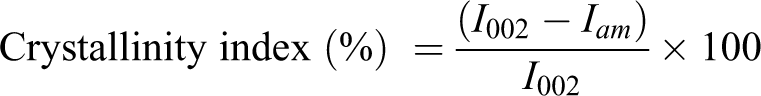Abstract
It was found beneficial to manufacture bacterial cellulose (BC) obtained from waste food sources to create value-added packaging products by firstly performing purification and disintegration processing of the BC and then reinforcing it with paper pulp. The purification treatment involved using sodium hydroxide (2% w/v at 100°C for 1 h) on BC pulp to remove the bacterial protein and the resulting cultured medium film was characterized with respect to its physical properties. An acid treatment on the purified BC pulp was applied to disintegrate cellulose network before forming a film. The results showed that the sodium hydroxide treatment increased the film burst index and brightness. Heating the BC pulp to 70–100°C in 1.25–5.00% v/v sulfuric acid treatment for 30 min degraded the film’s mechanical properties. Also, a study of the addition and mixing of paper pulps or modified cationic starch to the BC was found to improve the film properties with respect to packaging properties. Inclusion of short fiber paper pulp by 30 wt.% created a good synergistic effect by improving the mechanical properties of film especially for tear strength. Moreover, cationic-modified cassava starch (2% w/w) improved the tensile and burst index as well as resistance to oxygen permeation of the BC film. The results indicate that the BC pulp could be used to improve mechanical properties and resistance to water vapor and oxygen permeation of the short fiber paper pulp film which is ideal for packaging materials.
Introduction
Bacterial cellulose (BC) has been used as a foodstuff for a long time and it is called Nata de coco in the Philippines and many other countries. This cellulose is produced by Gluconacetobacter xylinus grown in a liquid medium by using coconut juice as the base medium containing sucrose, ammonium sulfate, and acetic acid. The cellulose is produced in the form of a gelatinous membrane known as a pellicle which when fully formed floats on the liquid medium. The cellulose content in the pellicle is about 0.7% w/w. From a scanning electron micrograph, the pellicle is composed of fabricated microfibrils, with each fibril having a diameter of approximately 3.5 nm. About 46 adjacent fibrils join to form a ribbon through hydrogen bonds, which has a width ranging from 40 to 60 nm 1 and branching occurs every 180–196 nm. 2 Together, the entangled mesh of these fibrils produces a pellicle in which bacterial cells are entrapped. This gel-like pellicle is also called a BC gel and contains hemicellulose, lignin, and pectin-free cellulose. The Young’s modulus of a BC sheet is about 20–30 GPa; 1,3,4 however, the modulus of a single BC fiber estimated by Raman spectroscopy techniques is 130 GPa. 5,6 This BC gel is typically cleaned and dried to be utilized in many applications. 1,7 Cellulose acetate membranes prepared from a BC gel have been used to separate copper ions or volatile organic compounds from their respective matrices. 8 –10 Carboxymethylcellulose derived from BC gel on the other hand has been used to coat bell peppers thus prolonging their postharvest life. 11 To improve the biocompatibility of a BC gel, biopolymers, such as gelatine, were added into the fermentation media. BC-biopolymer sheets have also been used as separation membranes, tissue-engineering scaffolds, and wound-dressing materials. 12
After forming the pellicle, the physical properties of BC gel can be altered by adjusting pH, changing ionic strength, heating, and freezing. 13 To form sheets and diaphragms, the BC pellicle was either treated with NaOH and then hydraulically pressed 3 or treated with acetone and subsequently vacuum dried 14 (or treated with NaClO). 3,15 These sheets and diaphragms were prepared from a BC gel without disintegration. In this research, the BC gel was disintegrated to remove the bacterial cells and impurities that were implanted in the gel. It is good to use renewable, edible, and biodegradable materials like BC pulp film as packaging materials (similar to sericin-based films) for protection of the environment. 16,17 The BC pulp can be used as a reinforcement material due to its high mechanical performance. For example, a nanocomposite was reported to be made from BC pulp and a polyurethane-based resin cured under ultraviolet light. 18 This nanocomposite made with 10–50% w/w BC pulp exhibited superior density and mechanical and dielectric properties. Woehl et al. 19 hydrolyzed NaOH-purified BC pulp with an enzyme and used the resulting product as a reinforcement material for bionanocomposites made from cassava starch. Lin et al. 20 focused on the effect of high-pressure homogenization (HPH) on the physical properties of BC aqueous suspensions to improve water swelling ability during storage of processed food. Beside that, BC was used as a new functional material in paper and nonwoven fabric-like products. 21,22 Due to small clusters of cellulose microfibrils of BC, it was added in paper as a binder, which enhances the strength and durability of pulp into paper. Development of BC for paper products in the paper industry was patented for Ajinomoto Co. with Mitsubishi Paper Mills in Japan (JP patent 63295793). 23 Incorporating 5% of modified BC with wood pulp was investigated by Basta and El-Saied 24 to improve kaolin retention, strength, and fire-resistance properties, compared to paper sheets produced from incorporating BC. During the decade 2000 to 2010, BC was studied as an active food packaging with the addition of nisin to control Listeria monocytogenes and other bacteria, on the surface of vacuum-packaged frankfurters. 25 The BC films containing with absorbed bovine lactoferrin (bLF) on surface were devolped for bio-based edible antimicrobial packaging systems. 26
There is a lot of BC waste from the food processing industry, which can be disintegrated and the resulting BC pulp can be utilized to make value-added products for paper packaging which can replace some plastic packaging. Thus, the present work focuses on BC pulp treatment and the mechanical properties of the produced film products. The effects of the BC pulp on physical properties of short fiber paper pulp film and effects of cationic-modified cassava starch on physical properties of the BC pulp film were also investigated.
