Abstract
Polymer nanocomposites offer enhancement in thermomechanical and physicochemical properties of polymers with the presence of a little amount of nanostructured fillers such as carbon nanotubes, graphene, and layered silicates. A facile and rapid preparation of hytrel (HTL)-graphene oxide (GO) nanocomposites is done
Introduction
Polymer nanocomposites offer enhancement in thermomechanical and physicochemical properties of polymers with the presence of a little amount of nanostructured fillers such as carbon nanotubes (CNTs), graphene, and layered silicates. 1 –4 Graphene, a two-dimensional, single atomic layer of sp 2 -hybridized carbon atoms arranged in hexagonal lattice has attracted a great attention of researchers in the last decade. 5 –7 It has been accounted as a leading reinforcement material due to its high electron conductivity (200,000 cm2 V−1 s−1), exceptional thermal conductivity (approximately 5000 Wm−1 K−1), ultimate Young’s modulus (1 TPa), elastic modulus (approximately 0.25 TPa), fracture strength (130 GPa), and large specific surface area (2630 m2 g−1). 8 –12 However, preparation of a large amount of graphene oxide (GO) and reduced graphene oxide (rGO) is easily achievable compared to single graphene sheet. 13,14 Also, GO contains hydroxyl and epoxy groups on the basal plane and carboxyl groups at the edges, and thus useful as a reinforcement material with homogeneous dispersion in polymer matrix. GO has been considered one of the best nanofillers, and provides enormous structural applications ranging from aerospace, construction, transportation industries to light-weighted commodity plastic. 15 –19
Bora et al. prepared unsaturated polyester resin (PE)/GO nanocomposite with 1–3 wt% of GO concentration with respect to PE
Herein, we report the facile and rapid preparation of hytrel (HTL)-GO nanocomposites
Materials and method
HTL grade 4056 was supplied by E.I. DuPont de Nemours & Co, Hyderabad, India. Sigma-Aldrich (Saint Louis, USA) kindly provided expanded graphite powder, whereas, potassium permanganate (KMnO4) was purchased from Central Drug House (P) Ltd, Delhi. Sodium nitrate (NaNO3) and hydrochloric acid (HCl) were obtained from Merck Life Science Private Ltd, Mumbai, India. Sulfuric acid (H2SO4) was purchased from Finar Limited. Hydrogen peroxide (H2O2) and chloroform (CHCl3) were supplied by Avantor Performance Materials India Ltd, Dehradun, Uttrakhand.
The oxygenated derivative of graphene was produced from graphite powder by modified Hummer’s method. 26 The preparation of GO/HTL nanocomposite films with different concentration of GO was complete as per the following process, where firstly 2 g HTL 4056 polymer was dissolved in 30 ml of chloroform under constant stirring for 6 h and then a certain quantity of GO was uniformly dispersed in 10 ml of ethanol with the help of magnetic stirring for 1 day. Regarding the impact of GO percentage on material properties, we have prepared GO/HTL nanocomposite films containing 0, 0.1, 0.5, 1, 2, and 5 wt% GO with respect to HTL, where firstly HTL was completely mixed in chloroform, followed by the addition of brown colored homogeneous suspension of GO, and the resultant mixture was kept in stirring for 2 days. After that, the homogeneous solution was slowly poured into petri dish and dried at room temperature for 1 day. The dried membranes were peeled off for testing. A similar approach was applied for the preparation of GO/HTL nanocomposite films with different GO loadings. The preparation route is also presented in Figure 1.
