Abstract
The flammability, thermal properties, and synergistic effects of modified expandable graphite (MEG) with magnesium hydroxide (MH) and aluminum hydroxide (ATH) on the linear low-density polyethylene/ethylene vinyl acetate (LLDPE/EVA) blends are investigated by Underwriters Laboratories-94 (UL-94) vertical combustion test, limiting oxygen index (LOI), thermogravimetric analysis (TGA), cone calorimeter test (CCT), and scanning electron microscopy (SEM). The results show that the MEG improves the flame-retardant efficiency of LLDPE/EVA blends. The addition of MEG apparently improves the LOI values and the UL-94 rating of LLDPE/EVA composites. The data obtained from the CCT show that the heat release rate (HRR), the total heat release (THR), and the gas production rate of composites with MEG decrease remarkably with increasing the content of MEG. When 10 phr of MEG is added, the char residues of LEMEG10 increase to 38.2% from 2.7% of LLDPE/EVA. The results of SEM and CCT present that MEG can improve the quality of char layers. The rate of char formation is enhanced also due to the existence of MEG, which plays an important role to improve the flame retardancy of the LLDPE/EVA composites.
Introduction
Linear low-density polyethylene (LLDPE) is widely used in cable and wire industry and life fields 1 ; however, the flammability greatly limits its application in some fields. 2,3 In addition, its poor stress and compatibility with inorganic fillers also hinder its application further. To solve these problems, ethylene vinyl acetate (EVA) is added to the LLDPE because of its excellent flexibility, low viscosity, partial polarity, and so on. EVA is a suitable candidate for blending with polyethylene (PE) to improve its flexibility, toughness, environmental stress cracking resistance, and transparency. 4,5 EVA can also be well miscible with PE due to the presence of polar ester groups in EVA. 6 Therefore, the addition of EVA into the flame-retardant LLDPE/inorganic fillers composites cannot only improve the compatibility between the polymer matrix and inorganic particles but also obtain complementary advantages. 7 –10
PE and EVA both containing C, H are flammable and will release a lot of smoke, heat, and toxic gases during combustion, which brings great harm and loss to humans and the environment. 4 To overcome the flammability, researchers added inorganic flame retardants into polymers. 11 Recently, halogen-containing flame retardant is replaced by halogen-free flame retardant, such as magnesium hydroxide (MH) and aluminum hydroxide (ATH), due to its good smoke suppression, nontoxic, and endothermic performances, 12 –16 which not only reduces the temperature of the solid phase through endothermic but also releases vapor into the gas phase to dilute combustible gas. 17 Ma et al. 11,18 studied the combustion characteristics and thermal viscoelastic, crystallization, and mechanical properties of high-density polyethylen (HDPE)/ethylene vinyl acetate (EVA) blends containing MH. Chen et al. 19 studied the thermal oxidative degradation kinetics of polypropylene (PP) and PP/Mg(OH)2 composites. In addition, the use of ATH is limited to those polymers processed below about 200°C while MH is stable above 300°C and thus can be used in polymers that are processed at higher temperatures. 20 This can broaden the range of decomposition temperatures of the flame retardant, which plays a synergistic role in polymer retardancy, greatly improving the flame-retardant properties. 21 Therefore, the combination of MH with ATH is widely used in the flame-retardant polymers.
However, MH and ATH have some shortcomings, such as poor flame retardancy, high addition, and poor adhesion with the polymer.
22
To improve the compatibility between MH and the matrix, Ding et al.
23
investigated MH modified by 1-
In this work, a novel organic–inorganic interfacial intumescent flame-retardant mode is proposed using grafting process containing phosphorous and applied to the LLDPE/EVA composite systems with MH/ATH, which exhibits excellent flame-retardant efficiency. The flammability and synergistic effect of modified EG (MEG) and MH/ATH on the flame-retardant LLDPE/EVA composites are investigated systematically by limiting oxygen index (LOI), Underwriters Laboratories-94 (UL-94) vertical burning test, cone calorimeter test (CCT), thermogravimetric analysis (TGA), Fourier transform infrared (FTIR), and scanning electron microscopy (SEM).
Experimental
Material
LLDPE (DFDA7042) is produced by Jilin Petrochemical, China. EVA (7240M) with VA content of 15 wt% is produced by Taiwan Plastic Company, China. MH (average particle size: 1.5 μm) and ATH (average particle size: 2.6 μm) are bought from Yantai Ai Weier Chemical Company, China. EG with particle size of 270 μm and carbon content of 90–99.9% is provided by Qingdao Kang Boer Graphite Products company. Phosphorus-containing flame retardant, 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxidation (DOPO), is provided by Guangdong Huizhou Shengshida Technology Company (Huizhou, China), and the γ-glycidyloxypropyltrimethoxysilane (silane coupling agent) is provided by Chengdu Chenbang Chemical Co., Ltd, China.
The surface modification of EG
Surface modification of EG is performed according to our previous work. 29 A certain amount of DOPO and silane coupling are mixed in round-bottomed flask and stirred 4 h at 160°C to prepare the DOPO-silane coupling agent. Then a mixed solution A is prepared with appropriate amount of deionized water and EG, and the pH is adjusted to 4 by adding ethylic acid. The mixture of DOPO-silane coupling agent and ethyl alcohol are slowly added into the solution A and stirred 4 h at 60°C. Finally, wash the product with ethanol aqueous solution to remove the excess reactants. The modified EG product is called MEG.
Preparation of the flame-retardant LLDPE/EVA composites
The LLDPE/EVA composites filled with the desired amounts of MH, ATH, and MEG are blended in a twin-screw extruder (TSE-20A1600-4-40, made in Nanjing, China) at 130–170°C with a rotation rate of 150 r min−1, and then the extrude granulates are pressed at 170°C for testing. The formulas of the specimen are listed in Table 1.
Flame properties of samples obtained based on LOI values and UL-94 test.
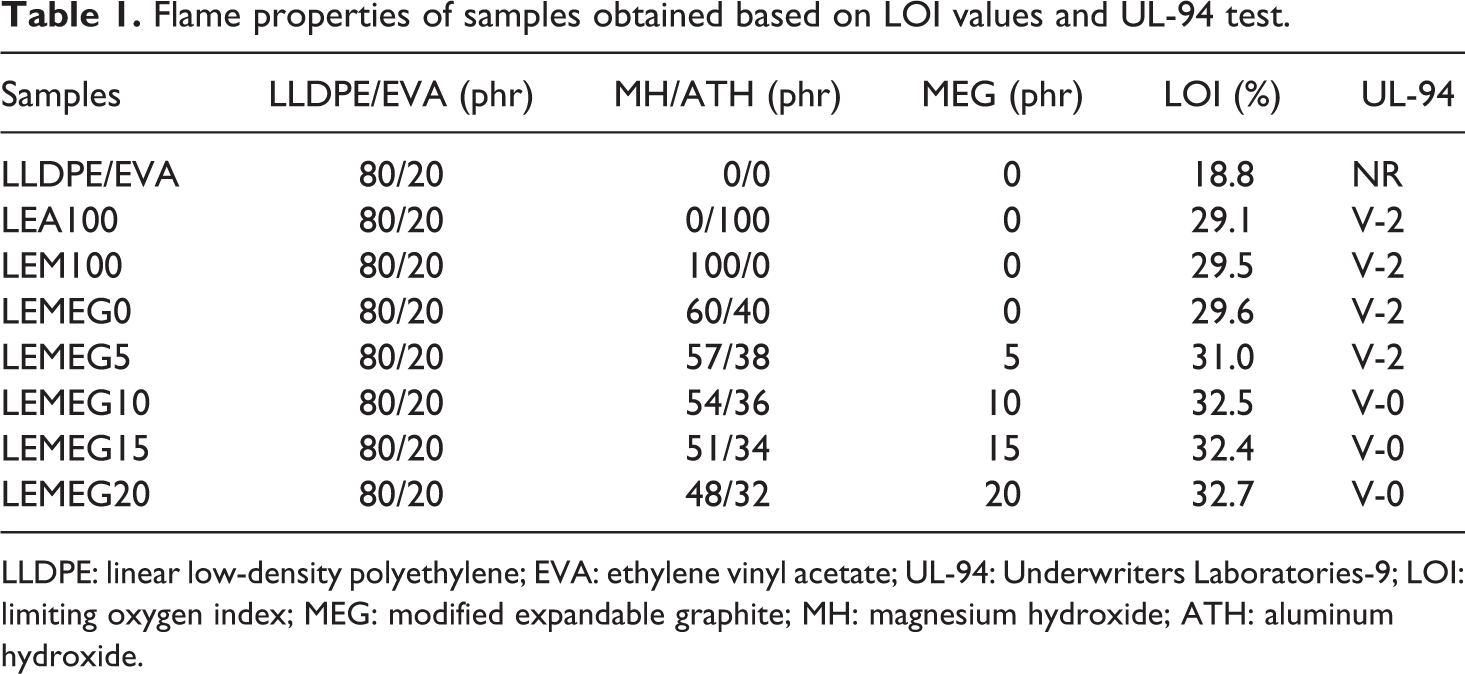
LLDPE: linear low-density polyethylene; EVA: ethylene vinyl acetate; UL-94: Underwriters Laboratories-9; LOI: limiting oxygen index; MEG: modified expandable graphite; MH: magnesium hydroxide; ATH: aluminum hydroxide.