Materials and methods
Purification of the BC pulp
The crude BC gel waste was provided for this study as wastage from the solid BC cube process (provided by Panafood Industry Co. Ltd., Bangkok, Thailand). This was combined with coconut juice as the base medium and G. xylinus bacterial strain to generate the BC pellicle and, in turn, was analyzed for protein, ash, moisture, fat, fiber, and carbohydrate content (AOAC, 1990). The BC gel was washed twice and then boiled in washing water (1:1 by weight) until the washing water pH was neutral. The BC gel was left to cool at room temperature and then water was added to obtain the weight ratio of BC gel to water of 1:2. Then the BC gel was disintegrated in a British disintegrator (British Pulp Evaluation Apparatus; Mavis Engineering, Ltd., England) at 3750 r min−1. Water was added to the BC pulp slurry to obtain the BC pulp concentration of 1.5% w/v.
The pulp slurry was mixed with 2% w/v NaOH solution at the weight ratio of 1:10 and then heated to 100°C for 1 h. The pulp was washed until the washing water pH was neutral. The films were formed from the unpurified and NaOH-purified BC pulp as described in “Film forming” section.
Acid treatments of the NaOH-purified BC pulp
The NaOH-purified BC pulp was dried at 60°C for 2 h and then added with H2SO4 solution at the constant w/v ratio of pulp to sulfuric acid of 1:67 and different concentrations of 1.25, 2, 3, 4, and 5% v/v. The slurry was heated at various temperatures (30, 70, and 100°C) for 30 min. After the slurry was cooled and the acid solution was discarded, the pulp was dispersed in 5% w/v NaOH solution at the w/v ratio of 1:67 and then boiled for 30 min. The NaOH was discarded. The pulp was neutralized with 1% v/v HCl once and with water until the pH of the rinsing water was nearly 7. A film of acid-treated pulp was formed.
Preparation of the BC and paper pulp mixture
The sulfite-treated short fiber paper pulps were bought from Bang Pa-in Paper Industry, Co. Ltd., Ayutthaya, Thailand. The paper pulp was prepared according to ISO 5269/1. The NaOH-treated BC pulp was mixed with prepared short fiber paper pulp at 0, 30, 50, 70, and 100% w/w.
Preparation of the BC pulp and modified starch mixture
The suspension of cationic-modified cassava starch TAPLINK-2 with a degree of substitution of 0.018–0.025 (Thaiwa Co. Ltd., Bangkok, Thailand) was heated at 95°C for 15 min and diluted to the concentration of 5% w/v. The starch suspension (of 0, 0.5, 1.0, 1.5, and 2.0 g) was then mixed with 100 g of dry NaOH-treated BC pulp.
Film forming
Films were cast from BC pulp, BC-paper pulp mixtures, and BC-starch mixtures prepared in “Purification of the BC pulp”, “Acid treatments of the NaOH-purified BC pulp”, “Preparation of the BC and paper pulp mixture”, and “Preparation of the BC pulp and modified starch mixture” sections using the standard method described in ISO 5269-1 (2005). A standard sheet former (Mavis Engineering Ltd.) and sheet press (Toyo Seiki Seisakusho, Tokyo, Japan) were used for film forming.
Film characterization
Prior to determination of physical properties, the BC films were conditioned under 27 ± 1°C and relative humidity of 65 ± 2% for 24 h. The physical properties of film to be determined were mass (ISO 536, 1995), thickness and apparent density (ISO 534, 2005), Young’s modulus, tensile strength and elongation (ISO 1924/2, 1994), tear resistance (ISO 1974, 1990), bursting strength (ISO 2758, 2001), and brightness (ISO 2740, 1999).
The water vapor permeability and oxygen gas permeability of the film were determined using Dish Method ASTM E-96 (1980) and ASTM D1434-82(1998), respectively.
Disintegration of the film was determined by soaking 0.05 ± 0.01 g film in 25 ml distilled water. The soaking time required for disintegration or tearing of the film was recorded.
The crystallinity of BC with NaOH purification was examined using a high-resolution X-ray diffractometer (Philips Panalytical X’Pert, the Netherlands). Scattered radiation was detected in the range of 2θ from 5° to 40° with a scanning rate of 5°/min. The crystallinity index was calculated by Segal’s method as shown in the following equation: 27
where crystallinity index represents the relative degree of crystallinity,
Experimental design and statistical analyses
A completely randomized experimental design (CRD) was used for all experiments. SPSS 11.0 for windows (SPSS Inc., Chicago, IL, USA) was used to calculate analysis of variance (ANOVA) and Duncan’s multiple range test with a significance level of 0.05.