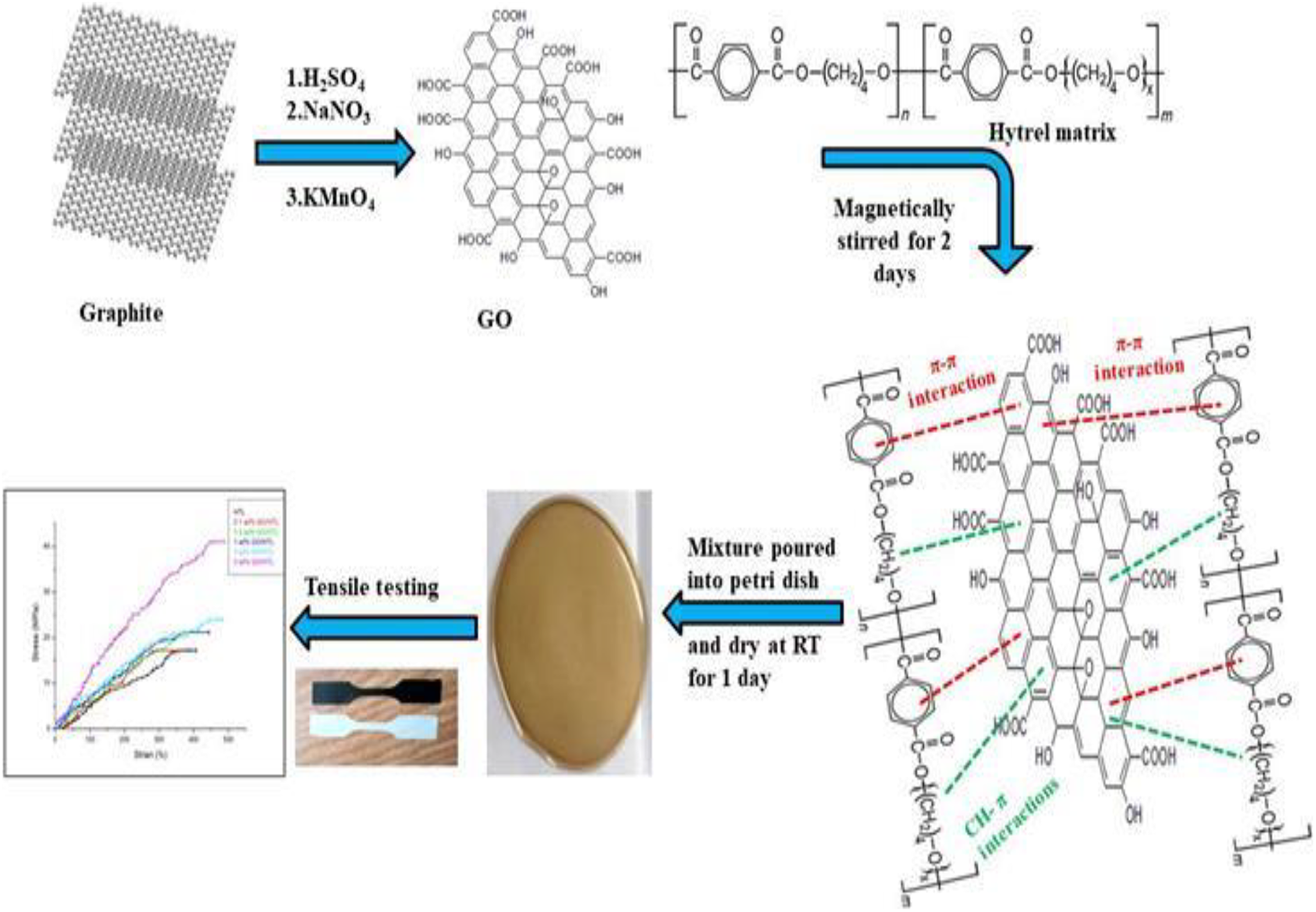
Representation of synthesis route of HTL nanocomposites.HTL: hytrel.
Characterization techniques
Raman spectroscopy (RIRM-LP1519, Research India; with 532 nm excitation) was performed to analyze the structural characteristics of GO including the interaction between GO sheets and HTL polymer matrix.
DSC analysis was conducted for the analysis of crystallization and melting behavior of nanocomposites by using PerkinElmer DSC 4000 thermal analyzer. The samples were heated from 0°C to 280°C, holding at 280°C for 1 min and then cooled to 0°C with 10°C min−1rate in all runs. The experimental procedure was performed under nitrogen atmosphere by taking approximately 3 mg of sample in sealed aluminum pan.
The thermal stability of polymer nanocomposites containing GO was illustrated with the help of TGA by using PerkinElmer TGA 4000 thermal analyzer. The loss in weight percent was observed from 30°C to 600°C with a heating rate of 10°C min−1 with respect to temperature.
Dynamic mechanical analyzer was used to evaluate the viscoelastic properties of nanocomposite samples by using PerkinElmer DMA 8000 analyzer. The samples were operated from 28°C to 135°C with a constant temperature and frequency scan (2°C min−1, 1 Hz).
The mechanical properties of GO/HTL composites were analyzed by universal tensile testing machine (UTM 1623, A. S. I. Sales Private Limited) with 20 kN load cell and jaw separated with 25 mm min−1 speed.
Results and discussion
Figure 2 presents the Raman spectra of GO and HTL nanocomposites, where the spectra of GO show two prominent peaks at 1315.22 cm−1 (D band) and 1553.864 cm−1 (G band).
