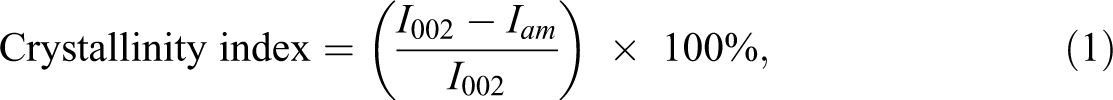Abstract
Flax fibre was treated with low concentrations of alkali and used to prepare poly(furfuryl alcohol) (PFA)-based composites through in situ polymerization of furfuryl alcohol. The properties of low concentrations of alkali-treated flax fibre and flax fibre–reinforced composites were characterized for their structure and properties by X-ray diffraction, scanning electron microscopy, Fourier transform infrared, thermogravimetric analysis, dynamic mechanical analysis, water absorption and flexural measurements. The alkali-treated flax fibre showed increased cellulose content and crystallinity index with smooth surfaces of the microfibres. PFA composites exhibited dispersed microfibres within PFA matrix with agglomerates. There was also a clear increase in thermal stability, storage modulus and damping factor for all treated PFA composites. Poor resistance to water uptake was also observed for the PFA composites, even though the highest alkali treatment improved the resistance compared to the untreated composite. PFA–flax biocomposites were characterized by improved flexural strength and modulus, except the untreated which showed the opposite.
Introduction
Natural fibres are gaining more use in polymer composite materials primarily due to diverse appealing properties such as eco-friendliness, low density, partial or complete biodegradability, high strength and low cost. 1 –3 Owing to a decline in fossil resources, current environmental concerns and high cost of petroleum-based materials, natural fibre–reinforced polymer composites are gaining more appreciation in advanced engineering applications such as aerospace, automotive and sports markets. 3,4 This is especially the case for petroleum-based thermosetting natural fibre composites. Furthermore, the use of natural fibres with bio-based thermosetting polymers such as poly(furfuryl alcohol) (PFA) produces ‘all green composites’ with acceptable mechanical, chemical, viscoelastic, moisture stability and fire-resistant properties. 5 PFA is a bio-based cross-linked thermoset material that is normally obtained by acid-catalysed polymerization of furfuryl alcohol (FA). FA is derived from reduction of renewable furfural, which is produced from agricultural and forestry wastes such as corn, wheat, cereals, sugar cane bagasse and sawdust. PFA exhibits excellent chemical and fire-resistant properties. 3,4 It is used in applications such as metal-casting cores and moulds, corrosion resistance coatings, polymer concrete, wood adhesives and binders as well as cellulosic substrate reinforcement for new alternative fuel. 3,4,6
Natural fibres such as coir, flax, kenaf and sisal have been used as reinforcement in polymeric materials including PFA. 3,4,6,7 –9 However, they are inherently marred by poor moisture resistance, low resistance to microbial attack, high tendency to form aggregates during processing and poor surface adhesion with polymer matrices. 1,7 These shortfalls mainly result into poor properties of the composites and require improving the fibre–matrix interfacial adhesion by chemical modification of fibre surfaces. The surface modification of natural fibres can be accomplished by various methods including alkali treatment (i.e. mercerization). Alkali treatment is a common successful method because of its low cost and easy-to-carry out procedure. 1,4,9,10 –23 Benyahia et al. 9 examined the influence of an increase in the concentration of sodium hydroxide (NaOH; 1, 3, 5 and 7%) on unsaturated polyester resin reinforced with short α-fibre. The authors found maximum enhancement in both tensile and flexural properties when 7% alkali-treated fibre was used. On the other hand, Jacob et al. 10 concluded that 4% NaOH treatment of sisal and oil palm fibres resulted in maximum tensile strength of their natural rubber–based composites. To our knowledge, little or no work has been reported on optimizing low concentrations of alkali treatment on flax fibre–PFA biocomposites. Besides, there is limited information about reinforcing PFA with flax through in situ polymerization.
This work is devoted to investigate the effect of low and desirable concentrations for alkali treatment on flax fibre and its reinforcing effect on PFA bio-based composites. Flax fibre was treated with different low concentrations, which was extreme relative to literature, of NaOH before being compounded into PFA matrix. Both the alkali-treated fibre and flax–PFA-reinforced composites were characterized for their physico-chemical properties. The effect of varying alkali content on flax-PFA-reinforced composites on the morphology, thermal, mechanical and water uptake properties of PFA was studied.
Experimental
Materials
NaOH pellets 98%, p-toluene sulfonic acid monohydrate 98.5% and sulphuric acid (H2SO4) 98% were obtained from Minema Chemicals (Pty) Ltd, South Africa. FA monomer was purchased from Fluka Analytical, South Africa. Retted flax fibres were obtained from a farm near Port Elizabeth in South Africa.