Results and discussion
BC gel used in this study contained 93.51% w/w water (wet basis). The fiber, protein, ash, fat, and carbohydrate contents (dry basis) of BC gel were 14.82, 13.72, 7.40, 0.93, and 63.13 wt.%. The noncellulose content in BC gel was about 85.18 wt.% (dry basis). This BC gel was used for film forming which was prepared with different treatments and with incorporation of other biomaterials.
Effect of purification of BC pulp on physical properties of the BC film
Upon purification of BC pulp by heating the pulp in 2% w/v NaOH solution at 100°C for 1 h, the burst index and brightness of the film increased 32.56 and 23.86%, respectively (Table 1). Other film physical properties were not affected by this NaOH purification. Nishi et al. 15 reported that purification of BC gel using this method resulted in an increase in mechanical properties of the dried BC sheet/film. Cellulose I, the native form of cellulose in BC gel, was altered to cellulose II by soaking it in an alkaline solution. 3 Moreover, NaOH can solubilize amorphous cellulose because the alkaline treatment removes proteins and nucleic acids of the bacterial cells which increases the crystallinity of the material. The higher interchain hydrogen bonding in cellulose II gave higher mechanical properties of the purified dried BC sheet/film. 3 The Young’s modulus of the BC film formed from BC pulp was only 50% of that of the intact dried BC film reported by Nichi et al. 15 In case of % crystallinity of BC pulp after purification analyzed by XRD (Figure 1), the result showed 80.62% which is high crystallinity after purification. 28 This result supported the higher mechanical properties after purification of BC pulp.
Effect of NaOH purification of BC pulp on physical properties of the BC film.a
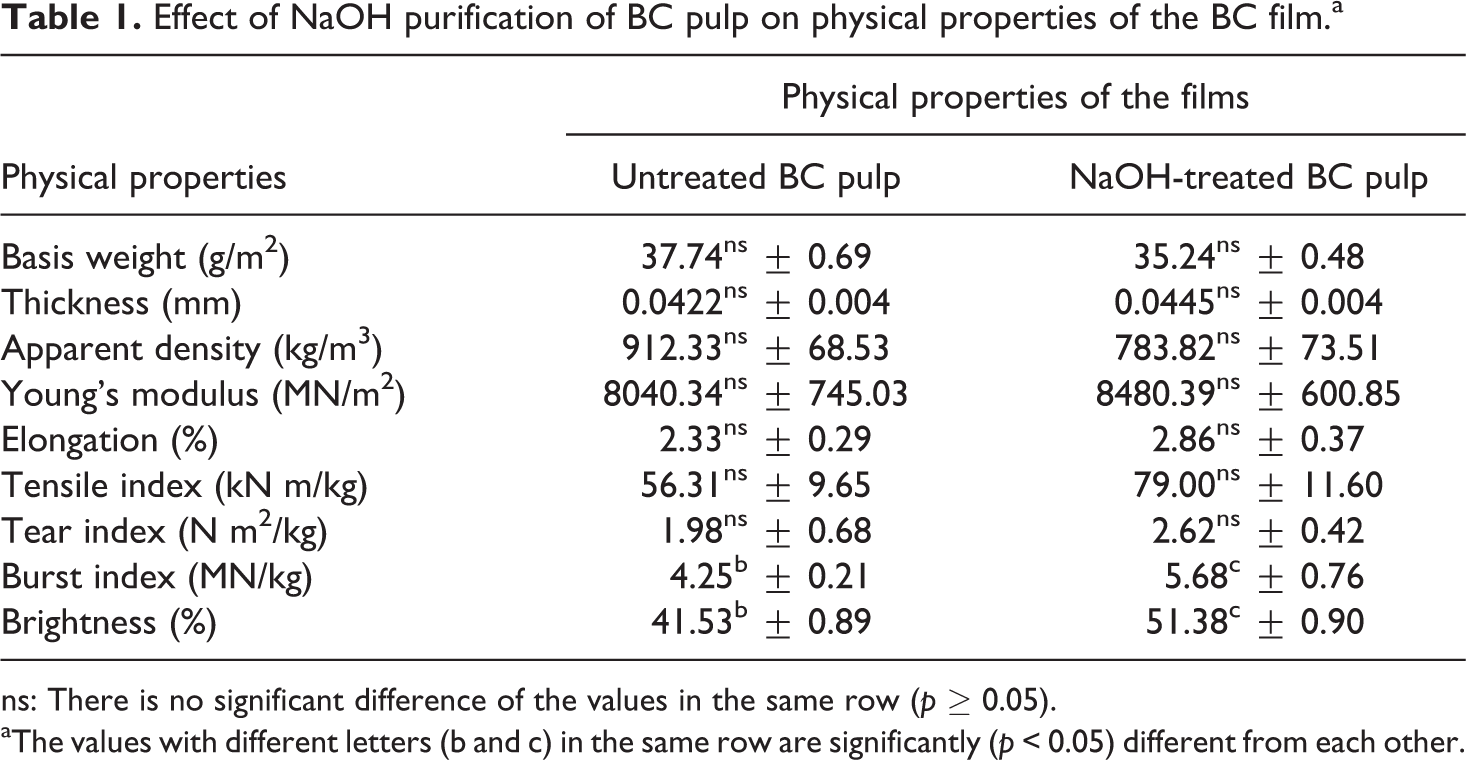
ns: There is no significant difference of the values in the same row (p ≥ 0.05).
aThe values with different letters (b and c) in the same row are significantly (p < 0.05) different from each other.
