Abstract
Siloxane polymer containing vinyl groups was blended with poly(vinyl chloride) (PVC) to convey plasticization and stability against decomposition, that is likely to happen during thermal processing or following exposure to the ultraviolet (UV) light. The immiscibility between silicone rubber and PVC was proved from images acquired using scanning electron microscopy (SEM), which revealed worsening of the situation at higher loading (10 wt%) of silicone polymer. The thermal degradation profiles indicated that the inclusion of silicone polymer did not affect the thermal resistance significantly between 200°C and 350°C. However, it protected the matter from intensive decomposition at higher temperatures and increased the residual weight. Fourier transform infrared spectra were collected for the blends after exposure to UV radiation for 168 h, from which the absence of appreciable dehydrochlorination or change in the nature of the material was ensured. The hampering of dehydrochlorination is expected to have proceeded by instant attack of any appearing radicals on PVC backbone onto vinyl groups of the silicone polymer and buildup of a network structure.
Introduction
The extensive request on poly(vinyl chloride) (PVC) is attributed particularly to its supply in a cost-effective price alongside other excellent properties such as high chemical stability and resistance to flammability, which meet the criteria for use in many fields.1–5
PVC is primarily stiff and brittle material, which affects its long-run use. This raises the concern to formulate PVC with some additives, among which is a plasticizer, to facilitate its processing and prolong its endurance.6,7 As well, PVC is suffering from sensitivity to heat and light, which causes worsening of its properties and eventual failure.8–10 Specifically, PVC degrades at a temperature below its processing temperature. 11 In the meantime, it undergoes rapid decomposition under ultraviolet (UV) radiation especially in the presence of oxygen and moisture. 12 Different structural defects are in charge of the low stability against heat and light of wavelength between 250 nm and 350 nm. This includes allylic chlorine, tertiary hydrogen and chlorine atoms, double bonds at the terminals, head-to-head arrangements, oxygen-containing groups in addition to peroxide residues. 9
The degradation of PVC starts by radical creation (chlorine atom removal). In the presence of impurities or residues, they act as receptors for these radicals and dehydrochlorination propagates, which lead to degraded chains of shorter length and polyene formation. In the absence of oxygen, the degradation can develop to cross-linking and gel formation, which is associated with failure and properties alteration.10,13–16 In the presence of oxygen, it combines with the radicals to form peroxides that can also break the double bonds formed on the backbone leading to brittleness and crack formation. 17
A number of strategies followed the blending approach with other polymers, such as epoxidized natural rubber, 18 polymethyl methacrylate, 19 and unfunctionalized silicone polymer,15,20 for stabilization of PVC and facilitating its processing.
The ongoing study is a further effort to a previous investigation which found that blending PVC and some silicone polymers can enhance some properties of PVC,14–16,20 considering that the silicone polymer with its siloxane linkages is characterized by high flexibility and resistance to heat and aging related to UV, ozone, and radiation. 21
For this purpose, we used a type of vinyl-functionalized siloxane polymer that renders it a focal center for blocking of any probable radicals formed during the degradation of PVC by immediate attack on the double bonds leading to network structure rather than polyene formation due to entropy decrease. Besides, even in case of unavoidable polyene formation, they can interact favorably with the double bonds of the silicone rubber (SiR) by Diels–Alder condensation 3 as this exchanges two n bonds for two σ bonds, thus prevent the degradation process from propagation.
Our preliminary investigation of the possible changes of the material strength, after short exposure to UV radiation (96 h), ensured confusing changes due to complication of the stabilization process and extended action of this novel route. 16 This inspired us to go further and detect the changes after exposure to UV radiation prolonged to 168 h. This will lead to deeper insight on the mechanism and extent by which the silicon polymer can protect PVC against the effect of heat mistreatment or exposure to harmful radiation.
Experimental
Materials
Commercial grade PVC with k-value of 67, dioctyl phthalate (DOP; plasticizer), zinc stearate (ZS) (processing aid), and dibutyltin maleate (DM) (heat stabilizer) were supplied from the local market. SiR with low vinyl density (ELASTOSIL R 401/10) was purchased from Wacker silicones, Germany.
Methods
Preparation of PVC-SiR formulations
PVC alongside different proportions of SiR were prepared by initial introduction of the SiR into the chamber of a melt-blender, then the temperature was elevated thoroughly to 180°C, whereas the rotor was set at its lowest speed. Once the SiR reached the melting state, the designated amount of PVC was added to the processing chamber combined with the specified parts of DM, ZS, and DOP (Table 1). The speed of the rotor was increased appreciably and the blending process was continued for additional 6 min, then the samples were withdrawn from the chamber and kept to reach the room temperature. Compression moulding of the samples was undertaken using a press preheated to 180°C at 25 MPa pressure to give sheets with thickness 0.7 mm. For concise depiction of the samples composition, PVC1 is a basic formulation of plasticized PVC, whereas PVC2 comprises 5 phr of silicone polymer. PVC3 is prepared very similar to PVC2, but a thermal stabilizer was additionally included. Doubling the SiR to 10 phr without (PVC4) or with a thermal stabilizer (PVC5) was also undertaken. The thermal stabilizer is added in some formulations for comparison to check if degradation can be avoided during processing in its absence.