Methods
Alkali treatment
Flax fibres were chemically treated with various concentrations of 1, 1.5 and 2% of NaOH and kept for 1 h at room temperature. The treated fibres were subsequently washed repeatedly with running tap water followed by deionized water until a neutral solution (pH = 7) was obtained to remove the remaining unreacted basic medium. Finally, the treated fibres were dried at 50°C for 72 h for removal of moisture.
Preparation of PFA–flax biocomposites
PFA was prepared by polymerization of cold FA monomer described elsewhere in Motaung et al. (2015). Typically, 0.6 g p-toluene sulfonic acid catalyst was dissolved in 10 ml deionized water. This aqueous solution was added drop-wise in 200 ml FA monomer, mechanically mixed with 15 mg flax fibre and continuously stirred for homogeneity. About 2 ml of the suspension was kept in an oven at 50°C overnight for drying. The amount of solid obtained was used to calculate the required volume of the suspension to obtain 5 wt% flax fibres in PFA bioresin and cured accordingly. The content has shown remarkable results in the latest literature of the similar preparation method. 16,17,23 –25 The final curing step was done by raising the temperature of the oven to 150°C and the end of the curing was confirmed using differential scanning calorimetry (DSC). Like in the study of Ahmad et al., 3 no residual exothermic process was observed in the DSC curve. For comparison, pure PFA samples were prepared under the same conditions.
Characterization
X-ray diffraction
X-ray diffraction (XRD) data was collected on a Philips PW 1830 X-ray diffractometer (the Netherlands)
where I002 is the maximum intensity of diffraction of the 002 peak and Iam is the intensity of diffraction of the amorphous material. 12
Chemical composition
The cellulose content of all samples was analysed by a reaction with H2SO4 using a standard method recommended in TAPPI-T222 and om-88 in TAPPI T19m-54.
Fourier transform infrared spectroscopy
Fourier transform infrared (FTIR) analyses were carried out using Spectrum 100 FTIR (PerkinElmer, Waltham, Massachusetts, USA) fitted with a PIKE Miracle™ attenuated total reflectance. The spectra were obtained at room temperature in the wave number range between 500 cm−1 and 4500 cm−1. A minimum of 32 scans were collected at a resolution of 4 cm−1
Scanning electron microscopy
Scanning electron microscopic (SEM) analyses were conducted to evaluate the dimensions of untreated and treated flax fibres and their distribution within PFA bioresin using the environmental SEM FEI Quanta 200 (FEI Co., Eindhoven, the Netherlands) electron microscope operated at 20 kV.
Thermogravimetric analysis
The thermal stability of the PFA-flax fibre-treated biocomposites was studied using a themogravimetric analyzer Q500 (TA Instruments, New Castle, Delaware, USA). Samples ranging between 5 mg and 10 mg were heated from room temperature to 900°C at a heating rate of 10°C min−1 under a nitrogen environment.
Dynamic mechanical analyses
Dynamic mechanical analyses (DMA) of samples were performed using a PerkinElmer DMA 8000 analyzer in the dual cantilever bending mode. The temperature dependence of the storage modulus (E′) and tan δ were tested at a frequency of 6.28 rad s−1 and the strain amplitude of 0.02%. The heating rate was set at 2°C min−1 in the temperature range from 25°C to 160°C.
Differential scanning calorimetry
DSC measurements were performed in a PerkinElmer DSC7 differential scanning calorimeter under a nitrogen atmosphere. Samples were heated from 25°C to 200°C at 20°C min−1.
Mechanical properties
Flexural tests were performed using a 3-point bending set-up according to the ASTM D790 standard. The span length to the specimen thickness ratio is maintained at 16:1. At least five specimens were tested for each processing condition. The samples with dimensions of 10 × 1.5 × 0.2 cm3 (length × width × thickness) were tested at the rate of 1 mm min−1.
Moisture absorption
Water absorption was determined according to the standard ASTM D570-98 test method. The PFA–flax fibre biocomposites were initially dried at 50°C for 1 h, weighed, and placed in a desiccator. Then, the initial mass of samples was recorded and the samples were immersed in deionized water and weighed every 24 h for three consecutive days.
Results and discussion
XRD spectra of untreated and alkali-treated flax fibres are depicted in Figure 1 and the results are shown in Table 1. It can be observed that the untreated flax fibre exhibited two well-resolved peaks with different intensities and other weak intense peaks. The first broad peak was recorded at 2θ = 15° and the intense one at 2θ = 25°. The former and the latter peaks were normally characteristics of amorphous cellulose and crystalline cellulose portions, respectively. 13,14 An increase in alkali treatment of the fibre increased both cellulose and CI values of the fibre, and it seemed to be more resolved for the crystalline peak. The similar behaviour was shown by other alkali-treated natural fibres, and it was attributed to a partial removal of amorphous materials such as wax, lignin and hemicellulose from crystalline cellulose. 15 The same reason is highly possible in this study and could be confirmed by the direct proportionality between the alkali concentration and cellulose content as seen in Table 1.
