Abstract
Polymer blends of polyamide 6 (PA6) and phenol novolac (PN) were prepared by melt mixing. Up to 30 wt% of high molecular weight PN (HPN) or low molecular weight PN (LPN) was blended with PA6, and the physical and mechanical properties were examined. The water absorption of PA6 is inhibited by PN, and this effect is independent of the molecular weight of PN. PA6 and PN are miscible, and their blends show a single glass transition temperature (T g) that is higher than that of PA6. HPN can enhance the T g of PA6 more efficiently than LPN because of its high T g. PA6 and the PA6/LPN blend after immersion in water had lower-than-room-temperature T gs transitioning to rubbery states. In contrast, the PA6/HPN blend after immersion in water had a higher-than-room-temperature T g. The PA6/HPN blend in water has excellent mechanical properties in its glassy state compared to those of PA6 in the dry state. Thus, the PA6/HPN blend can be used to broaden the applications of PA6, especially in humid conditions.
Introduction
Polyamides are widely used in industry because of their excellent mechanical properties, chemical resistance, and formability. Polyamide 6 (PA6) is one of the most popular and mass-produced polyamides. However, PA6 is easily plasticized by water molecules, and the glass transition temperature (T g) of PA6 can be depressed below room temperature (R.T.), which results in poor mechanical properties in humid conditions. Thus, it is important to improve the mechanical properties of PA6 under these conditions to enable its wide application.
There are many attempts to enhance the mechanical properties of PA6. For example, PA6 matrix composites incorporating nanofiller such as clays 1,2 or carbon nanotubes 3,4 were reported. On the other hand, there have been some more fundamental attempts, which are preventing water absorption and increasing the T g of PA6 by blending it with phenol-containing polymers such as phenol novolac (PN), 5 –7 polyvinylphenol, 8 and their derivatives. 9 These polymers can form strong hydrogen bonds between the carbonyl groups of PA6 and the hydroxyl groups of phenol-containing polymers. Quantum mechanical calculations have shown that the hydrogen bonds between PA6 and phenol-containing polymers are stronger than the hydrogen bond between PA6 and water molecules. 7 This is one explanation for the decreased water absorption of PA6 after blending with phenol-containing polymers. The good miscibility of PA6 and phenol-containing polymers is also attributed to the formation of strong hydrogen bonds. Most polymer blends are immiscible because the entropy of mixing is almost zero. However, some previous studies have shown that strong interactions through hydrogen bonds can promote the miscibility of polymer blends. 10 –12
In this study, we prepared polymer blends of PA6 and a phenol-containing polymer with a high T g to improve the mechanical properties of PA6 in humid conditions. An especially high molecular weight PN (HPN) was synthesized by the phosphorous acid phase separation (PAPS) process and used as a phenol-containing polymer. PN synthesized by the PAPS process has a low amount of free phenol and a narrow molecular weight distribution. Furthermore, it has a high softening point compared to standard PN. 13 To examine the influence of the molecular weight of PN, blends of PA6 with two types of PN having different molecular weights were prepared, and the effects of the PN components on the physical properties of the blends were investigated.
Experiment
Materials
PA6 used in this study was a standard grade PA6 (A1030BRL) purchased from Unitika Corporation, Japan. The number-average molecular weight of PA6 was 14,500. Two types of PNs synthesized by the PAPS process were purchased from Asahi Organic Chemicals Industry Corporation, Japan. Standard molecular weight grade (PAPS PN4) as a representative low molecular weight PN (LPN) and an ultrahigh molecular weight grade (PAPS PN70) as a representative HPN were used in this study. The number-average molecular weights of LPN and HPN were 760 and 2200, respectively.
Processing
All polymers were vacuum dried for 12 h at 80°C before melt mixing. The PA6/PN blends were melt mixed with a twin-screw extruder (KZW15-60MG, screw diameter (D) = 15 mm and length (L)/D = 60, Technovel Corporation, Japan). The compositions of the fabricated polymer blends are summarized in Table 1. The barrel temperature was set to 250°C and the screw rotating speed was set to 70 r min−1. The weight ratio of PA6/PN blends was controlled by the feeding speed of each polymer. The strands obtained upon extrusion were immediately cooled in water, and then cut into pellets by a pelletizer.
Sample IDs and weight fractions of prepared samples.
PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Injection molding
The obtained pellets were vacuum dried for 12 h at 80°C before molding. The dried pellets were molded into dumbbell-shaped specimens by the injection molding machine (Mobile-0813, Shinko Sellbic Corporation, Japan) at a cylinder temperature of 250°C and a mold temperature of 150°C.
Water absorption testing
At first, the dumbbell specimens were vacuum dried at 80°C for 12 h before measuring their weights. The dried specimens were immersed in distilled water at 80°C and weighed after 1, 4, and 9 h. The amount of water absorbed by the specimens was calculated from the increased weight. After immersion in distilled water maintained at 80°C for 1 day, the specimens were moved in to distilled water maintained at 23°C and immersed in water until a constant weight was obtained. The amount of water absorbed at this point is estimated to be the equilibrium moisture content.
Dynamic mechanical analysis
Measurement of PNs
The softening point is observed as a rapid decrease in the storage modulus; this and the T g were evaluated by dynamic mechanical analysis (DMA; DVA-225, ITK Corporation, Japan) for the two PNs. LPN and HPN were vacuum dried at R.T. for 24 h, and then molded into rectangular specimens (0.5 × 5 × 35 mm3) by press molding. The molding temperatures were 80°C and 140°C for LPN and HPN, respectively. DMA was carried out between 23°C and 200°C at a heating rate of 5°C min−1 and a frequency of 10 Hz. The dynamic strain was set to 0.01% because of the brittleness of the PNs.
Measurement of polymer blends
The softening points and T g values of the polymer blends were also evaluated by DMA (DVA-225, ITK Corporation). The pellets of each compound were vacuum dried at 80°C for 12 h, and molded into rectangular specimens (0.5 × 5 × 35 mm3) by hot press molding. To obtain highly crystallized specimens, two hot press machines were used. In the first heating, the temperature was set to 250°C, which is above the melting points of PA6 and the polymer blends (about 230°C), and the press molding time was set to 1 min. In the second step, the press temperature was set to 150°C to promote the crystallization of the molded specimens, and the specimen was pressed for 1 min. One specimen vacuum was dried at 80°C for 12 h to dryness, and the other was immersed in diluted water maintained at 23°C until equilibrium water absorption was reached. These samples were prepared for the purpose of comparing the mechanical properties of the specimens in these different states. DMA was carried out in a temperature range of −100°C to 150°C at a heating rate of 5°C min−1 at a frequency of 10 Hz and a dynamic strain of 0.05%.
Differential scanning calorimetry
The crystallization starting temperatures and the crystallization rate were evaluated by differential scanning calorimetry (DSC; Q1000, TA Instruments, USA). About 10 mg of the sample was cut from the center part of the dumbbell specimen and encapsulated in an aluminum DSC pan. The DSC measurements were carried out under nitrogen atmosphere at a heating rate of 10°C min−1 and a cooling rate of 20°C min−1 between 20°C and 250°C.
Mechanical testing
Three-point bending tests on the polymer blends were carried out using a universal testing machine (4302, Instron Corporation, USA). After testing, stress–strain (SS) curves were obtained for all specimens. The measurements were conducted at R.T. with a supporting distance of 32 mm and a test speed of 1 mm min−1. Bending tests were carried out for the dumbbell specimens in both the dry state (after vacuum drying at 80°C) and the water absorbed state (equilibrium state in water maintained at 23°C).
