Abstract
A monodisperse poly (N-phenylmaleimide–acrylonitrile–styrene) (PNAS) nanolatex was synthesized via seed microemulsion polymerization. The obtained PNAS nanolatex was then directly used as an organic nanofiller to prepare polyvinyl chloride (PVC)/PNAS hybrid composite through water blending and melt compounding. The characteristics of PNAS nanolatex were analyzed by Fourier transform infrared (FTIR) spectrometer, elemental analysis, scanning electron microscope, transmission electron microscope (TEM), dynamic laser lighting scattering (DLS), differential scanning calorimetry (DSC), and thermogravimetric analyzer (TGA). FTIR and elemental analysis confirmed the formation of PNAS copolymer with high monomers conversion; meanwhile, for the PNAS nanoparticles, the morphology of a well-defined core–shell spherical structure with average diameter ranging from 156 nm to 249 nm was observed. DSC analysis and TGA indicated that both polymers had excellent compatibility, and the corresponding heat resistance of PVC was greatly improved with the addition of PNAS. When PNAS loading was 50 wt%, the glass transition temperature value of PVC/PNAS hybrid composite was increased by 22.4°C, compared with that of pristine PVC. The mechanical properties of the PVC composite were also enhanced with the addition of PNAS.
Introduction
Preparation of structured latex particles with predetermined morphologies and a variety of functions has attracted interests of numerous scientists for many years. 1 –5 Due to their structural and chemical tailorability, these polymer materials are widely used in coating, adhesives, paints, and impact modifiers. 6 –9 Core–shell structural modifiers as typical latex particles are usually prepared by a two-stage emulsion polymerization process. 10 –12 Most core–shell structural copolymers were used as toughening modifiers, and few studies have been reported on the synthesis of core–shell structural latex used for heat-resistant modification of polymeric matrix.
However, it is not easy to achieve a well-defined core–shell structure via seed microemulsion polymerization. 13 Varying some polymerization parameters, such as initiator and emulsifier types, core/shell monomers ratio, monomer composition, cross-linking agent dosage, and monomers feeding mode at different polymerization stages, produces various morphologies of the resulting composite particles, which are dictated by thermodynamic/kinetic factors. 14 –19
The polymerization behavior of N-phenylmaleimide (NPMI) has stimulated considerable interests because of its special structure. 20 The five-membered planar ring and a strong polar carbonyl in the polymer chains can hinder the rotation of the backbone chain and result in greater structural stiffness and superior thermal stability, making it a promising option for heat-resistant solitary materials and modifiers. 21,22 So far, extensive studies have been carried out on NPMI copolymerization with monomers such as methyl methacrylate, 23 –26 styrene, 27,28 and vinyl chloride 29 used as heat-resistant modifiers, which present good compatibility with acrylonitrile-butadiene-styrene (ABS) 20 or PVC 22 resin.
In this study, a monodisperse core–shell structural poly (N-phenylmaleimide–acrylonitrile–styrene) (PNAS) nanolatex was prepared via seed emulsion polymerization. Fourier transform infrared (FTIR) spectrometer, elemental analysis, scanning electron microscope (SEM), transmission electron microscope (TEM), dynamic laser lighting scattering (DLS), and differential scanning calorimetry (DSC) were performed to characterize and investigate its chemical composition, morphology, and thermal property. As a heat-resistant modifier, the obtained latex was then directly used as an organic nanofiller to complex with PVC resin to prepare the corresponding PVC/PNAS hybrid composite. The effects of PNAS as a nanofiller on the heat-resistant and mechanical properties of PVC resin were also researched.
Experiment
Materials
Acrylonitrile (AN) and styrene (St), from Tianjin Fuchen Chemical Reagent Co., Ltd (Tianjin, China), were distilled under vacuum to remove their inhibitors. N-phenylmaleimide (NPMI) was provided by Taiyuan RHF Chemical Co., Ltd (Taiyuan, China), recrystallized from an ethanol/water mixture three times and then stored in the absence of light before use. The initiator ammonium persulfate (APS) together with the emulsifier including sodium dodecyl sulfonate (SDS), and OP-10, supplied by Tianjin Kermel Chemical Reagent Co., Ltd (Tianjin, China) were used as received. Acetone and methanol were supplied by Tianjin Fuyu Fine Chemical Co., Ltd (Tianjin, China). Distilled deionized water (DDI) was used in all experiments. Commercial-grade PVC resin with an average polymerization degree of 1000 was provided by Xi’an Petrochemical Corp. (Xi'an, China). Magnesium stearate (MgSt2) and calcium stearate (CaSt2) and dioctyl phthalate (DOP) were used as heat stabilizers and the plasticizer of PVC resin, respectively.
Synthesis of PNAS structural latexes
SDS and OP-10 (1:2, mass ratio) dissolved in DDI were added into a 250-mL three-necked flask equipped with an inlet for nitrogen purging and a mechanical stirrer. The solution was continuously stirred in an ultrasonic bath at room temperature for 25 min, followed by the addition of the monomers and the initiator. Then the mixture was stirred in an ultrasonic bath for another 30 min. One-fifth of the above mixture and a quarter of the initiator were seed polymerized in a 150-mL four-necked flask in a water bath at 75°C for 15 min under nitrogen atmosphere. Then, the remaining four-fifth of the above mixture was added gradually to the flask, with a dropping rate of 25 mL/h at a stirring rate of 180 r/min. After that, the residual initiator was added to the emulsion, and the reaction was carried out for another 2 h at 80°C.
For purification, the prepared emulsion was precipitated by freezing at –20°C. The precipitated copolymer was filtered, washed with methanol for 30 min to remove the residual monomers and the emulsifier, and then dried in a vacuum oven at 80°C for 48 h. The copolymerization recipes are given in Table 1.
Recipes for the synthesis of PNAS nanolatexes.
