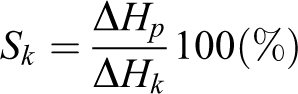Abstract
The structure and thermal characteristics of nanocomposites based on polyvinyl alcohol (PVA) and montmorillonite (MMT) intercalated with polyvinylpyrrolidone were investigated by X-ray diffraction analysis and differential scanning calorimetry. The modification of PVA with intercalated MMT reduces the degree of crystallinity of the resulting nanocomposites but significantly increases their thermal stability. Under ultrasound, the intercalated MMT was completely distributed in a PVA solution and formed a monocrystalline structure. Films based on PVA with modified MMT were cross-linked at 110°C in the presence of 5 wt% acrylic acid and 0.5 wt% Ferrous(II) sulfate as an initiator. The formed films have a homogeneous cross-linked structure.
Keywords
Introduction
Nanocomposites are a new class of alternative materials in which nanosized fillers are dispersed into a polymer matrix, resulting in improved elasticity, strength, thermal stability, fire resistance, and barrier properties.1,2 In this regard, nanocomposites are an attractive alternative to polymer-filled disperse powders for use in the packaging, medical, automotive, and other industries.3,4
Currently, layered silicates, in particular montmorillonite (MMT), hectorite, and saponite, are the most used nanofillers. 5 Polymer molecules can intercalate in a gallery of layered silicates, pushing the layers and creating a nanocomposite.6–8 The most common methods for producing aluminosilicate nanocomposites are polymer modification during polymerization, mixing with a polymer in the viscous-fluid state at high shear stress, or mixing in solution. 9 To achieve a high dispersion of nanofiller in a polymer matrix, high-pressure mixing, centrifugation, and ultrasonic treatment are used. Ultrasonic treatment is one of the most important methods for increasing the gallery interval between silicate layers in nanoclay, which considerably improves their dispersion in polymers.9,10 In addition, ultrasonic treatment also affects the polymer matrix, resulting in the formation of a material with a new structure and altered properties. 11
Polyvinyl alcohol (PVA) is used in the industrial, commercial, medical, and food sectors to produce many products, such as varnishes, resins, surgical yarns, and food packaging, that are often in contact with food. 12 Therefore, improving the mechanical and chemical stability of PVA is an urgent problem that can be successfully solved by combining PVA with layered silicates.
Nanocomposites on a PVA and layered silicate base have improved characteristics, explained by the formation of a new crystalline structure. Strawhecker and Manias 13 examined the structure of PVA-based nanocomposite with a 20 wt% MMT content using a transmitted electron microscope. In the intercalated structures, inorganic layers with parallel to each other orientation, characteristic of natural silicates, were separated by superfine (1.3–5 nm) layers of PVA. Due to the orderly parallel orientation of the silicate layers, the intercalated structures have clearly expressed peaks in the X-ray diffraction (XRD) curves. In layered structures, the silicate layers are much further apart (>>5 nm). In this case, the orientation of the layers is chaotic, and there is no parallel aggregation. 14
Aslam et al. 15 used XRD to show that with the increase in MMT content from 40 wt% to 90 wt% in PVA, the height of the interlayer space in the nanocomposite is uniformly reduced. At low concentrations of layered silicate, the diffraction peak, corresponding to the magnitude of the MMT interlayer space, moves to the 2θ < 3° area. At these concentrations, the structure of MMT intercalated silicate layers, which are characterized by a more than 5-nm interlayer space, remains ordered.
Using the differential scanning calorimetry (DSC) method,13,16,17 it was established that with the increasing MMT content in PVA, the intensity of peaks corresponding to the glass transition and melting points decreases. With a more than 60 wt% MMT content in the composite, the peaks completely disappear. 13 This suggests that in such materials, the polymer is under the influence of inorganic layers. At lower MMT concentrations (up to 20 wt%), two clear peaks, referring to the melting point, appear on the DSC curves. One of them is in the region of the unfilled polymer’s melting point, and the second one is at a higher temperature. The double (dual) melting point corresponds to two crystalline phases, one phase is formed predominantly by syndiotactic molecular chains and the second phase by atactic ones. The difference between the melting points is 15–22°C.
Rajeswari et al. 18 melted polymer nanocomposites using PVA with the different weight percentages of calcined clays in a twin-screw extruder. Scanning electron microscopy confirmed the degree of exfoliated nanocomposites. The polymer and functionalized nanoclay interactions were reflected in the enhanced thermal stability and mechanical properties.
