Abstract
The objective of this work is to evaluate the reinforced effect of wheat straw (WS) after oxidation in thermoplastic corn starch (TPS).The mechanical properties (tensile strength and elongation at break) of TPS and oxidized wheat straw (OWS)/TPS composites were evaluated by the tensile measurements. The composites were characterized further using scanning electron microscopy (SEM), dynamic mechanical thermal analysis, and torque measurements. In general, OWS played a key role for enhanced mechanical and thermal properties in composites, and the composites had slightly poorer rheological processing behaviors, compared to the TPS. The highest tensile strength (7.66 MPa) was achieved using WS oxidized with 0.045 mol·L−1 sodium periodate. The damage and erosion produced on WS after oxidation positively affected the properties of the composites. With respect to the findings of this study, it may be stated that OWS can be used as a promising natural filler for other composites.
Introduction
Wheat straw (WS) residues are agricultural byproducts. Large amounts of WS are generated globally every year after harvesting. However, most get burned on the field. Only a small percentage is being used as a source of fuel or animal feed and therefore do not add much value to the crops. 1 In addition, WS, to a limited extent, has traditionally been used for pulp and paper making. 2,3 Composite is another prospective commercial application area that would unlock the potential of these underutilized renewable materials and provide a non-food–based market for agricultural industry.
WS contains a high content of natural cellulose, hemicellulose, and lignin. Currently, two main methods have been applied to utilize the WS in composites. Direct using is the first one, such as, making low or medium particleboard, 4,5 by filling into high-density polyethylene and polypropylene to produce composites, 6 and in this case, its structure is not damaged. The other important aspect is that WS should be pretreated first, such as steam explosion, 7,8 fungal, 9,10 dilute alkali, 11 and coupling agent treatments, 12 in most of this case, producing microfibrillated or nanosized cellulose fibers is expected to be used further in cellulose-based composite materials.
Starch is one of the most promising candidates for biodegradable plastic, 13 because it is a cheap and abundant natural polymer. However, thermoplastic corn starch (TPS) cannot be used widely because of retrogradation and unsatisfactory mechanical properties, particularly in wet and dry environments. 14,15 Consequently, much work has focused on solving these problems. Nowadays, the addition of natural fillers to TPS is an attractive method to improve the mechanical properties of TPS, because they are cheaper and renewable other than traditional reinforcing materials. 16
As a natural filler, the WS before treatment is more hydrophobic, which is not favorable for reinforcement in TPS. Oxidation is another possible pretreatment of WS through chemical method. This method would possibly damage WS surface, make the hydroxyl groups in cellulose partially oxidized to aldehyde or ketone groups, 17,18 and contribute to the improvement in miscibility between WS and TPS. Therefore, the main goal of the present work is to explore the effect of WS oxidation on WS/TPS composites, especially the effects of OWS with different oxidation degree on the mechanical properties, thermal, and rheological process behavior of WS/TPS composites, to demonstrate the reinforcing potential of these OWS in composite applications.
Methods
Material
WS, obtained from a research field in Jiangsu, China, was cut into small pieces by a micro plant grinding machine (FZ102, Tianjin Taisite Co. Ltd, China), and its aspect ratio is about 2.6. They were stored in a storehouse with adequate ventilation to dry. Corn starch containing 28% amylase and 12% moisture was supplied by the Shandong Hengren Industrial and Trade Co., Ltd (China). The plasticizer, that is, glycerol, was provided by the Sinopharm Chemical Reagent Co., Ltd (China). Sodium periodate, purchased from the Xilong Chemical Co., Ltd (China) was used as the oxidant.