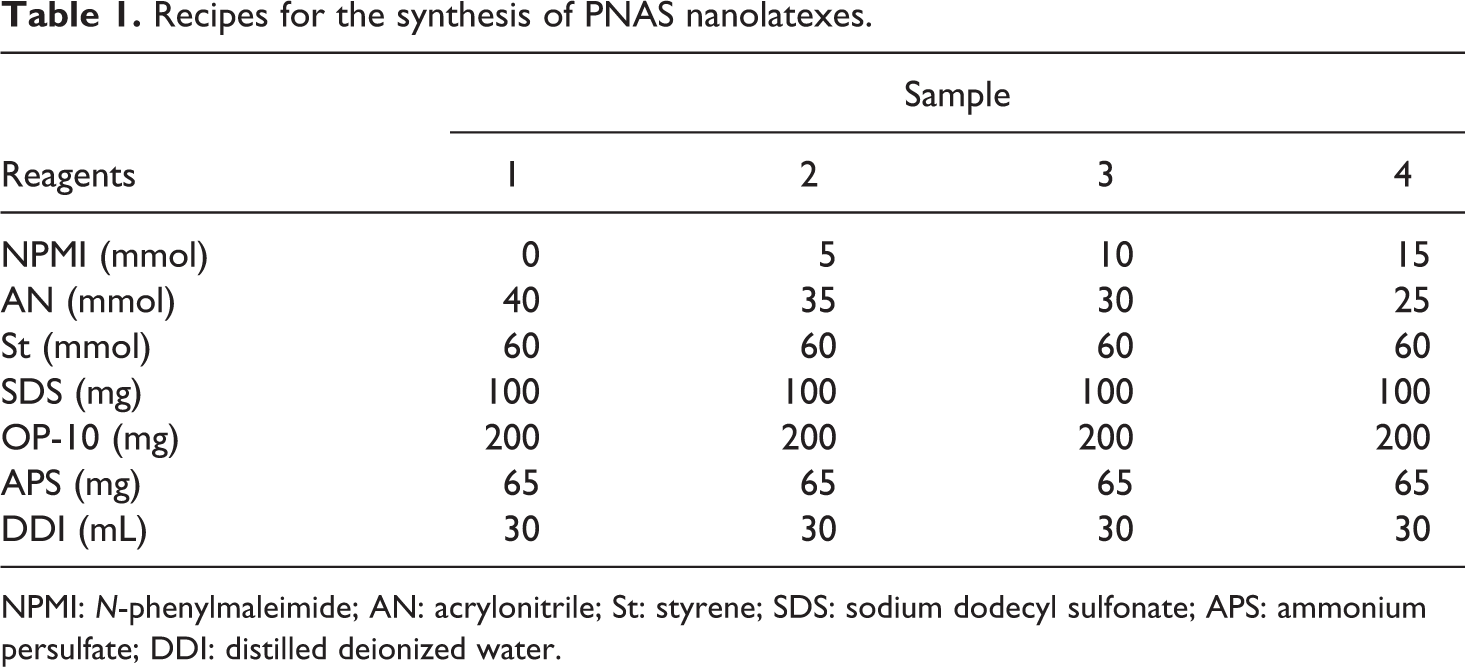
NPMI: N-phenylmaleimide; AN: acrylonitrile; St: styrene; SDS: sodium dodecyl sulfonate; APS: ammonium persulfate; DDI: distilled deionized water.
Preparation of PVC/PNAS hybrid composite
A certain amount of PNAS (Sample No. 3 in Table 1) latex was diluted with 5- to 10-fold volume of water, followed by the addition of a certain amount of PVC resin and various additives, as described in Table 2. The mixture was blended uniformly by an FA-25 homogeneous mixer, and then transferred into a vacuum drying oven at 80°C for 48 h. The obtained powder was mixed and molded by heating to prepare the requested test samples at 180°C with the molding pressure of 15 MPa.
Recipes for the preparation of PVC/PNAS hybrid composite.

PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene); MgSt2: magnesium stearate; CaSt2: calcium stearate; DOP: dioctyl phthalate.
Measurements
FTIR spectra of the samples were obtained on a Nicolet iS10 spectrometer (Thermo Fisher Scientific, USA) with a resolution of 4 cm–1 from 4000 cm–1 to 400 cm–1. The ratio of each monomer segment in PNAS molecular chains was calculated on the basis of the element content of the copolymer, determined on a PE2400 CHN elemental analyzer (PerkinElmer, USA). Particle size and distribution of PNAS nanolatexes were measured via a Zetasizer Nano-ZS DLS instrument (Malvern Panalytical, USA). External morphologies of PNAS nanoparticles were observed by Quanta 600 FEG field-emission SEM (Thermal Fisher Scientific, USA) at an accelerating voltage of 10 kV, while the internal microstructure of the PNAS nanospheres was observed using a Hitachi H-600 TEM (Hitachi, Japan) at an accelerating voltage of 75 kV. DSC measurements of the samples were determined with a Mettler Toledo DSC 1 system differential scanning calorimeter (Mettler Toledo, USA). The samples were scanned at a heating rate of 10°C min–1 with a scanning range from 25°C to 200°C under nitrogen atmosphere. TGA thermograms of the PVC/PNAS hybrid composites were obtained on a Q600 SDT TGA (TA, USA). The samples were heated at a rate of 10°C min–1 from room temperature to 600°C under nitrogen atmosphere. Tensile tests were performed using a CMT8000 universal testing machine (Sans, Shenzhen, China) according to ISO 527-2:1993, while impact tests were performed on a ZBC2503 impact testing machine (Sans, Shenzhen, China) according to ISO 180:2000.
Results and discussion
Chemical composition of PNAS copolymer
FTIR spectra of PNAS copolymer with different NPMI monomer proportions are shown in Figure 1. The characteristic absorption peak at 1383 cm–1 of NPMI appeared was ascribed to its imide structure. The appearance of bands at 1712 cm–1 and 1186 cm–1 was attributed to stretching vibration of carbonyl and C–N bond, respectively. The peaks at 1599, 1455, and 1495 cm–1 were associated with the phenyl groups in the NPMI and St units, reflecting the semicircle stretching and mixed C–H bending of a monosubstituted benzene ring. The existence of St was confirmed by the stretching vibrations of CH2 at 2929 cm–1. Another typical absorption at 2236 cm–1, belonged to the C≡N stretching vibration of AN fragments. It revealed that the AN monomers were successfully polymerized.