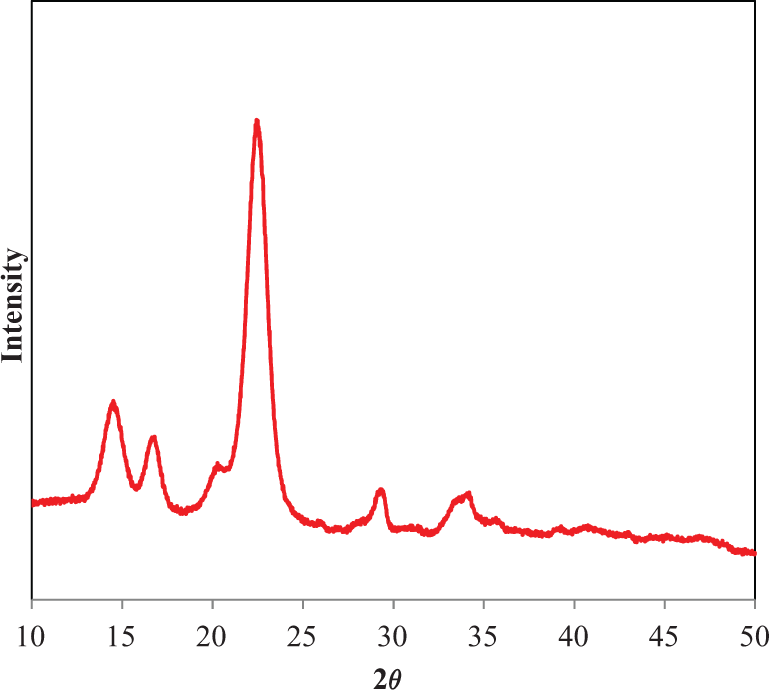
X-ray diffraction of the BC pulp after NaOH purification.
Effect of acid treatment of the NaOH-purified BC pulp on physical properties of the BC film
Regardless of acid concentration, sulfuric acid treatment of BC pulp at 30°C did not affect mechanical properties but increased brightness of the BC films (Table 2). This indicated that at low temperature, sulfuric acid did not destroy the cellulose chain of the BC pulp but bleached the pulp. Mechanical properties of the BC films decreased when BC pulp was treated with sulfuric acid at 70 and 100°C. An increase in temperature used for sulfuric acid treatment resulted in decreased elongation, tensile index, tear index, burst index, and brightness of the BC films. Acid treatment at 70 and 100°C might destroy the cellulose chain and micelle. 29 Therefore, the microfibril was shortened and the thickness of fiber was reduced. This is resulting in decrease in film mechanical properties which depend on the strength of the fibers. 30,31 The SEM images also show that fibers of film of the BC pulp treated with acid at 100°C were shorter and interfiber interactions were less than those of the film of untreated BC pulp (Figure 2). However, there was no significant difference in Young’s modulus of the BC films when the treatment temperature increased from 70 to 100°C, even though acid hydrolysis was the method commonly used for preparation of cellulose nanocrystals. 32 Low molecular weight hydrolysates and sugar might be produced by acid hydrolyzation of cellulose at high temperature. These hydrolysates could cause film browning which decreased the film brightness. At the constant temperature of either 70 or 100°C, increasing acid concentration tended to decrease burst index but did not affect other mechanical properties and brightness of the films.