Measurements and characterization
Limiting oxygen index
LOI value is measured with JF-4TG oxygen index instrument (manufactured by Jiangning Analysis Instrument Factory, Nanjing, China) on sheets 120 × 6.5 × 3.20 mm3 according to the standard oxygen index rest (ASTM D 2863-77).
Underwriters Laboratories-94test
The UL-94 vertical burning tests are carried out on a CZF-1 type instrument (Nanjing Jiangning Analytical Instrument Factory, China), with the bar dimensions of 127 × 12.7 × 2.7 mm3 according to the standard ASTM D3801-10.
Thermogravimetric analysis
TGA is performed on a Q50 thermogravimetric analyzer (TA Instruments, USA) under nitrogen with a flow rate of 50 mL min−1. About 10.0 mg sample is tested and heated from room temperature to 650°C at a heating rate of 10° C min−1.
Cone calorimeter test
The cone calorimeter (Stanton Redcroft, UK) tests are carried out according to ISO 5660 standard procedures. Each specimen of dimensions 100 × 100 × 3 mm3 is wrapped in aluminum foil and exposed horizontally to an external heat flux of 35 kW m−2.
Fourier transform infrared spectroscopy
The Fourier transform infrared (FTIR) spectra are recorded with a Nicolet 560 spectrophotometer (Nicolet Instrument Co., USA). Samples are mixed with a potassium bromide powders, and the mixture is pressed into a tablet.
Morphology observation
The surface morphologies of char layers from burned LEMEG0 and LEMEG10 composites coated conductive gold are observed on a SEM (JSM-7500F, Japan) with an acceleration voltage of 5.0 kV.
Results and discussion
Flammability: LOI and UL-94 test
The LOI values and UL-94 rating level of the composites are displayed in Table 1. As shown in Table 1 that the LOI values of flame-retarded LLDPE/EVA composites are higher than that of LLDPE/EVA. The LOI value of LLDPE/EVA is only 18.8%, and it is not classified in the UL-94 test. However, the addition of MH, ATH, or ATH/MH at a mass fraction of 100 phr can improve the LOI values of LEA100, LEM100, and LEMEG0 samples to 29.1, 29.5, and 29.6%, respectively. The LOI values of the LEA100 and LEM100 samples are both lower than that of LEMEG0. This is because that the thermal decomposition temperature of ATH and MH is different. In addition, LEA100 and LEM100 (containing only one flame retardant) only release water vapor and absorb heat in 250–350°C and 350–450°C, respectively, while LEMEG0 can release water vapor and absorb heat in 250–450°C, which can well dilute the concentration of combustible gas and oxygen and reduce the temperature around the composites. At the same time, it is found that the LEA100, LEM100, and LEMEG0 samples can achieve the UL-94 V-2.
The influence of modified EG content on the flame-retardant performance of LLDPE/EVA composites is also investigated. Keeping the total flame-retardant amount of 100 phr constant and adjusting the proportion, the LOI values of composites gradually increase with increasing the content of MEG, and even the LOI value of LEMEG20 reaches to 32.7% when the content of MEG is 20 phr. In addition, the similar results are also obtained in the UL-94 test. The LEMEG10 sample containing 10 phr MEG is classified UL-94 V-0, and when 20 phr MEG is added, the LEMEG20 sample cannot be ignited. This indicates that the addition of MEG can improve the flame retardancy of the composites. During the UL-94 test, the composites containing MEG form a worm-like structure, which prevents air and combustible gases from entering the underlying substrate. 30
The photographs of the samples obtained from LOI and UL-94 test are displayed in Figures 1 to 3. It is clearly observed from Figures 1 and 2 that LLDPE/EVA cannot produce carbon layer after LOI and UL-94 test. With addition of MH or/and ATH, a little char residue is found on the surface of samples (b) to (d). While various amounts of MEG are incorporated into the flame-retardant LLDPE/EVA composites, as shown in samples (e) to (h), the charred residues of the samples are swelled, and the expandable charred residues gradually increase with increasing the MEG content. The samples with 10–20 phr MEG have larger expanding volume and keep good shapes after tests. The flame retardancies are enhanced further, which shows that there is synergistic effect between three additives. At the same time, the burning process is significantly different as shown in Figure 3. The pure LLDPE/EVA burns sharply with dripping severely, which is dangerous and easy to cause “secondary combustion.” 31 When the flame retardants are added into the blends, the dripping disappears, and the burning gradually becomes slow. Especially, the samples (e) to (h) have very slow burning rate and can achieve a desired result with increasing MEG content.

Photographs of LLDPE/EVA and its flame-retardant composite after LOI test: (a) LLDPE/EVA, (b) LEA100, (c) LEM100, (d) LEMEG0, (e) LEMEG5, (f) LEMEG10, (g) LEMEG15, and (h) LEMEG20.

Photographs of LLDPE/EVA and its flame-retardant composite after UL-94 test: (a) LLDPE/EVA, (b) LEA100, (c) LEM100, (d) LEMEG0, (e) LEMEG5, (f) LEMEG10, (g) LEMEG15, and (h) LEMEG20.
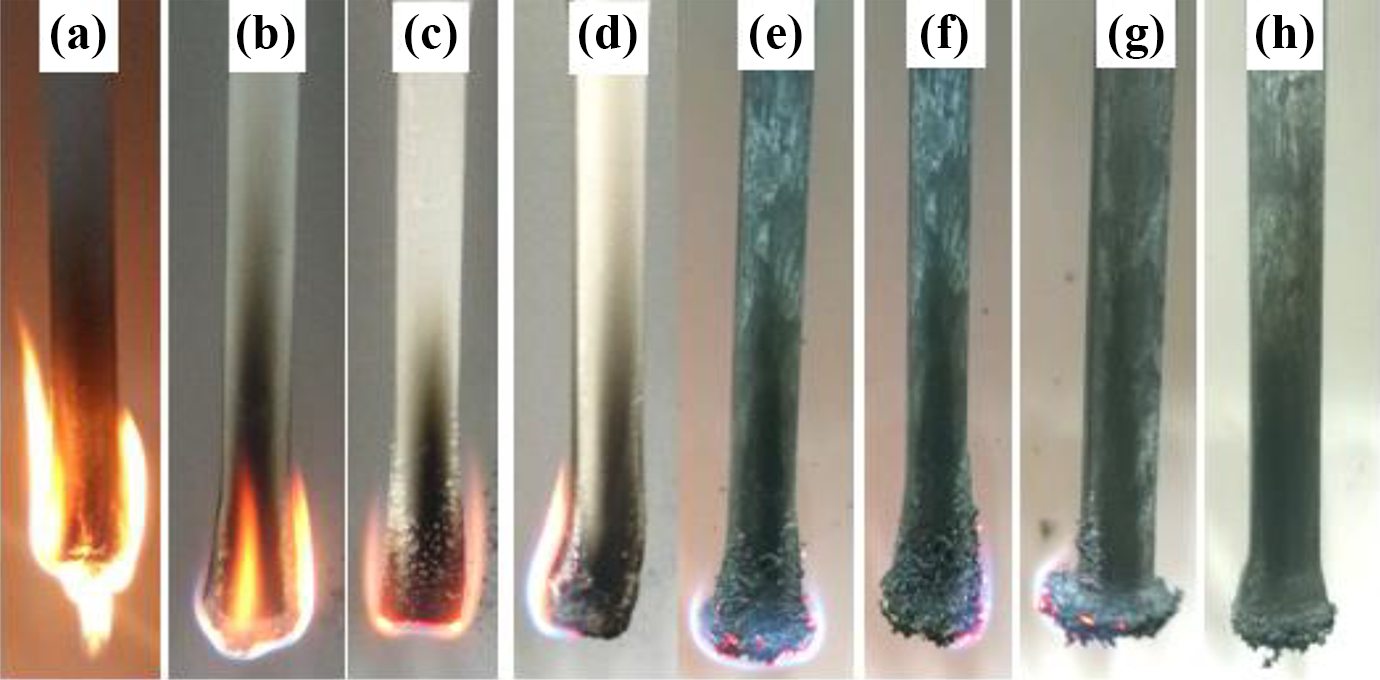
Digital photographs taken during UL-94 burning of LLDPE/EVA and its flame-retardant composite: (a) LLDPE/EVA, (b) LEA100, (c) LEM100, (d) LEMEG0, (e) LEMEG5, (f) LEMEG10, (g) LEMEG15, and (h) LEMEG20.