Results
Water absorption behavior
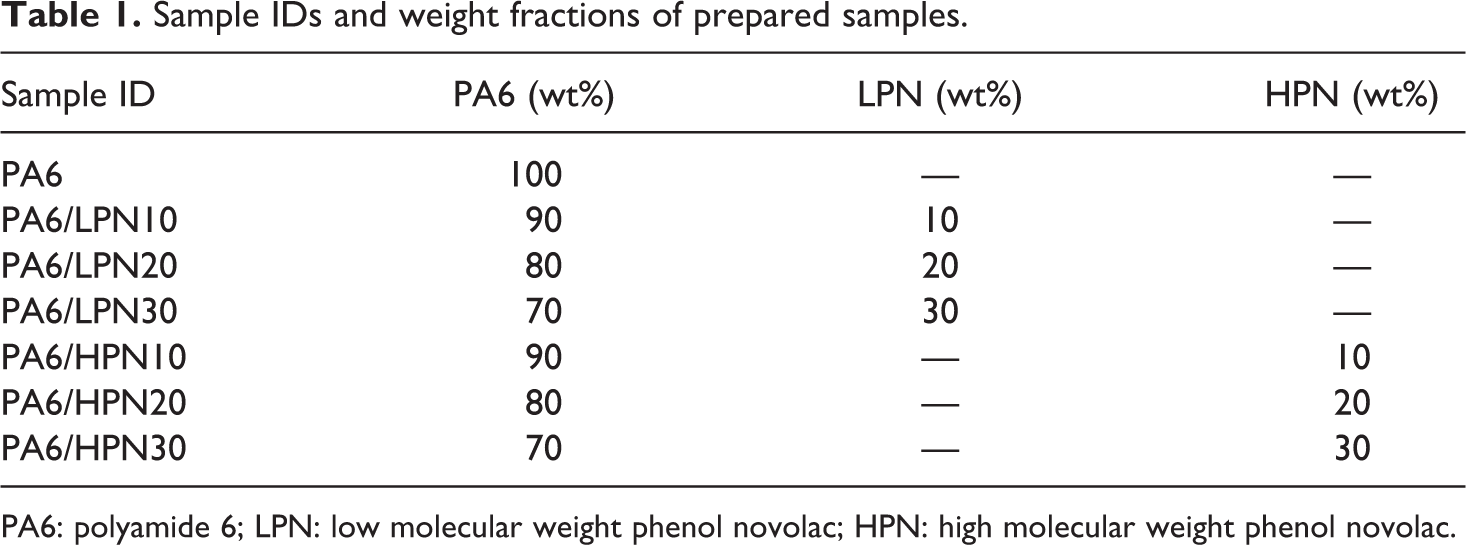
The water absorption behaviors of PA6/PN blends in distilled water at 80°C are shown in Figure 1. The slopes in the initial stages indicate the water absorption rate. The results for the PA6/LPN blends (Figure 1(a)) reveal that the PA6/LPN blends show slower water absorption behaviors in comparison with that of PA6, and the water absorption slowed with increasing PN content. PA6 reaches an equilibrium value after immersion for 16 h, but PA6/LPN30 had not reached water absorption saturation after immersion for 49 h. As shown in Figure 1(a) and (b), similar results were obtained for the PA6/HPN blends, independent of the molecular weight of the PNs. The saturated values of water absorption after the immersion in diluted water at 23°C of all specimens are listed in Table 2. The equilibrium water absorption of PA6 decreased after blending with PN, and the equilibrium values are independent of the molecular weight of the PN. The equivalent water absorption amounts of PA6/LPN30 and PA6/HPN30 were evaluated to be about half the value of PA6.
Water absorption variations of PA6 and the polymer blends. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Saturated water absorption values of PA6 and polymer blends after reaching a constant value in distilled water maintained at 23°C.

PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Viscoelasticity
Measurement of PNs
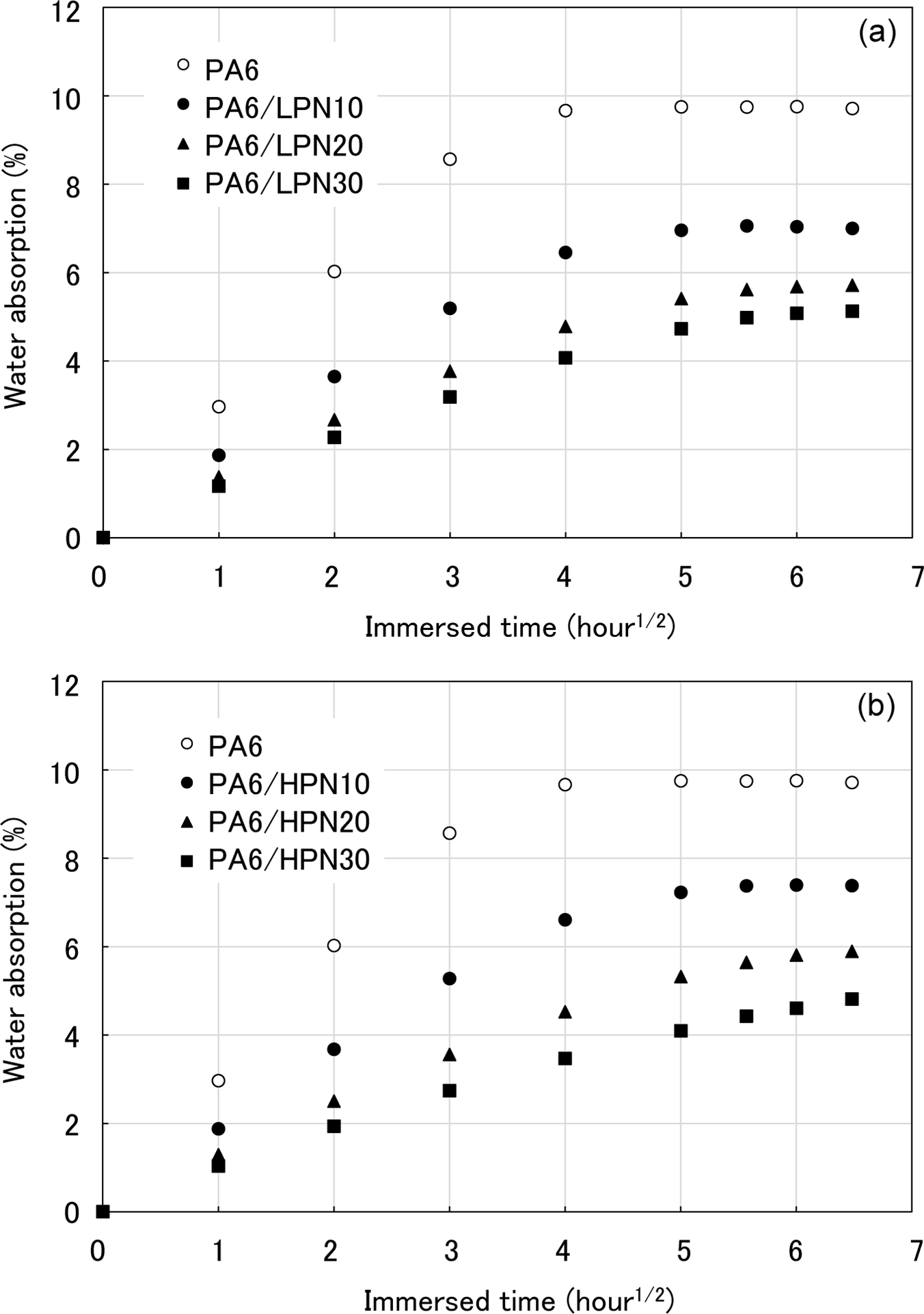
The temperature dependencies of the dynamic elastic modulus (E′) of the PNs are shown in Figure 2(a). The softening point defined in this study can be understood as the temperature at which E′ shows a notable decrease. As shown in Figure 2(a), different softening points were obtained for two types of PN. LPN shows a glass-to-liquid transition at 50°C and a drastic decrease in E′ occurs because PN is an amorphous polymer and has no crystalline region. On the other hand, HPN maintains the high E′ of the glassy state at about 50°C, which is the softening point of LPN. The transition from the solid to the liquid state of HPN, accompanied by a decrease in E′, occurs at around 90°C. Figure 2(b) shows the temperature dependence of the dissipation factor (tanδ) of the PNs. The T g of the polymers can be defined as the peak temperature of tanδ. The estimated T gs of LPN and HPN are 80°C and 125°C, respectively.