Effect of sulfuric acid treatment of the NaOH-purified BC pulp on physical properties of the BC film.a
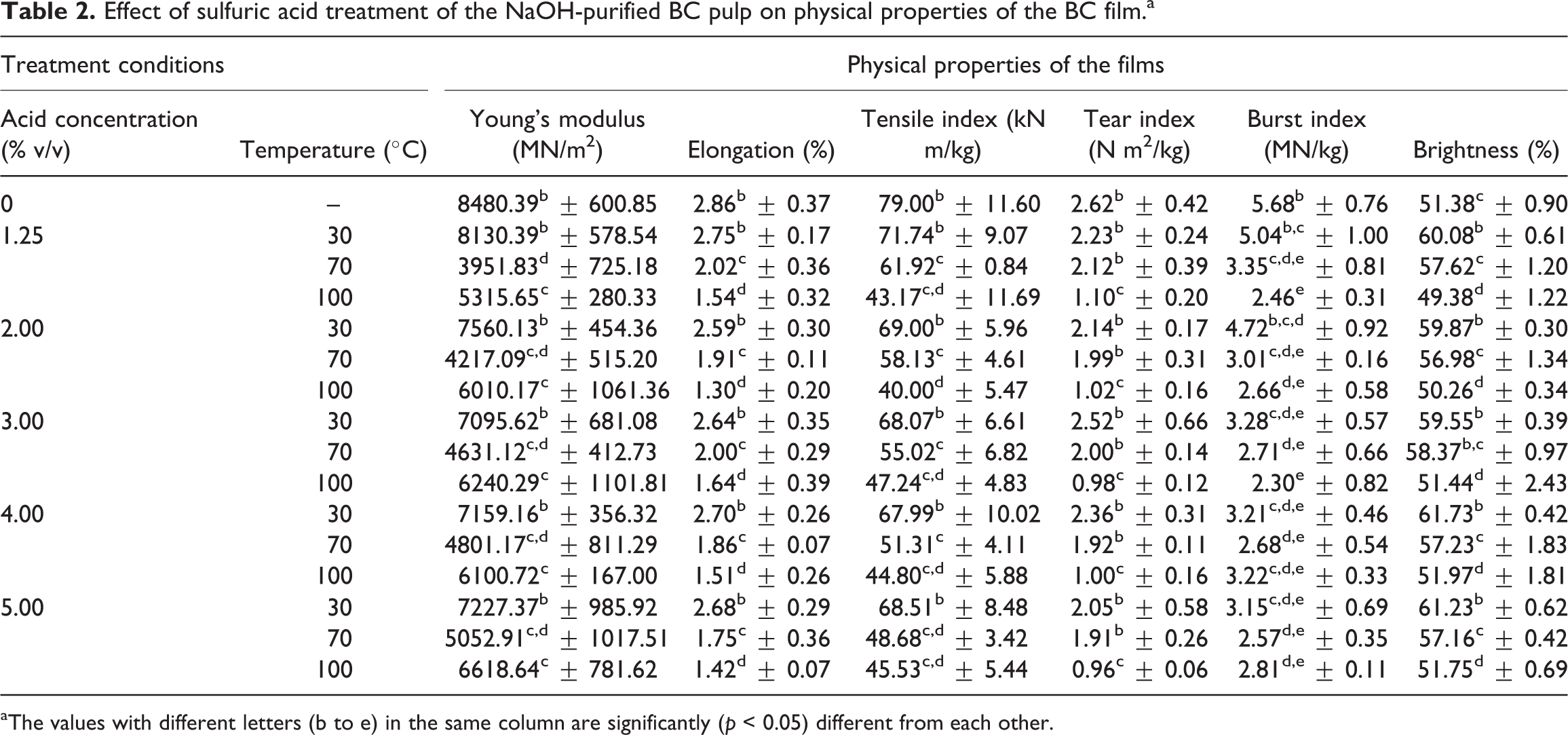
aThe values with different letters (b to e) in the same column are significantly (p < 0.05) different from each other.
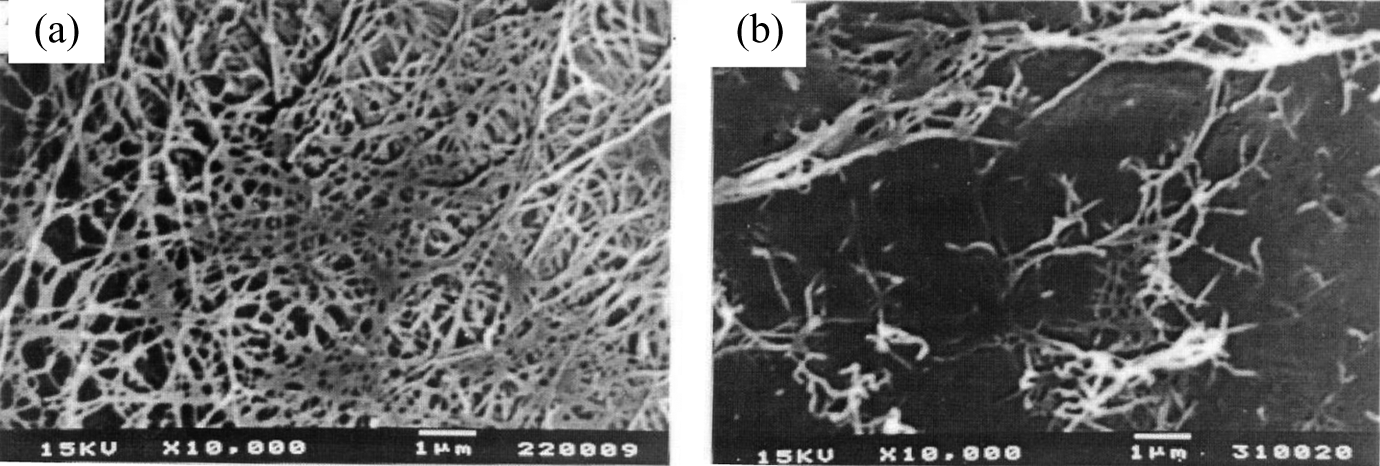
SEM photographs of the BC films (a) with no acid treatment, (b) with sulfuric acid treatment at 1.25 vol%, 100°C.