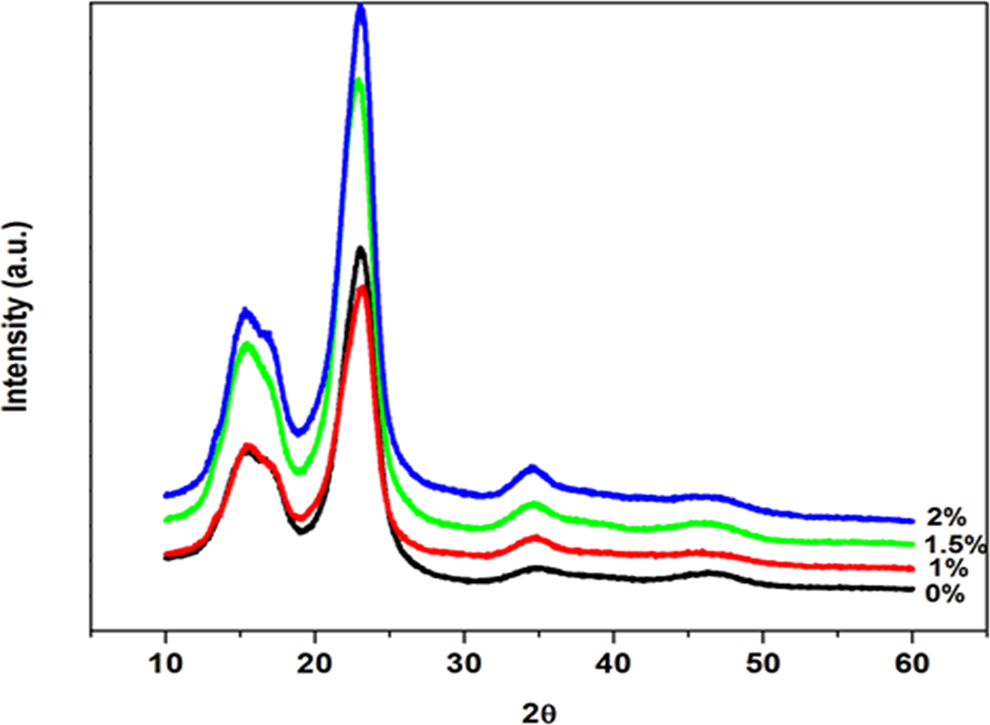
X-ray diffractograms of untreated and alkali-treated flax fibres.
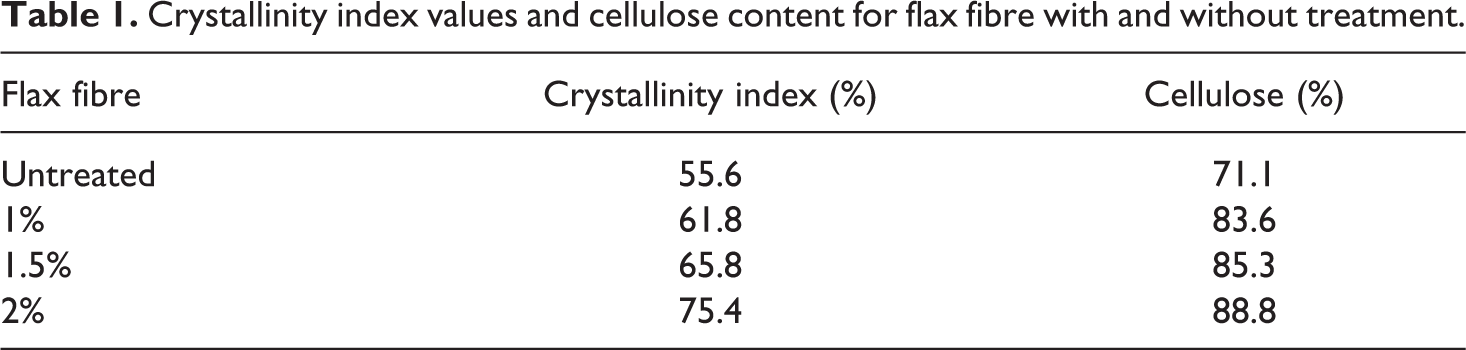
Crystallinity index values and cellulose content for flax fibre with and without treatment.
