Abstract
The effect of microcrystalline cellulose (MCC) on the thermal properties (melting and crystallization temperatures and percentage crystallinity) and degradation of low density polyethylene (LDPE)–MCC blends were investigated. Weight percentages of MCC were varied at 0, 0.5, 1, 2.5, 5, 10, 20, and 30 wt%. The thermal properties of the composites were studied using differential scanning calorimetry while the degradation test was carried out using soil burial method; the weight loss of LDPE/MCC composites was measured and analyzed over a period of 120 days. It has been found that the addition of MCC to LDPE increased the crystallization temperature from 99°C to 103.5°C and decreased the melting temperature from 117°C to 113.6°C. A rule of a nucleating agent has been given as an interpretation to this increase in the crystallization temperature and intensity of crystals by the increase of MCC content. The dramatic reduction was in the percentage crystallinity where the value reduced from 58% for neat LDPE to about 11% for LDPE/30 wt% MCC. On the other hand, the addition of MCC has a little effect on degradation of LDPE; the weight loss did not exceed 1.5% over a period of 120 days. It seems that even at high MCC concentration, LDPE long carbon chains restrict and increase the resistance to microorganism attack and hence, reduce the hydrolysis and degradability.
Introduction
Plastics are materials with a complex formation that have amazing properties compared to metals in manufacturing processes. The increase in the plastic production through the whole nation had introduced the world to environmental problems because most of the plastic materials have remained in the garbage deposits and landfills for decades. Among the most used polymers in our daily life is polyethylene (PE) which is a commodity polymer and has a simple structure of hydrocarbon polymer which has different types classified according to its density and branching. PE exists as high-density PE (HDPE), low-density PE (LDPE), and linear low-density PE (LLDPE). It is extremely hydrophobic, has high molecular weight and not biodegradable in its native form as it is not a good culture for microorganisms. 1 It is the first plastic to be used for food packaging and came into general use in the 1950s. 2 In the agriculture sector, the first use of a plastic film started in 1948 when Prof. E.M. Emmert exchanged the greenhouse with a PE film in order to save money. The amount of plastic materials used annually worldwide in the agricultural sector exceeds 6.5 million tons. 3
The more regrettable effect on the environment can be found in the huge amount of disposed plastics, where over 25 million tons of synthetic plastic are disposed on the ocean coasts and the environment around them. The high rate of plastic items credited to PE were 64% of the aggregate synthetic plastics, which can be found in the manufacturing of bottles, carry bags, disposable articles, rubbish holders, margarine tubs, milk containers, water pipes, and so on.
Here in Jordan, the average weight percentage of different sorts of PE (film and container) is around 15.1% of the total aggregate solid waste and this represents 71.6% of the different sorts of plastics waste. 4
However, a huge amount of plastics waste is driving a destructive impact on nature, bringing on ecological contamination as plastics have high resistance to biodegradation and this has negative consequences for human and creatures, principally influencing the endocrine system, growth issues, and may cause cancer. 5
Polymer biodegradation is a chemical degradation provoked by the action of microorganisms such as algae, bacteria, and fungi. 6 Biodegradation itself is considered a type of degradation involving biological activity where the primary degradation mechanism is started by the action of metabolism by microorganisms. Proteins, polysaccharides, and nucleic acids are natural polymers and degraded in biological systems by oxidation and hydrolysis. 7 On the other hand, synthetic biodegradable polymers degrade into biomass, carbon dioxide, and methane. 8 Although biodegradation is usually caused by biological activity, it will usually occur simultaneously with—and is sometimes even initiated by—abiotic degradation such as photodegradation and simple hydrolysis. 9
Temperature is one of the most important factors affecting microorganism growth. Also important are sources of carbon and nitrogen and pH. 10,11 The biodegradation process can be divided into aerobic and anaerobic degradation. If oxygen is present, aerobic biodegradation occurs and carbon dioxide is produced. If there is no oxygen, an anaerobic degradation occurs and methane is produced instead of carbon dioxide. 12,13 The chemical structure of polymers that is responsible for functional group stability, reactivity, hydrophilicity, and swelling behavior is the most important factor affecting the biodegradability of polymeric materials. Other important factors are inter alia, physical and physicomechanical properties such as molecular weight, porosity, elasticity, and morphology. 14
Nowadays, the inversion of PE from a nondegradable material into a biodegradable material has attracted the intention of many researchers more than ever before as the world’s regulations are to decrease the pollution to its most reduced level or even diminish it. 2 Added substances to LDPE like plasticizers, lubricants, stabilizers, and antioxidants might make it as a host medium to the microorganisms. 15
Cellulose is reckoned as the abundant polymer existing with a yearly natural production of 1.5 × 1012 tons, and is one of the strongest and stiffest fibers that has a high potential to perform as a reinforcing agent in biopolymers. It is considered as a sustainable source of raw materials and is hygroscopic and biodegradable; this can expand water absorption in PE/cellulose blends and hence increase the biodegradation qualities when presented to microscopic organisms or fungi. Cellulose-based polymer composites are described by low cost, low density, high specific stiffness, high tensile strength, biodegradability and flexibility during processing with no damage to the equipment, and good mechanical properties. 16
Microcrystalline cellulose (MCC) for industrial purposes is usually obtained from natural sources; the wood pulp and purified cotton linters. The need for environment-friendly degradable polymer products has attracted researcher’s attention, and cellulose is one among the different materials recommended as additives to help and assist in the degradation of polymers. 17 In the present study, the composites of interest are prepared by blending LDPE in a powder form with MCC. Since the addition of cellulose in any form is expected to alter the properties of the polymer, the main objective of this work is to investigate the effect of MCC concentration on the thermal properties and degradation of LDPE.
