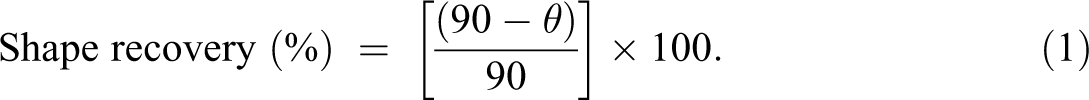Abstract
Novel segmented poly(urethane urea) (PUU) was prepared from polycaprolactone triol (soft segment), 4,4′-methylenebis(phenyl isocyanate) (hard segment), and hexamethylenediamine (chain extender). Graphitized carbon black (GCB) was used as filler. Conducting polythiophene (PTh) was infiltrated by chemical oxidative polymerization. The structure, morphology, mechanical properties, electrical conductivity, and voltage-triggered shape memory effect were demonstrated. The unique network morphology was observed by scanning electron microscope due to the generation of interpenetrating polymer networks (IPNs). The formation of PUU-PTh IPNs was due to the interpenetrating PTh and branched polycaprolactone triol structure. The PTh-modified PUU films reinforced with GCB showed 59% increase in tensile strength and 50% enhancement in Young’s modulus relative to the pristine matrix. The presence of PTh and increase in GCB content increased the conductivity of the composites. The conductivity of 10 wt% GCB-loaded PUU/PTh/GCB composite was raised to 0.089 Scm−1, which is higher than the neat PUU (0.91 × 10−2 Scm−1). The surface temperature of PUU/PTh/GCB 10 was also found to increase with time when a higher voltage was applied (40 V). Such a conductivity of composites was enough to show electroactive shape recovery up to 94% (40 V).
Keywords
Introduction
Shape memory polymers (SMPs) have ability to swiftly change their contour from a temporary shape to their permanent shapes. For the purpose, apposite stimulus such as electric field, magnetic field, temperature, or light is needed. 1,2 SMPs have several advantages such as lightweight, inexpensive, good processability, shape recoverability, shape deformability, and manageable transition temperature as compared to shape memory metallic alloys. 3,4 In this regard, thermosensitive SMPs have been widely studied and employed in industry. 5,6 Thermosensitive SMPs generally possess amorphous/crystalline hard segment, cross-linked structure, and low-temperature transition of phases. They are processed or thermally set to acquire original shape. In general, internal stress is very low in permanent shape. If the SMP is subject to deformation, large internal stress is stored in structure by cooling the polymer below its transition temperature. 7,8 In this regard, the block copolymers of polyurethane have been studied. By applying thermal heating, the shape recovery properties of polyurethane can be achieved. 9,10 The shape memory effect can also be prescribed by electric field. Generally, most of the polymers are structurally insulators having poor electrical conductivity. The combination of conventional matrices with conducting polymers or fillers has been used to fabricate materials with distinctive electrical properties. The shape recovery of polyurethane by applying voltage has been reported by previous studies. Shape recovery of conducting polyurethane/multiwalled carbon nanotube composites has been investigated by applying voltage. 11 Similarly, the conducting shape memory polyurethane incorporating carbon black (CB) has been reported. 12 Polymer composites with conducting CB might serve as polymer conductors or heat transferring media. 13,14 They have found a lot of applications in the fields of electric and electronics industry. 15 Addition of CB reinforcement in the polyurethane has found to influence the strain recovery properties, especially for those samples with CB content above the percolation threshold. 16 SMPs have wide range of applications in sensor and actuators, 17,18 water-vapor permeability materials, 19 medical devices, 20 and high-performance structures in spacecraft. 21,22 In this article, we have focused on the preparation of conductive, flexible composites from novel segmented poly(urethane urea) (PUU) and polythiophene (PTh) formed by chemical oxidative polymerization. We are herein interested in the composites of PUU/PTh blend with graphitized carbon black (GCB) filler to investigate the voltage-triggered shape memory. Effect of the structure, cross linking, and morphology on the conductivity and shape memory properties were studied. These materials can potentially be used in a variety of industrial applications where high conductivity and shape memory properties are the important consideration such as nanocomposite filaments for conductive textile relevance and magnetic and flame retardant SMP for technical applications. 23 –25
Experimental
Materials
4,4′-methylenebis(phenyl isocyanate) (98%), polycaprolactone triol (number-average molecular weight approximately 900), GCB (<200 nm), thiophene (≥99%), iron III chloride (FECl3, ≥99%), hexamethylenediamine (HDA, 98%), and N,N-dimethylformamide (DMF, ≥99%) were supplied by Sigma-Aldrich (St Louis, Missouri, USA).
