Abstract
In this work, the effect of inclusion of nanographite particles in a polypropylene (PP) matrix is studied. Nanographite particles were obtained through ultrasound exfoliation from graphite upon using a water-based hydrophobically modified alkali-swellable emulsion (HASE) associative polymer as a surfactant. Results indicate that exfoliation renders particle size distribution ranging from 3 to 3000 nanometers. Nanographite was blended with PP through two extrusion processes: twin screw and single screw, the latter includes the coupling to a static-mixer head, to generate extensional flows. Concurrently, ultrasonic waves are applied to the molten flow through ultrasonic transducers attached to the mixing head, which induces high particle dispersion and good particle distribution in the polymer matrix. It was found that at HASE concentration of 5% by weight and sonication time of 14 days (period of the exfoliation process), optimum tensile properties of the compound were achieved. Also, with respect to the PP matrix, the rate of thermal degradation decreased from 2.1 (PP) to 1.9 (% °C−1), melt temperature ranged from 442°C (PP) to 396°C, and melt index decreased from 7.4 (PP) to 6.2 (g/10 min). Raman spectroscopy confirmed the exfoliation process, rendering sizes ranged from graphite particles of few graphene layers to micron-sized particles. Rheological measurements of the compounds revealed that the extrusion-ultrasound process influences the viscosity, storage, and loss moduli. The dispersion and distribution of nanoparticles improved the electromagnetic radiation shield (approximately 35%). The dielectric constant changed from 2.21 (pristine PP) to 9.02 for the compounds, which enables a good level of electrostatic charge dissipation.
Introduction
Graphite is a stable allotropic form of carbon under standard conditions (1 atm and 25°C). Its laminar graphene structure comprises spatial arrangements where Van der Waals forces are predominant. Different from the structures observed in carbon nanotubes or fullerenes, 1 graphene possesses a unique structure, due to high electron mobility at ambient temperature, transparency, high Young’s modulus, and excellent thermal conductivity. These properties make graphene a good candidate for new polymeric composites, with applications in electronics, conversion and energy storage, sensors, and electromagnetic shield devices.
Until recently, high-quality graphite particle production (nanographite and graphene) made possible specific applications of these compounds in nanoelectronics. New methods for production have been developed in continuously emerging trends. 2 –5 For example, the ultrasound-assisted exfoliation in liquid phase has been studied using various organic, aqueous, or ionic-liquid solutions. Hernández et al. 6 have shown the method for exfoliation using N-methyl pyrrolidone to produce graphene monolayers without defects. Yang et al. 7 studied the dispersion of graphene in sodium salts of 1-pyrene sulfonate using water as a solvent, in which graphite exfoliation is efficient and fast. Lotya et al. 8 used sodium dodecyl benzene sulfonate (SDBS) as surfactant for graphite exfoliation in water. Similarly, Green and Hersam 9 have used planar amphiphilic sodium cholate as surfactant for graphite exfoliation to control graphene layers width under centrifugation with density gradient. This process produce graphene leaves with widths that increase with buoyant density. In addition, Ledesma et al. 10 studied the reinforcement of polymers with delaminated graphene layers produced from graphite oxide under ultrasound within the extrusion process. Yoshida et al. 11 obtained graphene by means of chemical deposition on metallic substrates. The synthesized graphene can be separated from the substrate for deposition elsewhere. Zhamu et al. 12 patented a method to obtain graphene sheets using a non-ionic ethoxylated fluorosurfactant (Zonyl from Dupont) to prepare a suspension or slurry under ultrasound for exfoliation and reduction of particle size in short periods (2 h).
The main problem to produce graphite nanometric layers is not only the exfoliation process of graphite tactoids, but coalescence. Therefore, the effect of surfactants on graphite exfoliation has been given much attention. Concurrently, associative polymers have been studied for applications in stabilization of water-based paints and coatings. As opposed to traditional thickeners, these water emulsions are ambient friendly. The so-called HASE polymers (hydrophobically modified alkali-swellable emulsion) are associative acrylic-based polymers synthesized in acid dispersion of acrylic and metacrylic copolymers. At low pH, HASE presents a dispersion of insoluble particles (hydrophobic) in aqueous media. Upon neutralization at high pH, the hydrophobic groups become water soluble inducing a molecular expansion into elongated micelles. The viscosity of the solution becomes high and intermolecular attractions are feasible.
In this work, HASE solutions are used as dispersants in the process to exfoliate or separate the graphite layers mechanically. The resulting system is a mixture of graphene laminae and graphite nanoparticles with size distribution between 3 to 3000 nm. The objective is the reinforcement of thermoplastic matrices, which is accomplished without separation of these ingredients. Results indicate that this mixture with added additives is capable of providing the polymeric matrix with high dielectric constant, antistatic, and electrically conductive properties.
Experimental
Materials
Polypropylene (PP; Formolene 2306 N from Formosa Plastics Corporation, USA, high-impact copolymer, melt index of 6 g/10 min, density of 0.90 g cm−3); carbon nanotubes prepared from Plasticyl Nanocyl (Nanocyl SA, Belgium) PP 2001, with a 20% p/p content. Bentonite clay was obtained from AIDSA Co. (Mexico) graphite mesh 325 from Abaquim SA Co. (Mexico). HASE Primal TT-935 was from Rohm and Haas (Mexico) with pH 2.8, solids content of 30 wt%; maleic anhydride (MAH) 95% from Aldrich (Mexico); benzoil peroxide from PCO Co. (México) with melting point of 104–106°C. L-lysine HCL from Sehyex, S.A. de C. V.
Equipment
Planetary mixer, sugar glass mill (Overena Machinery, Mexico), single-screw extruder Haake Rheocord, and torque rheometer (Therma Scientific Inc., USA) with adapted head containing six piezoelectric ultrasound transducers were used in this study. The screw diameter was 30 mm with power of 1.5 HP, variable speed control, and four heating zones, and L/D of 24:1 mm. The following equipment are also used in this study: twin-screw corotating Haake-Rheocord 90 extruder; pellets feeder Maguire MLG-4-34, Maguire Inc., USA; low pressure dryer 30 from Maguire Products Inc, USA; high-velocity cutter mill; square wave generator; injection-molding machine Mannesmann DEMAG, Ergotech 50 (Germany) with screw diameter of 25 mm and L/D of 20; Izod impact notched with the ASTM D256 norm; universal test machine (Instron-1125, USA) with tensile test norm ASTM D638; thermogravimetric analyzer, Dupont Inst; differential scanning calorimeter (DSC) from TA Instruments, DSC 2910, modulated DSC; controlled-stress rheometer ARG2 (TA Instruments, USA) with parallel-plate fixture of 429-mm external diameter and 400-mm internal diameter; and a DT-1130 portable digital display electromagnetic radiation detector EMF meter tester with maximum-reading low-frequency of 1999 V m−1 and high frequency of 1999 μW cm−2from Amazon (USA).
Procedure
Nanoclay preparation
Nine liters of deionized water and 5 kg of bentonite were mixed under high-shear stirring, after which 0.5 kg of L-lysine-HCL were added, previously diluted in 1.36-L deionized water at 70°C. Keeping same temperature, stirring continued for 1 h. The bentonite-lysine mix was dried at ambient temperature in a high-speed blender which renders a powder-like texture with grain size of less than 44 μm.
Clay-lys is a salt-grafted clay of the amino acid L-lysine and has a carboxylic group end that reacts with anhydride groups of PP-g-MAH, producing a hybrid material. At the process temperature, weight loss and degradation occur in the reaction with carbonyl groups to produce aldehydes and ketones.
