Abstract
There are several chemicals for using as ultraviolet (UV) absorbers. Considering that titanium phosphate (TiP) not yet studied for this purpose, we intended to investigate its effect on poly(ethylene terephthalate) (PET). TiP was synthesized and intercalated with two types of long-chain amine, octadecylamine (OETiP) and ether-amine-O-(2-aminopropyl)-O′-(2-methoxyethyl)polypropylene glycol) (JETiP) being the intercalation assisted by ethylamine. Nanocomposites of PET/TiP (filled with 5 wt%) (PET-TiP, PET-OETiP, and PET-JETiP) were prepared by melting extrusion. Thermogravimetric analysis, differential scanning calorimetry, wide-angle X-ray diffraction (WAXD), Fourier transform infrared spectroscopy, hydrogen low-field nuclear magnetic resonance (1H LFNMR), and UV-visible (Vis) absorption were used for characterizations. OETiP and JETiP induced the decrease of PET glass transition temperature (Tg) and heat crystallization temperature (Tch) while PET crystallinity degree (Xc), cold crystallization temperature (Tcc), and crystallization rate increased. WAXD suggested the intercalation of PET chains inside of fillers lamellae. According to 1H LFNMR, all fillers increased the PET molecular mobility. Diffuse reflectance revealed increase of the absorption at UV light region. The sample might be thought as a UV absorber.
Introduction
Since the advent of nanotechnology about 20 years ago, the areas of science and scientific research beyond business have been enormously transformed. Nanotechnology is related to the use of compounds and/or structures with dimensions at the level of atoms and molecules, commonly in the order of 1–100 nm. With this size range, nanoparticles have a higher surface area/volume ratio improving interaction with host polymers and then imparting properties.1,2 Nanoparticles mainly phyllosilicates, oxides, and ceramics have been experienced in polymeric nanocomposites.3–5 Due to their extraordinary properties, phosphates of tetravalent metal—M(RPO4)2n·H2O (M = Ti, Zr, Sn; R = hydrogen or organic group)—have gained importance as alternative to the phyllosilicates in polymeric nanocomposites. Extensively studied zirconium and titanium phosphates (TiPs) possess peculiar lamellar crystalline arrangements allowing intercalation with short- and long-chain amines.6–13 Uryupina and coauthors investigated the influence of TiP on stability of epoxy resin suspension. They mentioned the increase of suspension stability owing to the formation of the adsorption shell on nanoparticles. 14 Zirconium phosphate (ZrP) was investigated as flame retardancy agent in composites based on polyamide-6 (PA-6), poly(ethylene terephthalate) (PET), polypropylene (PP), and ethylene vinyl acetate copolymer. 15 Distinguishable morphologies and reduction of flammability were attained according to the polymer matrix. The action of octadecylamine-modified lamellar-ZrP and screw speed on the properties of PP nanocomposites were evaluated. Best dispersion was achieved at higher screw speed. Thermal stability increased while crystallinity degree was influenced by screw speed. 16 PET composites containing 2–5 wt% of ZrP and organic–inorganic hybrid layered zirconium phenylphosphonate (ZrPP) were studied. In both cases, the addition of the fillers increased PET crystallization rate and the modulus. 17 PET has remarkable importance in the universe of the thermoplastics due to its low cost, transparency, processability, mechanical properties, recyclability, and high performance either commodity or engineering polymer. With different optical appearance, production recipes and molecular weight, PET fiber-grade and bottle-grade dominate the global market of textile fiber and carbonated beverage bottles, respectively. More than 60% of the PET produced worldwide is applied in the textile sector.18,19
The textile industry has been always rated as a major economic driver and fashion launcher. In this context, nanotechnology has been applied to improve properties such as water repellence, crease resistance, bactericidal, ultraviolet (UV) radiation barrier, flame retardancy, dyeing, among others.20,21 According to Gupta et al., 22 the incorporation of titanium dioxide (TiO2) in cotton fabrics protects the garments from organic stains. TiO2 nanoparticles were anchored onto clothes improving the anti-UV absorption and the material life. Zanrosso and Lansarin immobilize TiO2 on cotton textile by dip-pad-cure method by aid of succinic and citric acid. The authors commented that TiO2 immobilization forms stable and active materials at prolonged UV irradiation periods. 23 Asgharian-Dastenaie et al. 24 investigated the effect of TiO2 nanoparticles and polycarboxylic agent onto cotton fabric samples. They pointed out that self-cleaning property against chemical and natural stains was improved. A method for producing self-cleaning textiles starting from a commercial colloidal nanosuspension (nanosol) of TiO2 was described by Ortelli and collaborators. They revealed that to cover textile surfaces with commercial TiO2 nanosol is feasible and to overcome problems of industrial scalability. 25
Our research group synthesizes and develops structural variation of zirconium and TiP viewing applications in several industrial sectors. As importance and motivation of this research was taken in consideration that there is not report on TiP as UV absorber. As the first approach, nanocomposites of PET filled with synthesized and modified TiP (5 wt%) were investigated. Thermal, crystallographic, molecular mobility, and UV-visible (Vis) absorption characteristics were assessed.
Experiment
Materials
Bottle-grade PET A80W resin (density = 1.39 g cc−1, intrinsic viscosity = 0.80 dl g−1) was supplied by Grupo Mossi & Ghisolfi—M&G (Poços de Caldas, Brazil). Phosphoric acid (H3PO4, 85% w/w), titanium isopropoxide (C12H28O4Ti), ethanol (C2H6O, 99%), ethylamine (C2H5NH2), O-(2-aminopropyl)-O′-(2-methoxyethyl)polypropylene glycol—Jeffamine M600 (C30H63NO10, oxy-ethylene/oxy-propylene ratio OE/PO = 1/9, molar mass = 600 g mol−1, density = 0.98 g mL−1), and octadecylamine (stearylamine, C18H39N 98%) were purchased of Sigma–Aldrich, Brazil. Herein, the abbreviations PET, ethylamine (E), Jeffamine (J), and octadecylamine (O) were used.
Synthesis and amine intercalation of TiP
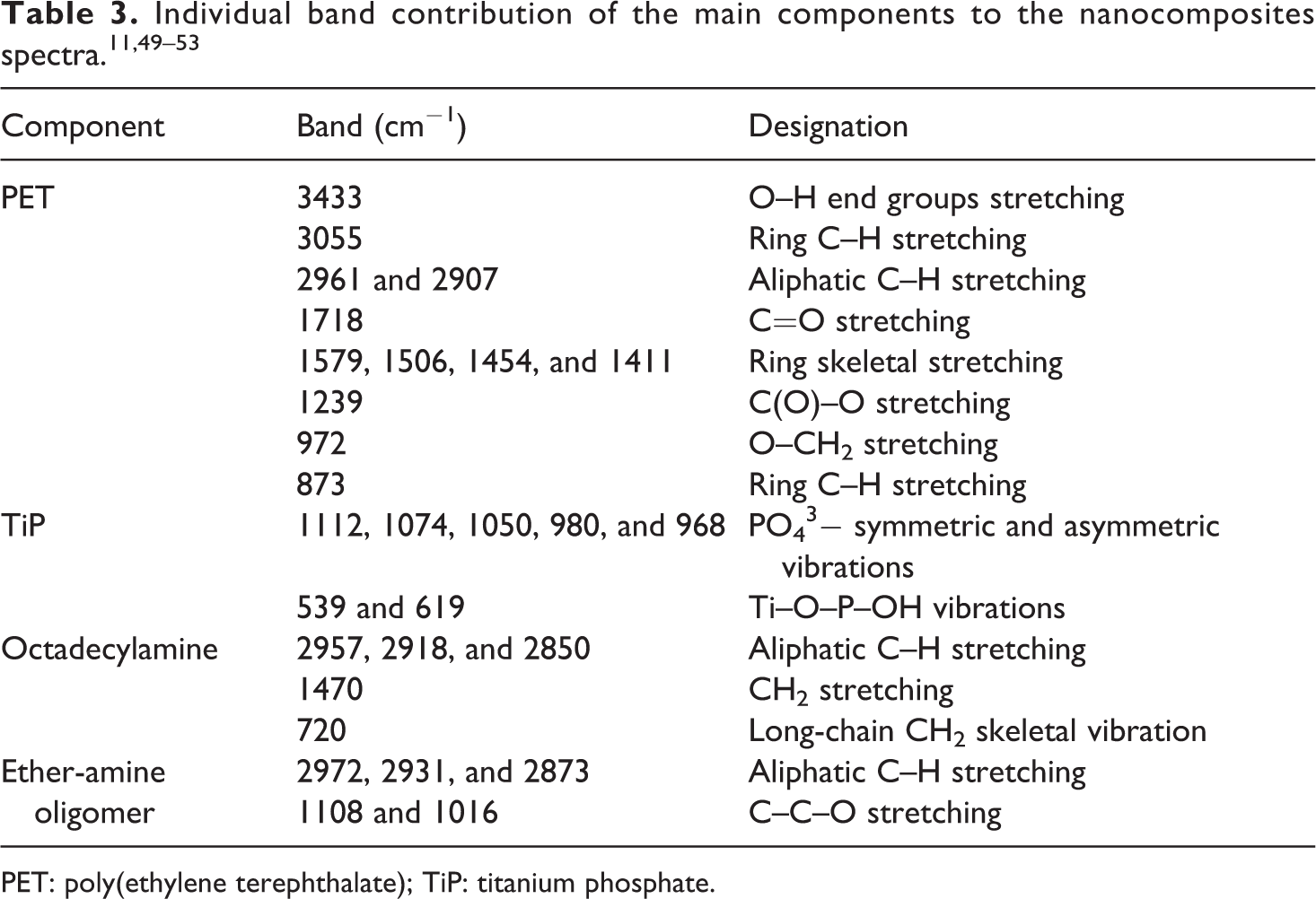
At molar ratio of 1:8 and under stirring, titanium isopropoxide (20 mL) was added dropwise to orthophosphoric acid (35 mL) followed the addition of deionized water (10 mL). The reaction medium was kept under agitation and reflux for 24 h.26–30 The reaction product was poured into dialysis bag which was immersed in a water bath to eliminate residual acid. When the pH reached value 6, the process was stopped. The final precipitate was dried at 90°C until constant weight. The TiP precursor was labeled as TiP.
