Abstract
Biocomposites are sustainable composite materials that, owing to their many benefits, have attracted interest of industry. In the present research, the durian husk fiber (DHF) was used as natural fiber in poly(lactic acid) (PLA) biocomposites. This study focused on the effects of fiber and processing aid content on the processing torque, tensile, thermal, and morphological properties of PLA/DHF biocomposites. The biocomposites with high fiber content have strength and modulus that are suitable for nonstructural application. The processing aid used was Ultra-Plast XP519. The addition of Ultra-Plast XP519 significantly improved the processing by lowering the torque, but it brought negative effect on mechanical properties.
Introduction
Biocomposites are composite materials that are produced from bio-based polymer (as matrix) and natural fiber. 1 Biocomposites are also a green and sustainable material since they are derived from natural and renewable resources. 2 Biocomposites have received significant interests in recent years due to new legislation that promotes more environmentally sustainable manufacturing technologies as well as reuse and recycling of materials. 3 The increase of environmental awareness and deletion of fossil resources have significantly driven the biocomposites market to high volume applications. In many cases, biocomposites offer numerous advantages, such as reduction in weight and cost, positive impact on agriculture, and less reliance on fossil resources. 3,4 Recently, biocomposites are widely used in many applications, including automotive, construction, furniture, and packaging industries. 3,5,6 The German company Tecnaro is the best example of biocomposites manufacturer for 15 years that have used various types of bio-based polymer as matrix and natural fibers to produce biocomposites. 5 In addition, biocomposites that are limited to nonstructural applications are usually constituted by bio-based polymer (e.g. poly(lactic acid) (PLA) and polyhydroxylkanoates) matrix biocomposites reinforced with short and randomly oriented natural fibers, such as wood fiber, palm oil empty fruit fiber, and rice husk fiber. 7,8 Generally, these biocomposites are produced by melt compounding and molding processes. Besides, such biocomposites for nonstructural applications are typically lower in mechanical strength but higher in stiffness compared to its matrix. 3
Durio zibethinus Murray, which is also commonly known as durian, is a tropical fruit that is highly produced in Southeast Asia region especially in Malaysia, Thailand, Indonesia, and Philippines. 9 Durian husk is the shell of the fruit which comprises 60% of the whole fruit. 10 The husk is a waste that is going to be disposed after the flesh of the fruit is consumed. The production of durian fruits are predicted to achieve 22,000 metric tons in year 2020. 11 Assuming that 60% of total amount of durian fruit produced is husk, it is estimated the durian husks waste produced in year 2020 will achieve 13,200 metric tons. The conventional disposal method of durian husks is either open burning or landfill which leads to serious environment pollution. 12 Thus, turning the huge amount of husk into benefit biomass saves the environment, cost, and energy. The husk contains 60.45% of cellulose, 15.45% of lignin, and 13.09% of hemicellulose, and the content is similar to wood fiber. 13 Therefore, this research aimed to produce biocomposites from durian husk fiber (DHF). Although some literatures of PLA/DHF biocomposites have been reported by Manshor et al. 14 and Penjumras et al., 10 the research on biocomposites with DHF is still limited. In these literatures, the biocomposites contained less than 35-wt% fiber content. This research utilized more DHF for the biocomposites.
The compounding process of biocomposite materials is challenging due to the high fiber content. It is difficult to ensure a good fiber dispersion and strong interfacial adhesion between fiber and matrix. Plastic additives such as coupling agent (e.g. maleated polymer), 15 lubricant (e.g. mineral oil), 16 and processing aid (e.g. stearic acid) 17 are commonly added upon the melt compounding process in order to improve the quality of mixture. Processing aid is an important plastic additive for melt compounding process of biocomposites. Generally, the processing aid is a polyolefin compound which is able to reduce the cost by improving the processability and improving the quality of compound. 18 The studies related to the effects of coupling agent and chemical treatment on the properties of PLA biocomposites were found in numerous of literatures. However, the effect of processing aid on PLA biocomposites is rarely reported.
The effects of fiber and processing aid content on the tensile, thermal, and morphological properties of PLA/DHF biocomposites are discussed in this article.
Methodology
Materials
The PLA resin was obtained from TT Biotechnologies Sdn. Bhd., Malaysia and processing aid used for the experiment was Ultra-Plast XP519, which was supplied by Performance Additives Sdn. Bhd., Malaysia. Moreover, the durian husks were collected from durian stalls at Petaling Jaya, Malaysia.
Preparation of DHF
First, the collected durian husks were cleaned and cut into small pieces. The small pieces of durian husks were dried using an oven at 70°C. The weight of durian husks was measured every 2 h until it became constant which indicated the husk had fully dried. After the durian husks were completely dried, they were grinded into short fibers by surface grinder and passed through a sieve with a mesh size of 600 µm to get a homogenous fiber size. The DHFs were dried again before compound with PLA resin.
Preparation of biocomposites
The biocomposites were prepared by melt compounding method using Haake Rheomix 600p (ThermoFisher Scientific, USA). The operating temperature was set at 180°C and the rotor speed was fixed at 80 r min−1. The total mixing time was 10 min. All the samples were compounded according to the formulation listed in Table 1. The compounding procedures are as follows: (i) the PLA resin was transferred into mixing chamber for 2 min; (ii) then, the DHFs were added to melted PLA and mixed for another 8 min; and (iii) finally, the compound was removed from the mixing chamber. The processing data (torque versus time) were recorded during the compounding.
Formulation of PLA/DHF composite with and without Ultra-Plast XP519.

PLA: poly(lactic acid); DHF: durian husk fiber; phr: part per hundred resin.