Different compositions of PVC-silicone polymer.
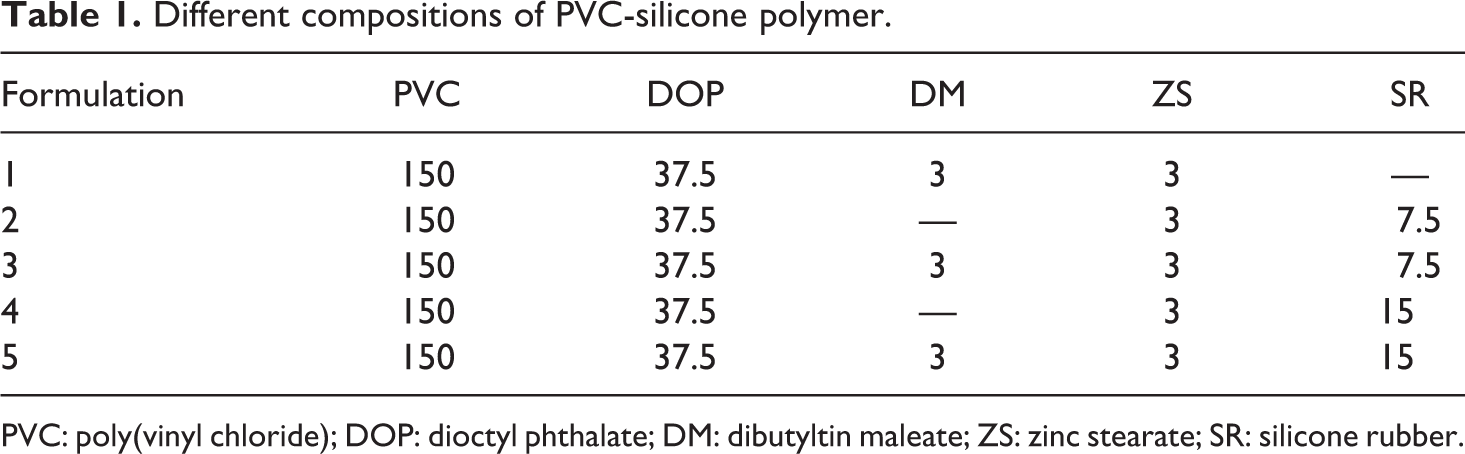
PVC: poly(vinyl chloride); DOP: dioctyl phthalate; DM: dibutyltin maleate; ZS: zinc stearate; SR: silicone rubber.
Characterizations
Imaging with scanning electron microscopy (SEM-Quanta FEG-250, Holland) was achieved, after gold plating using sputter coater (serial peripheral interface [SPI] module), at 20 kV operating voltage on PVC3, PVC4, and PVC5 in melt form as well as sheet sample of PVC5, after exposure to UV radiation for 96 h to check for any apparent or microscopic changes. The heat resistance of the samples was evaluated using Shimadzu-60 thermal gravimetric analyzer, Japan. A platinum pan was used to hold 3–5 mg of each sample, while the thermal run started from room temperature to 700°C, under nitrogen atmosphere, at a heating rate of 10°C min−1.
Fourier transform infrared (FTIR) spectra of sheet samples were collected in attenuated total reflectance mode, in the wavelength range 500–4000 cm−1, by undertaking a scanning speed of 2 mm s−1 with a total of 32 scans for each sample, at a resolution of 8 cm−1, using Brucker Optik spectrometer (Vertex70, GmbH), Germany.
The stress–strain behavior was studied, on five dumbell-shaped specimens cut from each sample, before and after exposure to UV radiation, using Zwick/Roell Z010 tensile testing machine, Germany. The machine was operated at a crosshead speed of 0.5 mm min−1. Average of five runs was considered for each sample.
Results and discussion
SEM analysis
Imaging of the samples in their blend forms was carried out using SEM before hot press to maintain the morphological features of the surface unaltered. It can be made sure that phase separation can develop between the PVC and the SiR (compare Figure 1(a) and (b)). This mismatching became more pronounced when the loading of the silicone polymer was doubled (Figure 1(d)). In both cases, the developed interfacial tension between the phases can complicate reaching a homogeneous distribution of the additives required for processing of PVC, for instance, DM as heat stabilizer (Figure 1(c)).

SEM mircographs of (a) PVC2, (b) PVC3, (c) PVC4, (d) PVC5, and (e) pressed PVC5 sheet after exposure to UV radiation for 96 h.