The intercalation of TiP with octadecylamine and Jeffamine was similar and assisted by ethylamine. Firstly, the dispersion/solution of the precursors were prepared as following: ethanolic dispersion of TiP (1 g in 15 mL absolute ethanol); ethanolic solution of amines (octadecylamine (0.52 g) plus 0.14 mL of ethylamine; Jeffamine (1.19 g) plus 0.14 mL ethylamine, both dissolved in 15 mL of absolute ethanol, maintaining molar ratio long-chain amine:ethylamine:TiP (0.5:0.1:1). After that, amine solution was slowly added upon ethanolic dispersion of TiP, at room temperature, under stirring, for 24 h. The reaction medium was centrifuged at 3000 r min−1, for 5 min. Finally, the reaction product was dried, at 90°C until constant weight. The samples were labeled as OETiP and JETiP, modified with octadecylamine and Jeffamine, respectively. Figure 1 summarizes the TiP synthesis and intercalation.
Scheme of TiP synthesis and intercalation.
Nanocomposites preparation
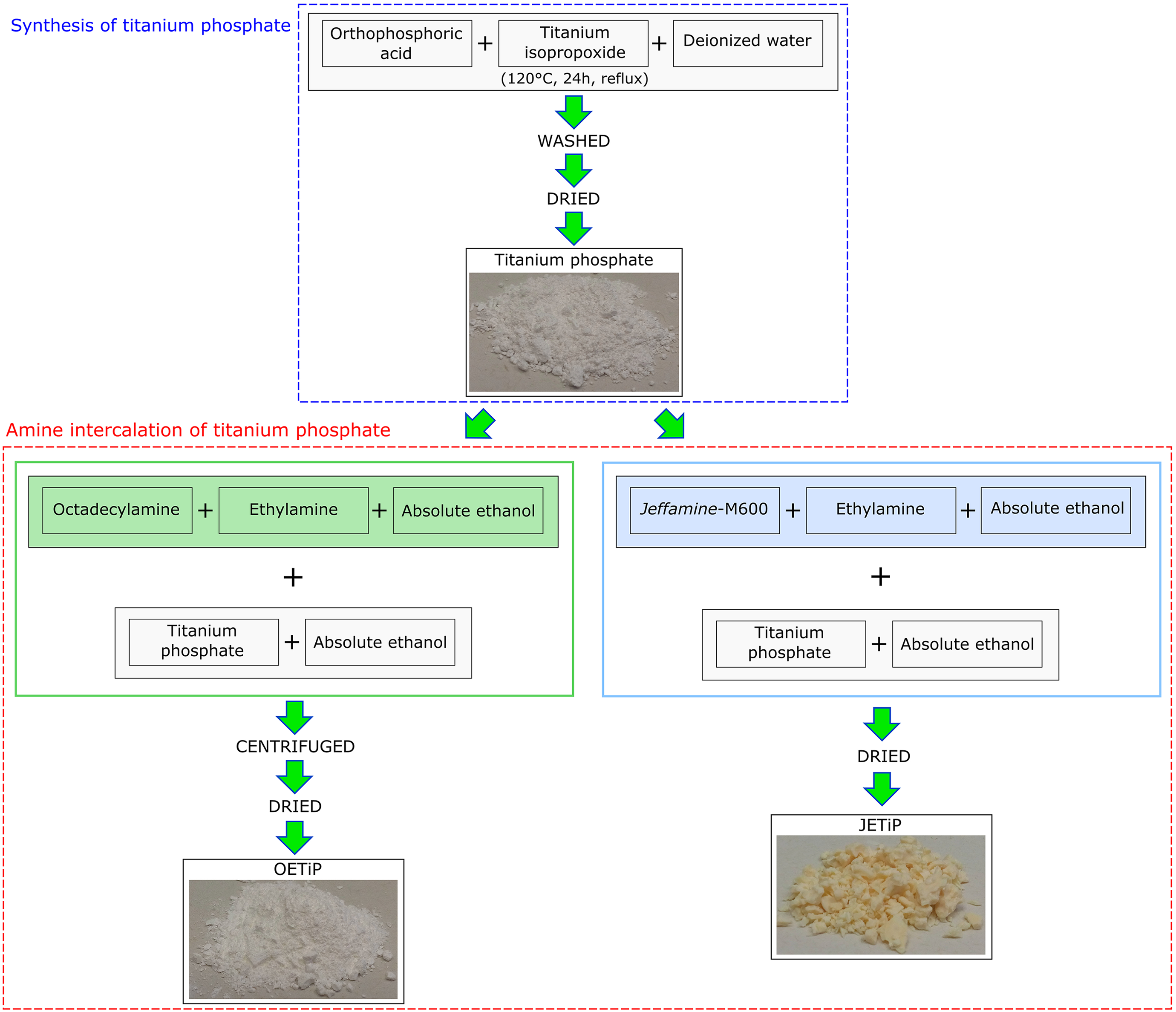
In order to facilitate the dispersion of the fillers in the PET matrix, a concentrate (masterbatch) of PET/filler containing 50 wt% of TiP, OETiP, and JETiP was processed in single-screw extruder with processing window of 250°C, 260°C, and 270°C, at 60 r min−1. The concentrate was milled for further application. Twin-screw extruder (Teck Trill DCT-20) was used in the range of 120–270°C, at screw speed of 300 r min−1. The concentrate was added to PET precursor in order to attain nanocomposites with 5 wt% of TiP, OETiP, and JETiP. After that, the nanocomposite was extruded in the pellet form. For comparison, PET precursor was processing in the same conditions. The samples were labeled as PET, PET-TiP, PET-OETiP, and PET-JETiP. Figure 2 depicts the masterbatch and nanocomposites preparation.
Schematic representation of masterbatch and nanocomposites preparation.
Characterization
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was performed in TA Instruments model Q500, between 30°C and 700°C, at 10°C min−1 and nitrogen as carrying gas. Tonset (initial degradation temperature) and Tmax (temperature at maximum degradation rate) were determined.
Differential scanning calorimetry
The differential scanning calorimetric (DSC) analysis was conducted in TA equipment model Q1000, at nitrogen atmosphere, according to the ASTM D3418.
31
Five thermal cycles were performed. Firstly, a heating cycle was performed from 50°C to 270°C, at 10°C min−1, keeping the sample for 2 min in order to eliminate the thermal history. Following, a first cooling cycle at high rate (quenching) was carried out. A second heating cycle was accomplished at the same conditions of first one. After that, a second cooling cycle at 10°C min−1 was conducted. Finally, a third cycle was done following the procedure of the first one. The glass transition temperature (Tg), cooling crystallization temperature (Tcc), and melting crystalline temperature were registered. PET degree of crystallinity (Xc) was determined by PET melting enthalpy of each sample through equation (1). ΔHm (J g−1) is the experimental melting enthalpy,
Wide-angle X-ray diffraction
Wide-angle X-ray diffraction (WAXD) analysis was performed using Rigaku Miniflex, 2θ range 2–60°, with CuKα radiation (λ = 1.5418 Å), 40 kV, 20 mA, and resolution of 0.05°. Calculation of average crystal size and dspacing was carried out through Scherrer and Bragg equations, respectively.