The FTIR spectra of untreated and alkali-treated flax fibre are shown in Figure 2. The untreated fibre showed a broad peak at approximately 3328 cm−1 and a strong absorption at 1000 cm−1. These correspond to OH, that is, H-bonded and C–O (i.e. out of plane bend), respectively. Similar peaks were also observed for alkali-treated fibres; however, they became less pronounced as a function of NaOH concentration particularly those at 2921, 1625, 1428 and 1300 cm−1. In fact, the peak at 1625 cm−1 was almost disappeared at the highest alkali concentration alongside with highest cellulose content. That absorption denoted a presence of aromatic compounds such as lignin, hemicellulose and pectins. 16 Therefore, it is clear that the results are in agreement with XRD in a way that some amorphous compounds were dissolved during alkalization for purer cellulose. However, some studies suggested that the increase in concentration of OH could also be responsible for the observations. 9
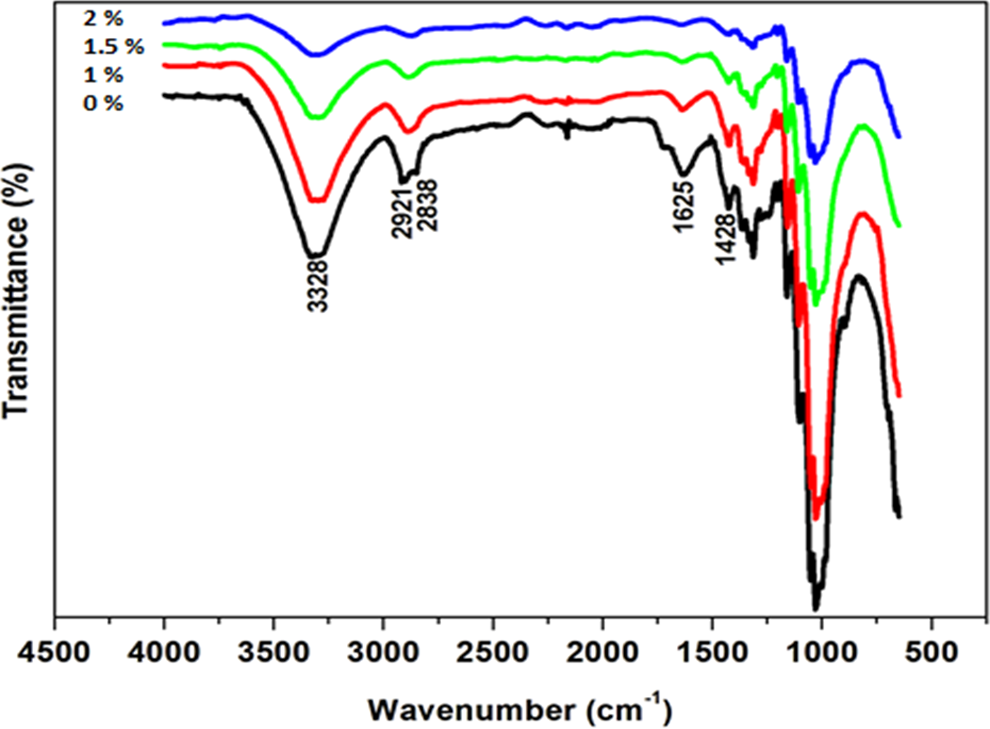
FTIR spectra of untreated and alkali-treated flax fibre.
The FTIR spectra of untreated and alkali-treated flax fibre–reinforced PFA composites are presented in Figure 3. The presence of the furan rings in all three spectra could be noticed by the absorption peaks at 3125, 1600, 1505, 1148, 1075 and 1016 cm−1. 17 An increase in alkaline content revealed more intense peaks, particularly those at 1324 and 2917 cm−1. In addition, there was a new absorption peak at approximately 1422 cm−1 with intensity also proportional to the alkaline concentration. The new peak and increased intensities could be related to the presence of the fibre in PFA and the increasing concentration of cellulose as speculated by XRD and chemical composition studies. However, considering literature from which PFA was reinforced with cellulosic fibres, the new peaks could also be related to a strong interfacial interaction between PFA and cellulose. 3
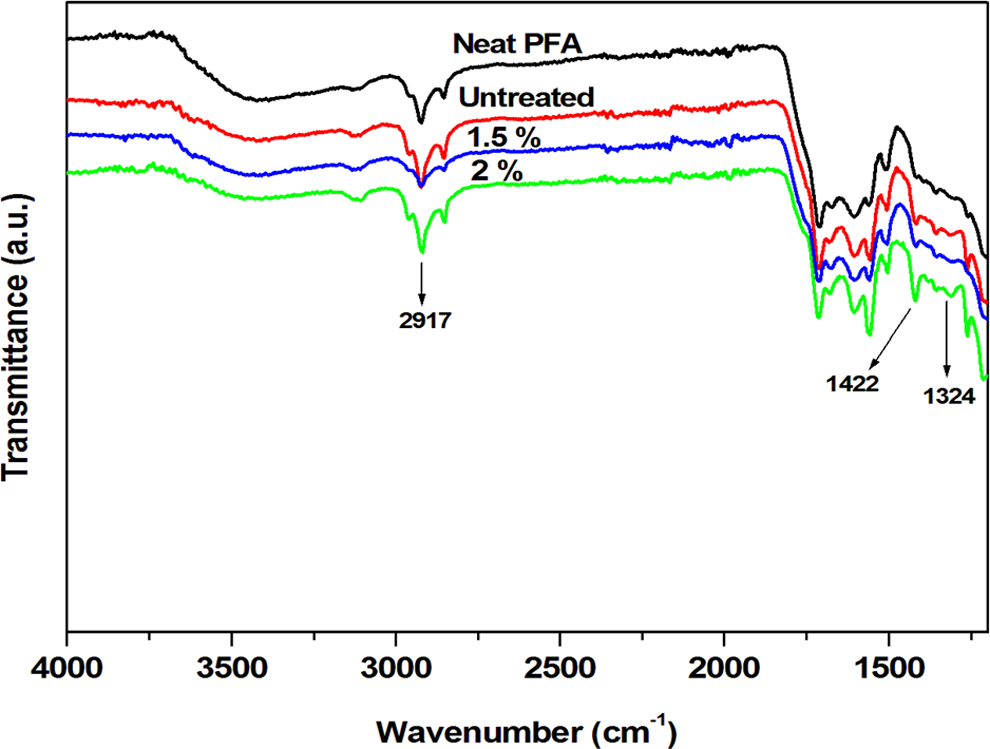
FTIR spectra of the neat PFA, PFA containing untreated, 1.5% treated, and 2% treated. FTIR: Fourier transform infrared; PFA: poly(furfuryl alcohol).
Figure 4(a) and (b) shows SEM images of untreated and alkali-treated flax fibres. There was a whitish substance on the surface of the untreated flax (of the size range 25–40 µm) and what looked like percolation lines, which showed an interface of fibrils within the bundle (shown by arrows in Figure 4(a)). That may be related to the amorphous materials as depicted in FTIR, XRD and chemical composition studies, since it is hardly visible in the treated counterparts (shown by arrows in Figure 4(a)). Furthermore, the treatments led to defibrillation to almost 5 µm after the alkali treatment. It was a challenge to monitor fibril lengths and aspect ratio due to the entanglement of the de-bundled fibrils as displayed in Figure 4(b).

