg) was 10 g/10 min, and the density in the solid state was 910 kg/m3.
The Al(OH)3 and Mg(OH)2 were used as the flame-retardant additives, which were white powders. The density and the mean diameter of the Al(OH)3 were 2.42 g/cm3 and 1.50 μm, respectively. The density and the mean diameter of the Mg(OH)2 were 2.39 g/cm3 and 1.74 μm, respectively. The ZB was served as a flame-retardant synergist, and its average diameter, density, and melting point temperature were 6 μm, 2.89 g/cm3, and 980°C, respectively. The Al(OH)3, Mg(OH)2, and ZB were supplied by the Jinge Fire-Fighting Materials Co., Ltd (China).
The nano-CaCO3 with trademark CC-A, supplied by the An Yuan Technological and Chemical Industry Co., Ltd (China), was used as the filler. Its average diameter and density were 40 nm and 2.5 g/cm3, respectively. The surface of the nano-CaCO3 was pretreated with stearic acid using a high-speed mixer. The POE, with trademark Engage-8180, was supplied by the Du Pont Co. (USA), and its density and melt flow index were 0.87 g/cm3 and 0.5 g/10 min, respectively.
Preparation
The flame-retardant additives, including Al(OH)3, Mg(OH)2, and ZB, and the POE and filler nano-CaCO3 particles were separately mixed with the PP master batches in the high-speed mixer, then the mixtures were melt-blended using a model TSE-20 corotating twin-screw extruder, and the extrudate was granulated to form the pellets of the unfilled PP and the three groups of PP composite systems: PP/Al(OH)3/Mg(OH)2/ZB, PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3, and PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites. The diameter and the length-to-diameter ratio of the screw were 22 mm and 42, respectively. The screw speed was 60 rotation/min and the extrusion temperature range was from 160°C to 180°C. For investigating the influence of the flame-retardant content on the tensile and combustion behavior of the composites, the weight fractions (
Both the specimens for the flame-retardant tests and the specimens for the tensile tests were molded using a plastics injection machine (model UN120A) supplied by the Yizhimi Pml Precision Mechanism Co., Ltd (China); the injection temperature range was from 180°C to 190°C. The size of the specimens for flame-retardant tests was 130 × 13 × 4 mm3; and the specimens for tensile tests were dumbbell sheets with the width of 10 mm and the thickness of 4 mm.
Apparatus and methodology
Tensile property measurement
The tensile tests of the three groups of PP composite systems were carried out at room temperature using a universal materials testing machine (model CMT4104) supplied by the Newsans Co., Ltd (China) according to standards ISO 527-1-1993, and the crosshead speed for the tensile tests was 50 mm/min. Each group of specimens contained five pieces, and the average values of the measured tensile properties were used for the reported data.
Morphology observation of fire residue surface and fracture surfaces
The burning tester used in this work was an LOI apparatus (model JF-3), and it was supplied by the Jianglin Analysis Instrument Co. (China). The experiments of the LOI for the three groups of PP composites were carried out according to standards ISO4589-2-2017 for studying the combustion behavior and flame-retarding mechanisms of these composites. In addition, the pictures of the specimens were taken after the LOI tests to observe the fire residue surface morphology of the specimens.
The room temperature fracture surfaces of specimen were examined using a scanning electron microscope (SEM, model S-3700N) supplied by the Hitachi Co., Ltd (Tokyo, Japan) to observe the interfacial debonding, interlayer structure morphology, and the dispersion or distribution of the filler particles in the matrix. Before the SEM examination, the fracture surfaces of the specimens were gold-coated.
Results and discussion
Tensile properties
Curves of tensile stress versus strain
Figure 1 shows the curves of tensile stress versus tensile strain for the neat PP and the PP/Al(OH)3/Mg(OH)2/ZB composites. It can be seen that the maximum tensile stress and the tensile strain at break decreased while the slope of the curves somewhat increased with increasing flame-retardant weight fraction (
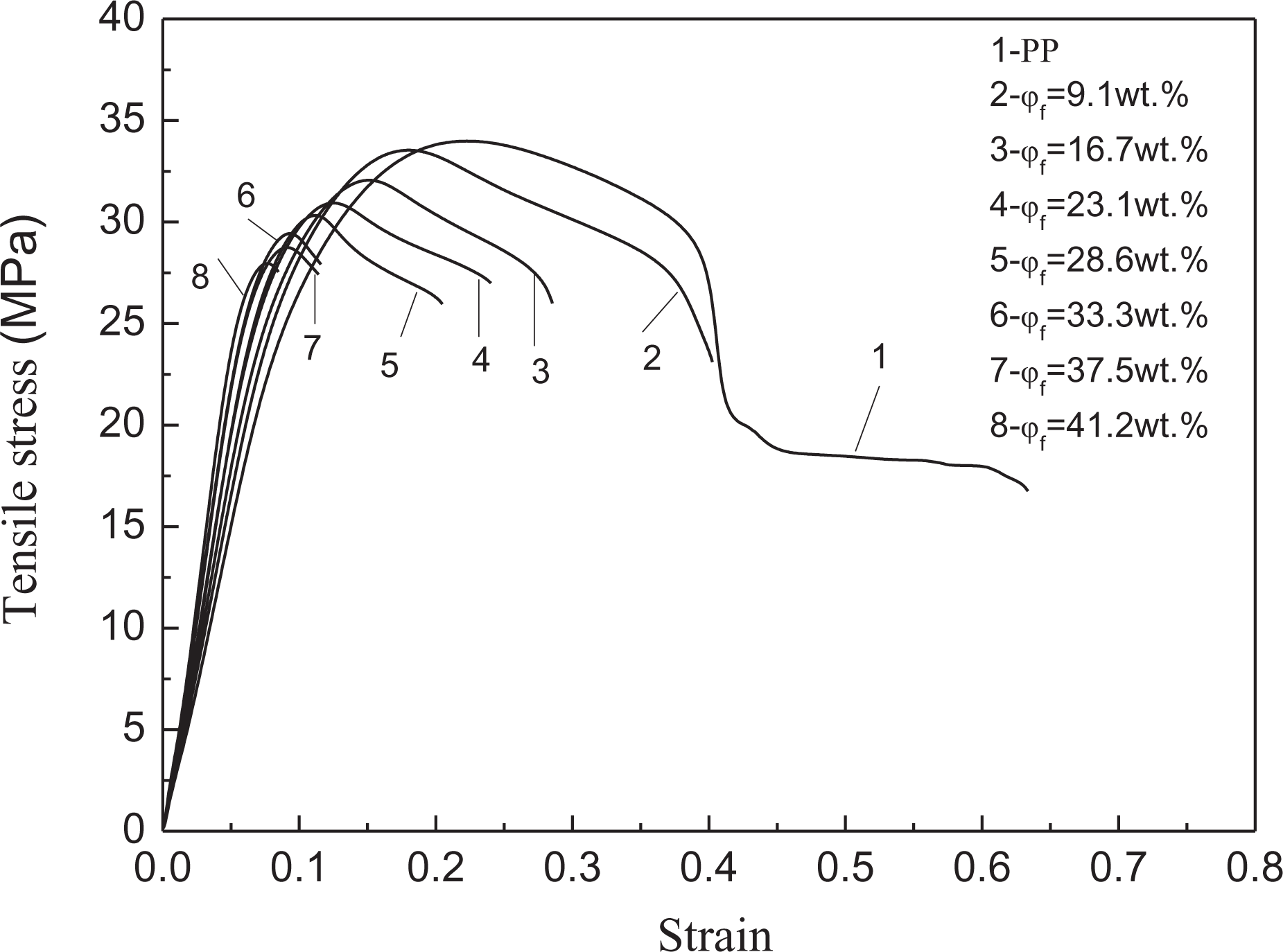
Curves of tensile stress versus tensile strain for neat PP and PP/Al(OH)3/Mg(OH)2/ZB composites.