Experimental
Materials
The utilized PE was LDPE (LDPE 2102TN00 W), having a melting temperature of 115.4°C, a density of 921 kg m−3, and a melt flow index of 1.9 g/10 min. This product, produced by SABIC, Saudi Arabia, is a general purpose grade without additives, and it has excellent processability and stretchability.
MCC in a white powder form was supplied by ALDRICH Chemistry, Germany, this MCC is biodegradable and carries hydrophilic hydroxyl group that interacts with oxygen atoms from another cellulose chain. The bulk density of this cellulose is 1.27–1.60 g cm−3 and the average particle size is 20 µm. The odorless cellulose has a high tensile strength and a good thermal stability, and has a melting point ranging between 260°C and 270°C.
Preparation of LDPE/MCC composites
In order to ensure good mixing with cellulose powder, LDPE was ground to powder. LDPE powder was mixed physically with the MCC and then fed to the extruder’s hopper by direct addition. A single screw extruder (Axon, Sweden) was used to prepare the LDPE/MCC composites of various compositions as shown in Table 1. Extruder’s temperature zones were set at 150°C near the feeder, 175 and 190°C in the middle zones, and 210°C at the die. On exit from the extruder’s die (the point that gives the final shape), the extrudate passed through a water trough at room temperature for further cooling and solidification. The cooled laces are chopped into small granules using a pelletizing machine (Axon Pelletizer) equipped with a steel blade. The obtained LDPE granules were dried in an oven at 100°C to remove any moisture.
Composition of LDPE/MCC composites.
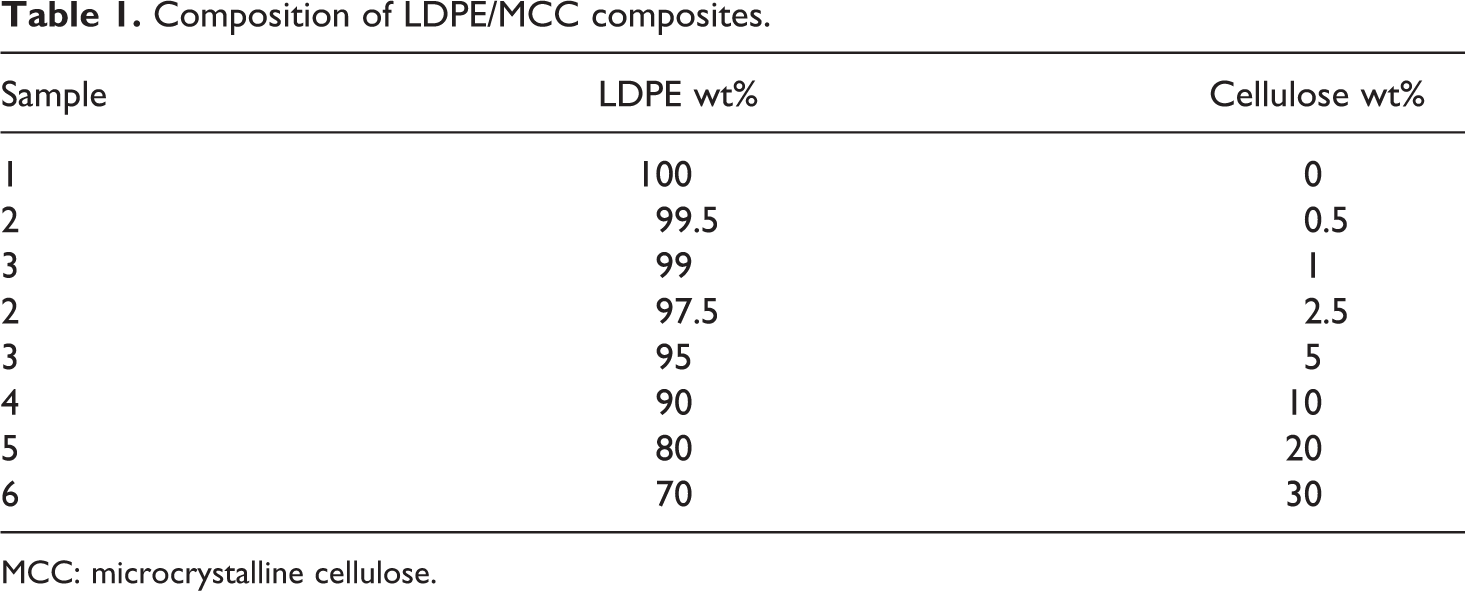
MCC: microcrystalline cellulose.
Thermal analysis
A differential thermal analysis is used to examine materials and detect the phase transitions and changes in thermodynamic properties under heating or cooling. 18
All samples were analyzed using a differential scanning calorimeter (DSC; METTLER TOLEDO, USA, model 823) prior to thermal testing; the instrument was calibrated using pure Indium and Zinc. The obtained heating/cooling thermal scans were used to determine the melting and crystallization temperatures in addition to the percentage crystallinity. Five to ten milligrams of LDPE/MCC composite were weighed into the aluminum pan using a six-digit analytical balance. The sample prepared in the pan and an empty pan were both placed in the instrument and analyzed according to the following program: The sample was heated from 20°C to 220°C at a rate of 10°C min−1. It was kept at 220°C for 3 min to remove thermal history. The sample was then cooled from 220°C to 20°C at a rate of 10°C min−1. The second heating scan took place by heating the sample from 20°C to 220°C at a rate of 10°C min−1.