SEM micrographs of (a) untreated flax fibre and (b) 2% NaOH treated flax fibres.
SEM images of fractured surfaces of PFA composites are shown in Figure 5. As expected, a relatively poor interfacial interaction resulted into fibre pull outs, which were randomly dispersed throughout the sample as shown by arrows (Figure 5(a)). Besides, the higher magnification indicated an example of pull out which is almost in the range of the untreated fibre size range as estimated in SEM of fibres (ovalled in Figure 5(b)). However, the treated counterpart had mostly shown a cluster of fibres that seemed broken in the polymeric matrix. This normally happens when there is an improvement in interfacial interaction between a polymer and a natural fibre. 17 –19 The same reason is highly possible in this study despite some minor fibre pull outs and what appeared as busted bubbles.

SEM images of untreated PFA composites (a) and (b) and 2% NaOH-treated PFA composite (c).
Figure 6 shows TGA and derivative thermogravimetric (DTG) curves of neat PFA and flax fibre–reinforced PFA composites. Generally, all samples showed two thermal degradation steps throughout the experimental temperature range as shown by two clearly separated peaks in DTG curves. The thermal degradation steps are normally attributed to the intrinsic thermal degradation characteristics of PFA. 3,17 The presence of flax fibres generally increased the thermal stability except the composite containing untreated fibre, which revealed the total opposite compared to the pristine PFA. Taking into consideration SEM results, it is possible that the fairly poor interfacial interaction is accountable for the low thermal stability of the untreated composites and the opposites may be accurate for increased thermal stability. However, Mwaikambo and Ansell 20 indicated that lignin and hemicellulose in natural fibres could also catalyse thermal degradation of natural fibre–based composites. So from the increased cellulose content as seen from chemical composition results, which most possibly occurred at an expense of the removal of hemicellulose and lignin, as confirmed by FTIR, it is safe to assume that a higher content of cellulose also contributed to the increased thermal stability.
