Abstract
In this work, laccase was employed to improve the properties of lignocellulosic jute fabrics and then the modified jute was used as the fiber reinforcement of polypropylene (PP) matrix composites to improve its reinforcing effect on the polymer resins and obtain composite materials with better performance. The decrease in the –C–O–H component and the increase in the –C–O–C component on the surface of laccase-treated jute fabrics suggested that phenolic hydroxyl groups of lignins on the jute fiber surface were oxidized by laccase and the produced phenoxyl radicals were coupled to form ether structures. The laccase-treated jute fiber surface became smooth with lignins attached tightly. Moreover, the surface hydrophobicity and tensile properties of the jute fabrics were increased via the laccase-mediated reactions of lignins. PP composites reinforced by the laccase-treated jute fabrics showed higher breaking strength, storage modulus, and melting temperature than the control. The fracture surface of the laccase-treated jute fabric/PP composites was neat and jute fibers on the section surface were surrounded by PP resins closely, which indicated better interfacial adhesion between the modified jute reinforcement and PP matrix.
Introduction
The application of abundant plant fibers in nature such as ramie fiber, sisal fiber, bamboo fiber, and coir fiber to replace glass fibers and synthetic fibers as the reinforcement of resin matrix composites has drawn a wide public attention in recent years. 1,2 Plant fiber-reinforced composites have the merits of low cost, light weight, high strength, and biodegradability. 3 –5 However, the plant fibers always have strong hydrophilicity, high moisture absorption, and numbers of structural defects, which decrease the interfacial compatibility with the nonpolar resins and deteriorate the mechanical properties of the composites. 6,7 In order to obtain high-performance fiber-reinforced composites, surface modification of plant fibers is needed to translate the original hygroscopicity into hydrophobic characteristics.
Traditionally, two approaches, that is, physical and chemical processing technologies, have been employed to modify natural fibers. Physical methods normally include heat treatment, 8 steam explosion, 9 plasma processing, 10 and irradiation, 11 while the chemical methods mainly consist of graft polymerization by initiators 12 or high-energy rays 13 and coupling with organic silicon compounds. 14 However, these processes were investigated to have some inherent weaknesses. The physical methods are usually difficult to industrialize and can seriously deteriorate the mechanical properties of the natural fibers. 9 –11 Grafting modification with vinyl monomers is able to increase the hydrophobicity of the fibers and improve the compatibility with the nonpolar resins, but it is also known that the homopolymerization of the monomers will occur inevitably. Although the treatment of fibers with silicon couplers has achieved much success in improving the surface hydrophobicity, it has disadvantages of complicated multi-step processes and environmental unfriendliness. 15
In response to shortcomings of the physico-chemical methods, the emergent enzyme technology offers new approaches in the modification of various lignins and natural lignocellulosic materials. 16 –21 Enzymatic processes have advantages of specificity, effectness, eco-friendliness, and working in mild conditions as compared to conventional methods. Laccases (EC 1.10.3.2, benzenediol: oxygen oxidoreductase) are the most investigated enzymes in this field. They are multi-copper glycoproteins that catalyze the mono-electronic oxidation of phenols and aromatic or aliphatic amines to reactive radicals and simultaneously reduce molecular oxygen to water in a redox reaction. 22 Lignin is a three-dimensional aromatic polymer with three structural units, guaiacyl, syringyl, and p-hydroxyphenyl, linked together in an irregular manner. 23 Studies showed that lignin is a suitable substrate for laccase and the phenolic sites of lignin macromolecules can be oxidized to phenoxyl radicals by laccase. 24 These reactive radicals can then undergo the covalent coupling to initiate the polymerization of lignins; while the degradation of lignin can occur simultaneously, mediated by the radical-induced cleavage of covalent bonds or aromatic rings. Previous research has shown that the molecular weight of lignin can be increased by laccase alone 25,26 or in the presence of some phenolic compounds 27,28 in response to enhanced cross-coupling. However, the depolymerization of lignin becomes dominant in laccase/mediator systems. These mediators can be synthetic mediators 29,30 such as 1-hydroxybenzotriazole and 2,2′-azino-bis-(3-ethylthiazoline-6-sulfonate) (ABTS) as well as natural mediators with higher redox potential 31,32 such as syringaldehyde and acetosyringone, which can be activated by laccase to oxidize the non-phenolic units dominating the lignin structure. In addition, laccase-oxidized (radical-containing) phenols or nonoxidized amines of foreign interest can also be grafted to the radicalized lignins or lignocellulosic surfaces to produce engineered materials with novel functions. 33 –35
Natural jute fiber occupies the second place in the world production of plant fibers after cotton. 36 –38 As an abundant lignocellulosic bioresource with a lignin content of 14–20%, it has the potential to be modified by this green biotechnology for endowing it with better performance or novel functions. 39,40 In our former work, the surface hydrophobization of jute fabrics via the laccase-mediated grafting reactions has been made to increase their interfacial adhesion with the nonpolar resins in fiber-reinforced polymer composites. 40,41 However, as far as our knowledge goes, the laccase-facilitated self-crosslinking of lignins has not seemingly been attempted for improving the surface and bulk properties of the jute fabrics to increase the reinforcing ability with the resin matrix.
