Abstract
Mesoporous diatomite nanoplatelets were employed to prepare various poly (styrene-co-butyl acrylate)/diatomite nanocomposites by in situ reverse atom transfer radical polymerization of styrene and butyl acrylate. Fourier-transform infrared spectroscopy, thermogravimetric analysis (TGA), and nitrogen adsorption/desorption isotherm were employed for evaluating some properties of the pristine diatomite nanoplatelets. Evaluation of pore size distribution and morphological studies were also performed by scanning and transmission electron microscopy. Conversion and molecular weight determinations were carried out using gas and size exclusion chromatography, respectively. Addition of 3 wt% pristine mesoporous diatomite nanoplatelets leads to an increase in conversion from 77% to 92%. Molecular weight of poly (styrene-co-butyl acrylate) chains increases from 17,348 g mol−1 to 21,346 g mol−1 with the addition of 3 wt% pristine mesoporous diatomite nanoplatelets; however, polydispersity index values increases from 1.38 to 1.65. Increasing thermal stability of the nanocomposites is demonstrated by TGA. Differential scanning calorimetry shows an increase in glass transition temperature from 35.5°C to 39.4°C with the addition of 3 wt% mesoporous diatomite nanoplatelets.
Keywords
Introduction
Organic/inorganic nanocomposite materials have been extensively studied during the last decades. 1 –3 Among various nanocomposites, polymer-based (nano)composites have attracted great attention and are manufactured commercially for many diverse applications (e.g. sporting goods, aerospace components, and automobiles). 1,4 Polymer nanocomposites are two-phase materials in which the polymers are loaded with high-surface-area reinforcing fillers. 5 Polymer nanocomposites present unique features in comparison with the neat polymer matrix, because they combine the advantages of the inorganic material (e.g. rigidity and thermal stability) and the organic polymer (e.g. flexibility, dielectric, ductility, and processability). 6 Polymer nanocomposites often exhibit appropriate improvement in several properties when compared with neat polymer or conventional micro- and macro-composites. 7,8 Enhanced barrier characteristics, increased moduli and strengths, high heat distortion temperatures, decreased thermal expansion coefficients, reduced gas permeabilities, decreased absorption of organic liquids, and enhanced ionic conductivities are some of the enhanced properties of nanocomposites. 9,10 The methods for the preparation of nanocomposites include in situ polymerization and solution and melt intercalation procedure. 3,11
Diatomaceous earth or diatomite is a mineral silicate material, which is mainly composed of amorphous hydrated silica (SiO2·nH2O). Diatomite is classified as noncrystalline opal-A according to the mineralogical classification. Diatomite presents many unique physical and chemical characteristics such as highly developed mesoporosity and/or macroporosity, strong acid resistance, high mechanical strength, low thermal conductivity, large surface area, small particle size, and so on. 12 –16 Due to these attractive features, diatomite is employed in various applications such as filtering-utility material, filling material, insulation material, adsorbent, abrasive material and surface cleaning material, catalyst carrier, light construction material, and silica source. 17,18
Although free radical polymerization can be performed in a mild reaction condition, it also has some deficiencies (e.g. lack of control over molecular weight, polymer composition, and end functionality). 19 –22 Controlled living radical polymerization (CRP) methods can be used to prepare tailor-made polymers with predetermined molecular weight and low polydispersity index (PDI) values. 23 Atom transfer radical polymerization (ATRP), nitroxide-mediated polymerization, and reversible addition fragmentation chain transfer are the three famous CRP methods. 24 Polymerization of a wide variety of monomers, commercial availability of its reagents, and its application in different polymerization systems and media are some of the unique advantages of ATRP over other CRP methods. 25,26
A review of literatures indicates that there is no obvious research on the application of diatomite as a filler to synthesize polymer/diatomite composites. However, Karaman et al. have prepared polyethylene glycol (PEG)/diatomite composite as a novel form-stable composite phase change material (PCM) in which the PCM was prepared by incorporating PEG in the pores of diatomite. 27 Li et al. have synthesized conducting diatomite by polyaniline on the surface of diatomite. Linkage of polyaniline on the surface of diatomite is attributed to the hydrogen bond between the surface of diatomite and polyaniline macromolecules. 28 Li et al. have also prepared fibrillar polyaniline/diatomite composite by one-step in situ polymerization. According to their results, the polyaniline/diatomite composite can be applied as fillers for electromagnetic shielding materials and conductive coatings. 29 In addition, other studies such as investigating the effects of extrusion conditions on die-swell behavior of polypropylene/diatomite composite melts and crystallization behaviors and foaming properties of diatomite-filled polypropylene composites have been performed. 30,31
In this research, pristine diatomite nanoplatelets were employed to synthesize tailor-made random poly (styrene-co-butyl acrylate)/diatomite nanocomposites. ATRP was employed to synthesize tailor-made random poly (styrene-co-butyl acrylate) matrices. Among different initiation techniques of ATRP, reverse ATRP (RATRP) was employed to prepare well-defined copolymer matrices. The effect of pristine diatomite nanoplatelets on conversion, molecular weights, and PDI values of the synthesized nanocomposites by in situ RATRP and thermal properties of the products are discussed.
Experimental
Materials
Diatomite earth sample was obtained from Kamel Abad-Azerbaijan-IR., Iran. It was dispersed in 100-mL of distilled water by magnetic stirring and then it was kept constant until some solid impurities were dispersed. The particles were separated with filter paper and dried at 100°C for 8 h. Styrene (Aldrich, Switzerland, 99%) and butyl acrylate (Merk, Germany, 99%) were passed through a basic alumina-filled column and dried over calcium hydride. Copper(II) bromide (CuBr2; Fluka, Switzerland, 99%), N,N,N′,N′′,N′′-pentamethyldiethylenetriamine (PMDETA; Aldrich, Switzerland, 99%), 2,2′-azobisisobutyronitrile (AIBN; Acros, Belgium), anisole (Aldrich, Switzerland, 99%), tetrahydrofuran (THF; Merck, Germany, 99%), and neutral aluminum oxide (Aldrich, Switzerland, 99%) were used as received.
Preparation of neat random poly (styrene-co-butyl acrylate) and its nanocomposites via in situ RATRP
Reverse atom transfer radical random copolymerization of St and BA was performed in a 150-mL lab reactor which was equipped with a reflux condenser, nitrogen inlet valve, and a magnetic stir bar that was placed in an oil bath thermostated at desired temperature. Typical batch of copolymerization was run at 100°C with the molar ratio of 200:1:1:1 for [M]:[AIBN]:[CuBr2]:[PMDETA], giving a theoretical molecular weight of 23,232 g mol−1 at 100% conversion. At first, St (8.34 mL), BA (8.33 mL and 0.05 mol), anisole (10 mL), CuBr2 (0.19 g and 0.87 mmol), and PMDETA (0.18 mL and 0.87 mmol) were added into the reactor and the reactor was degassed and back-filled with nitrogen three times and stirring was continued at room temperature. The solution turned green color, since the CuBr2/PMDETA complex was formed. When the majority of the metal complex had formed, the reaction temperature was increased to 100°C for 5 min. Subsequently, AIBN (0.14 g and 0.87 mmol) as a monomer soluble radical initiator (dissolved in 5-mL St) was injected into the reactor to start the copolymerization. After 10 h, the copolymerization process was stopped by opening the reactor and exposing the catalyst to air. For preparation of nanocomposites, a desired amount of pristine diatomite nanoplatelets was dispersed in 8.34 mL of St and the mixture was stirred for 22 h. Subsequently, the copolymerization procedure was applied accordingly. Designation of the samples with the percentage of pristine diatomite nanoplatelets is given in Table 1.
Designation of the samples.

RATRP: reverse atom transfer radical polymerization; NPSB: neat poly (styrene-co-butyl acrylate).
Separation of copolymer chains from pristine diatomite nanoplatelets and catalyst removal
For the separation of poly (styrene-co-butyl acrylate) chains from pristine diatomite particles, the nanocomposites were dissolved in THF. With high-speed ultracentrifugation (10,000 r min−1) and then passing the solution through a 0.2-μm filter, poly (styrene-co-butyl acrylate) chains were separated from pristine diatomite particles. Subsequently, copolymer solutions were passed through an alumina column to remove catalyst species.
