Abstract
The degradation of polyhedral oligomeric silsesquioxane (POSS)/polystyrene (PS) nanocomposites at different percentages of reinforcement was studied in order to verify their thermal stability in respect to the pristine PS. Analyzed compounds were prepared by in situ polymerization of styrene, in the presence of POSSs and characterized by 1H-NMR. The temperature at 5% mass loss and the activation energy of degradation, determined through kinetics literature methods, were taken into consideration in order to assess both the resistance and the kinetics of degradation. The results obtained for the synthesized polymer and composites were discussed and compared with each other and with those obtained in the past for similar alkyl-POSS/PS nanocomposites. A good improvement of the thermal stability, in respect to neat polymer, was found and showed that the presence of cyclopentyl groups at the vertices of the POSS cage led to a good interaction with the matrix. It was also drawn up a thermal stability classification among the synthesized materials and those studied in the past according to the organic nature of the substituents on the inorganic cage of the composite reinforcement.
Keywords
Introduction
Nowadays, both the academy and industry need to address a number of challenges related to health, sustainability, and environmental protection through the design and development of new functional materials with the aim to improve the quality of life and create new advanced technologies. The research in this area is a continually expanding frontier at the interface of chemistry and materials science and a significant advance has been the synthesis of nanocomposites, where the structural order within the material can be controlled on nanometer/submicron scales. 1 Moreover, the combination of functional polymers with inorganic nanostructured compounds have become a major area of research and technological development owing to the remarkable properties and multifunctionalities deriving from their nano and hybrid structure. These materials represent the natural interface between two worlds of chemistry (organic and inorganic) each with different characteristic properties, so the idea, in developing hybrid compounds, is to take advantage of the best properties of each component trying to decrease or eliminate their drawbacks. Despite the concept of “hybrid organic-inorganic” nanocomposites has had much emphasis in the second half of the 20th century with the expansion of the so-called soft chemistry, 2 the habit of combining dissimilar components to yield improved materials is not new. Since the antique civilization of Asia, Egypt, Europe, and Pre-Columbian America, for more than 5500 years, organic–inorganic composite materials created by men have been part of human culture and customs. 3,4 The litmus test that demonstrates the high interest of the scientific community and industrial sector toward polymer hybrids and/or polymer composites is the huge increase in the number of publications, reports, and patents appeared during the last years on the design, synthesis, properties, and applications of these materials. 5 –7 Among the different reinforcement for obtaining polymer-based hybrid nanocomposites, 8,9 the use of silsesquioxanes, a kind of inorganic–organic hybrid molecules, having caged structure and usually called polyhedral oligomeric silsesquioxanes (POSSs), has been largely developed. The most common POSSs for this use have the formulas (RSiO1.5)8 and are characterized by the presence, at the vertices of the cage, of silicon atoms covalently bound with organic aliphatic and/or aromatic groups, whose nature influence the main properties of final nanocomposite. Another aspect to be considered in design POSSs reinforced materials is their structure, by preferring the asymmetric ones that lead to an increase in solubility and compatibility into polymer matrices. 10,11 Following this indication, with the aim to increase the resistance to degradation and the overall thermal stability of polystyrene (PS), in recent years, we have carried out the synthesis and characterization of POSS molecules variously functionalized. In this context, and trying to achieve a classification of the thermal stability of the synthesized compounds in respect to the functionalization of the organic reinforcement periphery, PS-based nanocomposites were synthesized by in situ polymerization of styrene in the presence of 3% and 5% of POSS in the reactant mixtures. The structure of the used POSSs is shown in Figure 1 and numbered as follows
n decyl heptacyclopentyl POSSs/PS at 3% w/w
n decyl heptacyclopentyl POSSs/PS at 5% w/w
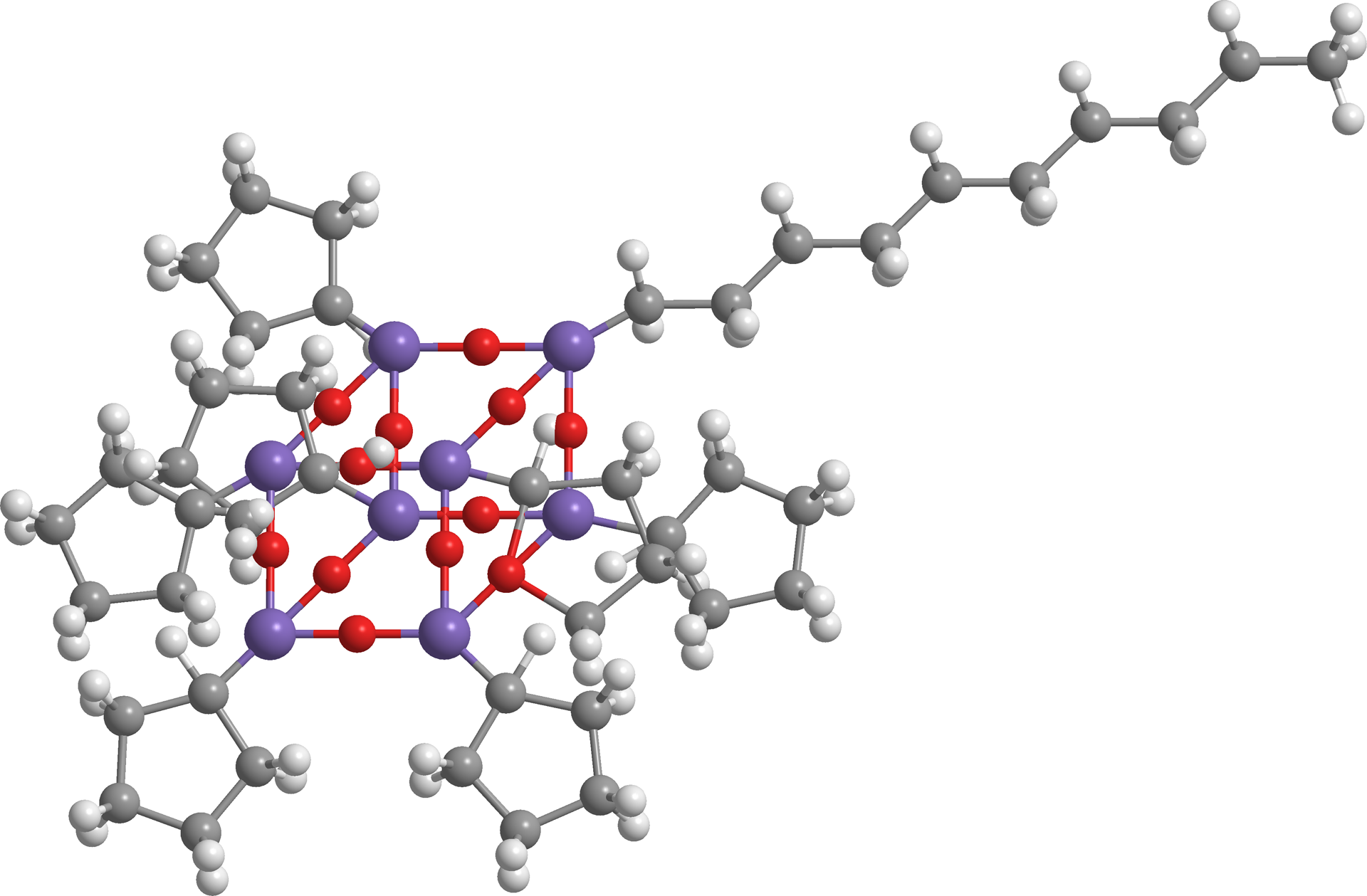
Molecular structure of n-decyl hepta cyclopentyl POSS. POSS: polyhedral oligomeric silsesquioxane.
