Abstract
The goal of this work was to synthesize and characterize ternary rubber blends based on polyisoprene (natural rubber (NR)), polybutadiene rubber (BR), and styrene–butadiene rubber (SBR) (NR/BR/SBR = 25/25/50) reinforced with different loading silica (SiO2) nanoparticles (0–100 part per hundred parts of rubber (phr)). The specimens were subjected to thermooxidative aging at 100°C, for two times: at 72 and 168 h, respectively, and then mechanically stretched to fracture by tension with a Zwick 1425 (Zwick GmbH, Ulm, Germany) universal tensile testing machine. Rheological and mechanical properties were used as characterization of the ternary rubber blends. The reinforcing performance of the filler was investigated using rheometric, mechanical, and swelling measurements, thermogravimetric analysis, scanning electron microscopy, and Fourier transform infrared spectroscopy with attenuated total reflectance. Hardness, tensile strength, elongation at break, and swelling degree were assessed before and after thermal aging. There was a remarkable decrease in the optimum cure time (t c90) and the scorch time (t s2), which was associated with a decrease in the cure rate index of (NR/BR/SBR = 25/25/50) ternary rubber blend with 60 phr of filler loading. Interaction between rubber blend and SiO2 nano-filler is confirmed by moving absorption band from 1450 cm−1 to 1480 cm−1.
Introduction
Elastomeric blends are frequently used in the rubber industry to obtain desirable physical properties, process ability, and lower cost. 1 Blending of rubbers also enhances the physical properties of the final vulcanized product. 2 The properties of polymer blends can be controlled by regulating blend morphology, blend compositions, and processing condition. 3 Normally, rubbers are vulcanized by systems based on sulfur or peroxide. The common feature of these systems is that they all require activation energy in the form of heat. This heat (150–180°C) may affect the final properties of the product by a variety of uncontrolled side reactions. 4
Natural rubber is also commonly used, and it has certain advantages, such as flexibility. Nevertheless, some of its properties fall short in certain applications, such as oil resistance, air permeability, ozone resistance, compression set, and thermal aging resistance. Several studies have already dealt with the blends of natural rubber (NR) and different rubber combinations. 5,6 Hamed and Zhao 7 studied the physical effects of NR/styrene–butadiene rubber (SBR) after thermal aging. Sirisinha et al. 8 explored NR/NBR viscoelasticity and oil resistance, and Koshy et al. 9 studied the changes in NR/EVA physical and ozone resistance properties after thermal aging.
Polybutadiene is classified as general-purpose rubbers intended for the manufacture of tires and general mechanical products. However, one type of rubber may not possess all the physical properties desired in a finished product. Much attention is currently being devoted to the simplest route for combining the outstanding properties of different existing polymers, that is, the formation of polymer blends. 10 For example, in tread compounds, the high abrasion resistance under certain conditions conferred by the use of BR is desirable, but the poor road holding and rib tearing properties are not; hence, blends of polybutadiene rubber (BR) with NR and SBR are employed. Compatibility is the fundamental property, deciding the practical utility of a polymer blend. If the two elastomers in a blend are incompatible, it will exist in the form of two separate phases and the cured blend will show inferior properties. In addition, there have been several reports on NR/BR rubber blends 11 mainly dealing with the physical properties of NR/BR blends such as wear resistance, bending resistance, tensile strength, and compression stiffness. Several researchers have also explored the effect of carbon blacks or additives on NR/BR blends 12 –14 and analyzed the changes in properties of NR/BR blends after thermal aging. Also, Mallik and Shah 15 studied the thermal aging effect on the physical properties of NR/BR blends at different blending ratios. Using thermal analyses (thermogravimetric analysis (TGA), derivative thermogravimetric analysis (DTG), and differential thermal analysis (DTA)), Budrugeac 16 explored the reactions of heat and heat resistance in nonisothermal conditions. Meanwhile, Ciutacu et al. 17 used infrared (IR) spectrometry methods to investigate the molecular chain reactions of blends at higher temperatures.
SBR is widely used as one of the components of the elastomeric matrix for automotive tires, wire, and cable applications due to its high fracture elongation, but unfortunately it has low elastic modulus and durability. In other words, SBR is not smooth or homogeneous and has poor physical properties, which means that it needs some additives such as antioxidants, accelerators, softeners, and fillers to improve its properties. 18
The reinforcement of rubbers is expressed by enhancement of the modulus, mechanical properties, and abrasion resistance of the vulcanizates. The main aim for filler addition is to improve certain properties and cheapen the compound. In the last two decades, research was focused on the development of other reinforcing agents to replace carbon black in rubber compounds. Nanocomposites exhibit greatly improved mechanical and thermal properties because of the nanometer-scale dispersion of the reinforcements and the high surface-to volume ratio. 19 The phenomenon of reinforcement of elastomers is of great importance for the structuring of materials in new technologies. Namely, adding the nanoparticle fillers in the elastomers obtained from different types of rubber leads to an increase in the modulus of elasticity and to an improvement of key properties such as tensile strength and elongation, as well as abrasion resistance. The surface hydroxyl groups of silica including isolated, vicinal, and geminal silanols are believed to play a key role in most of the aforementioned properties. 20 The presence of silanol groups on the silica surface increases particle–particle interaction which tends to favor agglomeration of filler and decreases rubber–filler interactions in the most rubber matrix. However, silica has been widely used in rubber industry, particularly in tire applications because it provides several advantages over carbon black, for instance, the improvement in rolling resistance, wear resistance, and wet traction when it is used in combination with silane coupling agent.
Adding nanoparticles influences the regime of cross-linking as well, especially in some types of precursor cross-linking. Reinforcement of elastomers is a particularly complicated process if cross-linked material contains more than one type of precursor cross-linking. By creating a multiphase system, characteristics of individual phases can be partly preserved or significantly changed due to the influence of intermolecular interaction. Therefore, the modern research and industrial practice of tire industry leaders are directed toward the use of existing starting polymers and the obtainment of new types of elastomeric materials with new modified macromolecules. 21 The cross-linking and chain degradation are dependent on chains structural characteristics and the presence of some curing system. A pronounced increase in the level of reinforcement of elastomers is expected. Materials which have a satisfactory thermal stability and mechanical properties required in the specific working conditions are obtained by selecting the optimum content of the filler.
The objective of this study is to synthesize and characterize nanosilica-filled (NR/BR/SBR = 25/25/50) ternary rubber blends. The effects of aging are measured in terms of the changes in tensile strength, elongation at break, and hardness. Rheometric characteristics and mechanical and swelling properties of nanocomposites are assessed before and after thermal aging.
Experimental
Materials
Polyisoprene rubber, NR SMR-20 (0.92 g/cm3) was supplied by Astlett Rubber Inc. (Oakville, Canada); BR SKD N (0.91 g/cm3), with 94% of 1,4-cis content, mooney viscosity Ml (1 + 4) 100°C = 44 M was supplied by Njižnjekamsk (Russia); SBR Europa Intol 1783 (density = 0.94 g/cm3), which is an emulsion SBR with 23.5% bound styrene, obtained by cold polymerization extended with 37.5% RAE oil, was supplied by Versalis (Italy). News 175G, Wuxi (China) (ρ = 2.0 g/cm3) was used as nanosilica filler with 22 nm average size of primary particles. Content of filler was 0, 20, 40, 60, 80, and 100 part per hundred parts of rubber (phr).
The curing system was N-cyclohexyl-2-benzothiazolsulfonamide-CBS (1.4 phr), diphenyl guanidine (1 phr), N-(cyclohexylthio)phthalimide, CTP 100 (0.2 phr), and sulfur (2 phr). In all rubber blend compounds, the network precursor ratio was 25/25/50 (w/w/w). Content of other ingredients were: zinc oxide 3 phr, stearic acid 2 phr and naphthenic oil as plastificator 10 phr. Formulations of the rubber compounds are shown in Table 1.
Formulation of ternary NR/BR/SBR (25/25/50) rubber blend reinforced with SiO2.
