Abstract
Three different types of styrene-ethylene-butylene-styrene block copolymer (SEBS) with varying ratios of styrene and rubber were melt-compounded with exfoliated graphite nanoplatelets at different loadings. The morphological, thermal, and mechanical properties of the nanocomposites were studied and compared. Morphological observation under SEM and AFM found that the xGnPs were dispersed at the sub-micron level throughout the SEBS matrix. Good interfacial adhesion between the xGnPs and the matrix was also observed. However, the behavior of dispersion was dependent on the styrene/rubber content. SEBS with higher styrene content showed better dispersion and strong interfacial adhesion between the xGnPs and SEBS matrix. These results contributed to the enhancement of the tensile strength of the nanocomposites. Low styrene content behaved like rubber that resulted in low tensile strength but higher elongation compared to SEBS of different amounts of styrene. The XRD patterns indicated that the melt compounding process did not change the d-spacing of xGnPs in all types of SEBS. From the thermal analysis, there was no change in the glass transition of the polymer and no improvement in the thermal stability of the nanocomposites.
Introduction
Graphite and graphitic fillers such as expanded graphite, graphite nanoplatelets, and graphene are the most studied fillers for elastomers because of low cost and remarkable functional properties that can be used in many engineering applications. The excellent properties of graphite in in-plane mechanical, structural, thermal, and electrical are significant at the nanoscale if the graphites are in the form of thin nanoplatelets and single graphene sheet.1-4 The polymers based nanocomposites incorporated with graphite and graphitic nano fillers have shown substantial improvement in mechanical, thermal, electrical properties.5-14 While the graphene used for barrier membrane applications exhibited excellent impermeability to oxygen and effective optical property together with ultrahigh electron mobility.15-17 The nanocomposites based on exfoliated graphite nanoplatelets (xGnPs) showed considerable improvement in mechanical, thermal, electrical and thermal conductivity.7,8,18-27 The graphite nanoplatelet was also used as reinforcement for nanofibers. 27 Graphite is easy to exfoliate due to its sheet-like structure composed of carbon layers arranged in alternating stacked sheets that are regularly spaced at 0.335 nm apart and weakly bonded by Van de Waals forces. The separated sheets/layers can be down to nanometer thickness, high aspect ratio around 200–1500 and high modulus graphite nanosheets can be formed. It has the susceptibility to undergo exfoliation under proper conditions, that is, to render pristine tactoids into graphite nanoplatelets (GnPs, several layers stacks). The pores in the expanded layered structure of graphite have been suggested to be capable of absorbing polymer molecules and lead to better interactions between the platelets and the host polymer in graphene/polymer nanocomposites. 28
Block copolymers are special group elastomers, consisting of more than two kinds of chemically distinct polymers linked by covalent bonds. They can undergo microphase separation into self-assembled ordered morphologies as a result of the incompatibility between the different blocks constituting the copolymer. 29 The mechanism of nanostructure development due to microphase separation leads to the understanding the self-organization of surfactants. 30 These self-assembled block copolymers are considered to be composite materials since they consist of physically cross-linked glassy and rubbery microdomains. Their morphological characteristics (microdomains, orientation) can be controlled by varying the types of blocks, volume fractions, molecular weight and history of processing conditions.31-34
Among the block copolymers elastomer, the styrene-ethylene-butylene-styrene block copolymer (SEBS) has attained great interest for many researchers due to high elasticity, high rigidity, thermo-oxidative stability, UV resistance, high service temperature and diversified application.
35
SEBS is obtained by hydrogenation of the central block of poly(styrene-b-butadiene-b-styrene) copolymer, which confers heat resistance due to it the absence of double bonds that are sensitive to thermal oxidative degradation.
36
SEBS is a thermoplastic elastomers having polystyrene content in the range between 20 to 35%. It has a glass transition temperature (
In this work, the nanocomposite polymer was prepared by embedding xGnPs into styrene-ethylene-butylene-styrene block copolymer by simple melt compounding approach. Their effects on the morphological properties, thermal behavior and mechanical properties were reported and discussed. The effect of the dispersed platelets on the microphase separation behavior of different ratios of styrene/rubber of SEBS matrix was also studied. The research hopes to contribute to the new advancement in the field of soft and stretchable polymer nanocomposites for dielectric elastomer and electronic applications.