Dynamic combustion behaviors
The CCT has been extensively used to evaluate the flammability of materials. 32,33 The heat release rate (HRR), total heat release (THR), time to ignition (TTI), CO, and CO2 production rate are shown in Figures 4 to 7, and the detailed data including total smoke rate (TSR) and average effect heat of combustion (average EHC) are listed in Table 2. As shown in Table 2, when flame retardants are added, TTI of the composites increases gradually. For instance, when 10 phr of MH and ATH are replaced by MEG, the TTI of LEMEG10 increases to 53 s from 30 s of LEMEG0. The TTI of LEMEG20 containing 20 phr MEG increases to 58 s. It is concluded that MEG can improve the flame retardancy of the composites.
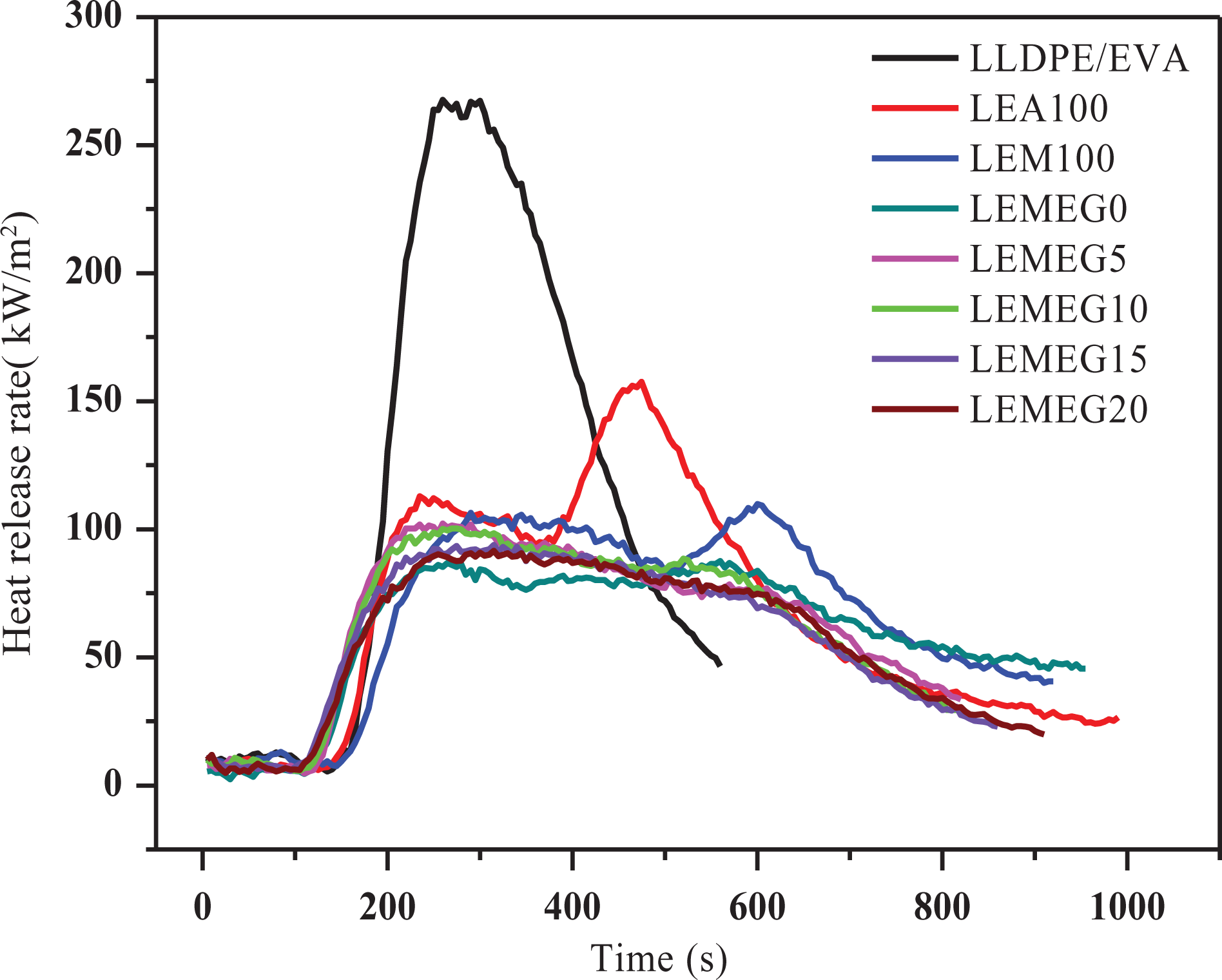
HRR curves of LLDPE/EVA and its flame-retardant composites.
The CCT data of LLDPE/EVA and its flame-retardant composites.