Graphite solutions preparation
Solutions were prepared with 18g graphite, 1 L of deionized water, and various HASE concentrations (1.5, 2, 5, 10, 15, 20, and30 wt%, with respect to graphite content). To fix the viscosity of the solution with 30 wt% solids, 90.6-mL 2-amino-2-metil-1-propanol (AMP)/for each 100 g of solution were prepared. Solutions were treated under ultrasound radiation at a frequency of 28 kHz with amplitude of 333 V. Residence time in the ultrasound bath was 7, 14, 21, and 28 days. Graphite solutions were dried by slow evaporation after ultrasound treatment. Particle size distribution of the graphite–water and graphite–water–HASE solutions was determined in a dynamic light-scattering Microtrac apparatus from Microtac Inc. (USA), model Nanotrac Wave (see Figure 1).
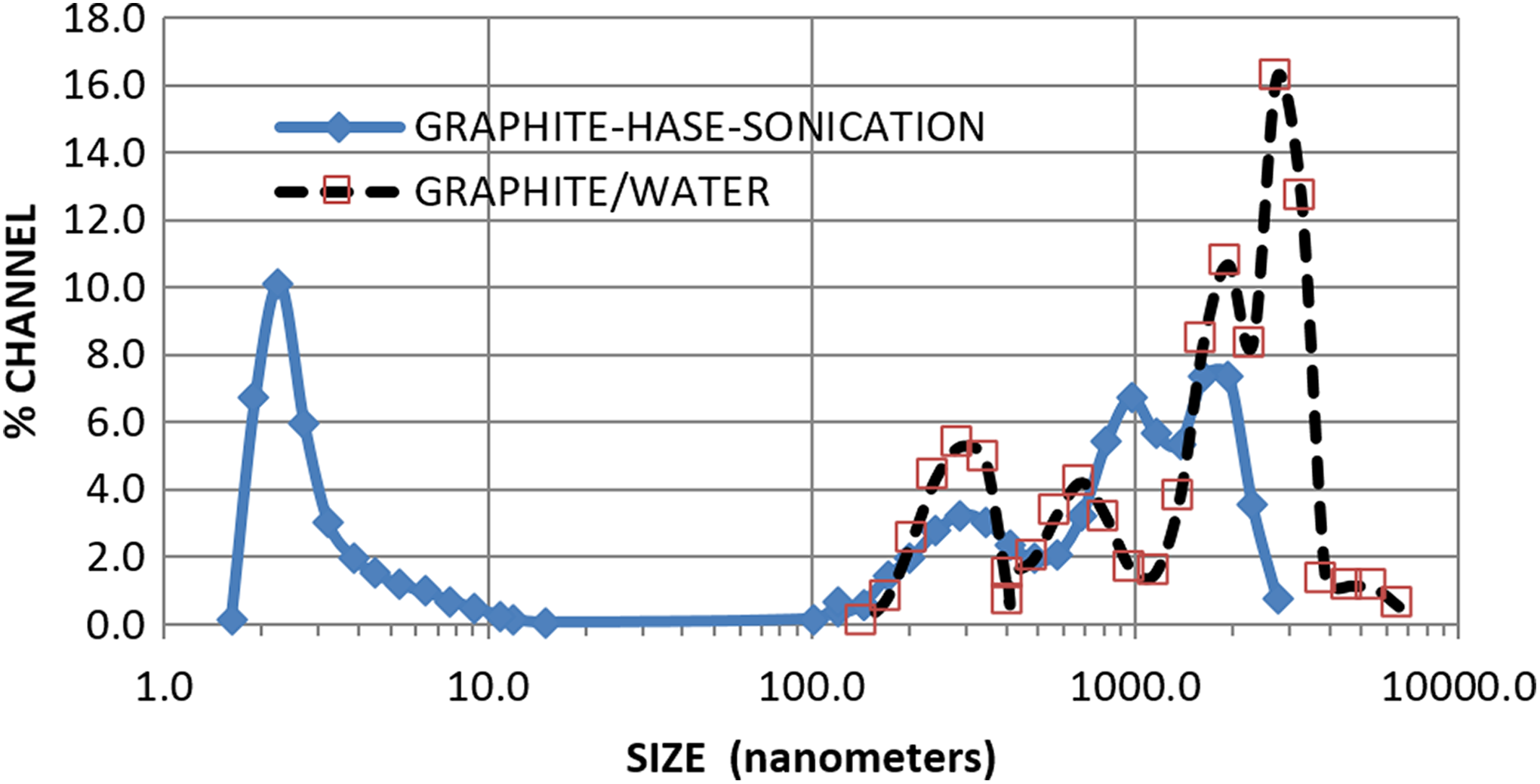
Distribution of particle size for graphite with water and graphite treated with ultrasound in a HASE solution.
During the graphite-HASE ultrasound treatment process, water evaporation is detected, even though the process is carried out at 23°C. At this temperature, no decomposition of the HASE associative polymer is found. The HASE surfactant decomposes at temperatures higher than 220°C, producing (like acrylic compounds) dioxide and carbon monoxide, low molecular weight compounds including monomers which can react with oxygen to produce ketones and formaldehyde. Since it presents thermal stability at the process temperature, it is likely that particles of nanographite are coated promoting adhesion (this effect is not measured in this work).
PP-graphite compounds
Blends of graphite (gr) and PP were prepared (with and without ultrasound) at a concentration of 3 phr, according to Table 1.
Sample description.
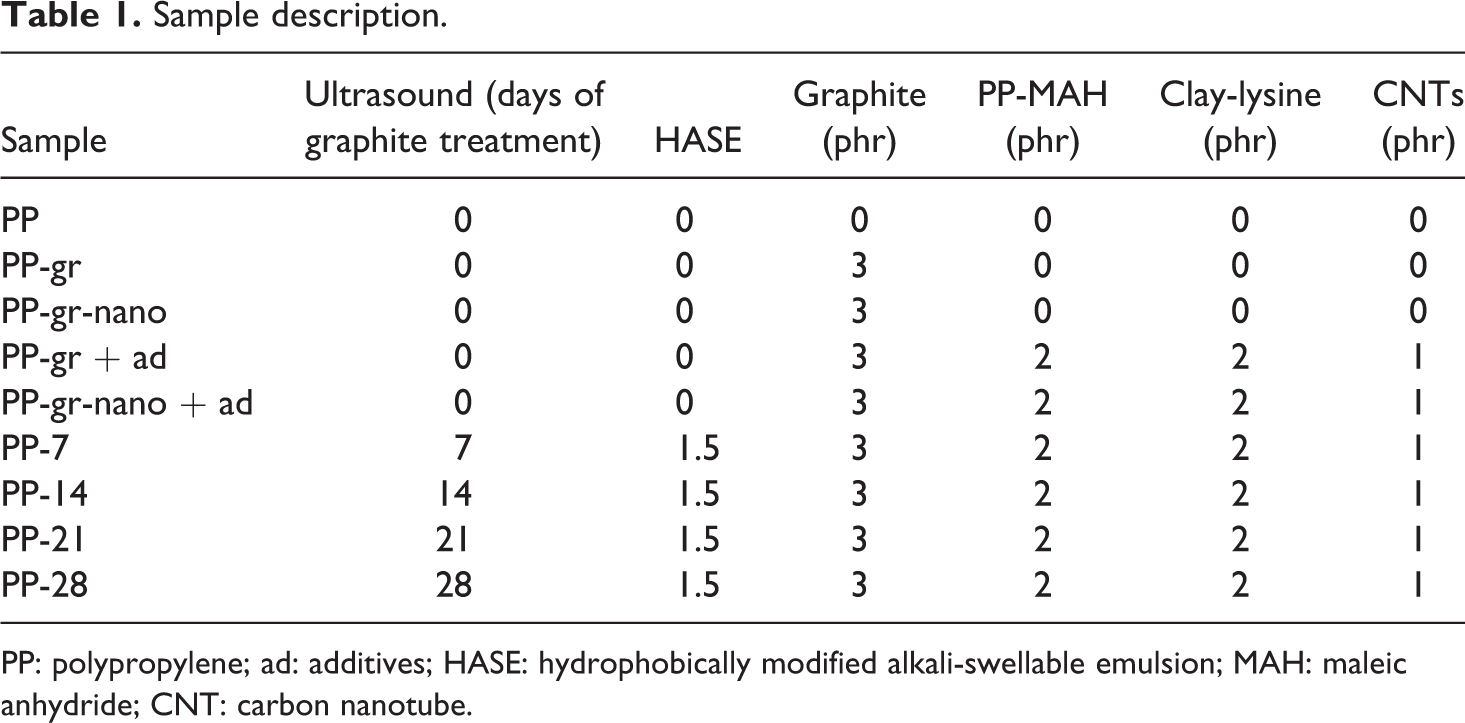
PP: polypropylene; ad: additives; HASE: hydrophobically modified alkali-swellable emulsion; MAH: maleic anhydride; CNT: carbon nanotube.