A preliminary suggestion was introduced previously for the interaction of the elastic siloxane polymer and stiff PVC has been initiated following the emergence of radicals on PVC. 16 The formed macroradicals can then hit the vinyl sites of the siloxane polymer leading to a cross-linked network as simplified in Figure 2. This parallel route is thought to have precedence to oxidative degradation route because more cross-links are added in a system, which will be accompanied by conformational entropy and energy decrease.
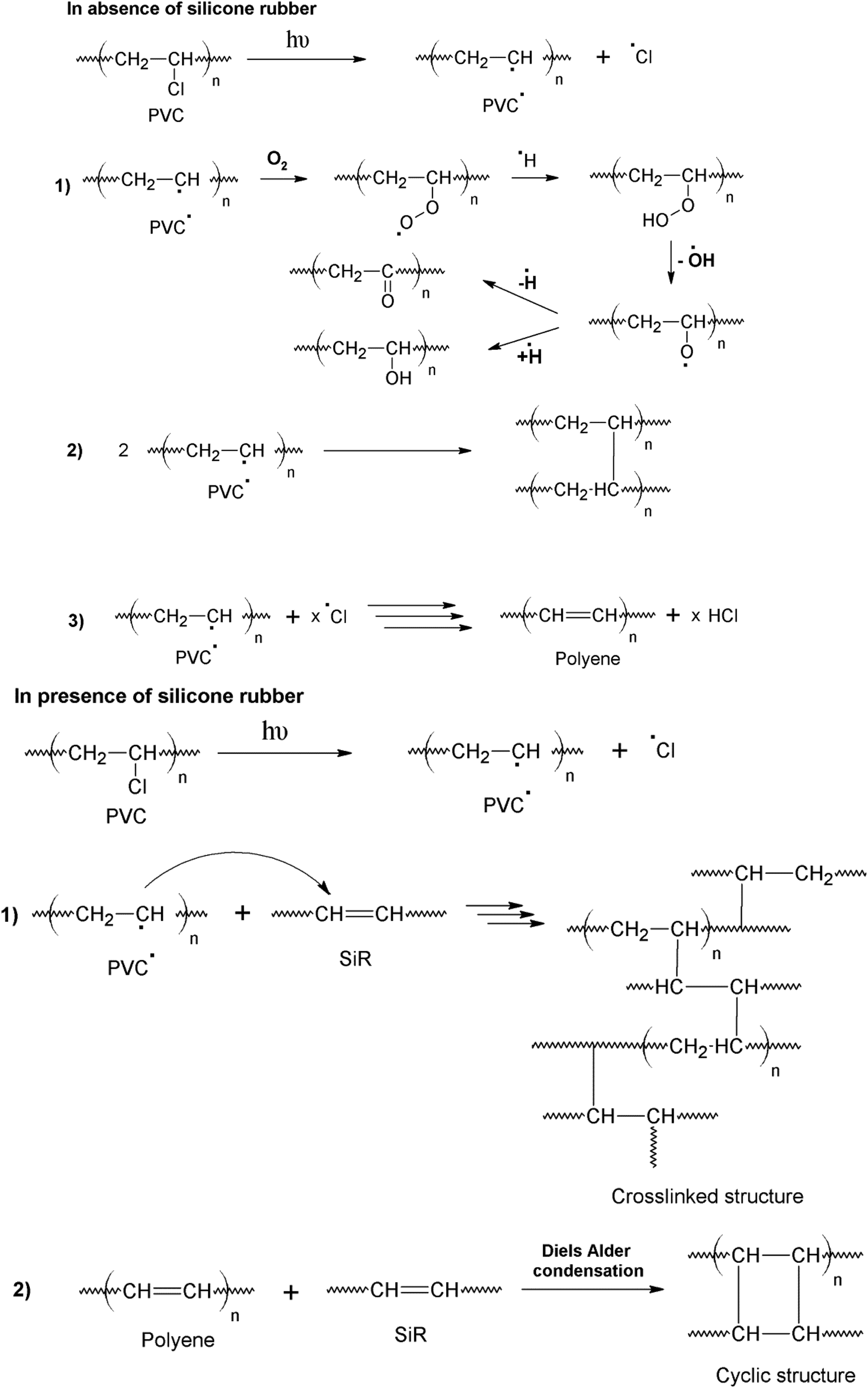
Chemical pathway for the involvement of SiR in network structure with PVC.
McNeill and Basan 15 explained that for PVC compositions with content 50% or more of polydimethylsiloxane, both polymers are stabilized and ensured no detectable change in the nature of degradation products. It was additionally suggested that since both polymers are not miscible, the agent in charge of this effect must be able to diffuse efficiently across the boundaries, for example, hydrogen chloride or chlorine radicals, leading to cross-linking.