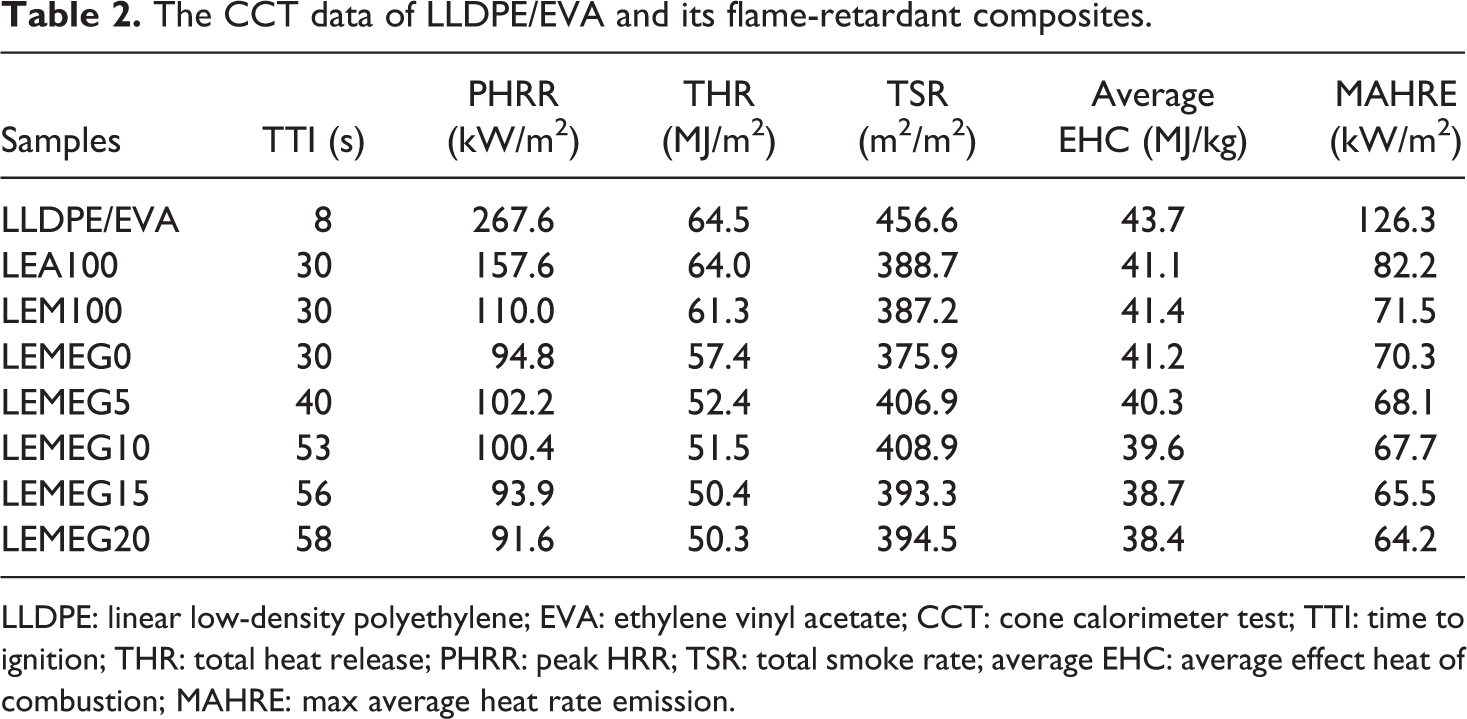
LLDPE: linear low-density polyethylene; EVA: ethylene vinyl acetate; CCT: cone calorimeter test; TTI: time to ignition; THR: total heat release; PHRR: peak HRR; TSR: total smoke rate; average EHC: average effect heat of combustion; MAHRE: max average heat rate emission.
Figure 4 represents the dynamic curves of HRR versus time for LLDPE/EVA and its flame-retardant composites. Between 200 and 400 s, LLDPE/EVA shows a sharp peak with the peak HRR (PHRR) value of 267.6 kW/m2, and its burning time is very short within 600 s. However, all the flame-retardant composites appear relatively smooth with the PHRR values below 150 kW/m2, while their burning times remain more than 900 s. These data show that the flame-retardant LLDPE/EVA composites burn more slowly than pure LLDPE/EVA. The LEA100 and LEM100 samples have two peaks, however, other flame-retarded composites with MEG have only one peak. The possible reason is that the char layer formed by only ATH or MH is so loose and easily destroyed at high temperature. There is a good synergy between MEG with ATH and MH, as well generates a compact carbon layer, which inhibits heat and air infiltrating into the substrate to prevent further combustion.
The values of THR are often used to assess the safety factor of materials under a combustion environment. 34 It can be observed from Figure 5 that the THR value decreases due to the addition of MH, ATH, and MEG. The THR results are closely related to HRR, and its values for all samples are 64.5, 64, 61.3, 57.4, 52.4, 51.5, 50.4, and 50.3 MJ/m2, respectively. The fire spread of samples LEMEG0 to LEMEG20 has decreased gradually, and even LEMEG20 is comparatively the lowest among all samples. These phenomena can be explained by the intumescent charred residues on the surface of composites, which can serve as a barrier from the flame zone to underlying materials and restrain flammable gases to flame zone. 35 In addition, it is found from Table 2 that the incorporation of MEG has a little effect on the average EHC values. From the aforementioned discussion, it is concluded that the flame-retardant mechanism of MH/ATH with MEG is a condensed phase mechanism.
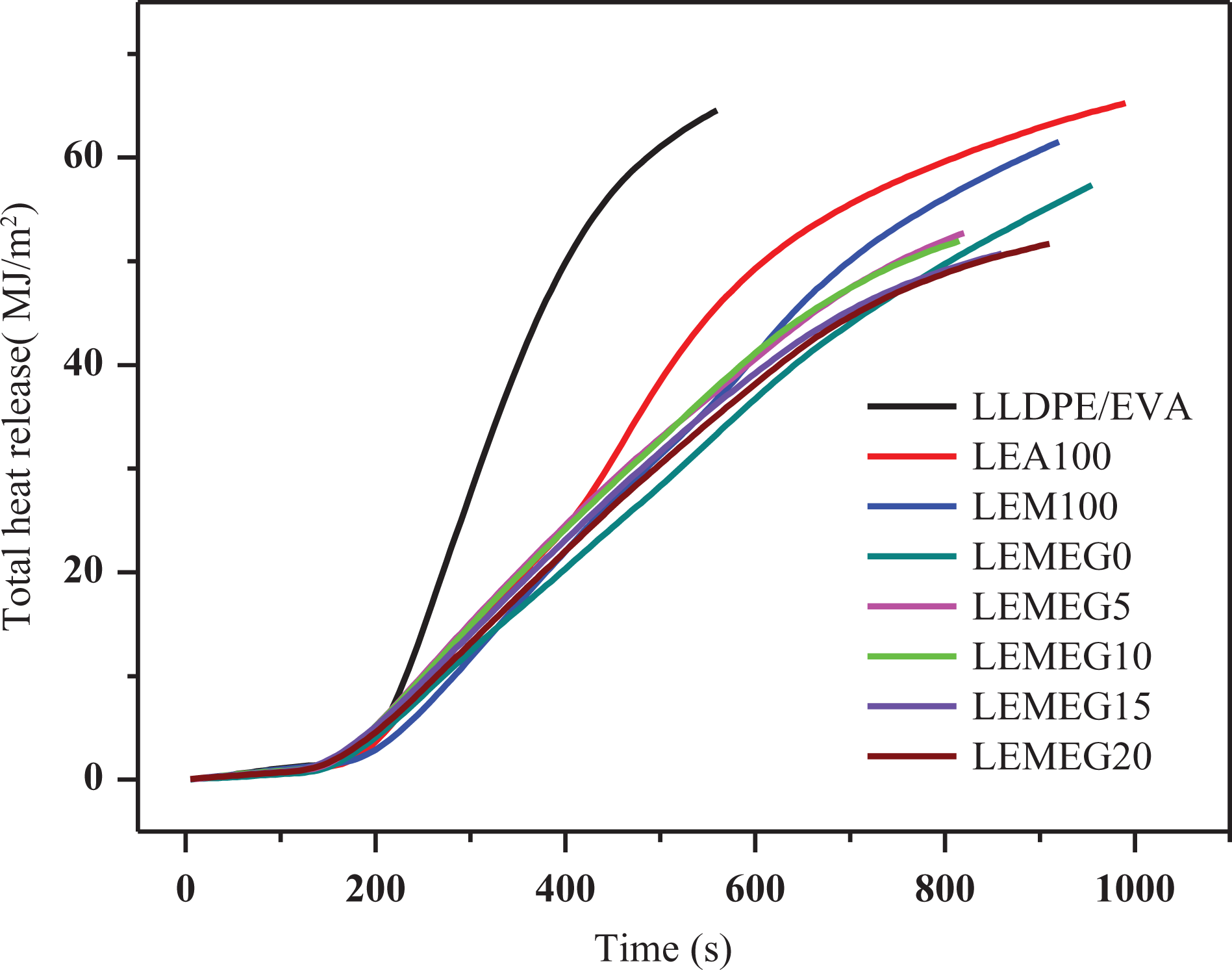
THR curves of LLDPE/EVA and its flame-retardant composites.
The CO and CO2 production can evaluate the gas release rate of flame-retardant materials in a certain extent. 36 Figures 6 and 7 are the CO and CO2 release rate curves of the above composites, respectively. The CO and CO2 production rate peaks of LLDPE/EVA are sharp and narrow, meaning that LLDPE/EVA burns very violently. It is obvious that the CO and CO2 production rate values of the flame-retardant composites decrease remarkably with the addition of MEG at the range of 0–700 s, unfortunately they are higher than that of the composites without MEG between 750 and 1000 s. This is also found from Table 2 that the values of TSR exhibit a similar trend of change. This implies that MEG apparently promotes to produce gases, which may be attributed to the following three reasons. First, the intumescent graphite layers formed by MEG in the heat flux inhibit the penetration of air into matrix, which may cause the incomplete combustion of substance. 37 This may be the main reason why the smoke emission increases by the addition of MEG. Second, the carbon is the main composition of MEG, which produces a lot of CO and CO2 during combustion. Third, the decomposition products of MH or ATH are main metal oxide and H2O, which can dilute the smoke and gases.
