Abstract
Ethylene vinyl alcohol (EVOH) with excellent barrier properties has insufficient thermomechanical properties. The introduction of magnesium chloride (MgCl2) as an initiator in EVOH blends improved its properties by cross-linking. Torque behavior and gel experiment analysis indicated that a cross-linking in EVOH was formed. The cross-linking mechanism was confirmed through 13C nuclear magnetic resonance spectroscopy (13C NMR) and Fourier-transform infrared (FTIR) spectrometry. In 13C NMR spectra, the splitting peaks of CH carbon and CH2 carbon tended to disappear, and the stretching vibration peak of –C=C– was observed in the FTIR spectra. The formation of hydrogen bond between MgCl2 and EVOH destroyed the intramolecular and intermolecular hydrogen bonds of EVOH, which contributed to the dehydration of –OH to form –C=C–, and –C=C– was the basis for a cross-linking reaction. The thermal analysis of blends demonstrated that the melting temperature and crystallization temperature decreased, and the crystallinity gradually disappeared when the MgCl2 content increased. Glass transition temperature significantly increased as the intermolecular force enhanced. Thermogravimetric analysis showed that a cross-linked structure could improve the thermostability of EVOH with an increase in the MgCl2 content. Mechanical test results revealed a remarkable increase in the tensile strength of EVOH as the MgCl2 content increased.
Introduction
Studies have been conducted to preserve flavor components in food throughout their required shelf life, and most of them aim to determine and choose packaging materials that are a good fit for a given product. 1 Ethylene vinyl alcohol (EVOH) is a key material of interest because of its outstanding barrier properties to oxygen and carbon dioxide,2–4 high resistance to hydrocarbons, and good processability. 5 A random copolymer consists of ethylene units, which contribute to processability and flexibility, and vinyl alcohol units, which are responsible for high barrier properties. 6 The barrier property of EVOH due to its small free volume in between molecules is from polyvinyl alcohol (PVA), which increases as vinyl alcohol content increases in terms of grade, and processing property is attributed to polyethylene. 7 These materials have been widely used in the packaging industry as barrier layers to protect foods from the ingress of oxygen and losses of flavors and consequently to increase package shelf life. However, some drawbacks in the mechanical properties and thermal properties of EVOH and its brittleness limit its application. The hydrogen bond formed by hydroxyl groups in EVOH is insufficiently strong, causing an adverse effect on the mechanical and thermal properties of EVOH. Some efficient approaches must be used for probing to improve the properties of EVOH and further exploit its usefulness in more fields in the future.
Polymer blending technology is one of the major areas of research and development. Publications, patents, and research theses have shown that blending existing polymers has an added advantage over conventional polymers. In this study, blending technology involves the modification of physical, mechanical, and chemical properties of commodity to engineer a range of products. 8 The methods of modifying EVOH by blending with other materials have been reported. The most common modification method is the addition of plasticizers9–11; however, the mechanical and barrier performance of EVOH slightly decrease with the introduction of plasticizers. Active hydroxyl groups in EVOH can interact with other polymers that contain amino, carboxyl, acid anhydride, and epoxy groups.12–14 In the present method, compatibility is important and can directly lead to a decreasing effect on the properties of EVOH. 15
Recent studies have focused on the blending modification of EVOH. Li et al. 16 prepared boric acid that induces cross-linked EVOH/graphene oxide nanocomposite films, but the preparation method is complicated and costly.
Accordingly, in the present study, cross-linking would be a simple way to overcome EVOH deficiencies. Thus, a cross-linked structure could improve the tensile strength and thermostability of EVOH. Magnesium chloride (MgCl2), an inorganic substance, was selected as a catalyst for the cross-linking reaction. The cross-linked EVOH was discussed in terms of viscosity, thermal properties, and mechanical properties.
Experimental
Materials
EVOH (F101A) was purchased in a solid form from Kuraray Corporation, Japan, and it had an ethylene concentration of 32% and a melt flow index of 1.6 g 10 min−1 (190°C, 2.16 kg). Anhydrous MgCl2 was obtained from Shanghai Aladdin Biochemical Technology Co., Ltd, China. N, N-dimethylacetamide (DMAc) was also procured from Shanghai Aladdin Biochemical Technology Co., Ltd, China.
Preparation of neat EVOH and EVOH/MgCl2 blends
EVOH was dried in a vacuum at 80°C for 8 h, and MgCl2, Mg(OH)2, CaCl2, BaCl2, CaO, and MgO were dried in an oven at 100°C for 4 h before blending. The blends of EVOH/metal compounds with compositions of 99/1 were prepared. Four samples of EVOH/MgCl2 with compositions of 100/0, 99/1, 95/5, and 90/10 (w/w) were prepared and marked as M0, M1, M5, and M10, respectively. The blends were introduced to a mixer and blended at 100 r min−1 for 6 min at 195°C. The torques during blending were automatically recorded to analyze processability and viscosity of the blends. After blending was completed, the samples were molded into a 0.5-mm-thick film by using a hot press and quenched to room temperature (25°C). The pressing temperature was 200°C, and the pressing time with a pressure of 10 MPa lasted 10 min.
Torque rheometry measurements
Torque during blending in the mixer was automatically recorded to analyze the rheological behavior of M0, M1, M5, and M10. In the internal mixer (XSS 300, Shanghai Kechuang Rubber & Plastic Machinery Equipment Co. Ltd., China), the mixing torque was continuously recorded as a function of the blending time.
Gel experiment
DMAc was selected as a suitable solvent to dissolve samples in a water bath at 85°C. Then, the dissolved samples were dried at 80°C and weighted.
The samples were boiled in water at 100°C, dried at 80°C, and weighed. The quantities of the samples before and after boiling were m1 and m2, respectively.
13C nuclear magnetic resonance spectroscopy
13C nuclear magnetic resonance (13C NMR) cross-polarization magic angle spinning spectra were recorded on a 500 MHZ/AVANCEIII spectrometer (Bruker, Switzerland). All the samples were solid. Analysis was performed using MestReNova software (Mestrelab Research, Spain).