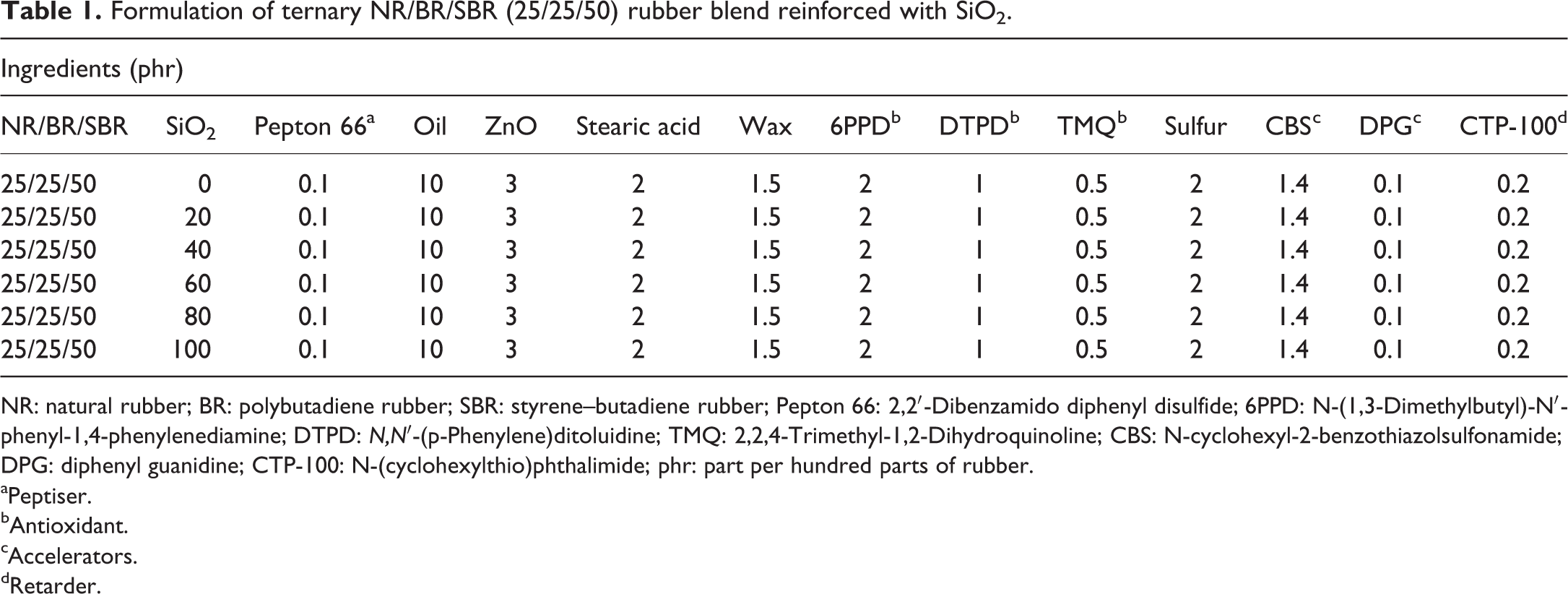
NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; Pepton 66: 2,2′-Dibenzamido diphenyl disulfide; 6PPD: N-(1,3-Dimethylbutyl)-N′-phenyl-1,4-phenylenediamine; DTPD: N,N′-(p-Phenylene)ditoluidine; TMQ: 2,2,4-Trimethyl-1,2-Dihydroquinoline; CBS: N-cyclohexyl-2-benzothiazolsulfonamide; DPG: diphenyl guanidine; CTP-100: N-(cyclohexylthio)phthalimide; phr: part per hundred parts of rubber.
aPeptiser.
bAntioxidant.
cAccelerators.
dRetarder.
Compounding and cross-linking
All samples are mixed in a laboratory mixer K-0 INTERMIX (Francis Shaw, Bradford, Manchester, UK), volume 1 L, rotor speed of 8–88 r/min, and laboratory roll mill (14201-Buzuluk, Komarov Czech Republic) dimension 400 mm × 200 mm at a speed of rollers n 1/n 2 = 17.4/14 and a temperature between 60°C and 70°C, according to the procedure ASTM D318489. Compounds were conditioned at 25°C during 24 h prior to property assessments. Rheometer curves were recorded using an Alpha technologist (Akron, Ohio, USA) Rheometer MDR 2000 at 160°C and are used to determine the start of cross-linking t s2, optimum of cross-linking t C90, as well as the maximum and minimum torques (M max and M min). 22 The cross-linking was carried out in an electrically heated hydraulic press (E-604 Metroohm, Herisau, Switzerland) under a pressure of 20 MPa and 150°C. The scorch time (t s2), the optimum cure time (t c90), the cure rate index (CRI), and maximum torque (M max) were determined from rheometer data.
Sample characterizations
Tensile strength and elongation at break were measured using dumbbell-type specimens according to ASTM D41298a. Tensile tests were carried out on a Zwick 1425 universal tensile testing machine at room temperature. Hardness tests were carried out using Shore A Durometer (Zwick GmbH, Ulm, Germany) according to ASTM D2240. The percentage of abrasion resistance was measured according to DIN5356. The given results are the mean value of three specimens. The error in these measurements was ±0.5%.
Thermal aging of elastomeric composites samples
The effect of thermal aging on the mechanical properties of the unfilled and filled ternary NR/BR/SBR blend was studied by keeping the dumbbell samples in an air oven at 100°C for 72 and 168 h, respectively. The NR/BR/SBR samples were then conditioned at ambient temperature for 24 h before testing according to ASTM D573. The changes in tensile strength, elongation at break, and hardness before and after the thermal aging conditions were then evaluated. Three samples were tested for each set of experiments. The aging resistance is expressed as a percentage of retention in tensile properties calculated by the following equation 23 :
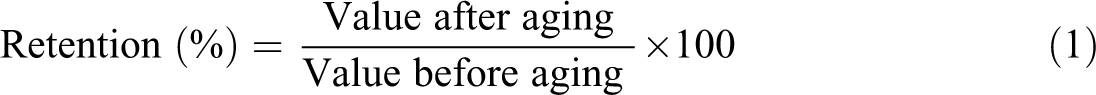
Swelling measurements
The investigated rubber composites were obtained as small sheets (1 g) weighed accurately and immersed in 25 cm3 benzene solvent in closed simple bottles at 25°C. After 72 h, the samples were removed from the solvent and weighed in a glass stopper bottle after removing the surface fluids by blotting with a filter paper. The volume swelling in percentage was calculated using the following equation:
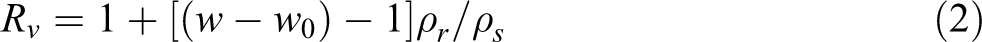


where Rv and Rw are the volume and mass ratio of rubber samples in the swollen gel, respectively; w and wo are weights of the sample before and after swelling, respectively; ρr and ρs are densities of the rubber and the solvent, respectively; and v 2 is the volume fraction of the sample in the swollen gel.
The elastic modulus (E) values were obtained on dumbbell samples by stress–strain measurements. 24 The values of E were obtained from the initial slope of the curves in neo-Hook’s stress–strain little Greek letter sigma relations versus (α−α − 2) in the following equation:

where α is the elongation ratio (L/Lo ).
The E modulus is related to cross-link density by the well-known equation from the kinetic theory of elasticity

where the factor g < 1, ν is concentration of cross-link density, R is the universal gas constant, and T is the absolute temperature


where
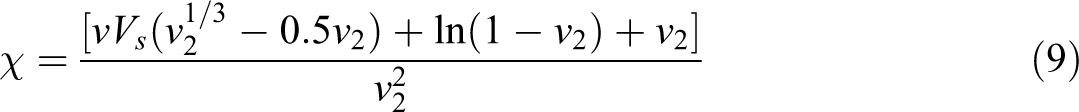
where Vs is the molar volume of the benzene solvent.
The cross-link density was determined from the swelling data. The samples were allowed to swell in the benzene solvent and the equilibrium uptake was noted. The molecular weight between the cross-link
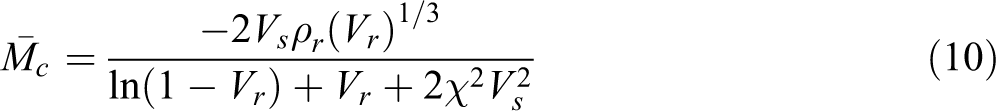
where
Equilibrium swelling was carried out on a uniform circular cut from the obtained sheets (5 mm diameter and 2 mm thick) by immersing in toluene at room temperature. The samples were allowed to swell for 2 days until the equilibrium was reached. Then, the test pieces were taken out and the solvent was rapidly removed by blot ting with filter paper. The swelling degree Q was calculated as

where m and mo are the weights of the specimen before and after swelling, respectively; ρ 2 and ρ 1 are the density of the cross-linked materials and solvent, respectively. The volume fraction of the samples at swelling equilibrium was used to determine the overall cross-linking density v in obtained materials.
Thermal analysis
The thermal stability was investigated by nonisothermal thermogravimetry (TG, DTA) using a Setaram Setsys Evolution 1750 instrument (France). The heating was carried out at temperature range from room temperature to 700°C with a heating rate of 20°C/min under nitrogen gas atmosphere with the gas flow rate of 20 cm3/min. The weight of samples ranged from 5 mg to 8 mg. The temperatures at maximum decomposition rate were determined from the peak maxima of the DTG curves.
Scanning electron microscopy
Samples were immersed in liquid nitrogen for more than 15 min to cool down and then fractured immediately. The dried samples were sputter coated with gold prior to scanning electron microscopy (SEM), model JEOL JSM 5300 (USA), examination. The samples were sputter coated with gold for 3 min under high vacuum with image magnifications of 2000×.
