Abstract
Ring-opening polymerization of anionic polyamide-6 (APA-6) requires both an activator and an initiator for the reaction to occur. Typical processing techniques for liquid-molded thermoplastic composite laminates involve infusion of the reinforcement with a premixed monomer solution containing both activator and initiator species. The technique described here is a step toward simplification and automation of the in situ polymerization process for composite laminates. By depositing the initiation functional group onto the reinforcement, infusion of a single stream of inert monomer solution is possible. The technique simplifies the processing equipment required and reduces the risk of contamination. Two separate methodologies derived from a silane and a diisocyanate were investigated. The soluble diisocyanate method was used to successfully demonstrate the single-stream APA-6 processing technique. Glass fiber surface-initiated polymerization was also demonstrated using the silane-derived initiator. The findings represent the first steps toward a new processing paradigm of APA-6 composites.
Keywords
Introduction
Thermoplastic composites possess mechanical properties that are optimal for current automotive, transportation, and energy industry needs. A thermoplastic composite with high specific properties of strength and stiffness, end-of-life recyclability, facile reparability, impact energy absorption, and good vibration damping must also be rapidly producible. The preceding requirements are best met when the reinforcing phase consists of continuous fibers in a near-net shape with minimal processing steps. The liquid molding of anionic polyamide-6 (APA-6) laminates has proved to be one of very few processes that can meet all requirements simultaneously as studied by several authors. 1 –4 Liquid molding of thermoplastic composites is possible only when the polymer is formed in situ due to the relatively low viscosity of monomer compared to polymer. The technology has tremendous potential, yet wide-scale industrial adoption of APA-6 processing has been met with obstacles related to the stringent processing conditions of temperature, chemical activity, and contamination.
One significant challenge of in situ polymerized APA-6 laminates is the reactivity of the monomer stream. Typically, two separate monomer solutions are prepared. One stream contains a small concentration of chemical activator, produced when the monomer, ε-caprolactam, is reacted with an alkali metal or similar species. The other stream contains a small concentration of initiator to start anionic ring-opening polymerization. When the two solutions are combined and heated above the reaction temperature of approximately 150°C, polymerization proceeds quickly. Typical processing windows can range from several minutes to several seconds. Once mixed, the induction time, the period of low solution viscosity appropriate for infusion, is limited, and excess monomer must be purged from the system. The gelation issue can be solved using a sophisticated metering system and mix head, which adds to the capital cost of production. The goal of the following work is to eliminate the difficulties in processing and equipment complexity of a two-stream system. By depositing the initiation species onto the reinforcement preform, a single stream of activated monomer may be used. Complex metering equipment becomes unnecessary. Successful application of the technology has widespread implications for developing a new processing paradigm for rapid reaction injection molding and new markets of continuous APA-6 products.
For reinforcements with weak interaction with thermoplastics, such as glass fibers, sizing greatly improves the mechanical performance. Typical sizing chemistries for bonding glass to amides are silanes with amine groups that interact with the matrix through secondary bonds. Improved matrix adhesion and moisture resistance were demonstrated by Goettler and Otaigbe with the use of γ-aminopropyltriethoxysilane. 1,5 Surface-initiated polymerization, however, has the added advantage of a stronger covalent bond with the reinforcement which could prove to be more effective.
With a similar processing goal in mind, Yan and Yang reacted hydroxylated multiwalled carbon nanotubes (MWCNTs) with toluene diisocyanate to initiate surface polymerization. 6 The effect was a more homogeneously dispersed composite. Yan and Yang did not seize the opportunity to use the nanotubes solely to initiate polymerization. In another study by Yang et al., isocyanate-functionalized MWCNTs were confirmed to be APA-6 initiation sites. 7 Both studies demonstrated the viability of chemical sizing with APA-6 initiation capability.
The previous examples were investigations on the nanometer scale. When considering the length scales of typical woven fabric reinforcement, surface initiation alone does not provide a sufficiently uniform distribution of starting points for complete polymerization within the bulk of the matrix. Additionally, the surface concentration of Si-OH functional groups on typical glass fibers is around 6–15 µM−2 which may limit the maximum concentration of initiation groups available. 8 Therefore, to completely convert all monomer into polymer in a uniform fashion, a soluble initiation species must be present. Cast nylon chemistries are based on this principle, with initiator reagents pre-dissolved in monomer pellets that are melted and premixed with the activated monomer solution. However, these processes must be carried out under an inert, moisture free atmosphere to prevent termination of the growing polymer chains by moisture contamination. 9 It follows that for practical usability of any initiator that has been placed directly onto the fiber reinforcement must be resistant to deactivation through moisture or contamination in the ambient air.
The variety of chemical initiators available for the anionic polymerization of polyamides is diverse. 9 The subset of initiators based on diisocyanates was chosen for this study due to its relatively high effectiveness. 10,11 Also, isocyanates are easily “blocked” with other chemical species to inhibit their reactivity prior to use. Isocyanates blocked with ε-caprolactam, for example, are resistant to hydrolysis by ambient moisture, which prevents the formation of unfavorable chemical groups when exposed to air. Isocyanates blocked with ε-caprolactam are coincidentally an initiation moiety for APA-6, which can be seen in Figure 1. When combined with a silane denoted as “R” in the figure, the initiator becomes a glass fiber sizing as shown in Figure 2.
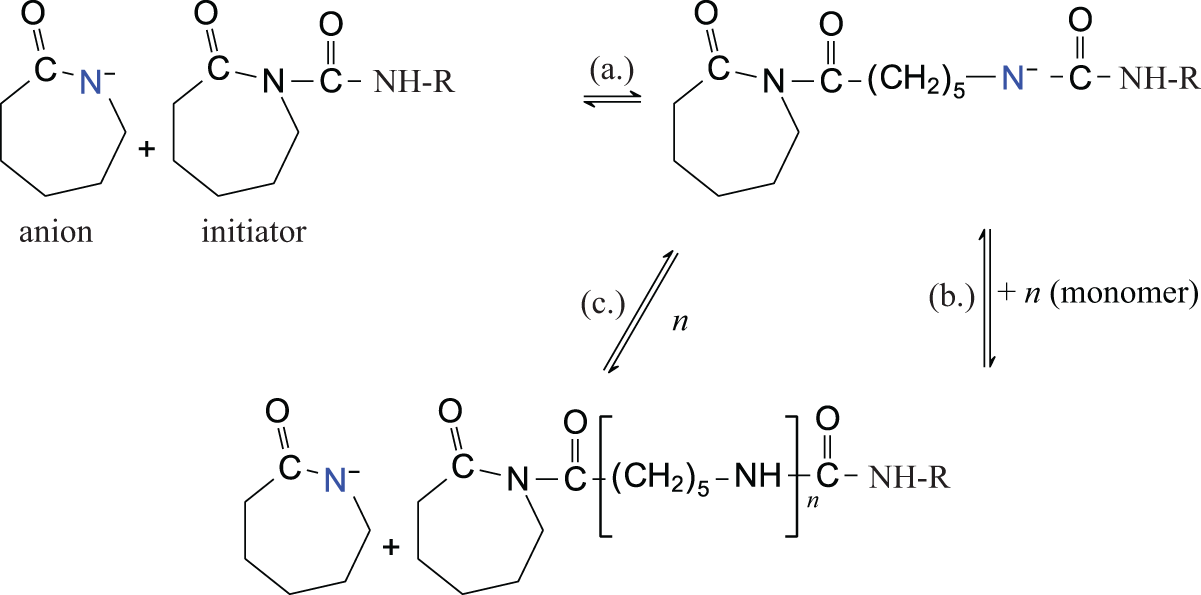
The “coactivated” anionic initiation and propagation reaction steps. An anion reacts with the initiator followed by a ring-opening step. The regenerated anion reacts with the ε-caprolactam monomer and the ring-opening process continues.