27
The G band is originated due to the stretching of sp
2
-hybridized carbon (C=C) frame and D band is due to sp
3
-hybridized carbon, generated by structural defects developed by the attachment of polar functional groups on graphene sheet,
28
where the intensity of both peaks (
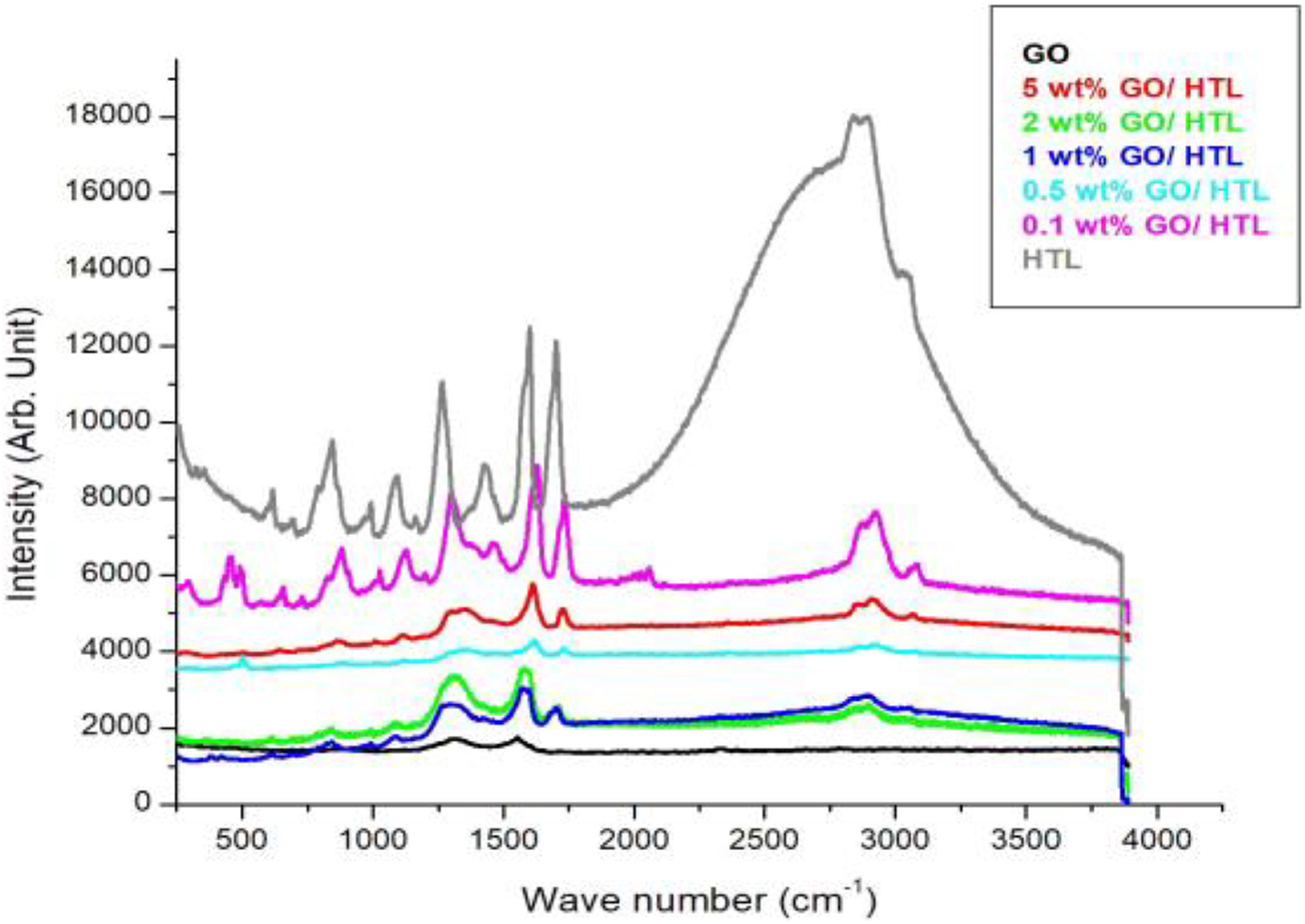
Raman spectra of GO and GO/HTL nanocomposites.HTL: hytrel; GO: graphene oxide.