ATR-IR spectroscopy
Spectra were collected at room temperature in a Thermo Nicolet 380 Fourier transform infrared (FTIR) spectrophotometer (Thermo-Nicolet, USA) equipped with an overhead attenuated total reflection accessory (ATR). Sixty-four scans were recorded for each spectrum in the spectral region between 4000 cm− 1 and 400 cm− 1 and nominal resolution of 4 cm− 1. Samples were applied on the surface of a diamond in a form of a powder and after measurement crystal was thoroughly washed. The cleaned crystal was carefully examined and checked with the background spectrum.
Results and discussion
The essence of cross-linking is a chemical reaction between the rubber macromolecules, which in the presence of selected fillers, softeners, activators, curing agents, accelerators, and retarders form a cross-link. Cross-linking of rubber macromolecules represents topologically critical phenomenon, when the ensemble of linear chains forms a three-dimensional cross-links of macromolecules. In order to design the raw composition of elastomeric material, which will have good properties, it is necessary to select a representative combination of cross-link percussion with structural and technological aspect. At the same time, bearing in mind the structure of elastomeric nanocomposites at different scales of observation cross-links and ensembles precursor silica aggregates, as active nanofillers, elastomer materials are obtained and then examined to check the characteristics needed for specific operating conditions and in terms of mechanical properties and aging of materials.
Cure characteristics
In the case of silica, however, strong filler/filler interactions resulting from polar surface functional groups such as siloxane 27 are believed to be primarily responsible for the increases recorded. It is interesting that the values of Mooney viscosities of some silica-filled rubber compounds at high loading of the filler have large increases. These workers ascribed the increases to strong filler/filler interaction of silica.
The cure characteristics as function of the silica loading of the ternary NR/BR/SBR (25/25/50) rubber blends are show in Table 2. The values for curing characteristics depend mainly on the nature of the network precursors employed in the blends. For filled compounds, type and content of filler affects the cure characteristics. 28,29
The cure characteristics of ternary NR/BR/SBR (25/25/50) rubber blends reinforced with nano-SiO2.
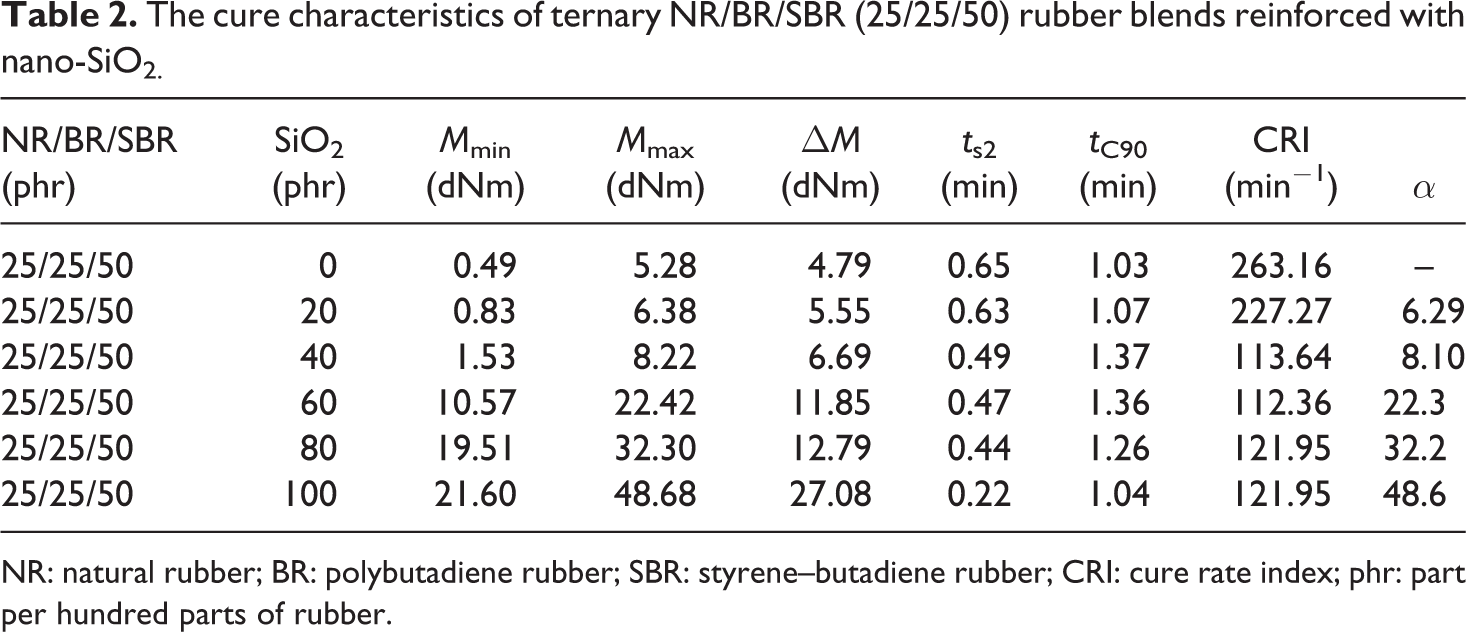
NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; CRI: cure rate index; phr: part per hundred parts of rubber.
Generally, acidic compounds retard the vulcanization of elastomers. For this reason, precipitated silica, which contains a large number of acidic silanol (Si–OH) groups, is not generally incorporated without an activator in NR. The surface of silica is acidic, and therefore, a strong hydrogen bond with polymer groups is formed. SBR rubber possesses phenyl groups, resulting in the hydrogen bonding of the silanol group of silica and a strong silica/SBR interaction. Silica particles also adsorb polar curative molecules on their surfaces, rendering the deactivation of vulcanization, and thus lower the curing state.
From the cure characteristics (Table 2) of the composites, it can be observed that the minimum torque M min, which is a measure of the viscosity and filler–filler inter-aggregations of the composites, increases with increase in the content of nano-SiO2 filler. Maximum torque M max, which is an indication of the shear modulus of the vulcanizates, increases with increase in the nano-SiO2 content. The increase in M max was attributed to the presence of silica nanoparticles in the matrix, which reduces the mobility of the molecules due to its hard particles and consequently increases the torque of the vulcanizates. 30 The scorch time (t s2) and optimum cure time values (t c90) of SiO2-filled composites are higher than unfilled. Due to the fact that the cure process of SBR rubber macromolecules is a slower compared to the NR and BR rubber macromolecules, a higher activation is needed to produce proper content of cross-link points. In addition, the limited efficiency of the accelerators used in the compound formulations can play a decisive role. The values during the scorch time of cross-linking (t s2) and optimum networking (t 90) with cured systems based on NR/BR/SBR ternary rubber blends decrease, with increase in nano-SiO2 content, that is, cross-linking process became faster. Lower values of the scorch time indicate that a “period dissolving” of the compound reduces, that is, cross-linking process starts sooner.
CRI was calculated according to the following equation:

It is obvious from data in Table 2 that CRI is higher for unfilled than nano-SiO2-filled ternary NR/BR/SBR rubber blend composites. For each system, the CRI decreases with increase in nano-SiO2 content. This influence supports the activation of the cure reaction up to the content of 60 phr. At higher loading, a deactivation of the cure process was observed. This can be explained by critical volume fraction of nano-SiO2 which has a tendency of agglomeration. Another procedure to evaluate the reinforcing potential of the filler is the use of α value as proposed by Wu and Tian, 31 where the following expression was employed:

where M
max and
The ratio between the increase in torque of the filled and unfilled compound was found to be directly proportional to the filler loading:
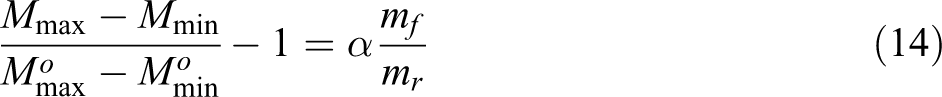
where M
max − M
min and
It can be concluded that the values of the minimum (M min) and maximum (M max) torque increase with increase in nano-SiO2 filler content. The values of M min, that is, resistance that provides a cross-linking system rotor rheometer, determine the safety of curing processing system at a given temperature.
Mechanical properties
Mechanical properties of nanocomposites generally depend on factors such as filler content, particle size and shape, the degree of adhesion between the filler and the polymer matrix, and the dispersion degree of the filler within the matrix.
32
Strengthening of the rubber reinforced with fillers changes the mechanical properties such as hardness, modulus, tensile strength, and so on. Interactions between macromolecules and filler particles occurred, where silica nanoparticles have a stronger interaction. This is reflected in the characteristics of cross-linked material and correlated with their ability reinforcements. The reinforcement of cross-linked materials is resulted as: particle size or specific surface area that, together with the content of the filler, determines the effective contact surface between the filler and the polymeric matrix; structure or filler particles degree of irregularity affecting the mobility of the macromolecular chain elastomers under deformations; and surface activity of the filler particles which affects the interaction of the filler–filler and filler–polymer.