Fang et al. 19 further improved and enhanced the performances and properties of polyvinyl acetate (PVAc); MMT and N-hydroxymethyl acrylamide (NMA) were simultaneously introduced to polymerize with vinyl acetate (VAc). Linear macromolecular chains of PVAc-NMA were formed in MMT layers. MMT was exfoliated into layers or sheets of nanoparticles and dispersed randomly in the PVAc-NMA matrix. PVAc-NMA-MMT had good dispersion, excellent storage stability, and high static tensile strength (6.47–6.85 MPa).
Bee et al. 20 investigated the effect of calcined eggshell and MMT on the physicomechanical properties and thermal characteristics of PVA blends. The addition of MMT particles significantly reduced the tensile strength and elongation at break of PVA blends. This indicates that the MMT particles did not provide a reinforcement effect to polymer matrix of PVA blends due to the poor dispersion and intercalation effect of MMT particles in PVA, as evidenced by shifting 2θ to higher values in the XRD analysis. However, the effective intercalated MMT particles in the PVA matrix were only observed in a polymer matrix with 2 phr calcined eggshell added PVA/MMT blends with the increased d-spacing of deflection peak (002). The incorporation of low-calcined eggshell loading levels (≤2 phr) showed a significant increase in the optimum melting temperature and enthalpy of melting for PVA and PVA/MMT blends. Additionally, there are known studies 21 in which the thermal stability of PVA is increased by adding zinc oxide nanoparticles.
The physicomechanical properties of PVA depend differently on the MMT content. The Young’s module of a nanocomposite with 4 wt% MMT is three times higher than that of unfilled PVA. The increase in MMT content decreases the dependence of the elasticity modulus on the MMT concentration. 22 The maximum tensile stress of PVA/MMT composites depends slightly on the MMT concentration. The higher the MMT concentration, the less fracture load is observed.23,24
A method for obtaining nanocomposite films based on PVA and MMT intercalated with polyvinylpyrrolidone (PVP) was previously developed.25,26 PVP initiates the polymerization of hydroxyalkylene methacrylates as an active complexing agent, 27 forms new polymeric random cross-linked matrices,28,29 and contributes to the formation of homogeneous polymer mixtures based on amphipolar polymers. 30 In this work, 31 the formation of the intercalated structure of MMT during PVP modification in aqueous solution by ultrasound is substantiated by X-ray and DTA analyses. Intercalated PVP MMT is easily distributed into PVA aqueous solutions without agglomerate formation. PVA:MMT = 12:1 is optimum in terms of film strength and water resistance. 25
The objective of this work was to investigate the influence of composition and production conditions on the structure and thermal characteristics of the developed nanocomposites using XRD and DSC analyses.
Research methods
SUNDY PVA 088-20 from the Chinese firm (PVA 088-20, 23 mPa·s; Sinopec Sichuan Vinylon Works, China) was used for producing films. Eight percent (by weight) solutions of PVA in water were used. Dissolution was carried by stirring out at 60°C with a magnetic stirrer for 2–3 h. Montmorillonite-polyvinylpyrrolidone mixture (MPM) (MMT:PVP = 1:5) obtained by Krasinskyi et al.’s method31,32 was added to the prepared PVA solutions in the amount such that the ratio of PVA:PVP elementary links should be 12:1. At the same time, compositions based on PVA with similar amounts of unmodified MMT were prepared. The resulting mixtures were treated for 3 min with 22 kHz ultrasound using a “Wave UZTA-0.4/22-OM” manufactured by "U-Sonic," Russia. To obtain films, the mixture was poured into special forms with a polyethylene lining. After formation, the film was dried in air at room temperature. The resulting films were separated from the form and subjected to heat treatment in an oven at 110°C for 30 min. To obtain water-insoluble films, 5 wt% acrylic acid and 0.5 wt% polymerization initiator (FeSO4) were added to these solutions. FeSO4 initiates the cross-linking reaction due to the formation of an active complex with charge transfer in the presence of PVP. 27 The behavior of the obtained films in water was investigated. Only a heat-treated film containing acrylic acid is water-insoluble.