In the present work, lignocellulosic jute fabrics were treated by laccase and then the modified jute was used as the reinforced materials to prepare polypropylene (PP) matrix composites. Initially, the surface elemental composition and morphology of the jute fabrics were characterized by X-ray photoelectron spectroscopy (XPS), scanning electron microscope (SEM), and atomic force microscope (AFM). Then, the surface hydrophobicity and tensile properties of the jute fabrics were measured. Lastly, the tensile properties, shear properties, dynamic mechanical properties, and fracture surface of the jute fabric/PP composites were investigated.
Experimental
Materials and reagents
ABTS and laccase from Trametes versicolor were provided by Sigma-Aldrich (Shanghai, China). The 100% raw jute fabrics (427 g m−2 in mass density, 7/7 (warp/weft) cm−1 in areal density, and 254 Tex in thread density) were supplied by Longtai Weaving Co., Ltd (Changshu, China). PP spunlace non-woven cloths (54 g m−2) were purchased from Yonghui Textile Technology Co. Ltd (Yangzhou, China). All the other chemicals used in the study were commercially available and of analytical purity grade.
Laccase assay
The activity of laccase was measured using a UV-1800 UV/Vis spectrophotometer (Shimadzu, Japan) by monitoring the oxidation of ABTS (ε 420 = 36,000 M−1 × cm−1) as a substrate at 420 nm in pH 4 acetate buffer (0.2 M) at 50°C. The enzyme activity was expressed in units defined as micromoles of ABTS oxidized per minute. 42
Pretreatment of jute fabrics
The jute fabrics were Soxhlet extracted with benzene/ethanol (2:1, v:v) for 12 h to remove lipophilic extractives and then boiled with distilled water for 3 h to remove water-soluble fractions. As a result, the oxidation of substrate molecules by laccase and analysis of modified fibers could be carried out without interference.
Treatment of jute fabrics with laccase
Jute fabrics of 1 g were incubated in 50 mL 0.2 M acetate buffer (pH 4) with 2.0 U/mL laccase in a shaking bath. The reaction was allowed to proceed at 50°C for 4 h. Control samples without laccase followed the same treatment condition as mentioned above. Afterwards, the jute fabrics were washed with distilled water at 50°C for 30 min twice and then air dried.
XPS analysis
XPS experiments of jute fabrics were carried out on an Amicus spectrophotometer (Kratos Analytical Ltd, UK) with magnesium Kα radiation. The X-ray anode was run at 180 W, and the high voltage was kept at 12.0 kV. The pass energy was fixed at 75 eV to ensure sufficient resolution and sensitivity. The base pressure of the analyzer chamber was about 1 × 10−6 Pa. The sample was directly pressed to a self-supported disk (10 × 10 mm) and mounted on a sample holder then transferred into the analyzer chamber. Both the whole spectra (0–1100 eV) and the narrow spectra of carbon with much high resolution were recorded. Binding energies were calibrated with the containment carbon (C1s = 284.6 eV). The spectrum analysis including background subtraction, deconvolution, integration, and quantitation was performed by using the XPS Peak4.1 software.
SEM analysis
The surfaces of the jute fabric samples were scanned using a SU1510 SEM (Hitachi, Japan) under 5.00 k voltages at 1.00 k and 5.00 k magnifications.
AFM analysis
The three-dimensional surface topography and root-mean square values of the jute fiber samples were examined using a CSPM 4000 AFM made by Benyuan Co., Ltd. (Beijing, China). The fibers were immobilized on the magnetic AFM sample stubs by the help of a double-sided tape and the scanning was carried out in tapping mode AFM with a silicon cantilever at room temperature in the atmosphere. The scanning was set at a size of 5000 × 5000 nm2, and the scanning frequency was set at 2.0 Hz.
Hydrophobicity measurements
The hydrophobicity of the jute fabrics was estimated by contact angle and wetting time. Before the measurements, jute fabric samples were conditioned to moisture equilibrium in a constant temperature and humidity environment (21 ± 1°C, 65 ± 5% relative humidity). Then, the contact angle of water was measured in 3 s after drop deposition using a JC2000D4 contact angle meter (Zhongchen, Shanghai). For each sample, five spots were measured, and the results were averaged. The wetting time was determined according to the AATCC Test Method 79-2007. A drop of water was allowed to fall from a height of 10 ± 1 mm onto the taut surface of the test fabric. The time required for the specular reflectance of the water drop to disappear was measured and recorded as the wetting time. Five readings were taken and averaged for each sample.
Tensile measurement of jute fabrics
Jute fabrics were cut into pieces of 200 mm in length and 50 mm in width. Then, the tensile properties were determined on a YG (B) 026D-250 electronic tensile testing machine (Textile Standard Instrument Co. Ltd, China). The testing speed was 100 mm min−1. Data of breaking strength and elongation were recorded. All measurements were performed using at least five samples.