PP-g-MAH is PP to which a MAH functional group has been grafted, becoming a compatibilizing material linking the organic (PP) and the inorganic material (clay-lys), hence producing a hybrid material. It promotes adhesion and cohesion (not measured directly in this work) and is stable up to the process temperature (220°C). Also, carbon nanotubes are stable up to the process temperature of 220°C.
Exposure time of PP was set at 7–28 days, after which solutions with various HASE contents were prepared (see formulations disclosed in Table 2). Melt extrusion of the blends comprises two stages in a cascade process: twin-screw extrusion followed by single-screw extrusion with adapted ultrasound special static-mixing head. This head has six piezoelectric transducers of 50 W each one to provide the ultrasonic waves, attached to a squared-wave generator that controls the amplitude and frequency of the radiation.
Twin-screw-processed samples with ultrasound-treated graphite (14 days) varying HASE content.

PP: polypropylene; HASE: hydrophobically modified alkali-swellable emulsion; MAH: maleic anhydride; CNT: carbon nanotube.
In Figure 2, the single-screw extruder is schematically illustrated. In the frontal picture, the transducers are observed while in the lateral view, the four heating zones are depicted. Temperature profiles for both extrusion processes were 190, 200, 210, and 220°C for the feed, compression, dosage, and die sections, respectively.
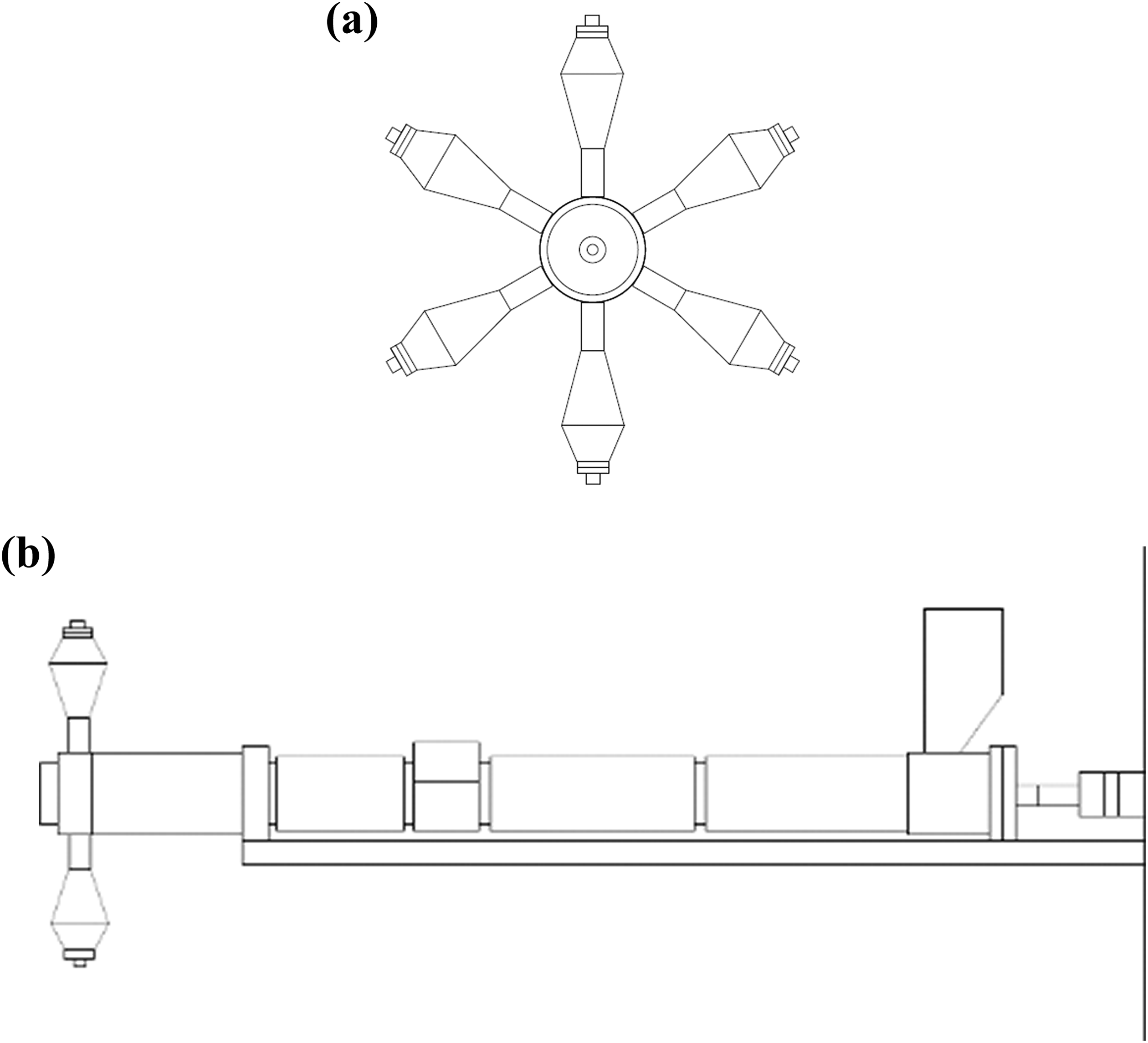
Static head mixer with ultrasound transducers: (a) frontal view, (b) side view.
Results and discussion
Mechanical analysis
Mechanical tests (specifically tensile under constant rate of deformation) determine fracture strain and tenacity. Such tests are very sensitive to distribution and dispersion of nanometric-sized particles in the polymer matrix, from which the compatibility of particle matrix can be assessed. According to tensile data in Table 3, the use of HASE in the formulations leads to a decrease of the mechanical properties. On the other hand, the effect of residence time under ultrasound of the graphite solutions is important. For instance, after 14 days, the fracture strain and tenacity of the composites increase with respect to the pristine polymer from 79% to 102% and from 14.8% to 19.2%, respectively. On this basis, the optimum residence time was determined for graphite exfoliation. Thereafter, the next step considered variable HASE concentrations in the graphite solutions. In Table 2, the samples are described, while in Table 4, tension data are disclosed. Accordingly, HASE content of 5 wt% related to the graphite weight presents the best fracture strain and tenacity values.
Impact and tensile tests, 1.5 wt% HASE.