Methods
Materials
Three different types of polystyrene ethylene butylene styrene (SEBS) block copolymers were used in this experiment with different styrene/rubber ratios coded as SEBS-1 with 35/65; SEBS-2 with 29/71 and SEBS-3 with 22/78. These materials were supplied by Kuraray Co. Ltd, Japan (SEBS 1 and 3) and Shell Co. Ltd, USA (SEBS-2). The expandable graphite flakes from Anthracite Industries, Asbury Graphite Mills, Inc (USA), is a graphite-sulfuric acid intercalated compound with expansion ratio of 290:1 and Table 1 presents the properties of these expandable graphite flakes.
Product description and typical particle size analysis of EG flakes.

Exfoliated graphite nanoplatelets (xGnPs)
The expandable graphite flakes consist of several layers of graphite sheets as seen in Figure 1(a). These graphite nanosheets contain functional groups such as –OH, –COOH after acid and high temperature treatments,44,45 thus causes exfoliation. The expandable graphite flakes were heat-treated at 600°C in a furnace for 30 seconds. This resulted in a sudden increase in dimension perpendicular to the carbon layers of the intercalated graphite, thus, forming vermicular or worm-like structure. The loosely connected graphite nanosheets, as shown in Figure 1(b) were later separated into individual nanosheets by sonication in acetone bath (20g/100 ml of acetone) for one hour at room temperature. The xGnPs were filtered using filter paper Whatman no. 41 and dried at room temperature then oven dried at 40°C. The layers of detached nanoplatelets can clearly be seen in Figure 1(c). The XRD patterns for the expandable graphite (EG) and xGnPs are shown in Figure 2. The typical d-spacings between the carbon layers in graphite at 3.35 Å, 2.52 Å, 2.02 Å, and 1.24 Å were detected. However the intensity of the d-spacing in 3.35 Å increased when it was exfoliated at high temperature and this suggests that the structure of xGnPs became more crystalline.
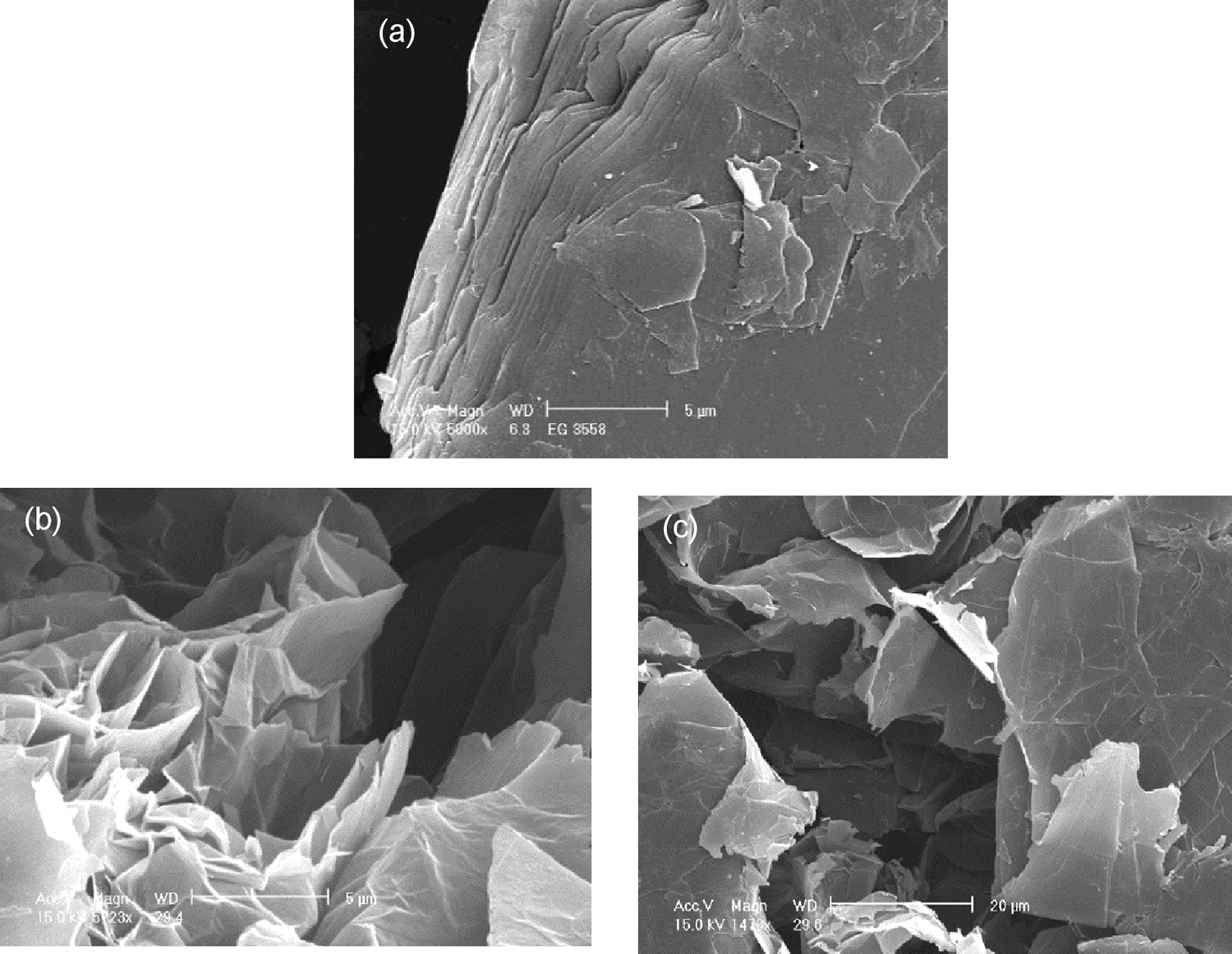