The melting and crystallization parameters of HTL and GO/HTL nanocomposites were obtained from DSC analysis, as mentioned in Table 1 and Figure 3(a) and (b). The pristine HTL shows
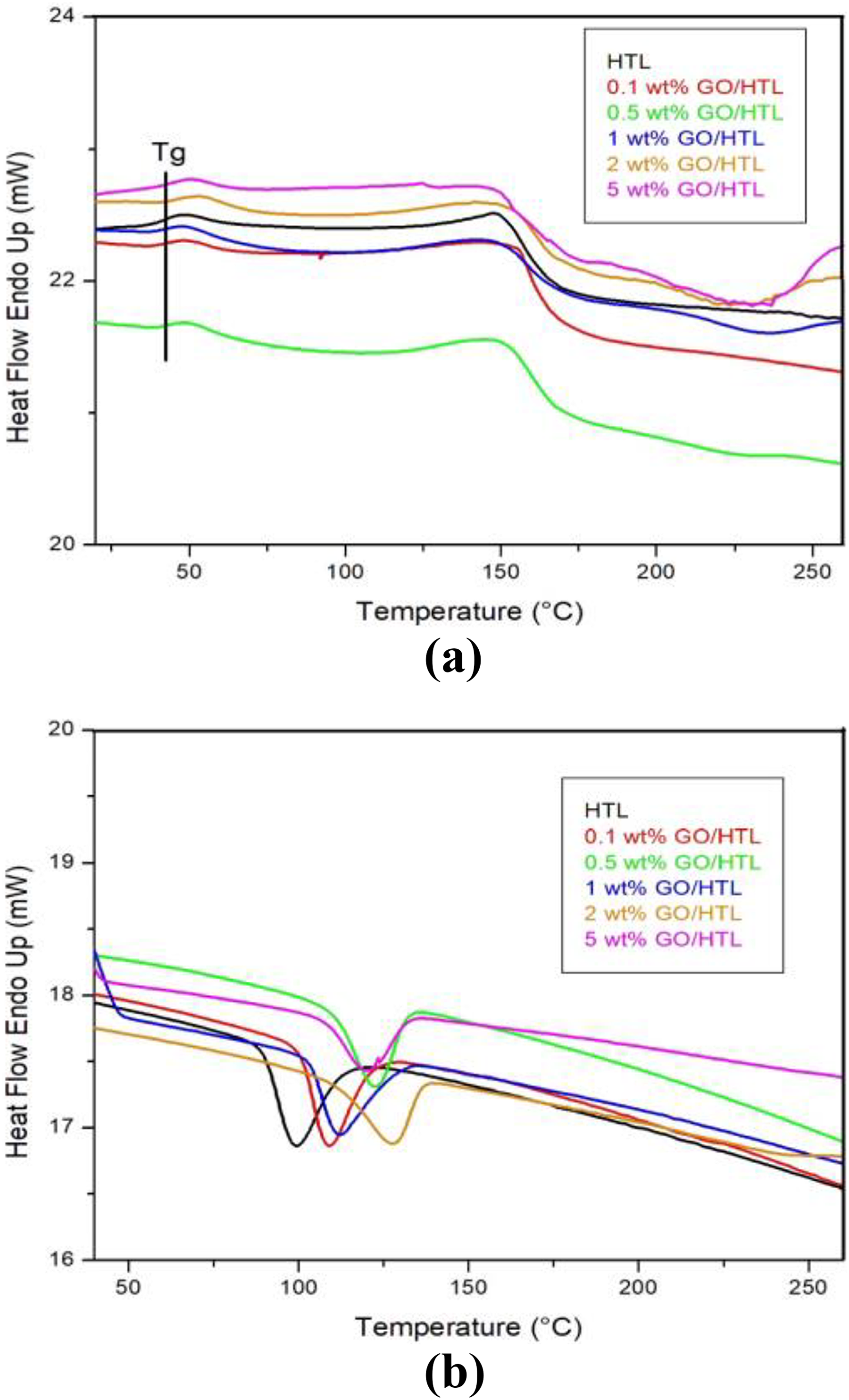
DSC thermogram of GO/HTL composites on (a) heating and (b) cooling.HTL: hytrel; GO: graphene oxide; DSC: differential scanning calorimetry.
DSC analysis data of HTL and its nanocomposites with different GO loadings.
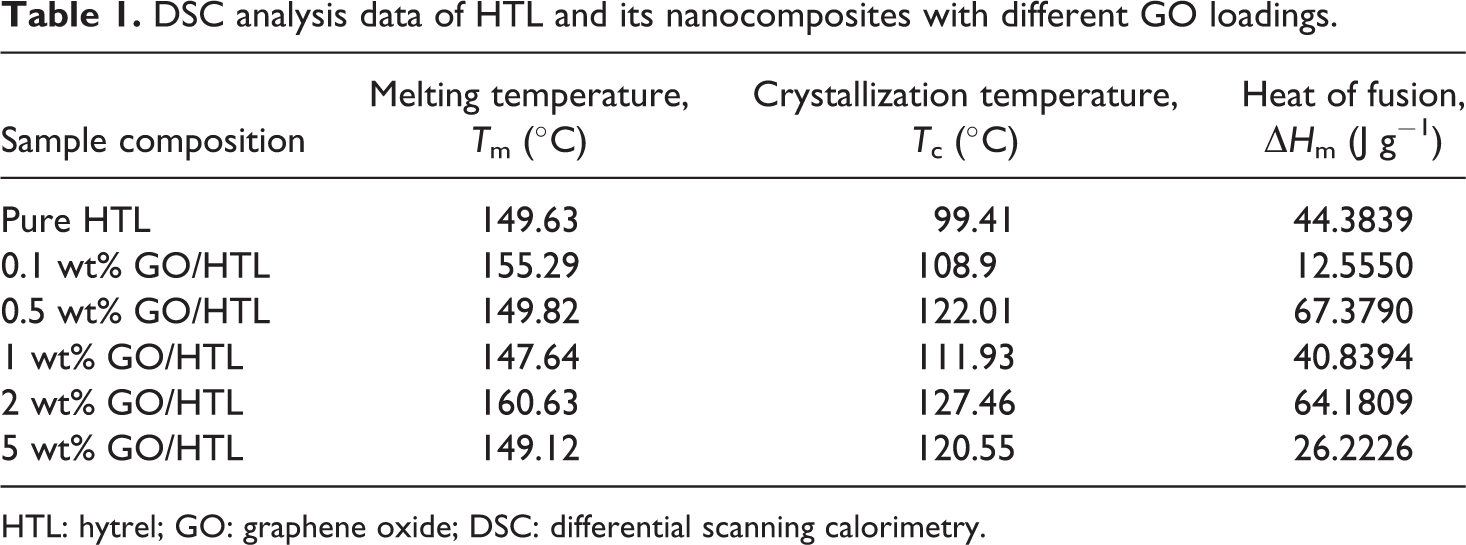
HTL: hytrel; GO: graphene oxide; DSC: differential scanning calorimetry.