Fourier transform infrared spectroscopy
Infrared spectroscopy was accomplished in Perkin Elmer equipment model Frontier. The spectra were taken from 4000 to 600 cm−1, with 60 scans and 4 cm−1 of resolution.
Hydrogen low-field nuclear magnetic resonance
This characterization permits to speculate qualitatively and by indirect way the filler dispersion in the matrix and interaction polymer/filler. Molecular mobility and polymer relaxation time was investigated in a Maran Ultra 23 low-field NMR device. The relaxation time (T1H) was measured in time intervals of 2 s, taking 40 points, at 30°C. The result was expressed in terms of domain curves.
UV-Vis-NIR spectroscopy
UV-Vis-NIR Cary 5000 Varian spectrometer was used to obtain the spectra, at wavelength range of 200–800 nm, using the diffuse reflectance technique.
Results and discussions
Thermogravimetric analysis
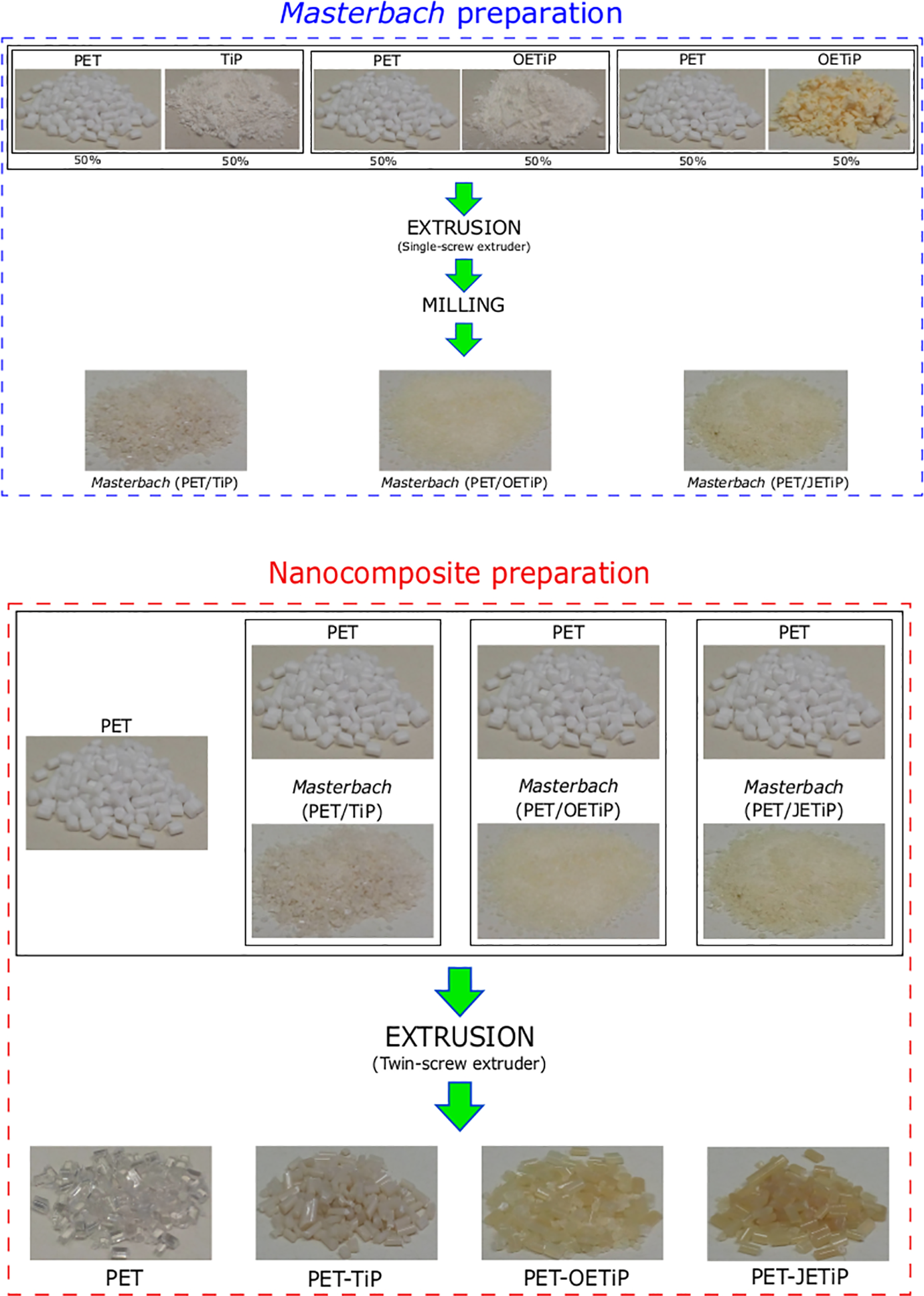
Figure 3 shows the loss weight and derivative curves of the samples. Table 1 summarizes the degradation temperatures. PET and PET-TiP nanocomposite presented one degradation step. PET-OETiP nanocomposite displayed a peak with a shoulder while for PET-JETiP one two sharp degradation steps appeared. The PET and PET-TiP curves are practically superimposed attaining similar value to Tonset but PET degradation chains at Tmax was slightly higher for PET-TiP probably due to the steric effect of the titanium lamellae. Lim et al. report on reprocessability of PET/organo-montmorillonite nanocomposites (PET/OMMT). They pointed out that the incorporation of OMMT increased PET thermal stability. 33 This is in agreement with our finding. Both PET-OETiP and PET-JETiP nanocomposites showed reduction of Tonset but more significant for PET-OETiP. For both nanocomposites, Tmax was split. PET-OETiP displayed a shoulder at 394°C and a main peak at 416°C while in PET-JETiP appeared at 305°C and 419°C. Brandão et al. reported the reduction of PET thermal stability in nanocomposites with ZrP and ZrPP attributed to the aminolysis of ester bonds by amines. 17 Mariano et al. 16 and Freitas et al. 34 pointed out the decrease of thermal stability in nanocomposites-based PP and PA-6, respectively, owing to the presence of ZrP modified with octadecylamine. By using filler with different crystalline arrangement of those studied herein, Chowreddy and coauthors revealed the increase of PET thermal stability in recycled PET/carbon nanotube composites. 32
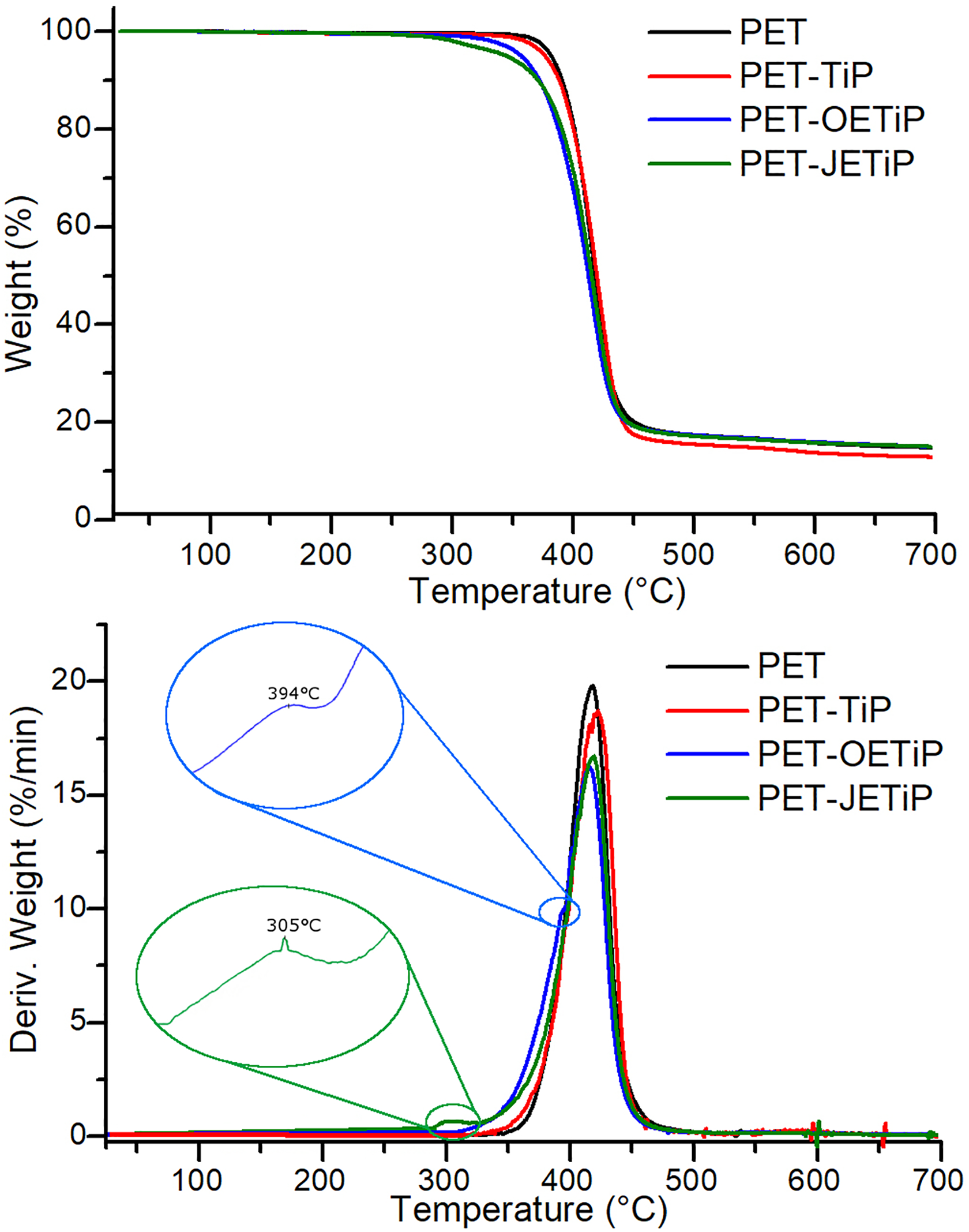
Loss weight and derivative curves of PET and PET-TiP, PET-OETiP, PET-JETiP nanocomposites.