The energy required to melt the sample is obtained by estimating the area under the endothermic DSC curve. The area under the second heating curve from the onset to the offset temperature represents the measured enthalpy of fusion which is used to calculate the percentage crystallinity: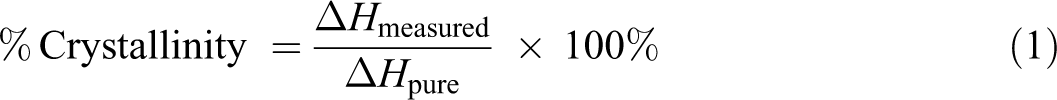
where Δ
Scanning electron microscope
Inspect (model F50, Canada) scanning electron microscope (SEM) was employed to examine the fractured surfaces of LDPE/MCC composites before and after burying in the soil. The signals created by utilizing this machine will analyze the information through the surface and provide data about the surface structure, external morphology, crystalline structure, dispersion of fibers in the matrix, the orientation of materials sample, and to recognize the presence of any defect.
Preparation of degradation testing samples
Degradation testing samples were prepared by a hot compression molding (Carver Auto Series Press, USA). Overhead projector sheets were used to prevent LDPE/MCC composites from sticking to the machine surface after molding. All samples were hot pressed for 8 min at 150°C except the composite with 30% cellulose content which was hot pressed at 180°C. A rectangular sample of 100 × 150 × 1.3 mm3 in dimensions was cut for each LDPE/MCC composite.
Degradation test
A soil burial test was carried out on a laboratory scale to examine LDPE/MCC degradability. The flowerpot containing the soil and samples were incubated at an almost constant temperature of 46°C and moisture content at 40–50% for 4 months. The 40–50% moisture content was found to be optimal for microbial activity. 20 Also, the flowerpots were covered with a plastic film to avoid water evaporation from the soil surface. Degradation was estimated by monitoring changes in weight as a function of burial time. The samples were removed from the soil every 20 days to calculate their weight loss. Debris on the specimens was removed by washing with water, and then the samples were dried in an oven at 100–105°C for 24 h. The dried samples were weighed using an electronic balance with a precision of 0.1 mg before they buried again in the soil. 1
Results and discussion
Thermal properties results
It is interesting to note in Figures 1 and 3 that the melting temperature (
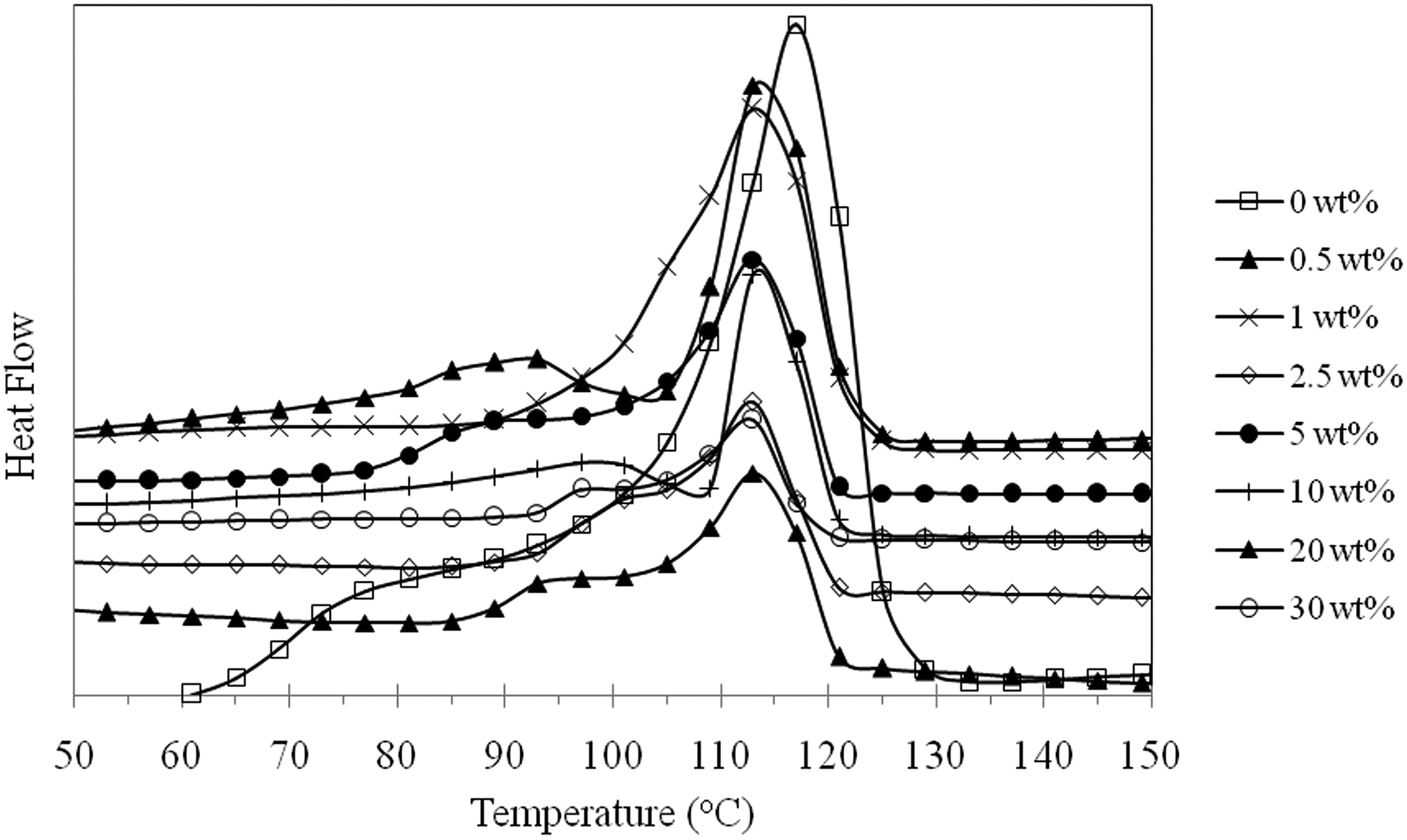
DSC melting endotherms for LDPE/MCC composites.