Cross-linking of unsaturated rubber macromolecules with sulfur takes place through complicated radical substitutions to form the mono-, di-, or polysulfide bonds and also possible is internal cyclization of the polymer chains which contain sulfur. The degree of cross-linking and the distribution of cross-linking agent in the polymeric matrix affect the physical properties and stability to aging of the elastomer; however, they do not only depend on the type and content of sulfur and accelerators but also on the temperature and time of cross-linking. The values of the mechanical properties of cross-linked materials obtained from an NR/BR/SBR ternary rubber blend reinforced with silica nanoparticles are given in Table 3.
Mechanical properties of ternary NR/BR/SBR rubber blend reinforced with SiO2.
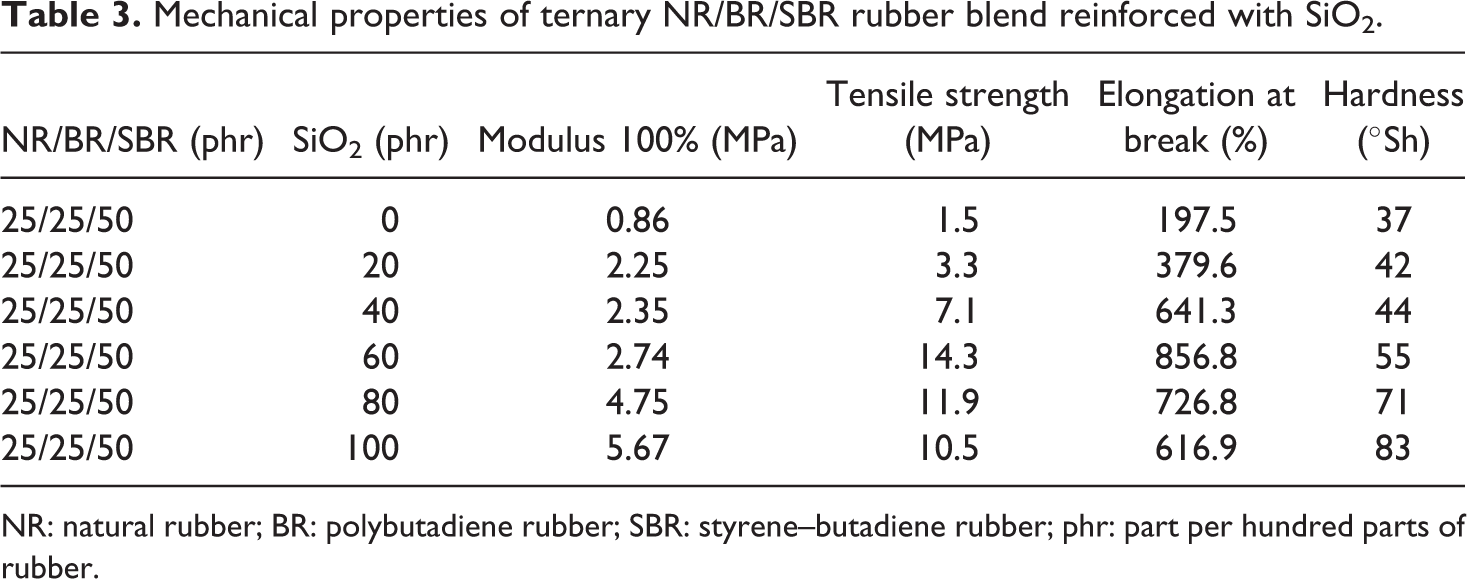
NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
Values of modulus and elongation at break are explained by the nature of filler (mainly the shape of the filler particles), but a very important factor is the bond strength between matrix and filler, which reduces the mobility of the polymer phase (better “wetting” of filler rubber macromolecules), the dispersion of fillers, and share of agglomerates of filler particles.
Tensile strength is a complex function consisting of the nature and type of cross-links, cross-link densities, and chemical structure of the used rubber. It is well known that if rubber is deformed by an external force, part of the input energy is stored elastically in the chains and is available (released upon crack growth) as a driving force for fracturing. The remaining energy is dissipated through molecular motions by heat; and as such, it is made unavailable to break the chains. At higher cross-linking levels, chain motions become restricted, and the dense network is incapable of dissipating as much energy. This results in a relatively facile brittle fracture at low elongation. The tensile strength increased with SiO2 content, showing the reinforcement effect. The tensile strength values of nanocomposites based on NR/BR/SBR/SiO2 ternary rubber blend increase with increase in content of filler, and the maximum value is obtained for the sample containing 60 phr of filler and then decreases (Figure 1(a)). The optimal charging for rubber is assumed that all agglomerates filler are dispersed to the aggregate. Silica particles tend to agglomerate due to the formation of hydrogen bond between the surface. At lower loading of SiO2, the nanoparticles were well dispersed, thereby increased the surface area for interaction. When the SiO2 content increases, the values of the elongation at break and the hardness values increase in ternary rubber composites.
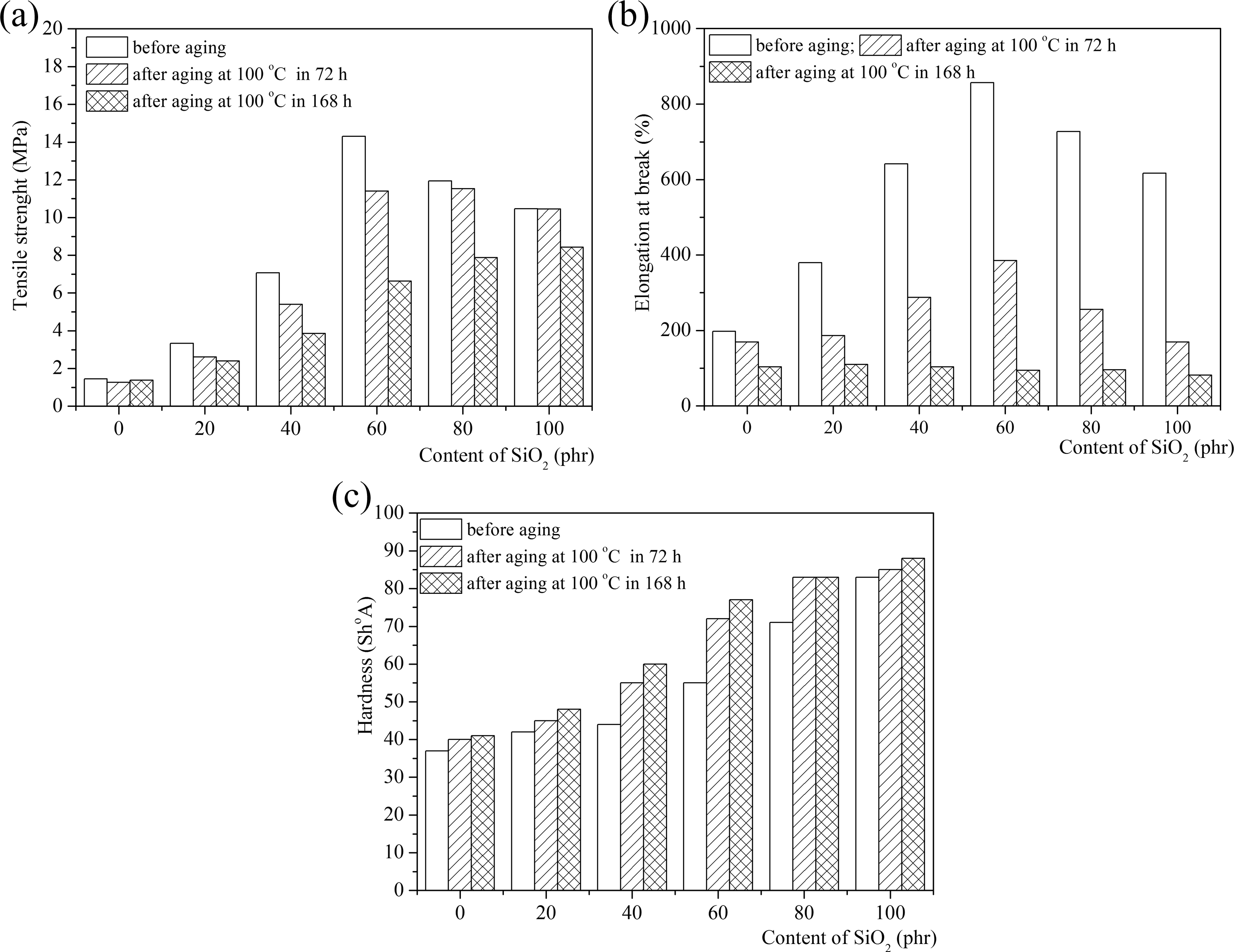
Mechanical characteristic before and after aging of ternary NR/BR/SBR rubber blend reinforced with different loading of nano-SiO2: tensile strength (a), elongation at break (b), and hardness (c). NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber.