Characterization
Fourier-transform infrared (FTIR) spectrum of the pristine diatomite was achieved using FTIR spectroscopy on a Bruker FTIR spectrophotometer (Germany), within a range of 400–4400 cm−1. The porosity of materials was characterized by nitrogen adsorption/desorption curves obtained with a Quntasurb QS18 (Quntachrom) apparatus (USA). The surface area and pore size distribution values were obtained with the corrected Brunauer–Emmett–Teller (BET) equation. In addition, specific surface area measurements were also performed with chemisorption analyzer (TPR, TPD, and TPO) and BET surface area (Toseye Hesgarsazan Asia Co., Iran). Surface morphology of the pristine diatomite was examined by scanning electron microscope (SEM) (Philips XL30) with an accelerating voltage of 20 kV. The transmission electron microscope, Philips EM 208 (The Netherlands), with an accelerating voltage of 120 kV was employed to study the morphology of the pristine diatomite sample. Gas chromatography (GC) is a simple and highly sensitive characterization method and does not require removal of the metal catalyst particles. GC was performed on an Agilent-6890 N (USA) with a split/splitless injector and flame ionization detector, using a 60-m HP-INNOWAX capillary column (USA) for the separation. The GC temperature profile included an initial steady heating at 60°C for 10 min and a 10°C min−1 ramp from 60°C to 160°C. The samples were also diluted with acetone. The ratio of monomer to anisole was measured by GC to calculate monomer conversion throughout the reaction. Size exclusion chromatography (SEC) was used to measure the molecular weight and molecular weight distribution. A Waters 2000 ALLIANCE with a set of three columns of pore sizes 10,000, 1000, and 500 Å was utilized to determine polymer average molecular weight and PDI. THF was used as the eluent at a flow rate of 1.0 mL min−1, and calibration was carried out using low polydispersity polystyrene standards. Proton nuclear magnetic resonance ( 1 H NMR) spectra were recorded on a Bruker 300-MHz 1H NMR instrument with deuterated chloroform as the solvent and tetramethylsilane as the internal standard. Thermogravimetric analysis (TGA) was carried out using a PL thermogravimetric analyzer (TGA 1000; Polymer Laboratories, UK). Thermograms were obtained from ambient temperature to 700°C at a heating rate of 10°C min−1. Thermal analysis was carried out using a differential scanning calorimetry (DSC) instrument (NETZSCH DSC 200 F3; Netzsch Co., Selb/Bavaria, Germany). Nitrogen at a rate of 50 mL min−1 was used as purging gas. Aluminum pans containing 2–3 mg of the samples were sealed using DSC sample press. The samples were heated from ambient temperature to 225°C at a heating rate of 10°C min−1.
Results and discussion
Kinetics of in situ polymerization and nanocomposite properties can be affected by the characteristics of the nano-filler, and therefore, the characterization of unique features of the pristine diatomite nanoplatelets is the first step for the preparation of poly (styrene-co-butyl acrylate)-based nanocomposites. 32
FTIR spectrum of the pristine diatomite nanoplatelets is shown in Figure 1. The peaks at 3434 and 1634 cm−1 correspond to the stretching vibrations of physically adsorbed water and zeolitic water, respectively. 33 Although the diatomite sample is rehydrated, during the preparation process and obtaining the spectrum, some water molecules may be readsorbed. 34 The strong peak at 1098 cm−1 is attributed to the stretching mode of siloxane (Si–O–Si). In addition, the peak at 471 cm−1 is associated with the asymmetric stretching mode of siloxane bonds. The peak at 796 cm−1 is also attributed to the vibration of O–H. 35,36
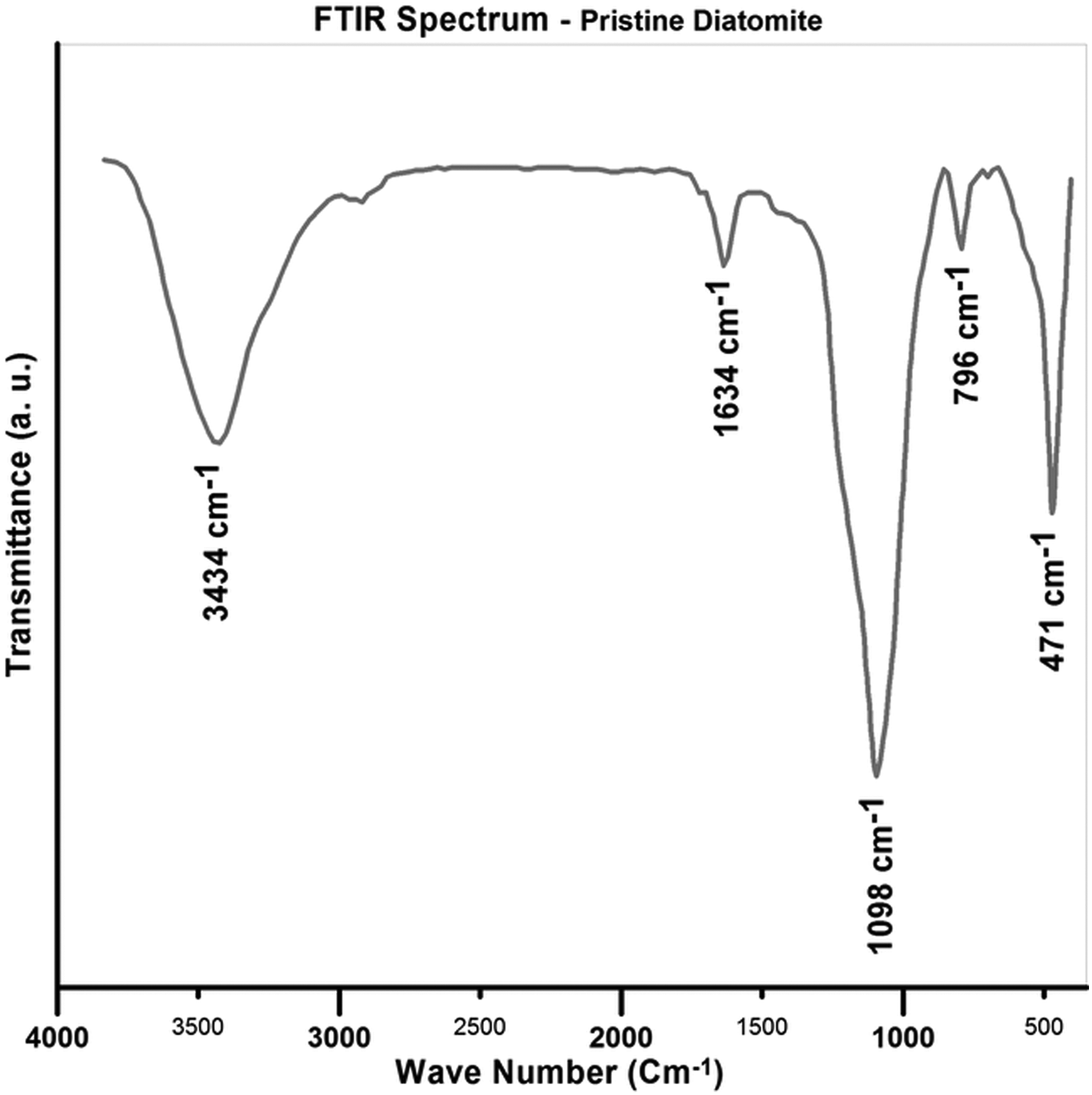
FTIR spectrum of the pristine diatomite nanoplatelets. FTIR: Fourier-transform infrared.