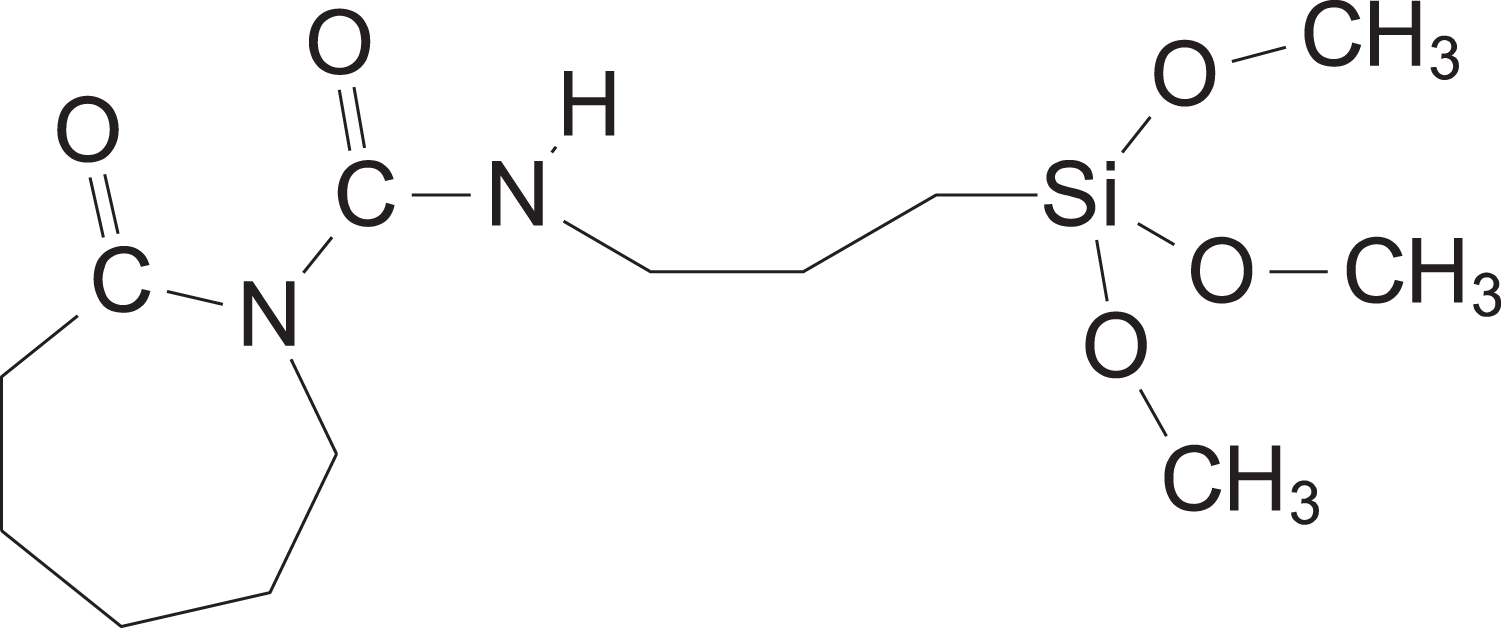
The APA-6 initiating silane molecule showing functionality for both glass fiber and APA-6 (CAS: 106996-32-1). APA-6: anionic polyamide-6.
Materials
E-glass fabric (588 g m−2 plain weave with proprietary sizing) was provided by PPG Industries Inc. (Carlisle, PA, USA). A commercially available APA-6 system consisting of an activator, caprolactam magnesium bromide (Nyrim C1); an initiator, hexamethylene-1,6-dicarba-moylcaprolactam (Bruggolen C20); and the monomer, ε-caprolactam was supplied by Brüggemann Chemical (Newtown Square, PA, USA). The diisocyanate chosen for the work was 4,4’-methylene diphenyl diisocyanate 98% (MDI) from Acros Organics (Geel, Belgium). The experimental fiber-sizing initiator was
Useful data to calculate concentrations required for a target APA-6 molecular weight of 53,000 g mol−1.

APA-6: anionic polyamide-6.
Characterization techniques
Microscopy
Scanning electron microscopy (SEM) images were acquired with a Quanta FEG 650 SEM equipped with a gaseous secondary electron detector (GSED). Images were acquired at low pressures of water vapor using the GSED rather than traditional sputter-coated high-vacuum imaging to avoid alteration of the fiber surface morphology and to prevent charging.
1H NMR spectroscopy
Proton nuclear magnetic resonance (1H NMR) spectra were recorded on a Bruker DRX-400 NMR spectrometer (Billerica, MA, USA). Deuterated solutions of the complexes were prepared under a stream of N2 gas, and all NMR experiments were performed at room temperature. The 1H NMR spectra were referenced to internal SiMe4.
Differential scanning calorimetry
A TA Instruments’ differential scanning calorimetry (DSC) equipped with a TA Q100 controller and refrigerated cooling system was used to measure the melting endotherm of the soluble diisocyanate initiator (SDI) product. Samples were heated at 10°C min−1. The deblocking onset temperature was shown by Parker et al. to coincide with the initial deviation from the tangent line at the start of the melting endotherm. 12
Fourier-transform infrared spectroscopy
Functional group analysis was done using a Bruker ALPHA Fourier-transform infrared (FTIR) Spectrometer in attenuated total reflectance mode. Sample aliquots dissolved in toluene were removed from the SDI synthesis flask at the reaction start and after 28 h. The presence of a peak at 2265 cm−1 corresponds to the presence of unreacted isocyanate groups. 12
Thermogravimetric analysis
A TA Instruments’ thermogravimetric analyzer (TGA 2950, New Castle, DE, USA) was used to determine the minimum temperature required to completely remove organic functionality from the surface of the E-glass fabric. Samples were heated at 20°C min−1 under nitrogen.