Then, the compound was molded into 1-mm thickness sheet using hot press machine (model Moore) at a temperature of 180°C. The molding procedures were (i) preheating to soften the compound for 1 min; (ii) fully compressed the softened compound under 100 kPa pressure for 1 min; and (iii) finally, cooling specimen under the same pressure until temperature was below 50°C and removed the sheet from the mold. All the biocomposite sheets were cut into tensile specimens with dimensions referring to ASTM D638 type IV.
Testing and analysis
Tensile tests were carried out according to ASTM D638 standard. The tensile properties of the biocomposite were tested using Instron Universal Testing Machine (model 5569, Norwood, Massachusetts, USA). A crosshead speed of 5 mm min−1 and a 50-kN load cell were used to perform the test. The load and extension of the specimens were detected and recorded by a computer.
The fracture specimens of biocomposites were examined using FEI Quanta 400F field emission scanning electron microscope (FESEM, Thermo Fisher Scientific Inc., USA). The fracture specimens were sputter coated with ultrathin layer of palladium to prevent charging of the specimen. The acceleration voltage was set at 10 keV.
Thermogravimetric analysis was carried out by Pyris Diamond thermogravimetric analysis (TGA; brand: PerkinElmer, Waltham, Massachusetts, USA). The specimen was cut into small pieces (7 ± 2 mg) and placed on a ceramic pan. The specimen was then sent under thermal scanning from 30°C to 600°C with a heating rate of 10°C min−1 under purified nitrogen atmosphere where the atmosphere flow rate was set at 20 ml min−1.
Differential scanning calorimetry (DSC) analysis was performed using DSC equipment (brand: PerkinElmer). The specimen was cut into small piece (7 ± 2 mg) and placed in a closed aluminum pan. Then, the sample was heated from 30°C to 160°C with a heating rate of 10°C min−1. The process was carried out under nitrogen atmosphere where the nitrogen gas flow rate was set at 20 ml min−1. The glass transition temperature (T g), melting temperature first peak (T m1) and second peak (T m2), cold crystallization temperature (T cc), enthalpy of cold crystallization (ΔHcc ), and enthalpy of fusion (ΔH f) were obtained from the DSC curves. In addition, the crystallinity of biocomposite (X c) was calculated using the following equation
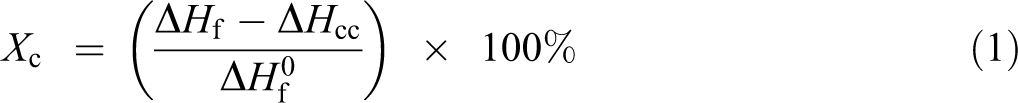
where
Results and discussion
Processing torque
The processing torque against time curves of PLA/DHF biocomposites with different fiber and Ultra-Plast XP519 content are shown in Figure 1. According to Figure 1(a), the first sharp peak was found at first minute and the processing torque raised rapidly once the solid PLA resin was added. Then, the torque gradually decreased after the solid PLA resin started to melt due to high processing temperature and continuous shearing action. The DHF was added at time 2 min resulting in a second processing torque peak. This is because the addition of DHF has disturbed the flow of melted PLA. After that, the processing torque decreased slowly and achieved a stabilized torque. This indicates the PLA/DHF had become a homogenized compound. Figure 1(a) shows that the processing torque at time 10 min increased with increasing fiber content. The friction between fiber and molecular chains increased with the increase of fiber content. Hence, the increasing amount of fiber will hinder the melt flow of melted PLA. A similar finding on processing torque was also found in other literatures. 20,21 From Figure 1(b), the stabilized torque of biocomposites was significantly decreased when added with more Ultra-Plast XP519. The addition of 2 phr of Ultra-Plast XP519 reduced 15% of processing torque. The Ultra-Plast XP519 is a compound mainly containing fatty acid amide which is able to work as lubricant for the compound. This fatty acid amide might be present between the fibers and the melted PLA and this highly reduced the friction between fiber and matrix. Besides, the fatty acid amide also lubricated the compound by reducing the friction with mixing chamber wall. For this reason, the processing torque of compound was significantly reduced. Chun et al. 20 also reported the addition of stearic acid lubricating the biocomposites compound during the process by lowering the processing torque.
Torque versus time curves of PLA/DHF biocomposites with (a) different fiber contents and (b) Ultra-Plast XP519 content.
