Abstract
In this work, the properties of poly(methyl methacrylate) (PMMA)/alumina composites prepared by melt blending were evaluated. Two types of alumina were used: one, that was synthesized (ALS) by the combustion reaction method, and the other that is commercially available (ALC). Extrusion proved to be a good processing technique for obtaining PMMA/alumina composites. PMMA/alumina composites containing ALC presented higher alumina average particles size. The addition of alumina to PMMA substantially decreased the wear index and increased the scratch resistance. The mechanical properties of the composites containing ALS were similar to those containing ALC. The storage modulus (
Introduction
Among the various commercial thermoplastic polymers, poly(methyl methacrylate) (PMMA) stands out for its excellent transparency and stiffness being called “acrylic glass.” The advantage in relation to glass is related to its lower density, which facilitates the handling and reduces the costs with transport, besides the fact of not generating shrapnel when broken. However, when compared to glass, PMMA has high flammability, low resistance to abrasion and to scratch. Due to these limitations, different types of fillers1–13 have been used in an attempt to improve the properties of PMMA, forming PMMA matrix composites.
Martínez-Pérez et al. 14 studied the characterization and sliding wear performance of the PMMA-reinforced silicon dioxide (SiO2) nanoparticles. The authors observe that the addition of the SiO2 nanoparticles to PMMA decreased the coefficient of friction and that the main wear mechanisms were adhesion wear, plastic deformation, ploughing, and pitting.
Mohanty and Nayak 15 investigated the effect of nanoclays on the mechanical, thermal, and flammability properties of PMMA nanocomposites. The nanoclays used were Cloisite 30B (C30B), Cloisite 20A (C20A), and Bentone 109 (B109). The authors reported an increase in tensile modulus of about 35% with the addition of 5% of CL30B to PMMA. The thermal stability of PMMA was improved with the addition of the nanoclays. An improvement in fire retardancy of PMMA was observed with the addition of 5% of B109 nanoclay.
Singh et al. 16 evaluated the strength of PMMA composites panels bonded with polymeric adhesives containing nanosilica (nanosil) particulates. The authors observed that the panels pressed with nanosil dispersed styrene acrylonitrile (SAN) adhesives exhibited the highest impact strength.
Li 17 investigated the effect of nanosilica (SiO2) and titanium dioxide particles on the tribological (friction and wear) properties of PMMA/particle-reinforced composites. The authors observed a great strengthening effect with the addition of SiO2 and TiO2 particles to PMMA and that the composites exhibited excellent tribological properties.
Another filler used to modify the properties of PMMA is alumina. Alumina (Al2O3) is a ceramic material that has been used in PMMA matrix composites to improve abrasion and scratch resistance, surface hardness, modification of thermal properties, among other characteristics, because its density is not very high and it can maintain a low weight of the composite when used in low concentrations. There are some works in the literature that used alumina as filler in PMMA matrix composites.
Alzarrug et al. 1 evaluated the effect of different types of alumina (nanoparticles, whiskers, electrospun) on the mechanical properties of PMMA/alumina composites. The authors compared the properties of the PMMA composites containing electrospun alumina (synthesized by the authors) with the ones commercially available (alumina nanoparticles and alumina whiskers). The composites were prepared by in situ-polymerization and characterized by dynamic mechanical thermal analysis (DMTA) and nanoindentation. The authors reported that the mechanical properties of the composites containing the electrospun alumina were similar to that of the composites containing commercially available alumina and that the composites with alumina whiskers showed better mechanical properties than the one with alumina nanoparticles.
Ash et al.
18
investigated the glass transition temperature (
Hasan et al. 19 evaluated the effect of size and shape of alumina in the PMMA/alumina composites. The composites were prepared by in situ polymerization. The types of alumina used were spherical and whiskers nanoparticles. The authors observed that both types of alumina improved the mechanical properties and that better mechanical properties were obtained for the composites containing alumina whiskers.
Sezavar et al. 20 studied the effect of alumina nanoparticles on the fracture behavior of PMMA/alumina composites. PMMA and alumina were mechanically mixed and then, the PMMA/alumina composites containing different amounts of alumina were prepared by compression molding. The authors reported that the elastic modulus of the composites increased with the addition of alumina to PMMA whereas the tensile strength and elongation at break were not affected. The authors also reported the change in the deformation and fracture mechanisms of PMMA with the addition of alumina.
Stapountzi et al. 21 evaluated the effect of the alumina trihydrate (ATH) on the elastic modulus of PMMA/alumina composites. The authors use the commercially available PMMA/alumina composites sheets containing 33–49 vol % of alumina. The authors observed an increase in the modulus of about 35% when the amount of alumina was increased from 33% to 49%.
Laachachi et al. 22 investigated the effect of alumina in the thermal stability and flammability of PMMA. The composites were prepared by melt blending in an internal mixer and samples for the tests were prepared by compression molding. The authors observed that the thermal stability was substantially improved with the addition of alumina filler to PMMA. The flammability decreased with an increase in alumina content.
As can be observed, in most of the works, PMMA/alumina composites are prepared via in situ polymerization process, which uses solvents. The use of solvents can lead to health hazards and to cause environmental impact, besides being of high cost. In this work, the PMMA/alumina composites were prepared by melt blending PMMA with alumina in a twin screw extruder. The advantage of this process is that the PMMA/alumina composites can be prepared in large scale and is eco-friendly since no solvent is necessary. Therefore, the aim of this work is to evaluate the properties of PMMA/alumina composites prepared by melt blending in a twin screw extruder.
Experimental
Materials
Polymer (methyl methacrylate)—PMMA Acrigel® LEP100, MFI = 3.8 g 10 min–1 (230°C/3.8 Kg) (ASTM D-1238) provided by Unigel Plastics S/A (Brazil) was used as the polymer matrix. Aluminum nitrate nonahydrate (Al (NO3) 3.9H2O) and urea ((NH2) 2CO) from Vetec (Brazil) used for the synthesis of the alumina (ALS). Alumina trihydrate—Hydrogard-GP (ALC) supplied by Alcoa Alumínio S.A., Brazil.
Synthesis of the alumina (ALS)
The alumina used in this work (ALS), in order to compare with the commercially available, was synthesized according to the method reported by Freitas et al. 23 The alumina synthesis process was carried out by means of the combustion reaction between aluminum nitrate nonahydrate (Al (NO3) 3.9H2O), which is the source of the cations for the production of the metallic oxide, and the urea ((NH2) 2CO), which is the reducing fuel. For the production of 100 g, 71.42 g of aluminum nitrate and 28.58 g of urea were used. In order to carry out the combustion reaction, the ratio of the initial mixture of each reactant was calculated according to the valences of the reactive elements established by stoichiometry according to the chemical concepts of propellants and explosives. The two components were mixed manually in a suitable aluminum vessel for the completion of the combustion process. The mixture was initially heated onto a heating plate, where it quickly boiled and became viscous. A large amount of gases began to break off and were released until auto-ignition (combustion) occurred. After the start of combustion (appearance of a light flame), the alumina powder begins to form in the form of flakes. During the whole combustion process, the reaction temperature near 450°C was observed with the aid of a pyrometer, in the combustion reaction. The synthesized alumina is called ALS.