TGA thermogram represents the influence of GO concentration on the thermal stability of HTL composites, as displayed in Figure 4. The thermal degradation of GO occurs in three steps, where the first degradation curve was observed due to the removal of water molecules below 100°C, the second curve could be due to the removal of polar functional groups (hydroxyls, carboxyls, and epoxides) at nearly 200°C, and the third one is due to the ignition of carbon skeleton.
33
Weight loss of 5 and 50% was considered to determine the thermal stability of nanocomposites (Table 2). The nanocomposites with 0.1, 0.5, 1, 2, and 5 wt% GO loadings show 71.21, 130.79, 140.12, 141.26, and 143.83°C increment in
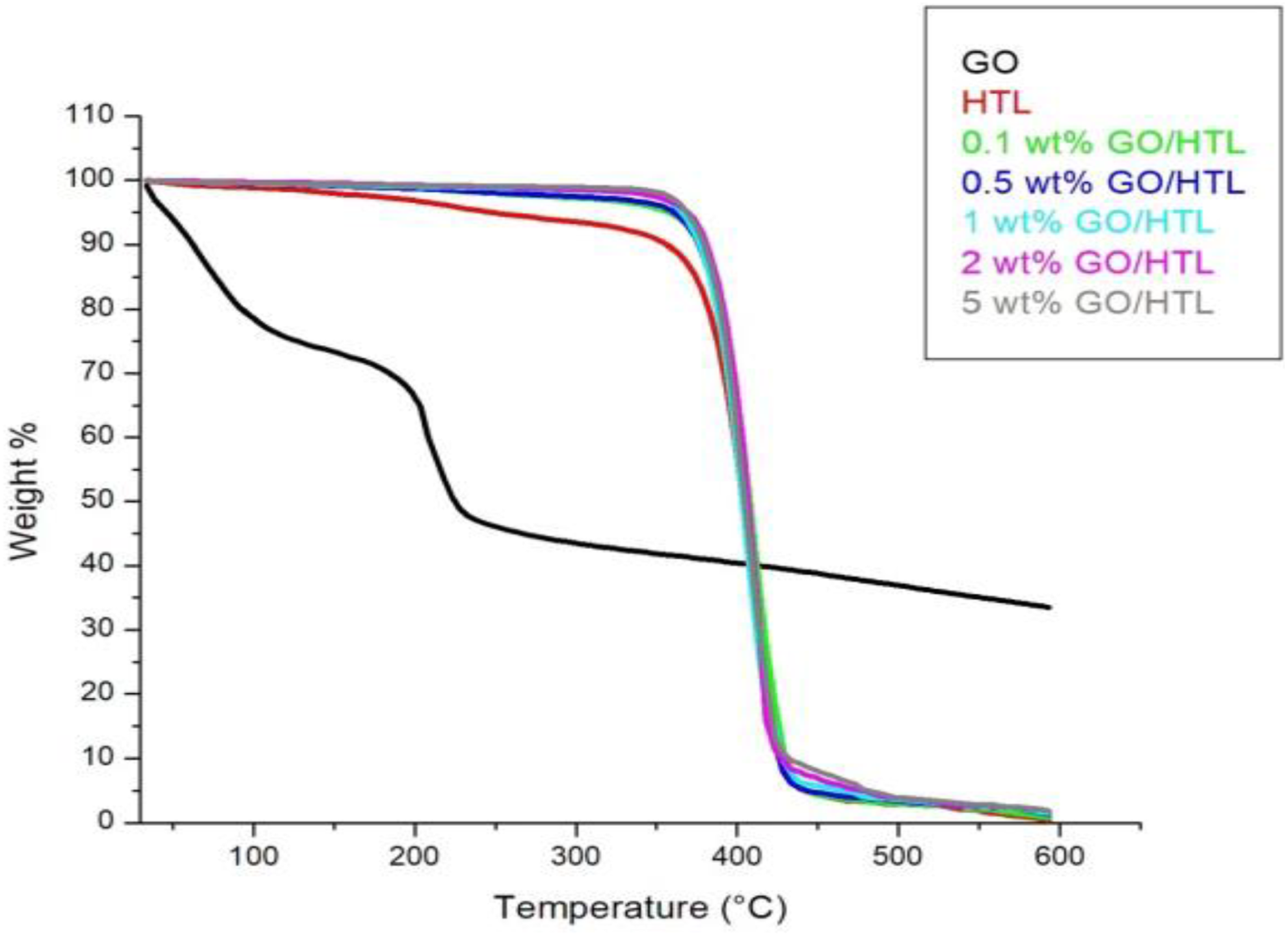
TGA thermogram of GO and GO/HTL nanocomposites.HTL: hytrel; GO: graphene oxide; TGA: thermogravimetric analysis.