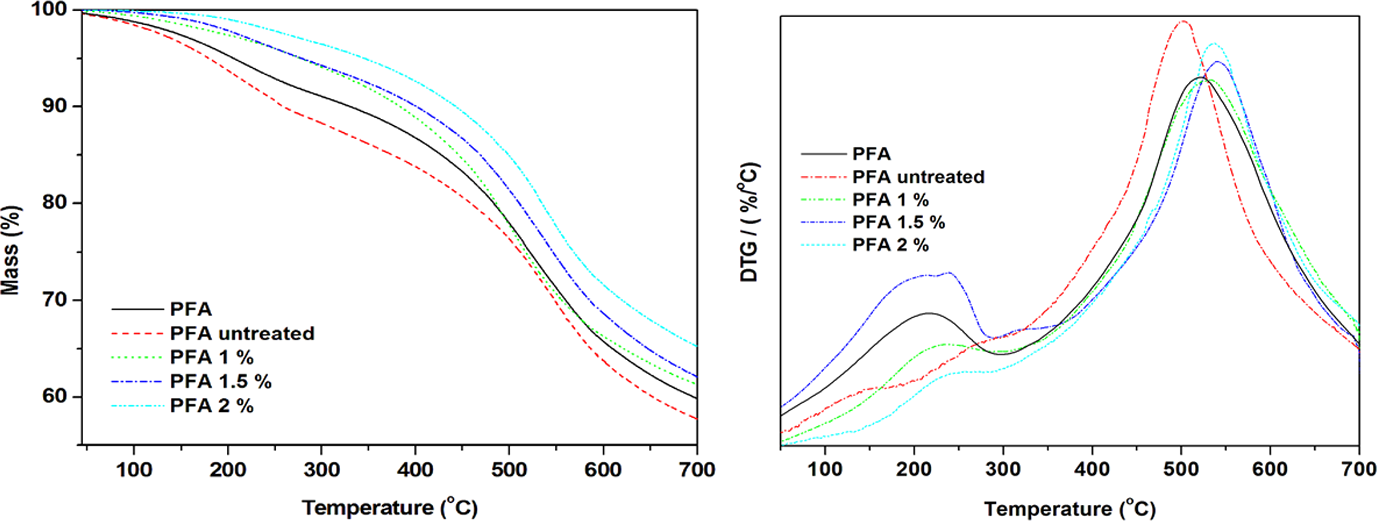
TGA and derivative thermogravimetric curves of neat PFA and flax fibre reinforced-PFA composites.
Storage modulus and tan δ of PFA and its composites are shown in Figure 7. The presence of the fibres generally indicated an increase in storage moduli, particularly at temperatures above 70°C compared to the pristine PFA (Figure 7(a)). The composite containing 1.5% treated fibre had a highest storage modulus, whereas, unexpectedly, 2% treated fibre containing composite revealed lowest storage modulus compared to all composites. The increase in modulus obviously emanated from reinforcement effect by the flax fibre. However, these reproducible results seemed to somewhat contradict SEM, which suggested a better interfacial interaction of 2% treated fibre composite compared to untreated composite. Nevertheless, busted bubbles are perhaps responsible for a poor stress transfer and therefore account for the observed apparent contradiction. In addition, other researchers have indicated that high alkalization at higher concentrations has a potential to destroy cellulose to a certain extent. 21 In this study, perhaps cellulose for 2% treated fibre composite was somewhat destroyed and rendered either a weak interaction or poor stress transfer to the polymeric matrix with an increase in temperature.
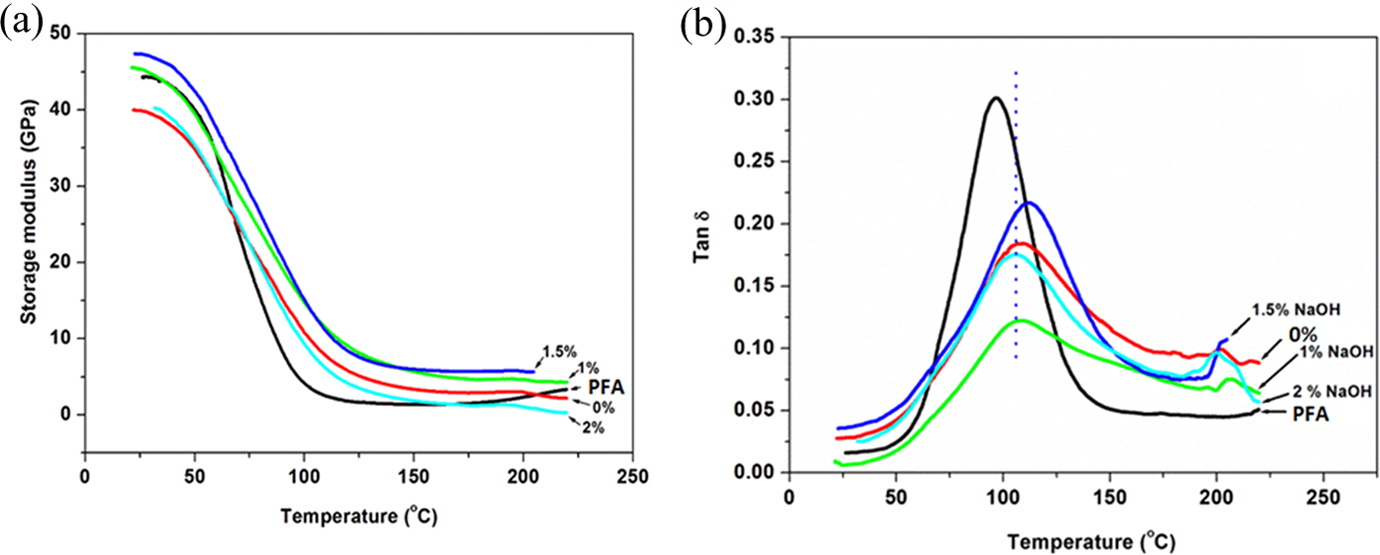
DMA results as a function of temperature (a) storage modulus and (b) tan δ curves of the PFA and its composites.