Characterization techniques
Infrared spectra were taken at room temperature with a resolution of 4 cm−1 using Excalibur Series Fourier transform infrared (FTIR) spectrometer (model no. FTSW 300 MX; Bio-Rad Hercules, California, USA). The scanning electron microscopic (SEM) images were obtained by SEM S-4700 (Hitachi Co. Ltd, Japan). The weight-average molecular weight (Mw) and polydispersity index (PDI) were calculated through gel permeation chromatography. Stress–strain behavior was examined using a universal testing machine (Instron 4466; Instron, Norwood, Massachusetts, USA) with a strain rate of 5 mm min−1 at 25°C according to ASTM D638 standard method. Sample dimension was equal to 40 × 10 × 1 ± 0.01 mm3. The electrical conductivity was measured at room temperature using 4-point method (Keithley 2401, Ohio, USA). The rectangular samples with dimensions 40 × 10 × 1 ± 0.01 mm3 were used for conductivity measurement. The shape memory effect of rectangular strips with dimension 40 × 10 × 1 ± 0.01 mm3 was observed at 40 V. The shape recovery was calculated using the following equation:
In equation (1), θ indicates an angle between tangential line at midpoint of a sample and a line connecting the midpoint and end of a curved sample.
Synthesis of PUU
Shape memory PUU was synthesized from its monomers by two-step process. 4,4′-Methylenebis(phenyl isocyanate) was first dehydrated at 60°C for 24 h. Polycaprolactone triol was also dehydrated at 60°C for 24 h. Afterward, the polyol and diisocyanate were sonicated in 10 mL DMF (NCO/OH = 1.1:1) with steady stirring for 6 h. HDA (equimolar) was dissolved separately in 10 mL of DMF with constant stirring for 6 h. The above mixtures were then mixed and refluxed at 80°C for 12 h. 26 Later, the mixture was cast in Teflon mold and cured at 80°C to obtain PUU film (Figure 1). Mw = 39 × 102 g mol−1; PDI = 2; % yield = 88%.
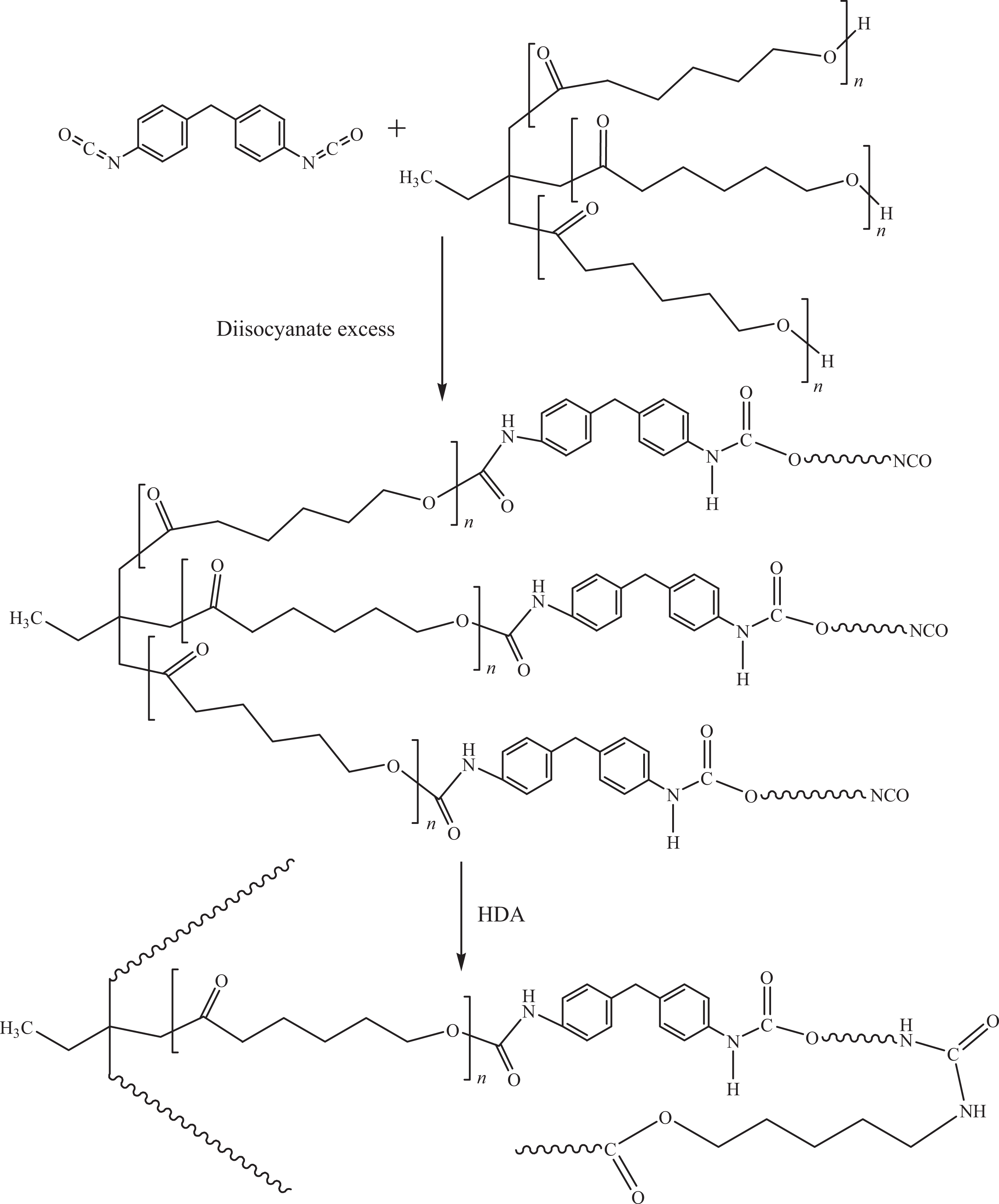
Schematic preparation of PUU. PUU: poly(urethane urea).