SEM images of (a) EG flakes, (b) expanded graphite (magnification: 5 µm), and (c) xGnPs (magnification: 20 µm).
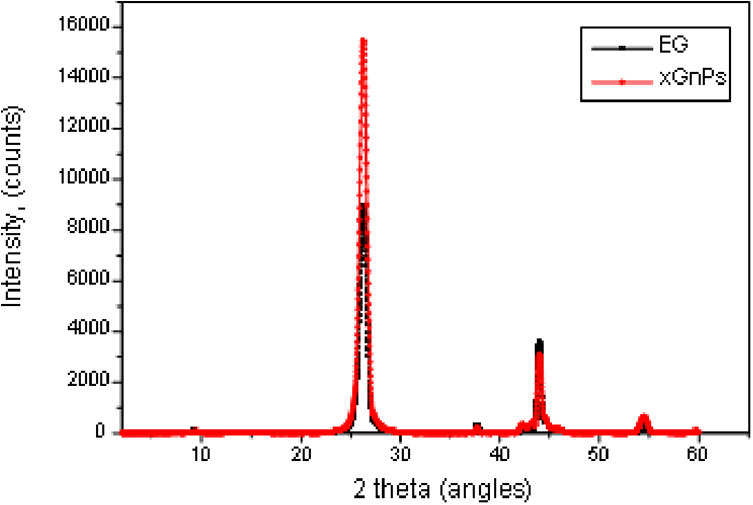
XRD of EG and xGnPs.
Melt-blending of SEBS and xGnPs
The nanocomposites were prepared by compounding polystyrene-b-poly(ethylene-co-butylene)- b-polystyrene block copolymer (SEBS) with different amounts of graphite powder at 1, 2, 3, 5, 7, and 10% using laboratory rolling machine that was heated at 180°C. The mixing time was 20 min after the formation of SEBS sheet on the anterior cylinder of the rolling machine and addition of the xGnPs powder. The mixtures were formed into sheet by pressing using mold plate with dimension of 120 mm × 180 mm × 2 mm at 180°C with a pressure of 50 kg/cm2 for 10 min, from which specimens were cut for the determination of their properties.
Characterization
The morphological behavior of the tensile fractured of nanocomposites was observed using a Scanning Electron Microscopy (FE-SEM, model Philips XL 30 S, Netherland) operated at 15 kV. Samples were coated with gold using JEOL JFC-110E, Tokyo, Japan, prior to scanning.
The atomic force microscopy (AFM) measurements were carried out with a XE-100 (PSIA) atomic force microscopy applying non-contact mode and scanned at 30 µm and 5 µm at different modes and techniques.
X-ray diffraction (XRD) profiles were recorded using Shimadzu X-ray Diffractometer Lab X-XRD 6000 in the angular range from 2° to 40° (2θ) at an operating voltage at 40 kV and a current of 20 mA with a wavelength of 1.7901 Å.
Differential scanning calorimetry (DSC) experiments were performed using a TA Instruments, TA DSCQ10, USA calibrated with indium standards. Hermetically sealed aluminum DSC pan containing 10 mg of samples was used. The thermal transitions of samples were also scanned from −150°C to 180°C with heating rate of 10°C/minutes. Glass transition temperature (corresponding to the middle point of the baseline shift) was measured.