Synthesized materials were spectroscopic investigated with 1H-NMR to check the presence of POSSs within the polymeric matrix and with Fourier transform infrared (FTIR) spectroscopy to study the composition of the nanocomposites residue at the end of degradation. The characteristic thermal parameters, namely temperature at 5% mass loss (T5%) and apparent activation energy (Ea) of degradation, were obtained by thermogravimetric analysis (TGA) and were compared with each other and with those of neat PS to evaluate their thermal behavior. Results were also compared with those obtained in the past for similar compounds, thus trying to realize a thermal resistance classification among them.
Experimental
Materials
Styrene, 2,2-azobis(isobutyronitrile) (AIBN), and toluene were purchased from Aldrich Co. and purified, the first one in an inhibitor removal column, AIBN re-crystallized twice from dry ethanol, out of direct light and at temperatures less than 313 K and the last one stirred over calcium hydride (24 h) and distilled in inert atmosphere (nitrogen). n-decyl heptacyclopentyl POSS was prepared by the procedure described elsewhere
12
while in situ polymerization of 3% and 5% w/w POSS/styrene mixtures in toluene was carried out to obtain nanocomposites at different POSS contents. As example, we report the procedure of free-radical polymerization for sample
1H-NMR spectroscopy
1H-NMR characterization was performed in a Unity Inova instrument 1H 500 MHz (Varian) by using CDCl3 as solvent and TMS as internal standard.
Thermogravimetric analysis
A Shimadzu DTG-60 simultaneous DTA-TG apparatus was used for TGA. The calibrations (repeated every 2 weeks) of temperature, heat flow, and mass were performed following the procedure of the instruction manual of equipment and reported elsewhere 13 using as standard materials: indium (NIST SRM 2232), tin (NIST SRM 2220), and zinc (NIST SRM 2221a) for temperature; indium (NIST SRM 2232) for heat flow and a set of exactly weighed samples supplied by Shimadzu for mass. Thermal degradations were carried out at various heating rates (Φ = 2, 5, 7.5, 10, 12.5, 15, 17.5 and 20 K·min−1), in the temperature range 298–973 K in both flowing nitrogen (0.02 L·min−1) and static air atmosphere. Samples of about 6 × 10−3 g, placed in a 40 μL alumina open pan, were used for experiments. The sample mass as a function of temperature was monitored and recorded by a personal computer connected with the DTG-60 apparatus. At the end of each experiment, these data were used to plot the percentage of undegraded sample, (1 − D)%, as a function of temperature, where D = (Wo − W)/Wo, and Wo and W were the masses at the starting point and during scanning. TGA measurements were performed by adopting the blank method to correct the error in the mass determination due to the reduction of the buoyancy force with increasing temperature. 14 All the considered values were averaged from those of three runs, the maximum difference between the average and the experimental values being within ± 1K.
IR spectroscopy
FTIR spectra were obtained with a Spectrum 100 spectrometer (Perkin Elmer, Waltham, USA) directly on the nanocomposites residues, by using a universal attenuated total reflection (ATR) sampling accessory. The measurements were performed at room temperature, from 4000 to 650 cm− 1 with a resolution of 4.0 cm− 1.