Fourier-transform infrared spectroscopy
The Fourier-transform infrared (FTIR) spectra of the samples were recorded using a Nicolet 6700 spectrometer (Thermo Fisher, Waltham, Massachusetts, USA). Sample power was mixed with KBr powder and ground into fine powder. Subsequently, the mixtures were compressed into a round transparent tablet. The frequency range of FTIR was from 4000 cm−1 to 400 cm−1 against KBr as the background.
Thermal properties
Differential scanning calorimetry
The differential scanning calorimetry (DSC) of the blends was determined in a modulated DSC 2910 (TA Instruments, New Castle, Delaware, USA) under a nitrogen flow of 20 mL min−1. The sample weights were approximately 8 mg. The scanning rate used throughout the investigation was 10°C min−1. Pure EVOH and the blend samples were first heated from 25°C to 230°C and then held for 3 min to eliminate heat. The crystallization behavior was determined from the cooling step, and melting points were evaluated from the second heating run at a rate of 10°C min−1.
Thermogravimetric analysis
The thermogravimetric analysis (TGA) measurements of the samples were performed in a TG209F1 TG/differential thermal analysis analyzer (Netzsch, Germany) under a nitrogen protective atmosphere. The samples were heated from 25°C to 600°C with a heating rate of 10°C min−1 and a flow rate of 50 mL min−1. Prior to the test, the samples were dried in a vacuum oven at 80°C for 24 h.
Dynamic mechanical analysis
Dynamic mechanical measurements at 1 Hz were made in a tensile mode by using a Q800 dynamic mechanical thermal analyzer (TA Instruments). Tests were performed at a heating rate of 5°C min−1 from −60°C to 150°C. Rectangular specimens with a width of 5 mm and a length of 30 mm were used.
Mechanical properties
A tensile test was performed at 25°C and relative humidity of 52% by using a universal testing machine (CMT6104, Sans, China). The crosshead speed was set at 10 mm min−1 with a gauge length of 50 mm in accordance with GB/T 1040-2006. Each sample included five tested replicates to obtain a reliable value of tensile strength and standard deviation.
Results and discussion
Torque behavior
Rheology is the theoretical basis for polymer processing, and most polymers are processed through melting and deformation. Through a rheological behavior test, information on the microstructure of polymers can be obtained, and the relationship between the microstructure and behavior can be established.
Torque is one characteristic of rheological behavior. The torque behavior of several common metal compounds is compared in Figure 1(a). A significant improvement in the torque behavior is observed via the addition of MgCl2. Other metal compounds slightly affect torque that decreases when CaO is added. With experimental data in Figure 1, MgCl2 is chosen to improve the properties of EVOH.
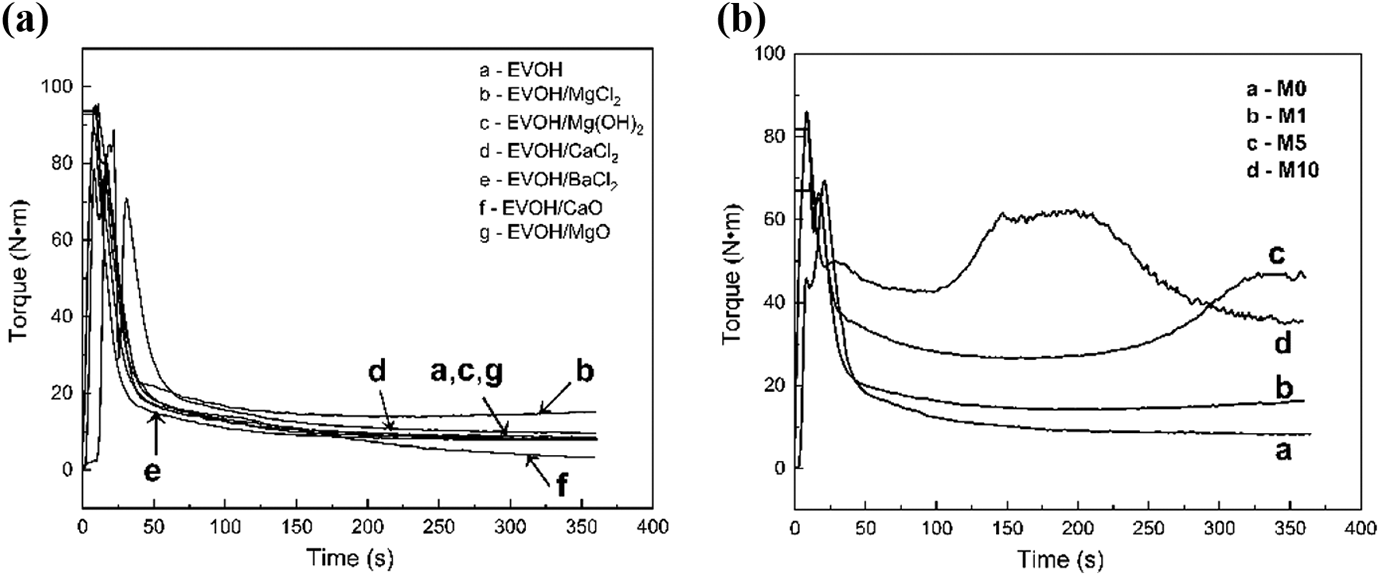
Variation in the torque versus the mixing time of (a) several metal compounds and (b) M0, M1, M5, and M10.
Figure 1(b) shows the torque curves of M0, M1, M5, and M10 measured with the internal mixer at 195°C. The torque of melting polymer is related to the melt viscosity and the melt processability of the blends. Torque increases as the weight percent of MgCl2 increases compared with that of pure EVOH.
Notably, the torques of M10 and M5 sharply increase at 150 and 300 s, respectively. The possible explanation for this behavior is that the cross-linking reaction of EVOH occurs when MgCl2 is added. As the viscosity of M10 increases significantly, the cross-linked molecular chains easily break into multiple cross-linked structures by the shear force in the mixer. These structures have a weak chemical and physical interaction among them, but they have a strong internal interaction. Thus, the torque of M10 subsequently decreases as the part of the cross-linked structure is destroyed; however, the cross-linking degree is still high.