Nitrogen adsorption/desorption isotherms of the pristine diatomite nanoplatelets are presented in Figure 2. The shape of isotherm is similar to the type-IV isotherms according to the International Union of Pure and Applied Chemistry classification and confirms that diatomite has mesoporous structure. 37,38 The hysteresis is associated with the filling and emptying of the mesopores by capillary condensation. 39 A sharp increase in the nitrogen adsorbed quantity near the relative pressure of 1 demonstrates the existence of macropores in the pure diatomite and therefore a nonuniform pore size distribution can be comprehended. 40
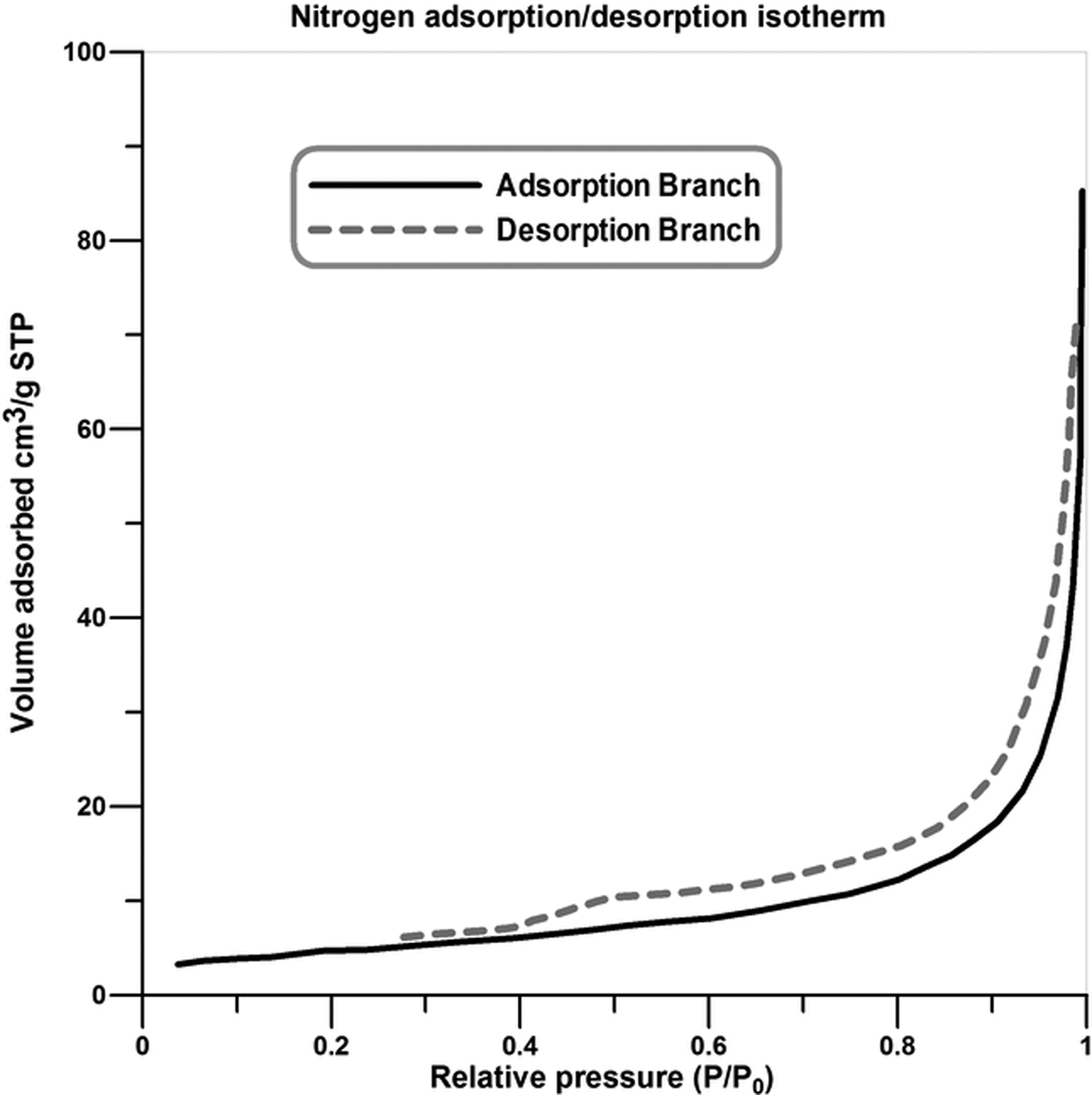
Nitrogen adsorption/desorption isotherm of the pristine diatomite nanoplatelets.
According to the extracted data from the nitrogen adsorption/desorption isotherms, the surface area of the pristine diatomite particles is calculated as 17.984 m2 g−1. Also, the average pore diameter is estimated to be around 27.42 nm.
The TGA curve of the pristine diatomite sample is illustrated in Figure 3. As it can be seen, two main mass losses are resolved in the TGA curve; the first mass loss is mainly ascribed to the dehydration of diatomite and the second weight loss may be associated with the dehydroxylation of the silanols of diatomite nanoplatelets.
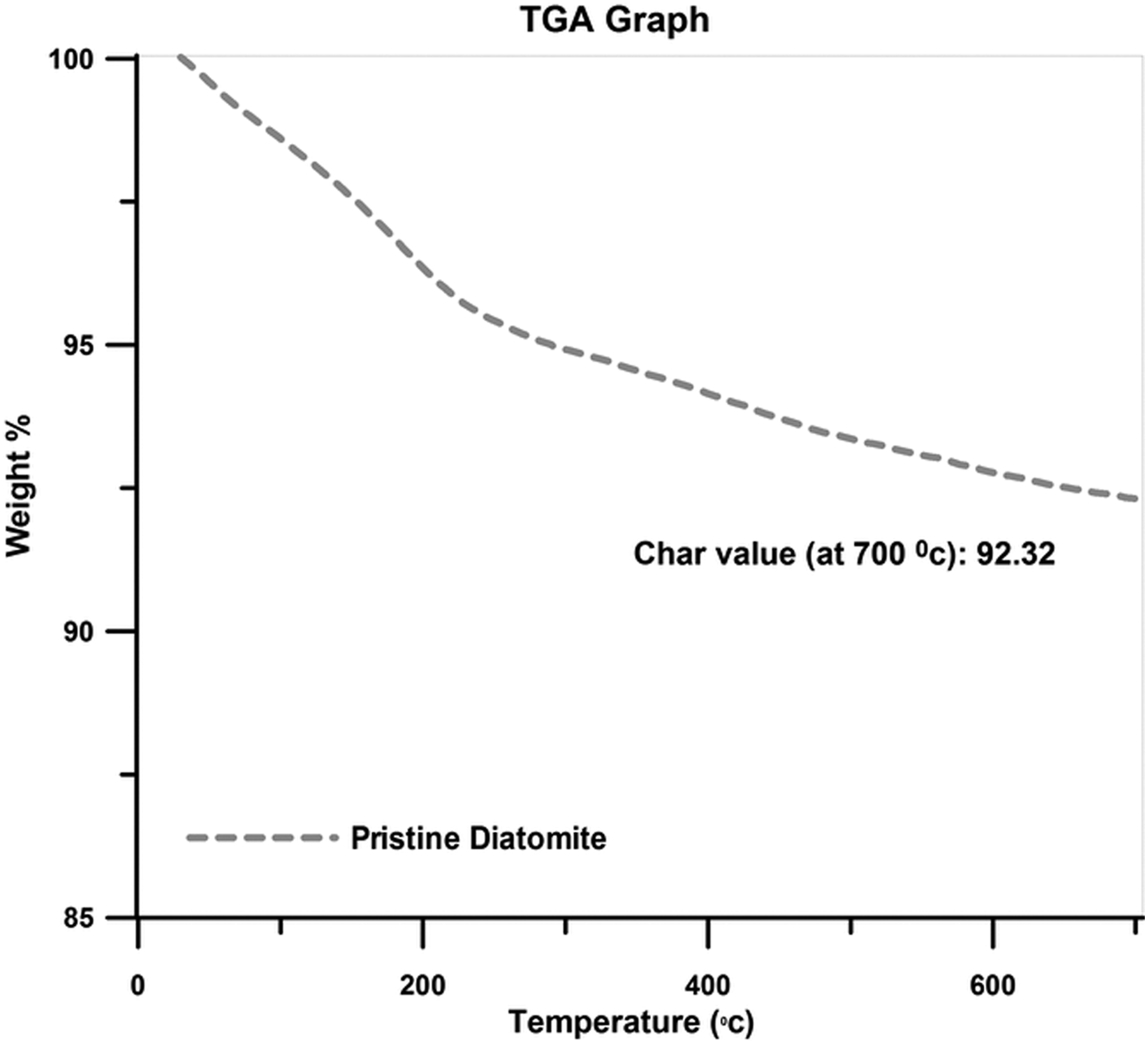
TGA graph of the pristine diatomite nanoplatelets.
The SEM image of the pristine diatomite nanoplatelets is exhibited in Figure 4. As it can be seen, the pristine diatomite sample is composed of plaque plate particles with spherical-shaped pores. These plates have regular pores and sometimes are aggregated.

SEM image of the pristine diatomite nanoplatelets.
The transmission electron microscopic (TEM) image of the pristine diatomite platelets is represented in Figure 5. According to this image, the pristine diatomite sample contains numerous regularly spaced rows of pores in its structure, and this observation is confirmed with SEM images. In addition, the average pore diameter from TEM images is estimated to be between 30 nm and 40 nm.