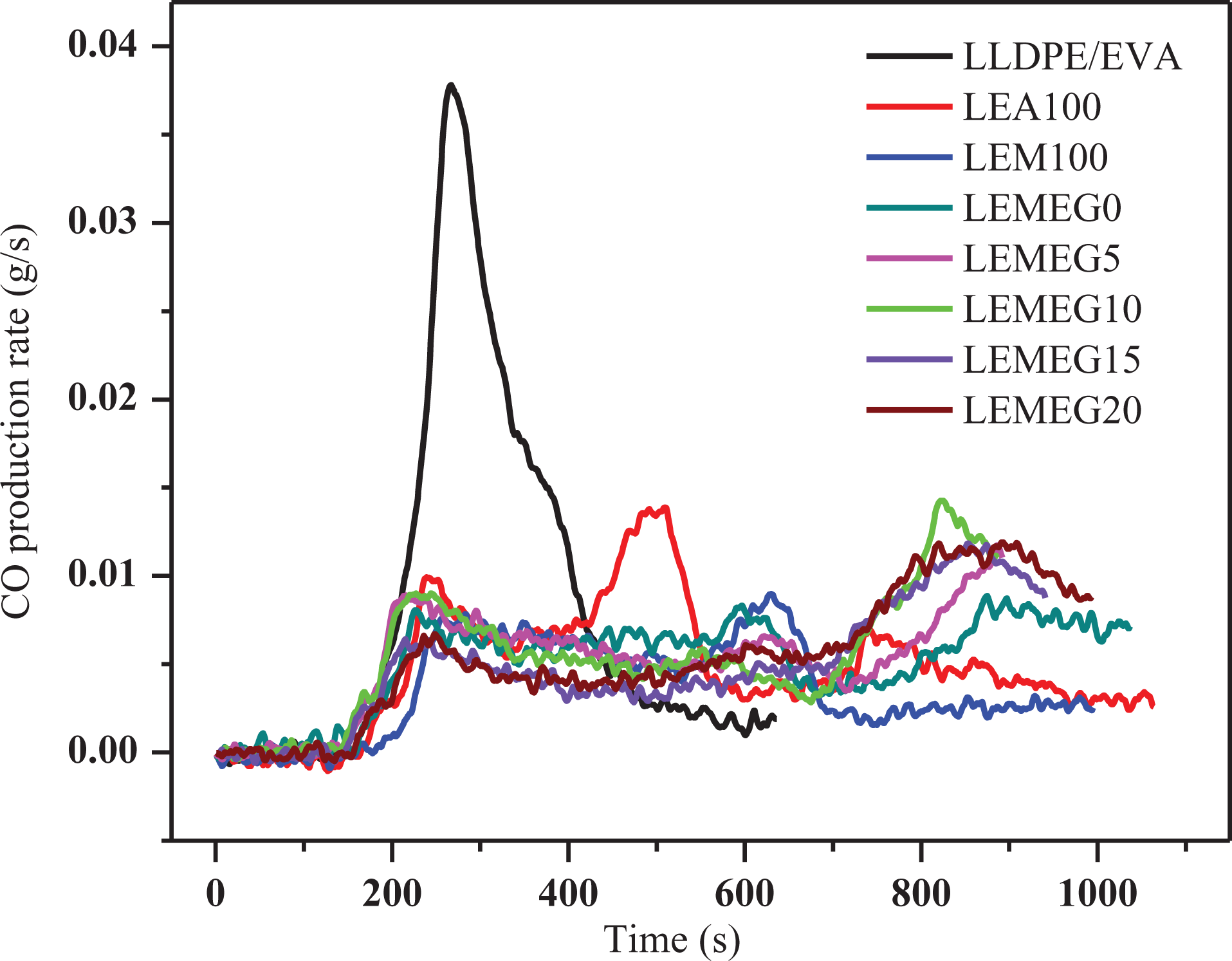
CO release curves of LLDPE/EVA and its flame-retardant composites.
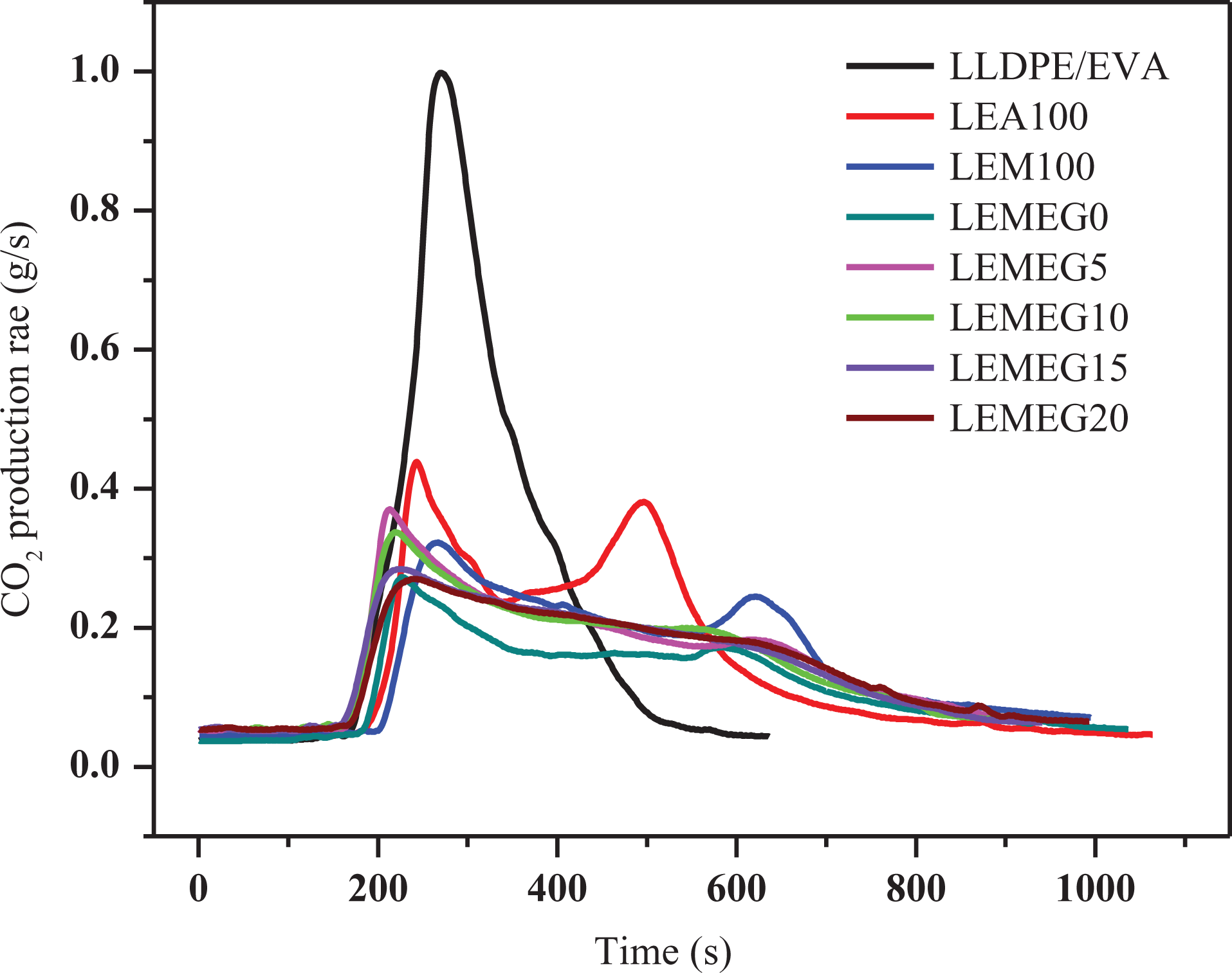
CO2 release curves of LLDPE/EVA and its flame-retardant composites.