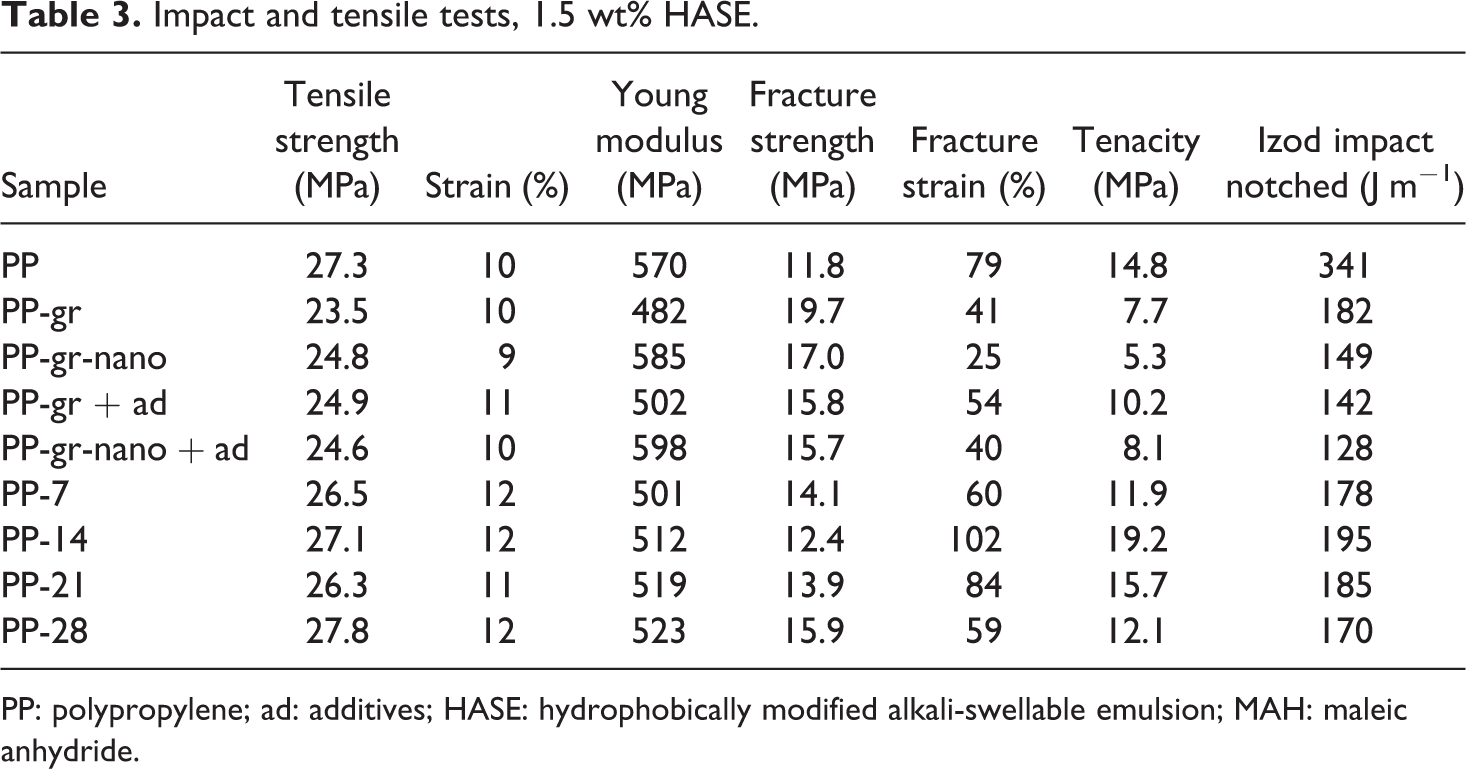
PP: polypropylene; ad: additives; HASE: hydrophobically modified alkali-swellable emulsion; MAH: maleic anhydride.
Impact and tensile tests varying HASE concentration (ultrasound-treated graphite: 14 days)
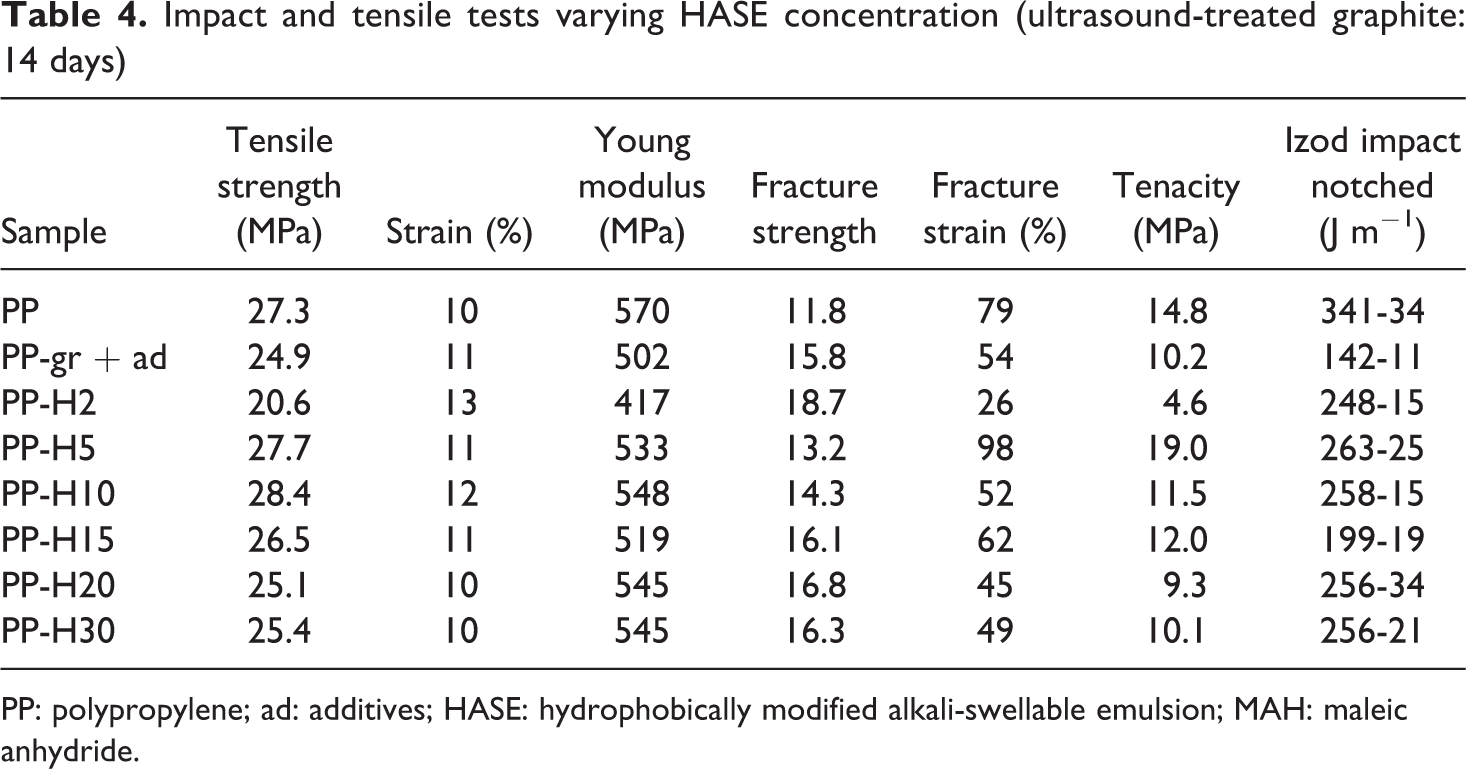
PP: polypropylene; ad: additives; HASE: hydrophobically modified alkali-swellable emulsion; MAH: maleic anhydride.
Standard deviations are written next to the mean value in Tables 3 and 4. In both, tensile strength, strain, and Young’s Modulus show standard deviations of less than 15%. Fracture strength, fracture strain, and tenacity exceed this value, producing significant values of the percentage of standard deviation around the mean value. Measurements of mechanical properties at large deformations induce large values of these statistical values. This effect may be caused by several factors: a wide molecular weight distribution of the polymer, a different crystallinity of the test specimens, effect of stress concentration by particle reinforcement and additives, a non-optimized dispersion of the nanoparticles in the polymer matrix (due to the particle size distribution), and so on. This is an interesting issue for future research.
Thermal analysis
Clay-L-lysine
Thermogravimetry was used from ambient temperature to 600°C. Pristine clay weight loss is 4.53 wt%, and after treatment with the salt of L-lysine, the weight loss is 1.96 wt% at the same temperature. Amino acid–clay interactions confer a high thermal stability at the processing temperature.
Graphite-HASE
Same analysis was performed on the graphite samples with varying HASE content, with exposure time of 14 days. Table 5 shows that the graphite sample without treatment with HASE has similar weight losses, which means that these losses are attributed to trapped water at the processing temperature (220°C).
Weight loss of graphite samples with varying HASE content.

HASE: hydrophobically modified alkali-swellable emulsion.