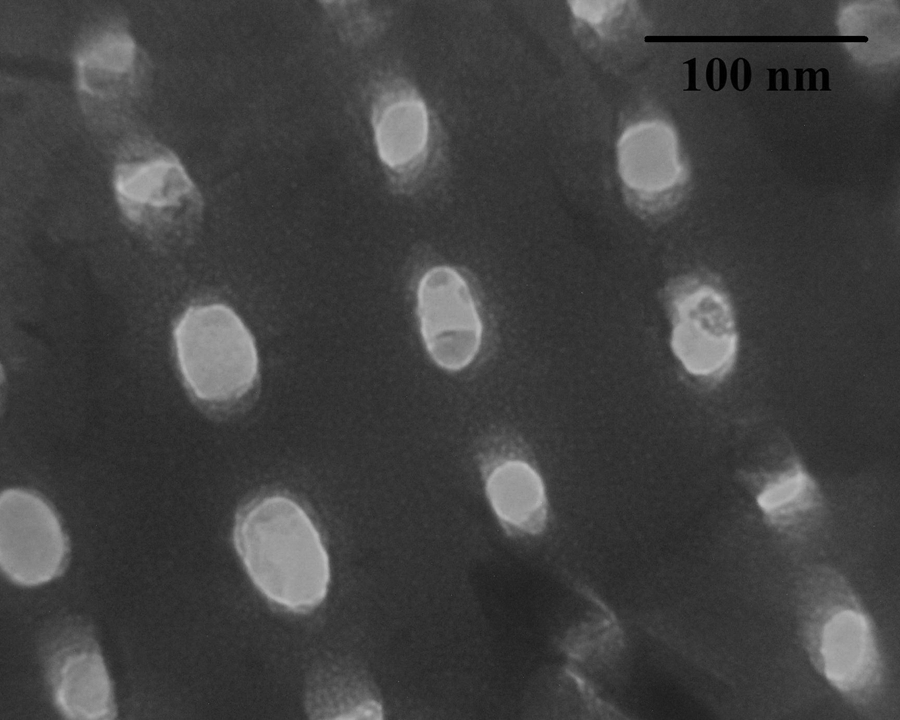
TEM image of the pristine diatomite nanoplatelets. TEM: transmission electron microscopy.
RATRP is a suitable method for circumventing the oxidation problems of normal ATRP, since reverse initiation technique employs oxidatively stable form of copper (Cu) species (CuBr2). Thus, reactants in this initiation process can be easily prepared, and therefore, synthetic procedure can be facilitated. 41,42 The general mechanism of RATRP is illustrated in Figure 6.
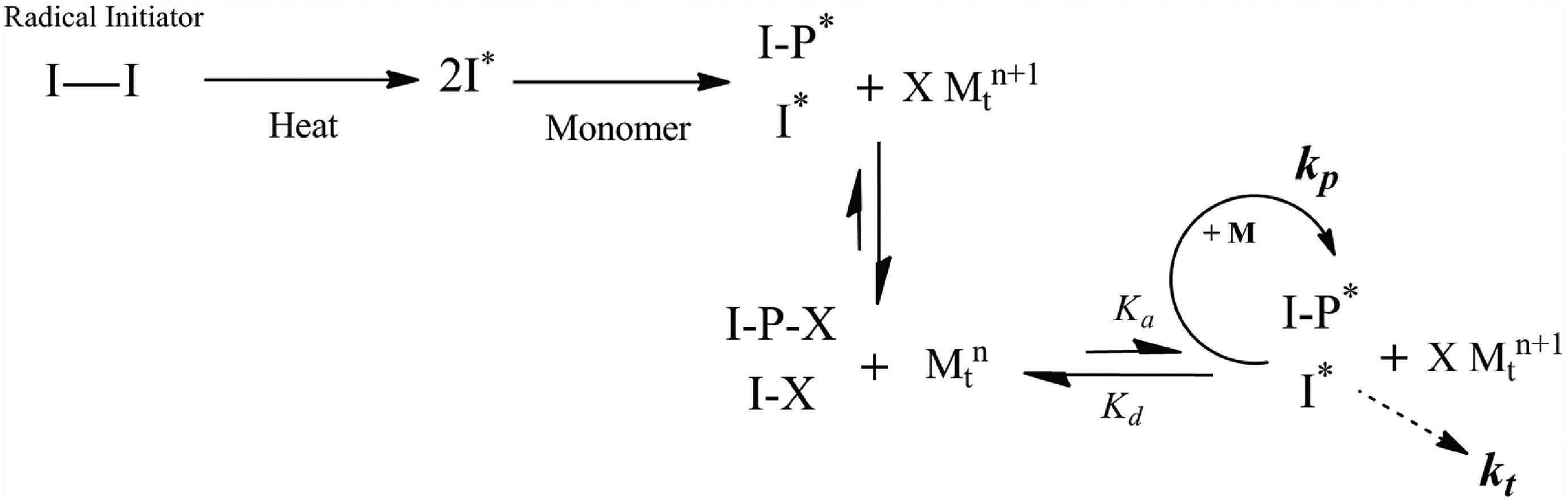
The general mechanism for RATRP. RATRP: reverse atom transfer radical polymerization.
The general procedure for the synthesis of tailor-made random poly (styrene-co-butyl acrylate) chains via RATRP in the presence of the pristine diatomite nanoplatelets is illustrated in Figure 7.
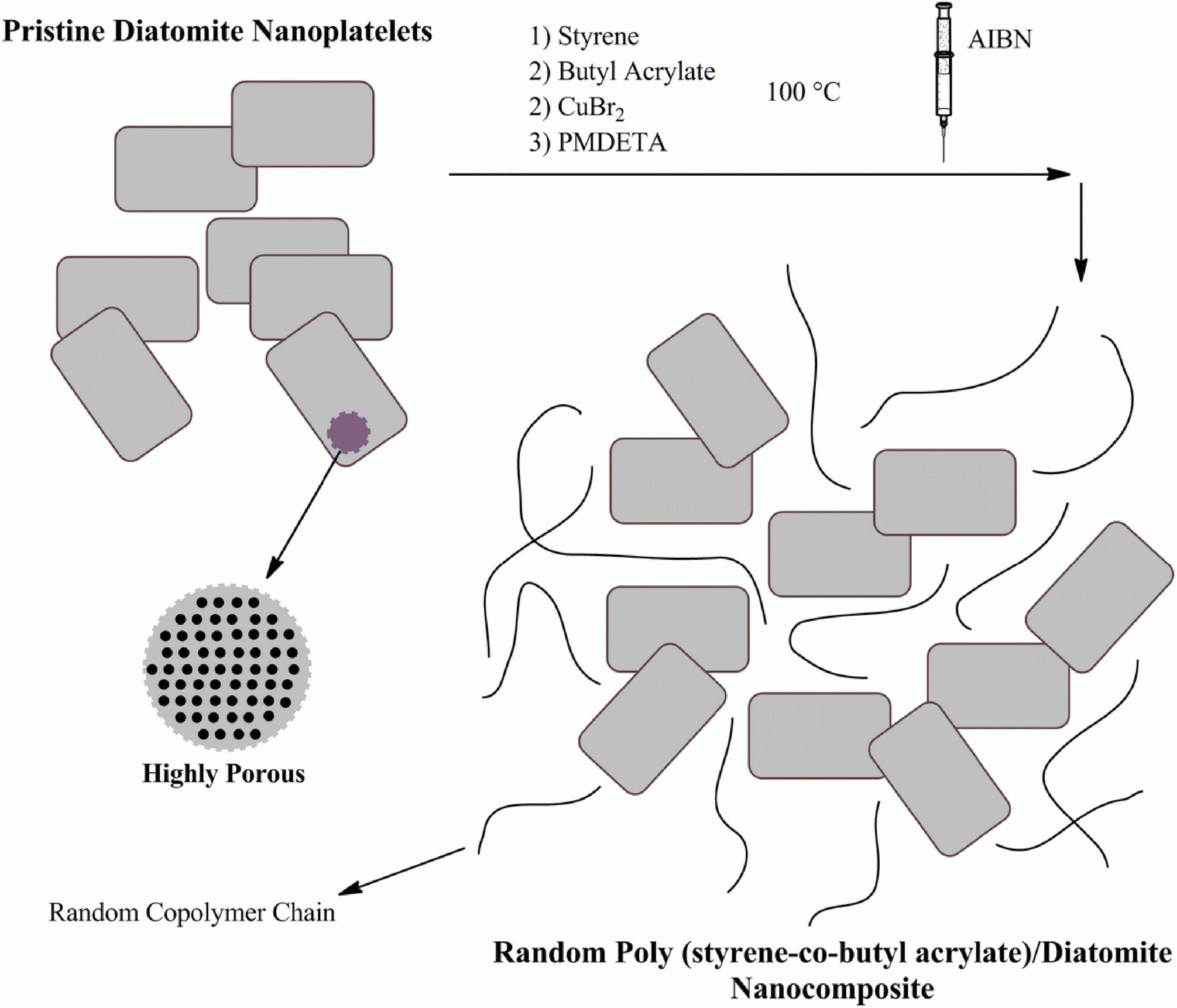
The general procedure for the synthesis of poly (styrene-co-butyl acrylate)/diatomite nanocomposites via in situ RATRP. RATRP: reverse atom transfer radical polymerization.
Figure 8 presents the SEC traces of NPSB and its nanocomposites. According to the results, NPSB and all the nanocomposites display monomodal peaks corresponding to the molecular weight values predetermined by the molar ratio of monomer to initiator. NPSB reveals narrow distribution and low PDI value.