For determining the degradation temperature of the nanocomposites, thermogravimetric analysis (TGA) was carried out in a DuPont TA2100 TGA in the temperature range of 50–700°C with a heating rate of 20°C/min.
The tensile properties were determined in accordance to ASTM D412 using a Tensilon Universal Testing Machine with the cross head speed was 500 mm/min and a load cell of 10 kN capacity. The reported values were the average value of five experimental results.
Results and discussion
Morphological characteristics
To determine the dispersion and reinforcing effect of xGnPs in SEBS/xGnPs nanocomposites, the tensile fractured of samples were observed by FE-SEM. A comparative examination of the SEM images of SEBS-1, SEBS-2, and SEBS-3 loaded with selected amounts of xGnPs are presented in Figure 3(a) to (l), respectively. The figures demonstrated the interaction between the polymers and xGnPs being attached to the polymeric matrix. Figure 3(a) to (d) shows the SEM images of SEBS-1/xGnPs with 2%, 5%, 7% and 10% of xGnPs. It was clearly seen that there existed some interactions between the matrix and graphite sheets and better dispersions of xGnPs in the polymer matrix can be achieved. The morphology of all the nanocomposites containing different amounts of xGnPs shows the intimate contact and high embedding with polymer matrices which indicates good interfacial bonding between nanoplatelets and the SEBS matrix. To further evaluate the adhesion between the xGnPs and the SEBS matrix, the fractured sample of 5% xGnPs was examined at higher magnification. As can be seen in Figure 3(b) insert, the matrix adhered to the surface of the xGnP and well-dispersion of xGnPs in polymeric matrix, indicating strong interaction adhesion between the xGnPs