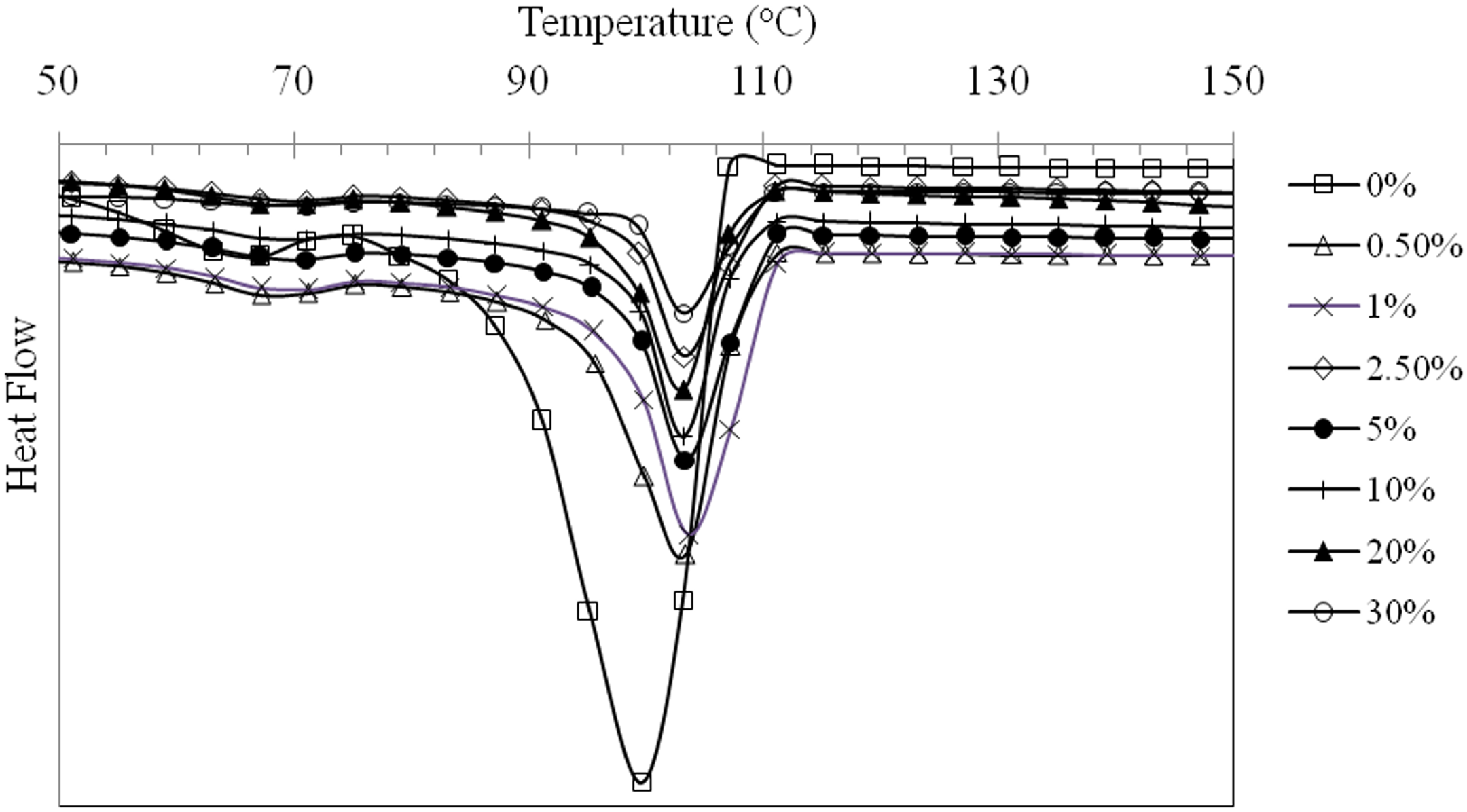
DSC crystallization exotherms for LDPE/MCC composites.
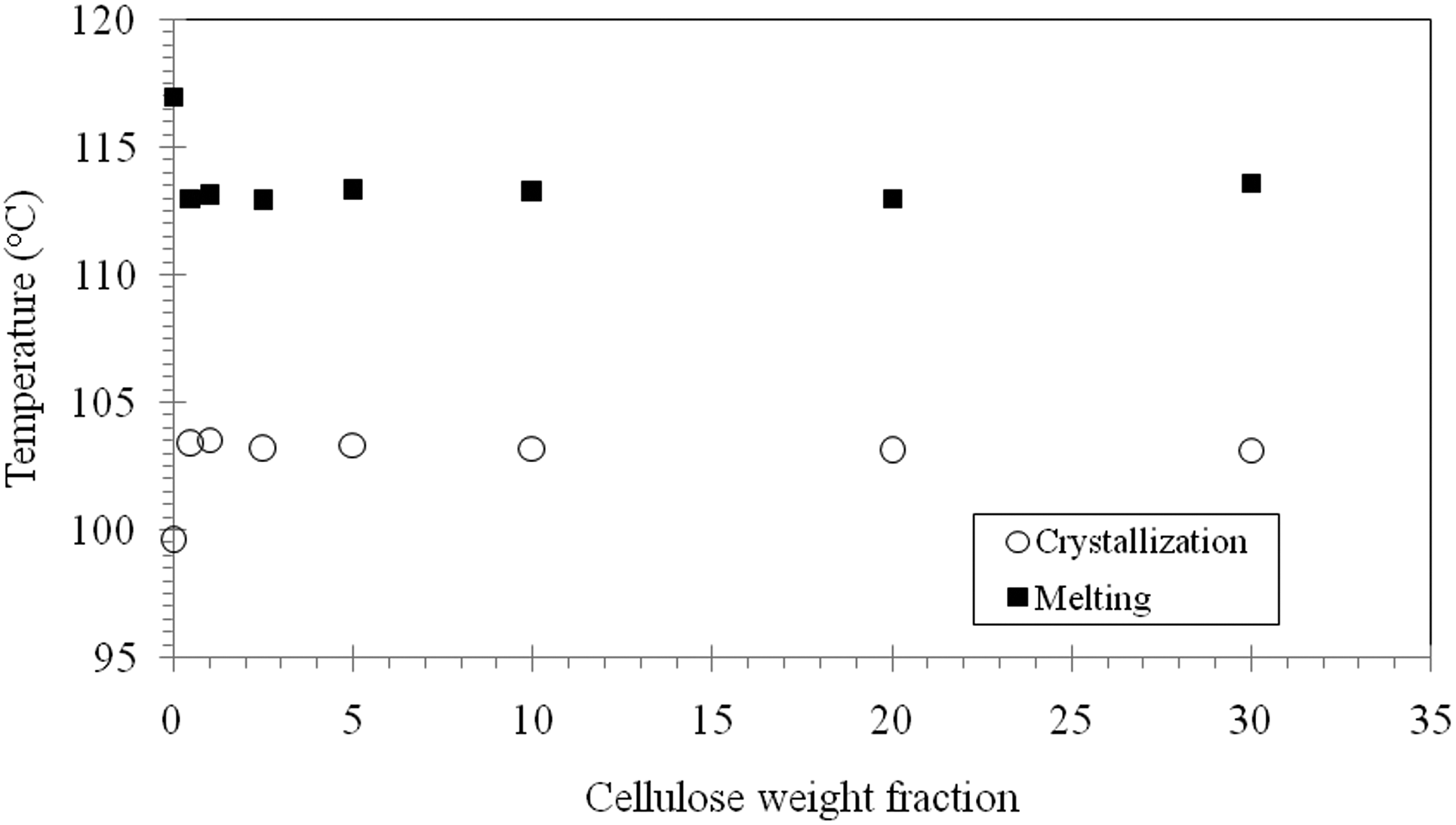
Melting and crystallization temperatures of LDPE/MCC composites.
The percentage crystallinity of LDPE composites as a function of MCC concentration is shown in Figure 4. The results reveal a sharp reduction in the percentage