Abstract
The polycarbonate (PC)/polymethyl methacrylate (PMMA) (10/90) blends with microcellular foams were prepared by the two-step process using supercritical carbon dioxide as physical foaming agent. The effects of saturation temperature, saturation pressure, foaming temperature, and foaming time on the cell morphology structure were investigated by scanning electron microscopy. The results indicated that the mean diameter of cells in foamed PC/PMMA films decreased with the increment of saturation temperature and saturation pressure but increased with the increment of the foaming time. Moreover, the mean diameter of cells decreased first, but then increased with the increment of the foaming temperature. The cell density (N c) increased with the increment of saturation temperature and saturation pressure but decreased with the increment of the foaming time. However, the N c increased first but then decreased with the increment of the foaming temperature.
Introduction
Microcellular foams are defined as foams with cell size less than 10 μm, and cell densities on the order of 10 9 –10 15 cells/cm 3 . 1 –3 Because of the inherent characteristics, such as high strength-to-weight ratio, excellent thermal and sound insulations, high energy or mass absorption, and materials saving, polymer foams have been used in many applications, including packaging, buoyancy, thermal insulation, acoustic attenuation, and vibration damping. 3,4 Supercritical fluid known as environmental-friendly technique is widely used to prepare microcellular foams. Supercritical fluid has many advantageous properties, such as a tunable solvent power, the plasticization of glassy polymers, and enhanced diffusion rates. 5 Especially, the use of carbon dioxide (CO2) has achieved many attentions in recent years due to the low critical conditions (31.1°C and 7.1 MPa). In general, supercritical carbon dioxide (scCO2) is inexpensive, abundant, and environmental benign. 6 ScCO2 is well known as a polymer foaming agent, used in both industrial process and polymer research. 7 –9 There are two main batch routes to produce microcellular polymer foams using scCO2, namely one-step process (continuous) and two-step process (noncontinuous).
In the one-step process, the polymer is first saturated with CO2 at a certain temperature and pressure (assuring the supercritical conditions of CO2) and kept for a fixed time. After that, the polymer is depressurized rapidly to atmosphere pressure. In this method, the cellular microstructure may be controlled by changing the saturation temperature and the depressurization rates. In the two-step process, the polymer is saturated with scCO2 in the supercritical regime. Next, the polymer/gas mixture is quenched into a glassy supersaturated state by reducing the pressure to atmosphere value. Finally, polymer is foamed by heating to a temperature above or near the glass transition temperature, leading to nucleation and cell growth. Foaming temperature and foaming time are the main parameters to adjust the cellular structures of the final product. Both in the one-step process and in the two-step process, microcellular foams are formed due to the thermodynamic instability of gas in a polymer system, which means the system is suddenly applied to a pressure drop, or a temperature increase, leading to sudden polymer/fluid immiscibility. 1 Four main steps are contained to obtain microcellular foams, which are shown in Figure 1. (1) Polymer/gas solution formation by saturating a sample with a high-pressure gas, (2) microcellular nucleation: sudden temperature drop, (3) cell growth, and (4) cell stabilization. The cell growth process is stopped by ending of the driving force for cell growth and the cellular foam is obtained. 10,11
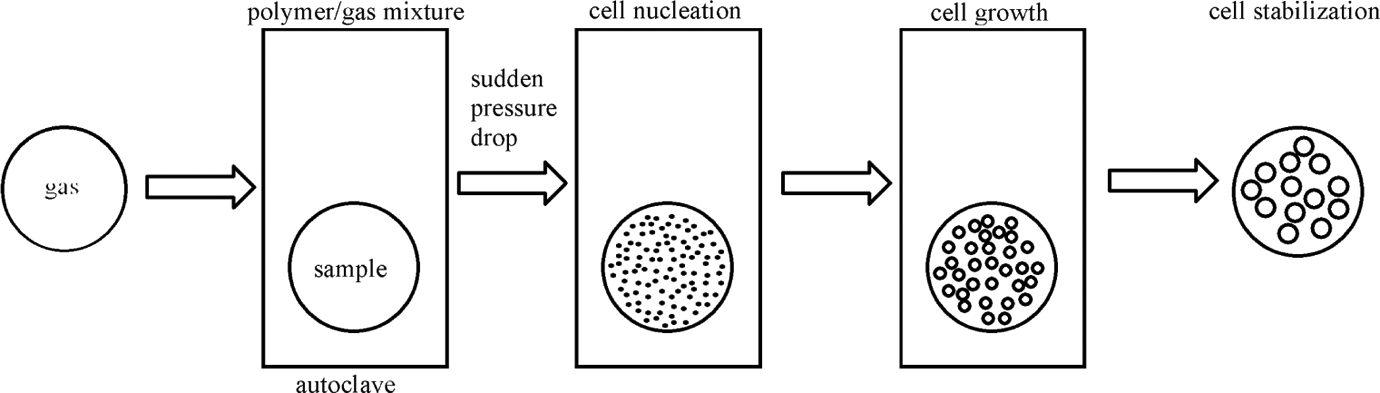
Schematic diagram of the foaming process in scCO2. scCO2: supercritical carbon dioxide.