Cross-linking can lead to an increased effect on viscosity, which is indicated by an increase in torque. Accordingly, a cross-linking reaction in EVOH might occur through the addition of MgCl2.
Gel experiment
DMAc was selected as a suitable solvent to dissolve the blends in a water bath at 85°C and to validate this hypothesis. The gel contents of M0, M1, M5, and M10 are 0%, 0%, 69.8%, and 74.4%, respectively. Notably, the gel gradually forms as the MgCl2 content in the blends increases. Gel formation indicates that the cross-linking reaction occurs. The degree of cross-linking of the blends increases as the gel content increases.
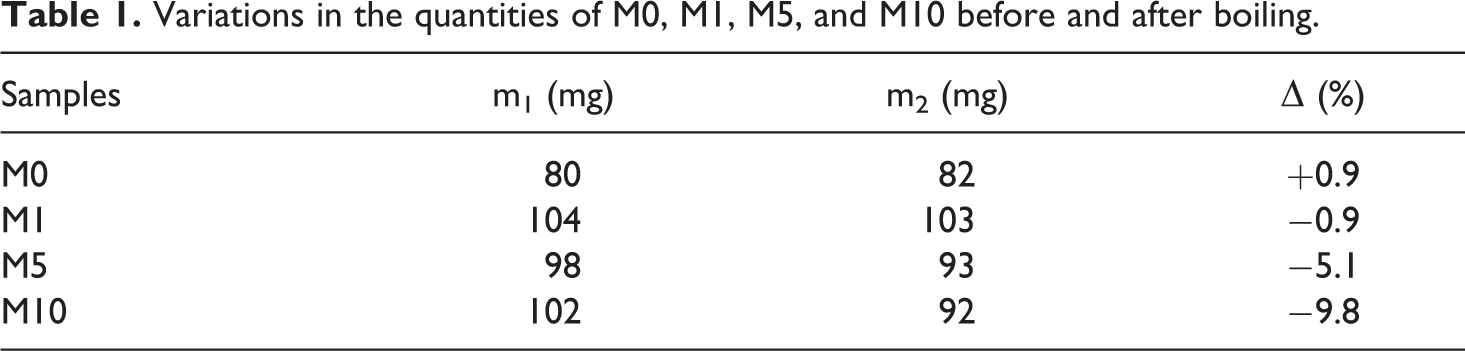
The results after boiling are shown in Table 1. The variation in the quantities of M1, M5, and M10 after boiling is significantly reduced by nearly 1%, 5%, and 10%, respectively. The quantity of MgCl2 may decrease, and MgCl2 only participates in the cross-linking reaction as a catalyst.
Variations in the quantities of M0, M1, M5, and M10 before and after boiling.
Cross-linking mechanism
The following assumptions are made on the basis of the variation in the torque and gel experiment: the cross-linking reaction occurs with the addition of MgCl2, and MgCl2 only as a catalyst participates in the cross-linking reaction. The cross-linking mechanism is analyzed to prove these hypotheses.
The structure of PVA is similar to that of EVOH. PVA is easily degraded, and the initial decomposition temperature is about 200°C, so the thermal decomposition mechanism of PVA is analyzed first. According to the TG and differential thermal gravity (DTG) of PVA, during the thermal degradation of PVA in the inert atmosphere, the shape of the mass loss curve is consistent with the generally accepted two-step mechanisms. The first step is the result of dehydration (elimination of water) accompanied with polyene formation, and the second one is pyrolization that produces some organic volatile products as shown in Figure 2.17-19 The mechanism of the thermal degradation of PVA comprises two groups of reactions: the ones that only affect functional groups at about 200°C (inter- or intramolecular dehydration) and those accompanied with a scission or a modification of the main chain at a high temperature. 20
The processing temperature of EVOH is 195°C, so the first step of PVA thermal degradation is considered. Water elimination accompanied with polyene formation in EVOH involves the same hydroxyl structure as PVA likely occurs.21,22 The cross-linking reaction of EVOH is based on the formation of –C=C–. With the addition of MgCl2, the new hydrogen bond is formed between Cl in MgCl2 and hydroxyl groups in EVOH, whereas the hydrogen bond in EVOH weakens, thereby contributing to dehydration accompanied with –C=C– formation. No interaction exists between Mg in MgCl2 and EVOH. According to the boiling testing result, MgCl2 as a catalyst may still remain in a molecular form. Then, the cross-linking reaction occurs with a –C=C– fracture at a high temperature, as shown in Figure 2.