The DMA results of PNs. (a) Temperature dependence of the storage modulus of the PNs and (b) temperature dependence of tanδ of PNs. DMA: dynamic mechanical analysis; PN: phenol novolac; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Measurement of polymer blends
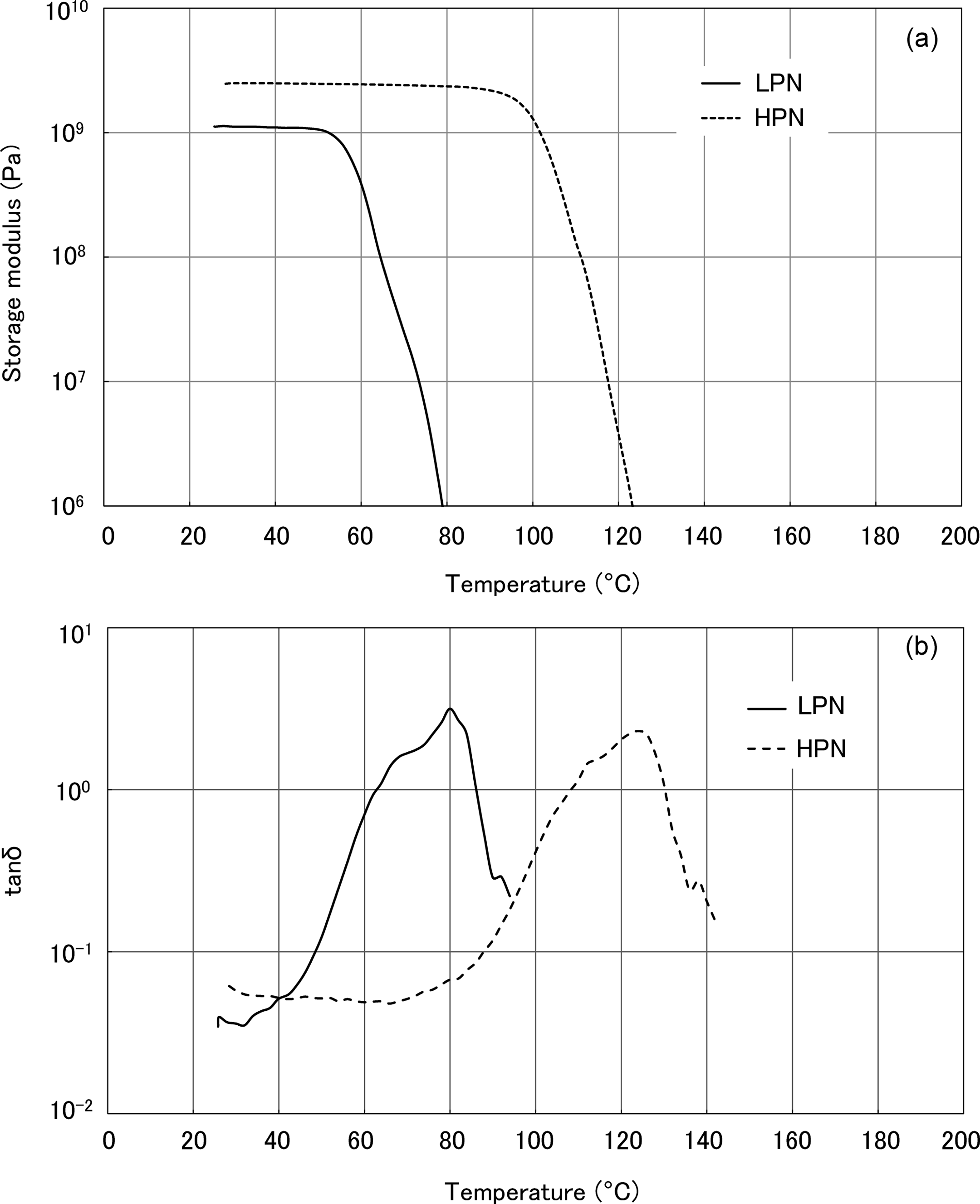
The DMA results of the polymer blends in the dry state are shown in Figures 3 and 4. Figure 3 shows the temperature dependence of E′. Figure 3(a) shows that the PA6/LPN blends exhibit a single softening point at higher temperatures than PA6. The softening point of the PA6/LPN blend shifted to higher temperatures with increasing PN content. In contrast, PA6/LPN30 has a softening point about 20°C higher than that of PA6. The results for the PA6/HPN blends are shown in Figure 3(b) and show a single, higher softening point than PA6. The PA6/HPN blends have a higher softening point than the PA6/LPN blends with the same PN content. PA6/HPN30 has a 40°C higher softening point than PA6.