The research objects were sample 1—PVA-based film, sample 2—PVA/MPM-based film (PVA:MPM = 12:1) dried at room temperature, sample 3—PVA/MPM-based film (PVA:MPM = 12:1) heat-treated at 110°C, sample 4—PVA/MMT-based film (PVA:MMT = 12:1) dried at room temperature, sample 5—PVA/MMT-based film (PVA:MMT = 12:1) heat-treated at 110°C, and sample 6—PVA/MPM-based film (PVA:MPM = 12:1) cross-linked at 110°C in the presence of acrylic acid (5 wt%) and FeSO4 (0.5 wt%) as the initiator.
X-ray studies were performed on a Dron-4-07 diffractometer manufactured by CRI "Burevestnik," Russia. Illuminating lamps with a copper anode and Ni-filter were used. The research was carried out in the 4–60° 2θ range, the step size was 0.1, and the pulse measurement time was 8 s.
Thermal research was carried out on a differential scanning calorimeter SDT Q600 (TA Instruments, New Castle, DE, USA). The changes in sample weight (thermogravimetric analysis) were registered simultaneously with the processes accompanied by release or absorption of heat (DSC/differential thermal analysis) in the temperature range from room temperature to 1500°C. Samples of nanocomposite films (3–4 mg) were investigated in an argon atmosphere at a 10°C min−1 heating to 20–450°C. According to the measured heat of melting ΔHp and PVA crystallites heat of melting ΔHk = 156.8 J g−1,33 the degree of crystallinity was calculated according to the formula
Research results and discussion
The thermolysis of samples 1–5 took place according to similar mechanisms, passing five stages (Table 1 and Figure 1). At the first stage, in the 44–46°C range, the composites’ glassy state transits to a highly elastic physical state, which is accompanied by insignificant endothermic effects on the DSC curves. It should be noted that the composites compositions and the conditions for obtaining films practically do not affect the samples’ glass transition temperatures.
Results of thermal analysis.
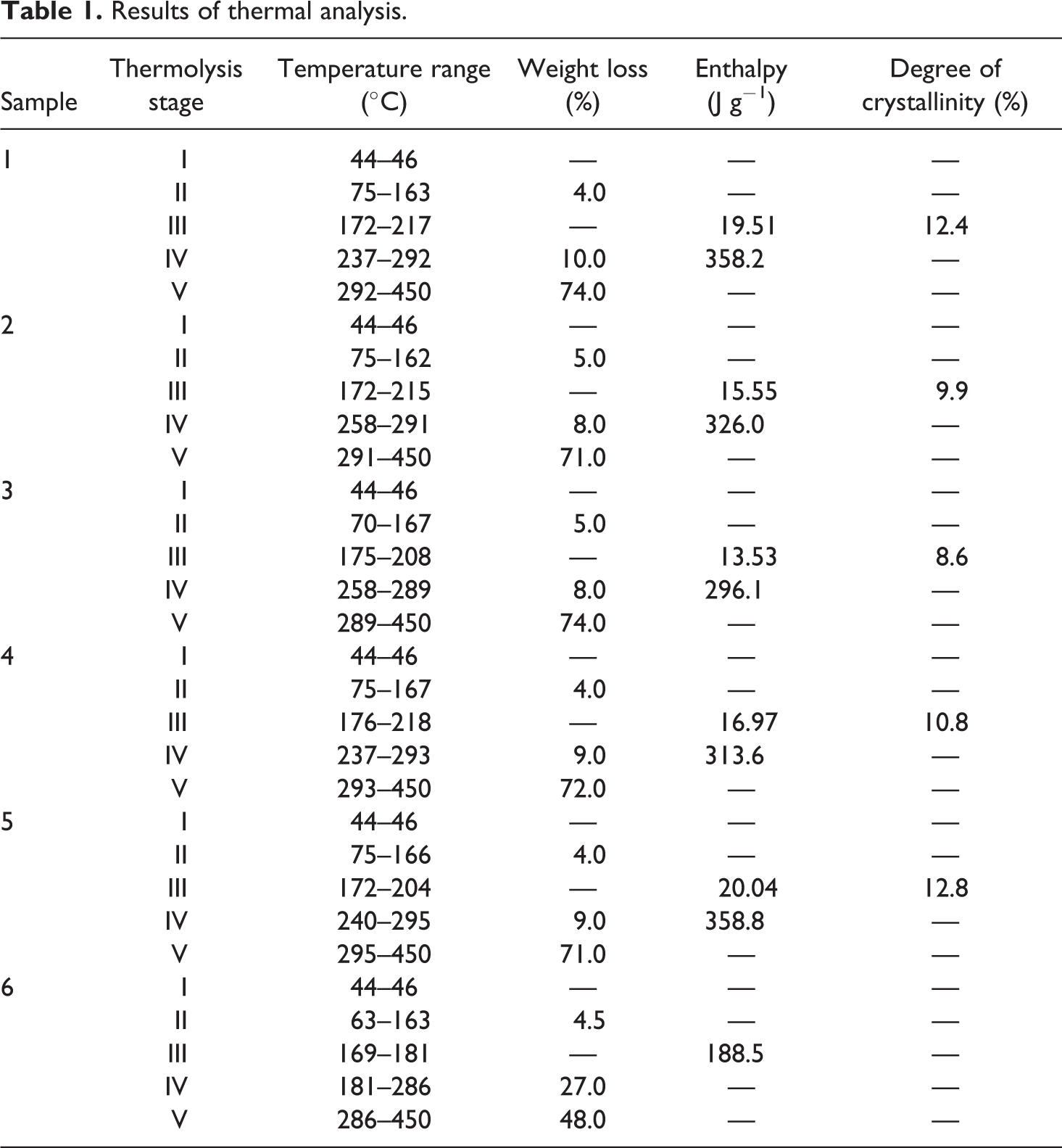
DSC curves of the composites based on modified PVA: (a) sample 1—a film based on the initial PVA, (b) sample 2—a film based on the PVA:MPM = 12:1 composition, dried at room temperature, (c) sample 3—a film based on the PVA:MPM = 12:1 composition, heat-treated at 110°C, (d) sample 4—a film based on the PVA:MMT = 12:1 composition, dried at room temperature, (e) sample 5—a film based on the PVA:MMT = 12:1 composition, heat-treated at 110°C.
