Abstract
In this article, polyhedral oligomeric silsesquioxane (POSS)-containing aliphatic polyester composites were synthesized using condensation polymerization. Firstly, a series of polyesters have been prepared from malonic acid, succinic acid, adipic acid, sebacic acid (diacids), and 1,4-butanediol. On the other step of this study, polyester composites were synthesized between 1,2-propanediolisobutyl POSS (POSS-Diol) and diacid. For comparison, the same reaction was repeated in the presence of 1,4-butanediol, POSS-Diol, and diacids. The resulting samples were characterized by proton nuclear magnetic resonance, X-ray photoelectron spectroscopy (XPS), and Fourier transform infrared spectroscopy. Thermal behaviors of molecules were analyzed using thermogravimetry (TG) analysis. Molecular weights of synthesized polymers were determined with gel permeation chromatography (GPC). The XPS results indicate that the POSS hybrid molecule was successfully incorporated into the polyester structure. The characteristic binding energies of organic silicium atoms and Si–O were observed at 102 eV and 104 eV, respectively. According to the results from GPC, POSS-containing polyester composites (POSS-Diol + diacid + 1,4-butanediol) have higher molecular weight. The number-average molecular weights (Mn) of polyester composites b1,b2,b3, and b4 are found to be 6800 g/mol, 7050 g/mol, 7200 g/mol, and 7250 g/mol, respectively. TG results show that the resistance to thermal degradation of POSS-containing polymer bonds is not strong as expected. The results support that the synthesized POSS-based polyesters can be used as high-performance commercial polymers.
Keywords
Introduction
Most of the recent studies have focused on the preparation of organic/inorganic hybrid materials with excellent properties using inorganically developed nanoscale substances such as carbon nanotubes, graphene, and polyhedral oligomeric silsesquioxanes (POSS).1–5 POSS, a class of unique inorganic compounds having a specific nanostructure, can be incorporated into polymer structures to produce hybrid materials with superior properties.6–9 The general formula of POSS is showed with (RSiO1.5) n , (n ≥ 6). The “ane” tag symbolizes the hydrocarbon group R which could be hydrogen or any alkyl, alkylene, aryl, arylene, or organofunctional derivative of alkyl, alkylene, aryl, or arylene groups. Among the POSS types, octa-silsesquioxanes (R8Si8O12) have been mostly investigated; they consist of a rigid, cubic inorganic silica core with a 0.53 nm side length and eight corner organic groups.10,11 Thus, the POSS molecules demonstrate some unique characters in the construction of hybrid polymers, as compared to other inorganic agents.12–14
Polyesters are one of the versatile synthetic polymers. They are widely used commercially as fibers,15,16 plastics,17,18 and coatings.19,20 Aliphatic polyesters are substantially resistant to oxidation by air or ozone under normal conditions, but are degraded rapidly by ammonia, hydrazine, warm alkali solutions, and primary or secondary amines, which cleave the ester linkage forming hydroxyl groups and the salt or amide derivatives of the carboxyl functionality. The addition of POSS hybride composite can improve the mechanical properties of the linear polyester such as strength,21,22 modulus,23,24 and rigidity25,26 and can reduce its flammability,27,28 heat evolution,29,30 and viscosity31,32 during processing. These developments have been investigated in a wide variety of commercial high-performance thermoset33,34 and thermoplastic composites.35,36 Spoljaric and Shanks 37 prepared POSS-functionalized Bol-torn-thermoplastic polyurethane blends and the results showed that the treated dendritic polymers enhanced its thermal stability and thermomechanical properties. In another POSS-containing thermoplastic composite study, McMullin et al. 36 developed thermoplastic biodegradable elastomers which have low modulus (<15 MPa), low melt processing temperatures, high strain to failure, and potential for controllable degradation.
In this article, POSS-containing composite polyesters were synthesized using 1,2-propanediol isobutyl POSS with two functional groups (POSS-diol), 1-4 butanediol and diacid monomers at different chain lengths. Traditional condensation polymerization method was used for this study. Considering all former studies on POSS, this study will bring innovation to the literature both technically and synthetically. The promising applications of the synthesized composite polymers mentioned in this study will boost further studies in the laboratory and commercial application areas.
Experimental method
Materials
Aminopropyl isobutyl POSS (POSS-NH2) (≥97.0%, Hybrid Plastics, Hattiesburg, Mississippi, USA); dimethylformamide (DMF, anhydrous; ≥99.9%, Sigma-Aldrich, UK); dicyclohexylcarbodiimide (DCC; ≥99.9%, Sigma-Aldrich, UK)/4-(dimethylamino) pyridine (≥99.9%, Sigma-Aldrich, UK); 2,2-bis(hydroxymethyl)propionic acid (98%, Sigma-Aldrich); 1,4-butandiol (Sigma-Aldrich, 99%); p-toluenesulfonic acid monohydrate (Sigma-Aldrich, ≥98.5%); toluene (Sigma-Aldrich, anhydrous 99.8%), 1,2-propanediol isobutyl POSS (≥97.0%, Hybrid Plastics); methanol (Sigma-Aldrich, anhydrous 99.8%); malonic acid (98%, BDH, UK); succinic acid (≥98.0%; Merck, Germany); adipic acid (≥99.5%; Fluka, Switzerland); and sebacic acid (≥98.0%, Merck) were used.
Instrumentation
Proton nuclear magnetic resonance (1H-NMR) measurements were recorded in deuterated chloroform with Si (CH3)4 as the internal standard, using Varian AS-400 (400 MHz) instrument (Palo Alto, California, USA). Fourier transform infrared (FTIR) spectra were recorded on a PerkinElmer FTIR Spectrum One-B spectrometer (Waltham, Massachusetts, USA). Molecular weights were determined by gel permeation chromatography (GPC) instrument equipped with a Waters styragel column (HR series 2, 3, 5E) with tetrahydrofuran as the eluent at a flow rate of 1 mL min−1 and a Waters 410 differential refractometer detector (UK). X-Ray photoelectron spectroscopy (XPS) analysis was performed with a Thermo Scientific instrument to interpret the analysis results. The expected atoms in the synthesized condensation polymers are silicium, oxygen, and carbon. Thermogravimetry (TG) measurements of powder polymer samples were obtained on PerkinElmer Diamond TA/TGA (USA) from 25°C to 600°C at 10°C heating rate at a constant flow rate of 100 mL min−1 under nitrogen atmosphere. The sample weights for all the experiments were taken in the range of 8–10 mg.
General procedure for synthesis of traditional aliphatic polyesters
1,4-Butanediol and diacid monomers of different chain lengths were allowed to react with p-toluenesulfonic acid (p-TSA), which was used as the catalyst, in toluene at 110°C. The reactions were stirred for 36 h. The final products were precipitated in cold methanol and were dried at vacuum oven. The synthesized polyester types and monomer mole ratios are shown in Figure 1 and Table 1, respectively. Also, the abbreviations which are given in Table 2 have been used to express the types of polyesters.
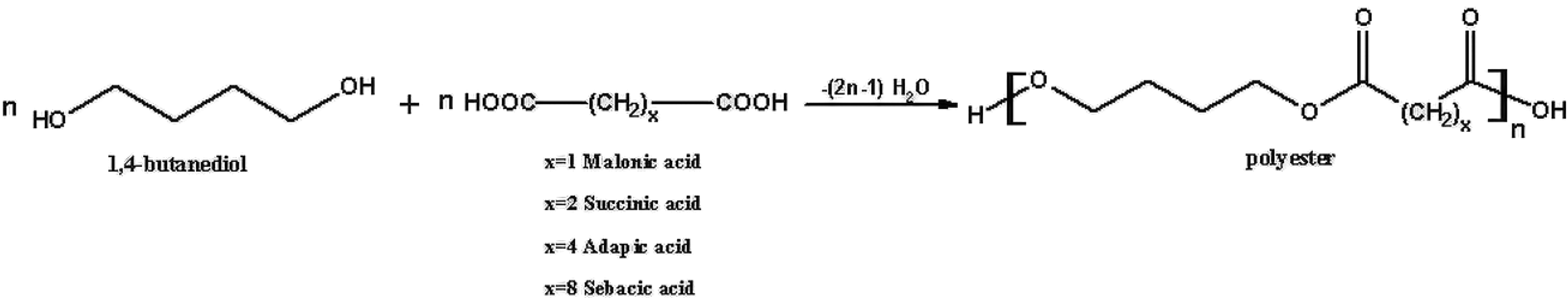
Synthesis of aliphatic polyester at different chain lengths.
The names and ratios of the monomers used in aliphatic polyester synthesis.

POSS: polyhedral oligomeric silsesquioxane; PTSA: p-toluenesulfonic acid.
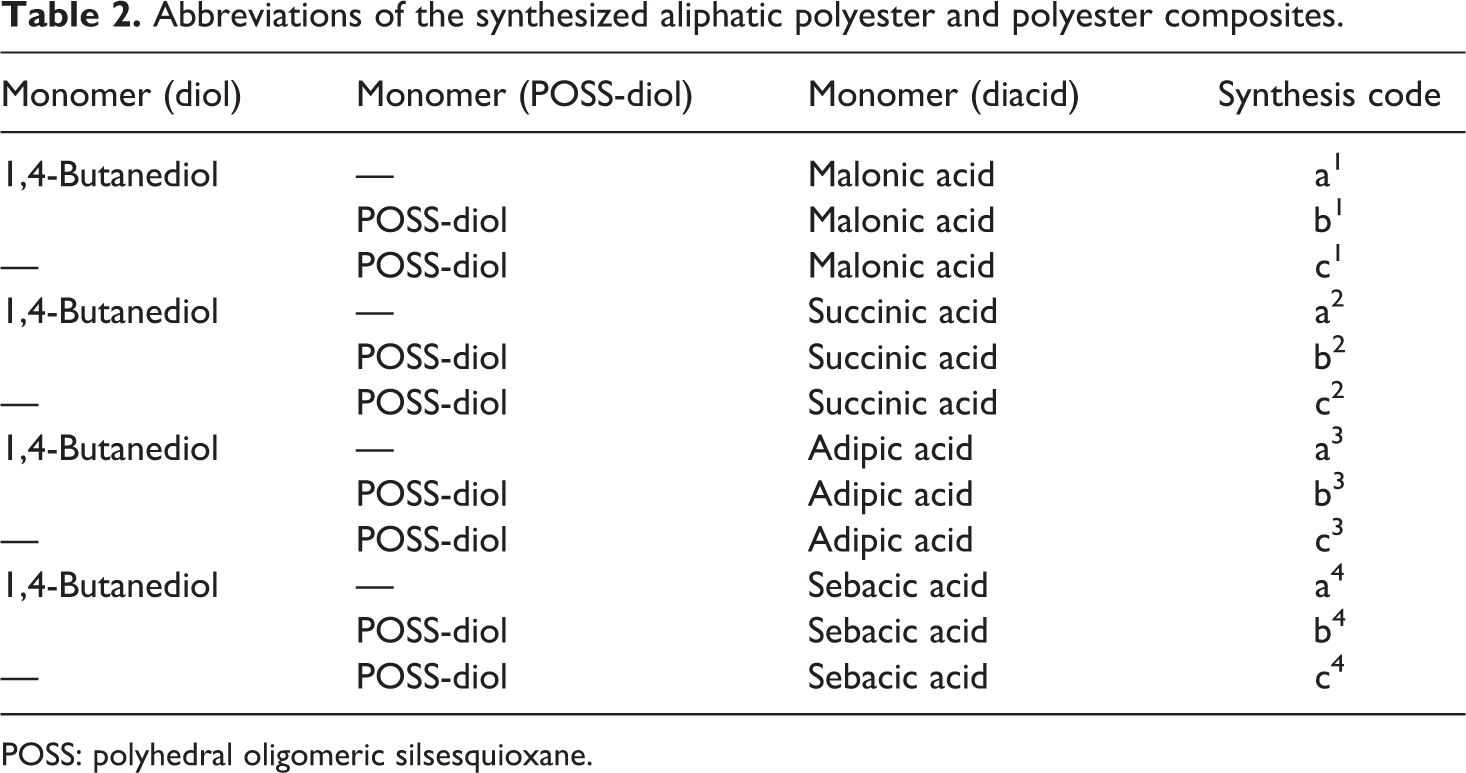
Abbreviations of the synthesized aliphatic polyester and polyester composites.