crystallinity from about 58% for pure LDPE to about 11% at a MCC concentration of 30 wt% within the matrix. This reduction in the percentage crystallinity agrees with what Šumigin et al. 23 and Sdrobis et al. 21 reported in their work.
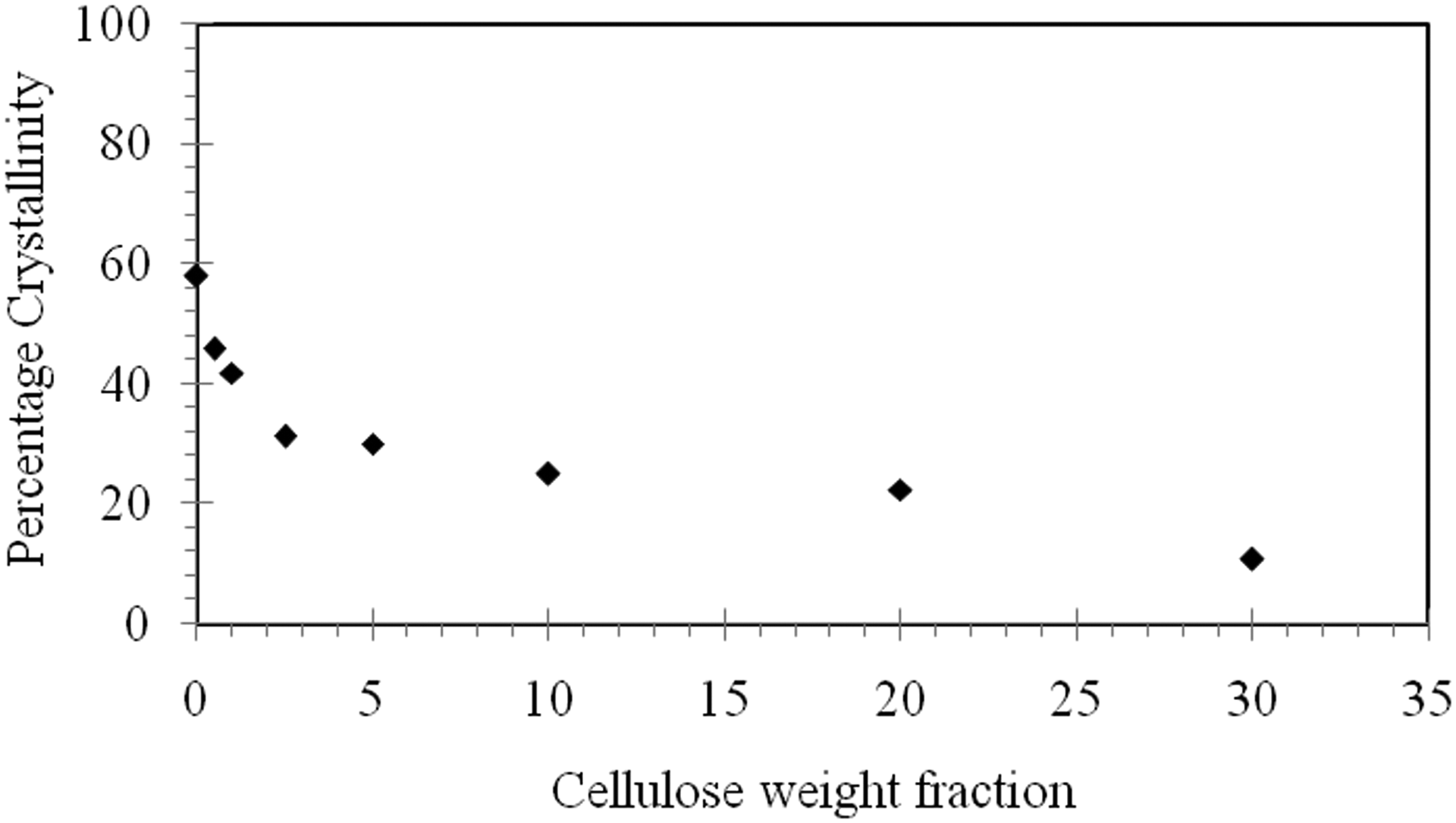
Percentage crystallinity of LDPE/MCC composites as a function of cellulose concentration.