In the 70–167°C range, at the second stage of thermolysis, the separation of physically bound water occurs. This process is accompanied by a slight weight loss (4–5%) and the appearance of insignificant endothermic effects on the DSC curves. Samples 2 and 3 are characterized by higher moisture content (5%) due to the presence of hygroscopic PVP in their composition.
In the 172–218°C range, during the third stage of thermolysis, an endothermic effect without weight loss is observed on the DSC curves, corresponding to composite melting (Figure 1). This effect is the most acute and deep for samples 1, 4, and 5, which do not contain amorphous PVP. Additionally, these samples have higher values of melting heat and, accordingly, degree of crystallinity (Table 1). Samples with unmodified MMT, which were heat-treated at 110°C, have the highest degree of crystallinity. The addition of PVP-modified MMT to PVA reduces the degree of crystallinity of the resulting composites. Heat treatment increases the degree of crystallinity by 2%, but it remains lower than that of the initial PVA. The absence of double melting peaks on the DSC curves of the created nanocomposites, in contrast to the endotherms obtained for PVA/MMT-based nanocomposites,13,16,17 indicates a complete distribution of MMT in PVA and the formation of a single-crystal structure. This fact also confirms the correctness of the chosen technology for PVA-based nanocomposites.
In the 237–295°C range (fourth stage of thermolysis), PVA cross-linking and the hydrolytic and oxidative composite degradation occur simultaneously. These processes are accompanied by weight loss and the appearance of endothermic effects on the DSC curves. The heat resistance of the composites containing intercalated MMT was 21°C higher and their weight loss was lower compared to the initial PVA (PVA thermal-oxidative degradation temperature is 237°C) and the composites containing nonintercalated MMT. The temperature of thermal-oxidative degradation beginning is about 258°C for the composites with modified MMT (Table 1 and Figure 1(b) and (c)). Thermal treatment of these samples at 110°C did not affect their thermal stability. Samples with unmodified MMT had a thermal-oxidative degradation temperature beginning at about 237°C, which increases to 240°C after heat treatment. Composites with intercalated MMT have the highest temperatures at the beginning of intense weight loss, that is, for the PVA:MPM = 12:1 composition, the temperature is 282°C, which decreases to 280°C after heat treatment. The intense weight loss of the PVA:MMT = 12:1 composite begins at 276°C and decreases to 268°C after heat treatment. The initial PVA intensively loses weight at 273°C.