POSS: polyhedral oligomeric silsesquioxane.
General procedure for synthesis of POSS-containing polyester composites
1,4-Butanediol, 1,2-propanediolisobutyl POSS (POSS-diol), and diacid monomers were used for the synthesis of polyester composites. The reaction conditions and the mole ratios of the reactants are presented in Table 1. For the purpose of comparison, the same reaction model was repeated using only POSS-diol and diacid monomers. A schematic representation of the obtained polyester composites is given in Figure 2.
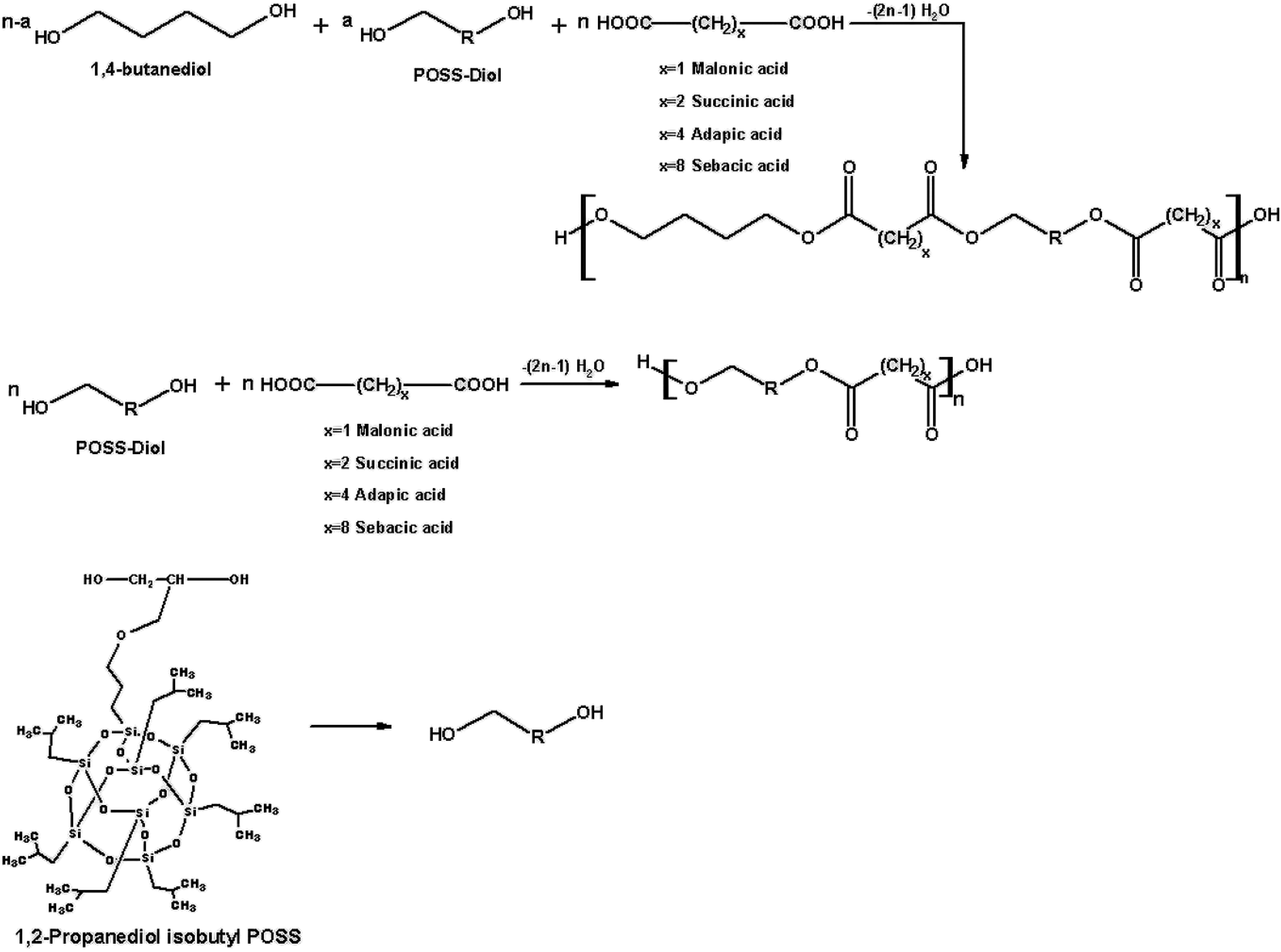
Synthesis of POSS-based aliphatic polyester composites.
Results and discussion
In this study, POSS-based aliphatic composite polyesters were synthesized at different chain lengths using the typical condensation polymerization method. For this purpose, firstly, POSS-free polyesters were synthesized. 1,4-Butanediol was allowed to react with malonic acida1, succinic acida2, adipic acida3, and sebacic acida4. POSS-containing polyesters were synthesized in the subsequent studies. 1,2-Propanediolisobutyl POSS (POSS-diol) and 1,4-butanediol were allowed to react with malonic acidb1, succinic acidb2, adipic acidb3, and sebacic acidb4, respectively. A similar study was repeated using POSS-diol and malonic acidc1, succinic acidc2, adipic acidc3, and sebacic acidc4. The structural characterization of these syntheses was supported by FTIR, 1H-NMR, and XPS. Thermal properties and molecular weights were investigated by TG and GPC methods, respectively.
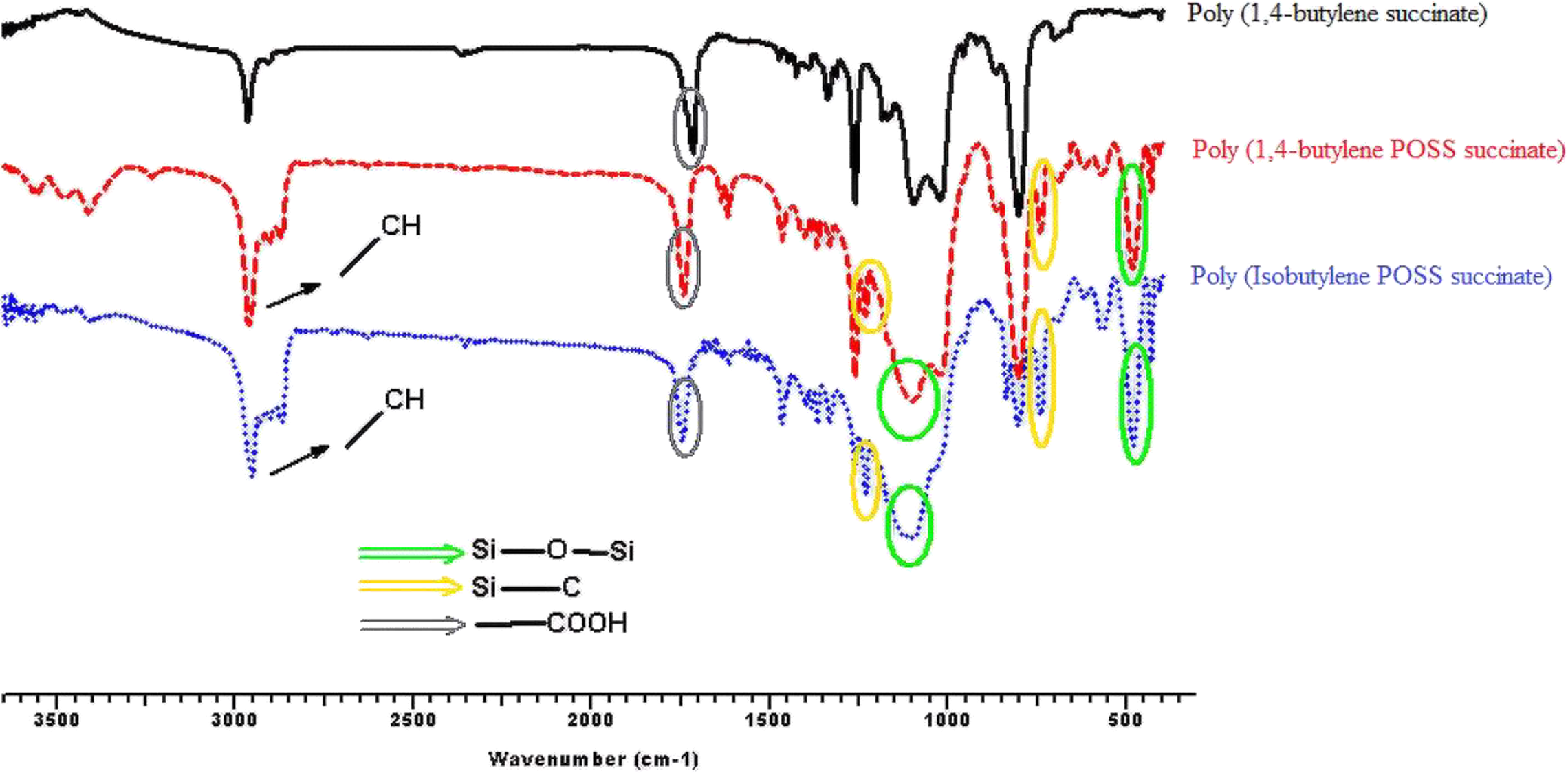
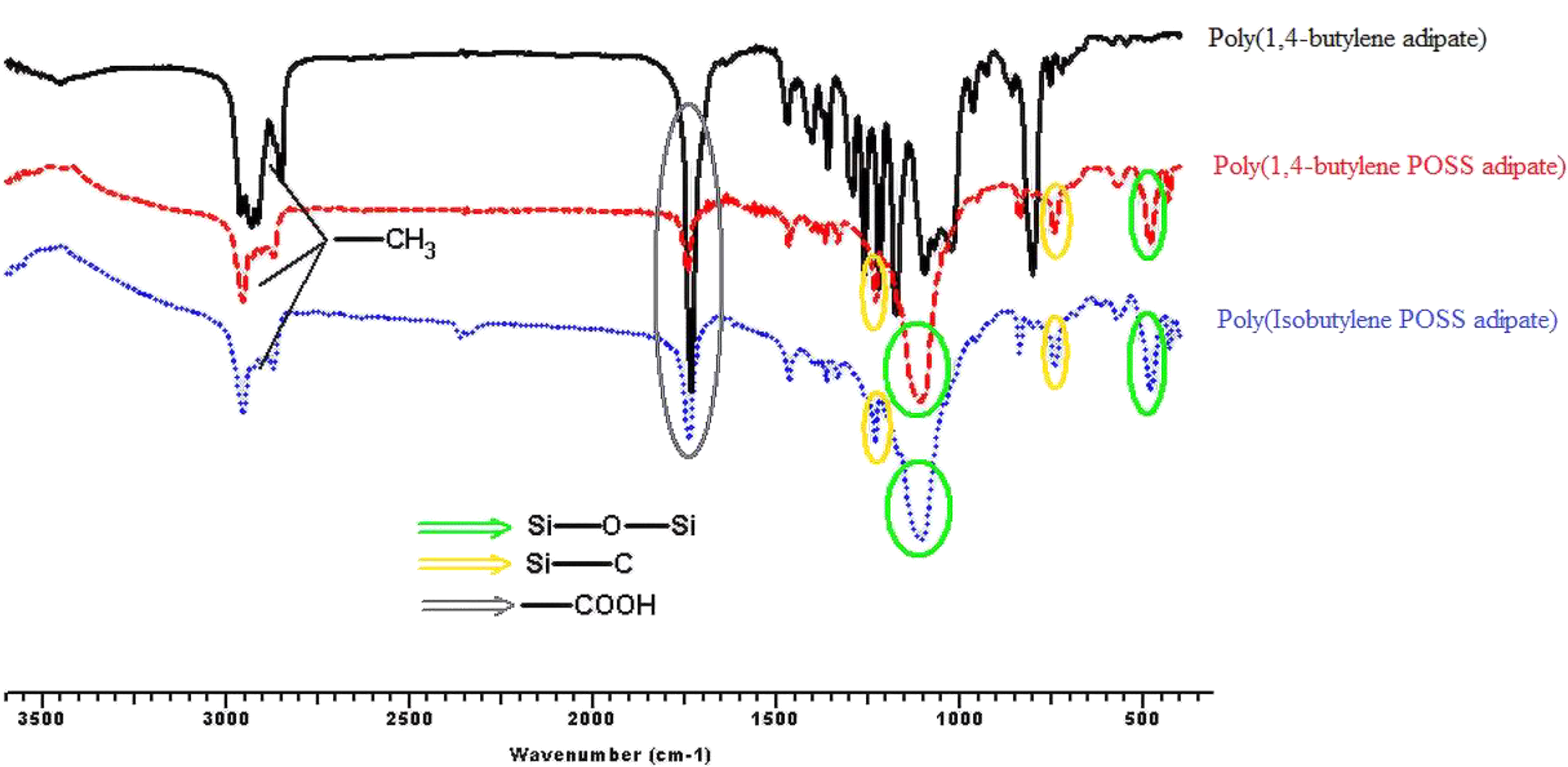
Figures 3, 4, 5, and 6 show the FTIR spectra of synthesis a1;b1;c1, a2;b2;c2, a3;b3;c3, and a4;b4;c4, respectively. According to the obtained FTIR results, the characteristic peaks of carbonyl group (–COOH) and aliphatic straight chain vibrations (–CH2–) in POSS-free polyesters are seen at 1750 cm−1and 2800–3000 cm−1, respectively. When the POSS hybride molecule is added to the structure, formation of characteristic peaks of Si–O, Si–O–Si, and Si–C are observed at 500, 1100, and 1200/700 cm−1, respectively. C–O–C ester stretching for all the syntheses appears at 1240 cm−1. Also, specific stretching peaks were marked on all the FTIR figures.