Graphite effect at the onset and final stages of the degradation process, with the rate of degradation, is exposed in Table 6. The rate of weight loss is associated with material structuring: a low rate means that temperature resistance is higher. In this case, the value given in the table corresponds to graphite treated with 5 wt% HASE solution with a degradation rate of 1.9% °C−1. Initial and final temperatures are lower as compared to those of the pristine polymer.
Temperature and rate of degradation for various blends obtained from TGA thermograms.
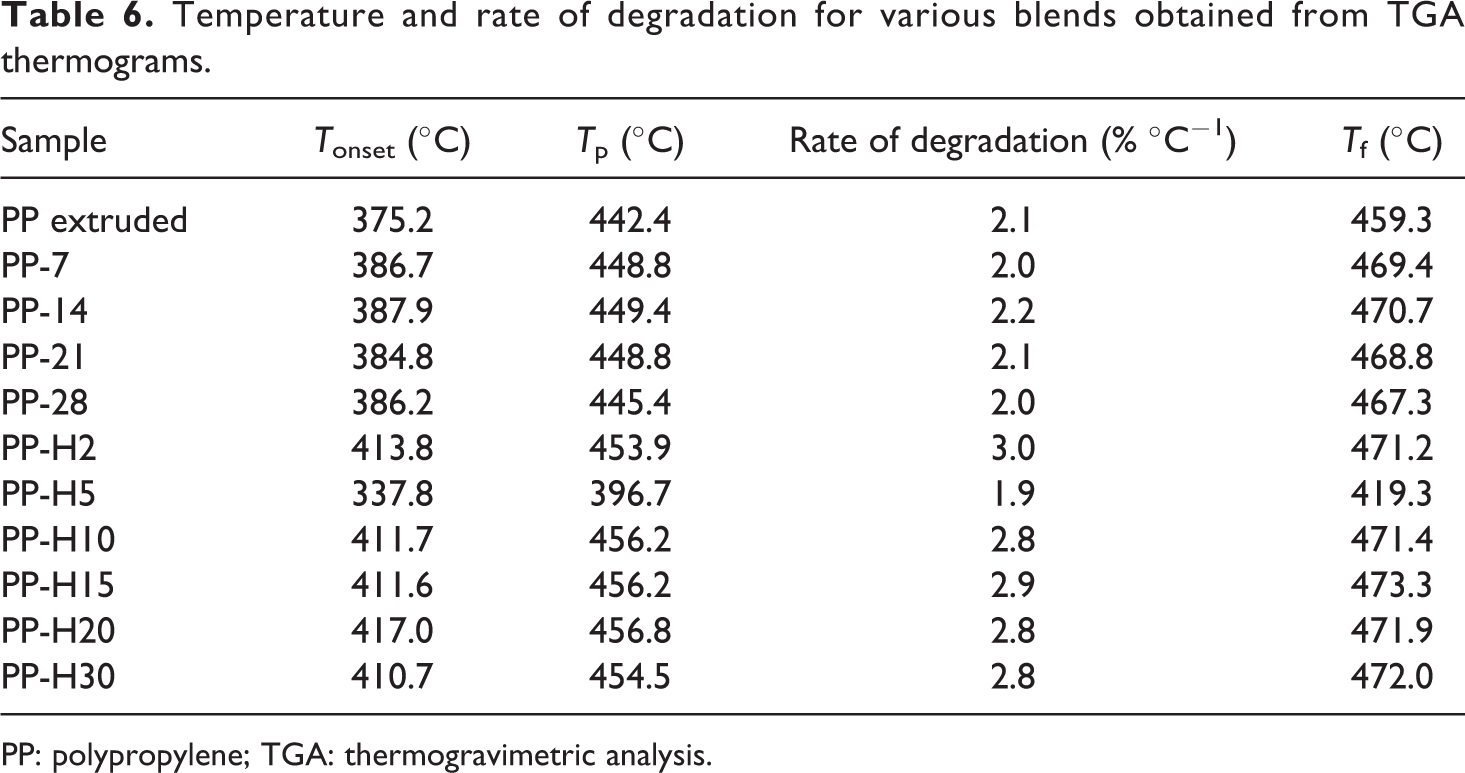
PP: polypropylene; TGA: thermogravimetric analysis.
Flow index
Small polymer chains have the ability to be more fluid-like, as opposed to large molecules which have more resistance to orient and extend under flow conditions, and therefore the flow index is inversely proportional to the molecular weight. Measurements of flow index were performed according to the ASTM D1238 norm, method A. Table 7 discloses data of flow index at a given temperature for all samples. It is interesting to observe that a lower flow index with respect to the pristine PP polymer is revealed in the graphite samples treated with HASE with concentrations equal or larger than 5 wt% under ultrasound (14 days). Particularly, the sample with 5 wt% HASE presents higher tenacity, fracture resistance, and an optimum value of the rate of degradation (6.2 g/10 min) which reflects a more structured material with higher flow resistance.
Flow index for various PP samples with additives.

PP: polypropylene.
Rheological analysis
Figure 3 shows the shear viscosity versus shear rate of the samples processed by twin-screw extrusion wherein the exposure time of graphite under ultrasound is varied (PP-14 and PP-21) with 1.5 wt% HASE. Comparison is made with samples containing non-treated graphite (PP-gr) and those containing additives (PP-gr + ad). Graphite formulations without ultrasound present shear thinning and lower viscosity than those processed with ultrasound, revealing a larger surface area of the smaller particles due to exfoliation. The effect of the additives in the samples without ultrasound treatment is not substantial (PP-g-MAH, clay-L-lysine, carbon nanotubes) in contrast to the large effect of the ultrasound in the graphite/HASE systems. Among these systems, sample PP-21 shows the larger viscosity, revealing a clear ultrasound-induced exfoliation with larger exposed area of the particles. Mechanical properties are similarly influenced.
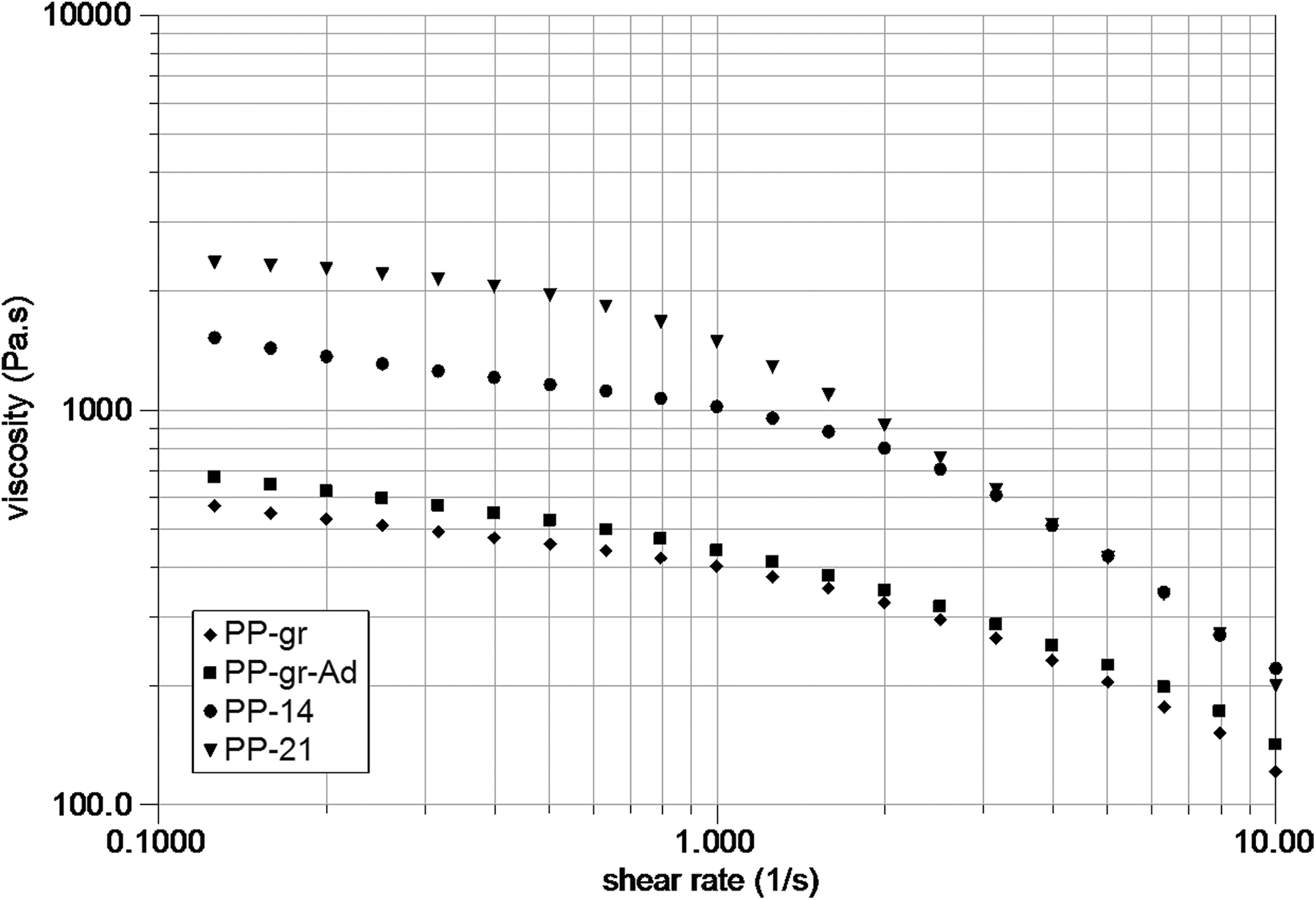
Viscosity versus shear rate in samples processed by twin-screw extrusion with treated graphite 14 and 21 days at HASE content of 1.5% (PP-14 and PP-21) and for pristine graphite with and without additives.