Preparation of OWS/TPS
The pHs of sodium periodate solutions of different concentrations (0.015, 0.03, 0.045, and 0.06 mol·L−1) were adjusted to 6 with hydrochloric acid (HCl) at 45°C. For convenience, each sample is described by sodium periodate solutions concentration (0.015, 0.03, 0.045, and 0.06) in the reaction feed rather than the actual oxidation degree. WS particles (10 g) were immersed in 500 mL of each solution, and the mixtures were stirred at 45°C for 6 h under lightproof conditions. Ethylene glycol (10 mL) was then added to each solution to terminate the reaction. The slurries were filtered and washed four times with distilled water, and the products were dried at 70°C for 8 h. OWS samples with different degrees of oxidation were obtained.
Corn starch granules were plasticized with glycerol and blended with OWS in an electric mixer, and then the mixture was fed into a twin-screw extruder (screw diameter 21.7 mm, length-to-diameter ratio 32:1; SHJ20, Nanjing Giant Machinery Co., Ltd, China). The temperature profile along the extruder barrel was set in the range 105–120°C, and the screw was operated at 150 r min−1. The ratio of starch to glycerol was 3:1 by weight, and the amount of OWS was 1 phr, based on the total weight of starch and glycerol. The extrudate were cut into pellets of length 3 mm and dried for 6 h at 80°C before injection molding. Specimens for tensile testing were prepared using an injection-molding machine (BV90, Apollo, China).
Determination of aldehyde contents
The aldehyde contents of the OWS were determined using the hydroxylamine hydrochloride method reported in the literature 19,20 based on the chemical reaction

The released HCl was titrated with sodium hydroxide–methanol solution during the reaction in methanol, and the aldehyde group content (
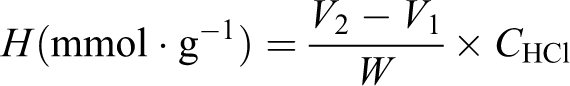
where
Surface morphology
The surfaces of the samples were studied on a Quanta 200 environmental scanning electron microscope (FEI, Hillsboro, Oregon, USA). The fractured surfaces of TPS and composites were obtained from the dumbbell-shaped specimens after the tensile measurements. The fractured part of specimen was cut into 10 ×5 ×4 mm3 near the fracture surface. The fractured faces were vacuum coated with gold prior to analysis, and the tungsten filament was operated at 20 kV.
Mechanical properties
Measurements of mechanical properties (tensile strength and elongation at break) were performed in accordance with the ASTM D638 standard. The tensile measurements were conducted on a testing machine (SANS, Shenzhen, China). Dumbbell-shaped specimens (2-mm thick) of the composites were obtained by injection molding. The samples were placed between the grips of the machine and stretched with strain rate of 20 mm min−1 at room temperature. Five to eight specimens were tested for each sample, and the average values of the measured properties were reported.
Dynamic mechanical thermal analysis
Dynamic mechanical thermal analysis (DMTA) measurements were performed using a Netzsch 242E instrument in three-point bending mode at a frequency of 1 Hz, corresponding to a maximum displacement amplitude of 60 μm. The temperature range was −110 to 110°C, and the heating rate was 3°C min−1.
Rheological characteristics
An RTIO55/20 torque rheometer (POTOP, China) was used to investigate the rheological behaviors of the composites. The temperature from the head board to the back board was set at 120°C, 130°C, or 140°C, and the rotors were operated at 20 r min−1.
Results and discussion
SEM of WS and OWS surface
SEM micrographs of WS before and after oxidation were used to examine the surface morphology. As Figure 1 shows, the surface of the untreated WS (the particles on its epidermis were scraps of straw, which were produced by crushing) was much smoother than that of the oxidized WS. As the degree of oxidation increased, small cracks and voids gradually appeared on the WS epidermis. The SEM image in Figure 1(b) clearly shows that the WS surface was eroded or destroyed, with apparent voids and interstices appearing on the WS epidermis along the fibrillar structures, especially using 0.045 mol·L−1 sodium periodate. Meanwhile, the aspect ratio of OWS is about 2.6. In addition, as is well known, WS contains natural cellulose (33–38%), hemicellulose (26–32%), and lignin (17–19%). Therefore, the changing surface morphology of OWS can be related to the oxidized and damaged chemical structure, including cellulose, hemicellulose, and lignin in WS.