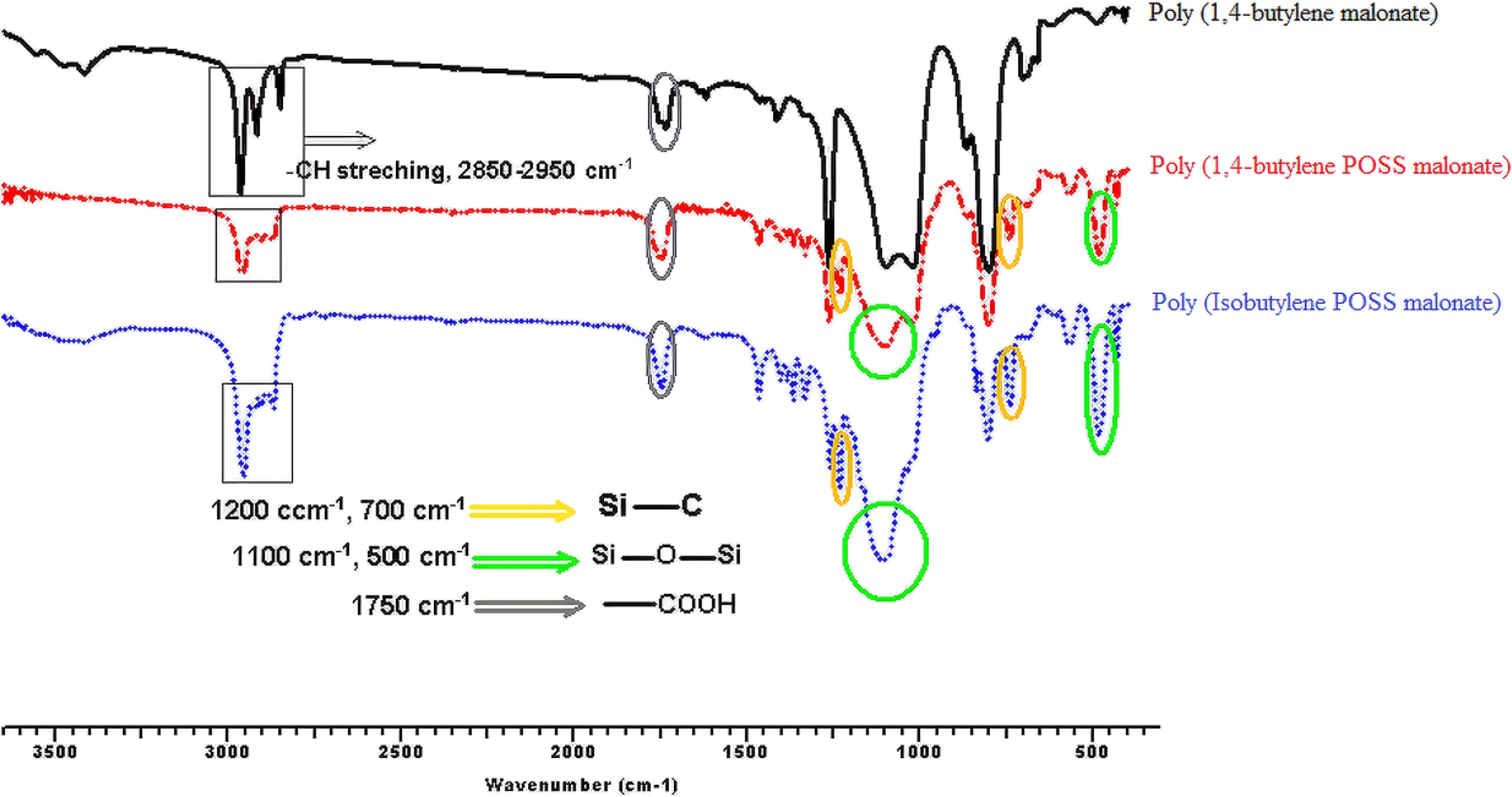
FTIR spectra of synthesis of a1;b1;c1.
FTIR spectra of synthesis of a2;b2;c2.
FTIR spectra of synthesis of a3;b3;c3.
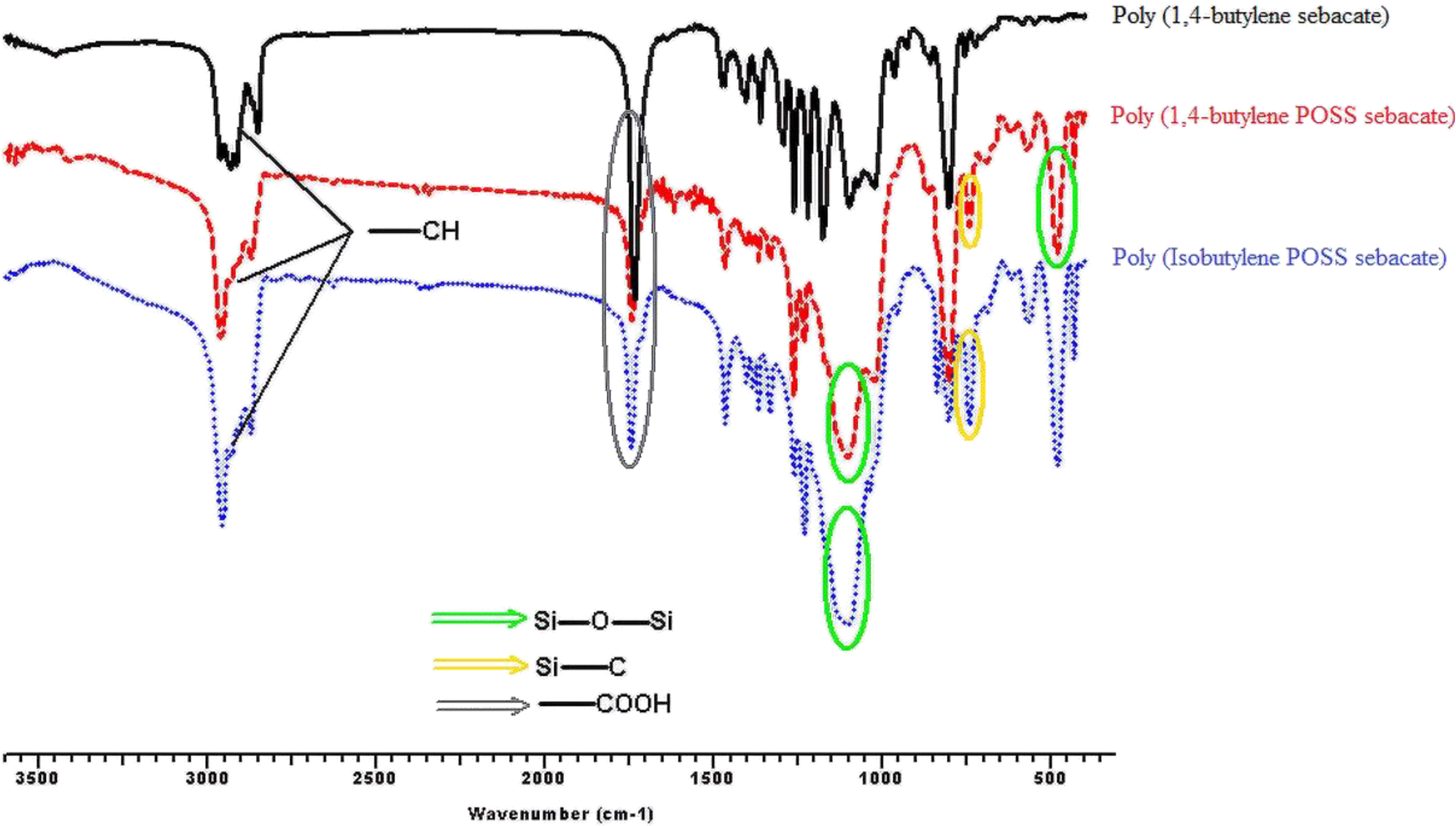
FTIR spectra of synthesis of a4;b4;c4.
1H-NMR of the synthesized poly(isobutylene POSS malonate) and poly(1,4-butylene POSS malonate) is shown in Figures 7 and 8, respectively. (All 1H-NMRs of the synthesized polymers and polymer composites are shown in Figures 7 to 14.) According to Figure 7, chemical shift value of the methylene group attached to silicium atom is observed at approximately 0.6 ppm. The peak of the methylene group between two carbonyl groups can be seen around 3.4 ppm. The characteristic –CH3 proton peak is observed at 1 ppm. The CH peak of the isobutyl group on the POSS molecule (–CH(CH3)2) is seen at 1.9 ppm; another CH peak bound to the oxygen on the repeated unit is observed at 5.1–5.2 ppm. The –CH2– peak on the POSS molecule can be seen at 2.1 ppm. According to Figure 8, when 1,4-butanediol was added to the resultant structure, the chemical shift value of the methylene group in the extended chain was determined at 4.2 ppm. Chemical shift values of the protons are shown in detail between Figures 7 and 14.