Degradation Temperatures of PET and PET-TiP, PET-OETiP, PET-JETiP Nanocomposites.
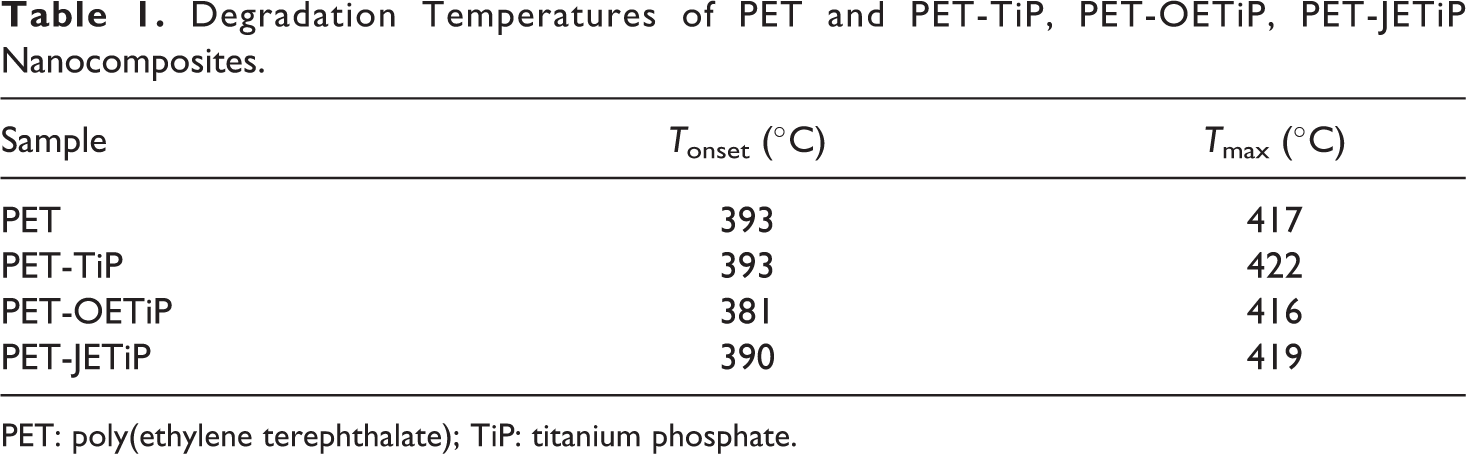
PET: poly(ethylene terephthalate); TiP: titanium phosphate.
Herein, the variations found in both degradation temperatures suggested that the intercalation of PET chains into the filler galleries was achieved. The insertion was facilitated due to the presence of octadecylamine and ether-amine as intercalation agents.
Differential scanning calorimetry
Figure 4 and Table 2 show the DSC curves and calorimetric data, respectively, according to ASTM D3418. 31 It is interesting to notice that after processing, PET was amorphous in all samples and crystallized during the first heating cycle (Figure 4(a)). Similar behavior was found by Vidotti et al., 35 investigating the effect of organomodified montmorillonite on the barrier properties of PET nanocomposites. DSC curves of second heating cycle (Figure 4(b)) showed that PET crystallized only in the nanocomposites. This is indication that the fillers increased the PET crystallization rate. PET glass transition temperature (Tg) presents value quite similar to that observed for PET-TiP nanocomposite. PET-OETiP and PET-JETiP nanocomposites display similar values of Tg but lower than the PET precursor. Cooling crystallization temperature showed strong increase in the nanocomposites as compared to the PET precursor. All melting peaks exhibit a sharp maximum at higher temperature and a shoulder at lower one. PET crystallinity degree increased by adding of the fillers. Xc was higher for all nanocomposites. The type of filler and the intercalating influence the thermal properties of PET. Lima et al. studied the effect of organically modified montmorillonite on the structure, rheological, and thermal behavior of PET/OMMT nanocomposites. 36 They mentioned the increase of Xc, Tg, and Tcc. 37 The results are slightly different of those found herein. Brandão et al. 17 reported that the high affinity between PET chain with phenyl groups on the platelet surface of ZrPP promoted the increase of its crystallization rate. Dong and coauthors studied the effect of layered double hydroxide (LDH) intercalated with stearic acid in order to overcome the PET low crystallization rate. 37 They mentioned that the nucleation mechanism of PET was induced by LDH. The addition of binary system magnesium hydroxide/exfoliated graphite (Mg(OH)2/EG) and magnesium hydroxide/carbon nanotubes (Mg(OH)2/CNT) to PET favored its crystallization, according to Gorrasi and coauthors. 38 Cui and coauthors investigated nanocomposites based on PET filled with LDH modified by terephthalate (TA). They suggested that LDH nanoparticles could effectively promote the nucleation and crystallization of PET. 39
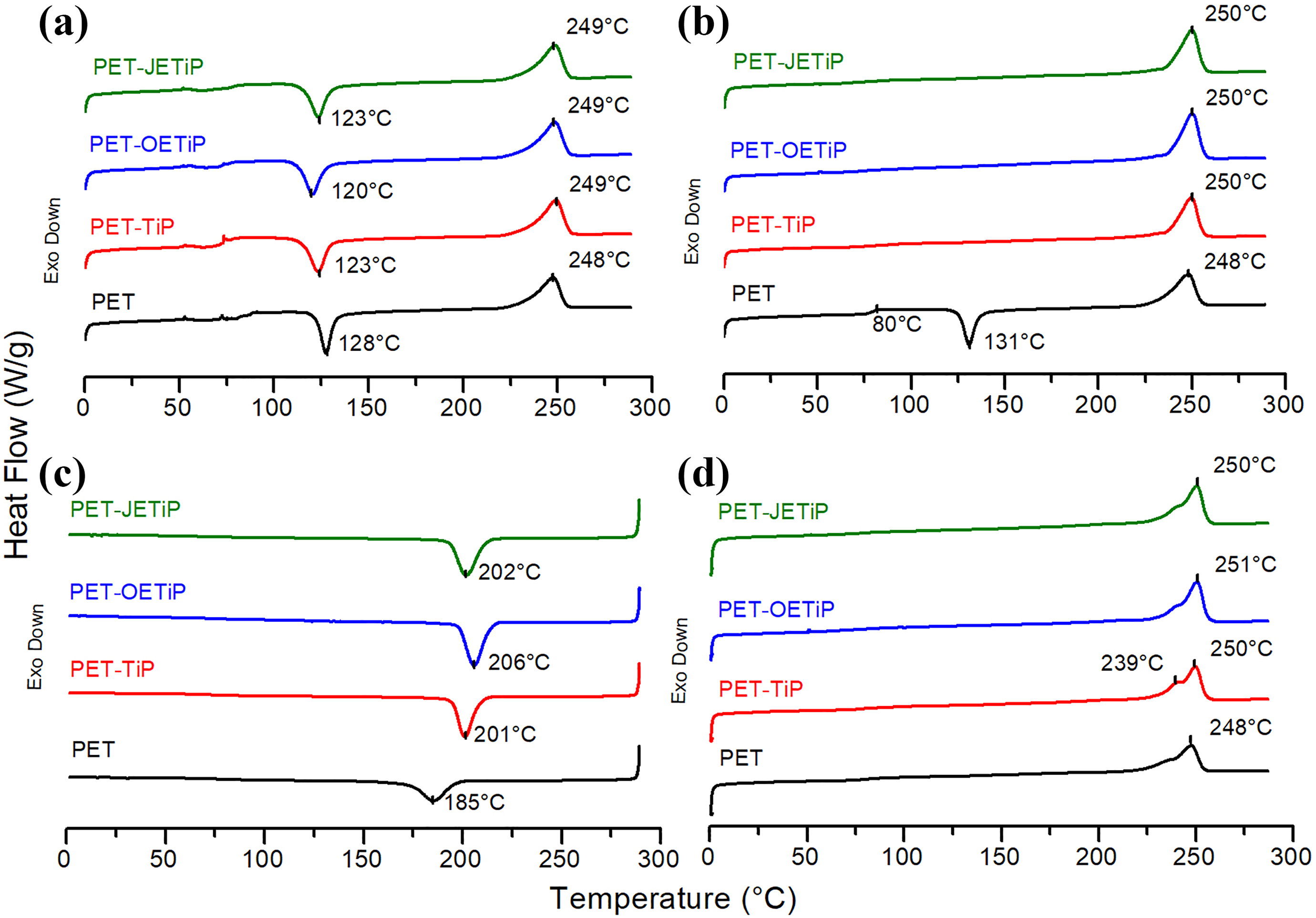
DSC curves of PET and PET-TiP, PET-OETiP, PET-JETiP nanocomposites: (a) first heating cycle, (b) second heating cycle, (c) second cooling cycle, and (d) third heating cycle.