SEM images of (a) WS and (b) OWS (0.045). WS: wheat straw; OWS: oxidized wheat straw.
Mechanical properties of OWS/TPS composite
The mechanical properties of TPS, WS, OWS (0.045), and OWS/TPS composites are shown in Table 1. As in Figure 2, the tensile strength of composites increased from 2.2 (TPS) to 7.66 MPa with increasing oxidant concentration, whereas the elongation at break decreased, that is, the highest tensile strength (7.66 MPa) was achieved at 0.045 mol·L−1 sodium periodate, the corresponding aldehyde content was 0.602 mmol·g−1. Considering the chemical mechanism, the aldehyde groups in OWS probably react with hydroxyl groups in starch molecules to form inter- and intra-molecular hemiacetals and acetal cross-links, 17 during OWS/TPS processing. Therefore, OWS having higher aldehyde content maybe more favorable to contribute to the reinforcement for OWS/TPS composites. 21 In addition, for the untreated WS, the average tensile strength is 9.98 MPa, the elongation at break is 5.38%, and the corresponding Young’s modulus is 185.5 MPa. Although the tensile strength of OWS (0.045) after oxidation treatment decreased to 6.25 MPa, the strong interaction between aldehyde and hydroxyl groups played a key role for the maximum tensile strength of OWS (0.045)/TPS composite. The corresponding microscopic structure changing and mechanism will be discussed further in the following section.