Temperature dependence of storage modulus of PA6 and the polymer blends in the dry state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
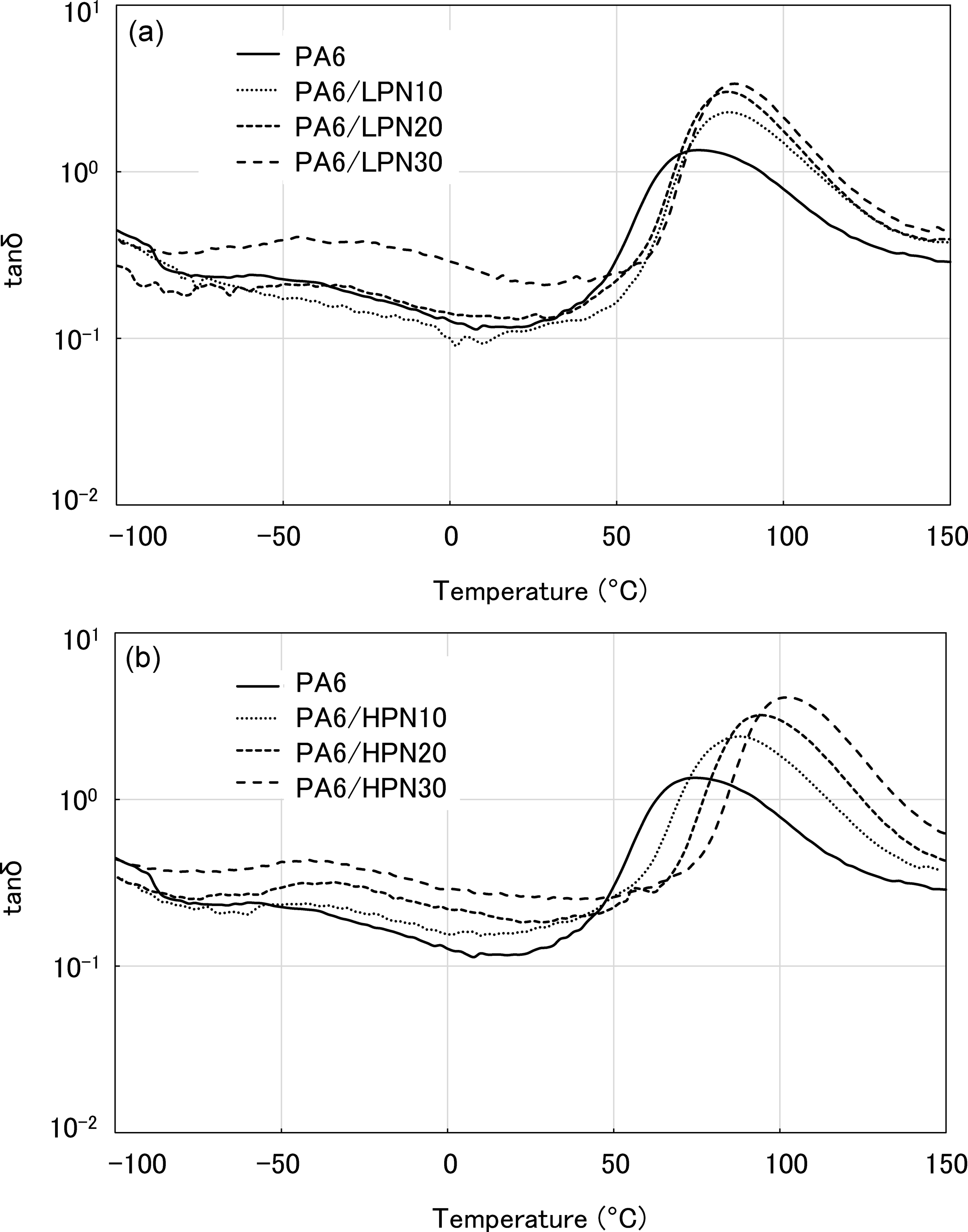
Temperature dependence of tanδ of PA6 and the polymer blends in the dry state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
The temperature dependencies of tanδ are shown in Figure 4. All the PA6/LPN and PA6/HPN blends have a single T g, which shifted to higher temperature with increasing PN content. The PA6/HPN blends have higher T gs than the PA6/LPN blends at the same PN content. These results show that the HPN component can shift the T g of PA6 to higher temperatures more efficiently than the LPN one.
The DMA results of the water absorbed states are shown in Figures 5 and 6. Figure 5 shows the temperature dependencies of E′ of the PA6/PN blends. The softening point of PA6 shifted below R.T. on the absorption of water. PA6 in the water absorbed state has a low E′ and is rubbery at R.T. Figure 5(a) shows that the softening point of PA6/LPN is single and higher than that of PA6. The softening point shifted to higher temperatures with increased PN content because T g of LPN is sufficiently higher than that of PA6 in the water absorbed state.
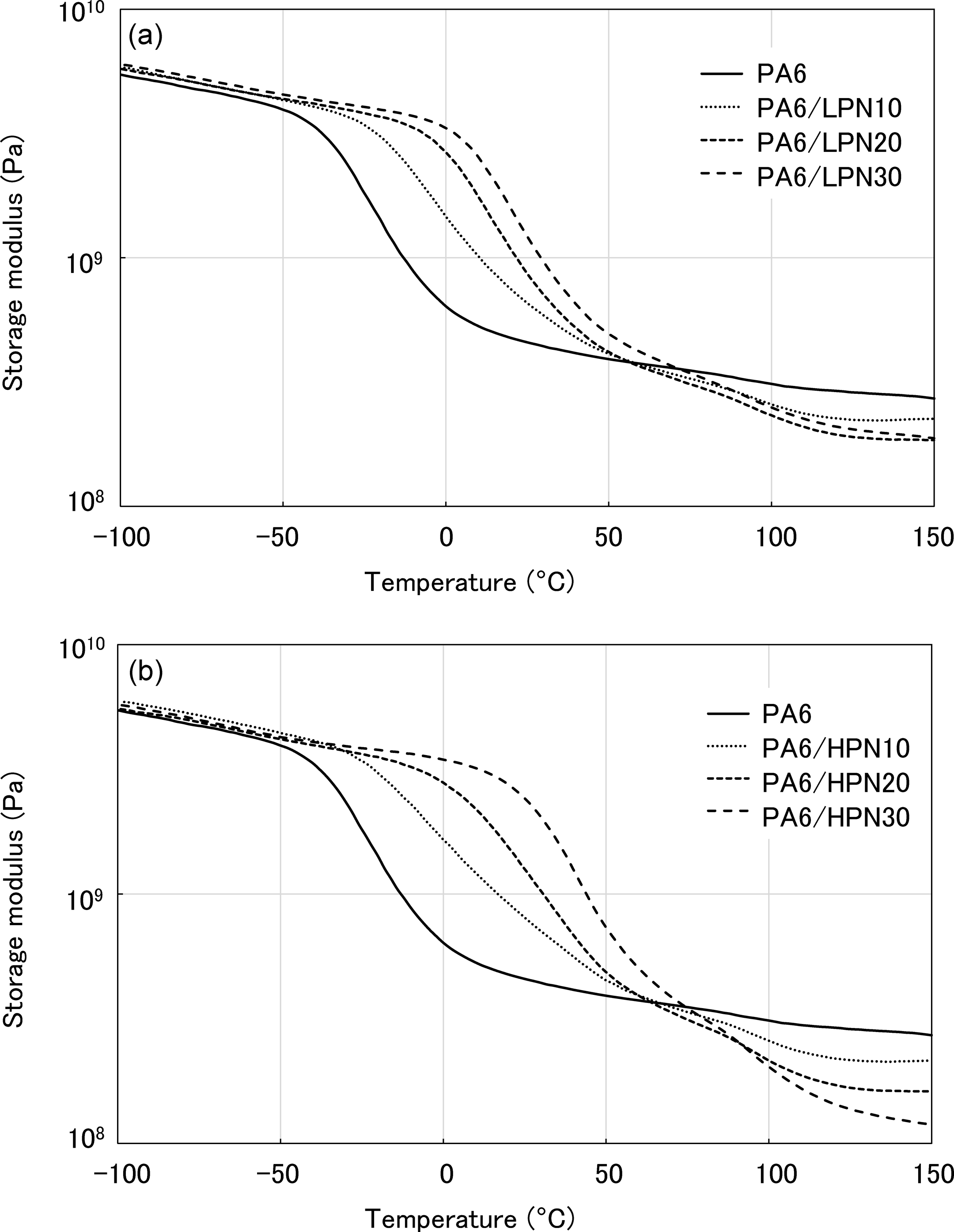
Temperature dependence of storage modulus of PA6 and the polymer blends in the water absorbed state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
The results for the PA6/HPN blends are shown in Figure 5(b) and show that the softening point of PA6 increased significantly after blending with HPN. The softening point of PA6/HPN30 was about 20°C. The E′ value of PA6/HPN30 in the water absorbed state remained high, and its E′ was the same as that of PA6 in the dry state at R.T. The temperature dependencies of tanδ for polymer blends are shown in Figure 6. The PA6/PN blends also show a single T g in which the T g of PA6 is shifted to a higher temperature on blending with PN. These results indicate that HPN increases the T g of PA6 more efficiently than does LPN, and PA6/HPN30 has a T g greater than R.T. These results show that PA6/HPN30 blends are maintained in the glassy state at R.T. and contain a small amount of absorbed water. On the other hand, PA6/LPN30 blends become rubbery at R.T.