Tensile properties
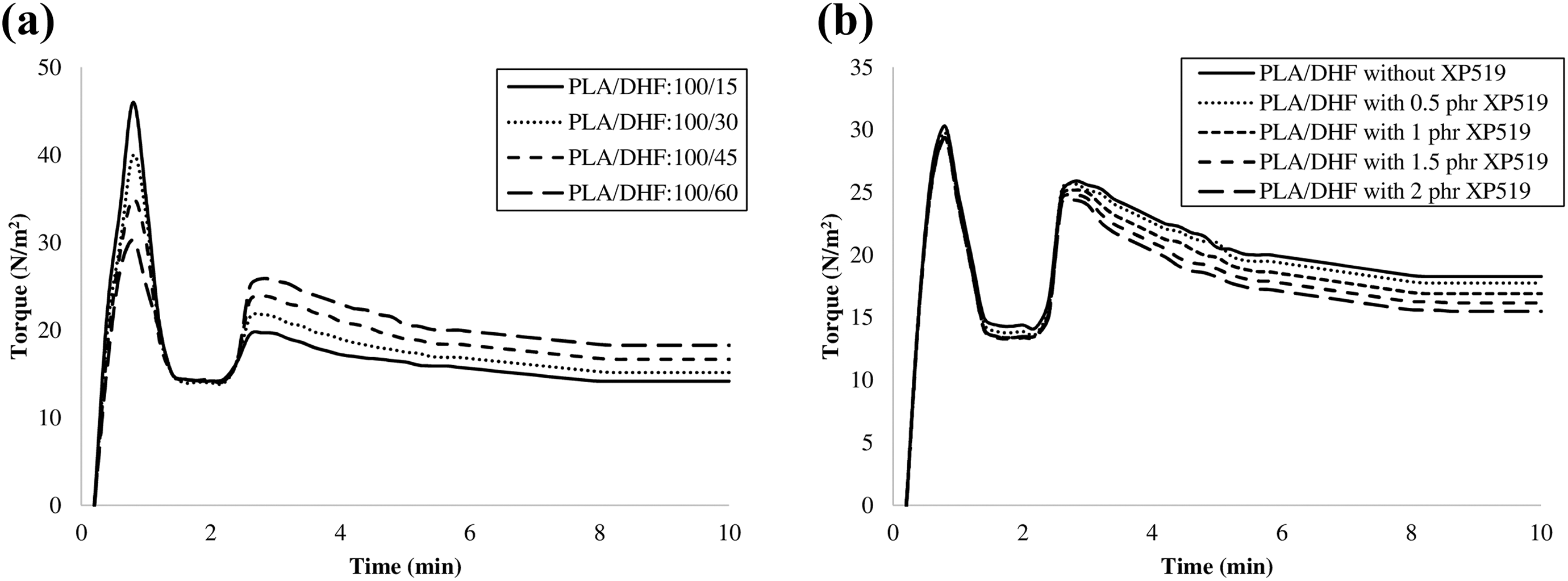
Figure 2(a) shows the tensile strength of PLA/DHF biocomposites with different DHF content. The fiber obtained from durian husk was short fiber. Generally, the efficiency of short fibers to act as reinforcement depends on fiber orientation. In general, short fibers have a random fiber orientation and only fibers that are aligned parallel to the direction of axial load are able to act as load bearing component in the composite. 22 From the results, the tensile strength of PLA/DHF biocomposites exhibited an increasing trend as the fiber content increases. Usually, natural fiber displays a higher mechanical strength compared to most plastic material. 23 Thus, the addition of more DHF will increase the strength of biocomposites. Moreover, the addition of more DHF might have more short fiber aligned parallel to the direction of stress. Hence, part of the short fibers can share the axial load subjected on the PLA matrix and increased the tensile strength of biocomposites. Similar findings have also been reported by other researchers. 14 –24 The tensile modulus of PLA/DHF biocomposites with different fiber content is displayed in Figure 1(b). The tensile modulus of the biocomposites gradually increased with increasing amount of DHF. It is because the modulus of DHF is higher than PLA matrix. Therefore, the presence of higher amount of DHF contributed to higher modulus of the biocomposites. Moreover, Gunti et al. 25 also agreed that the modulus of PLA biocomposites is highly depending on the amount of filler whereby the PLA/jute biocomposites with higher fiber content showed a higher tensile modulus.
(a) Tensile strength and (b) tensile modulus of PLA/DHF biocomposite with different fiber contents.
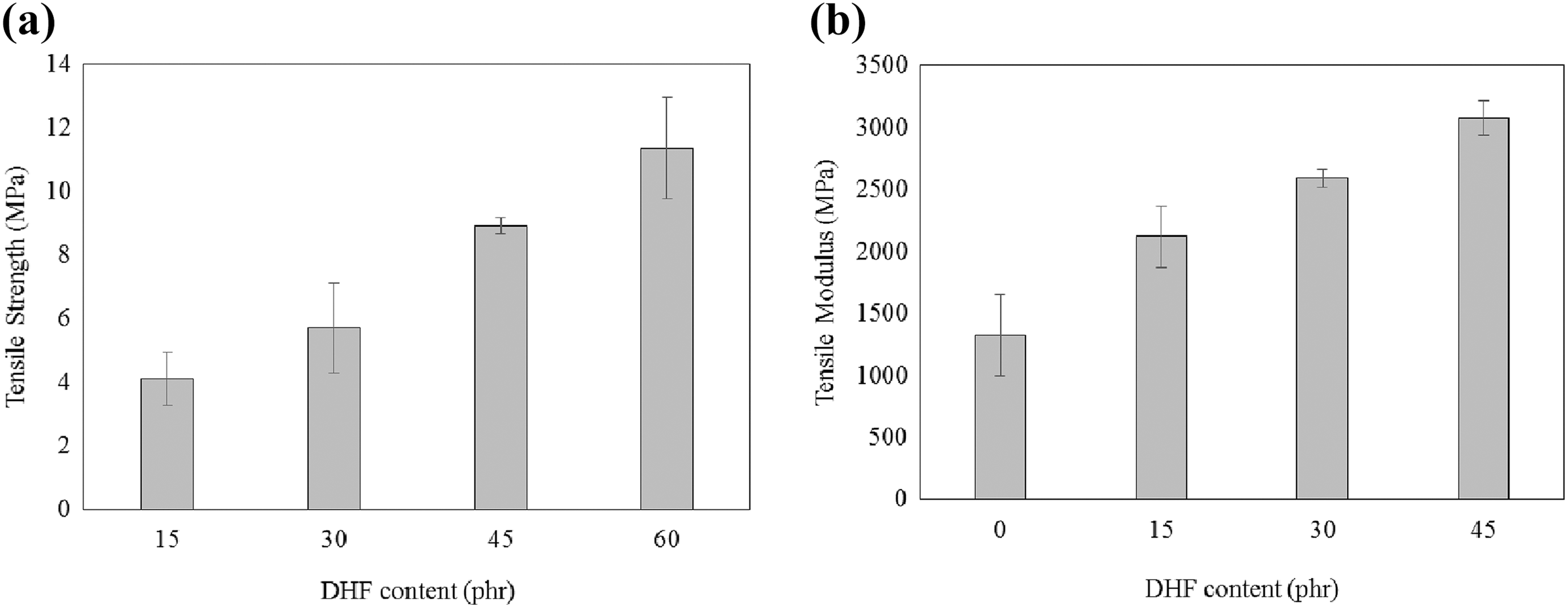
Figure 3(a) shows the effect of Ultra XP-519 on the tensile strength of PLA/DHF biocomposites with 60 phr of DHF. Overall, the results proposed an almost linear decrement with the increased Ultra-Plast XP-519 content. The Ultra-Plast XP519 is a compound of fatty acid amide which behaves as lubricant for biocomposites. The amide groups from the Ultra-Plast XP519 will attach on fiber surface by forming hydrogen bonding with hydroxyl groups from DHF, as shown in Figure 4(a). This statement also agreed with study reported by Chun et al. 26 Hence, the presence of Ultra-Plast XP519 will form a fatty acid layer that covers the fiber surface as illustrated in Figure 4(b). The presence of this fatty acid layer will also provide a lubricating effect at the fiber–matrix interface region. From Figure 5(a), a mechanical interlock is usually present between fiber and matrix, and it is also known as interfacial bonding. This bonding allows the stress transfers between matrix and fiber. However, the presence of fatty acid layer on fiber surface may have inhibited the formation of mechanical interlock between PLA matrix and DHF surface. As a result, the stress transfer at interface region became weak and caused the tensile strength of biocomposites to decrease. Chun et al. 26 also found that the strength of biocomposites tends to decrease when the filler surface was fully covered by fatty acid compound.