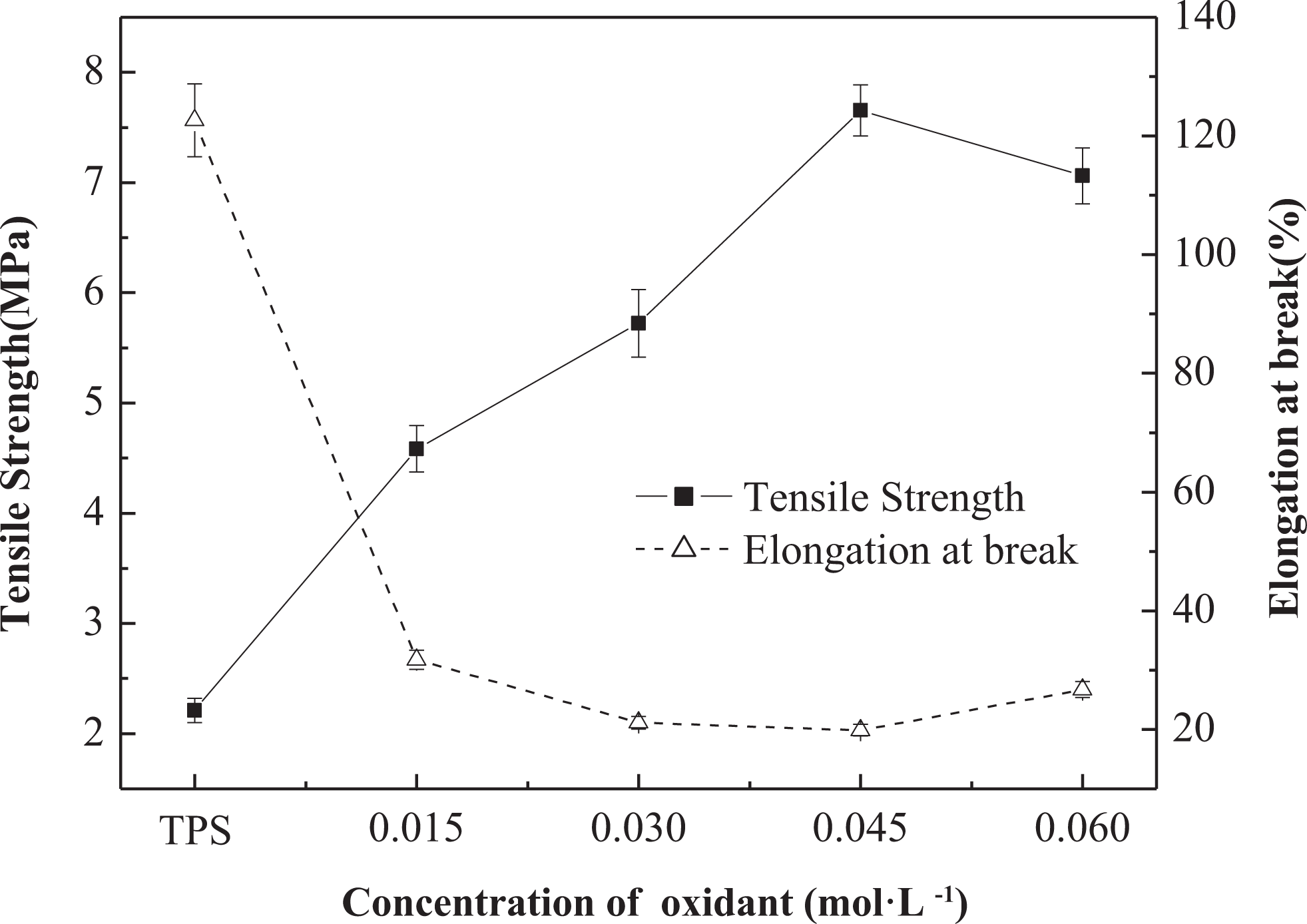
Effect of degree of oxidation on mechanical properties of TPS and OWS/TPS composites. TPS: thermoplastic corn starch; OWS: oxidized wheat straw.
Mechanical properties of TPS, WS, OWS (0.045), and OWS/TPS composites with varying oxidation concentration (0.015–0.06 mol·L−1).a
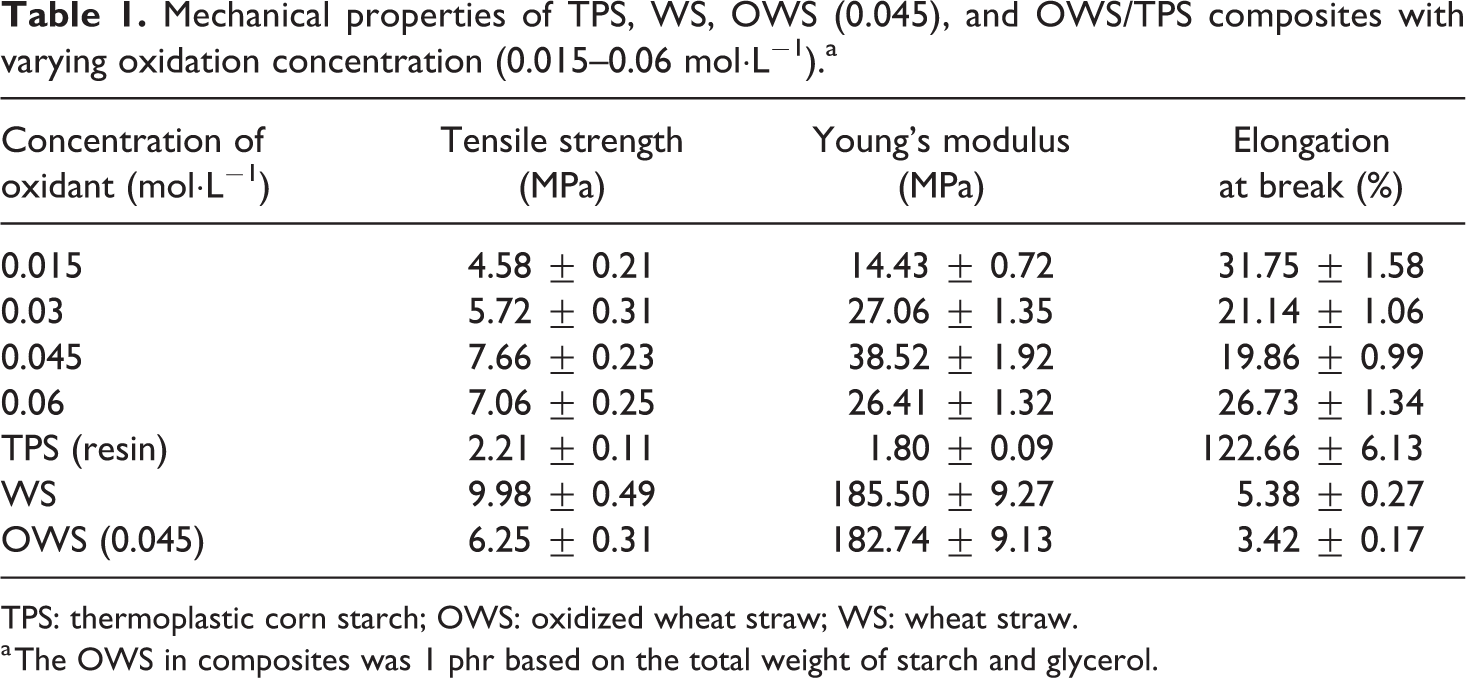
TPS: thermoplastic corn starch; OWS: oxidized wheat straw; WS: wheat straw.
a The OWS in composites was 1 phr based on the total weight of starch and glycerol.
SEM of fracture OWS/TPS composite surface
SEM was used to clarify the microscopic structure of the composite. The morphologies of fractured cross sections of TPS and the composites after the tensile tests are shown in Figure 3. TPS had a homogeneously smooth surface, which is consistent with literature reports, 22 although some small starch granules still remained at the interface. 23 The composites containing OWS had rougher surfaces (Figure 3(b) and (c)). The OWS orientation distribution in composite should be disordered after the process by twin-screw extruder and injection-molding machine.

SEM images of fractured surfaces of (a) TPS, (b) OWS (0.015)/TPS, and (c) OWS (0.045)/TPS. SEM: scanning electron microscopy; TPS: thermoplastic corn starch; OWS: oxidized wheat straw.
For the sample OWS (0.015)/TPS, two distinct phases of TPS and OWS were clearly seen in the composite fractured surface. The continuous phase was TPS, and the formed OWS bundles at the interface and obvious creaks between OWS and TPS matrix also can be observed. That is because, the OWS with lower oxidation probably cannot be miscible with TPS matrix. So the composite had poor mechanical properties. This morphological feature is similar to that reported by Girones for a similar matrix. 24
For the sample OWS (0.045)/TPS, as shown in Figure 3(c), the composite cross section was roughly homogenous, indicating that the OWS was homogenously dispersed and well miscible with the TPS matrix, as observed with red arrow in Figure 3(c). The reason for obvious morphological changes in the fractured surface may come from the erosion and damage produced on WS after deep oxidation. During the processing, some TPS can go into the holes produced by erosion. It leads to better miscibility between them. As a result, it probably increases the mechanical strength and reduces the elongation, which is in accordance with the mechanical test results.