Preparation of PUU/GCB films
PUU/GCB composite was prepared using the similar procedure mentioned in section “Synthesis of PUU”. However, after the addition and refluxing with HDA, the desired content of GCB (1, 3, 5, and 10 wt%) was added and the mixture was refluxed for 24 h before film casting.
Preparation of PUU/PTh/GCB composite
The composites were prepared by in situ oxidative polymerization of PTh on the surface layer of PUU/GCB. The above prepared PUU/GCB film was first immersed in an aqueous solution of PTh for 12 h at room temperature. The films were then removed from the PTh solution and immersed in an aqueous solution of FeCl3 (FeCl3/PTh molar ratio 4.6) for 24 h at room temperature. The isolated PUU/PTh/GCB films were blackish in appearance. Films were finally washed with distilled water and dried at 80°C for 24 h. 27
Results and discussion
FTIR analysis
FTIR spectrum of PUU is specified in Figure 2(a). Carbonyl peaks for urea and urethane structure emerged at 1701, 1731, and 1782 cm−1, respectively. Amide N−H stretching and bending vibrations also appeared at 3343 and 1599 cm−1, respectively. The aliphatic and aromatic protons were observed near 2922 and 3055 cm−1. The C–O stretching vibration was found at 1300 cm−1 and C–N stretch appeared at 1465 cm−1. Manifestation of hydroxyl stretching vibration at 3412 cm−1 was related to the unreacted O−H functionality of polyol. PUU/PTh/GCB 1 (Figure 2(b)) displayed nearly all characteristic bands of polyurethane found in the resulting spectrum. However, there was shift in the N–H stretching and bending vibrations to lower wave numbers due to the presence of interaction between the PUU, PTh, and CB particles. In addition, the carbonyl peaks were also shifted to lower wave numbers due to the presence of secondary interaction. Therefore, carbonyl functionalities appeared at 1686 and 1718 cm−1. The N−H stretching and bending vibration also appeared at lower wave numbers of 3222 and 1596 cm−1, respectively. Similar to PUU spectrum, the aliphatic and aromatic protons were experiential near 2912 and 3021 cm−1. The composite displayed peaks related to C–O at 1289 cm−1 and C–N stretch at 1452 cm−1. Moreover, FTIR spectrum shows the typical characteristic thiophene ring stretching vibration, C−C, stretching vibration of in-plane C−H, C−S, and C−H out-of-plane bending vibration absorption at about 1433 cm−1, 1216 cm−1, and 1072 cm−1 and 1043 cm−1, 839 cm−1, and 702 cm−1, respectively. 28 –30