During the fifth stage of thermolysis, in the 289–450°C range, thermal-oxidative degradation of hydrolysis and combustion products, accompanied by intense weight loss, takes place. The weight loss at this stage is more than 70% for all samples, but the weight loss of the resulting composites is lower than the initial PVA. Moreover, the degradation processes occurring in the composites were less deep than in the initial PVA, as evidenced by significantly lower exopics in this area.
Thus, it has been established that composites based on PVA with PVP intercalated MMT (ratio of 12:1) have higher thermal stability than the initial PVA or composites with unmodified MMT. There are studies 20 in which the thermal and mechanical properties of the PVA/MMT mixture were increased by the addition of calcined eggshells to the composite. The authors explain the improvement of the properties by blocking hydrogen atoms in the PVA hydroxyl groups. While in our studies, we can increase the heat resistance of the PVA/MMT composite using MMT intercalated by PVP. However, previous studies found that films of this composition were not completely cross-linked even after heat treatment at 110°C, which results in high water absorption. 22 Therefore, using DSC, we investigated an insoluble film based on the PVA:MPM = 12:1 composition, cross-linked at 110°C in the presence of 5 wt% acrylic acid and 0.5 wt% FeSO4 initiator (Figure 2 and Table 1, Sample 6).
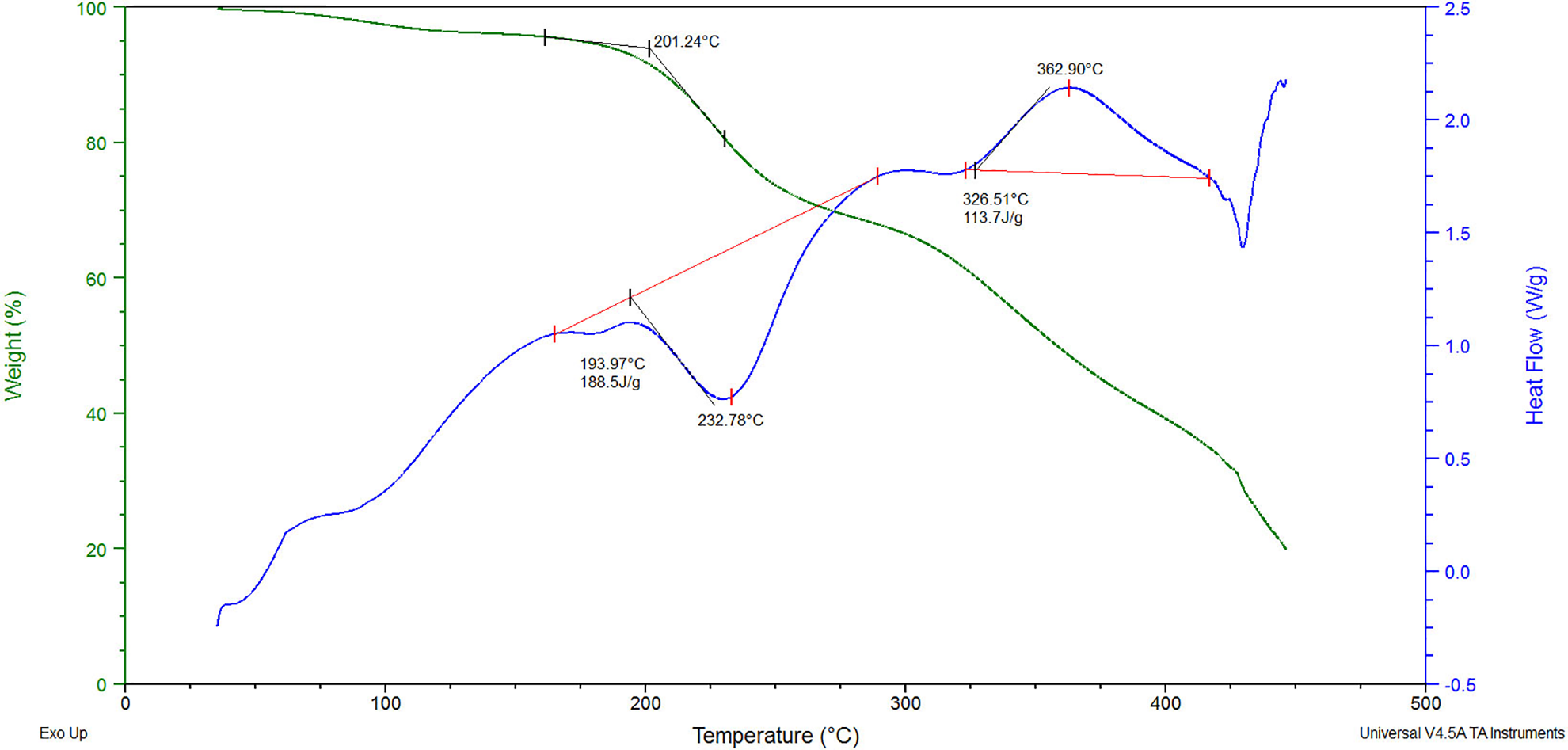
Thermal analysis of a film based on the PVA:MPM = 12:1 composition, cross-linked at 110°C in the presence of 5 wt% acrylic acid and 0.5 wt% FeSO4 initiator.