Calorimetric properties of PET and PET-TiP, PET-OETiP, PET-JETiP nanocomposites.
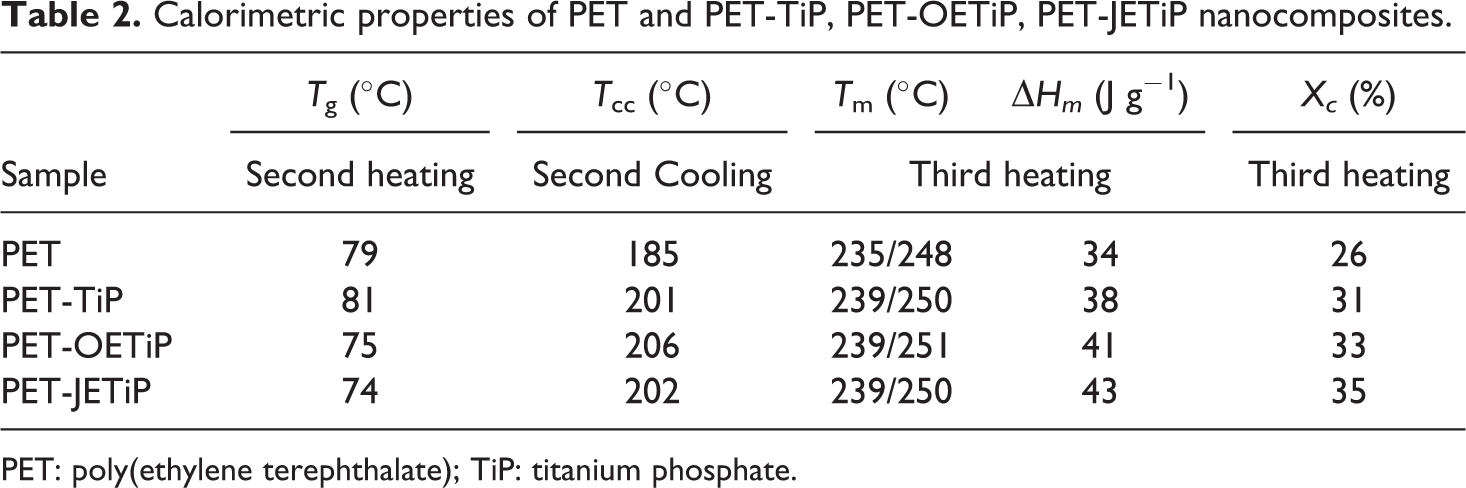
PET: poly(ethylene terephthalate); TiP: titanium phosphate.
OETiP and JETiP fillers drastically altered the PET calorimetric properties. We supposed that as intercalant both amines promoted the increase of TiP dspacing allowing the entrance of PET chains into the filler galleries. This favored the interaction between PET chains and long-chain amines leading to the reduction of PET Tg. In addition, the fillers had a role as nucleating agent increasing the PET crystallization rate. Macedo et al. 40 reported the phenomenon known as transcrystallization in their work on sustainable hybrid composites of recycled PP and construction debris. Modified montmorillonite induced the PET crystallization was described by Ou et al., 41 in the study on PET nanocomposites. Herein, the titanium crystalline lamellae acted as heterogeneous nucleation nuclei increasing the PET crystallization degree as shown in Figure 5.
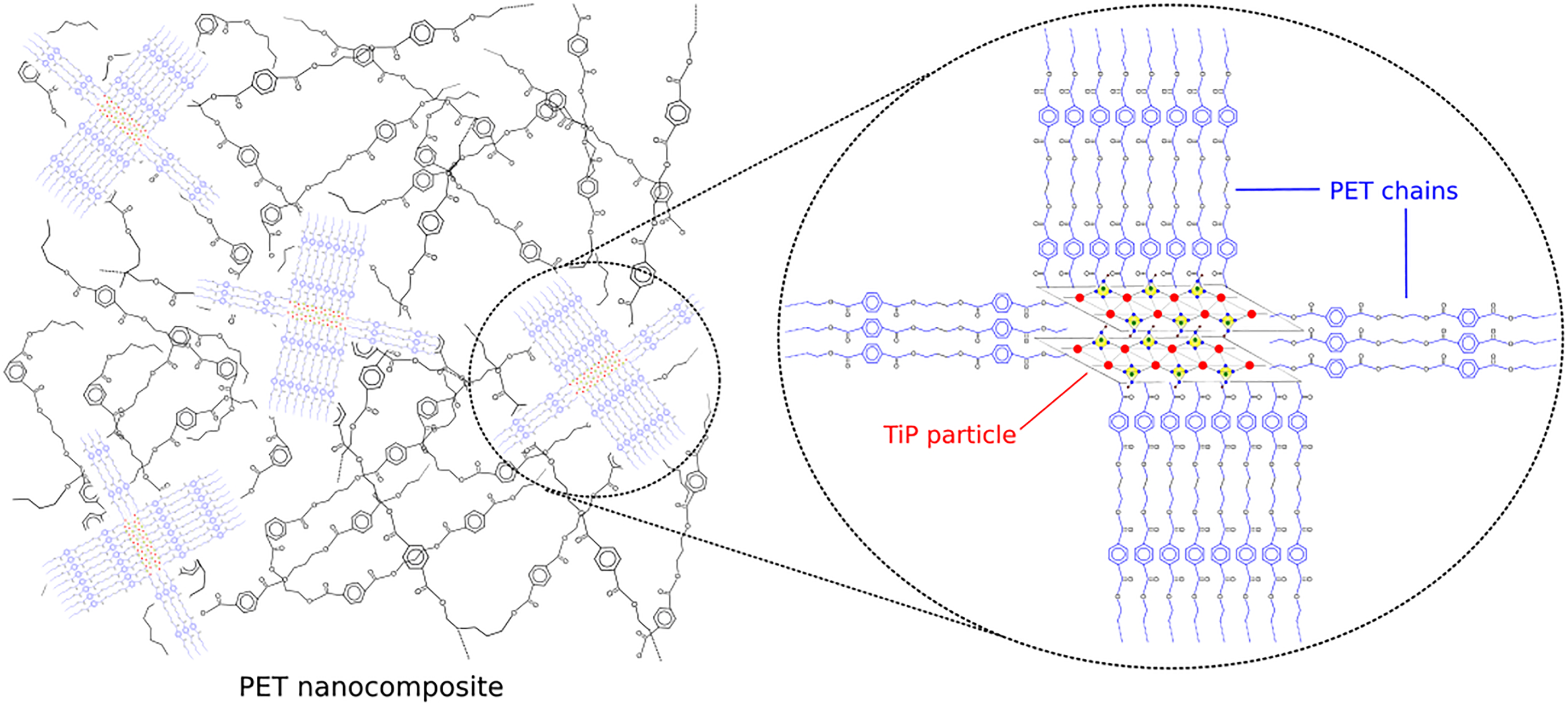
Schematic illustration of PET heterogeneous nucleation process by TiP.