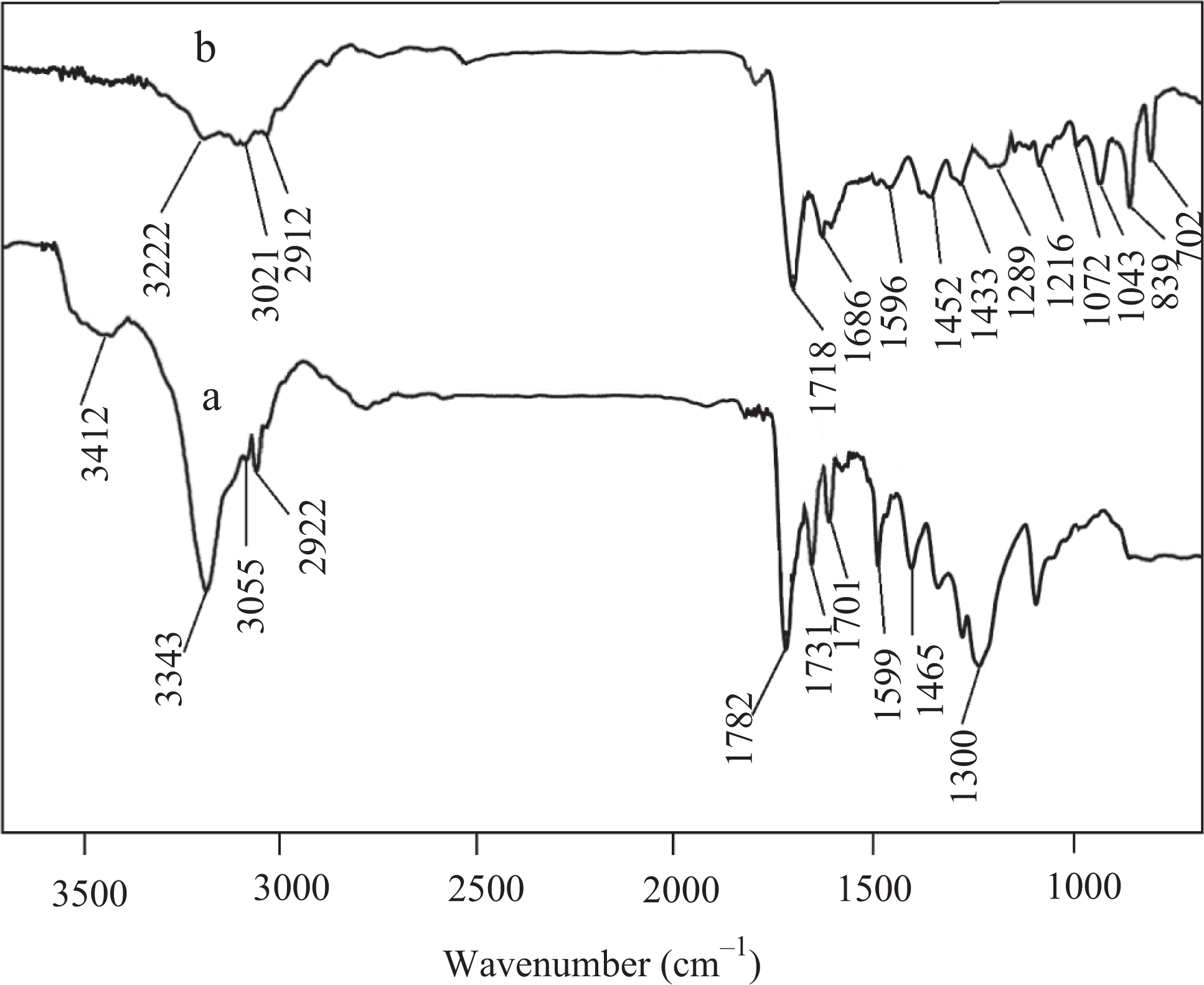
FTIR spectra of (a) PUU and (b) PUU/PTh/GCB 1 composite. FTIR: Fourier transform infrared; PUU: poly(urethane urea).
Morphological investigation
Figure 3 shows the SEM photographs for PUU/GCB 1. A single phase was observed in this case. However, some CB aggregated along with the matrix were visible at lower resolution (Figure 3(a)). At higher resolution (Figure 3(b)), particles seemed to be dispersed evenly in the matrix. The PUU/GCB 10 morphology is depicted in Figure 3(c) and (d). At 10 wt% level of GCB containing composite, PUU form uniform matrix. However, at higher resolution (Figure 3(d)), small extent of GCB spherical granules covered with matrix was observed. The SEM images of PUU/PTh/GCB composites are shown in Figure 4. The composite showed homogeneous microstructure with quite different morphology. In the presence of PTh, the composite has shown unique pattern of layered porous structure. The unique network pattern was more pronounced in Figure 4(c) and (d) with higher PTh content. It seemed that the concentration of PTh was high enough to form interconnective pathways. Such pathways are, in turn, responsible for the good conductivity of the composites. During the immersion of PTh containing PUU film in the aqueous solution of FeCl3, the PTh is oxidized by FeCl3 and a conducting network is generated on the surface of the film. 31 The depth of infiltration of the oxidation layer is clearly seen in Figure 4(c) and (d), where the interpenetrating network was significantly developed. The development of exclusive morphology suggested that the conductive phase was not only coated on the surface but also incorporated into the PUU. 32 It is apparent from the micrographs that by increasing the filler loading, the two polymers formed network structure. The porosity was observed due to generation of interpenetrating polymer network (IPNs). 33 This phenomenon is often exhibited by shape memory polyurethanes having cross-linked structure. In this case, the formation of PUU-PTh IPNs was due to the cross linked structure. The PUU is branched and cross-linked due to the polycaprolactone triol segment used. Whereas PTh is the interpenetrating polymer responsible for the generation of IPNs.

FESEM images of (a) PUU/GCB 1 (5 µm), (b) PUU/GCB 1 (1 µm), (c) PUU/GCB 10 (5 µm), and (d) PUU/GCB 10 (1 µm). FESEM: field-emission scanning electron microscopic; PUU: poly(urethane urea); GCB: graphitized carbon black.