(a) Tensile strength and (b) tensile modulus of PLA/DHF biocomposite with different Ultra-Plast XP519 contents.
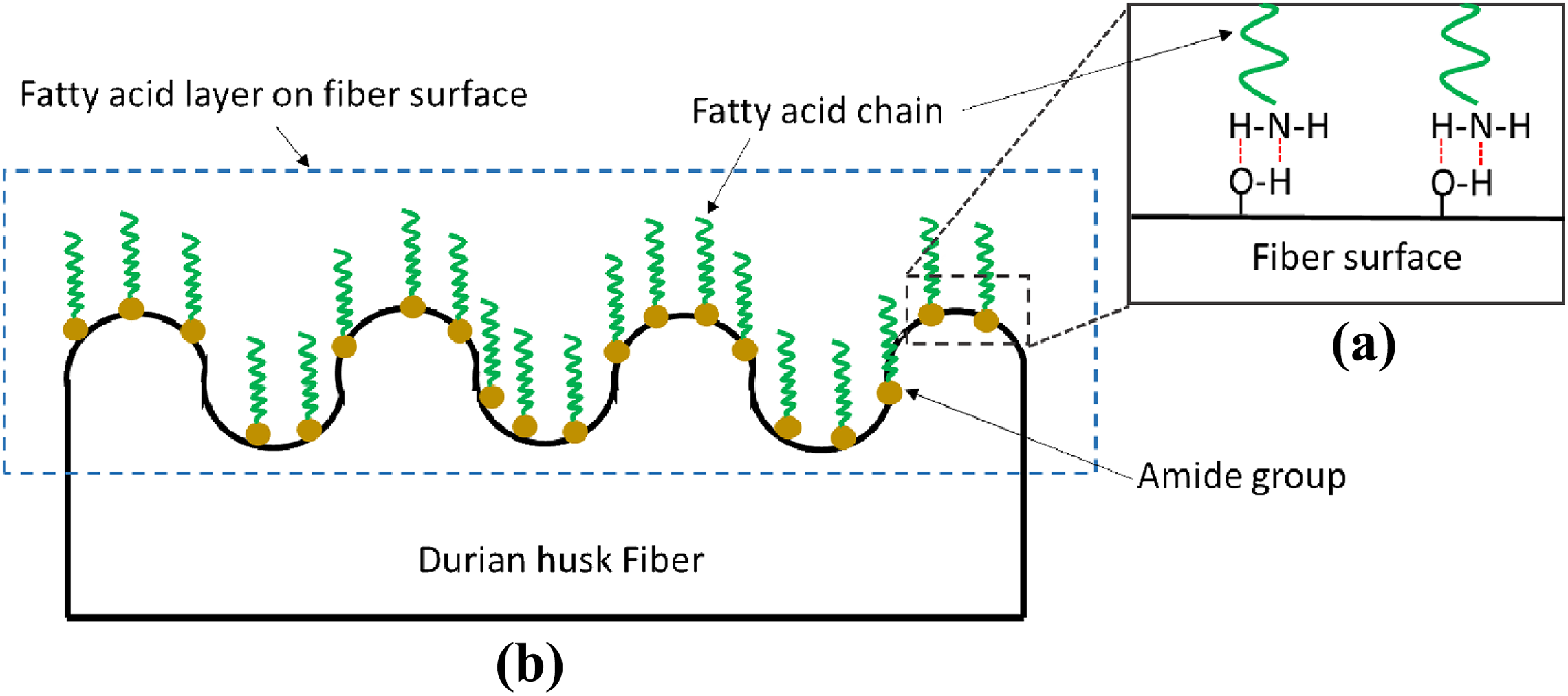
Proposed schematic for (a) formation of fatty acid layer when Ultra-Plast XP519 reacted with DHF and (b) the Ultra-Plast XP519 attached on fiber surface via hydrogen bonding.