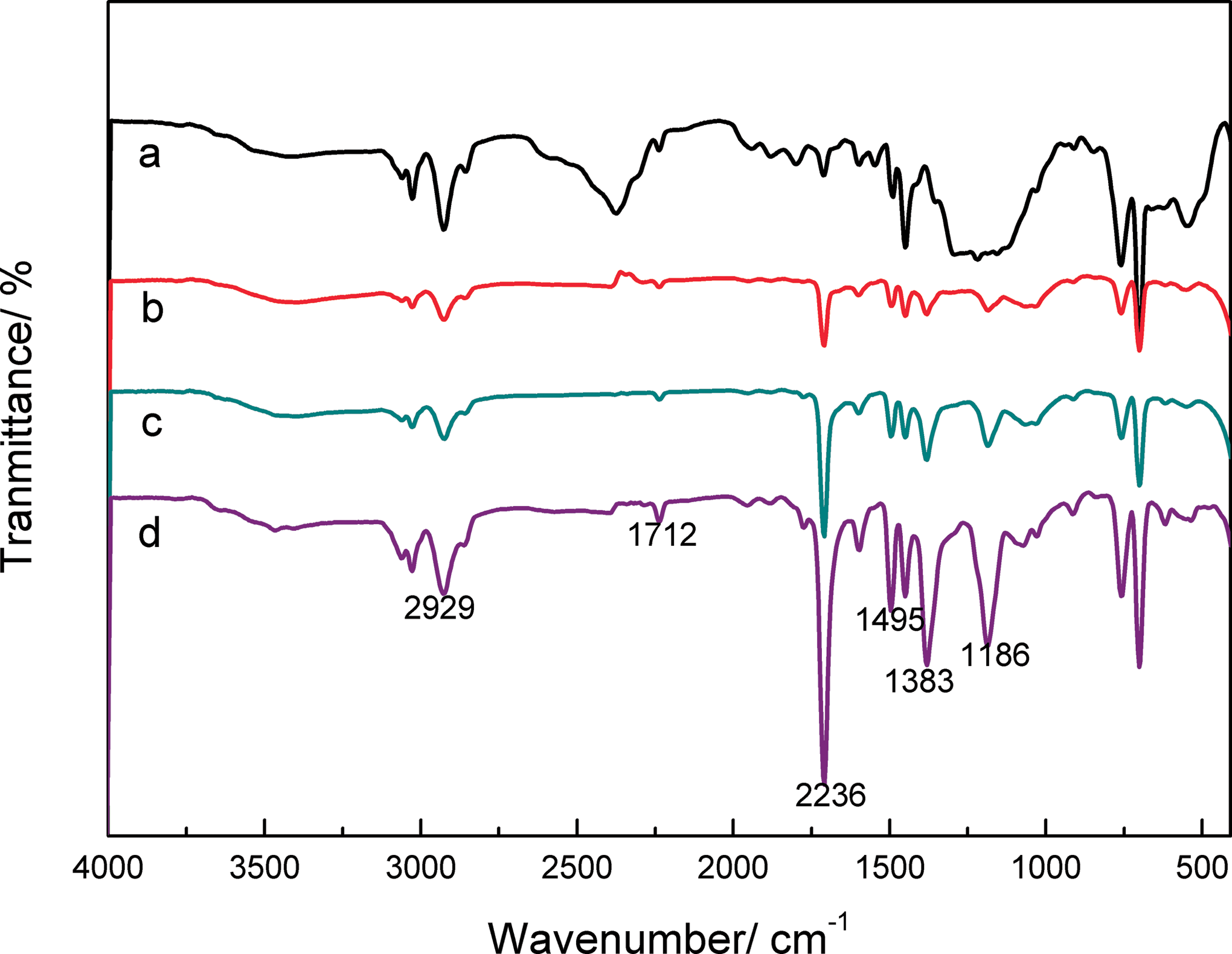
FTIR spectra of PNAS copolymer as a function of NPMI contents: (a) 0%, (b) 5%, (c) 10%, and (d) 15%. FTIR: Fourier-transform infrared; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene); NPMI: N-phenylmaleimide.
Elemental analysis results are described in Table 3. It can be found that the composition of the copolymers was almost the same as that of the feed monomers. Results showed that the monomers were almost consumed completely, and the formed coagula were always negligible. Thus, the composition of the copolymers can be estimated through that of the feed monomers.
Elemental analysis of PNAS copolymer with different compositions.
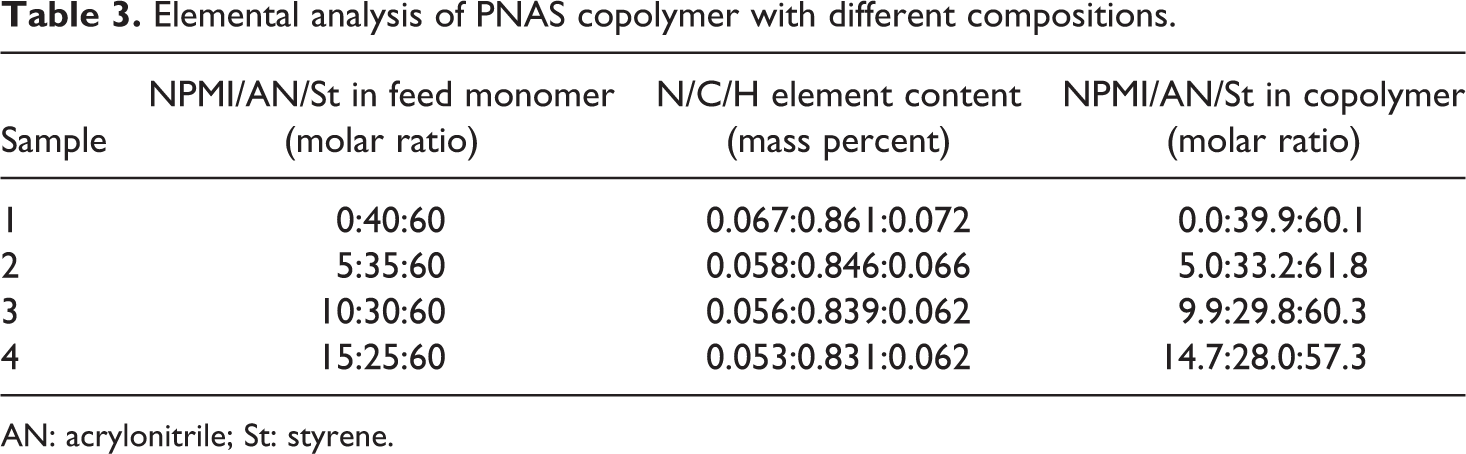
AN: acrylonitrile; St: styrene.
Morphologies of PNAS nanolatexes and PVC/PNAS composites
The particle size and distribution of PNAS nanolatexes with different emulsifier dosages (2.5, 5.0, and 7.5 g L–1) are shown in Figure 2. The corresponding average diameter of PNAS nanolatexes was 249.3, 177.2, and 155.6 nm, respectively. In addition, the particle size distribution index for PNAS nanolatexes was 0.015, 0.006, and 0.002, respectively. It was clearly seen that, with the increasing emulsifier dosage, the particle size became more uniform. The narrow size distribution indicated that the monomers were polymerized evenly on the seed.