Recently, micro-foaming technology has attracted more and more attentions in the research of polymer thin films using scCO2 as a physical blowing agent. 12 –14 Siripurapu et al. 15 studied the surface-constrained foaming of poly(methyl methacrylate) (PMMA) thin film. Their results demonstrated that the cell size can be systematically reduced to less than 100 nm under high saturation pressure, relatively low foaming temperature (near the glass temperature of the scCO2-plasticized polymer), and a rapid pressure quench. Besides, Siripurapu et al. 16 studied the synergistic use of surface barriers and nanoscale additives as alternate means to promote bubble nucleation, which can improve the porosity of PMMA thin films constrained between impenetrable plates. The foaming process is modified with nanosilica particles and commercial short-chain fluorosurfactants, or designer CO2-philic block/graft copolymers, and then exposed to high-pressure CO2. According to their report, the bubble nucleation of copolymer micelles is much better controllable because the hard nanoparticles only can increase cell density (N c) by more than two orders of magnitude at low CO2 pressures.
Polymer blends have always been considered as a cheap way to obtain new high-performance polymeric materials, whose physical properties may not only be superior to that of their components but also tunable as a function of the components of blends without synthesizing new polymers. Blends of polycarbonate (PC) and PMMA have received considerable attentions 17 –19 because of their potential applications as gas separation membrane, pearl material, substrate of the optical data storage discs, packaging material, and so on. 20,21
Many studies have reported the microcellular foaming of multiphase/multicomponent polymer blends, such as low-density polyethylene/ultra-high-molecular-weight polyethylene, polystyrene/PMMA, polyphenylene sulfide/polyether sulfone, and polyether ether ketone/polyetherimide, 22 –25 but the investigation of the control foaming of PC/PMMA blends is quite scarce in the past few decades. The aim of this work is to investigate the cell structures of foamed PC/PMMA blends. In this work, microspheres of PMMA prepared by soap-free emulsion polymerization are used to fabricate PC/PMMA blends as fillers. Foamed blends of PC and microspheres PMMA are prepared by the two-step process using scCO2 as physical foaming agent. The effects of process conditions on the cell morphologies and cell parameters are to be investigated.
Experimental
Materials
Methyl methacrylate (MMA, 98.0%, chemically pure grade; Shanghai Lingfeng Chemical Reagent Co., Ltd. China) was distilled under reduced pressure before use in order to remove polymerization inhibitors. The initiator, potassium persulfate (KPS, 99.5%, AR grade; Unite initiator (shanghai) Co., Ltd, China), and copper II sulfate pentahydrate (CuSO4·5H2O, 99.0%, analytical reagent (AR) grade; Shanghai Xinbao fine chemical plant, China) were used as-received without any further purification. Bisphenol-A-PC used in this study was commercial, which was purchased from Sam Yang Advanced Materials (Trirex 3026B (L); Korea). A laboratory grade of methylene chloride (MC, 99.5%, AR grade; China) was obtained from Sinopharm Chemical Reagent Co., Ltd (China) and used in the preparation of PC/PMMA blends. Industrial CO2 with a purity of 99.9% was supplied by Nanjing Wenda Special Gas Co., Ltd (China).
Synthesis of PMMA particles
The PMMA particles were prepared by soap-free emulsion polymerization. The synthesis was performed in a 500 mL three-necked round-bottom flask placed in the constant temperature water bath. The flask was equipped with a water cooled reflux condenser, a thermometer, and a nitrogen (N2) gas inlet/outlet. N2 gas was bubbled into flask throughout the reaction.
