Abstract
Different concentrations of fillers such as manganese dioxide (MnO2) and magnetite (Fe3O4) were incorporated into acrylonitrile butadiene rubber (NBR)-interlinked composites. The prepared composite systems were irradiated by electrons at a constant dose of 50 kGy to induce radiation cross-linking under atmospheric conditions. The effect of different contents of fillers and temperature variations on direct current (DC) electrical conductivity,
Introduction
The incorporation of fillers into rubbery polymers imparts many interesting and useful properties to the particle-filled composite materials. 1 –3 Fillers are introduced into rubbery polymers for many varied reasons, more generally to help in tailoring the physical and/or chemical properties of the rubber for various applications where flexibility is an important parameter. 4,5 The ability of fillers to interact physically and/or chemically with rubber compounds, under suitable conditions, is also an important aspect of reinforcement, primarily contributed by the cross-linking mechanism. 6 While the physical and chemical nature of the filler will determine its effectiveness in a functional role, the extent to which this occurs depends on many factors including the amount of filler present and possible interactive effects between the filler and rubbery polymer or between the filler particles themselves.
Electrical properties of such composites are of particular importance, not only from the application point of view but also from the fundamental point of view, as these composites are essentially very good dielectrics. 7,8 Therefore, study of these materials, particularly in an alternating current (AC) field, sheds light on the behavior of charge carriers’ mobility and the mechanism of conduction. A filled rubbery polymer differs substantially from a free one in a wide range of properties. The presence of filler affects both the electrical and mechanical properties. A survey of literature reveals that the conductivity studies on manganese oxide (MnO2)- and magnetite (Fe3O4)-incorporated rubber composites are rather scarcely or seldom reported.
Various rubbers are being widely used for preparation of such conductive composites, for example, natural rubber, styrene–butadiene rubber, acrylonitrile butadiene rubber (NBR), silicone conductive rubber, butyl rubber (IIR), ethylene propylene rubber, and ethylene propylene diene monomer. 9 –11 In the present study, we used NBR due to its good low temperature flexibility, heat and aging resistance, swelling resistance, and especially remarkable oil resistance because of the polar nitrile groups. MnO2 and Fe3O4 were prepared and used as conductive fillers because metals are unstable to oxidation. In addition, the relationship between NBR composites loaded with different concentrations of fillers and direct current (DC)/AC electrical conductivity, dielectric properties as well as mechanical properties were investigated.
Experimental
Materials
NBR with 34% acrylonitrile content was manufactured by EniChem Company Inc. (Italy, Europrene N3345). The antioxidant used is 1,2-dihydro-2,2,4-trimethyl quinoline (TMQ) and was obtained from Birla Tyres Limited (Kolkata, West Bengal, India). The activators namely zinc oxide (ZnO), stearic acid, and dioctyl phthalate used in the study are of commercial grade and purchased from Shandong Kexing Chemical Co. (China). The acrylate polyfunctional monomer, trimethylol propane trimethacrylate (TMPTMA), was obtained from Aldrich Chemical Company Inc. (Germany). Fe3O4, Fe = 88%, and MnO2, Mn = 92%, were supplied by El Nasr Chemicals Company (Egypt).
Mixing and compounding
The rubber composites were prepared on a two-roll opened laboratory mixer under identical conditions of mixing time, temperature, and gap between the rolls. First, NBR was masticated for 2 min followed by the addition of ingredients, antioxidant (TMQ), stearic acid, and ZnO, then the curing agent TMPTMA, and finally the fillers Fe3O4 and MnO2 were added. The nip gap, mill roll speed ratio, and the number of passes were kept constant for all mixtures. Compounds were finally sheeted again in the rolling direction into slabs of about 1 mm thickness, and the sheets were pressed in clean molds of an electric press. The molds were brought to 160°C and held at this temperature for 5 min at a pressure of 160 kg/cm2. The different formulations of mixes and the sample code are tabulated in Table 1.
Formulation of the mixes.

NBR: acrylonitrile butadiene rubber; TMQ: 1,2-dihydro-2,2,4-trimethyl quinoline; ZnO: zinc oxide; TMPTMA: trimethylol propane trimethacrylate; Fe3O4: magnetite; MnO2: manganese oxide.
Irradiation of samples
The samples were irradiated at a constant dose of 50 kGy using an electron beam accelerator in the presence of air at the National Center for Radiation Research and Technology, Cairo, Egypt. The irradiation was done at a beam current of 5 mA, an accelerator energy of 1.5 MeV, and a conveyor speed of 3.2 m/min.