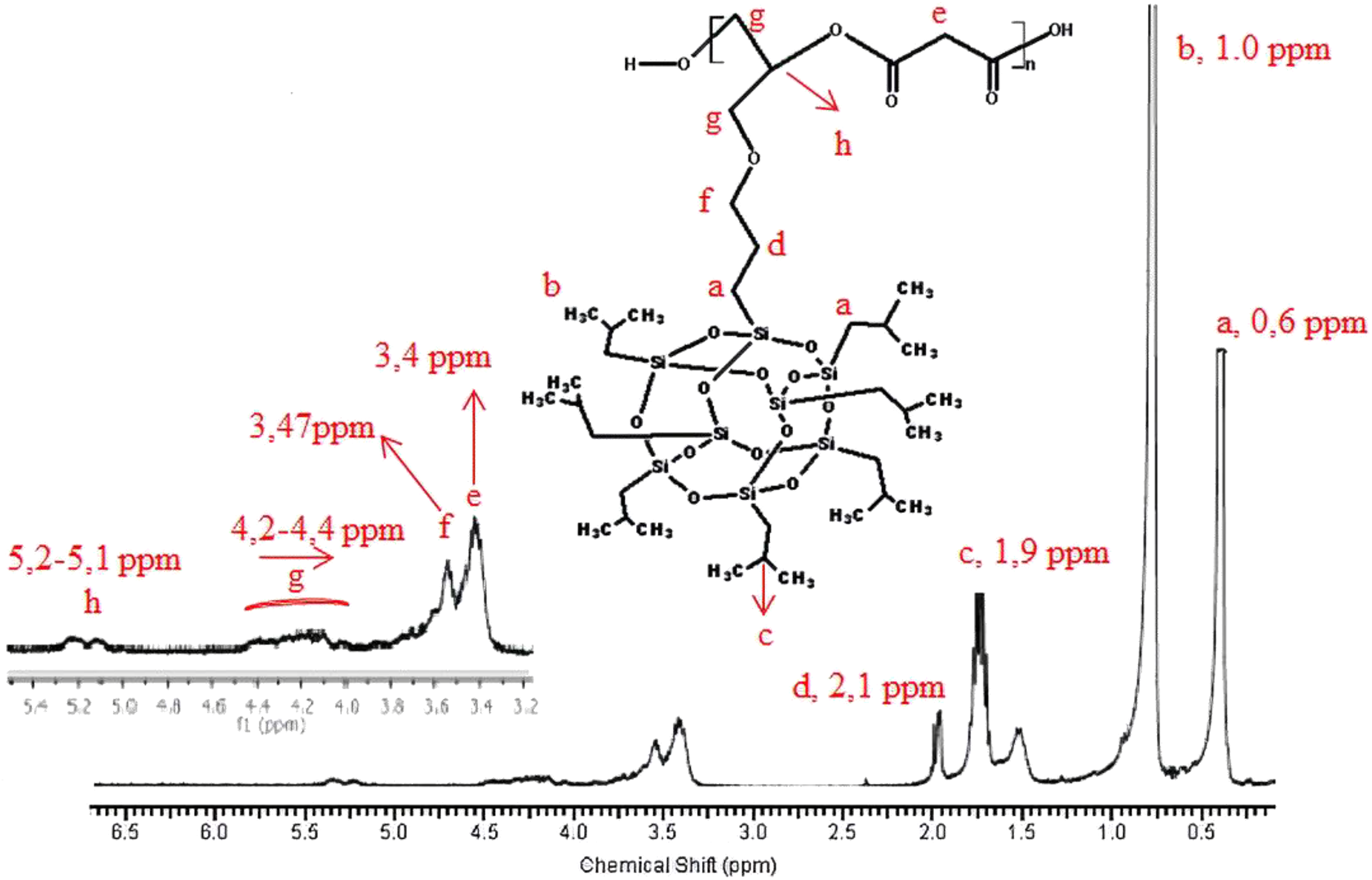
1H-NMR of poly(isobutylene POSS malonate).
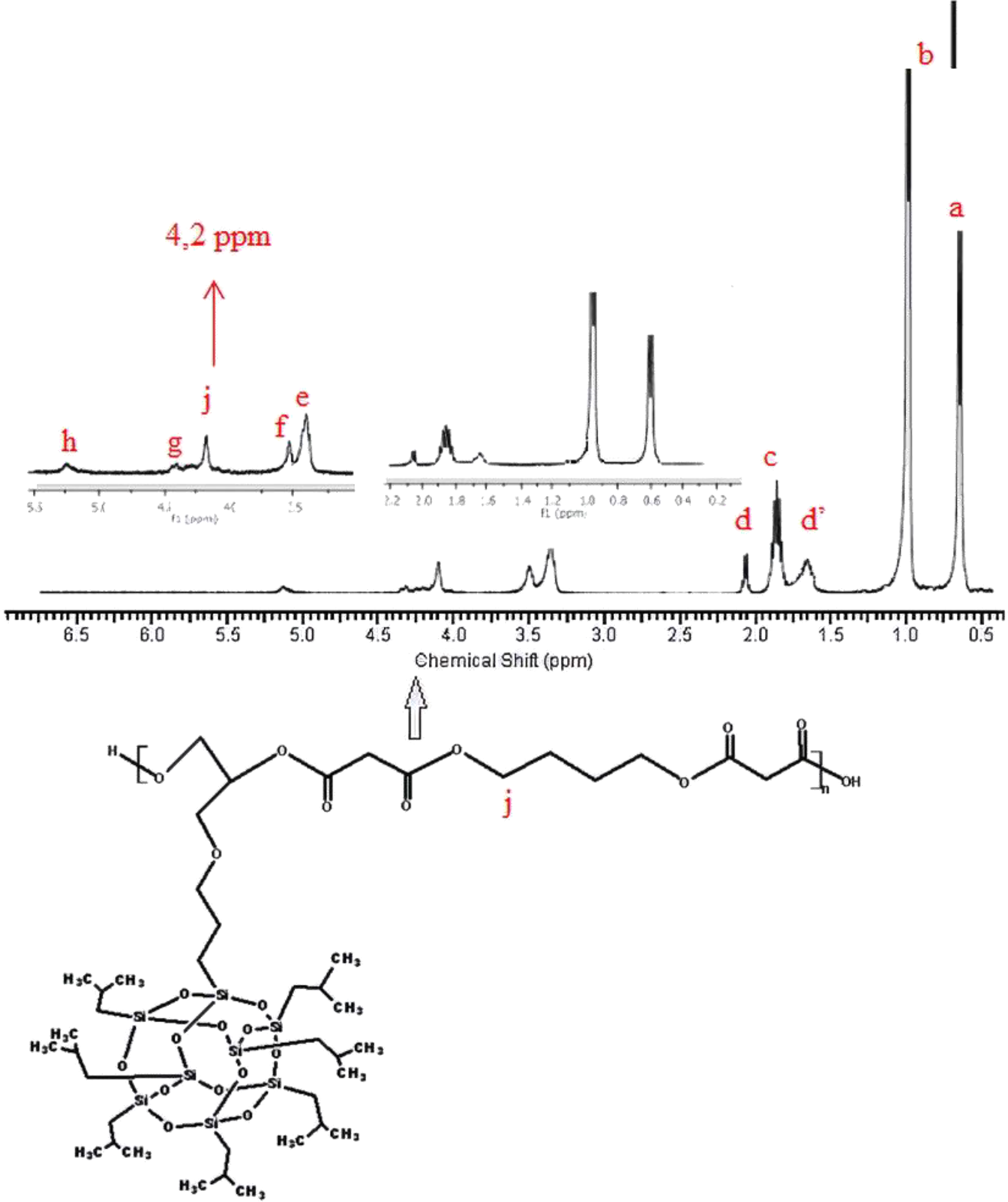
1H-NMR of poly(1,4-butylene POSS malonate).
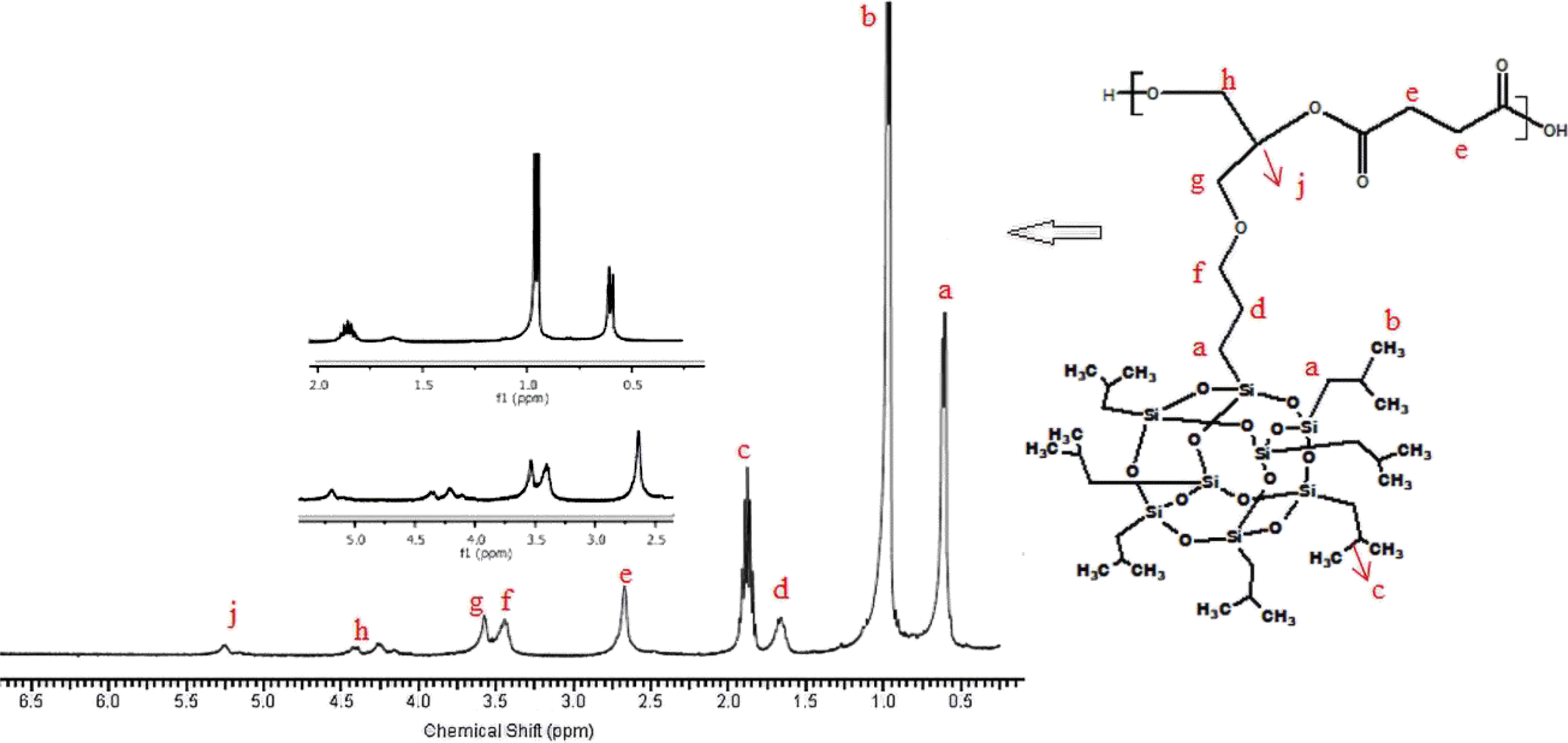
1H-NMR of poly(isobutylene POSS succinate).