Thermal gravimetric analysis
The thermal degradation traces as well as the relevant differential thermal gravimetric profiles of the samples lacking heat stabilizer (DM), PVC2, and PVC4 were then collected under inert atmosphere to investigate if there is a change in the thermal response as compared to PVC1 (Figure 3(a) and (b), Table 2). The degradation profiles were generally almost typical with no serious effect after the silicone polymer insertion. PVC2 exhibited a faster but modest extra loss with respect to PVC4 and PVC1 in the temperature range 200–350°C. The residual weight for PVC2 and PVC4 was 2–3% higher as compared to neat PVC1, which is in partial accordance with Zulfiqar and Ahmed, who claimed enhanced thermal resistance of compositions based on a type of polysilicone and PVC. 14 Otherwise, the residual weight would have been lessened significantly. This supports the incorporation of the silicone polymer did not actually hinder the dehydrochlorination process, but it combats its harmful effect on the properties of PVC by instantaneous buildup of a stable network structure instead of propagation in the degradation process (as will be discussed in the part of mechanical testing).
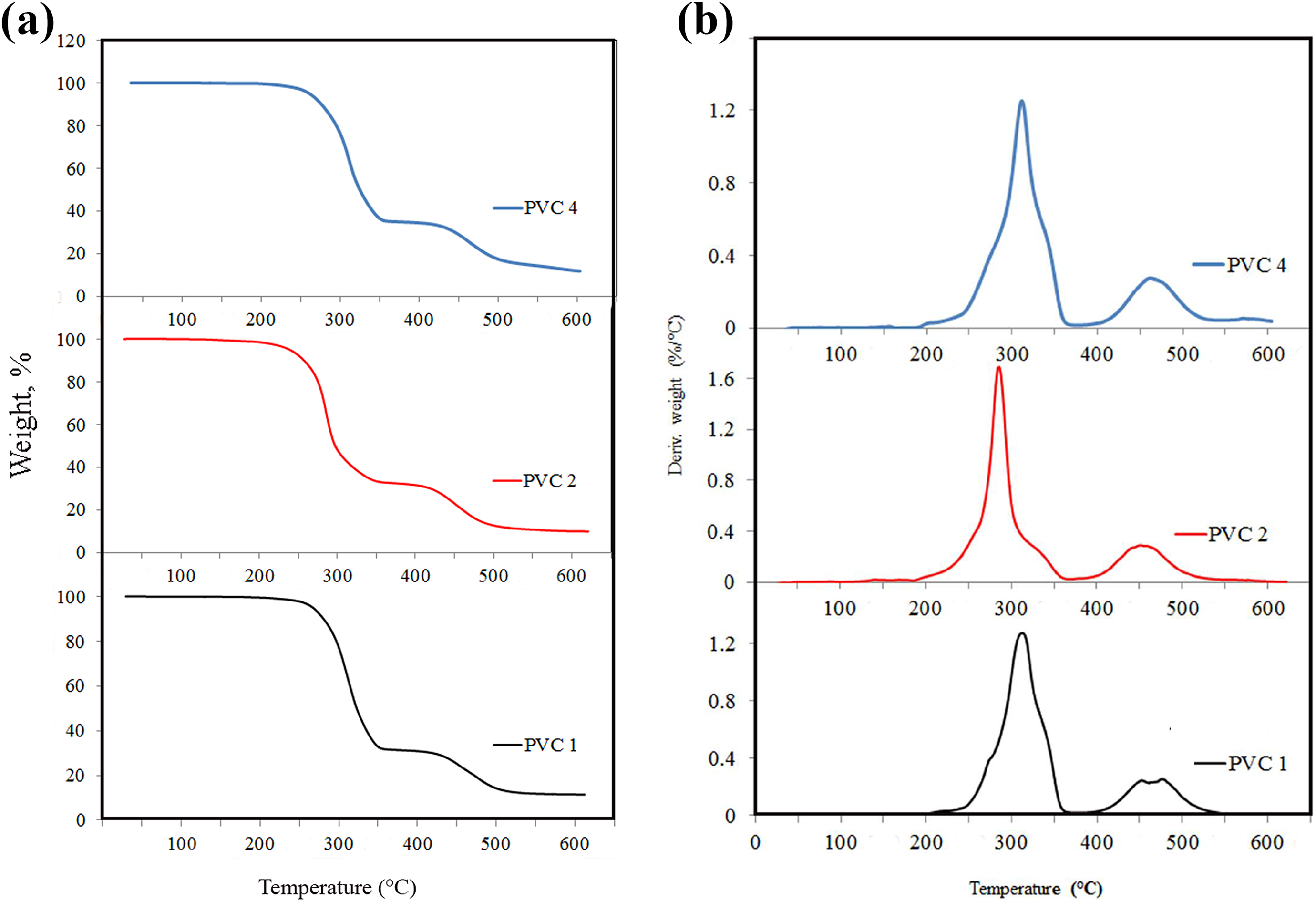
(a) TGA and (b) DTG profile for the PVC-SiR compositions.