The difference of tan δ of the PFA and its composites as a function of temperature is presented in Figure 7(b). The tan δ of PFA at around 90°C, related to the modulus drop, decreased after the introduction of flax fibre. The flax fibres noticeably improved the elasticity of the matrix and reduced the energy losses. On the other hand, the position of tan δ peak, which denotes a glass transition, for all composites shifted to almost the same higher temperature (120°C) in the presence of the flax fibre compared to the pristine PFA. However, the position of tan δ peak for 1.5% treated flax fibre containing composite slightly shifted to a higher temperature (123°C). It is clear that an effective immobilization of polymeric chain segments takes place in the presence of flax fibre, whereas the alkalization seemed to show a marginal effect. However, the shift by 1.5% treated flax fibre containing composite indicated either a certain extent of effectiveness of alkalization or an effect of undamaged cellulose, which somewhat increase glass transition temperature. The overall results match the trend displayed by the rigidity of composites.
The effect of fibres treatment on the flexural properties of PFA and the reinforced PFA is presented in Figure 8. Results showed that mechanical proprieties were strongly improved in the presence of flax fibres despite a generally high standard deviation, which may suggest somewhat an inhomogeneity. That may be related to a reinforcement effect of the fibre as suggested in DMA. In fact, the flexural strength of 2% and 1.5% treated fibre composites showed the best flexural strength properties, which are nearly 10% more than that of PFA. The observation may be attributed to bonding of the fibre with the PFA matrix as confirmed by FTIR and SEM thereby improving the fibre–matrix interaction. As for lower flexural strength of low alkaline contents, particularly the untreated composites, it is clear that the interfacial interaction was not strong enough, as predicted by SEM, to sustain an applied flexural stress. The similar trend is followed by flexural modulus and, as predicted in DMA, 1.5% treated fibre containing composite revealed highest flexural modulus.
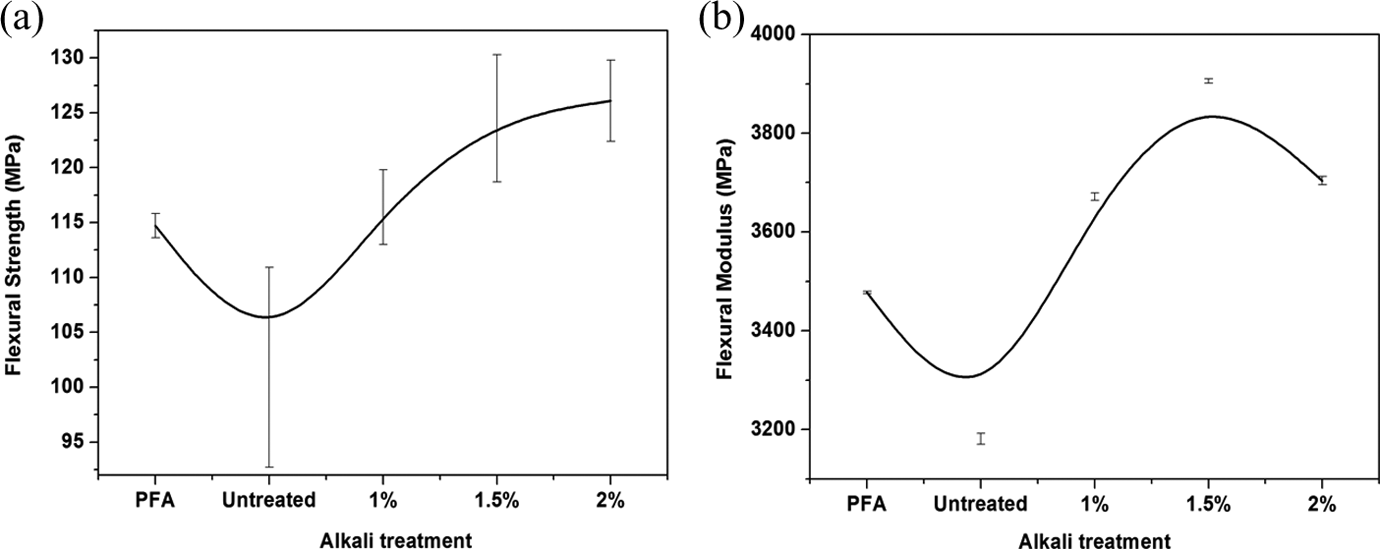
Flexural strength (a) and flexural modulus (b) of PFA and composites.