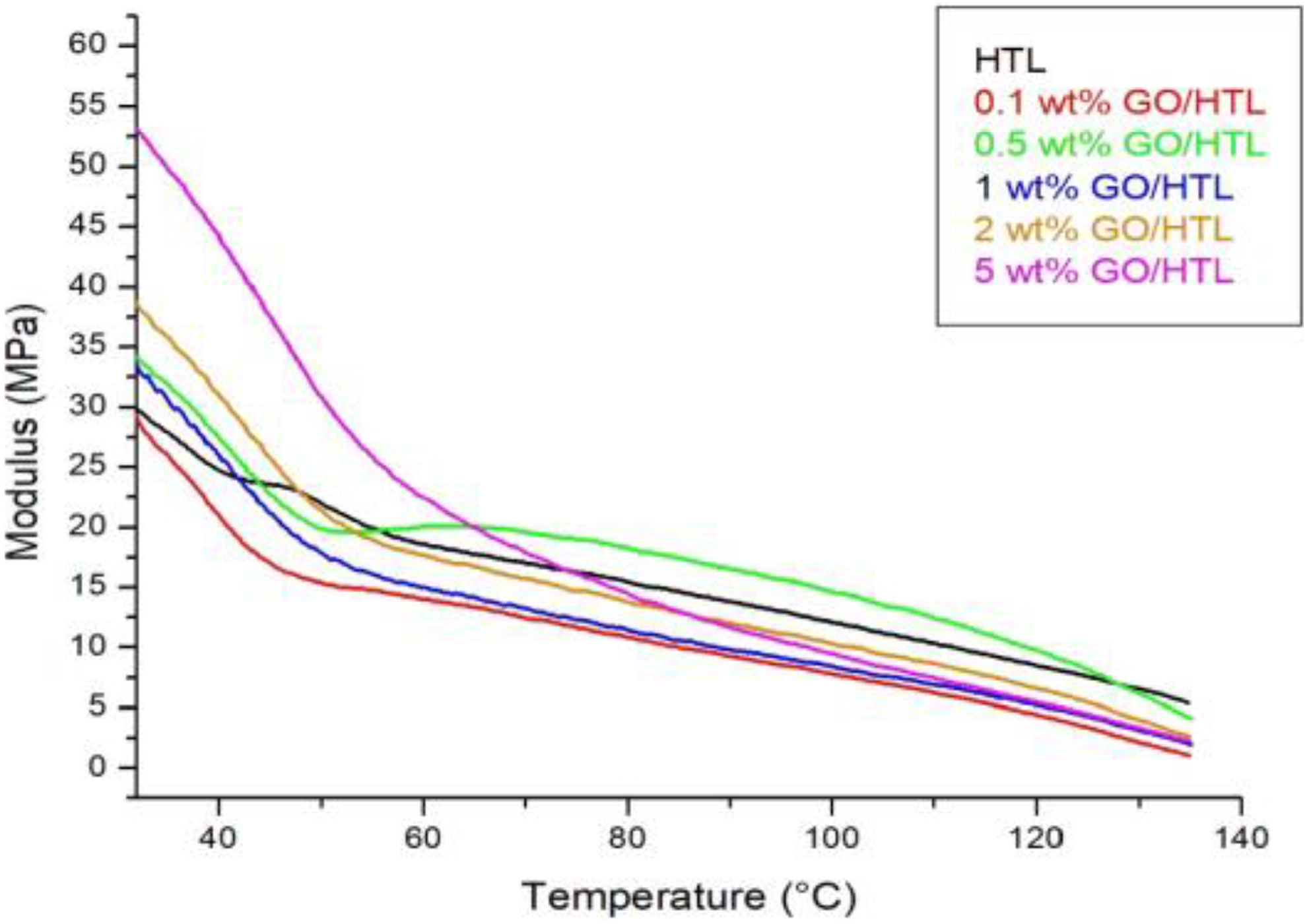
DMA plot of GO/HTL nanocomposites with constant frequency (1 Hz).HTL: hytrel; GO: graphene oxide; DMA: dynamic mechanical analysis.