A typical synthesis proceeded as follows: 10 mL MMA and 70 mL distilled water was added into the flask, which was then bubbled with N2 for 30 min. Under mechanical stirring (the stirring speed was maintained at a uniform speed during the reaction), the solution was heated to 80°C. Then, 0.027 g KPS were dissolved in 10 mL H2O. The reaction was initiated by adding 10 mL KPS solution into the flask. After approximately 2 min, the transparent solution became cloudy and then turned milky white within a few minutes. After 2 h reaction, 10 mL solution that contained 0.025 g CuSO4·5H2O was added into flask. The reaction was stopped after 3 h by adding the CuSO4 solution. After removing the unreacted initiator, the polymer emulsion was washed in distilled water by centrifugation. The microspheres PMMA were thus obtained by evaporating the water in an oven at 80°C for 24 h.
Blends preparation
A total of 1.5 g PC/PMMA mixtures were dissolved in 50 mL of MC (3.0% (w/v) solution) at room temperature for 24 h. Thin films of PC/PMMA blends were prepared with the concentration of 10PC/90PMMA (w/w) by casting solution. The solution was cast on glass plates and covered with petri dishes for slow evaporation overnight. Then, the films were placed in an oven at 80°C for another 24 h to remove excess solvent. All of these films were peeled from glass plates by immersing in water, and free-standing films were transferred to a vacuum at 80°C for an additional 12 h to remove residual water.
ScCO2 foaming
A scCO2 extraction system was applied for impregnation of CO2 at a supercritical condition. Primarily, the dried PC/PMMA thin films were cut into 1.2 × 2.0 cm2 rectangles for foaming. Second, the rectangular films were sandwiched in a metal. Finally, the specimen was set in a high-pressure vessel in stainless steel and the entire vessel was placed in a thermostat oven. The specimen was saturated with scCO2 for 5 h at CO2 pressures ranging from 10 MPa to 30 MPa and temperatures ranging from 40°C to 70°C. After scCO2 impregnation, the vessel was depressurized rapidly and the specimen was removed from the vessel to oil bath for foaming. During the foaming process, two main steps were employed. The schematic diagram of experiment for the two-step foaming process is shown in Figure 2. First, the foaming temperature in the hot oil bath was fixed at 100°C, with foaming times of 10, 30, 60, 90, and 120 s. In the second step, foaming time was fixed at 60 s, with different foaming temperatures of 80, 90, 100, 110, and 120°C. During heating process, CO2 bubble expanded in the PC/PMMA films. Then, the specimen was quenched in cool water at room temperature to fix the cell structure. Foamed samples were further dried under vacuum at 50°C for 24 h.
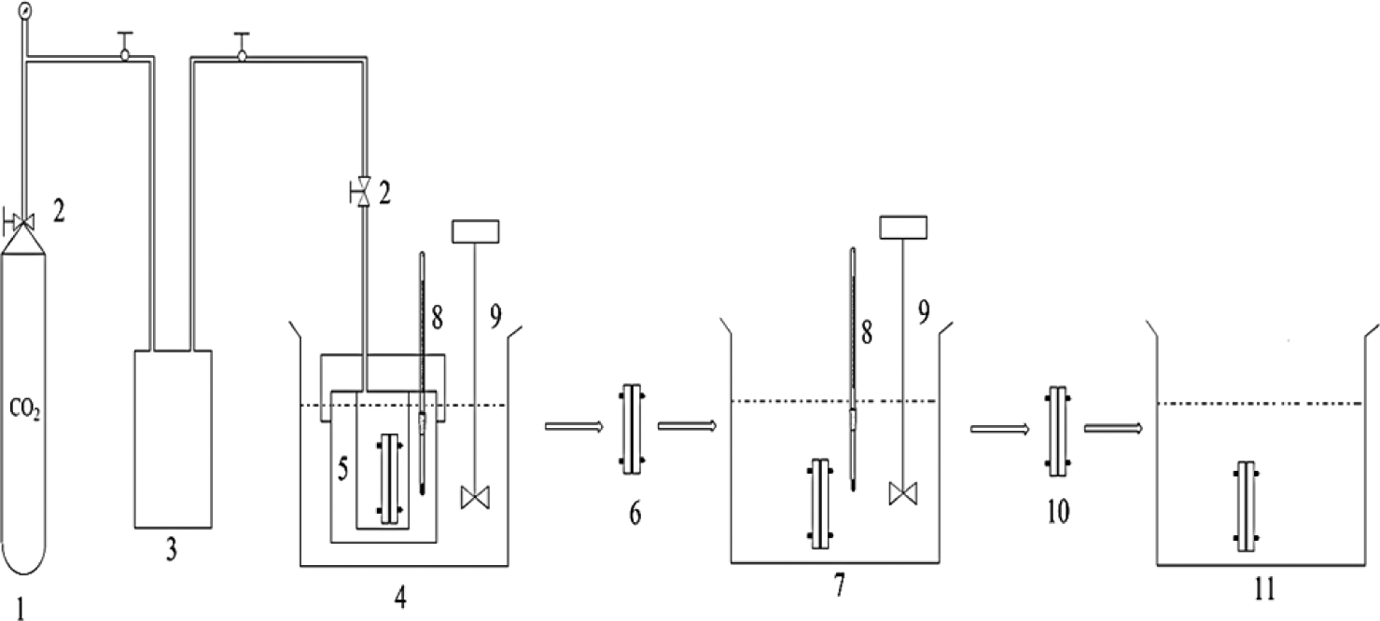
Schematic diagram of the experiment for the two-step foaming process: (1) CO2 cylinder, (2) valve, (3) high-pressure liquid pump, (4) constant temperature water bath, (5) high-pressure vessel, (6) saturated specimen, (7) constant temperature oil bath, (8) thermometer, (9) stirrer, (10) foamed specimen, and (11) cold water bath. CO2: carbon dioxide.
Scanning electron microscopy
