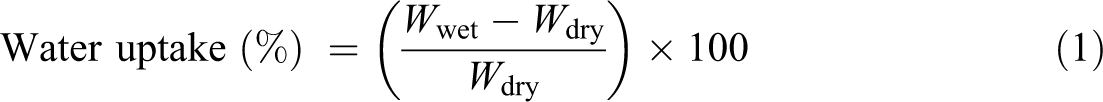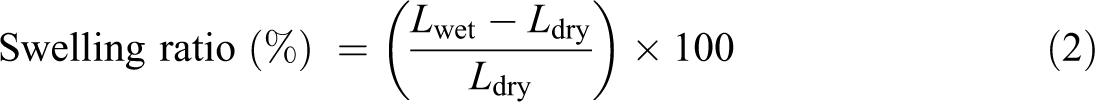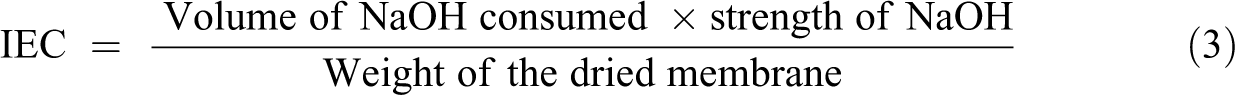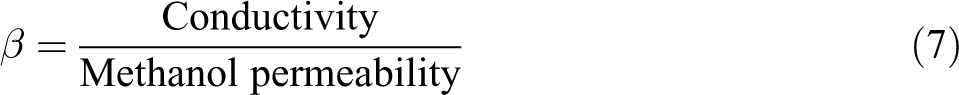Abstract
Composite membranes based on sulfonated poly (ether ether ketone) (SPEEK)/phosphomolybdic acid (PMA) have been prepared by solution casting technique. SPEEK/PMA membrane surfaces are modified with polyaniline (PANI) by in situ polymerization. The structural and electrochemical properties of the surface modified membranes are studied by atomic force microscopy (AFM), Fourier transform infrared, nuclear magnetic resonance, water uptake, ion-exchange capacity, proton conductivity, methanol permeability, and tensile strength. The AFM image shows that the hetero polyacids are better compatible with SPEEK matrix. The SPEEKs/PMAs membranes achieved acceptable proton conductivity with increasing in weight ratios of PMAs and reached highest proton conductivity of 5.5 × 10−3 S cm−1 at room temperature. With Lower swelling ratio, high thermal stability, proton conductivity, and relative selectivity, SPEEKs/PMAs-PANI membranes can be a promising material as Proton Exchange Membrane (PEM) for Direct Methanol Fuel Cell (DMFC) applications.
Keywords
Introduction
Fuel cell is an alternative green technology of power source which can partially solve the energy crisis issues, global warming, environmental pollution, and rapid depletion of fuel cells. 1 Fuel cell is a device which efficiently transforming chemical energy directly into electricity and is regarded as promising future clean power sources. 2 Nowadays, Nafion ionomer performed as an electrolyte in proton exchange membrane fuel cells and direct methanol fuel cells due to their high proton conductivity and good chemical, high thermal, and mechanical stability. However, it has several limitations such as high methanol permeability, high cost, and not suitable for high-temperature fuel cell operation. These limitations and catalyst cost are the major barriers for commercialization of DMFC. Nowadays, there has been a serious effort to develop a low-cost aromatic hydrocarbon to replace Nafion ionomer such as sulfonated poly (benzimidazole), sulfonated poly (arylene ether ketone), and sulfonated poly (imide). 3 –5 These are commercial polymers and exhibit excellent proton conductivity, good physicochemical properties, and remarkable mechanical and thermal stability. Among said polymers, poly (ether ether ketone) (PEEK) is a semicrystalline polymer and has a very high thermal stability as well as mechanical stability. Chemically, it is an aromatic polymer with non-fluorinated polymer backbone. 6 The PEEK can be functionalized by sulfonation, and the degree of sulfonation (DS) can be controlled by the reaction time and controlled temperature of concentrated sulfuric acid (H2SO4; 97%). Sulfonated aromatic polymers that are typically used in Proton Exchange Membrane Fuel Cell (PEMFC) are suitable materials as a host material for hetero polyacids (HPAs). HPAs are mostly used inorganic materials that contain transition metal oxygen anion clusters. They exhibit a wide range of well-define molecular structures, surface charge densities, and chemical and electronic properties. 7 The inherent Keggin structure of HPAs is known to be highly active catalysts for both acid catalyzed and oxidation reaction. 8,9 HPAs are used in hybrid composite membranes for fuel cells because of their good proton conductivity at room temperature (0.02–0.1 S cm−1). Phosphomolybdic acid (PMA) has been characterized as solid acids with high proton conductivity, very high dense, and hydrogen-bonded hydration network. 10 HPAs in the solid state are pure bronsted acids stronger than the conventional solid acids such as silicon dioxide, titanium dioxide, and some clays. 11 However, HPAs have one major drawback for the application of fuel cell, that is, high solubility in polar medium such as water. The higher solubility exhibits to the leaching problems that can cause a severe effect on the cell performance. As a result, a lot of research on these materials has focused to reduce the leaching problem and on keeping HPA within the membrane. However Nakamura et al. 12 reported on a fuel cell with H2/O2 utilizing PMA as the solid electrolyte and producing a maximum power density of 100 mA cm−2 at 0.5 V. Uma and Nogami 13 also studied on a glass electrolytes based on PMA as a solid proton conducting materials as a result high hydrogen permeability and high proton conductivity. Nagarale et al. 14 reported on sulfonated PEEK (SPEEK)-polyaniline (PANI) as a proton exchange membranes. The present work reports on SPEEK-PMA membrane using PANI to confine the PMA in the SPEEK matrix. The PANI coated in the material is not only resolved the leaching problem but also increase the proton conductivity. The properties of PANI-coated SPEEK-PMA membranes were evaluated in terms of ion exchange capacity, water uptake, swelling ratio, proton conductivity, methanol permeability, tensile strength, and atomic force microscopy (AFM) studies.
Experimental procedure
Materials
PEEK was obtained from Victrex Pvt Ltd (Mumbai, Maharashtra, India); concentrated H2SO4, PMA, aniline, and hydrochloric acid (HCl; purity 85%) were procured from SRL (Mumbai, Maharashtra, India), and N-methyl-2-pyrollidone (purity 90%) from Merck, India, has been used as a solvent for the preparation of membrane.
Sulfonation reaction process
Ten grams of PEEK were added into 200 ml of H2SO4 and the mixture was subjected under N2 atmosphere with a vigorous stirring at room temperature for 7 h. The obtained solution was poured into ice-cold deionized water to precipitate the sulfonated polymer (SPEEK), followed by filtration and thorough washing of the filtrate polymer using deionized water until the pH of water becomes 6–7. The obtained SPEEK was then dried in a vacuum oven at 90°C for 24 h. 15,16 The process is explained in Figure 1.