Thermal aging
The resistance of the rubber vulcanizates to thermal aging is considered as an essential requirement for the long service life of products. After thermal aging, tensile strength, elongation at break, and hardness on unfilled and filled ternary NR/BR/SBR rubber blend were measured. The retained tensile strength percentage and elongation at break values were then calculated before and after aging. Based on the results (Table 4), the tensile strength decreased. The sulfur vulcanization of the unsaturated rubbers occurs through complicated radical substitution, forming mono-, di-, or polysulfide bridges and sulfur-induced intracyclization of the polymer molecules. However, at higher temperatures, the sulfur cross-links are less effective and the physical properties are weaker due to the dissociation of sulfur bonds and rubber chains. The results of mechanical characteristics after aging of NR/BR/SBR ternary rubber blends are presented in Table 4.
Retention (%) of mechanical characteristic of nano-SiO2-filled NR/BR/SBR ternary rubber composites after aging at 100°C during 72 and 168 h.
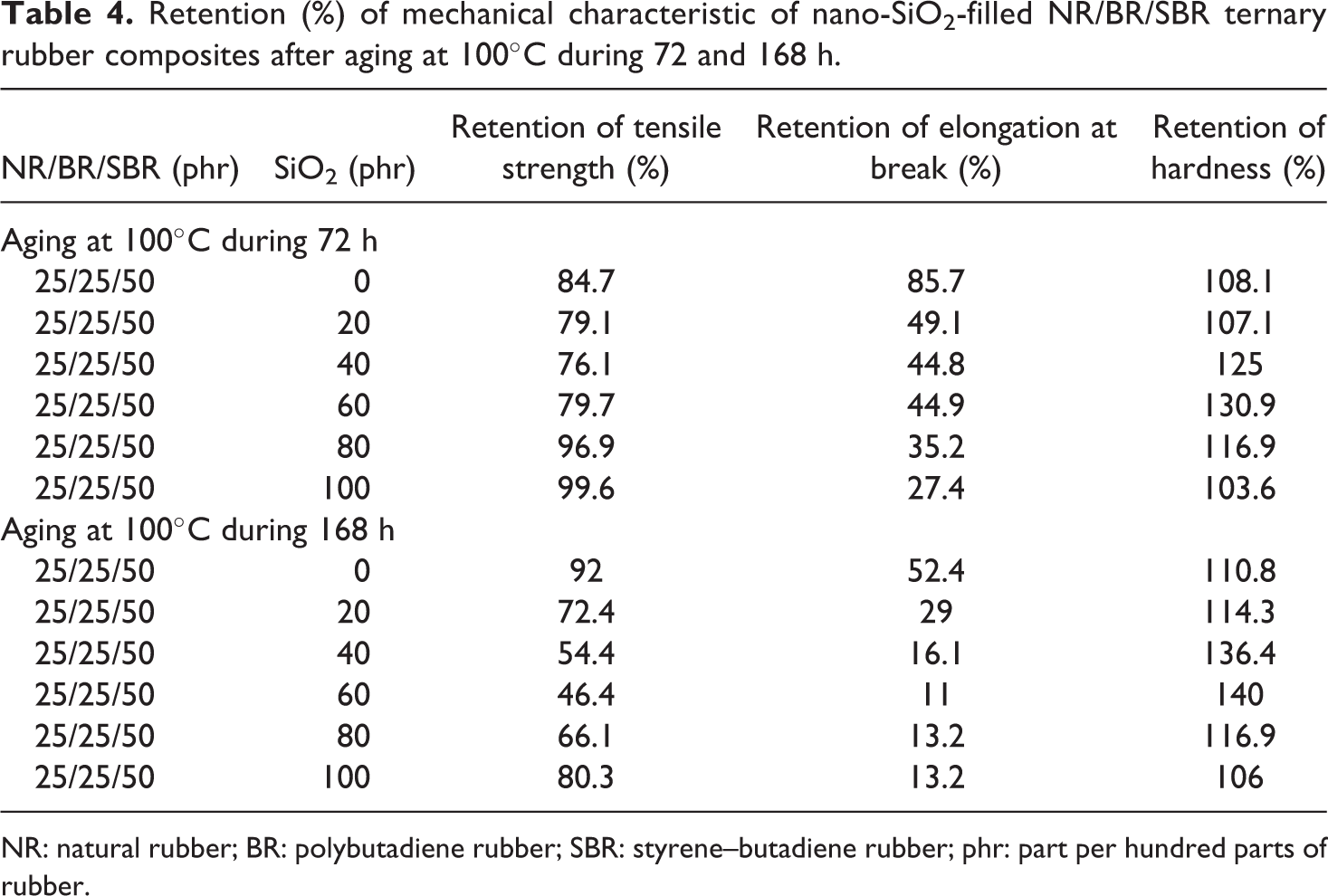
NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
The aging process is irreversible. Tensile strength is reduced, and the longer the aging process, the greater are the changes. However, thermal aging of rubbers can be very complicated because there are two competitive processes taking place almost simultaneously during the aging process: cross-linking and chain scission. 33 Cross-linking will lead to an increase in the elastic modulus and a consequent decrease in the extensibility of the material, whereas chain scission will result in the loss of the elastic modulus. Rubbers are susceptible to oxidative aging because of their unsaturated carbon–carbon double bonds in the backbone. Elevated temperatures usually promote oxidative aging. In some samples, tensile strength increases after aging as a result of the further process of cross-linking. It was found that the sulfur of polysulfide –C–SX–C– bond leads to further cross-linking.
During aging, existing material properties worsen, due to spontaneous changes occurring in the basic structure of the polymer. The aging process is defined as a set of irreversible physical and chemical changes in the observed material. Test procedures of aging consist in exposing samples to the effect of harmful factors in a given time interval.
In vulcanizates, the bond energy between the sulfur cross-link atoms and the polymer backbone greatly differs. There are three types of cross-links in the vulcanizates: polysulfidic, disulfidic, and monosulfidic. 33 As for rubber cross-linked with sulfur systems under conditions of elevated temperature, conversion of polysulfide bonds into monosulfidic is observed. The reaction is followed by separation of low molecular weight groups such as hydrogen sulfide, sulfur dioxide, and carbon disulfide. Monosulfidic connections are broken under the influence of stress, and they react with neighboring hydrogen atom, thus building a stable functional group. During subsequent cross-linking regardless of increase in cross-linking degree, due to the dominating process break cross-link fact, in most cases, the strength and elasticity decline. Cross-links formed during cross-linking, under the influence of heat and prolonged time networking, can be torn. In this case, first –S–S– bonds are broken, followed by –C–C– bonds, which is explained by a lower value of energy connection. Broken –S–S– bonds provide radicals that can react with each other giving the chains of smaller length of the starting molecules polymers and can react with each other by building the ring (cyclization) of different size and structure. The final properties of cross-linked materials strongly depend on the structure of cross connections. If the presence of polysulfide bridges is bigger, changing the mutual arrangement of parts chain between cross-link nodes is much easier.
Mechanical properties of NR/BR/SBR ternary rubber blend reinforced with SiO2 nanoparticles before and after aging (100°C for 72 and 168 h) are shown in Figure 1. After thermal aging, the values of the mechanical properties decrease and degradation process can be noticed. The changes in the tensile properties upon aging could be due to several reasons, such as change in the morphology of the system, degradation of rubber and cross-linking, and change in the level of interaction between components at elevated temperatures.
From Figure 1(a) and (b) and Table 4, we can observe that there is a marginal decrease in the tensile strength and elongation at break after aging for a period of 72 and 168 h for samples which contain 80 and 100 phr nano-SiO2. The decline in tensile strength for these materials could be attributed to the rubber which can react with oxygen, thus leading to chain scission or reduced cross-link formation between the rubber chains. These reductions in the properties are due to partial cross-linking of the elastomer backbone and degradation of the rubber taking place upon aging as observed by some of the researchers. 34 –36 It needs to be noted that with the increase in temperature, there is less cross linking of the polymer chain and the filler. The restriction in chain mobility during tensile testing might have also led to the reduction in tensile strength. The values of elongation at break decrease with increase in aging time.
Elongation at break (Figure 1(b)) decreases with tightening of the conditions of accelerated aging process. The reduction in value for elongation is explained by the degradation process of cross-linked materials and by reducing the polymer molecular weight due to breakage of elementary chains, which reduces the elasticity of the material.
Hardness (Figure 1(c)) of all tested samples increases with tightening of the conditions of accelerated aging process. The hardness of all rubber blend composites increases with time of aging being increased. This can be attributed to the cross-link density being increased after thermal aging. The well-dispersed nanoparticles of filler in rubber decrease the heat and mass transfer through the material, thereby preventing fast degradation. The well-dispersed filler acts as a mass transport barrier to oxygen and volatile decomposition products. A change in the hardness values increased with increase in SiO2 loading, which can be attributed to the increased cross-linking density after thermal aging. This can be explained by the sulfur networking addition process of the rubbers and the polysulfide cross-link density reduction process. The polysulfide reacts further to form mono-, di-, and cyclic-sulfide bonds during vulcanization via the dissociation, recombination, and rearrangement of the sulfur linkages.