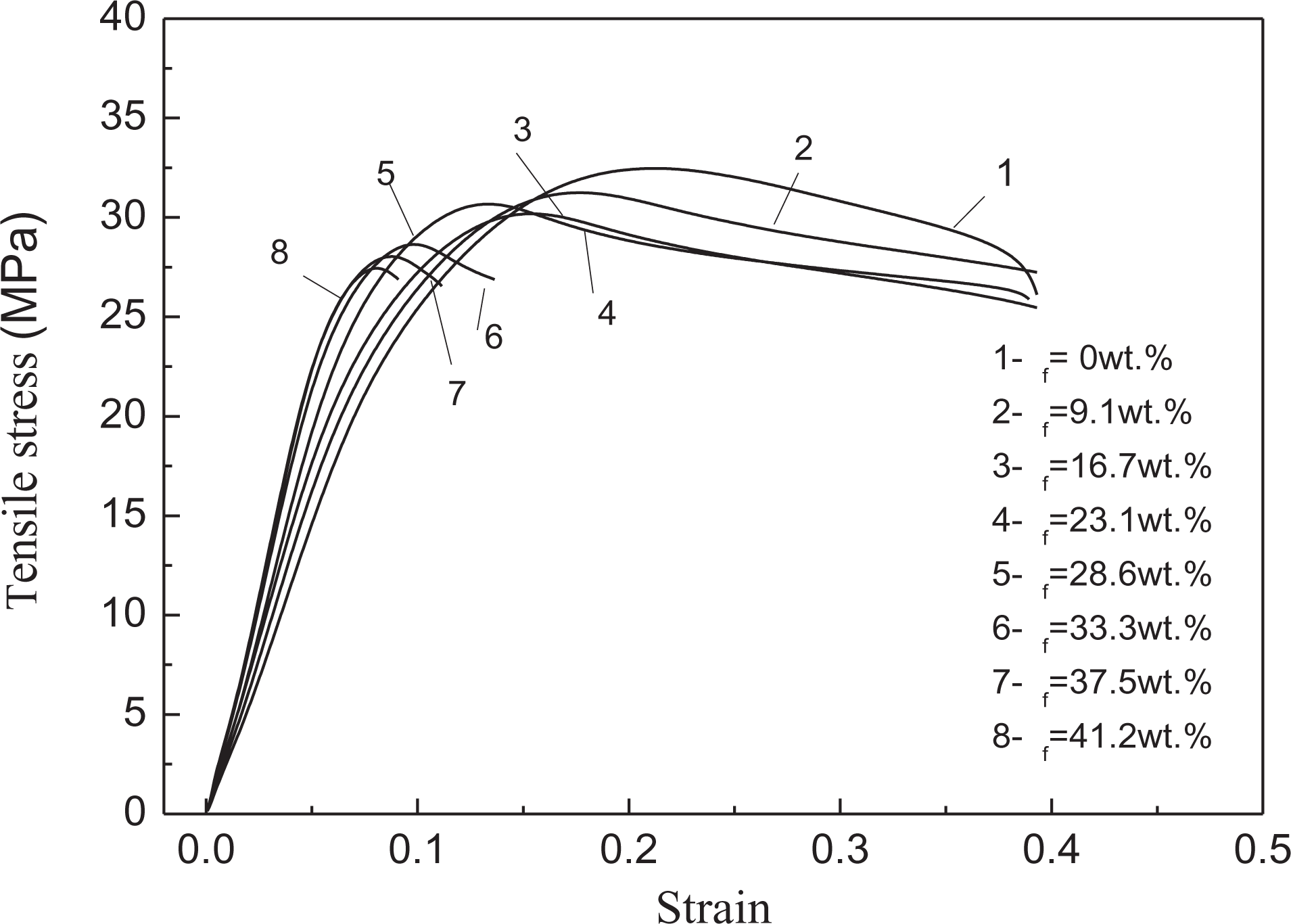
Curves of tensile stress versus tensile strain for PP/nano-CaCO3 composites and PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composites.