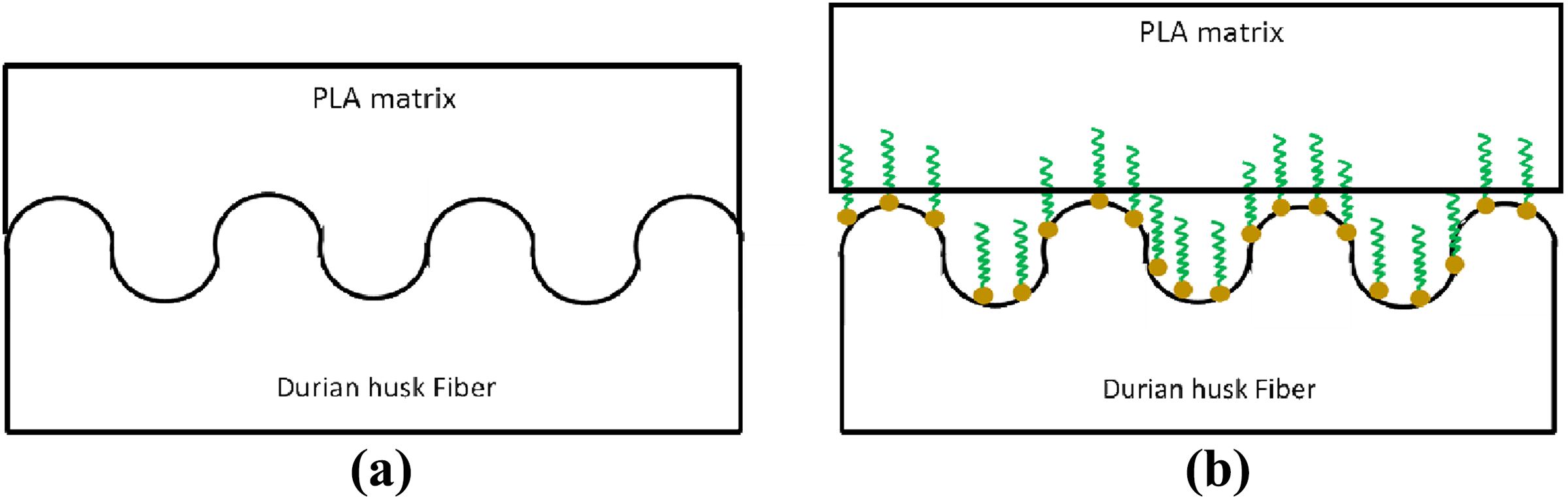
(a) Formation of mechanical interlock at fiber–matrix interface and (b) of fatty acid inhibited the formation of mechanical interlock at fiber–matrix interface.
The results of tensile modulus against processing aid content are illustrated in Figure 3(b). It can be clearly observed that the tensile modulus increased with the addition of Ultra-Plast XP519 up to 0.5 phr, but the tensile modulus decreased when the Ultra-Plast XP519 content further increased. Generally, the increase of tensile modulus in biocomposites is due to either (i) a good interfacial locking between fiber and matrix or (ii) a premature failure occurs. 27 The average tensile modulus of PLA/DHF biocomposites with Ultra-Plast XP519 was 2.8% higher compared to PLA/DHF biocomposites without Ultra-Plast XP519, which is not significant. In this case, the increase of tensile modulus in PLA/DHF biocomposites might be due to the presence of premature failure. At higher Ultra-Plast XP519 content, the tensile modulus decreased. This is because of the lubricating effect from fatty acid layer that occurred between fiber and matrix. Panthapulakkal et al. 28 also reported that processing aid reduced the adhesion between the matrix and filler and reduced the tensile modulus of biocomposites due to lubricating effect.
Morphological properties
Figure 6 shows the SEM micrographs of PLA/DHF biocomposites with 30 and 60 phr of fiber content. From Figure 6, the fiber was oriented parallel to the direction of axial load. The gaps found between fiber and PLA matrix indicated poor interfacial bonding. This is because the fiber is hydrophilic and PLA matrix is hydrophobic, thus both are incompatible to each other. Even though, there are weak fiber–matrix bonding, the fibers are still embedded in the matrix after fracture, which proves that the fibers are able to act as load bearing component in biocomposites. SEM micrographs of PLA/DHF biocomposites with different Ultra-Plast XP519 content are displayed in Figures 7 and 8. Fibers pull out were observed in Figures 7(a) and 8(a). According to Figures 7(b) and 8(b), a gap was also found at the interfacial region. Figure 8(b) shows that the gap was prolonged along the interface. This verified that the DHF had very poor interaction with PLA matrix when more Ultra-Plast XP519 was added. This observation agreed with the findings from tensile results as the presence of Ultra-Plast XP519 caused a weak interfacial adhesion between the fiber and the matrix. Besides, matrix cracking was observed in Figure 7(b) and the cracking propagated from the gap between fiber and matrix. It is believed that the cracking was initiated at poor interfacial region when subjected to axial load. This observation agreed that the poor interfacial mechanical interlock between fiber and matrix caused a premature failure of the biocomposites.

SEM micrographs of PLA/DHF biocomposite with (a) 30 phr and (b) 60 phr of fiber content.

SEM micrographs of PLA/DHF biocomposite with 1 phr of Ultra-Plast XP519 at a magnification of (a) 250× and (b) 800×.

SEM micrographs of PLA/DHF biocomposite with 2 phr of Ultra-Plast XP519 at a magnification of (a) 250× and (b) 800×.