DMTA of OWS/TPS composite
The changes in storage modulus (
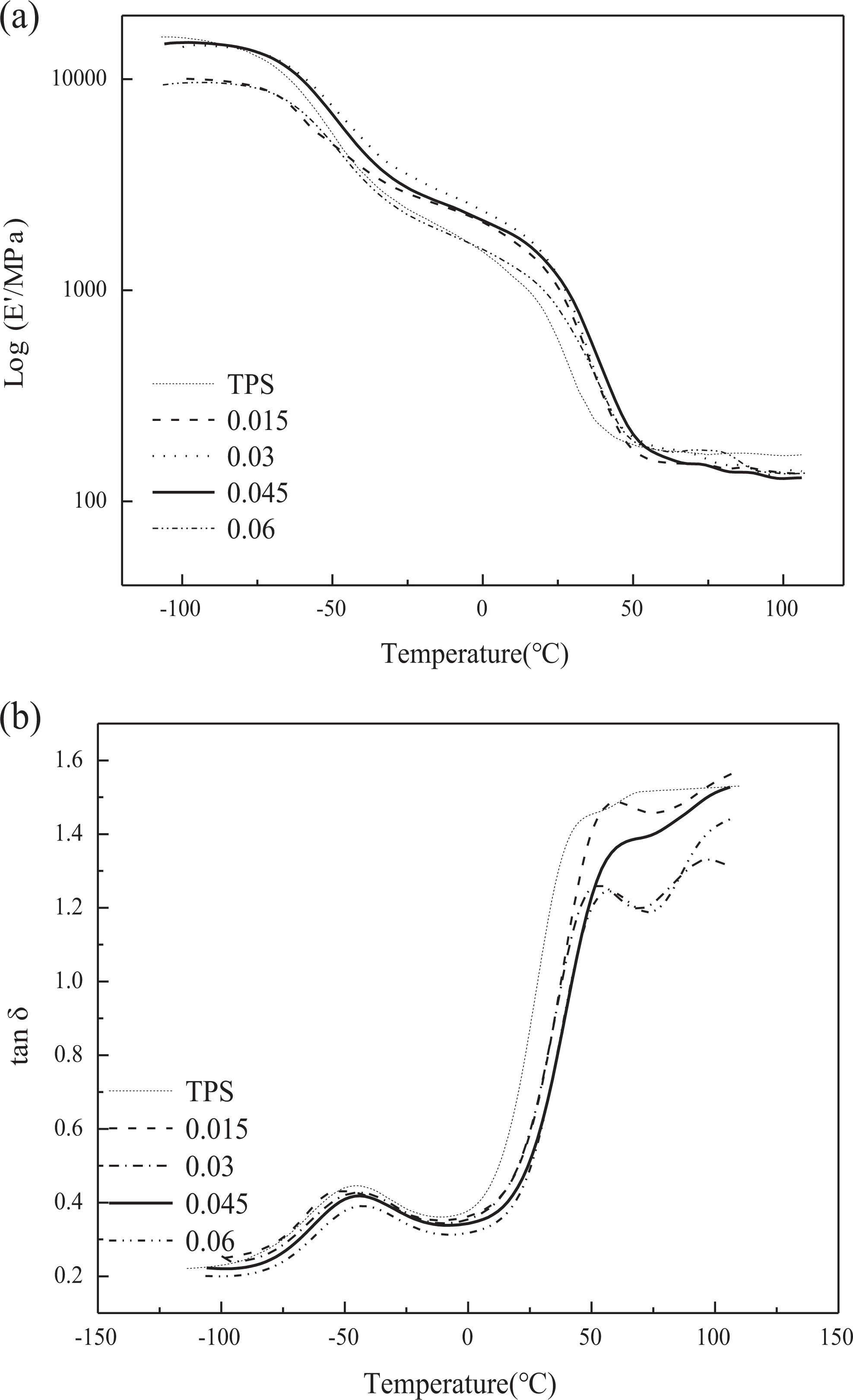
Temperature dependency of (a) storage modulus and (b) tan
Considering the tan
In the
Considering the storage modulus,
Rheological behavior of OWS/TPS composite
Processability is another important property for applications of TPS composites, so the torque characteristics of the OWS/TPS composites were studied. The torque rheological test simulates the processing conditions in an extruder and provides a basis for developing quantitative indices of processability as a function of mixing time. The peak and equilibrium torques of OWS/TPS were higher than those of TPS. Figure 5 and Table 2 show that on increasing the oxidation degree of OWS, the peak torque increased from 19.92 to 25.14 N·m. The equilibrium torque followed the same trend, and for the sample OWS (0.045)/TPS, the equilibrium torque of the sample reached a maximum, that is, 14.50 N·m. This can be ascribed to two aspects, one is better interpenetrating between OWS and TPS, the other is possible reaction between TPS and OWS with increased aldehyde content, resulting in poor melt processing behavior of the OWS/TPS composites. Furthermore, for the sample OWS (0.006)/TPS, the equilibrium torque decreased a little to 13.76 N·m. This implied that many more WS were destroyed deeply with further oxidation, resulting in the reduced viscous flow activation energy of OWS/TPS. Overall, the rheological processability of the OWS/TPS composite deteriorated after adding the OWS.