Effect of adding BC pulp on properties of short fiber paper pulp film
The BC film had apparent density, Young’s modulus, tensile index, and burst index higher than short fiber paper pulp film but had a lower tear index and brightness (Table 3). Table 3 also shows that adding BC pulp increased the physical properties of short paper pulp film except for brightness. The difference in elongation among these films was very small. Young’s modulus, tensile index, and burst index of the films increased with the BC pulp content. Tensile strength depended on strength of fibers and bonds between fibers. 30,31 Strength of interfiber bonds was more important than strength of fibers for Young’s modulus, tensile index, and burst index, but vice versa for tear index. 7,31 The bond strength between interfibers is related to the number of hydrogen bonds between fibers. 7 The fiber diameter was found to be 2–4 and 22,000–50,000 nm for the bacterial cellulose (BC) fiber and paper pulp, respectively. 1,7,33 The fine fibers of BC pulp provided larger contact areas among them resulting in higher fiber density and hydrogen bonds between fibers. 7 Interfiber bonds were greater as the BC pulp content increased. Tear index of films obtained from the mixtures of BC-short fiber paper pulp was higher than either the BC pulp film or short fiber paper pulp film (Table 3). This was the result of a combination effect of higher fiber strength of short fiber paper pulp and higher interfiber bonds of the BC pulp as confirmed by SEM photographs (Figure 3). It showed that the number of bonds between bacterial cellulose fiber and paper pulp (Figure 3(b)) was greater than the bond between the paper pulps (Figure 3(a)). Moreover, water vapor and oxygen permeability of the films decreased with the BC pulp content (Figure 4), which is a good property for packaging. This can be due to the fact that the BC pulp had higher fiber density and shorter distance between fibers than the paper pulp. The BC pulp may be useful as a wet-end additive for papermaking as reviewed by Keshk. 34
Physical properties of films from mixtures of BC and short fiber paper pulp.a
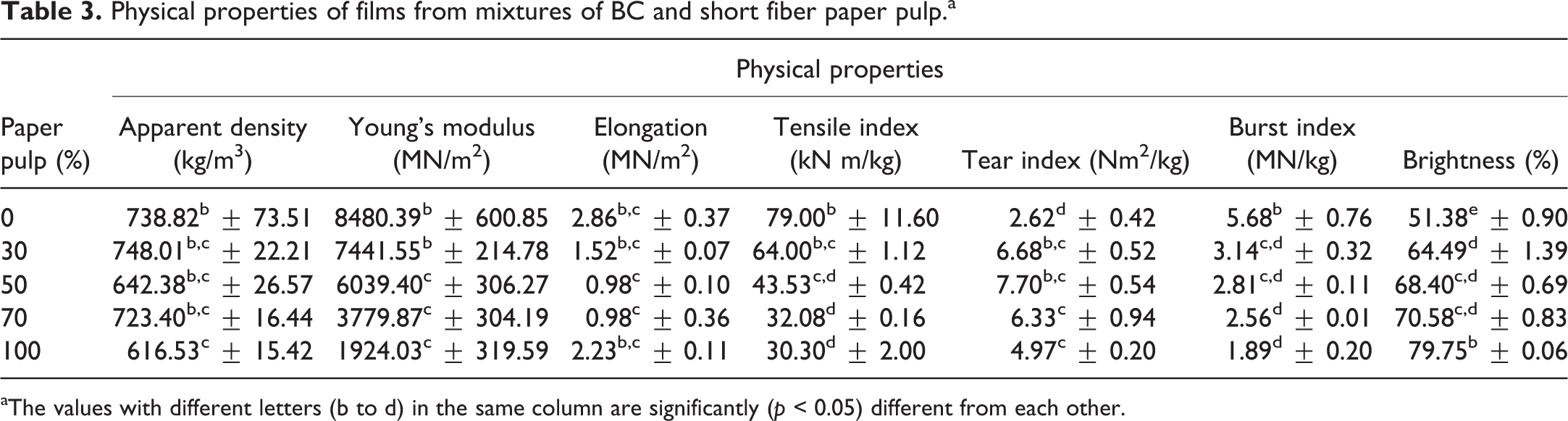
aThe values with different letters (b to d) in the same column are significantly (p < 0.05) different from each other.