Characterization of ALS and ALC
X-ray diffraction
X-ray diffraction (XRD) analysis was performed in a Shimadzu model XRD-6000 X-Ray diffractometer (CuKα radiation, Shimadzu Corporation, Japan), the voltage of 40 KV, a current of 30 mA and 2
Particles size distribution analysis
Particles size distribution analysis by laser diffraction used the method of dispersion of particles in liquid phase associated with an optical measurement process through laser diffraction. In this method, the proportional relation between the laser diffraction and the concentration and size of clusters is combined. The analysis was carried out in a Malvern Panalytical Mastersizer 2000 particle size analyzer (Malvern Panalytical, UK).
Scanning electron microscopy
The morphology analysis of ALS and ALC by scanning electron microscopy (SEM), in the powder form, was carried out in a Shimadzu SSX 550 Superscan Scanning Electron Microscope (Shimadzu Corporation, Japan), using a voltage of 15 kV in the filament. Before the analysis, the alumina particles were coated with gold using a Sanyu Electron SC-701 quick coater (Sanyu Electron, Japan).
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was used to evaluate the thermal stability of the ALS and ALC alumina. The analysis was performed in a Shimadzu TGA S1H thermogravimetric analyzer at a heating rate of 10°C min–1 and temperature ranging from room to 1000°C under N2.
Preparation of PMMA/alumina composites by melt blending
Before processing, PMMA was dried in an oven under vacuum at 60°C for 15 h. The PMMA/alumina composites were prepared in two steps: first, a concentrate containing 45 g of PMMA and 5 g of alumina (ALS or ALC) was prepared in a Thermo-scientific Haake Polylab QC Rheomix 600 internal mixer (Thermo Scientific HAAKE, Germany), equipped with roller rotors, at temperature of 200°C and rotors speed of 60 r min–1 during 10 min and, then, granulated. In the second step, for the preparation of the PMMA/alumina composites containing 1 and 3% wt of alumina, the concentrates were diluted into PMMA in a Coperion Werner & Pfleiderer ZSK 18 mm co-rotational modular twin screw extruder (
Preparation of PMMA/alumina composites samples
Samples for tensile and Izod impact strength tests were injection molded, using an Arburg Allrounder 270C golden edition injector (ARBURG, Germany), at a temperature of 250°C. The mold temperature was 40°C. Figure 1 shows the images of the samples obtained by injection molding: (a) PMMA; (b) PMMA-ALS 1%; (c) PMMA-ALS 3%; (d) PMMA-ALC 1%; and (e) PMMA-ALC 3%.

Images of PMMA and PMMA/alumina composites obtained by injection molding: (a) PMMA, (b) PMMA-ALS 1%, (c) PMMA-ALS 3%, (d) PMMA-ALC 1%, and (e) PMMA-ALC 3%. PMMA: poly(methyl methacrylate).
For the wear resistance tests, plates of 10 cm × 10 cm × 0.3 cm (length × width × height) were obtained by compression molding at 280°C in a hydraulic press from MH equipments, applying a pressure of 6 ton during 5 min.
Characterization of PMMA/alumina composites
Optical microscopy
The scratch analysis of the samples subjected to the abrasion tests was carried out in an OPTON model TNB-01T trinocular optical microscope equipped with a camera of 0.45×. Optical microscopy (OM) was also used to analyze the morphology of PMMA/alumina composites.
Scanning electron microscopy
The fracture surfaces subjected to the impact strength tests were coated with gold, using a Sanyu Electron SC-701 quick coater, and the morphology analyzed by SEM using a TECSCAN Vega 3 Scanning Electron Microscope. The voltage used in the filament was 20 kV.
Mechanical properties
Tensile tests were performed according to ASTM D 638, in a Shimadzu AG-IS 100kN universal testing machine with a crosshead speed of 50 mm min–1. Izod Impact strength tests on notched samples were performed according to ASTM D256 in a CEAST RESIL 5.5 apparatus, equipped with a hammer of 2.75J.
Wear resistance
To determine the wear resistance, a Taber, Rotary platform abraser, model 5135, was used. The test was performed according to ASTM D4060-14. The grinding wheel used was CS10, with a standard weight of 1000 g and the test was performed with 500 cycles. The wear index was calculated according to equation (1)

where
Scratch test
The scratch test was performed with a device developed for this test according to the methodology reported by Silva et al. 24 based on the Hoffman scratch test used in the paint and coatings industry. The equipment consists of a cart with a standard weight of 200 g, which contains a scratch tool, a steel tip hardened at a 45° angle to the surface. Three risks were performed in each sample.
Dynamic mechanical thermal analysis
DMTA was carried out in an Anton Paar model Physica MCR 301 rheometer, equipped with solid rectangular fixtures. The samples were heated from 28°C to 160°C at a heating rate of 5°C min–1, a frequency of 1 Hz and strain of 0.1%, under air atmosphere.
Rheological measurements under oscillatory shear flow
Rheological measurements under dynamic-oscillatory shear flow were carried out in an Anton Paar Physica MCR 301 rheometer, equipped with parallel plate geometry of 25 mm diameter and a gap between the plates of 1 mm, at a temperature of 240°C. Frequency sweeps between 0.1 and 600 rad s–1 were carried out at low strains (1%) within the linear viscoelastic region. These strains, under the linear viscoelastic region, were previously determined by the strain sweeps at 240°C.