Results and discussion
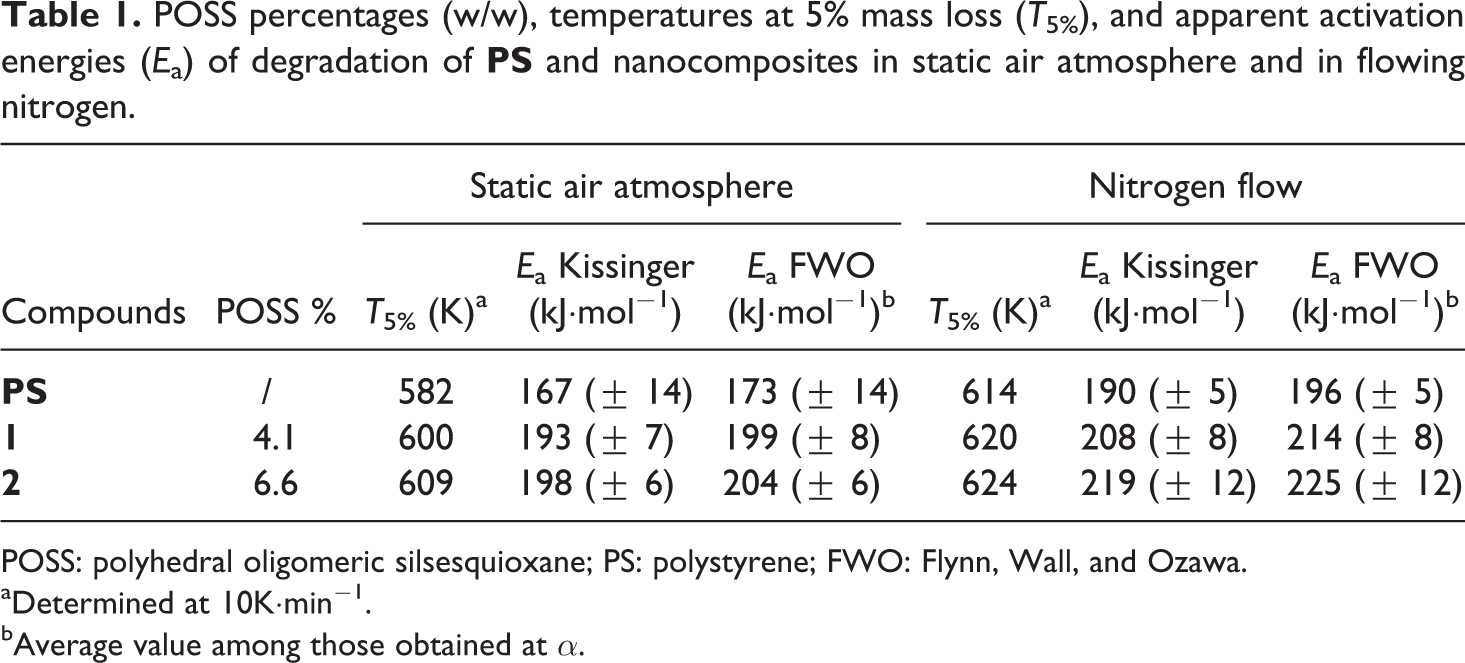
1H-NMR measurement was first performed to determine the presence of the reinforcement in the synthesized compounds, through the ratio between the POSS hydrogen atoms and the PS hydrogen ones. The exact POSSs content in the nanocomposites was slightly higher than that of starting mixtures (Table 1), probably due to the formation, during the in situ polymerization, of methanol soluble oligomers of PS. 15
POSS percentages (w/w), temperatures at 5% mass loss (T5%), and apparent activation energies (Ea) of degradation of
POSS: polyhedral oligomeric silsesquioxane; PS: polystyrene; FWO: Flynn, Wall, and Ozawa.
aDetermined at 10K·min−1.
bAverage value among those obtained at α.