Figure 4 displays the complex viscosity versus frequency in small amplitude oscillatory shear of samples with graphite treated with ultrasound and a reference curve of a sample with pristine graphite. Concurrently, the effect of different HASE concentration is exposed. Curves with treated graphite reveal higher viscoelasticity than those with non-treated graphite, and larger HASE contents induce larger values of viscosity. A material with particles of larger surface area is likely to have comparatively higher viscosity, and thus the dispersant action of HASE provides particle dispersion avoiding coalescence of nanographite particles. Simultaneously, the processing with ultrasound of the PP/graphite system is likely to produce free radicals that promote cross-linking and/or entanglements, increasing the system viscosity.
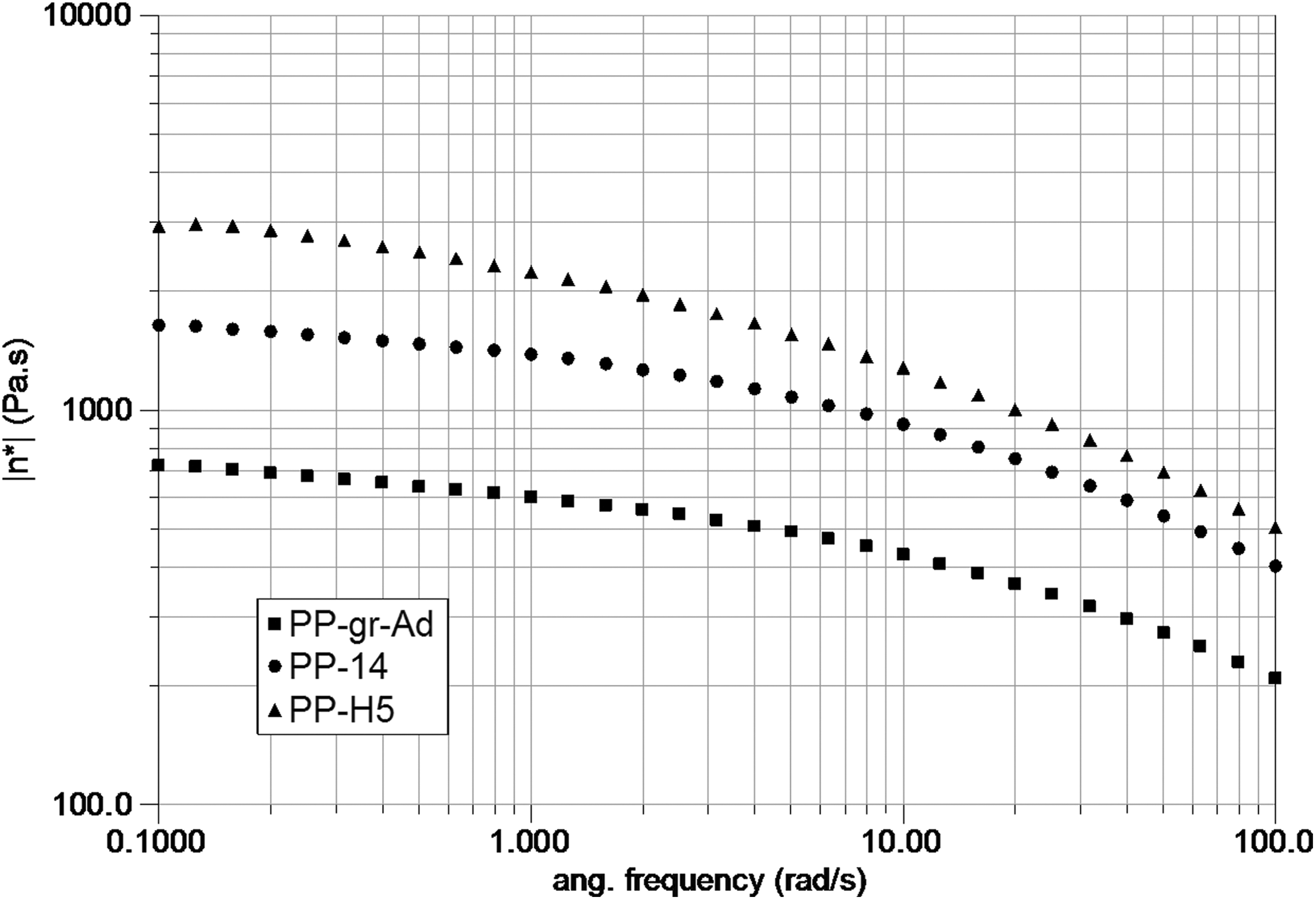
Complex viscosity versus frequency for twin-extruded samples with same additives. A reference sample contains pristine graphite while the other samples contain a graphite solution treated under ultrasound for 14 days with HASE contents of 1.5% (PP-14) and 5% (PP-H5).
Figure 5 shows the complex viscosity variation with frequency to illustrate the effect of varying HASE concentration. Graphite is treated under ultrasound for 14 days in all samples (PP-H5, PP-H10, PP-H15, and PP-H20). These results reveal that upon increasing HASE concentration, the value of the viscosity saturates and no substantial change is observed at high HASE contents.