The morphology of microsphere PMMA was examined by scanning electron microscope (SEM) using a QUANTA FEG250 (FEI, USA) apparatus operating at an accelerating voltage of 25 kV. The SEM image of PMMA microspheres is shown in Figure 3. It showed that these homogeneous particles were spherical; moreover, the surface of particles was smooth. The particle size of PMMA microspheres was about 300 nm.

SEM images of PMMA microspheres. SEM: scanning electron microscope; PMMA: polymethyl methacrylate.
In order to observe the microstructures of samples, foams were frozen under liquid N2 and fractured. After that, the samples were pasted on the conductive adhesive, then sputter coated with gold. The morphology of the foamed PC/PMMA blends was also examined by the same SEM at 5 kV . The mean cell diameter (d) and N c were determined by SEM images and analyzed by Image-Pro Plus software [version 6.0.0.260.]. The d was obtained using the following equation and at least 50 cells were selected randomly from SEM images:
The N c is defined as the number of cells per unit volume of the polymer, which was determined by the following equation 26 :
where n is the number of cells observed in a SEM image, A is the area of SEM image, and M is the magnification factor.
Results and discussion
Effect of saturation temperature on the cell morphology of foamed PC/PMMA blends
The SEM images in Figure 4 illustrate the cell morphology of foamed PC/PMMA blends with various saturation temperatures at a given saturation pressure of 10 MPa, a given foaming temperature of 100°C, a given foaming time of 60 s, and the solubility of scCO2 in PC/PMMA blends at various saturation temperatures is listed in Table 1. As shown in Figure 4, it was obvious that there were seldom cells at saturation temperature of 40°C or 50°C. The solubility of scCO2 in PC/PMMA blends increased with increasing the saturation temperatures. The high temperature improved the kinetic motion of scCO2, which accelerated the dissolution of scCO2 into PC/PMMA thin film. As the increment of the saturation temperature, the distribution became more uniform and the cell wall thickness decreased. The reason may be the increase in saturation temperature resulted in a higher solution of CO2 in PC/PMMA thin film, which was confined in metals. It was known that, foamability of polymer is affected by the gas sorption of polymer and the amount of the gas dissolved in the polymer influences the mechanism of cell nucleation and cell growth. When the saturation temperature was at 40°C and 50°C, the solubility of CO2 in the films was very little. It was not enough to drive all of these cells to grow. As the saturation temperature reached 60°C, there were many cells in the PC/PMMA thin films, but the distribution was not very uniform. While the saturation temperature was at 70°C, the solubility of the CO2 in the blend was enough to drive cell nucleate growth, so the distribution of the cells was uniform. The phase structure of PC/PMMA blend may be another reason that leads to the formation of cell structures. The higher amount of CO2 had stronger plasticization to PC, which improved the motion of molecular. In the heterogeneous nucleation process, the interface between two phases provided the nucleation sites and induced the formation of cells. When the saturation temperatures are 60°C and 70°C, the molecules of PC provided more nucleation sites, and it was in favor of the formation of cells. 24,27,28 That is to say, the 70°C was the best saturation temperature.