and the SEBS matrix. These features resulted to more effective absorption of SEBS matrix to the xGnPs. When the styrene content was reduced in the case of SEBS-2, some discrete xGnPs pulled out of the matrix from the tensile fractured, as shown in Figure 3(e) to (h). This behavior was also observed of similar styrene/rubber ratio by other researchers, 25 indicating that there is also strong interfacial adhesion between the xGnPs and the SEBS matrix. In the case of SEBS-3, a good adhesion between the SEBS matrix and the xGnPs was observed (Figure 3(i) to (l)). It was noticed that the 2% of xGnPs can hardly be seen by SEM (Figure 3(i)). Likewise, the improved dispersion and adhesion of the SEBS to xGnPs may be attributed not only to the modestly higher surface area, crystallite size and greater surface roughness of the nanofiller but as well the styrene and rubber ratio. The higher amount of styrene component in SEBS resulted to better dispersion of xGnPs than those of low amount of styrene. From the SEM images of the SEBS/xGnPs nanocomposites, it is apparent that the xGnPs particles also break apart during the shear mixing procedure, leading to smaller platelets. The thinner size and nanoporous morphology of the xGnPs render them more likely to be reduced in lateral dimension during shear mixing. This may lead to the predominantly smaller particles observed that can enhance dispersion with the polymer. Similar adhesion of polymer to carbon nanotubes during fracture pull-out was observed for MWCNTs in polycarbonate. 46
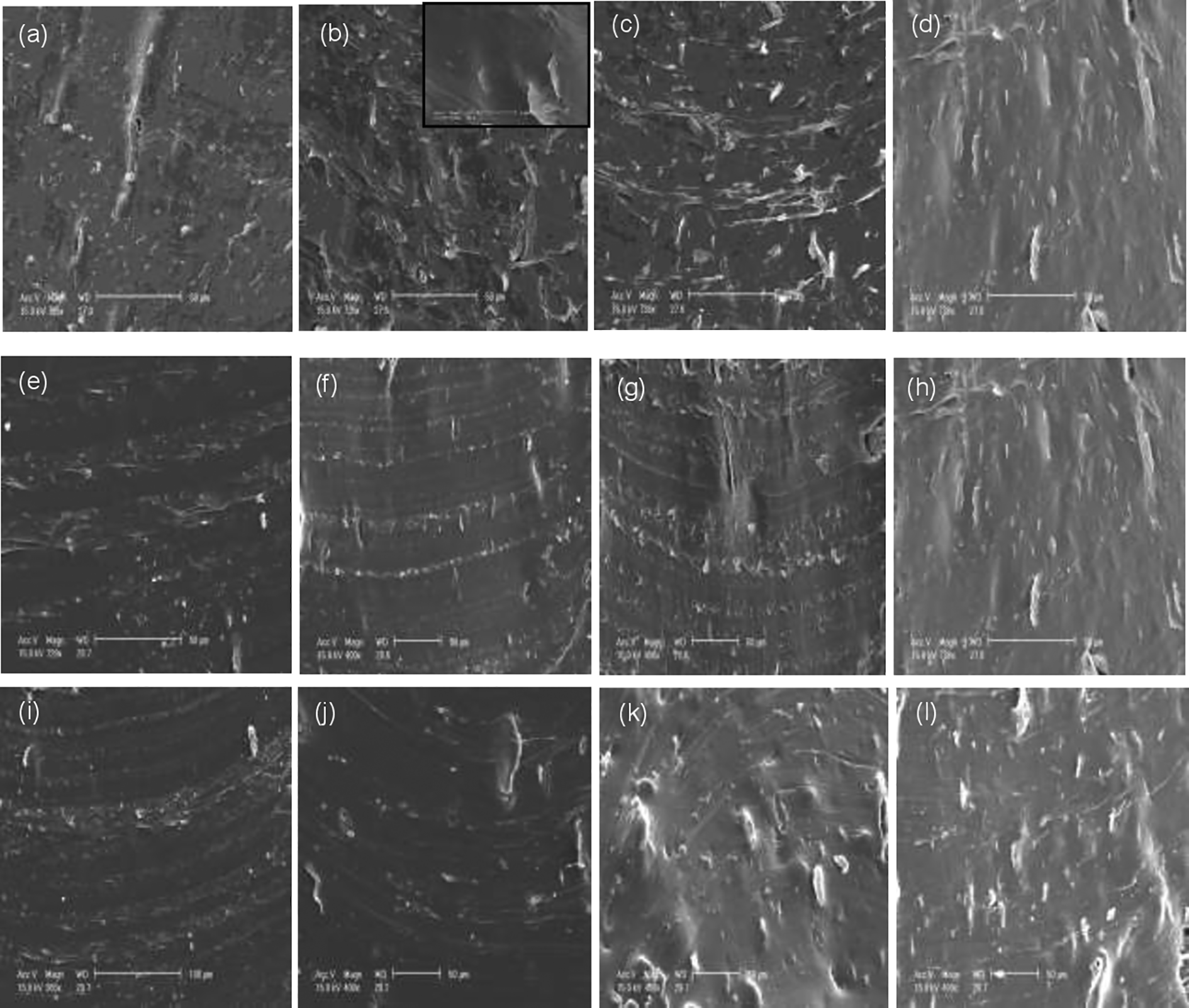
SEM micrographs of SEBS-1/xGnPs (a) 2%, (b) 5%, (c) 7%, (d)10%; SEBS-2/xGnPs (e) 2%, (f) 5%, (g) 7%, (h) 10%; SEBS-3/xGnPs (i) 2%, (j) 5%, (k) 7%, and (l) 10% (magnification 50 μm).