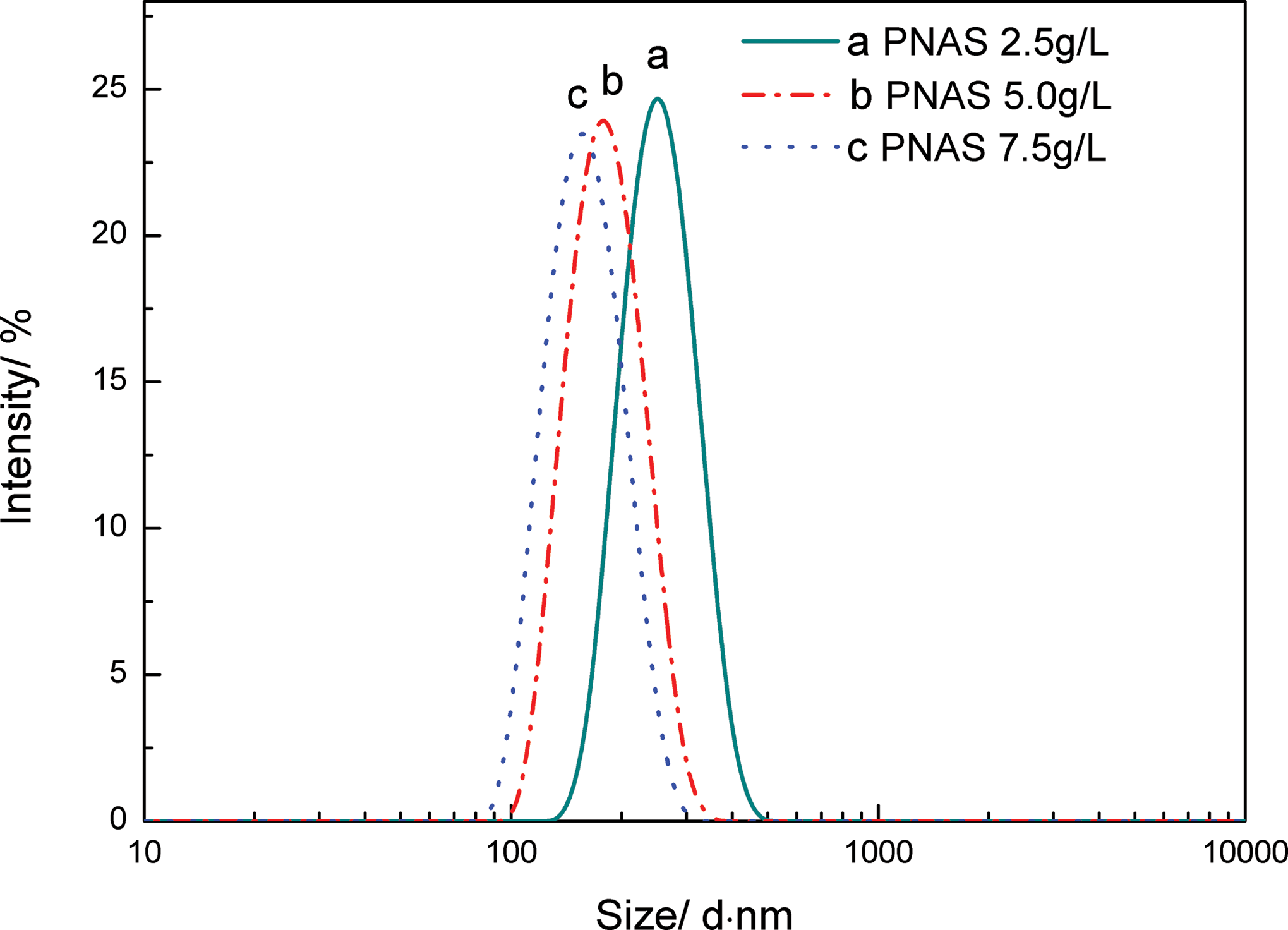
Particle size and distribution of PNAS nanolatexes with different emulsifier dosages: PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene). (a) 2.5 g L–1, (b) 5.0 g L–1, and (c) 7.5 g L–1.
SEM morphologies of PNAS nanolatexes with different emulsifier dosages are shown in Figure 3. It was seen that the obtained PNAS nanolatexes still presented a regular spherical shape as shown in Figure 3(b). There was no adhesion to each other for the nanoparticles, and the particle size was quite uniform. Furthermore, comparing with that of Figure 3(c), it was easy to find that the particle size decreased with the increasing emulsifier dosages. To our knowledge, one of the most direct approaches to change the latex particles morphology is to use different initiator and surfactant (emulsifier), 20 which can be ascribed to the thermodynamic/kinetic control limitations having the minimum possible Gibbs free energy of the whole system. On a thermodynamic basis, only small differences in Gibbs free energy arising from changing the interfacial tensions are required to vary composite latex particles from core–shell to partially engulfed structures. 14 In our work, starved feed mode of monomers, the stable system consisted of ionic initiator, nonionic surfactant OP-10 together with ionic surfactant SDS allowed the monomers grew uniformly on the seed, resulting in a regularly spherical morphology.

SEM images of PNAS seed and PNAS nanolatexes as a function of emulsifier dosages: (a) PNAS seed, (b) 2.5 g L–1, and (c) 5.0 g L–1. PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene); SEM: scanning electron microscope.
Figure 4 shows the TEM images of PNAS seed and PNAS nanolatexes. As shown in Figure 4(b) and (c), the inner area of the black loop corresponded to the core of the PNAS seed, while the regional black loop outside represented the shell arising from the remaining monomers growing continuously on the core of the PNAS seed. Furthermore, PNAS nanolatexes displayed a typical core–shell structure due to the seed emulsion polymerization method. Obviously, a fairly typical core–shell structure was observed for the PNAS nanolatexes mainly due to the starved monomer feed mode. In shell starve-feed polymerization, a gradual monomer addition prevented further plasticizing effect of the remaining monomers in the first-stage copolymer. On the other hand, the formation and growth of monomers around the core raised a kinetic barrier to prevent further monomers diffusion into the initial seeds, 30 which deviated from other phase-separated granular structure in the batch progress of seed emulsion polymerization. 31

TEM images of PNAS seed and PNAS nanolatexes as a function of emulsifier dosages: TEM: transmission electron microscope; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene). (a) PNAS seed, (b) 2.5 g L–1, and (c) 5.0 g L–1.
SEM images of impact fracture surface for PVC/PNAS composite at different magnifications were seen in Figure 5. The impact fracture surface exhibited a brittle fracture, and there was no obvious phase separation (Figure 5b), indicating good compatibility between PNAS copolymer and PVC resin 32 .