Processing
Calculation of stoichiometric quantities
The final molecular weight of APA-6 depends upon the concentration of initiation sites. 9 For a single-stream APA-6 process where the initiators are deposited onto the reinforcement prior to infusion, the final composite fiber weight fraction becomes an important variable. Stoichiometric quantities are calculated first in terms of moles of monomer. Second, the mole fractions are converted to a mass fraction with respect to 1 mole of monomer. Finally, the mass fraction must be adjusted by the target composite fiber weight fraction and calculated using the final mass of the composite. Since the anionic polymerization of ε-caprolactam into APA-6 conserves mass, that is, does not evolve any volatiles, the mass of monomer and final polymer are the same.
The final goal of the process is to produce APA-6 with a number average molecular weight

The functionality
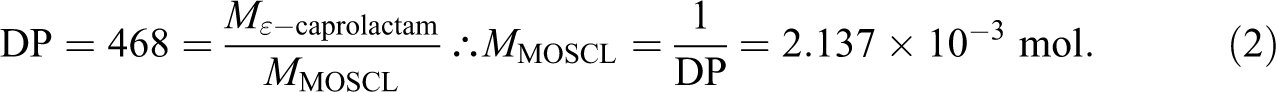
A mass fraction with respect to 1 mole of ε-caprolactam is a more practical quantity than a mole fraction. Thus, the mass fraction
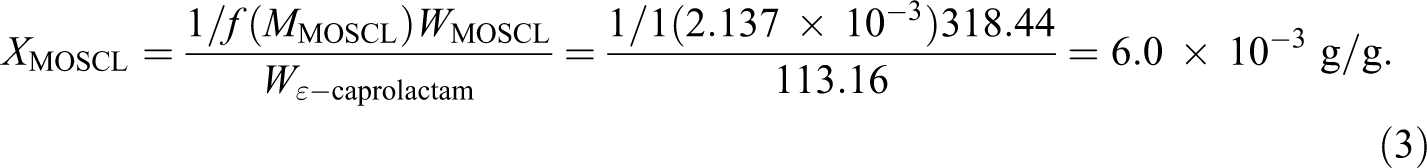
Thus to achieve a molecular weight of the matrix polymer of 53,000 g mol−1, the mass fraction of MOSCL with respect to the total mass of the infused monomer is 0.60 wt%. Achieving a sufficient concentration of APA-6 initiation groups using a monolayer of MOSCL is difficult due to the actual concentration of Si-OH functional groups on the fiber surface. 8 For this study, it was determined that a complementary, soluble initiator would be required for bulk polymerization.
A similar calculation is done for the MDI-based initiator. MDI has a functionality of 2, with two isocyanate (NCO) groups per molecule. Using the details in Table 1 to determine the required degree of polymerization, we find that the ratio of ε-caprolactam to NCO is 234 for a difunctional initiator. Each growing chain will have a molecular weight of 26,500 g mol−1 with a total molecule weight of 53,000 g mol−1. The baseline initiator, Bruggolen C20P, also has a functionality of 2. The molar ratio of NCO to ε-caprolactam is equivalent to the DP and is calculated as:
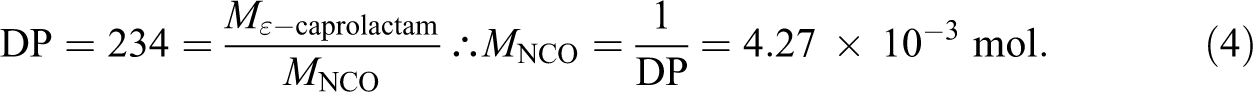
The mass fraction
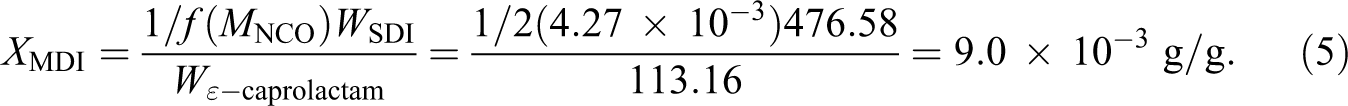
Thus, the concentration of SDI required to obtain the comparable polymer molecular weight of 53,000 g mol−1 is 0.90 wt%, with respect to the total mass of the infused monomer stream.
Synthesis of soluble diisocyanate initiator
A 1-L two-neck round-bottomed flask equipped with a reflux condenser was rinsed with organic solvents and flame-dried under vacuum. The reaction vessel was then moved to an oxygen-free, dry nitrogen glove box where 5.0 g of solid MDI, 10.0 g of ε-caprolactam, and 500 mL of anhydrous toluene were added. The molar ratio of ε-caprolactam to NCO was in excess of 2:1, such that the self-reaction of MDI was minimized. The flask was removed from the glove box under positive nitrogen pressure and placed into an oil bath. The mixture was then heated to 80°C and was maintained at this temperature under positive nitrogen pressure for approximately 38 h. A synthesis scheme for the reaction is shown in Figure 3.
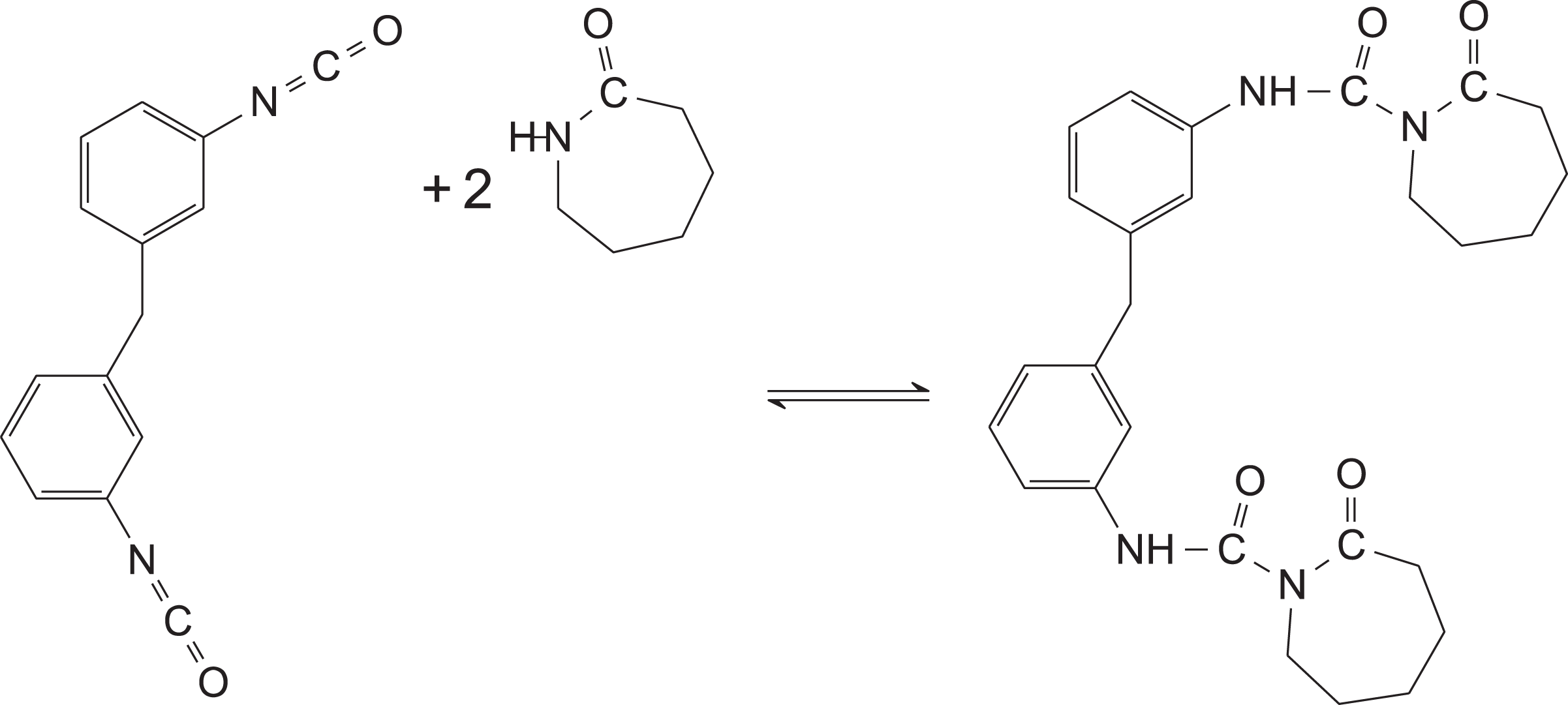
Synthesis scheme for the formation of the soluble diisocyanate initiator.