Thermal stability
The TGA and DTG curves of LLDPE/EVA, LEMEG0, and LEMEG10 samples are illustrated in Figures 8 and 9, respectively, and the detailed data are listed in Table 3. The TGA curve of LLDPE/EVA only has one main thermal degradation process with the final residue near to 2.7% at 600°C. As shown in Figure 8 and Table 3, its initial decomposition temperature corresponding to a 5% weight loss (
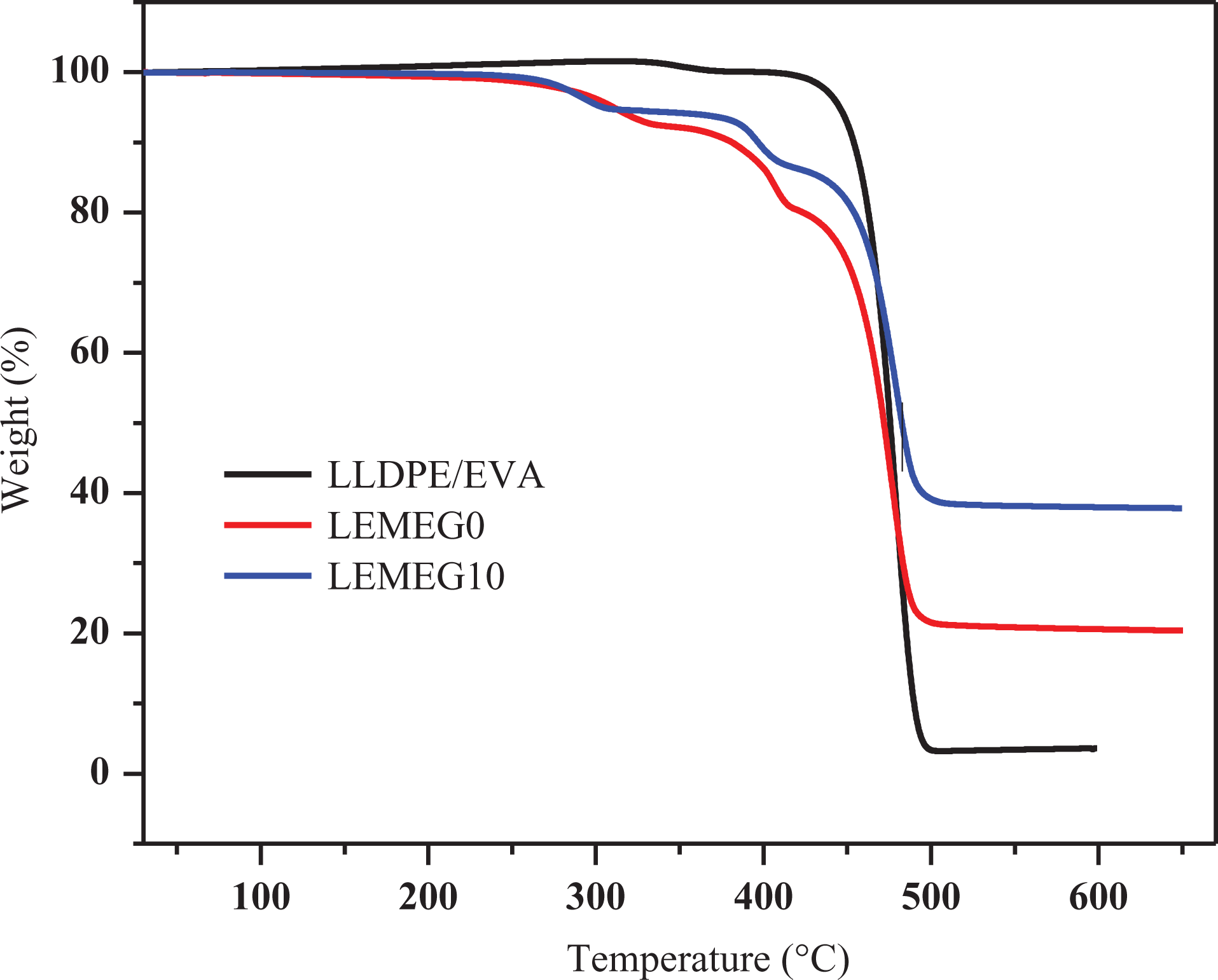
TGA curves of LLDPE/EVA, LEMEG0, and LEMEG10.
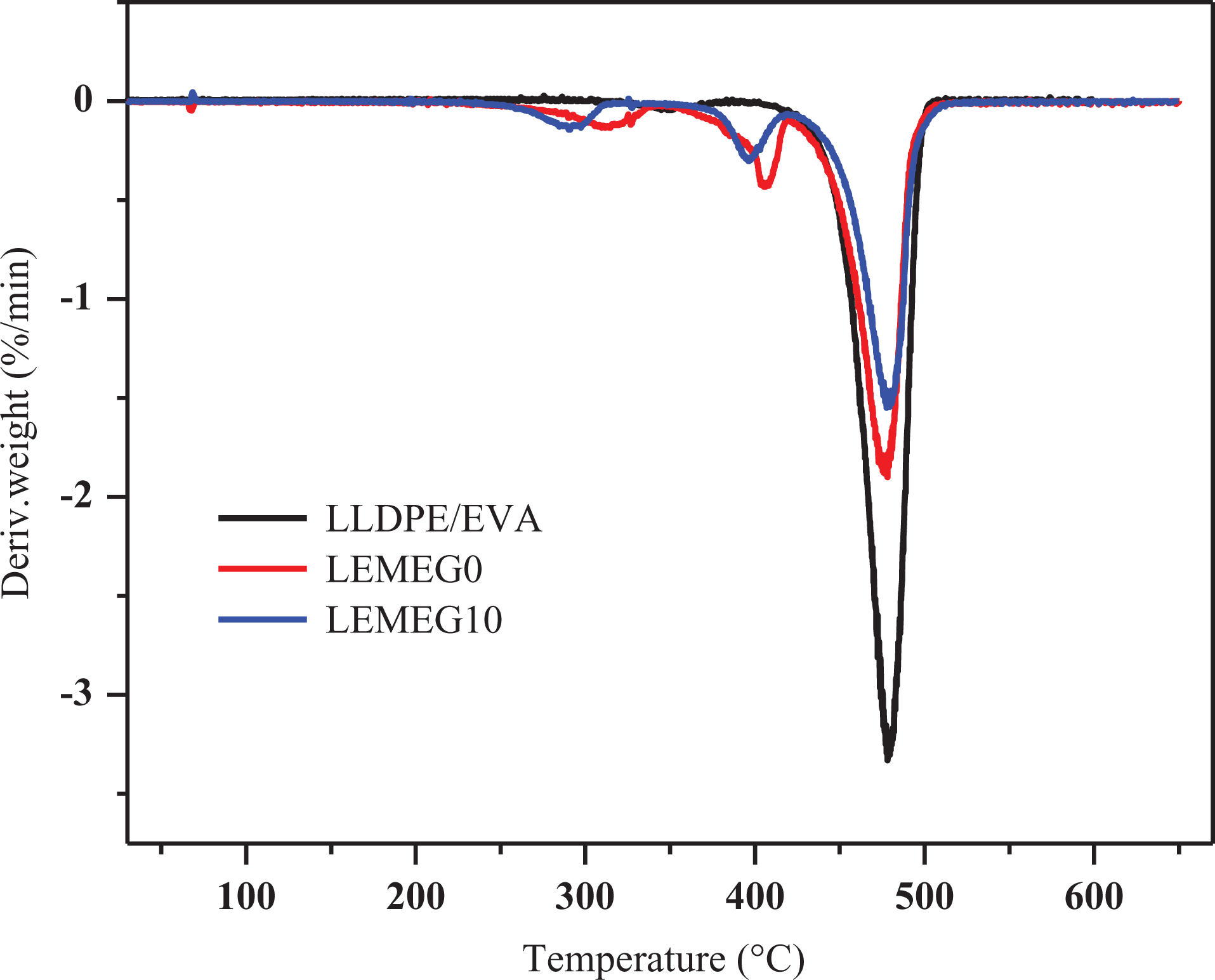
Derivative thermogravimetry (DTG) curves of LLDPE/EVA, LEMEG0, and LEMEG10.