The slightest change after aging is seen in a sample containing 80 phr SiO2 filler in it, and the greatest change is seen in the ternary NR/BR/SBR rubber blend a sample which contains 20 phr of SiO2 nanoparticles. The silica-filled ternary NR/BR/SBR rubber blend composite with 80 phr SiO2 possessed the highest retention in tensile strength.
Swelling properties
Swelling measurements are very useful to characterize the network degradation. In the case of unfilled rubbers, the swelling restriction is due to cross-links connecting the polymer chains, which avoid their extension and diffusion. Swelling provides information on the matrix network chain density (determined from the swelling ratio) and the percentage of soluble fraction (i.e. the proportion of chains that do not belong to the network). Because of the filler–rubber interactions, fillers can play the role of additional cross-links. Besides, their presence via filler–filler interactions can also restrict the swelling. Therefore, it is hazardous to relate the swelling ratio to the matrix network chain density, and the results of the measurements will be considered only as qualitative. 37
Cross-link density is an important factor affecting the thermal stability of vulcanized rubber. Higher cross-link density of vulcanized rubber leads to better thermal stability, because of the higher values of activation energy needed for thermal decomposition of vulcanizates. 38
Table 5 shows the variation of swelling characteristics (the volume rate of swelling (Rv
), the weight rate of swelling (Rw
), the volume fraction of the sample in the swollen gel (v
2), the Flory–Huggins parameter (χ), the cross-link density (ν), and the molecular weight of the rubber blends between two cross-links (
Swelling characteristics of NR/BR/SBR ternary rubber blend reinforced with nano-SiO2.
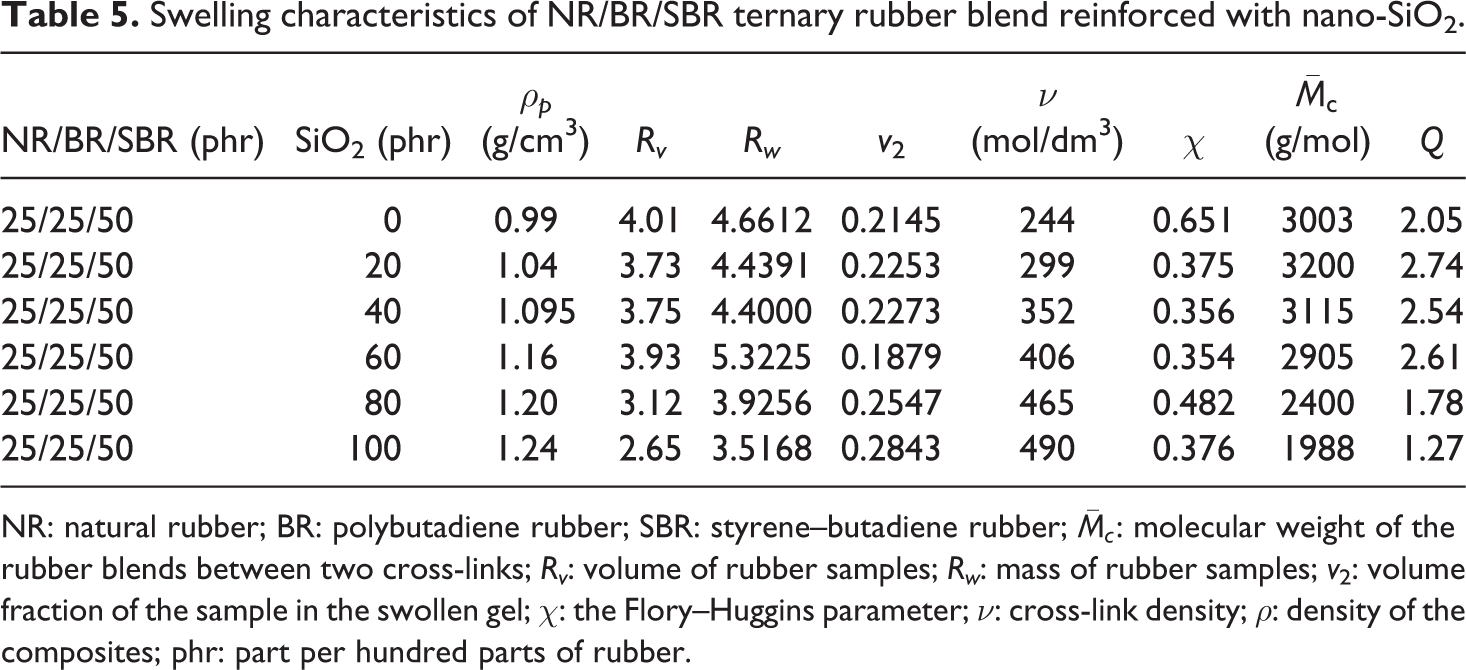
NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber;
All swelling parameters decreased with increase in the content of the SiO2 filler. The highest cross-link density values are obtained for 100 phr of SiO2 loading, and this is resulted as the swelling process restriction near the SiO2 particle surface.
The swelling process of the polymer matrix depends on diffusivity, as well as solubility of the solvent molecule, which is a rate-dependent process. When the filler is loaded in polymer matrix, the amount of the solvent absorbed became smaller, and reduction equilibrium swelling values in the SiO2 nanoparticles-reinforced NR/BR/SBR ternary blend are obtained. This swelling reduction near the surface of filler is explained in two ways: (a) A filler has a catalytic effect on curing process and is expected to have different states of networking, depending on whether the rubber matrix is filled, and (b) additional networking is formed between filler surface and polymer matrix. Fillers are classified as (a) filler that has poor adhesion to the surrounding polymer, which is damaging the forces between solvent and polymer (swelling pressure), and (b) the filler that has a strong adhesion to the polymer, which cannot be removed by the solvent molecules.
Thermogravimetric analysis
TGA can be used as a way to measure the thermal stability of a polymer and the thermal degradation of polymer blends due to the simplicity of the mass loss method. 39 The thermal stability of the unfilled and 60 phr SiO2-filled NR/BR/SBR ternary rubber blend composites was investigated using TGA, and the results are shown in Table 6 and Figures 2 and 3. Polymers are often exposed to high temperatures during processing and/or use. Thus, thermal stability is one of the most important properties of polymers for a wide range of applications. 40
DTG and DTA data of peak values and mass loss for unfilled and nano-SiO2-filled ternary NR/BR/SBR rubber blend composites.