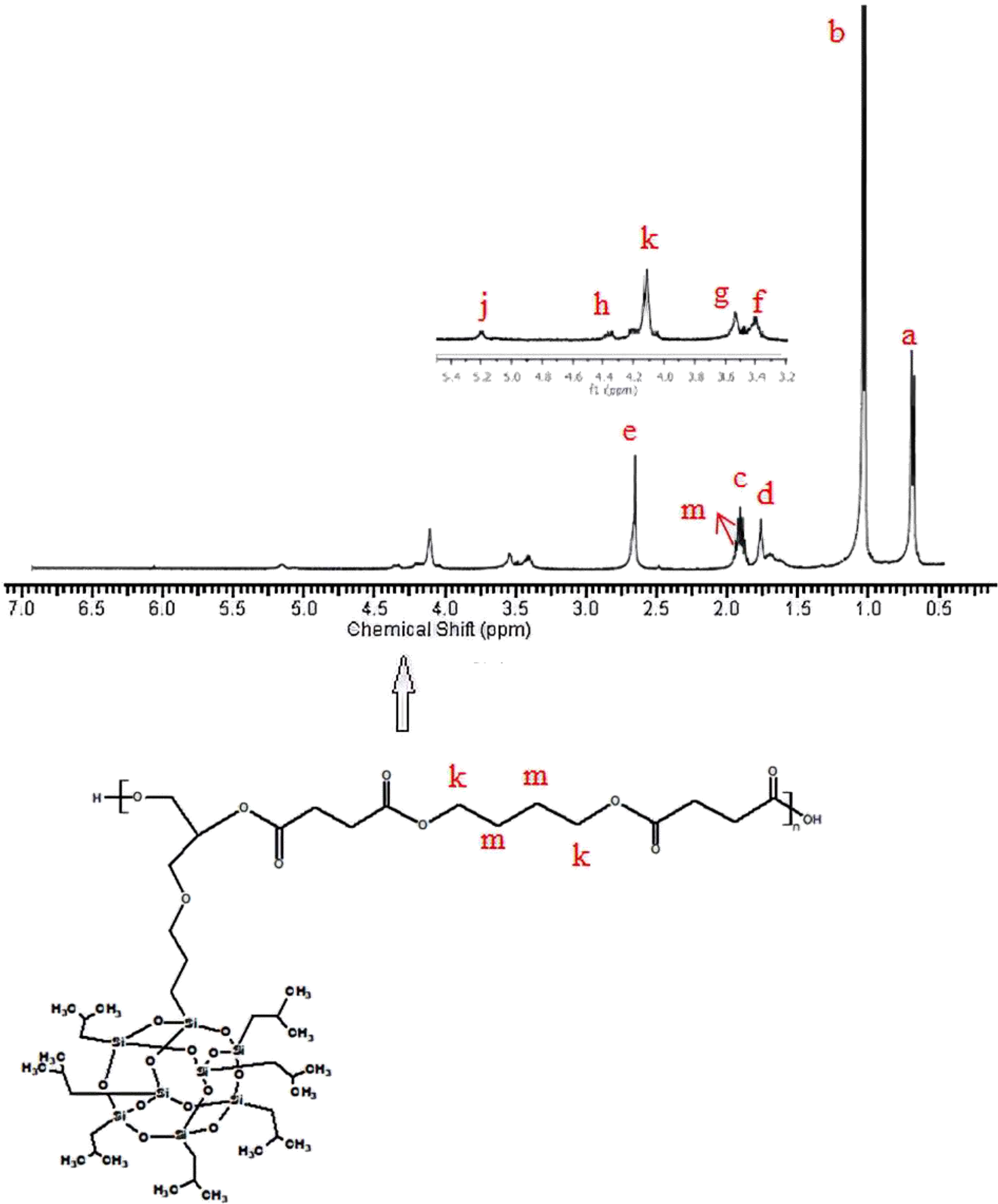
1H-NMR of poly(1,4-butylene POSS succinate).
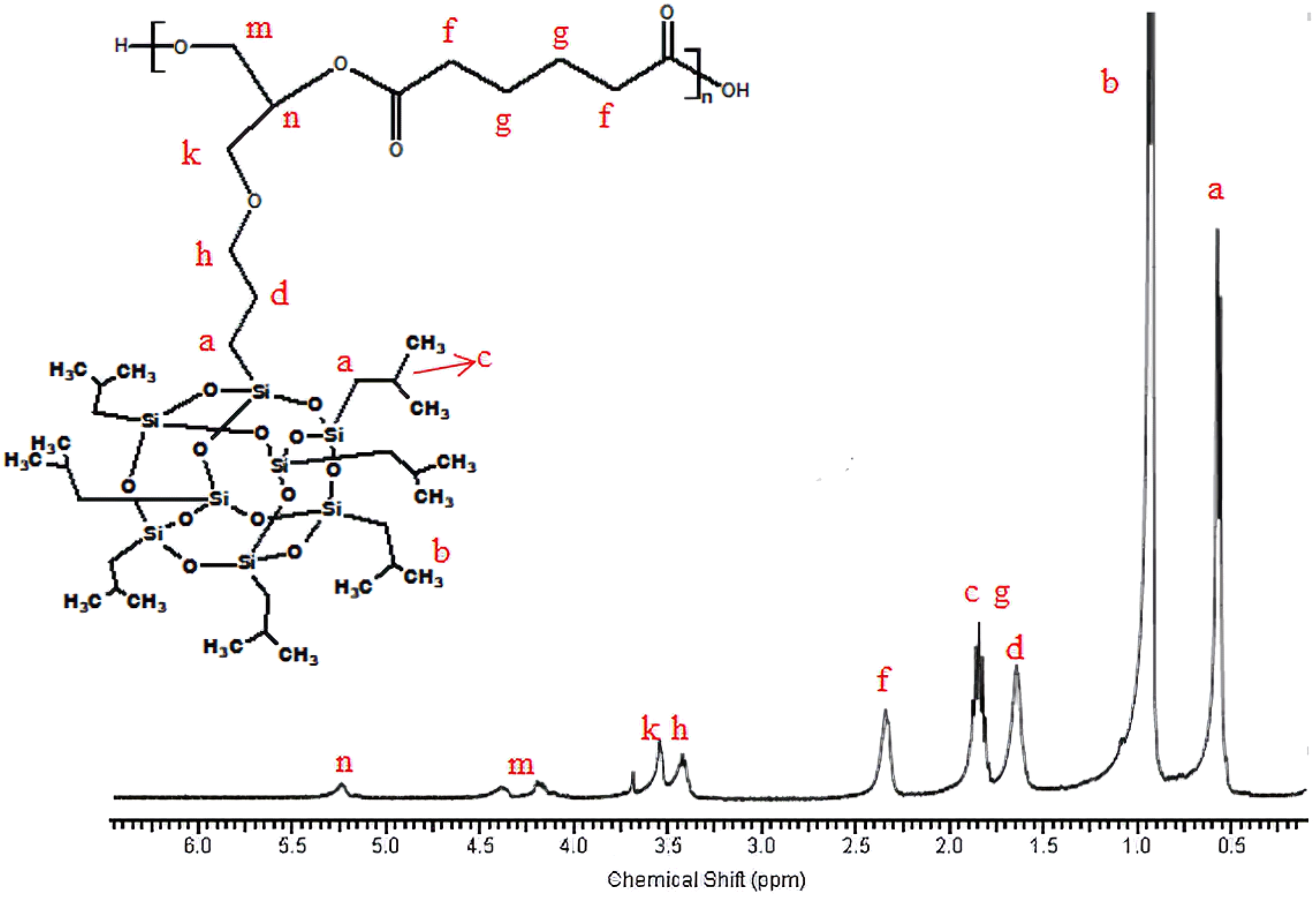
1H-NMR of poly(isobutylene POSS adipate).
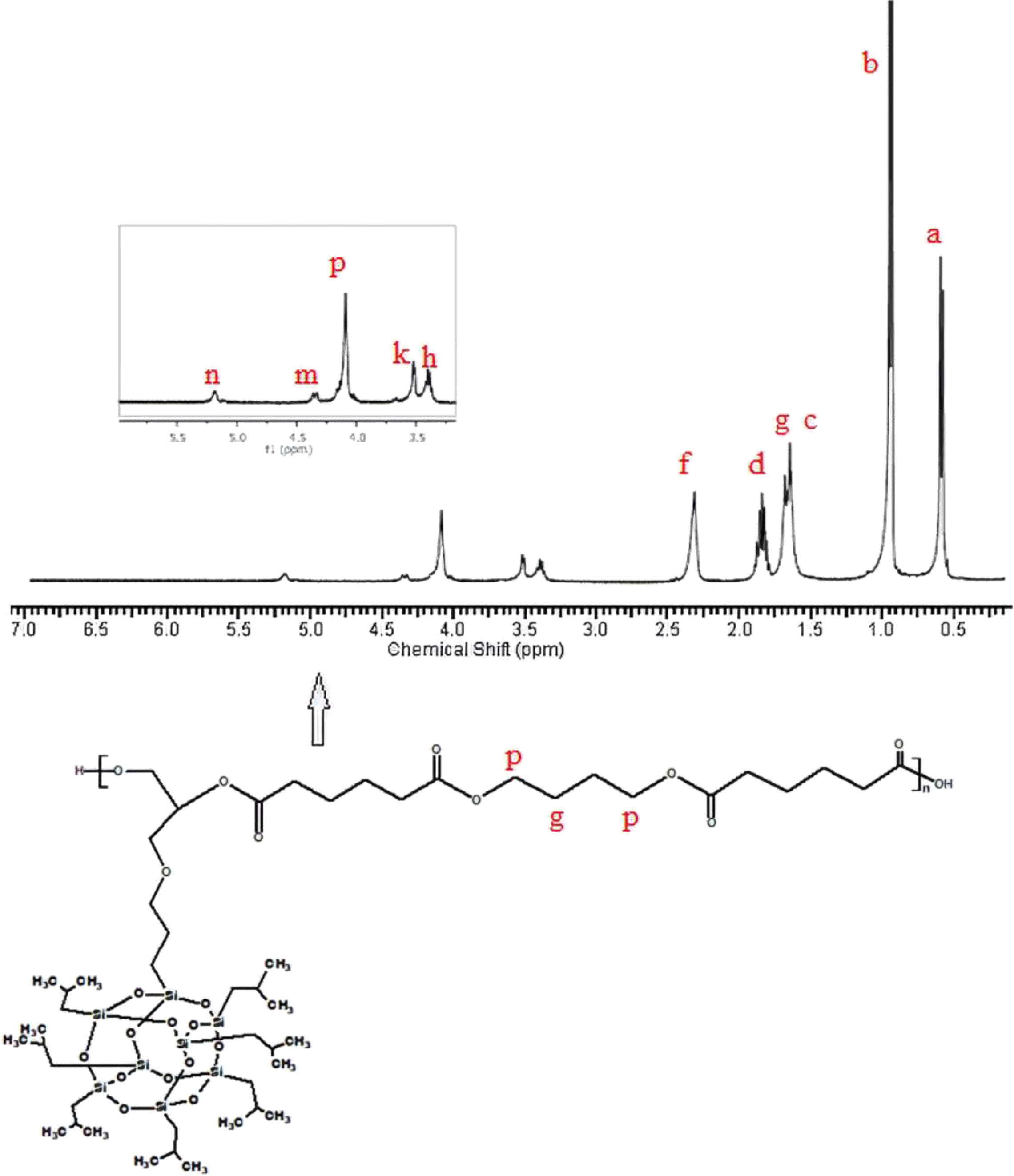
1H-NMR of poly(1,4-butylene POSS adipate).