Sample aliquots were taken from the reaction mixture at the start and after 28 h. Figure 4 shows the FTIR spectrum of an aliquot taken at 0 h (grey) and 28 h (black). The black spectrum in Figure 4 shows that the NCO peak (2265 cm−1) was no longer detected after 28 h. The reaction was continued for an additional 10 h. The peak at 1713 cm−1 that appeared after 28 h corresponds to the newly formed carbonyl peak between ε-caprolactam and the isocyanate group.
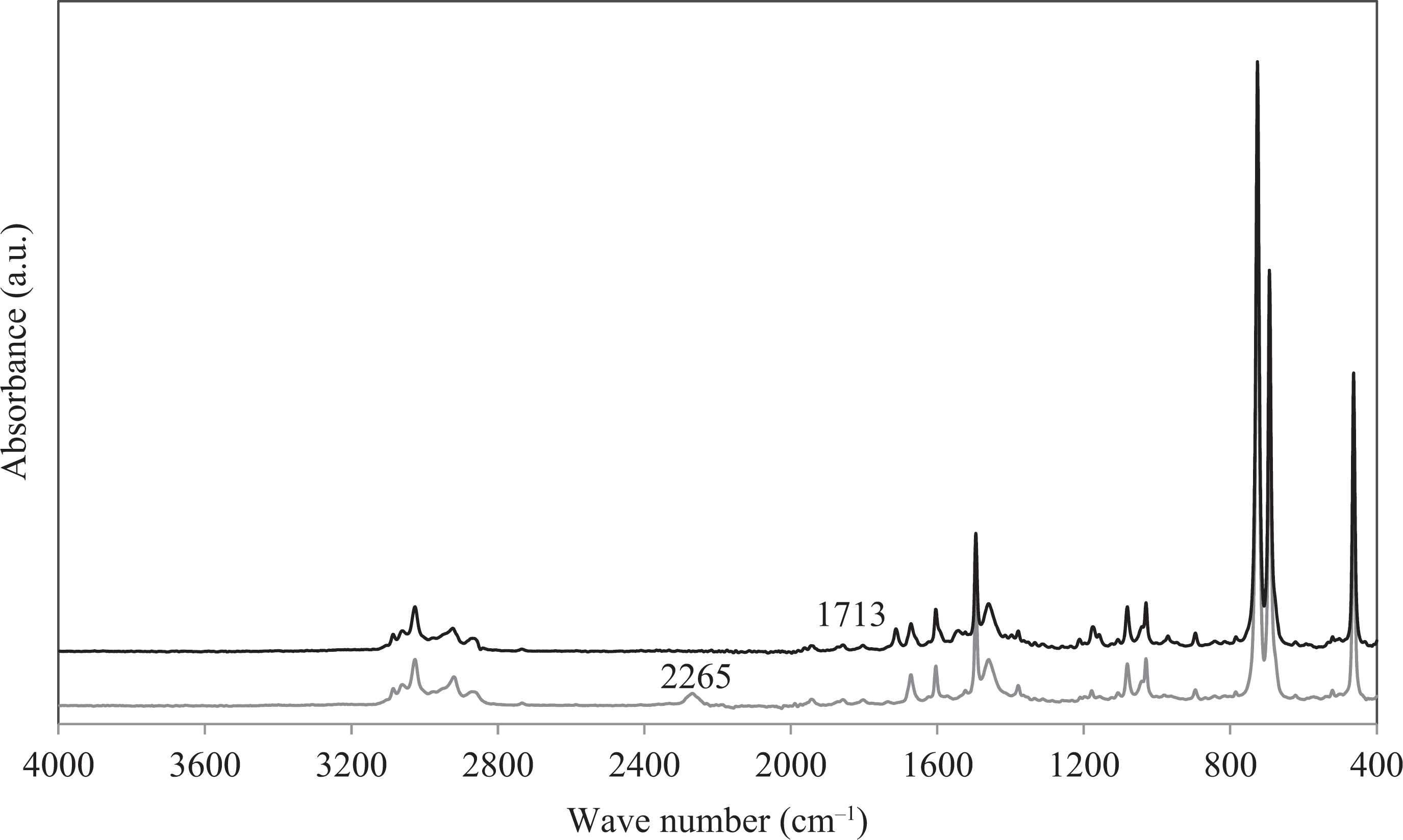
FTIR spectra of the solution over 28 h of reaction showing a complete disappearance of the NCO peak at wavenumber 2265 cm−1. A new C=O peak appeared at 1713 cm−1 when the NCO reacted with the ε-caprolactam. FTIR: Fourier-transform infrared.
After 38 h, the solvent was reduced to 10% of its original volume via rotary evaporation, at which time a white precipitate appeared. The solid was vacuum filtered through a 300-mL sintered glass funnel and washed three times with methanol. The product was then stirred in methanol for an additional 48 h to remove any remaining ε-caprolactam. The remaining solvent was decanted away. The solid was collected and dried in a vacuum oven at 120°C.
Initial single-stream processing trials using the as-produced SDI produced no polymer. The cause was hypothesized to be moisture absorbed by residual ε-caprolactam in the product which was effectively deactivating the anionic activator. For a final purification step to remove any remaining ε-caprolactam, the filtered white powder was redissolved in toluene and diluted with methanol 5:1. The dissolved SDI precipitated out of solution and was collected after 24 h and dried in vacuum at 120°C overnight to remove the excess solvent.