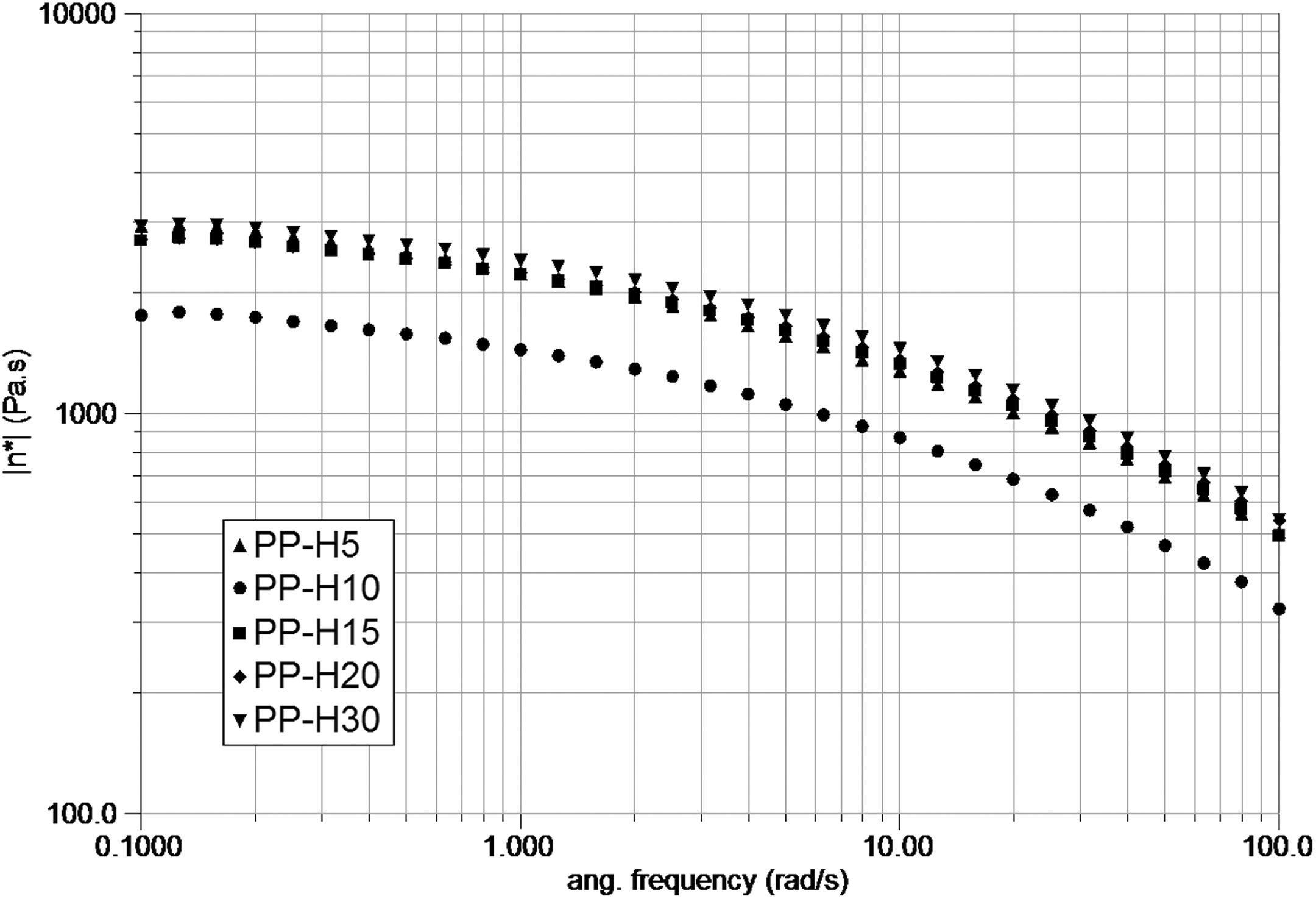
Complex viscosity versus frequency curve for ultrasound-treated graphite (14 days) with various HASE concentrations.
Figure 6 shows the variation of the storage and loss modulus with frequency. The degree of viscoelasticity corresponds to data shown in Figure 4, with predominant viscous modulus. The limiting slope at low frequency manifests the degree of particle/matrix interaction, and this is higher in the PP-H5 system, in accord with flow properties of a more structured material.
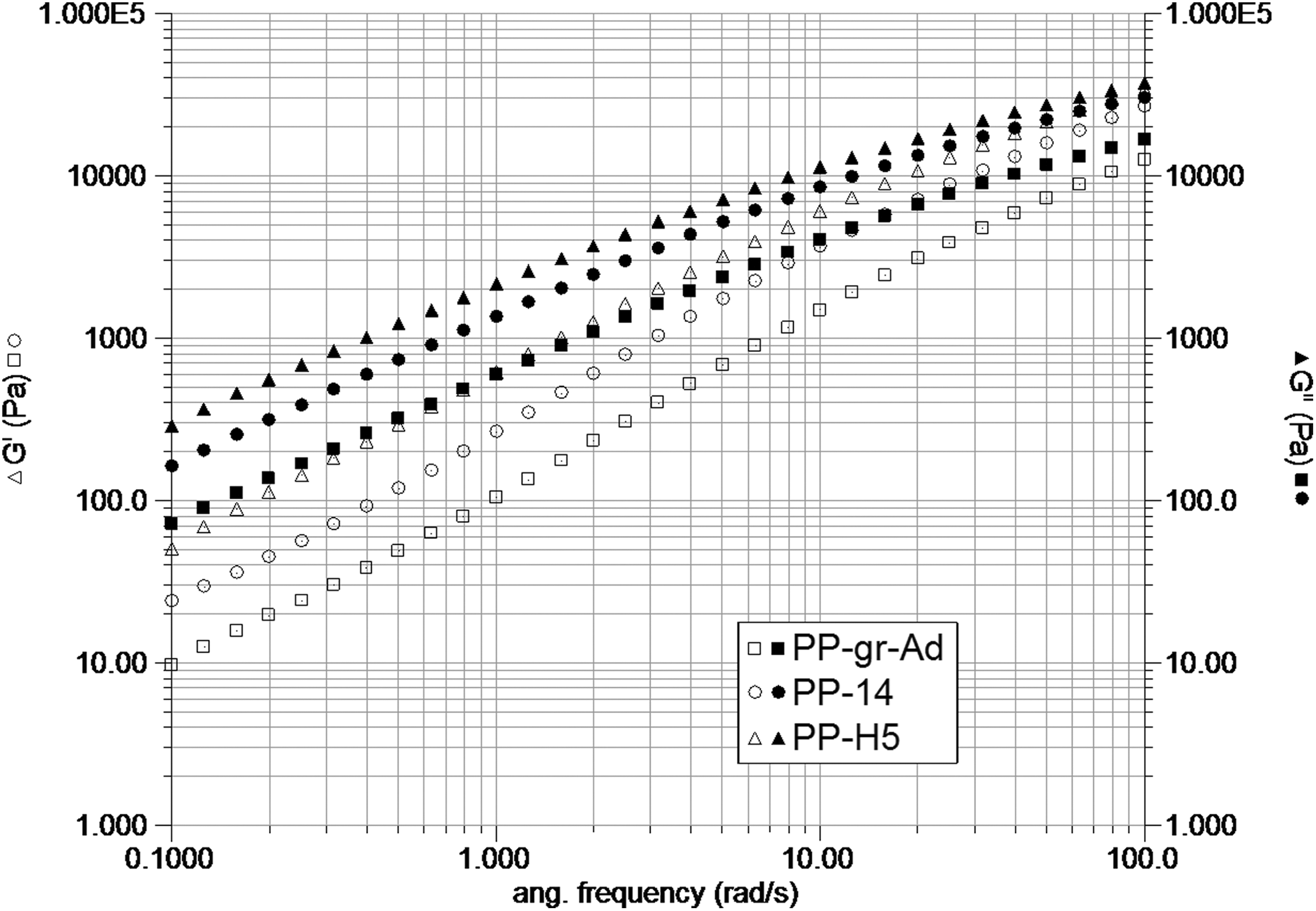
Storage and loss modulus versus frequency for pristine graphite with additives (PP-gr-ad), ultrasound-treated graphite with 1.5 wt% HASE (PP-14) and 5% HASE (PP-H5%).