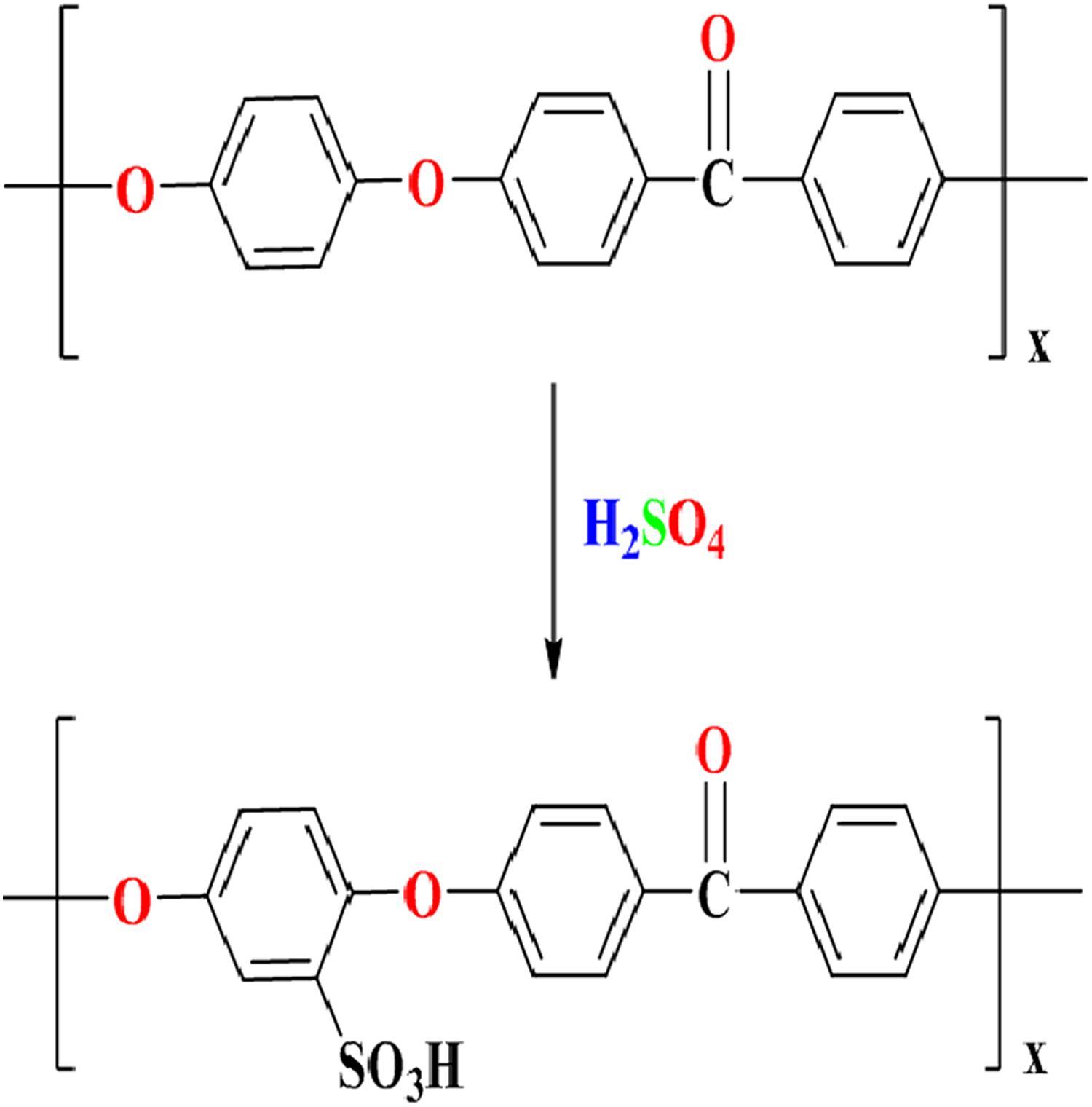
Sulfonation process for SPEEK. SPEEK: sulfonated poly (ether ether ketone).
Preparation of PANI-coated composite membranes
The surface modification of SPEEK-PMA composite membranes was carried out by in situ polymerization technique. The process is explained in Figure 2. Initially, the composite membranes were immersed in 0.05M aniline solution for 10 min at room temperature. Then, the membranes were rinsed with deionized water to remove the weakly bonded aniline monomer and subsequently immersed in mixture of 0.01M of ferric chloride and 0.01M of HCl solution for 10 min to induce the polymerization of aniline. The process was repeated to increase the number of aniline layer on the SPEEK/PMA membrane surface. The immersion time of the coated membranes was maintained constant throughout the in situ polymerization to get a uniform coating of PANI on both sides of SPEEKs/PMAs. It should be observed that PANI is an electronic conducting polymer and hence there was a panic that a continuous layer of PANI in the direction of proton transport may have resulted in electron conduction within the composite membrane that would have been detrimental to the fuel cell. Hence, the PANI concentration in this work was not more than 0.05M and the process time was only 10 min. These protections were achieved at preventing PANI infiltration into the polymer matrix and achieved a low level of PANI loading. The resulted hybrid membranes were identified as SPEEK/PMA-PANI (n) membrane, where n represents the number of process.
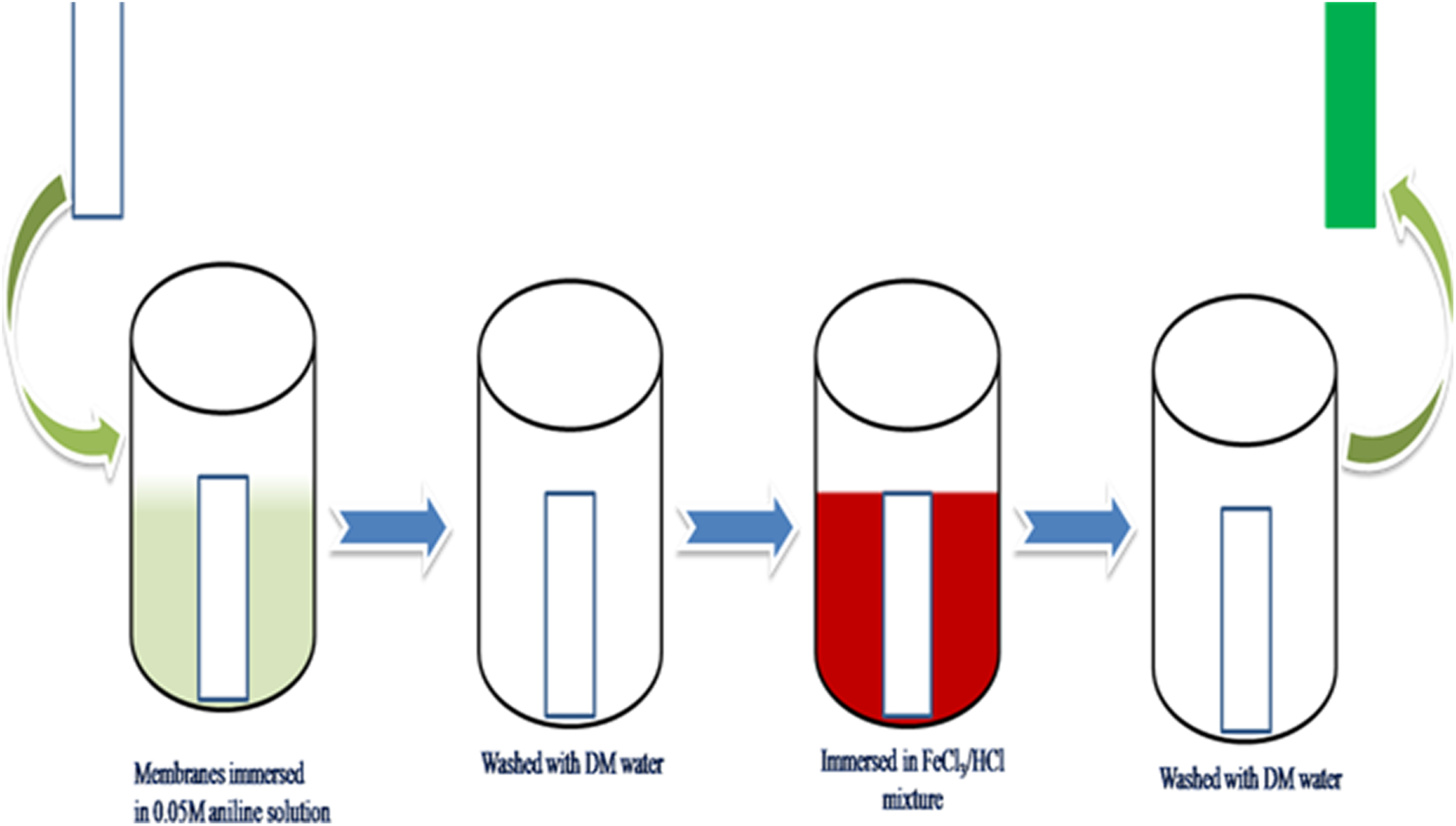
In situ polymerization of PANI-SPEEK. SPEEK: sulfonated poly (ether ether ketone); PANI: polyaniline.