Weight loss percentages obtained at different temperatures for the different PVC compositions.
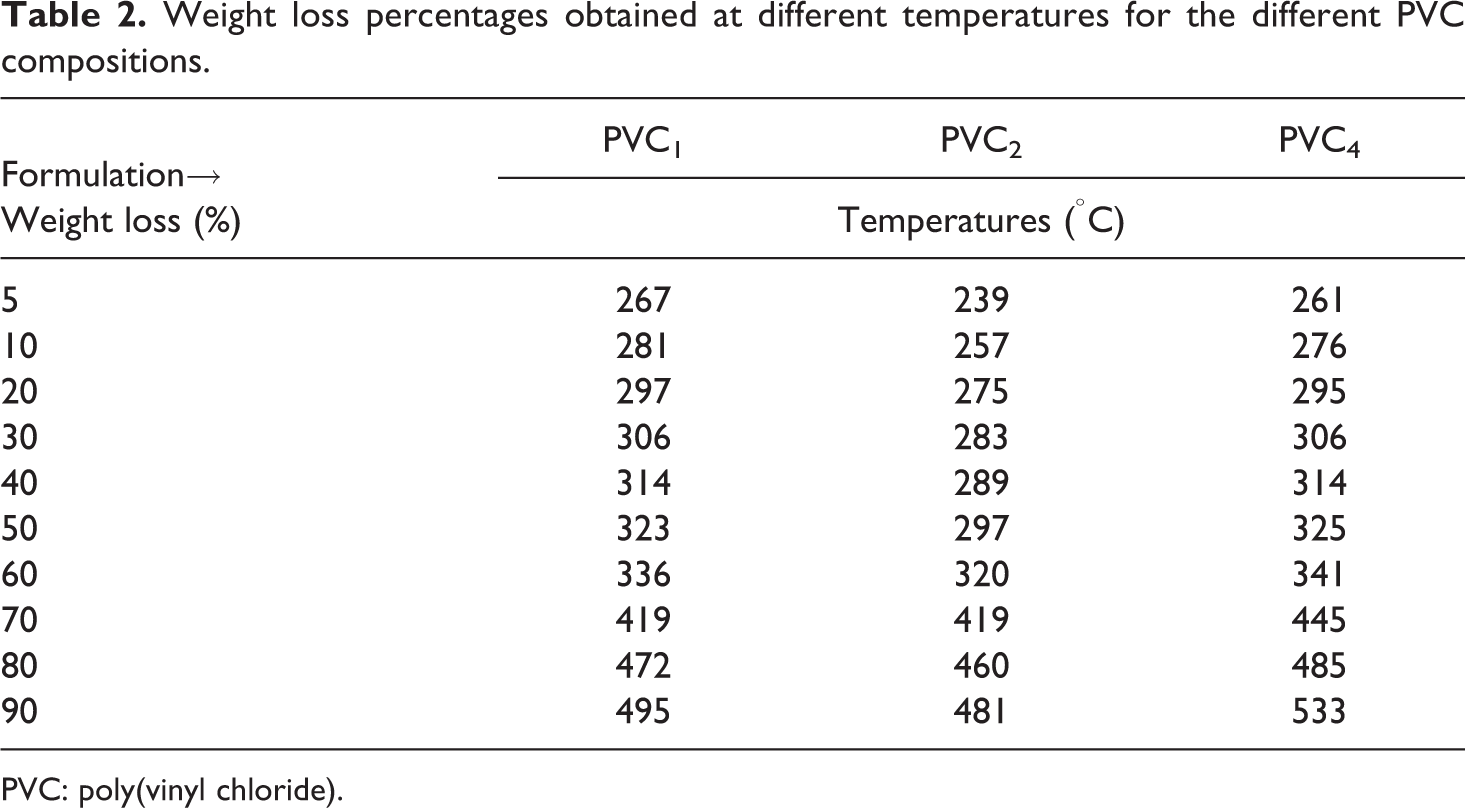
PVC: poly(vinyl chloride).
FTIR analysis
FTIR can also provide helpful information about the possible interaction between PVC macroradicals and the silicone polymer. FTIR spectra of the samples were collected after exposure to UV radiation for 168 h (Figure 4). PVC1 is characterized by C–Cl band at 650 cm−1 and a broadband at 890–1000 cm−1, revealing C–C bonds of the main chain. C–H groups neighboring Cl appeared at 1250 cm−1 while the bending mode of C–H is signified by a peak at 1400 cm−1. It can be observed that the IR spectra collected for PVC2, PVC3, PVC4, and PVC5 are similar to PVC1, considering the overlapping of some bands pertaining to silicone polymers such as CH3 deformation at 1250 cm−1 and its stretching at 2800–2900 cm−1 and Si–O stretching at 1000 cm−1.