Another technique used to determine the morphological aspects of SEBS and the dispersion of the filler in submicron level was done by AFM. The samples with selected loading of xGnPs were taken from the compressed-sheet form. Figure 4(a) to (f) shows the amplitude images of neat SEBS-1, SEBS-1/5% xGnPs, neat SEBS-2, SEBS2/5% xGnPs, neat SEBS-3, and SEBS-3/5% xGnPs, respectively. The triblock copolymers SEBS consists of hard (PS) and soft blocks (PEB) which it exhibits phase separation. The amplitude (Figure 4(a), (c), and (e)) of different types of SEBS shows that all the samples have phase-separated morphologies consisting of bright (PS blocks) and dark (PEB blocks) nano-domains. It showed that the hard phase of PS end blocks is dispersed as cylindrical nano-domains in SEBS-2 as shown in the enlarged portion at the left insert. The worm-like cylindrical structure is prominent in SEBS-1 and SEBS-3 as can be seen in the enlarged portion. Similar morphology of cylindrical nano-domain was reported for melt-mixing of SEBS (KratonG1652). 44 Likewise, worm-like cylindrical structure morphology of thin film of SEBS with 33% PS blocks was reported. 45 The neat SEBS showed self-assemble into a lamellar morphology with polystyrene and short range order for SEBS-1 and SEBS-2 (Figure 4(a) and (c)) and prominently long range order for SEBS-3 (Figure 4(e)). The nanocomposites of SEBS-1 having 5% xGnPs showed distinct characteristic of worm-like nano-domain and particles of xGnPs (Figure 4(b)). The formation of cylindrical nano-domain of SEBS-2 with 5% xGnPs was difficult to observe because of the presence of xGnPs (Figure 4(d)). However, the AFM image of SEBS-3 with 5% xGnPs showed the worm-like cylindrical behavior and presence of xGnPs (Figure 4(f)). At 5 µm taking at given amplitude revealed that these particles are dispersed although there are some part that flocculated into large-scale aggregates (Figure 4(b), (d), and (f)). This is because the xGnPs have a tendency to roll together and form some agglomeration during mixing due to the intrinsic Van Deer Waals attraction between the individual nanoplatelets. 22 Interestingly, the characteristics morphologies of the nanocomposites loaded with 5% xGnPs consist not only of the nanoplatelets of graphite but also of long worm-like structure. These worm-like structures were clearly seen in SEBS with higher polystyrene content. Such dissimilar may be caused by the difference in the free surface energies between the polystyrene and poly(ethylene butylenes). This result is similar to that of thin films reported by Motomatsu et al. 46 The same effect of thick films and gel forms for block copolymers has been also observed by TEM.42,47,48 As can be seen from the figures, when increasing the styrene content in SEBS matrix, the xGnPs particles were dispersed homogeneously and less agglomeration. This phenomenon was also observed in the SEM images. The nanoplatelets are covered with the matrix. To further understand the images, the topographic images for samples of neat SEBS (Figure 5(a), (c), and (e)) and 5% xGnPs (Figure 5(b), (d), and (f)) were also taken. The behavior of cylindrical and worm-like structure was confirmed in the topographic images. The AFM images are uniform, thus indicating that the surface of the analyzed sample is smooth and exhibited the dispersion of xGnPs in SEBS matrix. Comparing the other two composites, SEBS-1 exhibited less agglomeration of xGnPs than SEBS-2 and SEBS-3. The ratio of soft and hard segment of the matrix has effect on the dispersion of xGnPs (Figure 5(b), (d), and (f)). This is because of miscibility caused by the intrinsic properties of polymers. Additionally, the tangled structure of the polymer was easily ripped into pieces and can well penetrate into the pores of xGnPs when the temperature reaches its melting point due to lowering of the viscosity. This is possible because the xGnPs particles were destructed caused by ultrasonic exfoliation and shearing during processing.

AFM amplitude images of (a) neat SEBS-1, (b) SEBS-1/5% xGnPs, (c) neat SEBS-2, (d) SEBS-2/5% xGnPs, (e) neat SEBS-3, and (f) SEBS-3/5% xGnPs at 5 μm.

AFM topographic images of (a) neat SEBS-1, (b) SEBS-1/5% xGnPs, (c) neat SEBS-2, (d) SEBS2/5% xGnPs, (e) neat SEBS-3, and (f) SEBS-3/5% xGnPs at 5 μm.