Characterization of composite membranes
Elemental analysis
The elemental analysis was carried out using CHNS elemental analyzer using Elemental (Vario EL III, Germany). The elemental content of the membrane is reported as weight percent.
Structural analysis NMR—FTIR
The proton nuclear magnetic resonance (1H NMR) spectra were recorded on a Varian Unity Nova spectrometer (Nova Spectrometer) at a resonance frequency of 400 MHz for each analysis, and 2–5 wt% of polymer solution was prepared in Dimethyl sulfoxideDMSO-d6 and tetra methyl siloxane and was used as the internal standard. The Fourier transform infrared (FTIR) characterization was achieved using PerkinElmer spectrometer (RX1, Waltham, Massachusetts, USA).
Physicochemical characterization
The prepared composite membranes were dipped in distilled water to determine the water uptake of the membrane. The water uptake of the membrane was calculated from the following equation
Here, Wwet and Wdry are the weight of the wet and dry membranes, respectively.
The swelling ratios of the membranes were calculated as
where Lwet and Ldry are the lengths of the wet and dry membranes, respectively.
Classical back titration technique was used to determine the IEC of the membranes. First, the membranes in the acid form the H+ ions and were converted to sodium by immersing the membranes in 1M sodium chloride solution for 24 h at 40°C. Then, the exchanged H+ ions within the solutions were titrated with 0.01M sodium hydroxide (NaOH) solution using phenolphthalein as an indicator. The IEC values (meq g−1) of the membranes can be calculated from the following equation
The lambda value (λ) of all the composite membranes were calculated from the following equation
where Mw is the molecular weight of water (18.01 g mol−1).
The chemical stability was measured by measuring the weight loss of the proton conducting composite membrane (2 × 2 cm2) in Fenton’s solution (3-wt% hydrogen peroxide (H2O2) containing 2-ppm ferrous sulfate) at 60°C for 1 day.
Electrochemical characterization
The proton conductivity of the composite membrane was measured by the alternating current impedance technique using Multi Potentiostat (Biologic, SAS France) with an oscillating voltage of 10 mV and frequency range from 1 MHz to100 Hz. Before measuring the membrane, the samples were immersed in 1M H2SO4 and then the composite membranes were washed thoroughly with distilled water. The following equation was used for the calculation of proton conductivity
where A = 3.14 cm2 is the cross-sectional area of the PEM, l is the thickness of the membrane (mm), and Rb is the bulk resistance where obtained from the Nyquist plot.
Tensile strength measurement
The mechanical properties of the membranes were recorded using uniaxial tensile machine (Instron 3345, Buckinghamshire, UK). Membranes were cut into a rectangular shape with dimension of 6 × 1 cm2. At least three specimens from each sample were tested.
Thermal stability
Thermal stability of the polymer electrolytes was carried out using thermogravimetric analysis (TGA) model (SDT Q600) by heating under N2 atmosphere.
Atomic force microscopy
AFM studies were performed with a scanning probe microscope Agilent technology, USA, series S5500 in tapping mode. The scanning area is 1000 × 1000 nm2. The membrane samples were in dry condition, and scan rate ranging from 1 Hz to 2 Hz has been used to optimize the image quality.
Scanning electron microscopy
The morphology of composite membranes was examined by high-resolution scanning electron microscopy (SEM; FEI Quanta 250 Microscope, the Netherlands) after gold sputter coatings on a desired membrane samples.
Methanol permeability
To evaluate the methanol permeability, the polymer samples were carried out using a double chamber glass testing cell consisting of two reservoirs which is separated by an electrolyte membrane with a dense layer of SPEEK and composite membranes to reproduce a phenomenon of methanol cross over in DMFC system. The PEM is sandwiched between donor (A) and acceptor (B) compartments. Initially, the A compartment was filled with 50-ml aqueous 2M methanol solution with 50 ml of water. The solution in each bath was stirred using magnetic stirrer during measurement to keep uniform concentration. The methanol permeability was calculated by plotting methanol concentration in acceptor (B) and was recorded by measuring the refractive index using pocket refractometer. Methanol permeability was calculated from the following equation
where P is the methanol permeability (cm2 s−1), VB is the volume of the compartment B (cm3), A is the membrane area (cm2), CA is the methanol concentration in feed compartment (mol L−1), L is the thickness of the membrane (cm), and m is the slope value obtained from a plot between methanol concentrations versus time.
Relative selectivity
The relative selectivity of the composite membranes (β) can be determined using the following equation
Results and discussion
The hybrid membranes were modified by aniline using in situ as described in Figure 1. PANI is electron conducting polymer. The samples were treated by in situ aniline using Fe3+ as an oxidizing agent, and the aniline monomers were polymerized and precipitated on the surface of the SPEEK/PMA. After in situ modification with aniline, all surface modified membranes became dark green color. The elemental content of the carbon (C), hydrogen (H), and nitrogen (N) content as in weight percentage of the SPEEK and SPEEK/PMA-PANI membranes is given in Table 1. The reduction in carbon content by increasing the PANI layer is due to the addition of PANI and PMA. Nitrogen content value of SPEEK/PMA-PANI membrane increases with increasing the aniline layers as expected.
C, H, and N value for SPEEK and prepared composites.
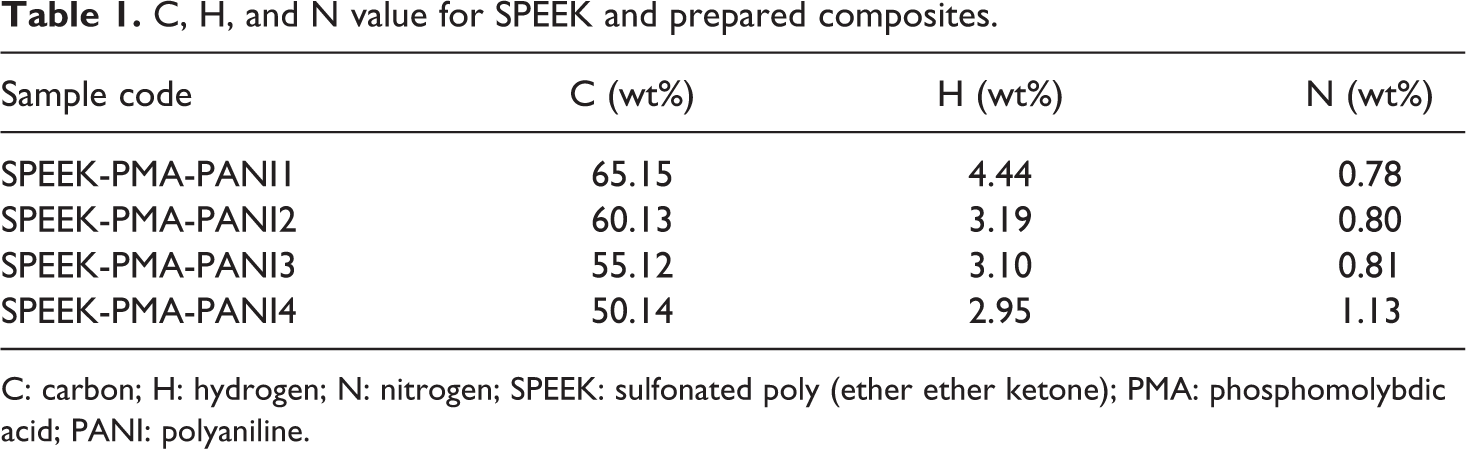
C: carbon; H: hydrogen; N: nitrogen; SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
Degree of sulfonation
IEC
The DS is defined as the percentage of repeating units of PEEK that has been sulfonated and calculated using the following equation
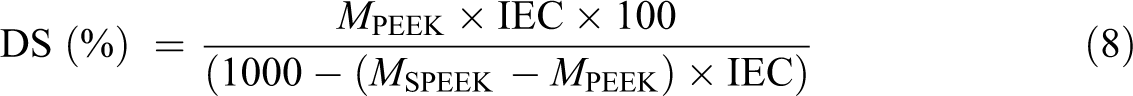
MPEEK and MSPEEK are the molecular weights of PEEK and SPEEK repeating units, and the values are 288 and 368, respectively, by substituting these values into the abovementioned equation, 17,18 the number (80) resulted from the difference between these two-unit molecular weights
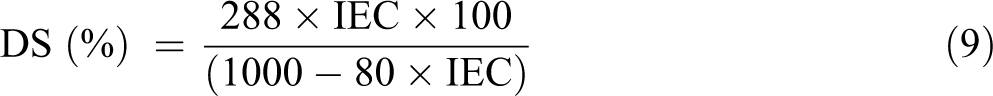
The DS was found to be 65%.