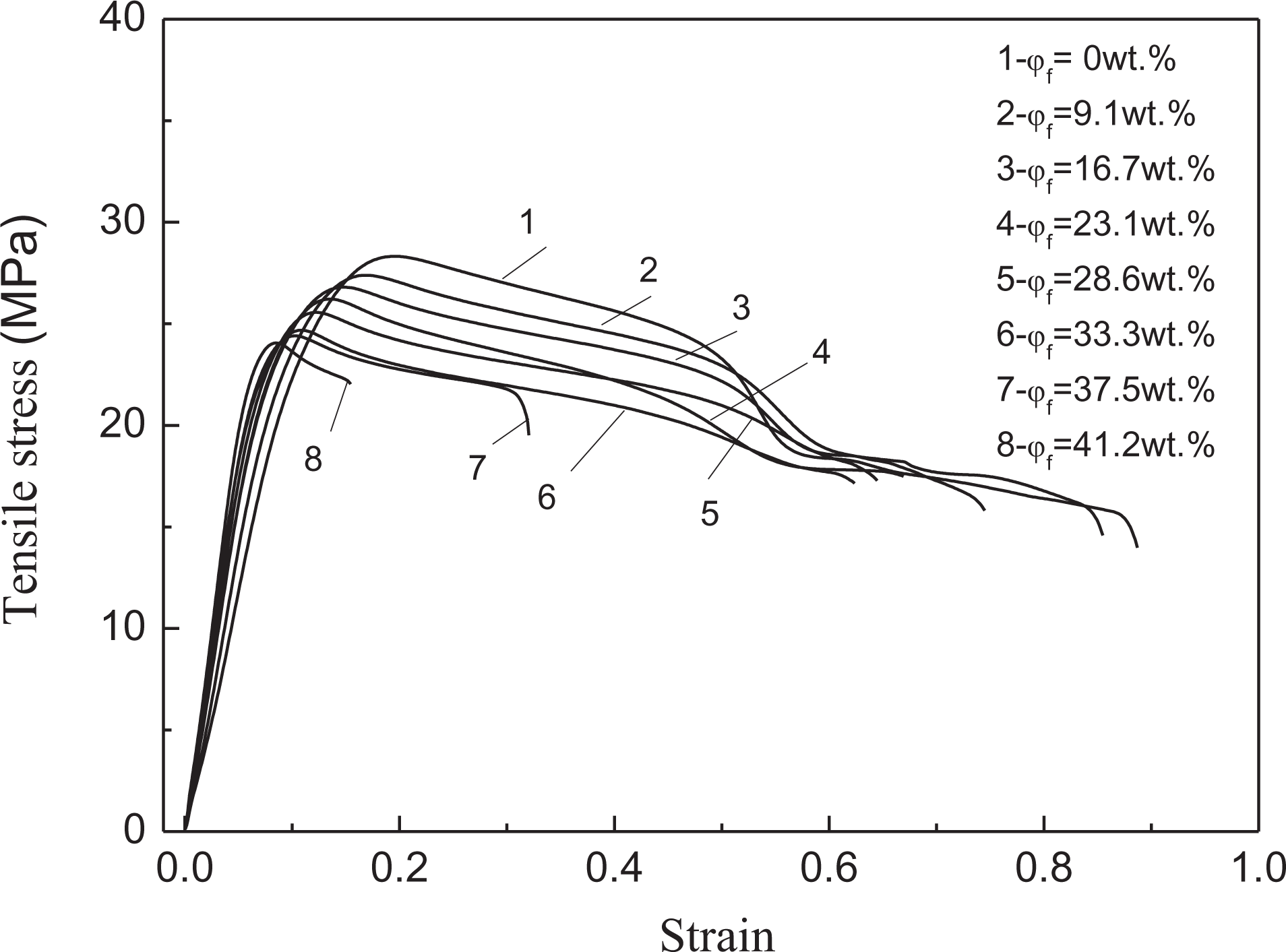
Curves of tensile stress versus tensile strain for PP/nano-CaCO3 composites and PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites.
Dependence of Young’s modulus on flame-retardant content
Young’s modulus describes the tensile stiffness of a material when a specimen is under load. Figure 4 illustrates the dependence of the Young’s modulus on the flame-retardant volume fraction for the three groups of PP composite systems. The flame-retardant volume fraction (

where

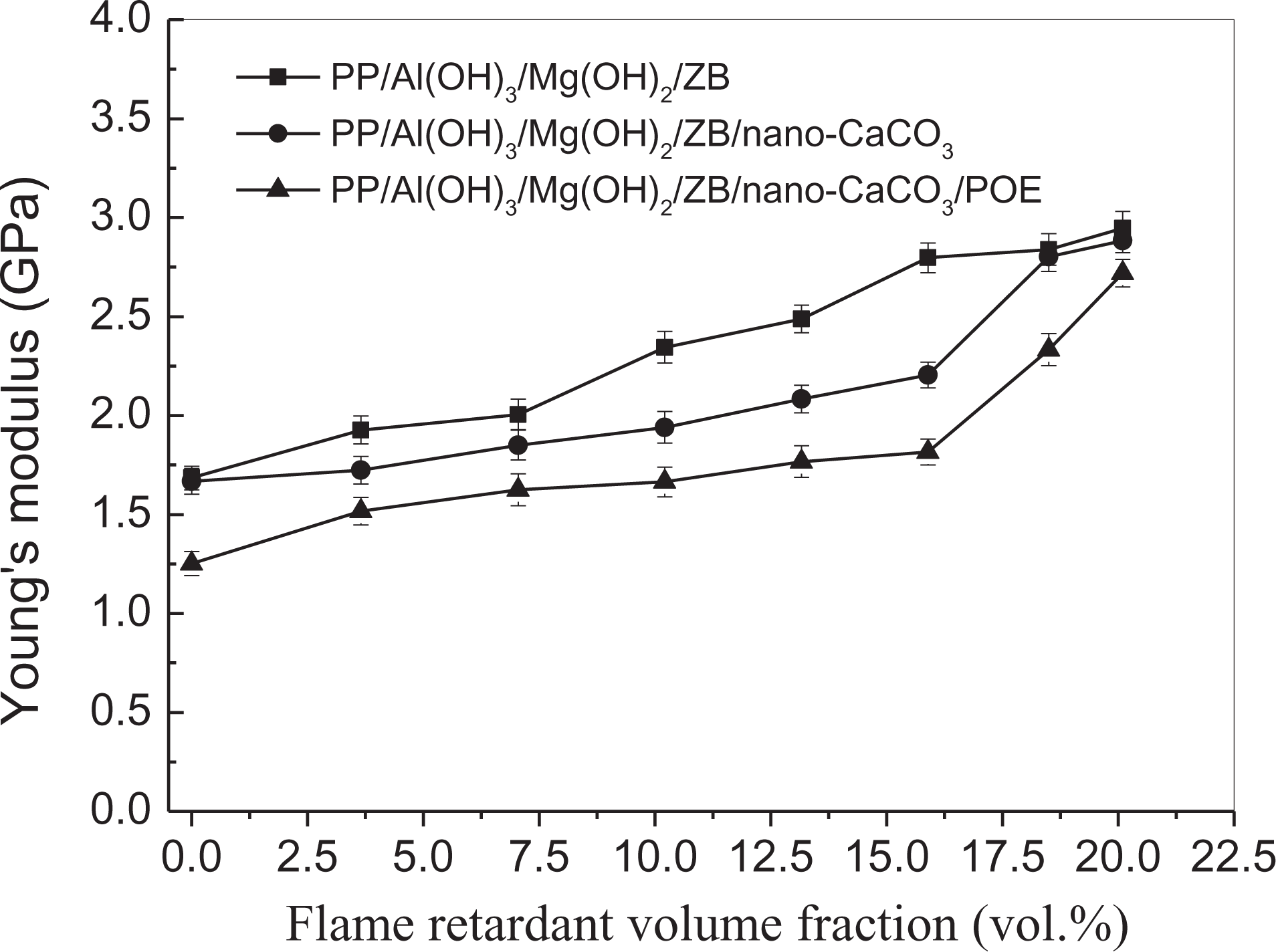
Dependence of Young’s modulus on flame-retardant volume fraction.