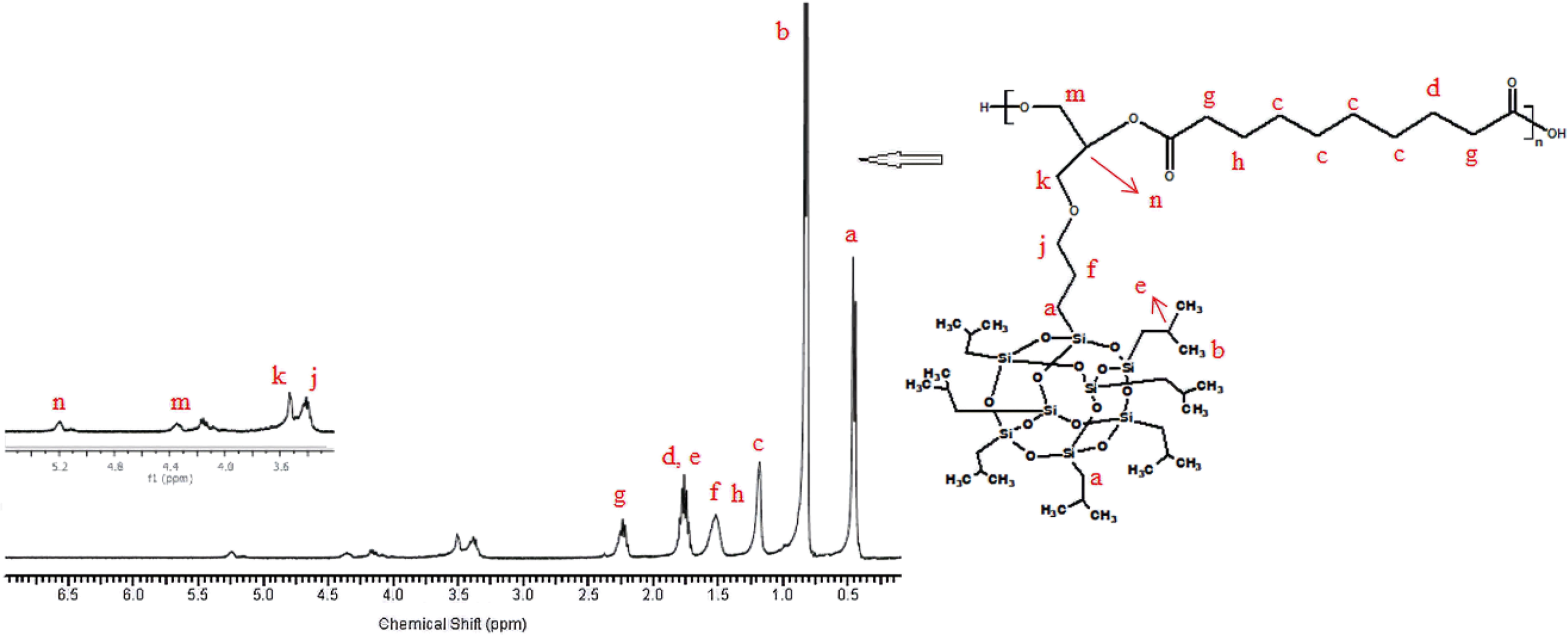
1H-NMR of poly(isobutylene POSS sebacate).
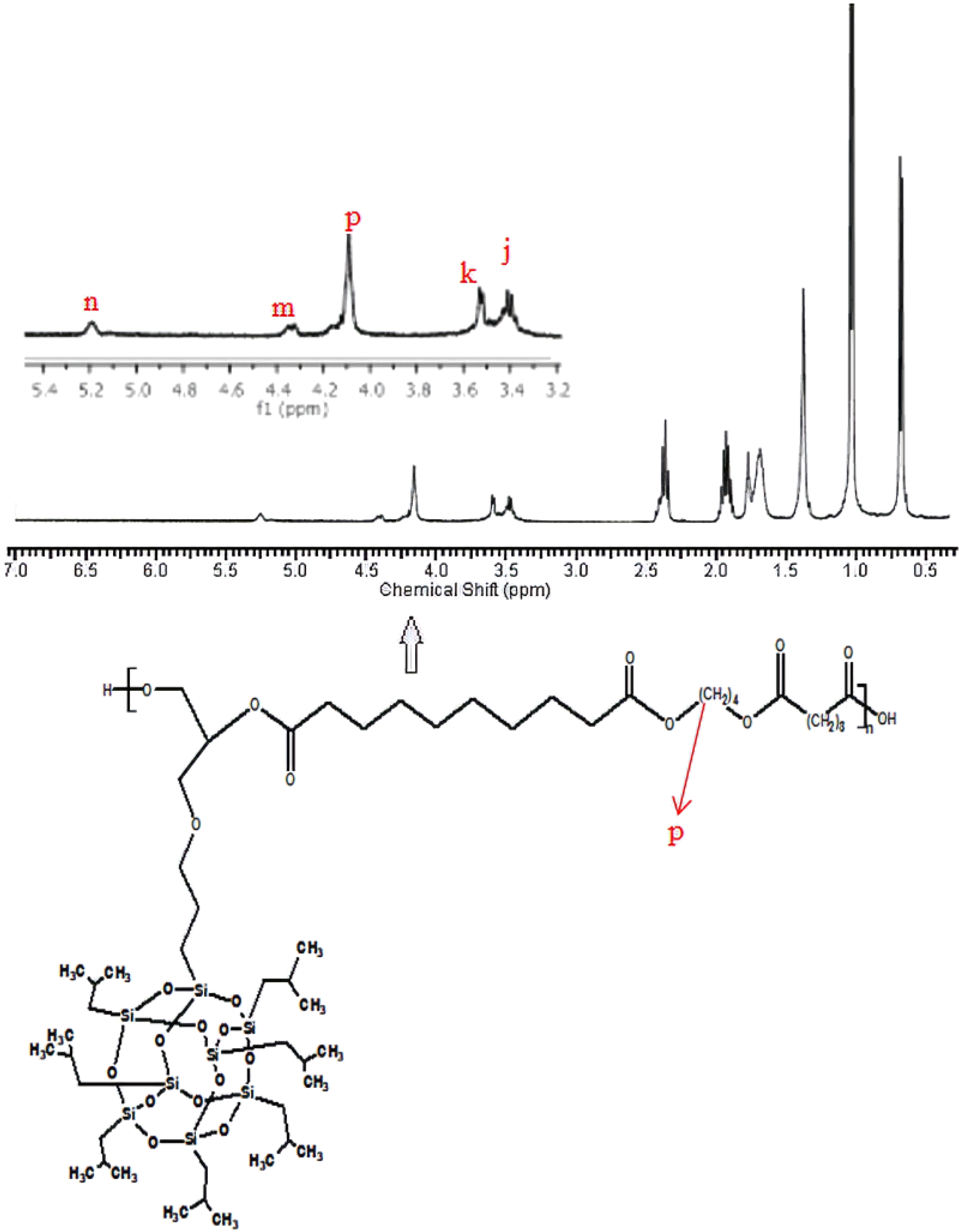
1H-NMR of poly(1,4-butylene POSS sebacate).
Thermogravimetic studies of the synthesized POSS-based aliphatic polyesters are illustrated in Figure 15. POSS-containing polymers and POSS-free polymers were compared with each other in these figures. When the TG curves are examined, it can be seen that the POSS molecule does not produce the expected thermal effect on the polyesters. This result may be because of two important reasons. One of them is the low molecular weight of the synthesized polyesters; the molecular weight of the polymers could not be raised sufficiently. Another important reason for the decrease in thermal stability may be the number of repeated units (segment). Because the POSS molecule has high molecular weight, it shows increased molecular weight of the polymers as expected. But when the segment number is calculated, it is seen that the number of repeating units of POSS-containing polymers is less. This decrease in segment number can cause the chains of POSS-containing polymers to break at lower temperatures.
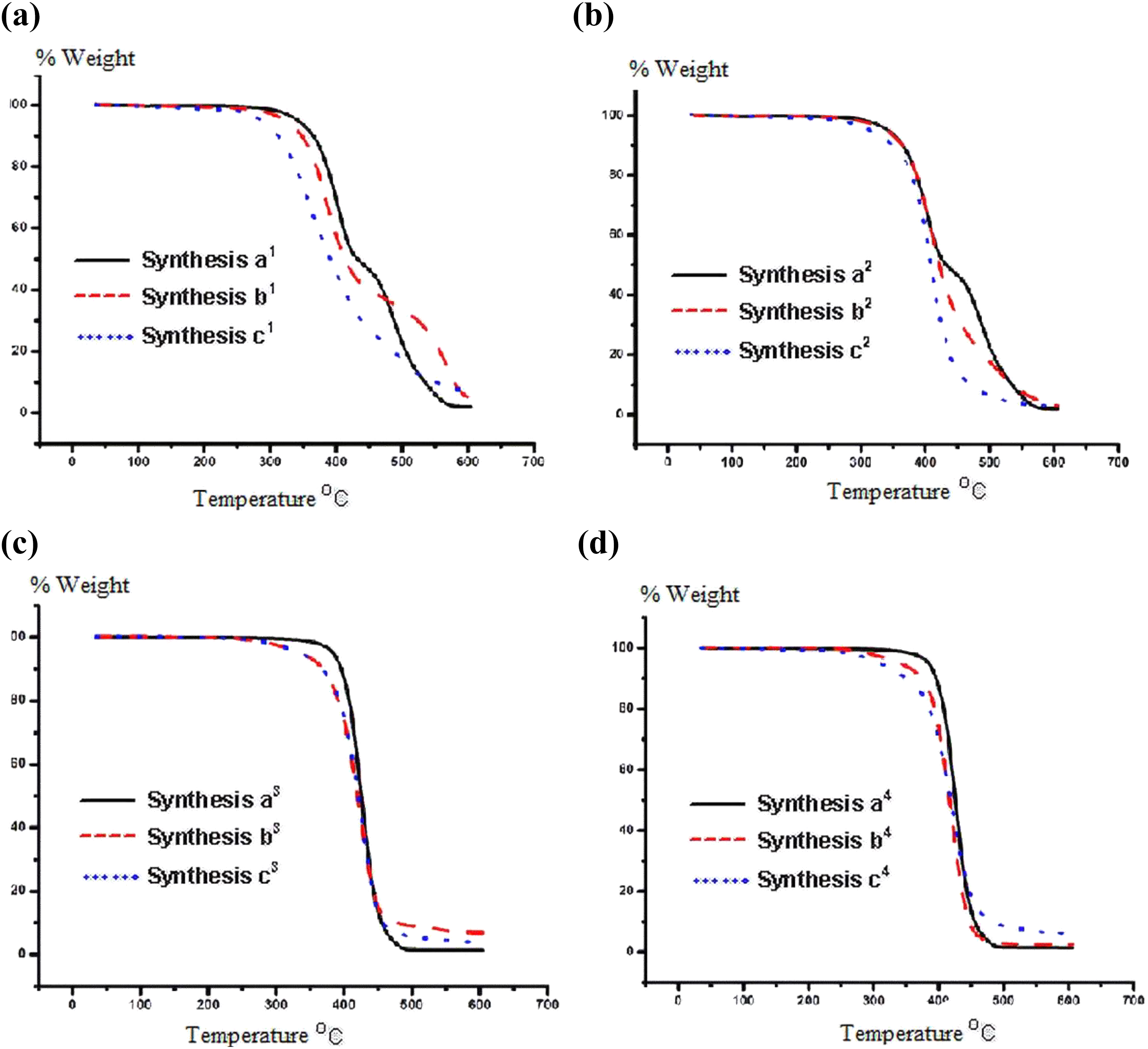
TG curves of synthesis of (a) a1;b1;c1, (b) a2;b2;c2, (c) a3;b3;c3, and (d) a4;b4;c4.