Polarized light microscope images (Figure 5) show the distribution of MCC particles within LDPE without agglomeration even at high MCC concentration, the images were taken while the sample was in a molten state and before crystallization. Although MCC may increase the nucleation and speed up the growth of LDPE crystals, their presence restricts and prevents LDPE chains movement and migration which results in a weaker crystallization process as compared to the neat LDPE.

Polarized light microscope images showing the distribution of MCC particles within molten LDPE; (a) neat, (b) 0.5 wt% MCC, (c) 1 wt% MCC, (d) 2.5 wt% MCC, (e) 5 wt% MCC, (f) 10 wt% MCC, (g) 20 wt% MCC, and (h) 30 wt% MCC.
Degradation results
From Figure 6, it can be observed that the weight loss for virgin LPDE is relatively very small that did not exceed 0.07 g on day 120. This small weight loss is expected because LDPE shows almost resistance to microorganism attack because of the long carbon chains in the hydrophobic backbones which give high resistance to hydrolysis.
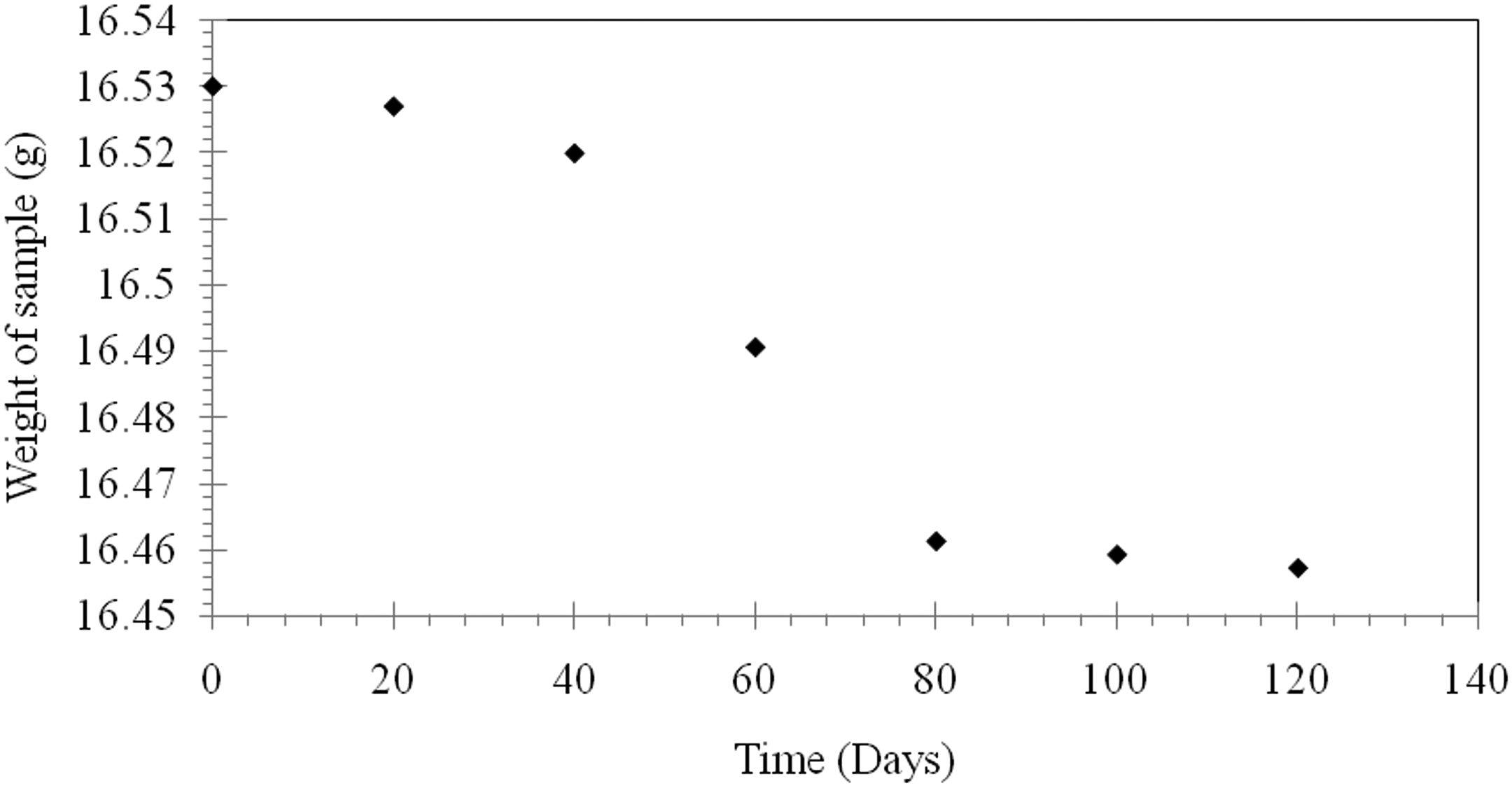
Weight of neat LDPE during burying in soil as a function of time.