Wide-angle X-ray diffraction
Figure 6 presents the X-ray diffractograms of PET and PET-TiP, PET-OETiP, PET-JETiP nanocomposites. Herein, the WAXD analysis was conducted with the samples after processing and PET was amorphous in all samples as already registered in the DSC first heating cycle. Diffraction patterns showed the outline of amorphous PET with 2θ peaks around 22° and 43° as pointed out by Ajji et al. 42 and Murthy et al., 43 in their studies on amorphous PET. The fillers reflection patterns were dissimilar in the nanocomposites. For PET-TiP nanocomposite, TiP precursor showed the 2θ angle at 11.7° (hkl 002), dspacing of 7.60 Å, and average crystalline size of 3.88 nm. As these parameters are consistent with several works on synthesis and characterization of TiP, it was concluded that PET chains did not enter into TiP lamellae.26–28,30 For the PET-OETiP nanocomposite, the TiP diffraction peak was not detected but three new diffraction angles at 2.4°, 5.3°, and 8.1° with dspacing of 37.6 Å, 16.7 Å, and 11 Å, respectively, were noticed. This is an indication that the primitive OETiP crystalline order was partially destroyed owing to the entrance of PET among filler lamellae forming at least an intercalated nanocomposite. Concerning to PET-JETiP nanocomposite, the absence of filler diffraction angle induced to infer that the PET promoted the complete damage of filler crystalline arrangement leading to the formation of exfoliated nanocomposite. Different levels of exfoliation were found by Boo et al., 44 controlling the alpha-ZrP lamellar spacing. Kim et al. 45 synthesized a thermal stable new organosilicate (DMIBrC) and prepared nanocomposites based on PET. The authors inferred that the PET/filler compatibility was improved and an exfoliated nanocomposite was achieved. Although prepared in-situ interlayer polymerization, PET did not enter into filler galleries on the study of nanocomposites containing organically modified mica. 46 Chang and coauthors found dissimilar result in PET nanocomposites with montmorillonite intercalated by dodecyl triphenyl phosphonium chloride. 47 The authors pointed out that the absence of clay peak is direct evidence that the PET/modified–MMT systems formed exfoliated nanocomposites. The effect of spherical TiO2 and platelet-like Closite20A nanoparticles on physical and mechanical properties of PET was studied by Farhoodi and coauthors. 48 They described that PET nanocomposite containing 1 wt% of nanoclay showed exfoliated structure. Also mentioned that in the nanocomposite with 3 and 5 wt% of Closite20A the displacement of filler hkl crystallographic plane and intercalated structures were noticed. The result found similarity to that described herein. In summary, the modified fillers lost the original layer order by the insertion of PET chains among their lamellae leading to suggest that intercalated and exfoliated nanocomposites could be attained. The results agree to those from TGA and DSC.
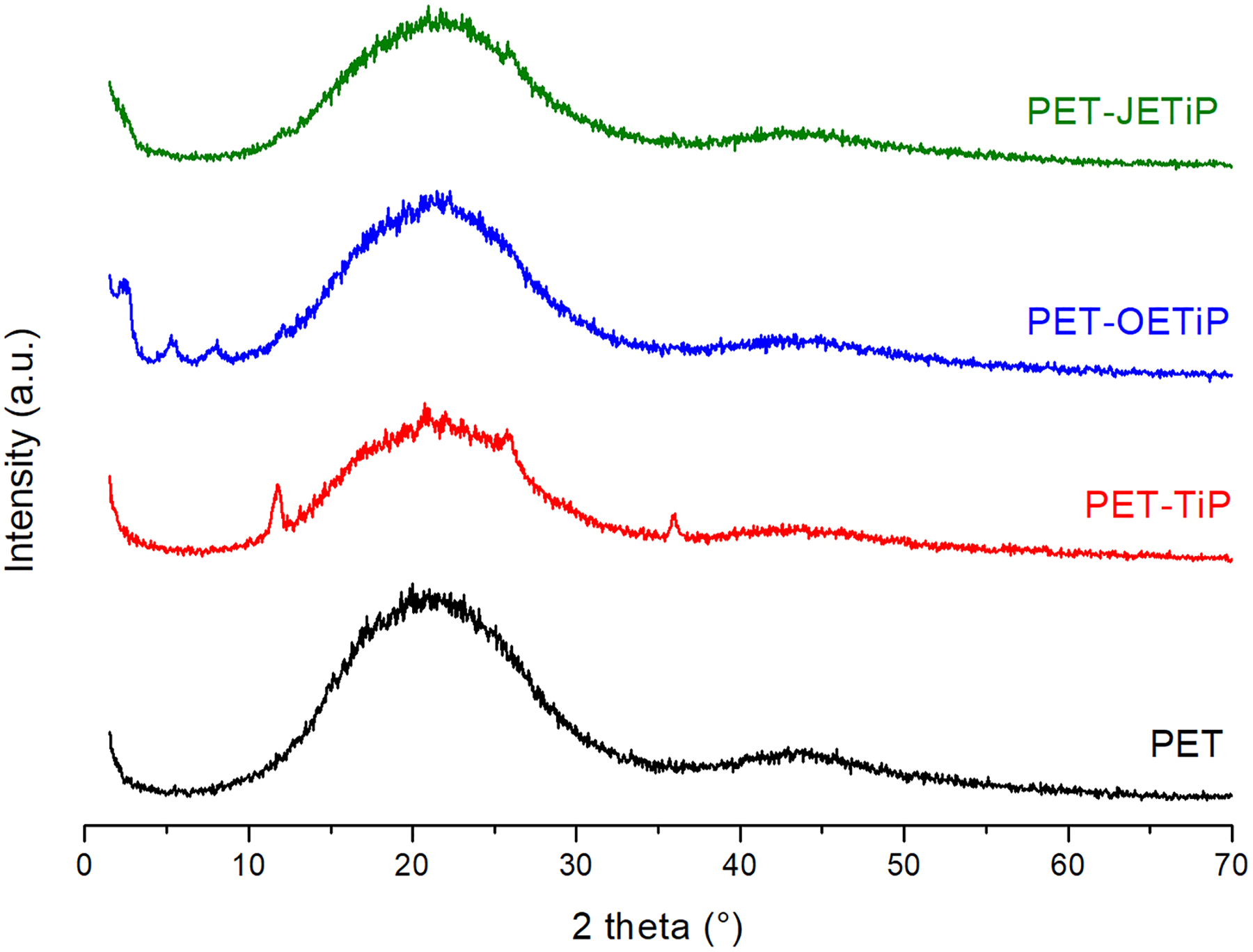
WAXD of PET and PET-TiP, PET-OETiP, PET-JETiP nanocomposites.
Fourier transform infrared absorption analysis
In order to make easier the infrared evaluation, the spectra were split in four spectral regions (Figure 7). The main absorption bands of each component are illustrated in Table 3.11,49–53 Even the nanocomposites filled with 5 wt.% of phosphates all spectra presented outline similar to the PET. For all nanocomposites, in the interval 1200–900 cm−1, PO43− symmetric and asymmetric vibrations of TiP are superimposed by PET ones. Ti–O–P–OH vibrations (539 and 619 cm−1) appeared in the PET/TiP sample. Octadecylamine absorption bands at 2957, 2918, 2850, 720 cm−1 were registered in the spectrum of PET-OETiP. Vibrations at 1108 and 1016 cm−1 related to the Jeffamine oxide bonds were hidden by PET’s.