After purification, a 1H NMR spectrum was obtained of the newly formed structure as shown in overlay in Figure 5. Integration was normalized by the amine proton peak, indicated by the far downfield broad singlet at 11.456 ppm. The normalized integration under each peak corresponds to the number of protons for each bond type which matches the assumed structure exactly. The sum of the normalized integration adds to 32, which is the number of protons in the SDI structure. Excess unreacted ε-caprolactam, for example, would have offset the lactam integration to greater than 12 protons. Thus, the crystallized product was determined to be essentially pure.
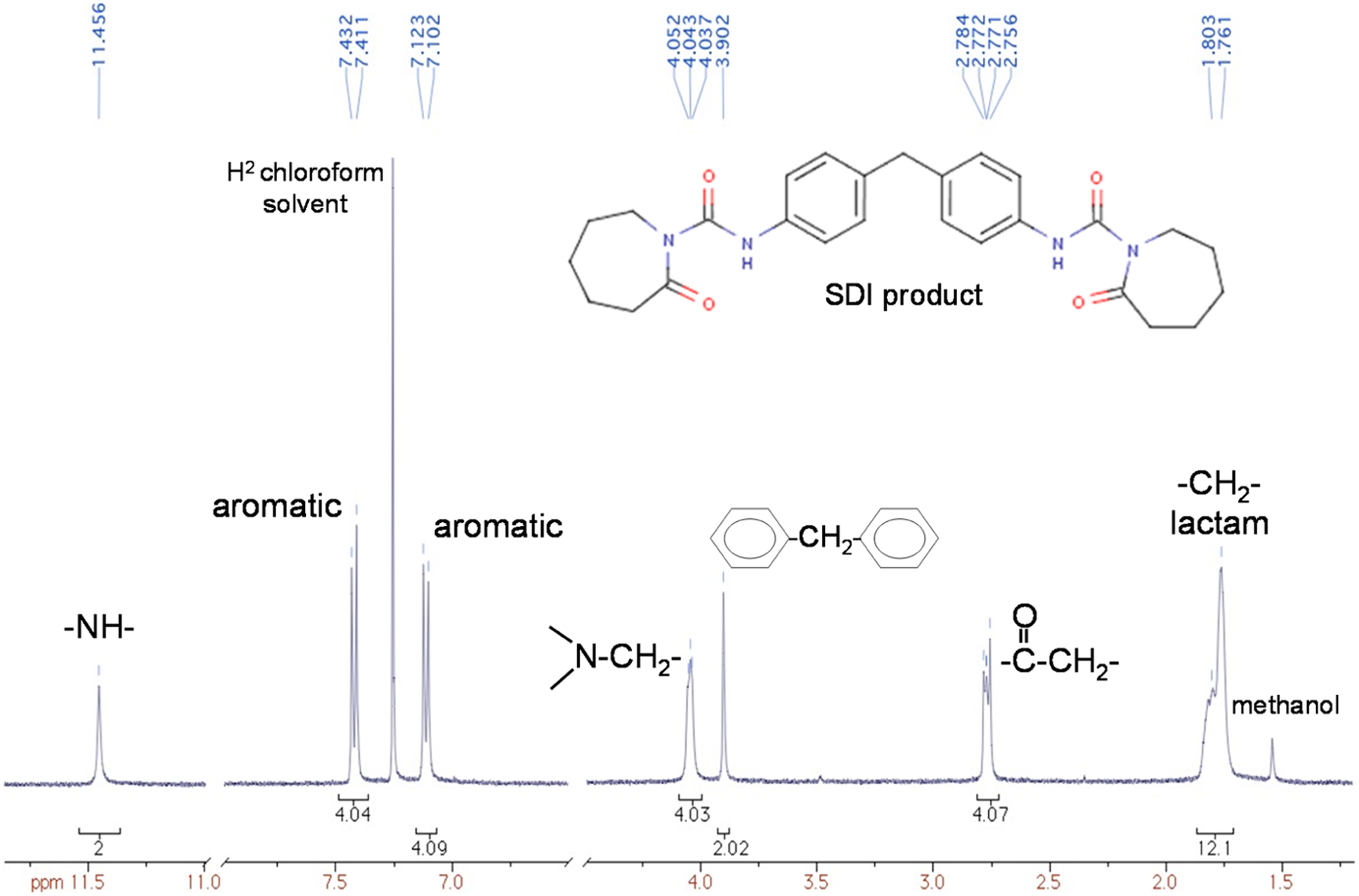
Proton NMR output of the purified SDI precipitate. NMR: nuclear magnetic resonance; SDI: soluble diisocyanate.
A comparison of the as-produced and purified SDI was done via DSC as shown in Figure 6. The heating rate was a standard ramp of 10°C min−1. The deblocking onset temperature was estimated following the procedure by Parker using the temperature of the first deviation from the tangent for the melting endotherm 12 . The DSC analysis confirmed that the purified and as-produced SDI product was stable to above the desired single-stream preprocess drying temperature range of 150–160°C.
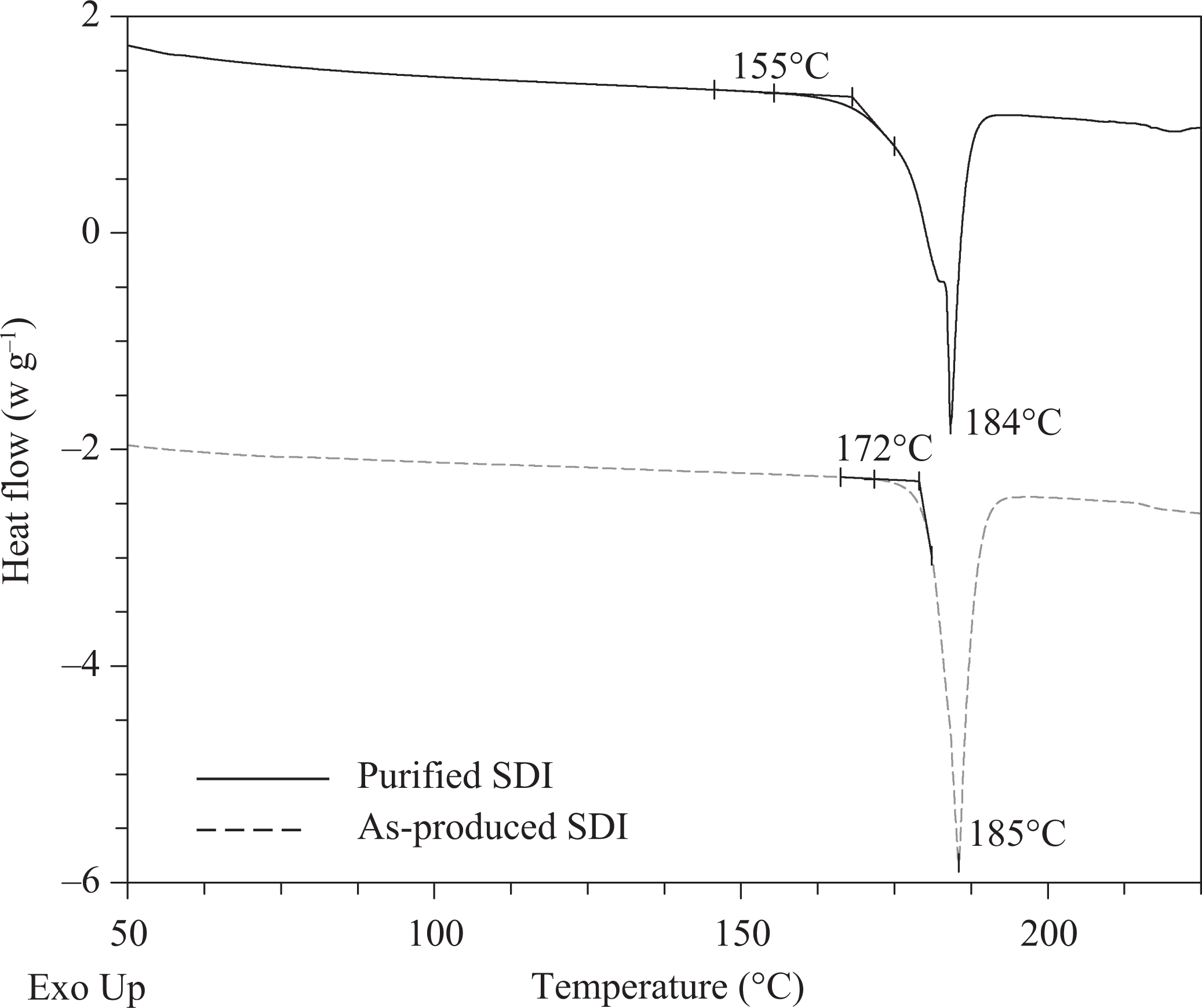