SEM images of the impact fracture surface of PVC/PNAS composite at different magnifications : (a)2000×, (b) 5000×. SEM: scanning electron microscope; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
Thermal properties of PNAS and PVC/PNAS hybrid composite
DSC curves of PNAS copolymer with different NPMI monomer ratio are shown in Figure 6. With the increasing contents of NPMI, the glass transition temperature (T g) values of PNAS copolymer was increased due to larger molecular chain stiffness. Furthermore, single T g value for each PNAS copolymer indicated a relatively homogenous structure of the copolymer, different from some previous work in which two different T g values of the NPMI copolymer were found. 20
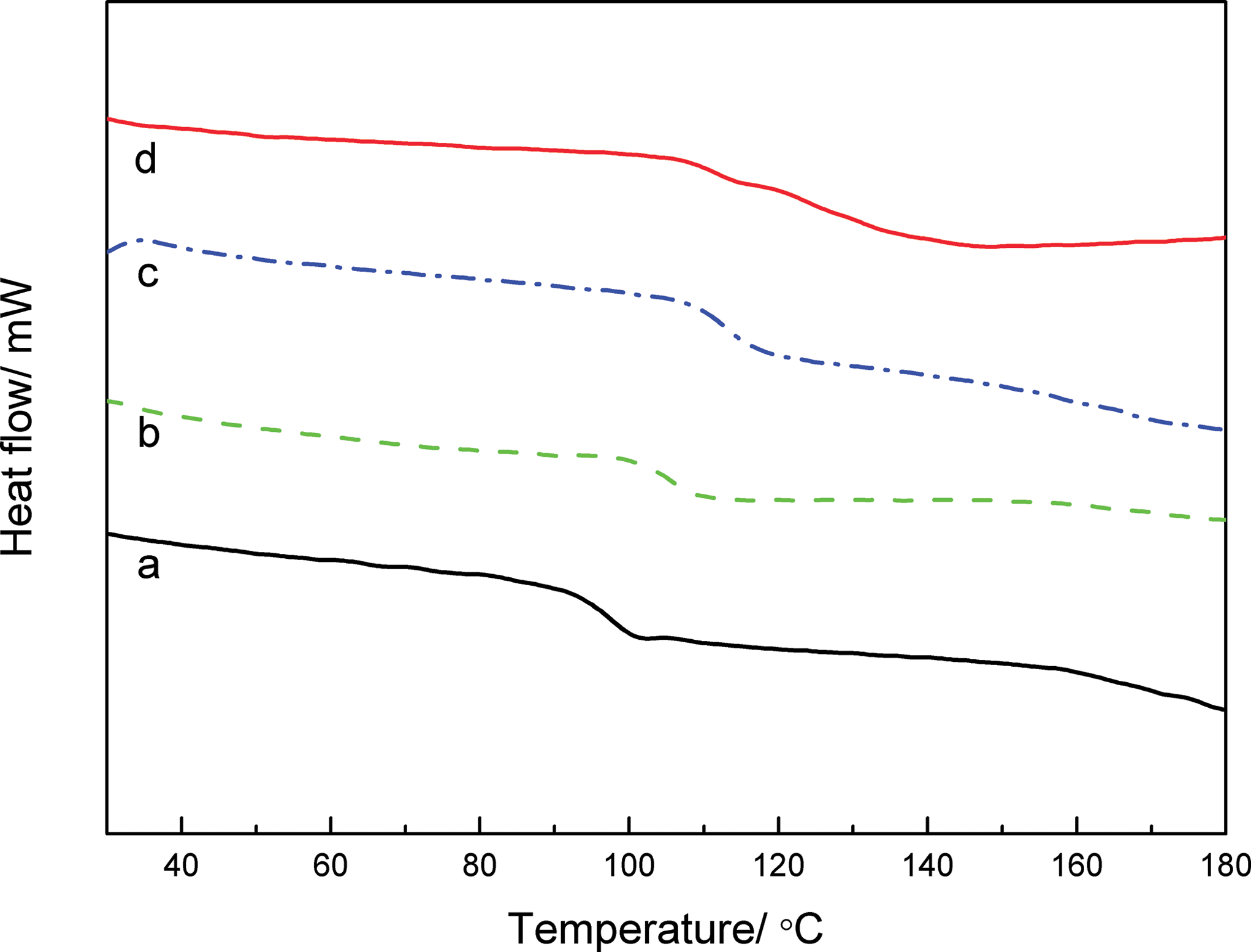
DSC curves of PNAS copolymer as a function of NPMI monomer ratio: (a) 0%, (b) 5%, (c) 10%, and (d) 15%. DSC: differential scanning calorimetry; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
In the conventional copolymerization (NPMI/AN as electron-acceptor and St as electron-donor monomer), St and NPMI/AN monomers possessed a huge trend to complex and form an alternating structure rather than a random or homogeneous one, 20 which was mainly affected by the polar effect owing to a high variable polarity between the monomers. Thus, a cross-propagation progress dominated in the first stage of copolymerization until the NPMI/AN monomers concentration became too low, and then the remaining St monomers continued to homopolymerize. The final product, even though with high conversion, was a mixture of polystyrene and poly (N-phenylmaleimide/acrylonitrile-alt-styrene). That was the reason for the appearance of double T g values. However, by means of dripping reactive monomers gradually at an appropriate rate, St monomers accounting for the majority could participate in the copolymerization reaction timely, ensuring the uniformity of the copolymer compositions. As a result, random copolymerization, which was controlled by a balanced combination of general reactivity and polar effects, was supposed to occur, and the resulting PNAS copolymer was believed to have a random molecular structure.