It can be seen that the Young’s modulus increases with increasing flame-retardant volume fraction. This is because the movement of the macromolecular chains of the polymer matrix is limited by the inclusions and by the physical cross-link points between the surfaces of the filler particles and the macromolecular chains of the matrix. Moreover, the filler particles play a role of a skeleton in the resin matrix. Thus, the tensile stiffness of the PP composites was improved, resulting in the increase of the Young’s modulus with increasing the flame-retardant volume fraction. In addition, PP is a semicrystalline resin, and the inorganic particles could play a role of heterogeneous nuclei in the matrix. 23 The crystal type and the crystallinity degree of PP can be changed in this case of loading inorganic particles, also resulting in variation of the stiffness of the composites. That is, both the skeleton effects of the flame-retardant additives in the matrix blocking the movement of the macromolecular chain and the induced crystallization were the main reinforcing mechanisms of these PP composites.24–26 For the same flame-retardant volume fraction, the Young’s modulus of the PP/Al(OH)3/Mg(OH)2/ZB composites was the highest, while the Young’s modulus of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites was the lowest. The reason could be that POE was a thermoplastic elastomer, its stiffness was lower than PP, and the nano-CaCO3 content is very small (only 3 wt%); thus, they were not enough to play the major role for promoting the skeleton effect and crystallization. Consequently, the Young’s modulus of these composite systems decreased.
Relationship between tensile strength and flame-retardant content
Tensile strength, including the tensile yield strength and tensile fracture strength, is an important characteristic of the use performance of materials. It is generally believed that the tensile strength of polymer composites depends, to great extent, upon both the interfacial adhesion status between the filler and the matrix and the dispersive status of the filler particles in the matrix. The better the interfacial adhesion status, the higher is the tensile strength of polymer composites.25–28 Figure 5 displays the relationship between the tensile yield strength and the flame-retardant volume fraction for the three groups of PP composite systems. It can be seen that the tensile yield strength decreased slightly with increasing flame-retardant volume fraction. It suggests the interfacial adhesion between the flame-retardant particles and the PP matrix is good; thus, the interface could somewhat transfer effectively the tensile stress, leading to only a slight reduction in the tensile yield strength with increasing flame-retardant content. Moreover, the tensile yield strengths of the PP/Al(OH)3/Mg(OH)2/ZB composites were close to those of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composites, while the tensile yield strengths of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites were the lowest at the same flame-retardant volume fraction. This indicates that the loading of the POE weakens the tensile yield strength of the PP composites.
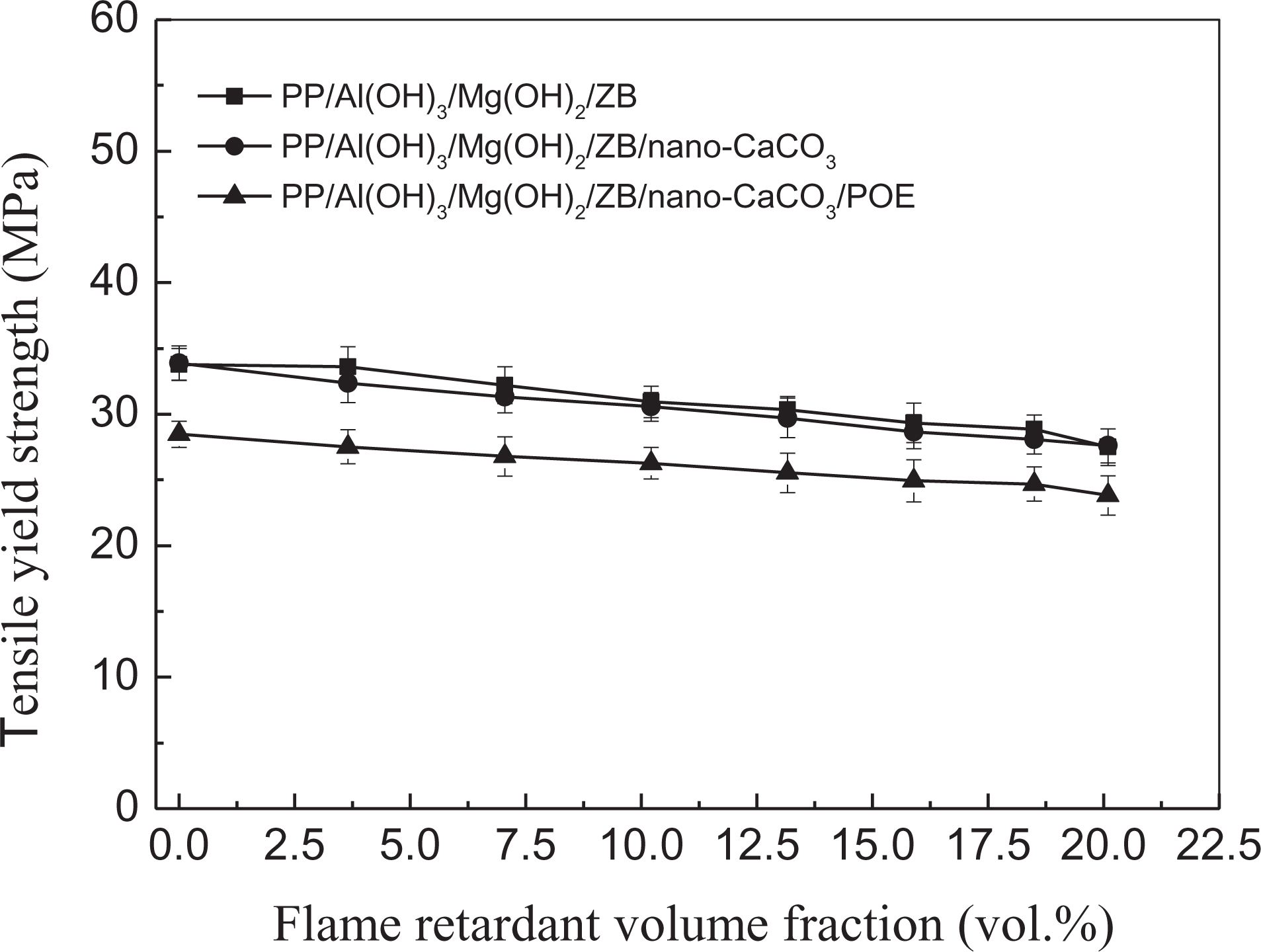
Relationship between tensile yield strength and flame-retardant volume fraction.
Tensile fracture strength is an important characteristic of the tensile fracture toughness or ductility properties of composites under tension load. The correlation between the tensile fracture strength and the flame-retardant volume fraction for the three groups of PP composite systems is shown in Figure 6. Contrary to the situation in Figure 5, the tensile fracture strength increased slightly with increasing flame-retardant volume fraction. This can also attributed to the relatively good interfacial adhesion between the flame-retardant particles and the PP matrix. Furthermore, the tensile fracture strength of the PP/Al(OH)3/Mg(OH)2/ZB composites was the highest while the tensile fracture strength of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites was the lowest for same flame-retardant volume fraction.