FESEM images of (a) PUU/PTh/GCB 1 (5 µm), (b) PUU/PTh/GCB 1 (1 µm), (c) PUU/PTh/GCB 10 (5 µm), and (d) PUU/PTh/GCB 10 (1 µm). FESEM: field-emission scanning electron microscopic; PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
Mechanical strength
The tensile strength of PUU/GCB and PUU/PTh/GCB composites is shown in Figure 5. In general, the mechanical properties of composites depend mainly on the compatibility of the phases, relative concentration of two phases, and concentration as well as nature of disperse phase. Results suggested that the mechanical property of the composites not only depends on the GCB filler content but also on the infiltration of the conducting polymer. First of all, filler addition in both the composite series increased the tensile strength progressively. In PUU/GCB 1-10, the ultimate tensile strength was increased from 22.8 to 27.3 MPa and in PUU/PTh/GCB 1-10 series the increase was from 29.1 to 34.4 MPa (Table 1). In both series, the maximum properties were depicted by the 10 wt% GCB loading. However, the PTh-modified PUU films reinforced with GCB showed higher tensile strength. Figure 6 shows the plot of Young’s modulus for PUU/GCB and PUU/PTh/GCB composite series. Here again, the PUU/PTh/GCB 1–10 series own higher values in the range of 521–667 MPa. The elongation at break was decreased at a higher rate with increase in GCB content up to 10 wt% in both series. The decrease in elongation at break was more prominent in PTh series because of the brittle PTh network. 34,35