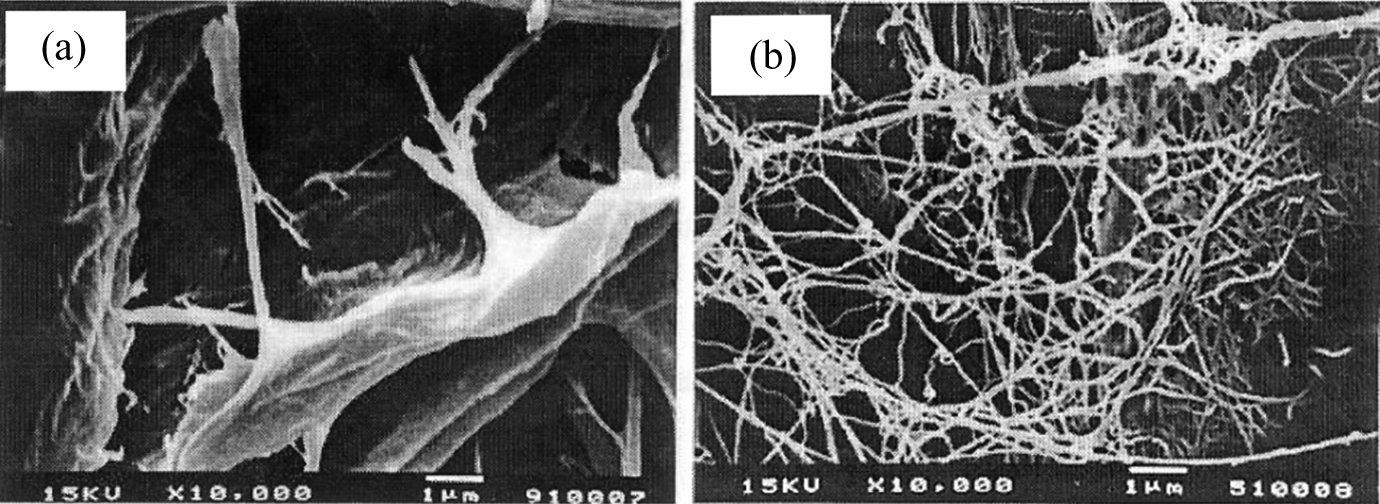
SEM photographs of the films formed from (a) short fiber paper pulp and (b) BC pulp mixed with 30 wt.% of short fiber paper pulp.
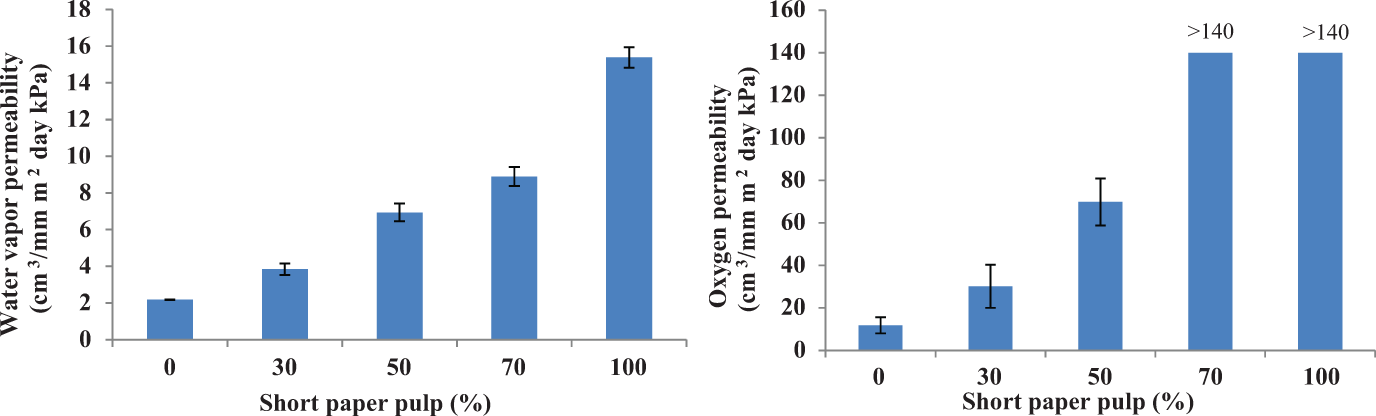
Water vapor and oxygen permeability of the films from mixtures of BC and paper pulp.
Effect of cationic modified cassava starch on properties of the BC pulp film
Cationic modified cassava starch improved the tensile index, burst index, and resistance to oxygen permeation of the BC film only when 2% of this starch was added (Table 4 and Figure 5). This starch concentration was reported to be an optimum concentration which gave the maximum starch retention on cellulose fiber. 35 Gaiolas et al. 36 and Zakrajsek et al. 35 reported that bonds between the free hydroxyl group on cationic starch and acidic groups on cellulose fibers improved physical properties of cellulose film. Starch did not only bind between fibers but also filled in the space between fibers resulting in lower oxygen permeation. As confirmed with burst index which increased with adding of starch at 2% in the BC pulp, addition of starch did not affect WVP because starch has a hydrophilic group; therefore, it did not act as a barrier against water vapor. Besides, the starch biocomposites with incorporated BC pulp enhanced the tensile properties and the resistance to moisture, which is applicable for packaging to protect food spoilage. 37 Additionally, natural biocomposites of BC are susceptible to biodegradability, which is one of the most promising aspects of its incorporation in packaging materials and industries. 38
Physical properties of films from mixtures of BC pulp and cationic-modified cassava starch.a