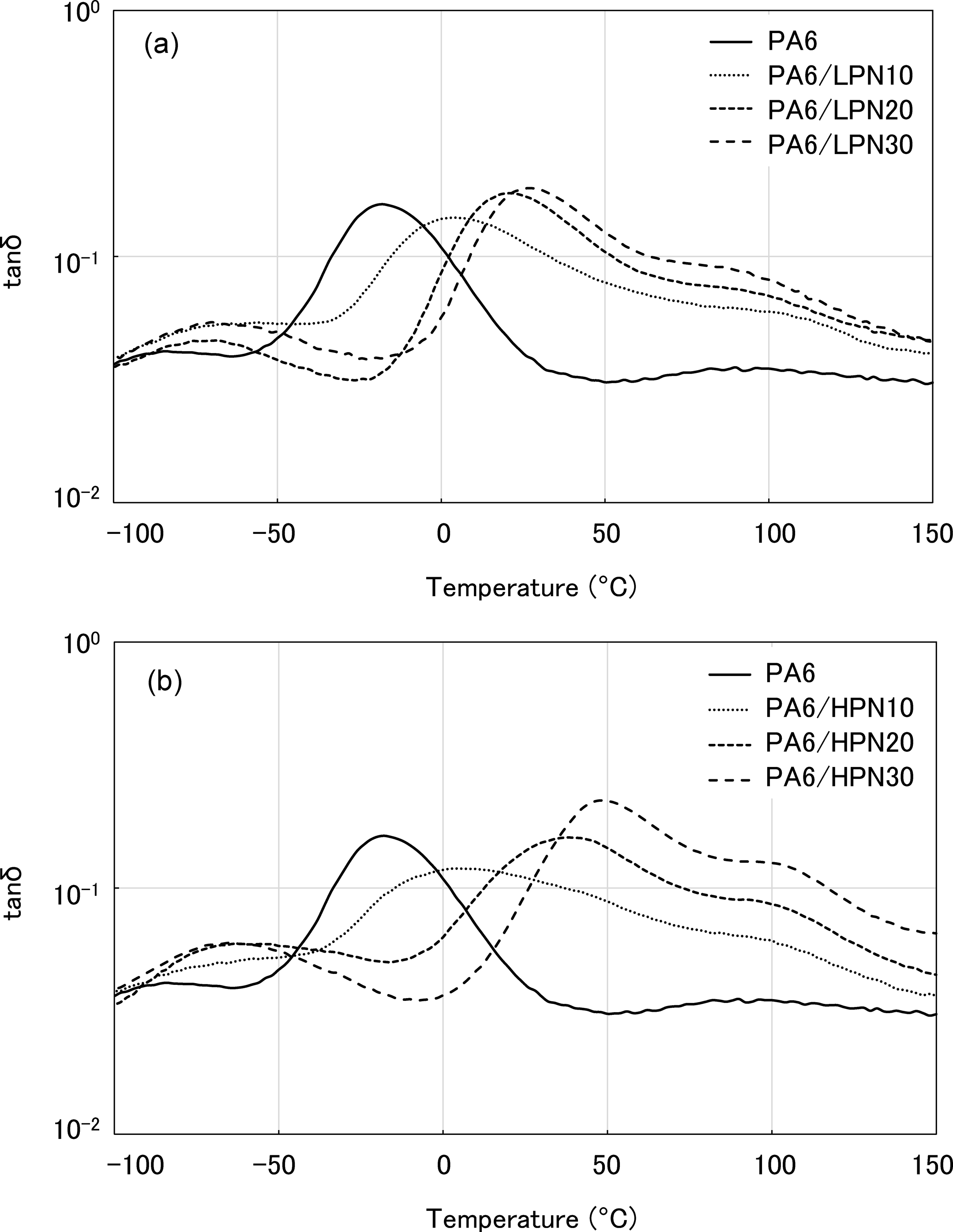
Temperature dependence of tanδ of PA6 and the polymer blends in the water absorbed state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Crystallization behavior
The DSC results of the first cooling scan are shown in Figure 7. The crystallization temperature of the polymer is defined by the exothermic peak in the cooling scan. Figure 7 shows that the crystallization temperature of PA6 is lowered by blending with PN. PA6/LPN30 has a 15°C lower crystallization temperature than that of PA6. The PA6/HPN blends had lower crystallization temperatures than those of the PA6/LPN blends, and PA6/HPN30 has a 25°C lower crystallization temperature than that of PA6. The half-width of the crystallization peak represents the crystallization rate. Figure 7 shows that the addition of PN reduces the crystallization rate of PA6. From the results for PA6/LPN and PA6/HPN, we confirmed that the PA6/HPN blends had lower crystallization rates than the PA6/LPN blends.
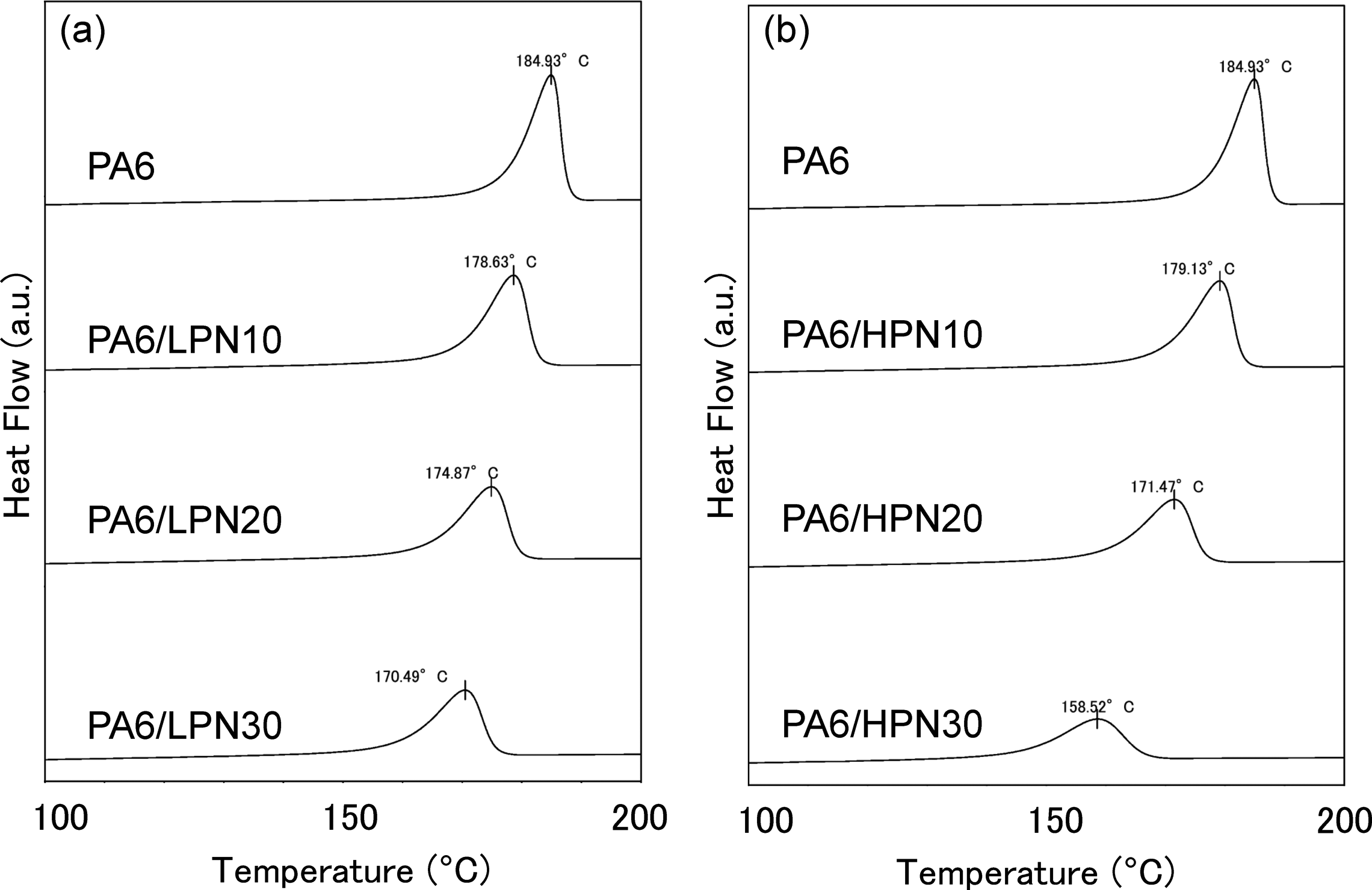
DSC curves of PA6 and the polymer blends obtained in the first cooling scan. (a) PA6/LPN blends and (b) PA6/HPN blends. DSC: differential scanning calorimetry; PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Mechanical properties
As a result of the bending tests, SS curves for the PA6/PN blends in both the dry state and the water absorbed state were plotted, as shown in Figures 8 and 9, respectively. The results for the PA6/LPN blends in the dry state show that the flexural modulus and strengths of the blends increased with increasing LPN content, as shown in Figure 8(a). PA6/LPN30 underwent brittle fracture, while other specimens continue to deform after their yield points. Figure 8(a) and (b) shows that these features are independent of the molecular weight of PN, and the PA6/HPN blends show similar behaviors. Both PA6/LPN30 and PA6/HPN30 have 30% higher flexural modulus and maximum strength