The thermolysis of the cross-linked composite also has five stages. At the first two stages, the processes are similar to those occurring in samples 1–5. In the 44–46°C range, the transition from glassy to a highly elastic physical state occurs, and in the 63–163°C range, the evaporation of physically bound water with a weight loss of 4.5% is observed. At the third stage of thermolysis, in the 169–181°C temperature range, sample softening occurs with the appearance of a slight endothermic effect on the DSC curve. This process is accompanied by a significant 188.5 J g−1 absorption of heat (the melting heat of the initial PVA is 19.51 J g−1), which confirms the high degree of composite cross-linking. In the 181–286°C range (fourth stage of thermolysis), additional PVA cross-linking, acrylic acid evaporation, and oxidative degradation of the composite occur simultaneously. These processes are accompanied by a 27% weight loss and a clear endothermic effect on the DSC curve. The effect of PVA physical and chemical cross-linking is expected in this temperature range. Physical cross-linking occurs due to the formation of hydrogen bonds between PVA hydroxyl groups and acrylic acid. At the same time, chemical structuring of PVA through the formation of inter-macromolecular bridges occurs due to polymerization with respect to double bonds under the influence of temperature and MMT as a catalyst. These processes lead to the formation of a homogeneous cross-linked structure. This is confirmed by XRD analysis (Figure 3) and the low depth of the destructive processes at the fifth stage of thermolysis