Preparation of jute fabric/PP composites
First, jute fabrics and PP foils were cut into samples of 20 mm width and 100 mm length. Then, the jute fabric/PP complex was prepared by successive laying of jute fabrics and PP foils in a mass ratio of 1:1 and was placed in the steel mould with a dimension of exactly 20 mm width and 100 mm length (shown in Figure 1(b)). Finally, the filled mould was covered by steel plates (seen in Figure 1(c)) on the double sides and transferred onto the molding press machine (shown in Figure 1(a)). The composite samples were compressed at 180°C in 10 ton pressure for 10 min. Composites with two layers of jute fabrics and three layers of PP foils prepared in the 1-mm-thick mold were used for the tensile measurement, dynamic mechanical analysis (DMA), and fracture analysis. Composites with four layers of jute fabrics and five layers of PP foils prepared in the 2-mm-thick mould were used for the shear measurement.

Equipments for the preparation of jute fabric/PP composites: (a) molding press machine, (b) steel mould, and (c) steel plate. PP: polypropylene.
Tensile measurement of jute fabric/PP composites
The tensile properties of the jute fabric/PP composites were determined using a 3385 H microcomputer-controlled electronic universal testing machine (Instron Corporation, Norwood, Massachusetts, USA). The composite samples had a dimension of 100 mm (length) × 20 mm (width). The gauge length was set at 60 mm and the testing speed was 2 mm min−1. A stress–strain curve of the composites and the data of breaking strength, modulus, and elongation were recorded. All measurements were performed using at least five samples.
Shear measurement of jute fabric/PP composites
The shear properties of the jute fabric/PP composites were determined by three-point bending techniques on a 3385 H microcomputer-controlled electronic universal testing machine (Instron Corporation). The composite samples had a dimension of 30 mm (length) × 20 mm (width). The testing speed was 20 mm/min. A stress–strain curve of the composites and the data of breaking strength, modulus, and elongation were recorded. All measurements were performed using at least five samples.
DMA of jute fabric/PP composites
A Q800 DMA analyzer (TA Instruments, New Castle, Pennsylvania, USA) was used to evaluate the dynamic mechanical properties of the jute fabric/PP composites. A three-point bending mode was employed. The samples were tested in a fixed frequency of 10 Hz and a heating rate of 2°C min−1 from 30°C to 170°C. Each sample had a thickness of 1 mm, a width of 13 mm, and a length of 60 mm.
Fracture analysis of jute fabric/PP composites
The fracture sections of the jute fabric/PP composites were scanned using a SU1510 SEM (Hitachi) under 5.00 k voltages at 1.00 k magnification.
Results and discussion
XPS analysis of jute fabrics
XPS spectroscopy was employed to understand the changes in the surface chemistry of jute fabrics after the laccase treatment. The deconvolution of C1s high-resolution signals in the XPS spectra was carried out by curve fitting to obtain the bonding states of carbon atoms on the jute surface. As shown in Figure 2, the C1s spectra were deconvoluted into five peaks, C1, C2, C3, C4, and C5. According to the literature, 43 the C5 peak with the lowest binding energy originates from a carbon atom bound only to carbon or hydrogen atoms (–C–C/–C–H). The C4 peak represents a carbon atom linked to an ether oxygen atom (–C–O–C). The C3 peak corresponds to a carbon atom bound to a hydroxyl oxygen atom (–C–O–H). The C2 peak represents a carbon atom linked to two non-carbonyl oxygen atoms (O–C–O). The C1 peak with the highest binding energy represents a carbon atom bound to one carbonyl oxygen atom (–C=O). The C2 component only exists in cellulose and hemicellulose of the jute fibers, while the C1 component can only be found in the structure of lignin or pectin.
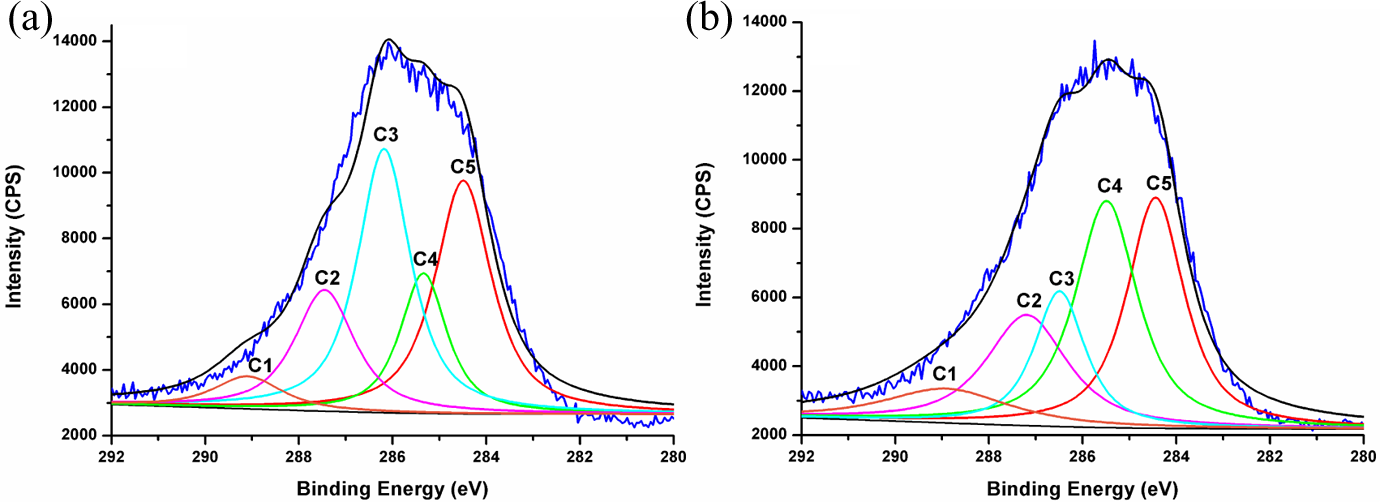
C1s peaks of control (a) and laccase treated (b) jute fabric samples.