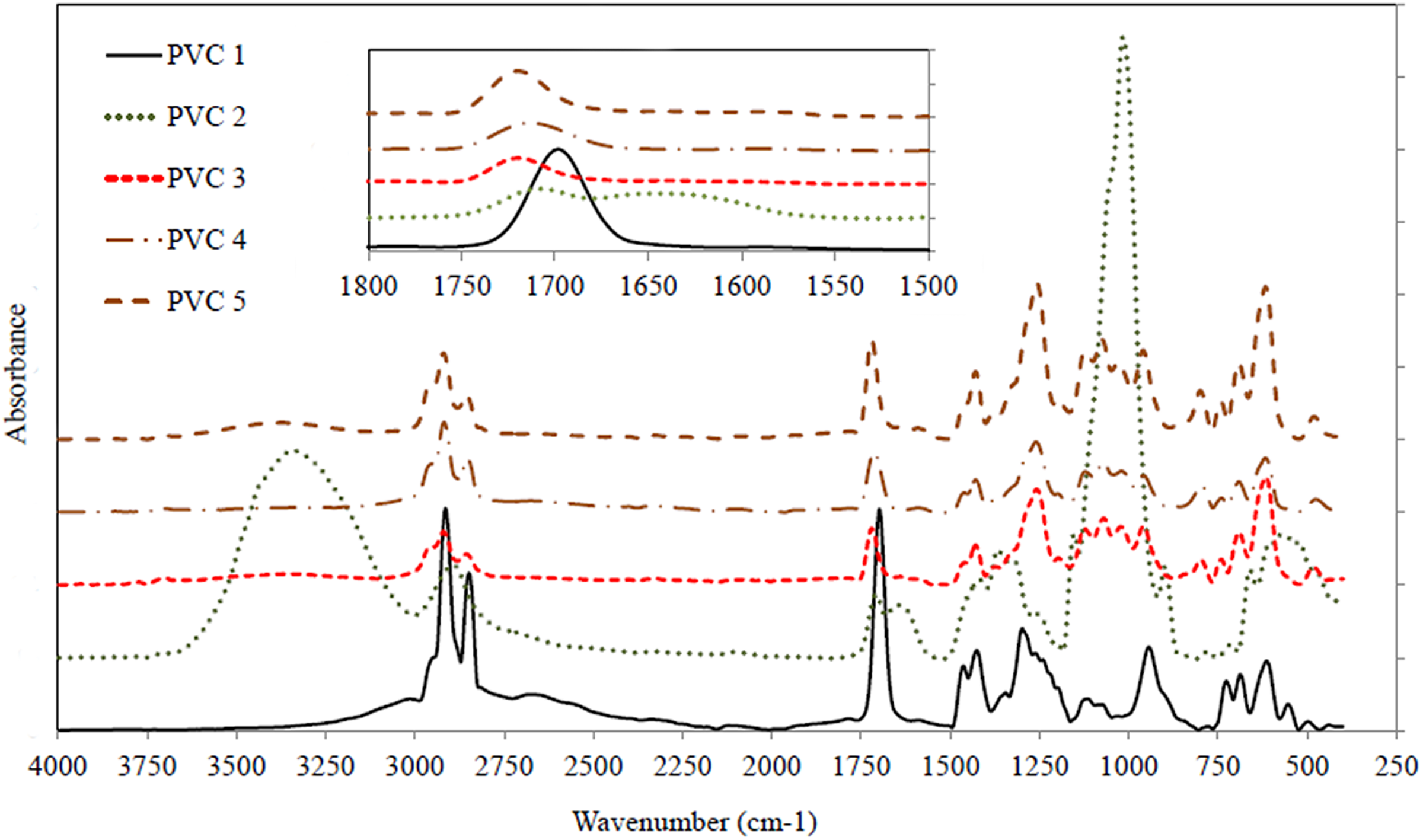
FTIR spectra collected for the samples after exposure to UV radiation.
A clear peak at 1670–1710 cm−1 can be firstly related to C=C resulting from the dehydrochlorination process and polyene formation (the inset of Figure 4). Nevertheless, C=C is generally visible in the range 1580–1610 cm−1. Thus, it is very likely that C=C was also succeeded by oxidative degradation and carbonyl formation, 10 which caused merging into one peak at around 1670–1710 cm−1 (the inset of Figure 4). Participation of the double bonds in Diels–Alder condensation is also suggested. The low intensity of this peak for the stabilized samples as compared to the sharp intensity in case of PVC1 presents an evidence of these scenarios. This is further supported by reshaping and broadening of the bands in the 2800–2900 cm−1 region, indicating the conversion of the double bonds either by radical attack or by more likely through Diels–Alder condensation. This highlights the promising stabilization route of PVC using vinyl-functionalized siloxane polymer as compared to other stabilizers committed for this purpose.22–26 Interestingly, a broad peak of limited/reasonable intensity in some cases (PVC2 and PVC5) can be detected in the region 3000–3500 cm−1, which is most likely attributed to tiny species formed haphazardly by limited thermooxidative degradation.