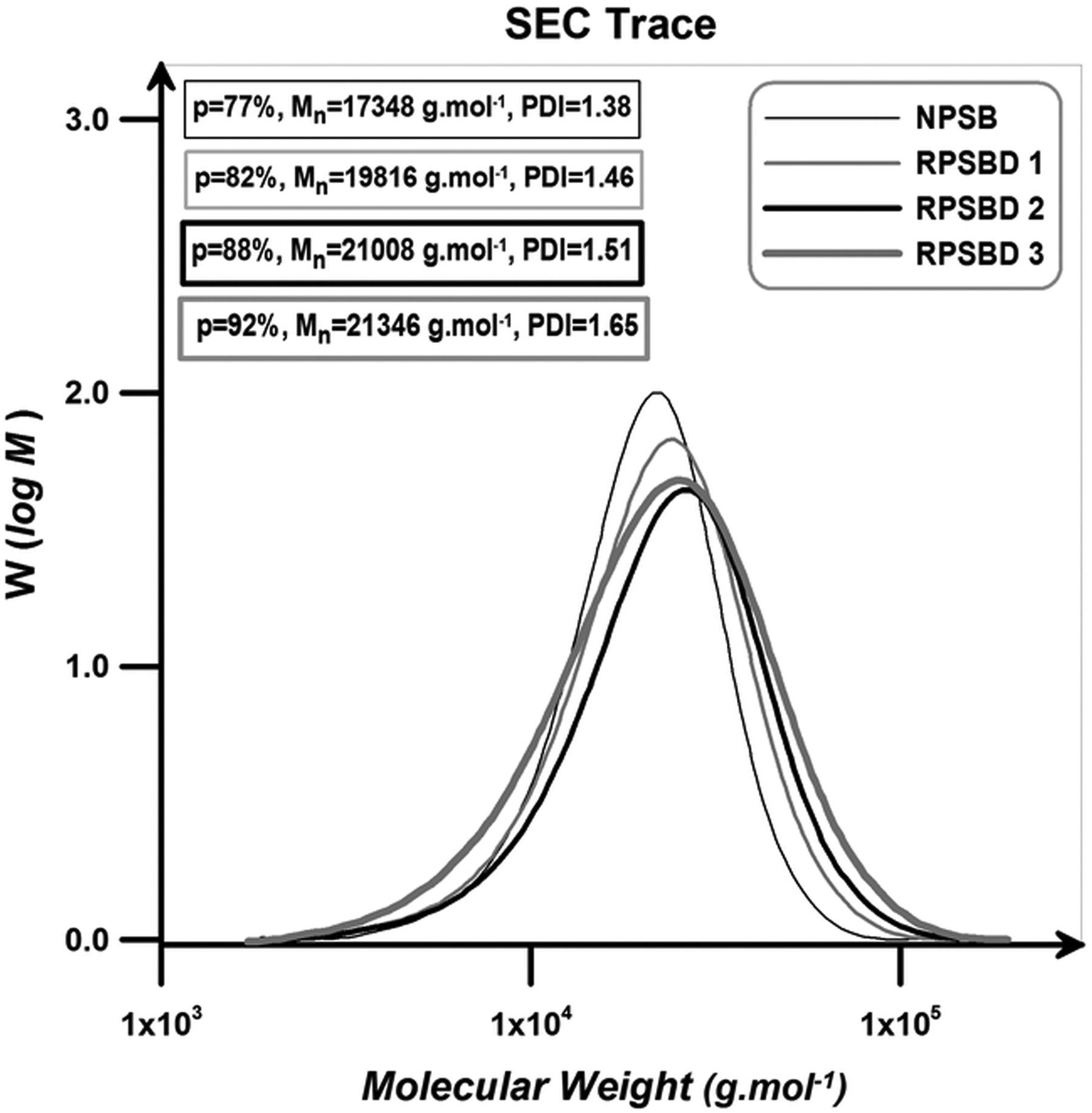
SEC traces of the NPSB and its different nanocomposites. NPSB: neat poly (styrene-co-butyl acrylate); SEC: size exclusion chromatography.
According to the results, RATRP of St and BA without diatomite nanoplatelets results in well-defined poly (styrene-co-butyl acrylate) chains with low PDI value. With the addition of 3 wt% pristine diatomite nanoplatelets, final conversion increases from 77% to 92%. The positive effect of the pristine diatomite nanoplatelets on the molecular weight of the samples is attributed to the pendant hydroxyl groups of the pristine diatomite. Polar solvents exert a rate acceleration effect on the polymerization systems for increasing radical activation rate and also reducing radical recombination rate. In addition, negatively charged surface could absorb and gather positively charged catalyst (Cu ions at our work) and consequently enhances the chain growth rate. The accelerating effect of other nano-fillers such as nanoclay and MCM-41 nanoparticles on the polymerization rate was also reported. 43 –45 PDI values of the copolymer chains increase with the addition of pristine diatomite loading. Pristine diatomite nanoplatelets act as an impurity in the polymerization medium and therefore cause the molecular weight distribution of the resultant copolymers to be increased; PDI value increases from 1.38 to 1.65 with 3 wt% loading of pristine diatomite. 46,47 Table 2 summarized the extracted data from the SEC traces of the NPSB and its different nanocomposites.
Molecular weights and PDI values of the extracted copolymers from SEC traces.
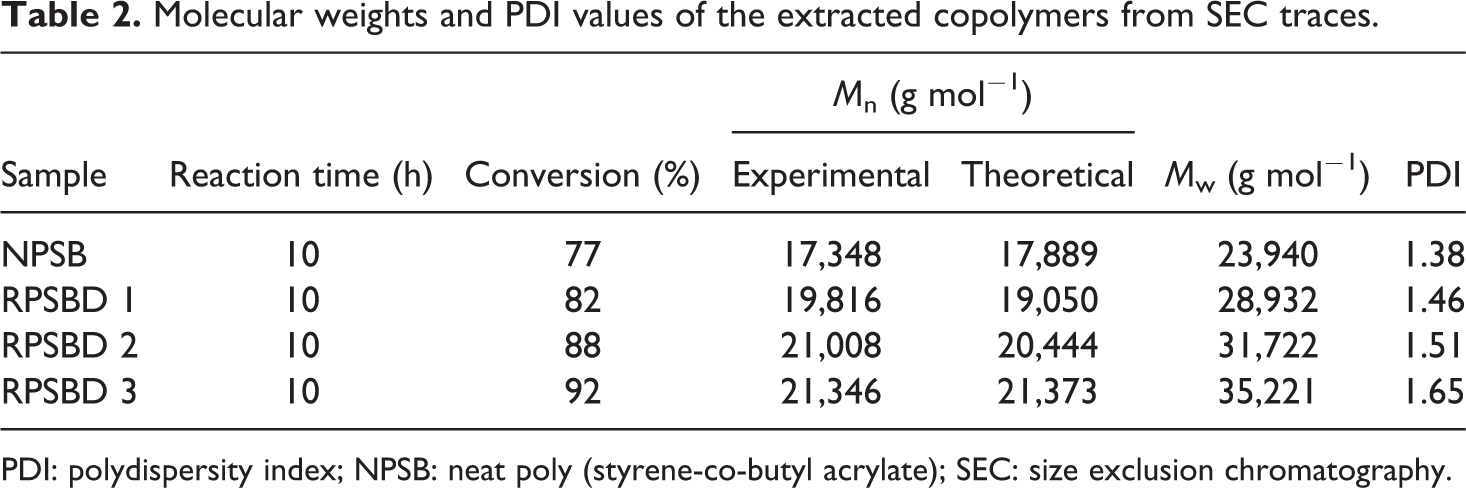
PDI: polydispersity index; NPSB: neat poly (styrene-co-butyl acrylate); SEC: size exclusion chromatography.
Figure 9 represents the 1H NMR spectra of the NPSB and its various nanocomposites.