Figure 7 shows that the weight loss increased slightly with the increase in cellulose content and it can be concluded that the higher the content of cellulose, the higher the weight loss over time with no clear trend. This reduction in weight can be attributed to the hydrophilic feature of cellulose which causes higher water absorption and leads to higher degradation of LDPE/cellulose. 2
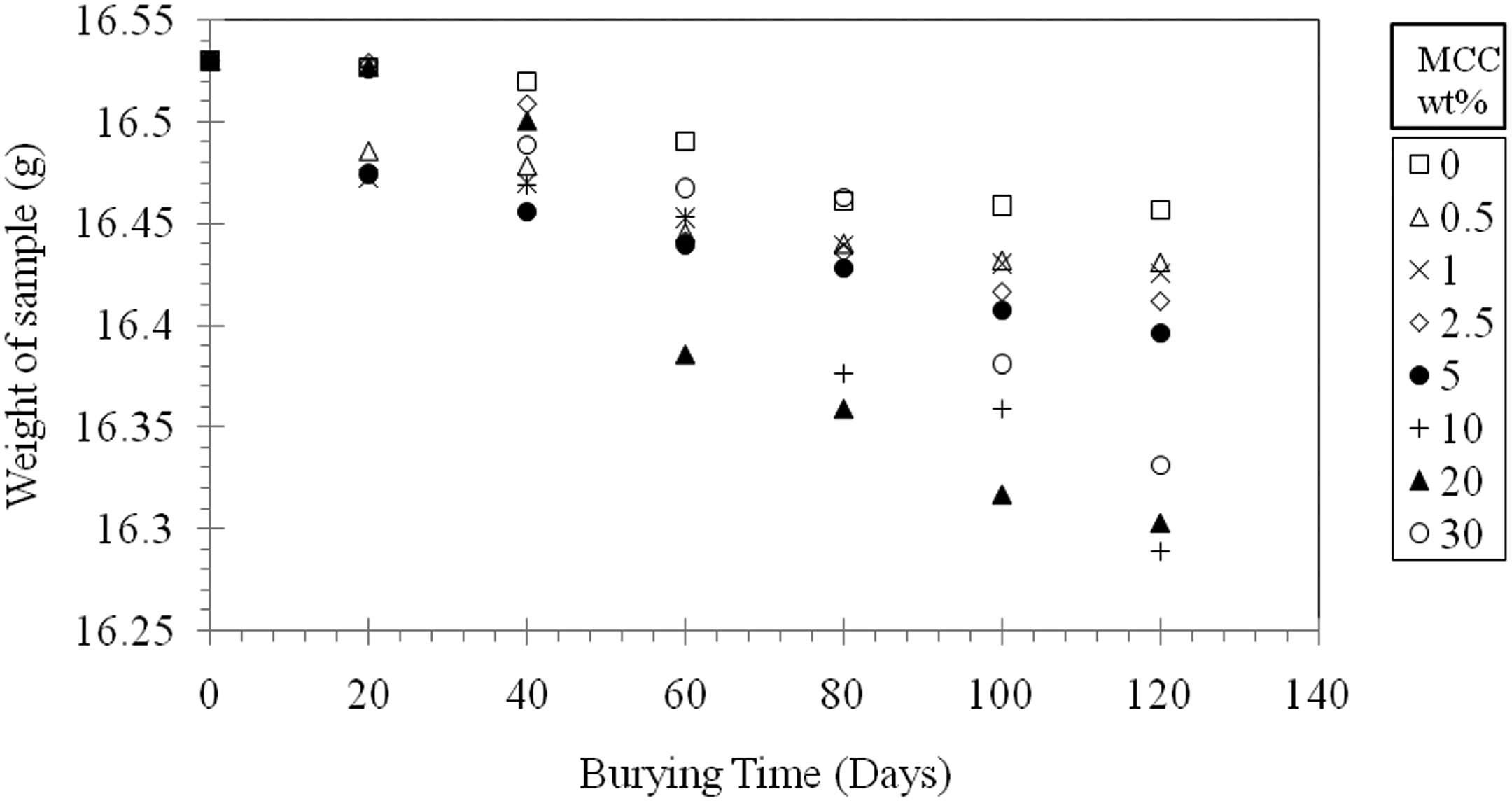
Weight of LDPE/MCC composites during burying in soil as a function of burying time. MCC: microcrystalline cellulose.
The maximum weight loss percentage achieved in the present study is about 1.5% as shown in Figure 8. This result is in a good agreement with what Tajeddin et al. 1 reported, the authors claimed that the weight loss is very small during this period of time and increased when cellulose content is higher than 30% and by adding PEG as a plasticizer. Also, the obtained result agrees with Okelo’s finding, where the author stated that the mass loss percentage for HDPE is 1.5% at a composition of 20% cellulose. On the other hand, Otieno et al. 25 mentioned in their publication that the biodegradation of recycled LDPE/cellulose composites depends strongly on cellulose content and it is much higher after 100 days at a composition of 20% cellulose and the weight loss was about 10%.