NMR analysis
The DS was determined quantitatively by 1H NMR using a variation of the method described in SPEEK polymers. The nomenclature of the aromatic protons for the SPEEK repeat unit is shown in Figure 3. The presence of each SO3H group results in a distinct signal for protons at the E position. The intensity of this HE signal yields an estimate of the HE content which is equivalent to the SO3H group content. The ratio between the peak area of the distinct HE signal (AHE) and the integrated peak area of the signals corresponding to all the other aromatic hydrogen’s (AHA, A′, B, and B′, C, D) is expressed as
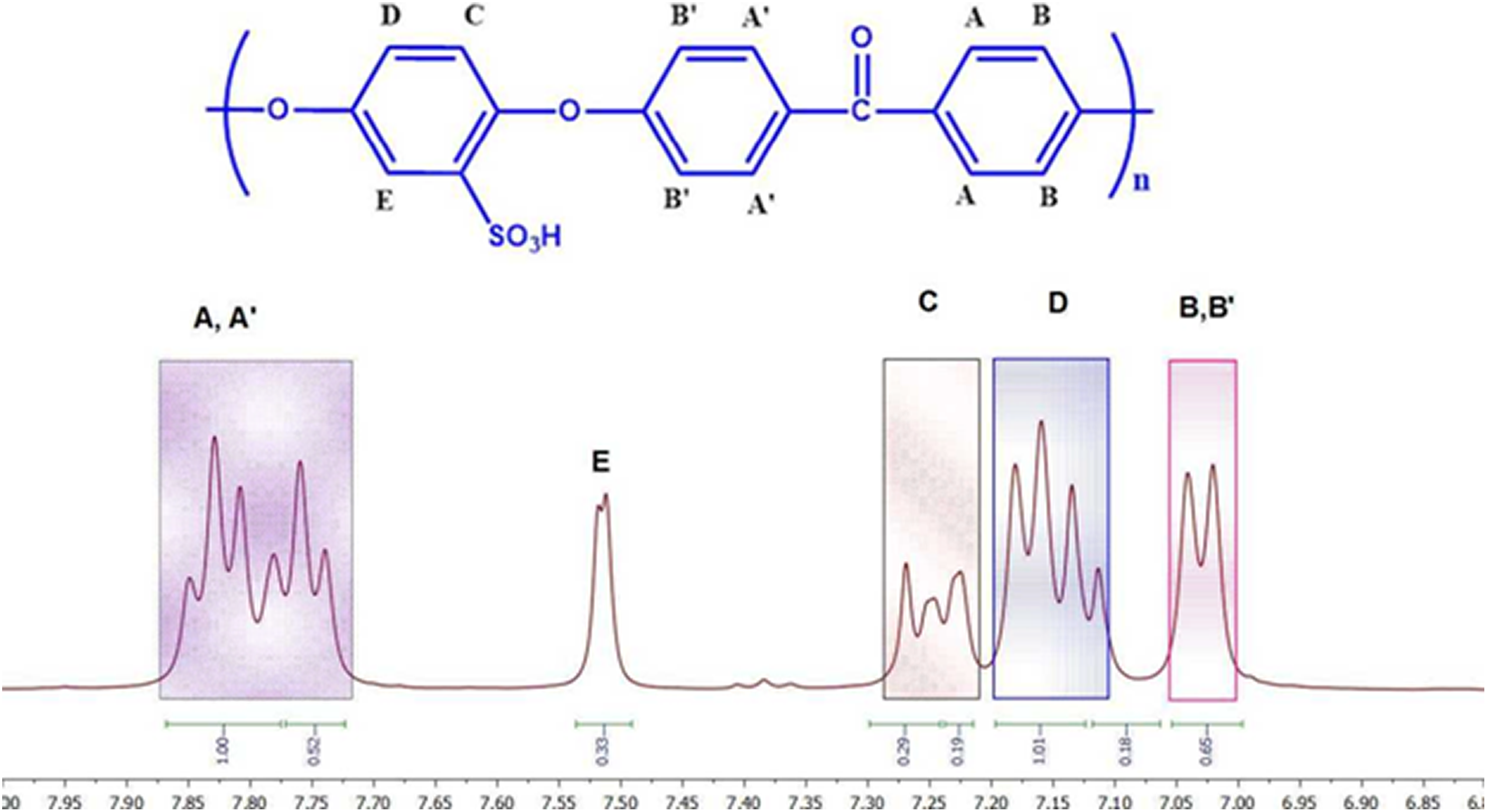
Proton NMR for SPEEK. NMR: nuclear magnetic resonance; SPEEK: sulfonated poly (ether ether ketone).
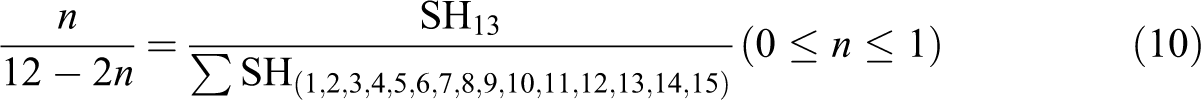
where n is the number of H13 repeat unit. SH13 is the area under the graph for H13 region and integrated peak area
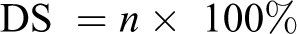
The presence of sulfonic acid group that causes a characteristic singlet (H13) at 7.55 ppm from the 1H NMR DS was also obtained as 65% for the prepared SPEEK.