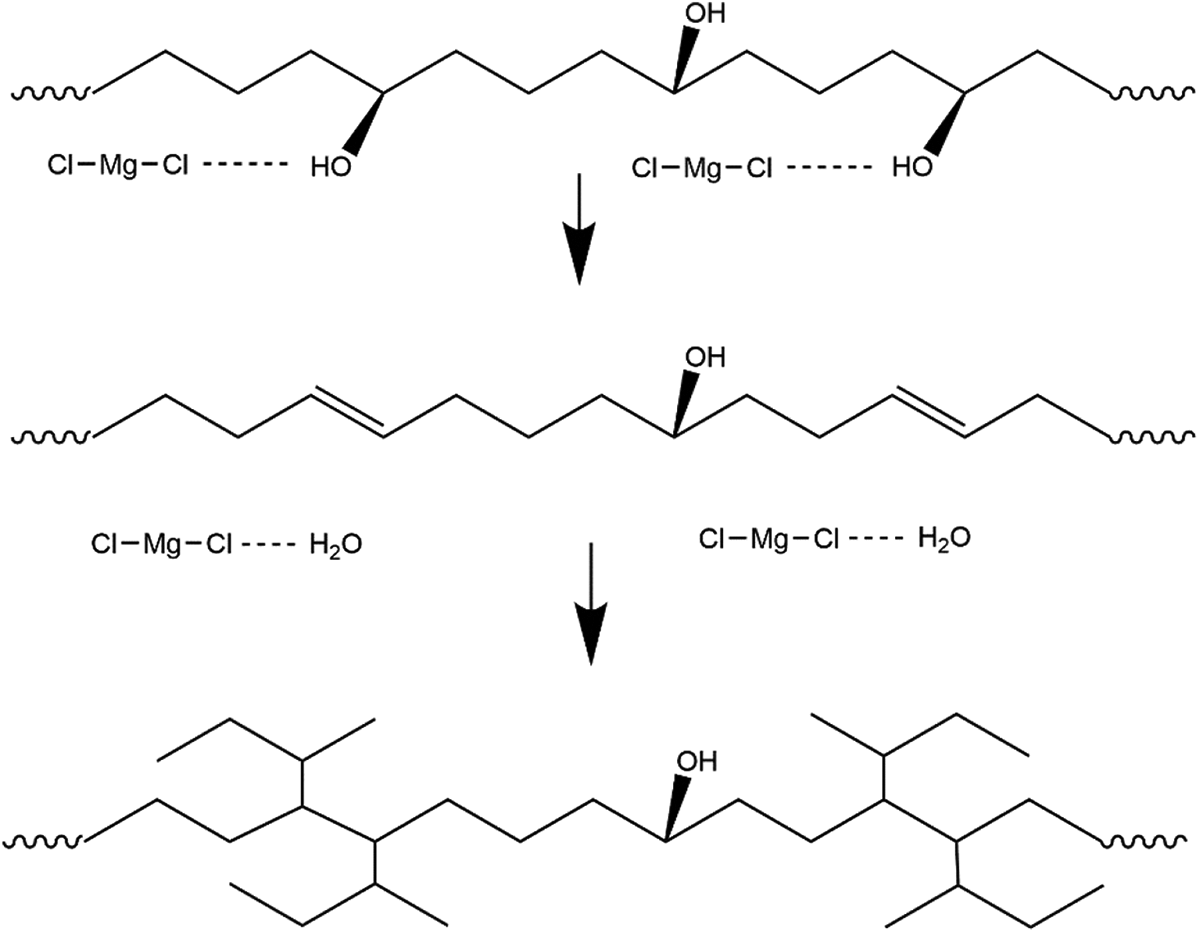
Cross-linking mechanism of EVOH.
The formation of –C=C– and cross-linking reaction was characterized through FTIR and 13C NMR to verify our assumptions.
13C NMR spectra of neat EVOH and EVOH/MgCl2 blends
The 13C NMR spectra of M0, M1, M5, and M10 are shown in Figure 3. In 13C NMR spectra of pure EVOH, two regions of absorption are presented: 0–50 ppm related to all methylene carbons –CH2–(C) and 50–80 ppm corresponding to all methine carbons CH–OH. The 13C NMR signal of CH2 carbon shows three main peaks: the most downfield peak (peak I, 39.03 ppm), the central peak (peak II, 34.48 ppm), and the most upfield peak (peak III, 24.31 ppm). Three splitting peaks are observed at around 24.31 ppm. Similarly, the 13C NMR signal of CH carbon has one main peak (64.14 ppm), and three splitting peaks are caused by the differences in sequence distribution and stereo configuration. 23 The splitting of the peak is attributed to the formation of intramolecular hydrogen bonding. 24 With the addition of MgCl2, the splitting peaks of CH carbon and CH2 carbon tend to disappear. The hydrogen bond of EVOH is destroyed because of the formation of a new hydrogen bond between MgCl2 and EVOH. Combined with the gel experiment, the 13C NMR signal indicates that the production of the cross-linking structure in EVOH reduces the differences in sequence distribution and stereo configuration when the MgCl2 content increases. The 13C NMR signal of –C=C– between 100 ppm and 150 ppm is also observed in Figure 3, which can verify the proposed assumption.
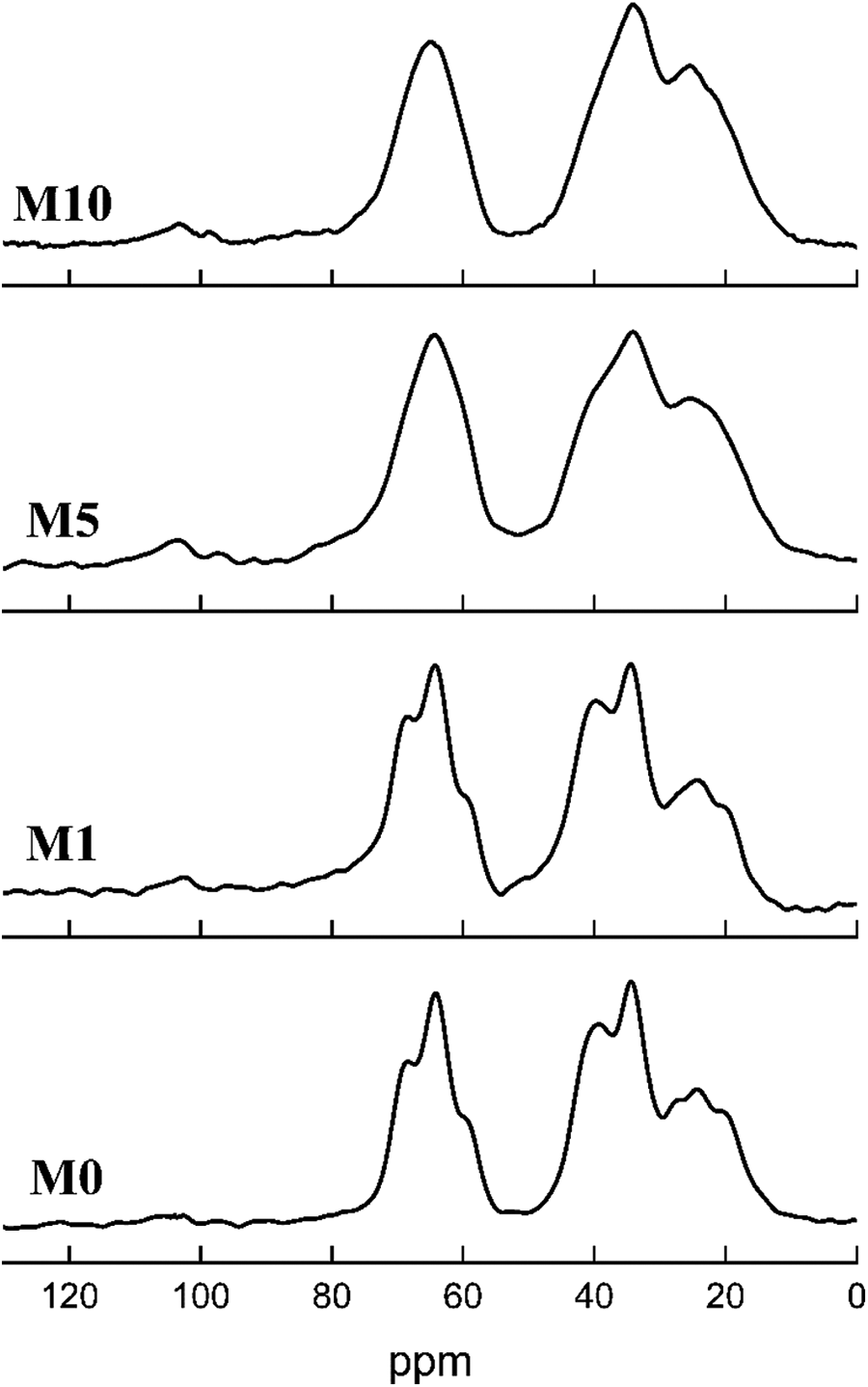
Solid-state 13C NMR of the samples: M0, M1, M5, and M10.