in the 286–363°C range (Figure 2). In this range, a fuzzy and insignificant endothermic effect is observed on the DSC curve. Only one intensive crystalline reflex is observed in the 2θ ∼ 16–23° region of diffraction angles (Figure 3). PVA has crystalline reflexes of 100, 101, 101, and 200 at the diffraction angles 2θ = 16.0°, 19.4°, 20.1°, and 22.7°, respectively (Figure 4). This indicates the formation of a homogeneous cross-linked structure in sample 6.
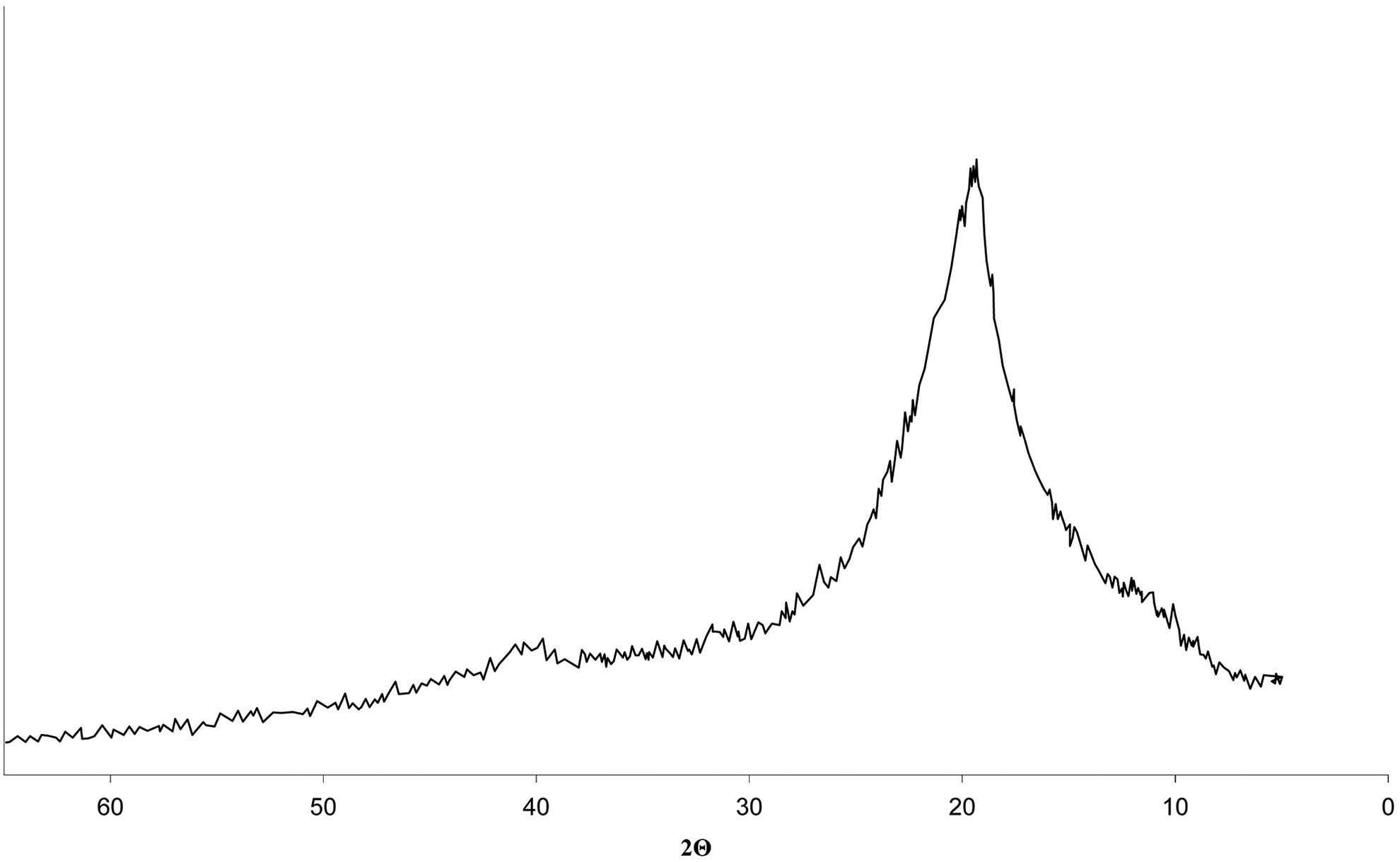
The diffractogram of the composite based on the PVA:MPM = 12:1 composition, cross-linked at 110°C in the presence of 5 wt% acrylic acid and 0.5 wt% FeSO4 initiator.
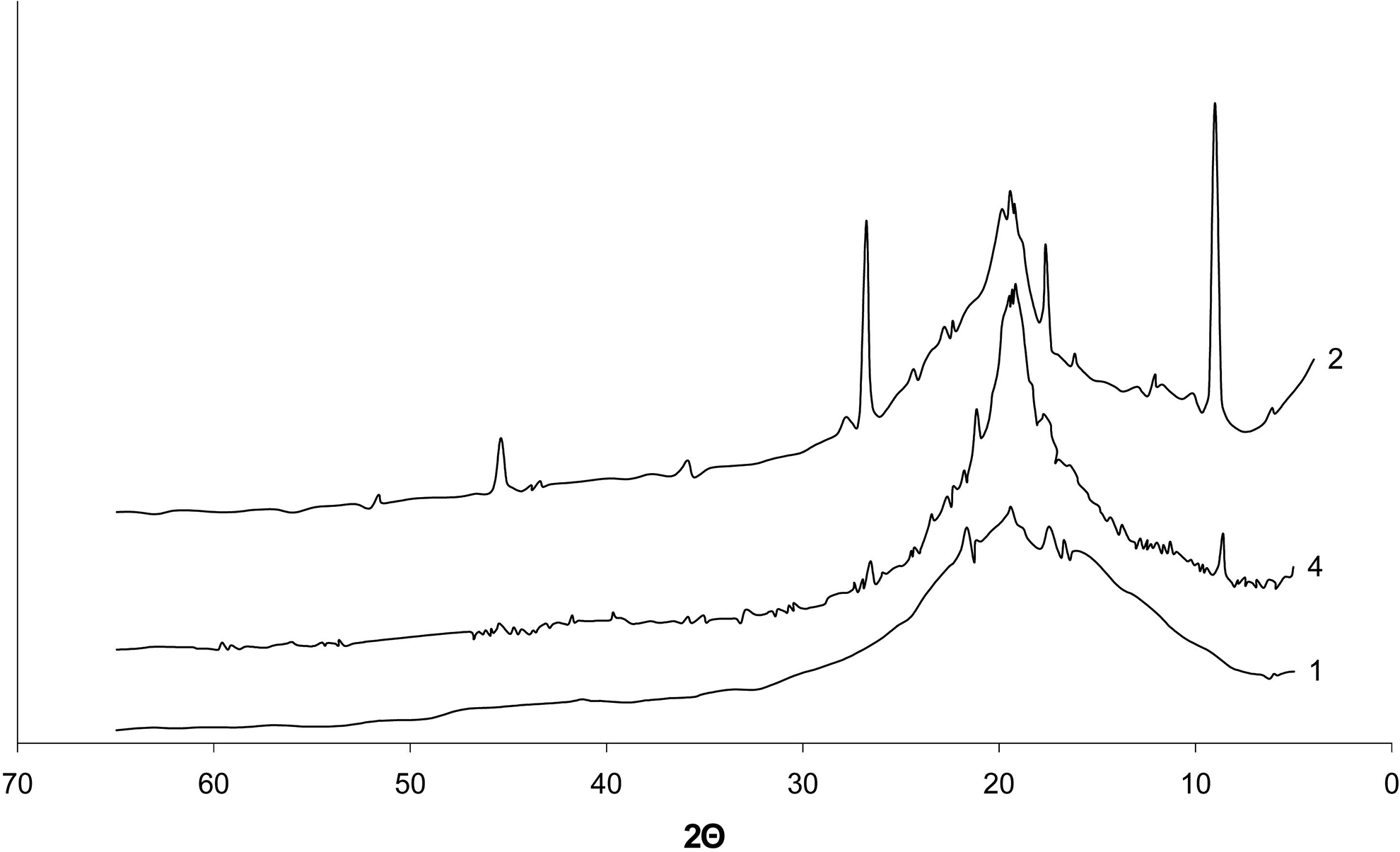
Diffractograms of the composites: 1—film based on the initial PVA, 2—film based on PVA:MPM = 12:1 composition, dried at room temperature, 4—film based on PVA:MMT = 12:1 composition, dried at room temperature.
In the diffraction patterns of samples 2 and 4 (Figure 4), in contrast with the initial PVA, crystalline reflexes are present at 2θ = 8.6°, 19.7°, and 26.8°, which are characteristic of MMT. 31 At the same time, the peaks of crystalline reflexes characteristic of PVA become less intense. The diffractogram of sample 2 (PVA:MPM = 12:1) is characterized by much more intense peaks of MMT crystalline reflexes in comparison with those for sample 4 (PVA:MMT = 12:1). This indicates that PVA macromolecules get into the interlayer space of the intercalated MMT, causing the peaks of the crystalline reflexes to be narrower and less intense at 2θ = 19.4°, 20.1°, and 22.7°. At the same time, the