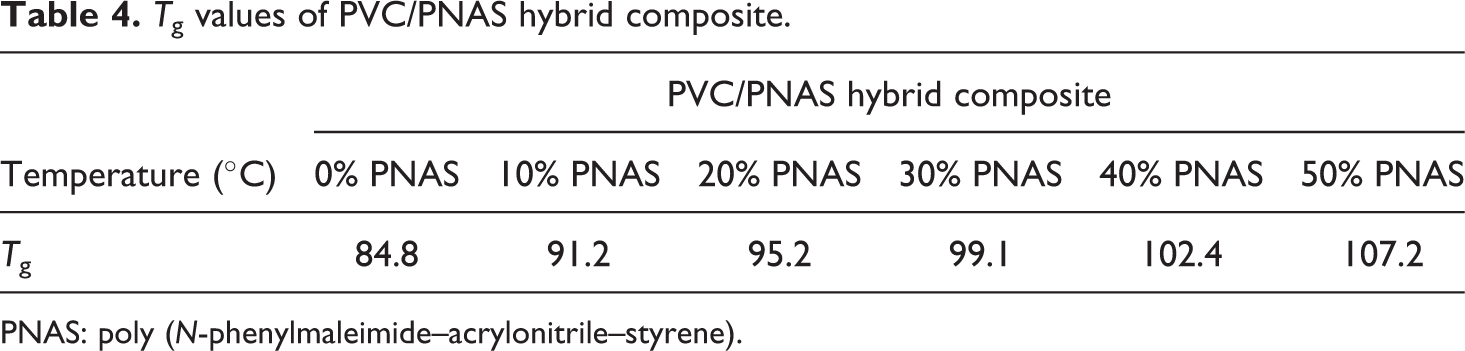
DSC curves of PVC/PNAS hybrid composite with different PNAS content are displayed in Figure 7, and the corresponding T g values are mentioned in Table 4. With the increasing contents of PNAS, the T g values of PVC/PNAS hybrid composite were increased due to higher T g value of the PNAS copolymer. Furthermore, only one T g value with narrow transition range for each composite was discovered, indicating excellent compatibility between the PVC resin and the PNAS copolymer. Furthermore, the T g values were increased from 84.8°C to 107.2°C with the increasing PNAS content. The heat resistance of the PVC resin was greatly improved, revealing that PNAS nanolatexes as heat-resistant modified fillers have been prepared successfully. The reason for this phenomenon might be attributed to the fact that the carbonyl groups in the five-membered planar ring formed hydrogen bonds with α-H in the PVC molecular chains, in favor of improving the compatibility between the PVC and the PNAS copolymer.
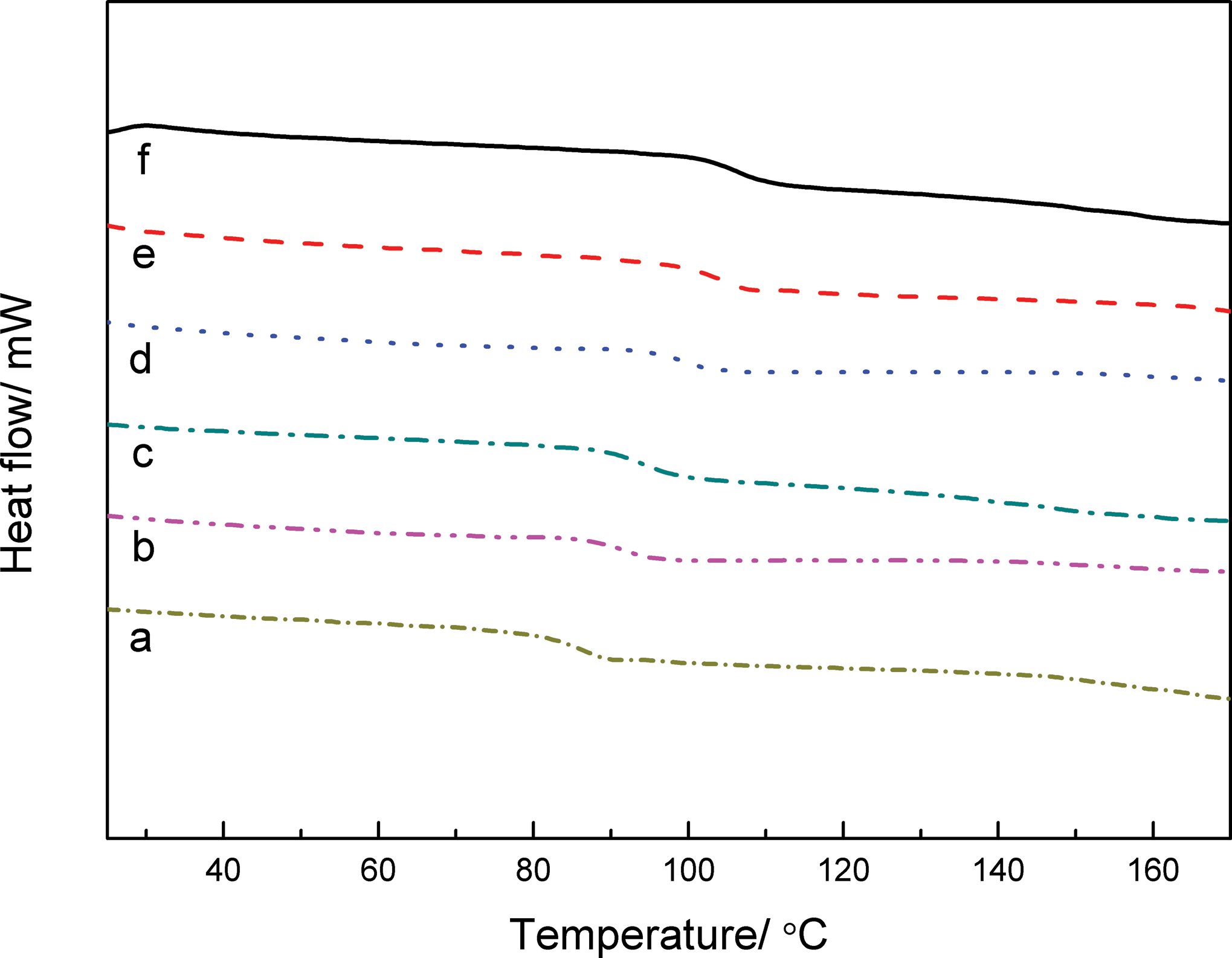
DSC curves of PVC/PNAS hybrid composite as a function of PNAS contents: (a) 0%, (b) 10%, (c) 20%, (d) 30%, (e) 40%, and (f) 50%. DSC: differential scanning calorimetry; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
T g values of PVC/PNAS hybrid composite.
PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
TGA curves of PVC/PNAS hybrid composite with different PNAS copolymer contents are shown in Figure 8, and the corresponding characteristic thermal data are also presented in Table 5. Pristine PVC presented a typical two-step degradation process ascribed to dehydrochlorination and fracture of carbon chains, respectively, while the poly(1,4-butylene adipate) (PBA)/PNAS copolymer displayed one-step decomposition from 345°C to 465°C, due to the cracking of molecular chains.