Electrical measurements
Electrical conductivity measurements were carried out in a cell with brass electrodes in the temperature range of 293–383 K, using an electric heater in an isolated chamber with a thermocouple placed very close to the sample. For the DC conductivity measurements, a programmable electrometer (model 617; Keithley Instruments, USA) was used for measuring the resistance of the samples at different temperatures within the range. The frequency-dependent measurements of impedance (
Mechanical measurements
Five individual dumbbell-shaped specimens were cut out from the sheets using a steel die of standard width (4 mm). The minimum thickness of the test specimens was determined using a gauge graduated to 1/100th of a millimeter. A benchmark of 1.5 cm was made on the working part of each test specimen. Tensile strength (TS) tests was carried out according to ASTM D412-66T, 1967 standard at a crosshead speed of 500 mm/min on a rubber tensile testing machine (Hounsfield, England) with modulus at 100% elongation estimated from stress–strain curve and expressed in mega pascal. Hardness was carried out using a durometer type A (Model 306L), from Pacific Transducer Corp. (Los Angeles, California, USA), according to ASTM d 2240, 2000 standard.
Results and discussion
DC conductivity
The DC electrical conductivity of NBR composites with different filler concentrations of MnO2 and Fe3O4 and irradiated at 50 kGy is computed at different temperatures. The electrical conductivity is calculated according to the following equation:
where 
where
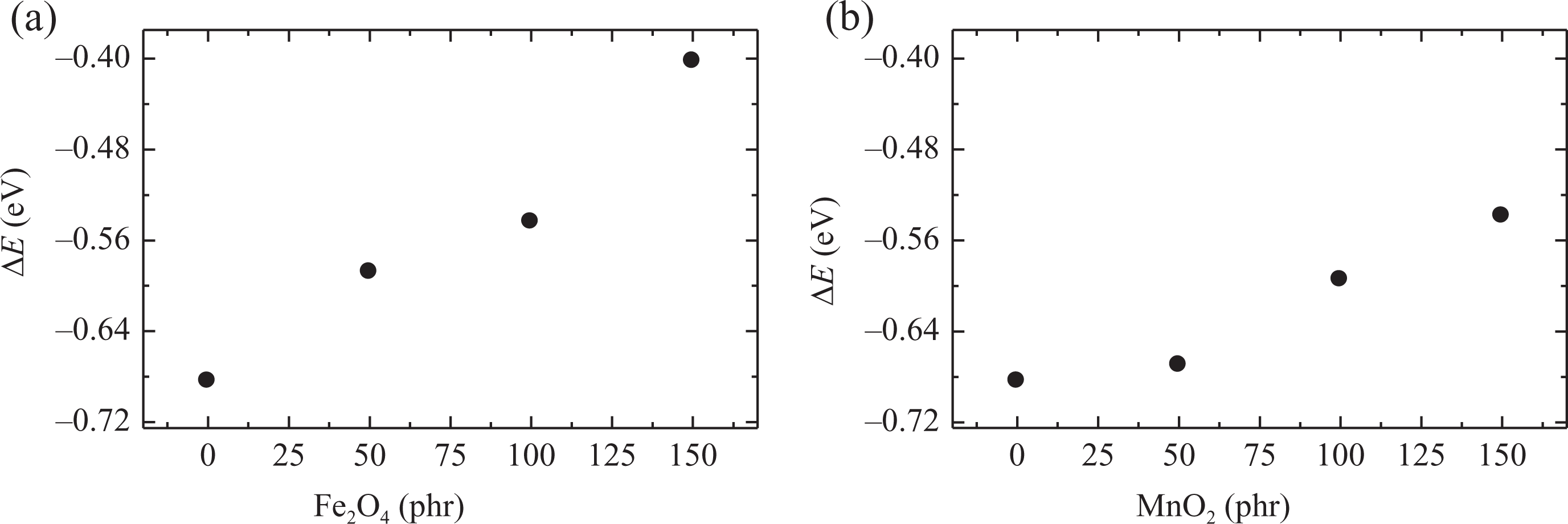
Activation energy of NBR composites loaded with different concentrations of fillers (a) Fe3O4 and (b) MnO2 and irradiated at 50 kGy. NBR: acrylonitrile butadiene rubber; Fe3O4: magnetite; MnO2: manganese oxide.