X-ray diffraction
Figure 6(a) to (c) shows the XRD patterns of the representative sample of SEBS-1/xGnPs, SEBS-2/xGnPs and SEBS-3/xGnPs. The XRD of neat SEBS is also shown for comparison. The broad feature at 18° is apparent to SEBS matrix. They are also observed in all SEBS/xGnPs nanocomposites that can be attributed to the polymeric phase. The maximum intensity peak at 2θ of 26.56° is attributed to graphene 25 with d-spacing of 3.37 Å, which is the characteristics of graphite.25,45,49 The occurrence of peaks at 18° and 26.56° in all the samples confirms not only the presence of graphite but also the individual xGnPs. This indicates that there is no difference at 002 peaks of graphite lattice. This behavior is consistent with the SEM (Figure 3(a) to (l)) and AFM (Figures (4) and (5)). The crystal structure of graphite does not change and no expansion happens to the gallery spaces of graphite carbon layer. It was found that the melt compounding technique used in this study did not affect the structure of the xGnPs in the SEBS matrix. This behavior was also reported in the melt-blending of SEBS/xGnPs. 25 The ratio of soft and hard segment of the matrix has less effect on the dispersion of xGnPs (Figure 6(a) to (c)). In the work of Chen et al., 18 there were no significant difference between the d-spacing and 2θ of 002 peaks of the original graphite worm (3.37 Å, 26.4), sonicated (3.3795 Å, 26.358) and the PS/Graphite nanosheet composite (3.3799 Å, 26.3588). This was confirmed in the XRD of EG and xGnPs as shown in Figure 2. From Figure 6, it was observed that the intensity peak of xGnPs increases with increasing amount of xGnPs content in the SEBS matrix, which is attributed to the higher number of graphite layers in the nanocomposites. Similar results have been reported. 25
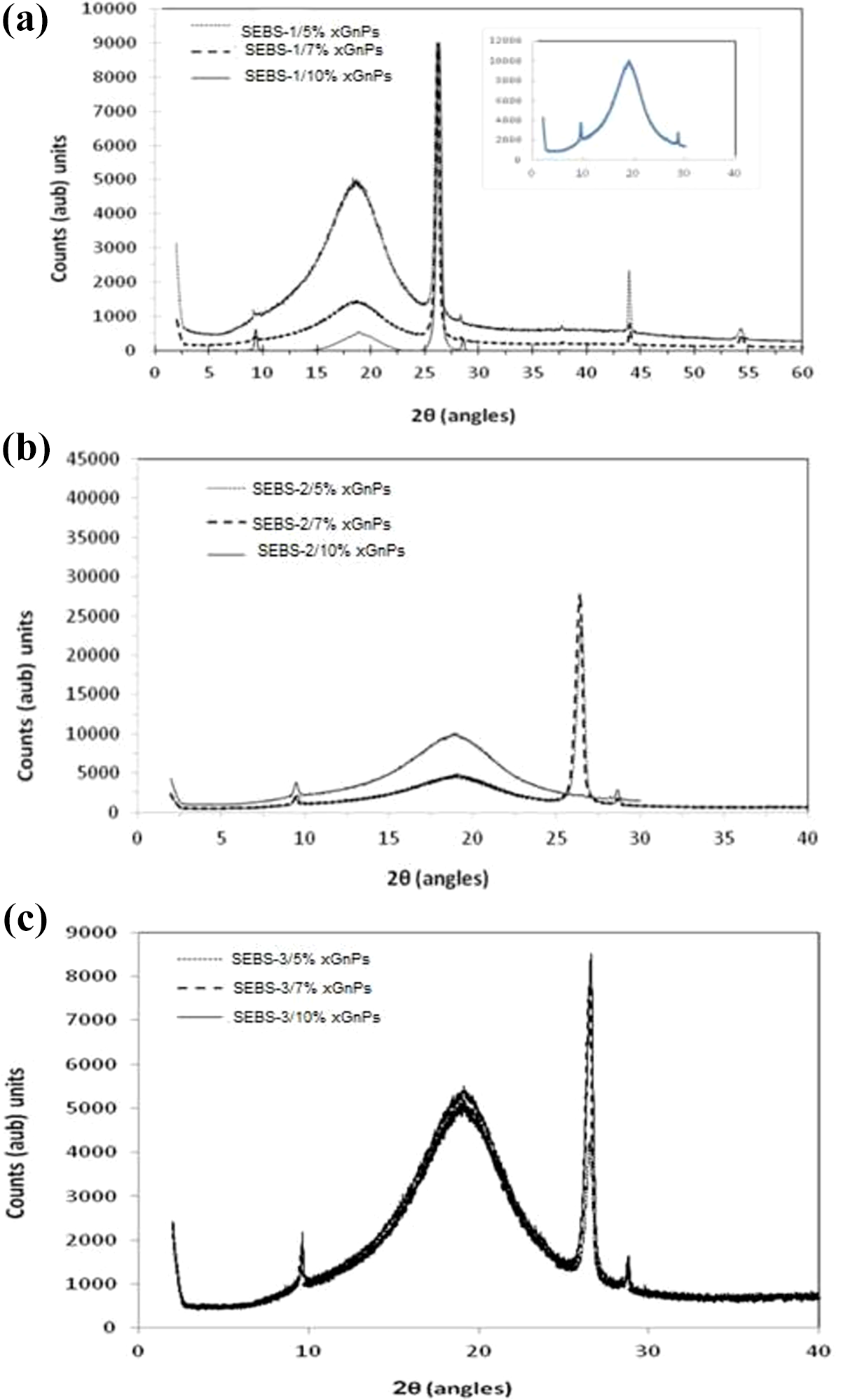
XRD patterns of (a) SEBS-1/xGnPs, (b) SEBS-2/xGnPs, and (c) SEBS3/xGnPs.