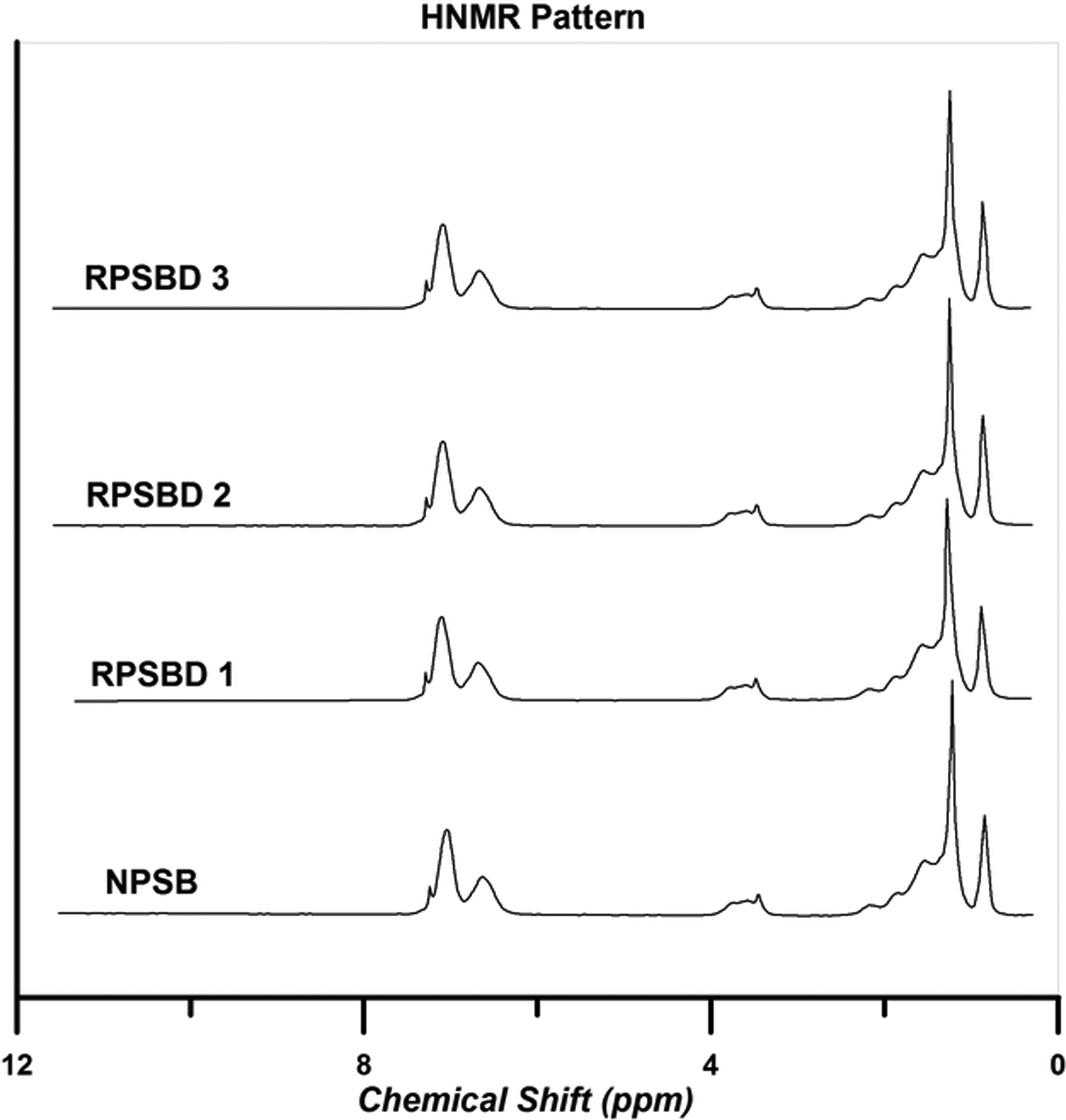
1H NMR spectra of the NPSB and its different nanocomposites. NPSB: neat poly (styrene-co-butyl acrylate); 1H NMR: proton nuclear magnetic resonance.
The molar ratio of each monomer in the copolymer chains can be determined by integrating aromatic peaks area (S Ph, 6.6–7.4 ppm, 5 H) which corresponds to the phenyl ring of St and methylene near to the ester group of BA (SM , 3.6–4.2 ppm, 2 H) by using the following
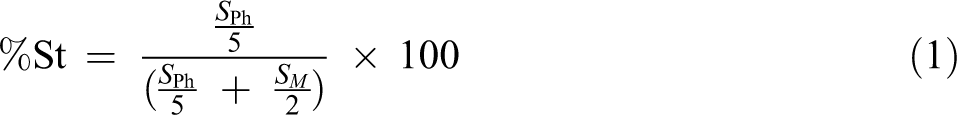
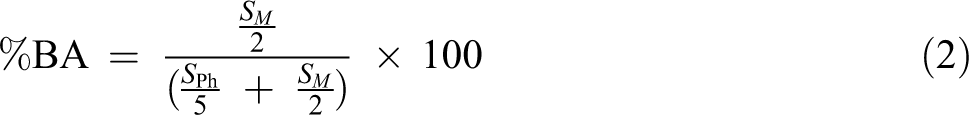
Extracted data from 1H NMR spectroscopy analysis is summarized in Table 3. According to the results, the molar ratio of each monomer in all the samples is approximately similar to the initial selected mole ratio of the monomers.
Extracted data from 1H NMR spectroscopy analysis of the pure poly (styrene-co-butyl acrylate) and its nanocomposites.
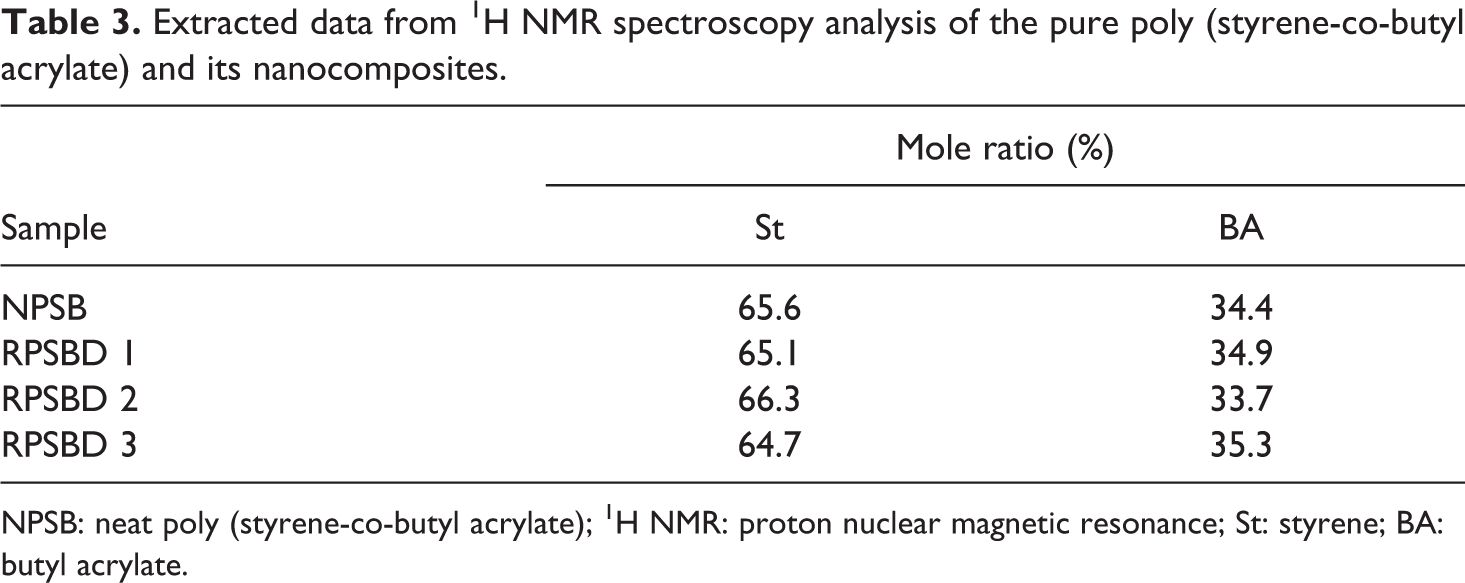
NPSB: neat poly (styrene-co-butyl acrylate); 1H NMR: proton nuclear magnetic resonance; St: styrene; BA: butyl acrylate.
Figure 10 presents the TGA thermograms of the NPSB and its different nanocomposites (weight loss as a function of temperature in the temperature window of 30–700°C).