Physicochemical characterizations (water uptake, swelling ratio, ion exchange capacity, durability)
Water uptake in PEM plays an important property in the transport characteristics and mechanical properties of the membrane. In a hydrated condition, sulfonated polymers can dissociate to generate immobile sulfonic acid groups and mobile protons in aqueous solution. 19 From Table 2, it is concluded that the PANI layer is increased the water uptake is decreased. From Table 2 water uptake of the prepared composites with decreasing trend in the water uptake values with increasing in the PANI layer. The decreasing tendency might be contributed to the hydrogen bond formation between the aniline and sulfonic acid groups of SPEEK. Surface modification of SPEEK/PMA with PANI led to a decrease water uptake level of the prepared polymer composites. Modifications of polymer composite membranes with a layer of PANI resulted in significant interaction with SO3H groups and so bring down the number of sites available for hydrogen bonding and form the surface less hydrophilic. 20 Water uptakes for different SPEEK-PMA-PANI composite membranes were comparable to those for Nafion 117 membranes. In Table 2, λ values decreased with increasing the number of PANI layer on the membrane surface. From the results, swelling ratio is reduced for SPEEK-PMA-PANI hybrid membrane, which is due to the hydrophobic nature of PANI layer. Ion exchange capacity indicates the charge density in the composites as presented in Table 3, and it is explained that the IEC of SPEEK-PMA composites membranes shows increasing trend with the increase in the content of HPAs than the SPEEK. Despite the sulfonic acid groups in the SPEEK matrix, the HPA particles might participate in the exchange process. The surface modification of SPEEK-PMA composite membranes by coating with PANI led to decrease in IEC values compared with that of SPEEK-PMA composite matrix. The decreasing trend was attributed to a lack of ion exchange groups in PANI and also hydrophobic nature of PANI. The IEC values of the prepared polymer composites ranged from 1.54 meq g−1 to 0.9 meq g−1 while Nafion 117 exhibited slightly lower (0.91 meq g−1). The oxidative stability is important property for the composite in fuel cell usage. The chemical stability test was performed in H2O2, and the observations are given in Table 3. From results, the PANI-coated membranes exhibit lower water uptake values than pristine SPEEK that also reduces the direct water contact to the polymer chain. As the PANI layer increases the free radicals were also increases and to attack the polymer chain. As the PANI layer increases becomes little harder for free radicals to attack the polymer chain. Therefore, compared with pure polymer membranes, hybrid membranes exhibit good oxidative stability.
Water uptake, swelling ratio and lamda value for prepared composites.

SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
Physicochemical properties of prepared composites.
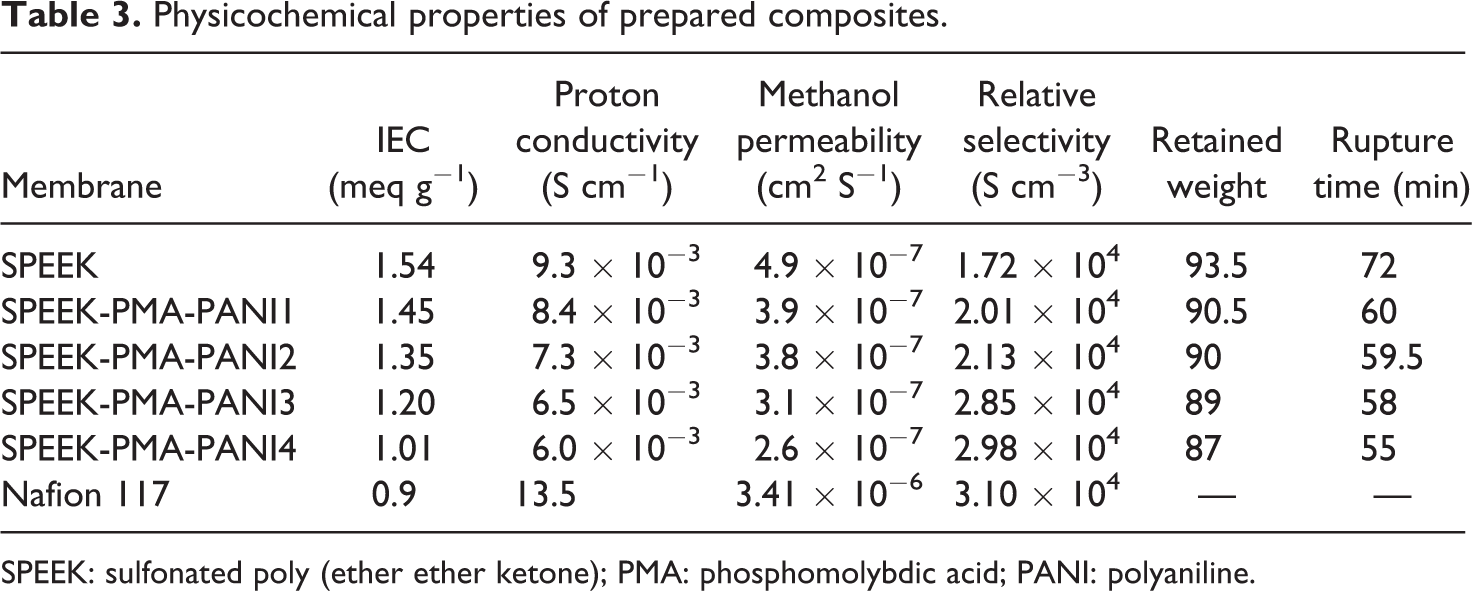
SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
FTIR analysis
Figure 4(a) shows that the broadband at 3427 cm−1 in the sulfonated polymers is indicated as the O–H stretching vibration of sulfonic acid groups. The presence of sulfonic acid groups such as 1220 cm−1 (asymmetric O=S=O), 1082 cm−1 (symmetric O=S=O), 1024 cm−1 (S=O) stretching, and 710 cm−1 (S–O) stretching confirms the successful sulfonation of SPEEK. 20 The characteristic band of PANI is 1590 cm−1 corresponding to N=Q=O (Q represents quinoid ring) and 1151 cm−1 corresponding to C–N. In the prepared samples, broadband observed in the high energy region of 3000–3900 cm−1 was due to the OH− stretch of PMA and SO3H group of SPEEK. The vibrational peaks at the wave number 1056, 1259, 1399, and 3500 cm−1 indicate the existence of PMA in the in situ polymerization. The abovementioned peaks confirmed the in situ polymerization in the prepared polymer electrolytes.