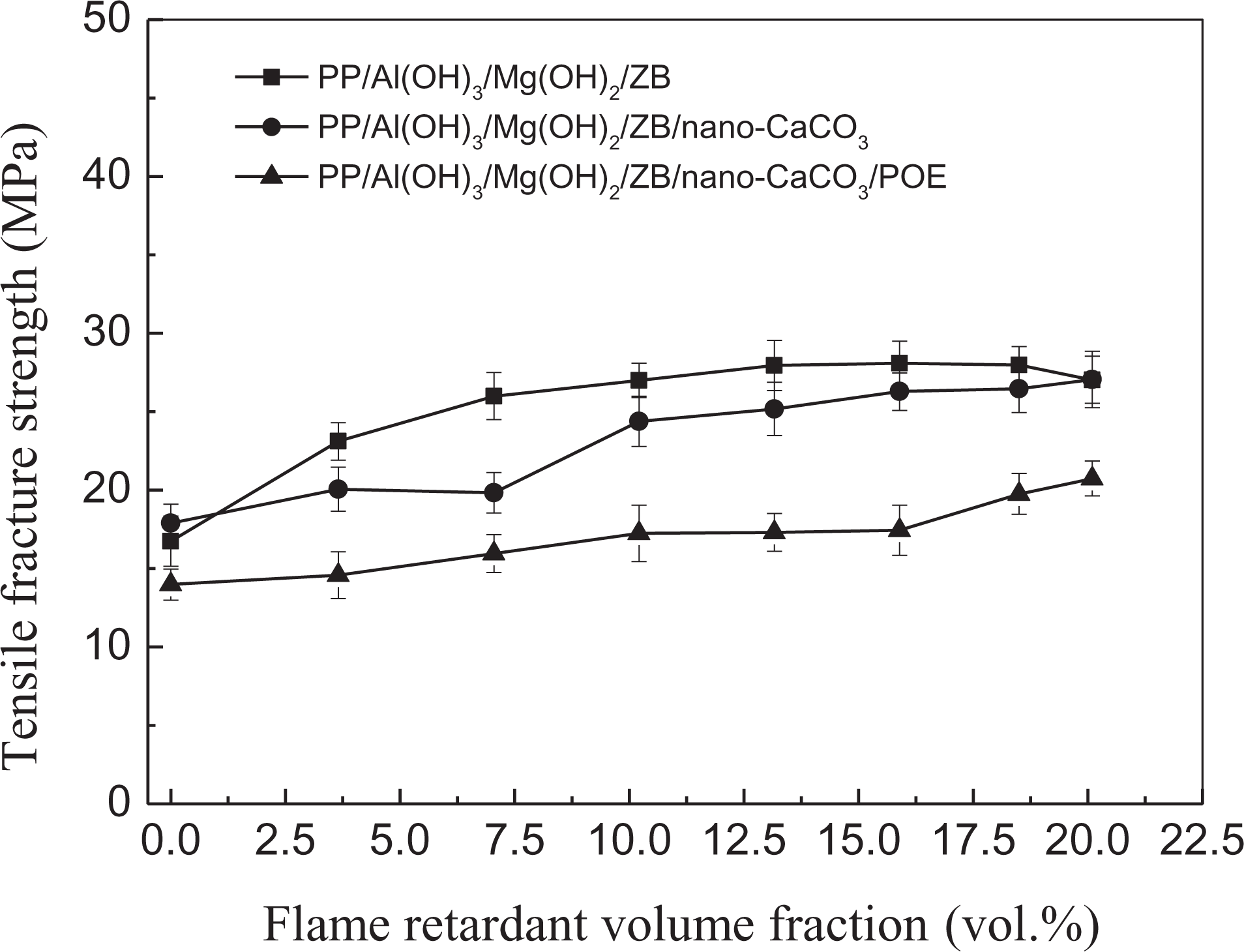
Correlation between tensile fracture strength and flame-retardant volume fraction.
In addition to good interfacial adhesion between the fillers and the matrix, the tensile strength of polymer composites depends, to a great extent, upon the dispersion status of the fillers in the matrix. Figure 7 shows an SEM photograph of the fracture surface of the PP/Al(OH)3/Mg(OH)2/ZB composite; Figure 8 shows an SEM photograph of the fracture surface of the specimen for the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composite; and Figure 9 shows an SEM photograph of fracture surface of the specimen for the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composite. From Figures 7 to 9, all the flame-retardant volume fraction was 7.05 vol.%. It can be seen that dispersion of the fillers, including the Al(OH)3/Mg(OH)2, the ZB, and the nano-CaCO3 particles, in the PP matrix was approximately uniform. Moreover, the interfaces between the fillers and the matrix were unclear, and some were pulled out. This means that the interfacial adhesion between the fillers and the matrix was good, leading to the improvement of the tensile strength of the composites (see Figures 5 and 6).

SEM photograph of specimen fracture surface of PP/Al(OH)3/Mg(OH)2/ZB composites (

SEM photograph of specimen fracture surface of PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composite (

SEM photograph of specimen fracture surface of PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composite (
Dependence of tensile elongation at break on flame-retardant content
Figure 10 shows the dependence of the tensile elongation at break on the flame-retardant volume fraction for the three groups of PP composite systems. For both the PP/Al(OH)3/Mg(OH)2/ZB composites and the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composites, the tensile elongation at break decreased with increasing flame-retardant volume fraction. When flame-retardant volume fraction was lower than 16 vol.%, the tensile elongations at break of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composites were obviously higher than those of the PP/Al(OH)3/Mg(OH)2/ZB composites, and then, the difference in the tensile elongations at break between them decreases. For the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites, when the flame-retardant volume fraction was lower than 16 vol.%, the tensile elongations at break were much greater than those for the other two systems, and it decreases slightly only with increasing flame-retardant volume fraction; then, it decreases considerably with increasing flame-retardant volume fraction when the flame-retardant volume fraction was higher than 16 vol.%. The reason is that the damage of the flame-retardant particles to the ductility of the PP composites was relatively low in the case of low flame-retardant content, while the POE could deform with the matrix under the tensile load owing to the good elasticity of the POE and the good interfacial adhesion between the POE and the matrix. Moreover, the POE could absorb the corresponding deformation energy during the specimen tension. Consequently, the tensile ductility of the composites could be improved significantly by adding the POE.