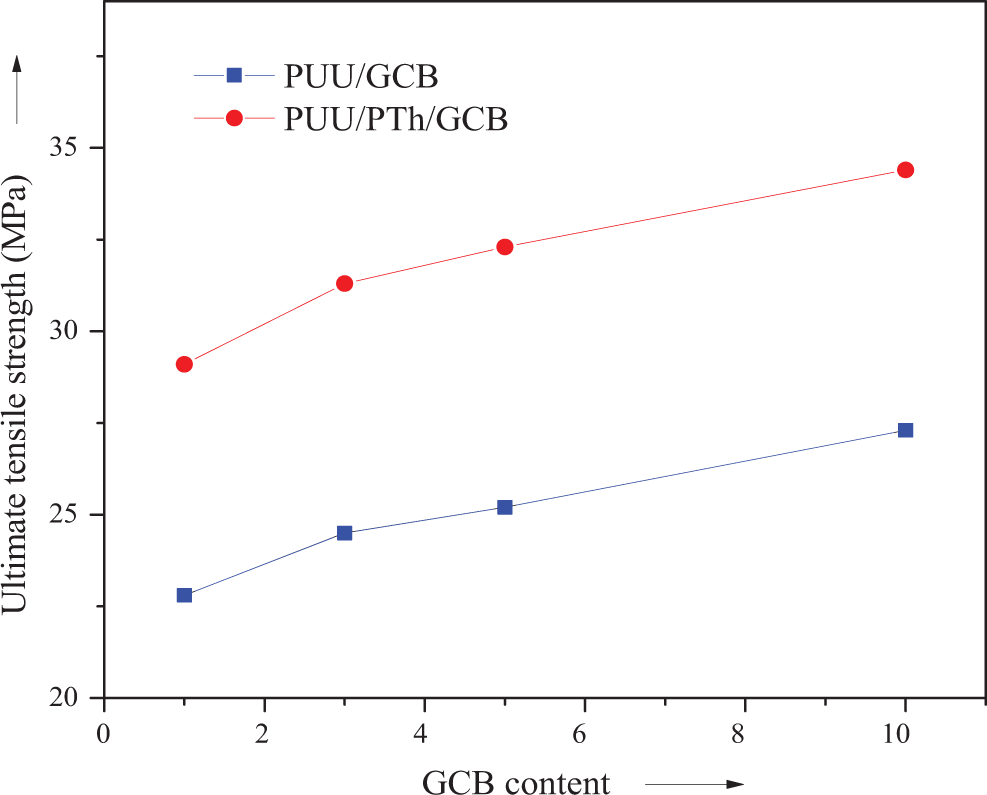
Plot of ultimate tensile strength of PUU/GCB and PUU/PTh/GCB composites. PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
Mechanical properties of composite films.
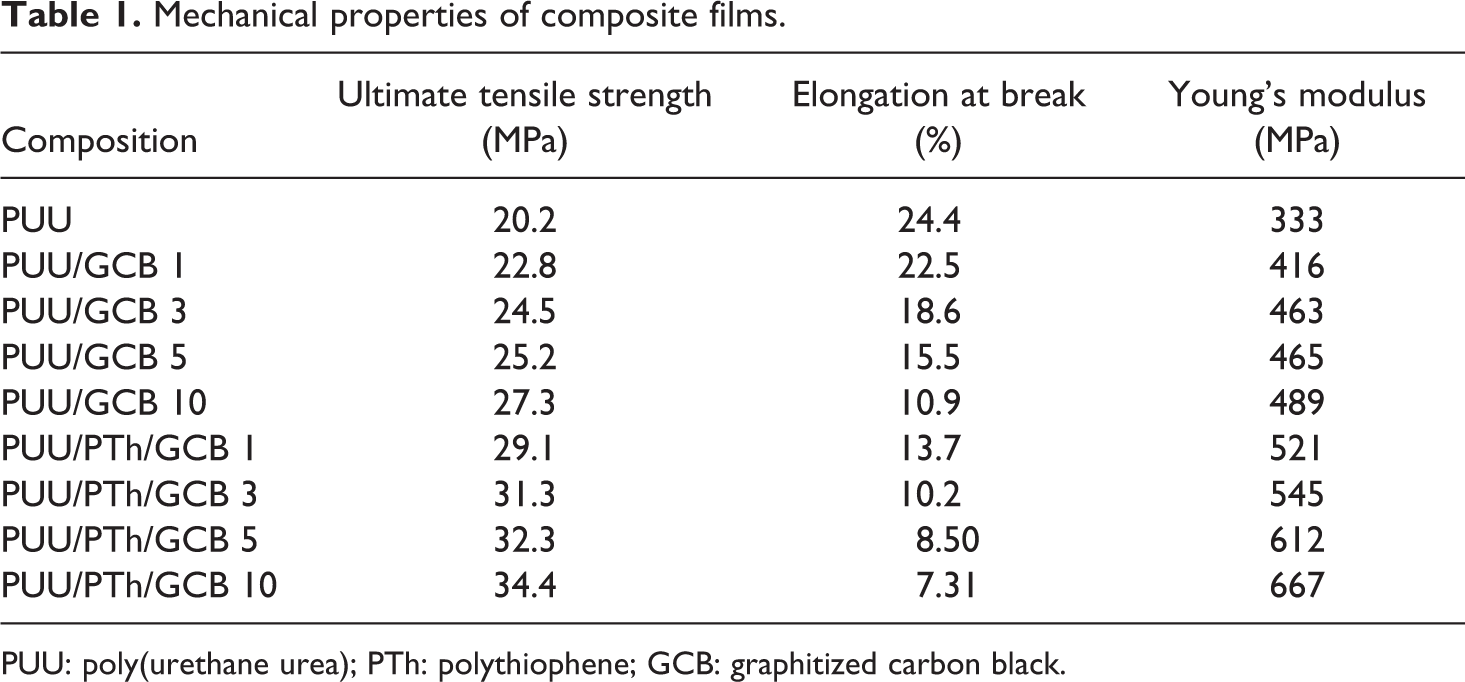
PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
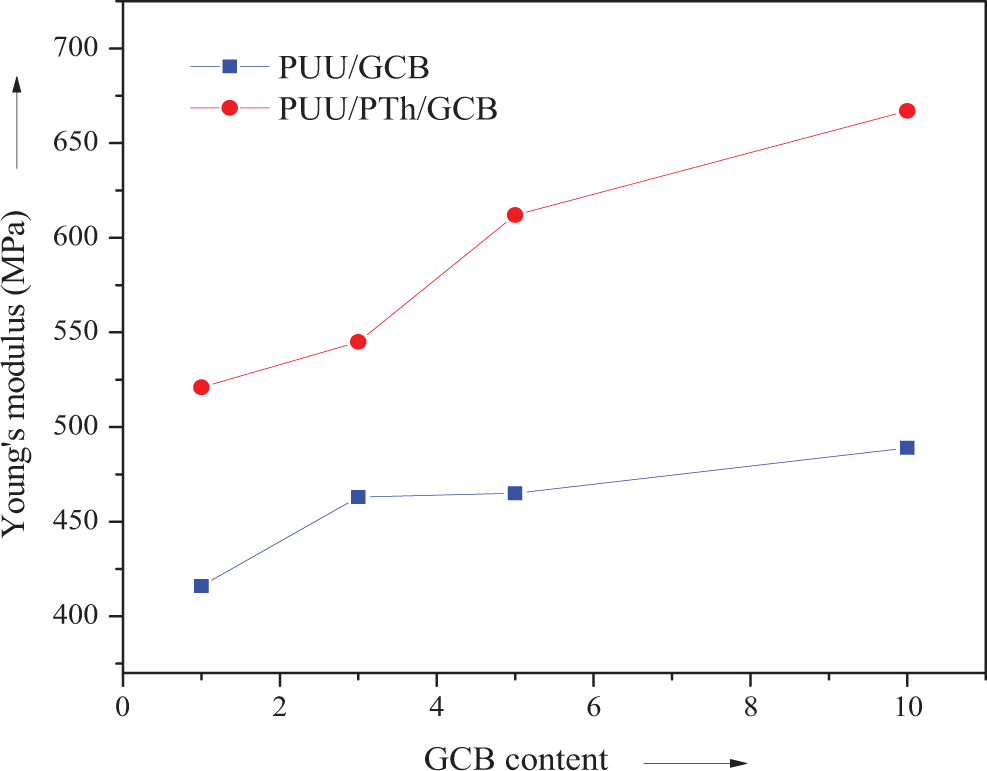
Plot of Young’s modulus of PUU/GCB and PUU/PTh/GCB composites. PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
Electrical conductivity and voltage effect
The influence of the amount of GCB on electrical conductivity of the composites was investigated (Table 2). The room temperature conductivity of PUU/GCB and PUU/PTh/GCB series is shown in Figure 7. The conductivity of the composites was increased with the increase in GCB content. The PUU/GCB 10 composite has electrical conductivity of 0.065 Scm−1, while PUU/PTh/GCB 10 attained a value of 0.089 Scm−1. The sharp increase in the conductivity for 10 wt% filler suggested the realization of percolation threshold in the surface layer. The relationship between the surface temperature and applied voltage for the PUU/PTh/GCB 10 composite is shown in Figure 8. It was observed that the surface temperature increased with time for 10 wt% GCB containing composite, when a higher voltage was applied (40 V). However, the same sample heated at 25 V showed decreased surface temperature with time. Consequently, a higher electrical conductivity is required to reach a good SMP actuator.