Dielectric studies
The frequency dependence of dielectric constant (
where
The values of the real part of the dielectric constant (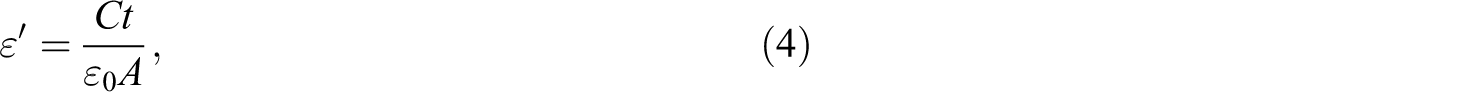
where 
The dissipation factor (tan
Figures 2 and 3 represent the experimentally obtained comparison plots of the temperature dependence of
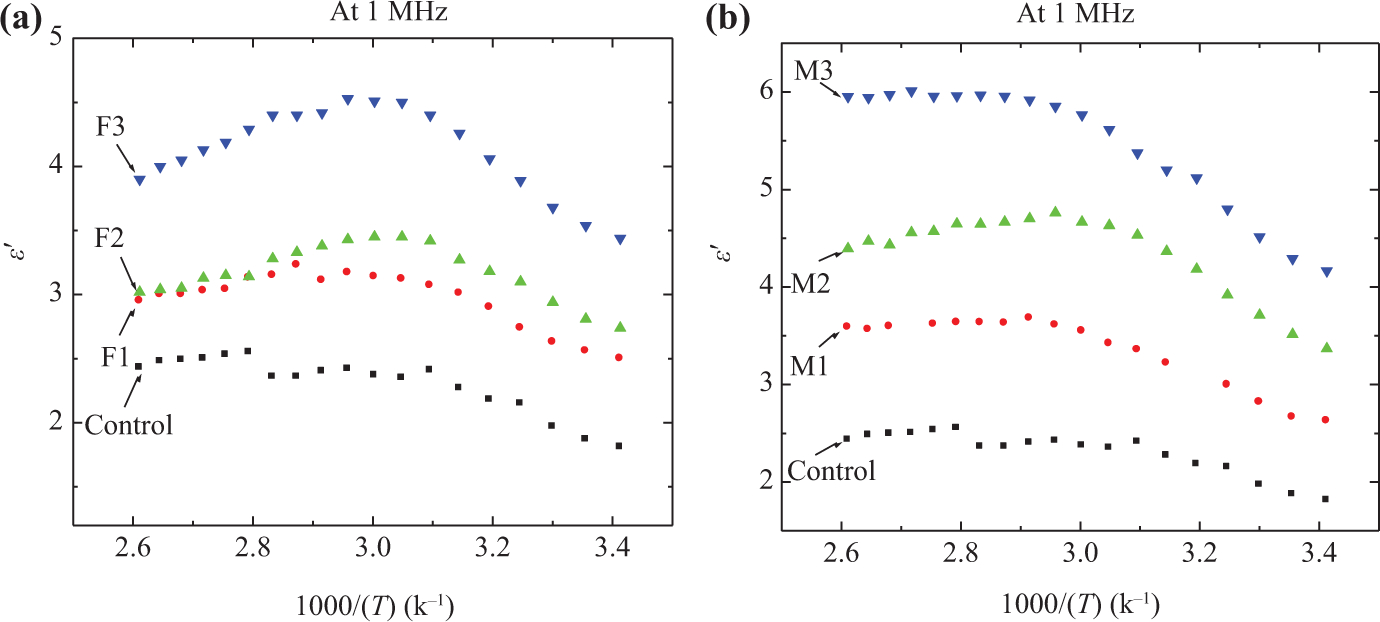
Temperature dependence of the dielectric constant,
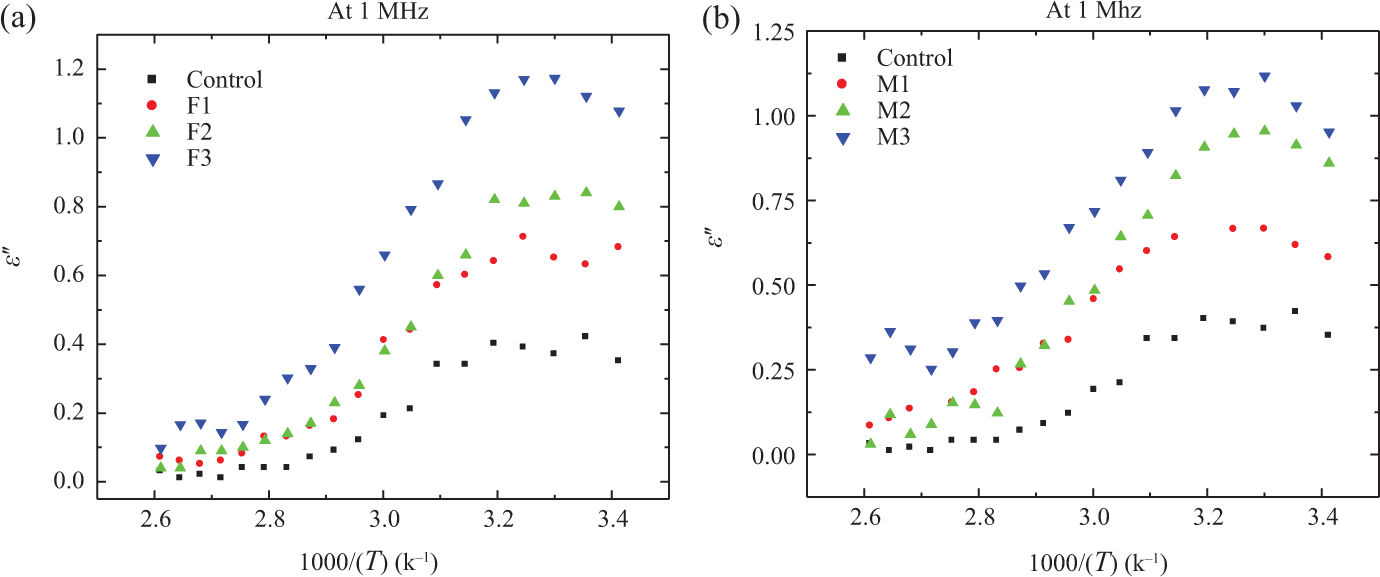
Temperature dependence of
The observed behavior of increasing
Figures 4 and 5 show the frequency dependence of
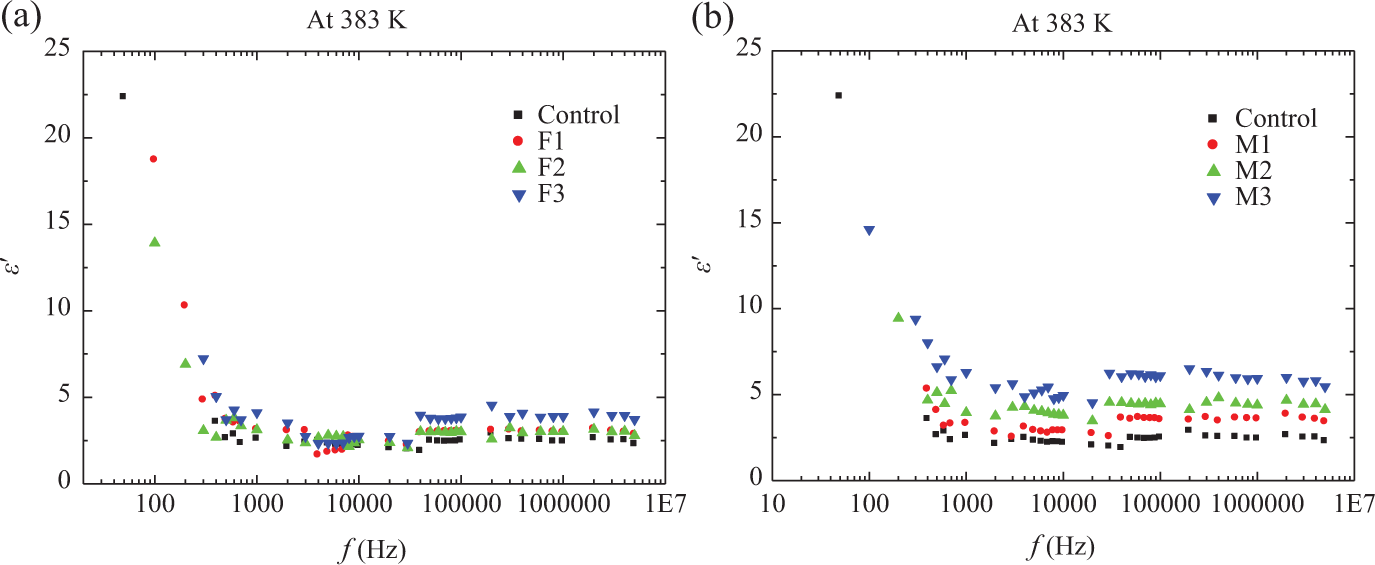
Frequency dependence of
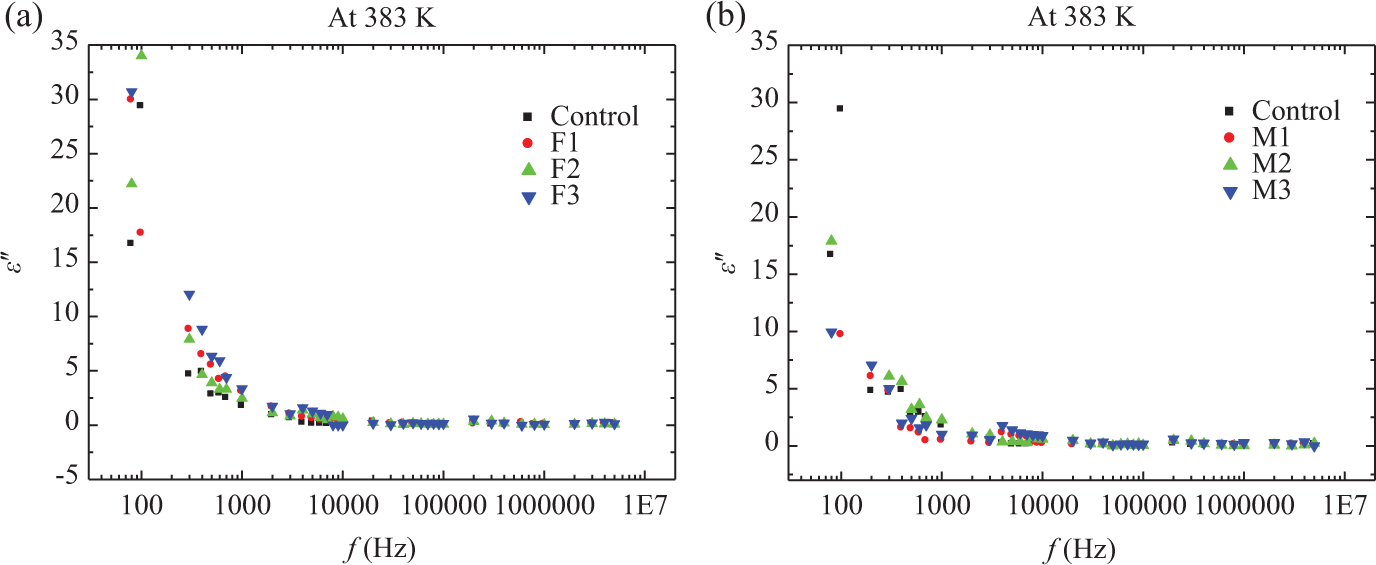