Rheological measurements at high shear rates
Rheological measurements, under steady shear flow, at high shear rates were carried out in a CEAST (Instron) SR20 capillary rheometer at a temperature of 240°C, capillary die with
Results and discussion
Characterization of the alumina (ALS and ALC)
X-ray diffraction
Figure 2 shows the XRD patterns of ALS and ALC. The ALS and ALC have different crystalline arrangements. The XRD pattern presented by ALS (Figure 2(a)) is similar to that of the patterns found in the ICDD 46-1212 e ICDD 42-1468 cards, where the major crystalline phase identified is α-Al2O3, the same crystalline structure presented by the mineral Corundum.23,25,26 It can also be observed that all the peaks present with high intensity and high basal width, in all the reflections observed, indicating that the samples have high crystallinity. The ALC (Figure 2(b)) corresponds to crystalline hydrated aluminum hydroxide. The XRD pattern presented by ALC corresponds to the crystalline structure of the Gibbsite mineral, as it resembles the patterns found in the ICDD 86-1410 card.27,28
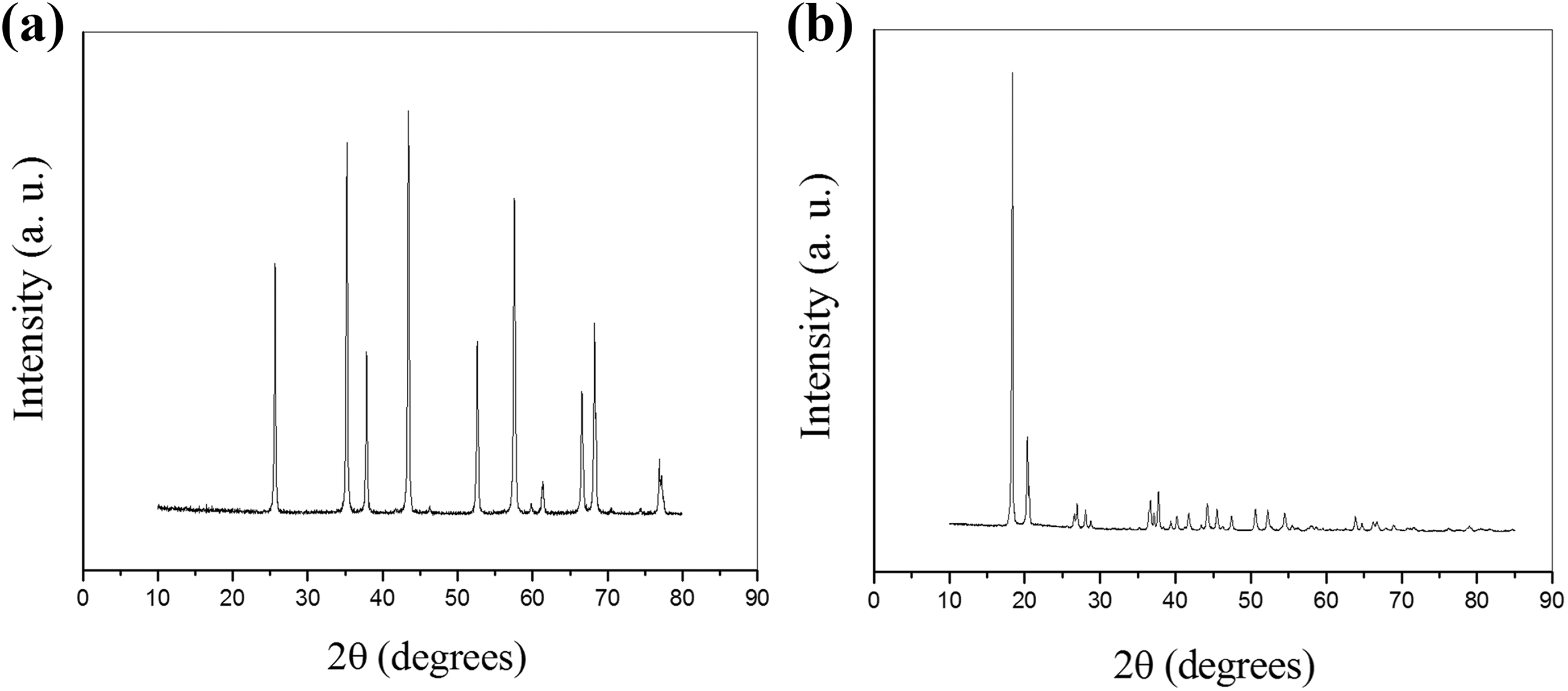
XRD patterns of the alumina: (a) ALS and (b) ALC.
Particles size distribution analysis
Figure 3(a) shows that the ALC has a smaller average diameter, values that are obtained by the cumulative curves in 50%. Figure 3(b) shows that the fillers exhibit monomodal behavior and that the ALS presents a more uniform distribution of its particles, a characteristic indicated by a curve with a base that is narrower than that of ALC.23,29,30 Since ALS was synthesized under controlled conditions, this allowed an aluminum oxide with greater uniformity in the size of its particles. 30
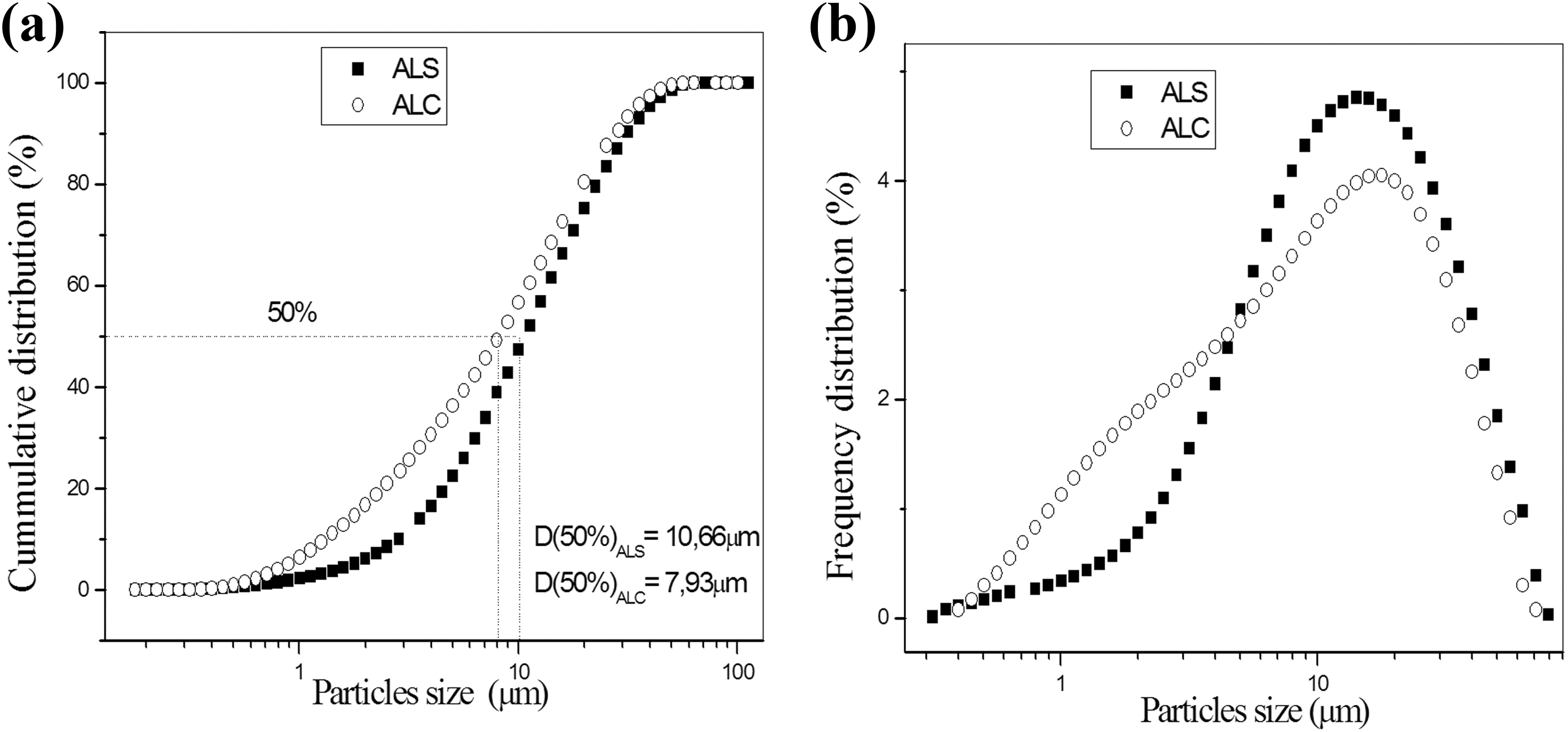
Particles size distribution of ALS and ALC.