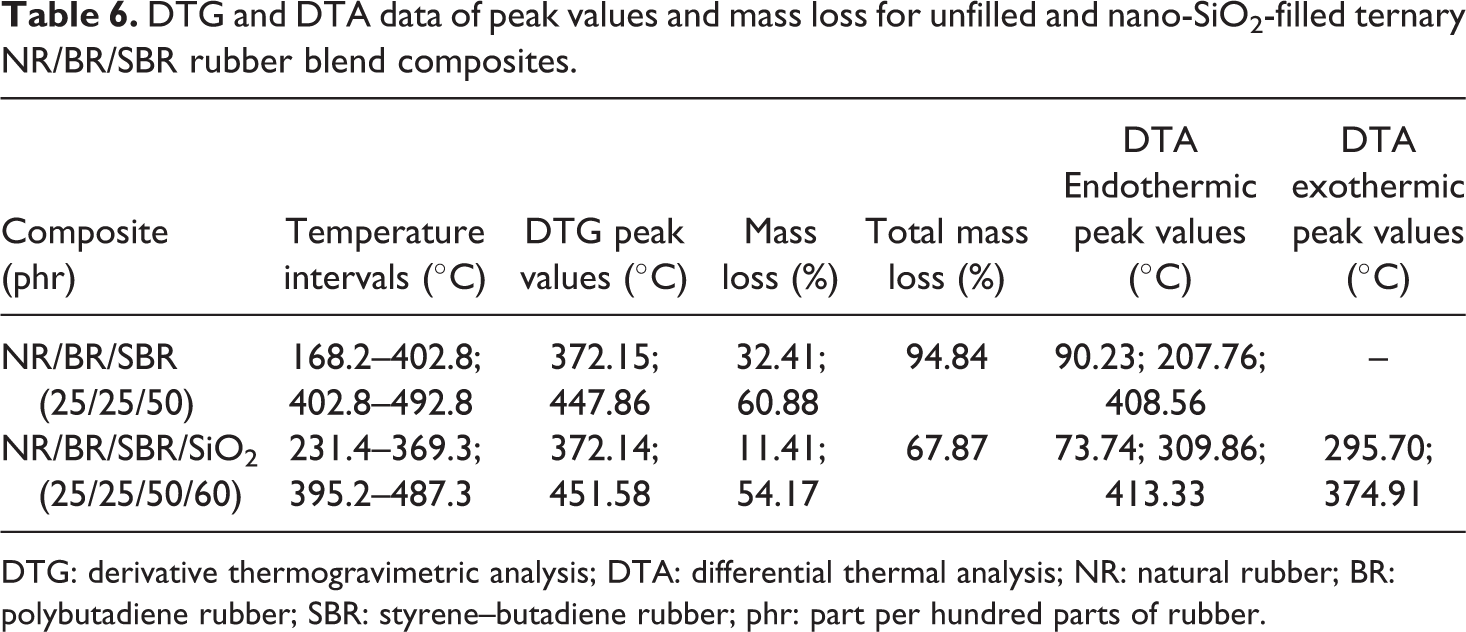
DTG: derivative thermogravimetric analysis; DTA: differential thermal analysis; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
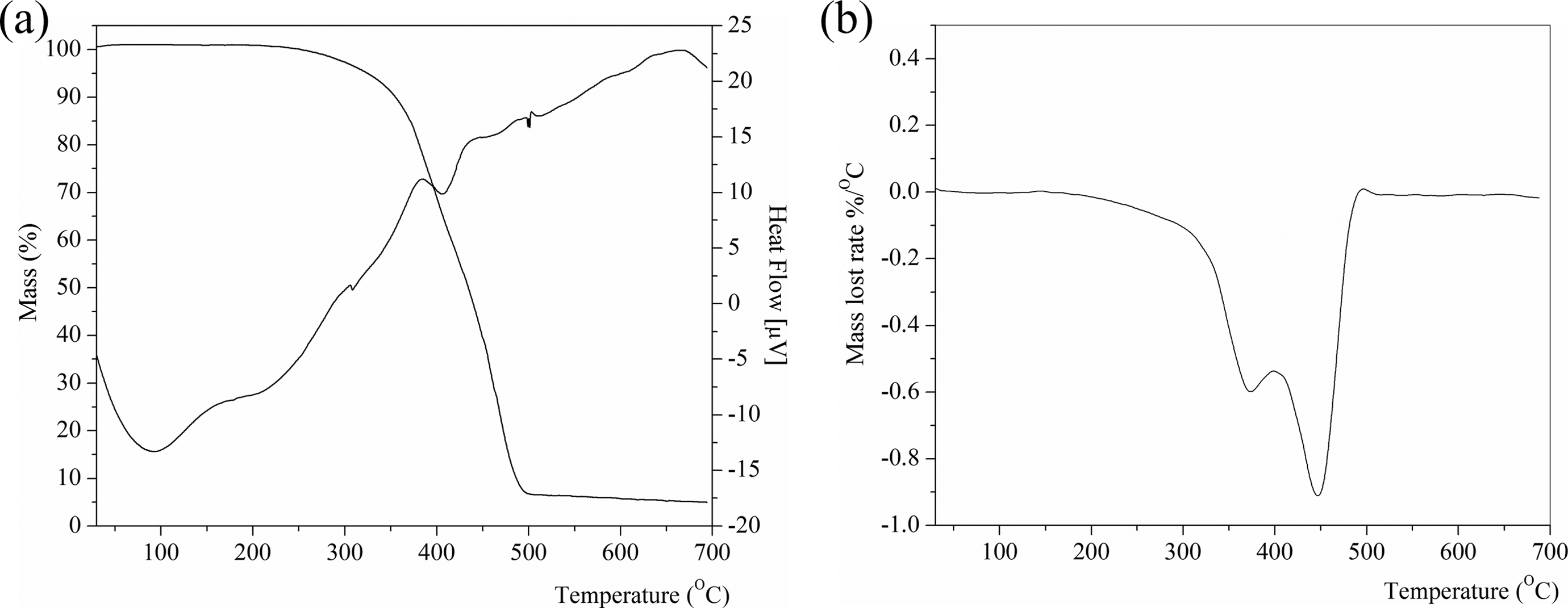
TGA/DTA (a) and DTG (b) thermograms of NR/BR/SBR ternary rubber blend. TGA: thermogravimetric analysis; DTA: differential thermal analysis; DTG: derivative thermogravimetric analysis; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber.
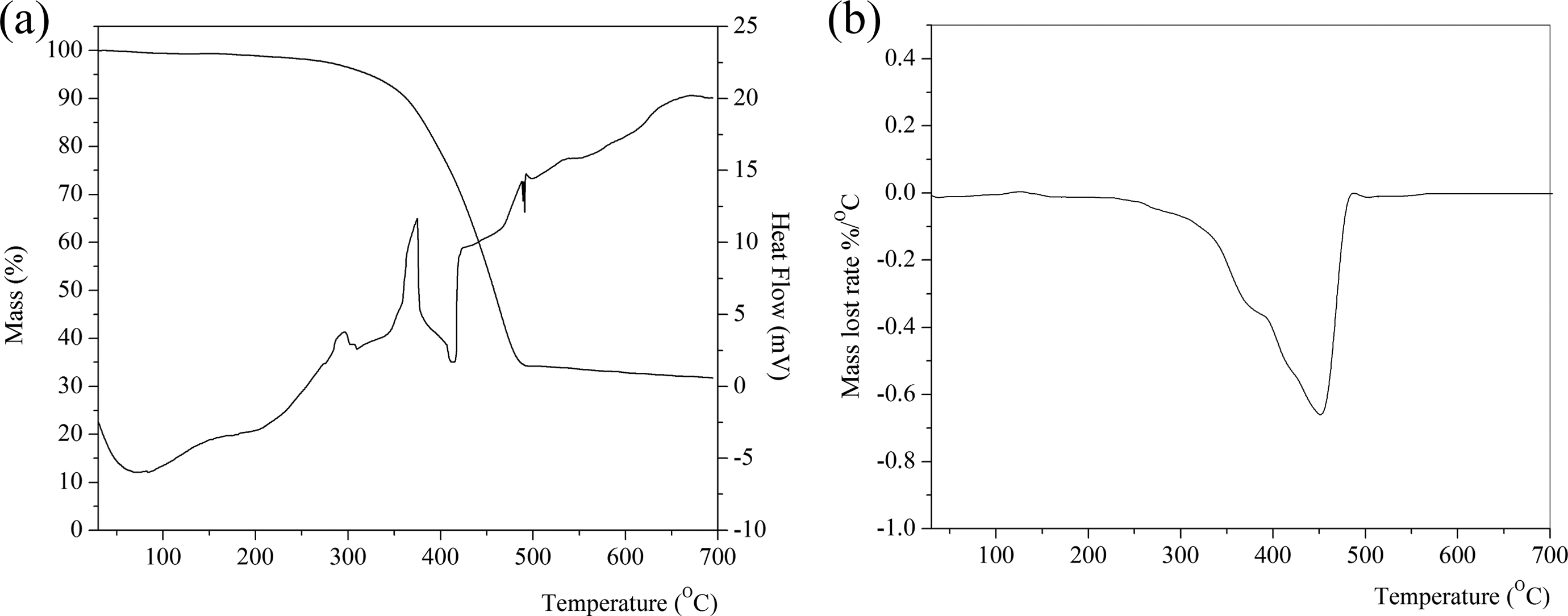
TGA/DTA (a) and DTG (b) thermograms of 60 phr SiO2-filled NR/BR/SBR ternary rubber blend. TGA: thermogravimetric analysis; DTA: differential thermal analysis; DTG: derivative thermogravimetric analysis; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
The TGA and DTG curves of vulcanized NR, SBR, and BR are given in the work of Lee et al. 39 As can be seen from Figures 2 and 3 and Table 6, the TGA curves of unfilled and filled NR/BR/SBR rubber blends show two-stage decomposition regions with these observations: The first thermal decomposition temperature region (168–402°C and 231–369°C for unfilled and filled rubber blends, respectively) comes from the volatilization of processing oil or any other low boiling point components that resulted in the mass loss of 32.4–11.4 wt%.
When the temperature reached 500°C, the decomposition of the mass loss remains constant. The second stage is occurred in temperature region at 402–492°C and at 395–487°C for unfilled and filled rubber blends, respectively. Maximum rate is at 372.2 and 447.8°C for unfilled and at 372.1 and 451.6°C for filled NR/BR/SBR ternary rubber blend NR, respectively.
However, in Figures 2(b) and 3(b), these steps could not be overlap interference of peaks. The results may be also due to covulcanization; a new “phase” is formed between NR, SBR, or BR rubbers. 41,42
The heat capacity as a function of temperature for NR/SBR, NR/BR, and BR/SBR blends of various compositions has been studied by Lee et al. 39
In the DTG thermogram, the two DTG regions appear at from 372°C to 381°C and the other at from 447.8°C to 451.6°C, which are originated from degradation process of the NR rubber and BR/SBR rubber blend, respectively. This is also confirmed by two DTA regions from 207.7°C to 309.9°C and from 408.5°C to 413.3°C for unfilled and 60 phr SiO2-filled NR/BR/SBR ternary rubber blend, respectively.