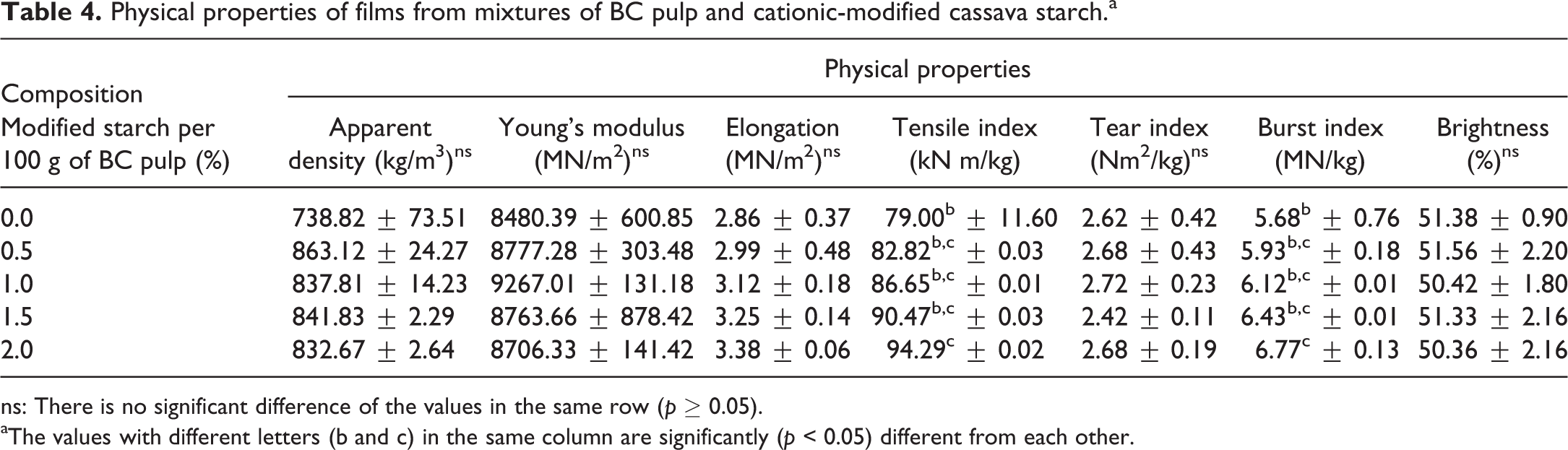
ns: There is no significant difference of the values in the same row (p ≥ 0.05).
aThe values with different letters (b and c) in the same column are significantly (p < 0.05) different from each other.
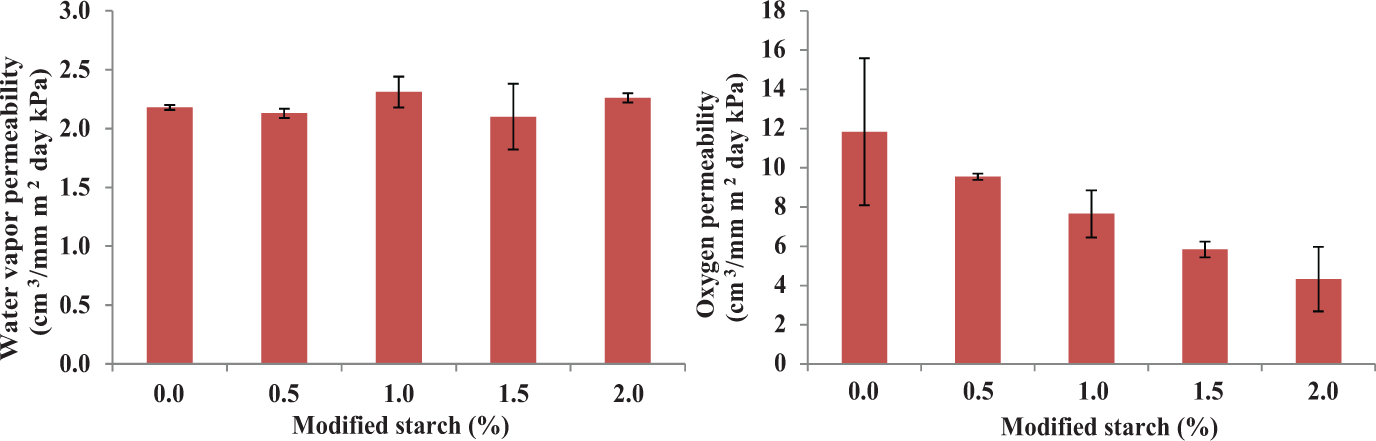
Water vapor and oxygen permeability of films from mixtures of BC pulp and cationic-modified cassava starch.
Conclusion
Purification of the BC pulp with 2% w/v NaOH was required to increase the burst index and brightness of the film. Treatment with sulfuric acid was not recommended for preparing the BC pulp. For the short fiber paper pulp film, inclusion of the BC pulp improved Young’s modulus, tensile and burst index, and resistance to water vapor and oxygen permeation. Addition of 2% cationic-modified cassava starch improved the tensile and burst index as well as resistance to oxygen permeation of the BC film. Therefore, the BC pulp after mild alkaline purification together with adding short paper pulp and/or cationic modification starch can be used to improve mechanical and barrier properties of paper packaging.
Footnotes
Acknowledgements
The authors would like to thank Panafood Industry Co. Ltd., Bangkok, for supplying the bacterial cellulose and Thaiwa Co. Ltd., for supplying modified starch used in this experiment. Formation of BC films was done at paper pulp unit of Research Division in Science Service Department, Ministry of Science and Energy, Thailand. Physical properties of films were determined at the Center of Packaging, the Institute of Science and Technology, Thailand.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by Ratchadaphiseksomphot Endowment Fund, Chulalongkorn University, Thailand.