Thermal properties
The TGA curves for pure PLA, DHF, and PLA/DHF biocomposites with different DHF content are shown in Figure 9(a). Table 2 lists the summary of data collected from the TGA curves. According to the results, the DHF has the lowest degradation temperature at 5% of weight loss (T d5%) compared to PLA. This is due to the weight loss from volatile compound and moisture in DHF. The T d5% was shifted to lower temperature when DHF content increased. This is because the PLA/DHF biocomposites exhibited an early thermal degradation compared to pure PLA. This early thermal degradation was due to the weight loss from components in DHF which includes (i) volatile compound and moisture and (ii) hemicellulose. In addition, the degradation temperature at 50% of weight loss (T d50%) of PLA/DHF biocomposites was lower than neat PLA. The increase of DHF content reduced the T d50% of biocomposites, which means that the thermal stability of biocomposites decreased with more DHF. The thermal degradation at T d50% was attributed to degradation of lignin and cellulose. At temperature of 600°C, the degradation of DHF yielded about 22.7% of char. Hence, the addition of more DHF in biocomposites results in more char residual at 600°C. Similar findings were also reported by Manshor et al. 14 The authors stated that the char residue is formed after the lignin and cellulose decomposed at high temperature. Also, the char residue of the PLA/DHF biocomposites is higher than the pure PLA due to the unremoved lignin and cellulose in the DHF.
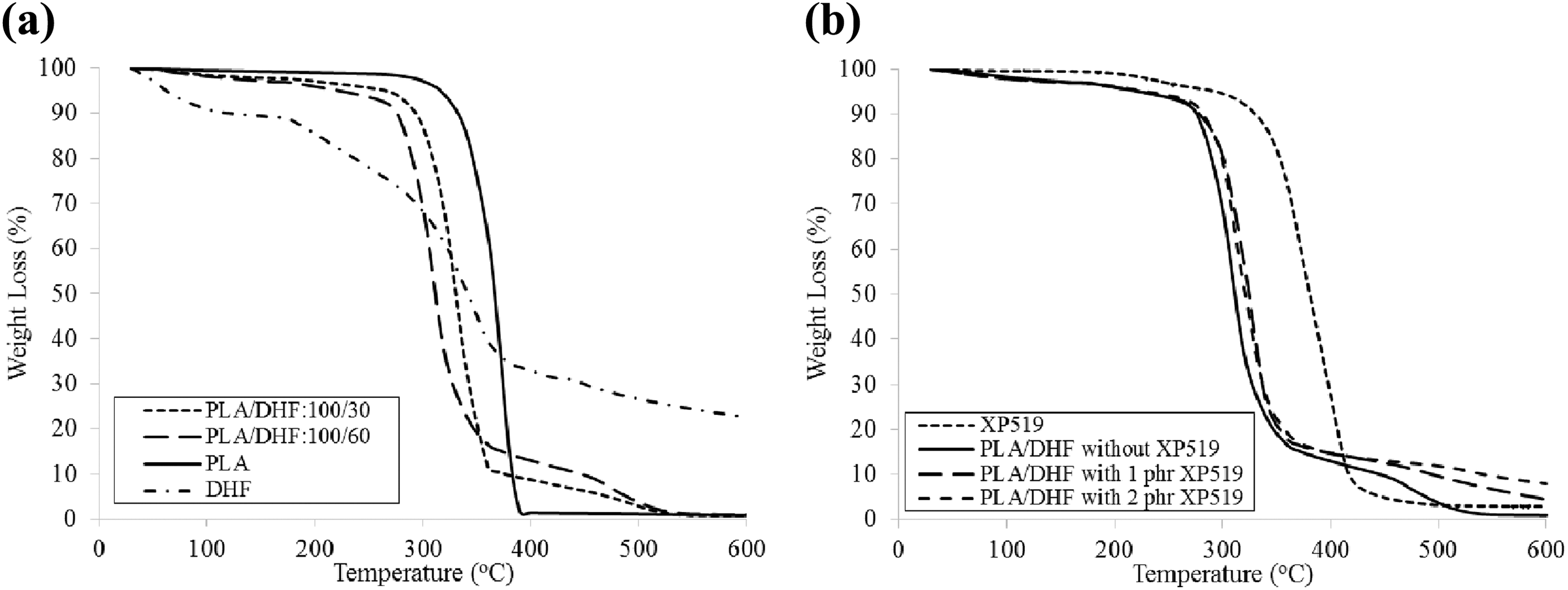
TGA curves of PLA/DHF biocomposites (a) without and (b) with Ultra-Plast XP519.
TGA data of PLA/DHF composite with and without Ultra-Plast XP519.
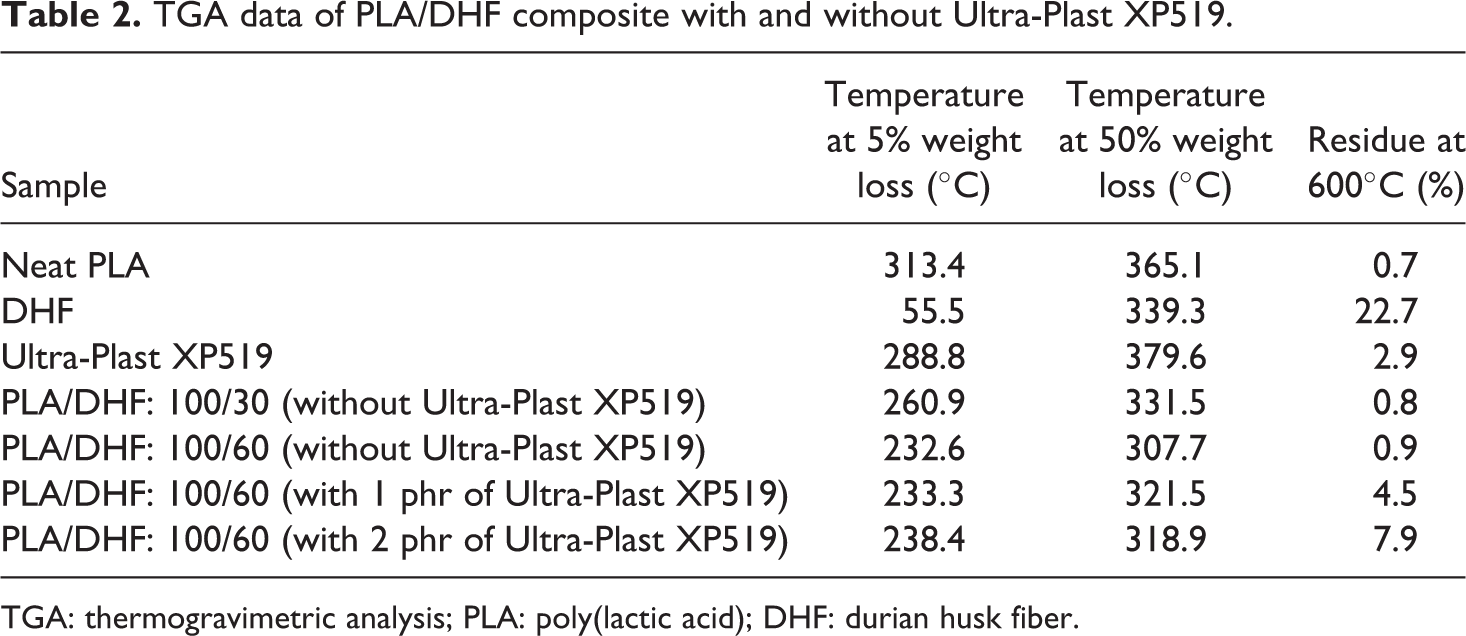
TGA: thermogravimetric analysis; PLA: poly(lactic acid); DHF: durian husk fiber.