compared to that of PA6. However, in the water absorbed state, there are significant differences in the mechanical properties of the LPN and HPN blends. In the water absorbed state, all PA6/LPN blends have inferior mechanical properties to PA6 and have almost identical SS curves in all compositions as shown in Figure 9(a). On the other hand, Figure 9(b) reveals that PA6/HPN30 has five times higher flexural modulus and three times higher maximum strength than PA6. The elastic flexural modulus of PA6/HPN30 in the water absorbed state is comparable to that of PA6 in the dry state. These results show that the flexural modulus and strength of PA6/LPN blends are not dependent on the PN content, but these PA6/HPN blends increased remarkably on blending with more than 20 wt% PN in humid conditions.
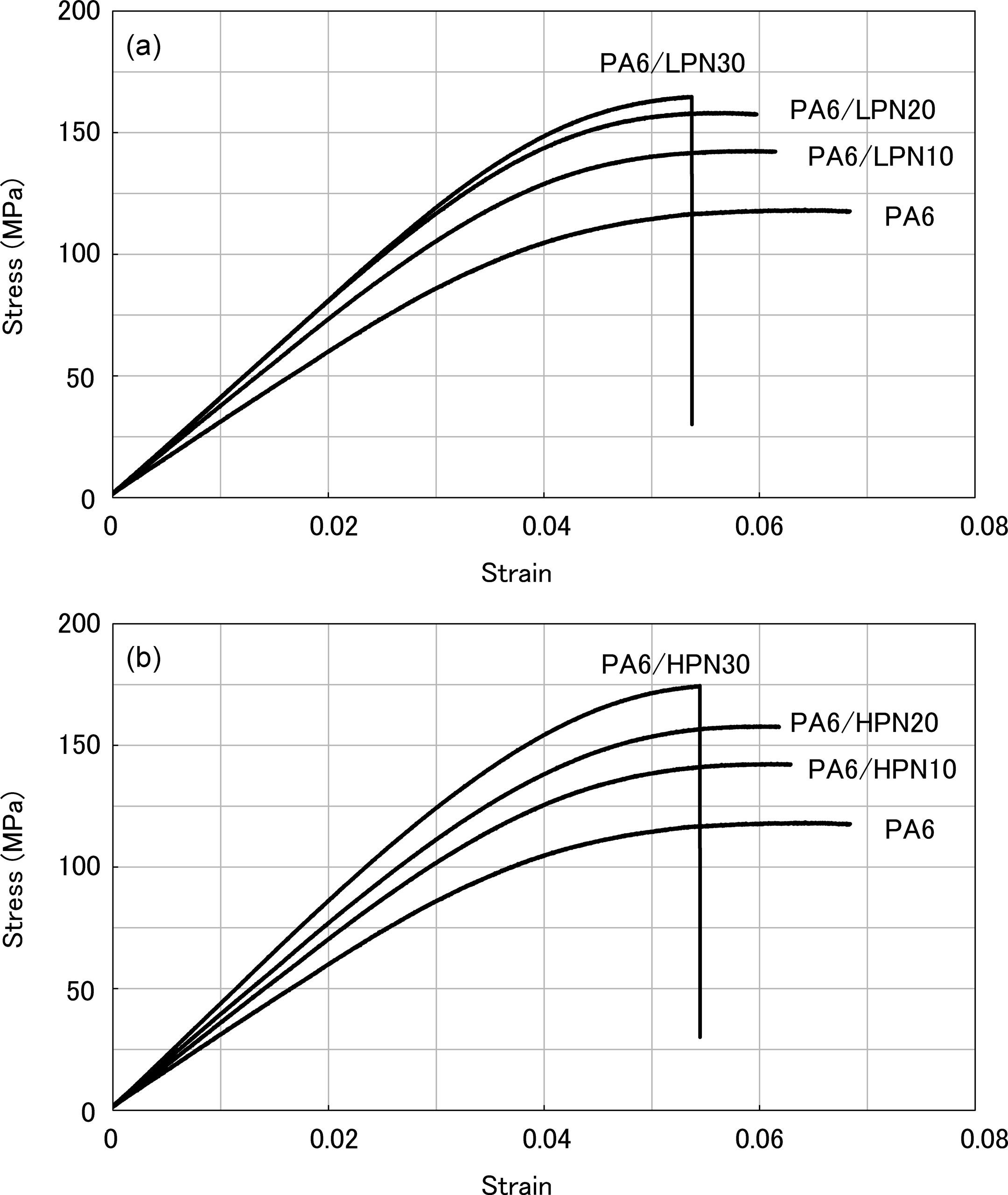
Stress–strain curves of PA6 and polymer blends at dry state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
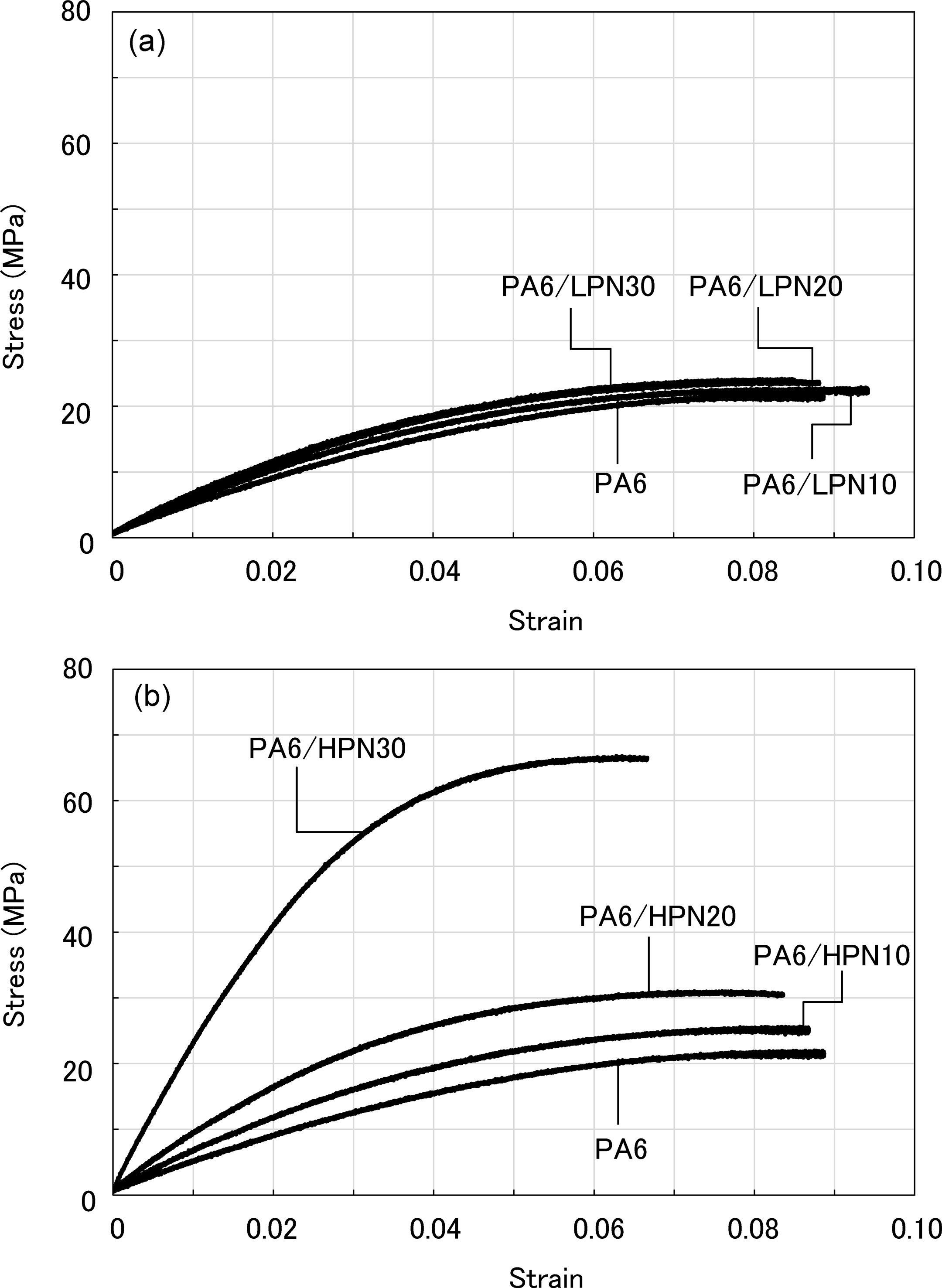
Stress–strain curves of PA6 and polymer blends in the water absorbed state. (a) PA6/LPN blends and (b) PA6/HPN blends. PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
Discussion
As discussed previously, the blends of PA6 and two types of PN have a single T g and lower water absorption for all PN contents in comparison with PA6.
As shown in Figure 1, there are no obvious differences in the water absorption behavior between the LPN and HPN blends. This result suggests that the strength of hydrogen bonding between PA6 and PN is unaffected by the molecular weight of PN. One plausible reason for the decreased water absorption on blending with PN is related to the strong hydrogen bonds between PA6 and PN.
The PA6/PN blends show lower storage modulus than PA6 at high temperature above their T g as shown in Figures 3 and 5. One of the possible causes is decreased amount of PA6 crystal, which contributes to maintaining high modulus at high temperature, by blending amorphous PN.