The proportions and binding energy positions of these groups are given in Table 1. A decrease in the C3 peak intensity from 32.42% to 14.48% was observed after the laccase treatment, indicating the decline of hydroxyl groups on the jute surface. Meanwhile, the C4 peak intensity was increased from 15.13% to 29.63%. This can be attributed to the laccase-mediated oxidation of phenolic hydroxyl groups in lignins on the jute surface and the coupling of the produced phenoxyl radicals to form ether structures. The atomic bonding results and speculation are also in accordance with the structure changes of lignin treated by laccase. 20,26
Binding energy and area of C1s on the surfaces of control and laccase-treated jute fabrics.
SEM and AFM analysis of jute fabrics
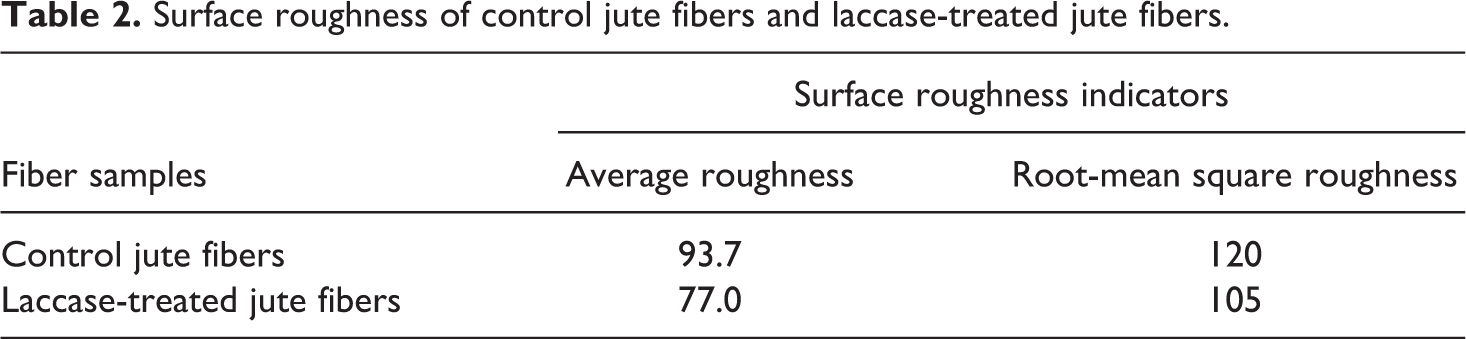
The changes in the surface morphology and topography of the control and laccase-treated jute fabrics were studied by SEM and AFM, respectively. The control jute fiber surface was rough and irregularly covered with numbers of lamelleted impurities (Figure 3(a) and (b)) and bulky convex under a greater magnification (Figure 4(a)). As the surface of jute fibers is mainly composed of middle lamella and primary cell wall, these components are considered as the mixture of lignin, hemicellulose, and pectin. 44 After the laccase treatment, the jute fiber surface became smooth and neat (Figure 3(c) and (d)) with lignin, and so on attached tightly. In the AFM image (Figure 4(b)), the natural convex turned to be smaller and the surface roughness of the jute fibers was decreased as shown in Table 2. This is probably because of the enzymatic dislodgement and redistribution of the bulgy lignins on the surface. At the beginning of the laccase treatment, some lignins on the jute fibers were degraded to low-molecular-weight pieces and dissolved into the aqueous medium. In the later stage of the reaction, the laccase-mediated polymerization of lignin became predominant, and the dissolved lignins were covalently attached back to the jute surface. 26 The degradation and subsequent polymerization of lignin by laccase redistributed the lignins on the jute surface and made the jute surface glossy.

SEM images of control jute fiber surfaces (a and b) and laccase-treated jute fiber surfaces (c and d) amplified at 1.00 k and 5.00 k, respectively. SEM: scanning electron microscope.