Thermal properties
The DSC behavior of neat SEBS and SEBS/xGnPs is shown in Figure 7(a) to (c.) The glass transition (Tg) values for different types of SEBS with selected xGnPs loading are compiled in Table 2. The neat and SEBS/xGnPs have very similar DSC traces. The observed Tg around −55°C pertains to soft (PEB) block of the triblock copolymers. The Tg of the hard block (PS) cannot be detected in the samples. The Tg of the nanocomposites was almost the same despite the addition of xGnPs. The TGA curves for representative composites are shown Figure 8(a) to (c) for different SEBS with different amounts of xGnPs loading. For all the SEBS/xGnPs nanocomposites, there were no significant difference in onset temperatures (396°C to 402°C) and maximum temperatures (455°C to 456°C) of degradation (derivative peaks are not shown here). This was confirmed by the loss in weight. The value was only varied due to the amount of xGnPs loaded in the nanocomposites. It appeared that the graphite platelets have no effect on thermal stability of the nanocomposites. Similar results were reported by Uhl et al. 19

DSC curves of (a) SEBS-1/xGnPs, (b) SEBS-2/xGnPs, and (c) SEBS-3/xGnPs.
Glass transition of SEBS/xGnPs nanocomposites.


TGA curves of (a) SEBS-1/xGnPs, (b) SEBS-2/xGnPs, and (c) SEBS-3/xGnPs.
Mechanical properties
Figure 9(a) shows typical tensile strength data for the nanocomposites. From XRD measurements, the blends do not show any expansion of the d-spacing of graphite so nanocomposites were not formed in the melt compounding process. Perhaps the microdispersion in the blends offers the same barrier properties as observed in the nanocomposites with graphite. Despite the results in XRD, the tensile strength greatly increased with increasing amount of xGnPs for all types of SEBS polymers. The modest increase in tensile strength was dependent on the type of SEBS. For SEBS-3 with low amount of styrene, lower tensile strength was obtained than the two SEBS based polymer composites. Initially, the tensile strength of SEBS-3/xGnPs composite increased with increasing content up to 7% followed by a reduction at 10% xGnPs loading. The higher amount of xGnPs particularly when the styrene content of SEBS is lower resulted to lower tensile strength, not because of poor adhesion between the polymer and particles but rather the xGnPs behave like a rigid particles and reinforced the filler into the polymer matrix. Consequently, the reduction of the tensile strength of the nanocomposites could not be avoided. This behavior was not observed in SEBS-1 and SEBS-2 having higher styrene content in the triblock copolymer. Figure 9(b) shows the elongation at break for all SEBS/xGnP nanocomposites. There is no significant improvement on the elongation at break but rather a slight decrease for all amounts of xGnPs was observed. However, in the case of SEBS-3/xGnP nanocomposites, the elongation at break increased at 7 wt% xGnP which is reasonable since the tensile strength has decreased.

(a) Tensile strength and (b) elongation at break of SEBS/xGnPs (error bar using standard error).
Conclusion
A process of dispersion of exfoliated graphite nanoplatelets was developed via simple melt compounding with styrene-ethylene-butylene-styrene block copolymer (SEBS) containing different ratio of styrene/rubber. Shearing process and ratio of styrene and rubber ratio of SEBS matrix strongly affected the dispersion of the nanocomposite as revealed by SEM and AFM images. The SEBS with higher amount of styrene resulted to better dispersion and strong adhesion of the xGnPs with SEBS matrix. It was found that the nanocomposites did not change the crystal structure and no expansion happens to the gallery spaces of xGnPs in the SEBS matrix. The Tg of SEBS/xGnPs nanocomposites was almost the same despite the addition of xGnPs and no significant difference in degradation temperature. The tensile strength of the nanocomposites has greatly improved with increasing amount of xGnPs. The increase of tensile strength depends on the amount of hard block (styrene) content on the SEBS matrix. The mechanical percolation threshold for SEBS containing less amount of styrene has been observed for a value (7%).
Footnotes
Acknowledgment
The authors would like to acknowledge the First in Colors, Inc., Quezon City, Philippines for allowing them to use their DSC and TGA and Gyeongsang National University, South Korea for AFM and SEM testing of the samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