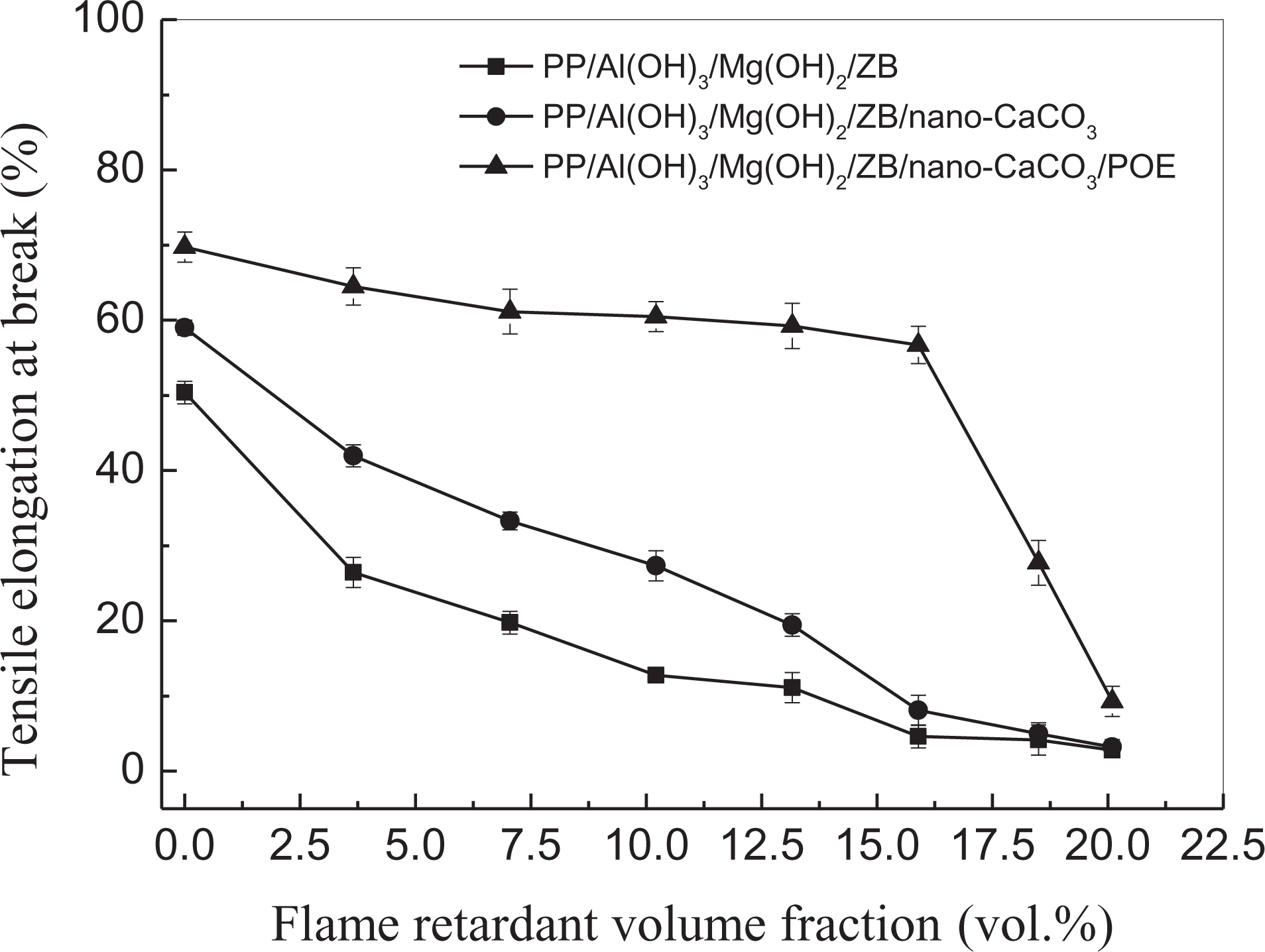
Dependence of tensile elongation at break on flame-retardant volume fraction.
It is generally believed that the introduction of various types of filler particles into polymer, if their surfaces are not properly modified, will form defects in the matrix. These defects will weaken, to some extent, the integrity and continuity of the matrix. Thus, in the cases of the higher flame-retardant contents, the defects in the composites would increase obviously, while the integrity and continuity would be reduced correspondingly. Therefore, the damage of the flame-retardant particles to the ductility of the PP composites in the absence of the POE would be increased. As a result, the tensile elongation at break of the composites decreases considerably.
Estimation of tensile yield strength
As stated earlier, in addition to the dispersion status of the inclusions in the matrix, the tensile strength of a polymer composite primarily depends on the interfacial adhesion status between the filler and the matrix. Thus, the evaluating of the interfacial adhesion strength is a key for predicting the tensile strength of polymer composites. In the case of poor interfacial adhesion between the inclusions and the matrix, the tensile yield strength (

where
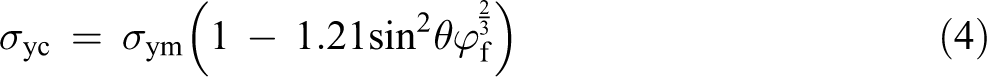
where
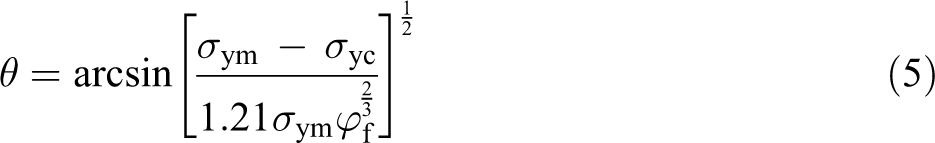
To investigate the interfacial adhesion status between the fillers (including the flame-retardant particles and the other inclusions) and the PP matrix, the tensile yield strength of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites was estimated using equations (3) and (4), and then, the calculations were compared with the measured data from the composites, the results of which are shown in Figure 11. It should be noted that the interfacial adhesion angle was about 40° for the flame-retardant particles, determined using equation (5) from the experimental data of the composites. It can be seen in Figure 11 that the values of the predicted tensile yield strength using equation (4) were close to the measured data from the PP composites, while the difference between the estimated tensile yield strength using equation (3) and the measured data increased with increasing flame-retardant volume fraction. On the basis of the abovementioned discussion, it can be concluded that the interfacial adhesion between all of the filler particles and the PP matrix for these composites was good.