FTIR spectra of pure EVOH and EVOH/MgCl2 blends
The infrared absorption peak of the free –OH (no hydrogen bond) is between 3610 cm–1 and 3640 cm–1, and the shape of peak is sharp. When a hydrogen bond forms, the infrared absorption peak shifts to a lower wave number, and the peak widens. The intensity of the hydrogen bond can be indicated by the variation in a hydrogen bond peak. When the hydrogen bond strengthens, the infrared absorption peak shifts to a lower wave number; conversely, this peak shifts to a higher wave number when the hydrogen bond weakens.
As shown in Figure 4, an intense broad band centered at approximately 3390 cm−1 that corresponds to hydroxyl stretching is found in the spectra of EVOH specimens, which are attributed to the intermolecular and intramolecular hydrogen bonds in EVOH. After 1% MgCl2 in EVOH is blended, the peak wave number of the absorption bands that correspond to the hydroxyl stretching of EVOH shifts from 3385 cm−1 to 3379 cm−1, which illustrates that the hydrogen bond strengthens upon the addition of MgCl2. Conversely, the absorption peak of –OH shifts to a higher wave number as the MgCl2 content increases, indicating that the hydrogen bond weakens. Thus, the weakened hydrogen bond inter- and intramolecular hydrogen bonding between the hydroxyl groups of EVOH and the characteristic peaks appear at 1650 cm−1, which is attributed to the –C=C– stretching vibration and confirmed the aforementioned assumption. No absorbance is noted above 3000 cm−1 for the double bond possibly because the peaks of –OH and –CH overlap with the –C=C– peak.
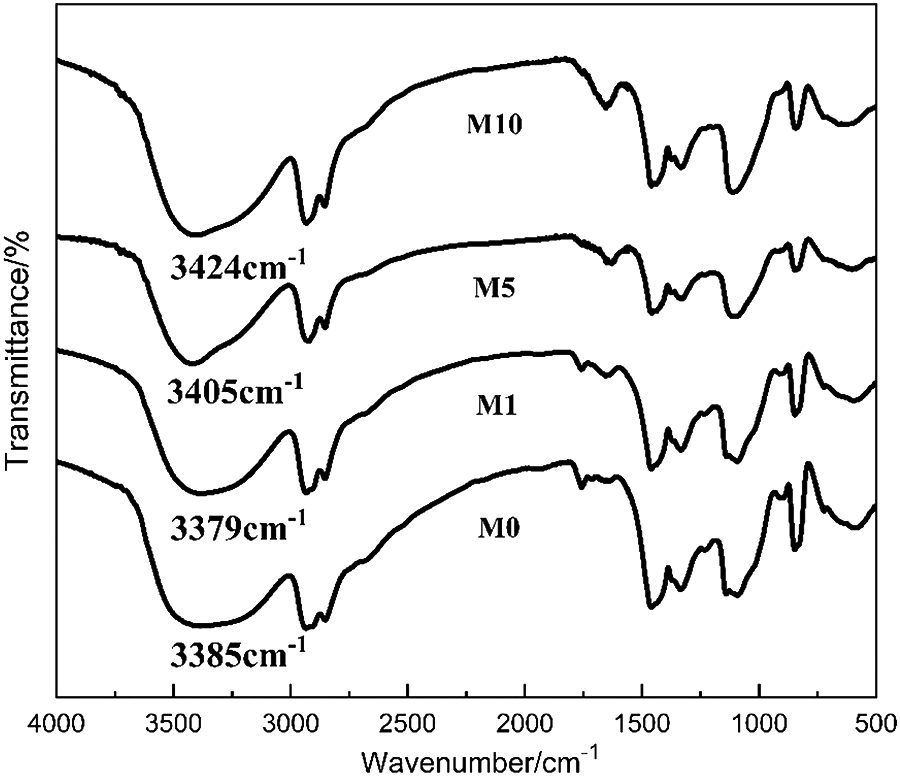
FTIR spectra of the samples: M0, M1, M5, and M10.