When Figure 15 is examined in more detail, it can be seen that thermal degradation of short-chain polyesters (a1, a2) is divided into two stages. The first degradation stageoccurs from 320°C to 425°C; during this step, breaking of the ester groups and small water escape may occur. The temperature in the second stage ranges from 425°C to 575°C. This degradation step could be the reason for the decomposition process of the methylene chain and –CH2OH. In accordance with the literature,38,39 long-chain polyesters (a3, a4) show a degradation step. As known, the POSS includes Si–C and Si–O bonds, and these bonds have higher energy than the C–C and C–H bonds. So, the thermal stability of the synthesized POSS-based polyester composites is expected to be higher, but there is no increase in the thermal stability of the products in this study. This result might be explained by the number of repeating units in the polymer. In addition, similar results are observed in this literature.29,40 There was no significant increase in the thermal stability of the product after having a high thermal stability polyester being modified with POSS.
Molecular weight dispersion of the synthesized condensation polymers was determined using GPC (Table 3). Based on the results obtained, POSS-free polymers have low molecular weights. When POSS molecule was added to the typical condensation polymers, it is observed that the molecular weight increases. Although this increase in the molecular weight is an expected result, this has not been effective in increasing the thermal stability of the synthesized polymers.
Molecular weight dispersion of the synthesized polyesters and polyester composites.
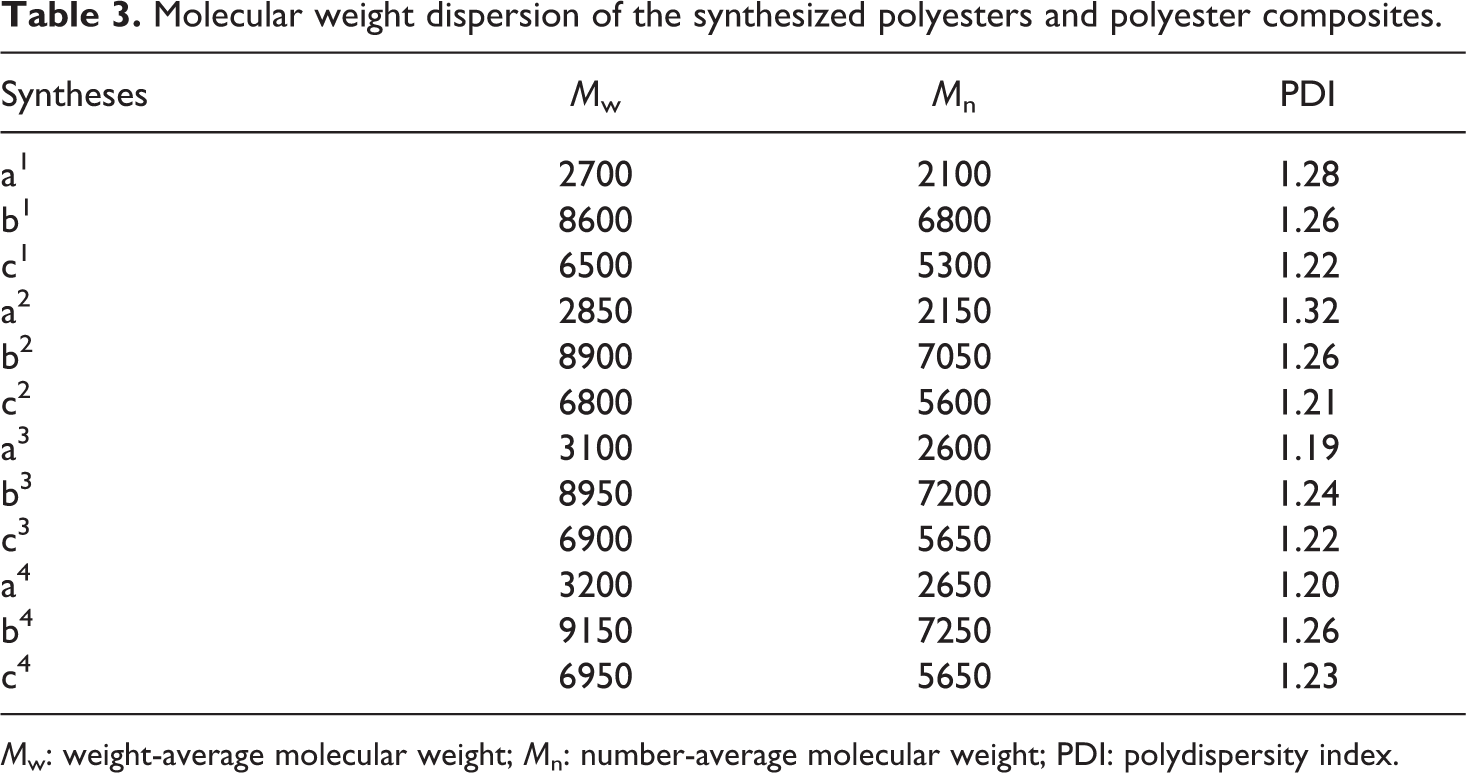
Mw: weight-average molecular weight; Mn: number-average molecular weight; PDI: polydispersity index.
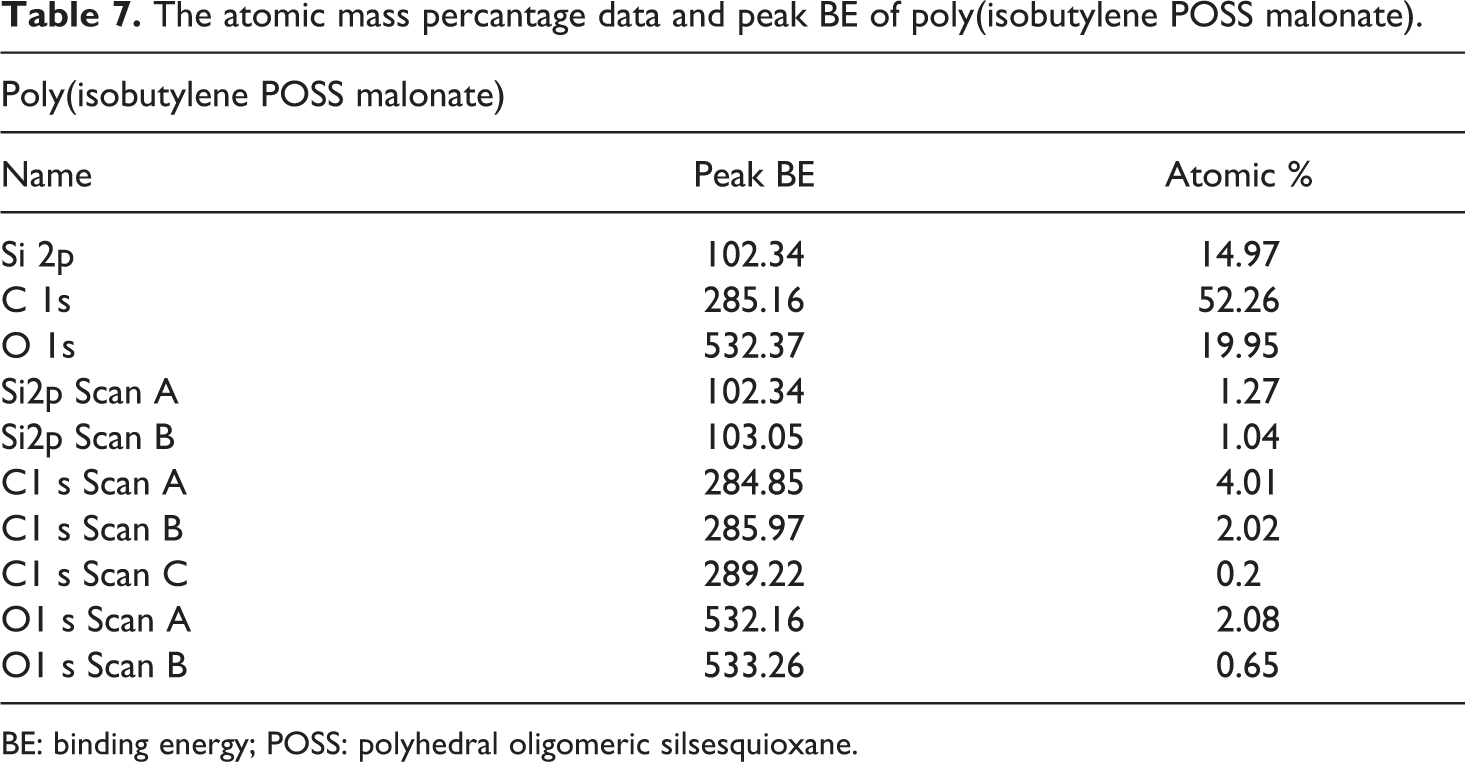
Synthesized POSS-based aliphatic polyester composites were supported by XPS studies. The binding energy values and the atomic mass percentage data obtained from these XPS analysis are provided between Table 4 and Table 7. The atomic mass percantage data and peak binding energy (BE) for the poly(isobutylene POSS adipate), poly(isobutylene POSS sebacate), poly(isobutylene POSS succinate), and poly(isobutylene POSS malonate) are presented in Tables 4, 5, 6, and 7, respectively. The binding energies and atomic mass percantage of the Si, C, and O atoms in all the synthesized polymers are close to the expected levels. According to the data on the tables, silicium atoms show at least two chemical environments (scan A and scan B). Binding energy peak of organic silicium atoms (scan A) was measured at approximately 102 eV. The other peak position (scan B) expresses the binding energy of Si–O, and these measurements were observed between 103 eV and 104 eV. XPS results support the successful synthesis of POSS-based aliphatic polyesters. The C1s spectrum typically has C–C, C–O-C, and O–C=O components. These three types of binding energy were seen in all the polyester composites synthesized in this study, as expected. Scan A, scan B, and scan C express the binding energies of C–C (284.5–285.5 eV), C–O–C (286–287.21 eV), and O–C=O (∼289 eV), respectively. According to the obtained XPS results, two chemical environments have been found for oxygen. One of the peak positions belongs to the binding energy of Si–O (532.16–532.7 eV). Scan B shows the organic C=O component and the measurement for it was observed between 533.2 eV and 533.3 eV. The data obtained are consistent with the previous studies.41,42
The atomic mass percantage data and peak BE of poly(isobutylene POSS adipate).