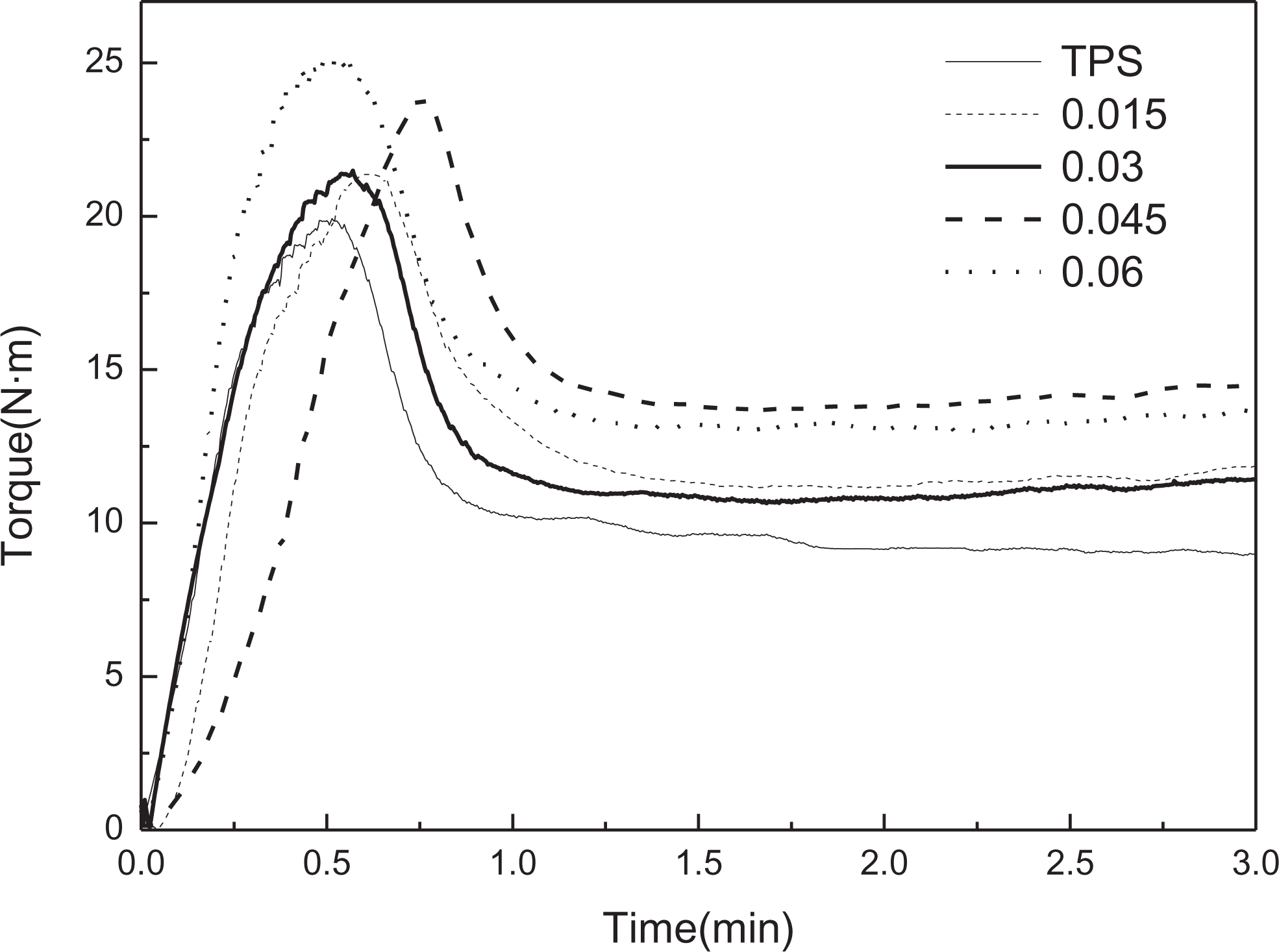
Torque rheological behavior of TPS and OWS/TPS. TPS: thermoplastic corn starch; OWS: oxidized wheat straw.