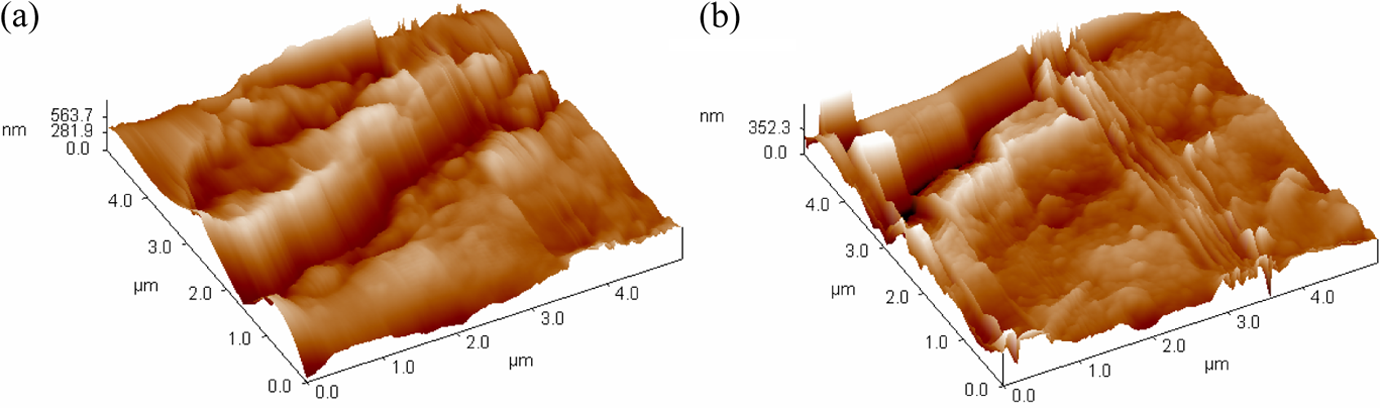
AFM images of control jute fiber surface (a) and laccase-treated jute fiber surface (b). AFM: atomic force microscope.
Surface roughness of control jute fibers and laccase-treated jute fibers.
Effect of laccase treatment on the surface hydrophobicity of jute fabrics
Wettability of plant fibers is an important factor for the interfacial adhesion with the hydrophobic resin matrix in composites as well as the water absorption behavior of the composites. The hydrophilic characteristics of jute fibers lead to poor wettability and weak interfacial bonding with resins and facilitate the intake of water when the composites are immersed in water, finally resulting in decreased mechanical and physical properties of the composites. 45 In order to evaluate the surface hydrophobicity of jute fabrics after the laccase treatment, the contact angle and wetting time of water were measured. The corresponding data are listed in Table 3. For the control jute fabrics, the contact angle is 98.7° and the water droplets on the surface disappeared with no specular reflectance in 22 s. In contrast, the laccase-treated samples showed increased contact angle of 109.2° and wetting time of 79.4 s in average. The increase in the surface hydrophobicity of jute fabrics after the laccase treatment was the results of oxidation of phenolic hydroxyl groups and formation of ethers by radical coupling, which have been proved by the XPS analysis.
Contact angle and wetting time of jute fabrics after control and laccase treatments.

Effect of laccase treatment on the tensile properties of jute fabrics
The properties of fiber-reinforced composites depend largely on the mechanical characteristics of the fiber reinforcements. The tensile strength and elongation of the control jute fabrics and laccase-treated jute fabrics were tested and shown in Figures 5 and 6. After the laccase treatment, the breaking strength of jute fabrics was increased by 4.9% in broad-wise and by 2.6% in warp-wise. By contrast, the breaking elongation increased more remarkably by 173% in broad-wise and by 151.7% in warp-wise. The coupling of lignins on the jute surface by laccase improved the combination between fibers and resulted in an enhancement in the tensile properties of jute fabrics. The possible reasons for the increase of elongation were that the cohesive force between fibers in jute yarns was decreased due to the smooth fiber surface after laccase treatment.
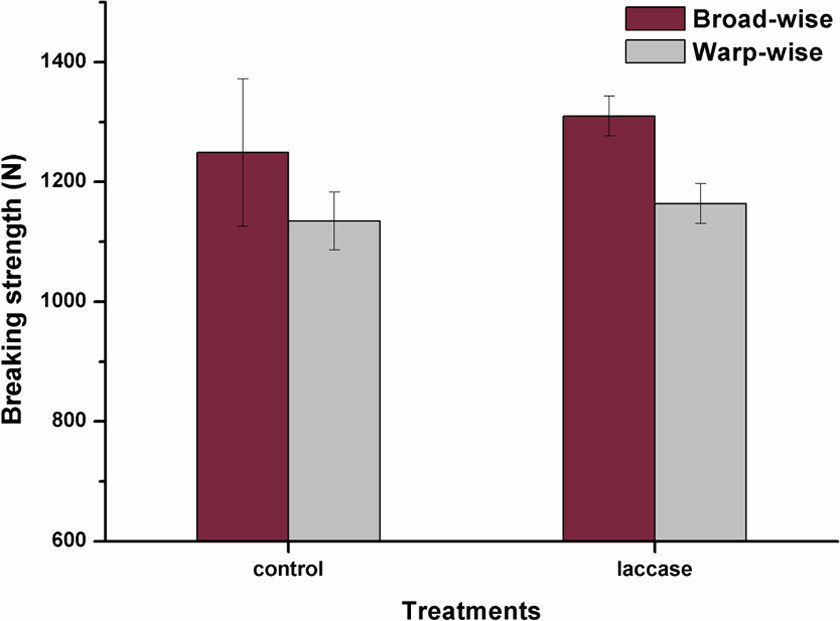
Breaking strength of control jute fabrics and laccase-treated jute fabrics.