Purified SDI (solid) and the as-produced SDI (dashed) DSC curves are shown, offset. The purified SDI exhibited a lower deblocking initiation temperature of 155°C and a well-defined melting endotherm peak at 184°C. The as-produced SDI deblocking onset occurred at 172°C with the melt endotherm at 185°C. DSC: differential scanning calorimetry; SDI: soluble diisocyanate initiator.
Glass fabric pretreatment
Woven E-glass was heated to 600°C for 1 h to remove any organic sizing present on the fibers. Although heating above 400°C most certainly degraded the mechanical performance of the fibers, 600°C was determined to be necessary to completely remove any trace organics, as shown in a TGA plot in Figure 7.
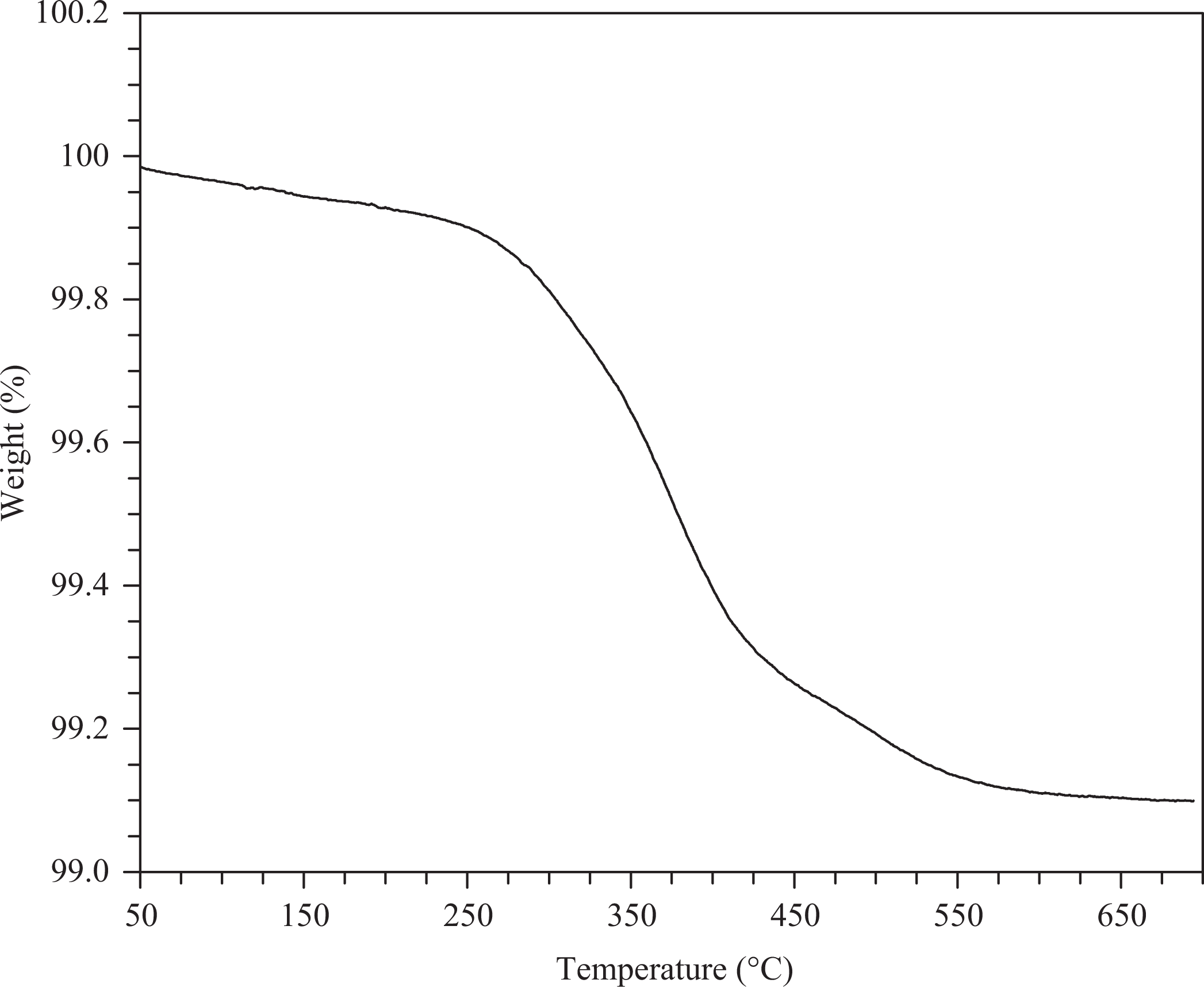
Thermogravimetric determination of minimum temperature required to completely remove organic sizing from test E-glass fabric.
Incorporation of SDI onto fabric preform
The appropriate number of glass fabric layers was cut and weighed. The stoichiometric quantity of initiator was calculated, based on a fiber fraction of 60 wt% using equation (5). For four layers of glass fabric, 1.92 g of SDI was combined with 300 ml of toluene. The solution was gently heated and stirred using a magnetic stir bar and hotplate until the solution became clear. The glass fabric layers were placed into a flat bottom tray and the solution was poured over the top. Individual layers were removed one at a time and placed onto wire mesh to dry in air in a fume hood.