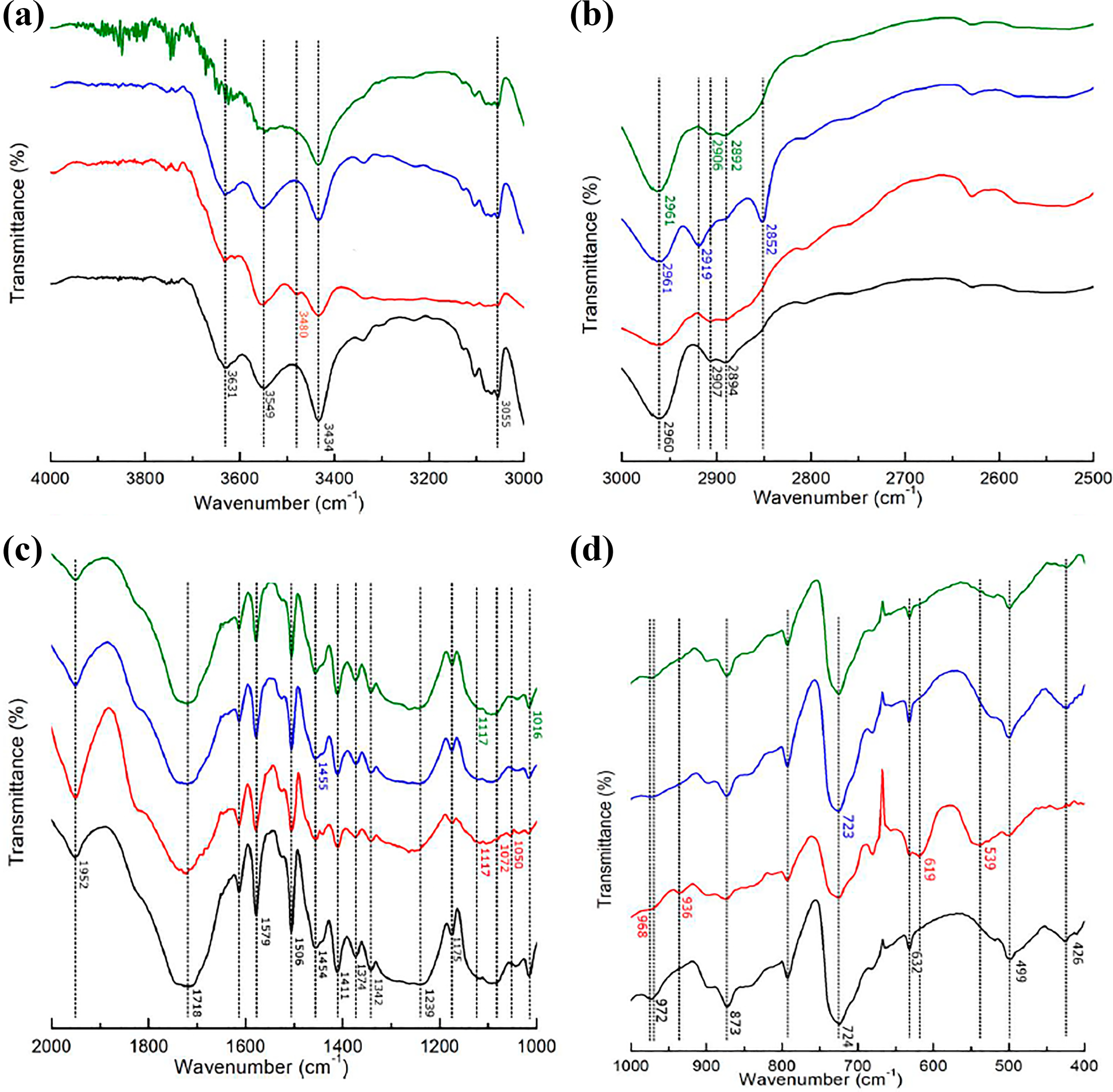
FTIR spectra of PET and nanocomposites at different spectral regions.
PET: poly(ethylene terephthalate); TiP: titanium phosphate.
Hydrogen low-field nuclear magnetic resonance
Figure 8 and Table 4 show PET and nanocomposites domain curves and relaxation time, respectively. The analysis was conducted in the samples after processing considering all of them in the amorphous state as already commented above. The presence of filler induced the enlargement of the nanocomposites domain curves. It is strongly possible to presume that there were high degree of filler dispersibility into the PET matrix and the polymer/filler interaction. Study on molecular dynamics of PET composites filled with muscovite mica by low-field NMR suggested filler action as reinforcing agent. Changes in T1H were associated with the content of amorphous phase in the composite. 54 To obtain information on intermolecular interaction in nanocomposites based on PP filled with crude Brazilian montmorillonite clay, Nogueira et al. 55 applied nuclear magnetic resonance with different nuclei 13C, 29Si, and 27Al. The clay interfered in the PP crystallization due to a specific interaction between components. PET-OETiP and PET-JETiP domain curves varied significantly. These two samples presented lower T1H as compared to the PET one. We suggested that the filler constituents, TiP and the amines, showed antagonistic effects on the PET matrix. While TiP lamellae induced a stiffening effect, the long-chain amines had the opposite one. For both nanocomposites, the amines effect was more prevalent due to the reduction of relaxation time. This assessment agrees with the lowest Tg values observed for those samples.
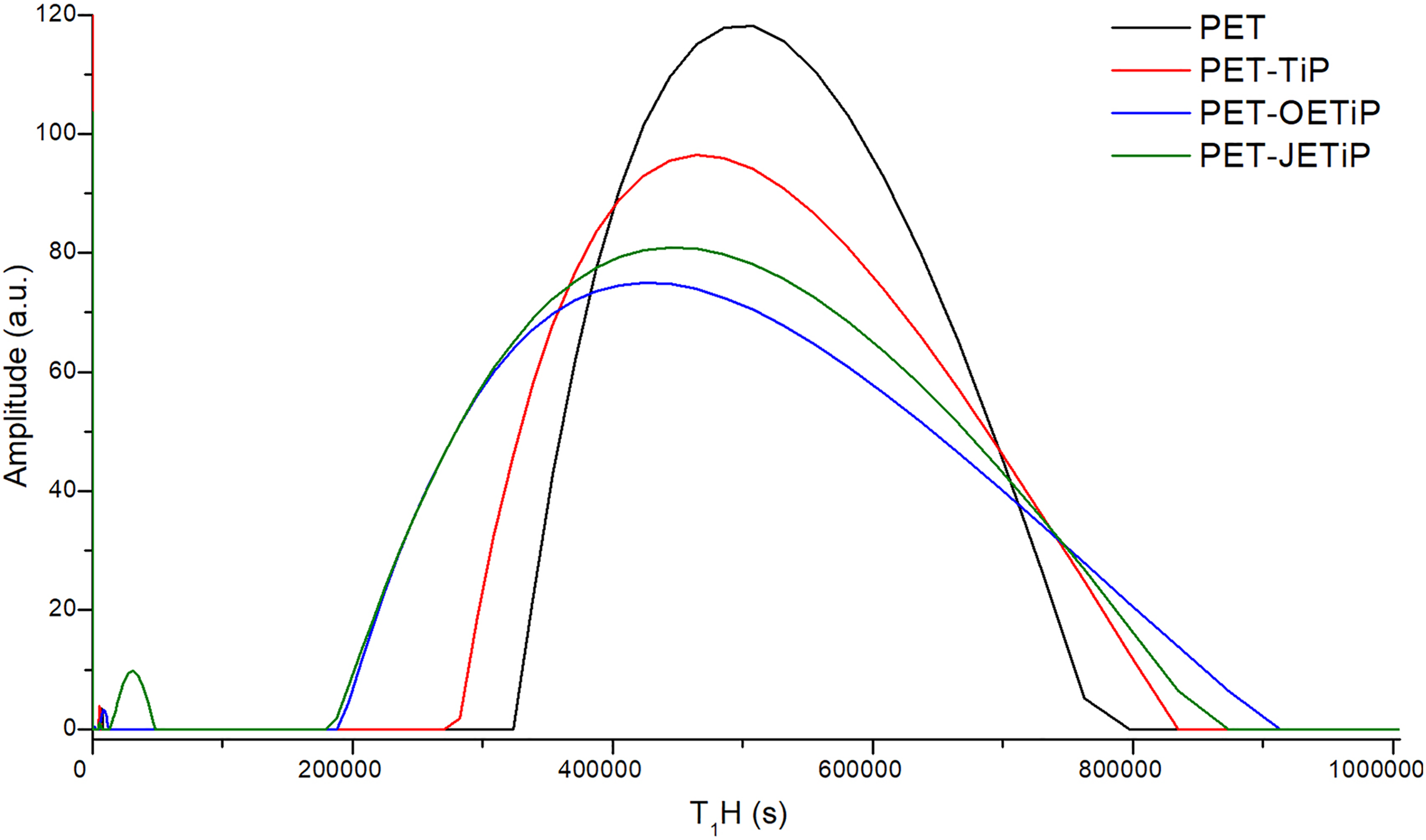
1H LFNMR domain curves of PET and nanocomposites.
1H LFNMR data of PET and nanocomposites.

PET: poly(ethylene terephthalate); TiP: titanium phosphate; 1H LFNMR: hydrogen low-field nuclear magnetic resonance.