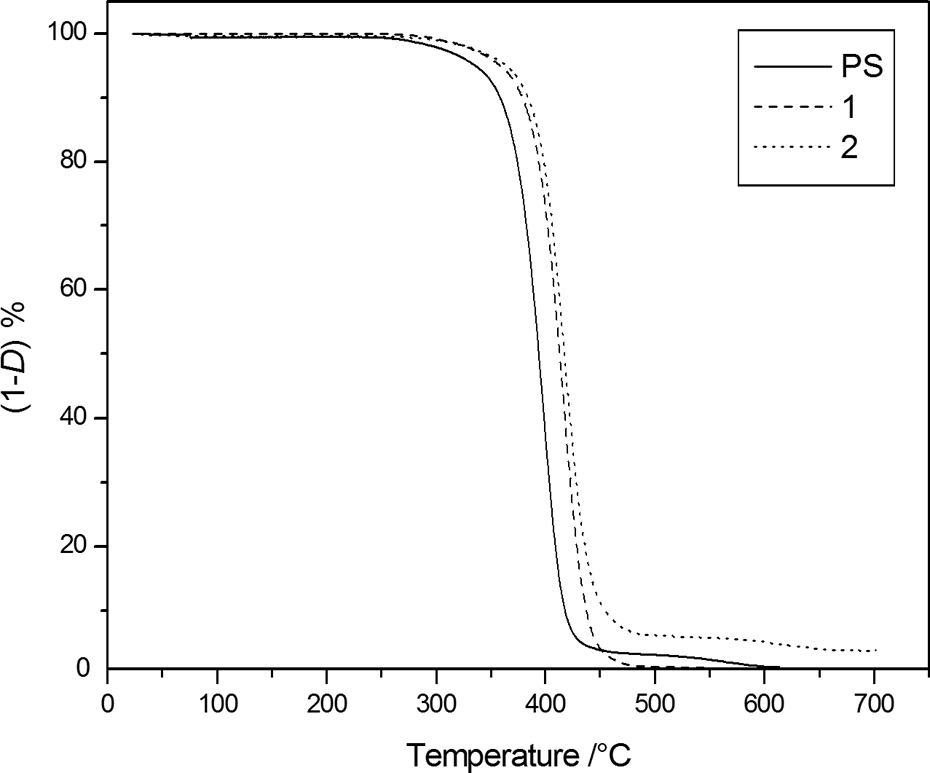
The thermogravimetric characterization was carried out firstly to evaluate the resistance to thermal degradation that seems to increase, at least qualitatively, in both, inert and oxidative, atmosphere as shown by the TGA degradation curves (Figures 2 and 3) of samples
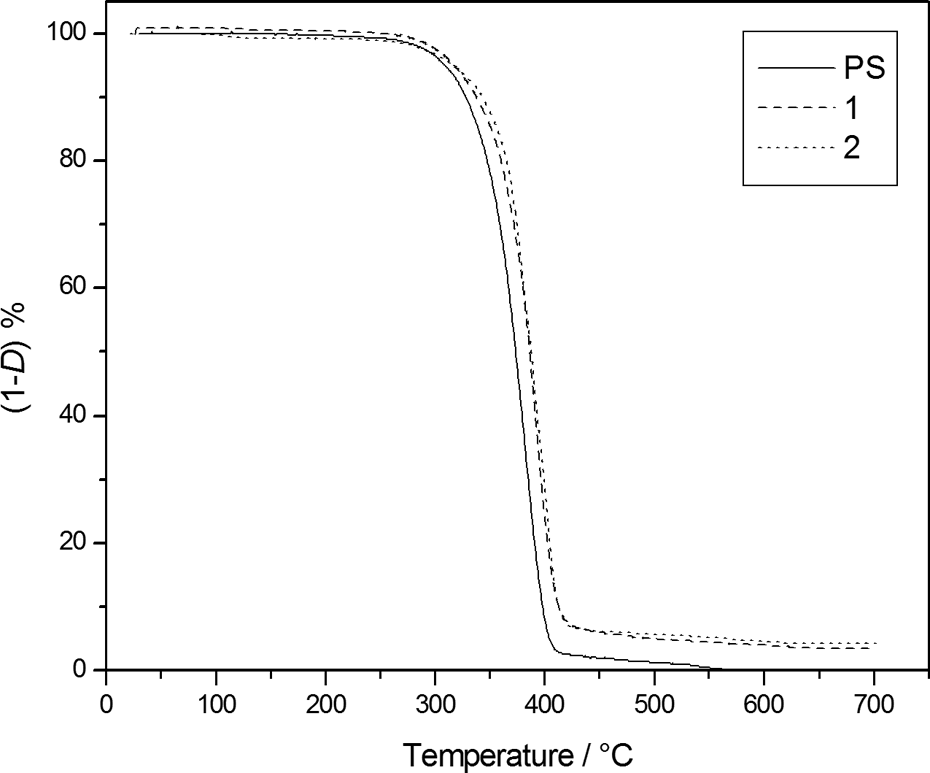
TG degradation curves, at 10K·min.−1, under nitrogen flow of
TG degradation curves, at 10K·min−1, in static air atmosphere of
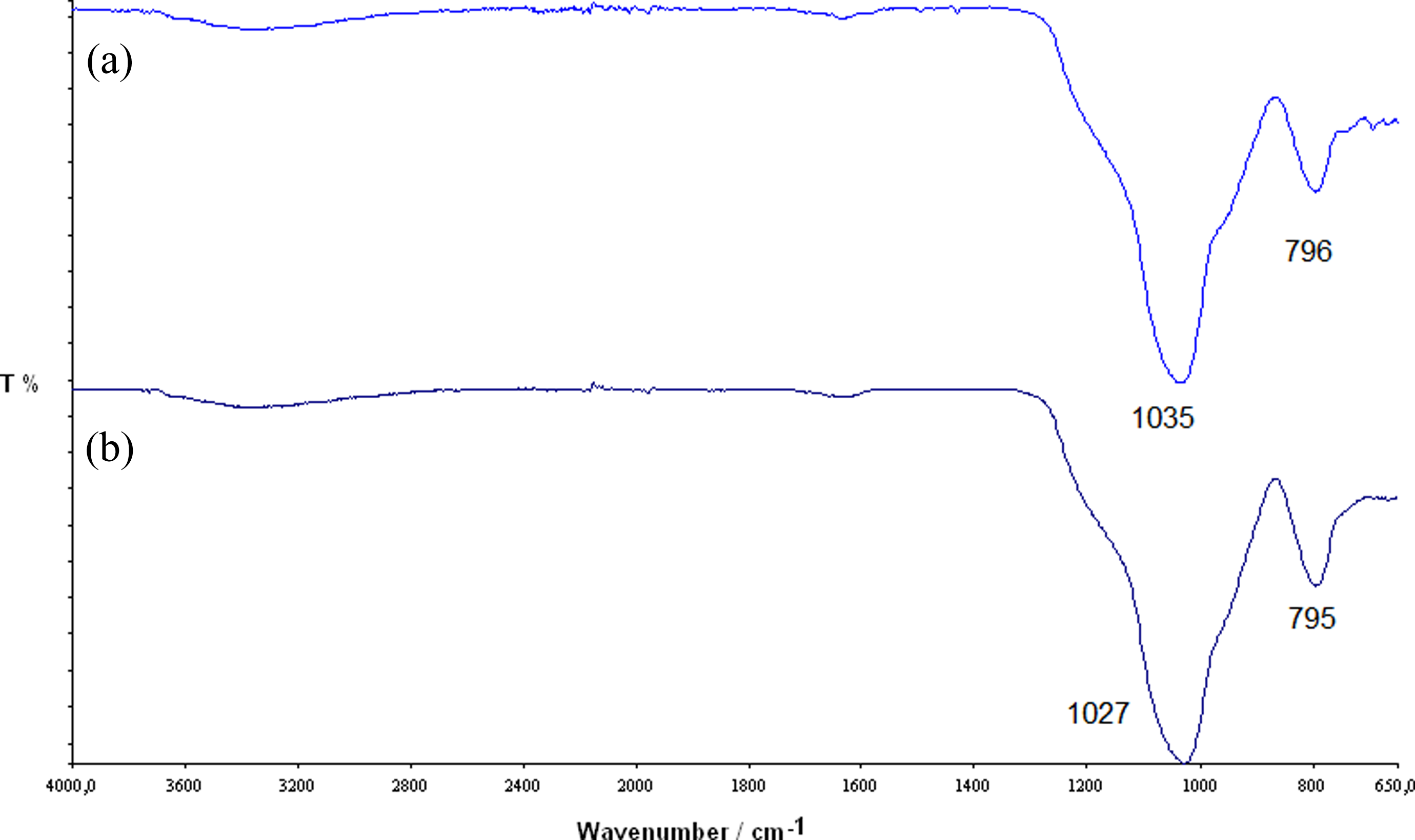
FTIR spectra of the residues, obtained at 973 K and in flowing nitrogen, of (a) sample
The evaluation of the thermal behavior of the synthesized nanocomposites, in respect to the pristine PS, was thus completed