The TGA curves for pure PLA and PLA/DHF biocomposites with different DHF content as well as different processing aid content are shown in Figure 9(b). According to Table 2, the Ultra-Plast XP519 exhibited an excellent thermal stability compared to PLA/DHF biocomposites. The T d5% of biocomposites was shifted to higher temperature at increased Ultra-Plast XP519 content. The existence of Ultra-Plast XP519 has formed a thermal protective coating on DHF. As the Ultra-Plast XP519 has better thermal stability, it is able to protect the DHF from earlier thermal degradation. Furthermore, the biocomposites with Ultra-Plast XP519 showed higher T d50% than biocomposites without Ultra-Plast XP519. This indicated that the Ultra-Plast XP519 is able to improve the thermal stability of biocomposites. In contrast, the T d50% of biocomposites decreased from 321.5°C to 318.9°C when Ultra-Plast XP519 content was increased. The presence of Ultra-Plast XP519 also promoted the process of char formation as the char residue increased at higher Ultra-Plast XP519 content. Hence, the presence of more Ultra-Plast XP519 would slightly reduce the thermal stability of biocomposites. This is because the Ultra-Plast XP519 might promote the thermal degradation of lignin and cellulose in DHF and yielded higher char content. In our previous studies, the addition of fatty acid compound was also found to increase the early thermal stability and char content of biocomposites as the fatty acid might promote char formation. 6,29
Figure 10(a) illustrates the DSC curves of PLA/DHF biocomposites with different DHF content. The data obtained from DSC curves were summarized in Table 3. The incorporation of DHF to PLA resin had decreased the T g and X c. From the literature, the degree of plasticity has been changed because the mobility of the non-crystalline chains was constrained by attaching to the immobile crystallites. 30 However, in this case, the mobility of PLA chains was increased because the noncrystalline chain parts have less constrain by the immobile crystallites structure. Therefore, the change of T g is directly proportional to X c. The decrement of X c is because the filler restrained the polymer chain from rearranging when the polymer tried to obtain crystallization. 19 An exothermic peak observed at temperature between 80°C and 100°C is attributed to the cold crystallization process and where T cc was obtained. The cold crystallization only occurs above T g because the amorphous region would reorganize into crystalline structure owing to the increment of the macromolecular flexibility and mobility due to the arising temperature. It can be observed that the PLA curve does not have the exothermic peak which indicated that the polymer chains of the neat PLA resin do not contain available amorphous region that can undergo cold crystallization. The crystallinity of PLA/DHF biocomposites had decreased with the increase of DHF content. This indicated that the biocomposites with more DHF content are easier to undergo cold crystallization process. This is because the addition of more DHF had restricted most of the PLA chains to form crystallites and resulting in more amorphous phase that is reserved for cold crystallization. Hence, the cold crystallization occurs at lower temperature. Similar findings were also found by Jandas et al. 30 where the PLA/banana fiber biocomposites with higher fiber content showed lower T cc due to the nucleating effect from fiber. From Figure 9(a), it can be observed that there are two peaks during the melting endotherms which might be due to two following reasons: (i) the lamellar rearrangement while cold crystallization of PLA occurred and (ii) the lower peak is the melting endothermic reaction of the initial crystallites while the higher peak is formed during the melting endothermic reaction of the recrystallized crystallites. 31 The melting temperature (T m) of biocomposites is correlated to X c as the matrix consists of more crystallites and it would require higher temperature to convert from solid crystal to viscous liquid. 32 For this reason, the T m1 and T m2 of the PLA/DHF biocomposites were shifted to lower temperature when the DHF content was increased.