Figure 7 shows that the PA6/PN blends have lower crystallization temperatures and slower crystallization rates than PA6. In some studies, it has been concluded that miscible polymer blends of semicrystalline polymers crystallize by pushing the additive polymer out of the crystallizing phase. 14,15 The results obtained in this study clearly support this since the HPN component prevented the crystallization of PA6, possibly because it is difficult to remove the large molecule from the crystalline lamellae. In engineering practice, semicrystalline polymers might be molded and crystallized in a nonequilibrium state during the injection molding process, which last for less than 1 min. Hence, a practical technology for improving the crystallization rate of PA6/PN blends is required.
The experimental results of the PA6/PN blends in both dry and water absorbed states are summarized in Table 3. Table 3 shows that the mechanical properties of the polymer blends in the dry state are not dependent on the molecular weight of PN. All blends in the dry state show T gs higher than R.T. These results may indicate that the increased flexural modulus and strength on the blending with PN are attributed to the stiffness and strength of PN.
T g and mechanical properties of PA6 and polymer blends in the dry state and the water absorbed state.
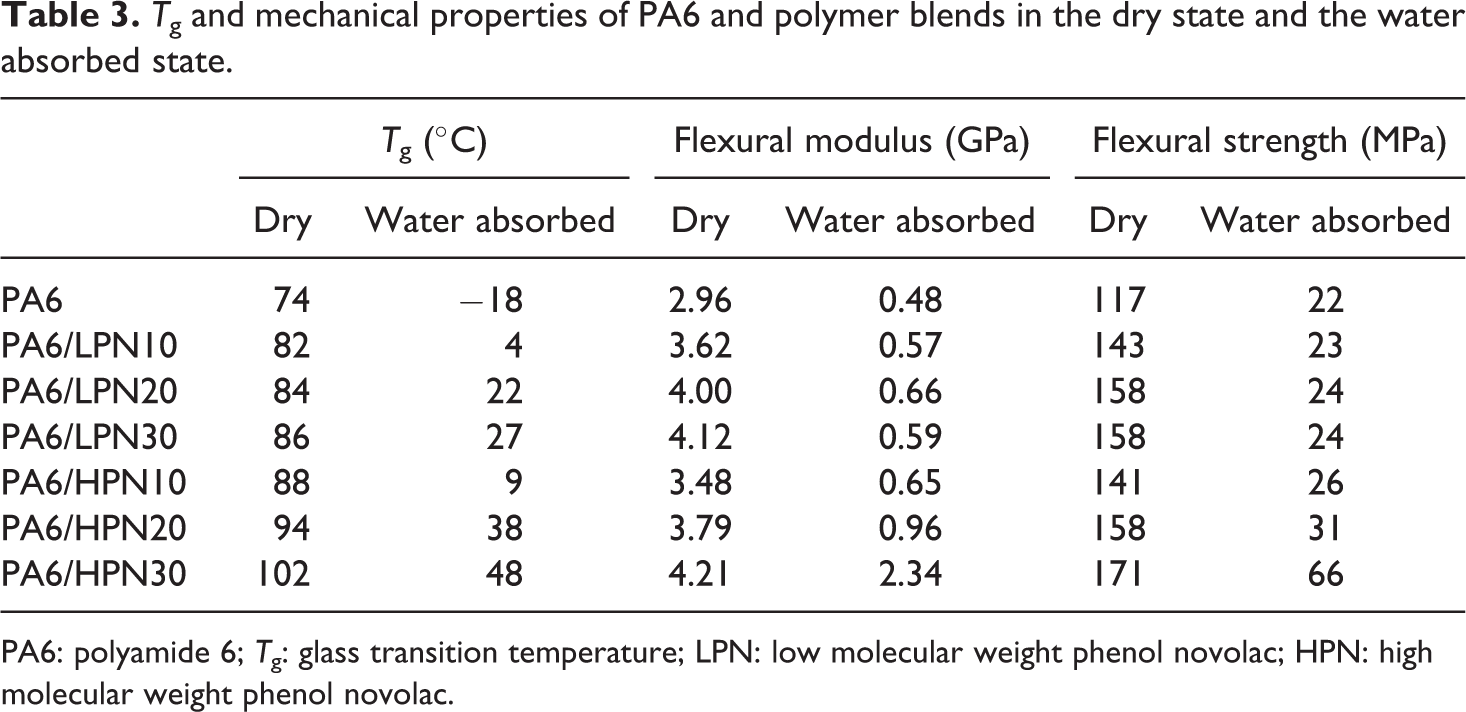
PA6: polyamide 6; T g: glass transition temperature; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
On the other hand, there is a remarkable difference in the mechanical properties between the PA6/LPN blends and PA6/HPN blends in the water absorbed state, as shown in Table 3. Table 3 clearly shows that the blends that have T gs above R.T. are associated with superior mechanical properties. These results suggest that the T gs of the polymer blends may have a strong effect on the mechanical properties of the PA6/PN blends.