The TGA data of LLDPE/EVA and its flame-retardant composites.
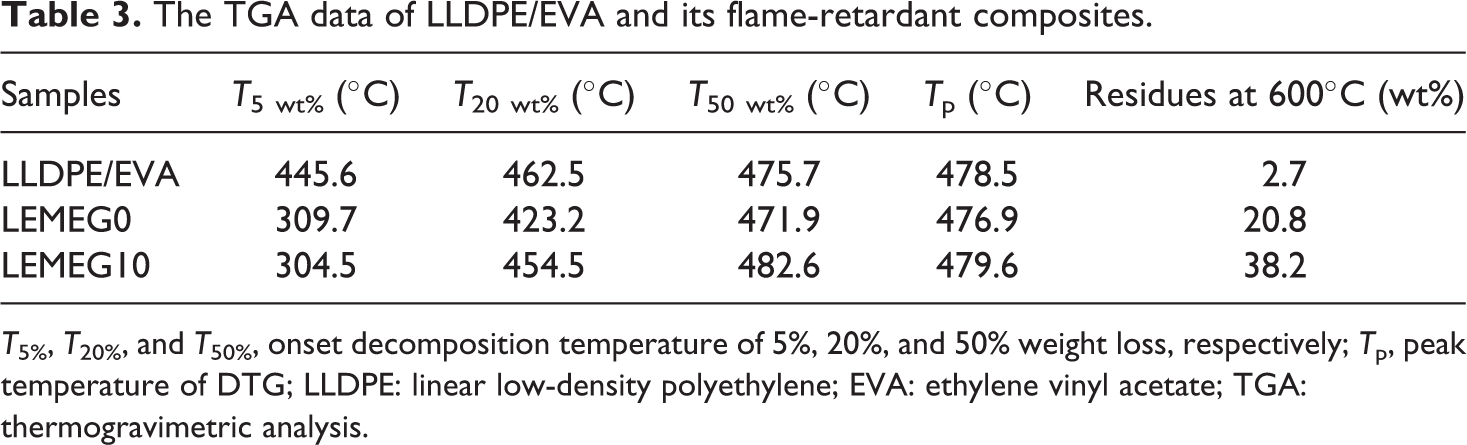
For the LEMEG0 and LEMEG10 samples, the TGA curves undergo three-step thermal degradation in the range of 30–600°C. The first-stage degradation occurs in the range of 260–330°C, and the second one occurs in the range of 370–410°C, which is attributed to the degradation of ATH and MH, respectively. The third one taking place in the range of 450–500°C involves the degradation of the decomposition of the polyacetylene-ethylene chains.
32
The LEMEG0 and LEMEG10 samples present a lower weight loss rate and higher charred residues at 600°C. Compared with LLDPE/EVA, it is observed that the
Morphology analysis of charred residues
The residual char of polymers can prevent the air and the flammable gases generated by the degradation of into underlying materials 37 ; hence, the char residue plays a very important role in the flame-retarded performances. Figure 10 shows the photographs of residues of the samples collected after CCT. LLDPE/EVA burns completely and does not left charred residue. However, the charred residues are formed in the flame-retardant LLDPE/EVA composites, as displayed in Figure 10(b) to (h). The LEA100, LEM100, and LEMEG0 samples left loose and brittle charred residues, whereas the composites containing MEG left relatively compact and integrated char layers, and there are more charred residues with increasing the MEG content, as shown in Figure 10(e) to (h). This suggests that the incorporation of MEG with MH/ATH can improve the structures of charred layers and reinforce thermal stability of charred layers, 36 resulting in the improvement of the flame retardancy.

Photographs of LLDPE/EVA composites after CCT: (a) LLDPE/EVA, (b) LEA100, (c) LEM100, (d) LEMEG0, (e) LEMEG5, (f) LEMEG10, (g) LEMEG15, and (h) LEMEG20.
SEM is used to analyze the surface morphology of residual char of LEMEG0 and LEMEG10 after flammability, and the results are shown in Figure 11. The surface of LEMEG0 sample is very loose and has many holes, and cannot form a compact protective layer, indicating that the quality of char layer is unsatisfactory, which leads to a poor flame retardancy of LEMEG0. This also reflects that LEMEG0 has a lower LOI value and cannot reach to UL-94 rating. However, the LEMEG10 sample forms a very dense and compact carbon layers. The transfer of heat and flammable gases and volatiles is effectively retarded by the insulative compact layer. 38 Therefore, the flame-retardant properties of the composites are improved for this sample.

SEM photographs of char residues surface after burning of (a) LEMEG0 and (b) LEMEG10 composites.
The dynamic FTIR spectra of LEMEG0 and LEMEG10 at different temperatures are shown in Figures 12 and 13, respectively. Some characteristic peaks of LEMEG0 and LEMEG10 disappear gradually with increasing pyrolysis temperature, which suggests that the composites have decomposed. It is obviously seen from Figure 12 that the peaks at 3700, 3675, 3515, 3446, 2923, 2853, and 1470 cm−1 assigning to the CH2 and CH3 vibration and the C–C stretching vibration, 38 and 1740, 1251, and 1020 cm−1 assigning to the C=O and C–O deformation vibration, 39 which are the characteristic absorption of LLDPE and EVA. These characteristic peaks do not change below 400°C, while the temperature rises to 400°C, the bonds at 3675, 3515, and 3446 cm−1 decrease. This can be explained by the decomposition of MH/ATH, as well the absorption peaks at 1740, 1251, and 1020 cm−1 disappear, which indicates that the main decomposition happens at this stage. All peaks nearly disappear at 400°C, suggesting that polymer matrix in LEMG0 sample decomposes completely.
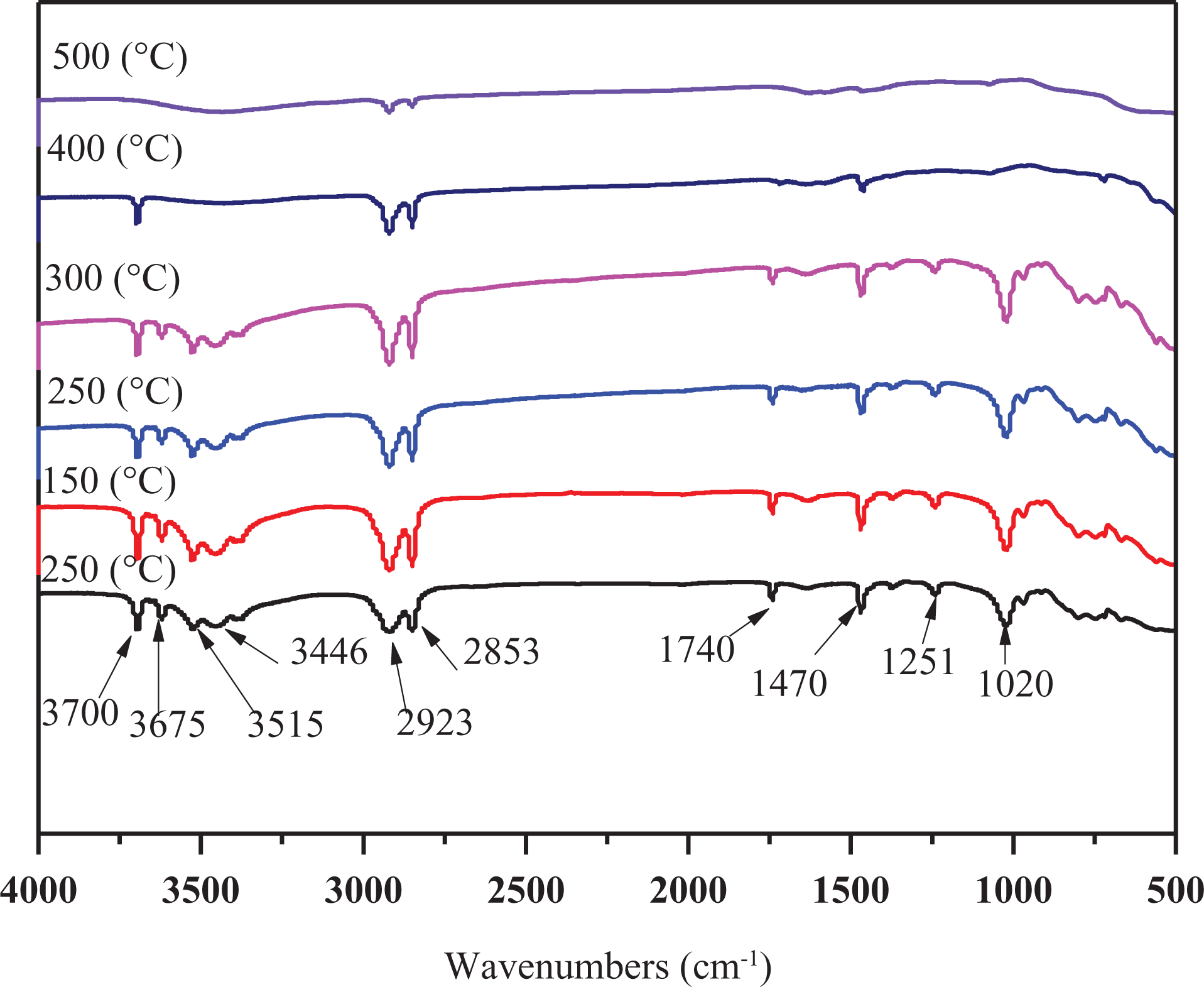
The dynamic FTIR spectra at different pyrolysis temperatures of LEMEG0.