Stress–strain behavior
Systematic examination of the mechanical behavior of the samples after short exposure to UV radiation indicated chaotic alteration because structural changes are still developing. 16 Accordingly, the stress–strain behavior of PVC1, PVC2, and PVC4 was recorded after intensive exposure to the UV light for 168 h to further elucidate the extent of interaction.
The occasion of degradation on plasticized PVC is associated with loss of its flexibility (compare Figure 5(a) and (b)). Incorporation of silicone polymer in PVC2 and PVC4 matches well with the classification of flexible materials (Figure 5(a), Table 3). However, they turned to stiffer materials after 168 h of exposure to UV radiation, indicating the involvement of the silicone polymer in a cross-linking reaction with emerging PVC macroradicals (Figure 5(b) and Table 3). Additional evidence of this assumption is the associated improvement in compatibility without cracks formation, even after only 96 h of exposure to UV radiation (PVC5, Figure 1(e)).
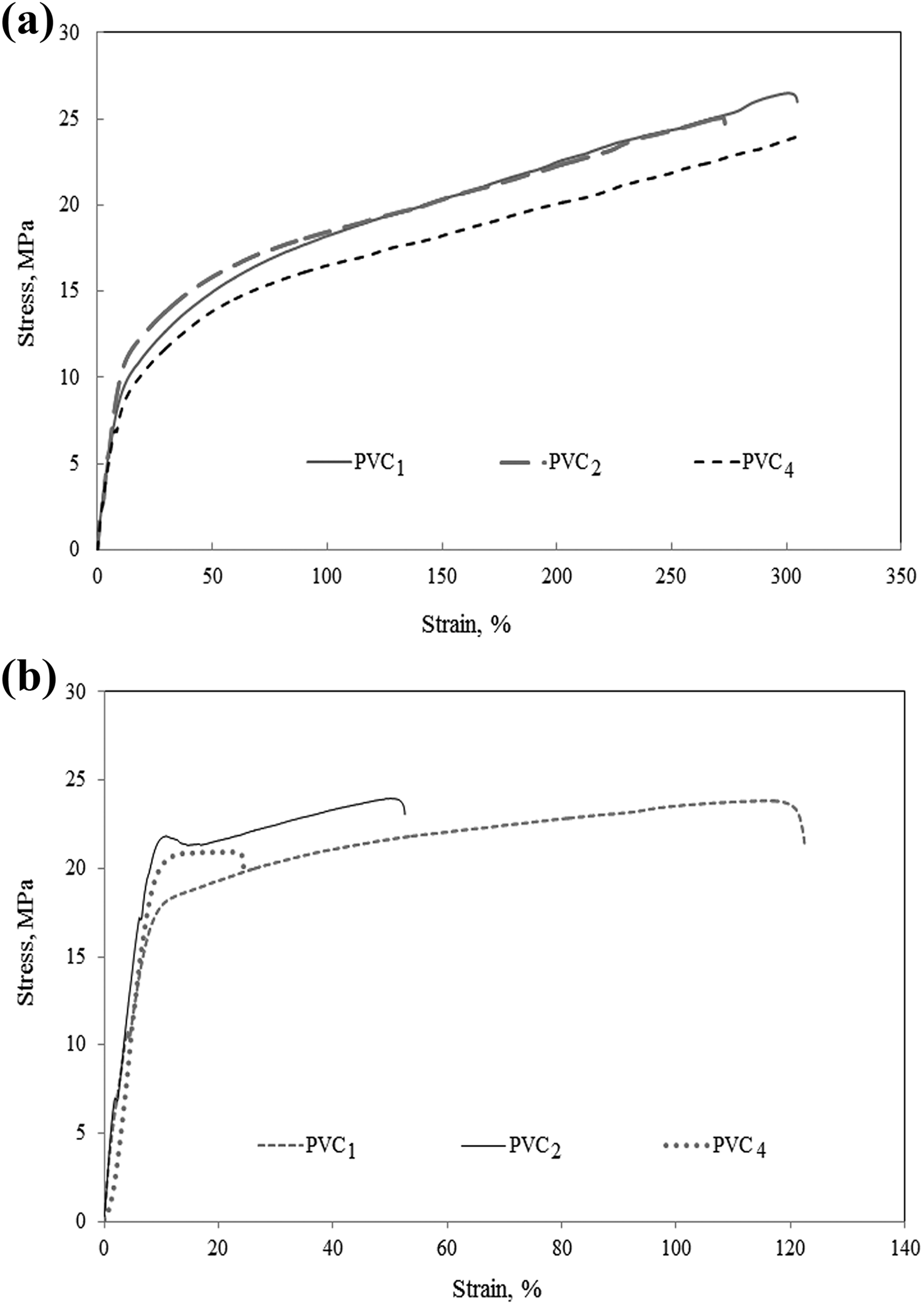
Stress–strain curves of PVC1, PVC2, and PVC4 (a) before and (b) after 168 h exposure to UV radiation.