Figure 9 represented the water absorption curves showing percentage of weight gain as a function of exposure time in water for PFA, untreated and 2% treated fibre composite. In all cases, the water absorption process was fairly steep initially and maintained close values to what could be equilibrium. The addition of flax fibres increased the moisture absorption of the composites, even though the treated counterpart revealed a lower absorption compared to the untreated. The factors affecting the water resistance of natural fibres with the similar chemical treatment are relative content of the crystalline regions and the aspect ratios. 22 The untreated flax fibre contained a substantial quantity of hydrophilic amorphous materials, which resulted in the increased water absorption. Nonetheless, the decreased water absorption of treated composite possibly resulted from the increased crystallinity region as observed in XRD and FTIR and perhaps aspect ratio of the flax fibre.
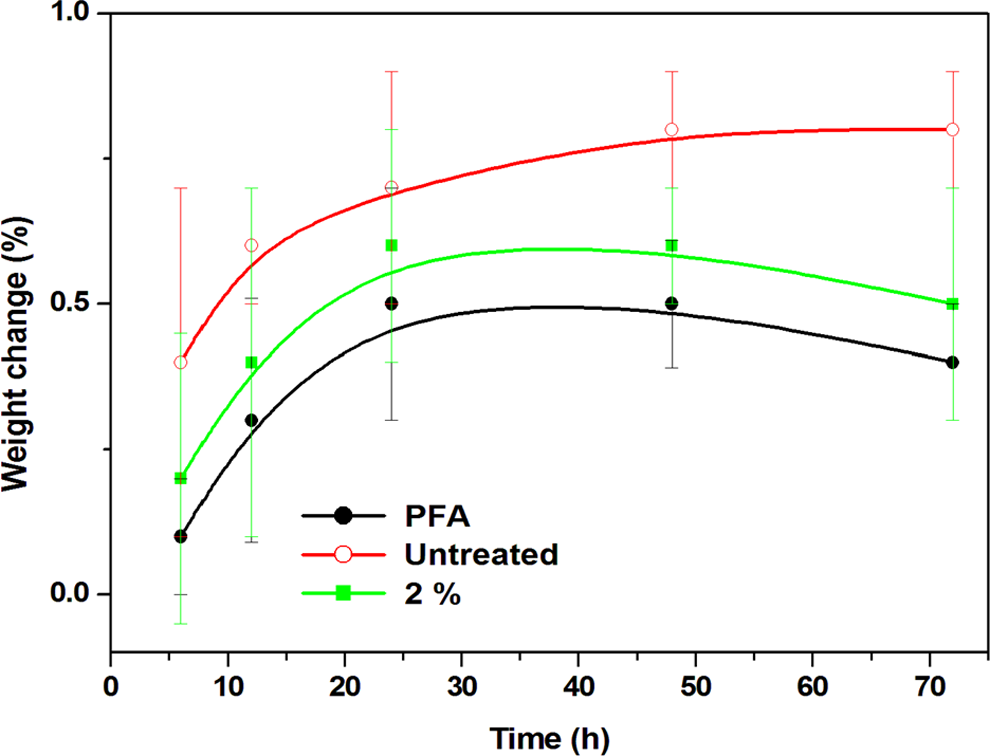
Water absorption of PFA and its composites.
Conclusions
The preparation and characterization of flax fibre and PFA-based composites were successful. The XRD, FTIR and chemical composition have shown that the alkali treatment increased the quantity of cellulose at an expense of lignin, pectin and hemicellulose. The increased crystallinity, intensities and new absorptions concur absolutely with SEM, which showed a removal of whitish substances on the flax fibre surfaces after alkali treatment. The presence of flax fibres increased the thermal stability significantly, except the poorly interfacial interaction in composite containing untreated fibre, which revealed the total opposite compared to the pristine PFA. The poor interfacial interface of the untreated composite had also cost its flexural strength and modulus, and somewhat its fairly poor water resistance. The glass transition for all composites shifted to higher temperature in the presence of the flax fibre compared to the pristine PFA, in which 1.5% treated fibre containing composites showed an exceptional properties. There could be many possibilities on why the composites behaved in that way. Therefore, it would be worth expanding the study of 1.5% treated on flax fibre in different polymeric composites.
Footnotes
Authors’ note
Opinions expressed and conclusions arrived at are those of the author and are not necessarily to be attributed to the Green Fund, Development Bank of Southern Africa (DBSA) or Department of Environmental Affairs (DEA).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article forms part of a research project, ‘Greener Cities in South Africa’, funded by the Green Fund, an environmental finance mechanism implemented by the Development Bank of Southern Africa (DBSA) on behalf of the Department of Environmental Affairs (DEA) and The Young Researchers Establishment Fund in Council of Scientific and Industrial Research (CSIR).