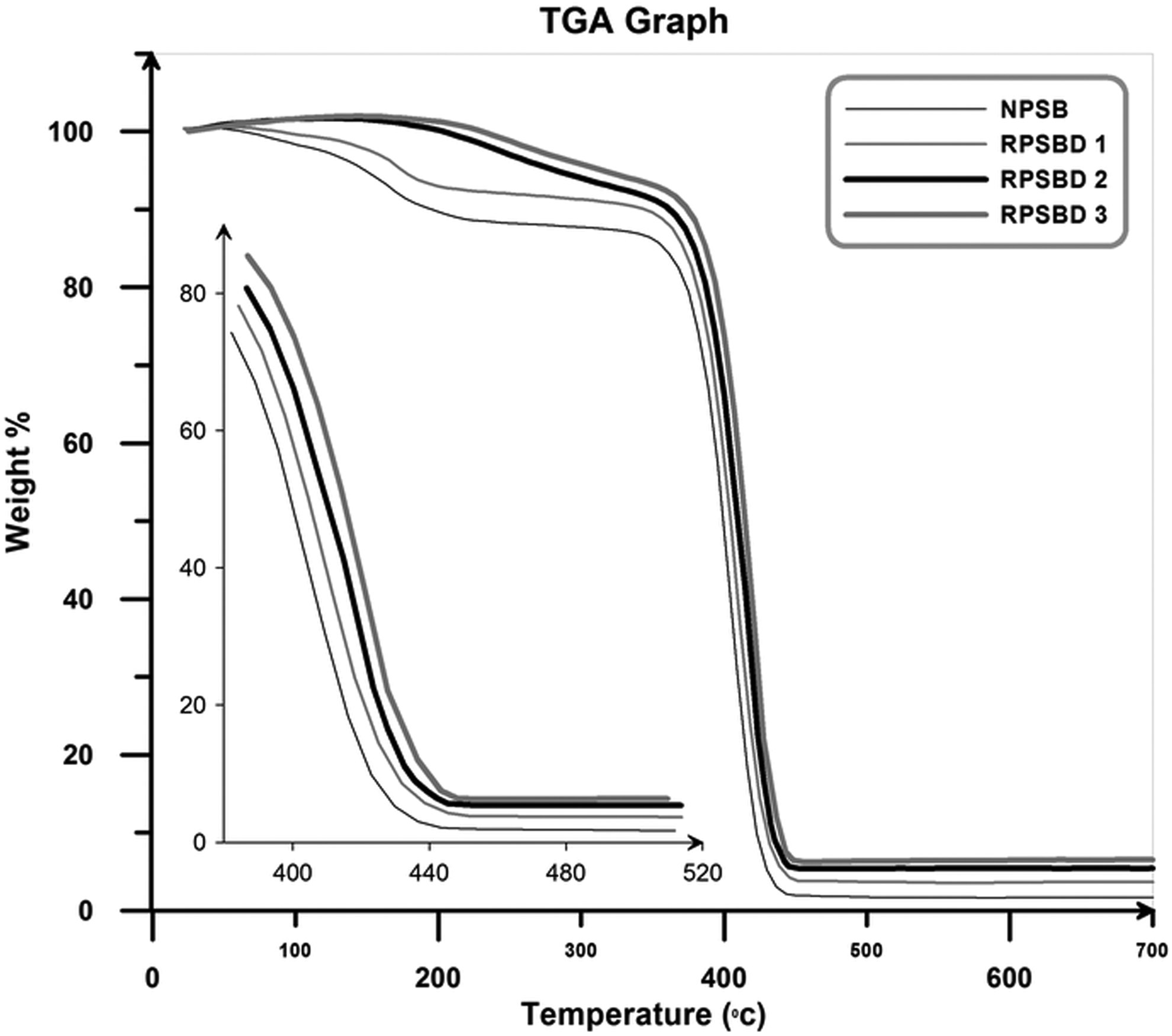
TGA thermograms of the NPSB and its different nanocomposites. NPSB: neat poly (styrene-co-butyl acrylate).
According to Figure 10, the thermal stability of the NPSB is lower than all of the nanocomposites. In addition, the thermal stability of the NPSB improves with the addition of pristine diatomite nanoplatelets, and by increasing the diatomite content, a rise in degradation temperatures was observed. In general, by increasing the temperature in the TGA graphs, three separate steps can be identified: (i) evaporation of the water molecules (at the temperature range of 100–150°C); (ii) degradation of volatile materials such as residual monomer and low molecular weight oligomers (at the temperature window of around 180–350°C); and (iii) degradation of the synthesized copolymer and nanocomposites (at the temperatures above 380°C). The extracted data from TGA graphs are graphically illustrated in Figure 11. The degradation temperature of the samples versus the amount of degradation shows that the addition of diatomite in the poly (styrene-co-butyl acrylate) matrix results in an improvement of thermal stabilities of the nanocomposites (T X: temperature threshold at which X% of NPSB and its nanocomposites is degraded). Char values for the NPSB and its nanocomposites at 650°C are given in Table 4. As it is expected, the char values increase with an increase in the pristine diatomite content.
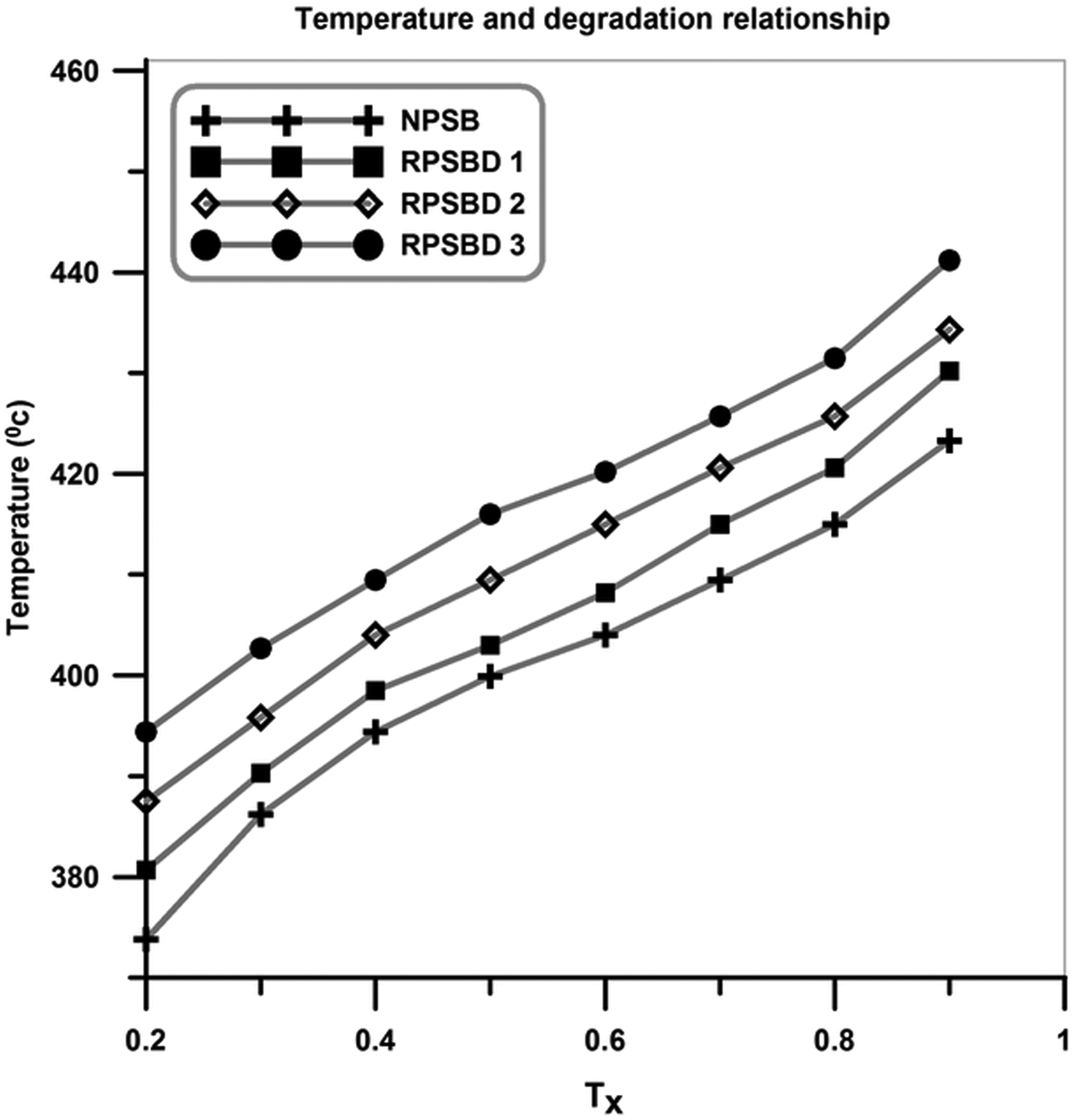
Graphical illustration of temperature and degradation relationship.
Char values of the NPSB and its composites (at 650°C).

NPSB: neat poly (styrene-co-butyl acrylate).
A rise in the degradation temperature of the nanocomposites with the addition of pristine diatomite nanoplatelets content is attributed to the high thermal stability of diatomite nanoplatelets and also interaction between diatomite nanoplatelets and copolymer matrix. 48,49 Additionally, the hindrance effect of the pristine diatomite nanoplatelets on the copolymer chains movement and restriction of oxygen permeation by these sheets are the other reasons for high thermal stability of the nanocomposites. Similar conclusions are also achieved in the case of polymer/clay nanocomposites. 45,50
Figure 12 displays the DSC thermograms of the NPSB and its different nanocomposites. Pristine diatomite nanoplatelets do not bear any transitions in this range of temperature, therefore only thermal transition of copolymers is observed. In these experiments, samples are heated from room temperature to 225°C. Then, they are cooled to room temperature and again heated from room temperature to 225°C to obtain T g values.