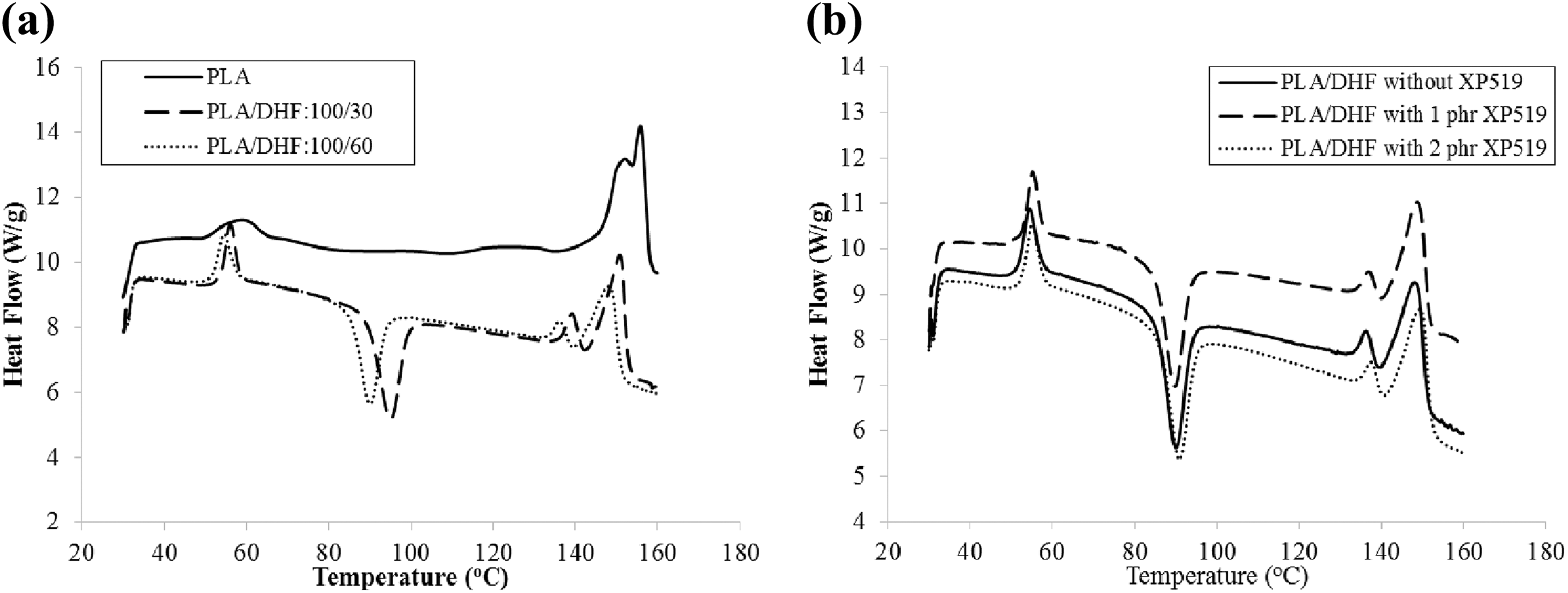
DSC curves of PLA/DHF biocomposites (a) without and (b) with Ultra-Plast XP519.
DSC data of PLA/DHF composite with and without Ultra-Plast XP519.

T g: glass transition temperature; T m1: melting temperature of the first peak; T m2: melting temperature of the second peak; T cc: cold crystallization temperature; X c: crystallinity of biocomposite; DSC: differential scanning calorimetry; PLA: poly(lactic acid); DHF: durian husk fiber.
The DSC curves of PLA/DHF with and without Ultra-Plast XP519 are displayed in Figure 10(b). From Table 3, the addition of Ultra-Plast XP519 has reduced the T g of the PLA/DHF biocomposite. As mentioned earlier, the Ultra-Plast XP519 has lubricating effect on biocomposite. The decrement in T g has proved that the addition of Ultra-Plast XP519 improved the flexibility of PLA chain due to the lubricating effect. Moreover, the addition of Ultra-Plast XP519 did not have significant influence on the T cc and T m of PLA/DHF biocomposite. On the other hand, the X c of PLA/DHF biocomposite had reduced when added with more Ultra-Plast XP519. The presence of Ultra-Plast XP519 might have inhibited the crystallization of PLA matrix. Thus, the crystallinity of PLA matrix was reduced. The finding is in agreement with observation reported by Qin et al 32 . They used polybutyl acrylate as processing aid in rice straw fiber reinforced PLA biocomposite and found that higher amount of polybutyl arcylate added to biocomposite results in decrement of crystallinity of the biocomposite.
Conclusions
The increase of fiber content increased the processing torque of PLA/DHF biocomposites. This means that the biocomposites will be difficult to process if fiber content is increased. The tensile strength and modulus of PLA/DHF biocomposites increased with increasing fiber content. At 60 phr of fiber content, the PLA/DHF biocomposites were able to achieve tensile strength of more than 11 MPa, suitable for nonstructural applications that do not require high mechanical strength. For thermal properties, the addition of more DHF decreased the thermal stability of the biocomposites. The PLA/DHF biocomposites showed an earlier thermal degradation above temperature 100°C. At the same time, increasing DHF leads to decrement of T g and X c of the biocomposites. The addition of Ultra-Plast XP519 had significantly reduced the processing torque of biocomposites. This will benefit the melt compounding process of biocomposites containing high fiber content. The addition of 0.5-phr Ultra-Plast XP519 did not show significant change in tensile modulus of PLA/DHF biocomposites and its tensile modulus reduced with more Ultra-Plast XP519 content. The tensile strength of PLA/DHF biocomposite decreased with increasing ltra-Plast XP519 content. Moreover, the addition of Ultra-Plast XP519 had improved the earlier thermal stability of the PLA/DHF biocomposites. However, the addition of more Ultra-Plast XP519 decreased the thermal stability of biocomposites at higher temperature. The T g and X c also further reduced due to the addition of Ultra-Plast XP519. Overall, the addition of Ultra-Plast XP519 provided positive effects on the processing and thermal properties of biocomposites, but it also brings negative effects on the mechanical properties.
Footnotes
Acknowledgements
The authors gratefully acknowledge use of services and facilities at Taylor’s University, funded by Taylor’s Research Grant Scheme. The authors also would like to acknowledge Performance Additives Sdn. Bhd. (Malaysia) which provided the processing aid for this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taylor’s Research Grant Scheme (project code: TRGS/ERFS/1/2017/SOE/033).