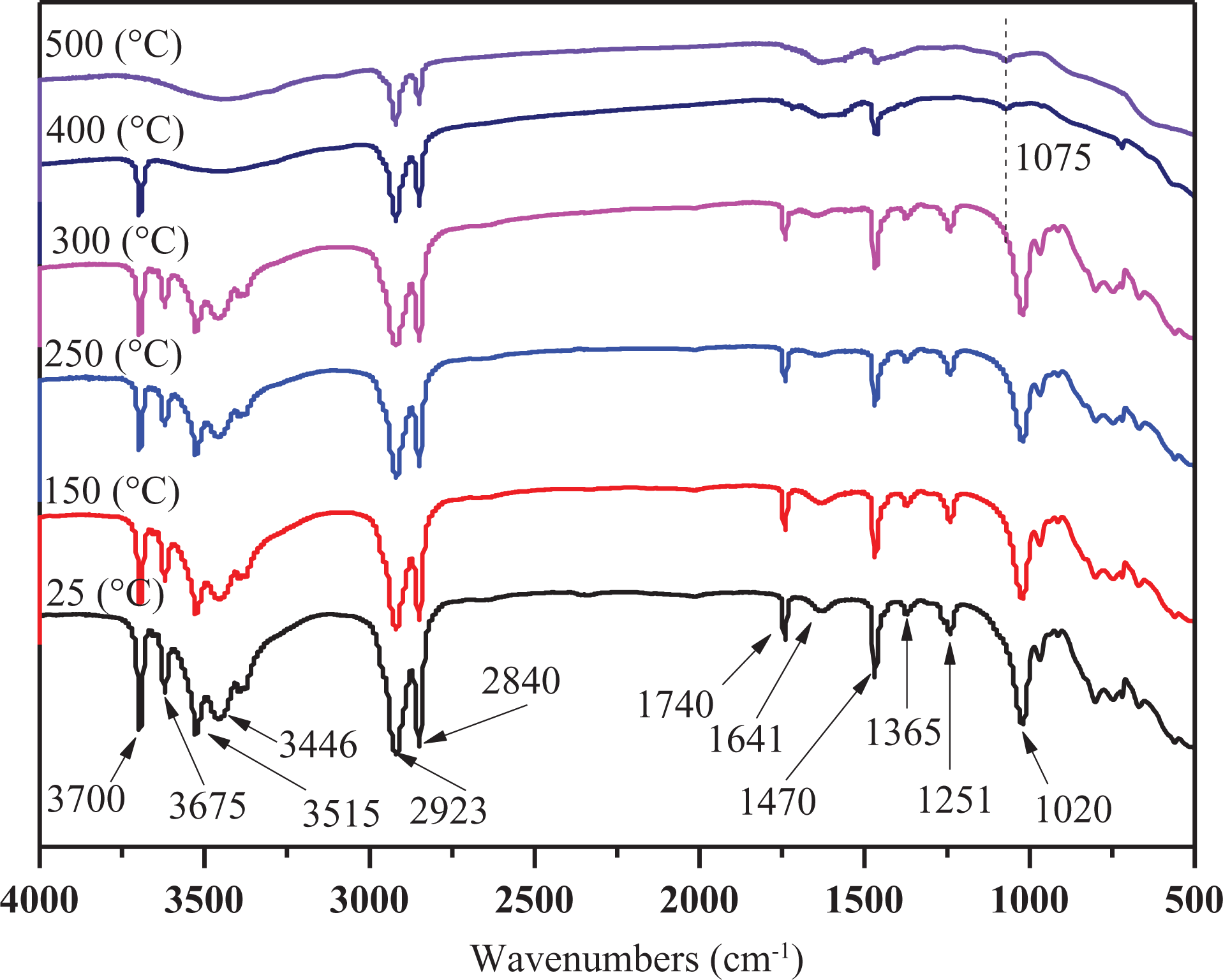
The dynamic FTIR spectra at different pyrolysis temperatures of LEMEG10.
Compared with LEMG0 sample, the thermos-oxidative degradation processes of LEMEG10 sample (Figure 13) are much more complicated. The peaks at 3700, 3675, 3515, 3446, 2923, 2853, 1741, 1470, 1251, and 1020 cm−1 are the characteristic absorption of LLDPE and EVA. The intensities of most peaks keep constant at 300°C, while the temperature rises to 400°C, the intensities of most peaks disappear, indicating that the main decomposition takes place at this stage. A new absorption peak at 1075 cm−1 appears, assigning to P–O vibrational peak decomposed by MEG, 40 and the phosphate catalyzes the formation of compact charred layer, which can delay the thermos-oxidative degradation during combustion. The intensities of all absorption peaks of LEMG10 sample are stronger than those of LEMG0 sample at the same pyrolysis temperature, indicating that the presence of MEG increasing the thermos-oxidative stability of the composites. This is consistent with the TGA results.
Conclusions
The flame retardancy and synergistic effect of MEG with MH/ATH in the flame-retardant LLDPE/EVA composites are investigated using LOI, UL-94, CCT, TGA, FTIR, and so on. It is concluded that MEG has a good synergistic effect with MH/ATH in the flame-retardant LLDPE/EVA composites. Compared with the composites without MEG, the LOI values and UL-94 rating of the composites with MEG are enhanced. The CCT data show the TTI of flame-retardant LLDPE/EVA composites is delayed, and the values of HRR and THR of the composites decrease remarkably with increasing the amount of MEG. The TGA and FTIR results reveal the presence of MEG greatly enhance the thermal oxidative stability and increase the charred residues of the composites. In addition, the char layers structure after combustion is improved due to the addition of MEG. The morphological observations exhibit the synergistic effects and flame-retardant mechanism of MEG with MH and ATH in the flame-retardant LLDPE/EVA composites. The improvement of the flame retardancy and thermos-oxidative stability of the composites is attributed to the stable and compact char layers promoted by MEG with MH and ATH acted as an effective heat barrier and thermal insulator. This prevents the underlying polymers from burning further, resulting in desired properties are achieved.
Footnotes
Authors’ note
Na Wang is the co-first author of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Opening Project of State Key Laboratory of Polymer Materials Engineering (Sichuan University) (2017-4-02), National Natural Science Foundation of China (51763002, 51003088), International Science and Technology Cooperation Project of Chengdu City (2015-GH02-00034-HZ, 2017-GH02-00051-HZ), and Dayi Science and Technology Supporting Project Foundation (2016-KC-00252-GX).