Raman spectroscopy
This technique is commonly used to characterize graphene layers, especially the 2-D peak that identifies graphene monolayers when is a single-band peak. In the case of multiple-band peaks (four bands, for instance), the spectra reveal arrangements AB. The shape of the 2-D peak thus changes with the number of graphite layers, although this band is not well characterized when 8–10 layers are present. In disordered systems, peak 2-D generates two additional bands: first order peak D (1350 cm−1) and peak D′ at 1620 cm−1. This technique was implemented on large number of small graphite leaves deposited on silicon substrate. In Figure 7, untreated graphite samples and graphite with various HASE contents (5, 10, 15, and 20 wt%) are exposed. The 2-D peak is observed at 2700 cm−1 associated to graphene or graphene layers, while the graphite peak is wider and shifted to the right. Graphene bands are normally observed at lower wave numbers, since the electronic clouds are less shifted with respect to those of graphite. Since the Raman spectrum arises from the interaction of an incident photon with the electronic cloud of graphene, a larger number of graphene layers induces wider peaks at higher wave numbers. Figure 7 shows the PP-H5 band, which is the first and more intense peak observed on the left of the remainder samples. This peak is thus associated with various graphene layers with structural defects, originated by interactions with HASE. The less intense signal corresponds to PP-H20, associated to lesser structural defects.
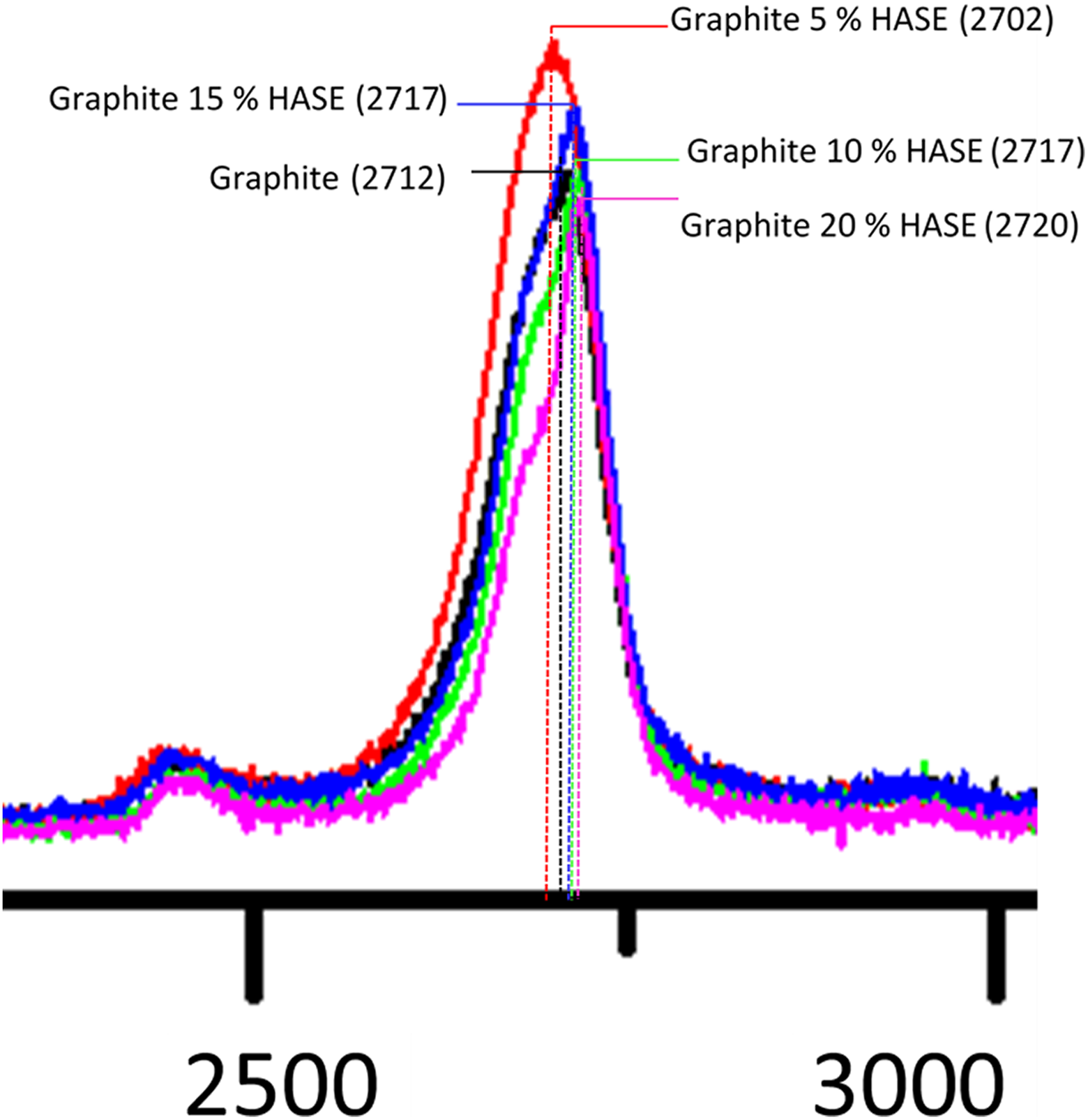
Raman spectrum in samples with ultrasound-treated graphite with varying HASE concentration.
Electromagnetic radiation
Electromagnetic fields generated by electric currents are given attention due to the possible negative effect of living organisms. The magnitude of these fields depends on the distance to electric sources, properties of the protective materials, walls, and so on. Since polymers are dielectric in general, the addition of conductive materials changes drastically their electric conductivity and electromagnetic properties. New materials are sought as useful compounds for protective electromagnetic shields, due to their corrosion resistance, processability, cost, and so on. Carbon black presents advantages for shield applications, but due to the high contents needed, even as high as 40 wt%, mechanical properties diminish considerably. In this regard, new materials are sought with conductive properties which can combine a good dispersion and distribution of nanoparticles to achieve an optimum percolation threshold without affecting substantially the mechanical properties of the material. In this work, the electromagnetic radiation was measured in an electrical conductor at 60 Hz and 120 V. The measuring device has an antenna placed at 1 cm from the radiation source. Samples of the material are placed between the radiation source and the antenna. Results are shown in Table 8, wherein the new materials (graphite with 5 wt% HASE ultrasound treated for 14 days, PP-H5) induce a decrease of the radiation level, or shield percentage, of 35%.
Electromagnetic radiation determination.

EMF: electromagnetic field; PP: polypropylene.
Dielectric analysis
The dielectric constant values range from 1 in vacuum to 2–7 for common dielectric materials and higher for electrical conductors beyond the percolation threshold. In this work, the dielectric constant was measured in a parallel-plate condenser following the ASTM D150 norm at 30°C for 120 min, with a frequency of 1 kHz and 1 V voltage. The reference PP dielectric constant amounts to 2.21, and the remainder compound values in Table 8 range between 8.76 and 9.02 (PP-H5).
Conclusions
The methodology previously exposed with ultrasound-treated graphite in a HASE solution promotes defoliation without observed coalescence of graphite particles, with values that range between nanometric to micrometric sizes. The optimum residence time under ultrasound in a 5 wt% HASE solution (with respect to graphite weight) is 14 days. Raman spectroscopy identified the amounts of nanographite and multilayer graphene. Upon mixing with the PP matrix, the resulting blend possesses larger values of fracture strain and tenacity with respect to those of the pristine polymer, which reveal high homogeneity and good dispersion and distribution of particles in the polymer matrix. The dispersant (HASE) concentration has an important effect at relative low contents (specially the 5 wt% in the PP-14 system) but saturates at higher concentrations. Higher viscosities are observed in samples treated with ultrasound, revealing a more structured material. The rate of degradation of this compound diminishes from 2.1 to 1.9% °C−1, exhibiting higher thermal resistance. The dispersant action of the associative polymer (HASE) induces lower fluidity index in the ultrasound-treated systems, due to the increase in the surface area of the particles. This material (PP-graphite/HASE/with ultrasound) is a good radiation-shield ingredient which lowers the electromagnetic radiation more than 35% with high dielectric constant, allowing its use in electronic devices.
Footnotes
Acknowledgements
The authors would like to thank Salvador López Morales for particle size and Raman measurements, Leonardo Moreno Morales for rheological characterization, Eliezer Hernández Mecinas for mechanical properties determinations, and Damaris Cabrero Palomino and Karla Eriseth Reyes Morales for thermal analysis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