Cell morphology of foamed PC/PMMA blends with different saturation temperature: (a) 40°C, (b) 50°C, (c) 60°C, and (d) 70°C (saturation pressure: 10 MPa; foaming temperature: 100°C; foaming time: 60 s). PC: polycarbonate; PMMA: polymethyl methacrylate.
Solubility of scCO2 in PC/PMMA blends at various saturation temperatures.

scCO2: supercritical carbon dioxide; PC: polycarbonate; PMMA: polymethyl methacrylate.
Effect of saturation pressure on the cell morphology of foamed PC/PMMA blends
As shown in Figures 5 and 6, the effect of saturation pressure on the cell morphology was investigated. It could be observed from Figure 6 that the cell distribution became uniform and the cell wall thickness decreased as the saturation pressure increased from 10 MPa to 30 MPa, whereas the saturation temperature, foaming temperature, and foaming time were maintained at 70, 100, and 60 s, respectively. What’s more, Figure 7 shows that the d decreased and the N c increased with the increment of saturation pressure. In addition, it could be observed from Figure 6 that the bubbles were easy to combine each other when the saturation pressure reached to 30 MPa. This may be the increment of saturation pressure, which improved the solubility of CO2 in the PC/PMMA polymer blends. Higher concentration of CO2 in polymer would lead to more super saturation of CO2 in the composites which, in turn, increased bubble nucleates. As a result, the cell size decreased and the N c increased. In addition, the higher concentration of CO2 in polymer increased the free volume in polymer and enhanced the motion of the molecular chain, which led to the decrease in viscosity and increase in the plasticization of the polymer blends.

Cell morphology of foamed PC/PMMA blends at different saturation pressure: (a) 10 MPa, (b) 20 MPa, and (c) 30 MPa (saturation temperature: 70°C; foaming temperature: 100°C; foaming time: 60 s). PC: polycarbonate; PMMA: polymethyl methacrylate.
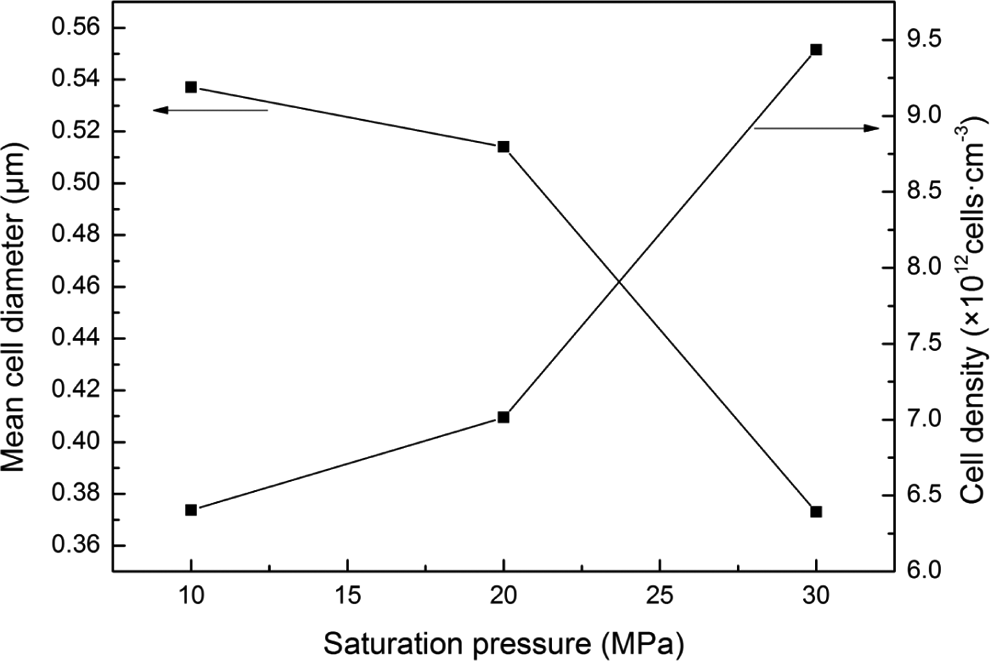
Cell parameters of foamed PC/PMMA blends at different saturation pressure (saturation temperature: 70°C; foaming temperature: 100°C; foaming time: 60 s). PC: polycarbonate; PMMA: polymethyl methacrylate.