Thermal behavior of EVOH and EVOH/MgCl2 blends
Crystallization characteristics and DSC curves
M0, M1, M5, and M10 were subjected to thermal characterization through DSC measurement. Figure 5(a) and (b) shows the cool and second heat scans of the DSC thermograms of four samples. The experimental data in Table 2 indicate that the crystallization temperature (Tc) and melting temperature (Tm) of EVOH are at 160.3°C and 183.4°C, respectively. Tm and Tc significantly decrease as MgCl2 is introduced, and they cannot be detected when the weight percent of MgCl2 increases. So does the crystallinity (χc). The cross-linking structure in EVOH upon MgCl2 addition from M0 to M1 may not be inferred precisely; however, decreasing trends in Tm, Tc, and χc can illustrate that crystallization and interactions between EVOH molecules are destroyed by the addition of MgCl2. These phenomena may be the result of slight cross-linking. The missing peaks of M5 and M10 can be used as a support for cross-linking in EVOH.
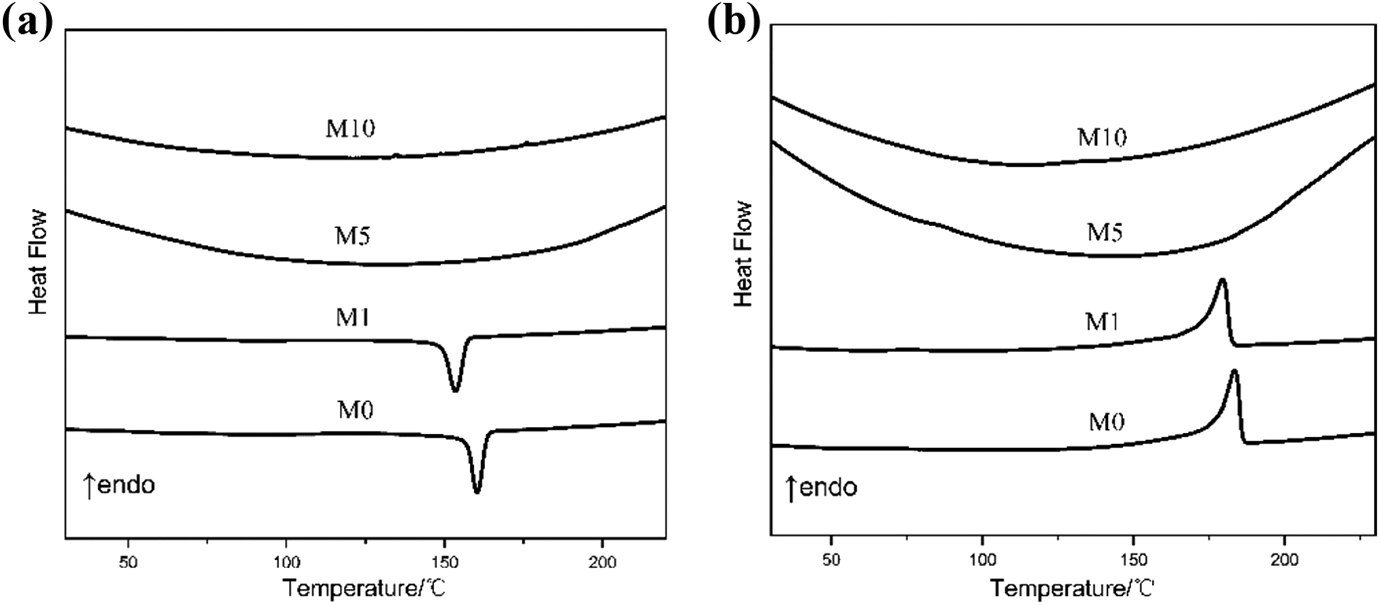
DSC curves of M0, M1, M5, and M10: (a) cooling curve and (b) second heating.
DSC results of M0, M1, M5, and M10.

DSC: differential scanning calorimetry; Tm: melting temperature; Tc: crystallization temperature; χc: crystallinity.
The possible explanation for the abovementioned behavior is that strong intermolecular interactions form, and molecular chain movement is restricted when MgCl2 is added; as a result, no significant variation in crystal enthalpy is observed. 25 The results agree with the NMR and FTIR spectra, implying that the cross-linking reaction occurs when MgCl2 is added to EVOH.
Thermogravimetric analysis
The TG and DTG curves of M0, M1, M5, and M10 are shown in Figure 6. Three degradation processes occur upon heating from 25°C to 600°C. The first step is dehydration to form –C=C–. The second one is the breakage of the main chains to produce some polymers with a low molecular weight, and the final one is carbonization. MgCl2 addition leads to the acceleration of the first reaction step. Then, the second reaction step occurs at a higher temperature than that of pure EVOH; therefore, the possible explanation for this behavior is that a cross-linked structure is present to improve the thermostability of EVOH. The result of this work clearly states that the cross-linking reaction contributes to the thermal properties of EVOH.