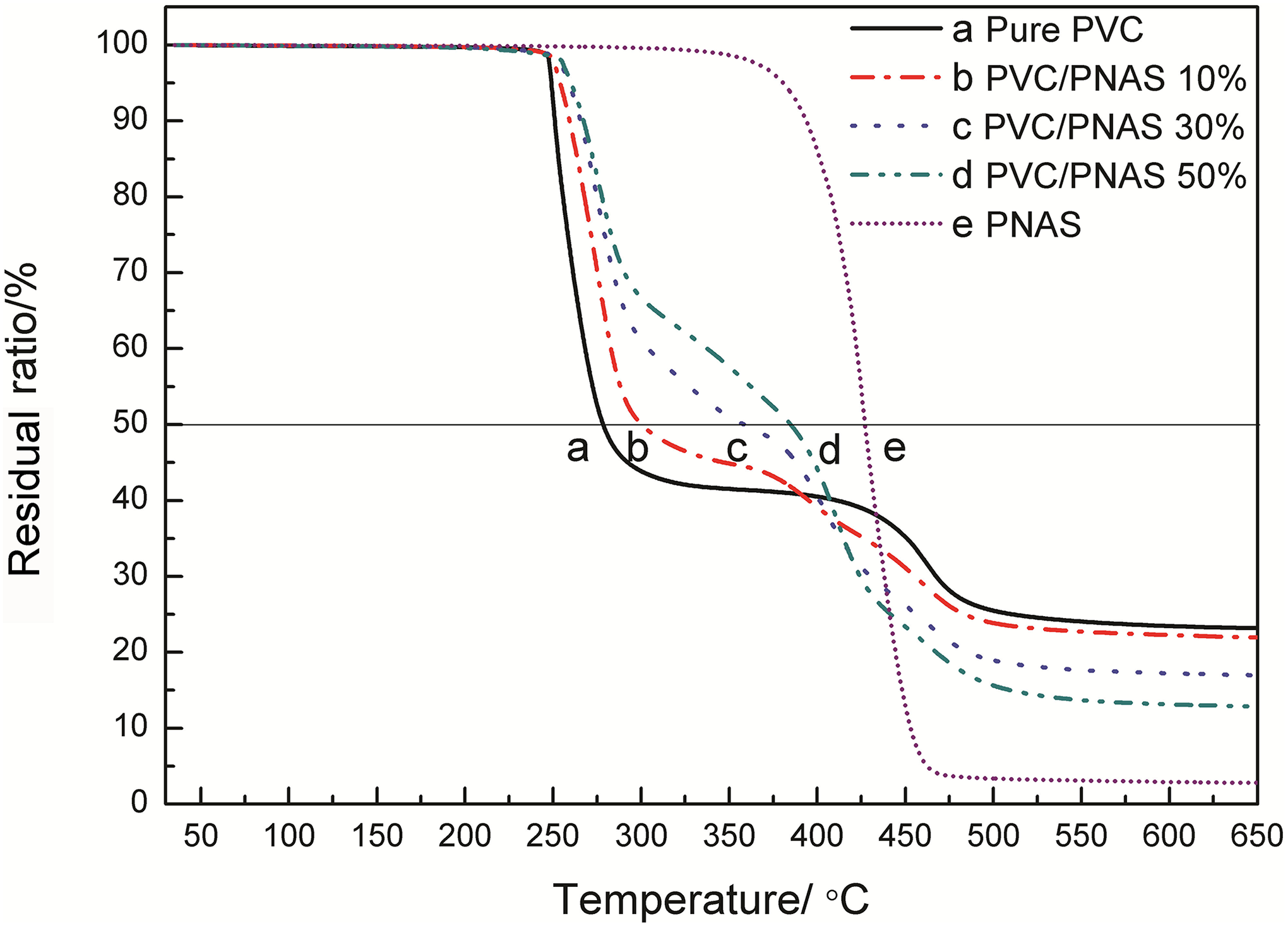
TGA curves of PVC/PNAS hybrid composite with different PNAS contents. TGA: thermogravimetric analyzer; PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
T i and T 50 of PVC/PNAS hybrid composite.
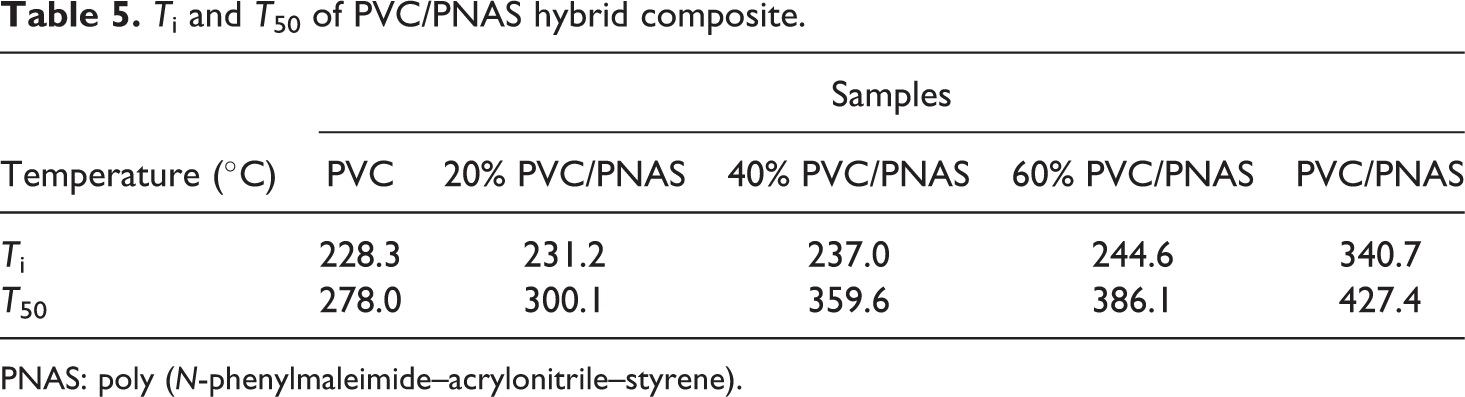
PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
The T i and T 50 of the composite became higher with the increasing content of PNAS, indicating improved thermal stability of PVC resins with the addition of the PNAS modifier. It is well-known that the weight loss of PVC resin in the first stage was mainly due to the HCl segment drawing off molecular chains, which was greatly affected by the concentration of hydrogen ion. The concentration of hydrogen ion was lowered by combining with the nitrogen atoms in the molecular chain to form a stable complex, which led to the improvement of thermal stability 33 of the PNAS/PVC hybrid composite.