Cell morphology of foamed PC/PMMA blends with various foaming time: (a) 10 s, (b) 30 s, (c) 60 s, (d) 90 s, and (e) 120 s (saturation temperature: 70°C, saturation pressure: 10 MPa; foaming temperature: 100°C). PC: polycarbonate; PMMA: polymethyl methacrylate.
Effect of foaming time on cell morphology of the foamed PC/PMMA blends
Figures 7 and 8 show the effect of the foaming time on the cell morphology of foamed PC/PMMA blends at a constant saturation temperature of 70°C, a constant saturation pressure of 10 MPa, and a constant foaming temperature of 100°C. The foaming time was set at 10, 30, 60, 90, and 120 s, respectively. As shown in Figure 7, there were a lot of bubbles in the foamed PC/PMMA blends at the foaming time of 10 s, but the cell size was very small. Because when the foaming time was 10 s, it was too short to drive the cell nucleates to grow. With the increment of foaming time, the cell size increased and the distribution of the cell became more homogeneous. In addition, it was found from Figure 8 that the d increased and the N c decreased with the increment of the foaming time. As the foaming time increased, there were more CO2 trapped in the cell nucleate, and it was enough to drive the cell nucleate growth. Moreover, it was found that the bubble was easy to combine each other at the foaming time of 120 s. It was due to the longer foaming time, which resulted the more CO2 trapped in the cell nucleate.
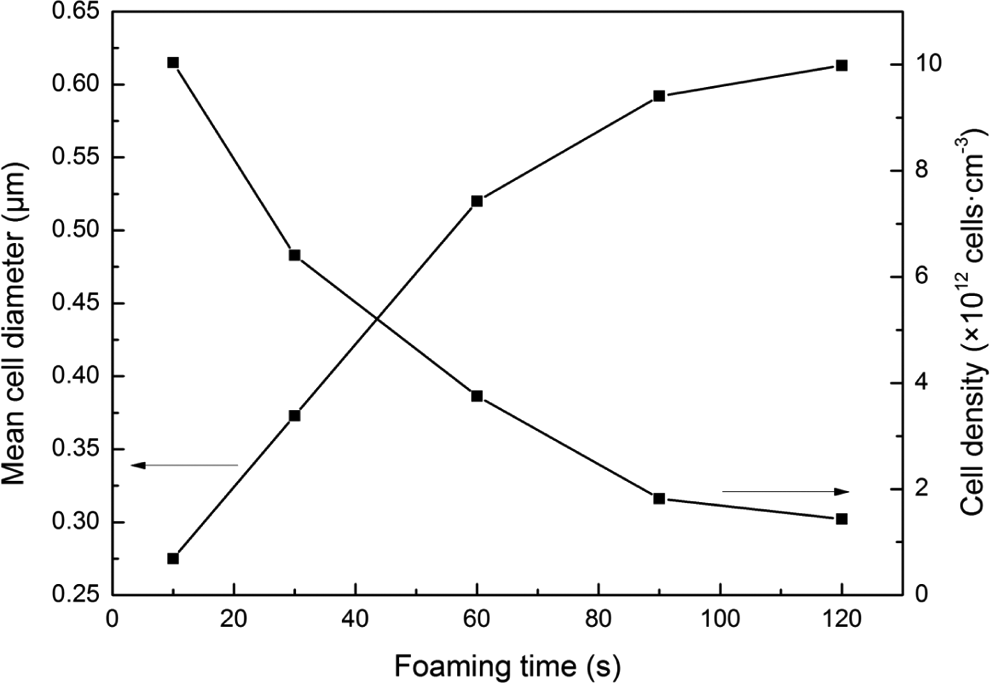
Cell parameters of foamed PC/PMMA blends with various foaming time (saturation temperature: 70°C; saturation pressure: 10 MPa; foaming temperature: 100°C). PC: polycarbonate; PMMA: polymethyl methacrylate.