by studying the kinetics of decomposition. The temperatures at maximum rate of weight loss (Tm) of all studied compounds, at all heating rates and in both atmosphere, were used to calculate the apparent Ea of degradation. To this aim, the least square treatment of the data in Tables 2 and 3 was made according to the Kissinger equation 17 :
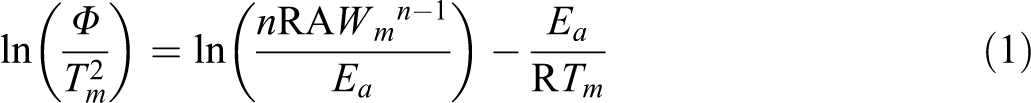
Temperatures at maximum rate of weight loss (Tm) for the degradation of
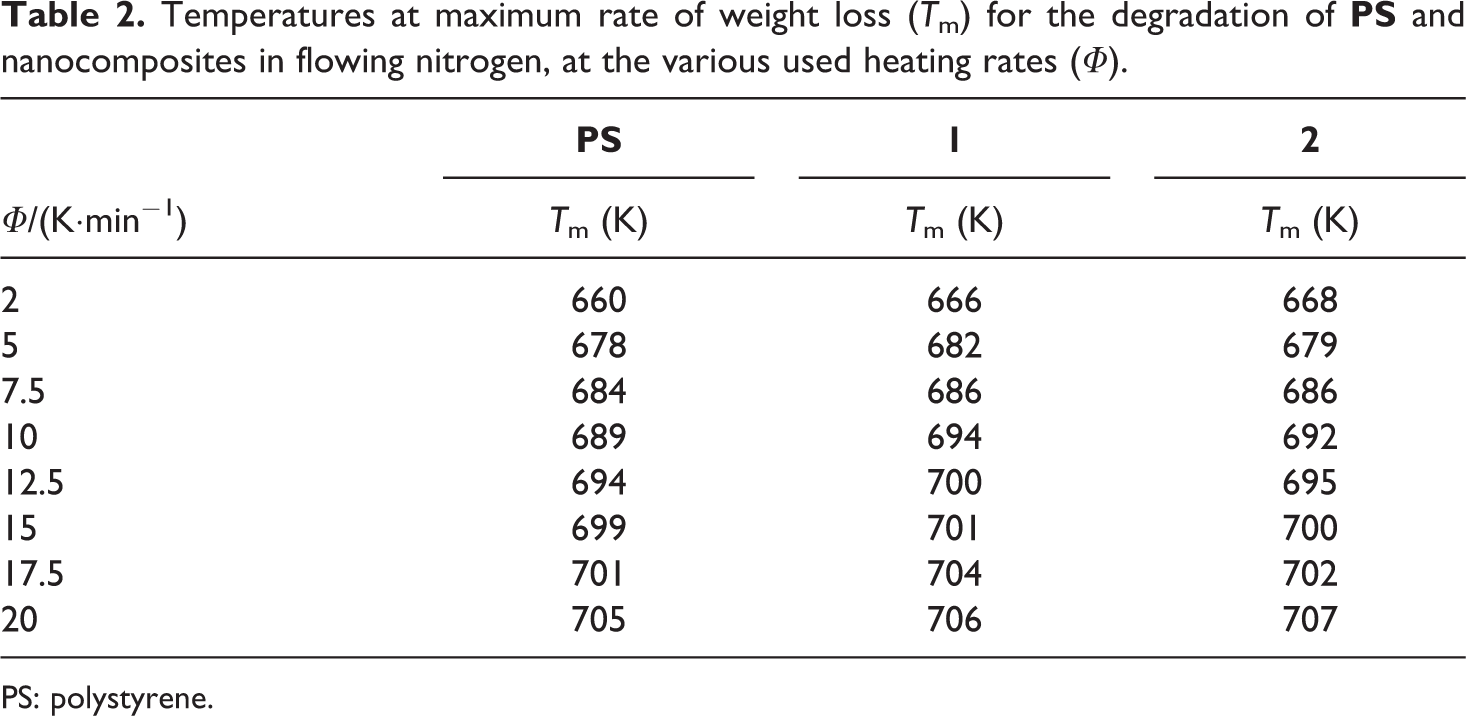
PS: polystyrene.
Temperatures at maximum rate of weight loss (Tm) for the degradation of PS and nanocomposites in static air atmosphere, at the various used heating rates (Φ).
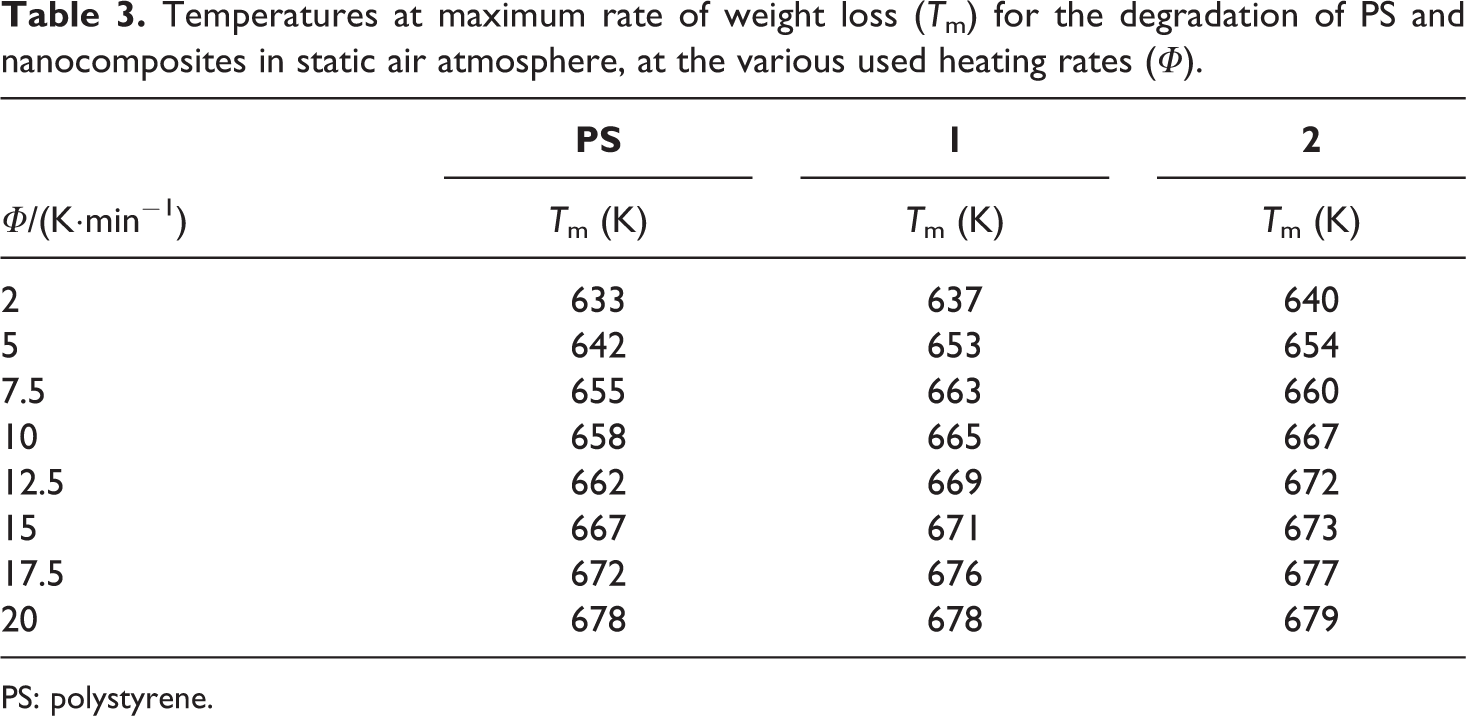
PS: polystyrene.