TGA analysis data of HTL and GO/HTL composites.

HTL: hytrel; GO: graphene oxide; TGA: thermogravimetric analysis.
Figure 6 demonstrates the mechanical performance of pristine HTL and its composites
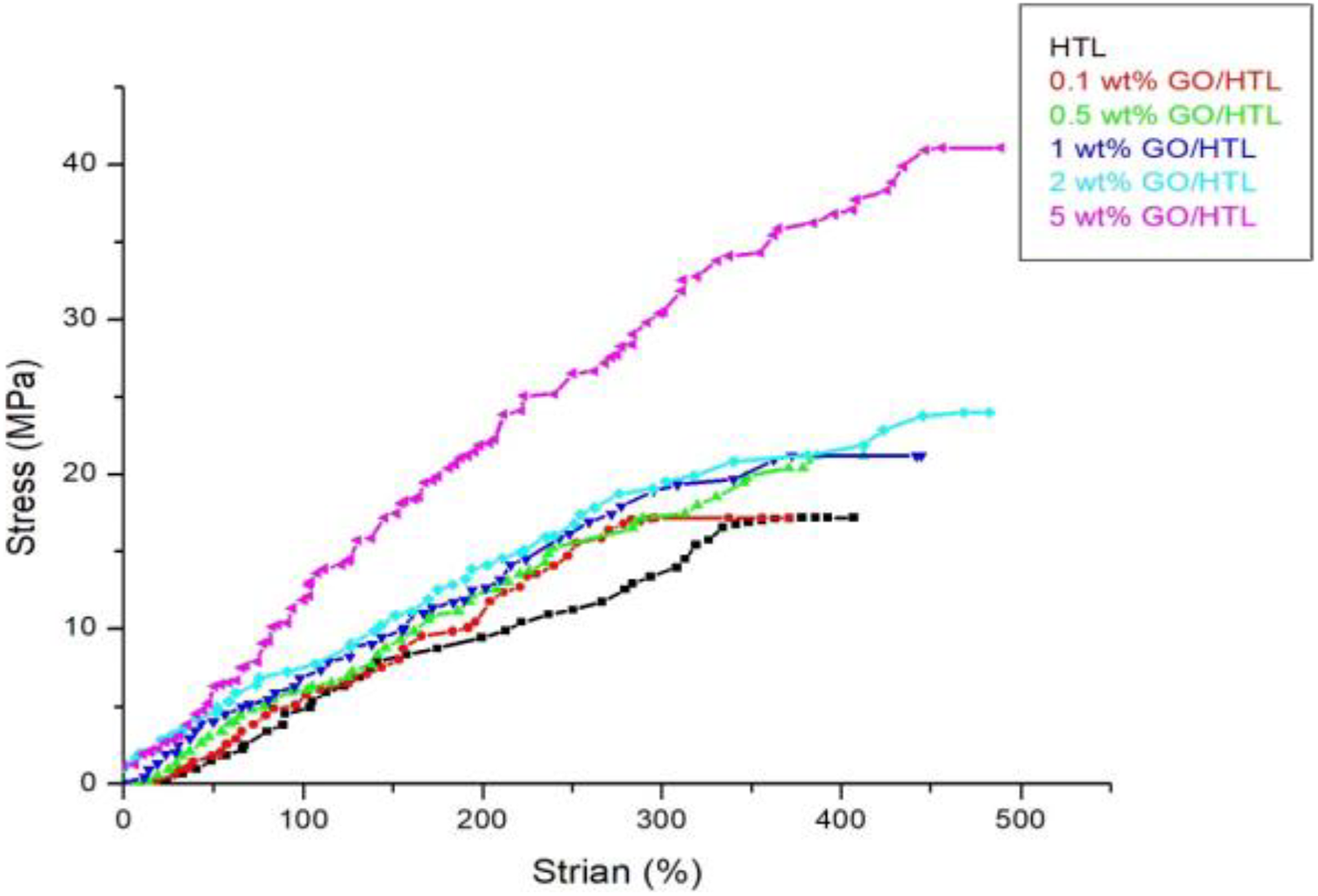
Stress–strain curve of GO/HTL nanocomposites.