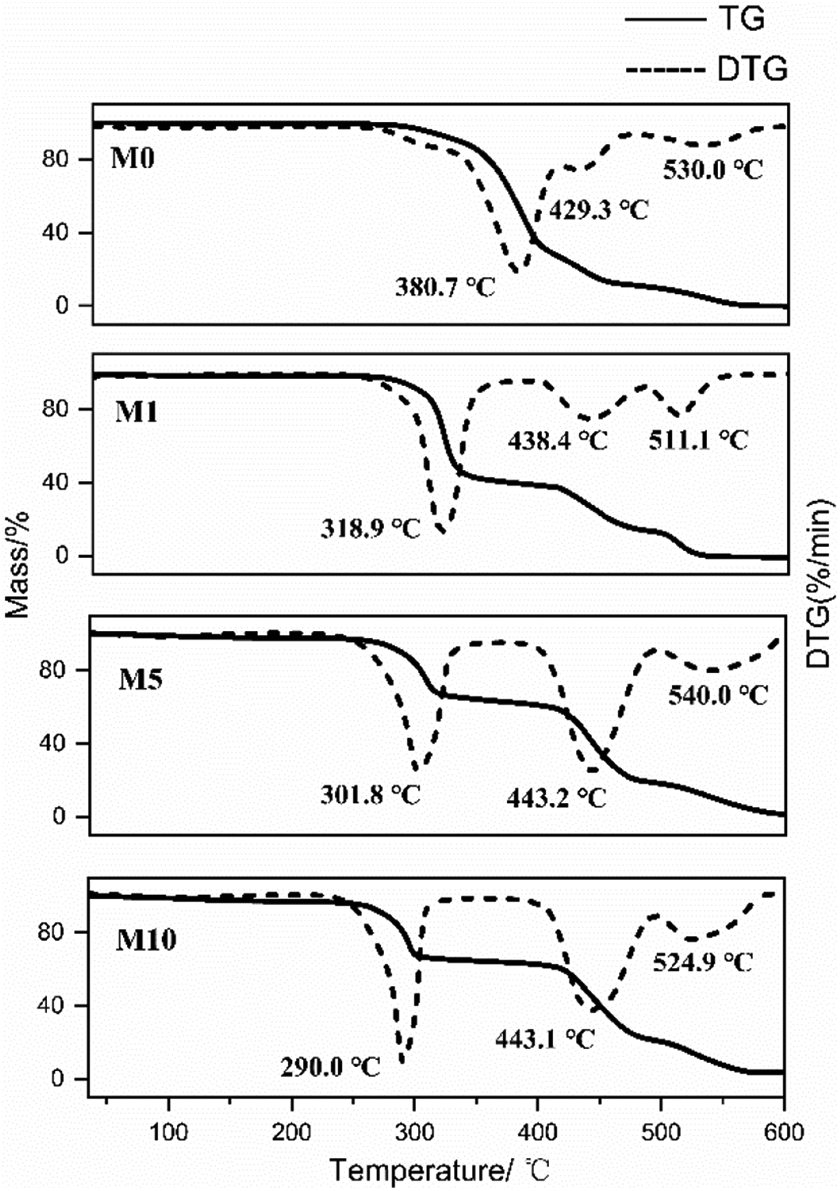
Thermogravimetric curves of M0, M1, M5, and M10.
Dynamic mechanical analysis
A series of storage modulus curves and loss factor (tan δ) as a function of temperature that ranges from −50°C to 150°C for M0, M1, M5, and M10 are depicted in Figure 7. It shows obvious platforms for rubber in M5 and M10, which can prove the formation of the cross-linked structure. Figure 7(b) shows a dynamic relaxation peak that appears in all the samples. According to Ishak and Berry, 26 the α relaxation peak is believed to be related to the breakage of the hydrogen bond between polymer chains that induce long-range segmental chain movement in an amorphous area. This area is assigned to the glass transition (Tg) temperature of the blends. Notably, Tg is significantly higher when the MgCl2 content increases. This result indicates that the interaction between the molecular chains of the blend remarkably enhances, and the movement of the molecular chain is restricted and leads to an increase in Tg. This result can be attributed to a cross-linking reaction. 27
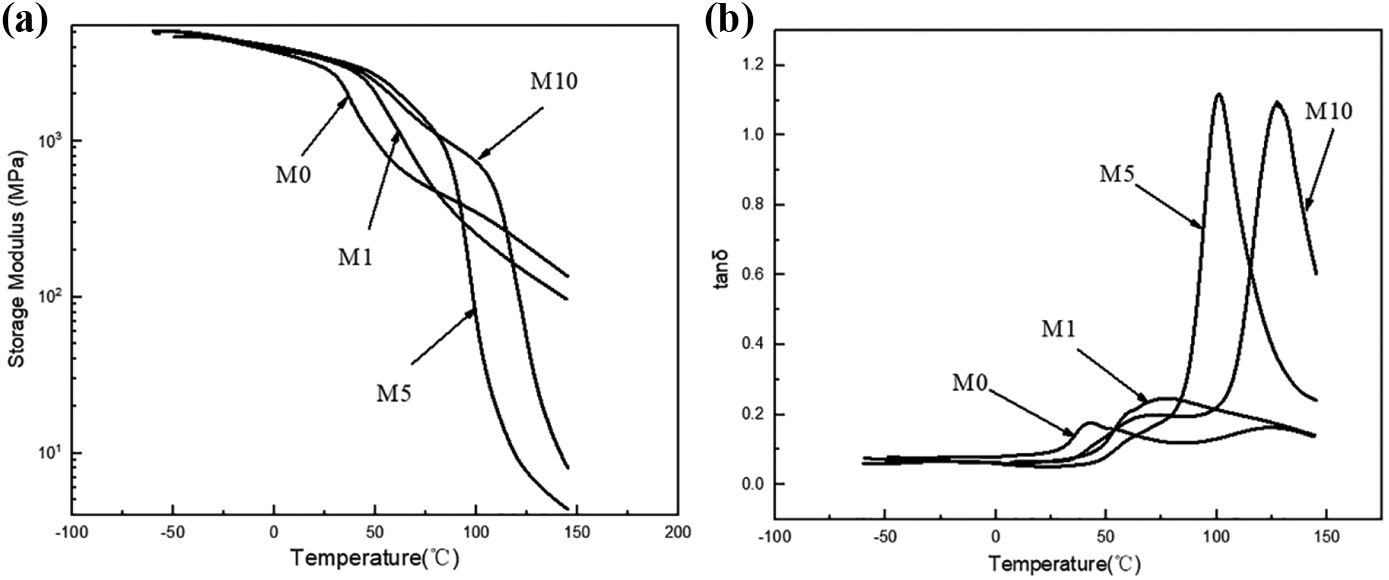
DMA curves of M0, M1, M5, and M10: (a) storage modulus-temperature curve and (b) tan δ-temperature curve.
Mechanical properties
Figure 8 shows the behavior of M0, M1, M5, and M10 under tensile loading, and Figure 9 presents their values. The tensile strength of EVOH increases as the MgCl2 content increases. However, the elongation of M10 decreases drastically.