Processing unsized SDI test panel
Once dried, the preform was placed into a resin transfer mold (RTM) and sealed under vacuum. The mold was heated to 160°C in a circulating air oven and held for 2 h to equilibrate in temperature. The infusion was done with a 5% Nyrim® C1 by weight of ε-caprolactam activated monomer solution at 120°C prepared under dry nitrogen. After infusion, the sample was held at temperature for 60 min. Once polymerization was ensured, the entire RTM was removed from the convection oven to cool.
The extent of SDI wash caused by the influx of monomer during infusion was inferred by measuring the matrix conversion along a profile in the panel following typical monomer extraction techniques. 4,15 Square samples of 19 mm on a side were cut with a band saw from the panel in a line directly through the center along the fiber 0° direction. The samples were weighed immediately to avoid any additional mass caused by ambient moisture absorption. The squares were placed in hot distilled water at approximately 60°C and allowed to soak for 72 h. The samples were then removed from the water and dried in a vacuum oven at 130°C for 8 h. The samples were weighed again, and the weight loss and remaining mass were recorded. The samples were then “burned-off” in a furnace at 550°C for 1 h to calculate the weight fraction of the fiber. The degree of conversion was calculated and is shown in Figure 8. The SDI washout and dilution by the incoming activated monomer stream is apparent by the dip in conversion at the center of the panel. However, nearly complete polymerization was observed along the perimeter of the panel. Polymerization was verified via DSC analysis as shown in Figure 9.
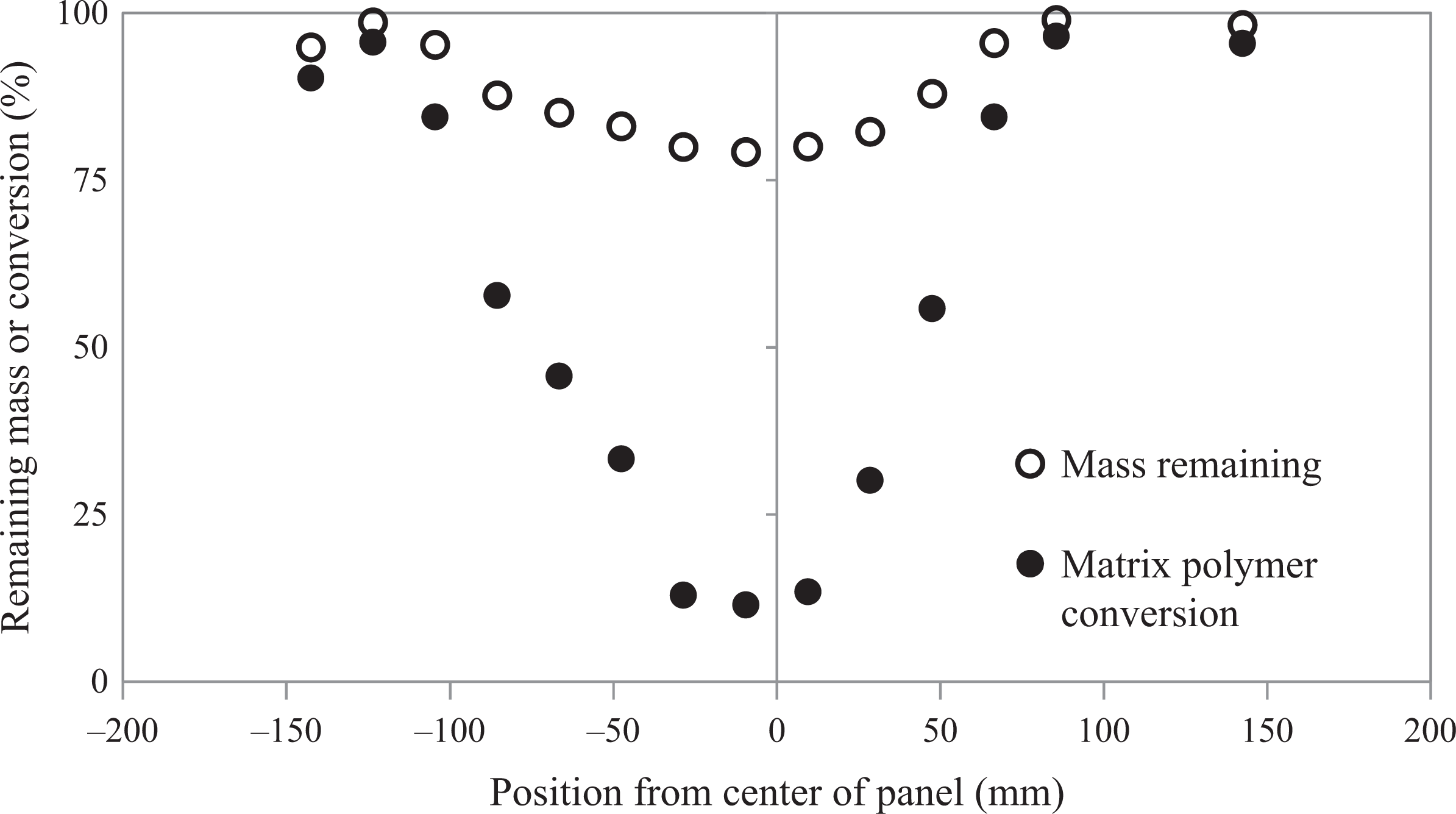
Remaining mass after soaking in distilled water (open circle). Approximate matrix polymer conversion (filled circle) with respect to position from the centerline of the panel along the 0° direction was calculated assuming a fiber content of 60 wt%.