where Φ is the heating rate, Tm is the temperature at maximum rate of weight loss, n is the apparent reaction order, R is the universal gas constant, A is the pre-exponential factor, and Wm is the weight of sample at the maximum rate of mass loss. After ascertaining that conversion does not vary with heating, in order to ensure a reliable estimation, 18,19 Ea values were calculated trough the obtained linear dependences of ln (Φ/Tm 2) on 1/Tm at various heating rates for both PS and nanocomposites. Degradation Ea values with their corresponding regression coefficients are reported in Tables 4 and 5 for nitrogen and air, respectively. In order to confirm the reliability of the obtained kinetics values, the sets of TGA data at the various heating rates, in both flowing nitrogen and static air atmosphere, were treated through the integral isoconversional method of Flynn, Wall, and Ozawa (FWO), 20,21 based on the following equation:
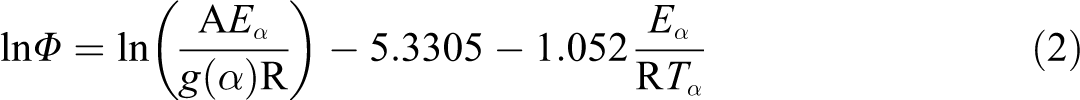
Regression coefficients and apparent activation energies (Ea) of degradation by the Kissinger equation for PS and nanocomposites in flowing nitrogen.
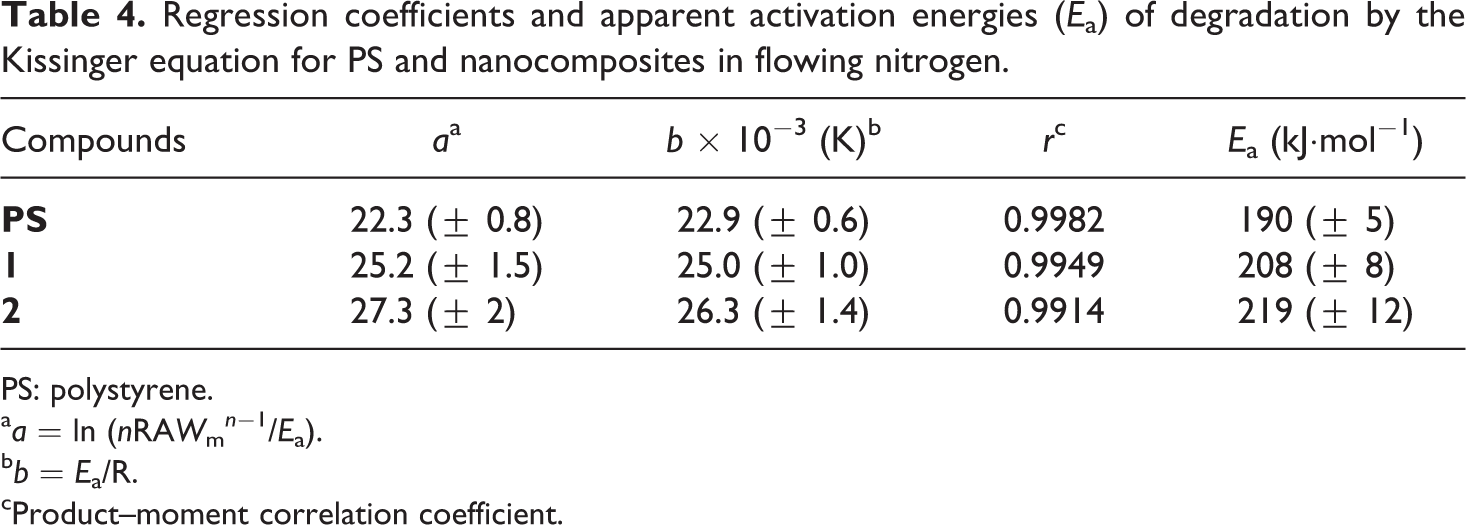
PS: polystyrene.
aa = ln (nRAWm n −1/Ea).
bb = Ea/R.
cProduct–moment correlation coefficient.
Regression coefficients and apparent activation energies (Ea) of degradation by the Kissinger equation for PS and nanocomposites in static air atmosphere.