DSC results show that the melting point of samples 1 and 2 is the same (Table 1). For sample 4, the peaks intensity of crystalline reflexes at 2θ = 19.4°, 20.1° decreases but a single peak at 2θ = 19.5° appears. This demonstrates the formation of a new crystalline structure with a higher melting point, which is confirmed by the DSC analysis (Table 1).
Conclusions
Depending on composition and reaction conditions, the structure and thermal characteristics of nanocomposites based on PVA and MMT intercalated with PVP were investigated using XRD analysis and DSC. It was established that the modification of PVA by intercalated MMT reduces the degree of crystallinity of the resulting nanocomposites but significantly increases their thermal stability. The composites with unmodified MMT, heat-treated at 110°C, have the highest degree of crystallinity. Under an ultrasound, intercalated MMT is completely distributed in a PVA solution to form a monocrystalline structure, as evidenced by the absence of double melting peaks on the DSC curves. The composites with intercalated MMT are characterized by a 21°C higher heat resistance and a lower weight loss compared to the initial PVA (temperature of PVA thermal-oxidative degradation is 237°C) and to composites containing nonintercalated MMT. Nanocomposite thermal treatment at 110°C does not practically affect their thermal stability.
Films based on PVA with modified MMT, cross-linked at 110°C in the presence of 5 wt% of acrylic acid and 0.5 wt% of initiator, have a homogeneous cross-linked structure formed due to the chemical and physical cross-linking.