Frequency dependence of
AC conductivity
The frequency dependence of the AC electrical conductivity, 
The AC electrical conductivity, for NBR composites loaded with different concentrations of fillers Fe3O4 and MnO2 and irradiated at 50 kGy, is computed for different frequencies and also at different temperatures. Figure 6 represents the frequency dependence of AC electrical conductivity for all composites under investigation at a temperature of 398 K, as an example. As can be seen from this figure, the electrical conductivity generally shows a sharp increase with increasing frequency. In addition, as it is expected, conductivity also increases with increasing filler Fe3O4 and MnO2 concentrations in NBR. This indicates that a continuous conductive network forms in the rubber permitting a higher percentage of electrons to flow through the samples. Similar behaviors are observed in the literature.
19,23
The increase in the electrical conductivity leads to an increase in the eddy current, which in turn increases the energy loss tan
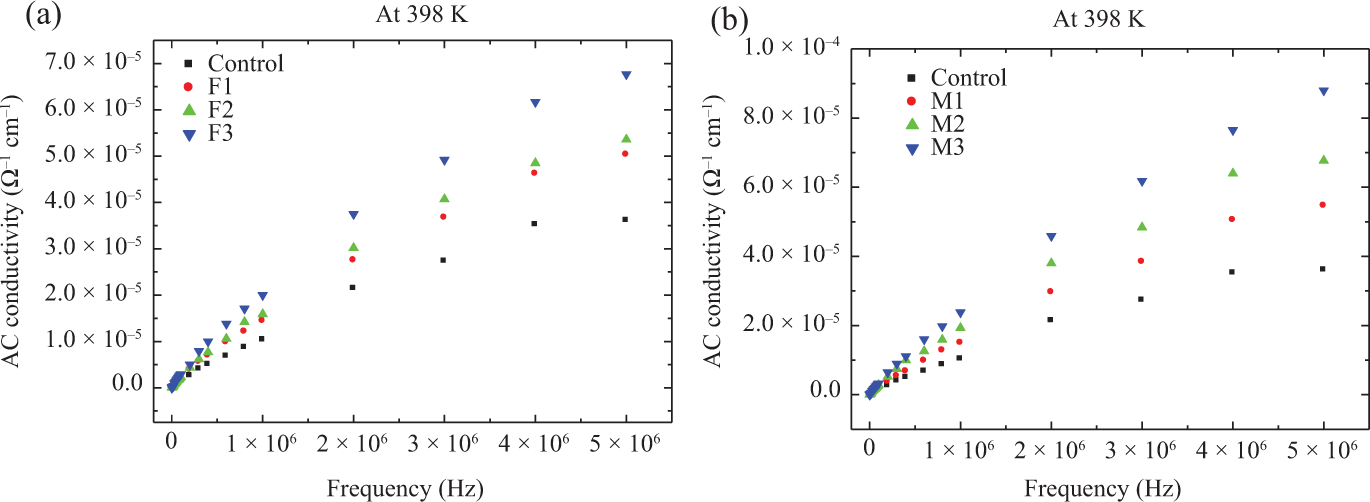
Frequency dependence of the AC conductivity measured at 398 K of NBR composites loaded with different filler concentrations of (a) MnO2 and (b) Fe3O4 and irradiated at 50 kGy. NBR: acrylonitrile butadiene rubber; Fe3O4: magnetite; MnO2: manganese oxide.