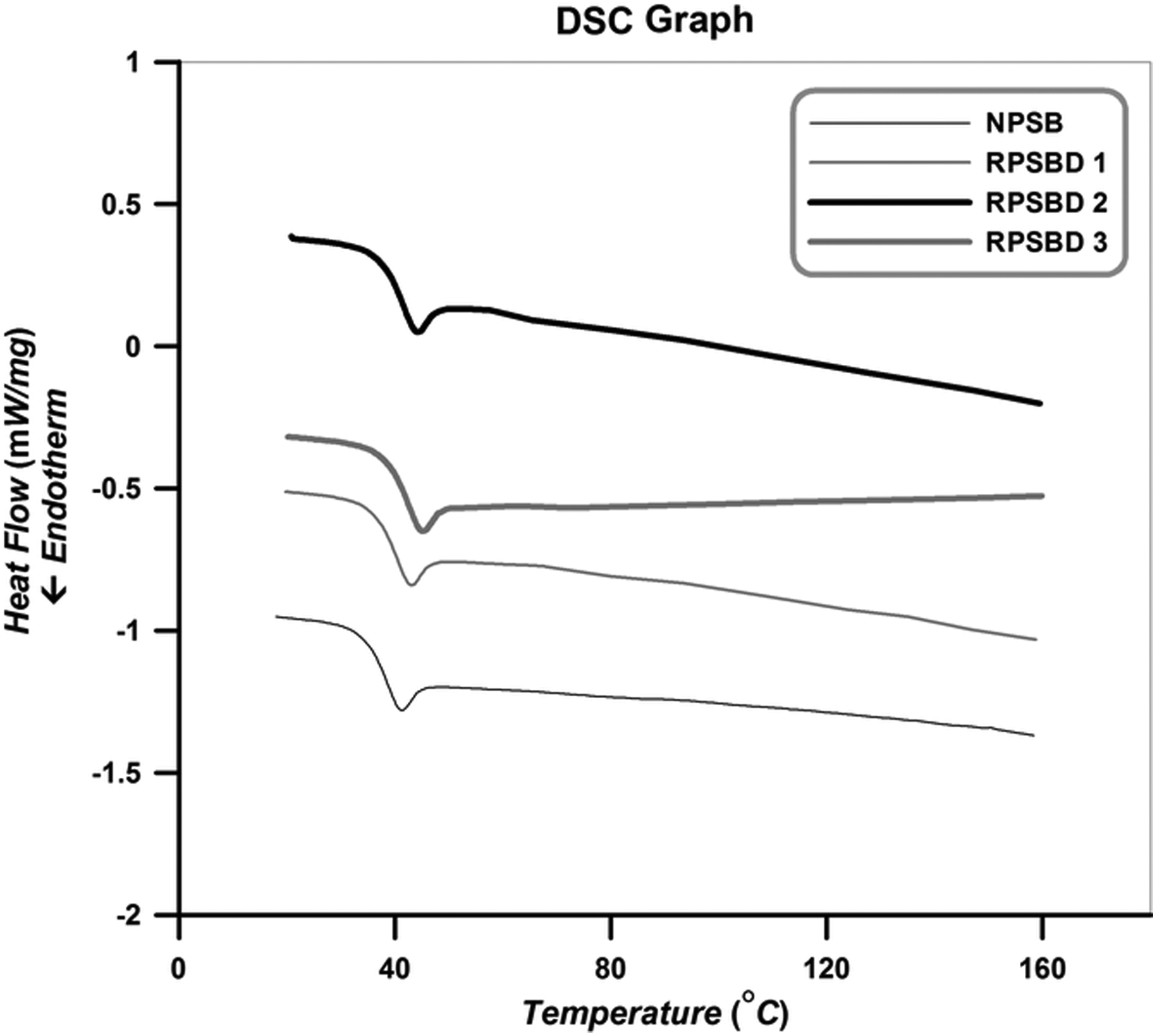
DSC thermograms of the NPSB and its nanocomposites. NPSB: neat poly (styrene-co-butyl acrylate); DSC: differential scanning calorimetry.
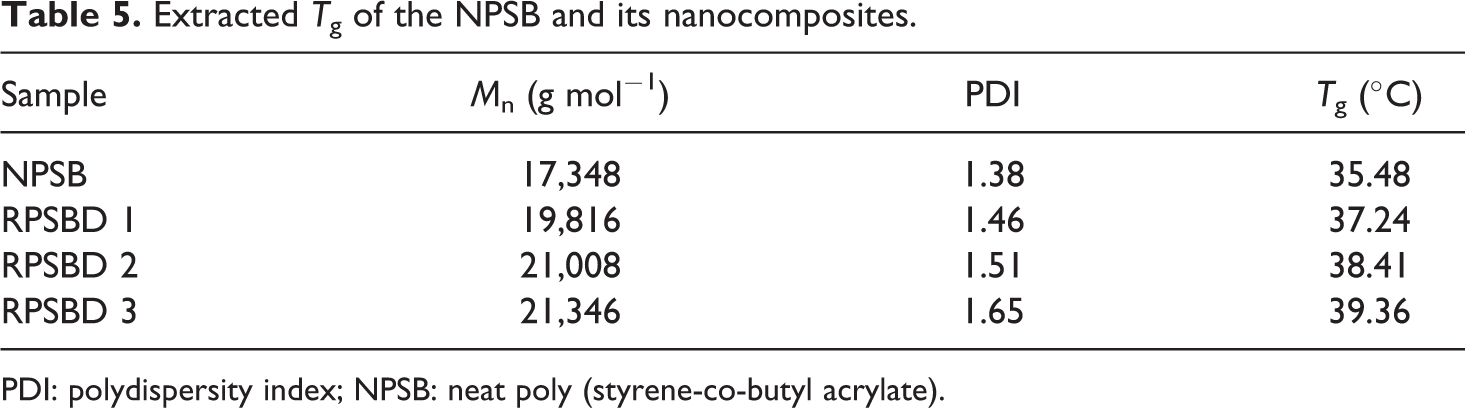
According to Figure 12, an obvious inflection in the heating path has occurred, which shows the T g values of the samples. Corresponding inflection in the cooling path has also appeared. No other peak in the cooling path is observed, which indicates that the structure of synthesized copolymer and its nanocomposites are mainly amorphous and they have not gone through crystallization phenomenon. Extracted T g values of the samples from DSC graphs are summarized in Table 5.
Extracted T g of the NPSB and its nanocomposites.
PDI: polydispersity index; NPSB: neat poly (styrene-co-butyl acrylate).
According to Table 5, the T g value of the NPSB is lower than all the nanocomposites and an increase in T g values occurred with an increase in the pristine diatomite nanoplatelets content. The increase in T g values with the addition of pristine diatomite nanoplatelets loading in the copolymer matrix can be attributed to the confinement effect of the diatomite nanoplatelets. The rigid two-dimensional diatomite platelets can restrict the steric mobility of copolymer chains and cause the inflection in the DSC graphs starting at higher temperatures. Similar conclusions are also reported in the case of polymer/clay nanocomposites. 45,50
The TEM image of the RPSBD 2 is shown in Figure 13. The TEM image was employed to study the structure of the prepared diatomite nanocomposite. TEM image of the RPSBD 2 shows that the pores of diatomite nanoplatelets have disappeared and a layer of poly (styrene-co-butyl acrylate) covers the surface of diatomite nanoplatelets.

TEM image of the RPSBD 2. TEM: transmission electron microscopy.
Conclusions
RATRP of St and BA was employed to synthesize tailor-made random poly (styrene-co-butyl acrylate) and its different nanocomposites in the presence of pristine mesoporous diatomite nanoplatelets. Mesoporous structure, the existence of plaque plate particles with spherical-shaped pores, and the existence of numerous regularly spaced rows in its structure are some features of the pristine diatomite nanoplatelets. In situ RATRP of St and BA in the presence of pristine mesoporous diatomite leads to an increment in conversion from 77% to 92%. Moreover, molecular weight and PDI values increase from 17,348 g mol−1 to 21,346 g mol−1 and from 1.38 to 1.65, respectively. 1H NMR spectroscopy results show that the composition of copolymers is approximately similar to the initial feed ratio of each monomer. Improvement in thermal stability of the nanocomposites and the increase in T g values from 35.48°C to 39.36°C were also observed by the incorporation of 3 wt% pristine mesoporous diatomite nanoplatelets.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