Scanning electron microscopy
From the SEM micrographs shown in Figure 4(a), the ALS shows the form of thin plates with irregular geometries of varying sizes. Since ALS is the result of a combustion reaction between aluminum nitrate nonahydrate and urea, its morphology is differentiated from the mineral corundum whose majority phase forms above 1000°C.23,26,28,30 ALC (Figure 4(b)), identified as being Gibbsite, commonly appears in the form of tabular or prismatic particles also having a hexagonal base. One can clearly perceive crystal clusters as well as the formation of some hexagonal and/or pseudo-hexagonal base prisms with sharp edges and irregular contours.25,27,31,32

SEM micrographs of the alumina: (a) ALS and (b) ALC.
Thermogravimetric analysis
Figure 5 shows the TG curves of ALS and ALC. ALS presents a weight loss at temperatures below 200°C, which can be related to the beginning of the decomposition of the components (aluminum nitrate nonahydrate and urea), whose melting points are 73°C and 133°C, respectively, that not reacted during the synthesis of this alumina. 33 Their decomposition temperature range is from 100 to 300°C. ALS continues to lose weight as the temperature is increased. Between 200°C and 800°C, the weight loss is related to the loss of organic matter, adsorbed water and it is possible that there is still the presence of chlorides, sulfates and metal oxides, which may decompose in this interval, present in small percentages as contaminants of the components.26,28,33 ALC present a substantial weight loss between 200°C and 315°C, related to the loss of water. Since one of the applications of ALC is as a flame retardant, due to the high percentage of water present in its composition, the sharp decrease in the weight loss was already expected. As the temperature increases, the sample continues to lose weight until the end of the experiment (1000°C), probably related to the loss of organic matter and the release of hydroxyls present in the gibbsite bauxite. 26
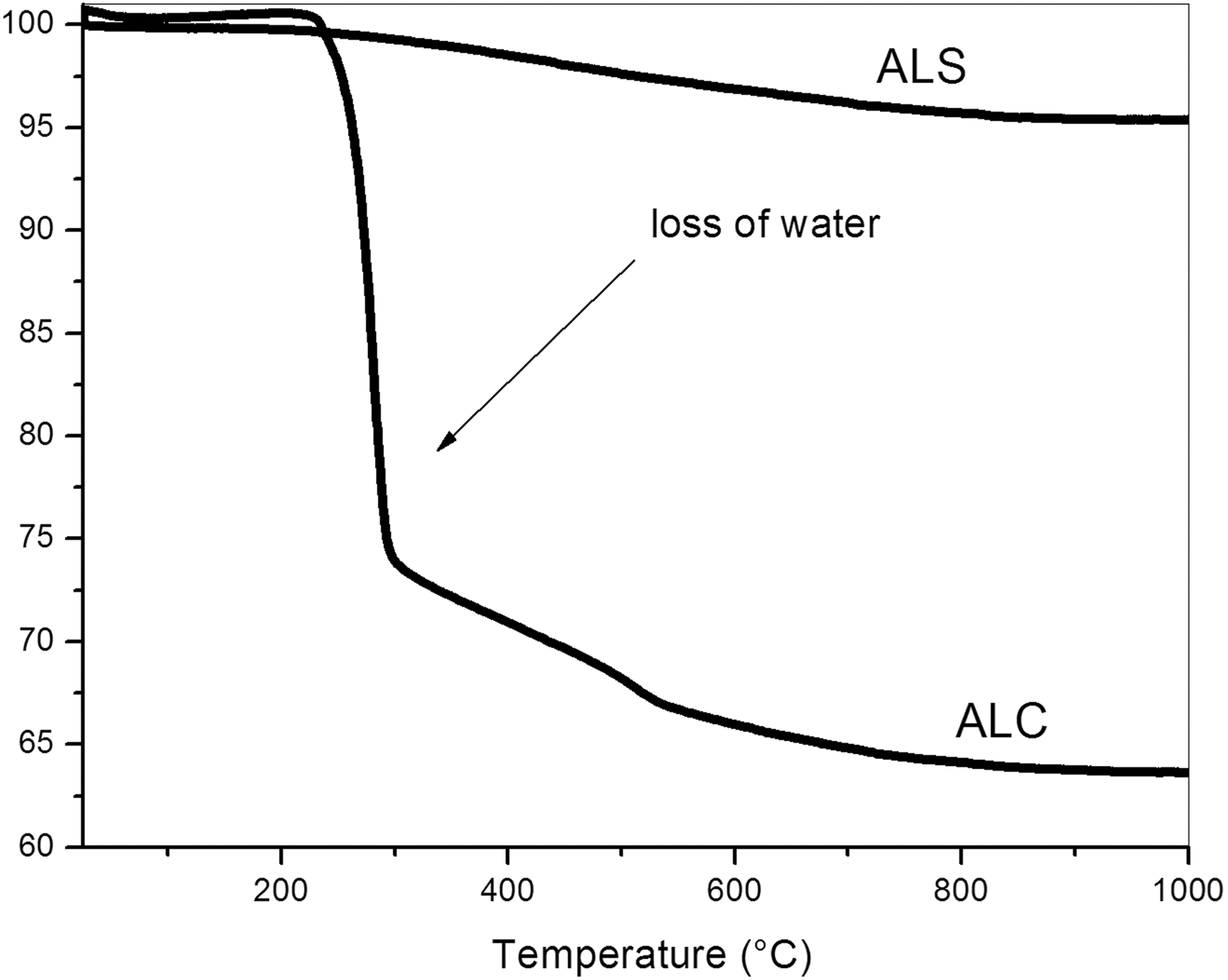
TG curves of the ALS and ALC.
Characterization of PMMA/alumina composites
Optical microscopy
Figure 6 shows the optical micrographs of PMMA/alumina composites. It is observed that in the PMMA/ALS composites (Figure 6(a) and (b)), the ALS particles are well dispersed in the PMMA matrix. On the other hand, in the PMMA/ALC composites (Figure 6(c) and (d)), the ALC particles are well distributed in the PMMA matrix and many agglomerates are observed. ALC particles size is higher than that of ALS particles. This may be ascribed to the shape (Figure 4(b)) and higher hardness of ALC particles, which during the processing (extrusion followed by injection molding), the shear rate was not enough to break the ALC agglomerates, thus not decreasing the ALC particles average size.

Optical micrographs of PMMA/alumina composites: (a) PMMA/ALS 1%, (b) PMMA/ALS 3%, (c) PMMA/ALC 1%, and (d) PMMA/ALC 3%.