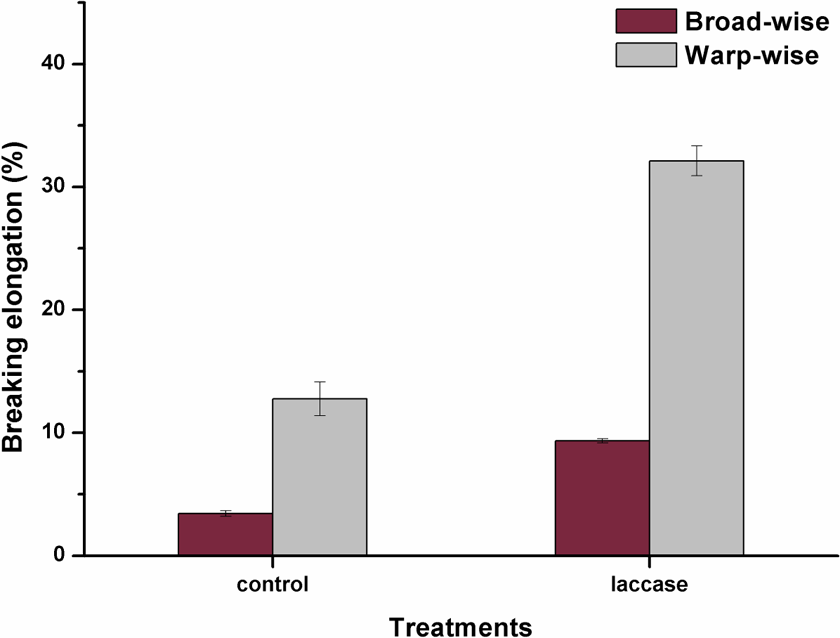
Breaking elongation of control jute fabrics and laccase-treated jute fabrics.
Interfacial behavior of jute fabric/PP composites
The fracture surfaces of the jute fabric-reinforced PP composites were produced by stretching on a universal testing machine and then observed by SEM. The micrographs of the fracture sections are displayed in Figure 7. In the case of the control jute fabric/PP composites (Figure 7(a)), jute fibers were pulled out of the polymer matrix during stretching the sample and the tips of fibers were protruded from the resins, which made the fracture surface of the composites uneven and irregular. For the laccase-treated jute fabric/PP composites (Figure 7(b)), PP matrix surrounding jute fibers was pulled out together with the fibers. Jute fibers were well adhered with PP resins to form a neat and smooth fracture surface, indicating stronger interfacial adhesion in the composites and adequate wetting of the modified fibers with PP resins. Therefore, it could be concluded that the modified jute fibers were better coated by PP and stronger fiber–matrix interaction was obtained owing to the laccase treatment of the jute surface.

SEM images of fracture sections of control jute fabric/PP composites (a) and laccase-treated jute fabric/PP composites (b). SEM: scanning electron microscope; PP: polypropylene.
Tensile and shear properties of jute fabric/PP composites
The tensile and shear properties of the jute fabric/PP composites were measured to estimate the effect of changes of jute fabrics caused by the laccase treatment on the mechanical behaviors of the composites. The breaking strength, Young’s modulus, and breaking elongation of the tensile and shear measurements are shown in Tables 4 and 5, respectively. After the fiber treatment, the breaking strength of the jute fabric/PP composites was increased by 4.5% in tensile behavior and by 14.7% in shear behavior. Young’s modulus showed a larger increase with 18.2% in tensile behavior and 32.6% in shear behavior. Meanwhile, the breaking elongation was decreased by 2.9% in tensile behavior and by 12.8% in shear behavior. The property improvement of the jute fabric/PP composites relied on several aspects. Firstly, the laccase treatment enhanced the surface hydrophobicity of the jute fabrics, which improved the interfacial compatibility with the resin matrix in composites. Secondly, the mechanical properties of the jute fabrics were increased via the laccase treatment. Lastly, the surface components of the jute fabrics (mainly lignin and hemicellulose) attached more tightly after the laccase treatment. Thus, the connection between the resin matrix, jute surface, and fiber body can be increased.
Tensile properties of control jute fabric/PP composites and laccase-treated jute fabric/PP composites.

PP: polypropylene.
Shear properties of control jute fabric/PP composites and laccase-treated jute fabric/PP composites.
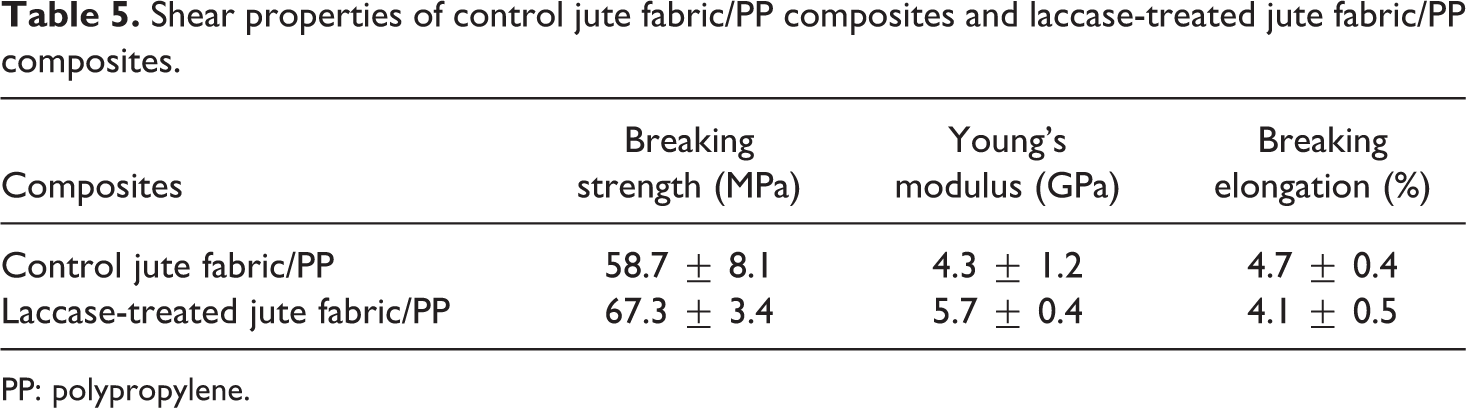
PP: polypropylene.