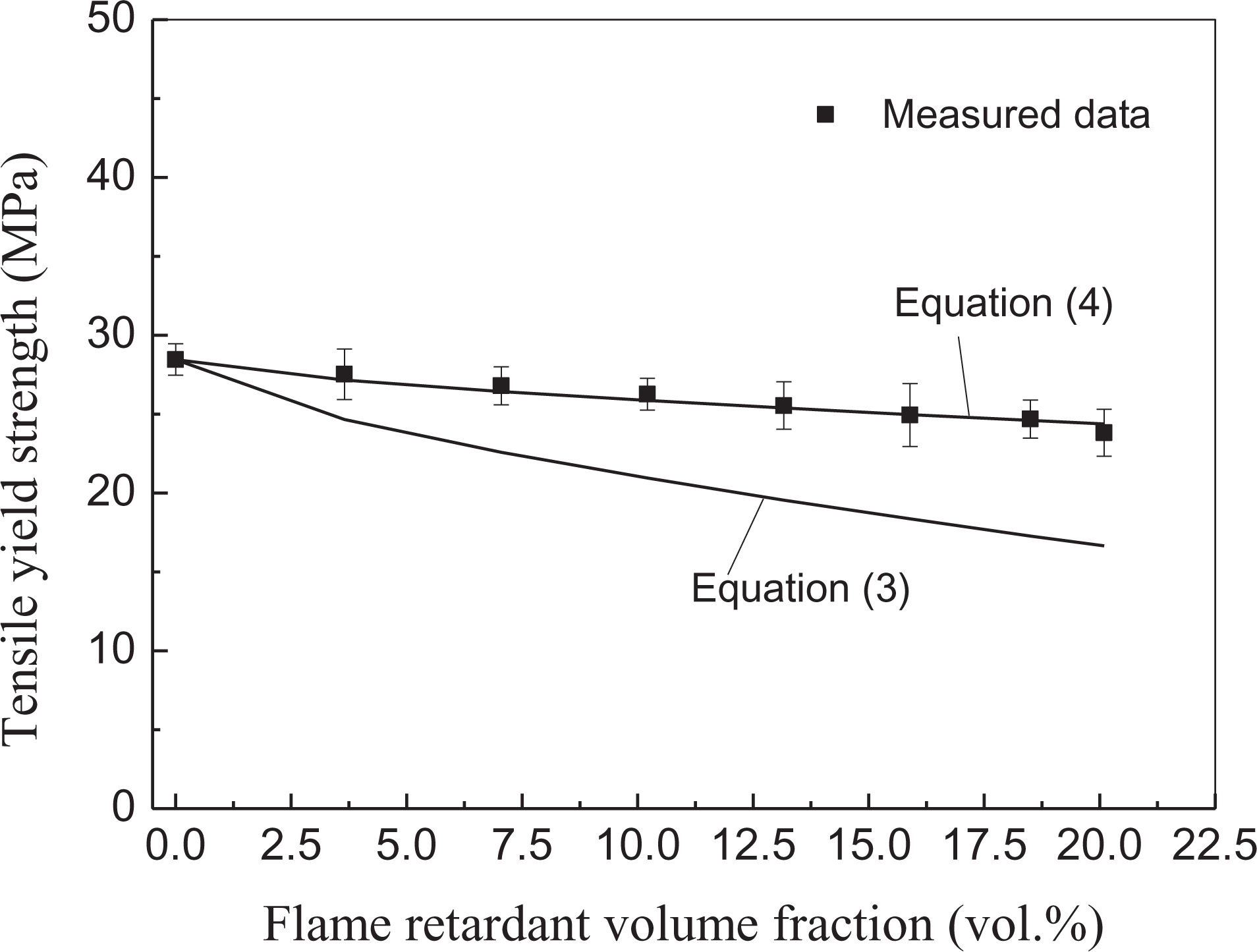
Comparison between estimations of tensile yield strength and measured data from PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites.
Fire residue morphology of the specimen
Effect of flame-retardant content on fire residue morphology
It is extensively believed that the fire residue morphology of specimens after combustion or burning is closely related to the flame-retardant behavior and mechanisms besides the fire behavior.16,18,29 The LOI is one of the important parameters for characterizing the flame-retardant properties of polymer materials. Table 1 lists the LOI of the neat PP and the three PP composites. It can be seen that the LOI increases with increasing the flame-retardant weight fraction, and the ranking of the LOI of the three groups of PP composite systems is as follows: PP/Al(OH)3/Mg(OH)2/ZB < PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 < PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE. Figure 12 shows the images of the burned surface morphology of the specimens for the neat PP and the PP/Al(OH)3/Mg(OH)2/ZB composites after the LOI testing. It can be observed that there was only melt morphology and drippings at the fire residue surface (the top zone of the specimens) of the neat PP; it means that the PP had poor flame-retarding effect (or mechanism). On the other hand, the PP/Al(OH)3/Mg(OH)2/ZB composites, even though at low flame-retardant content (e.g. 3.65 vol.%), had a black cover at the top zone of the specimens. This black cover is labeled the fire residue or carbonaceous char, and this fire residue area increased with increasing flame-retardant volume fraction. This is because the hydroxides in Al(OH)3 and Mg(OH)2, firstly, can release a crystalline liquid when they are heated, absorbing part of the heat quantity produced during the specimen combustion; thus, the surface temperature of the specimen was reduced and the degradation speed of the matrix resin is decreased. In general, the hydroxides release the water during decomposition of their OH groups; secondly, the heat insulation layers composed of MgO and Al2O3 could be generated at the surface of combustible matter after the Mg(OH)2 and Al(OH)3 released their water, and these heat insulation layers could not only block the entrance of heat and oxygen but also release combustible gas. Thus, the flame-retardant effect was produced. Furthermore, the concentration of the combustible polymer was reduced with the addition of Al(OH)3 and Mg(OH)2.3,4 This fire residue is a kind of amorphous carbon structure, which its essence is a carbon micro-crystallite. When the fire residue is formed, the combustion zone of the specimen will not burn and could arrest the heat transfer between the polymer and the heat source, leading to increasing the thermal degradation temperature of the polymer. Moreover, the porous fire residue can prevent the gas diffusion, blocking the gas diffusion generated in the pyrolyzation. Meanwhile, the porous fire residue can stop the external oxygen from diffuse to the surface of the no splitting decomposition polymer. As a result, the burned polymer composite will be self-extinguishing when the burning does not have sufficient oxygen and heat energy, leading to an increase of the LOI with increasing the Al(OH)3/Mg(OH)2 content.3,4 We previously measured the LOI of the PP/Al(OH)3/Mg(OH)2 composites and the PP/Al(OH)3/Mg(OH)2/POE/ZB nanocomposites and found that the LOI increased with increasing the Al(OH)3/Mg(OH)2 weight fraction.3,4 This indicates that the formation of the porous fire residue is one of the major flame-retardant mechanisms of these composites. In addition, ZB is also helpful to improve the flame-retardant properties of the PP composites. Younis 18 evaluated the flammability of the PP after using ZB and montmorillonite and found that the burning rate decreased by increasing the time of ignition with a slight increase in LOI.
LOI of the neat PP and the three PP composites.