Scanning electron microscopy
Figure 7 shows the SEM micrographs of PMMA/alumina composites. Figures 7(a) and 7 (b) show that ALS is finely dispersed in the PMMA matrix, regardless of ALS content (1% or 3%). The particles size distribution analysis indicated that ALS has a more uniform particle size distribution than ALC (Figure 3(b)). Figure 7(c) and (d) show that though the ALC particles are well distributed in the PMMA matrix, their average particles size is higher than that of ALS and some agglomerates are observed. These results are in agreement with the morphology observed by OM. It is also observed that the alumina particles are not wetted by the PMMA matrix. A similar result was observed by Ash et al. 2 that ascribed this result to the lack of affinity between PMMA and alumina particles. Mallakpour and Khadem 34 reported that alumina particles have high surface energies and tend to aggregate in order to minimize these energies.

SEM micrographs of the PMMA/alumina composites: (a) PMMA/ALS 1%, (b) PMMA/ALS 3%, (c) PMMA/ALC 1%, and (d) PMMA/ALC 3%.

Scratch in the surface of the samples: (a) PMMA, (b) PMMA/ALS 1%, (c) PMMA/ALS 3%, (d) PMMA/ALC 1%, and (e) PMMA/ALC 3%.
Wear resistance
PMMA, together with other glassy polymers such as polystyrene, exhibits a relatively low wear resistance when compared to glass. In particular, its wear is initiated by the formation of surface cracks parallel to the sliding direction as a result of a high coefficient of friction.
Figure 8 shows a wear index of PMMA and of PMMA/alumina composites. The wear index of PMMA substantially decreases with the addition of ALS and ALC. According to the literature, ceramic fillers, such as alumina, when added to the PMMA matrix, act as a barrier to the propagation of cracks on the surface of the matrix, increasing its resistance to wear.24,35–37 The composites containing 3% of ALS and 1% of ALC presented the lowest wear index.
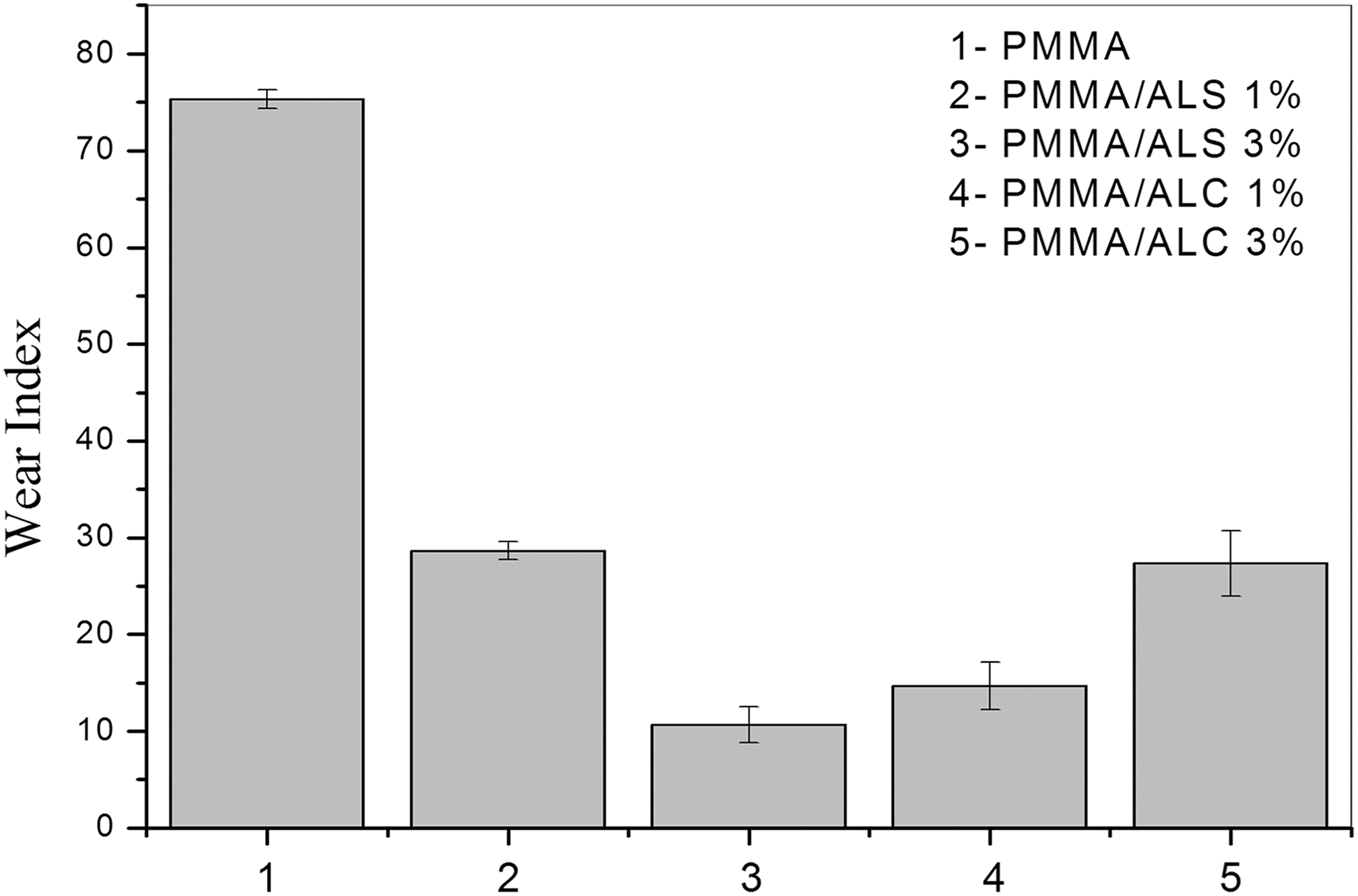
Wear index of PMMA and PMMA/alumina composites.
As for the different behavior of ALS and ALC in the wear resistance of PMMA, it can be qualitatively inferred by OM (Figure 6) and SEM (Figure 7) that the ALS is better dispersed and distributed, whereas the ALC, despite presenting agglomerates of alumina, these agglomerates are well distributed along the PMMA matrix and the size is less than 40 μm. In addition, ALS and ALC exhibit different crystalline structures and different particle sizes, which may also affect the wear index of the composites.
Scratch test
The resistance of PMMA/alumina composites to scratch was evaluated, based on the work of Silva et al. 24 through the analysis of the scratch width. Table 1 summarizes the width of the scratches of PMMA and PMMA/alumina composites. The scratch width increases with the addition of alumina particles (ALS or ALC) to PMMA.
Width of the scratch of PMMA and PMMA/alumina composites subjected to scratch tests.

PMMA: poly(methyl methacrylate).
Three wear mechanisms caused by scratch can occur 24 : microploughing; microcutting, and microcracking.