Dynamic mechanical properties of jute fabric/PP composites
DMA can provide reliable information over the relaxation behavior of the materials examined. 46 In order to estimate the influence of the laccase treatment of jute on the fiber/matrix interfacial adhesion, dynamic thermo-mechanical properties of the control jute fabric/PP composites and the laccase-treated jute fabric/PP composites were compared. The storage modulus (E′), loss modulus (E′′) and tan δ curves as a function of temperature are illustrated in Figure 8. E′ at 50°C and 170°C as well as the melting temperature (T m) obtained from tan δ curves are listed in Table 6.
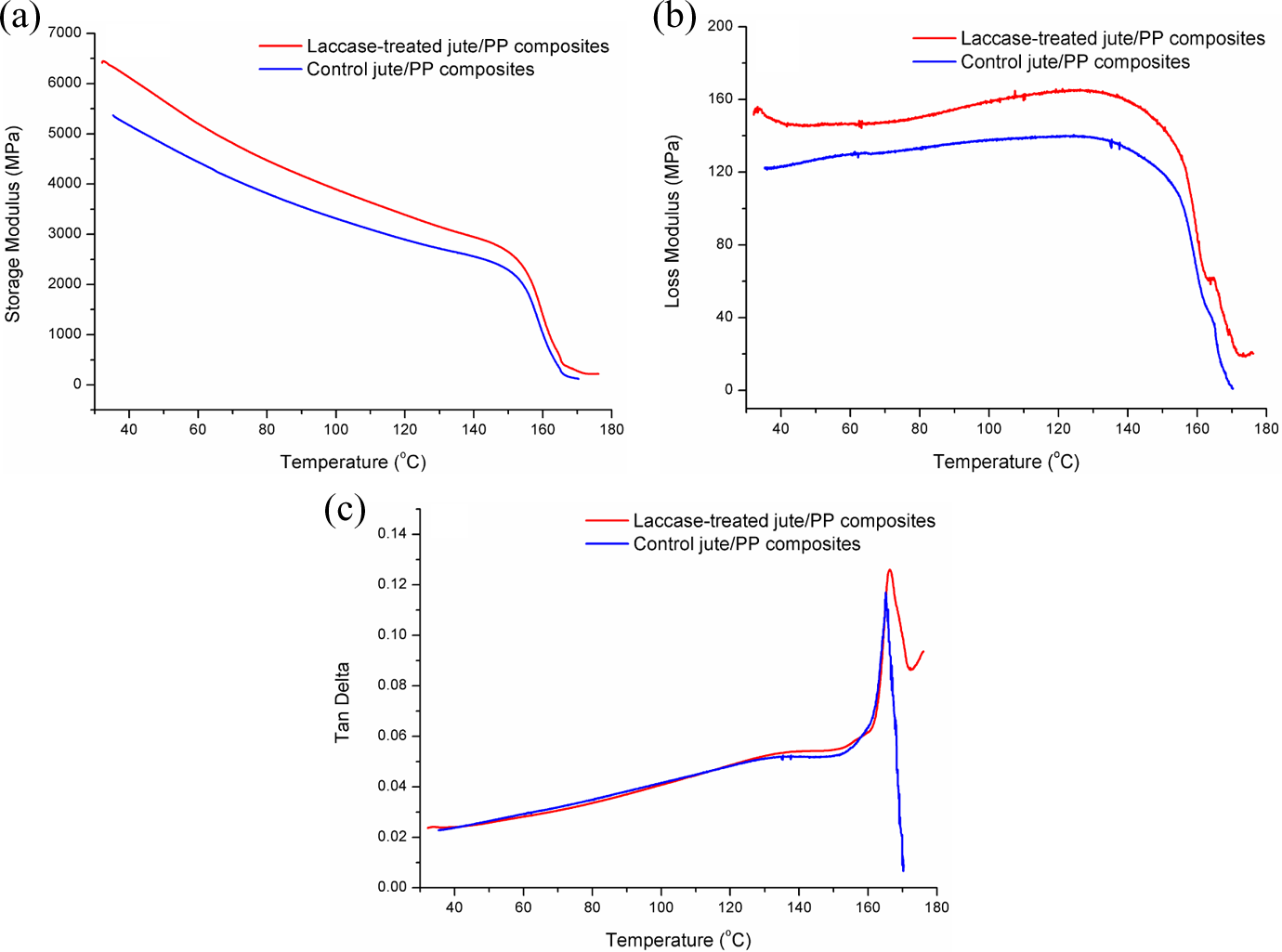
Dynamic mechanical curves of control jute fabric/PP composites and laccase-treated jute fabric/PP composites. PP: polypropylene.