Previous studies have discussed the T gs of polymer blends, 16 –18 and we used the Kwei equation to model the experimental results in this study because it is known to be suitable to understand the relationship between the average T g and the polymer–polymer interactions of miscible polymer blends. 19 The Kwei equation is shown in equation (1) and was established to represent positive deviations from the weight-averaged value
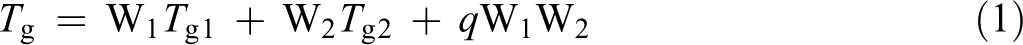
Here, T g1 and T g2 refer to the T gs of the two constituent polymers, and W1 and W2 refer to the weight fractions of the additive polymers. The q value is regarded as representative of the strength of the polymer–polymer interactions. The experimental relationships between the PN content and T g in the water absorbed state are shown in Figure 10. The dotted lines illustrated in Figure 10 correspond to the T gs predicted by the Kwei equation.
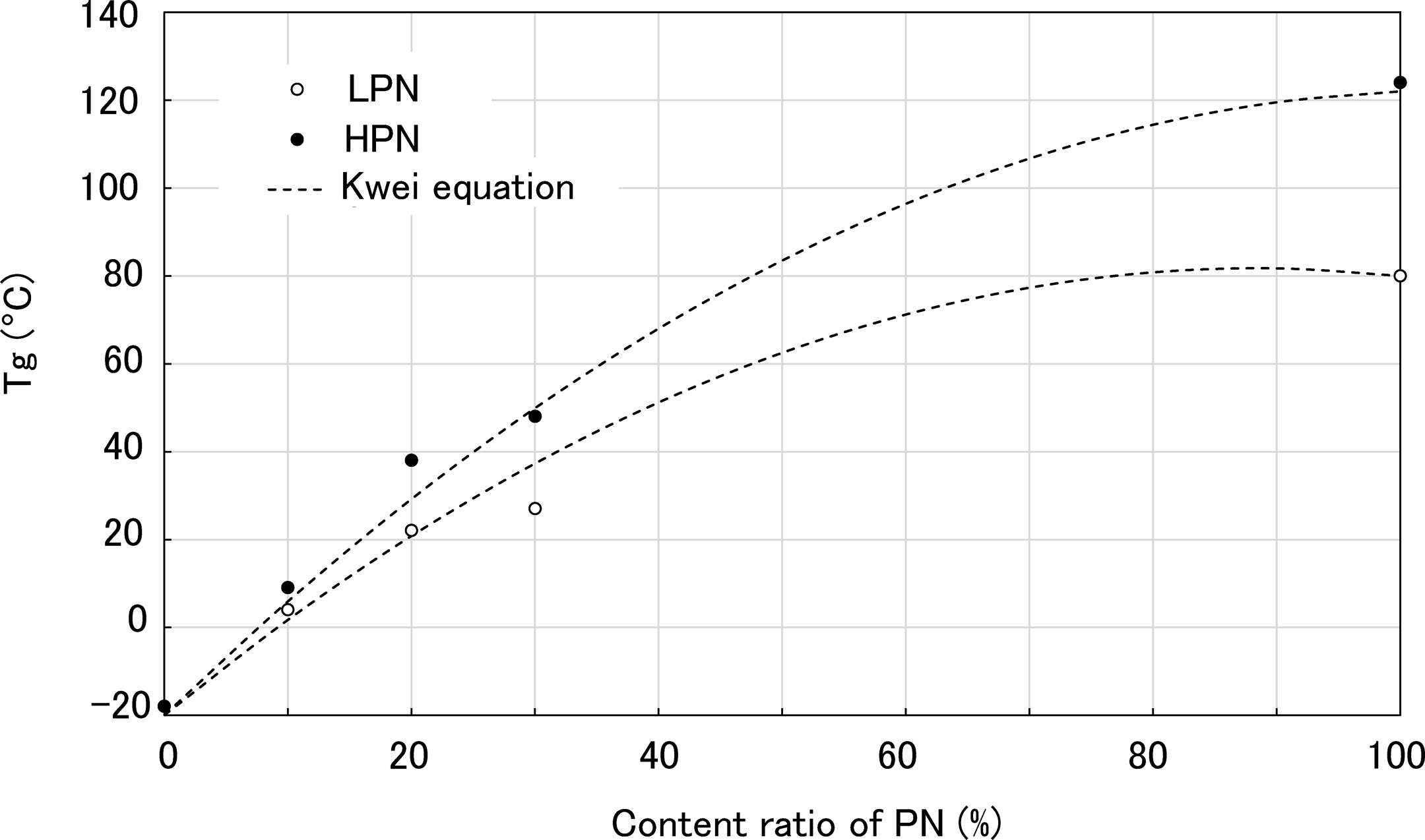
Relationship between PN content and T g of the polymer blends. Dotted lines indicate the theoretical value calculated using the Kwei equation. PN: phenol novolac; T g: glass transition temperature; PA6: polyamide 6; LPN: low molecular weight phenol novolac; HPN: high molecular weight phenol novolac.
The q value was set to be the same value in both PA6/LPN and PA6/HPN, in agreement with the experimental value. The PA6/PN blends with strong hydrogen bonds show higher T gs than the weight-averaged value. Thus, we found that the Kwei equation can predict the experimental results. Finally, the experimental results suggested that higher T g of HPN is attributed to the increase in the T g of PA6.
Conclusions
Polymer blends of PA6 with HPN have lower equilibrium water absorption values and excellent mechanical properties in the water absorbed state. The water absorption of PA6 is inhibited by PN, and the equilibrium water absorbed value of PA6/HPN30 is about half of that of PA6. HPN can increase the T g of PA6 more efficiently than LPN, and PA6/HPN30 has a T g greater than R.T. in the water absorbed state. PA6/HPN30 in the water absorbed state has a high elastic modulus in the glassy state, compared to that of PA6 in the dry state. These results indicate the possibility of the wider application of PA6, especially in humid conditions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