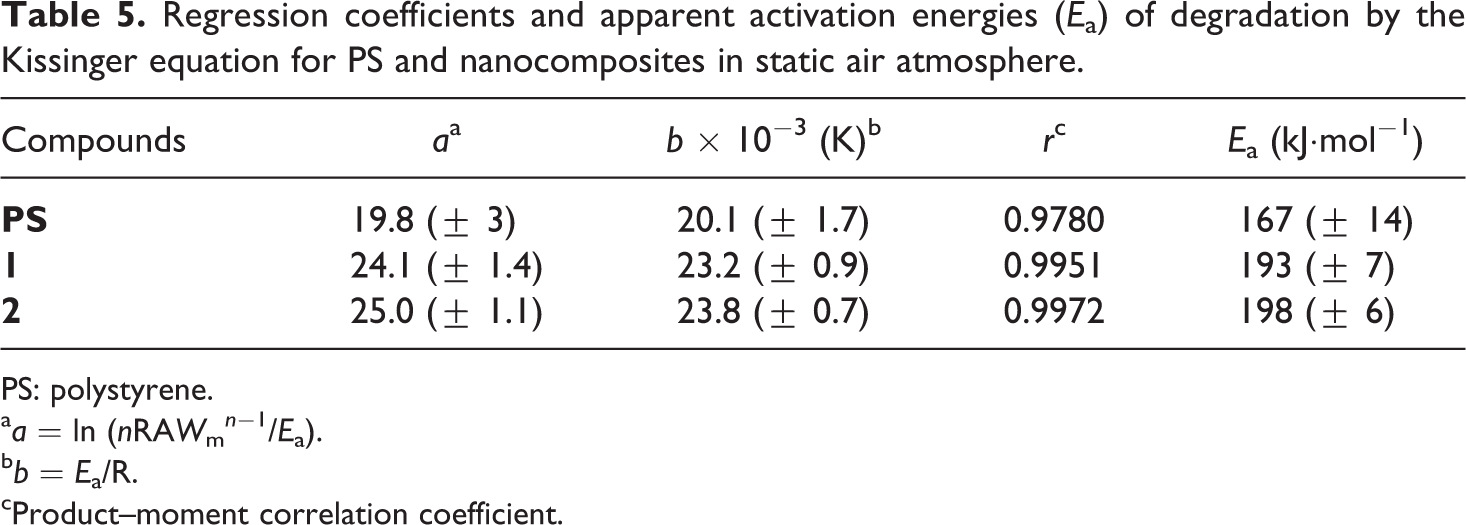
PS: polystyrene.
aa = ln (nRAWm n −1/Ea).
bb = Ea/R.
cProduct–moment correlation coefficient.
where Tα is the temperature at which the conversion α is experimentally achieved at the heating rate Φ. Through equation (2), the FWO method allows to obtain the value of the apparent activation energy Eα at a fixed value of α, from the slope of the straight line generated in the plot of ln (Φ) versus 1/Tα (Figures 5 (a) to (f)), for the number of TGA runs carried out at different heating rates (Φ), assuming that the Doyle’s approximation is valid over the entire range of α. 22,23 A very close agreement was found between the Ea values obtained through the Kissinger and FWO methods, respectively (Table 1).
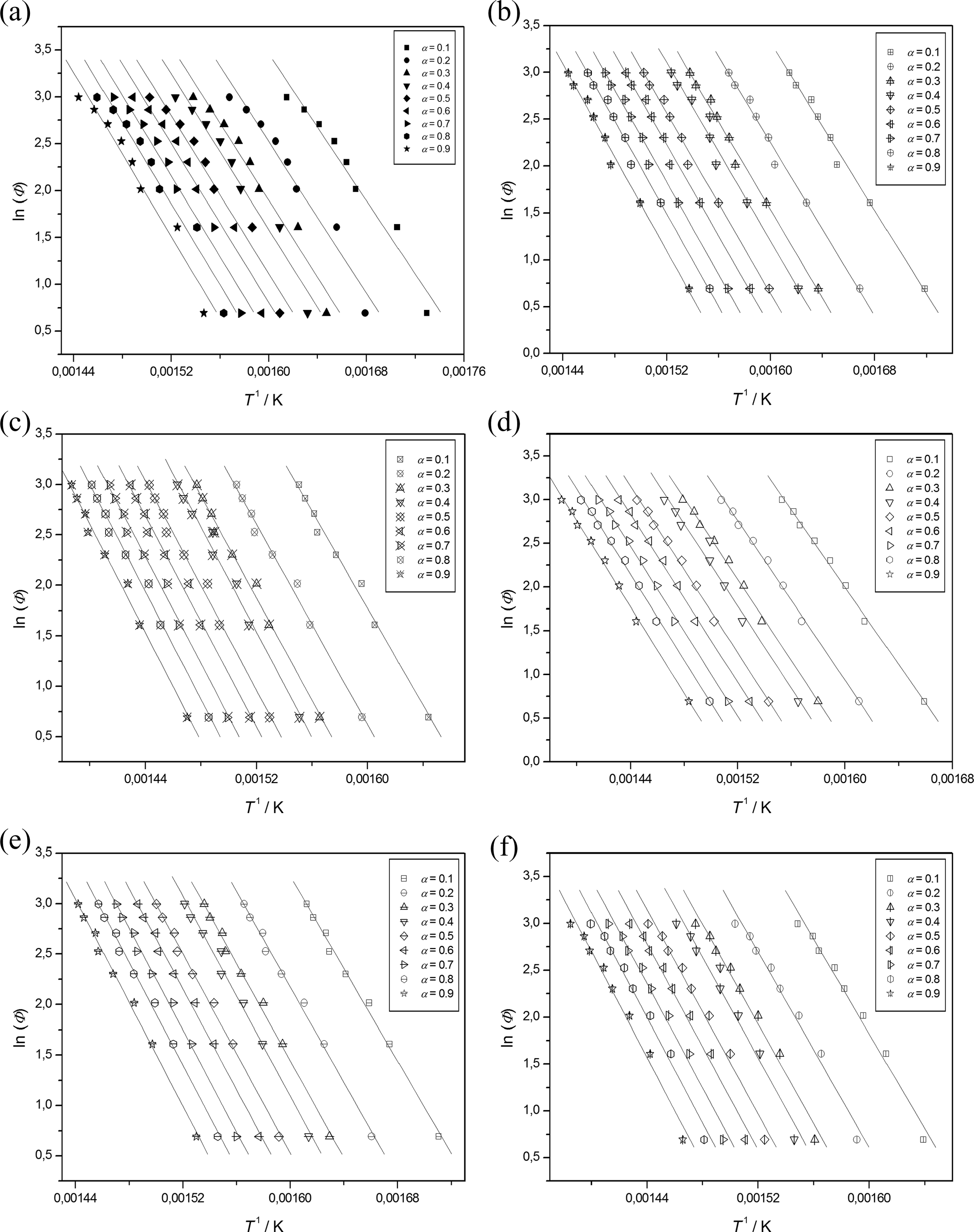
FWO straight lines for
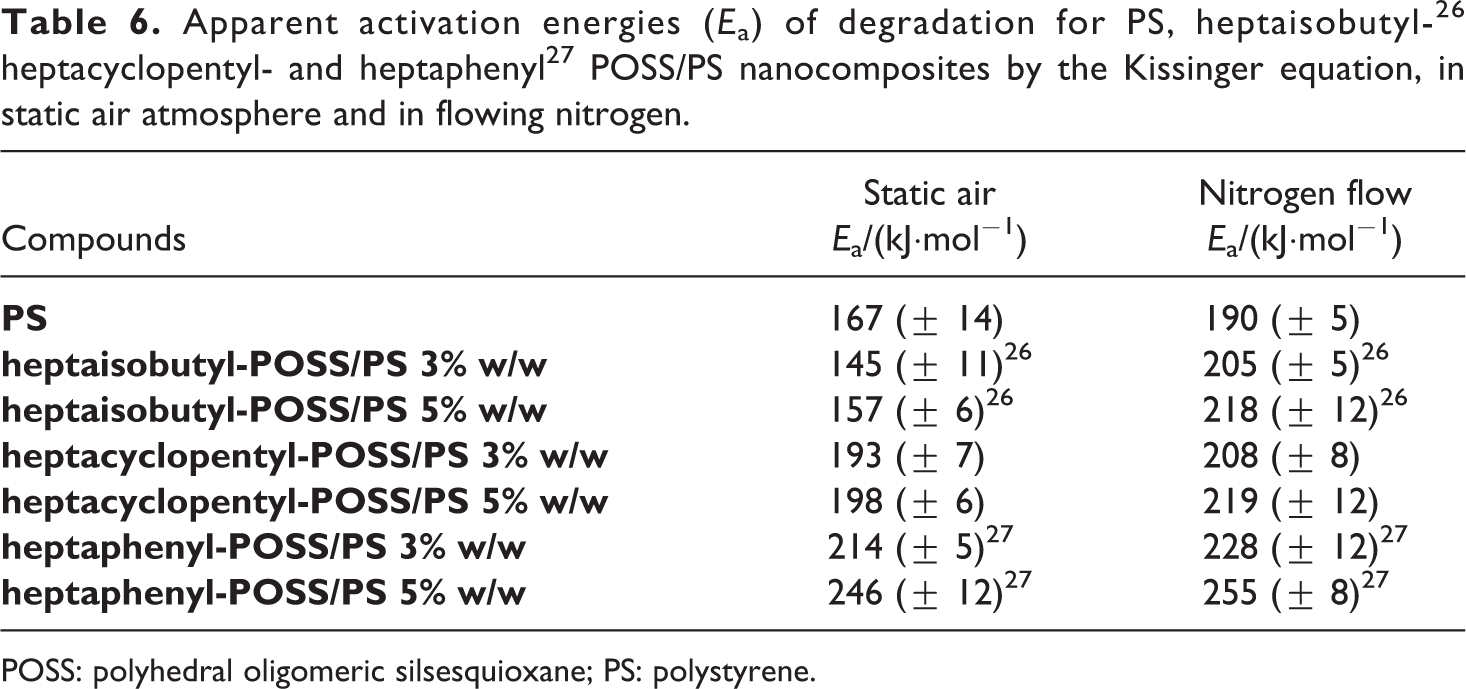
The largest increase of Ea in static air atmosphere, for the two synthesized nanocomposites in respect to PS, confirms the active role of POSSs in the thermoxidative degradation process of the hybrid systems. In agreement with literature report, 24,25 the increase in POSS content, ranging between 3% and 5% w/w, enhances not only the resistance to the thermal degradation but, by slowing the degradation rate, the overall thermal stability of nanocomposites. If we compare these results with those obtained in the past for similar compounds, but characterized by an isobutyl 26 or phenyl 27 periphery (Table 6), we can affirm that the increase and the decrease, recorded in this work, both for the initial decomposition temperature and for the activation energy of degradation in respect to the use of isobutyl and phenyl POSSs, respectively, is derived from a double action dependent on the nature of the substituent groups and its compatibility with the polymer matrix:
good dispersion in the matrix attributable to the asymmetry of the reinforcement molecules;
reduction of the filler auto-aggregation phenomena thanks to the use of POSS with less reactive groups at the vertices of the silicon cage.
POSS: polyhedral oligomeric silsesquioxane; PS: polystyrene.
We can conclude that the thermal stabilities of investigated POSSs/PS nanocomposites, as measured through T5% and Ea values, change according to the following order: phenyl-derivatives > cyclopentyl-derivatives > isobutyl-derivatives, thus indicating that the presence of less reactive and cumbersome groups makes POSSs molecules more suitable for dispersion in thermoplastic matrix.
Conclusions
The degradation experiments, in inert and oxidative atmosphere, of the PS-based nanocomposites investigated indicated a good improvement of the overall thermal stability with respect to neat PS and confirm the in situ polymerization as an effective, and quite simple and fast method to synthesize POSSs/polymer nanocomposites. Finally, it was clear that the presence of cyclopentyl groups at the vertices of the POSS cage, in respect to the isobutyl one previously studied, led to a better dispersion, but to a worse one in respect to phenyl groups, thus affecting both the T5% (and thus induction period) and the Ea (and thus kinetics of the degradation process).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.