The variations of conductivity with temperature for different compositions are shown in Figure 7. This figure shows that the conductivity increases up to a temperature of about 330 K. Further increase of temperature reduces the conductivity of ferrite samples, while the conductivity values of the MnO2 samples tend to be almost constant after this temperature. The influence of temperature on conductivity can be explained by considering the mobility of charge carriers responsible for hopping. As temperature increases, the mobility of hopping ions also increases thereby increasing the conductivity. 7 The electrons that are involved in hopping are responsible for electronic polarization in these ferrites and manganeses. The decrease in conductivity at higher temperature can be due to the thermal expansion of NBR. At higher temperatures, the NBR density is reduced by thermal expansion and this reduces the conductivity. 7
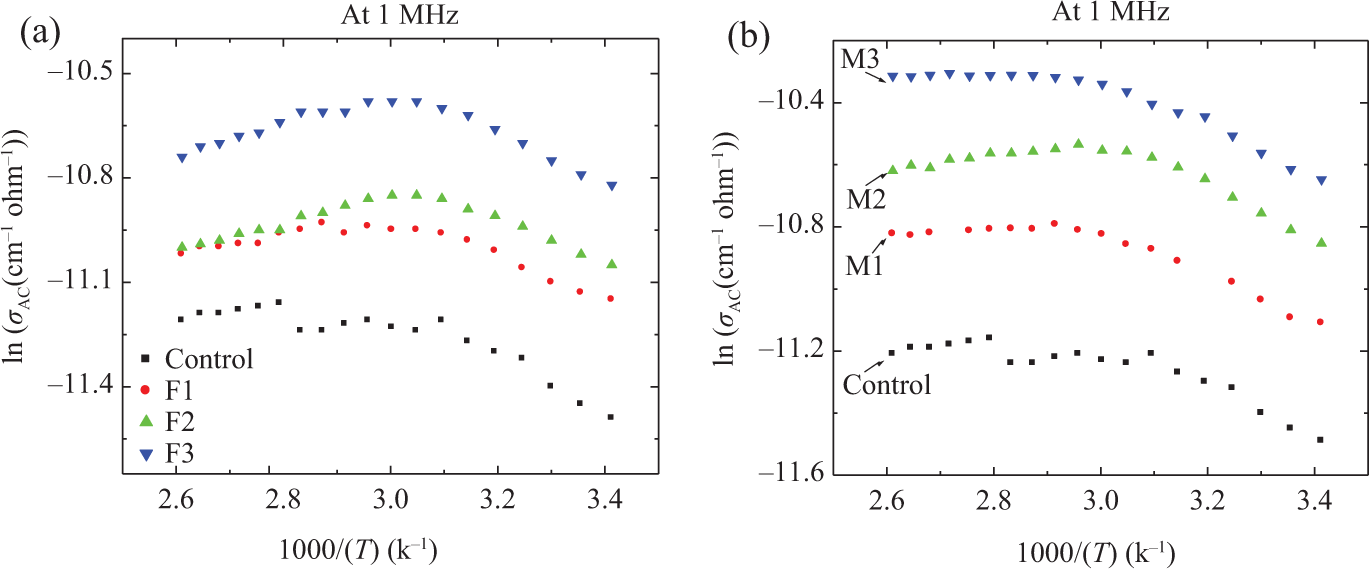
Variation of ln
Mechanical properties
Figure 8 shows the mechanical properties of NBR composites loaded with different concentrations of ferrite and irradiated at 50 kGy. From this figure it can be seen that the values of TS as well as hardness increase by increasing the filler content. On the other hand, the values of elongation at break decrease by increasing the content of the ferrite filler. These data indicate that the ferrite filler is considered as reinforced filler for NBR, and some type of adhesion occurs between the ferrite filler and rubber matrix.
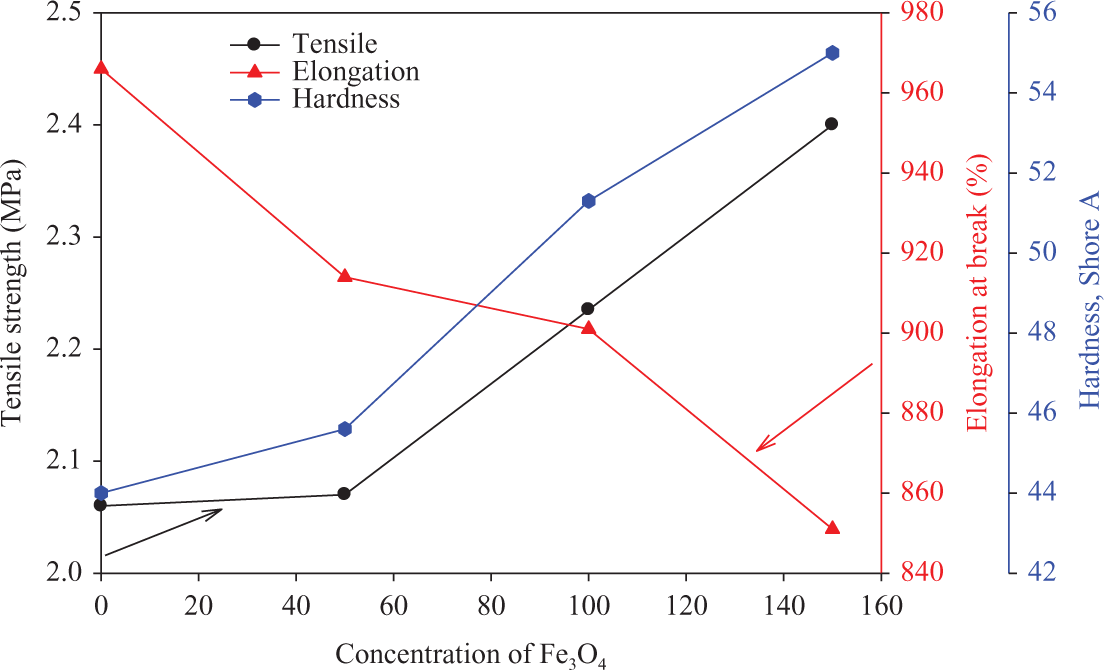
TS, elongation at break, and hardness of NBR composites loaded with different concentrations of ferrite and irradiated at 50 kGy. TS: tensile strength; NBR: acrylonitrile butadiene rubber.