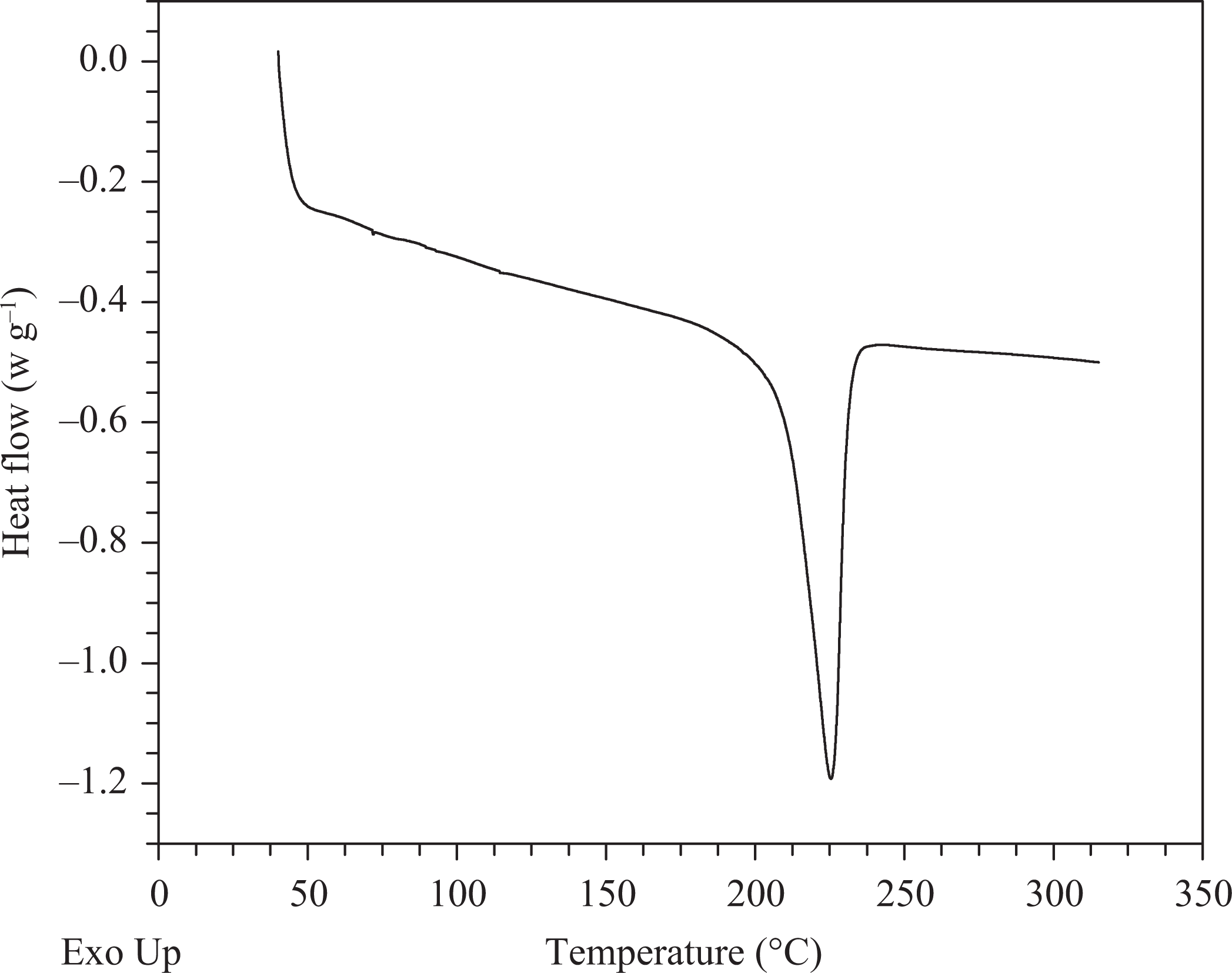
A DSC curve of a sample from the perimeter of the un-sized SDI panel. A large endothermic peak characteristic of APA-6 is shown near 225°C. For incomplete polymerization, residual monomer would appear around 68°C and/or the endothermic melting peak would become broad. The analysis confirms that near the perimeter of the panel, polymerization was successful. DSC: differential scanning calorimetry; SDI: soluble diisocyanate initiator; APA-6: anionic polyamide-6.
Silanization procedure
The procedure for silanization of the glass fabric used in the study was modeled after work done by Stenger et al. 16 and Hickman, 17 omitting the initial cleaning steps to instead use the fabric as-pyrolyzed. A solution was prepared with each concentration on a by-volume basis. Lab grade methanol (94%) was combined with 5.0% distilled water and 1.0% MOSCL. A stoichiometric quantity of solution was combined with woven glass fabric based on a nominal weight fraction of 60% fiber. The glass fabric was placed in a large flat basin and covered with the solution. The silanization was allowed to continue for 30 min to ensure even distribution throughout the fiber tows. Afterward, the fabric was removed from the solution and rinsed once in methanol. The fabric was then left to dry in air in a fume hood.
Surface characterization
The effectiveness of the MOSCL sizing as a surface polymerization initiator was tested using a similar procedure to the single-stream process but with a small fabric tow sample. A short section of MOSCL silanized glass tow was placed into a glass vial and dried under vacuum at 130°C for an hour. The furnace was back filled with air first passed through a canister of silica gel. The furnace was opened and the glass vial was quickly capped. Approximately 10 g of ε caprolactam and 0.5 g of Bruggolen C1, both in dry flake form, were added to the vial in a dry glove box. The vial was sealed and placed into a convection oven at 165°C for 2 h and removed. The content of the vial was dissolved in water to remove the unreacted ε-caprolactam. The fiber tow was recovered and placed in acetone and further washed in a common ultrasonic cleaner for 30 min. The fiber tow was then removed from the acetone and dried in a vacuum furnace at 130°C for 1 h.
Surface characterization was done by comparing the as-pyrolyzed glass and the MOSCL silanized glass fibers to the surface polymerization sample. SEM images were acquired with a Quanta FEG 650 (Hillsboro, OR, USA) scanning electron microscope under low vacuum. A low pressure of water vapor, approximately 80 Pa, was used to create the conduction path ordinarily provided by Au–Pd coating. A GSED was used. An accelerating voltage of 5 kV was used for imaging. A comparison of the surface characteristics of the as-pyrolyzed, MOSCL sized, and surface polymerized APA-6 fiber is shown in Figure 10. The fibers appeared to be clean after both the pyrolysis and after MOSCL sizing. Not until after surface polymerization was any noticeable change in surface roughness observed. The content of polymer was too small to be detected by DSC analysis.

(a) E-glass fibers are shown in the as-pyrolyzed condition. (b) The pyrolyzed fiber is shown after MOSCL silanization. (c) The fibers after surface-initiated polymerization of APA-6 are shown with an area of interest within the white dashed rectangle. (d) An expanded view of the fiber from within the rectangle in (c). The fiber surface shows evidence of polymerization occurring at several locations rather than a continuous monolayer of polymer. APA-6: anionic polyamide-6; MOSCL:
Conclusions
The viability of the single-stream processing technique for APA-6 was successfully demonstrated. Although the process has yet to be fully optimized, to the knowledge of the authors, this work represents the only such instance of a single-stream APA-6 process.
During infusion, the SDI initiator was washed by the incoming monomer stream. An undesirable concentration gradient of initiators resulted in incomplete conversion at the center of the panel. Despite the washing effect, complete polymerization was achieved around the perimeter of the sample panel. A more detailed study of the method by which the initiator is applied to the fiber preform, or the chemical characteristics of alternative initiator chemistry could alleviate the wash-out problem.
Glass fiber surface-initiated polymerization of APA-6 was demonstrated using the MOSCL sizing. As expected, bulk polymerization was not achieved from the fiber surface. However, only a relatively small quantity of polymer was observed possibly due to inefficient silanization of the fiber surface.
Footnotes
Acknowledgements
This work would not have been possible without the gracious support of Dr Gary Gray and Justin Martin from the UAB Department of Chemistry and the use of their facilities and expertise. Support from PPG Industries Inc. in supplying fabric materials and technical advice is also greatly appreciated. Special thanks go to the UAB SEM Laboratory for help with acquiring micrographs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding provided by the ALEPSCoR Graduate Research Scholars Program Rounds 7 and 8 was instrumental.