UV-Vis-NIR spectroscopy
Figure 9 illustrates the diffuse reflectance spectra of the PET and nanocomposites. Table 5 presents the absorbance at UV-Vis region. All samples absorbed at UVB and UVC region (320–200 nm). The presence of fillers intensified the absorption in UVB and UVC regions. The absorbance increased as following PET-JETiP > PET-OETIP > PET-TiP. Zhang et al. 56 accomplished electrospinning of hybrid nanofibers based on ZnO nanocrystal/PET. The UV-Vis diffuse reflectance spectra revealed the blue shift in the absorbance curve ascribed to the quantum confinement effects of ZnO nanoparticles in the hybrid materials. Bi-component filaments based on PET/PET-TiO2 were covered by metal photo-deposition. Through diffuse reflectance, gold and silver elements exhibited absorption at visible light region. 57 It is interesting to notice that absorption at UVA region (400–320 nm) appeared for PET-OETIP and PET-JETiP. The last one showed the highest absorption. As the amount of filler was constant (5 wt%) in all nanocomposites, the enhance of the absorbance was assumed to the filler dispersion and additionally a some interaction between polymer/filler. The study indicated that the fillers have potential for future application as UV radiation barrier. The results corroborate those found in DSC, WAXD, and hydrogen low-field nuclear magnetic resonance (1H LFNMR) sections.
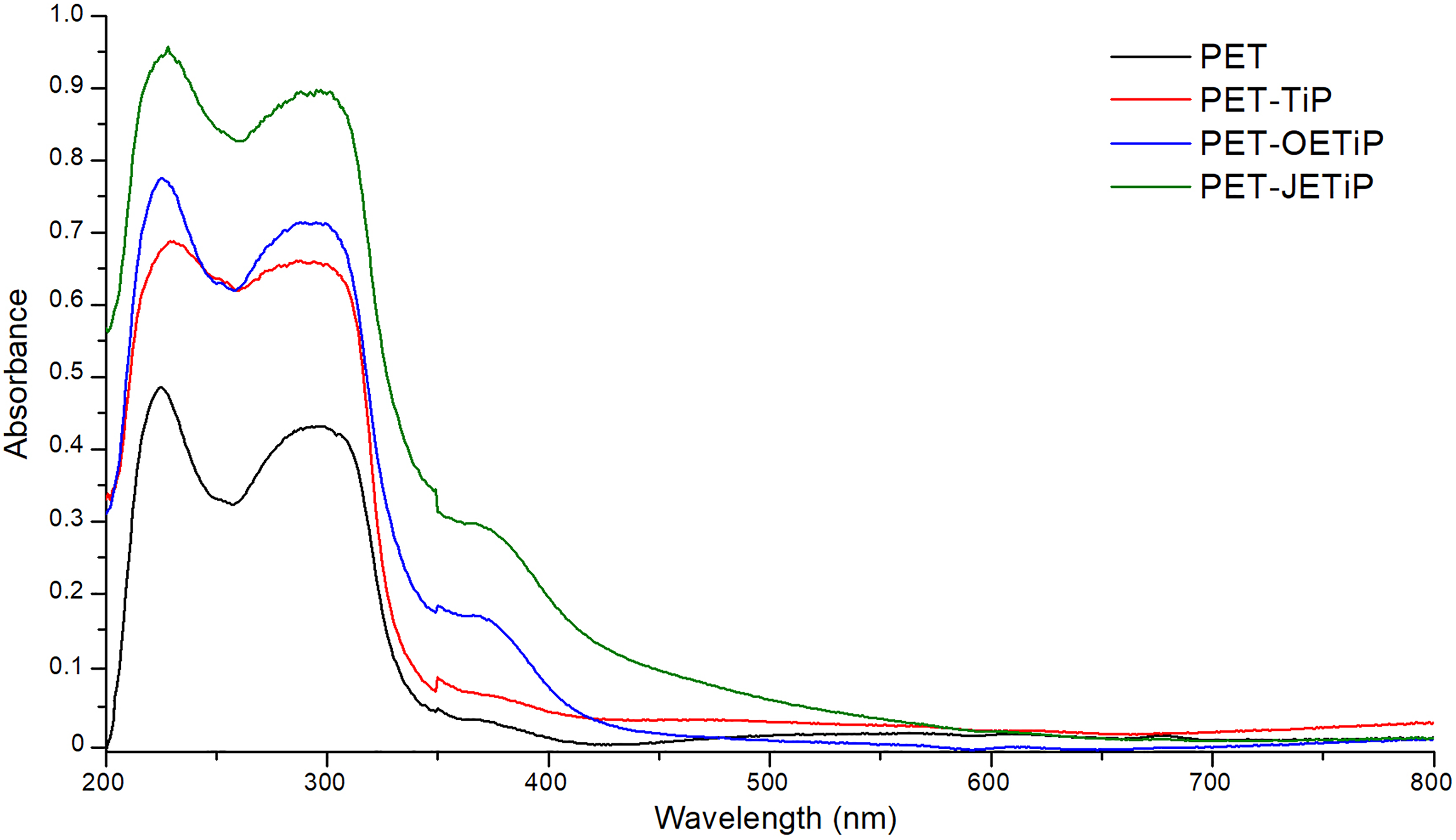
Diffuse reflectance spectra of PET and nanocomposites.
Diffuse reflectance at different UV region for pet and nanocomposites.
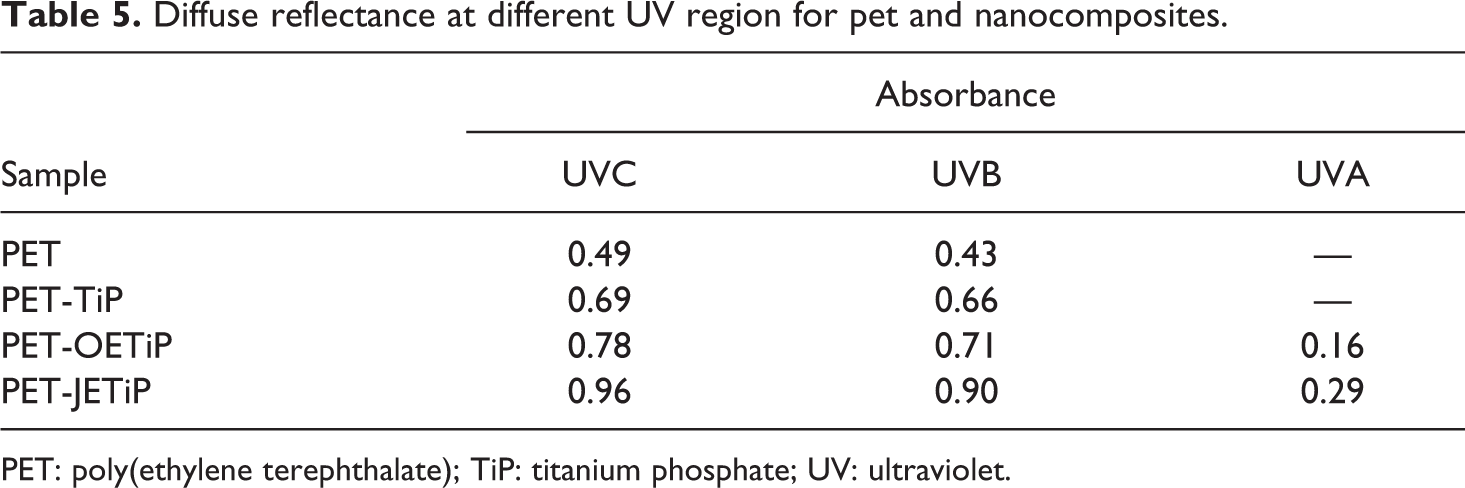
PET: poly(ethylene terephthalate); TiP: titanium phosphate; UV: ultraviolet.
Conclusion
Since TiP has not been studied as a UV absorber, in this work was investigated its effect on PET properties. The degradation temperatures suggested that the intercalation of PET chains into the filler galleries was achieved. PET glass transition temperature decreased due to the presence of amines inside of the TiP interlamellar region. The fillers promoted the increased of cold crystallization temperature owing to the heterogeneous nucleation. This led to the increase of PET crystallinity degree. WAXD suggested that intercalated and exfoliated structures were achieved. For all filler, the PET molecular mobility increased. Highest values of absorbance were attained by the PET-OETIP and PET-JETiP nanocomposites. The work continues at IMA-UFRJ.
Footnotes
Acknowledgements
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nıvel Superior (CAPES), Finance Code 1, and Universidade Federal do Rio de Janeiro for finance supporting of this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nıvel Superior (CAPES), Finance Code 1, and Universidade Federal do Rio de Janeiro.