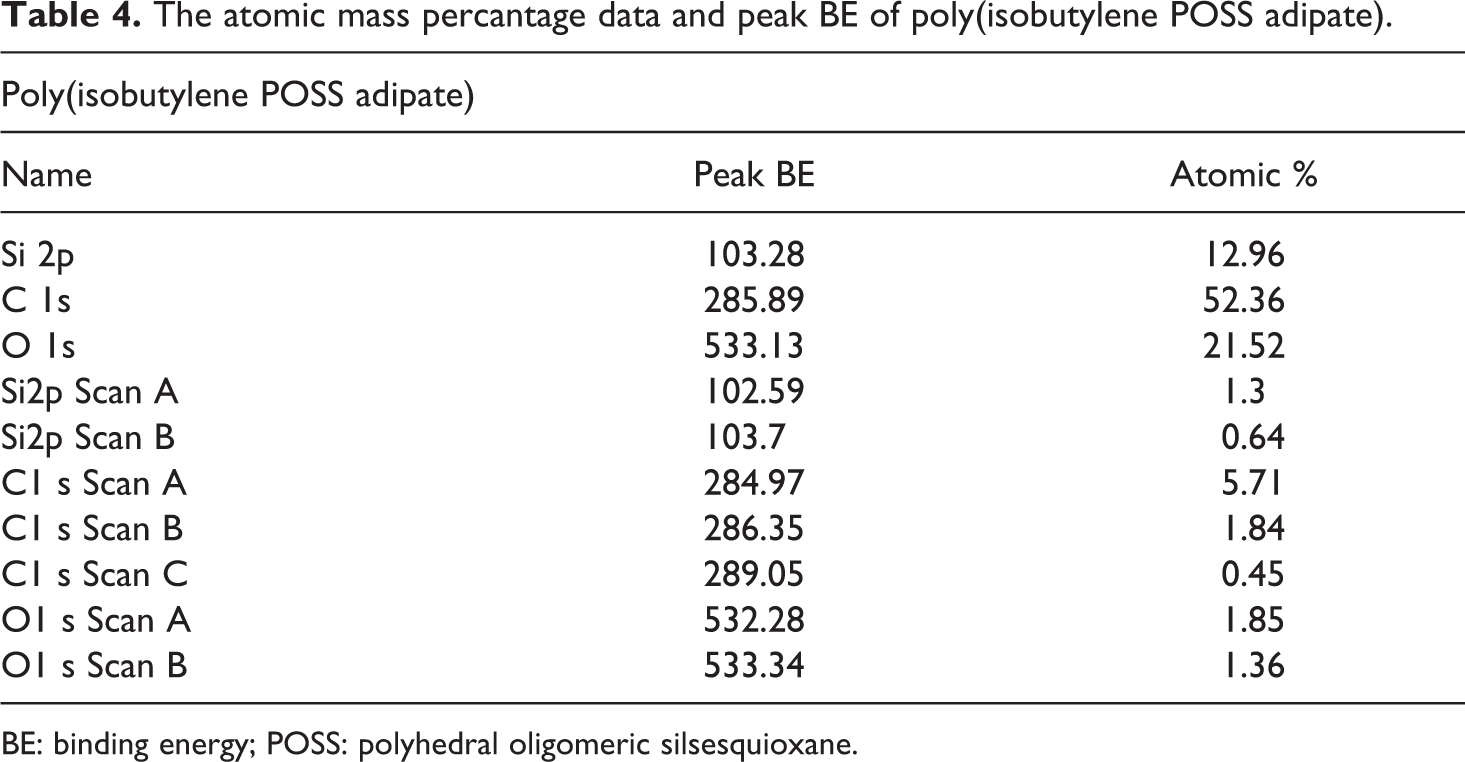
BE: binding energy; POSS: polyhedral oligomeric silsesquioxane.
The atomic mass percantage data and peak BE of poly(isobutylene POSS sebacate).
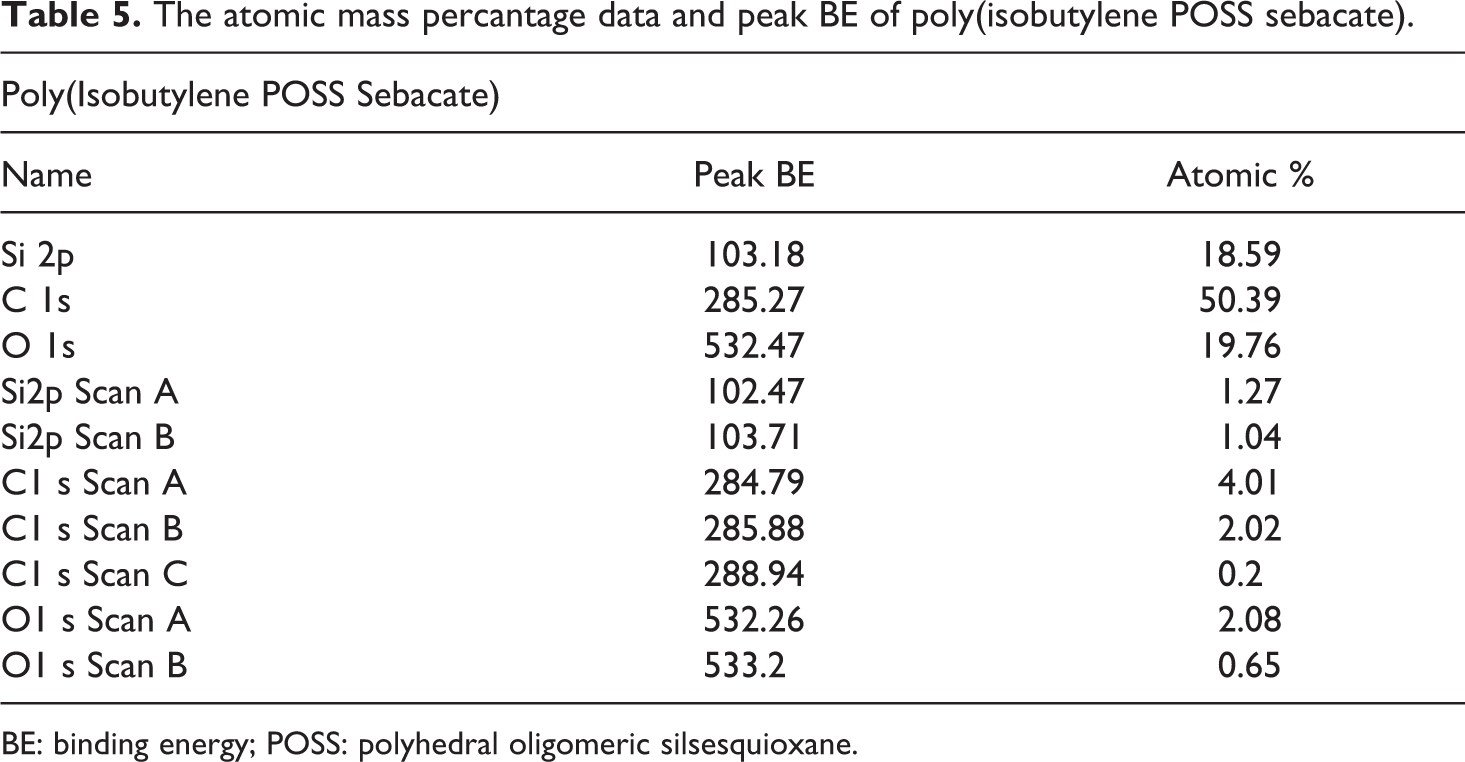
BE: binding energy; POSS: polyhedral oligomeric silsesquioxane.
The atomic mass percantage data and peak BE of poly(isobutylene POSS succinate).
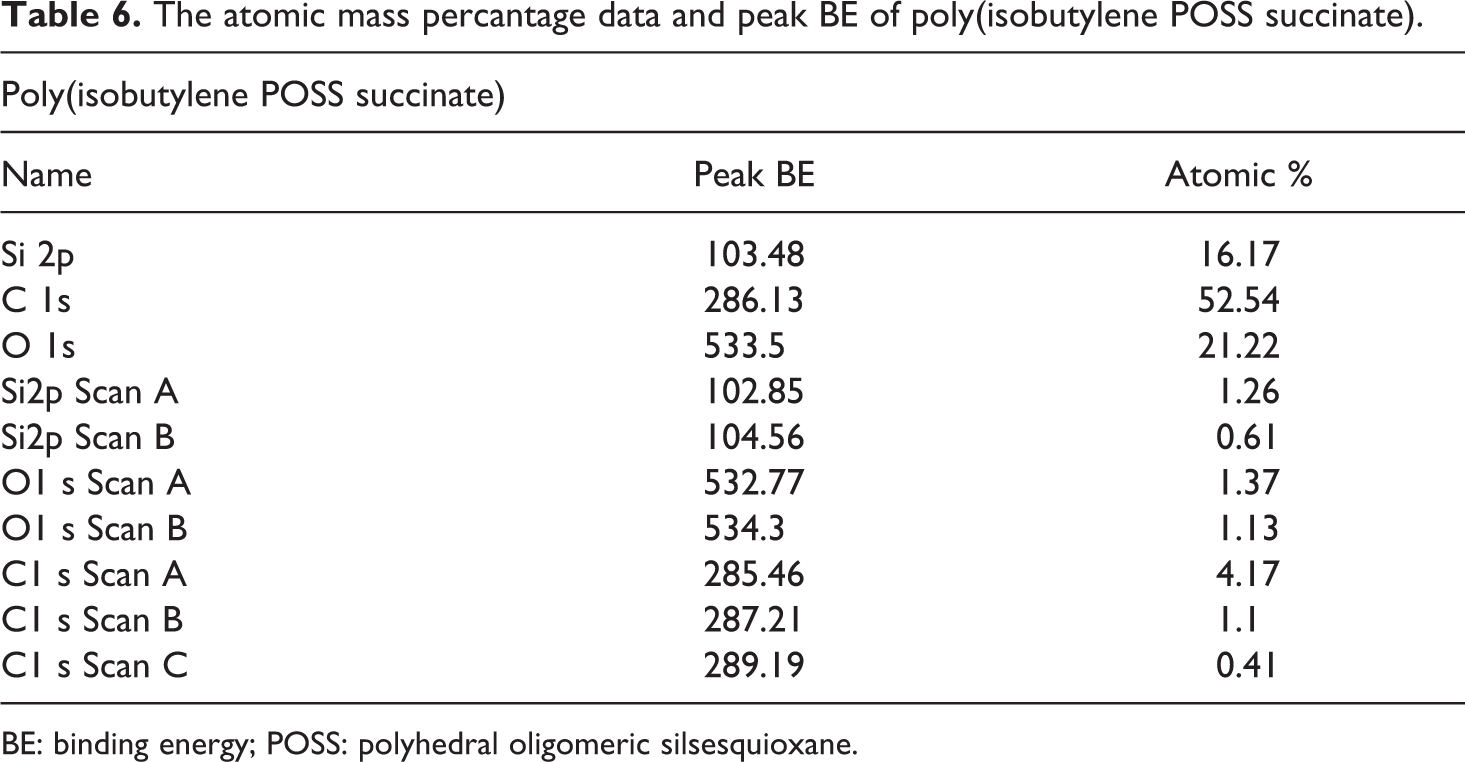
BE: binding energy; POSS: polyhedral oligomeric silsesquioxane.
The atomic mass percantage data and peak BE of poly(isobutylene POSS malonate).
BE: binding energy; POSS: polyhedral oligomeric silsesquioxane.
Conclusion
In this study, POSS-containing aliphatic polyesters were synthesized at different chain lengths using traditional condensation polymerization method. As stated in the introduction section, our synthetic strategy is based on designing a new composite molecule possessing both organic moiety and inorganic moiety. The results indicated that POSS-free polyesters have higher thermal stability than POSS-based polyesters. This may be due to lesser number of segments in POSS-based polyester. Therefore, chain breakage of the polymer composite was observed at lower temperatures. The XPS results show that the POSS molecule has been successfully incorporated into the condensation polymerization. The determined percentage of silicium atoms is theoretically compatible with the POSS molecule used in this experiment. According to the results obtained from GPC, the molecular weight of the polyester composites is not high as expected. The possible reason for this may be that the chains could not come side by side enough because of the bulky structure of the POSS molecule. Furthermore, NMR and FTIR results support the successful synthesis of this structure.