Effect of foaming temperature on cell morphology of the foamed PC/PMMA blends
Foaming temperature is one of the most important factors, which has the significant effects on the cell morphology on the foams. The effect of foaming temperature on the cell morphology of foamed PC/PMMA blends was shown in Figures 9 and 10. Figures 9 and 10 show that, with the increment of foaming temperature, the cell size and the d decreased first and then increased. But the N c increased first and then decreased. When the foaming temperature was 100°C, the cell size was the smallest, but the N c reached the biggest. Because the free volume in polymer blends increased and the motion of molecular chains enhanced with the increment of the foaming temperature, which led to the decrease of the melt viscosity of the polymer blends. But with the increment of foaming temperature, more and more cell nucleate formed in the polymer blends. When the foaming temperature was below 100°C, the melt viscosity decreased little but a lot of cell nucleates were formed, so the cell size decreased and the N c increased with the increment of foaming temperature. But when foaming temperature was more than 100°C, the melt viscosity decreased much but just a few of cell nucleates were formed, so the cell diameter increased and the N c decreased with the increment of foaming temperature.

Cell morphology of foamed PC/PMMA blends at different foaming temperature: (a) 80°C, (b) 90°C, (c) 100°C, (d) 110°C, and (e) 120°C (saturation temperature: 70°C; saturation pressure: 10 MPa; foaming time: 60 s). PC: polycarbonate; PMMA: polymethyl methacrylate.
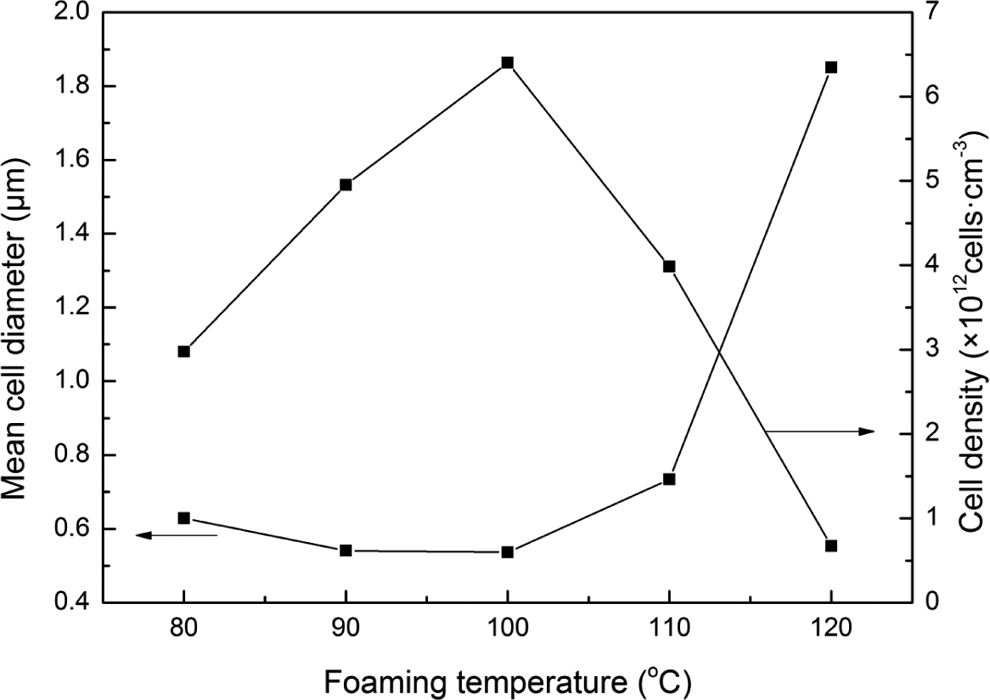
Cell parameters of foamed PC/PMMA blends at different foaming temperature (saturation temperature: 70°C; saturation pressure: 10 MPa; foaming time: 60 s). PC: polycarbonate; PMMA: polymethyl methacrylate.
Conclusion
The microcellular PC/PMMA (10/90) films were prepared by the two-step process using scCO2 as physical foaming agent. The morphology structures of foamed PC/PMMA films were investigated by SEM. The results showed that the cell distribution became uniform and the cell wall thickness decreased with the increment of saturation temperature and saturation pressure. In addition, the d decreased and the N c increased with the increment of saturation pressure. With the increment of foaming time, the d increased and the N c decreased. Moreover, it was found that bubbles were easy to combine each other with the foaming time of 120 s. It could also found that the cell size and the d decreased first and then increased with the increment of foaming temperature, but the N c increased first and then decreased.
Footnotes
Author's Contribution
Yuanxiang Luo and Yajun Ding contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work is partially supported by the major creative foundation of Nanjing Institute of Technology (CKJA201302), Youth natural science foundation of Jiangsu Province (BK20140763), and Youth foundation of Nanjing Institute of Technology (QKJA201202).