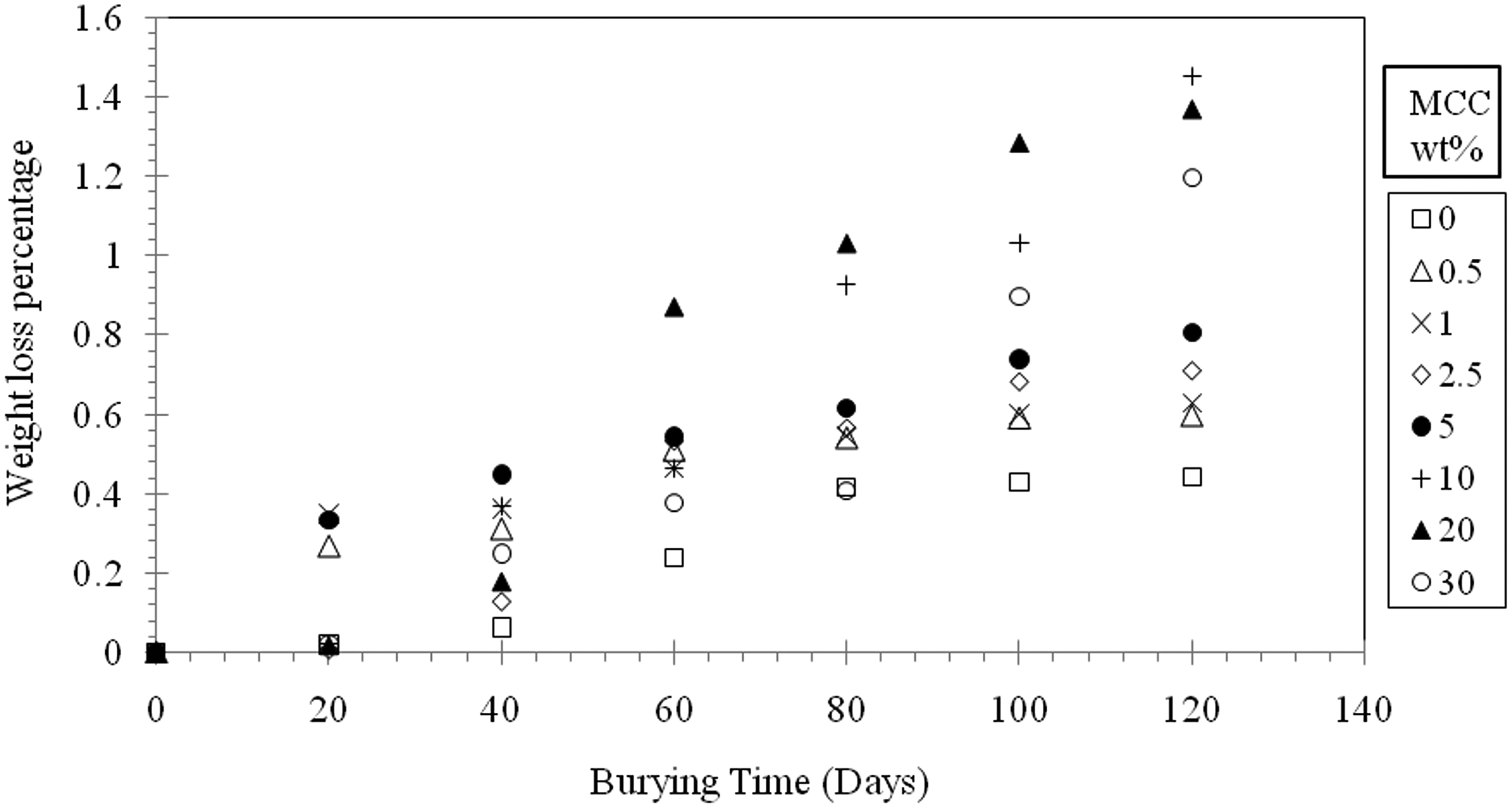
Weight loss percentage of LDPE/MCC composites during burying in soil as a function of burying time.
Figures 9 and 10 show the SEM images (at a magnification of 5000) of fractured surfaces for neat LDPE sample before and after burying in the soil. These images show that there are no critical contrasts between both specimens. SEM images of fractured surfaces for LDPE/30 wt% MCC composites before and after burying in the soil are presented in Figures 11 and 12.

SEM for fractured surface of neat LDPE before burying in soil with a magnification of 5000.

SEM for fractured surface of neat LDPE sample after burying in soil with a magnification of 5000.

SEM for fractured surface of LDPE/30 wt% MCC sample before burying in soil with a magnification of 5000.

SEM for fractured surface of LDPE/30 wt% MCC sample after burying in soil with a magnification of 5000.
The fractured surface of pure LDPE seems to be relatively smooth but the surface of LDPE/30 wt% MCC composite presents an uneven fracture surface with several hills and valleys that recommend a considerable matrix deformation. It can be seen that LDPE/MCC interface does not present homogeneity because of the different nature of the components hydrophobicity of LDPE and hydrophilicity of cellulose. 1 However, composite of LDPE with a composition of 30 wt% MCC is liable to degradation due to the presence of cellulose, an easily consumable ingredient for microorganism; thus, the marks of degradation are obviously noticeable. It is difficult to distinguish MCC particles from LDPE in the composite containing cellulose, which may suggest that the MCC particles are coated by the polymer. 26
Conclusions
Thermal analysis tests revealed that the addition of MCC to LDPE will cause the melting temperature of LDPE/MCC composites to decrease slightly from 117°C for the neat LDPE to about 113°C by the addition of about 1.0 wt% of MCC to the matrix. Increasing the weight fraction of MCC within LDPE matrix does not reduce the melting temperature further. An addition of about 1.0 weight fraction of MCC to LDPE matrix results in an increase in the crystallization temperature by about 4°C. Increasing the weight fraction of MCC beyond this concentration has no effect on the crystallization temperature. At its maximum weight fraction used in the present study (30 wt% MCC), the existence of MCC particles decreased the percentage crystallinity from about 58% for pure LDPE to about 11%, that is, over 80% reduction in the percentage crystallinity. Thermal characterization of the LDPE/MCC composites reveals that MCC particles play the rule of a nucleating agent. LDPE/MCC composites degradation tests were estimated by monitoring changes in weight as a function of burial time. Based on the results obtained in this work, it has been found that the addition of cellulose to LDPE composites has a little effect on degradation of LDPE. Over a period of 3 months, it is observed that the weight loss percentage did not exceed 1.5. The weight loss during the first 20 days is almost negligible for most LDPE/MCC composites, but then it increased slightly with the increase in cellulose content. SEM images show a relatively smooth fracture surface for neat LDPE in both cases, before and after burying in the soil. While on the other hand, an uneven fracture surface with several hills and valleys is shown for fracture surface of LDPE filled with 30 wt% MCC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