Mechanical properties of PVC/PNAS hybrid composite
Effects of PNAS copolymer contents on the mechanical properties of PVC/PNAS hybrid composite are shown in Figure 9. The numbers 1, 2, 3, 4, 5, and 6 represented the PVC/PNAS composite with different mass ratios of 100:0, 90:10, 80:20, 70:30, 60:40, and 50:50, respectively.
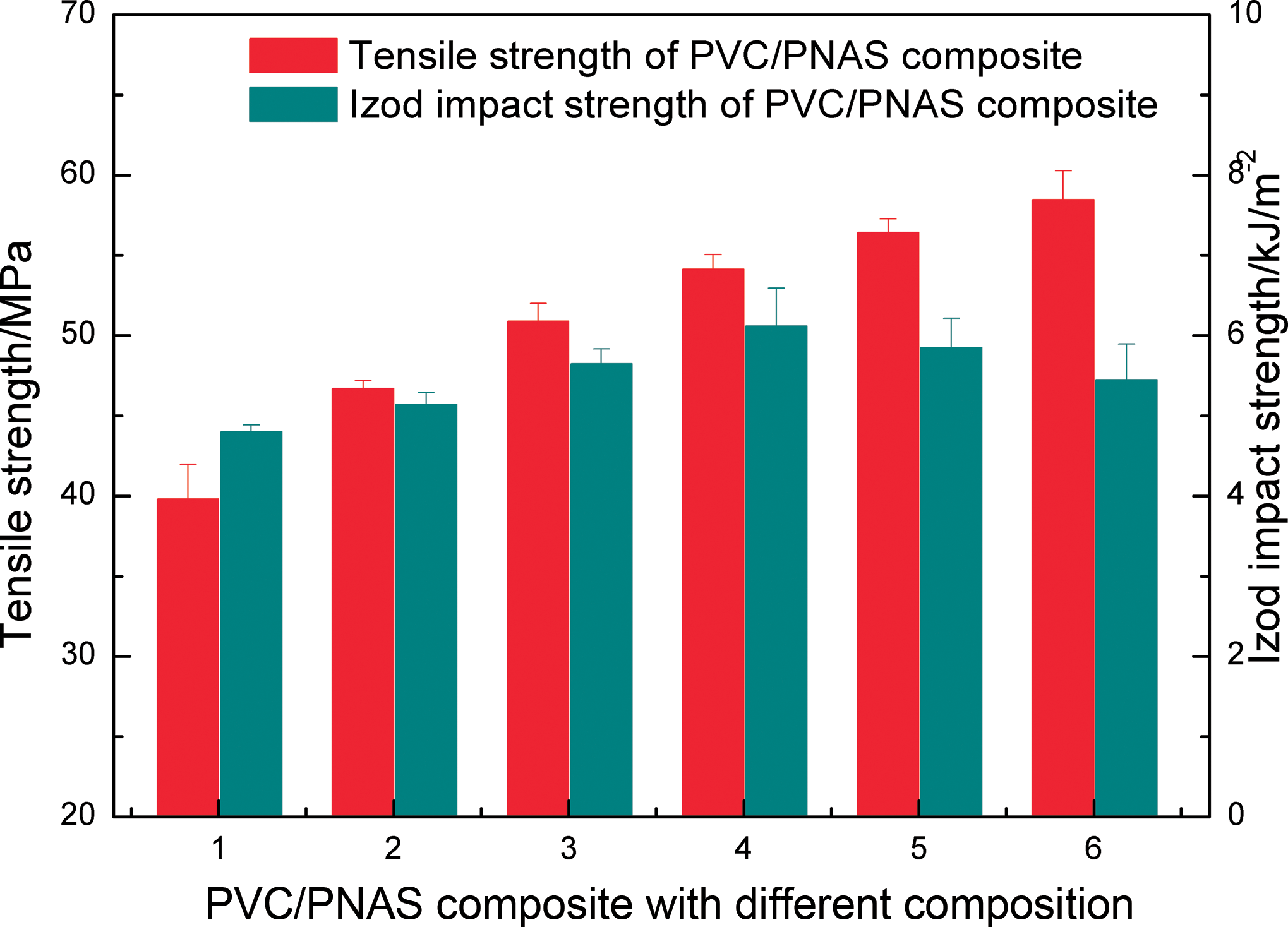
Mechanical properties of PVC/PNAS hybrid composite with different composition. PNAS: poly (N-phenylmaleimide–acrylonitrile–styrene).
It was clearly observed that the tensile strength of the PVC/PNAS hybrid composite was gradually improved with the increasing content of PNAS. The corresponding tensile strength (58.5 MPa) was optimal with 50 wt% PNAS, increased by 47.0% compared with that of pristine PVC, which might be due to the larger molecular chain rigidity.
In the meantime, the corresponding impact strength was slightly improved with the addition of PNAS, different from some studies reported before. 32,34,35 The reason was probably attributed to the PNAS nanoparticles debonded from the PVC resin and deformation of the resin materials around the PNAS nanoparticles. 36,37
Conclusions
In this study, a monodisperse PNAS nanolatex was synthesized via seed microemulsion polymerization, and subsequently, the latex used as a heat-resistant modifier was directly mixed with PVC resin to prepare a series of PVC/PNAS hybrid composite with different compositions of 100:0, 90:80, 70:30, 60:40, and 50:50. For the PNAS latexes, real core–shell structural nanospheres were obtained when a starved feeding mode of reaction monomers was applied. FTIR and DSC analyses revealed a homogeneous molecular structure of PNAS copolymer with high monomers conversion. DSC analysis and TGA revealed excellent compatibility between the two polymers and improved effects of PNAS on the thermal properties of PVC resin. When the PNAS loading was 50 wt%, the T g value of the blends increased by 22.4°C, compared with that of pristine PVC resin. Mechanical measurements showed that with the addition of PNAS, both the tensile strength and the impact strength of PVC enhanced. The resulting PNAS nanospheres may find applications as effective, adjustable, and economic heat-resistant modifiers.
Footnotes
Acknowledgements
The authors are grateful for the support and funding provided by the Foundation of National Natural Science Foundation of China, Space Supporting Fund from China Aerospace Science and Industry Corporation and for the financial support from the Foundation of Aeronautics Science Fund and the Fundamental Research Funds for the Central Universities.
Author Contributions
The authors Jin Wang and Hua Qiu contributed equally to this work and should be considered co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article The work was supported by the Foundation of National Natural Science Foundation of China (No. 51773169); Space Supporting Fund from China Aerospace Science and Industry Corporation (No. 2017-HT-XG); the Foundation of Aeronautics Science Fund (No. 2017ZF53071); and the Fundamental Research Funds for the Central Universities (No. 3102017jg02003).