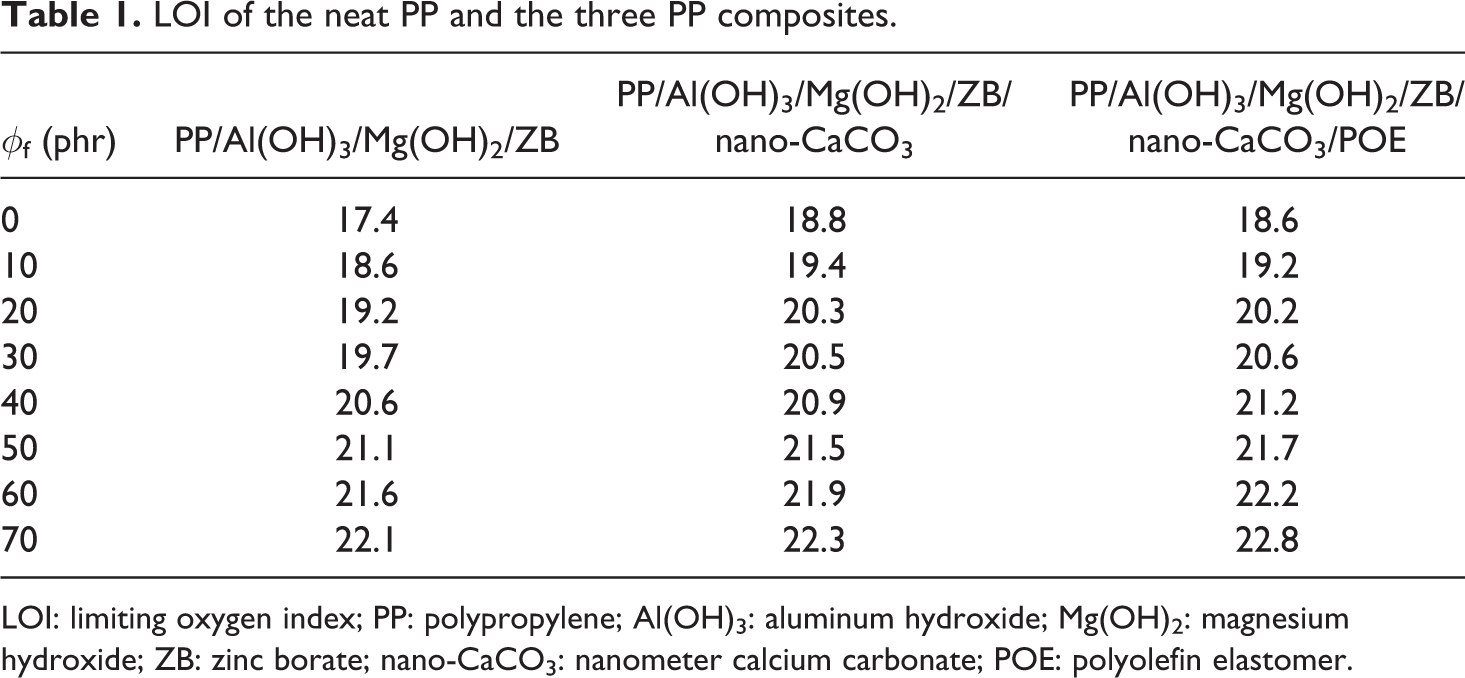
LOI: limiting oxygen index; PP: polypropylene; Al(OH)3: aluminum hydroxide; Mg(OH)2: magnesium hydroxide; ZB: zinc borate; nano-CaCO3: nanometer calcium carbonate; POE: polyolefin elastomer.

Images of fire residue surface morphology of specimens of PP/Al(OH)3/Mg(OH)2/ZB composites. (a) PP, (b) 3.65vol.%, (c) 7.05 vol.%, (d) 10.21 vol.%, (e) 13.16 vol.%, (f) 15.89 vol.%, (g) 18.50 vol.%, and (h) 20.10 vol.%.
Effect of composition on fire residue morphology
Figure 13 shows the pictures of the fire residue morphology of the specimens for the three groups of composite systems with flame-retardant volume fraction of 3.65 vol.% after the LOI testing; the fire residue area or thickness of the PP/Al(OH)3/Mg(OH)2/ZB composites was the smallest, while the fire residue area or thickness of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composite was the largest in the three groups of composite systems. This is because the nano-CaCO3 is a kind of nonflammable substance, the relative density of POE was small, and the flame-retardant property of POE was less than that of PP; in addition, the PP in the composites was reduced with the addition of the nano-CaCO3 and POE under the same conditions. Consequently, the flame-retardant property of the PP/Al(OH)3/Mg(OH)2/ZB composites was poorer than that of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3 composites and the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites due to loading with the nano-CaCO3 and the POE. Figure 14 shows the images of the specimen morphology of the three groups of composite systems with a flame-retardant weight fraction of 10.21 vol.% after the oxygen index testing. It can be seen that the each composite system had a fire residue at the combustion end of the specimen, with completely covering the end of the samples, and the phenomenon of carbon deposition is becoming obvious relative to the samples in Figure 13. Moreover, the fire residue images of these three groups of composite systems were similar. This indicates that the effect of the other compositions, the nano-CaCO3 and POE, on the flame retardant of the PP composites was insignificant in the case of higher concentrations of the Al(OH)3 and Mg(OH)2.
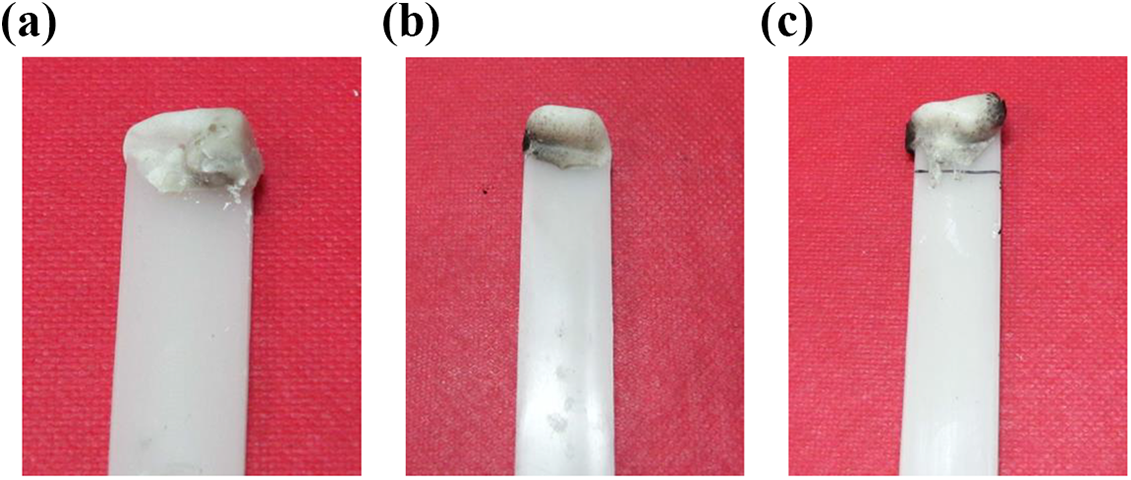
Images of fire residue surface morphology of specimens of three groups of composite systems (

Images of fire residue surface morphology of specimens of three groups of composite systems (
Conclusions
This report described the effects of the Al(OH)3/Mg(OH)2 content on the tensile properties and the fire residue morphology after burning for the unfilled PP and the three groups of filled PP composite systems within the flame-retardant volume fraction range being of 3–20 vol.%. For the same nano-CaCO3 and POE additive content, the Young’s modulus and the tensile fracture strength increased while the tensile yield strength and the tensile elongation at break decreased with increasing flame-retardant volume fraction. The tensile elongation at break of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites was obviously higher than that of other two composite systems. It was found that the fire residue area (or volume) in the pyrolysis zone of the specimens increased with increasing the flame-retardant weight fraction and also increased due to the loading of the nano-CaCO3 and POE, especially in the case of lower concentrations of the flame retardants. In addition, the tensile yield strength of the PP/Al(OH)3/Mg(OH)2/ZB/nano-CaCO3/POE composites was estimated using a tensile strength equation proposed previously by the author, and the predictions were close to the measured data. Thus, we concluded that there was a good interfacial adhesion between the filler and the matrix.