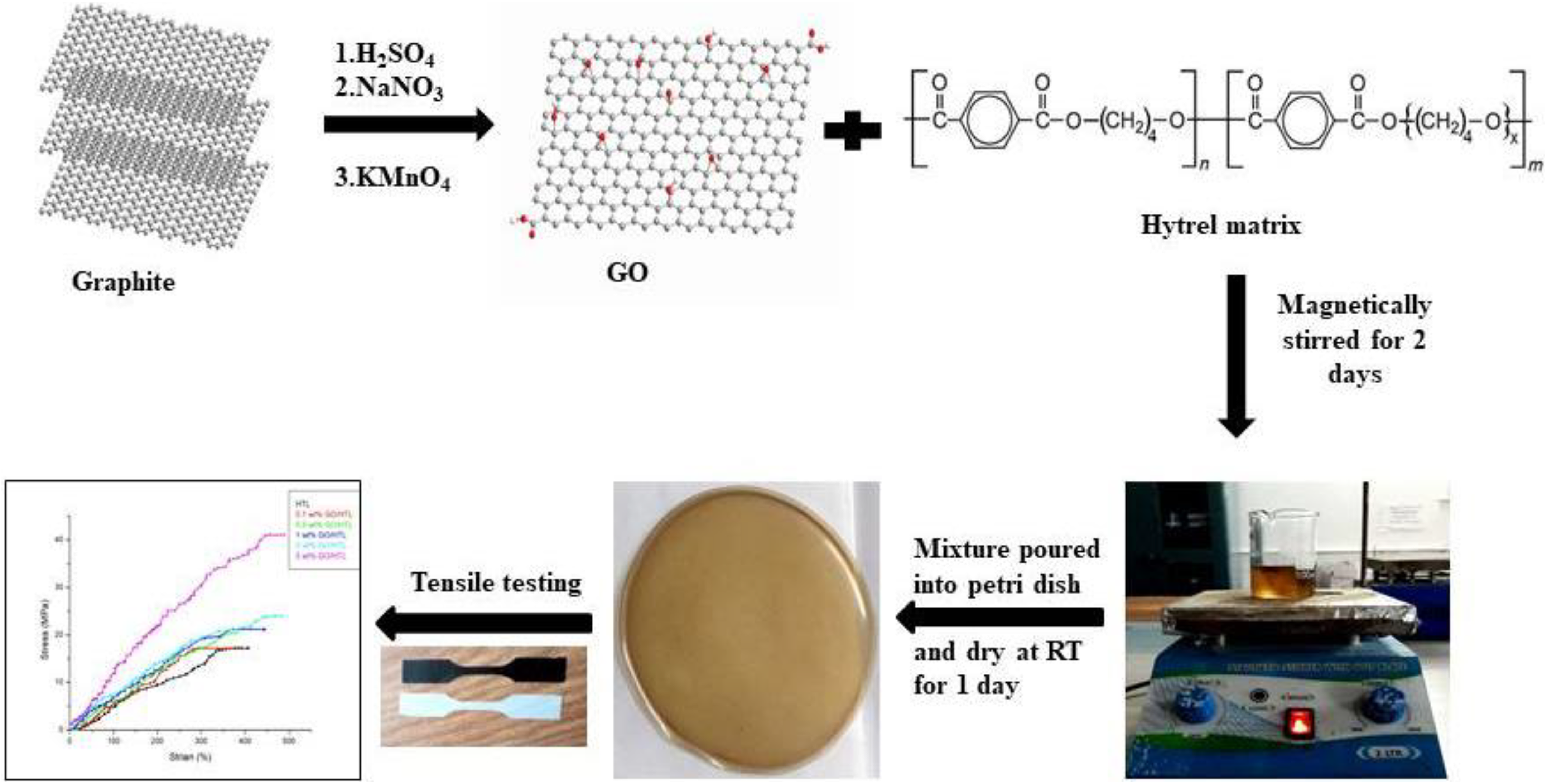
Synthesis of GO and GO/HTL nanocomposites.HTL: hytrel; GO: graphene oxide.
Conclusion
The incorporation of GO into HTL polymer was successfully achieved with enhanced thermal and mechanical properties due to the presence of strongest noncovalent interaction (π–π stacking) between the interface of nanocomposites. The thermal and mechanical analysis data of GO/HTL nanocomposites and pure HTL were compared to analyze the impact of GO addition in polymer. The DSC analysis reveals that
Footnotes
Author contributions
Alok Chaurasia and Nanda Gopal Sahoo contributed equally to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors acknowledge the financial support from NMHS research grant, GBPIHED, Kosi-Katarmal, Almora and DST-FIST, New Delhi. S.R. acknowledges a grant from Science and Engineering Research Board (SERB-DST), Government of India (Grant No. ECR/2016/001355).