Figure 9 illustrates the effect of concentration of MnO2 on the mechanical properties like TS, elongation at break, and hardness of NBR composites irradiated at 50 kGy. The data obtained from this figure showed that the values of TS increases with increasing the filler content up to 100 phr, and then it tends to decrease at higher content namely 150 phr. In addition, the values of hardness increase sharply up to 150 phr of filler. Meanwhile, the values of elongation at break decrease by increasing the filler content. The data obtained for TS may be explained also by the fact that MnO2 acts as reinforcing filler in NBR matrix up to 100 phr; also, some type of chemical adhesion occurs between the filler and rubber matrix during milling and pressing under heating and also by electron irradiation, resulting in increased cross-link density as shown in Figure 10. On the other hand, the decrease in the values of TS at a higher loading content of 150 phr may be due to agglomeration in the filler, which acts as nodes and separate macromolecules from attaching with each other, leading to a decrease in the values of TS at higher concentration of MnO2 filler. Generally, the hardness increases by increasing the cross-link density, while the elongation decreases by increasing the latter. 25
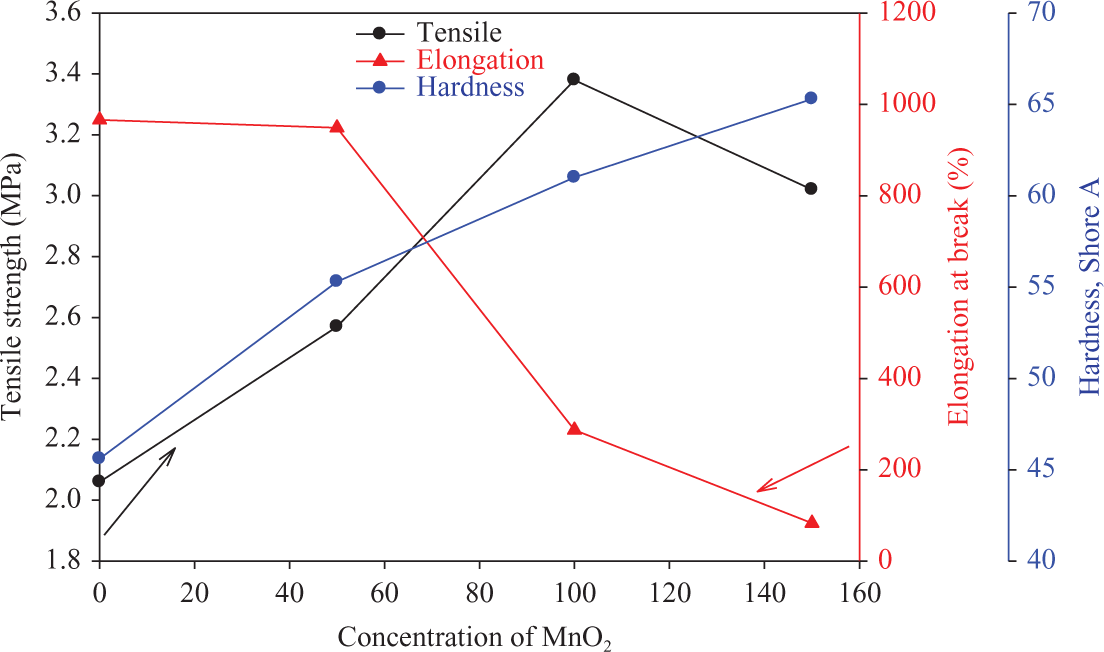
TS, elongation at break, and hardness of NBR composites loaded with different concentrations of MnO2 and irradiated at 50 kGy. TS: tensile strength; MnO2: manganese dioxide.