Electrical conductivity and shape recovery of composites.
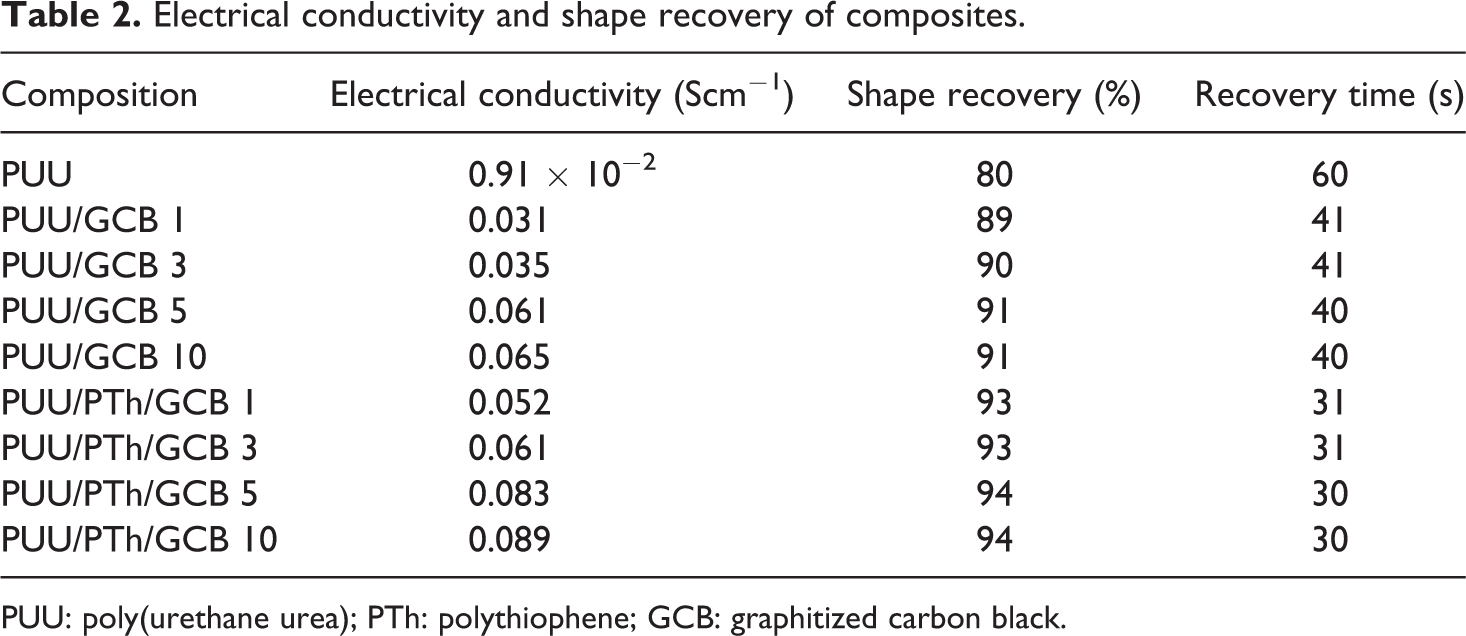
PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
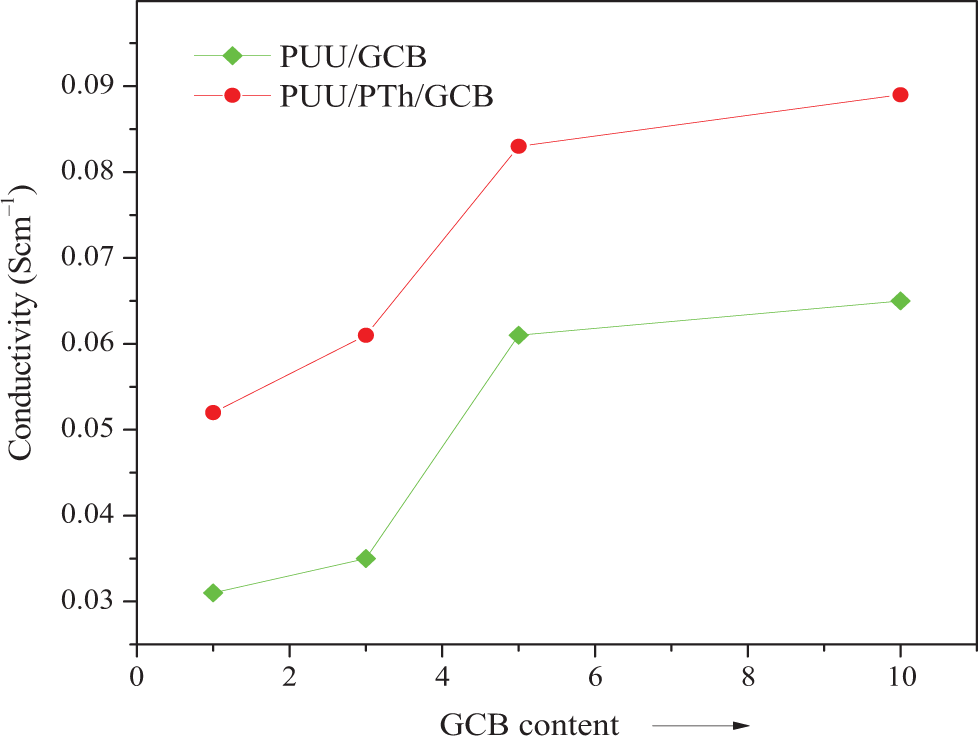
Conductivity of PUU/GCB and PUU/PTh/GCB composites. PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
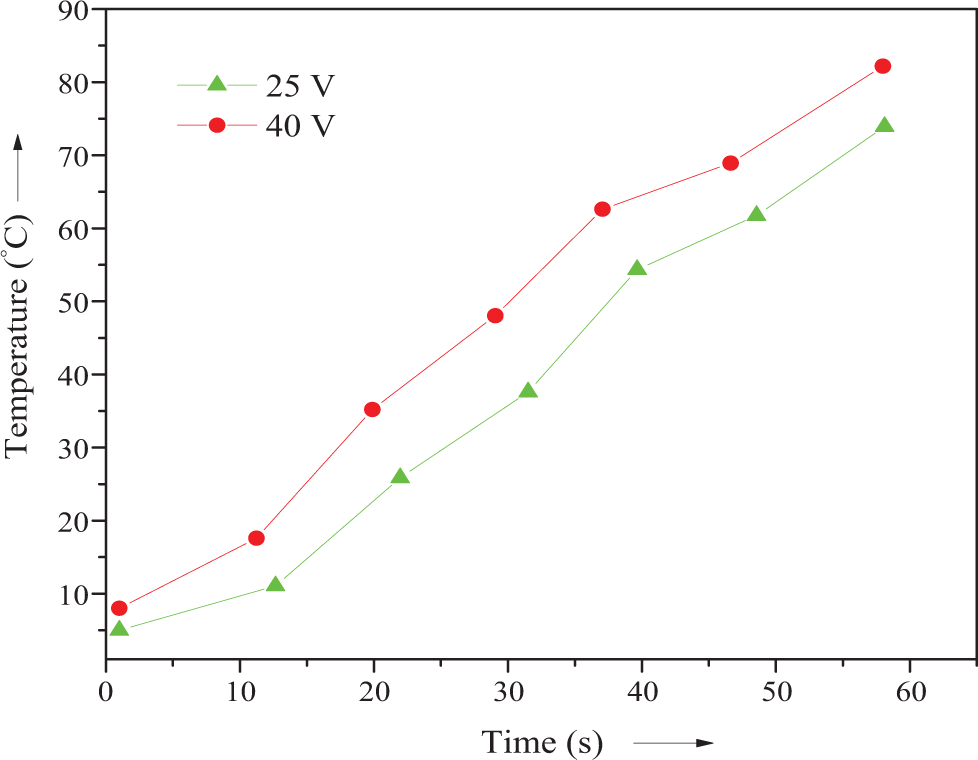
Temperature change of PUU/PTh/GCB 10 composite surfaces with time at various voltage. PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
Electroactive shape memory effect
Electric field-triggered shape recovery of the PUU/GCB and PUU/PTh/GCB composites is shown in Table 2. The sample was initially a rectangular strip and it was deformed into a round shape using liquid nitrogen. When constant voltage (40 V) was applied, its shape changed spontaneously (Figure 9). The original shape of the sample was almost 89–91% recovered in PUU/GCB series in 40 s. The PUU/PTh/GCB composites showed better shape recovery of 93–94% within 30 s. Therefore, the interpenetrating network due to PTh and cross-linking was required for active shape recovery. Moreover, the good electrical conductivity at higher filler loading and optimal mechanical properties was important to show electroactive shape memory behavior in composites compared with pure PUU.

Electroactive shape recovery behavior of PUU/PTh/GCB 10 composite at 40 V (a) 10 s, (b) 20 s, and (c) 30 s. PUU: poly(urethane urea); PTh: polythiophene; GCB: graphitized carbon black.
Conclusion
Shape memory composites of PUU/PTh/GCB were fabricated by the combination of solution casting and in situ technique. Morphology results showed the formation of PUU–PTh IPNs. The PUU own branched and cross-linked structure because of the employment of polycaprolactone triol soft segment. Infiltration of PTh in PUU was responsible for the generation of IPNs. The electroactive shape memory behavior of novel PUU composites was dependent on the network ability of PTh. IPNs, in turn, were responsible for fine ultimate tensile strength and electrical conductivity of the composites. Mechanical results showed that the Young’s Modulus and ultimate tensile strength increased significantly relative to pure PUU when the filler content reached 10 wt%. The electrical conductivity of PUU/PTh/GCB composites up to 0.089 Scm−1 was enough to show electroactive shape memory with bending mode. The original shape of the sample was almost 94% recovered in 30 s with an electric field of 40 V.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.