Storage modulus and melting temperature of control jute fabric/PP composites and laccase-treated jute fabric/PP composites.
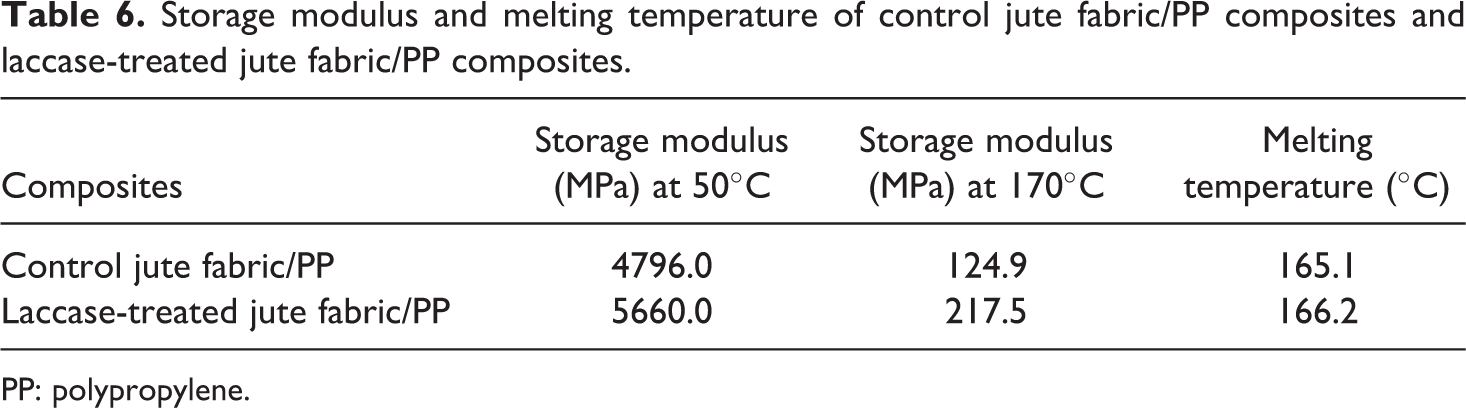
PP: polypropylene.
E′ is useful in assessing the molecular basis of the mechanical properties of materials because it is very sensitive to structural changes, such as fiber–matrix interfacial bonding. 47 As shown in Figure 8(a), the E′ values of all the composites were dropped with the increase of temperature. However, the PP composites reinforced by laccase-treated jute fabrics had a significant increase in the E′ over the whole range of testing temperatures in contrast with the control composites. The E′ of the laccase-treated jute fabric/PP composites is 5660 MPa at 50°C and 217.5 MPa at 170°C, while that of the control jute fabric/PP composites is only 4796 MPa at 50°C and 124.9 MPa at 170°C. This can be explained as the result of improved interfacial adhesion between the modified jute and the PP matrix, which allowed greater stress to transfer at the interface.
E′′ is proportional to the amount of energy that dissipated as heat by the material. Tan δ, the damping energy ratio, is an indication of the molecular motions of resins in composites. 48 The temperature where tan δ was the maximum indicates the T m of the composites. As shown in Figure 8(b), the laccase-treated jute/PP composites had higher E′′ values ranging from the whole testing temperatures than the control. The tan δ values in Figure 8(c) increased gradually before 160°C and then reached maximum rapidly at the T m. After the enzymatic treatment of jute fabrics, the T m of the jute fabric/PP composites shifted from 165.1°C (control) to higher temperature (166.2°C). E′′ and T m is attributed to the mobility of resin molecules. Larger E′′ and T m suggest a stronger restraining effect of fibers on the mobility of the resin matrix. 49 The corresponding increase in E′′ and T m indicated enhanced restriction after the laccase treatment of fibers and immobilization of polymer molecules near the modified jute fiber surface. The improved interfacial adhesion reduced the mobility of PP molecular chains at the interface.
Conclusions
The present work demonstrated that the laccase treatment of jute fabrics resulted in increases in the surface hydrophobicity and tensile properties of jute fabrics as well as the mechanical and interfacial properties of jute fabric/PP composites. The content of phenolic hydroxyl groups on the jute fabric surface was decreased after the laccase treatment, while the content of ether structures was increased. Thus, the modified jute fabrics obtained enhanced surface hydrophobicity with a contact angle of 109.2o and a wetting time of 79.4 s. Meanwhile, the breaking elongation of jute fabrics was increased by 173% (broad-wise) and 151.7% (warp-wise). In addition, the surface of jute fabrics treated by laccase became smooth with lignin adhered tightly. For the laccase-treated jute fabric/PP composites, the tensile and shear breaking strengths, storage modulus, as well as melting temperature were all increased. The fracture surface of the composites became neat and the jute fibers on the section surface were surrounded by PP resins closely, which suggested better interfacial adhesion between the jute reinforcement and the PP matrix.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundations of China (51603087 and 51673087); Program for Changjiang Scholars and Innovative Research Team in University (IRT_15R26), and Fundamental Research Funds for the Central Universities (JUSRP51717A).