Rheological data for TPS and OWS/TPS composites.
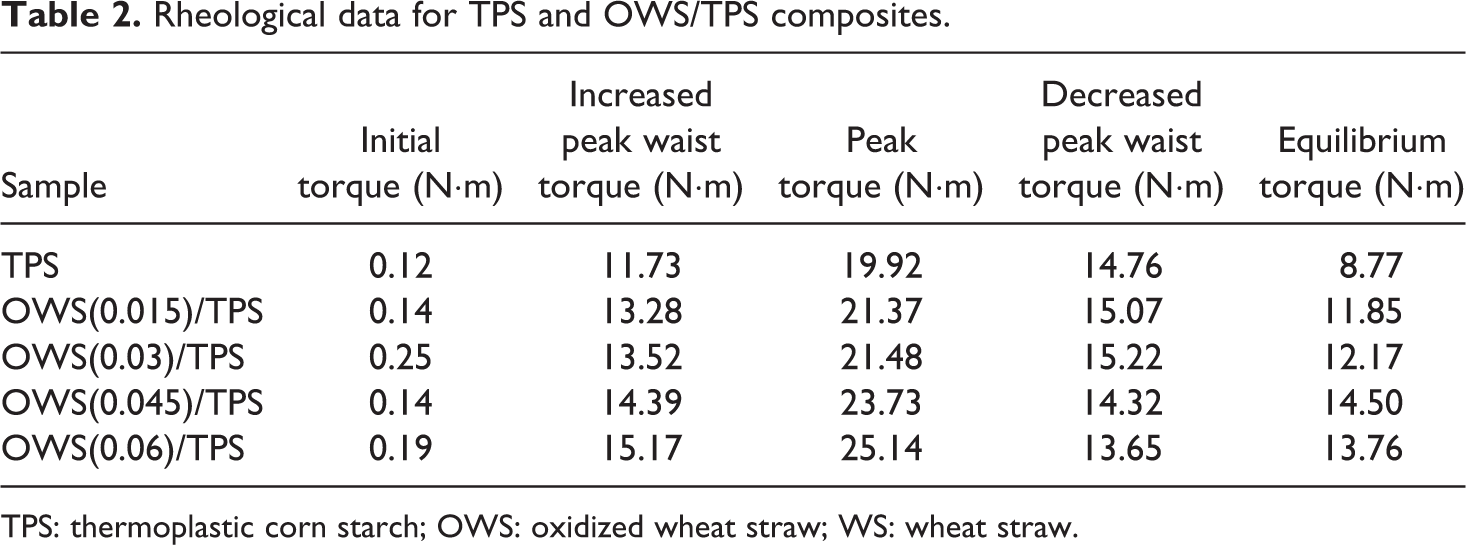
TPS: thermoplastic corn starch; OWS: oxidized wheat straw; WS: wheat straw.
Conclusions
In this study, OWS with different oxidation degree were blended with TPS as reinforcing fillers. Significant changes were observed on the WS surface as a result of oxidation pretreatment, and these changes promoted reactions and miscibility between OWS and TPS. On the basis of the data, it can be concluded that OWS can be used successfully as reinforcement in TPS composites. The above results suggest that it may be feasible to apply in other composites using oxidized WS, which is an underutilized agricultural by-product.
Footnotes
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work was supported by Jiangsu government Scholarship for overseas studies, China Postdoctoral Science Foundation (2012M521091), Natural Science Foundation of Jiangsu Province (grant no. BK20140967), and the Nanjing Forestry University Yong top talent program.