However, some characteristics differentiate each sample. By evaluating the pure PMMA (Figure 9(a)), it can be noticed that there is a splinter detachment of the material and a well-marked groove with a certain depth and with indications of plastic deformation at its extremities. The roughness of the harder surface presses the softer surface, inducing the formation of a flow of material from the soft surface around the harder surface, removing the softer material leaving a groove printed on the specimen. By evaluating the risks made in the PMMA/alumina samples, it can be observed an increase in the scratch width and that all samples present the characteristics of the microcracking type, where part of PMMA containing the filler is pulled out during the scratch test, as also observed by Silva et al. 24 studying PMMA/silica composites. In the case of PMMA/alumina composites containing ALC (Figure 9(d) and (e)), the formation of the groove is also observed, with a slight accumulation of material at the extremities, but the appearance of the impression left on the surface is wrinkled, with the removal of some flakes of material, indicating that the composites with ALC offered more resistance to the risk than the pure PMMA. This resistance was even greater in the case of the composites with ALS (Figure 9(b) and (c)) since the sharpest detachment of flakes of material can be clearly perceived and the groove did not present much depth. These results confirm what has already been observed in the abrasion test that the alumina fillers are bearing part of the applied load, offering resistance to PMMA surface penetration. An increase in the scratch width is observed when the alumina content is increased from 1% to 3%.
Mechanical properties