Morphological study
Morphology is a major factor of rubber blends, which determines the extent to which the blends are compatible. It is well known that the phase structure of the blend is influenced by several factors, including the surface characteristics, blend ratio, viscosity of each component, and compounding process. The primary factor that determines the final morphology of the mixes is their composition. SEM technique is an important tool for observing the surface morphology of crack initiation and the failure process in composite materials.
SEM micrographs of cryogenically fractured surface of elastomer blend without filler NR/BR/SBR (25/25/50) and SiO2-filled NR/BR/SBR/SiO2 (25/25/50/60) ternary rubber blend composite are presented in Figure 4(a) and (b). The SEM photomicrographs shown in Figure 4 confirm that smaller particle size of the filler provides a larger surface area for a better filler dispersion and interfacial bond between filler and rubber matrix. The fracture surface of NR/BR/SBR rubber blend composite without filler as shown in Figure 4(a) exhibits brittle fracture. Architectural homogeneity and the appearance of strong ridgelines indicate a better dispersion state with efficient interfacial cross-linking. The compact nature of blend systems, as revealed from the study of blend morphology, is manifested in the significant enhancement of the mechanical properties. Vertical steps and stairs are more frequent and more prevalent in cross-linked rubber blends with higher tensile strength values. 43 Good interactions between polymer matrix and SiO2 nanoparticles exist.

SEM micrographs of (a) unfilled NR/BR/SBR rubber blend and (b) 60 phr SiO2-filled NR/BR/SBR/ ternary rubber blend at 2000× magnification. SEM: scanning electron microscopy; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
ATR-IR study
FTIR spectra of unfilled NR/BR/SBR rubber blend and 60 phr SiO2-filled NR/BR/SBR ternary rubber blend are given in Figure 5(a) and (b). Assignment of the bands is shown in Table 7.
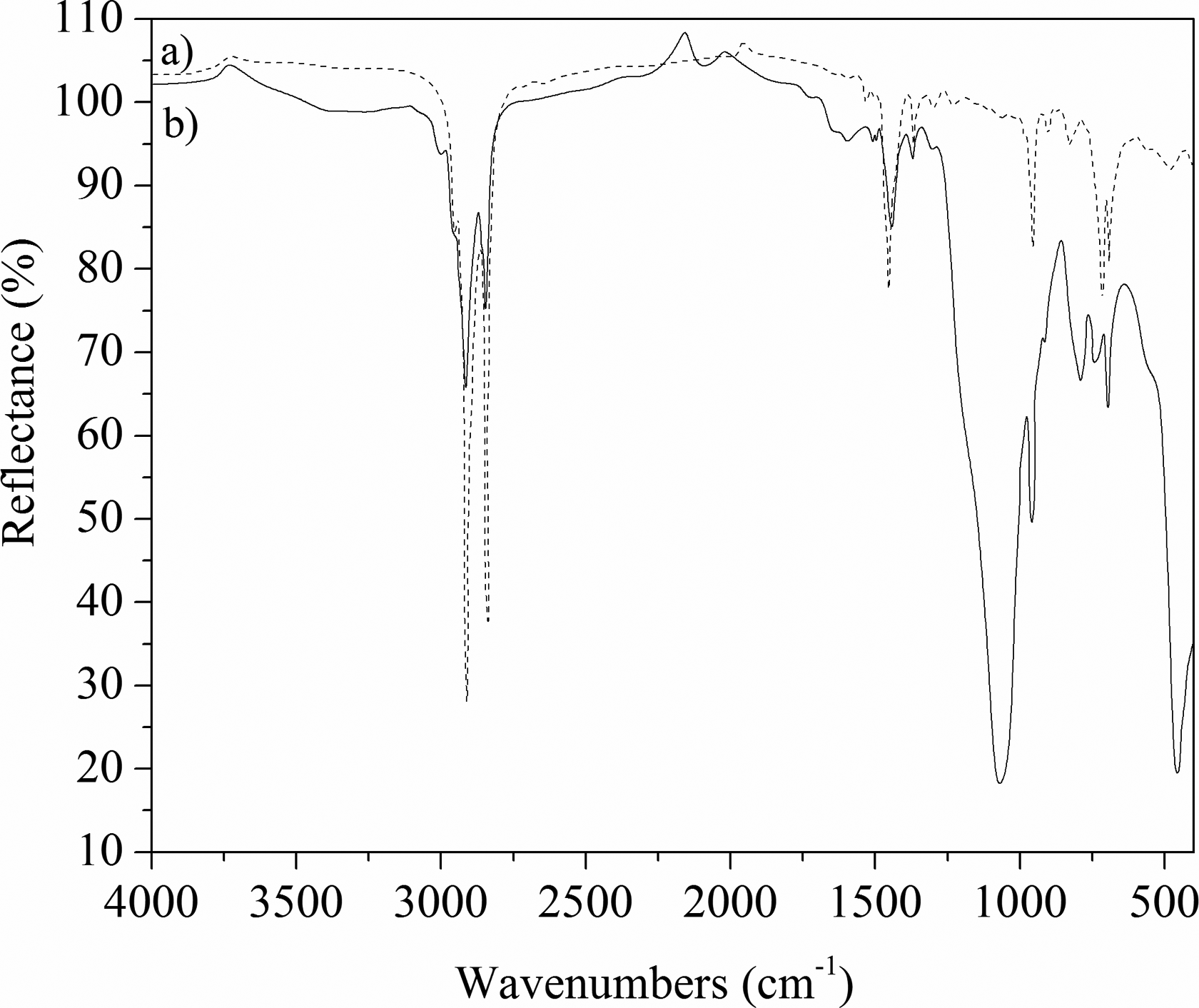
ATR-IR spectra of (a) unfilled and (b) 60 phr SiO2-filled ternary NR/BR/SBR rubber blends. ATR-IR: attenuated total reflection accessory infrared; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
Peaks assignments for the FTIR-ATR bands of unfilled and 60 phr SiO2-filled ternary NR/BR/SBR rubber blends.

FTIR-ATR: Fourier transform infrared spectroscopy with attenuated total reflectance; NR: natural rubber; BR: polybutadiene rubber; SBR: styrene–butadiene rubber; phr: part per hundred parts of rubber.
The presence of C-H stretching (2900 and 2830 cm−1) CH2 in-plane deformation (1450 cm−1), −CH2 wagging motion (1375 cm−1) and trans C=C stretching (970 cm−1) is apparent and characteristically for NR and BR rubbers. Typical styrene absorption occurs at 730 and 970 cm− 1 (C–H out-of-plane deformation of vinyl). The monosubstituted benzene ring at 700 cm−1 and the C–H stretching (out of plane) of butadiene double bond at 970 cm−1 from SBR, and the C=C stretching for the unconjugated linear olefin at 830 cm−1 of NR are shown (Figure 5(a)). The characteristic bands for all three types of SiO2 (quartz, tridimite, and cristobalite) (Figure 5(b)) are obtained at 1050 cm−1 for filled NR/BR/SBR rubber blend. The width of these bands depends on the chaotic state of solids. 44 –46 The movement of absorption band from 1450 to 1480 cm−1 and the interaction between rubber blend and SiO2 filler are confirmed.
Conclusion
The aim of this study was to design raw composition of elastomeric materials based on NR/BR/SBR (25/25/50) ternary rubber blend, as a precursor cross-link, reinforced with SiO2 nanoparticles. Cross-linking is achieved by sulfur system. Some conclusions can be drawn as follows: The values of scorch time (t
s2) and optimum time (t
90) in an NR/BR/SBR ternary rubber blend decrease with increase in SiO2 content, that is, ternary rubber blend cross-linked faster. By monitoring cross-linking regime in systems obtained from NR/BR/SBR ternary rubber blend, it was found that when SiO2 content increases, the values of the minimum (M
min) and maximum (M
max) torque increase. The cross-link density values are higher for SiO2-reinforced NR/BR/SBR ternary rubber blend than those of unfilled. The silica-filled ternary NR/BR/SBR rubber blend composite with 80 phr SiO2 content possessed the highest retention in tensile strength. The obtained TGA and DTG curves suggest two kinds of mass loss: (i) the first one appeared during the temperature interval at 200–400°C with mass loss of 32.4–11.4%, most probably indicating the evaporation of oil, and (ii) the second one appeared in the temperature interval at 400–500°C with mass loss of 94–67%, indicating polymer degradation. Good interactions between polymer matrix and SiO2 nanoparticles exist. The movement of absorption band from 1450 to 1480 cm−1 and the interaction between rubber blend and SiO2 filler are confirmed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was granted by the Ministry of Science and Technological Development of the Republic of Serbia (projects numbers: 45022, 45020 and 172056).