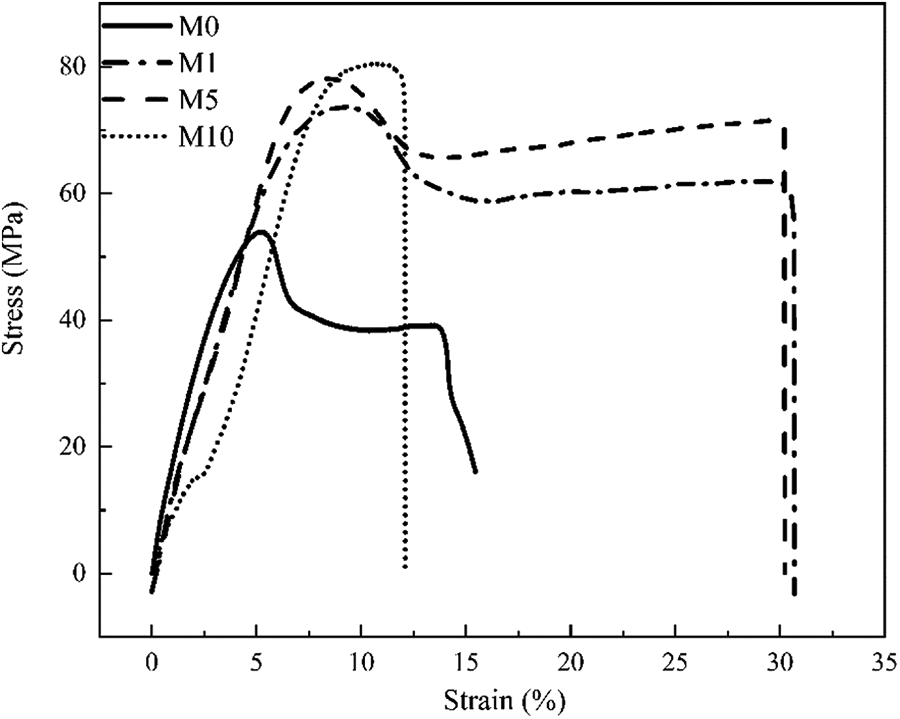
Stress–strain curves of M0, M1, M5, and M10.
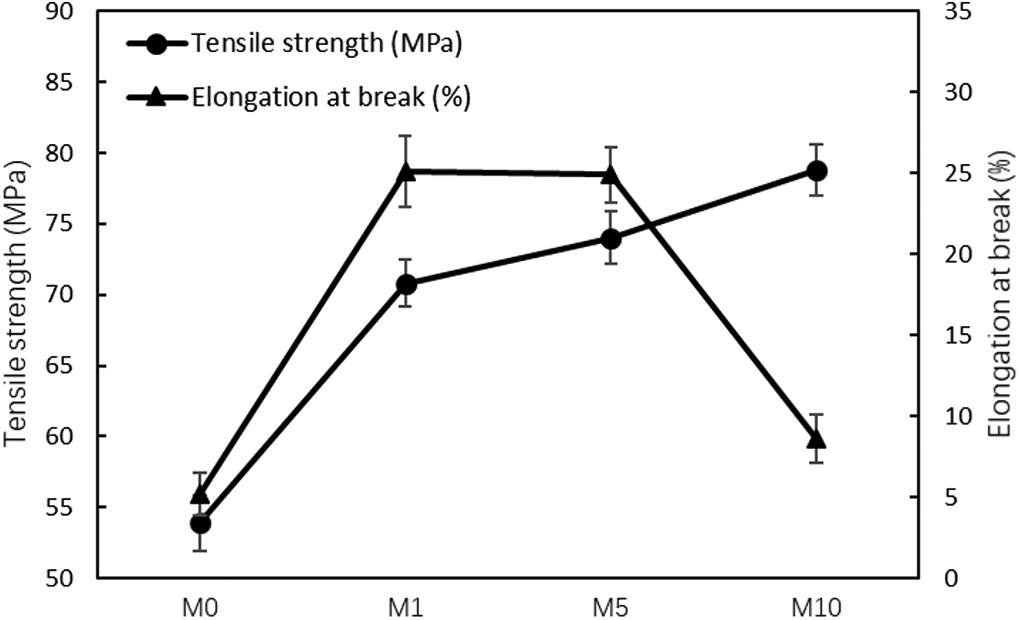
Tensile strength and elongation at break of M0, M1, M5, and M10.
As explained in the above section, the amount of cross-linking increases as the MgCl2 content increases, resulting in an increase in tensile strength. A dense network structure that results in the restriction of EVOH chain movement leads to a more brittle behavior. Since toughness is calculated by integrating the area under the stress–strain curve, a drastic increase in the elongation of M1 and M5 results in an increase in toughness; however, M10 decreases. Therefore, cross-linking moderation can improve the mechanical properties of EVOH.
Conclusion
In this study, cross-linked EVOH was prepared through MgCl2 introduction, and MgCl2 was used only as a catalyst. The properties of EVOH were improved via the cross-linking reaction. An abnormally increased torque was revealed in the torque behavior, which could explain the presence of a cross-linked structure, as shown by the gel experiment analysis. The cross-linking reaction of EVOH was based on the formation of –C=C–. With MgCl2 addition, the hydrogen bond of EVOH could be destroyed, while a new hydrogen bond was formed between Cl in MgCl2 and hydroxyl groups in EVOH. Such formation contributed to dehydration accompanied with –C=C– formation, which promoted the formation of the cross-linked structure. The mechanism of a cross-linking reaction was confirmed through 13C NMR and FTIR. The cross-linked structure destroyed the ordered arrangement of EVOH molecules, thereby causing the gradual disappearance of crystals from DSC curves. Thermal analysis showed that the cross-linked structure enhanced the intermolecular force. Therefore, Tg significantly increased when the MgCl2 content increased. Tensile tests indicated that tensile strength improved when the MgCl2 content increased. Therefore, this test proposed an effective and practical way to form the cross-linked structure to improve the tensile strength and thermostability of EVOH and provided a broad application area.
Supplemental Material
sj-pdf-1-jtc-10.1177_0892705720913313 – Supplemental Material for Effects of cross-linking by MgCl2 as an initiator on the properties of EVOH
Supplemental Material, sj-pdf-1-jtc-10.1177_0892705720913313 for Effects of cross-linking by MgCl2 as an initiator on the properties of EVOH by Bin Wang, Chong Lu, Jing Hu and Weixin Lu in Journal of Thermoplastic Composite Materials
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