Table 2 summarizes the results of the mechanical properties of PMMA and PMMA/alumina composites containing 1% and 3% of alumina. Overall, the elastic modulus of PMMA increases with the addition of ALS and ALC whereas the tensile and impact strength decreases. The decrease in the impact strength may be related to the agglomerated alumina particles that act as stress concentrators and to the lack of adhesion between PMMA and alumina particles, as can be observed by SEM (Figure 7). Joni et al. 38 reported that high loading of fillers can have a negative effect on the mechanical properties of composites, due to poor dispersion of the fillers or lack of affinity between the polymer matrix and the fillers. Among the composites, the best performance is observed for the PMMA/ALS containing 1% of ALS, which has balanced properties.
Mechanical properties of PMMA and of PMMA/alumina composites.
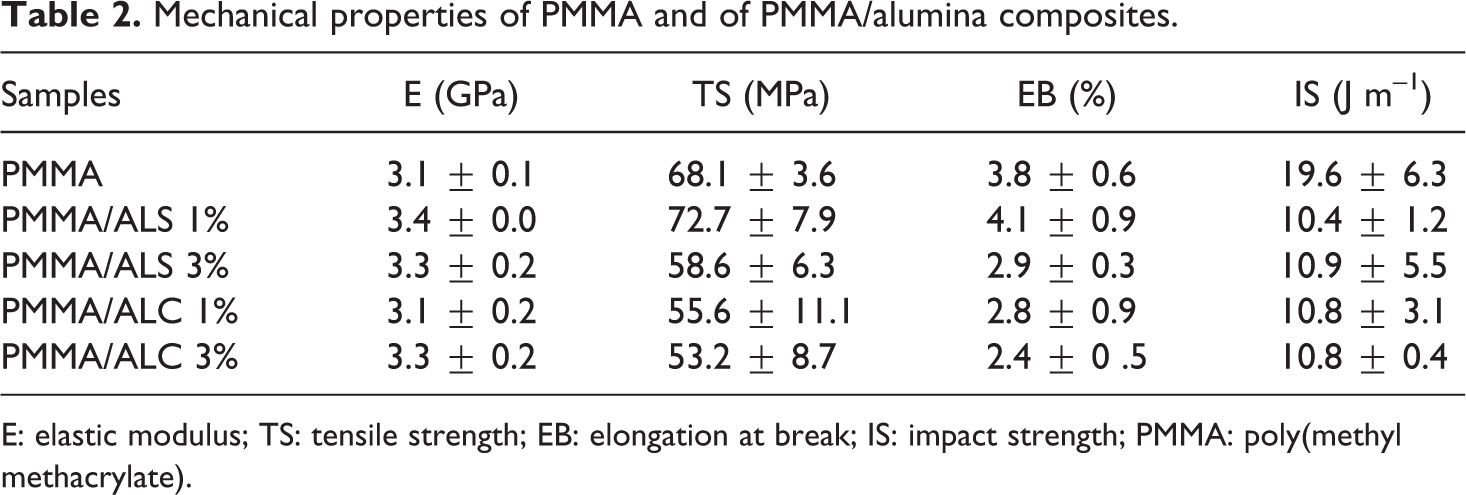
E: elastic modulus; TS: tensile strength; EB: elongation at break; IS: impact strength; PMMA: poly(methyl methacrylate).
Dynamic mechanical thermal analysis
Figure 10 shows the temperature dependency of the storage modulus (
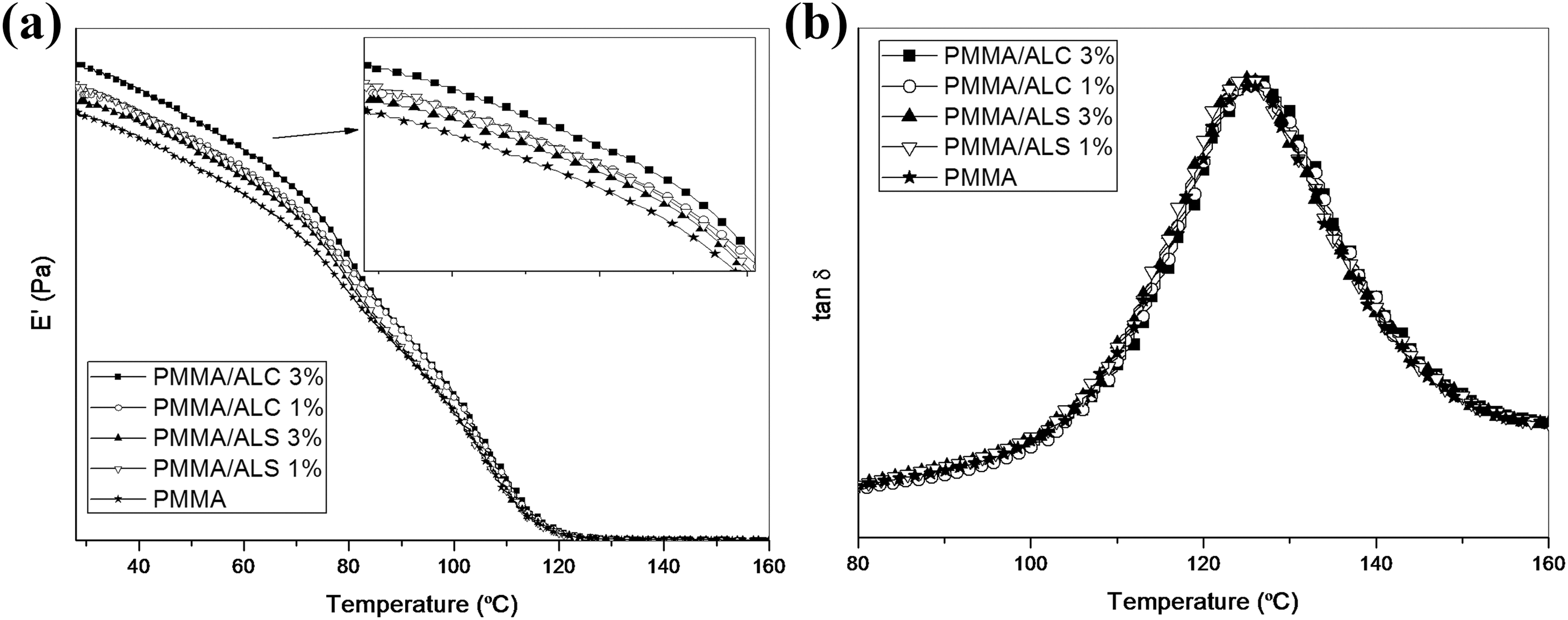
Temperature dependence of
Rheological measurements under oscillatory shear flow
Figure 11 shows the plots of complex viscosity (
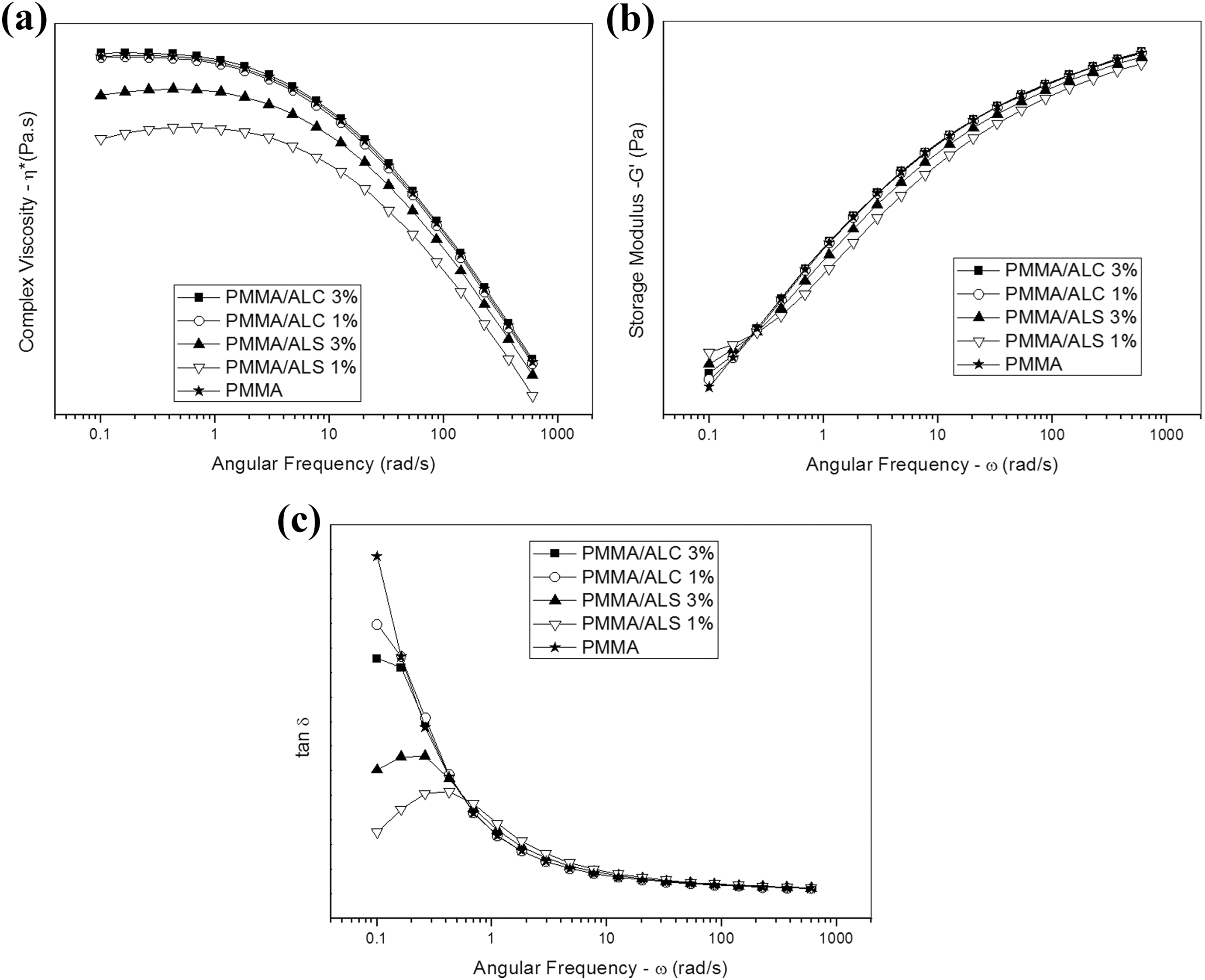
Plots of complex viscosity (
The zero shear viscosity (

where
Table 3 summarizes the parameter obtained by fitting the viscosity plots. It can be observed that the zero shear viscosity of PMMA/ALC composites is close to that of PMMA. On the other hand, there is a substantial decrease in the viscosity with the addition of 1% of ALS to PMMA. Further increase in the ALS content from 1% to 3% leads to an increase in the zero shear viscosity. The values of the power law index (
Cross fitting parameters for PMMA/ALS and PMMA/ALC composites.
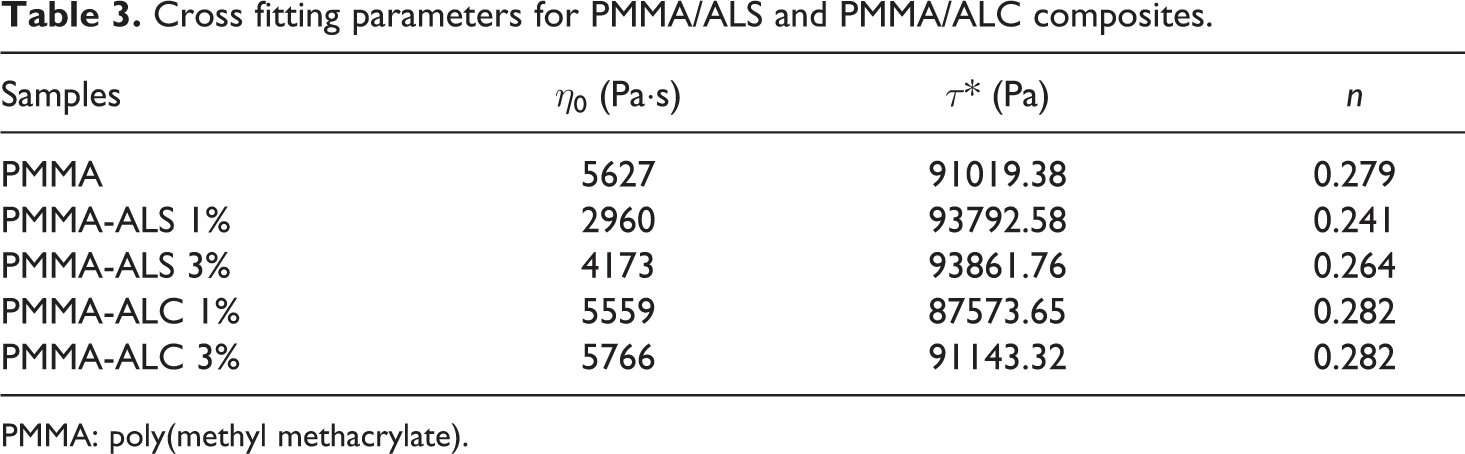
PMMA: poly(methyl methacrylate).
Figure 11(b) shows the plots of
Viscosity plots (a) and flow curves (b) of PMMA/ALS and PMMA/ALC composites. PMMA: poly(methyl methacrylate).