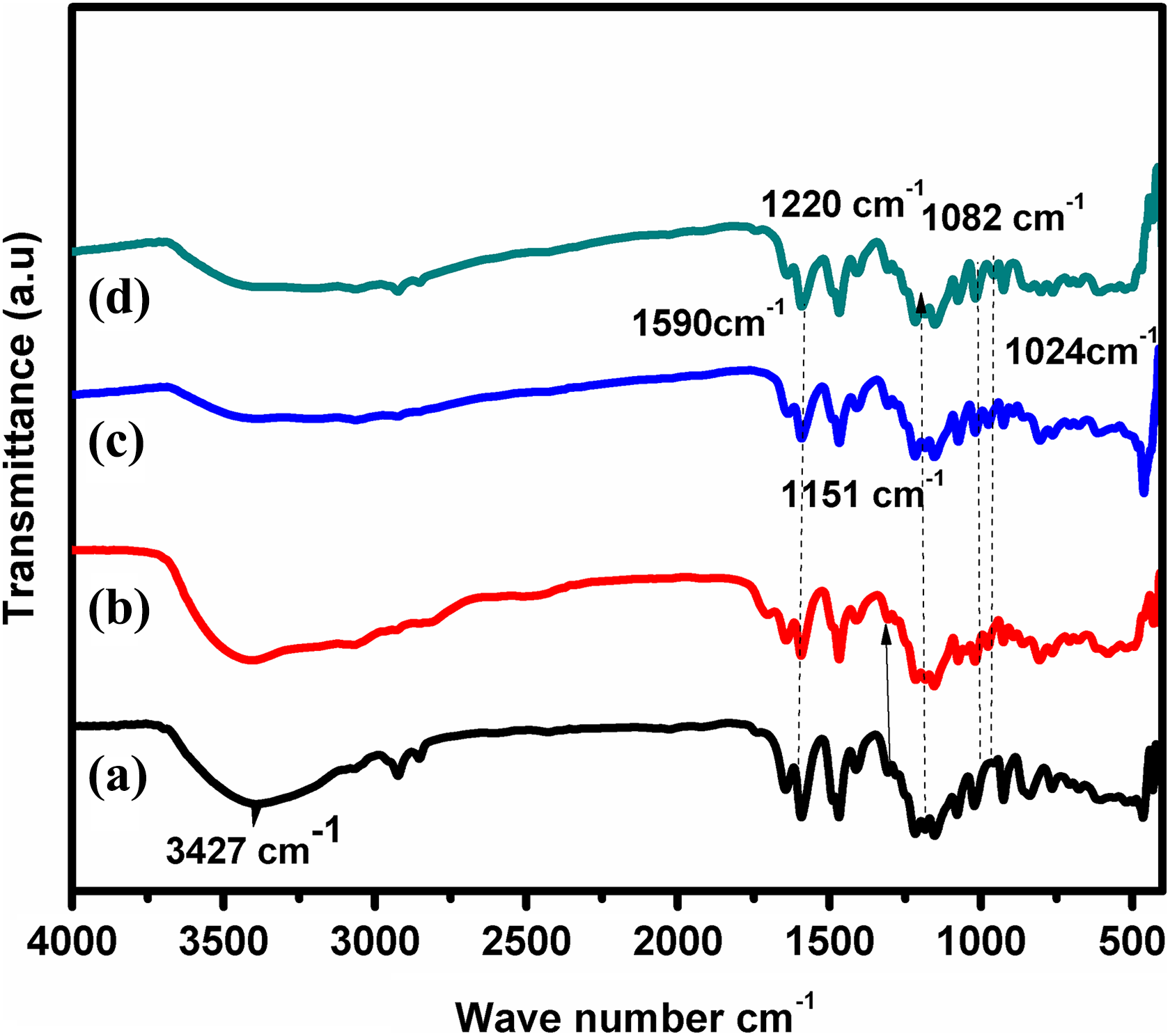
FTIR images for (a) SPEEK, (b) SPEEK-PMA-PANI1, (c) SPEEK-PMA-PANI3, and (d) SPEEK-PMA-PANI4. FTIR: Fourier transform infrared; SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
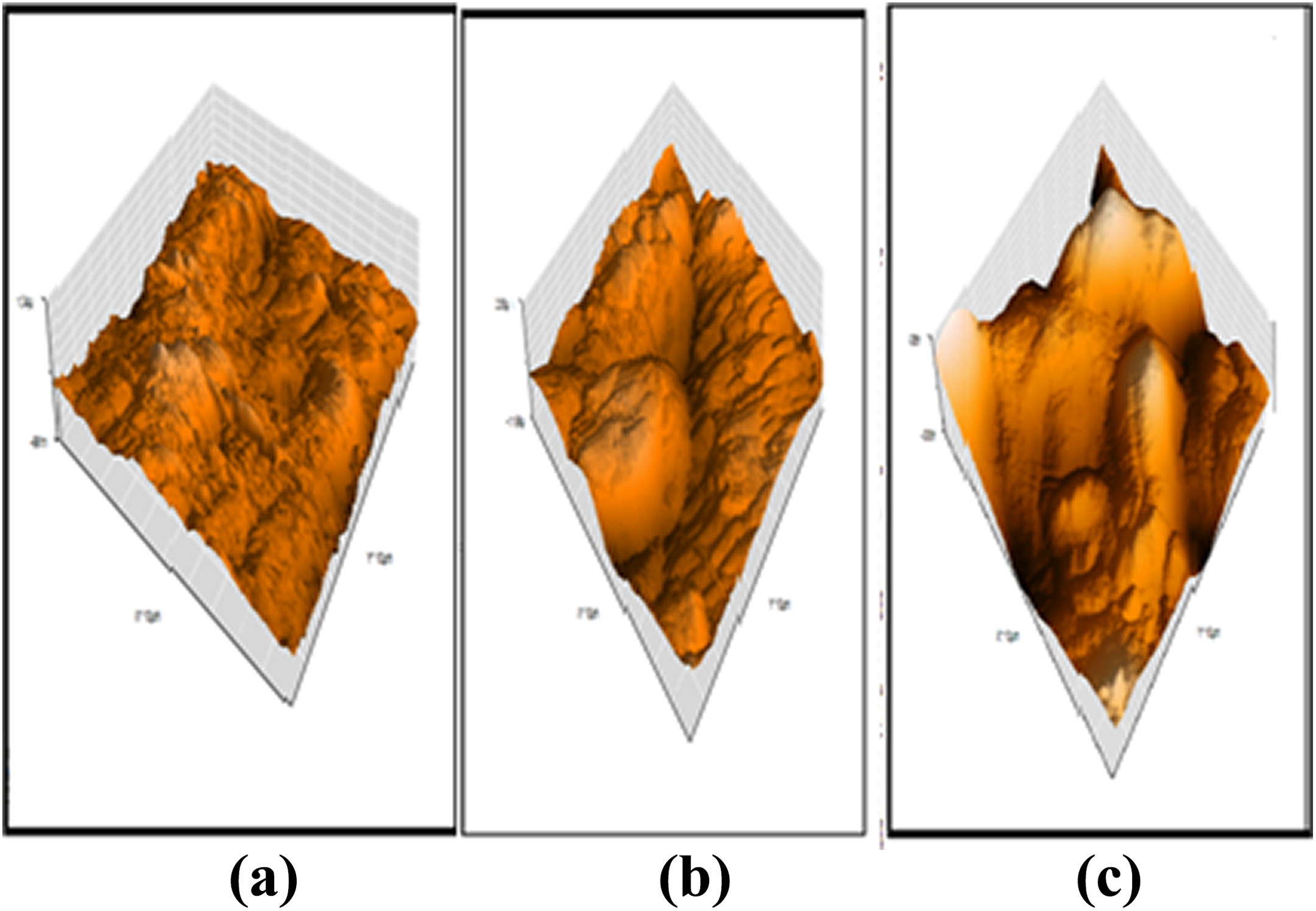
Three-dimensional atomic force microscopic image of (a) SPEEK, (b) SPEEK-PMA-PANI1, and (c) SPEEK-PMA-PANI4. SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
AFM analysis
AFM tapping mode is an excellent technique to identify the membrane surface. Surface roughness parameters were obtained from AFM image from three different locations of the samples and average values are reported. In the present study, three-dimensional topographical images of the samples SPEEK, SPEEK-PMA-PANI1, and SPEEK-PMA-PANI4 are shown Figure 5. In the present study, three-dimensional topographical images of the sample SPEEK, SPEEK-PMA-PANI1, and SPEEK-PMA-PANI4. The incorporation of PMA in the base matrix makes the surface rougher. The surface morphology of the membranes in terms of roughness (Ra) is given in Table 4. The roughness parameters of PANI-laminated composite membrane were lower than uncoated membranes. The formation of pores in the polymer composite is mainly due to the evaporation of the solvent, which is determined by the rate of solvent evaporation. 21 These pores are responsible for ion hopping conducting mechanism, and hence, it enhances the proton conductivity of the sample.
Roughness parameter and tensile strength.