Stress values (MPa) attained at different elongation levels (%) for the different compositions before and after exposure to UV radiation for 168 h, prior to break.
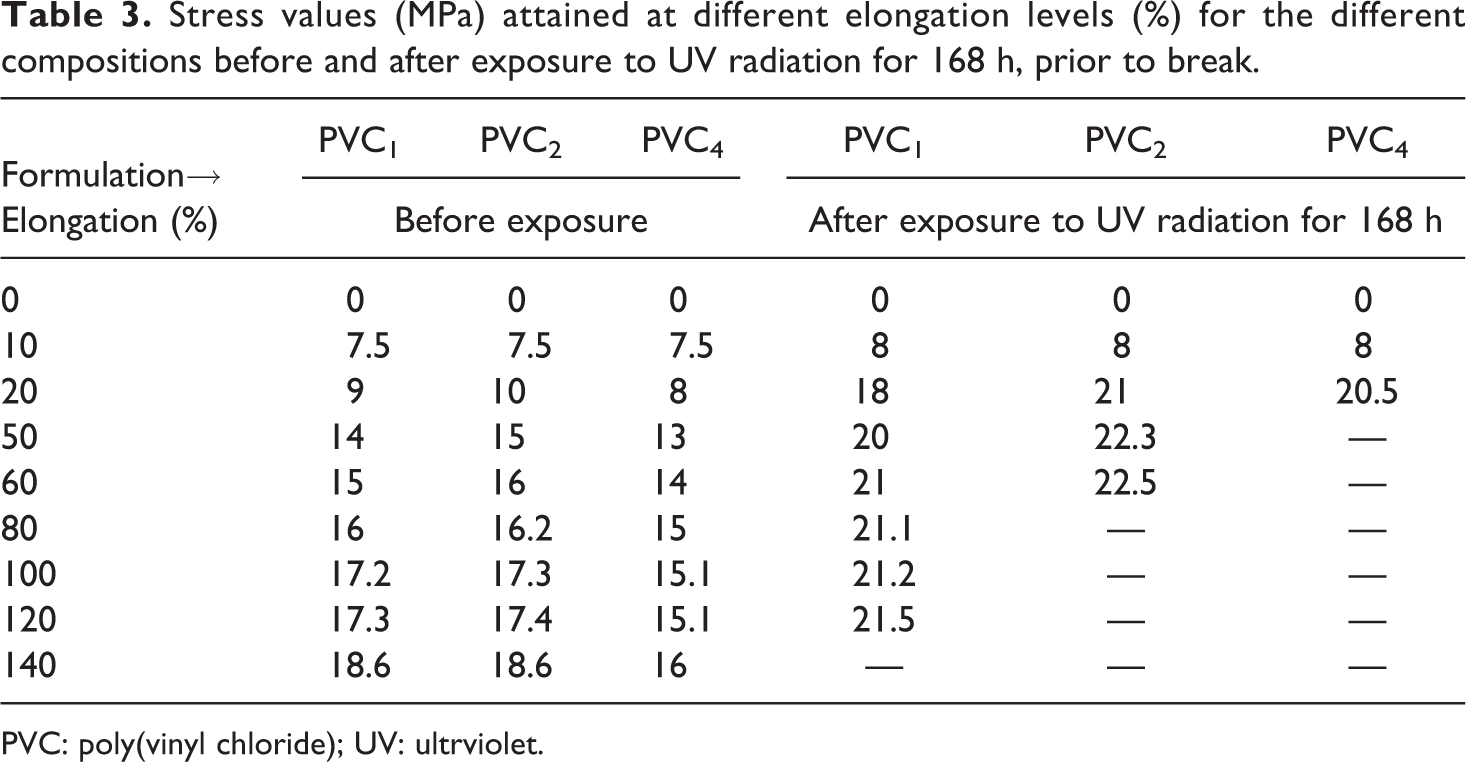
PVC: poly(vinyl chloride); UV: ultrviolet.
Conclusion
A SiR can be blended with PVC to provide enhancement of processing and stabilization against decomposition. Functionalization of SiR with polymerizable vinyl groups can facilitate the interaction between the immiscible phases via chemical coupling in case of emergence of radicals on PVC following some influences. Influences such as thermal treatment or exposure to the UV light are expected to induce active radicals on PVC backbone. In a consequence, these radicals can link to the vinyl groups of silicone polymer leading to network structure incorporating both phases instead of propagation of dehydrochlorination. The increased residual weight after induced thermal decomposition together with the enhanced miscibility between the silicone polymer and the PVC signify the effective incorporation of silicone polymer in network formation with PVC. This incorporation provoked improving the resistance to mechanical deterioration following exposure to UV radiation or heat.