Rheological measurements at high shear rates
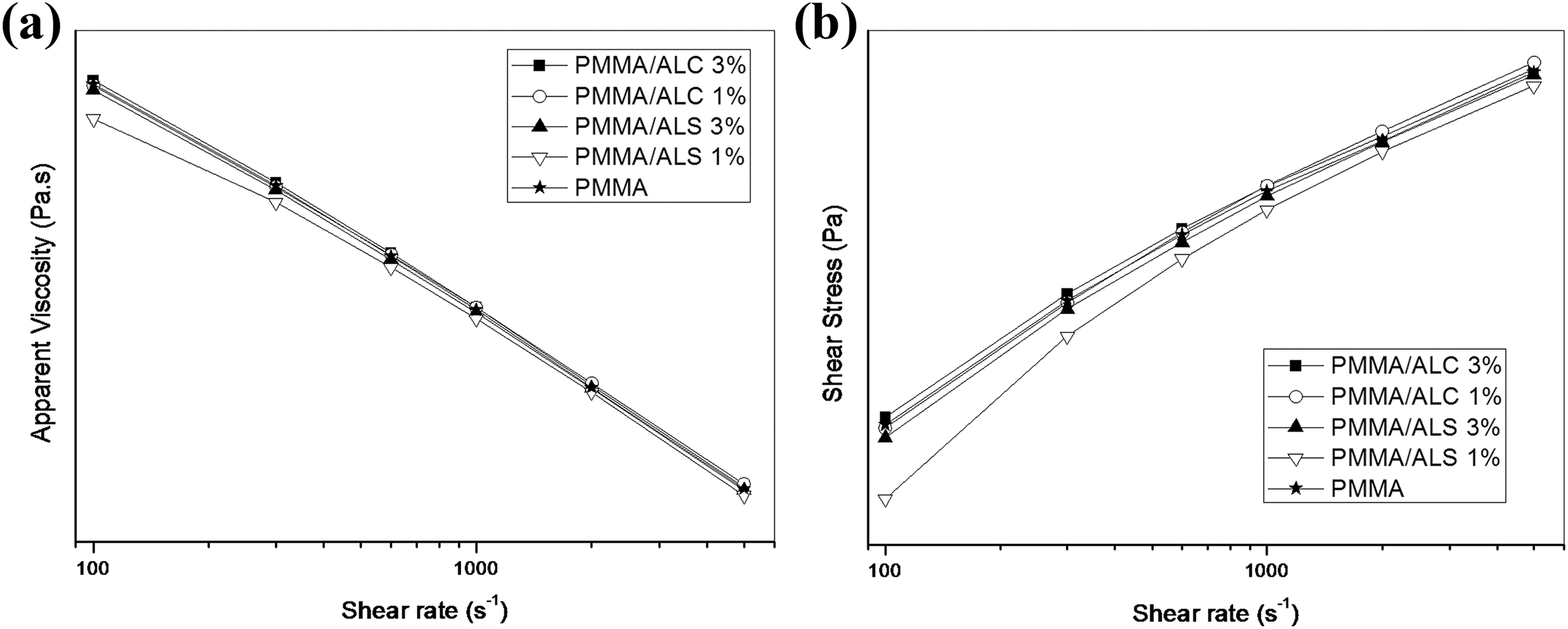
Figure 12 shows the viscosity and shear stress plots as a function of shear rate of PMMA/ALS and PMMA/ALC composites containing 1% and 3% of ALS or ALC composites. Figure 12(a) shows the plots of the viscosity as a function of shear rate. The viscosities of PMMA/ALC and PMMA/ALS composite containing 3% of ALS are close to that of PMMA at lower shear rates whereas the viscosity of PMMA/ALS composite containing 1% of ALS is lower than that of PMMA. At higher shear rates, the viscosities of PMMA/ALS and PMMA/ALC composites are similar to that of PMMA. This may be related to the orientation of ALS and ALC particles in the direction of flow. Figure 12(b) shows the flow curves (shear stress vs. shear rate) of PMMA/ALS and PMMA/ALC composites. It can be observed that the plots deviate from linearity indicating that PMMA and PMMA/ALS and PMMA/ALC composites present a shear-thinning behavior.
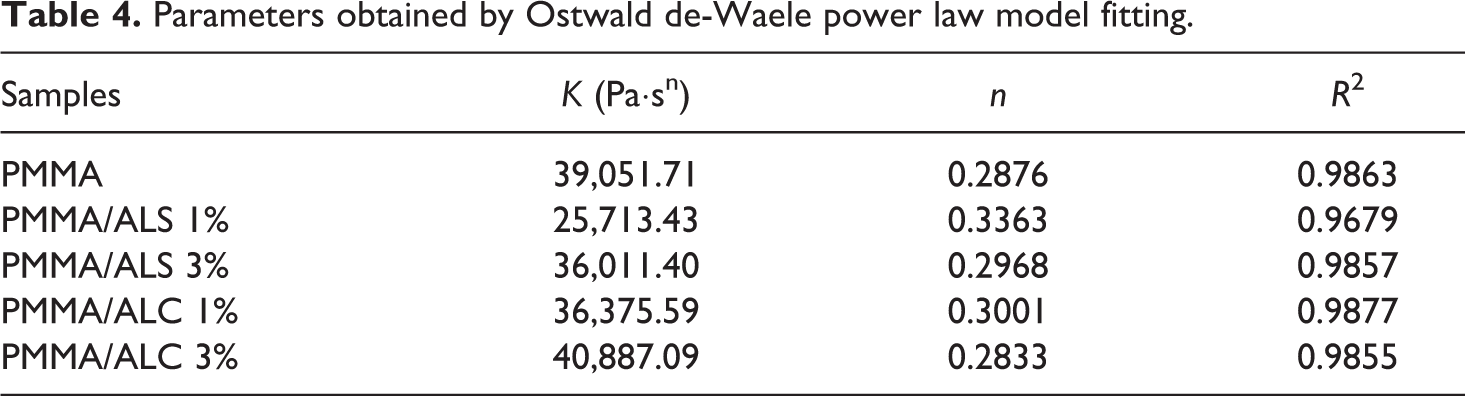
The plots were fitted using the Ostwald de-Waele power law model presented in equation (3)

where is the
Table 4 summarizes the parameters obtained by the fitting of the plots using the power law model. The consistency index (
Parameters obtained by Ostwald de-Waele power law model fitting.
Conclusions
The aim of this work was to evaluate the properties of PMMA/alumina composites, containing a synthesized (ALS) and commercially available alumina (ALC), obtained by melt blending in a twin screw extruder. The addition of alumina to PMMA led to a substantial decrease in the wear index and increased the scratch resistance. The tensile modulus was not affected with the addition of ALS and ALC to PMMA whereas the impact strength decreased. The type of alumina (ALS or ALC) and concentration (1% and 3%) did not affect the impact strength of PMMA/alumina composites. PMMA/alumina composites containing ALC presented higher alumina average particles size than those containing ALS. The storage modulus (