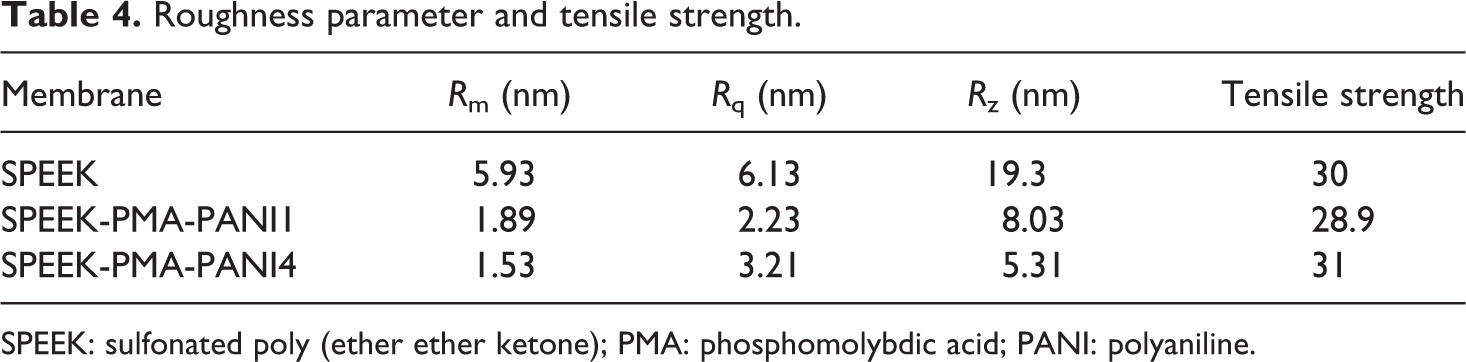
SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
SEM morphology
The morphology of the composite membranes as focused by SEM is shown in Figure 6 and Figure 6(a) indicates SPEEK which reveals no cracks in the system. Figure 6(b) to (e) explained that SPEEK-PMA-PANI4, PMA-loaded composite membrane shows smooth and uniform surface. In other words, PMA might be uniformly distributed over the SPEEK matrix which facilitates a desirable and efficient conductivity of protons through large interfacial areas between the SPEEK and super acid.

Scanning electron microscopic images for (a) SPEEK, (b) SPEEK-PMA-PANI1, (c) SPEEK-PMA-PANI2, and (d) SPEEK-PMA-PANI3, ESPEEK-PMA-PANI4. SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
Thermal analysis
The thermal behavior of the prepared composite in the present study was studied by TGA. TGA makes a continuous weighing of a given sample under a controlled N2 atmosphere with increase of temperature. From Figure 7, corresponding to SPEEK-PMA-PANI1, SPEEK-PMA-PANI4 thermogram has been obtained after heating the sample in temperature range of 40–1000°C. The first weight loss occurring at below 180°C is due to the evaporation of residual water present in the membrane. The second weight loss was occurred at 250–350°C which is attributed to the degradation of sulfonic acid groups. 22 The third major weight loss was observed at around 480°C and accredited to the polymer backbone degradation. From the thermogram, it was found that the thermal stabilities of the composite membranes followed by SPEEK-PMA-PANI1 > SPEEK-PMA-PANI4. It is observed that the thermal decomposition is well above the normal operating temperature (<100°C) for DMFCs.
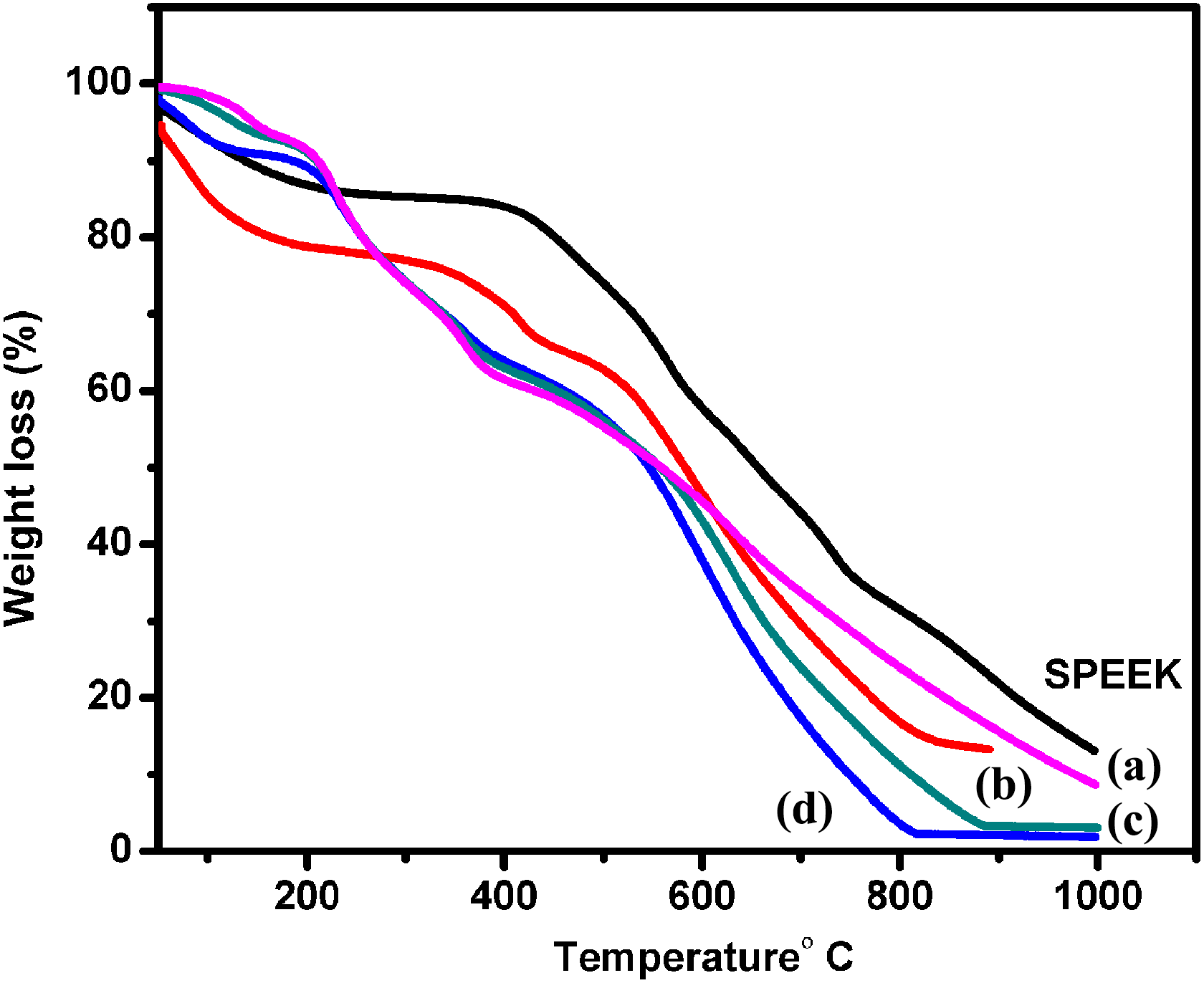
Thermogravimetric analysis spectra for SPEEK, (a) SPEEK-PMA-PANI1, (b) SPEEK-PMA-PANI2, (c) SPEEK-PMA-PANI3, and (d) SPEEK-PMA-PANI4. SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
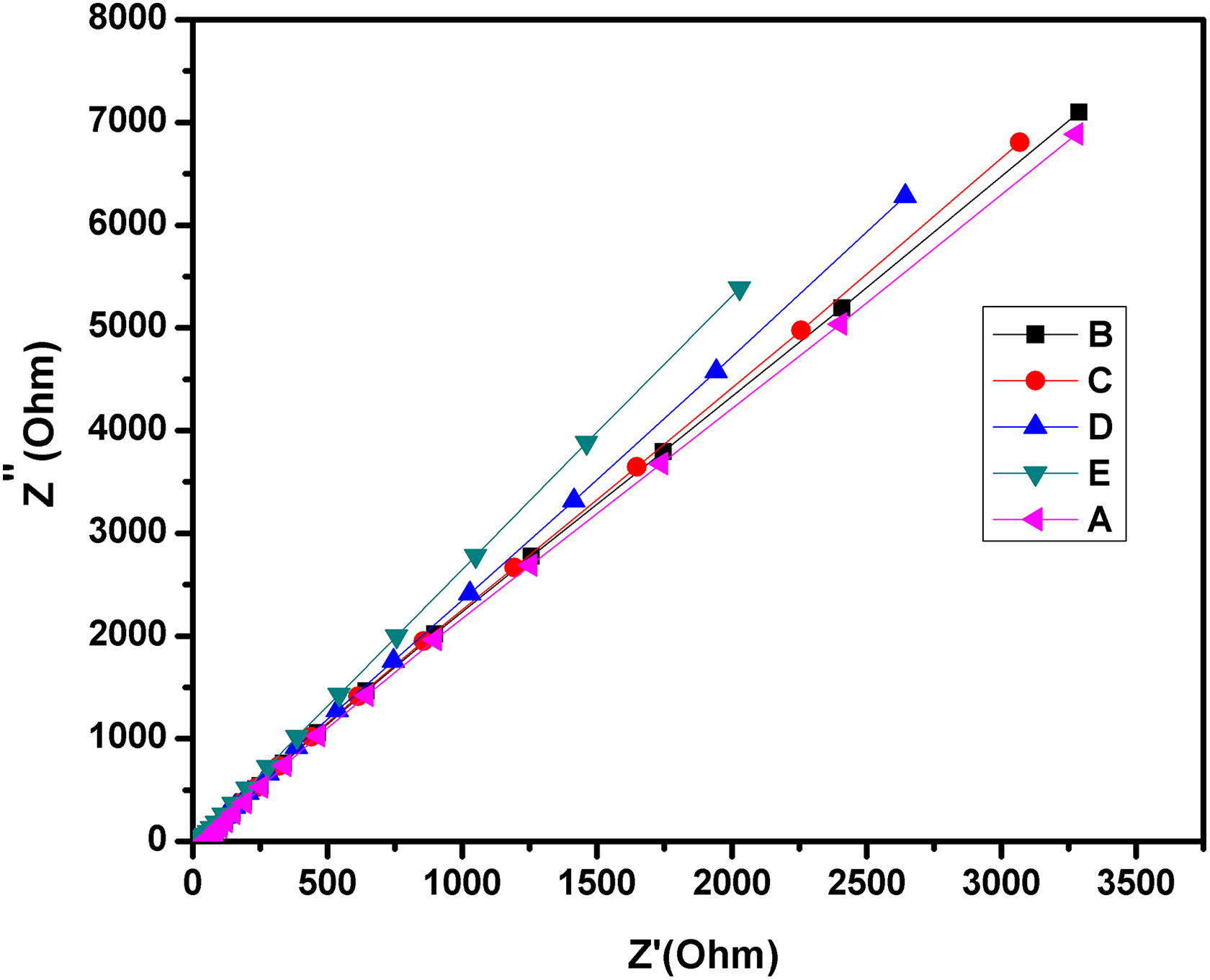
Complex impedance spectra for A: SPEEK, B: SPEEK-PMA-PANI1, C: SPEEK-PMA-PANI2, D: SPEEKPMA-PANI3, and E: SPEEK-PMA-PANI4. SPEEK: sulfonated poly (ether ether ketone); PMA: phosphomolybdic acid; PANI: polyaniline.
Mechanical stability
Tensile strength of the prepared membrane was tested at room temperature and the data are shown in Table 3. On increasing amount of PMA into the polymer electrolyte, the tensile strength is in increasing trend and was observed due to the more amount of hydrogen bonding interactions between the PMAs and the SPEEK polymer, producing a reinforcement process. The tensile strength of the SPEEK/PMA-PANI was lower than those of SPEEK matrix. The mechanical stability of the SPEEK-PMA-PANI hybrid membrane increased with increasing the number of PANI layer on the membrane matrix. All prepared polymer composites showed far better than Nafion 117 (15 MPa). These results indicate that the SPEEKs-PMAs-PANI membranes are tough enough for the PEM usage.
Proton conductivity
Complex impedance spectroscopy technique was used to determine the proton conductivity of the prepared composites. Generally, protons can be transported along hydrogen-bonded ionic channels and cationic mixtures such as H3O+, H5O2 +, and H9O4 + in the aqueous media. As it can be seen in Table 3, SPEEKs-PMAs composite membranes show much higher proton conductivity than those of pure SPEEK membrane because of the proton conductive ability of HPA present in these membranes. The hydrophilicity acid site density on inorganic nanoparticle surface and the amount of inorganic loading may have the influence on the conductivity of the composite membranes. 