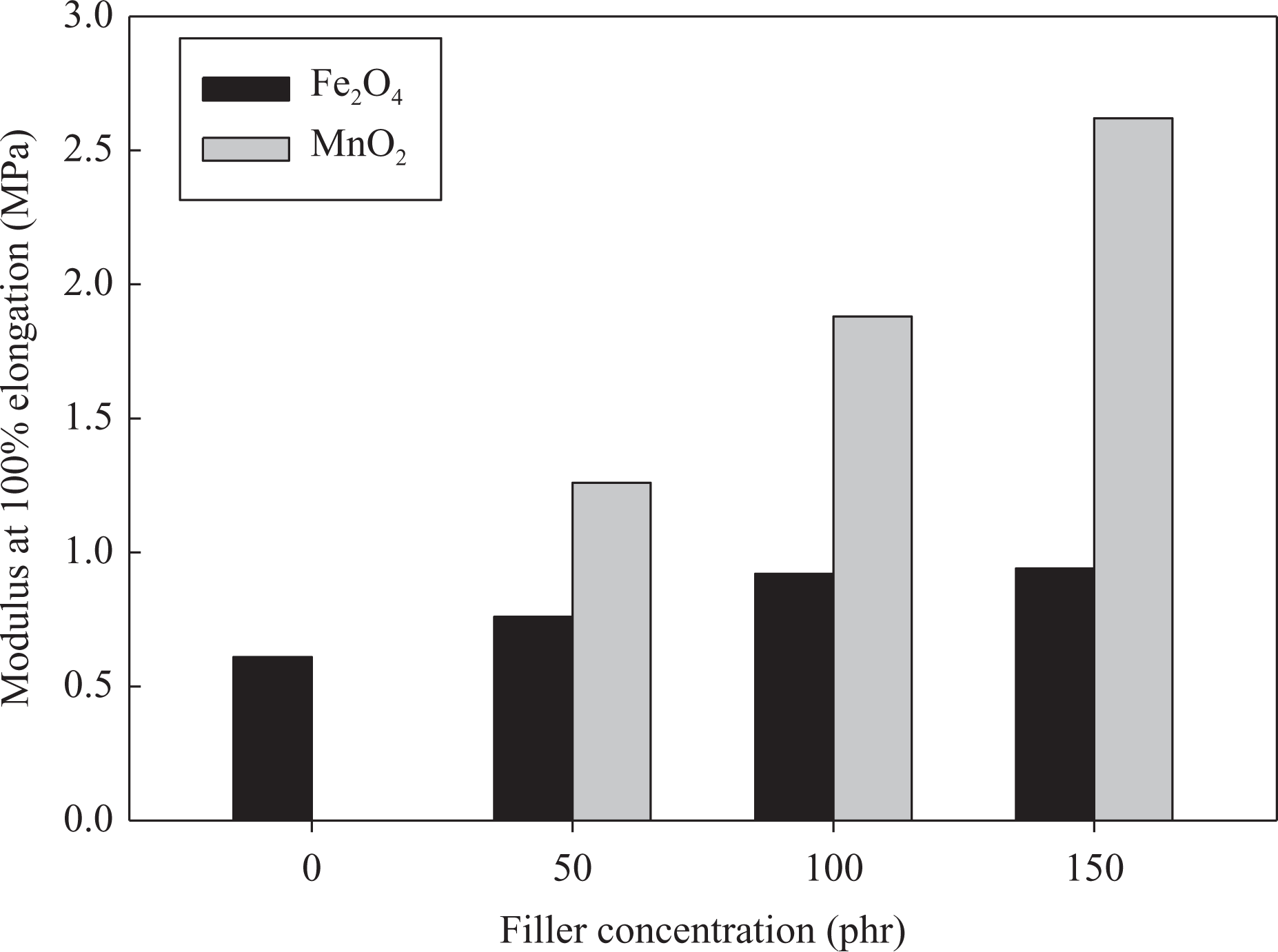
Modulus at 100% elongation of NBR composites loaded with different concentrations of fillers MnO2 and Fe3O4 and irradiated at 50 kGy. NBR: acrylonitrile butadiene rubber; Fe3O4: magnetite; MnO2: manganese oxide.
Conclusion
The DC conductivity results indicate that the increase in filler (MnO2 and Fe3O4) concentrations yielded a significant increase in the conductivity and lowering of the activation energy of the NBR matrix. The AC electrical conductivity is directly proportional to the frequency. It shows an increase in conductivity with increasing frequency for both MnO2 and Fe3O4 fillers. In addition, it has been observed that both dielectric constant and dielectric loss decrease exponentially with increasing frequency for all composites under investigation. This is ascribed to the decrease in electronic contribution and increase in dipolar contribution to the total polarizability. The DC/AC electrical conductivity proved that both types of fillers are dispersed in the NBR matrix uniformly to form an interconnected conducting network. The mechanical properties of composites show that the TS as well as hardness increase by increasing filler contents of MnO2 and Fe3O4. This increase is attributed to the improvement of interfacial bonding between the filler and NBR matrix.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