23 The highest proton conductivity value of 5.5 × 10−3 S cm−1 for SPEEK-PMA-PANI4 at room temperature was obtained and also shown in Figure 8. When the density of super acid groups is high, these ionic clusters become crowded in the hydrophilic membrane. As a result, the proton conductivity of the composite membrane is increased compared to pure SPEEK membrane. Proton transport in the SPEEKs-PMAs-PANIs system obeys Grotthuss mechanism, since the proton transport via Grotthuss hopping which simultaneously increases with increasing in the water content. PMAs demonstrated enhanced proton transport under increasingly dry conditions, which could imply that the PMAs facilitated Grotthuss hopping. All of these properties combined with high proton conductivity of PMA render it is a promising material for the development of proton conducting material.
Methanol permeability and relative selectivity ratio
From Table 3, for the SPEEKs/PMA-PANI membranes, methanol permeability values decrease with increasing the number of layers of PANI in the SPEEK matrix. The hydrophobic nature of PANI blocks the channel for methanol permeation through the composite membrane. Also, the modification with increasing the amount of PANI reduces the number of available sulfonic acid groups, which are essential for the formation of transport channel. The pure SPEEK membrane shows the methanol permeability of 4.9 × 10−7 cm2 S−1 at room temperature, whereas the SPEEKs-PMA-PANI (n) membranes show the permeability in the range of (3.9 × 10−7 cm2 S−1–2.1 × 10−7 cm2 S−1). The methanol permeability was decreased upon the introduction of PMA which is hydrophilic and requires no sulfonic functional groups for the formation of ion clusters and methanol transport channels. 24 The methanol permeability of Nafion 117, SPEEK, SPEEK-PMA-PANI1, SPEEK-PMA-PANI2, SPEEK-PMA-PANI3, and SPEEK-PMA-PANI4 is 3.41 × 10−6, 4.9 × 10−7, 3.9 × 10−7, 3.8 × 10−7, 3.1 × 10−7, and 2.6 × 10−7, respectively, all much lower than that of Nafion 117. It is notable that the SPEEKs/PMAs/PANIs composite membranes have less affinity toward methanol, compared to Nafion 117, which is favorable for long operation of DMFCs. These results exhibit better performance for DMFC applications. The relative selectivity ratio of proton conductivity and methanol permeability of SPEEK and composite membranes is given in Table 3, and the relative selectivity of (SPEEKs-PMA-PANI4) 2.98 × 104 is higher than the other prepared composite membranes. 25 Further increase in the PANI layer on SPEEKs-PMA membrane leads to decrease in the relative selectivity which is due to decrease in proton conductivity, and the composite membranes are attractive for DMFCs.
Conclusion
SPEEKs-PMAs-PANI hybrid PEM was successfully fabricated, in which PANI was coated on the surface of the SPEEKs-PMAs membrane by the in situ polymerization method. The SPEEK-PMA-PANI4 composite membranes showed the best results in terms of membrane selectivity defined by proton conductivity and methanol permeability which is relevant parameter for DMFC. The mechanical properties of the prepared composite membranes increase with increasing the PANI layer. In addition, the mechanical behavior of the prepared polymer composite membranes is better than commercial Nafion 117. It was confirmed that these composite membranes were suitable for direct methanol fuel cell application and as electrolyzers.
Footnotes
Acknowledgement
The authors would like to thank Alagappa University, Karaikudi, for funding RUSA 2.0, for funding
Authors’ contribution
M. Ramesh Prabhu and P. Sivakumar contributed equally.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Alagappa University, Karaikudi (RUSA 2.0).